Abstract
Background
Cannabinoid CB1 receptors play an essential role in drug addiction. Given the side effect profiles of orthosteric CB1 antagonism, new strategies have been attempted to modulate this target, such as CB1 receptor allosteric modulation. However, the effect of CB1 allosteric modulation in drug addiction is unknown. The present study examined the effects of the CB1 receptor allosteric modulator ORG27569 on the reinstatement of cocaine- and methamphetamine-seeking behavior in rats.
Methods
Rats were trained to self-administer 0.75 mg/kg cocaine or 0.05 mg/kg methamphetamine in 2-hr daily sessions for 14 days which was followed by 7 days of extinction sessions in which rats responded on the levers with no programmed consequences. On reinstatement test sessions, rats were administered ORG27569 (1.0, 3.2, 5.6 mg/kg, i.p.) or SR141716A (3.2 mg/kg, i.p.) 10 min prior to re-exposure to cocaine- or methamphetamine-paired cues or a priming injection of cocaine (10 mg/kg, i.p.) or methamphetamine (1 mg/kg, i.p.).
Results
Both cues and a priming injection of cocaine or methamphetamine significantly reinstated the extinguished active lever responding. Pretreatment with ORG27569 resulted in a dose-related attenuation of both cue- and drug-induced reinstatement of cocaine- and methamphetamine-seeking behavior. SR141716A also exhibited similar inhibitory action on reinstatement of drug-seeking behavior.
Conclusion
Negative allosteric modulation of CB1 receptors can produce similar functional antagonism as orthosteric CB1 receptor antagonists on reinstatement of drug-seeking behavior. Thus, ORG27569 or other negative allosteric modulators deserve further study as potentially effective pharmacotherapy for drug addiction.
Keywords: CB1 allosteric modulator, reinstatement, self-administration, cocaine, methamphetamine, rats
1. INTRODUCTION
Cocaine and methamphetamine are two common addictive psychostimulants with high prevalence and association with crime and violence (Anderson and Bokor, 2012). Although great progress has been made in understanding the neuropharmacology of these psychostimulants, to date there is no FDA-approved pharmacotherapy available for either cocaine or methamphetamine addiction. One of the major problems in the treatment of drug dependence is high rates of relapse even after prolonged drug abstinence (O’Brien, 1997). This relapse to compulsive drug use can be triggered by re-exposure to stimuli previously associated with drug-taking, or by re-exposure to drugs (deWit, 1996).
The endocannabinoid system has been implicated in a number of neuropsychiatric disorders, including depression and drug addiction (Caroti et al., 2013). In particular, a wealth of evidence demonstrates that the cannabinoid CB1 receptor is involved in brain reward function (Wiskerke et al., 2008). For example, the selective cannabinoid CB1 receptor antagonists SR141716A and AM251 reduced self-administration of palatable food and several drugs of abuse, as well as reinstatement of food- and drug-seeking behaviors (Carai et al., 2005; De Vries et al., 2001; Fattore et al., 2007), suggesting the potential utility of CB1 receptor antagonists in the treatment of drug addiction and obesity. Unfortunately, clinical research found that SR141716A can induce serious adverse psychiatric events, including depression, anxiety and suicidal ideation (Kirilly et al., 2012), which prompted the exploration of alternative strategies to modulate CB1 receptors.
Recently, a CB1 receptor allosteric modulating site has been discovered and several allosteric modulators have been characterized in vitro (Navarro et al., 2009; Price et al., 2005), including ORG 27569 (5-chloro-3-ethyl-1H-indole-2-carboxylic acid [2-(4-piperidin-1-yl-phenyl)ethyl]amide). ORG 27569 is a highly selective CB1 receptor negative allosteric modulator. Although the binding profile of ORG27569 on the CB1 receptor modulating site has been characterized, it did not demonstrate appreciable binding on any of the other receptors in a panel of 45 receptors and binding sites (Table 1S). ORG 27569 binds to a site that partially overlaps with but is significantly different from the binding site of the CB1 receptor orthosteric antagonist SR141716A (Shore et al., 2014). In in vitro studies, ORG27569 decreases the efficacy of CB1 orthosteric agonist CP55940-induced G protein signaling (Ahn et al., 2012). Biochemical characterization finds that ORG27569 induces conformational changes of the CB1 receptor, which results in cellular internalization and downstream activation of ERK signaling (Ahn et al., 2012, 2013; Iliff et al., 2011). However, it is unknown whether the allosteric modulators have behavioral significance in animal models of drug addiction. Our recent study showed that ORG 27569 decreased palatable and plain food intake and decreased the motivation of rats to self-administer palatable food reward (data not published).
In the present study, we employed a reinstatement model to investigate the effect of ORG 27569 on the reinstatement to cocaine- and methamphetamine-seeking behavior. In this procedure, following a period of extinction of drug-taking behavior that was previously maintained by cocaine or methamphetamine, the relapse behavior is induced by drug or cue previously paired with drug taking. For comparison purpose, effect of the cannabinoid CB1 receptor antagonist SR141716A was also examined.
2. METHODS
2.1. Subjects
Adult male Sprague-Dawley rats (Harlan, Indianapolis, IN) weighing 280-300 g were used in the study. Animals were housed individually on a 12/12 hr light/dark cycle (behavioral experiments were conducted during the light period) with free access to water and food except during experimental sessions. Animals were maintained and experiments conducted in accordance with the Institutional Animal Care and Use Committee, University at Buffalo, and with the 2011 Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources on Life Sciences, National Research Council, National Academy of Sciences, Washington DC).
