Abstract
Purpose.
Melanocytes are one of the major cellular components in the uvea. Interleukin-8/CXCL8 and monocyte chemoattractant protein-1 (MCP-1/CCL2) are the two most important proinflammatory chemokines. We studied the constitutive and lipopolysaccharide (LPS)-induced expression of IL-8 and MCP-1 in cultured human uveal melanocytes (UM) and explored the relevant signal pathways.
Methods.
Conditioned media and cells were collected from UM cultured in medium with and without stimulation of LPS. Interleukin-8 and MCP-1 proteins and mRNAs were measured using an ELISA kit and RT-PCR, respectively. Nuclear factor (NF)-κB in nuclear extracts and phosphorylated p38 mitogen-activated protein kinase (MAPK), extracellular signal-regulated kinases1/2 (ERK1/2), and c-Jun N-terminal kinase1/2 (JNK1/2) in cells cultured with and without LPS were measured by ELISA kits. Inhibitors of p38 (SB203580), ERK1/2 (UO1026), JNK1/2 (SP600125), and NF-κB (BAY11-7082) were added to the cultures to evaluate their effects.
Results.
Low levels of IL-8 and MCP-1 proteins were detected in the conditioned media in UM cultured without serum. Lipopolysaccharide (0.01–1 μg/mL) increased IL-8 and MCP-1 mRNAs and proteins levels in a dose- and time-dependent manner, accompanied by a significant increase of phosphorylated JNK1/2 in cell lysates and NF-κB in nuclear extracts. Nuclear factor–κB and JNK1/2 inhibitors significantly blocked LPS-induced expression of IL-8 and MCP-1.
Conclusions.
This is the first report on the expression and secretion of chemokines by UM. The data suggest that UM may play a role in the pathogenesis of ocular inflammatory diseases.
Keywords: uveal melanocyte, MCP-1, IL-8, LPS, p38 MAPK, JNK, NF-κB, uveitis
Cultured uveal melanocytes constitutively express IL-8 and MCP-1 at a low level, the expression is significantly increased by lipopolysaccharide stimulation via JNK1/2 and NF-κB pathways, suggesting that melanocytes may play a role in the pathogenesis of ocular inflammatory diseases.
Introduction
Uveal melanocytes (UM) are a major cell population in the uveal tract. In the past, little was known about the function of UM and their role in the pathogenesis of various ocular diseases. In the past few decades, we have developed protocols for the isolation and culture of human UM. Many pure cell lines of UM from the iris, ciliary body, and choroid have been established and have been used as an in vitro model for studying the role of UM in ocular physiology, immunology, and pathology.1–5 Previously, we have demonstrated that cultured human UM are capable of production of various melanins, matrix metalloproteinases (MMP), tissue plasminogen activator, IL-6, and VEGF in vitro.6–10
Chemokines, act as chemoattractants and activators of specific leukocytes and play a pivotal role in the influx of inflammatory cells into tissues and the pathogenesis of inflammation. CC and CXC are the two major families of chemokines. Interleukin-8/CXCL8 and monocyte chemattractant protein-1 (MCP-1/CCL2) are the most important and most studied chemokines and the prototypes of CXC and CC chemokines, respectively. Both are proinflammatory chemokines.11 Interleukin-8 is a potent chemoattractive factor mainly for neutrophils.11–16 Monocyte chemattractant protein-1 is chemotactic for monocytes, T lymphocytes, and natural killer cells.11,17,18 These two chemokines play an important role in various inflammatory diseases (e.g., arthritis, uveitis, and retinitis) and certain retinal diseases (e.g., diabetic retinopathy, proliferative vitreoretinopathy, AMD, and rhegmatogenous retinal detachment).11–38 Previous studies indicated that IL-8 and MCP-1 could be produced by a variety of cell types constitutively or after induction by various factors.11-18,39–49 However, the expression and secretion of IL-8 and MCP-1 by UM have not been reported in the literature.
Lipopolysaccharide (LPS) is an endotoxin, which can induce a strong inflammatory response by the human immune system.50 Lipopolysaccharide induces acute ocular inflammation in experimental animals (endotoxin-induced uveitis), which is an important animal model for acute anterior uveitis in the human.51–57 Both IL-8 and MCP-1 are critical chemokines in the development and regulation of endotoxin-induced uveitis. Lipopolysaccharide induces expression of IL-8 and MCP-1 in various cell types,50,58–63 but the effects of LPS on expression of these chemokines by UM have not been studied.
The purposes of the present study were to investigate the constitutive and LPS-induced expression of IL-8 and MCP-1 by UM and the relevant signal pathways involved in this process.
Methods
Cell Culture
Human UM were isolated from the iris, ciliary body, or choroid of adult donor eyes as previously described.1–5 Donor eyes were obtained from New York Eye Bank for Sight Restoration (New York, NY, USA).1 The Eye Bank obtained the donor's consent before the collection of the eyes. These donors were deceased due to various fatal diseases, such as myocardial infarction, stroke, and so on. The isolated UM were cultured with FIC medium, which is F12 medium supplemented with 10% fetal bovine serum (FBS), 2 mM glutamine, 20 ng/mL basic fibroblast growth factor, 0.1 mM isobutylmethylxanthine, 10 ng/mL cholera toxin, and 50 μg/mL gentamicin (FBS and F12 medium from Gibco, Invitrogen, Carlsbad, CA, USA; all others from Sigma-Aldrich Corp., St. Louis, MO, USA).1 After reaching confluence, the UM were detached using trypsin-EDTA solution (Sigma-Aldrich Corp.), diluted 1:3-1:6, and subcultured. Seven UM cell lines (two from the iris, one from the ciliary body, and four from the choroid) were used in the present study. One primary culture of human umbilical vein vascular endothelial cell line (HUVEC) with known MCP-1 and IL-8 production activity was tested for comparison. Human umbilical vein vascular endothelial cell lines were isolated as described previously and were cultured in 1% gelatin coated flask with Medium 199 (Gibco) supplemented with heparin (90 μg/mL), endothelial cell growth factor (20 μg/mL; all from Sigma-Aldrich Corp.) and 10% FBS.64–66 Cell cultures in the fifth passage were used in this study.
Basic Secretion of IL-8 and MCP-1 by UM and HUVEC
Early passages of seven cell lines of cultured UM were used in the present study. The ages of the donors that provided these UM cell lines ranged from 45 to 78 years, with a mean ± SD at 59.3 ± 11.4 (Table). Cells were plated into 24-well plates at a density of 1 × 105 per well. After 24 hours, the culture medium was withdrawn. The cultures were washed with PBS twice, and then serum-free culture medium was added. Conditioned medium was collected 24 hours later, centrifuged at 800g for 5 minutes, and the supernatants were transferred to vials and stored at −70°C until analysis. All experiments were performed in triplicate.
Table.
Secretion of IL-8 and MCP-1 in Uveal Melanocytes Cultured in Serum-Free Medium
|
Location |
Iris Color |
Age |
IL-8, pg/mL |
MCP-1, pg/mL |
|
| 1 | Iris | Hazel | 38 | 6.93 ± 1.01 | 24.7 ± 2.5 |
| 2 | Iris | Brown | 45 | 8.53 ± 1.33 | 23.3 ± 3.5 |
| 3 | Ciliary body | Green | 62 | 12.8 ± 2.68 | 24.0 ± 3.0 |
| 4 | Choroid | Green | 62 | 7.67 ± 0.42 | 22.7 ± 3.1 |
| 5 | Choroid | Blue | 62 | 5.50 ± 1.25 | 22.0 ± 2.7 |
| 6 | Choroid | Brown | 45 | 4.90 ± 1.39 | 19.3 ± 2.5 |
| 7 | Choroid | Hazel | 78 | 7.69 ± 0.34 | 25.0 ± 4.6 |
| Mean ± SD | 59.3 ± 11.4 | 7.72 ± 2.59 | 23.0 ± 3.1 |
Seven uveal melanocyte cell lines isolated from different locations of the uveal tract from donor eyes with various colored irides were plated into 24-well plates at a density of 1 × 105 per well. After 24 hours, the culture medium was withdrawn. The cultures were washed by PBS and serum-free culture medium. Conditioned medium was collected 24 hours later. The amount of IL-8 and MCP-1 protein in the conditioned medium was determined using the human IL-8 and MCP-1 Quantikine ELISA kit, respectively.
