Abstract
Background
This is an update of the original Cochrane review published in Issue 2, 2007. Some antiepileptic medicines have a place in the treatment of neuropathic pain (pain due to nerve damage). This updated review adds five new additional studies looking at evidence for Lamotrigine as an effective treatment for acute and chronic pain.
Objectives
To assess analgesic efficacy and adverse effects of the antiepileptic drug lamotrigine in acute and chronic pain.
Search methods
Randomised controlled trials (RCTs) of lamotrigine in acute, and chronic pain (including cancer pain) were identified from MEDLINE, EMBASE and CENTRAL up to January 2011. Additional studies were sought from the reference list of the retrieved papers.
Selection criteria
RCTs investigating the use of lamotrigine (any dose, by any route, and for any study duration) for the treatment of acute or chronic pain. Assessment of pain intensity or pain relief, or both, using validated scales. Participants were adults aged 18 and over. Only full journal publication articles were included.
Data collection and analysis
Dichotomous data (ideally for the outcome of at least 50% pain relief) were used to calculate relative risk with 95% confidence intervals. Meta-analysis was undertaken using a fixed-effect model. Numbers needed to treat to benefit (NNTs) were calculated as the reciprocal of the absolute risk reduction. For unwanted effects, the NNT becomes the number needed to harm (NNH) and was calculated.
Main results
Twelve included studies in 11 publications (1511 participants), all with chronic neuropathic pain: central post stroke pain (1), chemotherapy induced neuropathic pain (1), diabetic neuropathy (4), HIV related neuropathy (2), mixed neuropathic pain (2), spinal cord injury related pain (1), and trigeminal neuralgia (1); none investigated lamotrigine in acute pain. The update had five additional studies (1111 additional participants). Participants were aged between 26 and 77 years. Study duration was 2 weeks in one study and at least 6 weeks in the remainder; eight were of eight week duration or longer. There is no convincing evidence that lamotrigine is effective in treating acute or chronic pain at doses of about 200-400 mg daily. Almost 10% of participants taking lamotrigine reported a skin rash.
Authors’ conclusions
The additional studies tripled participant numbers providing data for analysis, and new, more stringent criteria for outcomes and analysis were used; conclusions about lamotrigine’s lack of efficacy in chronic pain did not change. Given availability of more effective treatments including antiepileptics and antidepressant medicines, lamotrigine does not have a significant place in therapy based on available evidence.
Medical Subject Headings (MeSH): Acute Disease, Analgesics [* therapeutic use], Chronic Disease, Pain [* drug therapy], Randomized Controlled Trials as Topic, Triazines [* therapeutic use]
MeSH check words: Humans
BACKGROUND
This review is an update to the previous review published in Issue 2, 2007 on this topic and contains five new studies providing data on an additional 1111 participants, of whom 767 received lamotrigine (Wiffen 2007).
Description of the condition
Pain is complex in terms of both nerve mechanisms and psychological perceptions. It is usually classified as acute pain lasting less than three months, or chronic pain, generally considered to be pain lasting for longer than three months. Chronic pain can also be due to nerve damage which is known as neuropathic pain, or to underlying disease including cancer. Chronic pain is a major health problem affecting one in five people in Europe (Breivik 2006; Pain in Europe 2004), though data to determine the incidence of neuropathic pain are more difficult to obtain. Different neuropathic pain disorders occur more or less commonly. The incidence of trigeminal neuralgia has been estimated at four in 100,000 per year (Katusic 1991; Rappaport 1994), and more recent data suggests incidences per 100,000 person years observation of 40 (95% CI 39-41) for post-herpetic neuralgia, 27 (26-27) for trigeminal neuralgia, 1 (1-2) for phantom limb pain and 15 (15-16) for painful diabetic neuropathy (Hall 2006). Chronic painful conditions are disabling, with significant reduction of quality of life that can be reversed by effective treatment (Moore 2010c).
Antiepileptic drugs have been used in pain management since the 1960s, very soon after they were first used in medicine. The clinical impression is that they are useful for neuropathic pain, especially when the pain is lancinating or burning (Jacox 1994). There is evidence for the effectiveness of a number of antiepileptics; these are considered in other reviews published by the Cochrane Pain, Palliative and Supportive Care group through The Cochrane Database of Systematic Reviews (Moore 2009; Wiffen 2005; Wiffen 2011). Antiepileptics are sometimes prescribed in combination with antidepressants, as in the treatment of post-herpetic neuralgia (Monks 1994). In the UK carbamazepine and phenytoin are licensed for the treatment of pain associated with trigeminal neuralgia, and gabapentin and pregabalin for the treatment of neuropathic pain. Lamotrigine is also being used for chronic pain but is not licensed in Europe for this condition.
Description of the intervention
Lamotrigine, a phenyltriazine, is chemically unrelated to other antiepileptic drugs. The drug is available as standard oral tablets (25 to 200 mg) and chewable, dispersible tablets (2 to 25 mg), and a new extended release tablet is available in some parts of the world.
How the intervention might work
Lamotrigine is an antiepileptic drug exerting its antiepileptic effect via sodium channels. There is some evidence that agents that block sodium channels are useful in the treatment of neuropathic pain (McCleane 2000). There is evidence from animal models supporting use of lamotrigine in neuropathic pain, and for an effect in experimental pain models such as cold-induced pain in humans (McCleane 2000). Lamotrigine is chemically unrelated to existing antiepileptic agents. There has also been discussion of the role of lamotrigine as a pre-emptive analgesic to reduce postsurgical pain (Bonicalzi 1997). More recently it has been shown that neuronal alpha-4-beta2-nicotinic acetylcholine receptors may be a target for lamotrigine, and this may mediate its antiepileptic effects (Zheng 2010).
Why it is important to do this review
Lamotrigine is not widely prescribed for neuropathic pain, though it is prescribed for some cases of painful HIV-related neuropathy. There is controversy over whether it is of value in other acute or chronic pain conditions, especially neuropathic pain.
Neuropathic pain is a complex and often disabling condition in which many people suffer moderate or severe pain for many years. Conventional analgesics are usually not effective in alleviating the symptoms, although opioids may be effective in some individuals. Treatment is usually by unconventional analgesics such as antidepressants or antiepileptics. The reason is that, unlike nociceptive pain (pain that arises from nerve endings detecting unpleasant or painful stimuli), such as arthritis or gout, neuropathic pain is caused by nerve damage, often accompanied by changes in the central nervous system (CNS).
There have been several changes in how efficacy of both conventional and unconventional treatments is assessed in chronic painful conditions. The outcomes used today are better defined, particularly with new criteria for what constitutes moderate or substantial benefit (Dworkin 2008); older studies may only report participants with ‘any improvement’. Newer studies tend to be larger, avoiding problems from the random play of chance. Newer studies also tend to be longer, up to 12 weeks, providing a more rigorous and valid assessment of efficacy in chronic conditions. New standards have evolved for assessing efficacy in neuropathic pain; we are now applying stricter criteria for inclusion of studies and assessment of outcomes, and we are more aware of problems that may affect our overall assessment.
To summarise, some of the recent insights that make a new review necessary, over and above including more studies are:
Pain relief results tend to have a U-shaped distribution rather than a bell-shaped distribution (see Moore 2005a for acute pain). This is true in acute pain, fibromyalgia, and arthritis (Moore 2010a); in all cases mean results usually describe the experience of almost no-one in the study. Continuous data expressed as means should be regarded as potentially misleading, unless it can be proved to be suitable. Systematic reviews now frequently report results for responders (Lunn 2009; Moore 2010a; Straube 2008; Sultan 2008).
This means we have to depend on dichotomous results usually from pain changes or patient global assessments, showing whether participants did or did not achieve some clinically useful level of pain reduction. The IMMPACT group has helped with their definitions of minimal, moderate, and substantial improvement (Dworkin 2008). In arthritis and fibromyalgia studies shorter than 12 weeks, and especially those shorter than eight weeks, overestimate the effect of treatment (Moore 2010a; Straube 2010); the effect is particularly strong for less effective analgesics. What is not always clear is how withdrawals are reported. Withdrawals can be high in some chronic pain conditions (Moore 2005b; Moore 2010b).
The proportion with at least moderate benefit can be small, falling from 60% with an effective medicine in arthritis, to 30% in fibromyalgia (Moore 2010a; Straube 2008; Straube 2010; Sultan 2008). A Cochrane Review of pregabalin in neuropathic pain and fibromyalgia demonstrated different response rates for different types of chronic pain (higher in diabetic neuropathy and postherpetic neuralgia and lower in central pain and fibromyalgia) (Moore 2009). This indicates that different neuropathic pain conditions should be treated separately from one another, and that pooling should not be done unless there are good grounds for doing so.
Finally, individual patient analyses indicate that patients who get clinically useful pain relief (moderate or better) have major benefits in many other outcomes, affecting quality of life in a major way (Barthel 2010; Hoffman 2010; Moore 2010c). Good response to pain predicts good effects for other troublesome symptoms like sleep, fatigue and depression.
This Cochrane review update will therefore assess evidence in ways that make both statistical and clinical sense; the requirements for doing so have been published (Moore 2010d). Studies included and analysed will need to meet a minima of reporting quality (blinding, randomisation), validity (duration, dose and timing, diagnosis, outcomes, etc.), and size (ideally a minimum of 500 participants in a comparison with NNTs of four or greater (Moore 1998)).
The review covers acute and chronic neuropathic pain (including fibromyalgia), concentrating for efficacy on dichotomous responder outcomes. We will consider conditions individually, as there is evidence of different effects in different neuropathic pain conditions for some interventions like pregabalin (Moore 2009), though less so for others (Lunn 2009). The review will also consider additional risks of bias. These will include issues of withdrawal (Moore 2010b), size (Moore 1998), and duration (Moore 2010a) in addition to standard risks of bias.
OBJECTIVES
To assess the analgesic efficacy of lamotrigine in acute and chronic pain.
To assess the adverse effects associated with the clinical use of lamotrigine for pain.
METHODS
Criteria for considering studies for this review
Types of studies
Studies were included in this review if they were randomised, double blind, placebo or active controlled trials (RCTs) that investigated the analgesic effects of lamotrigine in participants, with pain assessment as either the primary or secondary outcome. Full journal publication was required, abstracts were not included. Studies that were non-randomised, studies of experimental pain, case reports, clinical observations were also excluded. In the earlier review, we excluded studies of lamotrigine used to treat pain produced by other drugs; in this version we have included one study for chemotherapy-induced pain, but have not combined results from this study in the analysis (Rao 2008).
Types of participants
We included adult participants aged 18 years and above. Participants could have one or more of a wide range of acute or chronic painful conditions. For chronic neuropathic pain conditions we intended to include (but not limit to):
painful diabetic neuropathy (PDN);
postherpetic neuralgia (PHN);
trigeminal neuralgia;
phantom limb pain;
postoperative or traumatic neuropathic pain;
complex regional pain syndrome (CRPS);
cancer-related neuropathy;
HIV-neuropathy;
spinal cord injury;
fibromyalgia.
We also included studies of participants with more than one type of neuropathic pain. We analysed results according to the primary condition.
Types of interventions
Administration of lamotrigine, in any dose, by any route to achieve analgesia, with placebo or active comparators.
Types of outcome measures
Studies had to report pain assessment as either a primary or secondary outcome.
