Abstract
Temporomandibular disorders (TMD) and irritable bowel syndrome (IBS) are comorbid functional chronic pain disorders of unknown etiology that are triggered/exacerbated by stress. Here we present baseline phenotypic characterization of a novel animal model to gain insight into the underlying mechanisms that contribute to such comorbid pain conditions. In this model, chronic visceral hypersensitivity, a defining symptom of IBS, is dependent upon on three factors: estradiol, existing chronic somatic pain, and stress. In ovariectomized rats, estradiol replacement followed by craniofacial muscle injury and stress induced visceral hypersensitivity that persisted for months. Omission of any one factor resulted in a transient (1 week) visceral hypersensitivity from stress alone, or no hypersensitivity (no inflammation or estradiol). Maintenance of visceral hypersensitivity was estradiol dependent; resolving when estradiol replacement ceased. Referred cutaneous hypersensitivity was concurrent with visceral hypersensitivity. Increased spinal Fos expression suggests induction of central sensitization. These data demonstrate the development and maintenance of visceral hypersensitivity in estradiol-replaced animals following distal somatic injury and stress that mimics some characteristics reported in patients with TMD and comorbid IBS. This new animal model is a powerful experimental tool which can be employed to gain further mechanistic insight into overlapping pain conditions.
Keywords: comorbid pain, stress, visceral hypersensitivity, irritable bowel syndrome, temporomandibular disorder, estrogen
Introduction
In general women are more sensitive to pain than men (see 17,22 for review) and a greater number of chronic pain syndromes are more prevalent in women including irritable bowel syndrome (IBS) and temporomandibular disorders (TMD) 2,11,22,63. Both these conditions occur largely in premenopausal women and symptoms fluctuate across the menstrual cycle 25,29,40,56. Many patients with IBS or TMD report additional pain that is considered unrelated to the primary complaint resulting in comorbid or overlapping pain syndromes. For example, patients with TMD report symptoms consistent with IBS, chronic pelvic pain or fibromyalgia 1,21,66. For several frequently reported chronic pain syndromes, the rate of comorbidity exceeds 50%, making this a significant pain management problem.
Several hypotheses have been put forth to explain these seemingly unrelated comorbid conditions. Psychological factors including stress and/or depression can increase the severity of pain syndromes 20,26,44. While acute stress can be antinociceptive, e.g., stress induced analgesia 8, it is often is pronociceptive, especially for visceral stimuli 4,6,12,38. In contrast, chronic stress is typically pronociceptive. It exacerbates acute pain and triggers nociceptive episodes in patients with chronic pain syndromes 19,23. For example, functional gastrointestinal disorders including IBS are often comorbid with affective disorders such as depression, anxiety, panic, and posttraumatic stress disorder 10,35,44. Since stress modulates many pain syndromes it is not surprising that patients susceptible to one chronic pain condition have a high potential of experiencing multiple conditions during/after stressful situations. However, the underlying mechanisms are unknown.
Hormonal fluctuations and stress both modulate nociceptive sensitivity (see 60 for review). Since nociceptive stimuli originating in the deep tissues of rodents are especially susceptible to both the changing hormone levels during the estrous cycle and hormone replacement following gonadectomy, animal models can provide useful insight into pain conditions in women. Likewise, animal models of stress have been used to try to gain a mechanistic understanding of functional pain disorders, especially IBS 39. However, with the exception of diffuse noxious inhibitory controls (DNIC)/conditioned pain modulation (CPM), animal models to study the interaction of multiple pain conditions are lacking.
In the present study, we present a new animal model of chronic visceral hypersensitivity that is dependent upon estradiol, prior injury, stress and the temporal sequencing of these variables. This model mimics the clinical presentation of TMD patients with IBS symptoms, providing a platform to evaluate factors that contribute to these overlapping pain syndromes. Portions of this manuscript have been presented in abstract form 59.
Methods
Female Sprague-Dawley rats (225–250g) were purchased from Harlan (Indianapolis, IN) and double housed in the University of Maryland School of Dentistry animal facility with a 12h-12h light-dark cycle (lights on at 7am). Food and water were available ad libitum. All protocols were approved by the University of Maryland School of Medicine Institutional Animal Care and Use Committee and adhered to guidelines for experimental pain in animals published by the International Association for the Study of Pain.
Surgery
Rats were anesthetized with isoflurane and ovariectomized using a dorsolateral approach. Electromyogram (EMG) electrodes made from Teflon-coated 32 gauge stainless steel wire (Cooner Wire Company, Chatsworth, CA) were stitched into the ventrolateral abdominal wall. The electrode leads were tunneled subcutaneously and exteriorized at the back of the neck. Rats were treated pre- and postoperatively with buprenorphine (0.03 mg/kg, s.c., twice per day for 2 days). Upon recovery from anesthesia, rats were individually housed for the duration of the study.
Experimental protocol
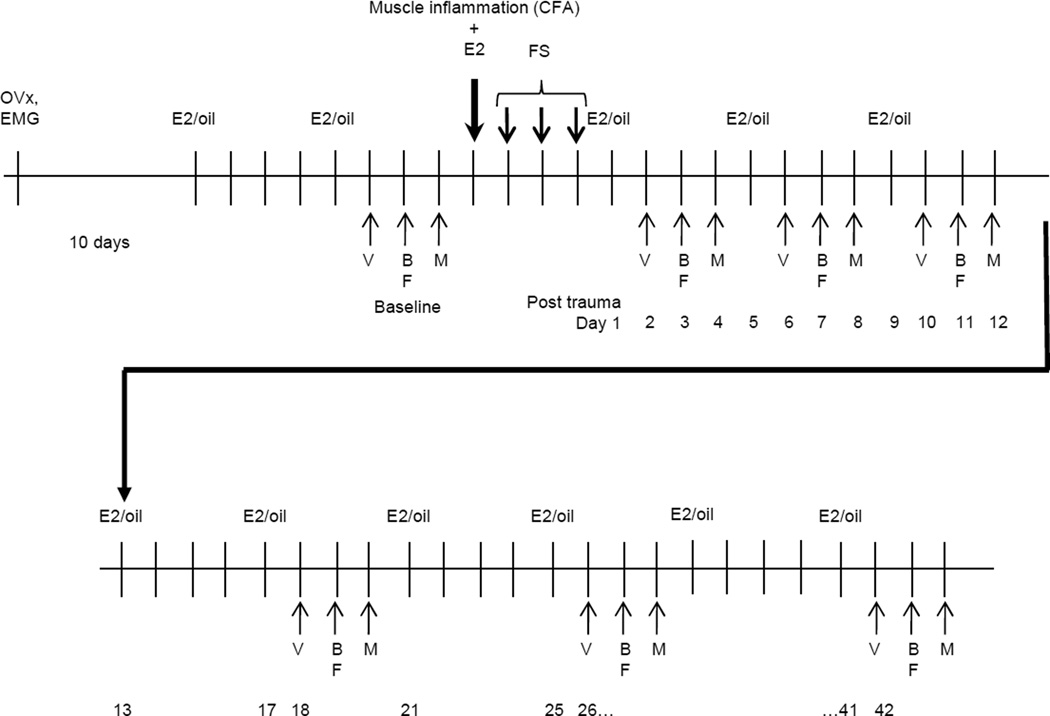
The experimental protocol is shown in Figure 1. The objective was to test the effects of prior injury (craniofacial masseter muscle inflammation, a model of TMD), stress (forced swim) and estradiol on visceral and somatic mechanosensitivity. Ten to 14 days following ovariectomy and electrode placement, rats were injected subcutaneously with 50 µg 17β-estradiol (E2) or 100 µl safflower oil (vehicle). The same injection was repeated at four day intervals. The bilateral masseter or biceps brachii muscles were injected with Complete Freund’s Adjuvant (CFA, 150 µl,1:1 in saline) or saline (control for CFA injection). Forced swim (FS) stress was produced by placing the rat in a cylindrical container (40×50 cm) containing 20 cm deep water at 26 °C for 10 minutes on the first day and 20 minutes on the next 2 days. The control for stress from the FS was to leave the rats undisturbed in their home cage. The day following the last forced swim was designated day 1. Baseline data were collected prior to the CFA injection/forced swim and then every 4–8 days for 6 weeks. This stress model was chosen because it could be completed between E2 injections or within one 4 day estrous cycle.
Figure 1.
