Abstract
Toxoplasmosis is caused by infection with the parasite Toxoplasma gondii. It is one of the most common parasitic infections in humans and is most typically asymptomatic. However, primary infection in a pregnant woman can cause severe and disabling disease in the developing fetus. Recent developments have included increased understanding of the role of parasite genotype in determining infectivity and disease severity. Risk factors for acquisition of infection have been better defined, and the important role of foodborne transmission has been further delineated. In addition, strategies have emerged to decrease mother-to-child transmission through prompt identification of acutely infected pregnant women followed by appropriate treatment. Refined diagnostic tools, particularly the addition of immunoglobulin G avidity testing, allow for more accurate timing of maternal infection and hence better decision making during pregnancy. Congenitally infected children can be treated, beginning in utero and continuing through the first year of life, to ameliorate the severity of disease. However, despite these many advances in our understanding of congenital toxoplasmosis prevention and treatment, significant areas of study remain: we need better drugs, well defined strategies for screening of pregnant women, improved food safety, and improved diagnostic tests.
Keywords: infant, toxoplasmosis, transmission
Toxoplasma gondii is a protozoan parasite that infects most species of warm-blooded animals, including humans. The taxonomy has recently changed: Phylum Apicomplexa, Class Coccidea, Subclass Coccidiasina, Order Eimeriida, Suborder Eimeriorina, Family Sarcocystidae, (Sarcocystis), Subfamily Toxoplasmatinae, Genus Toxoplasma [1].
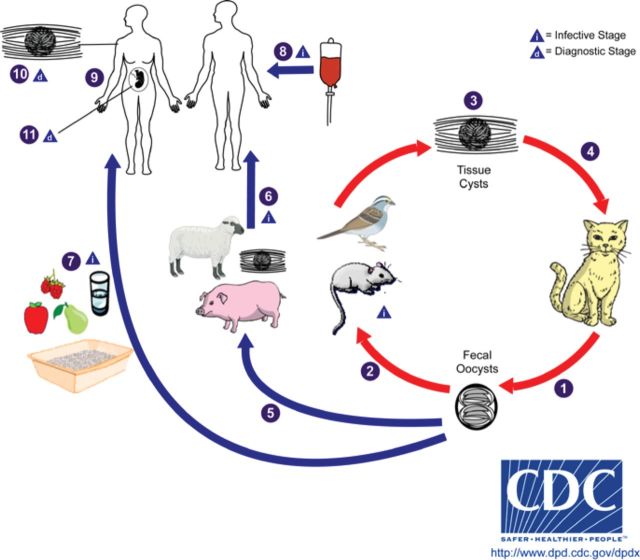
Members of the cat family Felidae are the only known definitive hosts for the sexual stages of T gondii and thus are the main reservoirs of infection. The 3 stages of this obligate intracellular parasite are as follows: (1) tachyzoites (trophozoites), which rapidly proliferate and destroy infected cells during acute infection; (2) bradyzoites, which slowly multiply in tissue cysts; and (3) sporozoites in oocysts. Tachyzoites and bradyzoites occur in body tissues; oocysts are excreted in cat feces (Figure 1). Cats become infected with T gondii by carnivorism or by ingestion of oocysts. Cats that are allowed to roam outside are much more likely to become infected than domestic cats that are confined indoors. After tissue cysts or oocysts are ingested by the cat, sporozoites are released and invade epithelial cells of the small intestine where they undergo an asexual cycle followed by a sexual cycle and then form oocysts, which are then excreted. The unsporulated (ie, noninfective) oocyst takes 1 to 5 days after excretion to become sporulated (infective). Although cats shed oocysts for only 1 to 2 weeks, large numbers may be shed, often exceeding 100 000 per gram of feces. Oocysts can survive in the environment for several months to more than 1 year and are remarkably resistant to disinfectants, freezing, and drying but are killed by heating to 70°C for 10 minutes [2].
Figure 1.
Lifecyle of Toxoplasma gondii.
- (1) Unsporulated oocysts are shed in the cat's feces, although oocysts are usually only shed for 1–2 weeks, large numbers may be shed.
- (2) Intermediate hosts in nature (including birds and rodents) become infected after ingesting soil, water, or plant material contaminated with oocysts.