A variety of outcome measures were used in the studies. The majority of studies used standard subjective scales for pain intensity or pain relief, or both. Particular attention was paid to IMMPACT definitions for moderate and substantial benefit in chronic pain studies (Dworkin 2008). These are defined as at least 30% pain relief over baseline (moderate), at least 50% pain relief over baseline (substantial), much or very much improved on Patient Global Impression of Change (PGIC) (moderate), and very much improved on PGIC (substantial). These outcomes are different from those set out in the previous version of this review, concentrating on dichotomous outcomes where pain responses are not normally distributed. People with chronic pain desire high levels of pain relief, ideally with more than 50% pain relief, and pain not worse than mild (O’Brien 2010).
In the process of completing the review, a more extensive list of potential biases was published (Moore 2010b). This added duration (less than 2 weeks, 2 to 6 weeks, 8 to 12 weeks), outcome (typically pain reduction less than 30% of baseline, pain intensity reduction ≥ 30% from baseline, pain intensity reduction ≥ 50% from baseline, though with other definitions), incomplete outcome assessment (how pain measures are dealt with on withdrawal), and treatment arm size (≤ 50, 50 to 199, and ≥ 200 participants per treatment arm) as potential sources of bias, with definitions that could be used in risk of bias tables in Cochrane reviews. This updated procedure has been used in this review.
Primary outcomes
Patient reported pain intensity reduction of 30% or greater.
Patient reported pain intensity reduction of 50% or greater.
Patient reported global impression of clinical change (PGIC) much or very much improved.
Patient reported global impression of clinical change (PGIC) very much improved.
Secondary outcomes
Any pain-related outcome indicating some improvement.
Withdrawals due to lack of efficacy.
Participants experiencing any adverse event.
Participants experiencing any serious adverse event.
Withdrawals due to adverse events.
Specific adverse events, particularly somnolence and dizziness.
During the updating process we discussed and reached consensus concerning a common core data set for pain reviews, and to reflect that we also used a working set of seven outcomes that might form a core data set. This overlapped to some extent with outcomes already identified:
at least 50% pain reduction;
proportion below 30/100 mm (no worse than mild pain);
patient global impression;
functioning;
adverse event (AE) withdrawal;
serious AE;
death.
We considered the possibility of using these outcomes, but aside from functioning they were already included in primary and secondary outcomes chosen (with death noted as a serious adverse event).
Search methods for identification of studies
Electronic searches
Studies were identified by several methods. RCTs of lamotrigine (and key brand names Lamictal, Lamictin, Neurium) in acute, chronic or cancer pain were identified using:
MEDLINE from 1966 to January 2011;
EMBASE 1994 to January 2011;
The Cochrane Library (issue 12, 2010).
Given the limited literature in this area, a sensitive search strategy was undertaken. See Appendix 1 for the MEDLINE search strategy, Appendix 2 for the EMBASE search strategy, and Appendix 3 for the CENTRAL search strategy. We also searched the PhRMA clinical study results database (www.clinicalstudyresults.org/) for trial results of lamotrigine in painful conditions.
Searching other resources
Additional studies were identified from the reference lists of the retrieved papers, and by contacting study authors.
There was no language restriction.
Data collection and analysis
Selection of studies
All studies identified by the search were read independently by two review authors and agreement on eligibility reached by discussion. The studies were not anonymised in any way before assessment.
Data extraction and management
Two review authors extracted data using a standard form, and agreed data before entry into RevMan 5 or any other analysis method. Data extracted included information about the pain condition and number of participants treated, drug and dosing regimen, study design, study duration and follow up, analgesic outcome measures and results, withdrawals and adverse events (participants experiencing any adverse event, or serious adverse event).
Assessment of risk of bias in included studies
We used the ‘Risk of bias’ tool to assess the likely impact on the strength of the evidence of various study characteristics relating to methodological quality (randomisation, allocation concealment, blinding, freedom from selective reporting), study validity (duration, outcome reporting, and handling of missing data), and size (Moore 2010b).
We also scored each study independently for quality using a three-item scale (Jadad 1996). We then met to agree a ‘consensus’ score for each study. Quality scores were not used to weight the results in any way.
The three-item scale is as follows:
-
Is the study randomised? If ‘yes’, then 1 point.
If described, is the randomisation appropriate? If ‘yes’ add 1 point, if not deduct 1 point.
-
Is the study double-blind? If ‘yes’, then 1 point.
Is the double-blind method appropriate? If ‘yes’ add 1 point, if not deduct 1 point.
-
Are withdrawals and drop-outs described? (i.e. the number and reason for drop-outs for each of the treatment groups).
If ‘yes’, add 1 point.
Scores of two and below have been associated with greater estimates of efficacy than studies of higher quality (Khan 1996).
Measures of treatment effect
Relative risk (or ‘risk ratio’, RR) was used to establish statistical difference. Numbers needed to treat (NNT) and pooled percentages were used as absolute measures of benefit or harm.
The following terms are used to describe adverse outcomes in terms of harm or prevention of harm:
When significantly fewer adverse outcomes occurred with lamotrigine than with control (placebo or active) we use the term the number needed to treat to prevent one event (NNTp).
When significantly more adverse outcomes occurred with lamotrigine compared with control (placebo or active) we use the term the number needed to harm or cause one event (NNH).
Unit of analysis issues
The control treatment arm will be split between active treatment arms in a single study if the active treatment arms are not combined for analysis.
Dealing with missing data
We used intention-to-treat (ITT) analysis. The ITT population consisted of participants who were randomised, took the assigned study medication and provided at least one post-baseline assessment. Missing participants were assigned zero improvement where this could be done. We were aware that imputation methods might be problematical and examined trial reports for information about them.
Assessment of heterogeneity
We dealt with clinical heterogeneity by combining studies that examined similar painful conditions, and not combining results from dissimilar painful conditions. We assessed statistical heterogeneity visually (L’Abbe 1987) and with the use of the I2 statistic.
Assessment of reporting biases
The aim of this review was to use dichotomous data of known utility (Moore 2010a). The review did not depend on what authors of the original studies chose to report or not report, though clearly there were difficulties with studies failing to report any dichotomous results. Continuous data, which probably poorly reflect efficacy and utility, were extracted and used only when useful for illustrative purposes.
We undertook no assessment of publication bias.
Data synthesis
We used dichotomous data to calculate relative risk (RR) or benefit with 95% Confidence Intervals (CIs) using a fixed-effect model, together with numbers needed to treat to benefit (NNTs) (Cook 1995). This was done for effectiveness, for adverse effects and for drug-related study withdrawal. We also undertook meta-analysis when appropriate data were available. We calculated NNTs as the reciprocal of the absolute risk reduction (McQuay 1998). For unwanted effects, the NNT becomes the NNH (number needed to treat to harm), and is calculated in the same way. In the absence of dichotomous data, summary continuous data are reported where available and appropriate, but no analysis was carried out. We undertook meta-analysis using a fixed-effect model.
Subgroup analysis and investigation of heterogeneity
No subgroup analyses were planned, beyond separate analysis of different conditions, as it was expected that there would be insufficient study data.
Sensitivity analysis
We planned no sensitivity analyses because the evidence base was known to be too small to allow reliable analysis.
RESULTS
Description of studies
See: Characteristics of included studies; Characteristics of excluded studies.
Results of the search
Twenty-three studies were identified in 23 publications.
Included studies
Twelve studies (12 publications), involving 1511 participants, satisfied inclusion criteria (Eisenberg 2001; Finnerup 2002; Jose 2007; McCleane 1999; Rao 2008; Silver 2007; Simpson 2000; Simpson 2003; Vestergaard 2001; Vinik 2007a; Vinik 2007b; Zakrzewska 1997). Two studies were reported in one publication (Vinik 2007a; Vinik 2007b), and an incomplete report of Eisenberg 2001 (Lurie 2000) provided no additional data, but is included and linked to the primary study. Included studies covered the following conditions: central post stroke pain (Vestergaard 2001), chemotherapy-induced peripheral neuropathic pain (Rao 2008), diabetic neuropathy (Eisenberg 2001; Jose 2007; Vinik 2007a; Vinik 2007b) HIV-related neuropathy (Simpson 2000; Simpson 2003), mixed neuropathic pain (McCleane 1999; Silver 2007), spinal cord injury related pain (Finnerup 2002), and trigeminal neuralgia (Zakrzewska 1997).
Eleven studies used a placebo comparator, and one (Jose 2007) used amitriptyline as the comparator. Two studies added lamotrigine or placebo to existing treatments for neuropathic pain (Silver 2007; Zakrzewska 1997). The studies included participants in the age range of 26 to 77 years. One study was for 2 weeks (Zakrzewska 1997); the remainder were at least 6 weeks, and eight were of 8 week duration or longer. Four were cross-over studies (Finnerup 2002; Jose 2007; Vestergaard 2001; Zakrzewska 1997). Details of all eligible studies are given in the ‘Characteristics of included studies’ and results for individual studies are in a separate table (Appendix 4).
There were seven studies in the original review (Eisenberg 2001; Finnerup 2002; McCleane 1999; Simpson 2000; Simpson 2003; Vestergaard 2001; Zakrzewska 1997). In this update five additional studies were added (Jose 2007; Rao 2008; Silver 2007; Vinik 2007a; Vinik 2007b), with 1111 participants, almost trebling the number of participants since the previous review. The studies added in this update were generally larger in size and of longer duration.
Excluded studies
Eleven studies were excluded (see ‘Characteristics of excluded studies’ table for reasons for exclusion) (Bonicalzi 1997; Breuer 2007; Carrieri 1998; Devulder 2000; di Vadi 1998; Eisenberg 1998; Eisenberg 2003; Eisenberg 2005; Lunardi 1997; Petersen 2003; Sandner-Kiesling 2002).
Risk of bias in included studies
Each study was scored for quality using the three-item Oxford Quality Score scale (Jadad 1996) and agreed by the review authors. All scored 3/5 or greater, with two scoring 3/5, four scoring 4/5, and six scoring 5/5.
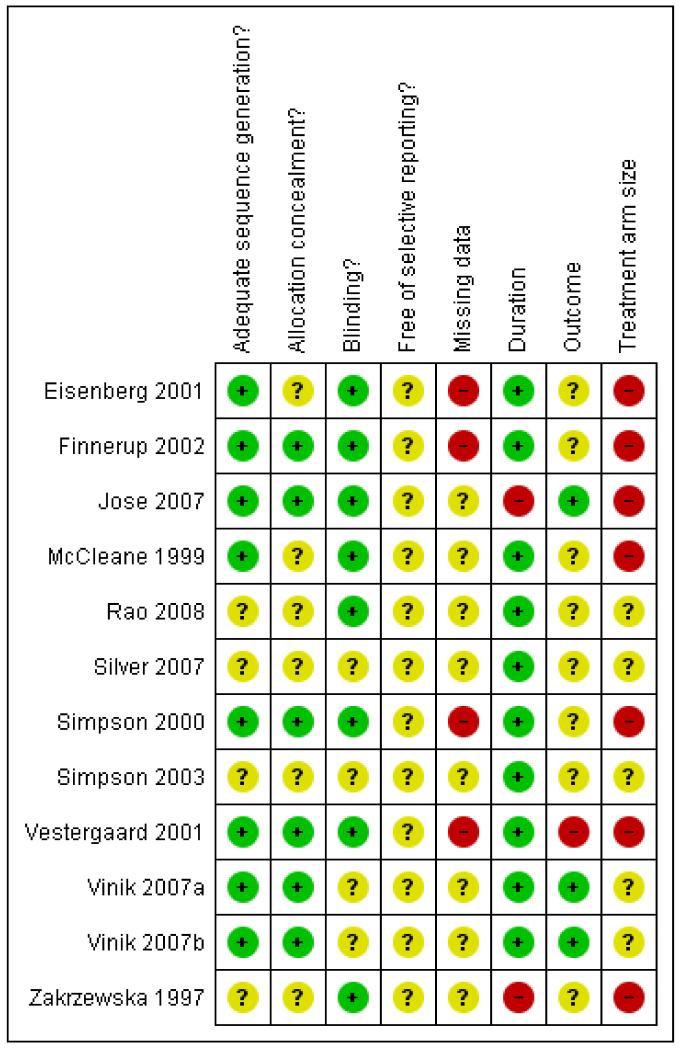
In this update we have used the new Risk of Bias tool. The comments on individual studies are reported in the Risk of Bias section of the ‘Characteristics of included studies’ table. The findings are displayed in Figure 1; no sensitivity analysis was undertaken. The greatest risks of bias came from freedom from reporting bias, missing data, outcomes used, and study size.
