Abstract
Glycerol-3-phosphate acyltransferase-1 is the first rate limiting step in de novo glycerophospholipid synthesis. We have previously demonstrated that GPAT-1 deletion can significantly alter T cell function resulting in a T cell phenotype similar to that seen in aging. Recent studies have suggested that changes in the metabolic profile of T cells are responsible for defining specific effector functions and T cell subsets. Therefore, we determined whether T cell dysfunction in GPAT-1 −/− CD4+ T cells could be explained by changes in cellular metabolism. We show here for the first time that GPAT-1 −/− CD4+ T cells exhibit several key metabolic defects. Striking decreases in both the oxygen consumption rate (OCR) and the extracellular acidification rate (ECAR) were observed in GPAT-1 −/− CD4+ T cells following CD3/CD28 stimulation indicating an inherent cellular defect in energy production. In addition, the spare respiratory capacity (SRC) of GPAT-1 −/− CD4+ T cells, a key indicator of their ability to cope with mitochondrial stress was significantly decreased. We also observed a significant reduction in mitochondrial membrane potential in GPAT-1 −/− CD4+ T cells compared to their WT counterparts, indicating that GPAT-1 deficiency results in altered or dysfunctional mitochondria. These data demonstrate that deletion of GPAT-1 can dramatically alter total cellular metabolism under conditions of increased energy demand. Furthermore, altered metabolic response following stimulation may be the defining mechanism underlying T cell dysfunction in GPAT-1 −/− CD4+ T cells. Taken together, these results indicate that GPAT-1 is essential for the response to the increased metabolic demands associated with T cell activation.
Introduction
Glycerol 3-phosphateacyltransferase-1 [GPAT-1] is an integral mitochondrial membrane protein responsible for conjugating fatty acyl-CoA with glycerol-3 phosphate in the first rate limiting step of de novo glycerophospholipid synthesis [1]. GPAT-1 catalyzes the conversion of glycerol-3 phosphate and acyl-CoA to lyosphosphatidic acid (LPA), which is then further acylated to phosphatidic acid, which subsequently serves as a precursor for all glycerophospholipid (GPL) and triglyceride synthesis. The acyl-CoA pool used by GPAT-1 can also be processed by carnitine palmitoyltransferase-1 (CPT1) for transport into the mitochondria for β-oxidation since both proteins are located in the mitochondria. GPAT-1 and CPT-1 compete for acyl-CoAs thereby playing a role in dictating whether a fatty acid is used for energy production (oxidation) or synthesis of more complex lipids (GPL and triglyceride). Therefore, it is not surprising that CPT-1 and GPAT-1 are sensitive to nutrient levels within the cell, specifically the ATP/AMP ratio [2]. AMP activated protein kinase (AMPK) activity increases when there is an abundance of AMP within the cell, signaling that ATP levels are low. Consequently, activated AMPK regulates both CPT-1 and GPAT-1 reciprocally. When cellular energy stores are low, AMPK is activated and down-regulates GPAT-1 activity, while promoting CPT-1 activity [2,3]. We have previously shown that GPAT-1 activity is up-regulated following T cell stimulation and that protein kinase C-theta (PKCθ) can directly activate T cell GPAT-1 [4]. Interestingly, we also found that stimulation induced up-regulation of GPAT-1 activity is significantly blunted in aged T cells, suggesting that T cell dysfunction with age may be at least partly attributed to altered cellular GPL levels.
Quiescent T cells must rapidly upregulate modes of energy production in response to stimulation by antigen in order to drive clonal expansion and cytokine production [5]. Immunometabolism is emerging as a key regulator of both T cell fate and function. Canonically, it is thought that this process primarily engages glycolytic pathways of energy production. However, evidence is emerging that the preferred energy substrate depends on the T cell subset in question. For example, it was recently shown that Treg subsets exhibit a metabolic preference for lipid oxidation while Th1 and Th2 subsets rely heavily on glycolysis and Th17 subsets engage both lipid and glycolytic pathways [6]. In addition, rapamycin treatment or fatty acid addition alone can enhance Treg differentiation while blocking lipid oxidation via etomoxir prevented Treg generation [6]. In another study, CD8+ memory T cells have recently been shown to possess substantially more spare respiratory capacity than CD8+ T effector cells, thereby conferring the ability to respond to increased stress and promote long term survival [7]. To date, the role of lipid metabolism on T cell function has focused primarily on fatty acid oxidation with little attention given to the role that lipid biosynthesis may play in dictating T cell functional phenotype.
In the current study, we examined how GPAT-1 deficiency alters CD4+ T cell metabolism and whether these changes may underlie T cell dysfunction. We detected a small but significant decrease in mitochondrial membrane potential from CD4+ T cells isolated from GPAT-1 knock out (KO) mice as compared to age matched controls. Although unstimulated GPAT-1 KO CD4+ T cells appeared to be metabolically equivalent to their WT counterparts, following stimulation, gross deficiencies in both respiratory and glycolytic metabolism in GPAT-1 KO CD4+ T cells were observed. These findings suggest that CD4+ T cells deficient in GPAT-1 are unable to meet the high energy demands concomitant with stimulation and shift their available metabolic resources for the purpose of promoting cell survival.