2.2. Apparatus
Self-administration chambers (30×20×20 cm, Med Associates, St Albans, Vermont, USA) were placed within sound-attenuating cubicles equipped with a house light and an exhaust fan. Each chamber contained two retractable levers and two stimulus lights. In addition, each chamber was equipped with a balanced metal arm and spring leash attached to a swivel. Tygon tubing extended through the leash and was connected to a 10 ml syringe mounted on an infusion pump located outside the sound attenuating cubicle. Before each session, vascular access harnesses were connected to the tygon tubing. Cocaine (2.5 mg/ml), methamphetamine (0.25 mg/ml) or saline was infused at a rate of 17.77 μl per sec driven by syringe pumps.
2.3. Drugs
ORG 27569 and SR141716A were dissolved in a 1:1:18 mixture of ethanol, emulphor and saline and administered i.p. Cocaine hydrochloride (2.5 mg/ml) and methamphetamine hydrochloride (0.25 mg/ml) were dissolved in 0.9% saline.
2.4. Catheterization surgery
Rats were anesthetized with ketamine/xylazine (72.0 mg/kg and 6.0 mg/kg, i.p., respectively). One end of the catheter was inserted 3 cm into the right external jugular vein. The other end ran subcutaneously and exited from a small incision in the animal’s back. This end attached to an infusion harness that provided access to an external port for intravenous drug delivery. Catheters were flushed every day until the completion of the experiments with 0.2-ml solution of enrofloxacin (4.54 mg/ml) mixed in a heparinized saline solution (50 IU/ml in 0.9% sterile saline) to preserve catheter patency.
2.5. Cocaine and methamphetamine self-administration training and extinction
Cocaine and methamphetamine self-administration were carried out according to our previous study (Thorn et al., 2014). After a one-week acclimatization period, animals were food-restricted (14 g/day standard rodent chow) and were given 1-hr (or until rats earned 100 reinforcers) daily sessions of autoshaping for 45 mg food pellets under a fixed ratio (FR) 1 time-out 5-sec schedule. The right lever was designated active and each response to the active lever resulted in the delivery of one food pellet. Responses on the inactive lever were recorded, but had no programmed consequence. The requirement was gradually increased to a terminal value of FR 5. Rats were trained under this schedule until stable levels of responding were obtained (less than 15% variation in total responding over 3 days). Two days following the lever-press training and free access to food, rats then were surgically implanted with a chronic indwelling jugular catheter.
Following a one-week recovery period, rats were trained to press the active lever for infusion of cocaine (0.75 mg/kg/inf) or methamphetamine (0.05 mg/kg/inf) under an FR 5 schedule during daily 2-hr sessions for 14 days. Reinforcer deliveries were accompanied by the presentation of a stimulus light over the active lever followed by a 30-s time-out period during which lever presses had no programmed consequence. A maximum of 40 reinforcers were available per session. All rats acquired reliable drug self-administration behavior in this study. One rat received a second catheter in the opposite jugular vein in this study due to the loss of patency of the original catheter. This rat was allowed 2 days of recovery after the new catheter was implanted.
Following acquisition of cocaine or methamphetamine self-administration, extinction of drug-seeking behavior took place during 2-hr daily sessions in which lever pressing produced saline instead of cocaine or methamphetamine injection, and the cocaine/methamphetamine-paired stimulus light was omitted. All other conditions remained unchanged. After 7 days of extinction, all rats reached the extinction criteria (reinforcers declined and stabilized at 5 or fewer saline infusions in each of 3 consecutive sessions).
2.6. Reinstatement of drug-seeking behavior
Cue-induced reinstatement test was conducted on the day following the last extinction session. Rats were pretreated with saline, ORG 27569 (1.0, 3.2, 5.6 mg/kg) or SR141716A (3.2 mg/kg) 10 min before the start of the test session. During the session, completion of the ratio requirement resulted in illumination of the previously drug-paired lights. Extinction sessions were conducted again for two consecutive days following cue-induced reinstatement test, and then the rats were tested for drug priming-induced reinstatement (Cox et al., 2013). The added two extinction sessions were sufficient to reduce the reinstated responding to meet the extinction criteria. Rats were pretreated with saline, ORG 27569 (1.0, 3.2, 5.6 mg/kg) or SR141716A (3.2 mg/kg) 10 min prior to a priming injection of either cocaine (10 mg/kg, i.p.) or methamphetamine (1 mg/kg, i.p.) administered immediately before the start of the reinstatement session.
2.7. Statistical analyses
Data are expressed as mean ± S.E.M. Differences in active lever responding between the last extinction session and reinstatement session were determined with paired t tests (within subjects comparison). The effects of ORG 27569 or SR141716A on reinstatement were analyzed by a one-way analysis of variance (ANOVA) followed by post hoc Bonferroni’s test (between subjects comparison). P < 0.05 was considered statistically significant.
