Abstract
Rationale and Objectives
Cancer cells generate more lactate than normal cells under aerobic and hypoxic conditions – exhibiting the so-called Warburg effect. However, the relationship between the Warburg effect and tumor metastatic potential remains controversial. We intend to investigate whether the higher lactate reflects higher tumor metastatic potential.
Materials and Methods
We employed hyperpolarized 13C-pyruvate magnetic resonance spectroscopy (MRS) to compare lactate 13C-labeling in vivo in breast cancer mouse xenografts of highly metastatic (MDA-MB-231) and relatively indolent (MCF-7) human cell lines. We obtained the kinetic parameters of the lactate dehydrogenase (LDH)-catalyzed reaction by three methods of analysis including the differential equation (DE) fit, q-ratio (qR) fit, and ratio fit (RF) methods.
Results
Consistent results from the three methods showed that the highly metastatic breast tumors exhibited a smaller apparent forward rate constant (k+ = 0.060 ± .004 s−1) than the relatively indolent tumors (k+ = 0.097 ± .013 s−1). The RF fit generates the highest statistical significance for the difference (p=0.02). No significant difference is found for the reverse rate constant.
Conclusion
The result indicates that the less metastatic breast tumors may produce more lactate than the highly metastatic ones from the injected 13C-pyruvate, and supports the viewpoint that breast tumor metastatic risk is not necessarily associated with the high levels of glycolysis and lactate production. More studies are needed to confirm whether and how much the measured apparent rate constants are affected by the membrane transporter activity and whether they are primarily determined by the LDH activity or not.
Keywords: pyruvate, Warburg effect, NMR, ratio fit, two site exchange model
Introduction
Metastasis results in about 90% of cancer deaths. However, tumor metastatic risk determination remains one of the greatest clinical challenges due to the lack of reliable biomarkers of metastatic potential (1). In recent years, cancer metabolism has received increasing research attention, and abnormally high level of glycolysis and lactate production (the Warburg effect) has been recognized as the hallmark of cancer (2–4). The Warburg effect has been demonstrated by a variety of techniques in vitro, ex vivo and in vivo with a large portion of these studies performed on cell culture models. A few methods have been used to demonstrate the Warburg effect in tissues as well. For example, FDG-PET detects the higher uptake and accumulation of FDG in tumors compared to normal tissue in vivo (5, 6) and the magnetic resonance spectroscopy and imaging (MRS/MRI) including hyperpolarized 13C-MR techniques (7–12) monitor higher 13C-lactate production from 13C-labeled hyperpolarized pyruvate in vivo.
It is clear that the Warburg effect discriminates cancer from non-malignant tissues. However, it is less clear if it also accounts for differences in cancer metastatic potential. Some studies using bioluminescence imaging of biopsies from human cancers indicate a positive correlation between tumor lactate concentration and incidence of metastasis in head and neck cancer patients and cervical cancer patients (13–16). Lactate levels measured by 1H-MRS were also shown as strong indicators of tumor grade and poor prognosis in various cancers, such as in brain (17), breast (18–20), lung (21), and liver (22). In addition, a positive correlation between the hyperpolarized lactate level and prostate tumor histological grades has been observed in transgenic mouse models by hyperpolarized NMR (23). The hypothesis that lactate enhances tumor survival, invasiveness, and metastatic potential was supported by evidence that lactate stimulates HIF-1α accumulation (24) and increases CD44 expression levels (associated with invasion) in both stromal fibroblasts and cancer cells (25, 26). On the other hand, other studies showed that lactate levels did not correlate with the invasive/metastatic potential of the breast cancer cells (27, 28) or histopathologic grade of oligodendrogliomas (29).
In the present study, we employed the hyperpolarized 13C-MRS technique (HP-MRS) to assess in vivo lactate fluxes or lactate pool size (concentration) in two well-established mouse models of human breast cancer lines, the less metastatic (MCF-7) and the more metastatic (MDA-MB-231) tumors (27, 30). In order to identify the best data-analysis approach to detecting the difference in enzymatic kinetics between the two tumor lines, we have applied three different analysis methods to quantify the apparent reaction rate constants of the LDH reaction. The RF and qR fit methods are based on fitting the time courses of the lactate to pyruvate ratios, whereas the DE fit method is a direct fit of the individual time courses of the metabolite signals. The three methods consistently showed that the more metastatic tumors had a lower apparent forward rate constant or lactate flux for the LDH reaction. The RF fit method has the highest statistical significance among the three approaches. The lactate flux in this paper represented by the forward rate constant is not the net flux and should be proportional to the lactate pool size or the concentration of lactate under steady state or near-equilibrium state. Our observation appears to indicate that the more metastatic breast tumors exhibited a smaller lactate pool size or lactate concentration, in other words, less Warburg effect compared to the less metastatic tumors. A preliminary report of this study was published in a conference proceeding (31).
Materials and Methods
The animal protocol of this study was approved by the Institutional Animal Care and Use Committee (IACUC) of the University Name. Human breast cancer lines MCF-7and MDA-MB-231 were propagated in culture and inoculated into the upper thighs of athymic nude mice (US National Cancer Institute, strain NCr nu/nu, 4–5 weeks) to produce tumor xenografts as described in our previous study (30). Tumor dimensions were measured weekly with a caliper. At the time of imaging, the tumor size ranged from 140–1400 mm3 and mouse weight ~20–30g.
All NMR experiments were performed with a 1.1-cm or 1.4-cm 1H/13C dual-tuned home-made surface coil in a 9.4-T Varian vertical bore NMR spectrometer as previously described (32, 33). The tumor-bearing mice were anesthetized by administering oxygen doped with 1% isofluorane while the body temperature was maintained at ~35.0±2.5 °C with heated air. Tail vein injection of 250 μL 75mM hyperpolarized (via the DNP method using a HyperSense, Oxford Instruments) 1-13C-pyruvate (~10 μL/g mouse body weight) was completed in ~10 sec. Single-pulse or slice-selective (for smaller tumors) 13C-NMR spectra were collected over a period of 2 minutes with a 9~15° nominal flip angle every 1 or 2 seconds. For the small subcutaneous tumors our slice selection is oriented tangentially to the body surface so that minimal normal tissues were included. For large tumors, the tumor thickness is larger than the diameter of the surface coil and the majority of the signals come from the tumor tissues. The number of tumors obtained with valid NMR data is 4 for MCF-7 line and 3 for MDA-MB-231 line, respectively.