3.3 Materials and Methods
Mice and T cell isolation/stimulation
Male C57BL/6 GPAT-1 KO mice were obtained from Dr. Rosalind Coleman (University of North Carolina Chapel Hill) and bred at the UT Austin animal facilities. Age matched male C57BL/6 mice were obtained from Charles River. At 5 months of age, mice were sacrificed and isolated spleens gently homogenized and single cell suspensions obtained. Cell suspensions were stained with Miltenyi anti CD4 (L3T4) coated microbeads and magnetically separated using a Miltenyi LS column. Unlabeled cells were washed through the column and CD4+ T cells were collected by removing the column from the magnetic field and forcing 5 mL of buffer through the column with a plunger. Typically, 1.3–1.5×107 viable CD4+ T cells were obtained from each mouse spleen as assessed by Trypan blue staining. These numbers did not significantly differ between WT and GPAT-1 KO mice. Freshly isolated CD4+ T-cells were plated at a density of 3×106 cells/mL and stimulated at 37°C for 20 hours in complete RPMI 1640 culture media (10% heat-inactivated fetal bovine serum plus 100 U/ml penicillin, 100 µg/ml streptomycin, 10 µM β-mercaptoethanol, and 100 mM L-glutamine) with 10 µg/ml plate-bound anti-CD3 and 1 µg/ml soluble anti-CD28 or left unstimulated.
Mitochondrial quantification
Freshly isolated T cells were incubated at 37°C with 100 nM Mitotracker Red for 30 minutes. Cells were then washed twice and resuspended in 1 mL of PBS. Cells were then analyzed using an Accuri C6 Flow cytometer and analysis carried out with Accuri C6 software.
BrdU Incorporation and CD4 staining
T cells were isolated and pooled from WT or GPAT-1 KO mice. Proliferation was measured using the BD biosciences APC BrdU Flow Kit. For stimulated conditions, 96 well plates were coated with 10 µg/ml of CD3 antibody in NaHCO3 buffer and incubated for 4 hours at 37°C. Subsequently, 1 mM BrdU diluted in PBS and 1 µg/mL of CD28 antibody was added to cultures in complete media at a density of 106 cells/mL and incubated at 37°C for 20 hours. We chose 20 hours and not longer stimulation times because we wanted to examine the influence of GPAT on the early events that lead to the first round of cell division as opposed to examining sustained proliferation at later time points. For unstimulated conditions, non-CD3 coated plates were used and CD28 antibody was not added. Following incubation, cells were fixed and permeabilized according to the manufacturer’s protocol and APC fluorescence was measured by flow cytometry.
Protocol optimization for Seahorse Extracellular Flux Analyzer
We determined that 500,000 murine T cells were needed to ensure oxygen consumption (OCR) and extracellular acidification rate (ECAR) readings fell within an interpretable range (data not shown). We next experimentally determined optimal concentrations of the mitochondrial inhibitors, oligomycin, carbonyl cyanide-4-(trifluoromethoxy) phenylhydrazone (FCCP), and rotenone using those reported previously for CD8+ T cells as a starting point [7]. Optimal concentrations were defined as those that gave replicable responses within the dynamic range of the Seahorse instrument. Dose response analysis with various inhibitor concentrations revealed the optimal combination of inhibitor concentrations to be: 2µM oligomycin, 3µM FCCP, and 4µM rotenone (data not shown). Prior to each assay, inhibitors were made fresh from frozen stocks and aliquoted into the microinjection ports of the XF-24 flux plate. We next determined the optimal palmitic acid/BSA conjugate concentration for the fatty acid oxidation assay in order to yield a discernible difference in OCR. Prior to each assay, freshly prepared palmitic acid/BSA conjugate or BSA only (control) were aliquoted into the microinjection port of the XF-24 flux plate.
Metabolic profiling
T cells were isolated and cultured as described above. After 20 hours, cells were harvested and washed in 5 mL of pre-warmed Seahorse XF assay medium and resuspended to a concentration of 10 × 106 cells/mL and 50 µL of this suspension was added to poly-l lysine coated dishes for a final count of 500,000 cells/well, and incubated at 37°C for 30 minutes in a non-CO2 incubator. XF assay medium (450 µL) was added to each well and the metabolic profile assessed using the XF-24 Extracellular Flux Analyzer (Seahorse Bioscience).
Fatty Acid oxidation profile
T cells were isolated and cultured as described above. After 20 hours, T cells were harvested and washed in 5 mL of pre-warmed minimal KHB assay medium (111 mM NaCl, 4.7 mM KCl, 2 mM MgSO4, 1.2 mM Na2HPO4, 2.5 mM glucose, and 0.5mMcarnitine) and resuspended to a concentration of 10 × 106 cells/mL. Approximately 50 µL of this suspension was added to a poly-l lysine coated24 well dish for a final count of 500,000 cells/well and incubated at 37°C for 30 minutes in a non-CO2 incubator. KHB assay medium (450µL) was added to each well and the metabolic profile assayed using the XF-24 Extracellular Flux Analyzer (Seahorse Bioscience).
Statistical analyses
Statistical significance was determined using a student’s T test or one-way analysis of variance due to the normal distribution of data. Treatment effects were considered significant when P <0.05. Post hoc analysis was conducted using Tukey’s multiple-comparison test with GraphPad Prism (San Diego, CA, USA).
Results
Proliferative response of CD4+ and CD8+ T cells
Since GPAT-1 deficiency may result in a loss of mitochondrial redox potential, we sought to assess proliferation by BrdU incorporation. To control for heterogeneities that can occur with multiple T cell isolations and rounds of stimulation, we pooled T cells from 10 WT or 10 GPAT-1 KO age matched mice. Total and CD4+ GPAT-1 KO T cells incorporated approximately 10% and 11% less BrdU than their WT counterparts, respectively (Figure S1). Furthermore, there appeared to be no difference in BrdU incorporation between WT and GPAT-1 KO CD8+ T cells (data not shown).
Metabolic profiles of WT and GPAT-1 KO CD4+ T cells
CD4+ T cells were isolated and stimulated with CD3/CD28 antibodies for 20 hours in complete RPMI media after which cells were harvested and plated onto poly-l lysine coated 24 well plates and their metabolic profiles assayed. Under basal conditions, stimulated GPAT-1 KO CD4+ T cells exhibited significantly (P<0.05) less respiratory and glycolytic activity than WT CD4+ T cells as determined by their respective oxygen consumption rate (OCR) and extracellular acidification rates (ECAR) (OCR: Figure 1, ECAR: Figure 2). In contrast, there was no significant (P<0.05) difference between the basal respiratory metabolic profile of unstimulated GPAT-1 KO and WT T cells. (Figure S2).