Experimental design. E2 or oil is injected every 4 days. Day 1 is the first day after the last forced swim (FS). V: visceromotor response; B, F, M: mechanosensitivity testing of the back, forepaw, masseter muscle.
Rats were tested for their visceromotor response (VMR) to graded intensities of colorectal distention (CRD), mechanosensitivity of the lower back (area of referred pain from the colorectum) and forepaw (test for whole body mechanosensitivity) using von Frey filaments, and threshold to withdrawal from stimulation of the masseter muscle region using an electro von Frey. The VMR was measured 24 hrs following an E2 or oil injection. Mechanosensitivity of the back and forepaw were tested 48 hrs following E2 or oil injection and threshold to orofacial stimulation was tested 72 hrs following the injection. Not all rats were tested for somatic mechanosensitivity. There was no difference in the magnitude of the VMR for rats tested for all stimuli (VMR and von Frey on consecutive days) and those only tested for visceral sensitivity so the VMR data were pooled.
The visceromotor response
The VMR is the EMG recorded from the abdominal muscles in response to graded intensities of CRD. On the appropriate day as indicated in Figure 1, after the E2 or oil injection, rats were fasted overnight. Water was available ad libitum. The following day rats were prepared to record the VMR. Rats were briefly sedated with isoflurane and a 5–6 cm balloon attached to Tygon tubing was inserted into the descending colon and rectum through the anus. The secured end of the balloon was at least 1 cm proximal to the external anal sphincter and the tubing was taped to the tail. Rats were loosely restrained in Plexiglas tubes and given 30 min to recover from sedation. The EMG signals were recorded with a CED 1401 and analyzed using Spike 2 for windows software (Cambridge Electronic Design, Cambridge, UK). CRD was produced by inflating the distention balloon with air. The pressure was monitored and kept constant by a pressure controller/timing device. Rats were given three graded intensity distention trials (each trial consisted of 20,20,40,40,60,60,80,80 mmHg distentions; each distention 20 sec duration, 3 min interstimulus interval). The EMG was rectified, and the mean EMG for the 20 sec prior to distention was subtracted from the response during distention. The response at each pressure for the last 2 trials was averaged to determine the stimulus response curve. The area under the curve (AUC: sum of responses at each pressure) was determined as the response of the rat for that day.
Mechanosensitivity
The day following the VMR measurement, rats were tested for mechanosensitivity. Rats were placed on an elevated tray (46×36 cm) with 1 cm sides and left for 30 min to acclimate. Von Frey filaments were applied to the back at the level of the base of the tail. Each filament was tested 5 times at 10 sec intervals. Filaments were tested with increasing force ranging from 1–200 g. Next rats were place on an elevated grated floor under inverted boxes. After a 30 min acclimation period rats were stimulated from underneath on the forepaws with von Frey filaments using the same protocol as on the back. The number of withdrawals to each filament was plotted.
On the 3rd day after the E2 injection, rats were tested for their response to craniofacial mechanical stimulation by probing the masseter muscle through the facial skin using a rigid von Frey filament coupled with a force transducer (Electrovonfrey, model no 2290, IITC, tip diameter 4mm). This large tip diameter is used to preferentially activate neuronal receptors located in deep tissues (Takahashi et al 2005). The force needed to elicit a withdrawal of the head was recorded following each of five stimulus presentations spaced at approximately one minute intervals.
Quantification of c-Fos in the dorsal horn
Separate groups of E2 and oil treated rats were treated with CFA and FS. One to two weeks after the last swim session rats were distended to 60 mmHg for 2 hrs (30 sec on, 90 sec off) and then perfused with 4% paraformaldehyde. The T13-L2 (TL) and L6-S2 (LS) spinal cord segments were removed, 30 µm sections cut and every 4th section labeled for cFos using standard immunocytochemical protocols.
Cells immunopositive for c-Fos were quantified using a non-biased stereological method 64. Tissue was examined with the observer blinded as to the experimental group. Spinal cord sections were randomly selected and stereological analysis of the dorsal horn was conducted using Stereoinvestigator software (Microbrightfield, USA). The upper and lower 1µm of each section was defined as the guard zone and was not analyzed due to inhomogeneity of the tissue. A 125µm × 125µm counting frame was utilized which yielded a CE of less than 0.1 and a ratio of CE2 to CV2 less than 0.5 (where CE=coefficient of error and CV=group variance) indicative of adequate sampling 57.
Data analysis
VMR: It was very difficult to maintain EMG electrodes in rats for up to 10 weeks. There was attrition in each treatment group over the course of the experiment. At a minimum, EMG electrodes had to remain in place at least through day 10 post FS (3 time points) to be included in any data analysis. Subsequently there were fewer rats at later time points necessitating analysis using one way ANOVAs without repeated measures. For analysis, the area under the curve for the stimulus response curve was calculated (AUC). The AUC at each timepoint starting on day 2 (see Figure 1) was normalized to each rat’s baseline response prompting nonparametric analysis (Kruskal-Wallis 1-way ANOVA) to assess changes from baseline across time within each treatment group. Multiple comparisons vs. baseline (Dunn’s test) following a significant ANOVA are reported in the figures.
Mechanosensitivity: The total number of withdrawals to the range of von Frey filaments (1–200g) was calculated and plotted across time for stimulation of the back and forepaw. The area under the curve was calculated and normalized to the baseline AUC. Threshold to withdrawal was determined for stimulation over the masseter muscle. Changes from baseline across time within a treatment group were determined by nonparametric 1 way ANOVA.
RESULTS
Prior injury and stress induce estrogen-dependent visceral hypersensitivity
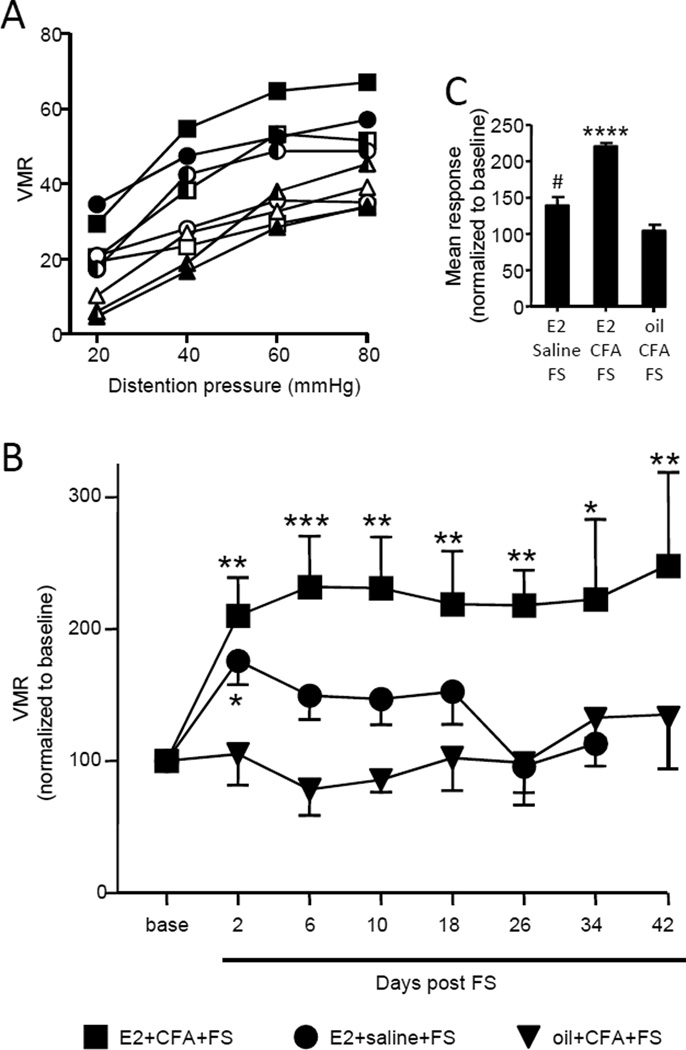
The effect on visceral sensitivity of bilateral masseter injection with CFA or saline followed by forced swim was examined in E2- or oil- treated ovariectomized rats. Stimulus-response curves for 3 time points (baseline, 6 days, 34 days) show a more vigorous response in E2+CFA+FS rats compared to E2+saline+FS rats or oil+CFA+FS rats (figure 2A). Collapsing the data to obtain the AUC at each timepoint over the 6 week test period, there was significant visceral hypersensitivity compared to baseline in the E2+CFA+FS rats (Kruskal-Wallis 1 way ANOVA, p<0.001, n=14; Figure 2B). In contrast, oil+CFA+FS failed to evoke any visceral hypersensitivity (Kruskal-Wallis 1 way ANOVA, p=0.4, n=7; Figure 2B). In the absence of masseter inflammation, E2+saline+FS, there was a significant effect of stress in E2-treated rats over 10 days (E2+saline+FS: Kruskal-Wallis 1 way ANOVA, p<0.05, n=10; Figure 2B). However, the p value edged up towards 0.07 over 34 days because the additional time points were not different from baseline. By 42 days all rats in this cohort had lost their electrodes. Collapsing the data post FS across time to get a mean total response, the magnitude of the VMR in the E2+CFA+FS rats was significantly greater than the E2+saline+FS and oil+CFA+FS rats (1 way ANOVA, p<0.0001; figure 2c). These data suggest that injury followed by stress induced profound and long lasting de novo visceral hypersensitivity that was estrogen dependent.