Data were analyzed with the assistance of customized Matlab® (MathWorks) programs. Line broadening (20 Hz) was applied before Fourier transformation of the NMR free induction decay (FID). The spectral baseline was removed by fitting it to a fourth order polynomial function. A coarse fitting was first performed by summing up the spectra of all time points; the sum was then fitted to Lorentzian functions to obtain an estimate of the peak positions and widths of the 13C-labeled pyruvate and lactate signals. Peak areas at each time point were then obtained by fitting the individual spectra to Lorentzian functions. The time courses of the pyruvate and lactate signals were smoothed among every three neighboring data points before further data analysis.
Three data analysis methods were used to extract the kinetic parameters, i.e. the reaction rate constants of the lactate dehydrogenase-catalyzed reaction as described by Li et al. (32, 33). The principles of the analysis are briefly summarized as follows. We assume that the LDH reaction is pseudo-first order, i.e.,
| (1) |
where k+ and k− are the apparent forward and reverse rate constants. From the two-site exchange model, we have the following differential equations
| (2a) |
| (2b) |
where P and L represent the NMR signals of hyperpolarized pyruvate and lactate, respectively and ρ is the T1 relaxation rate (assumed equal for pyruvate and lactate in vivo (33, 34)).
Method 1, differential equation fitting (DE), fits the time courses of pyruvate and lactate signals directly to Eq. 2 to extract the rate constants and the T1 relaxation rates of the metabolites. Method 2, the qR fit, determines the two rate constants from the slope and intercept of a linear correlation between q and the lactate-to-pyruvate ratio, R(t), derived from Eq. 2,
| (3) |
where R(t)=L(t)/P(t) and q = (dR/dt)/(1 + R). Method 3, the ratiometric fitting or ratio-fit (RF) analysis, fits the time course of R(t). From the two-site exchange model shown by Eq. 2, we solve for P(t) and L(t), giving:
| (4a) |
| (4b) |
where L(0) and P(0) are the signal intensities at t=0. Dividing Eq. 4b by Eq. 4a, we have
| (5) |
where r = k+/k−, s = k+ + k−. By fitting the ratio time course R(t) to Eq. 5, we obtain the fitting parameters k+ and k− as well as their ratio k+/k−.
As demonstrated previously (33), both the two-site exchange model and the linearity of q versus R are correct only for a specific time window. The exact initial and terminal time points of this window were determined from the linear correlation for q versus R and the SNR threshold. We did not use the early time points in which the pyruvate signal was increasing for any of the analyses since the two site exchange model may not be valid due to the initial process of blood transport of the hyperpolarized pyruvate into the tissue. Due to the low SNR, data from much later time points were also discarded. The SNR threshold was set at 2.5 for the pyruvate signals regardless of the SNR of the lactate signals. In practice, the initial time point determined as above varied from the pyruvate peak to the 2nd time point after for the datasets in this study. To compare the results of different analysis methods and their sensitivity to initial time points, we also varied the initial time point from the first to fourth time point after the pyruvate signal peaked while keeping the terminal time points unchanged as decided by the SNR threshold.
The forward and backward rate constant and their ratio were quantified for each tumor using the NMR signals within the specified time window. The mean value ± standard deviation (SD) was obtained by averaging the forward and backward rate constants and their ratios for each tumor line. The significance of the difference in the means between the two tumor lines was tested by unpaired Student’s t-test and p<0.05 is considered statistically significant. To test the difference between analysis methods, we pooled the data from two tumor lines for a paired Student’s t-test.
Results
In this investigation, we evaluated the hyperpolarized pyruvate and lactate signals to monitor lactate and pyruvate pool cross-labeling by the two breast tumor lines that have divergent growth rates and metastatic potentials. A representative time series of 13C-MR spectra of a MDA-MB-231 tumor is provided as Figure 1. Fig. 2 shows the typical time courses of hyperpolarized pyruvate and lactate signals, the DE, qR and RF fit results of a representative MDA-MB-231 tumor. Fig. 3 shows the results of the same fitting analyses for a representative MCF-7 tumor. For all tumors, the fitting correlation coefficient R2 ranges from 0.87–0.99 for the DE fit, 0.06–0.58 for the qR fit and 0.91–0.99 for the RF fit.
Fig. 1.

A representative time series of 13C-MR spectra from a breast tumor. The peak of 1-13C-pyruvate is nearby 171ppm and 1-13C-lactate is nearby 182 ppm. The time interval between adjacent spectra is 1s.
Figure 2.
The typical time courses of hyperpolarized pyruvate (*) and lactate (◊) and the DE fits (left), the qR fit (middle) and the ratio fit (right) results of a MDA-MB-231 tumor. All rates are in s−1.
Figure 3.
The typical time courses of hyperpolarized pyruvate (*) and lactate (◊), the DE fit (left) and the qR fit (middle) and the ratio fit (right) results of a MCF-7 tumor. All rates are in s−1.
The apparent rate constants k+ and k− as well as their ratio k+/k− are summarized in Table 1. The three different analysis methods (DE, qR and RF fits) give consistent values of the kinetic parameters. There is no significant difference in the mean values of rate constants and rate constant ratios comparing the RF or qR fit with the DE fit (p>0.1, paired t-test using the data of each tumor line or pooling the data of both lines). The SDs of the mean values of rate constants from the DE fit are larger than that from the RF or qR fits. The comparison of the results from RF and qR fits indicate significant difference in the mean values of rate constants and ratios when pooling the data of two tumor lines together (p<0.02 using paired t-test). The mean values of rate constants− from the RF fit appear to be ~10% for k+ and ~20% for k− less than those results from the qR fit, respectively. The mean value of the rate constant ratio k+/k− from the RF fit is about 8% higher than that from the qR fit (p<0.01 using paired t-test).