Figure 1. Risk of bias summary: review authors’ judgements about each risk of bias item for each included study.
Effects of interventions
Acute pain
One study (Bonicalzi 1997) examined the use of lamotrigine for acute pain but this was excluded as treatment was given pre-emptive and all participants were given the potent analgesic buprenorphine.
Chronic pain
The twelve included studies covered the following conditions: central post stroke pain (Vestergaard 2001), chemotherapy-induced neuropathic pain (Rao 2008) diabetic neuropathy (Eisenberg 2001; Jose 2007; Vinik 2007a; Vinik 2007b), HIV-related neuropathy (Simpson 2000; Simpson 2003), mixed neuropathic pain (McCleane 1999; Silver 2007), spinal cord injury related pain (Finnerup 2002) and trigeminal neuralgia (Zakrzewska 1997). Two studies added lamotrigine to exiting treatment for neuropathic pain (Silver 2007; Zakrzewska 1997), these are discussed as ‘add-on’ studies.
Central post stroke pain
Thirty participants took part in a single cross-over study, and only 20 completed both arms (Vestergaard 2001). The difference between lamotrigine 200 mg and placebo for clinical response was significant when assessed at eight weeks. Lower pain scores (reduction of 2/10 or more) were reported by 12 participants on lamotrigine and three on placebo.
Chemotherapy-induced peripheral neuropathic pain
In a study of 125 participants (Rao 2008), average pain scores decreased in both the active and placebo groups with no significant difference between the groups. The study authors concluded that lamotrigine was not effective in this condition.
Painful diabetic neuropathy (PDN)
Four studies (Eisenberg 2001; Jose 2007; Vinik 2007a; Vinik 2007b) looked at the role of lamotrigine for PDN (758 participants). None of these demonstrated any major benefits.
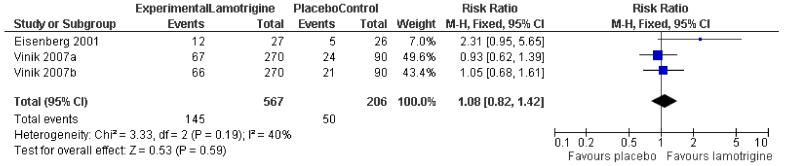
In one study (Eisenberg 2001), a 50% reduction in pain measured in last three weeks of treatment was reported by 12/27 on lamotrigine titrated up to 400 mg daily and 5/26 on placebo. In two large randomised studies of lamotrigine 200 to 400 mg daily there was no difference between lamotrigine and placebo for the outcome of at least 50% pain relief (Vinik 2007a; Vinik 2007b). There was no overall significant difference between lamotrigine 200 to 400 mg daily and placebo (relative risk 1.1 (95% CI 0.8 to 1.4)) (Figure 2). A similar non-significant difference was found for participants reporting “marked improvement”.
Figure 2. Forest plot of comparison: 1 Painful diabetic neuropathy, outcome: 1.1 50% pain relief.
In the fourth study, a 20% reduction in pain was reported by 19/46 on lamotrigine 200 mg daily and 13/46 on 50 mg amitriptyline at night (Jose 2007). There were insufficient data for analysis.
HIV-related neuropathy
There were two studies involving participants with HIV-related neuropathy. The first study of 42 participants (Simpson 2000) claimed effectiveness for lamotrigine 300 mg/day, but over 50% of the treatment group dropped out making results difficult to interpret. The second study (Simpson 2003) analysed the results according to whether participants were receiving antiretroviral therapy (ART) or not. For those who were receiving antiretroviral therapy there did appear to be some benefits in terms of attainment of moderate or better pain relief with lamotrigine (35/62, 57%) than placebo (7/30, 23%). For Patient Global Impression of Change, marked improvement was recorded by 29/62 (47%) of participants on lamotrigine and 4/30 (13%) on placebo with antiretroviral therapy.
Mixed neuropathic pain
One study of 100 participants examined the use of lamotrigine 200 mg daily in participants with intractable neuropathic pain diagnosed by symptoms of shooting/lancinating pain, burning, numbness, allodynia and paraesthesia/dysaesthesia (McCleane 1999). At least three of these symptoms were required for participation. Participants already taking an antiepileptic were excluded. No useful analgesic benefit was demonstrated. There was a reduction in the overall pain score of 1/100 mm. A second study used an ‘add-on’ design for lamotrigine titrated up to 400 mg daily on top of gabapentin, tricyclic antidepressant, or non-opioid analgesic where pain was inadequately controlled (Silver 2007). No additional analgesic benefit could be demonstrated over 14 weeks.
Spinal cord injury related pain
Thirty participants with neuropathic pain following traumatic spinal cord injury were included (Finnerup 2002). Doses of up to 400 mg daily for lamotrigine were used but the study authors reported no significant effects on pain intensity.
Trigeminal neuralgia
Fourteen participants participated in a cross-over ‘add-on’ study comparing lamotrigine with placebo in a cross-over study of two two-week phases separated by a three day long washout (Zakrzewska 1997). All participants continued on carbamazepine or phenytoin throughout the study period. Lamotrigine was slightly more effective than placebo in this small study (RR not significant); 10/13 participants stated that lamotrigine was better or much better, compared with 8/14 on placebo, using a global evaluation.
Adverse events
Adverse events were not consistently reported across studies. It was not possible to determine the incidence of mild and severe adverse effects. Rash can be problematic with lamotrigine. It was mentioned as an adverse event or adverse event withdrawal in 11 studies, and omitted from a long list of adverse events in the other (Zakrzewska 1997). Combining studies, the overall incidence of rash was 9.5% with lamotrigine and 5.6% with placebo, barely achieving statistical significance (relative risk 1.4; 95% CI 1.01 to 2.0) (Figure 3). This would indicate that rash with lamotrigine would affect about one person in 25 who would not have been affected with placebo.
Figure 3. Forest plot of comparison: 1 Painful diabetic neuropathy, outcome: 1.2 Rash.
DISCUSSION
Antiepileptic drugs have demonstrated a role in the treatment of neuropathic pain since carbamazepine was first used for trigeminal neuralgia in the 1960s. Subsequently other drugs in the class have been used so that gabapentin and pregabalin are now widely used, and other drugs such as valproate are considered to have a role in neuropathic pain (Moore 2009; Wiffen 2005a; Wiffen 2005). It was therefore inevitable that lamotrigine should also be investigated.
Overall there is no convincing evidence of benefit from large, high quality, long duration studies reporting clinically useful levels of pain relief for individual patients. There is very limited evidence for a possible effect of lamotrigine in central post stroke pain and in a subgroup of patients with HIV-related neuropathy receiving antiretroviral therapy. No benefit was demonstrated for diabetic neuropathy, in intractable neuropathic pain, spinal cord injury, or trigeminal neuralgia. The small number of studies and the small number of participants is insufficient to provide robust evidence for effect. This together with the difficulties of dose titration and adverse effects are likely to dissuade many clinicians from choosing lamotrigine to treat neuropathic pain. It is possible that those running the studies have chosen to include the more difficult participants in terms of severity and duration of pain, nevertheless, while neuropathic pain is difficult to manage there are more effective and safer medicines available (Wiffen 2005).
Safety is an important aspect of the choice of treatment even in difficult conditions. In this review, about 10% of participants developed a rash; this fits with wider epidemiological work (Hirsch 2006). The results are consistent with reports in the manufacturer’s summary of product characteristics. Serious potentially life threatening rashes such as Stevens Johnson Syndrome are estimated to occur at an incidence of one in 1000 (SPC 2007).
Summary of main results
No eligible studies were found in acute pain. There is no convincing evidence that lamotrigine is effective in treating chronic pain at doses of about 200 to 400 mg daily. It is possible that lamotrigine has some effect in some patients with painful HIV-related neuropathy undergoing antiretroviral therapy, but that comes from limited sub-group data in a single study (Simpson 2003). Assessment of benefits and risks was limited by small numbers and inconsistent reporting of adverse events, though rash appears to be more common with lamotrigine.
Overall completeness and applicability of evidence
Efficacy and adverse event outcomes were not consistently reported across the studies, and this limited the analyses to some extent.
Quality of the evidence
The studies included in this review covered a number of different painful conditions. For some, like HIV neuropathy for instance, it is unclear whether antiepileptic drugs are effective in the condition, and any indication of benefit is welcome. The main quality issues involve reporting of outcomes of interest, particularly dichotomous outcomes equivalent to IMMPACT, as well as better reporting of adverse events. The earliest study was published in 1997, and the past decade has seen major changes in clinical trial reporting. The studies themselves appear to be largely well-conducted, and individual patient analysis could overcome some of the shortcomings of reporting, though not the paucity of participants studied in each neuropathic pain condition.
Potential biases in the review process
The review was restricted to randomised double blind studies, thus limiting the potential for bias. Other possible sources of bias that could have affected the review included:
Duration - NNT estimates of efficacy in chronic pain studies tend to increase (get worse) with increasing duration (Moore 2010a). However, all studies were six weeks or longer, and most longer than eight weeks.
Outcomes may effect estimates of efficacy, but the efficacy outcomes chosen were of participants achieving the equivalent of IMMPACT-defined moderate or substantial improvement, and it is likely that lesser benefits, such as “any benefit” or “any improvement”, are potentially related to inferior outcomes, though this remains to be clarified. Most authors attempted to report dichotomous outcomes of interest, especially in the larger, more recent studies.
The question of whether cross-over trials exaggerate treatment effects in comparison with parallel-group designs, as has been seen in some circumstances (Khan 1996), is unclear but unlikely to be the source of major bias (Elbourne 2002). Withdrawals meant that any results were more likely to be per protocol for completers than for a true ITT analysis. Parallel group studies were larger than cross-over studies, and predominated analyses in terms of number of participants, with only about 100 participants in cross-over studies.
The absence of publication bias (unpublished trials showing no benefit of lamotrigine over placebo) can never be proven. However, publication bias is irrelevant where the published studies show no effect of treatment.
Imputation methods used when participants withdrew were typically either last observation carried forward (LOCF) or were not stated; no study reported clearly that participants achieving acceptable levels of pain relief were unequivocally on treatment at the end of the study (Moore 2010c). How this might affect estimates of efficacy is currently unclear.
Agreements and disagreements with other studies or reviews
This update is broadly in agreement with the previous Cochrane review (Wiffen 2007). A non-systematic review considered lamotrigine to be effective, but based on only a fraction of the results presented in the updated review (Jensen 2002).