- (3) Oocysts transform into tachyzoites that localize in neural and muscle tissue and develop into tissue cyst bradyzoites.
- (4) Cats become infected after consuming intermediate hosts harboring tissue cysts or by ingestion of sporulated oocysts.
- (5) Animals bred for human consumption and wild game may also become infected with tissue cysts after ingestion of sporulated oocysts in the environment.
- Humans can become infected by any of several routes:
- • eating undercooked meat of animals harboring tissue cysts (6)
- • consuming food or water contaminated with cat feces or by contaminated environmental samples (such as fecal-contaminated soil or changing the litter box of a pet cat) (7)
- • blood transfusion or organ transplantation (8)
- • transplacentally from mother to fetus (9)
- (10) Diagnosis is usually achieved by serology, although tissue cysts may be observed in stained biopsy specimens.
- (11) Diagnosis of congenital infections can be achieved by detecting T gondii DNA in amniotic fluid using molecular methods such as polymerase chain reaction.
Human infection may be acquired in several ways: (1) ingestion of undercooked contaminated meat containing T gondii cysts; (2) ingestion of oocysts from hands, food, soil, or water contaminated with cat feces; (3) organ transplantation or blood transfusion; (4) transplacental transmission; and (5) accidental inoculation of tachyzoites. Transmission through breast milk has not been described. The 2 major routes of transmission of Toxoplasma to humans are oral and congenital. In humans, ingesting either the tissue cyst or the oocyst results in the rupture of the cyst wall [3], which releases sporozoites that invade the intestinal epithelium, disseminate throughout the body, and multiply intracellularly. The host cell dies and releases tachyzoites, which invade adjacent cells and continue the process. The tachyzoites are pressured by the host's immune response to transform into bradyzoites and form tissue cysts, most commonly in skeletal muscle, myocardium, and brain; these cysts may remain throughout the life of the host. Recrudescence of clinical disease may occur if the host becomes immunosuppressed and the cysts rupture, releasing the parasites.
Congenital infection occurs predominantly after primary infection of a pregnant woman. However, well described cases of transmission from women infected shortly before pregnancy, transmission from immunosuppressed women undergoing reactivation, and transmission from women previously infected with one serotype developing a new infection with a second serotype during pregnancy have all been described [4].
The incidence of congenital toxoplasmosis varies with the trimester during which maternal infection was acquired. For untreated women, the transmission rate is approximately 25 percent in the first trimester, 54 percent in the second trimester, and 65 percent in the third trimester. Although the precise mechanism of movement of the parasite across the human placental is not fully understood, recent studies may offer new insights [5]. Human placental studies suggest that the extravillous trophoblasts (EVT) that anchor the placenta to the uterus are much more vulnerable to infection than are the syncytiotrophoblasts that are bathed in maternal blood [5]. Studies in animals have shown that initial infection occurs in the uterus [5]. Thus, it is likely that after primary infection a woman becomes parasitemic, leading to intracellular infection in the uterus, which then gradually leads to EVT infection, as the tachyzoites move from cell to cell, with eventual infection of the fetus. It is also possible that direct movement of tachyzoite infected maternal leukocytes to or across the placenta contributes to fetal infection. It seems likely that multiple mechanisms are involved; which could explain the apparent time delay between primary maternal infection and fetal infection.
In recent studies, researchers have begun to appreciate that differences in (1) reactivation and (2) disease severity may be explained in part by different genotypes of T gondii, of which there are 3 occurring in different parts of the world [4]. These studies are important because they may explain conflicting reports from different regions of the world on the relative public health importance of screening and treatment for congenital disease. Further research is needed to define the role of T gondii genotypes and the interplay with human innate immunity, particularly in the developing fetus and newborn infant. One study on transmission across the human placenta showed a trend towards increased transmission by 1 genotype; however, the differences were not statistically significant [5].
