Abstract
Background
Long‐acting inhaled ß2‐adrenergic agonists (LABAs) are recommended as 'add‐on' medication to inhaled corticosteroids (ICS) in the maintenance therapy of asthmatic adults and children aged two years and above.
Objectives
To quantify in asthmatic patients the safety and efficacy of the addition of LABAs to ICS in patients insufficiently controlled on ICS alone.
Search methods
We identified randomised controlled trials (RCTs) through electronic database searches (the Cochrane Airways Group Specialised Register, MEDLINE, EMBASE and CINAHL), bibliographies of RCTs and correspondence with manufacturers until May 2008.
Selection criteria
We included RCTs if they compared the addition of inhaled LABAs versus placebo to the same dose of ICS in children aged two years and above and in adults.
Data collection and analysis
Two review authors independently assessed studies for methodological quality and extracted data. We obtained confirmation from the trialists when possible. The primary endpoint was the relative risk (RR) of asthma exacerbations requiring rescue oral corticosteroids. Secondary endpoints included pulmonary function tests (PFTs), rescue beta2‐agonist use, symptoms, withdrawals and adverse events.
Main results
Seventy‐seven studies met the entry criteria and randomised 21,248 participants (4625 children and 16,623 adults). Participants were generally symptomatic at baseline with moderate airway obstruction despite their current ICS regimen. Formoterol or salmeterol were most frequently added to low‐dose ICS (200 to 400 µg/day of beclomethasone (BDP) or equivalent) in 49% of the studies. The addition of a daily LABA to ICS reduced the risk of exacerbations requiring oral steroids by 23% from 15% to 11% (RR 0.77, 95% CI 0.68 to 0.87, 28 studies, 6808 participants). The number needed to treat with the addition of LABA to prevent one use of rescue oral corticosteroids is 41 (29, 72), although the event rates in the ICS groups varied between 0% and 38%. Studies recruiting adults dominated the analysis (6203 adult participants versus 605 children). The subgroup estimate for paediatric studies was not statistically significant (RR 0.89, 95% CI 0.58 to 1.39) and includes the possibility of the superiority of ICS alone in children.
Higher than usual dose of LABA was associated with significantly less benefit. The difference in the relative risk of serious adverse events with LABA was not statistically significant from that of ICS alone (RR 1.06, 95% CI 0.87 to 1.30). The addition of LABA led to a significantly greater improvement in FEV1 (0.11 litres, 95% 0.09 to 0.13) and in the proportion of symptom‐free days (11.88%, 95% CI 8.25 to 15.50) compared to ICS monotherapy. It was also associated with a reduction in the use of rescue short‐acting ß2‐agonists (‐0.58 puffs/day, 95% CI ‐0.80 to ‐0.35), fewer withdrawals due to poor asthma control (RR 0.50, 95% CI 0.41 to 0.61), and fewer withdrawals due to any reason (RR 0.80, 95% CI 0.75 to 0.87). There was no statistically significant group difference in the risk of overall adverse effects (RR 1.00, 95% 0.97 to 1.04), withdrawals due to adverse health events (RR 1.04, 95% CI 0.86 to 1.26) or any of the specific adverse health events.
Authors' conclusions
In adults who are symptomatic on low to high doses of ICS monotherapy, the addition of a LABA at licensed doses reduces the rate of exacerbations requiring oral steroids, improves lung function and symptoms and modestly decreases use of rescue short‐acting ß2‐agonists. In children, the effects of this treatment option are much more uncertain. The absence of group difference in serious adverse health events and withdrawal rates in both groups provides some indirect evidence of the safety of LABAs at usual doses as add‐on therapy to ICS in adults, although the width of the confidence interval precludes total reassurance.
Keywords: Adolescent; Adult; Child; Child, Preschool; Humans; Administration, Inhalation; Adrenal Cortex Hormones; Adrenal Cortex Hormones/administration & dosage; Adrenergic beta‐Agonists; Adrenergic beta‐Agonists/administration & dosage; Albuterol; Albuterol/administration & dosage; Albuterol/analogs & derivatives; Anti‐Asthmatic Agents; Anti‐Asthmatic Agents/administration & dosage; Asthma; Asthma/drug therapy; Beclomethasone; Beclomethasone/administration & dosage; Chronic Disease; Drug Therapy, Combination; Drug Therapy, Combination/methods; Ethanolamines; Ethanolamines/administration & dosage; Formoterol Fumarate; Randomized Controlled Trials as Topic; Salmeterol Xinafoate
Plain language summary
Long‐acting beta2‐agonists versus placebo in addition to inhaled corticosteroids in children and adults with chronic asthma
The use of long‐acting ß2‐agonists (LABAs) as 'add‐on' medication to inhaled corticosteroids is recommended for poorly‐controlled asthma where asthma exacerbations may require additional treatment with oral steroids. The purpose of this review was to assess the efficacy and safety of adding long‐acting ß2‐agonists to inhaled corticosteroids in asthmatic children and adults. Based on the identified randomised trials, in people who remain symptomatic while on inhaled corticosteroids, the addition of long‐acting ß2‐agonists improves lung function and reduces the risk of asthma exacerbations compared to ongoing treatment with a similar dose of inhaled corticosteroids alone in adults. We could not find evidence of increased serious adverse events or withdrawal rates due to adverse health events with the combination of long‐acting ß2‐agonists at usual doses and inhaled corticosteroids in adults. This provides some indirect evidence, but not total reassurance, regarding the short‐ and medium‐term safety of this treatment strategy. There have not been enough children studied to assess the risks and benefits of adding LABAs in this age group.
Background
With the recognition of asthma as an inflammatory disease, the cornerstone of asthma management is the use of inhaled corticosteroids (Adams 2008; GINA 2007). Inhaled ß2‐agonists are powerful agents used to relieve the bronchoconstriction associated with asthma. They act by stimulating the ß2‐receptors located in airway smooth muscle resulting in smooth muscle relaxation (Nelson 1995). Inhaled ß2‐agonists can be differentiated by their onset and duration of action. Short‐acting ß2‐agonists, such as salbutamol and terbutaline, are hydrophilic and interact directly with ß2‐receptors, leading to a fast onset of action with a duration of effect of six hours or less (D'Alonzo 1997). Long‐acting ß2‐agonists (LABAs) provide longer symptom control, which is a particularly useful feature for preventing night‐time symptoms. There are two main LABAs, namely salmeterol and formoterol. Salmeterol is highly lipophilic and diffuses through the lipid bilayer in muscle cell membranes to reach the ß2‐receptors, explaining the slower onset and long duration of action (Nelson 1995). Formoterol, being less lipophilic, has a fast onset of action, similar to short‐acting ß2‐agonists, and is believed to be incorporated into the lipid bilayer to serve as a reservoir, accounting for its prolonged action (Nelson 1995).
Frequent use of short‐ or long‐acting ß2‐agonists generally indicates a significant inflammatory process that should be controlled with anti‐inflammatory drugs such as inhaled corticosteroids. The role of long‐acting ß2‐agonists in the management of asthma has previously been debated (Ernst 2006; Salpeter 2006). At present, the use of long‐acting ß2‐agonists as monotherapy clearly appears to be less effective than inhaled corticosteroids alone (Warner 1998), and has been associated with increased asthma deaths; these data resulted in an early trial termination (SMART). A recent systematic review, combining data from studies where patients received long‐acting ß2‐agonists as monotherapy or adjunctive therapy to ICS, raised concerns regarding the safety of LABAs (Salpeter 2006). However, a subsequent commentary based on Cochrane Reviews strongly suggested that only the use of LABA as monotherapy was associated with the serious adverse health events, while the use of LABAs in combination with inhaled corticosteroids was protective (Ernst 2006).
A previous Cochrane systematic review suggested that an increased risk of exacerbations may be limited to patients receiving long‐acting ß2‐agonists as monotherapy (Walters 2007). Although all national and international asthma consensus statements recommend the use of long‐acting ß2‐agonists only in combination with inhaled corticosteroids (BTS 2007; Canadian Paediatic Asthma Guideline 2005; GINA 2007; NAC 2006; NAEPP 2007), some uncertainty remains regarding the safety of combination therapy (Cates 2009a; Cates 2009b).
In adults with unsatisfactory asthma control on inhaled corticosteroids, international guidelines clearly favour the addition of LABAs to low or moderate doses of inhaled steroids over other options such as increasing the dose of steroids or adding other agents. Variations across guidelines highlight ongoing uncertainties regarding the optimal use of LABAs as add‐on treatment to inhaled steroids. First, the lowest dose of inhaled steroids to which LABAs could be considered as add‐on therapy varies across guidelines. In adults, LABAs can be added to chlorofluorocarbon‐propelled beclomethasone dipropionate (BDP) at a dose equivalent to or greater than 200 µg/day according to the American (NAEPP 2007), British (BTS 2007) and GINA guidelines (GINA 2007); 400 µg/day or more according to the Canadian consensus statement (Canadian Paediatic Asthma Guideline 2005); and 800 µg/day or more according to the Australian recommendations (NAC 2006). Recommendations also vary by age group. In children aged five years and over, the addition of a LABA is recommended if inadequate control is achieved with 200 µg/day of BDP according to the British (BTS 2007) and American guidelines (NAEPP 2007); 400 µg/day according to the GINA recommendations (GINA 2007); and 800 µg/day according to the Australian (NAC 2006) and Canadian (Canadian Paediatic Asthma Guideline 2005) statements. Secondly, the preference of adding LABA to inhaled steroids as 'step three' option over other alternative strategies varies by age. Indeed, the Canadian (Canadian Paediatic Asthma Guideline 2005) and Australian (NAC 2006) guidelines clearly favour increasing the dose of inhaled corticosteroid to 800 µg/day BDP‐equivalent before adding LABAs, as favoured by the British (BTS 2007) and American (NAEPP 2007) guidelines. In infants and preschool‐aged children, a LABA is not recommended as add‐on therapy, except by the American guidelines which suggest LABAs as add‐on to 100 to 400 µg/day of BDP or equivalent (NAEPP 2007). Finally, the criteria for considering the addition of LABA are vaguely described as inadequate control with no clear instruction as to whether the severity of baseline obstruction, duration of use, type and dose of LABA, dose difference between ICS as monotherapy and combination therapy, number of devices to deliver combination therapy or atopy may be important factors. We sought to update our systematic review of randomised controlled trials in order to clarify ongoing uncertainties about the optimal use of LABAs as add‐on therapy to inhaled steroids and the subgroups of patients that may benefit most from the intervention (Ni Chroinin 2005).