Figure 2.
The duration of visceral hypersensitivity is estrogen dependent. A: The magnitude of the visceromotor response to graded intensities of CRD for E2+CFA+FS rats (square), E2+saline+FS rats (circle) and oil+CFA+FS rats (triangle) at baseline (open symbols), 6 days post forced swim (filled symbols) and 34 days post forced swim (half-filled symbols). B: the area under the curve (AUC) of the VMR at all time points. Symbols represent same treatment groups shown in A. The data are normalized to the baseline response (base). *,**,*** p<0.05, 0.01, 0.001 vs. baseline in that treatment group. C: The data in B collapsed to the overall mean area under the curve. **** p<0.0001 vs. other groups; # p<0.05 vs. oil+CFA+FS.
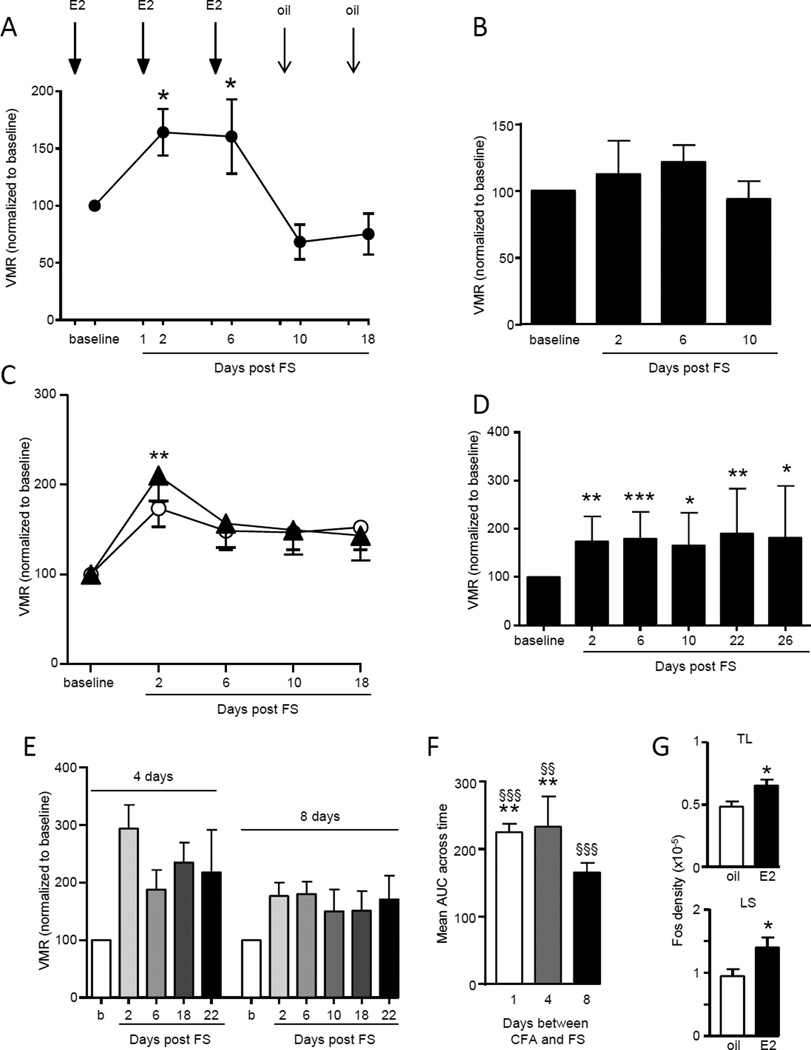
Six additional experiments were conducted to strengthen the conclusion that prior injury plus stress induced E2-dependent chronic visceral hypersensitivity. First, ovariectomy alone did not induce visceral hypersensitivity. Ovariectomized rats were left unchallenged in their home cage except to measure the visceromotor response to colorectal distention at the appropriate time points shown in Figure 1. There was no change in the magnitude of the VMR compared to the baseline response over 42 days (Kruskal-Wallis 1 way ANOVA, p=0.33, n=8). Second, the requirement of E2 in order for injury plus stress to induce chronic visceral hypersensitivity was demonstrated two ways. The oil+CFA+FS group (Figure 2) showed no evidence of visceral hypersensitivity. This was further confirmed by replacing the E2 injection with oil. When E2 was injected according to the schedule through day 6, visceral sensitivity increased similar to that observed in the E2+CFA+FS rats. Substitution of safflower oil for E2 on day 9 abrogated the visceral hypersensitivity (Kruskal-Wallis 1 way RM ANOVA, p<0.005, n=6; figure 3A). By day 18, 12 days after the last E2 injection, the magnitude of the VMR had further decreased, essentially eliminating the recovery of the VMR induced by E2 replacement.
Figure 3.
Chronic visceral hypersensitivity is dependent on estradiol (E2) and injury (CFA) preceding stress (FS). A: E2 administered one day prior to VMR testing in CFA+FS rats resulted in chronic visceral hypersensitivity. Substituting safflower oil for E2 at day 9 reversed the hypersensitivity below baseline. * p<0.05 vs. 10 day. B: In the absence of stress, E2+CFA failed to induce visceral hypersensitivity. C: Stress preceding injury (FS+CFA; closed triangles) induced the same magnitude and duration of visceral sensitivity as stress alone (E2+FS; open circles). ** p<0.01 vs. baseline. D: In E2 rats, biceps brachii inflammation preceding FS induced chronic visceral hypersensitivity. *,**,*** p<0.05, 0.01, 0.001 vs. baseline. E: There was chronic visceral hypersensitivity if the period between the CFA injection and the beginning of the FS was extended to 4 or 8 days. E2 injections were maintained every 4 days. F: The mean AUC across time for different intervals between CFA and FS. **p<0.01 vs. 8 days; §§, §§§ p<0.01, 0.001 vs. baseline. G: CRD-induced Fos expression in the lumbosacral (LS) and thoracolumbar (TL) spinal cord in E2+CFA+FS and oil+CFA+FS rats. * p<0.05 vs. oil.
Third, in E2-treated rats inflammation of the masseter muscles without FS (E2+CFA) had no effect on visceral sensitivity (VMR measured to day 10: Kruskal-Wallis 1 way ANOVA, p=0.54, n=6; Figure 3B). This suggests it is a combination of injury plus stress which results in the prolonged visceral hypersensitivity. Fourth, the order of the injury and stressor in the E2 treated rats is significant. Stress (forced swim) following an existing injury (masseter muscle inflammation) evoked the prolonged visceral hypersensitivity in E2-treated rats, but stress preceding the injury produced effects similar to stress alone. When CFA was injected the day following the last forced swim (day 1), there was a significant increase in visceral sensitivity only on day 2, returning to baseline by day 6 and continuing at the baseline level through day 18 (Kruskal-Wallis 1 way ANOVA, p<0.05, n=7; Figure 3C).
Fifth, the site of the initiating injury is not confined to the masseter muscles. Injection of CFA into the biceps brachii followed by forced swim induced visceral hypersensitivity (Kruskal-Wallis 1 way ANOVA, p<0.001; n=14; Figure 3D), of similar magnitude and duration as inflammation of the masseter muscles. In oil-treated rats, CFA injected into the biceps brachii plus FS did not result in chronic visceral hypersensitivity. Sixth, the chronic stress-induced visceral hypersensitivity was not dependent on the close interval between muscle inflammation and forced swim. Forced swim starting 4 or 8 days following masseter inflammation still resulted in visceral hypersensitivity in E2-treated rats (Kruskal-Wallis 1 way RM ANOVA, p<0.05 for both 4 day (n=6) and 8 day (n=7) interval; AUC: p<0.002; Figure 3E,F).