Early passages of a cultured cell line of HUVEC were plated into 24-well plates at a density of 1 × 105 per well. Conditioned medium was collected and stored as described above.
Effects of LPS on the Morphology, Viability, and Immunocytochemical Studies of UM
The effects of LPS on the morphology of UM were studied by phase-contrast microscopy of cells cultured with or without LPS at 0.01, 0.1, 1.0, and 10 μg/mL for 48 hours. Uveal melanocytes were seeded into 8-well plates and cultured with or without LPS at 0.01, 0.1, 1.0, and 10 μg/mL for 48 hours. Cultures were studied by immunocytochemical methods using anti-S-100, -cytokeratin and -α-smooth muscle actins antibodies as primary antibodies as described previously.1 The effects of LPS on the cell viability of UM were studied with the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) test as described previously.67 Briefly, UM were plated in 96-well plates at a density of 5 ×103 cells per well. After incubation for 24 hours, LPS (Sigma-Aldrich Corp.) was added to the wells at various final concentrations (0.01, 0.1, 1.0, and 10 μg/mL) and cultured for 24 hours. Next, 50 μL of tetrazolium bromide, MTT (1 mg/mL; Sigma-Aldrich Corp.) was added to each well and incubated for 4 hours. The medium was withdrawn and 100 μL of dimethyl sulfoxide (DMSO; Sigma-Aldrich Corp.) was added to each well. Optical density was read at 540 nm using a microplate reader (Multiskan EX; Thermo, Ventana, Finland). Cells cultured without LPS served as the controls. The effects of LPS on the cell viability were studied in one primary culture of UM. All groups were tested in triplicate.
Secretion of IL-8 and MCP-1 in LPS-Stimulated UM
Early passages of cultured UM (from the choroid) were plated into 24-well plates at a density of 1 × 105 per well. In a dose-effect study, after 24 hours culture, the cultured medium was withdrawn and replaced with serum-free medium after washing the cells. Lipopolysaccharide at different concentrations (0, 0.01, 0.1, and 1.0 μg/mL) was added to the media. After 24 hours, conditioned media were collected and stored as described above. In a time-effect study, cultured cells were plated as described above, the cultured medium was withdrawn and replaced with serum-free medium after washing the cells. Lipopolysaccharide (0.1 μg/mL) was added, and conditioned media (with and without LPS) were collected at 0, 6, 12, and 24 hours later and stored. All experiments were performed in triplicate.
Measurement of IL-8 and MCP-1 Protein
The amount of IL-8 and MCP-1 proteins in the conditioned media was determined using the human IL-8 and MCP-1 Quantikine ELISA kits (R&D Systems, Minneapolis, MN, USA) according to the manufacturer's instructions. Optical density was read by using a microplate reader at 450 nm and corrected with 540 nm. The amounts of IL-8 and MCP-1 (pg/mL) were calculated from a standard curve. The sensitivity of the IL-8 and MCP-1 kits was 3.5 and 5.0 pg/mL, respectively.
RNA Isolation and RT-PCR
Early passages of cultured UM were plated into 6-well plates at a density of 5 × 105. After 24 hours, the culture medium was replaced with serum-free culture medium. In a time-effect study, LPS at 0.1 μg/mL was added into the culture medium, and cells were collected 2, 6, and 24 hours later. After the culture medium was withdrawn, the cultures were washed with cold PBS and cells were harvested by scraping with a rubber policeman. Cells cultured without LPS were used as negative controls. After microcentrifuging at 800g for 5 minutes at 4°C, cell pellets were collected for mRNA extraction. Total RNA was isolated with the RNeasy mini kit (Qiagen, Valencia, CA, USA), according to the manufacturer's instructions. The SuperScript first-strand synthesis system for RT-PCR kit (Invitrogen, Camarillo, CA, USA) was used to perform cDNA synthesis. The PCR primers for glyceraldehyde-3-phosphate dehydrogenase (GAPDH) were TGAACTGAAAGCTCTCCACC and CTGATGTACCAGTTGGGGAA. Interleukin-8 primers were TTTTGCCAAGGAGTGCTAAAGA and AACCCTCTGCACCCAGTTTTC. Monocyte chemoattractant protein-1 primers were GATGCAATCAATGCCCCAGTC and TTTGCTTGTCCAGGTGGTCCAT. All primers were obtained from Invitrogen. The first-strand cDNA were synthesized from 0.5 μg of total RNA at 50°C for 50 minutes. Polymerase chain reaction amplification was conducted in a GeneAmp PCR system 9700 (Applied Biosystems, Foster City, CA) using the following parameters: first denaturation at 94°C for 5 minutes followed by 35 cycles of reactions of denaturation at 94°C for 30 seconds, annealing at 58°C for 45 seconds, and extension at 72°C for 45 seconds, and last extension for 5 minutes at 72°C. After amplification, samples were run on a 1% agarose gel (Invitrogen) in Tris-borate (TBE; 0.01 M), 0.001 M EDTA (Invitrogen) containing 2.0 μg/mL ethidium bromide (Invitrogen). Bands were visualized and photographed on a UV transilluminator (ChemiDoc XRS System; Bio-Rad, Hercules, CA, USA). In the dose-effect study, LPS at different concentrations (0, 0.01, 0.1, and 1.0 μg/mL) were added to the medium. After 24 hours, cells were collected, treated, and RT-PCR performed as described above.
p38, ERK, and JNK Map Kinase Assays in Cultured UM With and Without LPS
Uveal melanocytes were plated into 6-well plates at a density of 1 × 106. After 24 hours, LPS (0.1 μg/mL) was added. After 60 minutes the cultures were washed with cold PBS, and cells were harvested by scraping with a rubber policeman. Cells cultured without LPS were used as negative controls. After microcentrifuging for 5 minutes at 4°C, pellets were treated with ice-cold cell extraction buffer (Biosource, Camarillo, CA, USA) with protease inhibitor cocktail (Sigma-Aldrich Corp.) and phenylmethanesulfonyl fluoride (PMSF; Biosource) for 30 minutes, with subsequent vortexing at 10-minute intervals. Cell extractions were microcentrifuged for 30 minutes at 4°C. The supernatants were collected into vials and stored at −70°C until analysis. Phosphorylated p38 mitogen-activated protein kinase (MAPK), extracellular signal-regulated kinases1/2 (ERK1/2), and c-Jun N-terminal kinase1/2 (JNK1/2) measurements were performed in triplicate by using p38 MAPK, ERK, and JNK ELISA kits (Biosource), respectively, according to the protocol outlined by the manufacturer and were expressed as percentages of the control (cells not exposed to LPS). The sensitivity of these kits was 0.8 U/ml.
Assay of NF-κB in Nuclear Extracts in Cultured UM With and Without LPS
Uveal melanocytes were plated into 6-well plates at a density of 1 × 106 cells per well. After 24 hours incubation, the medium was replaced and LPS (0.1 μg/mL) was added to the medium as described above. Cells cultured without LPS were used as negative controls. After 30 minutes, the culture medium was withdrawn. Cells were washed with cold PBS and then scraped from the well. Cells were treated with hypotonic buffer (BioSource) and centrifuged. The pellet (nuclear fraction) was collected and treated with cell extraction buffer (BioSource), vortexed, and centrifuged. The supernatants (nuclear extracts) were stored at −70°C until analysis. The amount of nuclear factor–kappa B (NF-κB) in cell nuclear extracts was measured by using NF-κB ELISA kits (Invitrogen) according to the manufacturer's instructions. The levels of NF-κB in nuclear extracts were calculated using a standard curve and expressed as percentages of the negative controls. The sensitivity of this kit was less than 50 pg/ml. All tests were performed in triplicate.
