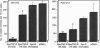Abstract
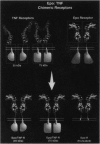
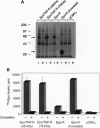
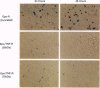
Many hormone and cytokine receptors are crosslinked by their specific ligands, and multimerization is an essential step leading to the generation of a signal. In the case of the tumor necrosis factor (TNF) receptors (TNF-Rs), antibody-induced crosslinking is sufficient to trigger a cytolytic effect. However, the quaternary structural requirements for signaling--i.e., the formation of dimers, trimers, or higher-order multimers--have remained obscure. Moreover, it has not been clear whether the 55-kDa or 75-kDa TNF-R is responsible for initiation of cytolysis. We reasoned that an obligate receptor dimer, targeted to the plasma membrane, might continuously signal the presence of TNF despite the actual absence of the ligand. Such a molecule, inserted into an appropriate vector, could be used to project receptor-specific "TNF-like" activity to specific cells and tissues in vivo. Accordingly, we constructed sequences encoding chimeric receptors in which the extracellular domain of the mouse erythropoietin receptor (Epo-R) was fused to the "stem," transmembrane domain, and cytoplasmic domain of the two mouse TNF-Rs. Thus, the Epo-R group was used to drive dimerization of the TNF-R cytoplasmic domain. These chimeric proteins were well expressed in a variety of cell lines and bound erythropoietin at the cell surface. Both the 55-kDa and the 75-kDa Epo/TNF-R chimeras exerted a constitutive cytotoxic effect detected by cotransfection or clonogenic assay. Thus, despite the lack of structural homology between the cytoplasmic domains of the two TNF-Rs, a similar signaling endpoint was observed. Moreover, dimerization (rather than trimerization or higher-order multimerization) was sufficient for elicitation of a biological response.
Full text
PDF
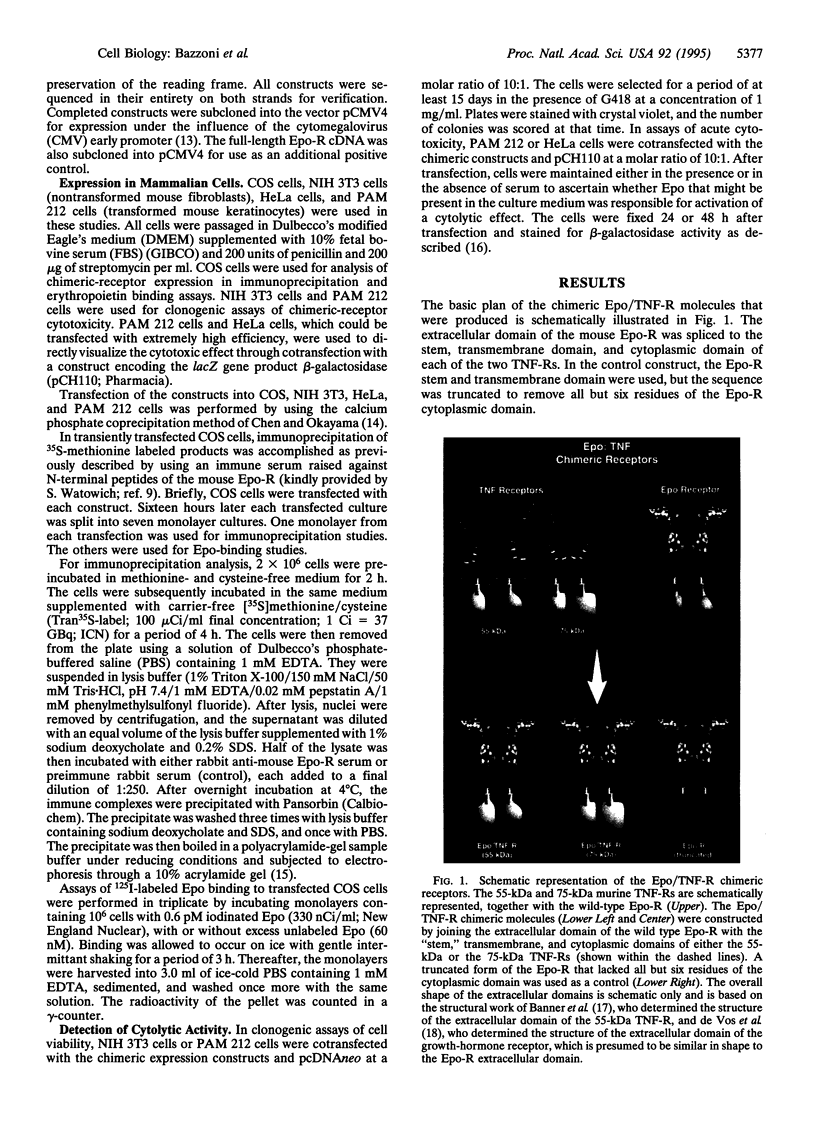
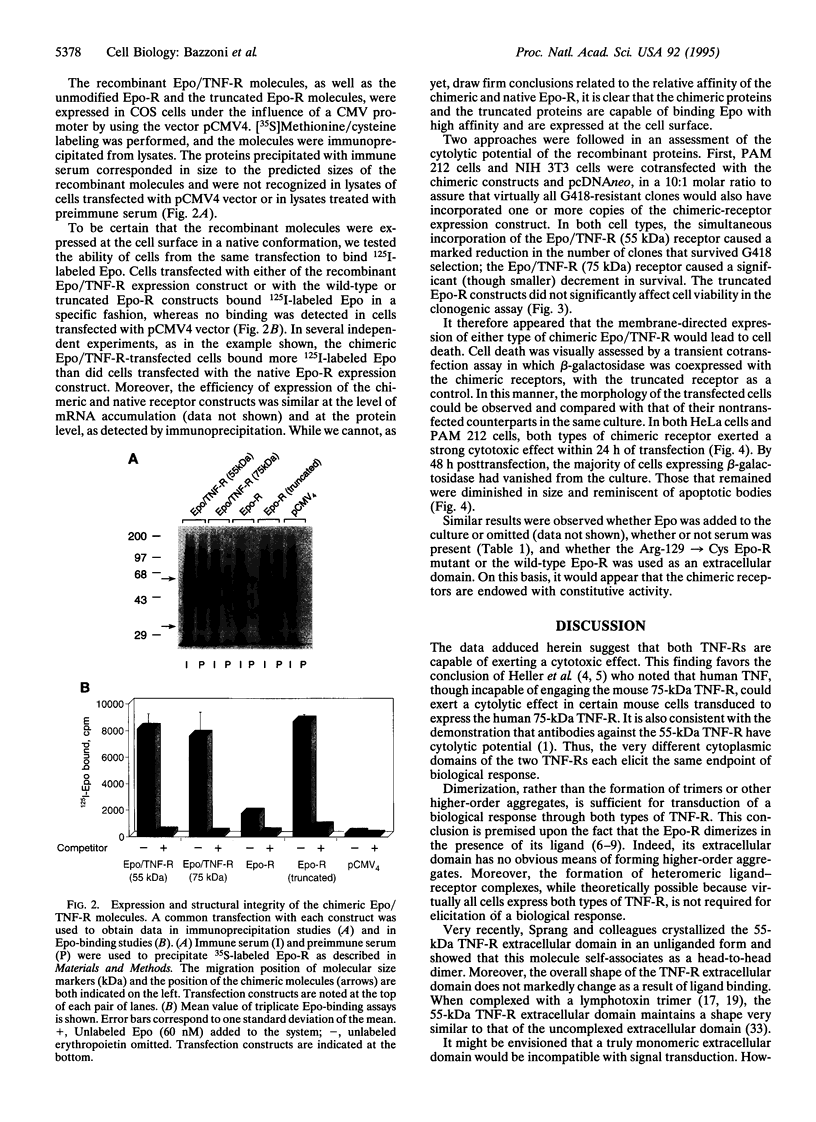