Table 1.
The apparent rate constants (s−1) and rate ratios (mean ± SD) quantified by the three analysis methods* (N=3 for MDA-MB-231 line and N=4 for MCF-7 line)
| Method | Tumor Phenotype | k+ (s−1) | k−(s−1) | k+/k− | Size (mm3) |
|---|---|---|---|---|---|
| DE fit | MDA-MB-231 | 0.059 ± .020 | 0.024 ± .012 | 2.64 ± 1.03 | 611 ± 711 |
| MCF-7 | 0.111 ± .038 | 0.027 ± .010 | 4.17 ± .67 | 283 ± 120 | |
| #Both lines | 0.089 ± .040 | 0.026 ± .010 | 3.51 ± 1.12 | ||
| p | 0.068 | 0.77 | 0.11 | 0.51 | |
| QR fit | MDA-MB-231 | 0.063 ± .013 | 0.028 ± .011 | 2.63 ± 1.54 | |
| MCF-7 | 0.093 ± .006 | 0.022 ± .007 | 4.58 ± 1.27 | ||
| Both lines | 0.080 ± .018 | 0.024 ± .009 | 3.75 ± 1.64 | ||
| p | 0.047 | 0.44 | 0.15 | ||
| RF fit | MDA-MB-231 | 0.057 ± .009 | 0.023 ± .009 | 2.83 ± 1.68 | |
| MCF-7 | 0.086 ± .005 | 0.019 ± .007 | 4.88 ± 1.32 | ||
| Both lines | 0.074 ± .017 | 0.021 ± .007 | 4.00 ± 1.73 | ||
| p | 0.02 | 0.52 | 0.16 |
Analysis methods include the DE (differential equation) fit, QR(q-ratio) fit, and RF(ratio-fit) fit.. The p values are from the Student’s t test comparing the results of two tumor lines using the same analysis method.
The rows of “both lines” show average values pooling the data of both tumor lines.
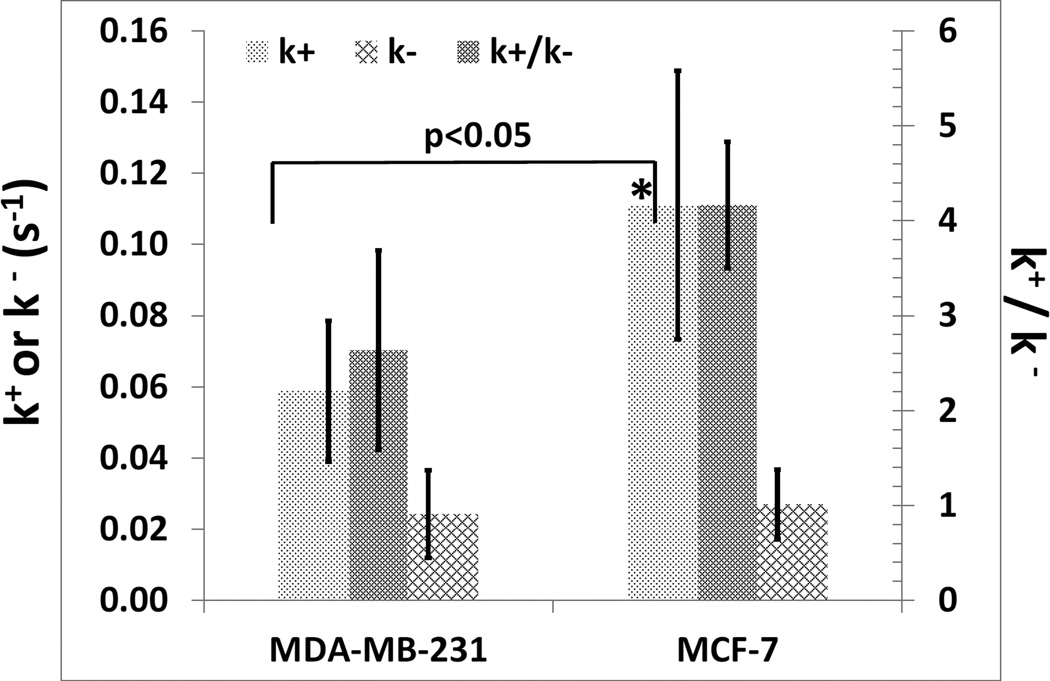
Table 1 also shows that only the forward rate constant k+ is significantly different between two tumor models. The DE method gives k+ = 0.059 ± .020 s−1 and 0.111 ± .038 s−1 for MDA-MB-231 and MCF-7 tumors, respectively, with p=0.068 when comparing the difference between the two tumor lines. The qR method yields k+ = 0.063 ± .013 s−1 and 0.093 ± .006 s−1 for MDA-MB-231 and MCF-7 tumors, respectively, with p=0.047 for the difference. The RF analysis produces k+ = 0.057 ± .009 s−1 and 0.086 ± .005 s−1 for MDA-MB-231 and MCF-7 tumors, respectively, with p=0.018 for the difference. The statistical significance levels (as reflected by the p values) increase in the rank order DE<qR<RF. The average value of k+ from the three methods is 0.060± .004 s−1 for MDA-MB-231 and 0.097± .013 s−1 for MCF-7 tumors. These results demonstrate a significant difference in the average values of the forward rate constants between the two tumor lines. There is no significant difference in the reverse rate constant k− or in the (k+/k−) ratio between two groups (Table 1 and Figure 4), which is likely due to the relatively large error (~30–50%) in the determination of k−.
Figure 4.
The comparison of rate constants (k+, k−) and ratios (k+/k−) between the MCF-7 and MDA-MB-231 tumor xenografts. These results were obtained with the RF (ratiofit) method shown in Table 1.