Application of MAPK and NF-κB Inhibitors on LPS-Stimulated UM
Uveal melanocyte cells were plated into 24-well plates at a density of 1 × 105 cells per well. After 24 hours incubation, the medium was changed, and various MAPK or NF-κB inhibitors were added to the medium separately, including 3 μM BAY11-7082 (NF-κB inhibitor), 10 μM UO1026 (ERK inhibitor), 10 μM SP600125 (JNK inhibitor), and 10 μM SB203580 (p38 MAPK inhibitor; all from Calbiochem, San Diego, CA, USA). Thirty minutes later, LPS was added to the medium at a final concentration of 1.0 μg/ml. Cells cultured without LPS were used as negative controls. Cells cultured with LPS and without any inhibitors were used as positive controls. After 24 hours incubation, the conditioned media were collected and stored.
Statistical Analysis
Statistical significances of difference of means throughout this study were calculated by one-way ANOVA test in comparing data from more than two groups and Student's t-test in comparing data between two groups. A difference at P less than 0.05 was considered to be statistically significant.
Results
Expression and Secretion of IL-8 and MCP-1 in UM Cultured With Serum-Free Culture Medium
Low levels of IL-8 and MCP-1 protein were detected in the conditioned medium from all seven UM cell lines cultured with serum free culture medium (Table). The range of IL-8 and MCP-1 levels was 5.50 to 12.8 pg/mL and 19.3 to 25.0 pg/mL in these seven cell lines, with an average of 7.72 ± 2.59 and 23.0 ± 3.1 pg/mL (mean ± SD), respectively. The baseline measurements of these two chemokines might represent the basic (constitutive) secretion of IL-8 and MCP-1 by normal UM in vitro.
Interleukin-8 was below detectable levels in the conditioned culture medium from HUVEC. Monocyte chemoattractant protein-1 levels in the conditioned culture medium from HUVEC were 31.3 ± 3.1 pg/ml.
Effects of LPS on the Morphology, Viability, and Immunocytochemical Studies of UM
The morphology of the UM cultured with LPS at 0.01 to 10 μg/mL was not affected as observed by phase-contrast microscopy (Fig. 1A). Uveal melanocytes expressed S-100, but no cytokeratin and α-smooth muscle keratin as previously described.1 The expression of cytokeratin, α-smooth muscle keratin and S-100 of UM did not change after the treatment of LPS at 0.01 to 10 μg/mL (data not shown). The viability of the UM cultured with LPS at 0.01 to 10 μg/mL was not affected by MTT test (Fig. 1B). Therefore, LPS at 0.01 to 1.0 μg/mL was used for the studies of effects of LPS on the expression and release of IL-8 and MCP-1 from UM.
Figure 1.
Effect of LPS on morphology and viability of UM. (A) Human UM were seeded to 12-well plates, cultured with LPS at 10 μg/mL for 48 hours and cell morphology was observed by phase-contrast microscopy (0, without LPS, and LPS, treated with LPS 10 μg/mL). (B) Human UM were seeded into 96-well plates and treated with LPS at 0.01, 0.1, 1.0, and 10 μg/mL for 24 hours and cell viability was determined by MTT assay (see Methods). Lipopolysaccharide at all tested levels did not affect the viability of UM. Data are mean ± SD (n = 3).
Effects of LPS on IL-8 and MCP-1 Production by UM
When different concentrations (0.01–1.0 μg/mL) of LPS were added to the culture medium of UM for 24 hours, both IL-8 and MCP-1 protein levels in the conditioned medium were significantly increased in a dose-dependent manner (Fig. 2). Interleukin-8 and MCP-1 levels in conditioned medium from cells cultured without LPS was 6.93 ± 1.01 and 22.0 ± 2.7 pg/mL (mean ± SD), respectively. Interleukin-8 levels in conditioned medium from cells cultured with LPS at 0.01, 0.1, and 1.0 μg/mL were 7.17, 28.2, and 50.6 times control values, respectively (Fig. 2A). Monocyte chemoattractant protein-1 levels in conditioned medium from cells cultured with LPS at 0.1, 1.0, and 10 μg/mL were 6.76, 8.71, and 10.8 times control values, respectively (Fig. 2B). The differences of IL-8 and MCP-1 levels were statistically significant (higher) between LPS-treated cells and the controls (P < 0.05).
Figure 2.
Dose-dependent effects of LPS on secretion of IL-8 (A) and MCP-1(B) by cultured human UM. Cells were plated into 24-well plates. After 24-hours incubation, LPS at 0, 0.01, 0.1, and 1.0 μg/mL was added to the culture and incubated for 24 hours. Conditioned culture media were collected and the amount of IL-8 and MCP-1 protein in the conditioned medium was determined by using the human IL-8 and MCP-1 Quantikine ELISA kit. Interleukin-8 and MCP-1 levels in conditioned culture medium were expressed as pictograms per milliliter (mean ± SD in triplicate tests). *P < 0.05, compared with the controls (cells cultured without LPS).
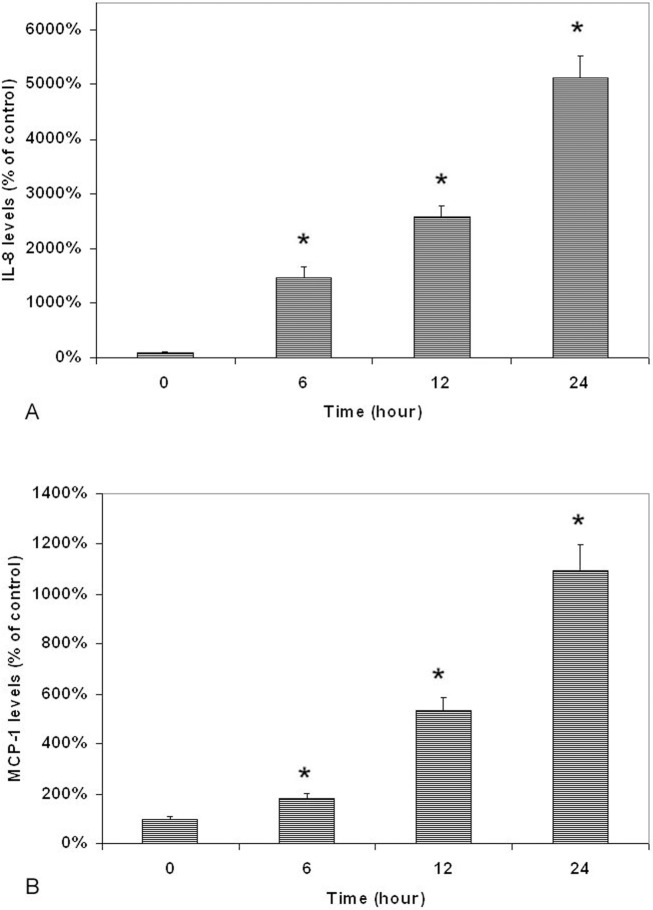
Lipopolysaccharide-induced increase of secretion of IL-8 and MCP-1 by UM was also time-dependent (Fig. 3). The differences of IL-8 (Fig. 3A) and MCP-1 levels (Fig. 3B) between LPS-treated cells and the controls were statistically significantly (higher) in cells treated with 0.1 μg/mL LPS for 6, 12, and 24 hours (P < 0.05).
Figure 3.
Time-dependent effects of LPS on secretion of IL-8 (A) and MCP-1 (B) by cultured human UM. Cells were plated into 24-well plates. After 24 hours incubation, LPS (0.1 μg/mL) was added to the culture and cultured for 6, 12, and 24 hours. Conditioned culture media were collected and the amount of MCP-1 protein in the conditioned medium was determined by using the human IL-8 and MCP-1 Quantikine ELISA kit. Interleukin-8 and MCP-1 levels in conditioned culture medium were expressed as pictograms per milliliter (mean ± SD in triplicate tests). *P < 0.05, compared with the controls (cells cultured without LPS).