AUTHORS’ CONCLUSIONS
Implications for practice
These results further confirm that lamotrigine is not suitable for use in the treatment of acute and chronic pain. Based on current evidence, the routine use of lamotrigine is unlikely to be of benefit in the treatment of neuropathic pain; there may be a role for experimental use or in patients who have failed to obtain pain relief from other treatments. The incidence of skin rash is not trivial and must be considered before initiating therapy.
Implications for research
Reasonable levels of evidence exist for the benefit of other antiepileptic drugs and antidepressants in the treatment of acute and chronic pain and therefore there is probably no justification for further research given the lack of evidence and the potential for harm due to skin rash, which can occasionally be serious.
PLAIN LANGUAGE SUMMARY.
Lamotrigine (an antiepileptic drug) for acute and chronic pain
Nerves which have been damaged by injury or disease can continue to produce pain. This type of pain is called neuropathic pain. Some antiepileptic medications can help neuropathic pain. Lamotrigine is an antiepileptic medication. The aim of this review was to assess how effective lamotrigine is for neuropathic or other chronic pain, or acute pain. The review identified 12 included studies which included a total of 1511 participants. Studies were only available for neuropathic pain, with no evidence that lamotrigine was effective in this type of pain. This may be because lamotrigine works in a different way to other antiepileptic medications. Lamotrigine also seems to cause more cases of skin rash, which can be serious. Based on current evidence, lamotrigine is unlikely to help with neuropathic pain. Other antiepileptic drugs such as pregabalin, gabapentin, and carbamazepine have been shown to be of value in neuropathic pain.
ACKNOWLEDGEMENTS
Jayne Edwards contributed as an author to the original review.
SOURCES OF SUPPORT
Internal sources
Pain Research Oxford, UK.
UK Cochrane Centre, UK.
External sources
No sources of support supplied
CHARACTERISTICS OF STUDIES
Characteristics of included studies [ordered by study ID]
| Methods | Randomised DB placebo controlled, parallel group study for 11 weeks. One week screening phase, 8 week treatment phase, 2 week post treatment phase | |
| Participants | 59 participants with painful diabetic neuropathy. Age 50 to 60 years Excluded: participants who had received antiepileptics or antidepressants for reasons other than pain and those who had received opioids |
|
| Interventions | Lamotrigine 25 mg dispersible tablets or matching placebo 25 mg daily for 2 weeks, 50 mg daily for 2 weeks, then 100 mg, 200 mg, 300 mg and 400 mg for 1 week at each dose level Rescue analgesia as paracetamol, dipyrone or NSAIDs |
|
| Outcomes | Daily pain intensity, McGill, Beck depression, Pain disability index, Global assessment. Responder: 50% reduction in pain measured in final 3 weeks of treatment | |
| Notes | Oxford Quality Score: R2, DB2, W1 = 5 | |
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Low risk | ‘Randomisation was done in blocks of four according to a computer generated random code’ |
| Allocation concealment? | Unclear risk | Not stated |
| Blinding? Assessors |
Low risk | ‘Patients in the placebo group received equal numbers of identical looking placebo tablets’ |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | High risk | Completer analysis - data from withdrawals not carried forward |
| Duration | Low risk | ‘Eight weeks treatment phase’ |
| Outcome | Unclear risk | Looked for reduction in pain intensity but reports numbers with 50% reduction. No mention of imputation method |
| Treatment arm size | High risk | 59 participants: 29 active, 30 placebo |
| Methods | Randomised DB placebo controlled, crossover study. One week baseline assessment, two 9 week treatment periods separated by 2 week washout | |
| Participants | 30 participants with neuropathic pain after traumatic spinal cord injury (SCI). Age 27 to 63 years | |
| Interventions | Lamotrigine tablets or identical placebo. Dose escalation to 400 mg a day. Weeks 1 and 2 at 25 mg daily, weeks 3 and 4 at 50 mg, 1 week each at 100 mg, 200 mg, and 300 mg then 2 weeks at 400 mg. Concomitant treatment with spasmolytics, sedatives for insomnia, and simple analgesics for other pain allowed in constant unchanged dose Rescue analgesia: paracetamol up to 3g daily |
|
| Outcomes | Average daily pain on 11 point numeric scale. Change in median weekly pain score from baseline to final week. Participant preference, other measures included details of types of pain, impact on sleep, and use of rescue medication | |
| Notes | Oxford Quality Score: R2, DB2, W1 = 5 | |
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Low risk | ‘assignment to treatment was random via a computer generated randomisation list with blocks of four’ |
| Allocation concealment? | Low risk | ‘The primary investigator was provided with sealed code envelopes- one for each patient- containing information on the treatment given … and envelopes were returned unopened to the monitor after the study termination.’ |
| Blinding? Assessors |
Low risk | ‘lamotrigine and identical placebo’ |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | High risk | completer |
| Duration | Low risk | Nine week per arm treatment period |
| Outcome | Unclear risk | Looked for reduction in pain intensity but reports numbers with 50% reduction |
| Treatment arm size | High risk | 30 participants total, 22 completers |
| Methods | Randomised DB active controlled, crossover study. Two 6 week treatment periods separated by 2 week washout | |
| Participants | 53 participants, of whom 46 received both treatments, with Type 2 Diabetes and painful diabetic neuropathy for at least 1 month Excluded: participants taking antidepressants, antiepileptics, opioids and local anaesthetic agents |
|
| Interventions | Lamotrigine dose escalation to 100 mg twice daily over 6 weeks or amitriptyline to 50 mg at night with matching placebo in the morning. Two week washout using placebo between treatment periods Rescue analgesia: paracetamol up to 3 g daily |
|
| Outcomes | Patient global assessment (> 50% pain relief = good, > 25% pain relief), VAS PI, short form McGill, 5 point categorical scale for pain and Hamilton depression scale | |
| Notes | CONSORT flow chart indicated 23 patients randomised to lamotrigine and 30 to amitriptyline on first crossover arm, 23 each on second. 46 patients included in ITT analysis. Outcomes reported for both arms of crossover, with 46 as denominator for efficacy Oxford Quality Score: R2, DB2, W1 = 5 |
|
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Low risk | ‘numbers generated using random number tables by block randomisation’ |
| Allocation concealment? | Low risk | ‘blinding and randomisation carried out by independent person unrelated to the study’, ‘drug codes were maintained under lock and key’ |
| Blinding? Assessors |
Low risk | ‘drugs were blinded, packed and numbered serially’ |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | Unclear risk | LOCF used |
| Duration | High risk | Six weeks dose escalation then cross over |
| Outcome | Low risk | ‘VAS score showing improvement of > 50%, > 25% and < 25%’ used |
| Treatment arm size | High risk | 53 participants; 23 per treatment arm, with 46 completers |
| Methods | Randomised DB placebo controlled, parallel group study. Eight week treatment period | |
| Participants | 100 participants with intractable neuropathic pain. Mean age placebo group 44.7 years, lamotrigine group 47.1 years. All had failed response to codeine or NSAID based analgesics Excluded: participants taking antiepileptics |
|
| Interventions | Lamotrigine 25 mg dispersible tablets or matching placebo 25 mg daily for 1 week, then 50 mg daily for 2 weeks, then 100 mg daily for 1 week, then 150 mg daily for 1 week, then 200 mg daily for 3 weeks Rescue analgesia not reported |
|
| Outcomes | Daily patient recorded VAS for PI, shooting pain, burning pain, paraesthesia, numbness, QOL, mobility, sleep and mood. Daily analgesic consumption Results: Scores aggregated at 56 days. No useful analgesic benefit demonstrated by lamotrigine in doses up to 200 mg. From a baseline of 6.76 (on 10 cm VAS) overall pain on lamotrigine reduced by 0.01 and on placebo increased by 0.03. “No patient achieved 50% reduction in overall pain with lamotrigine” |
|
| Notes | Eighteen withdrew: eight nausea (five placebo, three lamotrigine); two skin rash (one lamotrigine); two bad taste of tablets (one lamotrigine); six due to lack of analgesia two placebo four lamotrigine) . Eight failed to attend final assessment Oxford Quality Score: R2, DB2, W1 = 5 |
|
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Low risk | ‘Patients randomly assigned in equal numbers to one of two groups using computer generated random number lists’ |
| Allocation concealment? | Unclear risk | Not stated |
| Blinding? Assessors |
Low risk | ‘patients received either lamotrigine.. … or identical looking dispersible placebo tablets’ |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | Unclear risk | Not stated |
| Duration | Low risk | Eight week study |
| Outcome | Unclear risk | VAS recorded |
| Treatment arm size | High risk | 74 participants; placebo 38, lamotrigine 36 |
| Methods | Randomised DB placebo controlled, parallel group study. Ten week treatment period, followed by 4 week tapered withdrawal | |
| Participants | 125 participants (63 received lamotrigine) with diagnosis of symptomatic chemotherapy induced peripheral neuropathy > 1 month due to neurotoxic agents. Age 29 to 84 years. Average pain > 4 on NRS Excluded: participants taking drugs for treating neuropathic pain, including antiepileptics, opioids or topical analgesics at study entry; NSAIDs were permitted |
|
| Interventions | Lamotrigine or matching placebo. 25 mg once daily for 2 weeks, then 25 mg, 50 mg, 100 mg, 150 mg twice daily for 2 weeks at each dose, then 4 weeks taper down | |
| Outcomes | Average daily pain score using NRS and ENS (Eastern Cooperative Oncology neuropathy scale) . No significant difference between groups on any analgesia scale. Adverse events more common with lamotrigine (33%) than placebo (18%). Rash 3 lamotrigine vs 0 placebo | |
| Notes | Oxford Quality Score: R1, DB2, W1 = 4 | |
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | stated to be randomised |
| Allocation concealment? | Unclear risk | Not statement |
| Blinding? Assessors |
Low risk | ‘an identical appearing placebo’ |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | Unclear risk | Not stated |
| Duration | Low risk | 10 weeks |
| Outcome | Unclear risk | Average daily pain scores |
| Treatment arm size | Unclear risk | 125 participants; lamotrigine 63, placebo 62 |
| Methods | Randomised DB placebo controlled, parallel group, ‘add on study’. Fourteen week treatment period consisting of 8 weeks dose escalation and 6 weeks at fixed dose, followed by 1 week tapered withdrawal | |
| Participants | Neuropathic pain defined as DN, PHN, nerve injury, spinal cord injury, MS or HIV neuropathy. Mean age 60 years (SD 12). Mean weekly pain score > 4 on 11 point scale. Participants on stable (≥ 4 weeks) treatment with gabapentin, tricyclics or non opioid analgesics Excluded: back and neck pain |
|