The effect of E2 on visceral sensitivity was further tested by examining Fos expression in the spinal cord. One to two weeks after CFA+FS, when there was a clear distinction in the VMR between E2 and oil treated rats, there was significantly more CRD-induced Fos expression in both regions of spinal cord that receive colonic afferent input (TL and LS segments) of the E2 treated rats compared to the oil treated rats (t-test for TL, LS, p<0.05, n=7–8/group; Figure 3G). This suggests the visceral hypersensitivity is at least partially dependent on increased nociceptive processing at the level of the spinal cord.
Mechanosensitivity
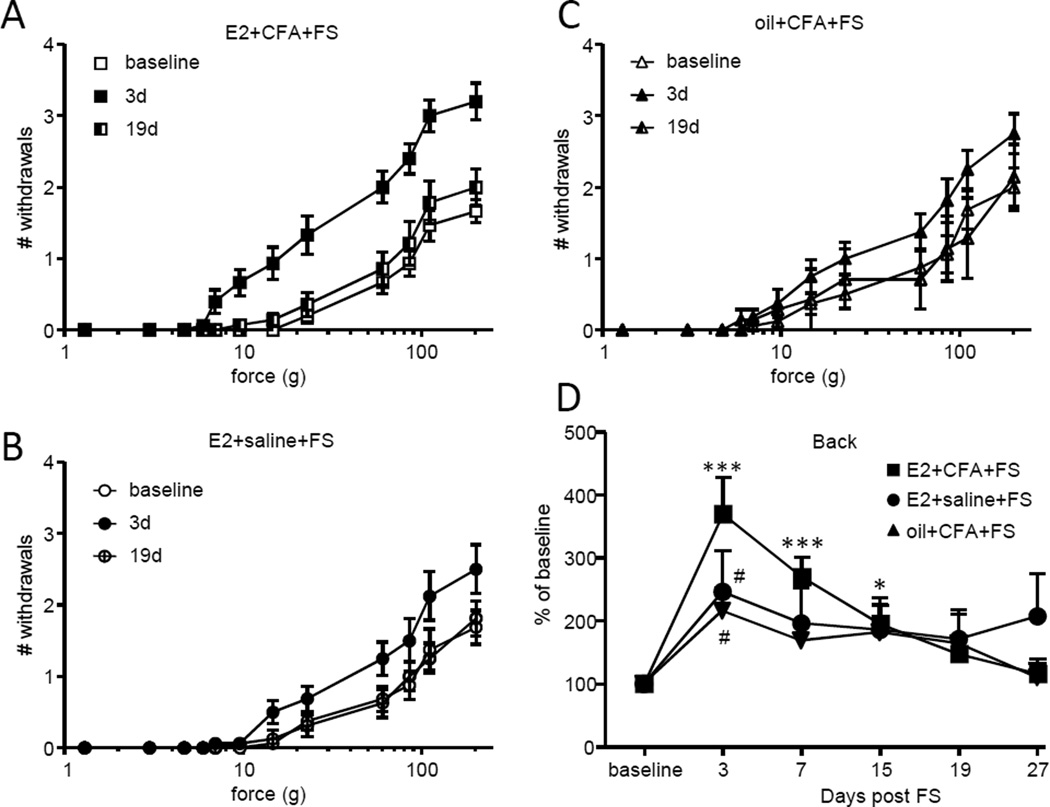
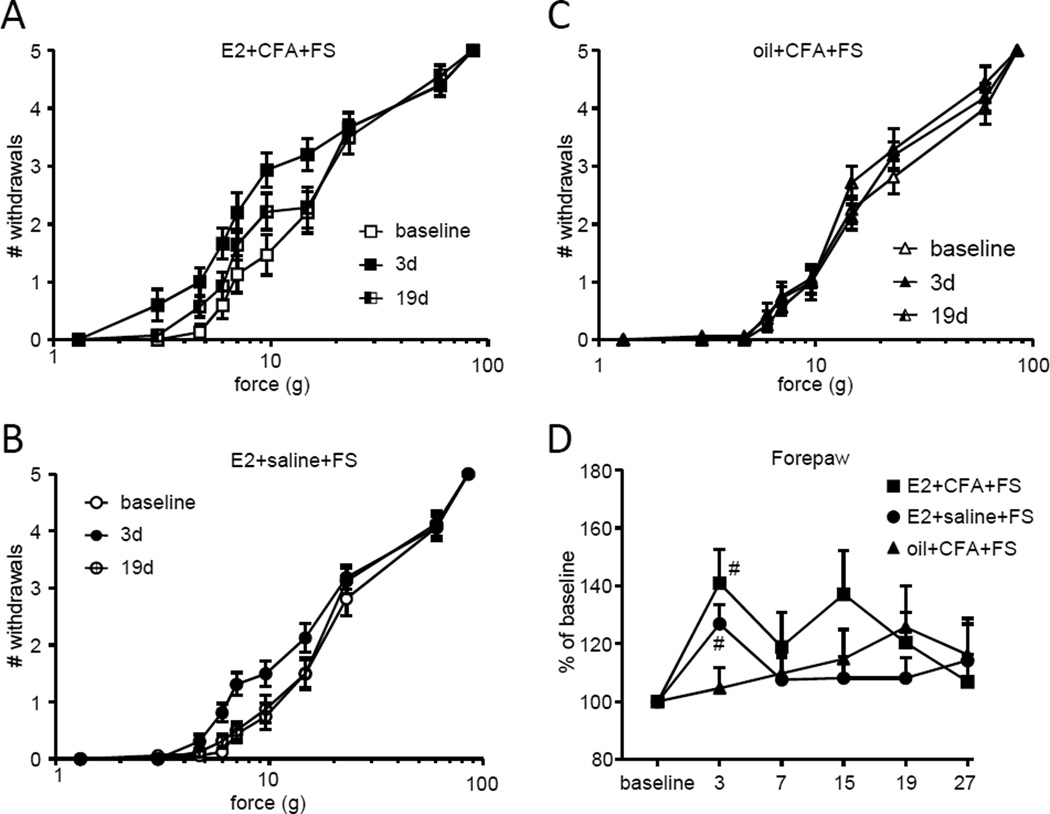
Colonic inflammation increases mechanosensitivity in the area over the lower back; a region of referred pain from the colon 61. Mechanosensitivity on the lower back was tested with von Frey filaments to determine if visceral hypersensitivity was associated with referred pain. In E2+CFA+FS rats there was a clear increase in the number of withdrawals to increasing forces of von Frey filaments on day 3 (Figure 4A). The increased mechanosensitivity returned to baseline on day 19. There was a much smaller increase in mechanosensitivity on day 3 in the E2+saline+FS rats (Figure 4B) and the oil+CFA+FS rats (Figure 4C). Comparing the area under the stimulus-response curve across time points showed significant mechanosensitivity through at least 15 days in the E2+CFA+FS rats (Kruskal-Wallis 1 way ANOVA, p<0.0001; Figure 4D). There was no overall increase in mechanosensitivity in the other cohorts (E2+saline+FS, p=0.36; oil+CFA+FS, p=0.19), although mechanosensitivity was increased by stress in these cohorts when comparing the 3 day timepoint to baseline (Wilcoxon Signed Rank test, p<0.005 for both).
Figure 4.
Referred pain is estrogen dependent. A,B,C: The number of withdrawals to graded intensities of von Frey stimulation to the lower back in E2+CFA+FS rats (A), E2+saline+FS rats (B) and oil+CFA+FS rats (C). D: The total number of withdrawals on each day normalized to the baseline response. *, **, *** p<0.05, 0.01, 0.001 vs. baseline, Dunn’s test following significant ANOVA; # p<0.005 vs. baseline, Wilcoxin Signed Rank test.