The sensitivity of the analyses on the selection of the initial time points for the fitting time window is shown in Table 2. For all four conditions of initial time points (the 1st–4th point after the pyruvate peak), the mean values of the forward and reverse rate constants are consistent within their standard deviations for all three analysis methods. For the backward rate constant k−, no significant difference is detected between the MDA-MB-231 and MCF-7 tumor lines for all conditions, consistent with the results in Table 1. However, a significant difference is consistently observed between the forward rate constants k+ of the two cell lines as long as the initial fitting point is fairly close to the time of maximum signal (first, second or third point after the pyruvate maximum). For these cases, the statistical significance levels increase in the rank order of DE<qR<RF consistent with Table 1. The DE method has borderline significance (0.1>p>0.05) when the initial time point after the pyruvate peak varies from 1 to 3. The qR method has borderline significance when initial time point is 1 and becomes significant when the initial time point is 2 or 3. The highest levels of significance are shown by the RF method when the initial time point ranging 1–3. If more points are eliminated from the fit (e.g., initial time point chosen at the 4th point after pyruvate maximum), all fitting methods lose their significance due to low signal-to-noise levels.
Table 2.
Comparison of different analysis methods with various initial time points after the pyruvate peak
| *Methods | Tumor | 1st | 2nd | 3nd | 4th | ||||
|---|---|---|---|---|---|---|---|---|---|
| k+(s−1) | k−(s−1) | k+(s−1) | k−(s−1) | k+(s−1) | k−(s−1) | k+(s−1) | k−(s−1) | ||
| DE | 231 | 0.061 ± .022 | 0.026 ± .011 | 0.056 ± .015 | 0.022 ± .006 | 0.062 ± .012 | 0.028 ± .010 | 0.069 ± .014 | 0.034 ± .016 |
| MCF-7 | 0.109 ± .039 | 0.025 ± .011 | 0.108 ± .042 | 0.025 ± .012 | 0.101 ± .029 | 0.023 ± .011 | 0.101 ± .037 | 0.023 ± .012 | |
| P | 0.096 | 0.98 | 0.081 | 0.73 | 0.067 | 0.54 | 0.18 | 0.36 | |
| QR | 231 | 0.065 ± .014 | 0.029 ± .010 | 0.062 ± .008 | 0.028 ± .012 | 0.067 ± .005 | 0.033 ± .017 | 0.072 ± .008 | 0.037 ± .021 |
| MCF-7 | 0.093 ± .006 | 0.022 ± .007 | 0.090 ± .005 | 0.021 ± .008 | 0.086 ± .010 | 0.020 ± .011 | 0.087 ± .012 | 0.020 ± .012 | |
| P | 0.060 | 0.38 | 0.010 | 0.45 | 0.028 | 0.34 | 0.12 | 0.30 | |
| RF | 231 | 0.057 ± .010 | 0.024 ± .008 | 0.054 ± .008 | 0.022 ± .011 | 0.058 ± .005 | 0.026 ± .015 | 0.060 ± .004 | 0.028 ± .018 |
| MCF-7 | 0.086 ± .004 | 0.019 ± .007 | 0.084 ± .005 | 0.018 ± .007 | 0.081 ± .009 | 0.018 ± .009 | 0.060 ± .042 | 0.014 ± .014 | |
| P | 0.029 | 0.47 | 0.012 | 0.62 | 0.0081 | 0.48 | 0.99 | 0.32 | |
DE: differential equation fit; QR: q-ratio fit; RF: ratio-fit. Ratio refers to the lactate to pyruvate ratio. The k+ and k− refer to the forward and reverse rate constant of the LDH reaction, respectively.
Discussion
Owing to its ~10,000-fold enhanced signal in detecting the 13C-enriched compounds and their metabolites compared to that of standard NMR techniques, HP-NMR provides a non-invasive method for dynamic measurement of tissue metabolism in vivo by probing enzymatic reactions in preclinical and clinical studies (11, 35). Because of its direct association with the Warburg effect and a variety of technical considerations, most HP-NMR work to date has probed the lactate dehydrogenase-catalyzed conversion of pyruvate to lactate. Although tumor cells generate or retain more lactate than normal tissues, it is still unclear if and how the Warburg effect is related to tumor metastatic risk. In this study, we set out to use MRS of hyperpolarized 13C-pyruvate to characterize and compare the kinetic fluxes of the LDH reaction in two breast cancer xenografts MDA-MB-231 and MCF-7. MDA-MB-231 tumors are highly metastatic, and MCF-7 tumors exhibit low levels of metastasis (27).
We employed three different methods on the basis of two-site exchange models to quantify the apparent rate constants of the LDH reaction and obtained consistent results for the two breast cancer xenografts. The DE fit is a direct fit to the time courses of pyruvate and lactate signals. Both the qR and RF fits are based on the lactate-to-pyruvate signal ratios, which are less sensitive to technical parameters and instrument settings, such as the degree of polarization and flip angles of the radiofrequency pulses compared to the individual signals (33). The T1 relaxation rates of 13C in pyruvate and lactate were assumed to be equal in these analyses. The accuracy of the DE and the RF methods was shown to vary with various factors including the width of transport function, relaxation rate, noise level, and rate constant ratio (33). The fit results of these methods for the same NMR data set can be quite different based on these specific conditions. Nevertheless, in this study the fitting results from the three analysis methods consistently show a difference in the forward rate constant of LDH reaction between the two tumor lines. This consistency might be understood according to our previous simulations of the HP-NMR signals and the quantification of the rate constants by the RF and DE methods (33). The time profiles of the pyruvate and lactate signals indicate that five out of the seven tumors in this study (except two MCF-7 tumors) had sharp profiles of blood transport functions, and the errors in the rate constant quantifications would be comparable between the RF and DE methods when the rate constant ratio k+/k− is 1~5 (See Fig. 10 in Ref 33). The consistency of the analysis results of the three methods is further supported by the insensitivity of the results to the selection of the initial time points for the fit window (Table 2). In all cases, the analysis results are consistent with the conclusion that the more metastatic MDA-MB-231 xenograft has a smaller apparent forward rate constant or a lower lactate 13C labeling flux than the less metastatic MCF-7 xenograft. Note that the RF method produced the highest statistically significance among the three methods indicating the advantage of this ratiometric fitting method.