Effects of LPS on IL-8 and MCP-1 mRNA Expression by UM
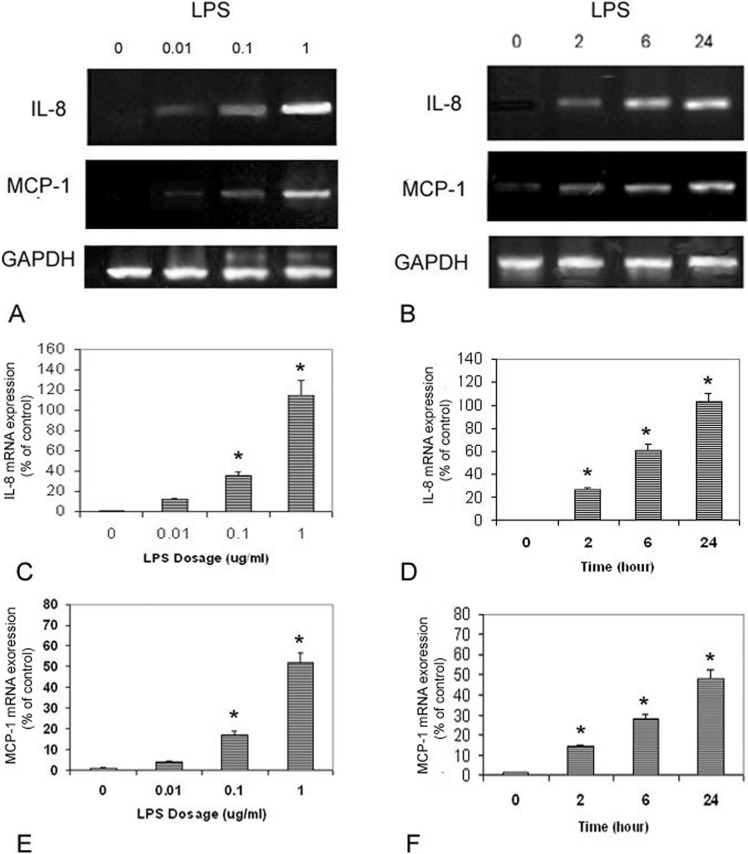
The RT-PCR experiment demonstrated that IL-8 and MCP-1 mRNAs were lower expressed in UM cultured without LPS (Fig. 4). Lipopolysaccharide upregulated IL-8 mRNA expression and MCP-1 mRNA expression in the UM. In the time-dependent study, IL-8 and MCP-1 mRNAs expression in LPS (0.1 μg/mL)-treated cells increased significantly after 2, 6, and 24 hours treatments (P < 0.05). In the dose dependent study, IL-8 and MCP-1 mRNAs expression in cells treated with 0.01, 0.1, and 1.0 μg/mL LPS were also significantly increased (P < 0.05).
Figure 4.
Dose- and time-dependent effects of LPS on IL-8 and MCP-1 mRNAs expression by human UM. Representative RT-PCR profiles from three experiments showed the mRNAs expression of IL-8 and MCP-1 by cells exposed to LPS at different concentrations and time periods (A, B). Glyceraldehyde-3-phosphate dehydrogenase was used as an internal loading control. Cells were plated into 6-well plates. Lipopolysaccharide at 0, 0.01, 0.1, and 10 μg/mL was added to the culture. Twenty-four hours later, cells were collected, mRNA was extracted and RT-PCR analysis was performed as described in the text ([C] IL-8 and [E] MCP-1). Cells were plated into 6-well plates. Lipopolysaccharide (0.1 μg/mL) was added, cells were collected at 2, 6, and 24 hours later, mRNA was extracted and RT-PCR analysis was performed as described in the text ([D] IL-8 and [F] MCP-1).
Effects of LPS on NF-κB and Phosphorylated MAPK Levels in UM
Lipopolysaccharide treatment (0.1 μg/mL with 30 minutes incubation) increased phosphorylated ERK1/2, JNK1/2, and p38 MAPK levels in UM (Fig. 5). The levels of phosphorylated ERK1/2, JNK1/2, and p38 MAPK in UM cultured with LPS increased to 1.26 ± 0.09 (mean ± SD), 4.27 ± 0.50, and 1.55 ± 0.14 times control values (UM cell cultured without LPS). The differences of phosphorylated ERK1/2, JNK1/2, and p38 MAPK levels between cells treated with and without LPS were also statistically significant (P < 0.05).
Figure 5.

Effects of LPS on NF-κB in nuclear extracts and phosphorylated ERK, JNK, and p38 MAPK in cultured UM. Cells were plated into 24-well plates. After 24-hours incubation, LPS (0.1 μg/mL) was added to the medium. Cells were collected 30 minutes later. The amount of NF-κB levels in nuclear extracts and phosphorylated p38 MAPK, JNK, and ERK1/2 in cell lysates were measured using the relevant NF-κB ELISA kit and phosphorylated MAPK ELISA kits. The levels of NF-κB in nuclear extracts and phosphorylated p38, ERK, and JNK in cell lysates were expressed as the percentages of the controls (cells cultured without LPS). *P < 0.05, compared with the controls.
Lipopolysaccharide treatment increased NF-κB levels in nuclear extracts of the UM (Fig. 5). The levels of NF-κB in nuclear extracts in cells cultured with LPS were 3.73 ± 0.23 times control values. The difference of NF-κB levels between cells treated with and without LPS was statistically significant (P < 0.05).
Effects of MAPK and NF-κB Inhibitors on LPS-Induced Release of IL-8 and MCP-1 by UM
Interleukin-8 protein levels in conditioned media from cells cultured with and without LPS were 386 ± 17.8 (positive control) and 7.23 ± 0.65 pg/mL (negative control), respectively (Fig. 6A).
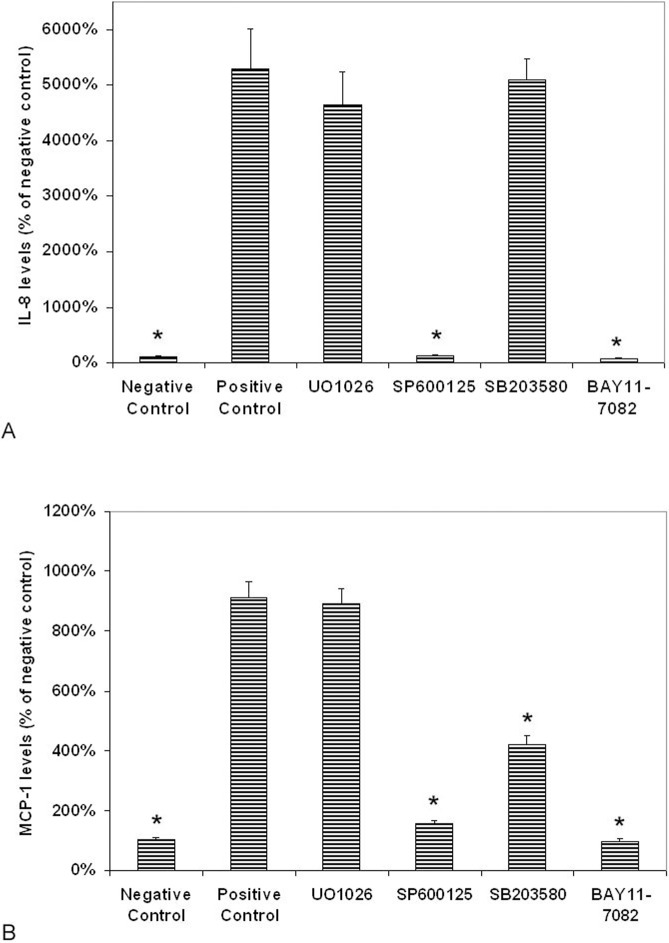
Figure 6.
Effects of NF-κB and MAPK inhibitors on LPS-induced production of IL-8 and MCP-1 by uveal melanocytes. Cells were plated into 24-well plates. After 24-hours incubation, various MAPK and NF-κB inhibitors were added to the medium separately, including BAY11-7082 (NF-κB inhibitor), UO1026 (ERK inhibitor), SP600125 (JNK inhibitor), and SB203580 (p38 MAPK inhibitor) at a final concentration of 10 μM with exception of BAY11-7082 (3 μM). Thirty minutes later, LPS (1.0 μg/mL) was added to the medium. Cells cultured without LPS were used as negative controls. Cells cultured with LPS but without inhibitors were used as positive controls. After 24 hours incubation, culture medium was collected and IL-8 (A) and MCP-1 (B) levels were measured with human IL-8 and MCP-1 Quantikine ELISA kit and expressed as pictograms per milliliter (mean ± SD in triplicate tests). *P < 0.05, compared with the positive controls (cells cultured with LPS but without inhibitors).