Objectives
The objective of this review was to assess the safety and clinical efficacy in asthma control resulting from the addition of long‐acting ß2‐agonists to inhaled corticosteroids in asthmatic patients. We also wished to examine whether the efficacy of long‐acting ß2‐agonists was influenced by age, severity of airway obstruction, dose of inhaled corticosteroids to which long‐acting ß2‐agonists were added, number of devices to deliver combination therapy, the dose and type of long‐acting ß2‐agonist and the duration of intervention. Additionally we wished to assess carefully the safety profile (and its possible determinants) of long‐acting ß2‐agonists administered as add‐on therapy to inhaled corticosteroids.
Methods
Criteria for considering studies for this review
Types of studies
Only randomised controlled trials conducted in adults, children or both, in whom long‐acting ß2‐agonists were added to inhaled corticosteroids.
Types of participants
Children aged two years and above or adults with chronic asthma and having received daily inhaled corticosteroids for at least four weeks prior to study entry.
Types of interventions
Long‐acting ß2‐agonist (salmeterol or formoterol) or placebo administered daily at a fixed dose for at least 28 days. The dose of inhaled corticosteroids had to be similar between the intervention (LABA + ICS) and the control (ICS monotherapy) groups. Other co‐interventions such as xanthines, anticholinergics and other anti‐asthmatic medications were accepted, provided that the dose remained unchanged throughout the study. Rescue inhaled short‐acting ß2‐agonists and short courses of oral steroids were permitted.
Types of outcome measures
Primary outcomes
The primary outcome was the number of patients with asthma exacerbations of moderate intensity; that is requiring a short course of oral corticosteroids.
Secondary outcomes
Other measures reflecting the severity of acute exacerbations, such as hospital admissions.
Measures reflecting chronic asthma control, including changes in pulmonary function tests, symptoms, days and nights without symptoms, functional status, quality of life and use of rescue short‐acting ß2‐agonists.
Changes in measures of inflammation, such as serum eosinophils, serum eosinophil cationic protein and sputum eosinophils.
Withdrawals.
Rates of serious adverse events, clinical and biochemical adverse effects.
Search methods for identification of studies
We carried out the most recent searches in May 2008.
Electronic searches
We carried out a search in the Cochrane Airways Group Specialised Register of asthma trials, which is derived from systematic searches of bibliographic databases including the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, EMBASE and CINAHL and handsearching of respiratory journals and meeting abstracts. This Register also contains a variety of studies published in foreign languages. We did not exclude trials on the basis of language.
The Register was searched using the following terms: (((beta* and agonist*) and long‐acting or "long acting") or ((beta* and adrenergic*) and long‐acting or "long acting") or (bronchodilat* and long‐acting or "long acting") or (salmeterol or formoterol or advair or symbicort)) and (((steroid* or glucocorticoid* or corticosteroid*) and inhal*) or (budesonide or beclomethasone or fluticasone or triamcinolone or flunisolide).
Searching other resources
We searched the clinical trial websites of manufacturers of long‐acting beta‐agonists: GSK (GlaxoSmithKline (GSK) Clinical Trials Register), AstraZeneca (AstraZeneca Clinical Trials Register) and Novartis (Novartis Clinical Trials Database). We consulted an additional website listing results of published and unpublished clinical trials (Clinical Study Results).
We checked reference lists of all included studies and reviews to identify potentially relevant citations.
We also made enquiries regarding other published or unpublished studies known to the authors of the included studies or to pharmaceutical companies, namely GlaxoSmithKline and AstraZeneca who manufacture the agents.
Data collection and analysis
Selection of studies
From the title, abstract or descriptors, one of the review authors (IRG or MNC and FMD or TL) independently reviewed the literature searches. We excluded all studies that were clearly not randomised controlled trials or that clearly did not fit the inclusion criteria. Two review authors reviewed all other citations independently in full text, assessing for inclusion based on study design, population, intervention and outcome.
Data extraction and management
Two review authors (IRG or MNC and FMD or TL) independently extracted data from the trials and entered these into a designated Excel workbook for double‐checking. Data were transferred by TL to the Review Manager software (RevMan 2008). Where necessary, we performed expansions of graphic reproductions and estimations from other data presented in the paper.
We reported the mean daily dose of inhaled corticosteroids in both the intervention and control groups, in chlorofluorocarbon (CFC)‐propelled beclomethasone‐equivalents, where 1 µg of beclomethasone dipropionate equates to 1 µg of budesonide and 0.5 µg of fluticasone propionate (NAEPP 2007). All doses of inhaled medications were reported based on ex‐valve rather than ex‐inhaler values.
Assessment of risk of bias in included studies
For the 2008 update of this review we undertook an assessment of the risk of bias for eligible studies, based on the recommendations described in the Cochrane Handbook for Systematic Reviews of Interventions (Cochrane Handbook). This entailed describing potential sources of bias in eligible trials (allocation, blinding, missing data and the availability of our primary outcome), and providing our judgement of how the design of each study protects against each potential source of bias. We have collated our judgements in a graphical overview. The methodology applied in the previous version of this review is given in Appendix 1.
Measures of treatment effect
The analysis focused on long‐acting ß2‐agonist (LABA) and inhaled corticosteroids (ICS) (LABA + ICS) versus a similar dose of inhaled corticosteroids (ICS monotherapy) as second‐line treatment, that is in patients already on inhaled corticosteroids.
Unit of analysis issues
We included data from cross‐over studies in this review provided that we could obtain estimates of within‐patient differences, and their associated standard errors from either back‐calculating 95% confidence intervals or from P values from appropriate statistical tests.
When a trial had more than one intervention or control group, additional intervention‐control comparisons were considered, if appropriate for this review. If two intervention‐control comparisons used the same group twice as comparator (for example a three‐arm study had two LABA + ICS arms and one ICS monotherapy arm) the number of participants in the group used twice (in this instance, the ICS monotherapy group) was halved to avoid over‐representation (Buhl 2003a; Buhl 2003b; Zetterstrom 2001a; Zetterstrom 2001b; Zimmerman 2004a; Zimmerman 2004b). For event rates, the numerator was also halved in the control group.
Dealing with missing data
We asked primary authors or sponsors to confirm the methodology and data extraction and to provide additional information and clarification for the trial, as needed. We contacted study authors/sponsors to obtain data on our primary outcome of exacerbations requiring oral steroids, and if possible hospital admissions and serious adverse events where they were not available in the primary study reports.
Assessment of heterogeneity
We tested homogeneity of effect sizes between studies being pooled with the I2 statistic, with a value greater than 25% as the cut‐off for heterogeneity (Higgins 2003). In the absence of heterogeneity we used the fixed‐effect model (Greenland 1985). If heterogeneity was suggested by the I2, we applied the Dersimonian and Laird random‐effects model (DerSimonian 1986) to the summary estimates. Unless otherwise specified the fixed‐effect model is reported. Equivalence was assumed if the relative risk estimate and its confidence interval were between 0.9 and 1.1.
Data synthesis
We calculated treatment effects for dichotomous variables using a relative risk (RR) with 95% confidence intervals (CI). For continuous outcomes, such as pulmonary function tests, we pooled data with weighted mean differences (WMD) for outcomes on the same scale or standardised mean differences (SMD) if the same construct was measured but done so with different scales. For both WMDs and SMDs we reported the mean difference with 95% confidence intervals.
We derived numbers needed to treat (NNT) from the pooled risk ratios using Visual Rx (an online calculator at http://www.nntonline.net) (Cates 2002). In order to reflect the variation in control group event rates, we also calculated NNTs for an average risk across the lower, middle and upper quartiles of the events rates, weighting the control event rates (CERs) by sample size.
We undertook a fail‐safe N test to determine how many negative studies would be required to overturn the results (Gleser 1996).
Subgroup analysis and investigation of heterogeneity
We planned subgroup analyses to explore possible reasons for heterogeneity of the primary outcome and in the absence of heterogeneity, to identify potential effect modifiers for which the magnitude of effect may change according to the value of the characteristic (for example, severity of airway obstruction). We examined the following a priori defined subgroups to explore their influence on the magnitude of effect (effect modification).
Magnitude of airway obstruction at baseline as determined by the mean percent predicted forced expiratory volume in one second (FEV1): classified as mild (FEV1 80% or more), moderate (FEV1 61% to 79%) or severe (FEV1 60% or less) (GINA 2007).
Children (less than 18 years of age) versus adults.
Mean dose (ex‐valve) of inhaled corticosteroids in both groups, reported in CFC‐propelled beclomethasone‐equivalent doses (µg/day), portrayed as the user‐defined number.
Usual versus higher than usual dose (reported as ex‐valve in µg) of the long‐acting ß2‐agonist (salmeterol or formoterol).
Type of long‐acting ß2‐agonist (salmeterol versus formoterol).
Use of one or two devices to deliver the combination of ICS plus LABA.
Trial duration (≤16 and > 16 weeks).
Since the publication of the original protocol in 1999 and prior to data analysis, we have added the last three subgroup analyses. Subgroup six was added because of recent data (Nelson 2003) suggesting a differential effect when using one or two devices to deliver the combination of LABA plus ICS. We added subgroup seven to investigate the risk of tachyphylaxis.
We examined differences in the magnitude of effect attributable to these subgroups with the residual Chi2 test from the Peto odds ratios or with the t‐test for weighted mean differences (Deeks 2001). We conducted a multivariate meta‐regression to examine the simultaneous impact of, and interaction between, the above‐named variables on the variance in the risk of patients with exacerbations requiring oral steroids. Backward and forward models were built using these subgroups as well as using FEV1 (litres) and dose of inhaled corticosteroids (µg/day) as continuous variables (Small Stata for Windows, Version 11 2009, Stata Corporation, Texas, USA).
Sensitivity analysis
We used funnel plots to examine the possibility of publication bias (Egger 1997). We undertook a sensitivity analysis by source of data, by removing the study data which were made available from unpublished sources. We did this since a considerable number of unpublished studies were identified from pharmaceutical company trial registers, or data from published studies were made available through correspondence with pharmaceutical companies.
Results
Description of studies
Results of the search
Electronic and handsearches yielded a total of 376 citations between April 2004 and May 2008 (see Table 1 for previous all‐year search results). A flow diagram depicting the inclusion and exclusion of studies for this update is given in Figure 1.