The forward flux rate of the LDH reaction converting pyruvate to lactate depends on the concentration of NADH, the coenzyme of lactate dehydrogenase. The intracellular lactate pool size (i.e. the concentration of both 12C-lactate and 13C-lactate) also affects hyperpolarized pyruvate to lactate conversion. Non-labeled lactate co-administered with hyperpolarized pyruvate can increase the forward flux (from 13C-pyruvate to 13C-lactate) (34). This pool size effect can be understood by considering the increase of NADH concentration resulting from the backward reaction flux (from lactate to pyruvate). Usually the LDH reaction is treated as a pseudo first order reaction with the NADH and NAD+ concentrations absorbed into the forward and backward rate constants, k+ and k−, respectively. The LDH reaction is expected to reach near equilibrium within seconds even with the abnormally-high exogenous pyruvate in-flux (33, 34), i.e, k+ Pm = k− Lm, where Pm and Lm represents the total (both 13C and 12C) steady state concentrations of pyruvate and lactate, respectively. Therefore, the forward LDH reaction rate constant, k+ measured in the hyperpolarized experiments reflects the rate constant under the new equilibrium, and is expected to reflect or be proportional to the total lactate concentration or lactate pool size.
Thus, the higher k+ of the MCF-7 tumors and lower k+ of the MDA-MB-231 tumors obtained from our preliminary data indicate that the less metastatic breast tumors have relatively higher lactate pool size or lactate concentration than the more metastatic breast tumors, contradicting the commonly held belief that lactate concentration correlates with tumor metastatic potential. The lactate concentrations of these two tumor lines have been measured by various methods and the results are somewhat inconsistent. For example, 1H-MRS measurement of PCA extracts of cell cultures showed that the lactate concentration ratio of MDA-MB-231 to MCF-7 was ~60% (27, 28). Spectrophotometric measurement of the cell line extractions gave the lactate ratio of ~48% (36). It was also reported that the orthotopic breast tumors (>200 mm3) of these two lines yield a concentration ratio of ~33% as assessed by lactate MRS using the multiple-quantum-coherence spectral editing technique (37). Our hyperpolarized MRS measurements gave an apparent rate constant ratio of MDA-MB-231 to MCF-7 of ~62% using the average values from the three analysis methods. Although variations do exist in the reported lactate concentrations, these studies point to the same conclusion: that intracellular lactate concentration in MCF7 cells exceeds that of MDA-MB-231 cells.
Notably, some other investigations showed completely different results. For example, Robey et al. reported that MDA-MB-231 cells produced over 3 times more lactate than the MCF-7 cells under normoxic conditions (20% oxygen) (38). Under similar in vitro conditions, other investigators reported 1.5 times higher lactate in MDA-MB-231 cells than in MCF-7 cells (39). However, these studies consistently showed that hypoxia significantly increased MCF-7 cells’ lactate production, but it did not significantly alter the lactate level in MDA-MB-231 cells. This may partially explain the apparent contradiction among aforementioned studies. In solid tumors, cancer cells are constantly under hypoxic conditions, which may increase the lactate level of MCF-7 cells but that of MDA-MB-231 cells may not be much affected. It is ultimately desirable to perform PCA extraction of lactate from tumor tissues for a direct quantification of lactate concentration and compare the results with that determined by our HP-MRS study. Alternatively, the lactate generation from infused glucose can be measured in tumors by conventional 13C-MRS (40).
Although divergent measurements of lactate production have been obtained by various methods, some studies concluded that the lactate level is associated with malignancy (cancer versus non-cancer), and appears not to be associated with tumor metastatic potential (27, 28, 41). Our in vivo results from this study further support the conclusion that the lactate level is not necessarily associated with breast tumor metastatic risk. Our previous studies have shown tumor metastatic risk is highly correlated with the mitochondrial redox state in both melanoma and breast cancer mouse models, and the more metastatic tumors have a more oxidized mitochondrial redox state (30, 42, 43). We also showed that the mitochondrial redox state discriminates colon tumors with wild type p53 from those with p53−/− (44). Additionally, the mitochondrial redox indices distinguish premalignant tissue caused by PTEN deletion from normal tissue in a transgenic mouse model (45, 46) and differentiate between malignant specimens and normal controls in breast cancer patients (47, 48). Together these results suggest that the mitochondrial metabolism may be more associated with tumor metastatic risk (49) than lactate levels, which mainly reflect cytosolic metabolism.
The advantage of the hyperpolarized 13C-MRS is that lactate labeling flux can be measured non-invasively in vivo within a very short period of time. However, this approach has some limitations. The lactate signal in tumors as observed by the hyperpolarized 13C-MRS is affected by a few factors (34, 50–52). Hyperpolarized 13C-pyruvate enters cells via monocarboxylate transporters (MCTs). MCTs are composed of a catalytic unit and an accessory unit (CD147, a cell surface glycoprotein) and are responsible for the efflux of intracellular lactate (53). MCT activity may or may not play a significant role in determining the kinetics of hyperpolarized 13C-lactate signals depending on the biological models and experimental circumstances (52, 54). MCF-7 and MDA-MB-231 cells differentially express MCTs (36, 55, 56). MCF-7 cells express more MCT-1 and MDA-MB-231 cells express more MCT-4. The exact difference in the MCT transport activities between these two tumor xenografts is unknown. Another consideration is the differential expression pattern of the LDH isoforms in these two lines. LDH activity was found to be higher in highly metastatic MDA-MB-231 cells than in poorly metastatic MCF-7 cells. However, no apparent association between LDH activity and lactate level was observed for four breast tumor lines (including these two used in the present study) (27, 57). Other factors include the transport of hyperpolarized pyruvate to tumor through the blood system, and the relative tumor extracellular volume fraction, and the effects of these factors should be more carefully investigated in the future.