Treatment of cells with SB203580 (p38 MAPK inhibitor) before the addition of LPS did not cause significant changes of IL-8 levels in conditioned medium as compared with cells cultured with LPS alone (the positive control; P > 0.05; Fig. 6A). Treatment of cells with SP600125 (JNK1/2 inhibitor) nearly completely blocked the LPS-induced secretion of IL-8 (P < 0.05 as compared with the positive controls; Fig. 6A). UO1026 (ERK inhibitor) only caused a slight decrease of secretion of IL-8 as compared with the positive controls; the difference between these two groups was not statistically significant (P > 0.05; Fig. 6A).
Treatment with BAY11-7082 (NF-κB inhibitor) completely blocked the LPS-induced secretion of IL-8 (P < 0.05 as compared with the positive controls; Fig. 6A). The difference in the amount of IL-8 in medium between cells treated with and without BAY11-7082 (positive control) was significant (P < 0.05).
Monocyte chemoattractant protein-1 protein levels in conditioned media from cells cultured with and without LPS were 198 ± 11.6 (positive control) and 21.7 ± 2.1 pg/mL (negative control), respectively (Fig. 6B).
Treatment of cells with UO1026 (ERK inhibitor) before the addition of LPS did not cause significant changes of MCP-1 levels in conditioned medium as compared with cells cultured with LPS alone (P > 0.05; Fig. 6B). Treatment of cells with SP600125 (JNK1/2 inhibitor) caused a prominent decrease of the secretion of MCP-1 as compared with the positive controls (Fig. 6B). SB203580 (p38 MAPK inhibitor) caused a significant and moderate decrease of secretion of MCP-1 as compared with the positive controls (P < 0.05; Fig. 6B).
Treatment with BAY11-7082 (NF-κB inhibitor) decreased the release of MCP-1 by the UM to 10% of that in cells treated by LPS only (Fig. 6B). The difference in the amount of MCP-1 in medium between cells treated with and without BAY11-7082 (positive control) was significant (P < 0.05).
Discussion
Uveal melanocytes is one of the major resident cells in the uveal tract, but the investigation of UM in vitro has been hampered by an inability to obtain a sufficient number of pure UM for study. Since 1990, we have developed the methods for the isolation, cultivation, purification, and investigation of human UM. This in vitro model of UM has been used for studying the growth, function, and melanogenesis of these cells over the past 20 years.1–5 Several bioactive factors have been found to be expressed in cultured human UM, including one cytokine (IL-6).6–10 However, the production of chemokines by UM has not been studied previously.
Chemokines are small heparin-binding proteins, which act as chemoattractants and activators of leukocytes and play an important role in the influx of inflammatory cells. More than 50 human chemokines and 20 chemokine receptors have been identified. Chemokines could be classified into four subfamilies based on the number and location of the cysteine residues at the N-terminus of the molecule and are named CXC, CC, CX3C, and C, in agreement with the systematic nomenclature. In the two main subfamilies (CC and CXC), CXC chemokines are important in the attraction of neutrophils and CC chemokines have powerful chemattractants and activators for monocytes and lymphocytes.11,17,18
Interleukin-8 (CXCL8) is encoded by the IL8 gene and is a prototype of CXC chemokine family with chemoattractant properties for neutrophils. It also triggers the migration and adhesion of T cells, monocytes, and basophils to vascular endothelium and leads to extravasation of these cells into the tissues.11–13 Expression of IL-8 has been observed in various tissues during acute inflammation caused by bacteria and virus infections and is also associated with severe trauma, cancer, and various chronic inflammatory conditions such as rheumatoid arthritis, psoriasis, Behçet's disease, polyarteritis, and osteoarthritis.11,12,14–16 Interleukin-8 has potent angiogenic activity. It stimulates migration and proliferation of vascular endothelial cells and induces neovasculariztion.11,13
Monocyte chemoattractant protein-1 is encoded by the CCL2 gene and is a member of CC chemokine family with chemoattractant properties for monocytes, memory T cells, natural killer cells, and basophils.11,17,18 It triggers the adhesion of monocytes to vascular endothelium and leads to extravasation of monocytes into the tissues. Monocyte chemoattractant protein-1 is one of the key chemokines that regulate the migration and infiltration of monocytes and is also a key factor involved in the initiation of inflammation.11,17,18 Expression of MCP-1 has been observed in a large number of tissues in various inflammatory diseases and also in atherosclerosis and cancer.11,17,18
In the eye, IL-8 and MCP-1 have been detected in the aqueous humor and vitreous in nonclassical inflammatory eyes, such as cataracts, and idiopathic epiretinal membrane.21,22,26,32,34,36 Interleukin-8 and MCP-1 levels are significantly increased in aqueous humor or vitreous from eyes with uveitis, including both infectous or noninfectuous uveitis, idiopathic iridocyclitis,21 Behçet's diseases,21,26 Fuchs' heterochromatic cyclitis,21 Vogt-Koyanagi-Harada disease,26 sarcoidosis,27 lens-induced uveitis,23 Posner-Schlossman syndrome,24 herpes uveitis,21 tuberculous uveitis25 and toxoplasmosis.22 Serum IL-8 or MCP-1 are reported to increase in ocular sarcoidosis and Behçet's diseases.15,27 In experimental animal studies, elevated IL-8 or MCP-1 levels have been found in autoimmune anterior uveitis induced by melanin associated antigen.68,69 Intraocular injection of IL-8 induces uveitis with many leukocytes in the aqueous humor.70 These results indicate that IL-8 and MCP-1 contribute to the recruitment of inflammatory cells into the eye during the development of ocular inflammation and play an important role in the pathogenesis of uveitis.
Interleukin-8 and MCP-1 levels are significantly increased in the aqueous humor or vitreous from eyes with chronic low-grade inflammation or neovascularization (e.g., diabetic retinopathy,28–33 retinal vein occlusion,29,32,36 rhegmatogenous retinal detachment,32–34 proliferative vitreoretinopathy,31,34,35 and AMD38) indicating that IL-8 and MCP-1 play an important role in the pathogenesis of these vitreoretinal disorders.
Interleukin-8 is secreted by fibroblasts, keratinocytes, chondrocytes, synovial cells, vascular endothelial cells, glial cells, monocytes, T cells, neutrophils, tumor cells, and various epithelial cells including RPE cells.11,12,19,41,45–49,58 Most cell types do not produce or only produces a low level of IL-8 constitutively, but production of IL-8 can be rapidly induced by proinflammatory cytokines, viruses, bacteria and their products (e.g., LPS) or hypoxia.11,19 However, the expression of IL-8 by UM has not been reported previously.
Monocyte chemoattractant protein-1 is secreted by vascular endothelial cells, fibroblasts, glial cells, monocytes, T cells, vascular smooth muscle cells, and epithelial cells including RPE cells, either constitutively or induced by endotoxin, inflammatory cytokines, or oxidative stress.17,18,39–43,47,58 However, the expression of MCP-1 by UM has not been reported previously.
In the present study, cultured human UM consistently express IL-8 and MCP-1 mRNAs and secrete low levels of IL-8 and MCP-1 at basal conditions (cultured with serum-free culture medium), indicating a constitutive expression of IL-8 and MCP-1 in normal UM. The secretion levels of IL-8 and MCP-1 by UM are comparable with those in the HUVEC. The main significance of constitutively expressed IL-8 and MCP-1 at low levels might be to direct the normal trafficking of leucocytes under normal physiological conditions.11 Interleukin-8 and MCP-1 also have other biological functions such as angiogenic effects.11,18 Therefore, they could interact with other chemokines, cytokines, and growth factors to maintain homeostasis in a constant microenviroment.11
Lipopolysaccharide is the major component of the outer membrane of gram-negative bacteria and can induce a strong response from the human immune system. Various LPS-bearing bacteria can infect humans and cause certain infectious diseases.50 High plasma levels of LPS have been detected in patients with chronic infections.58 Circulating gram-positive bacteria can cause metastatic sepsis in the eye, a condition which accounts for approximately 32% to 37% of endogenous endophthalmitis cases in western and 70% in eastern countries.58 Lipopolysaccharide induces the expression of IL-8 and MCP-1 in various cell types.58–63,71 Lipopolysaccharide can induce endotoxin-induced uveitis, an acute ocular inflammation in various experimental animal models.51–57 Lipopolysaccharide-induced uveitis could be partially blocked by anti-IL-8 or anti–MCP-1 antibodies treatment.57 Lipopolysaccharide-induced uveitis was strongly reduced in MCP-1-deficient mice (Ccl2−/−) compared with wild-type control mice.54 The mechanism of LPS action is via Toll-like receptor 4 (TLR-4).50 Therefore, IL-8 and MCP-1 may play a crucial role in LPS-induced uveitis. In the present study, LPS increases the expression of IL-8 and MCP-1 mRNAs and the secretion of IL-8 and MCP-1 proteins by human UM in vitro in a dose- and time-dependent manner. This indicates that LPS is one of the important factors that regulate the secretion of IL-8 and MCP-1 in cultured human UM.