To determine if the increase in mechanosensitivity was confined to the dermatomes associated with referred pain from the colorectum or was a general increase in mechanosensitivity, the forepaws were tested with von Frey filaments. There was a tendency towards an increase in mechanosensitivity of the forepaw in the E2+CFA+FS rats (Figure 5A). When the data were pooled (AUC of stimulus-response curve) there was no overall significant effect (Kruskal-Wallis 1 way ANOVA, p=0.0942; Figure 5D), but the 3 day timepoint was significantly different from baseline (Wilcoxon Signed Rank test, p<0.005). Similarly, there was no overall increase in forepaw mechanosensitivity in the E2+saline+FS rats (Kruskal-Wallis 1 way ANOVA, p=0.14; Figure 5B,D), but sensitivity at 3 days was significantly greater than baseline (Wilcoxon Signed Rank test, p<0.005). There was no increase in mechanosensitivity in the oil+CFA+FS rats (Kruskal-Wallis 1 way ANOVA, p=0.94; Figure 5C,D). These data suggest that stress induces mild, short-lasting mechanosensitivity that is E2 dependent, but unrelated to the longer duration referred mechanohypersensitivity associated with the colorectal hypersensitivity.
Figure 5.
There is no mechanohypersensitivity in the forepaw. A,B,C: The number of withdrawals to graded intensities of von Frey stimulation to the forepaw in E2+CFA+FS rats (A), E2+saline+FS rats (B) and oil+CFA+FS rats (C). D: The total number of withdrawals on each day normalized to the baseline response. # p<0.005 vs. baseline, Wilcoxin Signed Rank test.
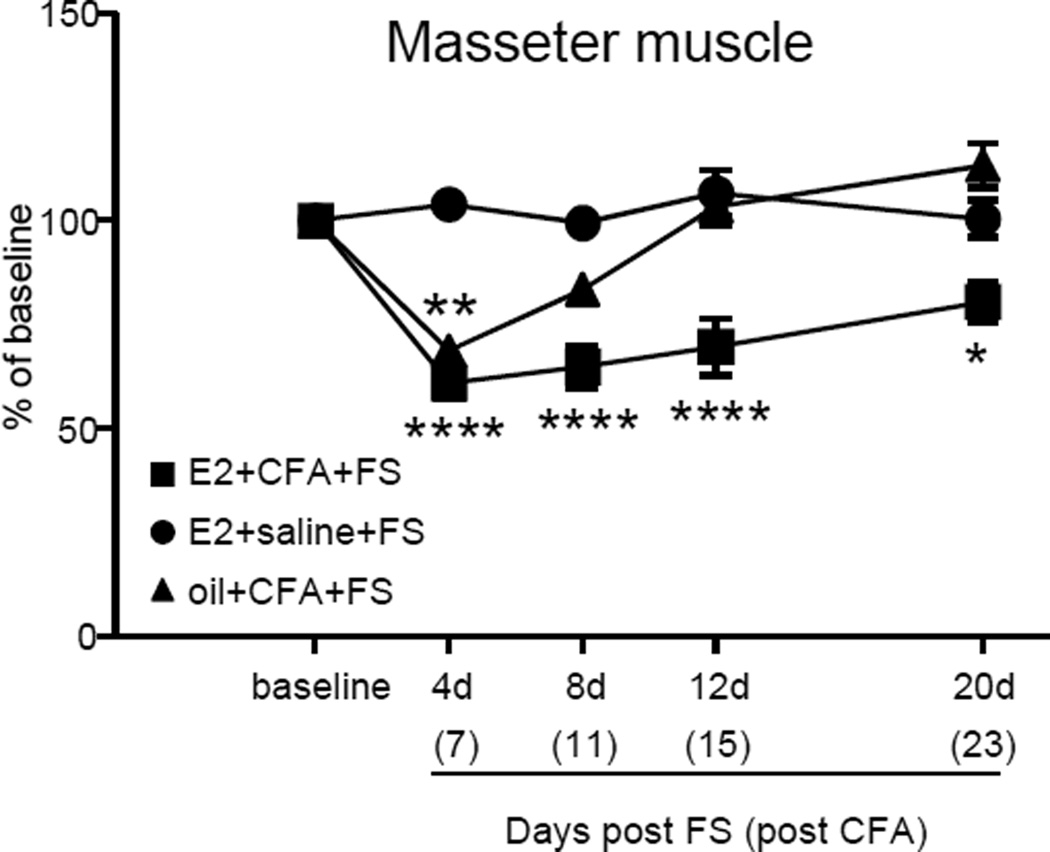
CFA injection into the masseter muscle increased mechanosensitivity to von Frey stimulation in the orofacial region. In rats injected with CFA there was approximately a 50% decrease in threshold 4 days after FS (7 days after CFA injection (see timeline in Figure 1); Figure 6). In E2+CFA+FS rats, the increased mechanosensitivity persisted approximately 20 days post FS (Kruskal-Wallis 1 way ANOVA, p<0.001). In contrast, in oil+CFA+FS rats, mechanosensitivity returned to baseline levels by 8 days post FS (Kruskal-Wallis 1 way ANOVA, p<0.001). Rats injected with saline (E2+saline+FS) in the masseter muscles showed no change in mechanosensitivity at any time point.
Figure 6.
Threshold for withdrawal from mechanical stimulation over the masseter muscles. E2 had no effect on the peak magnitude but prolonged the duration of hypersensitivity. Days post the end of the forced swim are shown on the abscissa, days post CFA injection in parentheses. *, **, **** p<0.05, 0.01, 0.0001 vs. baseline.
Discussion
Women are generally more susceptible to functional pain syndromes, many that can be exacerbated by stress. Since many functional pain syndromes overlap in their presentation and their underlying mechanisms are unclear, satisfactory pain management is difficult to achieve 1,62,66. It has been suggested that functional pain syndromes have altered CNS processing as a contributing factor, suggesting a potential mechanism for extensive comorbidities (see 43 for review). Indeed, patients with IBS or TMD have less central pain modulation than healthy controls 28,36. In animal studies, stress or gonadal hormone manipulation modulates nociceptive processing, especially of deep tissue, by acting at multiple levels of the neuraxis (see 30,35,39,60 for review). In the current study, we phenotype a new clinically relevant animal model to help extend mechanistic studies on overlapping pain conditions.
We report that muscle injury followed by forced swim stress induces visceral hypersensitivity that persists weeks longer than visceral hypersensitivity induced by forced swim alone. This same subchronic stress paradigm induced somatic thermal and chemical hypersensitivity that lasted approximately 8 days and sensitized trigeminal nucleus caudalis neurons to noxious stimulation of the temporomandibular joint 47,50,51. The unique aspect of our model is that requisite muscle injury prior to stress transitioned the stress-induced visceral hypersensitivity from acute or transient to chronic pain, persisting at least 6 weeks. Furthermore, the experiments were conducted in ovariectomized rats to allow examination of hormonal modulation. Estradiol replacement was necessary for the development and maintenance of the chronic visceral hypersensitivity, consistent with acute visceral pain studies, suggesting a pronociceptive effect of fluctuating levels of estradiol. Therefore, this model has face validity for the comorbidity of female TMD patients with chronic abdominal pain and thus can be useful for further studies to discern mechanisms underlying overlapping pain conditions.
Three conditions were necessary to induce the chronic comorbid visceral hypersensitivity. The first was muscle injury. Muscle inflammation alone did not induce visceral hypersensitivity. However, injury to muscle can be an important primer for chronic pain 14,15. For example, acute injury of the gastrocnemius muscle combined with stress evoked muscle hyperalgesia that persisted for more than a month and became bilateral 46. Injury to craniofacial muscle may provide an even more powerful and persistent trigger for chronic pain because masticatory muscles possess a much reduced ability to regenerate and repair following injury when compared to extracranial muscle 13,49. Since TMD affects 4–12% of the population and more than half of these patients complain of myalgia 42,58, craniofacial muscle injury provides a clinically relevant site of injury for this model.
The second was stress. Stress is considered a trigger for the exacerbation of IBS and the accompanying abdominal pain, as well as TMD. In rodents, stress induced visceral hypersensitivity of varying duration, mostly in animal models with a high anxiety genetic predisposition 7,24,55,67. Different stressors have slightly different effects but exacerbation of the visceral hypersensitivity by prior injury has never been previously demonstrated. In the current model when the stress paradigm began during the period of muscle inflammation-induced hyperalgesia the visceral hypersensitivity transitioned from transient to chronic, extending the duration of the visceral hypersensitivity.