3. RESULTS
3.1. Effect of ORG 27569 on cocaine associated cue-induced reinstatement
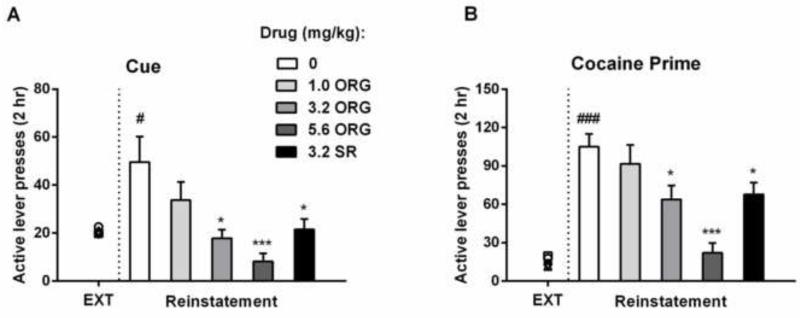
Rats earned an average of 26 (±0.8) infusions of cocaine (0.75 mg/kg/inf) during the 2-hr session under an FR 5 schedule on the final day of self-administration. Replacing cocaine infusions with saline infusions and omission of the associated stimulus lights significantly reduced responding on the active lever during extinction sessions. On the last extinction session, the animals earned an average of 3 (±0.3) saline infusions. Rats were then divided into five groups. There were no significant differences in the total number of injections on the last self-administration session (one-way ANOVA: F (4, 38) =1.161, p > 0.05) (Table S21), total active lever responses on the first extinction session (one-way ANOVA: F (4, 38) =0.106, p > 0.05) (Table S22) and total active lever responses on the last extinction session (range of group average responses: 19-22) [one-way ANOVA: F (4, 38) =0.141, p > 0.05, left side of dash line, Fig 1A]. When rats were re-exposed to cocaine-associated cues, responding on the active lever was reinstated [t=2.801, p < 0.05, right side of dash line in Fig 1A]. A one-way ANOVA revealed the significant differences in active-lever presses among groups [F (4, 38) =6.042, p < 0.001]. Post hoc tests showed that ORG 27569 at both the 3.2 and 5.6 mg/kg doses significantly attenuated reinstatement as compared to saline treatment (p < 0.05 and p < 0.001, respectively). SR141716A also attenuated cue-induced reinstatement of cocaine-seeking behavior (p < 0.05).
Figure 1.
Effects of ORG 27569 or SR141716A on cue- and cocaine prime-induced reinstatement of extinguished cocaine seeking behavior. (A) ORG 27569 and SR141716A significantly blocked cue-induced reinstatement of active lever responding as compared with saline treatment. (B) ORG 27569 and SR141716A significantly blocked cocaine prime-induced reinstatement of active lever responding as compared with saline treatment. #p<0.05 and ###p<0.001 as compared to corresponding saline group on the last extinction session; *p<0.05 and ***p<0.001 as compared to saline group. EXT, last extinction session; ORG, ORG 27569; SR, SR141716A.
3.2. Effect of ORG 27569 on cocaine priming-induced reinstatement
Two extinction sessions were conducted again following cue-induced reinstatement, and then the rats were tested for cocaine priming-induced reinstatement. A one-way ANOVA revealed that there were no differences in baseline active lever responding (the responses on the last extinction session before cocaine-induced reinstatement) among five groups (range of group average responses: 13-19) [one-way ANOVA: F (4, 38) =1.031, p > 0.05, left side of dash line, Fig 1B]. A single injection of 10 mg/kg cocaine robustly reinstated responding on the previously active lever [t=5.939, p < 0.001, right side of dash line in Fig 1B]. ORG 27569 attenuated cocaine priming-induced reinstatement in a dose-dependent manner (one-way ANOVA: F (4, 38) = 8.237, p < 0.001), with 3.2 and 5.6 mg/kg of ORG 27569 showing a significant decrease in active lever responding (p < 0.05 and p < 0.001, respectively). SR141716A also significantly reduced the reinstatement of cocaine-seeking behavior induced by cocaine priming (p < 0.05).
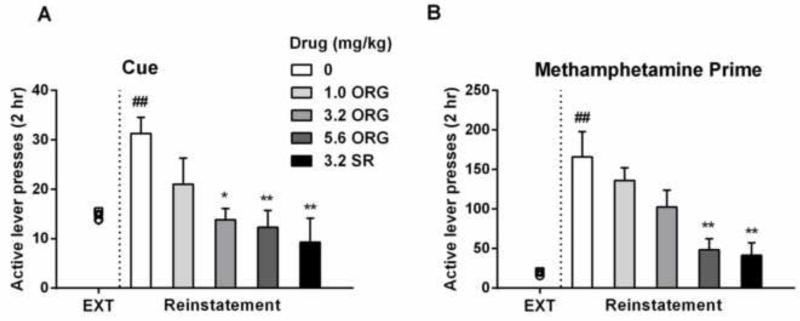
3.3. Effect of ORG 27569 on methamphetamine associated cue-induced reinstatement
On the last self-administration session, rats earned 14 (±1.9) infusions of methamphetamine (0.05 mg/kg/inf). After 7 days of extinction, the reinforcers dropped to 2 (±0.3) saline infusions, demonstrating a clear behavioral extinction. There were no significant differences in the total number of injections on the last self-administration session (one-way ANOVA: F (4, 29) =0.014, p > 0.05) (Table S3), total number of active lever responses on the first extinction session (one-way ANOVA: F (4, 29) =0.082, p > 0.05) (Table S3), and total number of active-lever responses on the last extinction session among all treatment groups (range of group average responses: 14-15) [one-way ANOVA: F (4, 29) =0.033, p > 0.05, left side of dash line, Fig 2A]. As expected, the presence of methamphetamine-associated cues significantly increased the active lever responding [t=4.384, p < 0.01, right side of dash line in Fig 2A]. A one-way ANOVA revealed that ORG 27569 dose-dependently attenuated cue-induced active lever responding [F (4, 29) =5.61, p < 0.01]. Post hoc analyses indicated that 3.2 and 5.6 mg/kg ORG 27569 significantly attenuated cue-induced reinstatement of methamphetamine seeking behavior (p < 0.05 and p < 0.01, respectively). Like ORG 27569, SR141716A also reduced cue-induced reinstatement behavior as compared to saline treatment (p < 0.01).