| Interventions | Lamotrigine 200-400 mg daily or placebo in addition to other (inadequate) treatments as above Rescue analgesia: paracetamol up to 3 g daily |
|
| Outcomes | Numerical PR (11 point), sleep interference, short form McGill, neuropathic pain scale, patient global impression of change | |
| Notes | Oxford Quality Score: R1, DB1, W1 = 3 | |
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | ‘randomised in a 1:1 ratio’ |
| Allocation concealment? | Unclear risk | Not stated |
| Blinding? Assessors |
Unclear risk | Stated to be double blind |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | Unclear risk | LOCF |
| Duration | Low risk | 14 week treatment |
| Outcome | Unclear risk | Change in daily pain intensity |
| Treatment arm size | Unclear risk | 111 participants lamotrigine, 109 placebo |
| Methods | Multicentre randomised DB placebo controlled, parallel study. Fourteen week treatment period | |
| Participants | 42 participants with painful HIV associated polyneuropathy. Mean age 44 years Excluded: participants taking valproate |
|
| Interventions | Lamotrigine or placebo. Week 1 and 2 at 25 mg daily, weeks 3 and 4 at 50 mg daily, week 5 at 100 mg daily, week 6 at 100 mg twice daily, then weeks 7 to 14 at 150 mg twice daily | |
| Outcomes | Average and peak neuropathic pain using Gracely Pain Scale. Difference in weekly mean pain scores. Pain assessed in weeks 1 and 14, also slope of change in pain scores |
|
| Notes | Oxford Quality Score: R2, DB2, W1 = 5 | |
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Low risk | ‘The biostatistician generated a list of treatment assignments in random order using a program written in SAS’ |
| Allocation concealment? | Low risk | ‘The biostatistician had no contact with patients nor did he communicate these to anyone other than the pharmacists’ (to supply the medicines) |
| Blinding? Assessors |
Low risk | ‘Lamotrigine and matching placebo’ |
| Free of selective reporting? | Unclear risk | LOCF used for part of the analysis |
| Missing data | High risk | Combination of LOCF and completer |
| Duration | Low risk | 14 weeks including dose escalation |
| Outcome | Unclear risk | Difference in weekly mean pain scores between baseline and final week |
| Treatment arm size | High risk | 42 participants in total at start |
| Methods | Randomised DB placebo controlled parallel multicentre trial. One week screening phase, then 11 week treatment period. Randomisation stratified according to use of neurotoxic antiretroviral therapy (ART) | |
| Participants | 227 participants with HIV associated sensory neuropathy. Age 32 to 67 years Excluded: participants with previous or current use of lamotrigine |
|
| Interventions | Lamotrigine or placebo. Weeks 1 and 2 at 25 mg on alternate days (daily if taking enzyme inducing drugs), then dose escalation over 5 weeks to a target dose of 400 mg daily (up to 600 mg daily allowed if taking enzyme inducing drugs), followed by 4 week maintenance phase. Concomitant medication allowed if stable (≥ 4 weeks) and unchanged Rescue analgesia: opioid and non-opioid analgesics as needed |
|
| Outcomes | Daily pain rating of average pain and worst pain on Gracely Pain Scale. VAS PI and short form McGill at end of baseline and beginning and end of maintenance phase, PGIC | |
| Notes | Oxford Quality Score: R1, DB1, W1 = 3 | |
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | ‘randomised’ |
| Allocation concealment? | Unclear risk | Not stated |
| Blinding? Assessors |
Unclear risk | ‘double blind placebo controlled’ |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | Unclear risk | observed scores used - meaning unclear |
| Duration | Low risk | 13 weeks including dose escalation |
| Outcome | Unclear risk | ‘average pain and worse pain’ recorded |
| Treatment arm size | Unclear risk | 227 participants; 150 lamotrigine, 77 placebo |
| Methods | Randomised DB placebo controlled, crossover study. Two 8 week treatment periods, separated by 2 week washout | |
| Participants | 30 participants with central post stroke pain with score of > 4 on an 11 point scale. Age 37 to 77 years | |
| Interventions | Lamotrigine soluble tablets or matching placebo. Initial dose of 25 mg daily increased every 2nd week to 200 mg daily. No concomitant use of antidepressants, antiepileptics or analgesics allowed Rescue analgesia: paracetamol 500 mg as needed |
|
| Outcomes | Average daily pain score during last week of treatment (11 point Likert scale). Clinical responders defined as 2/10 reduction on lamotrigine compared with placebo period. CAT PR and CAT PI. Use of rescue medication |
|
| Notes | Oxford Quality Score: R2, DB2, W1 = 5 | |
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Low risk | ‘patients were randomised to treatment according to a computer generated randomisation list with a cluster size of six’ |
| Allocation concealment? | Low risk | ‘code envelopes were kept by the investigator during the trial and returned unopened to the monitor at the termination of the study. The blinding was maintained throughout’ |
| Blinding? Assessors |
Low risk | ‘soluble lamotrigine and identical placebo’ |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | High risk | completer |
| Duration | Low risk | Two eight week crossover periods |
| Outcome | High risk | clinical response stated to be 2 or more points lower for lamotrigine compared to placebo |
| Treatment arm size | High risk | 30 participants; 16 lamotrigine, 13 placebo at initial randomisation, with 20 completers |
| Methods | Randomised DB placebo controlled parallel group. Nineteen week treatment period comprising 7 week dose escalation and 12 week fixed dose maintenance phase. Study no NPP30004 | |
| Participants | 360 participants with diabetic neuropathy (typel or type 2 diabetics). Pain > 6 months and pain score > 4 on 11 point Likert scale. Mean age 59 years (SD 11) | |
| Interventions | Lamotrigine at daily dose of 200 mg, 300 mg, 400 mg, or placebo. Dose doubled initially every 2nd week, then weekly to target dose. Concomitant gabapentin and TCAs allowed Rescue analgesia: paracetamol up to 4 g daily 91/360 received gabapentin, 17/360 received TCAs |
|
| Outcomes | Average pain intensity (11 point pain NRS). Sleep disturbance. Short form McGill | |
| Notes | Oxford Qulaity Score: R2, DB1, W1 = 4 | |
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Low risk | ‘in accordance with a computer generated randomisation schedule. A central randomisation procedure was used’ |
| Allocation concealment? | Low risk | ‘the study center called into a central system’ |
| Blinding? Assessors |
Unclear risk | stated to be double blind |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | Unclear risk | LOCF |
| Duration | Low risk | Seven week dose escalation and 12 weeks fixed dose |
| Outcome | Low risk | 50% reduction in pain intensity |
| Treatment arm size | Unclear risk | 360 participants; 90 patients randomised per group |
| Methods | Randomised DB placebo controlled parallel group. Nineteen week treatment period comprising 7 week dose escalation and 12 week fixed dose maintenance phase. Study no NPP30005 | |
| Participants | 360 participants with diabetic neuropathy (type 1 or type 2 diabetics). Pain > 6 months and pain score > 4 on 11 point Likert scale. Mean age 60 years (SD 11). Gabapentin and TCAs allowed. Paracetamol as rescue |
|
| Interventions | Lamotrigine at daily dose of 200 mg, 300 mg, 400 mg, or placebo. Dose doubled initially every 2nd week, then weekly to target dose. Concomitant gabapentin and TCAs allowed Rescue analgesia: paracetamol up to 4 g daily 76/360 received gabapentin, 23/360 received TCAs |
|
| Outcomes | Average pain intensity (11 point pain NRS). Sleep disturbance. Short form McGill Greater than 50% reduction in average pain intensity: 200 mg 22/90; 300 mg 22/90; 400 mg 22/90; placebo 21/90. Authors claim that 400 mg produced a significant reduction in pain |
|
| Notes | Withdrawals: 200 mg 10/90; 300 mg 9/90; 400 15/90; placebo 9/90 AE withdrawals: 200 mg 5/90; 300 mg 8/90; 400 mg 10/90; placebo 4/90 Rash 200 mg 9/90; 300 mg 10/90; 400 mg 14/90; placebo 8/90 Oxford Quality Score: R2, DB1, W1 = 4 |
|
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Low risk | ‘in accordance with a computer generated randomisation schedule. A central randomisation procedure was used’ |
| Allocation concealment? | Low risk | ‘the study center called into a central system’ |
| Blinding? Assessors |
Unclear risk | stated to be double blind |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | Unclear risk | LOCF |
| Duration | Low risk | Seven week dose escalation and 12 weeks fixed dose |
| Outcome | Low risk | 50% reduction in pain intensity |
| Treatment arm size | Unclear risk | 360 participants; 90 patients randomised per group |
| Methods | Randomised DB placebo controlled, crossover, ‘add on study’. Two 2 week treatment periods separated by 3 day washout. Lamotrigine added to existing antiepileptic treatment | |
| Participants | 14 participants with refractory trigeminal neuralgia. Age 44 to 75 (mean 60 years) | |
| Interventions | Lamotrigine or placebo added to existing stable regimen of carbamazepine or phenytoin, or both. Day 1 at 50 mg, day 2 at 100 mg, day 3 at 200 mg, then days 4 to 14 at 400 mg Rescue analgesia: increased dose of carbamazepine or phenytoin used for uncontrollable pain |
|
| Outcomes | No of pain paroxysms. CAT PI, CAT PR and global assessment at the end of each treatment period | |
| Notes | Withdrawals. One on placebo for uncontrolled pain. 7/13 reported AEs on lamotrigine and 7/14 on placebo Oxford Quality Score: R1, DB2, W1 = 4 |
|
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | ‘randomised’ |
| Allocation concealment? | Unclear risk | Not stated |
| Blinding? Assessors |
Low risk | ‘dispersible lamotrigine and identical placebo’ |
| Free of selective reporting? | Unclear risk | Not stated |
| Missing data | Unclear risk | unclear |
| Duration | High risk | 2 weeks per arm |
| Outcome | Unclear risk | composite efficacy index |
| Treatment arm size | High risk | 14 participants |
AEs = adverse effects, DB = double blind, CAT PI = categorical scale of pain intensity, CAT PR = categorical scale of pain relief, NNT = number needed to treat, NRS- numerical rating scale PI = pain intensity, QOL = quality of life, R = randomisation, VAS = visual analogue scale, W = withdrawals
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Bonicalzi 1997 | Pre emptive study but all participants also received a known analgesic-buprenorphine |
| Breuer 2007 | RCT of multiple sclerosis pain. Not neuropathic pain |
| Carrieri 1998 | Case study |
| Devulder 2000 | Survey not RCT |
| di Vadi 1998 | Case report only |
| Eisenberg 1998 | Not randomised |
| Eisenberg 2003 | Not randomised |
| Eisenberg 2005 | Review article |
| Lunardi 1997 | Case series |
| Petersen 2003 | RCT but healthy volunteers |
| Sandner-Kiesling 2002 | Case report |
DATA AND ANALYSES
Comparison 1. Painful diabetic neuropathy.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 50% pain relief | 3 | 773 | Risk Ratio (M-H, Fixed, 95% CI) | 1.08 [0.82, 1.42] |
| 2 Rash | 12 | 1715 | Risk Ratio (M-H, Fixed, 95% CI) | 1.43 [1.01, 2.03] |
Analysis 1.1. Comparison 1 Painful diabetic neuropathy, Outcome 1 50% pain relief.
Review: Lamotrigine for acute and chronic pain
Comparison: 1 Painful diabetic neuropathy
Outcome: 1 50% pain relief
 |
Analysis 1.2. Comparison 1 Painful diabetic neuropathy, Outcome 2 Rash.