1. Search history.
| Year | Detail |
| All years to April 2004 | Citations identified: 594 Of these, 545 reports were excluded for the following mutually exclusive reasons: (1) duplicate references (N = 208) (2) not a randomised controlled trial (N = 68) or an ongoing trial (N = 14) (3) participants were not asthmatics (N = 4) (4) no consistent intervention with inhaled corticosteroids in all participants (N = 41) (5) intervention was not daily inhaled long‐acting ß2‐agonists (N = 19) (6) control intervention was not inhaled corticosteroids alone (N = 63) (7) duration of intervention was less than 30 days (N = 45) (8) outcome measures did not reflect asthma control (N = 8) (9) the treatment and intervention groups compared the same medications either in combination or with different delivery devices (N = 30) (10) co‐intervention with a non‐permitted agent (N = 1) (11) patients were steroid‐naive on study entry (N = 20) (12) control group had a higher dose of inhaled corticosteroid than the intervention group (N = 21) (13) the dose of inhaled corticosteroid did not remain stable during the trial (N = 3) Due to the large number of citations considered, the reasons for exclusion are provided only for published randomised controlled trials 33 treatment‐control comparisons derived from 28 trials met the entry criteria of the review |
1.

Flow diagram for literature search results April 2004 to May 2008.
Included studies
We have included 77 randomised treatment‐control comparisons (referred to hereafter as studies) represented by 179 citations. This represents the addition of 44 studies to the original review, adding data from 14,043 participants to the 7205 patients recruited in studies included in the previous version (Ni Chroinin 2005). A full description of each study is given in the Characteristics of included studies table.
Fourteen trials contributed two treatment‐control comparisons, as they assessed more than one mode of delivering combination therapy, or assessed more than one dose of inhaled steroids as a control intervention (Aubier 1999a; Aubier 1999b; Buhl 2003a; Buhl 2003b; D5896C0001a; D5896C0001b; Jenkins 2006a; Jenkins 2006b; Morice 2008a; Morice 2008b; Noonan 2006a; Noonan 2006b; O'Byrne 2001a; O'Byrne 2001b; Pauwels 1997a; Pauwels 1997b; Pohunek 2006a; Pohunek 2006b; SD 037 0344a; SD 037 0344b; SD 039 0725a; SD 039 0725b; SD 039 0726a; SD 039 0726b; Zetterstrom 2001a; Zetterstrom 2001b; Zimmerman 2004a; Zimmerman 2004b). Each comparison, hereafter counting as a separate study with the adjustment, is described in the methods to avoid overrepresentation of subjects.
Of the included studies 17 have not been published as full‐text journal articles (Hultquist 2000; SAM40008; SAM40012; SAS40024; SAS40036; SAS40037; SD 037 0344a; SD 037 0344b; SD 039 0714; SD 039 0718; SD 039 0719; SD 039 0725a; SD 039 0725b; SD 039 0728; SFA100316; SFCF4026; SMS40012).
Participants
There were 21,248 participants (16,623 adults; 4625 children) randomised to the included studies. Study size varied from 16 (Simons 1997) to 2252 (GOAL). Twenty‐eight (36%) studies recruited adults exclusively (> 18 years); 24 (31%) recruited children and 21 (27% of trials) studies permitted the enrolment of an unspecified number of adolescents aged 12 years or more. In two trials the lower age cut‐off was 15 years (SAS40036; SAS40037). In Houghton 2007 and SD 039 0349 the mean age suggested that the participants were predominantly adults with an unspecified age limit for enrolment.
In adult trials, the mean age of participants was relatively homogeneous, varying from 35 years (Li 1999) to 48 years (Zetterstrom 2001a). In paediatric studies it ranged from eight years (Malone 2005) to 14 years (Langton Hewer 1995). The gender distribution varied widely from 30% males in Norhaya 1999 to 71% in Langton Hewer 1995.
In all but three trials, participants clearly had inadequate asthma control (that is, ongoing symptoms and use of rescue short‐acting ß2‐agonists in addition to maintenance inhaled steroids) at the time of enrolment. In the remaining three trials (Meijer 1995; Shapiro 2000; Simons 1997) participants appeared asymptomatic and well‐controlled according to the Canadian consensus guidelines (Canadian Paediatic Asthma Guideline 2005).
In over half of the studies (N = 45, 58%) the mean severity of baseline airway obstruction was moderate (that is, FEV1 or PEF predicted of 61% to 79%); while it was mild (80% or more of predicted) in 23 (30%) studies, and unavailable in the nine (12%) remaining studies (D'Urzo 2001; SAM40008; SAM40012; SAS40037; SD 037 0344a; SD 037 0344b; SFA100314; SFA100316; SFCF4026).
The presence of atopic disease at baseline was reported in only 13 studies, all of which reported atopy in 58% to 100% of participants (Akpinarli 1999; GOAL; Koopmans 2006; Langton Hewer 1995; Li 1999; Meijer 1995; Russell 1995; Simons 1997; Stelmach 2007; Tal 2002; van der Molen 1997; Verberne 1998; Wallin 2003).
Type, dose and delivery of inhaled therapy
The long‐acting ß2‐agonist was formoterol in 42 (54%) studies and salmeterol xinafoate in 35 (46%). The overwhelming majority (N = 73) of studies tested recommended doses of the long‐acting ß2‐agonist (that is, salmeterol 50 µg twice daily, formoterol 6 or 12 µg twice daily). In four studies a higher than usual dose of salmeterol (100 µg twice daily in Boyd 1995 and Langton Hewer 1995) or formoterol (24 µg twice daily in SD 039 0728; van der Molen 1997) was used.
All but five studies examined the combination of LABA plus ICS versus ICS monotherapy in a twice‐daily regimen: Buhl 2003a; D5896C0001b; Kuna 2006 and SD 039 0725a; SD 039 0725b examined the two options as a once‐daily administration regimen.
Within each study the dose of inhaled corticosteroid to which LABA was added was similar to ICS monotherapy in the control groups and fixed for all patients. It was a uniform dose in 57 (73%) studies and a range or unspecified dose for the remaining 20 studies. Of the studies assessing LABA in conjunction with fixed‐dose ICS, 36 (49%) tested the addition of LABA to low‐dose inhaled corticosteroids (200 to 400 µg/day of beclomethasone, or equivalent), five (7%) added LABA to a medium dose of ICS (401 to 799 µg/day of beclomethasone, or equivalent) and 16 (21%) studies used a high dose of ICS (800 µg/day or more of beclomethasone, or equivalent). LABA was added to budesonide (31 studies), beclomethasone (four studies) budesonide or beclomethasone (one study) or fluticasone propionate (22 studies). The remaining trials only indicated that the ICS used was usual ICS therapy.
Thirty‐four (44%) studies used two inhaler devices to deliver ICS and LABA, while 27 (42%) studies used one device. Five trials (represented by ten studies) tested both one and two delivery devices against inhaled steroids (Aubier 1999a; Aubier 1999b; Jenkins 2006a; Jenkins 2006b; Noonan 2006a; Noonan 2006b; Pohunek 2006a; Pohunek 2006b; Zetterstrom 2001a; Zetterstrom 2001b). Wallin 2003 failed to report the number of devices used. Compliance was assessed or monitored in 13 studies.
Study duration
The duration of the intervention in 43 studies (56%) studies was between 12 and 16 weeks. Sixteen (21%) studies lasted between four and eight weeks while the remaining 18 studies (23%) lasted from 24 to 54 weeks. Of note, only available data from the initial 12 weeks of the GOAL study were included to isolate the portion of this 52‐week trial that corresponded to the specific criteria of this review, and thus ensure homogeneity of included trial protocols. Indeed, GOAL used three 'step up' phases, each of 12 weeks duration in the initial phase of the study, during which the dose of inhaled corticosteroids was increased until either total asthma control was achieved or a pre‐specified maximal dose was achieved. Upon achieving pre‐defined asthma control by the end of the 'step', participants entered the second phase during which the background fluticasone dose was maintained for the remainder of the trial. Since the dose of ICS varied upward after the initial 12 weeks, we extracted data for the first 12 weeks while on a fixed dose.
Permitted co‐treatment
Co‐intervention with other prophylactic medications was explicitly permitted in six studies provided that doses remained unchanged throughout the trial. These included oral steroids, anticholinergics and xanthines (Langton Hewer 1995), cromoglycate and xanthines (Norhaya 1999) and immunotherapy (Zimmerman 2004a; Zimmerman 2004b). Two studies (Ind 2003; Russell 1995) permitted co‐intervention with other agents but did not mention specifically which drugs. Patients taking prophylactic medications were excluded in 16 other trials and this factor was unreported in the remaining trials. Rescue medications such as inhaled short‐acting ß2‐agonists and oral steroids were permitted in all the trials.
Outcomes
Data for the main outcome measure, the number of participants with one or more exacerbations requiring oral steroids, were reported or were made available to the authors on request for 30 studies.
Most studies reported changes in lung function, use of rescue ß2‐agonists, withdrawals due to any reason, withdrawals due to poor asthma control and overall adverse health events. There was a large variation in the way improvement in symptoms (symptom score, percent symptom‐free days, percent days with symptoms, percent night awakenings) and use of rescue fast‐acting ß2‐agonist were reported, both using various parameters (average value, final value at endpoint, percent change and change in percent values). Wide variations in reporting prevented the aggregation of some of the available data.
Funding
The overwhelming majority (86%) of the studies were sponsored by producers of both LABA and ICS: 33 studies were supported by GSK; 32 by AstraZeneca; three by Astra Draco (Pauwels 1997a; Pauwels 1997b; van der Molen 1997); one by Allen & Hanburys, a subsidiary of GSK in the United Kingdom (Boyd 1995); and one by Novartis (Fitzgerald 1999). Only one trial was independently supported by a charity organisation (Langton Hewer 1995) and a further study was supported by a University grant (Stelmach 2007). One study acknowledged AstraZeneca in the provision of active and placebo inhalers, but did not indicate that a grant had been awarded for the trial from the company (Green 2006). Four studies failed to declare a source of funding (Gardiner 1994; Teper 2005; Zimmerman 2004a; Zimmerman 2004b).
Excluded studies
A total of 315 studies did not meet the eligibility criteria of the review. They are listed in the section: Characteristics of excluded studies.
Risk of bias in included studies
Correspondence with GSK established the procedures used to allocate participants to treatment groups in studies sponsored by GSK (see Appendix 2). We have collated our judgements on the risk of bias for each study and present them in Figure 2.
2.

Allocation
Information regarding the allocation of participants to treatment groups was sufficient to grade 45 (58%) studies as being of low risk of selection bias. Information regarding the remaining studies did not allow us to make judgements.
Blinding
Only one study had an open label design (Molimard 2001), the remainder being double‐blind with an appropriate means of masking treatment (identical inhaler device, or double dummy design), except for D'Urzo 2001, Reddel 2007 and Teper 2005 where we could not establish how blinding was maintained.