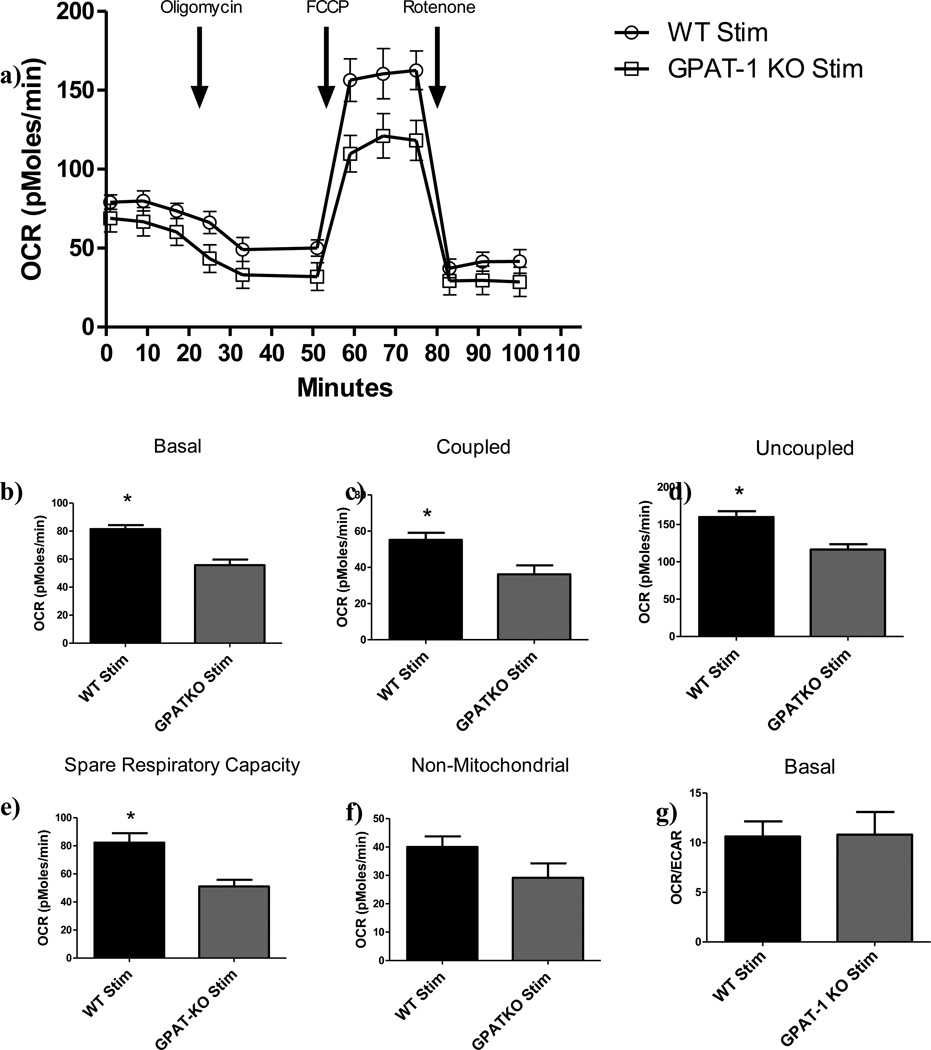
Figure 1. Respiratory response and spare respiratory capacity in CD4+ T cells.
CD4+ T cells were isolated from age matched WT or GPAT-1 KO mice stimulated with CD3/CD28 antibodies in complete medium. a) Cells were harvested and plated onto poly-l lysine coated dishes and basal oxygen consumption rate was analyzed followed by time specific injection of oligomycin, FCCP, and rotenone. Each point represents the mean +/− the SEM of five individual mice in triplicate. The data in a) were used to calculate: b)Basal OCR; c) Coupled OCR, following oligomycin treatment; d) Uncoupled OCR following FCCP Treatment; e) Spare respiratory capacity, calculated by subtracting uncoupled from basal OCR; f) Non-mitochondrialmetabolism following rotenone treatment; and g) Basal OCR/ECAR. Each bar represents the mean +/− SEM of five individual mice, in triplicate (for inhibitor addition) or sextuplet (for basal readings), and averaged across three time points. *Significantly different (p<0.05). Significance was determined using two-tailed student’s T- test. N=5.
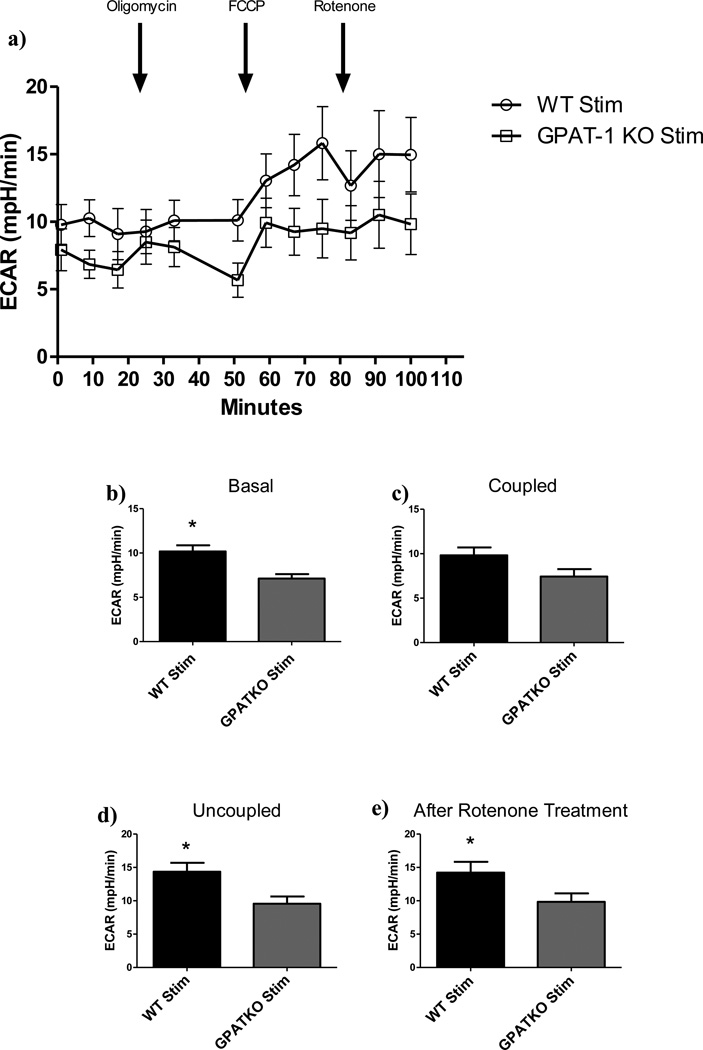
Figure 2. Glycolytic response in CD4+ T cells.
