Abstract
Angiogenesis is an important tissue-level program supporting the growth of highly aggressive cancers and early-stage metastases. However, rapid emergence of resistance to antiangiogenic therapies, such as bevacizumab, greatly limits the clinical utility of these promising approaches. The mechanisms of resistance to antiangiogenic therapy remain incompletely understood. The tumor microenvironment has been demonstrated to be a source of broad therapeutic resistance in multiple cancers. Much of the interaction between the cells comprising a tumor and their microenvironment is driven by integrins. Notably, signaling downstream of integrins in tumor cells promotes fundamental programs vital to aggressive cancer biology, including proliferation, growth, invasion, and survival signaling. These functions then can contribute to malignant phenotypes, including metastasis, therapy resistance, epithelial-to-mesenchymal transition, and angiogenesis. Accordingly, we found β1 integrin to be functionally upregulated in tumor specimens from patients after bevacizumab failure and in xenograft models of bevacizumab resistance. Inhibition of β1 in tumor cells with stable gene knockdown or treatment with OS2966, a neutralizing β1 integrin monoclonal antibody, attenuated aggressive tumor phenotypes in vitro and blocked growth of bevacizumab-resistant tumor xenografts in vivo. Thus, β1 integrins promote resistance to antiangiogenic therapy through potentiation of multiple malignant programs facilitated by interactions with the tumor microenvironment. The elucidation of this mechanism creates an outstanding opportunity for improving patient outcomes in cancer.
Introduction
Aggressive cancers require adequate vasculature to support continued tumor progression. Tumor vasculature can be derived from preexisting host blood vessels (vessel cooption) or through new vessel growth (angiogenesis). There is likely to be a spectrum of vessel utilization along this continuum depending on various contexts (1). The recognition of the critical importance of vascularization in cancer progression has led to the hope for a new standard of care through the development of antiangiogenic agents such as bevacizumab. However, these drugs, mostly focused on the VEGF pathway, have not produced the clinical outcomes expected on the basis of the dramatic preclinical results in mice. This was most recently demonstrated in a randomized phase III trial in patients with newly diagnosed glioblastoma (2). The reasons for the disparity between preclinical and clinical results with this therapeutic modality are multiple; however, the use of animal models, which themselves rely heavily upon angiogenesis for growth, such as subcutaneous cell line–derived xenografts, likely represents a selection bias for highly angiogenic, VEGF-dependent tumors.
Integrins and Their Role in Cellular Interaction with the Microenvironment in Inflammation and Cancer
Although angiogenesis during wound healing is tightly regulated and self-limiting, angiogenesis associated with chronic inflammation and cancer is often persistent and abnormal. However, many of the molecules that regulate angiogenesis in wound healing, such as integrin αvβ3, also regulate pathologic angiogenesis associated with chronic inflammation and cancer. Increasing evidence points to causative links between inflammation and cancer and suggests that inflammation promotes the angiogenic switch in tumors (3).
This connection between inflammation and cancer underscores the complexity of tumorigenesis and the fact that models that define cancer as driven primarily by a minor population of genetically aberrant cells violates the basic notions of systems biology, evolution, and pathophysiology. Cancer is a systems-level process defined by growth of malignant cells in an organ, which breaches normal tissue boundaries established by the basement membrane. In other words, cancer requires tumor cells directly interacting with cellular and structural components of the surrounding stroma. Dolberg and Bissell demonstrated the importance of this “tumor microenvironment” when they studied cells infected with the rous sarcoma virus (4). In vitro and in early embryonic-stage eggs, the cells grew aggressively. However, when transplanted into late-stage eggs, the genetically malignant program was overridden by the microenvironmental context and the cells incorporated into normal tissue.