EPIDEMIOLOGY
Serologic prevalence data indicate that toxoplasmosis is one of the most common infections of humans throughout the world, and prevalence increases with age [6]. Due to environmental factors impacting the survival of the oocysts, infection is more common in warm climates and at lower altitudes than in cold climates and mountainous regions. Variations in mode of exposure also lead to variations in prevalence. Analysis of National Health and Nutrition Examination and Survey (NHANES) data showed T gondii seroprevalence had declined in US-born persons 12–49 years old from 14.1% in 1988–1994 to 9.0% in 1999–2004 [7]. The incidence of congenital toxoplasmosis depends upon the proportion of women entering pregnancy without prior immunity and the rate of exposure to Toxoplasma during pregnancy. Estimates of congenital infection in the United States have ranged from 1 in 3000 to 1 in 10 000 live births.
CLINICAL MANIFESTATIONS
Toxoplasmosis can be categorized into 4 groups: (1) acquired in the immunocompetent patient; (2) acquired or reactivated in the immunodeficient patient; (3) ocular; and (4) congenital. Diagnosis and treatment may be different for each clinical category. In general, diagnosis is accomplished using serologic tests, because isolation of the parasite can be difficult. Molecular testing, such as polymerase chain reaction, plays a critical role in diagnosing infection in the fetus. Interpretation of diagnostic tests in pregnant women and newborns can be difficult and should be undertaken using a reliable reference laboratory (Toxoplasma Serology Laboratory, Palo Alto, CA; http://www.pamf.org/serology/) [1].
Acquired infection with Toxoplasma in immunocompetent individuals is generally an asymptomatic infection. However, 10%–20% of patients with acute infection may develop cervical lymphadenopathy or a flu-like illness. The clinical course is benign and self-limited; symptoms usually resolve within weeks to months. Recent data have suggested an association between T gondii infection and various neurologic or psychiatric syndromes, including schizophrenia, Alzheimer disease, and even suicide [8–10]. These findings are intriguing but require further study to validate.
Immunodeficient patients often have central nervous system (CNS) disease but may have myocarditis or pneumonitis. In patients with acquired immune deficiency syndrome, toxoplasmic encephalitis is the most common cause of intracerebral mass lesions and is thought to be due to reactivation of chronic infection. Toxoplasmosis in patients being treated with immunosuppressive drugs may be due to either newly acquired or reactivated latent infection [11, 12].
Ocular toxoplasmosis, an important cause of chorioretinitis in the United States, may be the result of congenital or acquired infection [13]. Congenitally infected patients can be asymptomatic until the second or third decade of life, when lesions develop in the eye presumably due to cyst rupture and subsequent release of tachyzoites and bradyzoites. Chorioretinitis is more often bilateral (30%–80%) in congenitally infected individuals than in individuals with acute acquired T gondii infection. Further defining the interaction of human immunity, timing of infection, and parasite genotype is an important area of ongoing research in understanding ocular toxoplasmosis.
Congenital toxoplasmosis has a wide spectrum of clinical manifestations, but it is subclinical in approximately 75% of infected newborns. The severity of clinical disease in congenitally infected infants is related inversely to the gestational age at the time of primary maternal infection—with first-trimester maternal infection leading to more severe manifestations. When clinically apparent, it may mimic other diseases of the newborn. In a proportion of cases, spontaneous abortion, prematurity, or stillbirth may result. Involvement of the CNS is a hallmark of congenital Toxoplasma infection. The presence of chorioretinitis, intracranial calcifications, and hydrocephalus is considered the classic triad of congenital toxoplasmosis. Fever, hydrocephalus or microcephaly, hepatosplenomegaly, jaundice, convulsions, chorioretinitis (often bilateral), cerebral calcifications, and abnormal cerebrospinal fluid are the classic features of severe congenital toxoplasmosis. Other occasional findings included rash (maculopapular, petechial, or both), myocarditis, pneumonitis and respiratory distress, hearing defects, an erythroblastosis-like picture, thrombocytopenia, lymphocytosis, monocytosis, and nephrotic syndrome.
Some infected children without overt disease as neonates may escape serious sequelae of the infection; however, a significant number (14 to 85%) develop chorioretinitis, strabismus, blindness, hydrocephalus or microcephaly, cerebral calcifications, developmental delay, epilepsy, or deafness months or years later.