a) CD4+ T cells were isolated from age matched WT or GPAT-1 KO mice and stimulated with CD3/CD28 antibodies in complete medium. Cells were harvested and plated onto poly-l lysine coated dishes and basal oxygen consumption rate was analyzed followed by timed injection of oligomycin, FCCP, and rotenone. Each point represents the mean +/− the SEM of five individual mice in triplicate on the plate. The data in a) was used to calculate glycolytic response: b)Basal; c) Coupled respiration following oligomycin treatment; d); Uncoupled respiration following FCCP treatment; e) Following rotenone treatment. Each bar represents the mean +/− SEM of five individual mice in triplicate (for inhibitor addition) or sextuplet (for basal readings), and averaged across three time points. *Significantly different (p<0.05). Significance was determined using two-tailed student’s T- test. N=5.
Injection of oligomycin, an ATP synthase inhibitor, followed by intermittent metabolic monitoring over a 24 minute time frame revealed a proportional decrease in OCR and ECAR in stimulated GPAT-1 KO CD4+ T cells compared to WT controls, indicating a blunted metabolic profile for the GPAT-1 KO (OCR: Figure 1b, ECAR: Figure 2b). Likewise, treatment with FCCP, an uncoupler, resulted in decreased maximal respiratory capacity (Figure 1c) and ECAR (Figure 2c) in stimulated GPAT-1 KO T cells. When maximal respiration was subtracted from basal respiratory levels in stimulated samples, GPAT-1 KO CD4+ T cells exhibited a significantly reduced spare respiratory capacity (SRC) (Figure 1e).These findings suggest the presence of an inherent mitochondrial defect reducing the ability to up-regulate energy production in response to stimulation. In complementary experiments, exposure to rotenone, an electron transport inhibitor [8], was used to measure non-mitochondrial respiration. No significant differences between GPAT-1 KO CD4+ T cells and WT were detected (OCR: Figure 1f) following rotenone treatment. However, ECAR was decreased in stimulated GPAT-1 KO CD4+ T cells as compared to the stimulated WT (ECAR: Figure 2e). These data suggest that decreased oxygen consumption rates observed in the GPAT-1 KO are the result of an inability to up-regulate mitochondrial respiratory metabolism in response to stimulation.
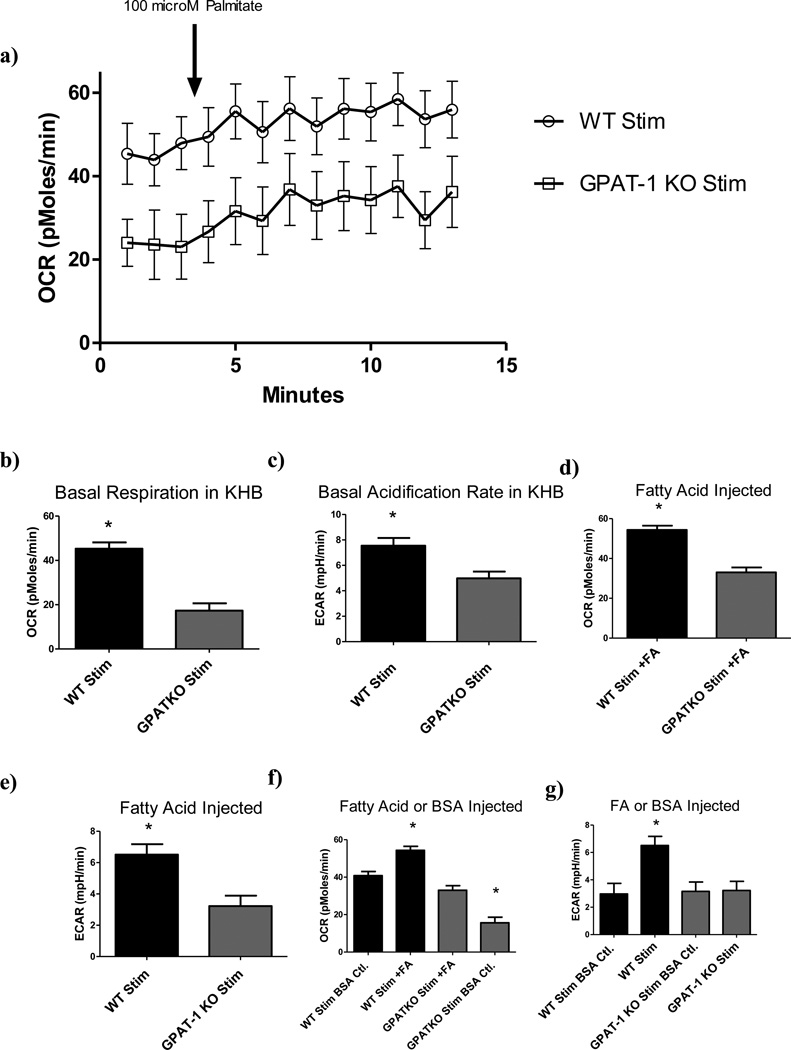
We have previously reported elevated CPT-1a protein levels in GPAT-1 KO thymocytes [9], supporting the hypothesis that loss of GPAT-1 may increase fatty acid oxidation. Therefore, in the current study we sought to determine whether loss of GPAT-1 in peripheral CD4+ T cells also correlated with increased lipid oxidation rates. Basal readings were acquired for 24 minutes followed by injection of 100 µM palmitate/BSA conjugate and intermittent monitoring of OCR and ECAR for 80 minutes in WT and GPAT-1 KO CD4+ T cells (Figure 3a). Basal OCR and ECAR readings differed significantly between stimulated GPAT-1 KO and WT T cells (Figures S3a, 3b, and 3c). Interestingly, no difference in OCR was observed between stimulated GPAT-1 KO and unstimulated WT controls (Figure S3b), implying that cell activation is unable to overcome the metabolic defect in GPAT-1 KO T cells in minimal KHB media. Following palmitate/BSA injection, OCR readings for stimulated GPAT-1 KO T cells were significantly reduced compared to WT (Figure 3d). ECAR readings following palmitate /BSA injection were significantly different between all samples tested (Figure 3e).