Integrins are a major mediator of these interactions between the cells comprising a tumor and their microenvironment. The cells engaging in these interactions include monocytes, in which intratumoral trafficking and subsequent promotion of angiogenesis are mediated by integrin α4β1 (VLA4; ref. 5). Integrins also contribute to macrophage polarization, with β3 integrin promoting the M1 cytotoxic immunostimulating macrophage phenotype rather than the M2 immunosuppressive, proinvasive phenotype (6). For tumor cells, these interactions with the microenvironment are crucial to the development of malignant features, with reversion of the malignant phenotype demonstrated in breast cancer cells by inhibition of β1 integrin in culture and in vivo (7). These findings emphasize the importance of interactions between the cells comprising a tumor with microenvironmental integrin ligands in the extracellular matrix for tumor progression. Integrins are also crucial to VEGF-dependent and VEGF-independent angiogenesis. On endothelial cells, reciprocal interactions between integrin αvβ3 and VEGFR2 are particularly important during tumor vascularization (8). The αv integrin expression on endothelial cells is also stimulated by VEGF-independent angiogenic growth factors such as bFGF, TNF-α, and interleukin (IL)-8 (9). Finally, mesenchymal aspects of angiogenesis, including endothelial cell invasion and vascular remodeling, rely on α5β1 integrin (10).
We now understand the multiple parallel signaling pathways downstream of integrin engagement that promote tumor growth, including FAK, ERK/MAP kinase, Src, Akt, and Ras (11–14). These pathways are upregulated as β1 levels increase. Indeed, several studies have demonstrated the expression of β1 integrin to correlate with malignant features, including metastasis (15–17). Accordingly, β1 integrin signaling in tumor cells has been shown to promote resistance to multiple treatment modalities, including cytotoxic drugs, radiotherapy (18), targeted therapies such as trastuzumab (19) and lapatinib (20), and oncolytic viruses (21).
β1 Integrin and Resistance to Antiangiogenic Therapy
Rubenstein and colleagues demonstrated that treatment of experimental glioma xenografts with bevacizumab increased invasiveness of tumor cells into normal adjacent brain parenchyma via vessel cooption (22). Nearly a decade later, similar observations were made by Paez-Ribes and colleagues, including increased metastasis (23). These findings can be understood in the context of the vascularization continuum (Fig. 1). In the face of VEGF neutralization and inhibition of neovascularization via angiogenesis, tumor cells must adapt by becoming motile and hijacking the existing vasculature to survive and grow. As part of this invasive process, some cells may intravasate and enter the bloodstream, which may explain the findings of increased metastasis in many studies (15, 16, 23). Importantly, β1 integrins have been recognized to play a major role in metastases (17, 23), tumor growth (24, 25), and invasion (26). Furthermore, Carbonell and colleagues recently identified the β1 integrin–dependent mechanism of vessel cooption driven by adhesion to the vascular basement membrane (27). Vessel cooption provides an instant vasculature for newly metastatic or locally invasive cancer cells. Thus, resistance to antiangiogenic therapy can be explained by cellular stress triggering the Darwinian survival imperative facilitated by enhanced β1 integrin activities. We therefore explored the hypothesis that resistance to antiangiogenic agents such as bevacizumab is promoted by the spectrum of malignant features driven by β1 integrin signaling through interactions with the tumor microenvironment.
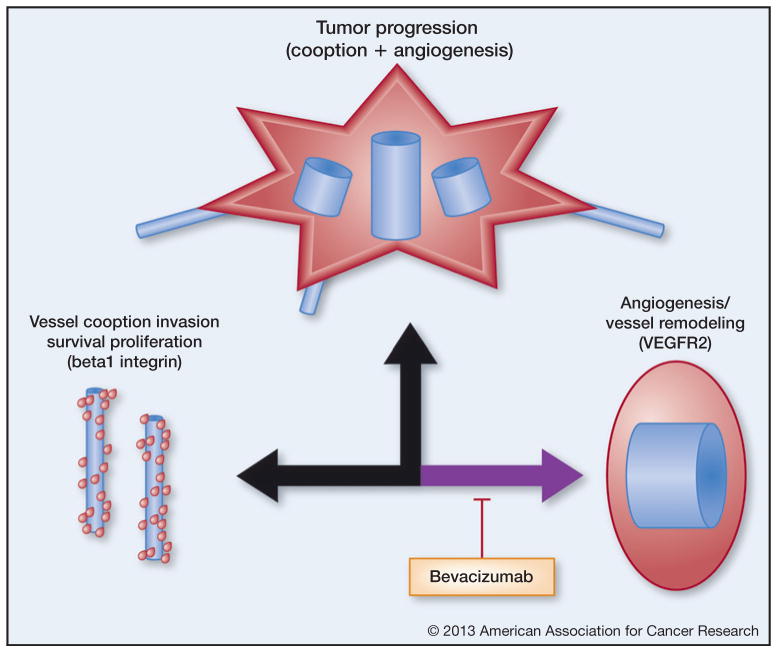
Figure 1.