Review: Lamotrigine for acute and chronic pain
Comparison: 1 Painful diabetic neuropathy
Outcome: 2 Rash
 |
Appendix 1. MEDLINE search strategy
(pain* or analgesi* or neuralgi* or headache* or toothache* or earache* or sciatica or causalgi* or arthralgi* or colic* or dysmenorrhoea or dysmenorrhea).mp. [mp=title, original title, abstract, name of substance word, subject heading word] (482954)
(lamotrigin* or lamictal* or lamictin* or neurium* or labileno or crisomet).mp. [mp=title, original title, abstract, name of substance word, subject heading word] (2850)
*lamotrigine/(0)
3 or 2 (2850)
4 and 1 (312)
(2006**** or 2007**** or 2008**** or 2009****).ed. (2536672)
6 and 5 (123)
randomized controlled trial.pt. (275280)
controlled clinical trial.pt. (79863)
randomized.ab. (184599)
placebo.ab. (113625)
drug therapy.fs. (1328219)
randomly.ab. (133908)
trial.ab. (191715)
groups.ab. (918498)
or/8-15 (2427837)
(animals not (humans and animals)).sh. (3316879)
16 not 17 (2058235)
18 and 7 (87)
Appendix 2. EMBASE (via OVID) search strategy
(pain* or analgesi* or neuralgi* or headache* or toothache* or earache* or sciatica or causalgi* or arthralgi* or colic* or dysmenorrhoea or dysmenorrhea).mp. [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name] (507181)
(lamotrigin* or lamictal* or lamictin* or neurium* or labileno or crisomet).mp. [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name] (10342)
*lamotrigine/(2142)
(2006** or 2007** or 2008** or 2009** or 2010**).em. (2113688)
3 or 2 (10342)
1 and 5 (2247)
4 and 6 (1011)
random*.ti,ab. (402107)
factorial*.ti,ab. (8426)
(crossover* or cross over* or cross-over*).ti,ab. (39987)
placebo*.ti,ab. (111613)
(doubl* adj blind*).ti,ab. (85818)
(singl* adj blind*).ti,ab. (7584)
assign*.ti,ab. (110664)
allocat*.ti,ab. (35027)
volunteer*.ti,ab. (100438)
CROSSOVER PROCEDURE.sh. (21495)
DOUBLE-BLIND PROCEDURE.sh. (73001)
RANDOMIZED CONTROLLED TRIAL.sh. (170607)
SINGLE BLIND PROCEDURE.sh. (8290)
or/8-20 (671990)
ANIMAL/or NONHUMAN/or ANIMAL EXPERIMENT/(3483821)
HUMAN/(6540195)
22 and 23 (547985)
22 not 24 (2935836)
21 not 25 (585134)
7 and 26 (206)
Appendix 3. CENTRAL search strategy
#1 (lamotrigin* OR lamictal* Or lamictin* OR neurium* OR labileno OR crisomet):ti,ab,kw
#2 (pain* OR analgesi* OR neuralgi* or headache* OR toothache* OR earache* OR sciatica OR causalgi* OR arthralgi* OR colic* OR dysmenorrhoea or dysmenorrhea):ti,ab,kw
#3 (#1 AND #2)
#4 (#3 AND CENTRAL)
Appendix 4. Results in individual studies
Appendix 4. Results in individual studies.
| Study ID | Maximum daily dose of lamotrigine Titration/fixed |
Comparator Numbers in trial |
Withdrawals | Efficacy | Adverse events (general) |
Adverse events (specific) |
|---|---|---|---|---|---|---|
| Central post stroke pain | ||||||
| Vestergaard 2001 | Lamotrigine titrated from 25 mg daily to 200 mg daily. Crossover at 8 weeks. 2 week washout |
Placebo N = 30 |
lamotrigine 4 (3 due to AEs, 1 lack of efficacy) placebo 4 (3 lack of efficacy, 1 protocol violation) | ‘Responders’ on lamotrigine 200 mg 12/27 No difference 12/27 Lower pain scores on placebo 3/27 lamotrigine up to 100 mg no different from placebo |
17 events in participants with lamotrigine, 18 with placebo, and 10 during washout period | Three participants on lamotrigine withdrew (rash, headache and severe pain). Five participants had skin related AEs with lamotrigine and 2 with placebo |
| Chemotherapy induced peripheral neuropathy | ||||||
| Rao 2008 | Lamotrigine titrated up to 150 mg twice daily over 10 weeks then 4 weeks taper down | Placebo N = 125 |
lamotrigine 29 withdrew (13 refusals, 7 due to AEs, 9 other) placebo 16 withdrew (10 refusals, 1 AE, 5 other) | Small reduction in 11 point Likert pain scale of less than one point in both groups. No difference between groups | 26 events in participants with lamotrigine, 28 with placebo | Mainly dizziness and fatigue Three on lamotrigine reported skin rash |
| Diabetic Neuropathy | ||||||
| Eisenberg 2001 | Dose titrated from 25 mg daily up to 400 mg daily | Placebo N = 59 (46 completed) |
lamotrigine 5/29 (2 rash, 3 other) placebo 8/30 (2 AE, 6 other) | 50% reduction in pain measured in final 3 weeks of treatment: lamotrigine 12/27, placebo 5/26 (not ITT) Global assessment: ‘highly effective’ lamotrigine 7122, placebo 2/21 ‘moderate or better’ lamotrigine 16/22, placebo 9/21 |
17 events in participants with lamotrigine, 21 with placebo | Most common: nausea, drowsiness and dizziness Withdrew: lamotrigine 2/27 (rash), placebo 2/26 (1 impotence, 1 diarrhoea) |
| Jose 2007 | Dose titration up to 100 mg twice daily over 6 weeks. Crossover with 2 weeks washout |
Amitriptyline up to 50 mg at night N = 53 |
Six did not attend for 1st visit and one for 2nd visit | ≥ 50% reduction on VAS global assessment (good improvement) : lamotrigine 19/46, amitriptyline 13/46 Increased sleep: lamotrigine 0/46, amitriptyline 19/46 |
11 events in participants with lamotrigine, 33 with amitriptyline | 3 participants developed rash with lamotrigine |
| Vinik 2007a | Dose titration over 7 weeks to max in assigned group (200 mg, 300 mg, 400 mg) | Placebo N = 360 |
All: lamotrigine 200 mg 10/90, 300 mg 10/90, 400 mg 15/90, placebo 5/90 AE withdrawals: lamotrigine 200 mg 2/90,300 mg 5/90, 400 mg 9/90, placebo 2/90 |
50% or greater reduction in pain intensity: lamotrigine 200 mg 21/90, 300 mg 30/90, 400 mg 16/90, placebo 24/90 Not significant |
206 events with lamotrigine (270 participants), 62 events with placebo group (90 participants) | Most common: Headache: 40/270 lamotrigine, 3/90 placebo Rash: 31/270 lamotrigine, 8/90 placebo |
| Vinik 2007b | Dose titration over 7 weeks to max in assigned group (200 mg, 300 mg, 400 mg) | Placebo N = 360 |
All: lamotrigine 200 mg 10/90, 300 mg 9/90, 400 15/90, placebo 9/90 AE withdrawals: lamotrigine 200 mg 10/90, 300 mg 9/90,40013/90, placebo 9/90 |
50% or greater reduction in pain intensity: lamotrigine 200 mg 22/90, 300 mg 22/90, 400 mg 22/90, placebo 21/90 Not significant |
206 events with lamotrigine (192 participants), 54 events with placebo (90 participants) | Most common: Headache: 47/270 lamotrigine, 6/90 placebo Rash: 33/270 lamotrigine, 8/90 placebo |
| HIV related neuropathy | ||||||
| Simpson 2000 | Lamotrigine titrated by 25 mg doses to 300 mg at 7 weeks | Placebo N = 42 |
lamotrigine 11/20 (6 AE, 5 lost to follow up) placebo 3/22 (2 personal reason, 1 lost to follow up) | Large dropout-only 9 completed in lamotrigine group. Higher falls in pain scores in lamotrigine group | Not reported | lamotrigine 6/20 (5 rash, 1 GI infection) |
| Simpson 2003 | Lamotrigine titrated by 25 mg every other day to a target of 400 mg/day | Placebo N = 227 |
lamotrigine 34/150 (10 AE, 6 withdrew consent, 8 lost to follow up, 8 protocol violations, 2 other) placebo 21/77 (7 AE, 1 withdrew consent, 4 lost to follow up, 5 protocol violations, 4 other) |
Moderate or better pain relief (>30%): Participants receiving ART: lamotrigine 35/62 (57%) , placebo 7/30 (23%) Participants not receiving ART: lamotrigine 46/88 (52%) (>30%), placebo 21/47 (45%) For PGIC (marked improvement) : lamotrigine 76/150, placebo 34/77 (29/62 vs 4/30 ART group) |
87 events with lamotrigine (150 participants) 39 with placebo (77 participants) | Most common: lamotrigine events (causing withdrawal) : rash 21 (2) , infection 17, nausea 17 (1) , diarrhoea 16, headache 16 (1) placebo events (causing withdrawal): rash 9(1), infection 7, nausea 8 (1), diarrhoea 7 (1), headache 8 |
| Mixed neuropathic pain | ||||||
| McCleane 1999 | Lamotrigine titrated to 200 mg daily. Study duration 8 weeks | Placebo N = 100 |
lamotrigine 14 (10 AE, 4 lack of efficacy) placebo 8 (6 AE, 2 lack of efficacy) Eight patients refused to attend the end of study review |
No participants achieved 50% pain relief | No further data available | Withdrawals: lamotrigine (3 nausea, 2 skin rash, 1 bad taste) placebo (5 nausea, 1 bad taste) |
| Silver 2007 | Lamotrigine 200-400 mg daily titrated over 8 weeks then 6 week fixed dose. Add-on study to existing (inadequate) treatment |
Placebo N = 220 |
lamotrigine 47/111 (28 due to AEs) placebo 31/109 (11 due to AEs) | No statistically significant difference between lamotrigine or placebo in mean change in pain intensity between baseline and week 14 | 79/111 lamotrigine participants and 61/109 placebo participants reported an adverse event | Most common: Rash 20/111 lamotrigine, 14/109 placebo Dizziness 10/111 lamotrigine, 11/109 placebo Somnolence 7/111 lamotrigine, 2/109 placebo |
| Spinal cord injury | ||||||
| Finnerup 2002 | Lamotrigine 400 mg titrated over 8 weeks. Crossover study with 2 weeks washout | Placebo N = 30 |
lamotrigine 3/15 (1 AE, 1 new trauma, 1 left country) placebo 5/15 (2 AE, 1 consent withdrawn, 1 protocol violation, 1 escape medication) |
No significant difference between groups for 50% or 30% pain relief, though authors claim benefit in subgroup who had incomplete SCI. ? post hoc analysis | 13/27 lamotrigine participants and 14/28 placebo participants reported an adverse event | Most common (events): CNS 12 lamotrigine, 9 placebo, Skin 4 lamotrigine, 4 placebo GI 4 lamotrigine, 3 placebo 1 placebo patient withdrew due to rash |
| Trigeminal neuralgia | ||||||
| Zakrzewska 1997 | Lamotrigine 400 mg titrated up over 4 days. Crossover. 2 weeks treatment, 1 week washout then 2 weeks treatment. Add-on study in participants also receiving either carbamazepine or phenytoin |
Placebo N= 14 |
One placebo participant on day 14 (uncontrolled pain) | 7/14 claimed to be ‘much better’ with lamotrigine, 1/14 with placebo 10/13 ‘better or much better’ with lamotrigine, 8/14 with placebo |
7/13 lamotrigine and 7/14 placebo participants reported AEs | Most common (events): Dizziness 5 lamotrigine, 1 placebo GI 8 lamotrigine, 3 placebo No skin rash reported |
WHAT’S NEW
Last assessed as up-to-date: 17 January 2011.
| Date | Event | Description |
|---|---|---|
| 18 January 2011 | New citation required but conclusions have not changed | This review update involves new authors and up to date methodology to confirm the conclusions. It is unlikely that the conclusions of this review will change in the foreseeable future |
| 18 January 2011 | New search has been performed | Five new studies (Jose 2007; Rao 2008; Silver 2007; Vinik 2007a; Vinik 2007b) were added with 1111 additional participants, increasing substantially the amount of information on this drug and further confirming its conclusions |
HISTORY
Protocol first published: Issue 2, 2006
Review first published: Issue 2, 2007
| Date | Event | Description |
|---|---|---|
| 30 October 2008 | Amended | Dates section corrected |
| 7 July 2008 | Amended | Converted to new review format. |
DIFFERENCES BETWEEN PROTOCOL AND REVIEW
The updated review conforms to more stringent evidence standards than those pertaining at the time of the original protocol.