The third condition was circulating estradiol. IBS is more prevalent in women than men and estrogens partially contribute to the severity of hypersensitivity. Several retrospective studies report a correlation between the menstrual cycle and IBS symptoms, the increase in symptoms occurring most often during menses or in the perimenstrual period when estradiol and progesterone levels drop 29,34,45,65. Furthermore, the incidence of IBS decreases following menopause, and increases with hormone replacement therapy in post-menopausal women 48,54, suggesting that either elevated gonadal hormones or fluctuating levels exacerbate abdominal pain.
Likewise, currently available animal models of pain arising from the viscera or the temporomandibular joint fluctuate across the estrous cycle and are sensitive to hormonal manipulation 3,9,18,31,33,37,41,47,53. Estrogen is pronociceptive in many visceral pain models in rodents. Ovariectomy decreased the magnitude of the visceromotor response to colonic or bladder distention which was restored by bolus estradiol replacement 31,32,53. An important observation from the current study is consistent with fluctuating plasma E2 concentration as a necessary component for the chronic hypersensitivity. Plasma E2 levels peak within a few hours following E2 injection in ovariectomized rats and decline over several days so repetitive injections every 4 days mimic the fluctuation of a normal estrous cycle 31. In rats with masseter inflammation plus stress, E2 was necessary to develop and maintain the chronic hypersensitivity. When the E2 injections ceased, the hypersensitivity reversed to the baseline level in 5 days (1 day after the first oil injection). By 10 days, the magnitude of the VMR further decreased as the plasma E2 concentration approached that in ovariectomized rats. Although E2 reverses the decrement in visceral sensitivity induced by ovariectomy, the sensitivity is not of comparable magnitude to the stress-induced visceral hypersensitivity observed in injured rats. The fact that rats repeatedly injected with E2 and stressed, but not inflamed, only had a transient hypersensitivity argues that the repetitive E2 injections maintained normal visceral sensitivity, but did not induce or maintain chronic visceral hypersensitivity.
In addition, the duration of the CFA+stress-induced muscle hyperalgesia was also E2-dependent. The masseter hypersensitivity persisted almost 2 weeks longer in the presence of E2 compared to rats with masseter inflammation plus stress that were treated with safflower oil.
Prior injury transitions stress-induced visceral hypersensitivity from transient to chronic
How the nervous system transitions from acute to chronic pain is poorly understood. A model of hyperalgesic priming in primary afferent neurons proposes that injury or inflammatory mediators sensitize the cell to subsequent proinflammatory mediators resulting in abnormally prolonged mechanical hyperalgesia 14,16,27. Projection of primary afferent-derived hypersensitivity into the spinal cord would implicate development of central sensitization.
The priming event in the current model was muscle injury. Since injury outside the temporomandibular joint region also contributed to comorbid visceral hypersensitivity this is likely not a phenomenon exclusively related to the orofacial region. While this study was initially designed to model comorbidity between TMD and IBS, fibromyalgia is also comorbid with IBS 52,62,66. Although biceps brachii inflammation is not a model of fibromyalgia, it suggests that a more generalized mechanism of injury plus stress inducing chronic visceral pain must be considered.
The severity and persistence of muscle inflammation induces central sensitization in the trigeminal nucleus caudalis or spinal dorsal horn immediately downstream from the muscle nociceptive afferents and in supraspinal sites involved in nociceptive processing. Several of these supraspinal nuclei overlap with sites affected by stress (e.g., hypothalamus, amygdala). An overlap between sites of central sensitization and central effects of psychological/physical stress could alter the response from one stimulus alone. Since there were indications of central sensitization in the spinal cord that process colorectal input (increased Fos expression) a change in descending modulation is suggested. CRD alone induced Fos in the spinal cord and as expected, Fos expression in the LS segments was greater than the TL segments. However, in the visceral hypersensitive rats, there was significantly greater spinal cord Fos expression consistent with increased processing of noxious visceral stimuli at the level of the spinal cord.
Central sensitization at the level of the spinal cord is further supported by the observations on referred pain and hypersensitivity. Cutaneous mechanohypersensitivity was observed in the area of referred pain from the colorectum. This referred hypersensitivity persisted at least 2 weeks in the E2+CFA+FS rats, considerably shorter than the duration of the visceral hypersensitivity. One possible explanation is that withdrawal from the cutaneous stimulus is largely, if not exclusively, spinally mediated while the VMR is dependent on supraspinal processing. Differences in plasticity of spinal vs. supraspinal structures could contribute to differences in hypersensitivity between visceral and cutaneous tissue. In addition, there was a shorter duration mechanohypersensitivity induced by stress that was independent of E2 or CFA. E2+stress also induced a short duration mechanohypersensitivity in the forepaw, but it was of lesser magnitude than the back. These data suggest that visceral hypersensitivity is associated with referred pain, supporting reports of referred pain following colonic inflammation and validating the usefulness of our model 5,61.
In summary, a unique model of overlapping pain conditions is presented. It has face validity with features described in clinical reports of comorbid pain conditions observed the clinic. Future studies will utilize this model to investigate the peripheral and central mechanisms underlying overlapping pain conditions.
Perspective.
The majority of patients with TMD report symptoms consistent with IBS. Stress and female prevalence are common to both conditions. In a new experimental paradigm in ovariectomized rats with estradiol replacement, masseter inflammation followed by stress induces visceral hypersensitivity that persists for months modeling these comorbid pain conditions.
Acknowledgments
Grant support: NIH R21 DE022235, NIH R01 NS037424
Abbreviations
- AUC
area under the curve
- CFA
complete Freund’s adjuvant
- CRD
olorectal distention
- E2
estradiol
- FS
forced swim
- VMR
visceromotor response
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclosures: The authors declare they have nothing to disclose.
Author contributions:
RJT: study concept and design, data analysis, drafting of manuscript, obtained funding, study supervision
DYC: data acquisition, data analysis
JK: data acquisition, technical support
SP: data acquisition, technical support
YJ: critical revision of the manuscript for intellectual content
SGD: study concept and design, critical revision of the manuscript for intellectual content
DD: study concept and design, data analysis, obtained funding, critical revision of the manuscript for intellectual content
Reference List
- 1.Aaron LA, Burke MM, Buchwald D. Overlapping conditions among patients with chronic fatigue syndrome, fibromyalgia, and temporomandibular disorder. Arch Intern Med. 2000;160:221–227. doi: 10.1001/archinte.160.2.221. [DOI] [PubMed] [Google Scholar]
- 2.Berkley KJ. Sex differences in pain. Behav Brain Sci. 1997;20:371–380. doi: 10.1017/s0140525x97221485. [DOI] [PubMed] [Google Scholar]