Figure 2.
Effect of ORG 27569 or SR141716A on cue- and methamphetamine prime-induced reinstatement of extinguished methamphetamine seeking behavior. (A) ORG 27569 and SR141716A significantly attenuated cue-induced reinstatement of active lever responding. (B) ORG 27569 and SR141716A significantly attenuated methamphetamine prime-induced reinstatement of active lever responding. ##p<0.01 as compared to corresponding saline group on the last extinction session; *p<0.05 and **p<0.01 as compared to saline group. See Figure 1 for other details.
3.4. Effect of ORG 27569 on methamphetamine priming-induced reinstatement
Following cue-induced reinstatement of methamphetamine-seeking behavior, two more extinction sessions further decreased the active lever responses. A priming injection of 1 mg/kg methamphetamine (i.p.) significantly reinstated the active lever responding [t=4.283, p < 0.01, left side of dash line, Fig 2B]. There were no differences in active-lever responses among all groups before the priming reinstatement test (range of group average responses: 16-20) [one-way ANOVA: F (4, 29) =0.268, p > 0.05]. A one-way ANOVA revealed that there were significant differences among all treatment groups [F (4, 29) = 6.847, p < 0.001]. Post hoc analyses indicated that 5.6 mg/kg ORG 27569 and 3.2 mg/kg SR141716A significantly reduced methamphetamine-primed reinstatement of methamphetamine-seeking behavior (p < 0.01 for both).
4. DISCUSSION
The primary finding of the current study was that a CB1 receptor negative modulator ORG27569 dose-dependently attenuated both cue- and drug-induced reinstatement of cocaine- and methamphetamine-seeking behaviors. This represents the first in vivo assessment of a CB1 receptor negative modulator in an animal model of drug relapse and suggests that functional modulation of the CB1 receptor allosteric binding site may be a useful strategy against drug addiction.
A large body of literature demonstrates that the endocannabinoid system and cannabinoid receptors play a key role in reward mechanisms in brain (Cheer et al., 2000; Gardner, 2005). Drugs acting at cannabinoid receptors can modulate addiction-related behavioral effects in rodents and nonhuman primates, especially reinstatement to drug-seeking behavior. For example, CB1 receptor agonist HU210 reinstates cocaine seeking after prolonged withdrawal period, which is reversed by the selective CB1 receptor antagonist SR141716A (De Vries et al., 2001). Blockade of CB1 receptors by SR141716A or AM251, or CB1 receptor knockout mice reduces the reinforcing effects and the motivation for drug intake (Orio et al., 2009; Soria et al., 2005; Xi et al., 2008) of a variety of addictive drugs, including heroin (Navarro et al., 2001; Solinas et al., 2003), morphine (Navarro et al., 2001), nicotine (Cohen et al., 2002) and methamphetamine (Schindler et al., 2010; Vinklerova et al., 2002) in laboratory animals. In addition, SR141716A or AM251 prevents cue-induced reinstatement of cocaine- (Adamczyk et al., 2012; De Vries et al., 2001; Filip et al., 2006; Ward et al., 2009), heroin- (Alvarez-Jaimes et al., 2008), methamphetamine- (Anggadiredja et al., 2004) and nicotine-seeking (Cohen et al., 2005, 2002) behavior in rats and mice, and attenuates reinstatement of cocaine- (Adamczyk et al., 2012; De Vries et al., 2001), heroin- (Fattore et al., 2005) and methamphetamine-seeking (Schindler et al., 2010) behavior induced by priming drug injection in rats and nonhuman primates.
Although accumulating evidence supports the potential anti-relapse effect of SR141716A, severe adverse events led to the withdrawal of rimonabant from the market worldwide (Sanofi Aventis, 2008). The recent discovery of CB1 negative allosteric modulators marks a significant progress in the neuropharmacology of the endocannabinoid system. However, the behavioral pharmacology of CB1 receptor allosteric modulators is largely unclear. So far, there is only one in vivo study of such compounds (Gamage et al., 2014). The present study examined whether the allosteric modulator ORG 27569 inhibits reinstatement of responding by cocaine and methamphetamine in rats with a history of drug self-administration. Both cocaine and methamphetamine maintained reliable and high level of self-administration behavior. After 7 days of extinction, the reinforcers earned declined to lower than 5 infusions. When rats were re-exposed to drug paired cues or priming injection, they exhibited significant reinstatement responding. Different doses of ORG 27569 were administered before the reinstatement test. Besides, SR141716A was employed as a positive control based on previous reports (De Vries et al., 2001; Ward et al., 2009). Our findings demonstrated that ORG 27569 significantly and dose-dependently attenuated cocaine- and methamphetamine-seeking behavior elicited either by drug-paired cues or by priming injection of the drugs. SR141716A also reduced reinstatement to cocaine- and methamphetamine-seeking behavior in both conditions, which is consistent with the literature (Anggadiredja et al., 2004; De Vries et al., 2001). It is important to note that the reduction of ORG 27569 on active lever responding was not due to general behavioral suppression. In a different study, we found that at a higher dose than the doses used in this study (10 mg/kg) ORG 27569 had no significant effect on food maintained operant responding in rats (data not shown). Likewise, a recent report showed that 30 mg/kg ORG 27569 has no effect on the performance of operant responding in a drug discrimination paradigm in mice (Gamage et al., 2014).