Footnotes
DECLARATIONS OF INTEREST
PW is a full-time employee of the UK Cochrane Centre, funded by the UK National Institute of Health Research and has no relevant interests to declare.
RAM has received research support from charities, government and industry sources at various times, and consulted for and received lecture fees from various pharmaceutical companies related to analgesics and other healthcare interventions.
SD has received research support from charities, government and industry sources at various times.
References to studies included in this review
- Eisenberg 2001 {published data only} .Eisenberg E, Lurie Y, Breker C, Daoud D, Ishay A. Lamotrigine reduces painful diabetic neuropathy: a randomized, controlled study. Neurology. 2001;57(3):505–9. doi: 10.1212/wnl.57.3.505. [DOI] [PubMed] [Google Scholar]
- Lurie Y, Brecker C, Daoud D, Ishay A, Eisenberg E. Lamotrigine in the treatment of painful diabetic neuropathy: a randomized placebo controlled study. Progress in Pain Research and Management. 2000;16:857–62. [Google Scholar]
- Finnerup 2002 {published data only} .Finnerup NB, Sindrup SH, Bach FW, Johannesen IL, Jensen TS. Lamotrigine in spinal cord injury pain: a randomized controlled trial. Pain. 2002;96(3):375–83. doi: 10.1016/S0304-3959(01)00484-5. DOI: 10.1016/S0304-3959(01)00484-5. [DOI] [PubMed] [Google Scholar]
- Jose 2007 {published data only} .Jose VM, Bhansali A, Hota D, Pandhi P. Randomized double-blind study comparing the efficacy and safety of lamotrigine and amitriptyline in painful diabetic neuropathy. Diabetic Medicine. 2007;24:377–83. doi: 10.1111/j.1464-5491.2007.02093.x. DOI: 10.1111/j.1464-5491.2007.02093.x. [DOI] [PubMed] [Google Scholar]
- McCleane 1999 {published data only} .McCleane G. 200 mg daily of lamotrigine has no analgesic effect in neuropathic pain: a randomised, double-blind, placebo controlled trial. Pain. 1999;83(1):105–7. doi: 10.1016/s0304-3959(99)00095-0. DOI: 10.1016/S0304-3959(99)00095-0. [DOI] [PubMed] [Google Scholar]
- Rao 2008 {published data only} .Rao RD, Flynn PJ, Sloan JA, Wong GY, Novotny P, Johnson DB, et al. Efficacy of lamotrigine in the management of chemotherapy-induced peripheral neuropathy: a phase 3 randomized, double-blind, placebo-controlled trial, N01C3. Cancer. 2008;112:2802–8. doi: 10.1002/cncr.23482. DOI: 10.1002/cncr.23482. [DOI] [PubMed] [Google Scholar]
- Silver 2007 {published data only} .Silver M, Blum D, Grainger J, Hammer AE, Quessy S. Double-blind, placebo-controlled trial of lamotrigine in combination with other medications for neuropathic pain. Journal of Pain and Symptom Management. 2007;34:446–54. doi: 10.1016/j.jpainsymman.2006.12.015. DOI: 10.1016/j.jpainsymman.2006.12.015. [DOI] [PubMed] [Google Scholar]
- Simpson 2000 {published data only} .Simpson DM, Olney R, McArthur JC, Khan A, Godbold J, Ebel-Frommer K. A placebo-controlled trial of lamotrigine for painful HIV-associated neuropathy. Neurology. 2000;54(11):2115–9. doi: 10.1212/wnl.54.11.2115. [DOI] [PubMed] [Google Scholar]
- Simpson 2003 {published data only} .Simpson DM, McArthur JC, Olney R, Clifford D, So Y, Ross D, et al. Lamotrigine HIV Neuropathy Study Team Lamotrigine for HIV-associated painful sensory neuropathies: a placebo-controlled trial. Neurology. 2003;60(9):1508–14. doi: 10.1212/01.wnl.0000063304.88470.d9. [DOI] [PubMed] [Google Scholar]
- Vestergaard 2001 {published data only} .Vestergaard K, Andersen G, Gottrup H, Kristensen BT, Jensen TS. Lamotrigine for central poststroke pain: a randomized controlled trial. Neurology. 2001;56(2):184–90. doi: 10.1212/wnl.56.2.184. [DOI] [PubMed] [Google Scholar]
- Vinik 2007a {published data only} .Vinik AI, Tuchman M, Safirstein B, Corder C, Kirby L, Wilks K, et al. Lamotrigine for treatment of pain associated with diabetic neuropathy: results of two randomized, double-blind, placebo-controlled studies. PAIN. 2007;128:169–79. doi: 10.1016/j.pain.2006.09.040. DOI: 10.1016/j.pain.2006.09.040. [DOI] [PubMed] [Google Scholar]
- Vinik 2007b {published data only} .Vinik AI, Tuchman M, Safirstein B, Corder C, Kirby L, Wilks K, et al. Lamotrigine for treatment of pain associated with diabetic neuropathy: results of two randomized, double-blind, placebo-controlled studies. PAIN 2007. 2007;128:169–79. doi: 10.1016/j.pain.2006.09.040. DOI: 10.1016/j.pain.2006.09.040. [DOI] [PubMed] [Google Scholar]
- Zakrzewska 1997 {published data only} .Zakrzewska JM, Chaudhry Z, Nurmikko TJ, Patton DW, Mullens EL. Lamotrigine (lamictal) in refractory trigeminal neuralgia: results from a double-blind placebo controlled crossover trial. Pain. 1997;73(2):223–30. doi: 10.1016/S0304-3959(97)00104-8. DOI: 10.1016/S0304-3959(97)00104-8. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
- Bonicalzi 1997 {published data only} .Bonicalzi V, Canavero S, Cerutti F, Piazza M, Clemente M, Chio A. Lamotrigine reduces total postoperative analgesic requirement: a randomized double-blind, placebo-controlled pilot study. Surgery. 1997;122(3):567–70. doi: 10.1016/s0039-6060(97)90129-x. DOI: 10.1016/S0039-6060(97)90129-X. [DOI] [PubMed] [Google Scholar]
- Breuer 2007 {published data only} .Breuer B, Pappagallo M, Knotkova H, Guleyupoglu N, Wallenstein S, Portenoy RK. A randomized, double-blind, placebo-controlled, two-period, crossover, pilot trial of lamotrigine in patients with central pain due to multiple sclerosis. Clinical Therapeutics. 2007;29:2022–30. doi: 10.1016/j.clinthera.2007.09.023. DOI: 10.1016/j.clinthera.2007.09.023. [DOI] [PubMed] [Google Scholar]
- Carrieri 1998 {published data only} .Carrieri PB, Provitera VV, Lavorgna L, Bruno R. Response of thalamic pain syndrome to lamotrigine. European Journal of Neurology. 1998;5(6):625–6. doi: 10.1046/j.1468-1331.1998.560625.x. [DOI] [PubMed] [Google Scholar]
- Devulder 2000 {published data only} .Devulder J, De Laat M. Lamotrigine in the treatment of chronic refractory neuropathic pain. Journal of Pain Symptom Management. 2000;19(5):398–403. doi: 10.1016/s0885-3924(00)00131-7. DOI: 10.1016/S0885-3924(00)00131-7. [DOI] [PubMed] [Google Scholar]
- di Vadi 1998 {published data only} .di Vadi PP, Hamann W. The use of lamotrigine in neuropathic pain. Anaesthesia. 1998;53(8):808–9. doi: 10.1046/j.1365-2044.1998.00564.x. DOI: 10.1046/j.1365-2044.1998.00564.x. [DOI] [PubMed] [Google Scholar]
- Eisenberg 1998 {published data only} .Eisenberg E, Alon N, Ishay A, Daoud D, Yarnitsky D. Lamotrigine in the treatment of painful diabetic neuropathy. European Journal of Neurology. 1998;5(2):167–73. doi: 10.1046/j.1468-1331.1998.520167.x. DOI: 10.1046/j.1468-1331.1998.520167.x. [DOI] [PubMed] [Google Scholar]
- Eisenberg 2003 {published data only} .Eisenberg E, Damunni G, Hoffer E, Baum Y, Krivoy N. Lamotrigine for intractable sciatica: correlation between dose, plasma concentration and analgesia. European Journal of Pain. 2003;7(6):485–91. doi: 10.1016/S1090-3801(03)00020-X. DOI: 10.1016/S1090-3801 (03)00020-X. [DOI] [PubMed] [Google Scholar]
- Eisenberg 2005 {published data only} .Eisenberg E, Shifrin A, Krivoy N. Lamotrigine for neuropathic pain. Expert Review of Neurotherapeutics. 2005;5:729–35. doi: 10.1586/14737175.5.6.729. DOI: 10.1586/14737175.5.6.729. [DOI] [PubMed] [Google Scholar]
- Lunardi 1997 {published data only} .Lunardi G, Leandri M, Albano C, Cultrera S, Fracassi M, Rubino V, Favale E. Clinical effectiveness of lamotrigine and plasma levels in essential and symptomatic trigeminal neuralgia. Neurology. 1997;48(6):1714–7. doi: 10.1212/wnl.48.6.1714. [DOI] [PubMed] [Google Scholar]
- Petersen 2003 {published data only} .Petersen KL, Maloney A, Hoke F, Dahl JB, Rowbotham MC. A randomized study of the effect of oral lamotrigine and hydromorphone on pain and hyperalgesia following heat/capsaicin sensitization. Journal of Pain. 2003;4(7):400–6. doi: 10.1016/s1526-5900(03)00718-1. [DOI] [PubMed] [Google Scholar]
- Sandner-Kiesling 2002 {published data only} .Sandner-Kiesling A, Rumpold Seitlinger G, Dorn C, Koch H, Schwarz G. Lamotrigine monotherapy for control of neuralgia after nerve section. Acta Anaesthesiologica Scandinavica. 2002;46(10):1261–4. doi: 10.1034/j.1399-6576.2002.461014.x. DOI: 10.1034/j.1399-6576.2002.461014.x. [DOI] [PubMed] [Google Scholar]
Additional references
- Barthel 2010 .Barthel HR, Peniston JH, Clark MB, Gold MS, Altman RD. Correlation of pain relief with physical function in hand osteoarthritis: randomized controlled trial post hoc analysis. Arthritis Research & Therapy. 2010;12(1):R7. doi: 10.1186/ar2906. DOI: 10.1186/ar2906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breivik 2006 .Breivik H, Collett B, Ventafridda V, Cohen R, Gallacher D. Survey of chronic pain in Europe: prevalence, characteristics and mechanisms. European Journal of Pain. 2006;10(4):287–333. doi: 10.1016/j.ejpain.2005.06.009. DOI: 10.1016/j.ejpain.2005.06.009. [DOI] [PubMed] [Google Scholar]
- Cook 1995 .Cook RJ, Sackett DL. The number needed to treat: a clinically useful measure of treatment effect. BMJ 1995. 1995;310(6977):452–4. doi: 10.1136/bmj.310.6977.452. 310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dworkin 2008 .Dworkin RH, Turk DC, Wyrwich KW, Beaton D, Cleeland CS, Farrar JT, et al. Interpreting the clinical importance of treatment outcomes in chronic pain clinical trials: IMMPACT recommendations. The Journal of Pain. 2008;9(2):105–21. doi: 10.1016/j.jpain.2007.09.005. DOI: 10.1016/j.jpain.2007.09.005. [DOI] [PubMed] [Google Scholar]