Current treatment regimens work primarily against the actively dividing tachyzoite form of T gondii and do not eradicate encysted organisms (bradyzoites). A significant paradigm shift over the past 20–30 years has been the realization that therapy begun prenatally and within the first 1–2 months after delivery can significantly ameliorate subsequent neurologic damage in infected children. For congenital infection, treatment regimens that include pyrimethamine and sulfadiazine with leukovorin—prolonged for at least 1 year and often initiated before birth—seem to be associated with substantially less frequent and severe sequelae. Further refining the optimal treatment regimen is an area requiring further research, especially as newer drugs (azithromycin, atovoquone, etc) with activity against the various stages of Toxoplasma are developed. Continued research is required to determine the impact of therapy during infancy upon recrudescent ocular disease in adolescents who were congenitally infected.
PREVENTION OF TRANSMISSION
Prevention of congenital toxoplasmosis infection involves prevention of primary infection in pregnant women or prevention of transplacental transmission once primary infection has occurred in the mother. Secondary prevention of severe disease in the infected fetus and newborn relies upon early detection and initiation of appropriate therapy for the child, as described above.
Primary prevention of maternal infection depends upon an understanding of modes of acquisition of infection for a pregnant woman. Risk factors for T gondii infection identified in epidemiologic studies include eating raw or undercooked pork, mutton, lamb, beef, ground meat products, oysters, clams, or mussels and wild game meat, kitten ownership, cleaning the cat litter box, contact with soil (gardening and yard work), and eating raw unwashed vegetables or fruits [2, 3, 14–16]. Recommendations for prevention of toxoplasmosis in pregnant women are available on the Centers for Disease Control and Prevention web page: http://www.cdc.gov/parasites/toxoplasmosis/prevent.html. These recommendations include the following: (1) whole cuts of meat should be cooked to safe temperatures of at least 145oF with a 3 minute rest, ground meat should be cooked to 160o F, and poultry should be cooked to at least 165oF with a 3 minute rest; (2) fruits and vegetables should be peeled or washed thoroughly before eating; (3) cutting boards, dishes, counters, utensils, and hands should always be washed with hot soapy water after they have contacted raw meat, poultry, seafood, or unwashed fruits or vegetables; (4) pregnant women should wear gloves when gardening and during any contact with soil or sand, because cat waste might be in soil or sand, and wash hands afterwards; (5) pregnant women should avoid changing cat litter if possible. If no one else is available to change the cat litter, pregnant women should use gloves, then wash their hands thoroughly. The litter box should be changed daily because T gondii oocysts require more than 1 day to become infectious. Pregnant women should be encouraged to keep their cats inside and not adopt or handle stray cats. Cats should be fed only canned or dried commercial food or well cooked table food, not raw or undercooked meats. Several outbreaks have been reported in association with drinking untreated water contaminated by oocysts. Freezing meats for several days at subzero (0oF) greatly reduces the risk of infection and may partially explain the declining prevalence of Toxoplasma infection in the United States, because increasing proportions of meats are frozen for prolonged periods before human consumption. An important area of ongoing study is improved food safety–including the role of irradiation, monitored freezing of meats (as is done for Trichinella), and development of vaccines for use in cats or intermediate host animals.
Spiramycin (available through the US Food and Drug Administration, phone 301-796-0563 or 301-796-1400, or if no response then 301-796-3763) is recommended for pregnant women with acute toxoplasmosis when fetal infection has not yet occurred, in an attempt to prevent transmission of T gondii from the mother to the fetus [17, 18]. Although randomized prospective studies of treatment during acute infection in pregnant women have not been performed, several large prospective and retrospective cohort reviews have been analyzed. Some researchers have questioned, or been unable to demonstrate, the effectiveness of treatment during pregnancy in preventing congenital infection [19, 20] or sequelae in infants [20]. However, a multicenter observational study found that the treatment of acute T gondii infection in pregnancy was associated with a reduction of sequelae in infants, but not a reduction in maternal-fetal transmission [21]. Alternative medications, such as azithromycin, have been used but have not been as extensively studied as spiramycin.
Prevention of transmission from a pregnant woman who has experienced primary infection to her fetus requires prompt identification of what is most often an asymptomatic infection in the woman. Several strategies have been attempted or proposed to identify acutely infected pregnant women. Most strategies involve routine serologic screening for Toxoplasma infection before and during pregnancy. Two important general areas of ongoing research related to routine screening of pregnant women for Toxoplasma infection have been the refinement of diagnostic tools and the economic impact of strategies to apply those tools.