Summary of the vascularization continuum. The vascularization continuum recognizes the multiple mechanisms by which tumor cells will vascularize to survive and grow (simplified here to only consider dominant mechanisms of vessel cooption and remodeling). Aggressive tumor growth is characterized by angiogenesis, often with cooption at the growth fronts. When VEGF-dependent angiogenesis is blocked with agents such as bevacizumab, the vascularization continuum shunts toward vessel cooption and related β1 integrin-mediated tumor cell functions. Blue denotes blood vessels, and red denotes cancer cells.
We have previously reported diverging radiologic morphology in bevacizumab-resistant glioblastomas (BRG). Some BRGs show a nonenhancing infiltrating pattern, which we found to have upregulated levels of β1 integrin when analyzed with microarray and PCR (28). These results were further validated with immunhistochemistry, which demonstrated increased staining for β1 integrin in bevacizumab-resistant patient specimens, when matched with their respective pre–bevacizumab-treated specimens. Furthermore, flow cytometry of cells from the same bevacizumab-resistant tumors also showed increased levels of β1 integrin in primary cultures compared with cells from bevacizumab-naïve glioblastomas. These findings were replicated in our uniquely developed U87MG cell line–derived xenograft model of bevacizumab resistance.
Because integrin signaling reflects integrin turnover in focal adhesions, we supplemented our static assays of β1 integrin expression with dynamic assessment of β1 integrin turnover. By engineering bevacizumab-resistant and -naïve cell lines to express the β1 integrin–GFP fusion protein, we were able to apply fluorescence recovery after photobleaching to quantify the rate of integrin turnover in the focal adhesions formed. Complementary to our findings above, β1 integrin turnover as well as mobility was increased in the BRGs when compared with the naïve control cell lines. Cell adhesion assays further demonstrated this phenomenon when BRG cell lines showed increased adhesion to the extracellular matrix as compared with the naïve cell lines. Our findings therefore support the functional upregulation of β1 integrin in glioblastomas that evolve to become resistant to antiangiogenic therapy.
We found increased hypoxia in our BRG patient samples compared with their paired naïve samples, which directly correlated with the increased β1 integrin in the BRGs. In vitro studies of U87MGs and two non–central nervous system carcinoma cell lines, HT-1080 and MDA-MB-231, supported this finding with a significant increase in the expression of β1 integrin following hypoxia, suggesting a conserved survival mechanism. The hypoxia that results from antiangiogenic therapy with bevacizumab therefore may play a major role in β1 integrin upregulation.
The role for β1 integrin in the aggressive mesenchymal phenotype of BRGs was further characterized by allowing bevacizumab-resistant cell lines to express short hairpin RNA (shRNA)-targeting β1 integrin (shBETA1) in vitro. This led to a more epithelioid morphology with increased cell size in shBETA1 clones. Functionally, these cells lacked mesenchymal characteristics, as shown by static and dynamic in vitro assays demonstrating decreased adhesion, cell spreading, and migration. Furthermore, the knockdown cells displayed reduced in vitro proliferation. Spheroid formation in acidic media, a vital adaptive response used by tumor cells, was attenuated in U87-shBETA1 and BRG-shBETA1 cells compared with controls. Inhibiting glioma cells with OS2966 in vitro had similar outcomes as the knockdown in all of the above assays.
We then established preclinical proof-of-concept for the critical role of β1 integrin in BRGs in multiple xenograft models. Systemic treatment of subcutaneous BRG-derived xenografts with two different doses of OS2966 led to significantly smaller tumors than placebo. This treatment also proved effective in orthotopic BRG–derived xenografts. Full-dose combination therapy with bevacizumab and OS2966 led to cutaneous ulceration of 100% of U87MG subcutaneous xenografts, suggestive of synergy. We then alternated low-dose (1 mg/kg) bevacizumab and OS2966 and compared it with the standard dose of 10 mg/kg of bevacizumab alone. The combination therapy proved equally effective, with similar rates of complete regression in both groups. This may help reduce the morbidity of antiangiogenic approaches.