- Elbourne 2002 .Elbourne DR, Altman DG, Higgins JP, Curtin F, Worthington HV, Vail A. Meta-analyses involving cross-over trials: methodological issues. International Journal of Epidemiology. 2002;31:140–149. doi: 10.1093/ije/31.1.140. DOI: 10.1093/ije/31.1.140. [DOI] [PubMed] [Google Scholar]
- Hall 2006 .Hall GC, Carroll D, Parry D, McQuay HJ. Epidemiology and treatment of neuropathic pain: the UK primary care perspective. Pain. 2006;122:156–62. doi: 10.1016/j.pain.2006.01.030. DOI: 10.1016/j.pain.2006.01.030. [DOI] [PubMed] [Google Scholar]
- Hirsch 2006 .Hirsch LJ, Weintraub DB, Buchsbaum R, Spencer HT, Straka T, Hager M, Resor SR., Jr Predictors of lamotrigine-associated rash. Epilepsia. 2006;47(2):318–22. doi: 10.1111/j.1528-1167.2006.00423.x. DOI: 10.1111/j.1528-1167.2006.00423.x. [DOI] [PubMed] [Google Scholar]
- Hoffman 2010 .Hoffman DL, Sadosky A, Dukes EM, Alvir J. How do changes in pain severity levels correspond to changes in health status 3 and function in patients with painful diabetic peripheral neuropathy? Pain. 2010;149(2):194–201. doi: 10.1016/j.pain.2009.09.017. DOI: 10.1016/j.pain.2009.09.017. [DOI] [PubMed] [Google Scholar]
- Jacox 1994 .Jacox A, Carr DB, Payne R, Agency for Health Care Policy and Research . Management of cancer pain. Clinical Practice Guideline No. 9. US Department of Health and Human Services, Public Health Service; Rockville, Maryland: 1994. [Google Scholar]
- Jadad 1996 .Jadad AR, Moore RA, Carroll D, Jenkinson C, Reynolds DJM, Gavaghan DJ, et al. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Controlled Clinical Trials. 1996;17(1):1–12. doi: 10.1016/0197-2456(95)00134-4. DOI: 10.1016/0197-2456(95)00134-4. [DOI] [PubMed] [Google Scholar]
- Jensen 2002 .Jensen TS. Anticonvulsants in neuropathic pain: rationale and clinical evidence. European Journal of Pain. 2002;6(Suppl A):61–8. doi: 10.1053/eujp.2001.0324. DOI: 10.1053/eujp.2001.0324. [DOI] [PubMed] [Google Scholar]
- Katusic 1991 .Katusic S, Williams DB, Beard CM, Bergstralh EJ, Kurland LT. Epidemiology and clinical features of idiopathic trigeminal neuralgia and glossopharyngeal neuralgia: similarities and differences, Rochester, Minnesota, 1945-1984. Neuroepidemiology. 1991;10:276–81. doi: 10.1159/000110284. [DOI] [PubMed] [Google Scholar]
- Khan 1996 .Khan KS, Daya S, Jadad A. The importance of quality of primary studies in producing unbiased systematic reviews. Archives of Internal Medicine. 1996;156(6):661–6. [PubMed] [Google Scholar]
- L’Abbe 1987 .L’Abbé KA, Detsky AS, O’Rourke K. Meta-analysis in clinical research. Annals of Internal Medicine. 1987;107:224–33. doi: 10.7326/0003-4819-107-2-224. [DOI] [PubMed] [Google Scholar]
- Lunn 2009 .Lunn MP, Hughes RA, Wiffen PJ. Duloxetine for treating painful neuropathy or chronic pain. Cochrane Database of Systematic Reviews. 2009;(4) doi: 10.1002/14651858.CD007115.pub2. DOI: 10.1002/14651858.CD007115.pub2. [DOI] [PubMed] [Google Scholar]
- McCleane 2000 .McCleane GJ. Lamotrigine in the management of neuropathic pain. The Clinical Journal of Pain. 2000;16:321–6. doi: 10.1097/00002508-200012000-00008. [DOI] [PubMed] [Google Scholar]
- McQuay 1998 .McQuay H, Moore R. An evidence-based resource for pain relief. Oxford University Press; Oxford: 1998. ISBN: 0-19-263048-2. [Google Scholar]
- Monks 1994 .Monks R. Psychotropic drugs. In: Wall PD, Melzack R, editors. Textbook of Pain. Churchill Livingstone: 1994. pp. 963–89. [Google Scholar]
- Moore 1998 .Moore RA, Gavaghan D, Tramèr MR, Collins SL, McQuay HJ. Size is everything - large amounts of information are needed to overcome random effects in estimating direction and magnitude of treatment effects. Pain. 1998;78(3):209–16. doi: 10.1016/S0304-3959(98)00140-7. DOI: 10.1016/S0304-3959(98)00140-7. [DOI] [PubMed] [Google Scholar]
- Moore 2005a .Moore RA, Edwards JE, McQuay HJ. Acute pain: individual patient meta-analysis shows the impact of different ways of analysing and presenting results. Pain. 2005;116(3):322–31. doi: 10.1016/j.pain.2005.05.001. DOI: 10.1016/j.pain.2005.05.001. [DOI] [PubMed] [Google Scholar]
- Moore 2005b .Moore RA, Derry S, Makinson GT, McQuay HJ. Tolerability and adverse events in clinical trials of celecoxib in osteoarthritis and rheumatoid arthritis: systematic review and meta-analysis of information from company clinical trial reports. Arthritis Research and Therapy. 2005;7(3):R644–65. doi: 10.1186/ar1704. DOI: 10.1186/ar1704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore 2009 .Moore RA, Straube S, Wiffen PJ, Derry S, McQuay HJ. Pregabalin for acute and chronic pain in adults. Cochrane Database of Systematic Reviews. 2009;(3) doi: 10.1002/14651858.CD007076.pub2. DOI: 10.1002/14651858.CD007076.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore 2010a .Moore RA, Moore OA, Derry S, Peloso PM, Gammaitoni AR, Wang H. Responder analysis for pain relief and numbers needed to treat in a meta-analysis of etoricoxib osteoarthritis trials: bridging a gap between clinical trials and clinical practice. Annals of the Rheumatic Diseases. 2010;69(2):374–9. doi: 10.1136/ard.2009.107805. DOI: 10.1136/ard.2009.107805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore 2010b .Moore RA, Straube S, Derry S, McQuay HJ. Topical review: chronic low back pain analgesic studies - a methodological minefield. Pain. 2010;149(3):431–4. doi: 10.1016/j.pain.2010.02.032. DOI: 10.1016/j.pain.2010.02.032. [DOI] [PubMed] [Google Scholar]
- Moore 2010c .Moore RA, Straube S, Paine J, Phillips CJ, Derry S, McQuay HJ. Fibromyalgia: Moderate and substantial pain intensity reduction predicts improvement in other outcomes and substantial quality of life gain. Pain. 2010;149(2):360–4. doi: 10.1016/j.pain.2010.02.039. DOI: 10.1016/j.pain.2010.02.039. [DOI] [PubMed] [Google Scholar]
- Moore 2010d .Moore RA, Eccleston C, Derry S, Wiffen P, Bell RF, Straube S, McQuay H, ACTINPAIN Writing Group of the IASP Special Interest Group on Systematic Reviews in Pain Relief, Cochrane Pain, Palliative and Supportive Care Systematic Review Group Editors Evidence” in chronic pain--establishing best practice in the reporting of systematic reviews. Pain. 2010;150(3):386–9. doi: 10.1016/j.pain.2010.05.011. DOI: 10.1016/j.pain.2010.05.011. [DOI] [PubMed] [Google Scholar]
- O’Brien 2010 .O’Brien EM, Staud RM, Hassinger AD, McCulloch RC, Craggs JG, Atchison JW, et al. Patient-centered perspective on treatment outcomes in chronic pain. Pain Medicine. 2010;11(1):6–15. doi: 10.1111/j.1526-4637.2009.00685.x. DOI: 10.1111/j.1526-4637.2009.00685.x. [DOI] [PubMed] [Google Scholar]
- Pain in Europe 2004 .Fricker J. Pain in Europe. A report. 2006 Feb;:2004. Accessed on line at www.painineurope.com.
- Rappaport 1994 .Rappaport ZH, Devor M. Trigeminal neuralgia: the role of self-sustaining discharge in the trigeminal ganglion. Pain. 1994;56:127–38. doi: 10.1016/0304-3959(94)90086-8. DOI: 10.1016/0304-3959(94)90086-8. [DOI] [PubMed] [Google Scholar]
- SPC 2007 . [Accessed 5.2.2007];Summary of product characteristics for Lamictal. Section 4.8. www.medicines.org.uk
- Straube 2008 .Straube S, Derry S, McQuay HJ, Moore RA. Enriched enrollment: definition and effects of enrichment and dose in trials of pregabalin and gabapentin in neuropathic pain. A systematic review. British Journal of Clinical Pharmacology. 2008;66(2):266–75. doi: 10.1111/j.1365-2125.2008.03200.x. DOI: 10.1111/j.1365-2125.2008.03200.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Straube 2010 .Straube S, Derry S, Moore RA, Paine J, McQuay HJ. Pregabalin in fibromyalgia--responder analysis from individual patient data. BMC Musculoskeletal Disorders. 2010;11:150. doi: 10.1186/1471-2474-11-150. DOI: 10.1186/1471-2474-11-150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sultan 2008 .Sultan A, Gaskell H, Derry S, Moore RA. Duloxetine for painful diabetic neuropathy and fibromyalgia pain: systematic review of randomised trials. BMC Neurology. 2008;8:29. doi: 10.1186/1471-2377-8-29. DOI: 10.1186/1471-2377-8-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiffen 2005 .Wiffen PJ, McQuay HJ, Edwards JE, Moore RA. Gabapentin for acute and chronic pain. Cochrane Database of Systematic Reviews. 2005;(3) doi: 10.1002/14651858.CD005452. DOI: 10.1002/14651858.CD005452. [DOI] [PubMed] [Google Scholar]
- Wiffen 2011 .Wiffen PJ, Derry S, Moore RA, McQuay HJ. Carbamazepine for acute and chronic pain in adults. Cochrane Database of Systematic Reviews. 2011;(1) doi: 10.1002/14651858.CD005451.pub2. DOI: 10.1002/14651858.CD005451.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng 2010 .Zheng C, Yang K, Liu Q, Wang MY, Shen J, Valles SA, et al. The anticonvulsive drug lamotrigine blocks neuronal{alpha}4{beta}2-nicotinic acetylcholine receptors. The Journal of Pharmacology and Experimental Therapeutics. 2010 Aug 5; doi: 10.1124/jpet.110.171108. Epub ahead of print. [DOI: 10.1124/jpet.110.171108] [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
- Wiffen 2007 .Wiffen PJ, Rees J. Lamotrigine for acute and chronic pain. Cochrane Database of Systematic Reviews. 2007;(2) doi: 10.1002/14651858.CD006044.pub2. DOI: 10.1002/14651858.CD006044.pub2. [DOI] [PubMed] [Google Scholar]
- * Indicates the major publication for the study