Diagnosing acute infection from a single specimen has proven difficult to accomplish with precision, largely due to the prolonged presence of immunoglobulin (Ig)M antibodies after acute infection. A pregnant woman who is found to be Toxoplasma IgG and IgM positive early in pregnancy may have been infected up to 12 months earlier and therefore unlikely to transmit infection to her developing fetus. Most recently, IgG avidity testing has allowed more precise defining of the acuity of infection [1]. Serial testing once a pregnant woman is found to be IgG negative is accurate but can be costly.
Recent models have suggested that it would be cost-effective to screen all pregnant women in the United States [22] depending upon the cost of the test and the incidence of infection. From a public health perspective, further refinements of the variation in the epidemiology of toxoplasmosis across populations within the United States are needed to inform decisions regarding universal screening.
CONCLUSION
Congenital toxoplasmosis is a severe disease that can be treated and prevented. Despite many years of investigation, several important questions remain to be explored (Table 1). Does parasitic genotypic variation explain the variability seen across the globe in disease prevalence and severity? Are there better treatment options available, both for prevention of transmission from an infected pregnant woman to her developing fetus and for the treatment of congenitally infected children, particularly newer drugs that have shown activity against related parasites? Is it time to seriously consider universal screening of pregnant women in the United States? Do we understand the epidemiology well enough to institute such screening? Can more be done to improve food safety? In 1988, McCabe and Remington [23] wrote an editorial stating that the time had come to seriously address toxoplasmosis in pregnancy to prevent congenital infection. Perhaps now the time has really come.
Table 1.
Research Needs in Maternal and Congenital Toxoplasmosis
| • Defining role of parasitic genotypic variation in the variability of disease prevalence and severity |
| • Better treatment options for prevention of transmission of Toxoplasma infection from a pregnant woman to her fetus |
| • Better drugs for treatment of infants and children with congenital toxoplasmosis |
| • Effective approaches for toxoplasmosis screening in pregnant women |
| • Strategies to improve food safety |
Acknowledgments
Financial support. This work was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development—National Institutes of Health.
Potential conflicts of interest. Author: No reported conflicts.
Author has submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest.
References
- 1.McAuley JB, Jones JL, Singh K. Manual of Clinical Microbiology. 11th ed. Washington DC: ASM Press; 2014. Toxoplasma. [Google Scholar]
- 2.Centers for Disease Control and Prevention. Preventing congenital toxoplasmosis. MMWR Recomm Rep. 2000;49(RR-2):57–75. [PubMed] [Google Scholar]
- 3.Munoz-Zanzi CA, Fry P, Lesina B, Hill D. Toxoplasma gondii oocyst-specific antibodies and source of infection. Emerg Infect Dis. 2010;16:1591–5. doi: 10.3201/eid1610.091674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lindsay DS, Dubey JP. Toxoplasma gondii: the changing paradigm of congenital toxoplasmosis. Parasitology. 2011;138:1829–31. doi: 10.1017/S0031182011001478. [DOI] [PubMed] [Google Scholar]
- 5.Robbins JR, Zeldovich VB, Poukchanski A, et al. Tissue barriers of the human placenta to infection with Toxoplasma gondii. Infect Immun. 2011;80:418–28. doi: 10.1128/IAI.05899-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Robert-Gangneux F, Darde ML. Epidemiology of and diagnostic strategies for Toxoplasmosis. Clin Microbiol Rev. 2012;25:264–96. doi: 10.1128/CMR.05013-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jones JL, Kruszon-Moran D, Sanders-Lewis K, Wilson M. Toxoplasma gondii infection in the United States, 1999–2004, decline from the prior decade. Am J Trop Med Hyg. 2007;77:405–10. [PubMed] [Google Scholar]