Figure 3. Fatty acid oxidation respiratory response in CD4+ T cells.
a) CD4+ T cells were isolated from age matched WT or GPAT-1 KO mice and stimulated with CD3/CD28 antibodies in complete medium. Cells were harvested, transferred to KHB assay specific media and plated onto poly-l lysine coated dishes and basal oxygen consumption rate was analyzed followed by injection of 100 µM fatty acid (palmitic acid/BSA conjugate). Each point represents the mean +/− the SEM of five individual mice in triplicate. The data in panel a) was used to calculate respiratory response following: b) Basal OCR (average of 3 time points preceding injection); c) Basal ECAR (average of 3 time points preceding injection); d) OCR for FA injected (average of 10 time points following injection);e)ECAR for FA injected (average of 6 time points following injection); f) OCR for FA injected compared to BSA only injected controls (average of 10 time points following injection); g) ECAR for FA injected compared to BSA only injected controls (average of 6 time points following injection). Each bar represents the mean +/− SEM of five individual mice, in quintuplet (for basal readings) or triplicate (for FA injected) or duplicate (for BSA only injected), and averaged across multiple time points as indicated above. * Significantly different (p<0.05). Significance was determined using two-tailed student’s T- test or one-way ANOVA and Tukeys post-hoc analysis. N=5.
FA/BSA injection also had the unexpected effect of increasing ECAR in stimulated samples (Figure 3g). FA/BSA injection significantly increased ECAR readings in stimulated WT samples compared to BSA injected alone, while no increase was observed in GPAT-1 KO samples.
CD4+ T cell mitochondrial membrane potential
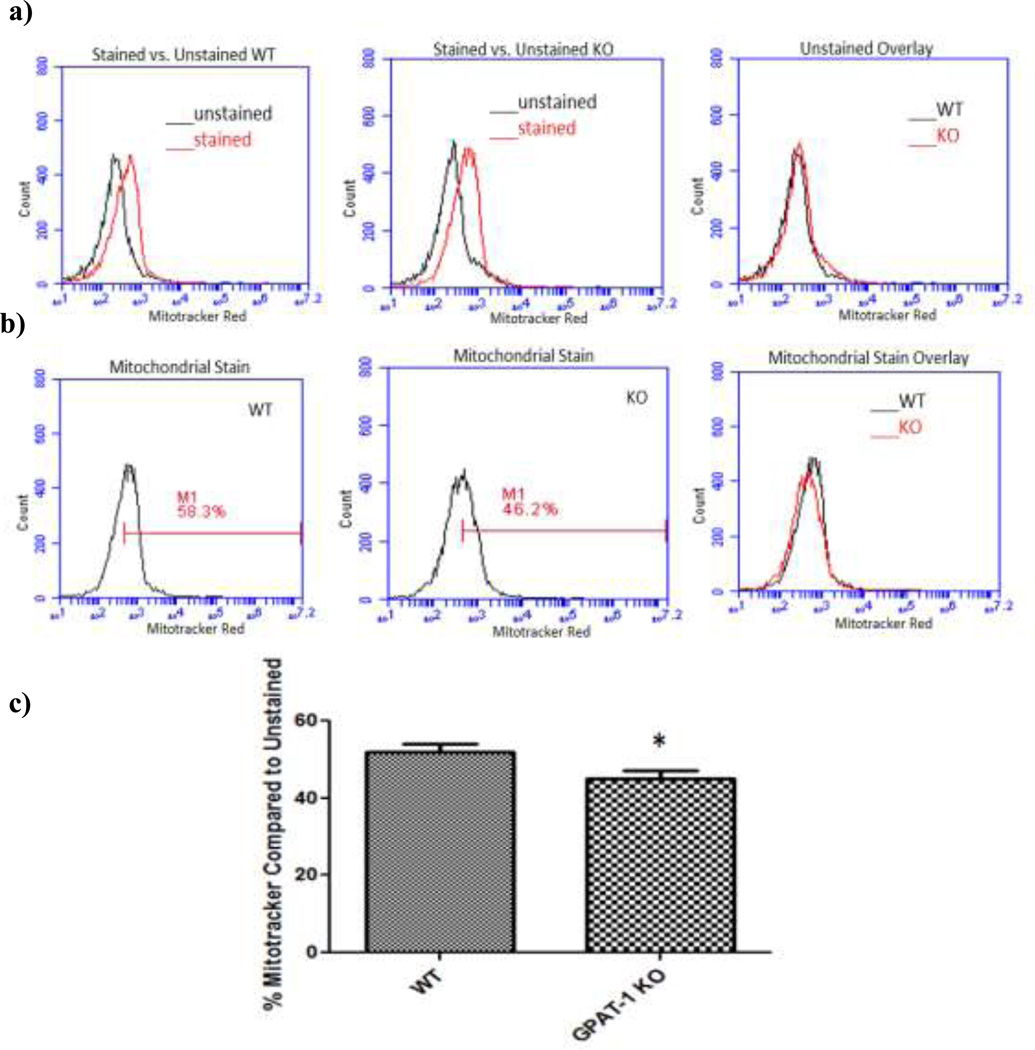
Total mitochondrial membrane potential was assessed using Mitotracker Red staining. GPAT-1 KO CD4+ T cells exhibited a small but significant (p<0.05) decrease in Mitotracker Red accumulation compared to WT controls (Figure 4). This difference could be the result of reduced mitochondrial number, smaller mitochondria, or dysfunctional mitochondria.