Incomplete outcome data
The handling of withdrawals and drop‐outs from analyses were not adequately described in many of the studies since the definition of 'intention‐to‐treat' population varied or was not adequately defined. Methods such as last observation carried forward and imputation were applied in only a few studies, with the majority of trials not defining what the intention‐to‐treat population was.
Selective reporting
We could not find definitive evidence of selective reporting in the studies, which we considered only in relation to our primary outcome of oral steroid‐treated exacerbations. Although a number of studies did collect data on exacerbations, we were unable to obtain further information on which of these were managed in accordance with our stated primary endpoint. Some data specific to exacerbations requiring oral steroid were made available to us for a number of studies (see risk of bias tables in the Characteristics of included studies).
Other potential sources of bias
One study stated how many patients were screened for eligibility (Green 2006). Thirty‐four studies reported the percent of run‐in participants that were successfully randomised, ranging from 43% to 95% of recruited patients.
Effects of interventions
Primary outcome: exacerbations of asthma requiring oral steroids
Thirty studies (39% eligible studies) reported data for this outcome (two studies reported no events occurring in either group). The addition of a LABA to ICS therapy led to a 23% reduction (from 15% to 11%) in the relative risk of patients experiencing one or more exacerbations requiring oral corticosteroids (RR 0.77, 95% CI 0.68 to 0.87, P < 0.0001, N = 6808) (Figure 3). We did not observe any more statistical heterogeneity than would be expected due to the play of chance (I2 = 0%). The result of the overall fail‐safe N test was that 222 studies with negative findings would be needed to bring the estimate back to null (Gleser 1996).
3.

Forest plot of comparison: 1 Long‐acting beta2 vs placebo: both groups receiving similar dose ICS, outcome: 1.1 # patients with exacerbations requiring oral steroids.
The overall number needed to treat to prevent one rescue oral corticosteroids was 41 (29, 72) based on studies of between four and 54 weeks duration, and a pooled control group event rate of 15%. However, the baseline risk (i.e. the control group event rate) of exacerbations requiring treatment with oral corticosteroids varied between the studies, with quartiles of low (0% to 2%), low/medium (3% to 6%), medium/high (7% to 23%) and high (23% to 38%) risk (Table 2). Applying the pooled risk ratio to middle values across these quartiles, the NNTs were between 17 and 435:
2. Control group risk status for primary outcome.
| Study ID | Control group % event rate | Control group N | Duration (wk) |
| Akpinarli 1999 | 0 | 16 | 6 |
| Kavaru 2000 | 0 | 90 | 12 |
| Weiler 2005 | 0 | 90 | 4 |
| SFA100314 | 1 | 124 | 4 |
| SFA100316 | 1 | 118 | 4 |
| SAS40024 | 2 | 100 | 4 |
| Shapiro 2000 | 2 | 84 | 12 |
| Malone 2005 | 3 | 102 | 12 |
| Nathan 2006 | 3 | 91 | 12 |
| Noonan 2006a | 4 | 55 | 12 |
| Noonan 2006b | 4 | 54 | 12 |
| SAS40037 | 4 | 161 | 16 |
| SAS40036 | 4 | 159 | 16 |
| Li 1999 | 6 | 16 | 12 |
| Simons 1997 | 6 | 16 | 4 |
| Fitzgerald 1999 | 7 | 91 | 24 |
| Aubier 1999b | 8 | 83 | 28 |
| Aubier 1999a | 9 | 82 | 28 |
| Norhaya 1999 | 10 | 30 | 4 |
| Verberne 1998 | 18 | 57 | 54 |
| Russell 1995 | 18 | 99 | 12 |
| O'Byrne 2001b | 20 | 312 | 52 |
| Kemp 1998 | 23 | 254 | 12 |
| Boyd 1995 | 23 | 64 | 12 |
| Langton Hewer 1995 | 25 | 12 | 8 |
| O'Byrne 2001a | 25 | 322 | 52 |
| Wallin 2003 | 26 | 19 | 12 |
| Pauwels 1997b | 28 | 214 | 52 |
| van der Molen 1997 | 28 | 114 | 24 |
| Pauwels 1997a | 38 | 213 | 52 |
| Risk status | Median control group event rate (% (range)) | Median study duration (weeks (range)) | Mean FEV1 (% predicted (range)) | NNT benefit |
| Low | 1 (0 to 2) | 4 (4 to 12) | 70 (64 to 78) | 435 |
| Low/medium | 4 (3 to 6) | 12 (4 to 16) | 77 (68 to 93.4) | 109 |
| Medium/high | 14 (7 to 23) | 26 (4 to 54) | 76 (73 to 87) | 32 |
| High | 26 (23 to 38) | 24 (8 to 52) | 75 (66 to 86) | 17 |
Sensitivity analyses by risk of bias (allocation sequence generation, allocation concealment, blinding and completeness of follow up) did not alter the direction of the results and made little impact on the upper limit of the confidence interval (Analysis 2.9; Analysis 2.10; Analysis 2.11; Analysis 2.12).
2.9. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 9 Sensitivity analysis: exacerbations requiring oral steroids: studies with low risk of bias of selection bias (adequate allocation sequence generation).
2.10. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 10 Sensitivity analysis: exacerbations requiring oral steroids: studies with low risk of bias of selection bias (adequate allocation concealment).
2.11. Analysis.
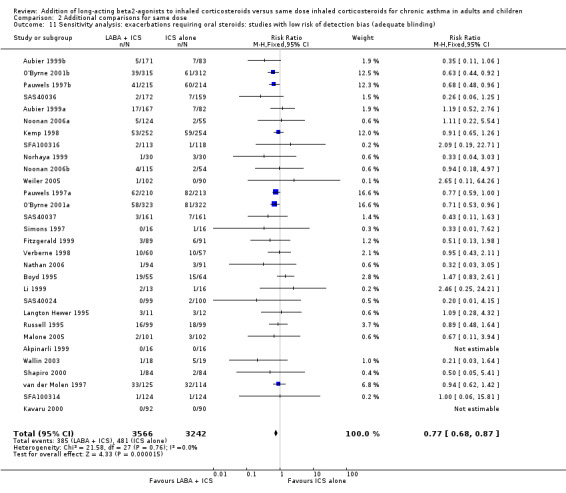
Comparison 2 Additional comparisons for same dose, Outcome 11 Sensitivity analysis: exacerbations requiring oral steroids: studies with low risk of detection bias (adequate blinding).
2.12. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 12 Sensitivity analysis: exacerbations requiring oral steroids: studies with low risk of bias of attrition bias (complete follow up of study participants).
Despite the absence of heterogeneity, we conducted a priori subgroup analyses to examine the impact of the following variables on the variance of the magnitude of effect observed (effect modification). There was no significant difference in the relative risks between subgroups based on airway obstruction (P = 0.20), age group (P = 0.53), dose of inhaled steroids (P = 0.34), type of LABA (P = 0.16), treatment duration (P = 0.13), number of devices (P = 0.45) or funding sources (P = 0.63). The effect in paediatric trials was not statistically significant (RR 0.89, 95% CI 0.58 to 1.39) and the confidence interval not only encompasses the limits of the estimate from the adult studies, it includes possibility of significant benefit of ICS alone over LABA in this age group. There were differences between usual and higher than licensed doses which reached statistical significance in favour of usual LABA doses (usual dose: RR 0.74 (95% CI 0.65 to 0.84) versus higher than usual dose: RR 1.10(95% CI 0.79 to 1.52, P = 0.03) (Analysis 2.5). The meta‐regression suggested the independent effect‐modifying effects of the LABA dose, baseline FEV1 and treatment duration, where higher than usual dose, higher baseline FEV1 or longer duration was associated with less benefit of combination therapy; all three variables were correlated (0.29 to ‐0.63).
2.5. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 5 # patients with exacerbations requiring oral steroids by whether LABA dose is usual or higher than usual.
There was no evidence of systematic bias identified by the test for funnel plot asymmetry (intercept 0.264, 95% CI ‐0.233 to 0.761). Since data for our primary outcome were not available in a number of full‐text articles and had to be requested through correspondence, we undertook a sensitivity analysis to assess the robustness of the result to data source. We removed studies which were only available as short reports from manufacturer trials registers, or for which we had to correspond to obtain relevant data, leaving only those studies for which data were available in a full‐text article. The resultant analysis restricted to published data gave a near identical result to the primary analysis (RR 0.77, 95% CI 0.67 to 0.88) (Analysis 2.13), although a funnel plot for this outcome did suggest some asymmetry without the unpublished data included (Figure 4). Since all but one trial contributing to the primary outcome was funded by manufacturers of LABA, we could not assess the impact of study sponsorship on the pooled effect size reliably.
2.13. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 13 Sensitivity analysis: exacerbations requiring oral steroids by data publication status (data available from published source).
4.

Funnel plot of outcome: 2.9 Sensitivity analysis: exacerbations requiring oral steroids by data publication status (data available from published source).
Secondary outcomes
Exacerbations requiring hospitalisation, serious adverse events and withdrawal
There was no significant group difference in the risk of exacerbations requiring admission to hospital (RR 1.13, 95% CI 0.70 to 1.82, 24 studies (of which 15 contribute numerical data)) (Figure 5).
5.

Forest plot of comparison: 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, outcome: 1.2 # patients with exacerbations requiring hospitalisation.
The risk of all‐cause serious adverse events (events requiring or prolonging hospital admission or causing death) was similar in the two treatment groups (RR 1.06, 95% CI 0.87 to 1.3, 53 studies (of which 48 contribute numerical data)) (Figure 6).
6.

Forest plot of comparison: 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, outcome: 1.3 Serious adverse event including respiratory.
The use of LABA significantly reduced the risk of overall withdrawals by 20% (all reasons included): RR 0.80 (95% CI 0.75 to 0.87, 53 studies) (Analysis 1.4) and reduced the risk of withdrawals due to poor asthma control by 50% (RR 0.50, 95% CI 0.41 to 0.61, 38 studies) (Analysis 1.5).
1.4. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 4 Total # withdrawals.
1.5. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 5 # withdrawals due to poor asthma control or exacerbation.