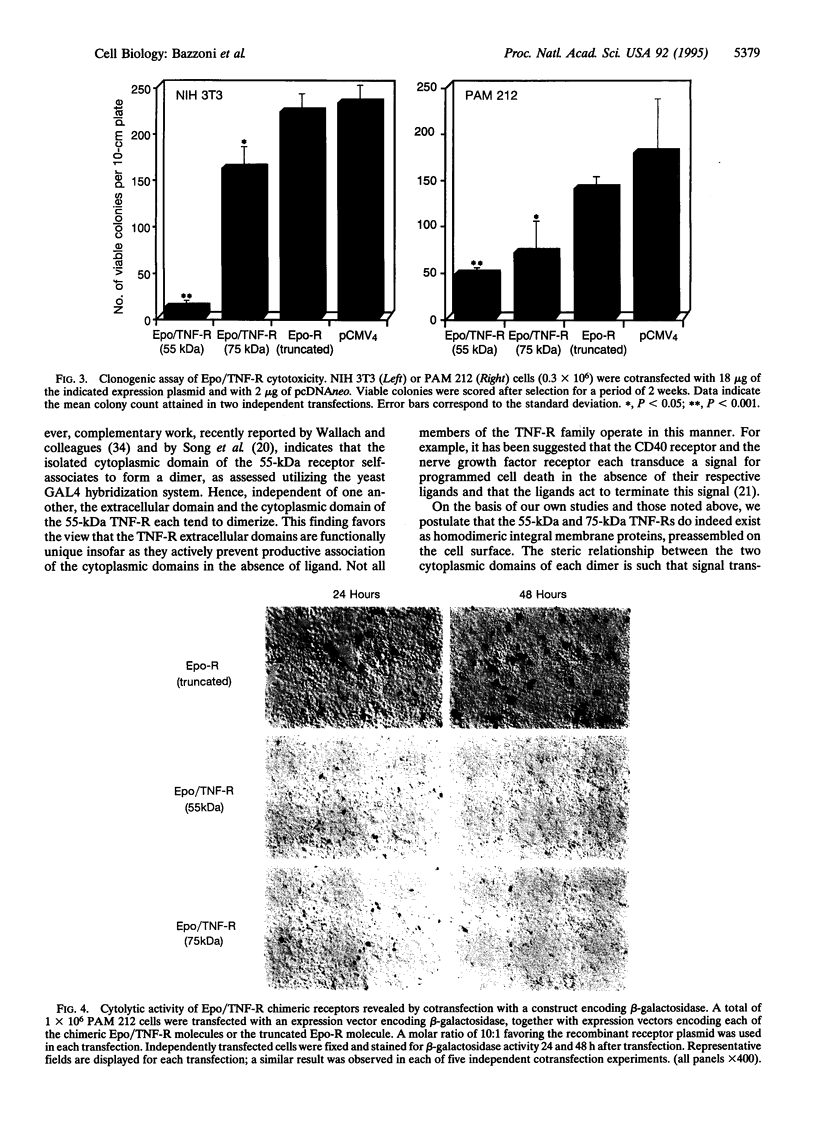

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andersson S., Davis D. L., Dahlbäck H., Jörnvall H., Russell D. W. Cloning, structure, and expression of the mitochondrial cytochrome P-450 sterol 26-hydroxylase, a bile acid biosynthetic enzyme. J Biol Chem. 1989 May 15;264(14):8222–8229. [PubMed] [Google Scholar]
- Banner D. W., D'Arcy A., Janes W., Gentz R., Schoenfeld H. J., Broger C., Loetscher H., Lesslauer W. Crystal structure of the soluble human 55 kd TNF receptor-human TNF beta complex: implications for TNF receptor activation. Cell. 1993 May 7;73(3):431–445. doi: 10.1016/0092-8674(93)90132-a. [DOI] [PubMed] [Google Scholar]
- Bevilacqua M. P., Pober J. S., Majeau G. R., Fiers W., Cotran R. S., Gimbrone M. A., Jr Recombinant tumor necrosis factor induces procoagulant activity in cultured human vascular endothelium: characterization and comparison with the actions of interleukin 1. Proc Natl Acad Sci U S A. 1986 Jun;83(12):4533–4537. doi: 10.1073/pnas.83.12.4533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boldin M. P., Mett I. L., Varfolomeev E. E., Chumakov I., Shemer-Avni Y., Camonis J. H., Wallach D. Self-association of the "death domains" of the p55 tumor necrosis factor (TNF) receptor and Fas/APO1 prompts signaling for TNF and Fas/APO1 effects. J Biol Chem. 1995 Jan 6;270(1):387–391. doi: 10.1074/jbc.270.1.387. [DOI] [PubMed] [Google Scholar]
- Chen C., Okayama H. High-efficiency transformation of mammalian cells by plasmid DNA. Mol Cell Biol. 1987 Aug;7(8):2745–2752. doi: 10.1128/mcb.7.8.2745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng J., Turksen K., Yu Q. C., Schreiber H., Teng M., Fuchs E. Cachexia and graft-vs.-host-disease-type skin changes in keratin promoter-driven TNF alpha transgenic mice. Genes Dev. 1992 Aug;6(8):1444–1456. doi: 10.1101/gad.6.8.1444. [DOI] [PubMed] [Google Scholar]