Figure 4. GPAT-1 KO mice accumulate less Mitotracker Red than their WT counterparts.
Freshly isolated WT and GPAT-1 KO T cells were stained with 100 nM mitotracker red and fluorescence was analyzed by flow cytometry. a) and b) are representative histograms depicting stained and unstained cells; c) fluorescence intensity of stained T cells compared to unstained cells. Values represent the mean +/− SEM of five individual mice.*Significantly different (p<0.05). Significance was determined using two-tailed student’s T- test. N=5.
Discussion
Conventional thought is that activated T cells become heavily reliant on glycolysis following stimulation and switch to an “anabolic” program of metabolism that supports clonal expansion even in the presence of adequate oxygen to support mitochondrial oxidative phosphorylation (OXPHOS) [10,11]. However, the exact role of mitochondrial OXPHOS during T cell stimulation remains unclear. Our data clearly show that CD4+ T cells up regulate both OXPHOS and glycolysis in response to stimulation. Furthermore, the OCR/ECAR ratio is significantly increased in response to stimulation demonstrating that OXPHOS is indeed significantly up-regulated in CD4+ T cells. In contrast, unstimulated T cells appear to rely primarily on glycolysis.
No difference in the respiratory profile of unstimulated WT and GPAT-1 KO CD4+ T cells was observed (Figure S2), suggesting that T cell metabolic dysfunction in the absence of GPAT-1 only becomes apparent under conditions of increased energy demand. The lack of a compensatory increase in glycolysis suggests that the GPAT-1 KO CD4+ T cells do not respond appropriately to stimulation and thus cannot engage normal modes of metabolism. Furthermore, a failure to up-regulate glycolysis implies that GPAT-1 KO CD4+ T cells suffer from a severe energy sink, which may contribute to the decreased IL-2 producing phenotype we have previously reported [12]. Collectively, the decrease in SRC observed in the GPAT-1 KO T cell (Figure 1e) is indicative of an inherent defect in the mitochondrial stress response, more so than a proportional decrease in the metabolic profile. It is therefore likely that GPAT-1 deficiency impacts T cell metabolic function at multiple levels.
Our data demonstrate that GPAT-1 is necessary for regulating the increased energy demand placed on T cells following stimulation. In the absence of stimulation we observed no general difference in the metabolic profile of GPAT-1 KO CD4+ T cells compared to the WT counterpart (Figure S2), suggesting that GPAT-1 dependent lipid biosynthesis plays a critical role in regulating the stimulation induced metabolic switch. One possible explanation is that GPAT-1 KO T cells have smaller or fewer mitochondria as indicated in (Figure 4), a consequence of which is decreased respiratory capability. However, it is more likely that GPAT-1 deficiency results in compromised mitochondria rather than physically fewer mitochondria in light of observations in the GPAT-1 KO liver showing increased susceptibility to Ca2+ induced opening of the mitochondrial permeability transition pore (MPTP) [13]. Since substantial increases in intracellular Ca2+ are necessary for activation of critical factors responsible for driving cytokine production and pro proliferative signaling (reviewed in [14]), it is possible that GPAT-1 plays a critical role in maintaining mitochondrial integrity, most likely by providing a source of GPL for mitochondrial membranes.
In addition to direct effects on mitochondrial function, GPAT-1 may also induce changes in plasma membrane phospholipid composition which are thought to interfere with proper activation of membrane based signaling complexes as seen with age [15]. It is noteworthy that GPAT-1 KO T cells have significantly reduced levels of nearly all of the major GPLs, and analysis of molar lipid mass per cell makes it unlikely that this reduction in GPL is solely due to altered mitochondrial membrane composition. Instead, it is more probable that GPAT-1 deficiency results in decreased GPL mass in all cellular membranes. Defects in cellular membranes are capable of interfering with membrane based signal transduction such as that associated with T cell stimulation through CD3 and the CD28 co receptor which mimics the in vivo antigenic stimulation. This could then lead to a defect in transmission of the stimulatory signal.
We have previously shown that GPAT-1 KO T cells have a dysfunctional phenotype similar to aged cells, which are characterized by decreased GPL to cholesterol ratios, decreased IL-2 production, and a pro-inflammatory polarization of T cell subsets [12,16]. Therefore, the reduced GPAT-1 activity in T-cells from aged rats suggests that life span related decline in GPAT-1 activity may be a primary contributor to immunosenesence through a mechanism mediated by alterations in GPL [17]. Decreased GPL to cholesterol ratios are thought to be a principle contributor to decreased membrane based signal transduction in senescent cells as described by the “membrane gate” theory of aging [15]. Interestingly, we have also shown that GPAT-1 KO T cells secrete increased levels of IL-17 [16], a proinflammatory cytokine associated with increased autoimmunity in aging. In addition, these mice exhibit elevated levels of CD4+CD25+FoxP3+ T cells (Treg) (unpublished observation), a phenotype also observed in aging [18][19]. Elevated IL-17 secretion in combination with elevated Treg is highly significant based on the fact that in the absence of TGF-β, Treg are capable of polarizing CD4+ T cells toward a Th17 phenotype [20]. In addition, GPAT-1 KO mice are more susceptible to coxsackie virus B3 infection which has been directly linked to dysfunctional T cells, suggesting that the in vivo adaptive immune response is indeed impacted by ex vivo dysfunction [21].