The exact mechanism by which ORG 27569 inhibits reinstatement of both cocaine- and methamphetamine-seeking behavior remains unknown. There exists a robust interaction between the cannabinoid and dopaminergic systems. Cocaine and methamphetamine share the common property of activating the mesolimbic dopamine reward system (Mailleux and Vanderhaeghen, 1992), and increased dopamine in this system is involved in reinstatement of cocaine and methamphetamine seeking behavior (Parsegian and See, 2014; Stewart, 2004). CB1 receptors are densely distributed in mesocorticolimbic brain area (Mailleux and Vanderhaeghen, 1992), which is a major neural substrate for the reinforcing effects of most drugs of abuse. Previous studies have shown that there is a robust crosstalk between the CB1 receptor and D2 receptor. For example, chronic administration of SR141716A up-regulates striatal D2 receptor (Crunelle et al., 2013). Blockade of CB1 receptor by SR141716A increases Fos expression in rat mesocorticolimbic areas, and dopamine D2 agonist quinpirole inhibits SR141716A-induced Fos expression (Alonso et al., 1999). Activation of either CB1 receptor or D2 receptor increases extracellular signal-regulated kinases 1 and 2 (ERK1/2) phosphorylation, while co-stimulation of these two kinds of receptors elicit an additive effect on the phospho-ERK1/2 signal (Chiang et al., 2013). In addition, in vivo studies have demonstrated a strong modulatory interaction between CB1 and dopaminergic systems. CB1 receptor blockade reduces amphetamine-induced dopamine release in the striatum (Polissidis et al., 2014) and CB1 knockout mice demonstrates reduced cocaine-stimulated dopamine release in the nucleus accumbens (NAc) (Li et al., 2009). It is likely that ORG27569 increased dopamine D2 receptor function through negatively modulating CB1 receptors, which then inhibited the reinstatement effects of cocaine and methamphetamine (Xue et al., 2011).
Alternatively, evidence in animals has consistently shown that cocaine and amphetamine inhibit GABA transmission in the striatum (Centonze et al., 2002), and enhancement of GABAergic activity reduces cocaine and methamphetamine addiction (Hip et al., 2007; Voigt et al., 2011). Since a majority of the striatal CB1 receptors are located presynaptically on inhibitory GABAergic terminals in a position to modulate neurotransmitter release (Julian et al., 2003), a functional interaction between CB1 and GABAergic transmission in striatum cannot be ruled out. It has been hypothesized that SR141716A can increase GABA release via disinhibition of GABAergic interneurons in the ventral tegmental area (Cohen et al., 2002), which results in the inhibition of dopamine neurons and suppression of dopamine-mediated reinforcing effects. There is also evidence showing that SR141716A and AM251 are positive allosteric modulators of GABAA receptor (Baur et al., 2012). ORG27569 shares similar chemical structures with these compounds and possibly also shares similar pharmacological effects via GABAergic modulation.
In addition, a role for cannabinoid-glutamate interactions should not be excluded. A previous study found that blockade of CB1 receptors increases glutamate levels in the NAc, which inhibits cocaine-induced glutamate levels in the NAc and attenuates cocaine-induced reinstatement of drug-seeking behavior (Xi et al., 2006). Further, mGluR2/3 antagonist LY341495 significantly reversed AM251-induced attenuation of cocaine prime-induced reinstatement of cocaine-seeking behavior (Xi et al., 2006). ORG 27569 may also exert its effects on cocaine- and methamphetamine-induced reinstatement via this mechanism.
ORG 27569 is a pharmacologically highly selective CB1 receptor negative allosteric modulator (Table 1S). This highly selective binding profile of ORG 27569 suggests that the observed effects of ORG27569 are most likely mediated through negative modulation of CB1 receptors. The observed effects of ORG 27569 on the relapse-like behaviors to cocaine and methamphetamine addiction prompt future studies to examine the potential role of CB1 receptor allosteric modulators in the abuse-related effects of other drugs of abuse, and to understand the safety profiles of such new allosteric modulators, particularly their effects on emotion and mood modulation (Kirilly et al., 2012).
In conclusion, these findings extend previous studies showing that negatively modulating CB1 receptors may represent a promising pharmacotherapeutic strategy to prevent relapse to psychostimulant seeking, and provide a novel pathway for the use of cannabinoid CB1 allosteric modulators in the prevention of relapse to drug use.
Supplementary Material
Highlights.
Acknowledgements
Receptor binding profiles were generously provided by the National Institute of Mental Health’s Psychoactive Drug Screening Program, Contract # HHSN-271-2008-00025-C (NIMH PDSP). The NIMH PDSP is Directed by Bryan L. Roth MD, PhD at the University of North Carolina at Chapel Hill and Project Officer Jamie Driscol at NIMH, Bethesda MD, USA.
Role of Funding Source
Funding for this study was provided by NIH grants DA034806 and DA033426; the NIH had no further role in designing the study, collecting the data, interpreting the results, writing the manuscript, or submitting the paper for publication.
Footnotes
Supplementary material can be found by accessing the online version of this paper at http://dx.doi.org and by entering doi:…
Supplementary material can be found by accessing the online version of this paper at http://dx.doi.org and by entering doi:…
Supplementary material can be found by accessing the online version of this paper at http://dx.doi.org and by entering doi:…
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Authorship Contributions
Li Jing and Jun-Xu Li conceived of the study and wrote the manuscript. Li Jing executed study and collected the data. Yanyan Qiu contributed to the experimental design and data collection. Yanan Zhang synthesized ORG27569 and edited the final draft of the manuscript. All authors read and approved the final draft of the manuscript.