In sum, we have established that β1 integrin is a critical driving force in therapeutic resistance and is a potentially promising adjunctive target in combination with antiangiogenic therapies such as bevacizumab. Thus, targeting β1 integrins holds outstanding potential for improving patient outcomes in glioblastoma.
Current Challenges and Limitations of Targeting β1 Integrin
Successful pharmacologic targeting of β1 integrin will require the development of specialized molecular inhibitors or humanized monoclonal antibodies. Although the ubiquitous cellular expression of integrins may raise concerns about the safety of this approach, chronic administration of up to 20 mg/kg hamster anti-mouse β1 integrin to nude mice resulted in no observable toxicity for up to 4 weeks (7). Accordingly, OS2966, a first-in-class, ligand-selective, humanized and deimmunized neutralizing β1 integrin monoclonal antibody, is currently under development for clinical testing.
In the case of glioblastoma, if penetration of β1 integrin blocking antibodies through the blood–brain barrier (BBB) after parenteral administration proved suboptimal, intratumoral delivery through strategies such as convection-enhanced or selective endovascular delivery could be considered as well. However, we have also shown in pilot studies that systemically delivered OS2966 successfully targets orthotopic U87 xenografts through the BBB (A. Jahangiri and M. Aghi; unpublished data).
Implications and Future Directions
Our findings underscore the importance of β1 integrin in resistance to antiangiogenic therapy and are part of an emerging literature showing the importance of β1 integrin in the evolution of tumor resistance to multiple therapeutic modalities (18–20). This is consistent with the ability of β1 integrin to stimulate invasion and proliferation and its central role in engaging the microenvironment owing to its ability to bind multiple extracellular matrix and cell adhesion molecules.
Future work will need to identify more detailed mechanisms by which β1 integrin is upregulated during therapeutic resistance. These mechanisms could be transcriptional or post-translational, and regulators could include hypoxia or the VEGF depletion caused by antiangiogenic therapy. It will also need to be determined which α heterodimer partners and respective ligands (23) are important for β1 integrin-mediated therapeutic resistance and to what extent endogenous integrin modulators contribute to the witnessed therapeutic resistance.
On the basis of the prior literature, it is likely the relevance of these findings are much broader than those described in the present study. Our BRG primary and cell lines were derived from patient tumors that failed standard therapy with temozolomide and radiation, as well as bevacizumab. Thus, these lines are likely resistant to multiple therapies and may represent a clinically predictive model. These results also raise the possibility that other angiogenic cancers, such as breast and colon, may benefit from the addition of anti-β1 integrin strategies to antiangiogenic therapy. This may particularly create a new opportunity for such agents in metastatic breast cancer considering the recent revocation of accelerated approval of bevacizumab in these patients.
In conclusion, very few candidate molecules have been demonstrated to be a critical gatekeeper for nearly all components of the “Hallmarks of Cancer” as β1 integrin (29). From its fundamental role in tumorigenesis, angiogenesis, exponential growth, survival in the face of various therapeutic regimens and cellular stresses, mesenchymal phenotypes, and local invasion and metastasis, targeting β1 integrin has the potential to significantly improve outcomes for patients with cancer.
Footnotes
Disclosure of Potential Conflicts of Interest
M.K. Aghi is a consultant/advisory board member of Oncosynergy. W.S. Carbonell is the President and CEO of OncoSynergy, Inc. for which he also has ownership interest (including patents). No potential conflicts of interest were disclosed by the other author.