Lung function
The addition of LABA to ICS provided significantly greater improvement in lung function over using the same dose of ICS as monotherapy (outcomes Analysis 1.8 to Analysis 1.17), irrespective of whether the group differences were reported as endpoint or change from baseline: change from baseline in FEV1 in litres (0.11 L/sec, 95% CI 0.09 to 0.13, random‐effects model, 32 studies) (Analysis 1.9), change in percent predicted (3.73% predicted, 95% CI 2.66 to 4.8, random‐effects model, eight studies) (Analysis 1.10 ), FEV1 at endpoint in litres (0.12 L/sec, 95% CI 0.07 to 0.17, 10 studies) (Analysis 1.8), FEV1 percent predicted at endpoint (5.34% predicted, 95% CI 3.29 to 7.38, four studies)(Analysis 1.11), change from baseline in morning peak expiratory flow (PEF) (19.24 litres/min, 17.08 to 22.20, random‐effects model, 51 studies) (Analysis 1.14); or in evening PEF (17.89 litres/min, 95% CI 14.82 to 20.95, random‐effects model, 33 studies) (Analysis 1.16) and morning PEF at endpoint (26.21 litres/min, 95% CI 13.31 to 39.1, random‐effects model, eight studies) (Analysis 1.13). There were insufficient data (fewer than two trials) to aggregate the change in PEF variability or evening PEF at endpoint.
1.8. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 8 FEV1 (L) at endpoint.
1.17. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 17 Change in PEF variability at endpoint.
1.9. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 9 Change in FEV1 at endpoint stratifying on baseline FEV1.
1.10. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 10 Change in FEV1 predicted endpoint stratifying on baseline FEV1.
1.11. Analysis.
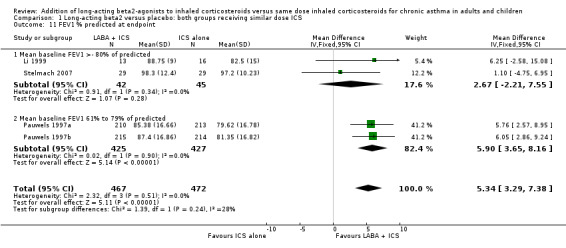
Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 11 FEV1 % predicted at endpoint.
1.14. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 14 Change in morning PEF (L/min) at endpoint stratifying on baseline FEV1.
1.16. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 16 Change in evening PEF at endpoint.
1.13. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 13 Morning PEF (L/min) at endpoint.
While subgroup analysis of the change in FEV1 with respect to duration of study (P = 0.67) (Analysis 1.12) did not identify statistically significant differences, the change in FEV1 was significantly influenced by baseline % predicted FEV1 with greater effect of LABA in patients with lowest baseline values (>= 80% predicted: 0.09 L (95% CI 0.03 to 0.14) versus 61% to 79% predicted: 0.12 L (95% CI 0.09 to 0.14, P = 0.03) (Analysis 1.9) .
1.12. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 12 Change in FEV1 (L or % predicted) stratifying on trial duration.
Symptoms and rescue medication use
Use of LABA significantly reduced daytime symptoms (SMD ‐0.33, 95% CI ‐0.42 to ‐0.23, eight studies) (Analysis 1.19); night‐time symptoms (SMD ‐0.22, 95% CI ‐0.33 to ‐0.11, five studies) (Analysis 1.20) and overall 24‐hour symptoms (SMD ‐0.23, 95% CI ‐0.34 to ‐0.12, six studies) (Analysis 1.18). The superiority of LABA and ICS over ICS monotherapy was also observed in the percent of symptom‐free days during the observation period (WMD 7.31, 95% CI 0.50 to 14.12, random‐effects model, six studies) (Analysis 1.21); the change from baseline in symptom‐free days (11.88%, 95% CI 8.25 to 15.50, random‐effects model, 16 studies) (Analysis 1.22) and in symptom‐free nights (SMD 0.51, 95% CI 0.28 to 0.74, random‐effects model, four studies) (Analysis 1.25). The favourable effect of LABA was observed in the change in "asthma‐control" days (15.81%, 95% CI 10.85 to 20.77, four studies) (Analysis 1.26). There were no significant group differences for the change in percent nights with no awakening (1.01%, 95% CI ‐1.06 to 3.08, five studies) (Analysis 1.35) and in night‐time awakening (SMD ‐0.10, 95% CI ‐0.21 to 0.01, five studies) (Analysis 1.37).
1.19. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 19 Change in daytime symptom score at endpoint.
1.20. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 20 Change in night‐time symptom score at endpoint.
1.18. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 18 Change in 24‐hour symptom score at endpoint.
1.21. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 21 % symptom‐free days.
1.22. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 22 Change in % symptom‐free days at endpoint.
1.25. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 25 Change in % symptom‐free nights at endpoint.
1.26. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 26 Change in asthma control days %.
1.35. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 35 Change in % nights with no awakening.
1.37. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 37 Change in night‐time awakening (number of nights) at endpoint.
The addition of LABA to ICS also reduced the need for rescue short‐acting ß2‐agonists whether reported as daytime use at endpoint (‐0.73 puffs/day, 95% CI ‐1.24 to ‐0.22, random‐effects model, two studies) (Analysis 1.29); night‐time use at endpoint (‐0.44 puffs/night, 95% CI ‐0.81 to ‐0.07, random‐effects model, two studies) (Analysis 1.30); change in overall 24‐hour use (‐0.58 puffs/24 hours, 95% CI ‐0.80 to ‐0.35, random‐effects model, 14 studies) (Analysis 1.27); change in night‐time use (‐0.3 puffs/night, 95% CI ‐0.48 to ‐0.11, random‐effects model, seven studies, Analysis 1.31); change in rescue‐free days (6.43%, 95% CI 1.2 to 11.66, two studies) (Analysis 1.34) or change in daytime use (‐0.68 puffs/day, 95% CI ‐0.94 to ‐0.42, random‐effects model, 13 studies) (Analysis 1.28 ). The change in mean rescue‐free days (17.05%, 95% CI 13.75 to 20.35, six studies) (Analysis 1.32) and in quality of life (as measured by the AQLQ) also favoured LABA (0.26, 95% CI 0.04 to 0.47, random‐effects model, three studies) (Analysis 1.38). There was no group difference in the percent of nights with awakening (WMD ‐1.37, 95% CI ‐2.75 to 0.02, fixed‐effect model, two studies) (Analysis 1.36).
1.29. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 29 # daytime rescue inhalations (puffs per day) at endpoint.
1.30. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 30 # night‐time rescue inhalations (puffs per night) at endpoint.
1.27. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 27 Change in # overall daily rescue inhalations at endpoint.
1.31. Analysis.
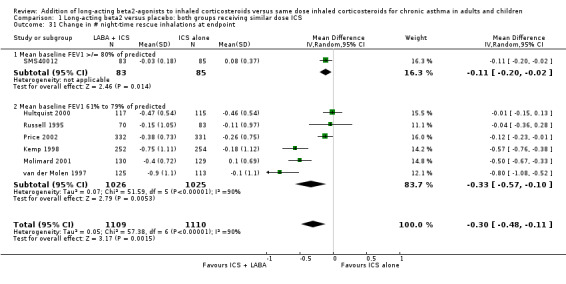
Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 31 Change in # night‐time rescue inhalations at endpoint.
1.34. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 34 Change in % rescue medication‐free days.
1.28. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 28 Change in # daytime rescue inhalations at endpoint.
1.32. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 32 Change in mean rescue‐free days.
1.38. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 38 Change in quality of life (AQLQ score) at endpoint.
1.36. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 36 % nights with awakening.
Non‐serious adverse events
There was no apparent group difference in the risk of overall adverse effects (RR 1.00, 95% CI 0.97 to 1.04, 41 studies) (Analysis 1.39), meeting our a priori defined limits of equivalence. There was also no group difference in the risk of specific side effects including headache (RR 0.99, 95% CI 0.87 to 1.13, 37 studies) (Analysis 1.40); hoarseness (RR 1.17, 95% CI 0.44 to 3.1, random‐effects model, six studies) (Analysis 1.41); oral thrush (RR 1.65, 95% CI 0.71 to 3.86, nine studies) (Analysis 1.42); tachycardia or palpitations (RR 2.11, 95% CI 0.83 to 5.37, 12 studies) (Analysis 1.44); cardiovascular adverse effects such as chest pain (RR 0.90, 95% CI 0.32 to 2.54, four studies) (Analysis 1.46) or tremor (RR 1.74, 95% CI 0.72 to 4.20, random‐effects model, 16 studies) (Analysis 1.43). There was no statistically significant difference from three studies reporting death (RR 2.46, 95% CI 0.48 to 12.65) (Analysis 1.45). However, the wide confidence interval (including the upper limit) for some adverse events was high for tachycardia, palpitations, tremor and death, indicating uncertainty. More dramatic was the scarce documentation of the impact on growth (in children), adrenal function and bone mineral density, preventing any aggregation due to the paucity (0 to 2) of trials measuring or reporting these outcomes. Withdrawal due to adverse events showed no significant difference between treatment options (RR 1.04, 95% 0.86 to 1.26, 52 studies) (Analysis 1.6).
1.39. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 39 Total # adverse events.
1.40. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 40 # patients with headache.
1.41. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 41 # patients with hoarseness.
1.42. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 42 # patients with oral thrush.
1.44. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 44 # patients with tachycardia or palpitations.
1.46. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 46 # patients with adverse cardiovascular events.
1.43. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 43 # patients with tremor.
1.45. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 45 Deaths.
1.6. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 6 # withdrawals due to adverse events.
Discussion
The strength of the evidence allows us to confirm the efficacy of adding a long‐acting ß2‐agonist (LABA) to inhaled corticosteroids (ICS) in reducing the risk of exacerbations requiring rescue oral corticosteroids in adults. In children the evidence in favour of LABAs is far less certain, with wide confidence intervals including both superiority and inferiority of LABA to ICS alone. The studies have largely recruited adults and older children with suboptimal asthma control on monotherapy with inhaled corticosteroids. The addition of a LABA to ICS reduced the relative risk of patients requiring oral steroids for an asthma exacerbation by 23% (from 15% to 11%) in studies of four to 54 weeks duration. The efficacy of adding a LABA to inhaled steroids was also supported by several secondary outcomes, namely the significantly greater improvement in FEV1 (by 110 ml) and morning PEF (20 litres/min), in symptom‐free days (12%), in rescue‐free days (6%) and a reduction by half in the risk of withdrawal due to poor asthma control compared to ICS monotherapy. The addition of a LABA was not associated with an increase in serious adverse events (SAEs), although the width of the confidence interval was large and could not exclude as much as a 30% increase in risk or a 13% reduction in risk of SAEs. There was no group difference in any documented specific adverse events with the wide confidence intervals around the estimates of risk for tachycardia or palpitation and death, indicating remaining uncertainty. However, the overall relative risk of adverse events between groups reached our a priori definition of equivalence, and there was no group difference in the rate of withdrawals due to side effects.