There are two main signal pathways involved in LPS-induced expression of IL-8 and MCP-1 by other cell types: MAPK and NF-κB signal pathways.60,62,63,71 The MAPK pathways mainly consist of ERK1/2, JNK1/2 and p38 MAPK pathways. The ERK1/2 pathway responds to growth factors and has an important role in modulating the survival and growth of cells, whereas p38 MAPK and JNK pathways response to various stresses, including proinflammatory cytokines and chemokines, and modulate differentiation and apoptosis of cells.10 Lipopolysaccharide is reported to stimulate the production of IL-8 and MCP-1 in various cells through the activation of one or more of these three MAPK pathways (mainly through the JNK1/2 pathway).60,62,63,71 Nuclear factor–κB is a major transcription factor and is present in the cytoplasm in an inactive NF-κB complex via their nonconvalent interaction with inhibitory proteins known as inhibitors of κB (IκBs). Various stimuli, including LPS, activate the latent cytoplasmic NF-κB/IκBα complex by phosphorylation on conserved serine residues in the N-terminal portion of IκB. Activated NF-κB translocates to the nucleus and induces the expression of relevant genes, including various cytokines and chemokines.10 It has been reported that LPS-induced expression of IL-8 and MCP-1 is through the activation and translocation of NF-κB to the nucleus.63,71
The involvement of MAPK or NF-κB pathway in LPS-induced IL-8 or MCP-1 expression is cell-type specific.60,62,63,71 Interestingly, cultured human epidermal melanocytes express a panel of functional TLRs, including TLR-4. These melanocytes can produce large amounts of IL-8 and MCP-1 via NF-κB signaling pathway after TLR stimulation by LPS.72 The present study found that LPS causes a prominent increase of JNK1/2 and NF-κB, but only a slight to moderate increase of ERK1/2 and p-38. Lipopolysaccharide-induced secretion of IL-8 was significantly inhibited by JNK1/2 and NF-κB inhibitors, but not by p38 and ERK inhibitors. LPS-induced secretion of MCP-1 was significantly inhibited by JNK1/2 and NF-κB inhibitors, and to a less degree, by a p38 inhibitor, but not by ERK inhibitors. Therefore, LPS-induced secretion of IL-8 and MCP-1 by UM is mainly through the activation of JNK1/2 and NF-κB signal pathways. This is consistent with previous reports regarding the signal pathways involved in LPS-induced secretion of IL-8 and MCP-1 by other cell types.60,62,63,71
In conclusion, the present studies demonstrate that UM constitutively express and secrete IL-8 and MCP-1 at a low level, and chemokines production is increase by LPS stimulation via JNK1/2 and NF-κB pathway. This suggests that UM, as the major cell component of the uveal tract, can secrete chemokines and recruit neutrophils, monocytes, and lymphocytes into the eye after proinflammatory stimulation and may play a role in the development of ocular inflammatory processes, especially in the development of uveitis.
Acknowledgments
Supported by the Pathology Research Funds of New York Eye and Ear Infirmary (New York, NY, USA).
Disclosure: D.-N. Hu, None; M. Bi, None; D.Y. Zhang, None; F. Ye, None; S.A. McCormick, None; C.-C. Chan, None
References
- 1. Hu DN, McCormick SA, Ritch R, Pelton-Henrion K. Studies of human uveal melanocytes in vitro: isolation, purification and cultivation of human uveal melanocytes. Invest Ophthalmol Vis Sci. 1993; 33: 2210–2219 [PubMed] [Google Scholar]
- 2. Hu DN, McCormick SA, Ritch R. Studies of human uveal melanocytes in vitro: growth regulation of cultured human uveal melanocytes. Invest Ophthalmol Vis Sci. 1993; 34: 2220–2227 [PubMed] [Google Scholar]
- 3. Hu DN, McCormick SA, Orlow SJ, Rosemblat S, Lin AY, Wo K. Melanogenesis in cultured human uveal melanocytes. Invest Ophthalmol Vis Sci. 1995; 36: 931–938 [PubMed] [Google Scholar]
- 4. Hu DN, McCormick SA, Orlow SJ, Rosemblat S, Lin AY. Regulation of melanogenesis by human uveal melanocytes in vitro. Exp Eye Res. 1997; 64: 397–404 [DOI] [PubMed] [Google Scholar]
- 5. Hu DN. Regulation of growth and melanogenesis of uveal melanocytes (Keynote Lecture). Pigment Cell Res. 2000; 13( suppl 8); 81–86 [DOI] [PubMed] [Google Scholar]
- 6. Wang Y, Hu DN, McCormick SA, Savage HE, O'Rourke J. Tissue plasminogen activator is released into cultured medium by cultured human uveal melanocytes. Pigment Cell Res. 2002; 15: 373–378 [DOI] [PubMed] [Google Scholar]
- 7. Chu SC, Hu DN, Yang SF, et al. Uveal melanocytes produce matrix metalloproteinases-2 and -9 in vitro. Pigment Cell Res. 2004; 17: 636–642 [DOI] [PubMed] [Google Scholar]
- 8. Wakamatsu K, Hu DN, McCormick SA, Ito S. Characterization of melanin in human iridal and choroidal melanocytes from eyes with various colored irides. Pigment Cell Res. 2008; 21: 97–105 [DOI] [PubMed] [Google Scholar]
- 9. Hu DN, McCormick SA. Biochemical pathways: different gene expression and cellular pathways determining tumor phenotype comparison of uveal melanocytes and uveal melanoma cells in vitro. In: DM Albert, Polans A. eds Ocular Oncology. New York, NY: Marcel Dekker; 2003: 189–210 [Google Scholar]
- 10. Hu DN, Chen M, Zhang DY, Ye F, McCormick SA, Chan CC. Interleukin-1β increases baseline expression and secretion of interleukin-6 by human uveal melanocytes in vitro via p38 MAPK/NF-κB pathway. Invest Ophthalmol Vis Sci. 2011; 52: 3767–74 [DOI] [PubMed] [Google Scholar]
- 11. Thomson AW, Lotze MT. eds The Cytokine Handbook. 4th ed San Diego, CA: Elsevier Science Ltd; 2003: 809–822, 1049–1081, 1083–1100 [Google Scholar]
- 12. Baggiolini M, Clark-Lewis I. Interleukin-8, a chemotactic and inflammatory cytokine. FEBS Lett. 1992; 307: 97–101 [DOI] [PubMed] [Google Scholar]
- 13. Mukaida N, Harada A, Matsushima K. Interleukin-8 (IL-8) and monocyte chemotactic and activating factor (MCAF/MCP-1), chemokines essentially involved in inflammatory and immune reactions. Cytokine Growth Factor Rev. 1998; 9: 9–23 [DOI] [PubMed] [Google Scholar]
- 14. Hoch RC, Schraufstätter IU, Cochrane CG. In vivo, in vitro, and molecular aspects of interleukin-8 and the interleukin-8 receptors. J Lab Clin Med. 1996; 128: 134–145 [DOI] [PubMed] [Google Scholar]
- 15. Katsantonis J, Adler Y, Orfanos CE, Zouboulis CC. Adamantiades-Behçet's disease: serum IL-8 is a more reliable marker for disease activity than C-reactive protein and erythrocyte sedimentation rate. Dermatology. 2000; 201: 37–39 [DOI] [PubMed] [Google Scholar]
- 16. Freire Ade L, Bertolo MB, de Pinho AJ Jr, Samara AM, Fernandes SR. Increased serum levels of interleukin-8 in polyarteritis nodosa and Behçet's disease. Clin Rheumatol. 2004; 23: 203–205 [DOI] [PubMed] [Google Scholar]