- 3.Berkley KJ, McAllister SL, Accius BE, Winnard KP. Endometriosis-induced vaginal hyperalgesia in the rat: effect of estropause, ovariectomy, and estradiol replacement. Pain. 2007;132:S150–S159. doi: 10.1016/j.pain.2007.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Botelho AP, Gameiro GH, Tuma CE, Marcondes FK, Arruda Veiga MC. The effects of acute restraint stress on nociceptive responses evoked by the injection of formalin into the temporomandibular joint of female rats. Stress. 2010;13:269–275. doi: 10.3109/10253890903362645. [DOI] [PubMed] [Google Scholar]
- 5.Bourdu S, Dapoigny M, Chapuy E, Artigue F, Vasson MP, Dechelotte P, Bommelaer G, Eschalier A, Ardid D. Rectal instillation of butyrate provides a novel clinically relevant model of noninflammatory colonic hypersensitivity in rats. Gastroenterology. 2005;128:1996–2008. doi: 10.1053/j.gastro.2005.03.082. [DOI] [PubMed] [Google Scholar]
- 6.Bradesi S, Svensson CI, Steinauer J, Pothoulakis C, Yaksh TL, Mayer EA. Role of spinal microglia in visceral hyperalgesia and NK1R up-regulation in a rat model of chronic stress. Gastroenterology. 2009;136:1339–1348. doi: 10.1053/j.gastro.2008.12.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bradesi S, Schwetz I, Ennes HS, Lamy CMR, Ohning G, Fanselow M, Pothoulakis C, McRoberts JA, Mayer EA. Repeated exposure to water avoidance stress in rats: a new model for sustained visceral hyperalgesia. Am J Physiol Gastrointest Liver Physiol. 2005;289:G42–G53. doi: 10.1152/ajpgi.00500.2004. [DOI] [PubMed] [Google Scholar]
- 8.Butler RK, Finn DP. Stress-induced analgesia. Prog Neurobiol. 2009;88:184–202. doi: 10.1016/j.pneurobio.2009.04.003. [DOI] [PubMed] [Google Scholar]
- 9.Cairns BE, Sim Y, Bereiter DA, Sessle BJ, Hu JW. Influence of sex on reflex jaw muscle activity evoked from the rat temporomandibular joint. Brain Res. 2002;957:338–344. doi: 10.1016/s0006-8993(02)03671-5. [DOI] [PubMed] [Google Scholar]
- 10.Chang L, Drossman DA. Irritable Bowel Syndrome and Related Functional Disorders. In: Mayer EA, Bushnell MC, editors. Functional Pain Syndromes: Presentation and Pathphysiology. Seattle: IASP Press; 2009. pp. 87–120. [Google Scholar]
- 11.Chang L, Mayer EA, Labus JS, Schmulson M, Lee OY, Olivas TI, Stains J, Naliboff BD. Effect of sex on perception of rectosigmoid stimuli in irritable bowel syndrome. Am J Physiol Regul Integr Comp Physiol. 2006;291:R277–R284. doi: 10.1152/ajpregu.00729.2005. [DOI] [PubMed] [Google Scholar]
- 12.Chen J, Winston JH, Sarna SK. Neurological and cellular regulation of visceral hypersensitivity induced by chronic stress and colonic inflammation in rats. Neuroscience. 2013;248:469–478. doi: 10.1016/j.neuroscience.2013.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dessem D, Lovering RM. Repeated muscle injury as a presumptive trigger for chronic masticatory muscle pain. Pain Res Treat. 2011:647967–642011. doi: 10.1155/2011/647967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dina OA, Green PG, Levine JD. Role of interleukin-6 in chronic muscle hyperalgesic priming. Neuroscience. 2008;152:521–525. doi: 10.1016/j.neuroscience.2008.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dina OA, Levine JD, Green PG. Muscle inflammation induces a protein kinase Cepsilon-dependent chronic-latent muscle pain. J Pain. 2008;9:457–462. doi: 10.1016/j.jpain.2008.01.328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ferrari LF, Bogen O, Levine JD. Role of nociceptor aCaMKII in transition from acute to chronic pain (hyperalgesic priming) in male and female rats. J Neurosci. 2013;33:11002–11011. doi: 10.1523/JNEUROSCI.1785-13.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fillingim RB, King CD, Ribeiro-Dasilva MC, Rahim-Williams B, Riley JL., III Sex, gender, and pain: a review of recent clinical and experimental findings. J Pain. 2009;10:447–485. doi: 10.1016/j.jpain.2008.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fischer L, Torres-Chavez KE, Clemente-Napimoga JT, Jorge D, Arsati F, Arruda Veiga MC, Tambeli CH. The Influence of Sex and Ovarian Hormones on Temporomandibular Joint Nociception in Rats. J Pain. 2008;9:630–638. doi: 10.1016/j.jpain.2008.02.006. [DOI] [PubMed] [Google Scholar]
- 19.Fukudo S. Stress and Visceral Pain: Focusing on Irritable Bowel Syndrome. Pain. 2013;154:s63–s70. doi: 10.1016/j.pain.2013.09.008. [DOI] [PubMed] [Google Scholar]
- 20.Gameiro GH, da Silva AA, Nouer DF, Ferraz de Arruda Veiga MC. How may stressful experiences contribute to the development of temporomandibular disorders? Clin Oral Investig. 2006;10:261–268. doi: 10.1007/s00784-006-0064-1. [DOI] [PubMed] [Google Scholar]
- 21.Green PG, Alvarez P, Gear RW, Mendoza D, Levine JD. Further validation of a model of fibromyalgia syndrome in the rat. J Pain. 2011;12:811–818. doi: 10.1016/j.jpain.2011.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Greenspan JD, Traub RJ. Gender differences in pain and its relief. In: McMahon S, Koltzenburg M, Tracey I, Turk D, editors. Wall and Melzack's Textbook of Pain: Expert Consult-Online and Print. 6th edition. Amsterdam: Elsevier; 2013. pp. 221–231. [Google Scholar]
- 23.Harter MC, Conway KP, Merikangas KR. Associations between anxiety disorders and physical illness. Eur Arch Psychiatry Clin Neurosci. 2003;253:313–320. doi: 10.1007/s00406-003-0449-y. [DOI] [PubMed] [Google Scholar]
- 24.He YQ, Chen Q, Ji L, Wang ZG, Bai ZH, Stephens RL, Jr, Yang M. PKCgamma receptor mediates visceral nociception and hyperalgesia following exposure to PTSD-like stress in the spinal cord of rats. Mol Pain. 2013;9:35. doi: 10.1186/1744-8069-9-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Heitkemper MM, Chang L. Do fluctuations in ovarian hormones affect gastrointestinal symptoms in women with irritable bowel syndrome? Gend Med. 2009;6(Suppl 2):152–167. doi: 10.1016/j.genm.2009.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Heitkemper MM, Jarrett M, Caudell KA, Bond E. Women with gastrointestinal symptoms: implications for nursing research and practice. Gastroenterol Nurs. 1998;21:52–58. doi: 10.1097/00001610-199803000-00005. [DOI] [PubMed] [Google Scholar]
- 27.Hendrich J, Joseph EK, Chen X, Bogen O, Levine JD. Electrophysiological correlates of hyperalgesic priming in vitro and in vivo. Pain. 2013:2207–2215. doi: 10.1016/j.pain.2013.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Heymen S, Maixner W, Whitehead WE, Klatzkin RR, Mechlin B, Light KC. Central processing of noxious somatic stimuli in patients with irritable bowel syndrome compared with healthy controls. Clin J Pain. 2010;26:104–109. doi: 10.1097/AJP.0b013e3181bff800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Houghton LA, Lea R, Jackson N, Whorwell PJ. The menstrual cycle affects rectal sensitivity in patients with irritable bowel syndrome but not healthy volunteers. Gut. 2002;50:471–474. doi: 10.1136/gut.50.4.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Imbe H, Iwai-Liao Y, Senba E. Stress-induced hyperalgesia: animal models and putative mechanisms. Front Biosci. 2006;11:2179–2192. doi: 10.2741/1960. [DOI] [PubMed] [Google Scholar]
- 31.Ji Y, Murphy AZ, Traub RJ. Estrogen modulates the visceromotor reflex and responses of spinal dorsal horn neurons to colorectal stimulation in the rat. J Neurosci. 2003;23:3908–3915. doi: 10.1523/JNEUROSCI.23-09-03908.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ji Y, Tang B, Traub RJ. Estrogen increases and progesterone decreases behavioral and neuronal responses to colorectal distention following colonic inflammation in the rat. Pain. 2005;117:433–442. doi: 10.1016/j.pain.2005.07.011. [DOI] [PubMed] [Google Scholar]
- 33.Ji Y, Tang B, Traub RJ. The visceromotor response to colorectal distention fluctuates with the estrous cycle in rats. Neuroscience. 2008;154:1562–1567. doi: 10.1016/j.neuroscience.2008.04.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kane SV, Sable K, Hanauer SB. The menstrual cycle and its effect on inflammatory bowel disease and irritable bowel syndrome: a prevalence study. Am J Gastroenterol. 1998;93:1867–1872. doi: 10.1111/j.1572-0241.1998.540_i.x. [DOI] [PubMed] [Google Scholar]