There was no evidence of statistical heterogeneity between trials in the primary outcome, despite the inclusion of populations of different ages, baseline severity of airway obstruction, use of different ICS doses and different LABAs. The overall number needed to treat (NNT) was 41 patients. We do acknowledge variation of baseline risk of rescue oral steroids between the control groups. When baseline risk is broken down further by quartile (see Results), the highest NNT of 435 was estimated for studies with the lowest baseline risk (between 0% and 2%). These studies had a short duration (four weeks was the median), although mean baseline FEV1 was the lowest for all of the risk quartiles at 70%. As event rates in the control group increased across the medium and high‐risk quartiles, the NNTs fell to 109, 32 and 17 respectively. Although the median study durations for studies in these risk quartiles were longer at 12, 26 and 24 weeks respectively, and the mean FEV1 was higher (77%, 76% and 75 % predicted respectively), these two factors probably do not well identify the risk groups defined by the baseline rate of exacerbations. In view of the wide confidence interval from the paediatric studies the similarity of the subgroup estimates for the primary outcome could reflect lower statistical power for the studies in this subgroup than for the adult studies, with children representing less than 10% of the overall number of study participants (605 children versus 6203 adults).
While the results seem to apply to particularly to adults, irrespective of baseline characteristics and variation of intervention, the meta‐regression and subgroup analyses both suggested that higher than usual dose of LABA significantly reduced the beneficial effect of LABA. Perhaps other factors such as baseline FEV1 and treatment duration modify the magnitude of effect but until further confirmation it is safe to assume that the addition of LABA to ICS probably yields similar benefit irrespective of the baseline FEV1, starting dose of ICS, type of LABA, number of devices to deliver the combination therapy or treatment duration. Whether the addition of LABA to ICS is superior to increasing the dose of ICS is addressed in another review (Ducharme 2010).
The improvement in lung function with the use of LABAs might be anticipated from their physiologic action, although most studies obtained these measurements at the trough of the dosage interval (12 hours or more after the last LABA inhalation). Improvements were seen in all lung function tests (FEV1 and PEF) whether measured in the respiratory laboratory or at home, in the morning or evening. While the addition of LABA reduced the need for rescue short‐acting ß2‐agonists slightly more than ICS, the effect was small: an average reduction of 0.58 puffs per 24 hours, ‐0.3 puffs/night and ‐0.68 puffs/day and a 6% increase in the percentage of rescue‐free days. Similarly, a difference in the improvement of symptoms with LABAs was present but modest (SMD of between ‐0.2 to ‐0.3 for changes in symptom scores measured over 24 hours, day only or night only). Perhaps study eligibility criteria, which cited a requirement for frequent but not daily symptoms and short‐acting ß2‐agonist use during the run‐in periods, explain this phenomenon. An alternative explanation may lie in the effectiveness of inhaled steroids alone for reducing symptoms and short‐acting ß2‐agonist use (Adams 2008; Manning 2008), perhaps magnified by enhanced compliance to inhaled corticosteroids in the context of these studies.
When used without ICS, salmeterol and formoterol increase the risk of serious adverse events (Cates 2008a; Cates 2008b). In this review, the outcomes relating to hospital admission and all cause serious adverse events did not provide evidence that the combination of use of LABAs and ICS increased the risk of these serious events. Our findings lend qualified support to the commentary by Ernst and colleagues (Ernst 2006), which attributed the adverse effect of LABA reported by Salpeter 2006 to its use without concomitant ICS. Due to the rarity of these events in the included studies, our pooled estimates were imprecise and the confidence intervals are not narrow enough to exclude either protection or harm confidently. This finding is concordant with separate analysis of salmeterol and formoterol as an additive treatment to ICS (Cates 2009a; Cates 2009b). Although the large confidence intervals around some other specific adverse effects highlight remaining uncertainties, the equivalence between groups in overall adverse effects is reassuring. Moreover, the absence of group difference in withdrawal due to adverse effects provides some support to the safety of adding LABA to ICS, when used up to 52 weeks.
The absence of data on airway inflammation that could be aggregated was disappointing. The concern that use of a LABA masks symptoms of poor asthma control and lead to deterioration of the airways is not supported by the evidence in our review. However, such a concern pertains to using a LABA as a steroid‐sparing strategy, when used in combination with a lower dose of inhaled steroids than in the comparison group; in other words, when the dose of inhaled corticosteroids is not equivalent to both treatment options. With similar improvement in FEV1 irrespective of study duration, there was no evidence of tachyphylaxis associated with prolonged use of LABA.
Our review provides complementary information to other reviews examining the overall efficacy of LABA in paediatrics (Bisgaard 2003) and in adults (Walters 2007) when used as monotherapy and/or inconsistent co‐treatment with ICS. There is enough power in our primary outcome to conclude firmly the efficacy of LABA as add‐on to ICS when compared to a similar dose of ICS as monotherapy in adults: 222 studies with negative findings would be needed to reverse this finding. The efficacy of adding LABA to ICS applies to adults who are symptomatic on a ICS, as low as 200 µg/day of beclomethasone or equivalent. In view of the subgroup result for paediatric studies we cannot currently be sure how the overall effect applies to children, and the priority remains for researchers to generate a more definitive evidence base for the effects of this strategy in children.
The generalisability of the findings to a clinic population must be considered with care. One of the main eligibility criteria of the studies was the presence of significant (12% to 15% or more) reversibility in FEV1 with a ß2‐agonist. However, such reversibility is demonstrated in less than 10% of patients at a given point in time (Storms 2003). Major exclusion criteria included smoking, pregnancy or lactation, as well as childbearing age without appropriate contraception. This may have excluded up to a half of our usual clinic patients. Finally, patients with severe airway obstruction, recent exacerbations, or both, were generally excluded. To how many of our patients would the results of these aggregated trials apply? Unfortunately only a limited amount of data were presented in the trials on the proportion of participants randomised to those screened for enrolment in the run‐in period. Only 34% of studies reported the proportion of patients enrolled in the run‐in period that were successfully randomised (varying between 43% and 95%). There was little reporting of adherence to treatment during the intervention period, mentioned in only 13 studies, with no adjustment or stratification in the analyses. Whether treatment with LABA plus ICS leads to improved compliance and thus better asthma control than ICS monotherapy could not be assessed in this review. The results of this review may not be generalised to the majority of our patients and in particular those with symptoms but poor reversibility in FEV1.
Whether the addition of a LABA is more effective and safer than increasing the dose of inhaled corticosteroids (Ducharme 2010), adding anti‐leukotrienes (Ducharme 2006) or whether it exerts a steroid‐sparing effect (Gibson 2005) are addressed in other Cochrane Reviews.
Authors' conclusions
Implications for practice.
In symptomatic adults with mild to moderate airway obstruction, who remain symptomatic despite a low, moderate or high dose of inhaled corticosteroids, the addition of a long‐acting beta2‐agonist at licensed doses is superior for reducing the rate of exacerbations requiring oral steroids, and for improving lung function, symptoms and quality of life, than remaining on similar doses of inhaled corticosteroids as monotherapy. There is little evidence to support this treatment option in children as a means of reducing requirement for oral steroids, precluding firm recommendations regarding the use of LABA for children. The available evidence indicates that the risk of serious adverse events between treatments is not statistically significant, although imprecision of the estimate includes the possibility of both an increase and a decrease in the risk of serious adverse events with the addition of a LABA in all age groups.
Implications for research.
Given the nature of the evidence, preschool‐aged children and school‐aged children warrant further investigation. Similarly, patients who are symptomatic on inhaled steroids, despite good compliance, but with little airway reversibility to short‐acting ß2‐agonists should be targeted for inclusion in future studies. Stratified subgroup analyses on baseline FEV1 and reporting effect size at different points in time would be useful to explore the potential modifying effect of these factors on response to therapy.
Future trials should be designed to take account of the following:
double‐blinding, adequate randomisation and complete reporting of withdrawals and drop‐outs with intention‐to‐treat analysis;
an intervention period of 12 weeks or more to assess properly the impact on exacerbations requiring oral corticosteroids;
clear reporting of the percent (and reasons) of non‐eligibility of approached patients and of those enrolled in the run‐in period;
complete reporting of continuous (denominators, mean change and mean standard deviation of change) and dichotomous (denominators and rate) data.
Outcomes of particular importance to assess include:
exacerbations requiring oral corticosteroids, to examine the effect of different patient characteristics, such as baseline lung function and baseline dose of ICS on this outcome;
careful monitoring and reporting of compliance to ICS prior to randomisation and to ICS and LABA post‐randomisation. The impact of compliance to combination therapy versus ICS monotherapy on the magnitude of the effect size should be examined;
reporting of the cost‐effectiveness of use of combination inhalers as compared to inhaled corticosteroids monotherapy;
long‐term side effects of long‐acting ß2‐agonists.
What's new
| Date | Event | Description |
|---|---|---|
| 19 June 2008 | New citation required and conclusions have changed | 44 new studies included; additional unpublished data available for primary outcome which had the effect of narrowing the confidence intervals in adults. |
| 2 May 2008 | New search has been performed | New literature search performed. |
History
Protocol first published: Issue 1, 2000 Review first published: Issue 4, 2005
| Date | Event | Description |
|---|---|---|
| 30 April 2008 | Amended | Converted to new review format. |
| 24 June 2005 | New citation required and conclusions have changed | Substantive amendment. |
Acknowledgements
We thank the Cochrane Airways Group, namely Stephen Milan, Karen Blackhall, Liz Arnold, Susan Hansen, Veronica Stewart and Bettina Reuben for the literature search and ongoing support; and Christopher Cates, Peter Gibson and Paul Jones for their constructive comments. We are indebted to the following corresponding authors or sponsors, namely G Boyd, X Li, A Tal, E Simons, F Price, JP Kemp, RA Pauwels, G Russell, Van der Molen and AAPH Verberne, J Holbrook, as well as to Richard Follows, Shailesh Patel, Karen Richardson, and Rob Pearson from GlaxoSmithKline, Robin von Maltzan, Steve Edwards, Nils Grundstrom and Roger Metcalf from AstraZeneca who assisted us in obtaining unpublished data and information from studies funded by their respective companies. We are indebted to Jenny Bellorini who copyedited the review.
Appendices
Appendix 1. Archive of methodological approach to study quality assessment (1999 to 2004)
Studies to be included underwent quality assessment, performed independently by two review authors, using two methods. First, using the Cochrane approach to assess allocation of concealment, trials were scored using the following principles.
Grade A: adequate concealment. Grade B: unclear concealment. Grade C: clearly inadequate concealment.