Thus, accurate explanations of the hyperpolarized lactate flux difference between the investigated two tumor phenotypes are still difficult due to the complexity of tumor tissue microenvironments and metabolism as well as insufficient knowledge about the activity or levels of the MCTs, LDH, NAD+ coenzyme and the metabolites. We need to quantify the amount of the metabolites, coenzymes, enzymes, and pH in order to determine the LDH activity in vivo. Note that the in vitro tissue enzymatic activity does not really provide sufficient information for understanding the enzymatic activity in vivo. The results from the in vitro biochemical assays depend on the co-enzymes and the substrates added in vitro, which does not reflect the in vivo condition. These in vitro assays can only provide the amount of active enzymes in the tissue specimen but not the enzymatic reaction fluxes in vivo. Therefore, the ultimate goal is to develop an in vivo technique to determine the flux, and it seems that HP-NMR has the potential for achieving this goal. The HP-NMR can also be implemented as a spectroscopic imaging method to investigate spatial heterogeneities of tumor metabolism, which may provide more insight into tumor progression in vivo.
Conclusions
We have reported the results of in vivo HP-MRS studies of lactate dehydrogenase-catalyzed conversion between hyperpolarized pyruvate and lactate in two breast tumor models with different metastatic potentials. Our preliminary results show that the less metastatic MCF-7 tumors have a significantly larger apparent forward rate constant and thus a higher level of lactate labeling than the highly metastatic MDA-MB-231 tumors, indicating that high lactate levels are not necessarily associated with high breast tumor metastatic risk. Future studies with more mice are needed to take into account various factors such as membrane-transporter activity, coenzyme levels and inter- and intra-tumor heterogeneities.
Acknowledgement
This work is supported by National Institute of Health R01 CA155348 (L.Z. Li). We also appreciate the support from Mr. Ben Pullinger and Ms. Lily Moon.
References
- 1.Sleeman J, Steeg PS. Cancer metastasis as a therapeutic target. European Journal of Cancer. 2010;46(7):1177–1180. doi: 10.1016/j.ejca.2010.02.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cairns RA, Harris IS, Mak TW. Regulation of cancer cell metabolism. Nat Rev Cancer. 2011;11(2):85. doi: 10.1038/nrc2981. [DOI] [PubMed] [Google Scholar]
- 3.Cantor JR, Sabatini DM. Cancer cell metabolism: one hallmark, many faces. Cancer discovery. 2012;2(10):881–898. doi: 10.1158/2159-8290.CD-12-0345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ward PS, Thompson CB. Metabolic reprogramming: a cancer hallmark even warburg did not anticipate. Cancer cell. 2012;21(3):297–308. doi: 10.1016/j.ccr.2012.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jadvar H, Alavi A, Gambhir SS. 18F-FDG uptake in lung, breast, and colon cancers: molecular biology correlates and disease characterization. J Nucl Med. 2009;50:1820–1827. doi: 10.2967/jnumed.108.054098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gambhir SS. A tabulated summary of the FDG PET literature. J Nucl Med. 2001;42 [PubMed] [Google Scholar]
- 7.Tessem M-B, Swanson MG, Keshari KR, et al. Evaluation of lactate and alanine as metabolic biomarkers of prostate cancer using 1H HR-MAS spectroscopy of biopsy tissues. Magn Reson Med. 2008;60(3):510. doi: 10.1002/mrm.21694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Swanson MG, Zektzer AS, Tabatabai ZL, et al. Quantitative analysis of prostate metabolites using 1H HR-MAS spectroscopy. Magn Reson Med. 2006;55(6):1257. doi: 10.1002/mrm.20909. [DOI] [PubMed] [Google Scholar]
- 9.Cornel EB, Smits GA, Oosterhof GO, et al. Characterization of human prostate cancer, benign prostatic hyperplasia and normal prostate by in vitro 1H and 31P magnetic resonance spectroscopy. J Urol. 1993;150(6):2019–2024. doi: 10.1016/s0022-5347(17)35957-8. [DOI] [PubMed] [Google Scholar]
- 10.Park I, Larson PE, Zierhut ML, et al. Hyperpolarized 13C magnetic resonance metabolic imaging: application to brain tumors. Neuro-oncology. 2010;12(2):133–144. doi: 10.1093/neuonc/nop043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kurhanewicz J, Vigneron DB, Brindle K, et al. Analysis of cancer metabolism by imaging hyperpolarized nuclei: prospects for translation to clinical research. Neoplasia. 2011;13(2):81–97. doi: 10.1593/neo.101102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brindle KM, Bohndiek SE, Gallagher FA, Kettunen MI. Tumor imaging using hyperpolarized 13C magnetic resonance spectroscopy. Magn Reson Med. 2011;66(2):505–519. doi: 10.1002/mrm.22999. [DOI] [PubMed] [Google Scholar]
- 13.Walenta S, Wetterling M, Lehrke M, et al. High lactate levels predict likelihood of metastases, tumor recurrence, and restricted patient survival in human cervical cancers. Cancer research. 2000;60(4):916–921. [PubMed] [Google Scholar]
- 14.Walenta S, Salameh A, Lyng H, et al. Correlation of high lactate levels in head and neck tumors with incidence of metastasis. The American journal of pathology. 1997;150(2):409–415. [PMC free article] [PubMed] [Google Scholar]
- 15.Schwickert G, Walenta S, Sundfor K, Rofstad EK, Mueller-Klieser W. Correlation of high lactate levels in human cervical cancer with incidence of metastasis. Cancer research. 1995;55(21):4757–4759. [PubMed] [Google Scholar]