Aged T cells are also more susceptible to the damaging effects of oxidative stress which has been linked to sensitization of mitochondria and reduced spare respiratory capacity [22]. Increased oxidative stress is thought to contribute significantly to mitochondrial DNA mutations with age and has been suggested to play a role in degenerative disease and cancer development [23]. This is important in the context of GPAT-1 for three key reasons. First, GPAT-1 is an integral mitochondrial membrane protein and its activity may serve to locally supply the mitochondria with lysophosphatidic acid for glycerophospholipid synthesis, especially cardiolipin. Therefore, loss of GPAT-1 may structurally alter mitochondrial integrity and compromise mitochondrial membrane potential resulting in T cell unresponsiveness. We have previously shown that the mitochondrial membrane lipid; cardiolipin, found exclusively in the mitochondria, is decreased in T-cells of GPAT-1 KO mice in addition to a global decrease in most major GPL [12]. Second, we have demonstrated that GPAT-1 KO CD4+ T cells are less proliferative as assessed by MTT assay (an assay measuring mitochondrial redox ability) and more prone to an “apoptosis like” program of cell death following stimulation [12]. These observations are supported by previous work examining GPAT-1 KO hepatocytes, where stimulation induced cell death was observed [13]. Third, mitochondria from GPAT-1 KO hepatocytes lose mitochondrial outer membrane potential (MOMP) rapidly and form extensive mitochondrial permeability transition pores (MPTP) in response to addition of exogenous Ca2+, suggesting that GPAT-1 plays a critical role in regulating mitochondrial integrity [13]. It is also noteworthy that MPTP formation is significantly increased following TCR stimulation in old murine T-cells [24]. These observations construct a scenario in which stimulation of the T cell increases intracellular Ca2+ levels inducing MPTP formation and subsequent loss of MOMP. Since both GPAT-1 activity and cardiolipin content are reduced with aging, this mechanism may be a major contributing factor to mitochondrial mediated immunosenescence.
With respect to beta-oxidation, addition of palmitate as a fuel source, even in the presence of glucose, significantly increased mitochondrial oxygen consumption (Figure 3f). This effect was demonstrated in both stimulated and unstimulated cells. This observation may be explained by a recent report showing that in skeletal muscle cells, palmitate treatment rapidly up-regulates GLUT4 expression and stimulates glucose uptake [25]. To the best of our knowledge this is the first time that a similar phenomenon has been observed in primary murine T cells and may be an important mechanism by which diseases linked to high fat content, like obesity, may trigger a proinflammatory T cell response.
In summary, the data presented here show that CD4+ T cells deficient in GPAT-1 have a blunted metabolic profile and reduced SRC as compared to their WT counterparts. In addition, these defects are only apparent when the T cell is stimulated, suggesting that GPAT-1 is necessary to meet the increased energy demands placed on the cell following stimulation. In light of our previously reported finding that GPAT-1 activity is reduced in aged rats [17], it is possible that T cell dysfunction associated with aging can be at least partly attributed to an inherent loss of GPAT-1 activity. Thus, modulation of GPAT-1 activity may emerge as a principle target in the treatment of T cell dysfunction as we age and/or in developing strategies to reduce immune mediate diseases.
Supplementary Material
Highlights.
GPAT-1 deletion can significantly alter T cell function.
We determined if GPAT-1 −/− CD4+ T cells have changes in cellular metabolism.
GPAT-1 −/− CD4+ T cells exhibit several key metabolic defects.
Altered metabolism may be key in GPAT-1 −/− CD4+ T cell dysfunction.
GPAT-1 is essential for the metabolic response to T cell activation.
Acknowledgements
This work was supported in part by NIH (CA159383) to CAJ.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Literature Cited
- 1.Igal RA, Wang S, Gonzalez-baró M, Coleman RA. Mitochondrial glycerol phosphate acyltransferase directs incorporation of exogenous fatty acids into triacylglycerol, J. Biol. Chem. 2001;276:42205–42212. doi: 10.1074/jbc.M103386200. [DOI] [PubMed] [Google Scholar]
- 2.Wendel Aa, Lewin TM, Coleman Ra. Glycerol-3-phosphate acyltransferases: rate limiting enzymes of triacylglycerol biosynthesis. Biochim. Biophys. Acta. 2009;1791(6):501–506. doi: 10.1016/j.bbalip.2008.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Winder WW, Thomson DM. Cellular energy sensing and signaling by AMP-activated protein kinase. Cell Biochem. Biophys. 2007 May;47(3):332–347. doi: 10.1007/s12013-007-0008-7. [DOI] [PubMed] [Google Scholar]
- 4.Collison LW, Jolly Ca. Phosphorylation regulates mitochondrial glycerol-3-phosphate-1 acyltransferase activity in T-lymphocytes. Biochim. Biophys. Acta. 2006;1761(1):129–139. doi: 10.1016/j.bbalip.2005.12.002. [DOI] [PubMed] [Google Scholar]
- 5.Frauwirth Ka, Thompson CB. Regulation of T lymphocyte metabolism. J. Immunol. 2004 Apr;172(8):4661–4665. doi: 10.4049/jimmunol.172.8.4661. [DOI] [PubMed] [Google Scholar]