In addition, each study was assessed using a 0 to 5 scale described by Jadad (1995) and summarised as follows. 1. Was the study described as randomised (yes = 1; no = 0)? 2. Was the study described as double‐blind (yes = 1; no = 0)? 3. Was there a description of withdrawals and drop‐outs (yes = 1; no = 0)? 4. Was the method of randomisation well‐described and appropriate (yes = 1; no = 0)? 5. Was the method of double‐blinding well‐described and appropriate (yes = 1; no = 0)? 6. Deduct one point if methods for randomisation or blinding were inappropriate.
Appendix 2. Randomisation procedures for GSK studies
The procedures for randomising GSK sponsored studies have been detailed in correspondence between Richard Follows and TL, the details of which are given below:
The randomisation software is a computer‐generated, centralised programme (RandAll). After verification that the randomisation sequence is suitable for the study design (cross‐over, block or stratification), Clinical Supplies then package the treatments according the randomisation list generated. Concealment of allocation is maintained by a third party, since the sites phone in and are allocated treatments on that basis. Alternatively a third party may dispense the drug at the sites. Unblinding of data for interim analyses can only be done through RandAll, and are restricted so that only those reviewing the data are unblinded to treatment group allocation.
Data and analyses
Comparison 1. Long‐acting beta2 versus placebo: both groups receiving similar dose ICS.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 # patients with exacerbations requiring oral steroids | 30 | 6808 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.68, 0.87] |
| 1.1 Mean baseline FEV1 >/= 80% of predicted | 8 | 1713 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.70 [0.56, 0.86] |
| 1.2 Mean baseline FEV1 61% to 79% of predicted | 18 | 4095 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.81 [0.70, 0.94] |
| 1.3 Mean baseline FEV1 not reported | 4 | 1000 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.24, 1.65] |
| 2 # patients with exacerbations requiring hospitalisation | 24 | 7297 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.70, 1.82] |
| 2.1 Mean baseline FEV1 >/= 80% of predicted | 3 | 193 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.18, 5.39] |
| 2.2 Mean baseline FEV1 61% to 79% of predicted | 18 | 5685 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.11 [0.67, 1.84] |
| 2.3 Mean baseline FEV1 not reported | 3 | 1419 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.12, 72.71] |
| 3 Serious adverse event including respiratory | 57 | 16213 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.87, 1.30] |
| 3.1 Mean baseline FEV1 >/= 80% of predicted | 14 | 4219 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.64, 1.21] |
| 3.2 Mean baseline FEV1 61% to 79% of predicted | 32 | 8397 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.20 [0.89, 1.62] |
| 3.3 Mean baseline FEV1 not reported | 11 | 3597 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.18 [0.74, 1.87] |
| 4 Total # withdrawals | 58 | 14718 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.75, 0.87] |
| 4.1 Mean baseline FEV1 >/= 80% of predicted | 16 | 2501 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.67, 1.04] |
| 4.2 Mean baseline FEV1 61% to 79% of predicted | 35 | 9644 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.75, 0.90] |
| 4.3 Mean baseline FEV1 not reported | 7 | 2573 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.60, 0.85] |
| 5 # withdrawals due to poor asthma control or exacerbation | 38 | 9505 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.50 [0.41, 0.61] |
| 5.1 Mean baseline FEV1 >/= 80% of predicted | 6 | 596 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.20, 1.17] |
| 5.2 Mean baseline FEV1 61% to 79% of predicted | 27 | 6879 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.42, 0.66] |
| 5.3 Mean baseline FEV1 not reported | 5 | 2030 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.40 [0.24, 0.66] |
| 6 # withdrawals due to adverse events | 52 | 14038 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.86, 1.26] |
| 6.1 Mean baseline FEV1 >/= 80% of predicted | 9 | 1647 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.74 [0.41, 1.34] |
| 6.2 Mean baseline FEV1 61% to 79% of predicted | 35 | 9199 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.14 [0.90, 1.45] |
| 6.3 Mean baseline FEV1 not reported | 9 | 3192 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.62, 1.36] |
| 7 # withdrawals due to serious non‐respiratory event | 2 | Risk Ratio (M‐H, Random, 95% CI) | Totals not selected | |
| 7.1 Mean baseline FEV1 61% to 79% of predicted | 2 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 FEV1 (L) at endpoint | 10 | 2045 | Mean Difference (IV, Fixed, 95% CI) | 0.12 [0.07, 0.17] |
| 8.1 Mean baseline FEV1 >/‐ 80% of predicted | 2 | 615 | Mean Difference (IV, Fixed, 95% CI) | 0.10 [0.03, 0.17] |
| 8.2 Mean baseline FEV1 61% to 79% of predicted | 6 | 914 | Mean Difference (IV, Fixed, 95% CI) | 0.15 [0.07, 0.22] |
| 8.3 Mean baseline FEV1 not reported | 2 | 516 | Mean Difference (IV, Fixed, 95% CI) | 0.12 [‐0.02, 0.26] |
| 9 Change in FEV1 at endpoint stratifying on baseline FEV1 | 32 | 9784 | L (Random, 95% CI) | 0.11 [0.09, 0.13] |
| 9.1 Mean baseline FEV1 >= 80% of predicted | 5 | 1036 | L (Random, 95% CI) | 0.09 [0.03, 0.14] |
| 9.2 Mean baseline FEV1 61% to 79% of predicted | 24 | 7917 | L (Random, 95% CI) | 0.12 [0.09, 0.14] |
| 9.3 Mean baseline FEV1 predicted not reported | 3 | 831 | L (Random, 95% CI) | 0.13 [0.05, 0.21] |
| 10 Change in FEV1 predicted endpoint stratifying on baseline FEV1 | 8 | % (Random, 95% CI) | 3.73 [2.66, 4.80] | |
| 10.1 Mean baseline FEV1 >= 80% of predicted | 6 | % (Random, 95% CI) | 4.06 [2.96, 5.16] | |
| 10.2 Mean baseline FEV1 61% to 79% of predicted | 1 | % (Random, 95% CI) | 3.46 [1.40, 5.52] | |
| 10.3 Mean baseline FEV1 predicted not reported | 1 | % (Random, 95% CI) | ‐0.40 [‐5.03, 4.23] | |
| 11 FEV1 % predicted at endpoint | 4 | 939 | Mean Difference (IV, Fixed, 95% CI) | 5.34 [3.29, 7.38] |
| 11.1 Mean baseline FEV1 >‐ 80% of predicted | 2 | 87 | Mean Difference (IV, Fixed, 95% CI) | 2.67 [‐2.21, 7.55] |
| 11.2 Mean baseline FEV1 61% to 79% of predicted | 2 | 852 | Mean Difference (IV, Fixed, 95% CI) | 5.90 [3.65, 8.16] |
| 12 Change in FEV1 (L or % predicted) stratifying on trial duration | 14 | 4008 | Std. Mean Difference (IV, Random, 95% CI) | 0.34 [0.26, 0.42] |
| 12.1 Change in FEV1 (L) or (% predicted) at 6 +/‐ 2 weeks of treatment | 2 | 299 | Std. Mean Difference (IV, Random, 95% CI) | 0.41 [0.18, 0.64] |
| 12.2 Change in FEV1 (L) or (% predicted) at 12 +/‐ 4 weeks of treatment | 11 | 2003 | Std. Mean Difference (IV, Random, 95% CI) | 0.36 [0.24, 0.49] |
| 12.3 Change in FEV1 (L) or (% predicted) at 24 +/‐ 4 weeks of treatment | 2 | 352 | Std. Mean Difference (IV, Random, 95% CI) | 0.30 [0.09, 0.51] |
| 12.4 Change in FEV1 (L) or (% predicted) at 52 +/‐ 4 weeks of treatment | 3 | 1354 | Std. Mean Difference (IV, Random, 95% CI) | 0.28 [0.12, 0.44] |
| 13 Morning PEF (L/min) at endpoint | 8 | 1787 | Mean Difference (IV, Random, 95% CI) | 26.21 [13.31, 39.10] |
| 13.1 Mean baseline FEV1 >/= 80% | 1 | 29 | Mean Difference (IV, Random, 95% CI) | 86.0 [17.11, 154.89] |
| 13.2 Mean baseline FEV1 61% to 79% of predicted | 5 | 1127 | Mean Difference (IV, Random, 95% CI) | 19.14 [2.93, 35.34] |
| 13.3 Mean baseline FEV1 not reported | 2 | 631 | Mean Difference (IV, Random, 95% CI) | 34.26 [17.83, 50.70] |
| 14 Change in morning PEF (L/min) at endpoint stratifying on baseline FEV1 | 53 | 14365 | L/min (Random, 95% CI) | 19.64 [17.08, 22.20] |
| 14.1 Mean baseline FEV1 >/= 80% of predicted | 12 | 3364 | L/min (Random, 95% CI) | 11.96 [8.68, 15.24] |
| 14.2 Mean baseline FEV1 61% to 79% of predicted | 32 | 8348 | L/min (Random, 95% CI) | 23.41 [19.84, 26.98] |
| 14.3 Mean baseline FEV1 not reported | 9 | 2653 | L/min (Random, 95% CI) | 17.09 [12.99, 21.18] |
| 15 Evening PEF (L/min) at endpoint | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 15.1 Mean baseline FEV1 61% to 79% of predicted | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 16 Change in evening PEF at endpoint | 33 | 8248 | L/min (Random, 95% CI) | 17.89 [14.82, 20.95] |
| 16.1 Mean baseline FEV1 > /= 80% of predicted | 7 | 1345 | L/min (Random, 95% CI) | 13.37 [5.98, 20.76] |
| 16.2 Mean baseline FEV1 61% to 79% of predicted | 23 | 6058 | L/min (Random, 95% CI) | 19.70 [16.36, 23.03] |
| 16.3 Mean baseline FEV1 not reported | 3 | 845 | L/min (Random, 95% CI) | 13.85 [5.05, 22.64] |
| 17 Change in PEF variability at endpoint | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 17.1 Mean baseline FEV1 61% to 79% of predicted | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 18 Change in 24‐hour symptom score at endpoint | 6 | 1473 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.23 [‐0.34, ‐0.12] |
| 18.1 Mean baseline FEV1 61% to 79% of predicted | 6 | 1473 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.23 [‐0.34, ‐0.12] |