- 17. Deshmane SL, Kremlev S, Amini S, Sawaya BE. Monocyte chemoattractant protein-1 (MCP-1): an overview. J Interferon Cytokine Res. 2009; 29: 313–326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Daly C, Rollins BJ. Monocyte chemoattractant protein-1 (CCL2) in inflammatory disease and adaptive immunity: therapeutic opportunities and controversies. Microcirculation. 2003; 10: 247–257 [DOI] [PubMed] [Google Scholar]
- 19. Baggiolini M, Moser B, Clark-Lewis I. Interleukin-8 and related chemotactic cytokines. The Giles Filley Lecture. Chest. 1994; 105: 95S–98S [DOI] [PubMed] [Google Scholar]
- 20. Yoshida Y, Yamagishi S, Ueda S, Yoshimura K, Okuda S, Yamakawa R. Aqueous humor levels of asymmetric dimethylarginine (ADMA) are positively associated with monocyte chemoattractant protein-1 (MCP-1) in patients with uveitis. Br J Ophthalmol. 2008; 92: 93–94 [DOI] [PubMed] [Google Scholar]
- 21. Curnow SJ, Falciani F, Durrani OM, et al. Multiplex bead immunoassay analysis of aqueous humor reveals distinct cytokine profiles in uveitis. Invest Ophthalmol Vis Sci. 2005; 46: 4251–4259 [DOI] [PubMed] [Google Scholar]
- 22. Lahmar I, Abou-Bacar A, Abdelrahman T, et al. Cytokine profiles in toxoplasmic and viral uveitis. J Infect Dis. 2009; 199: 1239–1249 [DOI] [PubMed] [Google Scholar]
- 23. Banerjee S, Savant V, Scott RA, Curnow SJ, Wallace GR, Murray PI. Multiplex bead analysis of vitreous humor of patients with vitreoretinal disorders. Invest Ophthalmol Vis Sci. 2007; 48: 2203–2207 [DOI] [PubMed] [Google Scholar]
- 24. Li J, Ang M, Cheung CM, et al. Aqueous cytokine changes associated with Posner-Schlossman syndrome with and without human cytomegalovirus. PLoS One. 2012; 7: e44453 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Ang M, Cheung G, Vania M, et al. Aqueous cytokine and chemokine analysis in uveitis associated with tuberculosis. Mol Vis. 2012; 18: 565–573 [PMC free article] [PubMed] [Google Scholar]
- 26. El-Asrar AM, Al-Obeidan SS, Kangave D, et al. CXC chemokine expression profiles in aqueous humor of patients with different clinical entities of endogenous uveitis. Immunobiology. 2011; 216: 1004–1009 [DOI] [PubMed] [Google Scholar]
- 27. Nagata K, Maruyama K, Uno K, et al. Simultaneous analysis of multiple cytokines in the vitreous of patients with sarcoid uveitis. Invest Ophthalmol Vis Sci. 2012; 53: 3827–3833 [DOI] [PubMed] [Google Scholar]
- 28. Maier R, Weger M, Haller-Schober EM, et al. Multiplex bead analysis of vitreous and serum concentrations of inflammatory and proangiogenic factors in diabetic patients. Mol Vis. 2008; 14: 637–643 [PMC free article] [PubMed] [Google Scholar]
- 29. Suzuki Y, Nakazawa M, Suzuki K, Yamazaki H, Miyagawa Y. Expression profiles of cytokines and chemokines in vitreous fluid in diabetic retinopathy and central retinal vein occlusion. Jpn J Ophthalmol. 2011; 55: 256–263 [DOI] [PubMed] [Google Scholar]
- 30. Murugeswari P, Shukla D, Rajendran A, Kim R, Namperumalsamy P, Muthukkaruppan V. Proinflammatory cytokines and angiogenic and anti-angiogenic factors in vitreous of patients with proliferative diabetic retinopathy and Eales' disease. Retina. 2008; 28: 817–824 [DOI] [PubMed] [Google Scholar]
- 31. Elner SG, Elner VM, Jaffe GJ, Stuart A, Kunkel SL, Strieter RM. Cytokines in proliferative diabetic retinopathy and proliferative vitreoretinopathy. Curr Eye Res. 1995; 14: 1045–1053 [DOI] [PubMed] [Google Scholar]
- 32. Yoshimura T, Sonoda KH, Sugahara M, et al. Comprehensive analysis of inflammatory immune mediators in vitreoretinal diseases. PLoS One. 2009; 4: e8158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Capeans C, De Rojas MV, Lojo S, Salorio MS. C-C chemokines in the vitreous of patients with proliferative vitreoretinopathy and proliferative diabetic retinopathy. Retina. 1998; 18: 546–550 [PubMed] [Google Scholar]
- 34. Mitamura Y, Takeuchi S, Yamamoto S, et al. Monocyte chemotactic protein-1 levels in the vitreous of patients with proliferative vitreoretinopathy. Jpn J Ophthalmol. 2002; 46: 218–221 [DOI] [PubMed] [Google Scholar]
- 35. El-Ghrably IA, Dua HS, Orr GM, Fischer D, Tighe PJ. Intravitreal invading cells contribute to vitreal cytokine milieu in proliferative vitreoretinopathy. Br J Ophthalmol. 2001; 85: 461–470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Funk M, Kriechbaum K, Prager F, et al. Intraocular concentrations of growth factors and cytokines in retinal vein occlusion and the effect of therapy with bevacizumab. Invest Ophthalmol Vis Sci. 2009; 50: 1025–1032 [DOI] [PubMed] [Google Scholar]
- 37. Melth AD, Agron E, Chan CC, et al. Serum inflammatory markers in diabetic retinopathy. Invest Ophthalmol Vis Sci. 2005; 46: 4295–4301 [DOI] [PubMed] [Google Scholar]
- 38. Jonas JB, Tao Y, Neumaier M, Findeisen P. Cytokine concentration in aqueous humour of eyes with exudative age-related macular degeneration. Acta Ophthalmol. 2012; 90: e381–e388 [DOI] [PubMed] [Google Scholar]
- 39. Hollborn M, Enzmann V, Barth W, Wiedemann P, Kohen L. Changes in the mRNA expression of cytokines and chemokines by stimulated RPE cells in vitro. Curr Eye Res. 2000; 20: 488–495 [PubMed] [Google Scholar]
- 40. Yoshida A, Elner SG, Bian ZM, Kunkel SL, Lukacs NW, Elner VM. Differential chemokine regulation by Th2 cytokines during human RPE-monocyte coculture. Invest Ophthalmol Vis Sci. 2001; 42: 1631–1638 [PubMed] [Google Scholar]
- 41. Higgins GT, Wang JH, Dockery P, Cleary PE, Redmond HP. Induction of angiogenic cytokine expression in cultured RPE by ingestion of oxidized photoreceptor outer segments. Invest Ophthalmol Vis Sci. 2003; 44: 1775–1782 [DOI] [PubMed] [Google Scholar]
- 42. Cavicchio VA, Pru JK, Davis BS, Davis JS, Rueda BR, Townson DH. Secretion of monocyte chemoattractant protein-1 by endothelial cells of the bovine corpus luteum: regulation by cytokines but not prostaglandin F2alpha. Endocrinology. 2002; 143: 3582–3589 [DOI] [PubMed] [Google Scholar]
- 43. Gawaz M, Neumann FJ, Dickfeld T, et al. Activated platelets induce monocyte chemotactic protein-1 secretion and surface expression of intercellular adhesion molecule-1 on endothelial cells. Circulation. 1998; 98: 1164–1171 [DOI] [PubMed] [Google Scholar]
- 44. Strunnikova NV, Barb J, Sergeev YV, et al. Loss-of-function mutations in Rab escort protein 1 (REP-1) affect intracellular transport in fibroblasts and monocytes of choroideremia patients. PLoS One. 2009; 4: e8402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Cao S, Walker GB, Wang X, Cui JZ, Matsubara JA. Altered cytokine profiles of human retinal pigment epithelium: oxidant injury and replicative senescence. Mol Vis. 2013; 19: 718–728 [PMC free article] [PubMed] [Google Scholar]