Conflict of Interest
The authors declared no financial conflict of interest about this research.
REFERENCES
- Adamczyk P, Miszkiel J, McCreary AC, Filip M, Papp M, Przegalinski E. The effects of cannabinoid CB1, CB2 and vanilloid TRPV1 receptor antagonists on cocaine addictive behavior in rats. Brain Res. 2012;1444:45–54. doi: 10.1016/j.brainres.2012.01.030. [DOI] [PubMed] [Google Scholar]
- Ahn KH, Mahmoud MM, Kendall DA. Allosteric modulator ORG27569 induces CB1 cannabinoid receptor high affinity agonist binding state, receptor internalization, and Gi protein-independent ERK1/2 kinase activation. J. Biol. Chem. 2012;287:12070–12082. doi: 10.1074/jbc.M111.316463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahn KH, Mahmoud MM, Shim JY, Kendall DA. Distinct roles of beta-arrestin 1 and beta-arrestin 2 in ORG27569-induced biased signaling and internalization of the cannabinoid receptor 1 (CB1) J. Biol. Chem. 2013;288:9790–9800. doi: 10.1074/jbc.M112.438804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alonso R, Voutsinos B, Fournier M, Labie C, Steinberg R, Souilhac J, Le Fur G, Soubrie P. Blockade of cannabinoid receptors by SR141716 selectively increases Fos expression in rat mesocorticolimbic areas via reduced dopamine D2 function. Neuroscience. 1999;91:607–620. doi: 10.1016/s0306-4522(98)00675-7. [DOI] [PubMed] [Google Scholar]
- Alvarez-Jaimes L, Polis I, Parsons LH. Attenuation of cue-induced heroin-seeking behavior by cannabinoid CB1 antagonist infusions into the nucleus accumbens core and prefrontal cortex, but not basolateral amygdala. Neuropsychopharmacology. 2008;33:2483–2493. doi: 10.1038/sj.npp.1301630. [DOI] [PubMed] [Google Scholar]
- Anderson PD, Bokor G. Forensic aspects of drug-induced violence. J. Pharm. Pract. 2012;25:41–49. doi: 10.1177/0897190011431150. [DOI] [PubMed] [Google Scholar]
- Anggadiredja K, Nakamichi M, Hiranita T, Tanaka H, Shoyama Y, Watanabe S, Yamamoto T. Endocannabinoid system modulates relapse to methamphetamine seeking: possible mediation by the arachidonic acid cascade. Neuropsychopharmacology. 2004;29:1470–1478. doi: 10.1038/sj.npp.1300454. [DOI] [PubMed] [Google Scholar]
- Baur R, Gertsch J, Sigel E. The cannabinoid CB1 receptor antagonists rimonabant (SR141716) and AM251 directly potentiate GABA(A) receptors. Br. J. Pharmacol. 2012;165:2479–2484. doi: 10.1111/j.1476-5381.2011.01405.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carai MA, Colombo G, Gessa GL. Rimonabant: the first therapeutically relevant cannabinoid antagonist. Life Sci. 2005;77:2339–2350. doi: 10.1016/j.lfs.2005.04.017. [DOI] [PubMed] [Google Scholar]
- Caroti E, Cuoco V, Marconi M, Ratti F, Bersani G. The endocannabinoid system and its possible role in neurobiology of psychiatric disorders. Rivista di psichiatria. 2013;48:375–385. doi: 10.1708/1356.15064. [DOI] [PubMed] [Google Scholar]
- Centonze D, Picconi B, Baunez C, Borrelli E, Pisani A, Bernardi G, Calabresi P. Cocaine and amphetamine depress striatal GABAergic synaptic transmission through D2 dopamine receptors. Neuropsychopharmacology. 2002;26:164–175. doi: 10.1016/S0893-133X(01)00299-8. [DOI] [PubMed] [Google Scholar]
- Cheer JF, Kendall DA, Marsden CA. Cannabinoid receptors and reward in the rat: a conditioned place preference study. Psychopharmacology (Berl.) 2000;151:25–30. doi: 10.1007/s002130000481. [DOI] [PubMed] [Google Scholar]
- Chiang YC, Lo YN, Chen JC. Crosstalk between dopamine D(2) receptors and cannabinoid CB(1) receptors regulates CNR1 promoter activity via ERK1/2 signaling. J. Neurochem. 2013;127:163–176. doi: 10.1111/jnc.12399. [DOI] [PubMed] [Google Scholar]
- Cohen C, Perrault G, Griebel G, Soubrie P. Nicotine-associated cues maintain nicotine-seeking behavior in rats several weeks after nicotine withdrawal: reversal by the cannabinoid (CB1) receptor antagonist, rimonabant (SR141716) Neuropsychopharmacology. 2005;30:145–155. doi: 10.1038/sj.npp.1300541. [DOI] [PubMed] [Google Scholar]
- Cohen C, Perrault G, Voltz C, Steinberg R, Soubrie P. SR141716, a central cannabinoid (CB(1)) receptor antagonist, blocks the motivational and dopamine-releasing effects of nicotine in rats. Behav. Pharmacol. 2002;13:451–463. doi: 10.1097/00008877-200209000-00018. [DOI] [PubMed] [Google Scholar]