- D'Andrea A. D., Lodish H. F., Wong G. G. Expression cloning of the murine erythropoietin receptor. Cell. 1989 Apr 21;57(2):277–285. doi: 10.1016/0092-8674(89)90965-3. [DOI] [PubMed] [Google Scholar]
- D'Arcy A., Banner D. W., Janes W., Winkler F. K., Loetscher H., Schönfeld H. J., Zulauf M., Gentz R., Lesslauer W. Crystallization and preliminary crystallographic analysis of a TNF-beta-55 kDa TNF receptor complex. J Mol Biol. 1993 Jan 20;229(2):555–557. doi: 10.1006/jmbi.1993.1055. [DOI] [PubMed] [Google Scholar]
- Engelmann H., Holtmann H., Brakebusch C., Avni Y. S., Sarov I., Nophar Y., Hadas E., Leitner O., Wallach D. Antibodies to a soluble form of a tumor necrosis factor (TNF) receptor have TNF-like activity. J Biol Chem. 1990 Aug 25;265(24):14497–14504. [PubMed] [Google Scholar]
- Gamble J. R., Harlan J. M., Klebanoff S. J., Vadas M. A. Stimulation of the adherence of neutrophils to umbilical vein endothelium by human recombinant tumor necrosis factor. Proc Natl Acad Sci U S A. 1985 Dec;82(24):8667–8671. doi: 10.1073/pnas.82.24.8667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris K. W., Mitchell R. A., Winkelmann J. C. Ligand binding properties of the human erythropoietin receptor extracellular domain expressed in Escherichia coli. J Biol Chem. 1992 Jul 25;267(21):15205–15209. [PubMed] [Google Scholar]
- Heller R. A., Song K., Fan N., Chang D. J. The p70 tumor necrosis factor receptor mediates cytotoxicity. Cell. 1992 Jul 10;70(1):47–56. doi: 10.1016/0092-8674(92)90532-h. [DOI] [PubMed] [Google Scholar]
- Klebanoff S. J., Vadas M. A., Harlan J. M., Sparks L. H., Gamble J. R., Agosti J. M., Waltersdorph A. M. Stimulation of neutrophils by tumor necrosis factor. J Immunol. 1986 Jun 1;136(11):4220–4225. [PubMed] [Google Scholar]
- Kolls J., Peppel K., Silva M., Beutler B. Prolonged and effective blockade of tumor necrosis factor activity through adenovirus-mediated gene transfer. Proc Natl Acad Sci U S A. 1994 Jan 4;91(1):215–219. doi: 10.1073/pnas.91.1.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lodish H. F., Hilton D., Longmore G., Watowich S. S., Yoshimura A. The erythropoietin receptor: dimerization, activation, and tumorigenesis. Cold Spring Harb Symp Quant Biol. 1992;57:95–106. doi: 10.1101/sqb.1992.057.01.013. [DOI] [PubMed] [Google Scholar]