- 16.Brizel DM, Schroeder T, Scher RL, et al. Elevated tumor lactate concentrations predict for an increased risk of metastases in head-and-neck cancer. International journal of radiation oncology, biology, physics. 2001;51(2):349–353. doi: 10.1016/s0360-3016(01)01630-3. [DOI] [PubMed] [Google Scholar]
- 17.Czernicki Z, Horsztynski D, Jankowski W, Grieb P, Walecki J. Malignancy of brain tumors evaluated by proton magnetic resonance spectroscopy (1H-MRS) in vitro. Acta neurochirurgica Supplement. 2000;76:17–20. doi: 10.1007/978-3-7091-6346-7_4. [DOI] [PubMed] [Google Scholar]
- 18.Sharma U, Mehta A, Seenu V, Jagannathan NR. Biochemical characterization of metastatic lymph nodes of breast cancer patients by in vitro 1H magnetic resonance spectroscopy: a pilot study. Magnetic resonance imaging. 2004;22(5):697–706. doi: 10.1016/j.mri.2004.01.037. [DOI] [PubMed] [Google Scholar]
- 19.Gribbestad IS, Fjosne HE, Haugen OA, et al. In vitro proton NMR spectroscopy of extracts from human breast tumours and non-involved breast tissue. Anticancer research. 1993;13(6A):1973–1980. [PubMed] [Google Scholar]
- 20.Yamamoto N, Watanabe T, Katsumata N, et al. Construction and validation of a practical prognostic index for patients with metastatic breast cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 1998;16(7):2401–2408. doi: 10.1200/JCO.1998.16.7.2401. [DOI] [PubMed] [Google Scholar]
- 21.Yokota H, Guo J, Matoba M, Higashi K, Tonami H, Nagao Y. Lactate, choline, and creatine levels measured by vitro 1H-MRS as prognostic parameters in patients with non-small-cell lung cancer. Journal of magnetic resonance imaging :JMRI. 2007;25(5):992–999. doi: 10.1002/jmri.20902. [DOI] [PubMed] [Google Scholar]
- 22.Yang Y, Li C, Nie X, et al. Metabonomic studies of human hepatocellular carcinoma using high-resolution magic-angle spinning 1H NMR spectroscopy in conjunction with multivariate data analysis. Journal of proteome research. 2007;6(7):2605–2614. doi: 10.1021/pr070063h. [DOI] [PubMed] [Google Scholar]
- 23.Albers MJ, Bok R, Chen AP, et al. Hyperpolarized 13C Lactate, Pyruvate, and Alanine: Noninvasive Biomarkers for Prostate Cancer Detection and Grading. Cancer research. 2008;68(20):8607–8615. doi: 10.1158/0008-5472.CAN-08-0749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lu H, Forbes RA, Verma A. Hypoxia-inducible factor 1 activation by aerobic glycolysis implicates the Warburg effect in carcinogenesis. The Journal of biological chemistry. 2002;277(26):23111–23115. doi: 10.1074/jbc.M202487200. [DOI] [PubMed] [Google Scholar]
- 25.Stern R, Shuster S, Neudecker BA, Formby B. Lactate stimulates fibroblast expression of hyaluronan and CD44: the Warburg effect revisited. Experimental cell research. 2002;276(1):24–31. doi: 10.1006/excr.2002.5508. [DOI] [PubMed] [Google Scholar]
- 26.Edward M, Gillan C, Micha D, Tammi RH. Tumour regulation of fibroblast hyaluronan expression: a mechanism to facilitate tumour growth and invasion. Carcinogenesis. 2005;26(7):1215–1223. doi: 10.1093/carcin/bgi064. [DOI] [PubMed] [Google Scholar]
- 27.Aboagye EO, Mori N, Bhujwalla ZM. Effect of malignant transformation on lactate levels of human mammary epithelial cells. Advances in enzyme regulation. 2001;41:251–260. doi: 10.1016/s0065-2571(00)00019-4. [DOI] [PubMed] [Google Scholar]
- 28.Aboagye OE, Bhujwalla MZ. Book Characterization of Lactate Levels in Normal and Malignant Human Mammary Epithelial Cells. City. 1999. Characterization of Lactate Levels in Normal and Malignant Human Mammary Epithelial Cells. [Google Scholar]
- 29.Jenkinson MD, Smith TS, Joyce K, et al. MRS of oligodendroglial tumors: correlation with histopathology and genetic subtypes. Neurology. 2005;64(12):2085–2089. doi: 10.1212/01.WNL.0000165998.73779.D9. [DOI] [PubMed] [Google Scholar]
- 30.Xu HN, Nioka S, Glickson JD, Chance B, Li LZ. Quantitative mitochondrial redox imaging of breast cancer metastatic potential. J Biomed Opt. 2010;15(3):036010. doi: 10.1117/1.3431714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xu HN, Kadlececk S, Profka H, et al. Is higher lactate generation rate an indicator of tumor metastatic risk? - A pilot study using hyperpolarized 13C-NMR. Proceedings of the Annual Meeting of the International Society of Magnetic Resonance in Medicine. 2013:701. [Google Scholar]
- 32.Xu HN, Kadlececk S, Pullinger B, et al. In vivo metabolic evaluation of breast tumor mouse xenografts for predicting aggressiveness using the hyperpolarized 13C-NMR technique. Adv Exp Med Biol. 2013;789:237–242. doi: 10.1007/978-1-4614-7411-1_32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li LZ, Kadlececk S, Xu HN, et al. Ratiometric analysis in hyperpolarized NMR (I): test of the two-site exchange model and the quantification of reaction rate constants. NMR in Biomedicine. 2013 doi: 10.1002/nbm.2953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Day SE, Kettunen MI, Gallagher FA, et al. Detecting tumor response to treatment using hyperpolarized 13C magnetic resonance imaging and spectroscopy. Nature medicine. 2007;13(11):1382–1387. doi: 10.1038/nm1650. [DOI] [PubMed] [Google Scholar]
- 35.Nelson SJ, Kurhanewicz J, Vigneron DB, et al. Metabolic Imaging of Patients with Prostate Cancer Using Hyperpolarized[1-13C]Pyruvate. Science translational medicine. 2013;5(198):198ra08. doi: 10.1126/scitranslmed.3006070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hussien R, Brooks GA. Mitochondrial and plasma membrane lactate transporter and lactate dehydrogenase isoform expression in breast cancer cell lines. Physiological genomics. 2011;43(5):255–264. doi: 10.1152/physiolgenomics.00177.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Annarao S, Thakur S. Book Quantification of lactate concentrations in orthotopic breast tumors with different growth rates. City. 2012. Quantification of lactate concentrations in orthotopic breast tumors with different growth rates. [Google Scholar]