- 6.Michalek RD, Gerriets Va, Jacobs SR, Macintyre AN, MacIver NJ, Mason EF, Sullivan Sa, Nichols AG, Rathmell JC. Cutting edge: distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J. Immunol. 2011 Mar;186(6):3299–3303. doi: 10.4049/jimmunol.1003613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.van der Windt GJW, Everts B, Chang C-H, Curtis JD, Freitas TC, Amiel E, Pearce EJ, Pearce EL. Mitochondrial respiratory capacity is a critical regulator of CD8+ T cell memory development. Immunity. 2012 Jan;36(1):68–78. doi: 10.1016/j.immuni.2011.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brand MD, Nicholls DG. Assessing mitochondrial dysfunction in cells. Biochem. J. 2011 Apr;435(2):297–312. doi: 10.1042/BJ20110162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gulvady Aa, Murphy EJ, Ciolino HP, Cabrera RM, Jolly Ca. Glycerol-3-phosphate acyltransferase-1 gene ablation results in altered thymocyte lipid content and reduces thymic T cell production in mice. Lipids. 2013 Jan;48(1):3–12. doi: 10.1007/s11745-012-3741-7. [DOI] [PubMed] [Google Scholar]
- 10.Wang T, Marquardt C, Foker J. Aerobic glycolysis during lymphocyte proliferation. Nature. 1976;261(5562):702–705. doi: 10.1038/261702a0. [DOI] [PubMed] [Google Scholar]
- 11.Pearce EL. Metabolism in T cell activation and differentiation. Curr. Opin. Immunol. 2010 Jun;22(3):314–320. doi: 10.1016/j.coi.2010.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Collison LW, Murphy EJ, Jolly Ca. Glycerol-3-phosphate acyltransferase-1 regulates murine T-lymphocyte proliferation and cytokine production. Am. J. Physiol. Cell Physiol. 2008;295(6):C1543–C1549. doi: 10.1152/ajpcell.00371.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hammond LE, Albright CD, He L, Rusyn I, Watkins SM, Doughman SD, Lemasters JJ, Coleman RA. Increased oxidative stress is associated with balanced increases in hepatocyte apoptosis and proliferation in glycerol-3-phosphate acyltransferase-1 deficient mice. Exp. Mol. Pathol. 2007;82:210–219. doi: 10.1016/j.yexmp.2006.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Smith-Garvin JE, Koretzky Ga, Jordan MS. T cell activation. Annu. Rev. Immunol. 2009;27:591–619. doi: 10.1146/annurev.immunol.021908.132706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yeo E-J, Park SC. Age-dependent agonist-specific dysregulation of membrane-mediated signal transduction: emergence of the gate theory of aging. Mech. Ageing Dev. 2002 Nov;123(12):1563–1578. doi: 10.1016/s0047-6374(02)00092-1. [DOI] [PubMed] [Google Scholar]
- 16.De Angulo A, Faris R, Cavazos D, Jolly C, Daniel B, Degraffenried L. Age-related alterations in T-lymphocytes modulate key pathways in prostate tumorigenesis. Prostate. 2013 Jun;73(8):855–864. doi: 10.1002/pros.22631. [DOI] [PubMed] [Google Scholar]
- 17.Collison LW, Kannan L, Onorato TM, Knudsen J, Haldar D, Jolly Ca. Aging reduces glycerol-3-phosphate acyltransferase activity in activated rat splenic T-lymphocytes. Biochim. Biophys. Acta. 2005;1687(1–3):164–172. doi: 10.1016/j.bbalip.2004.11.013. [DOI] [PubMed] [Google Scholar]
- 18.Lages CS, Suffia I, Velilla Pa, Huang B, Warshaw G, Hildeman Da, Belkaid Y, Chougnet C. Functional regulatory T cells accumulate in aged hosts and promote chronic infectious disease reactivation. J. Immunol. 2008 Aug;181(3):1835–1848. doi: 10.4049/jimmunol.181.3.1835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dominguez AL, Lustgarten J. Implications of aging and self-tolerance on the generation of immune and antitumor immune responses. Cancer Res. 2008;68(13):5423–5431. doi: 10.1158/0008-5472.CAN-07-6436. [DOI] [PubMed] [Google Scholar]
- 20.Xu L, Kitani A, Fuss I, Strober W. Cutting edge: regulatory T cells induce CD4+CD25-Foxp3- T cells or are self-induced to become Th17 cells in the absence of exogenous TGF-beta. J. Immunol. 2007 Jun;178(11):6725–6729. doi: 10.4049/jimmunol.178.11.6725. [DOI] [PubMed] [Google Scholar]
- 21.Karlsson EA, Wang S, Shi Q, Coleman RA, Beck MA. Glycerol-3-Phosphate Acyltransferase 1 Is Essential for the Immune Response to Infection with Coxsackievirus B3 in Mice 1, 2. Online. 2009;(16):779–783. doi: 10.3945/jn.108.101683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Desler C, Hansen TL, Frederiksen JB, Marcker ML, Singh KK, Juel Rasmussen L. Is There a Link between Mitochondrial Reserve Respiratory Capacity and Aging? J. Aging Res. 2012 Jan;2012:192503. doi: 10.1155/2012/192503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wallace DC. A mitochondrial paradigm of metabolic and degenerative diseases, aging, and cancer: a dawn for evolutionary medicine. Annu. Rev. Genet. 2005 Jan;39:359–407. doi: 10.1146/annurev.genet.39.110304.095751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Mather MW, Rottenberg H. The inhibition of calcium signaling in T lymphocytes from old mice results from enhanced activation of the mitochondrial permeability transition pore. Mech. Ageing Dev. 2002 Mar;123(6):707–724. doi: 10.1016/s0047-6374(01)00416-x. [DOI] [PubMed] [Google Scholar]
- 25.Pu J, Peng G, Li L, Na H, Liu Y, Liu P. Palmitic acid acutely stimulates glucose uptake via activation of Akt and ERK1/2 in skeletal muscle cells. J. Lipid Res. 2011 Jul;52(7):1319–1327. doi: 10.1194/jlr.M011254. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.