- Pober J. S., Gimbrone M. A., Jr, Lapierre L. A., Mendrick D. L., Fiers W., Rothlein R., Springer T. A. Overlapping patterns of activation of human endothelial cells by interleukin 1, tumor necrosis factor, and immune interferon. J Immunol. 1986 Sep 15;137(6):1893–1896. [PubMed] [Google Scholar]
- Pober J. S., Lapierre L. A., Stolpen A. H., Brock T. A., Springer T. A., Fiers W., Bevilacqua M. P., Mendrick D. L., Gimbrone M. A., Jr Activation of cultured human endothelial cells by recombinant lymphotoxin: comparison with tumor necrosis factor and interleukin 1 species. J Immunol. 1987 May 15;138(10):3319–3324. [PubMed] [Google Scholar]
- Probert L., Keffer J., Corbella P., Cazlaris H., Patsavoudi E., Stephens S., Kaslaris E., Kioussis D., Kollias G. Wasting, ischemia, and lymphoid abnormalities in mice expressing T cell-targeted human tumor necrosis factor transgenes. J Immunol. 1993 Aug 15;151(4):1894–1906. [PubMed] [Google Scholar]
- Rabizadeh S., Oh J., Zhong L. T., Yang J., Bitler C. M., Butcher L. L., Bredesen D. E. Induction of apoptosis by the low-affinity NGF receptor. Science. 1993 Jul 16;261(5119):345–348. doi: 10.1126/science.8332899. [DOI] [PubMed] [Google Scholar]
- Song H. Y., Dunbar J. D., Donner D. B. Aggregation of the intracellular domain of the type 1 tumor necrosis factor receptor defined by the two-hybrid system. J Biol Chem. 1994 Sep 9;269(36):22492–22495. [PubMed] [Google Scholar]
- Stolpen A. H., Guinan E. C., Fiers W., Pober J. S. Recombinant tumor necrosis factor and immune interferon act singly and in combination to reorganize human vascular endothelial cell monolayers. Am J Pathol. 1986 Apr;123(1):16–24. [PMC free article] [PubMed] [Google Scholar]