- Cox BM, Young AB, See RE, Reichel CM. Sex differences in methamphetamine seeking in rats: impact of oxytocin. Psychoneuroendocrinology. 2013;38:2343–2353. doi: 10.1016/j.psyneuen.2013.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crunelle CL, van de Giessen E, Schulz S, Vanderschuren LJ, de Bruin K, van den Brink W, Booij J. Cannabinoid-1 receptor antagonist rimonabant (SR141716) increases striatal dopamine D2 receptor availability. Addict. Biol. 2013;18:908–911. doi: 10.1111/j.1369-1600.2011.00369.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Vries TJ, Shaham Y, Homberg JR, Crombag H, Schuurman K, Dieben J, Vanderschuren LJ, Schoffelmeer AN. A cannabinoid mechanism in relapse to cocaine seeking. Nat. Med. 2001;7:1151–1154. doi: 10.1038/nm1001-1151. [DOI] [PubMed] [Google Scholar]
- deWit H. Priming effects with drugs and other reinforcers. Exp. Clin. Psychopharmacol. 1996;4:5–10. [Google Scholar]
- Fattore L, Spano MS, Deiana S, Melis V, Cossu G, Fadda P, Fratta W. An endocannabinoid mechanism in relapse to drug seeking: a review of animal studies and clinical perspectives. Brain Res. Rev. 2007;53:1–16. doi: 10.1016/j.brainresrev.2006.05.003. [DOI] [PubMed] [Google Scholar]
- Fattore L, Spano S, Cossu G, Deiana S, Fadda P, Fratta W. Cannabinoid CB(1) antagonist SR 141716A attenuates reinstatement of heroin self-administration in heroin-abstinent rats. Neuropharmacology. 2005;48:1097–1104. doi: 10.1016/j.neuropharm.2005.01.022. [DOI] [PubMed] [Google Scholar]
- Filip M, Golda A, Zaniewska M, McCreary AC, Nowak E, Kolasiewicz W, Przegalinski E. Involvement of cannabinoid CB1 receptors in drug addiction: effects of rimonabant on behavioral responses induced by cocaine. Pharmacol. Rep. 2006;58:806–819. [PubMed] [Google Scholar]
- Gamage TF, Ignatowska-Jankowska BM, Wiley JL, Abdelrahman M, Trembleau L, Greig IR, Thakur GA, Tichkule R, Poklis J, Ross RA, Pertwee RG, Lichtman AH. In-vivo pharmacological evaluation of the CB1-receptor allosteric modulator Org-27569. Behav. Pharmacol. 2014;25:182–185. doi: 10.1097/FBP.0000000000000027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gardner EL. Endocannabinoid signaling system and brain reward: emphasis on dopamine. Pharmacol. Biochem. Behav. 2005;81:263–284. doi: 10.1016/j.pbb.2005.01.032. [DOI] [PubMed] [Google Scholar]
- Hip M, Frankowska M, Przegalinski E. Effects of GABA(B) receptor antagonist, agonists and allosteric positive modulator on the cocaine-induced self-administration and drug discrimination. Eur. J. Pharmacol. 2007;574:148–157. doi: 10.1016/j.ejphar.2007.07.048. [DOI] [PubMed] [Google Scholar]
- Iliff HA, Lynch DL, Kotsikorou E, Reggio PH. Parameterization of Org27569: An allosteric modulator of the cannabinoid CB(1) G protein-coupled receptor. J. Computat. chem. 2011 doi: 10.1002/jcc.21794. Epub ahead of print. doi: 10.1002/jcc.21794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Julian MD, Martin AB, Cuellar B, Rodriguez De Fonseca F, Navarro M, Moratalla R, Garcia-Segura LM. Neuroanatomical relationship between type 1 cannabinoid receptors and dopaminergic systems in the rat basal ganglia. Neuroscience. 2003;119:309–318. doi: 10.1016/s0306-4522(03)00070-8. [DOI] [PubMed] [Google Scholar]
- Kirilly E, Gonda X, Bagdy G. CB1 receptor antagonists: new discoveries leading to new perspectives. Acta Physiologica Scand. 2012;205:41–60. doi: 10.1111/j.1748-1716.2012.02402.x. [DOI] [PubMed] [Google Scholar]
- Mailleux P, Vanderhaeghen JJ. Distribution of neuronal cannabinoid receptor in the adult rat brain: a comparative receptor binding radioautography and in situ hybridization histochemistry. Neuroscience. 1992;48:655–668. doi: 10.1016/0306-4522(92)90409-u. [DOI] [PubMed] [Google Scholar]
- Navarro HA, Howard JL, Pollard GT, Carroll FI. Positive allosteric modulation of the human cannabinoid (CB) receptor by RTI-371, a selective inhibitor of the dopamine transporter. Br. J. Pharmacol. 2009;156:1178–1184. doi: 10.1111/j.1476-5381.2009.00124.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Navarro M, Carrera MRA, Fratta W, Valverde O, Cossu G, Fattore L, Chowen JA, Gomez R, del Arco I, Villanua MA, Maldonado R, Koob GF, de Fonseca FR. Functional interaction between opioid and cannabinoid receptors in drug self-administration. J. Neurosci. 2001;21:5344–5350. doi: 10.1523/JNEUROSCI.21-14-05344.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Brien CP. A range of research-based pharmacotherapies for addiction. Science. 1997;278:66–70. doi: 10.1126/science.278.5335.66. [DOI] [PubMed] [Google Scholar]