- 8.Kusbeci OY, Miman O, Yaman M, et al. Could Toxoplasma gondii have any role in Alzheimer disease? Alzheimer Dis Assoc Disord. 2011;25:1–3. doi: 10.1097/WAD.0b013e3181f73bc2. [DOI] [PubMed] [Google Scholar]
- 9.Ling VJ, Lester D, Mortensen PB, et al. Toxoplasma gondii seropositivity and suicide rates in women. J Nerv Ment Dis. 2011;199:440–4. doi: 10.1097/NMD.0b013e318221416e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Arias I, Sorlozano A, Villegas E, et al. Infectious agents associated with schizophrenia: a meta-analysis. Schizophr Res. 2012;136:128–36. doi: 10.1016/j.schres.2011.10.026. [DOI] [PubMed] [Google Scholar]
- 11.Siberry GK, Abzug MJ, Nachman S, et al. Guidelines for the prevention and treatment of opportunistic infections in HIV-exposed and HIV-infected children: recommendations from the National Institutes of Health, Centers for Disease Control and Prevention, the HIV Medicine Association of the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the American Academy of Pediatrics. Pediatr Infect Dis J. 2013;32(Suppl 2):i–KK4. doi: 10.1097/01.inf.0000437856.09540.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Schmidt-Hieber M, Zweigner J, Uharek L, et al. Central nervous system infections in immunocompromised patients - update on diagnosis and therapy. Leuk Lymphoma. 2009;50:24–36. doi: 10.1080/10428190802517740. [DOI] [PubMed] [Google Scholar]
- 13.Jones J, Holland GN. Short report: annual burden of ocular toxoplasmosis in the United States. Am J Trop Med Hyg. 2010;82:464–5. doi: 10.4269/ajtmh.2010.09-0664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Boyer KM, Holfels E, Roizen N, et al. Risk factors for Toxoplasma gondii infection in mothers of infants with congenital toxoplasmosis: implications for prenatal management and screening. Am J Obstet Gynecol. 2005;192:564–71. doi: 10.1016/j.ajog.2004.07.031. [DOI] [PubMed] [Google Scholar]
- 15.Jones JL, Dargelas V, Roberts J, et al. Risk factors for Toxoplasma gondii infection in the United States. Clin Infect Dis. 2009;49:878–84. doi: 10.1086/605433. [DOI] [PubMed] [Google Scholar]
- 16.Jones JL, Lopez A, Wilson M, et al. Congenital toxoplasmosis: a review. Obstet Gynecol Surv. 2001;56:296–305. doi: 10.1097/00006254-200105000-00025. [DOI] [PubMed] [Google Scholar]
- 17.Remington JS, McLeod R, Thulliez P, Desmonts G. Infectious Diseases of the Fetus and Newborn Infant. In: Remington JS, Klein JO, editors. Toxoplasmosis. 6th ed. Philadelphia, PA: The W. B. Saunders Co.; 2006. pp. 947–1092. [Google Scholar]
- 18.Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet. 2004;363:1965–76. doi: 10.1016/S0140-6736(04)16412-X. [DOI] [PubMed] [Google Scholar]
- 19.Robert-Gangneux F, Gavinet MF, Ancelle T, et al. Value of prenatal diagnosis and early postnatal diagnosis of congenital toxoplasmosis: retrospective study of 110 cases. J Clin Microbiol. 1999;37:2893–8. doi: 10.1128/jcm.37.9.2893-2898.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gilbert R, Dunn D, Wallon M, et al. Ecological comparison of the risks of mother-to-child transmission and clinical manifestations of congenital toxoplasmosis according to prenatal treatment protocol. Epidemiol Infect. 2001;127:113–20. doi: 10.1017/s095026880100560x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Daffos F, Forestier F, Capella-Pavlovsky M, et al. Prenatal management of 746 pregnancies at risk for congenital toxoplasmosis. N Engl J Med. 1988;318:271–5. doi: 10.1056/NEJM198802043180502. [DOI] [PubMed] [Google Scholar]
- 22.Stillwaggon E, Carrier CS, Sautter M, McLeod R. Maternal serologic screening to prevent congenital toxoplasmosis: a decision-analytic economic model. PLoS Negl Trop Dis. 2011;5:e1333. doi: 10.1371/journal.pntd.0001333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.McCabe R, Remington JS. Toxoplasmosis: the time has come. N Engl J Med. 1988;318:313–5. doi: 10.1056/NEJM198802043180509. [DOI] [PubMed] [Google Scholar]