- 35.Kennedy PJ, Clarke G, Quigley EMM, Groeger JA, Dinan TG, Cryan JF. Gut memories: Towards a cognitive neurobiology of irritable bowel syndrome. Neuroscience & Biobehavioral Reviews. 2012;36:310–340. doi: 10.1016/j.neubiorev.2011.07.001. [DOI] [PubMed] [Google Scholar]
- 36.King CD, Wong F, Currie T, Mauderli AP, Fillingim RB, Riley JL., III Deficiency in endogenous modulation of prolonged heat pain in patients with irritable bowel syndrome and temporomandibular disorder. Pain. 2009;143:172–178. doi: 10.1016/j.pain.2008.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kramer PR, Bellinger LL. The effects of cycling levels of 17beta-estradiol and progesterone on the magnitude of temporomandibular joint-induced nociception. Endocrinology. 2009;150:3680–3689. doi: 10.1210/en.2008-1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Larauche M, Mulak A, Kim YS, Labus J, Million M, Tache Y. Visceral analgesia induced by acute and repeated water avoidance stress in rats: sex difference in opioid involvement. Neurogastroenterol Motil. 2012;24 doi: 10.1111/j.1365-2982.2012.01980.x. 1031–e547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Larauche M, Mulak A, Tache Y. Stress and visceral pain: from animal models to clinical therapies. Exp Neurol. 2012;233:49–67. doi: 10.1016/j.expneurol.2011.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.LeResche L, Mancl L, Sherman JJ, Gandara B, Dworkin SF. Changes in temporomandibular pain and other symptoms across the menstrual cycle. Pain. 2003;106:253–261. doi: 10.1016/j.pain.2003.06.001. [DOI] [PubMed] [Google Scholar]
- 41.Liu B, Eisenach JC, Tong C. Chronic estrogen sensitizes a subset of mechanosensitive afferents innervating the uterine cervix. J Neurophysiol. 2005;93:2167–2173. doi: 10.1152/jn.01012.2004. [DOI] [PubMed] [Google Scholar]
- 42.Machado LP, Nery CG, Leles CR, Nery MB, Okeson JP. The prevalence of clinical diagnostic groups in patients with temporomandibular disorders. Cranio. 2009;27:194–199. doi: 10.1179/crn.2009.029. [DOI] [PubMed] [Google Scholar]
- 43.Mayer EA, Bushnell MC, editors. Functional pain syndromes: presentation and pathophysiology. Seattle: IASP Press; 2009. [Google Scholar]
- 44.Mayer EA, Craske M, Naliboff BD. Depression, anxiety, and the gastrointestinal system. J Clin Psychiatry. 2001;62(Suppl 8):28–36. [PubMed] [Google Scholar]
- 45.Moore J, Barlow D, Jewell D, Kennedy S. Do gastrointestinal symptoms vary with the menstrual cycle? Br J Obstet Gynaecol. 1998;105:1322–1325. doi: 10.1111/j.1471-0528.1998.tb10014.x. [DOI] [PubMed] [Google Scholar]
- 46.Nasu T, Taguchi T, Mizumura K. Persistent deep mechanical hyperalgesia induced by repeated cold stress in rats. Eur J Pain. 2010;14:236–244. doi: 10.1016/j.ejpain.2009.05.009. [DOI] [PubMed] [Google Scholar]
- 47.Okamoto K, Thompson R, Katagiri A, Bereiter DA. Estrogen status and psychophysical stress modify temporomandibular joint input to medullary dorsal horn neurons in a lamina-specific manner in female rats. Pain. 2013;154:1057–1064. doi: 10.1016/j.pain.2013.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Palsson O, Whitehead W, Barghout V, Levy R, Feld A, Von Korff M, Garner M, Drossman D, Turner M. IBS severity and health-related quality of life improve with age in women but not in men. Am J Gastroenterol. 2003;98:S272. [Google Scholar]
- 49.Pavlath GK, Thaloor D, Rando TA, Cheong M, English AW, Zheng B. Heterogeneity among muscle precursor cells in adult skeletal muscles with differing regenerative capacities. Dev Dyn. 1998;212:495–508. doi: 10.1002/(SICI)1097-0177(199808)212:4<495::AID-AJA3>3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- 50.Quintero L, Cuesta MC, Silva JA, Arcaya JL, Pinerua-Suhaibar L, Maixner W, Suarez-Roca H. Repeated swim stress increases pain-induced expression of c-Fos in the rat lumbar cord. Brain Res. 2003;965:259–268. doi: 10.1016/s0006-8993(02)04224-5. [DOI] [PubMed] [Google Scholar]
- 51.Quintero L, Moreno M, Avila C, Arcaya J, Maixner W, Suarez-Roca H. Long-lasting delayed hyperalgesia after subchronic swim stress. Pharmacol Biochem Behav. 2000;67:449–458. doi: 10.1016/s0091-3057(00)00374-9. [DOI] [PubMed] [Google Scholar]
- 52.Riedl A, Schmidtmann M, Stengel A, Goebel M, Wisser AS, Klapp BF, Monnikes H. Somatic comorbidities of irritable bowel syndrome: a systematic analysis. J Psychosom Res. 2008;64:573–582. doi: 10.1016/j.jpsychores.2008.02.021. [DOI] [PubMed] [Google Scholar]
- 53.Robbins MT, Mebane H, Ball CL, Shaffer AD, Ness TJ. Effect of estrogen on bladder nociception in rats. J Urol. 2010;183:1201–1205. doi: 10.1016/j.juro.2009.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ruigomez A, Garcia Rodriguez LA, Johansson S, Wallander MA. Is hormone replacement therapy associated with an increased risk of irritable bowel syndrome? Maturitas. 2003;44:133–140. doi: 10.1016/s0378-5122(02)00321-3. [DOI] [PubMed] [Google Scholar]
- 55.Schwetz I, Bradesi S, McRoberts JA, Sablad M, Miller JC, Zhou H, Ohning G, Mayer EA. Delayed stress-induced colonic hypersensitivity in male Wistar rats: role of neurokinin-1 and corticotropin-releasing factor-1 receptors. Am J Physiol Gastrointest Liver Physiol. 2004;286:G683–G691. doi: 10.1152/ajpgi.00358.2003. [DOI] [PubMed] [Google Scholar]
- 56.Sherman JJ, LeResche L, Mancl LA, Huggins K, Sage JC, Dworkin SF. Cyclic effects on experimental pain response in women with temporomandibular disorders. J Orofac Pain. 2005;19:133–143. [PubMed] [Google Scholar]
- 57.Slomianka L, West MJ. Estimators of the precision of stereological estimates: an example based on the CA1 pyramidal cell layer of rats. Neuroscience. 2005;136:757–767. doi: 10.1016/j.neuroscience.2005.06.086. [DOI] [PubMed] [Google Scholar]
- 58.Stohler CS. Muscle-related temporomandibular disorders. J Orofac Pain. 1999;13:273–284. [PubMed] [Google Scholar]
- 59.Traub RJ, Cao D-Y, Dorsey SG, Dessem D. Stress and estrogen induce persistent visceral hypersensitivity following masseter muscle inflammation: a new model of overlapping pain conditions. Society for Neuroscience Abstract. 2013 [Google Scholar]
- 60.Traub RJ, Ji Y. Sex differences and hormonal modulation of deep tissue pain. Front Neuroendocrinol. 2013;34:350–366. doi: 10.1016/j.yfrne.2013.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Traub RJ, Tang B, Ji Y, Pandya S, Yfantis H, Sun Y. A rat model of chronic post-inflammatory visceral pain induced by deoxycholic acid. Gastroenterology. 2008;135:2075–2083. doi: 10.1053/j.gastro.2008.08.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Warren JW, Langenberg P, Clauw DJ. The number of existing functional somatic syndromes (FSSs) is an important risk factor for new, different FSSs. J Psychosom Res. 2013;74:12–17. doi: 10.1016/j.jpsychores.2012.09.002. [DOI] [PubMed] [Google Scholar]
- 63.Warren MP, Fried JL. Temporomandibular disorders and hormones in women. Cells Tissues Organs. 2001;169:187–192. doi: 10.1159/000047881. [DOI] [PubMed] [Google Scholar]
- 64.West MJ, Slomianka L, Gundersen HJ. Unbiased stereological estimation of the total number of neurons in thesubdivisions of the rat hippocampus using the optical fractionator. Anat Rec. 1991;231:482–497. doi: 10.1002/ar.1092310411. [DOI] [PubMed] [Google Scholar]
- 65.Whitehead WE, Cheskin LJ, Heller BR, Robinson JC, Crowell MD, Benjamin C, Schuster MM. Evidence for exacerbation of irritable bowel syndrome during menses. Gastroenterology. 1990;98:1485–1489. doi: 10.1016/0016-5085(90)91079-l. [DOI] [PubMed] [Google Scholar]
- 66.Whitehead WE, Palsson O, Jones KR. Systematic review of the comorbidity of irritable bowel syndrome with other disorders: what are the causes and implications? Gastroenterology. 2002;122:1140–1156. doi: 10.1053/gast.2002.32392. [DOI] [PubMed] [Google Scholar]
- 67.Winston JH, Xu GY, Sarna SK. Adrenergic stimulation mediates visceral hypersensitivity to colorectal distension following heterotypic chronic stress. Gastroenterology. 2010;138:294–304. doi: 10.1053/j.gastro.2009.09.054. [DOI] [PMC free article] [PubMed] [Google Scholar]