- Orio L, Edwards S, George O, Parsons LH, Koob GF. A role for the endocannabinoid system in the increased motivation for cocaine in extended-access conditions. J. Neurosci. 2009;29:4846–4857. doi: 10.1523/JNEUROSCI.0563-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsegian A, See RE. Dysregulation of dopamine and glutamate release in the prefrontal cortex and nucleus accumbens following methamphetamine self-administration and during reinstatement in rats. Neuropsychopharmacology. 2014;39:811–822. doi: 10.1038/npp.2013.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Price MR, Baillie GL, Thomas A, Stevenson LA, Easson M, Goodwin R, McLean A, McIntosh L, Goodwin G, Walker G, Westwood P, Marrs J, Thomson F, Cowley P, Christopoulos A, Pertwee RG, Ross RA. Allosteric modulation of the cannabinoid CB1 receptor. Mol. Pharmacol. 2005;68:1484–1495. doi: 10.1124/mol.105.016162. [DOI] [PubMed] [Google Scholar]
- Sanofi Aventis . Sanofi-Aventis to discountinue all clinical trials with Rimonabant. Paris: 2008. [Google Scholar]
- Schindler CW, Panlilio LV, Gilman JP, Justinova Z, Vemuri VK, Makriyannis A, Goldberg SR. Effects of cannabinoid receptor antagonists on maintenance and reinstatement of methamphetamine self-administration in rhesus monkeys. Eur. J. Pharmacol. 2010;633:44–49. doi: 10.1016/j.ejphar.2010.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shore DM, Baillie GL, Hurst DH, Navas F, 3rd, Seltzman HH, Marcu JP, Abood ME, Ross RA, Reggio PH. Allosteric modulation of a cannabinoid G protein-coupled receptor: binding site elucidation and relationship to G protein signaling. J. Biol. Chem. 2014;289:5828–5845. doi: 10.1074/jbc.M113.478495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solinas M, Panlilio LV, Antoniou K, Pappas LA, Sr G. The cannabinoid CB1 antagonist N-piperidinyl-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methylpyrazole-3-carboxamide (SR-141716A) differentially alters the reinforcing effects of heroin under continuous reinforcement, fixed ratio, and progressive ratio schedules of drug self-administration in rats. J. Pharmacol. Exp. Ther. 2003;306:93–102. doi: 10.1124/jpet.102.047928. [DOI] [PubMed] [Google Scholar]
- Soria G, Mendizabal V, Tourino C, Robledo P, Ledent C, Parmentier M, Maldonado R, Valverde O. Lack of CB1 cannabinoid receptor impairs cocaine self-administration. Neuropsychopharmacology. 2005;30:1670–1680. doi: 10.1038/sj.npp.1300707. [DOI] [PubMed] [Google Scholar]
- Stewart J. Pathways to relapse: Factors controlling the reinitiation of drug seeking after abstinence. Nebr. Sym. Motiv. 2004;50:197–234. [PubMed] [Google Scholar]
- Thorn DA, Jing L, Qiu Y, Gancarz-Kausch AM, Galuska CM, Dietz DM, Zhang Y, Li JX. Effects of the Trace Amine-Associated Receptor 1 Agonist RO5263397 on Abuse-Related Effects of Cocaine in Rats. Neuropsychopharmacology. 2014 doi: 10.1038/npp.2014.91. epub ahead of print doi: 10.1038/npp.2014.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vinklerova J, Novakova J, Sulcova A. Inhibition of methamphetamine self-administration in rats by cannabinoid receptor antagonist AM 251. J. Psychopharmacol. 2002;16:139–143. doi: 10.1177/026988110201600204. [DOI] [PubMed] [Google Scholar]
- Voigt RM, Herrold AA, Riddle JL, Napier TC. Administration of GABA(B) receptor positive allosteric modulators inhibit the expression of previously established methamphetamine-induced conditioned place preference. Behav. Brain Res. 2011;216:419–423. doi: 10.1016/j.bbr.2010.08.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward SJ, Rosenberg M, Dykstra LA, Walker EA. The CB1 antagonist rimonabant (SR141716) blocks cue-induced reinstatement of cocaine seeking and other context and extinction phenomena predictive of relapse. Drug Alcohol Depend. 2009;105:248–255. doi: 10.1016/j.drugalcdep.2009.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiskerke J, Pattij T, Schoffelmeer AN, De Vries TJ. The role of CB1 receptors in psychostimulant addiction. Addict. Biol. 2008;13:225–238. doi: 10.1111/j.1369-1600.2008.00109.x. [DOI] [PubMed] [Google Scholar]
- Xi ZX, Gilbert JG, Peng XQ, Pak AC, Li X, Gardner EL. Cannabinoid CB1 receptor antagonist AM251 inhibits cocaine-primed relapse in rats: role of glutamate in the nucleus accumbens. J. Neurosci. 2006;26:8531–8536. doi: 10.1523/JNEUROSCI.0726-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xi ZX, Spiller K, Pak AC, Gilbert J, Dillon C, Li X, Peng XQ, Gardner EL. Cannabinoid CB1 receptor antagonists attenuate cocaine’s rewarding effects: experiments with self-administration and brain-stimulation reward in rats. Neuropsychopharmacology. 2008;33:1735–1745. doi: 10.1038/sj.npp.1301552. [DOI] [PubMed] [Google Scholar]
- Xue Y, Steketee JD, Rebec GV, Sun W. Activation of D2-like receptors in rat ventral tegmental area inhibits cocaine-reinstated drug-seeking behavior. Eur. J. Neurosci. 2011;33:1291–1298. doi: 10.1111/j.1460-9568.2010.07591.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.