Authors’ Contributions
Conception and design: A. Jahangiri, M.K. Aghi, W.S. Carbonell
Acquisition of data (provided animals, acquired and managed patients, provided facilities, etc.): M.K. Aghi
Analysis and interpretation of data (e.g., statistical analysis, biostatistics, computational analysis): W.S. Carbonell
Writing, review, and/or revision of the manuscript: A. Jahangiri, M.K. Aghi, W.S. Carbonell
Administrative, technical, or material support (i.e., reporting or organizing data, constructing databases): A. Jahangiri, W.S. Carbonell
References
- 1.Holash J, Maisonpierre PC, Compton D, Boland P, Alexander CR, Zagzag D, et al. Vessel cooption, regression, and growth in tumors mediated by angiopoietins and VEGF. Science. 1999;284:1994–8. doi: 10.1126/science.284.5422.1994. [DOI] [PubMed] [Google Scholar]
- 2.Gilbert MR, Dignam J, Won M, Blumenthal DT, Vogelbaum MA, Aldape KD, et al. RTOG 0825: phase III double-blind placebo-controlled trial evaluating bevacizumab (Bev) in patients (Pts) with newly diagnosed glioblastoma (GBM) [abstract]. Proceedings of the 49th American Society of Clinical Oncology (ASCO) Annual Meeting; 2013 May 31–Jun 4; Chicago, IL: ASCO; 2013. p. Abstract nr 1. [Google Scholar]
- 3.Lin EY, Pollard JW. Tumor-associated macrophages press the angiogenic switch in breast cancer. Cancer Res. 2007;67:5064–6. doi: 10.1158/0008-5472.CAN-07-0912. [DOI] [PubMed] [Google Scholar]
- 4.Dolberg DS, Bissell MJ. Inability of Rous sarcoma virus to cause sarcomas in the avian embryo. Nature. 1984;309:552–6. doi: 10.1038/309552a0. [DOI] [PubMed] [Google Scholar]
- 5.Jin H, Su J, Garmy-Susini B, Kleeman J, Varner J. Integrin alpha4beta1 promotes monocyte trafficking and angiogenesis in tumors. Cancer Res. 2006;66:2146–52. doi: 10.1158/0008-5472.CAN-05-2704. [DOI] [PubMed] [Google Scholar]
- 6.Zhang L, Dong Y, Cheng J, Du J. Role of integrin-beta3 protein in macrophage polarization and regeneration of injured muscle. J Biol Chem. 2012;287:6177–86. doi: 10.1074/jbc.M111.292649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Park CC, Zhang H, Pallavicini M, Gray JW, Baehner F, Park CJ, et al. Beta1 integrin inhibitory antibody induces apoptosis of breast cancer cells, inhibits growth, and distinguishes malignant from normal phenotype in three dimensional cultures and in vivo. Cancer Res. 2006;66:1526–35. doi: 10.1158/0008-5472.CAN-05-3071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Somanath PR, Malinin NL, Byzova TV. Cooperation between integrin alphavbeta3 and VEGFR2 in angiogenesis. Angiogenesis. 2009;12:177–85. doi: 10.1007/s10456-009-9141-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brooks PC, Clark RA, Cheresh DA. Requirement of vascular integrin alpha versus beta 3 for angiogenesis. Science. 1994;264:569–71. doi: 10.1126/science.7512751. [DOI] [PubMed] [Google Scholar]
- 10.Kim S, Bell S, Mousa S, Varner J. Regulation of angiogenesis in vivo by ligation of integrin a5b1 with the central cell-binding domain of fibronectin. Am J Pathol. 2000;156:1345–62. doi: 10.1016/s0002-9440(10)65005-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hughes PE, Renshaw MW, Pfaff M, Forsyth J, Keivens VM, Schwartz MA, et al. Suppression of integrin activation: a novel function of a Ras/ Raf-initiated MAP kinase pathway. Cell. 1997;88:521–30. doi: 10.1016/s0092-8674(00)81892-9. [DOI] [PubMed] [Google Scholar]
- 12.Sawhney RS, Cookson MM, Omar Y, Hauser J, Brattain MG. Integrin alpha2-mediated ERK and calpain activation play a critical role in cell adhesion and motility via focal adhesion kinase signaling: identification of a novel signaling pathway. J Biol Chem. 2006;281:8497–510. doi: 10.1074/jbc.M600787200. [DOI] [PubMed] [Google Scholar]