- 38.Robey IF, Lien AD, Welsh SJ, Baggett BK, Gillies RJ. Hypoxia-inducible factor-1alpha and the glycolytic phenotype in tumors. Neoplasia. 2005;7(4):324–330. doi: 10.1593/neo.04430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sakamoto T, Niiya D, Seiki M. Targeting the Warburg effect that arises in tumor cells expressing membrane type-1 matrix metalloproteinase. The Journal of biological chemistry. 2011;286(16):14691–14704. doi: 10.1074/jbc.M110.188714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Artemov D, Bhujwalla ZM, Glickson JD. In vivo selective measurement of (1-13C)-glucose metabolism in tumors by heteronuclear cross polarization. Magn Reson Med. 1995;33(2):151–155. doi: 10.1002/mrm.1910330202. [DOI] [PubMed] [Google Scholar]
- 41.Gillies RJ, Robey I, Gatenby RA. Causes and consequences of increased glucose metabolism of cancers. Journal of nuclear medicine : official publication, Society of Nuclear Medicine. 2008;49(Suppl 2):24S–42S. doi: 10.2967/jnumed.107.047258. [DOI] [PubMed] [Google Scholar]
- 42.Li LZJ, Zhou R, Zhong T, et al. Predicting melanoma metastatic potential by optical and magnetic resonance imaging. Adv Exp Med Biol. 2007;599:67–78. doi: 10.1007/978-0-387-71764-7_10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Li LZ, Zhou R, Xu HN, et al. Quantitative magnetic resonance and optical imaging biomarkers of melanoma metastatic potential. Proceedings of the National Academy of Sciences of the United States of America. 2009;106(16):6608–6613. doi: 10.1073/pnas.0901807106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xu HN, Feng M, Moon L, Dolloff N, El-Deiry W, Li LZ. Redox imaging of the p53-dependent mitochondrial redox state in colon cancer ex vivo. J Innov Opt Health Sci. 2013:1350016. doi: 10.1142/S1793545813500168. 0(0) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xu HN, Nioka S, Chance B, Li LZ. Heterogeneity of mitochondrial redox state in premalignant pancreas in a PTEN null transgenic mouse model. Adv Exp Med Biol. 2011;701:207–213. doi: 10.1007/978-1-4419-7756-4_28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Xu HN, Nioka S, Li LZ. Imaging heterogeneity in the mitochondrial redox state of premalignant pancreas in the pancreas-specific PTEN-null transgenic mouse model. Biomarker Reseaerch. 2013;1:6. doi: 10.1186/2050-7771-1-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xu HN, Tchou J, Chance B, Li LZ. Imaging the redox states of human breast cancer core biopsies. Adv Exp Med Biol. 2013;765:343–349. doi: 10.1007/978-1-4614-4989-8_48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Xu HN, Tchou J, Li LZ. Redox imaging of human breast cancer core biopsies: a preliminary investigation. Academic radiology. 2013;20(6):764–768. doi: 10.1016/j.acra.2013.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Li LZ. Imaging mitochondrial redox potential and its possible link to tumor metastatic potential. J Bioenerg Biomembr. 2012;44:645–653. doi: 10.1007/s10863-012-9469-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Witney TH, Kettunen MI, Day SE, et al. A comparison between radiolabeled fluorodeoxyglucose uptake and hyperpolarized (13)C-labeled pyruvate utilization as methods for detecting tumor response to treatment. Neoplasia. 2009;11(6):574–582. doi: 10.1593/neo.09254. 1 p following 82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lodi A, Woods SM, Ronen SM. Treatment with the MEK inhibitor U0126 induces decreased hyperpolarized pyruvate to lactate conversion in breast, but not prostate, cancer cells. NMR Biomed. 2013;26(3):299–306. doi: 10.1002/nbm.2848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Harris T, Eliyahu G, Frydman L, Degani H. Kinetics of hyperpolarized 13C1-pyruvate transport and metabolism in living human breast cancer cells. Proceedings of the National Academy of Sciences of the United States of America. 2009;106(43):18131–18136. doi: 10.1073/pnas.0909049106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Halestrap AP. The monocarboxylate transporter family--Structure and functional characterization. IUBMB life. 2012;64(1):1–9. doi: 10.1002/iub.573. [DOI] [PubMed] [Google Scholar]
- 54.Witney TH, Kettunen MI, Brindle KM. Kinetic modeling of hyperpolarized 13C label exchange between pyruvate and lactate in tumor cells. The Journal of biological chemistry. 2011;286(28):24572–24580. doi: 10.1074/jbc.M111.237727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Slomiany MG, Grass GD, Robertson AD, et al. Hyaluronan, CD44, and emmprin regulate lactate efflux and membrane localization of monocarboxylate transporters in human breast carcinoma cells. Cancer research. 2009;69(4):1293–1301. doi: 10.1158/0008-5472.CAN-08-2491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Gallagher SM, Castorino JJ, Wang D, Philp NJ. Monocarboxylate transporter 4 regulates maturation and trafficking of CD147 to the plasma membrane in the metastatic breast cancer cell line MDA-MB-231. Cancer research. 2007;67(9):4182–4189. doi: 10.1158/0008-5472.CAN-06-3184. [DOI] [PubMed] [Google Scholar]
- 57.Weber G. Carbohydrate metabolism in cancer cells and the molecular correlation concept. Die Naturwissenschaften. 1968;55(9):418–429. doi: 10.1007/BF00602650. [DOI] [PubMed] [Google Scholar]