- Tartaglia L. A., Goeddel D. V., Reynolds C., Figari I. S., Weber R. F., Fendly B. M., Palladino M. A., Jr Stimulation of human T-cell proliferation by specific activation of the 75-kDa tumor necrosis factor receptor. J Immunol. 1993 Nov 1;151(9):4637–4641. [PubMed] [Google Scholar]
- Tartaglia L. A., Rothe M., Hu Y. F., Goeddel D. V. Tumor necrosis factor's cytotoxic activity is signaled by the p55 TNF receptor. Cell. 1993 Apr 23;73(2):213–216. doi: 10.1016/0092-8674(93)90222-c. [DOI] [PubMed] [Google Scholar]
- Taverne J., Sheikh N., de Souza J. B., Playfair J. H., Probert L., Kollias G. Anaemia and resistance to malaria in transgenic mice expressing human tumour necrosis factor. Immunology. 1994 Jul;82(3):397–403. [PMC free article] [PubMed] [Google Scholar]
- Teng M. N., Turksen K., Jacobs C. A., Fuchs E., Schreiber H. Prevention of runting and cachexia by a chimeric TNF receptor-Fc protein. Clin Immunol Immunopathol. 1993 Nov;69(2):215–222. doi: 10.1006/clin.1993.1172. [DOI] [PubMed] [Google Scholar]
- Watowich S. S., Hilton D. J., Lodish H. F. Activation and inhibition of erythropoietin receptor function: role of receptor dimerization. Mol Cell Biol. 1994 Jun;14(6):3535–3549. doi: 10.1128/mcb.14.6.3535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watowich S. S., Yoshimura A., Longmore G. D., Hilton D. J., Yoshimura Y., Lodish H. F. Homodimerization and constitutive activation of the erythropoietin receptor. Proc Natl Acad Sci U S A. 1992 Mar 15;89(6):2140–2144. doi: 10.1073/pnas.89.6.2140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshimura A., Longmore G., Lodish H. F. Point mutation in the exoplasmic domain of the erythropoietin receptor resulting in hormone-independent activation and tumorigenicity. Nature. 1990 Dec 13;348(6302):647–649. doi: 10.1038/348647a0. [DOI] [PubMed] [Google Scholar]
- de Vos A. M., Ultsch M., Kossiakoff A. A. Human growth hormone and extracellular domain of its receptor: crystal structure of the complex. Science. 1992 Jan 17;255(5042):306–312. doi: 10.1126/science.1549776. [DOI] [PubMed] [Google Scholar]