- 13.Schlaepfer DD, Hunter T. Focal adhesion kinase overexpression enhances ras-dependent integrin signaling to ERK2/mitogen-activated protein kinase through interactions with and activation of c-Src. J Biol Chem. 1997;272:13189–95. doi: 10.1074/jbc.272.20.13189. [DOI] [PubMed] [Google Scholar]
- 14.King WG, Mattaliano MD, Chan TO, Tsichlis PN, Brugge JS. Phosphatidylinositol 3-kinase is required for integrin-stimulated AKT and Raf-1/mitogen-activated protein kinase pathway activation. Mol Cell Biol. 1997;17:4406–18. doi: 10.1128/mcb.17.8.4406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Park CC, Bissell MJ, Barcellos-Hoff MH. The influence of the microenvironment on the malignant phenotype. Mol Med Today. 2000;6:324–9. doi: 10.1016/s1357-4310(00)01756-1. [DOI] [PubMed] [Google Scholar]
- 16.Uhm JH, Gladson CL, Rao JS. The role of integrins in the malignant phenotype of gliomas. Front Biosci. 1999;4:D188–99. doi: 10.2741/uhm. [DOI] [PubMed] [Google Scholar]
- 17.Weaver VM, Lelievre S, Lakins JN, Chrenek MA, Jones JC, Giancotti F, et al. Beta4 integrin-dependent formation of polarized three-dimensional architecture confers resistance to apoptosis in normal and malignant mammary epithelium. Cancer Cell. 2002;2:205–16. doi: 10.1016/s1535-6108(02)00125-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nam JM, Chung Y, Hsu HC, Park CC. beta1 integrin targeting to enhance radiation therapy. Int J Radiat Biol. 2009;85:923–8. doi: 10.3109/09553000903232876. [DOI] [PubMed] [Google Scholar]
- 19.Mocanu MM, Fazekas Z, Petras M, Nagy P, Sebestyen Z, Isola J, et al. Associations of ErbB2, beta1-integrin, and lipid rafts on Herceptin (trastuzumab) resistant and sensitive tumor cell lines. Cancer Lett. 2005;227:201–12. doi: 10.1016/j.canlet.2005.01.028. [DOI] [PubMed] [Google Scholar]
- 20.Huang C, Park CC, Hilsenbeck SG, Ward R, Rimawi MF, Wang YC, et al. Beta1 integrin mediates an alternative survival pathway in breast cancer cells resistant to lapatinib. Breast Cancer Res. 2011;13:R84. doi: 10.1186/bcr2936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shafren DR, Sylvester D, Johansson ES, Campbell IG, Barry RD. Oncolysis of human ovarian cancers by echovirus type 1. Int J Cancer. 2005;115:320–8. doi: 10.1002/ijc.20866. [DOI] [PubMed] [Google Scholar]
- 22.Rubenstein JL, Kim J, Ozawa T, Zhang M, Westphal M, Deen DF, et al. Anti-VEGF antibody treatment of glioblastoma prolongs survival but results in increased vascular cooption. Neoplasia. 2000;2:306–14. doi: 10.1038/sj.neo.7900102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Paez-Ribes M, Allen E, Hudock J, Takeda T, Okuyama H, Vinals F, et al. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell. 2009;15:220–31. doi: 10.1016/j.ccr.2009.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Guo W, Giancotti FG. Integrin signalling during tumour progression. Nat Rev Mol Cell Biol. 2004;5:816–26. doi: 10.1038/nrm1490. [DOI] [PubMed] [Google Scholar]
- 25.Li N, Zhang Y, Naylor MJ, Schatzmann F, Maurer F, Wintermantel T, et al. Beta1 integrins regulate mammary gland proliferation and maintain the integrity of mammary alveoli. EMBO J. 2005;24:1942–53. doi: 10.1038/sj.emboj.7600674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Friedl P, Wolf K. Tumour-cell invasion and migration: diversity and escape mechanisms. Nat Rev Cancer. 2003;3:362–74. doi: 10.1038/nrc1075. [DOI] [PubMed] [Google Scholar]
- 27.Carbonell WS, Ansorge O, Sibson N, Muschel R. The vascular basement membrane as “soil” in brain metastasis. PloS ONE. 2009;4:e5857. doi: 10.1371/journal.pone.0005857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.DeLay M, Jahangiri A, Carbonell WS, Hu YL, Tsao S, Tom MW, et al. Microarray analysis verifies two distinct phenotypes of glioblastomas resistant to antiangiogenic therapy. Clin Cancer Res. 2012;18:2930–42. doi: 10.1158/1078-0432.CCR-11-2390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–74. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]