- 46. Lueck K, Hennig M, Lommatzsch A, Pauleikhoff D, Wasmuth S. Complement and UV-irradiated photoreceptor outer segments increase the cytokine secretion by retinal pigment epithelial cells. Invest Ophthalmol Vis Sci. 2012; 53: 1406–1413 [DOI] [PubMed] [Google Scholar]
- 47. Chen Y, Kijlstra A, Chen Y, Yang P. IL-17A stimulates the production of inflammatory mediators via Erk1/2, p38 MAPK, PI3K/Akt, and NF-κB pathways in ARPE-19 cells. Mol Vis. 2011; 17: 3072–3077 [PMC free article] [PubMed] [Google Scholar]
- 48. Kurji KH, Cui JZ, Lin T, et al. Microarray analysis identifies changes in inflammatory gene expression in response to amyloid-beta stimulation of cultured human retinal pigment epithelial cells. Invest Ophthalmol Vis Sci. 2010; 51: 1151–1163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Kioka N, Minami K, Tamura A, Yoshikawa N. Chemokine expression in human astrocytes in response to shiga toxin 2. Int J Inflam. 2012; 2012: 135803 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Raetz CR, Whitfield C. Lipopolysaccharide endotoxins. Annu Rev Biochem. 2002; 71: 635–700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Rosenbaum JT, McDevitt HO, Guss RB, Egbert PR. Endotoxin-induced uveitis in rats as a model for human disease. Nature. 1980; 286: 611–613 [DOI] [PubMed] [Google Scholar]
- 52. Kasner L, Chan CC, Whitcup SM, Gery I. The paradoxical effect of tumor necrosis factor alpha (TNF-alpha) in endotoxin-induced uveitis. Invest Ophthalmol Vis Sci. 1993; 34: 2911–2917 [PubMed] [Google Scholar]
- 53. Ohgami K, Ilieva IB, Shiratori K, et al. Effect of human cationic antimicrobial protein 18 peptide on endotoxin-induced uveitis in rats. Invest Ophthalmol Vis Sci. 2003; 44: 4412–4418 [DOI] [PubMed] [Google Scholar]
- 54. Tuaillon N, Shen DF, Berger RB, Lu B, Rollins BJ, Chan CC. MCP-1 expression in endotoxin-induced uveitis. Invest Ophthalmol Vis Sci. 2002; 43: 1493–1498 [PubMed] [Google Scholar]
- 55. de Vos AF, Klaren VN, Kijlstra A. Expression of multiple cytokines and IL-1RA in the uvea and retina during endotoxin-induced uveitis in the rat. Invest Ophthalmol Vis Sci. 1994; 35: 3873–3883 [PubMed] [Google Scholar]
- 56. Chen FT, Liu YC, Yang CM, Yang CH. Anti-inflammatory effect of the proteasome inhibitor bortezomib on endotoxin-induced uveitis in rats. Invest Ophthalmol Vis Sci. 2012; 53: 3682–3694 [DOI] [PubMed] [Google Scholar]
- 57. Mo JS, Matsukawa A, Ohkawara S, Yoshinaga M. Role and regulation of IL-8 and MCP-1 in LPS-induced uveitis in rabbits. Exp Eye Res. 1999; 68: 333–340 [DOI] [PubMed] [Google Scholar]
- 58. Leung KW, Barnstable CJ, Tombran-Tink J. Bacterial endotoxin activates retinal pigment epithelial cells and induces their degeneration through IL-6 and IL-8 autocrine signaling. Mol Immunol. 2009; 46: 1374–1386 [DOI] [PubMed] [Google Scholar]
- 59. Zhang JZ, Cavet ME, VanderMeid KR, Salvador-Silva M, López FJ, Ward KW. BOL-303242-X, a novel selective glucocorticoid receptor agonist, with full anti-inflammatory properties in human ocular cells. Mol Vis. 2009; 15: 2606–2616 [PMC free article] [PubMed] [Google Scholar]
- 60. Thorley AJ, Grandolfo D, Lim E, Goldstraw P, Young A, Tetley TD. Innate immune responses to bacterial ligands in the peripheral human lung–role of alveolar epithelial TLR expression and signalling. PLoS One. 2011; 6: e21827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Nagarajan S, Burris RL, Stewart BW, Wilkerson JE, Badger TM. Dietary soy protein isolate ameliorates atherosclerotic lesions in apolipoprotein E-deficient mice potentially by inhibiting monocyte chemoattractant protein-1 expression. J Nutr. 2008; 138: 332–337 [DOI] [PubMed] [Google Scholar]
- 62. Salerno DM, Tront JS, Hoffman B, Liebermann DA. Gadd45a and Gadd45b modulate innate immune functions of granulocytes and macrophages by differential regulation of p38 and JNK signaling. J Cell Physiol. 2012; 227: 3613–3620 [DOI] [PubMed] [Google Scholar]
- 63. Zhu C, Zhang A, Huang S, Ding G, Pan X, Chen R. Interleukin-13 inhibits cytokines synthesis by blocking nuclear factor-κB and c-Jun N-terminal kinase in human mesangial cells. J Biomed Res. 2010; 24: 308–316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Cavicchio VA, Pru JK, Davis BS, Davis JS, Rueda BR, Townson DH. Secretion of monocyte chemoattractant protein-1 by endothelial cells of the bovine corpus luteum: regulation by cytokines but not prostaglandin F2alpha. Endocrinology. 2002; 143: 3582–3589 [DOI] [PubMed] [Google Scholar]
- 65. Gawaz M, Neumann FJ, Dickfeld T, et al. Activated platelets induce monocyte chemotactic protein-1 secretion and surface expression of intercellular adhesion molecule-1 on endothelial cells. Circulation. 1998; 98: 1164–1171 [DOI] [PubMed] [Google Scholar]
- 66. Strunnikova NV, Barb J, Sergeev YV, et al. Loss-of-function mutations in Rab escort protein 1 (REP-1) affect intracellular transport in fibroblasts and monocytes of choroideremia patients. PLoS One. 2009; 4: e8402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Lu C, Song E, Hu DN, et al. Curcumin induces cell death in human uveal melanoma cells through mitochondrial pathway. Curr Eye Res. 2010; 35: 352–360 [DOI] [PubMed] [Google Scholar]
- 68. Fang IM, Yang CH, Lin CP, Yang CM, Chen MS. Expression of chemokine and receptors in Lewis rats with experimental autoimmune anterior uveitis. Exp Eye Res. 2004; 78: 1043–1055 [DOI] [PubMed] [Google Scholar]
- 69. Yang CH, Fang IM, Lin CP, Yang CM, Chen MS. Effects of the NF-kappaB inhibitor pyrrolidine dithiocarbamate on experimentally induced autoimmune anterior uveitis. Invest Ophthalmol Vis Sci. 2005; 46: 1339–1147 [DOI] [PubMed] [Google Scholar]
- 70. Ferrick MR, Thurau SR, Oppenheim MH, et al. Ocular inflammation stimulated by intravitreal interleukin-8 and interleukin-1. Invest Ophthalmol Vis Sci. 1991; 32: 1534–1539 [PubMed] [Google Scholar]
- 71. Anton L, Brown AG, Parry S, Elovitz MA. Lipopolysaccharide induces cytokine production and decreases extravillous trophoblast invasion through a mitogen-activated protein kinase-mediated pathway: possible mechanisms of first trimester placental dysfunction. Hum Reprod. 2012; 27: 61–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Yu N, Zhang S, Zuo F, Kang K, Guan M, Xiang L. Cultured human melanocytes express functional toll-like receptors 2-4, 7 and 9. J Dermatol Sci. 2009; 56: 113–120 [DOI] [PubMed] [Google Scholar]