| 19 Change in daytime symptom score at endpoint | 8 | 1767 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.33 [‐0.42, ‐0.23] |
| 19.1 Mean baseline FEV1 >/= 80% | 1 | 54 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.27 [‐0.80, 0.27] |
| 19.2 Mean baseline FEV1 61% to 79% of predicted | 7 | 1713 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.33 [‐0.43, ‐0.23] |
| 20 Change in night‐time symptom score at endpoint | 5 | 1319 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.22 [‐0.33, ‐0.11] |
| 20.1 Mean baseline FEV1 >/= 80% | 1 | 54 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.53 [‐1.08, 0.01] |
| 20.2 Mean baseline FEV 1 61‐79% of predicted | 4 | 1265 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.21 [‐0.32, ‐0.10] |
| 21 % symptom‐free days | 6 | 2169 | Mean Difference (IV, Random, 95% CI) | 7.31 [0.50, 14.12] |
| 21.1 Mean baseline FEV1 >/= 80% of predicted | 1 | 627 | Mean Difference (IV, Random, 95% CI) | 4.60 [0.69, 8.51] |
| 21.2 Mean baseline FEV1 61% to 79% of predicted | 4 | 1460 | Mean Difference (IV, Random, 95% CI) | 10.35 [0.05, 20.65] |
| 21.3 Mean baseline FEV1 not reported | 1 | 82 | Mean Difference (IV, Random, 95% CI) | ‐1.0 [‐4.47, 2.47] |
| 22 Change in % symptom‐free days at endpoint | 16 | 4186 | Mean Difference (IV, Random, 95% CI) | 11.88 [8.25, 15.50] |
| 22.1 Mean baseline FEV1 >/= 80% of predicted | 1 | 203 | Mean Difference (IV, Random, 95% CI) | 3.20 [‐8.08, 14.48] |
| 22.2 Mean baseline FEV1 61% to 79% of predicted | 13 | 3344 | Mean Difference (IV, Random, 95% CI) | 13.37 [9.31, 17.44] |
| 22.3 Mean baseline FEV1 not reported | 2 | 639 | Mean Difference (IV, Random, 95% CI) | 6.57 [1.11, 12.03] |
| 23 Change in # of symptom‐free nights at endpoint | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 23.1 Mean baseline FEV1 61% to 79% of predicted | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 24 % symptom‐free nights at 12 +/‐ 4 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 24.1 Mean baseline FEV1 61% to 79% of predicted | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 25 Change in % symptom‐free nights at endpoint | 4 | 1052 | Std. Mean Difference (IV, Random, 95% CI) | 0.51 [0.28, 0.74] |
| 25.1 Mean baseline FEV1 61% to 79% of predicted | 4 | 1052 | Std. Mean Difference (IV, Random, 95% CI) | 0.51 [0.28, 0.74] |
| 26 Change in asthma control days % | 4 | 813 | Mean Difference (IV, Fixed, 95% CI) | 15.81 [10.85, 20.77] |
| 26.1 Mean baseline FEV1 61% to 79% of predicted | 4 | 813 | Mean Difference (IV, Fixed, 95% CI) | 15.81 [10.85, 20.77] |
| 27 Change in # overall daily rescue inhalations at endpoint | 14 | 4654 | Mean Difference (IV, Random, 95% CI) | ‐0.58 [‐0.80, ‐0.35] |
| 27.1 Mean baseline FEV1 >/= 80% of predicted | 2 | 1272 | Mean Difference (IV, Random, 95% CI) | ‐0.17 [‐0.29, ‐0.05] |
| 27.2 Mean baseline FEV1 61% to 79% of predicted | 12 | 3382 | Mean Difference (IV, Random, 95% CI) | ‐0.73 [‐1.05, ‐0.41] |
| 28 Change in # daytime rescue inhalations at endpoint | 13 | puffs per day (Random, 95% CI) | ‐0.68 [‐0.94, ‐0.42] | |
| 28.1 Mean baseline FEV 1 >/= 80% of predicted | 3 | puffs per day (Random, 95% CI) | ‐0.27 [‐0.62, 0.07] | |
| 28.2 Mean baseline FEV1 61% to 79% of predicted | 10 | puffs per day (Random, 95% CI) | ‐0.82 [‐1.18, ‐0.46] | |
| 28.3 Mean baseline FEV1 not reported | 0 | puffs per day (Random, 95% CI) | 0.0 [0.0, 0.0] | |
| 29 # daytime rescue inhalations (puffs per day) at endpoint | 2 | 277 | Mean Difference (IV, Random, 95% CI) | ‐0.73 [‐1.24, ‐0.22] |
| 29.1 Mean baseline FEV1 61% to 79% of predicted | 2 | 277 | Mean Difference (IV, Random, 95% CI) | ‐0.73 [‐1.24, ‐0.22] |
| 30 # night‐time rescue inhalations (puffs per night) at endpoint | 2 | 546 | Mean Difference (IV, Random, 95% CI) | ‐0.44 [‐0.81, ‐0.07] |
| 30.1 Mean baseline FEV1 61% to 79% of predicted | 2 | 546 | Mean Difference (IV, Random, 95% CI) | ‐0.44 [‐0.81, ‐0.07] |
| 31 Change in # night‐time rescue inhalations at endpoint | 7 | 2219 | Mean Difference (IV, Random, 95% CI) | ‐0.30 [‐0.48, ‐0.11] |
| 31.1 Mean baseline FEV1 >/= 80% of predicted | 1 | 168 | Mean Difference (IV, Random, 95% CI) | ‐0.11 [‐0.20, ‐0.02] |
| 31.2 Mean baseline FEV1 61% to 79% of predicted | 6 | 2051 | Mean Difference (IV, Random, 95% CI) | ‐0.33 [‐0.57, ‐0.10] |
| 32 Change in mean rescue‐free days | 6 | 1698 | % (Fixed, 95% CI) | 17.05 [13.75, 20.35] |
| 32.1 Mean baseline FEV1 61% to 79% of predicted | 5 | 1381 | % (Fixed, 95% CI) | 17.63 [14.03, 21.23] |
| 32.2 Mean baseline FEV1 not reported | 1 | 317 | % (Fixed, 95% CI) | 14.0 [5.77, 22.23] |
| 33 Rescue medication‐free days | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 33.1 Mean baseline FEV1 61% to 79% of predicted | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 34 Change in % rescue medication‐free days | 2 | 667 | Mean Difference (IV, Random, 95% CI) | 6.43 [1.20, 11.66] |
| 34.1 Mean baseline FEV1 61% to 79% of predicted | 1 | 475 | Mean Difference (IV, Random, 95% CI) | 5.20 [‐1.78, 12.18] |
| 34.2 Mean baseline FEV1 not reported | 1 | 192 | Mean Difference (IV, Random, 95% CI) | 8.0 [0.10, 15.90] |
| 35 Change in % nights with no awakening | 5 | 1158 | Mean Difference (IV, Fixed, 95% CI) | 1.01 [‐1.06, 3.08] |
| 35.1 Mean baseline FEV1 61% to 79% of predicted | 5 | 1158 | Mean Difference (IV, Fixed, 95% CI) | 1.01 [‐1.06, 3.08] |
| 36 % nights with awakening | 2 | 913 | Mean Difference (IV, Fixed, 95% CI) | ‐1.37 [‐2.75, 0.02] |
| 36.1 Mean baseline FEV1 >/= 80% of predicted | 1 | 627 | Mean Difference (IV, Fixed, 95% CI) | ‐1.50 [‐3.19, 0.19] |
| 36.2 Mean baseline FEV1 61% to 79% of predicted | 1 | 286 | Mean Difference (IV, Fixed, 95% CI) | ‐1.10 [‐3.51, 1.31] |
| 37 Change in night‐time awakening (number of nights) at endpoint | 5 | 1308 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.10 [‐0.21, 0.01] |
| 37.1 Mean baseline FEV1 61% to 79% of predicted | 5 | 1308 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.10 [‐0.21, 0.01] |
| 38 Change in quality of life (AQLQ score) at endpoint | 3 | Mean Difference (Random, 95% CI) | 0.26 [0.04, 0.47] | |
| 38.1 Mean baseline FEV1 61% to 79% of predicted | 3 | Mean Difference (Random, 95% CI) | 0.26 [0.04, 0.47] | |
| 39 Total # adverse events | 41 | 10622 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.00 [0.97, 1.04] |
| 39.1 Mean baseline FEV1 >/= 80% of predicted | 7 | 1424 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.09 [1.01, 1.19] |
| 39.2 Mean baseline FEV1 61% to 79% of predicted | 25 | 6555 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.94, 1.02] |
| 39.3 Mean baseline FEV1 not reported | 9 | 2643 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.01 [0.92, 1.10] |
| 40 # patients with headache | 37 | 10020 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.87, 1.13] |
| 40.1 Mean baseline FEV1 >/= 80% of predicted | 4 | 779 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.78, 1.33] |
| 40.2 Mean baseline FEV1 61% to 79% of predicted | 26 | 6644 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.81, 1.13] |
| 40.3 Mean baseline FEV1 not reported | 7 | 2597 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.12 [0.74, 1.70] |
| 41 # patients with hoarseness | 6 | 1602 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.44, 3.10] |
| 41.1 Mean baseline FEV1 61% to 79% of predicted | 5 | 1284 | Risk Ratio (M‐H, Random, 95% CI) | 1.02 [0.36, 2.88] |
| 41.2 Mean baseline FEV1 not reported | 1 | 318 | Risk Ratio (M‐H, Random, 95% CI) | 5.0 [0.24, 103.33] |
| 42 # patients with oral thrush | 9 | 1379 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.65 [0.71, 3.86] |
| 42.1 FEV1 >/= 80% predicted | 3 | 356 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.04 [0.46, 35.52] |
| 42.2 Mean baseline FEV1 61% to 79% of predicted | 6 | 1023 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.34 [0.52, 3.46] |
| 43 # patients with tremor | 16 | 3833 | Risk Ratio (M‐H, Random, 95% CI) | 1.74 [0.72, 4.20] |
| 43.1 Mean baseline FEV1 >/= 80% of predicted | 2 | 530 | Risk Ratio (M‐H, Random, 95% CI) | 5.30 [0.26, 109.66] |
| 43.2 Mean baseline FEV1 61% to 79% of predicted | 14 | 3303 | Risk Ratio (M‐H, Random, 95% CI) | 1.63 [0.64, 4.15] |
| 44 # patients with tachycardia or palpitations | 12 | 3491 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.11 [0.83, 5.37] |
| 44.1 Mean baseline FEV1 >/= 80% of predicted | 1 | 116 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 44.2 Mean baseline FEV1 61% to 79% of predicted | 10 | 2464 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.13 [0.77, 5.88] |
| 44.3 Mean baseline FEV1 not reported | 1 | 911 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.00 [0.18, 22.03] |
| 45 Deaths | 3 | 1673 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.46 [0.48, 12.65] |
| 45.1 Mean baseline FEV1 61% to 79% of predicted | 1 | 336 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.90 [0.12, 70.57] |
| 45.2 Mean baseline FEV1 not reported | 2 | 1337 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.31 [0.34, 15.63] |
| 46 # patients with adverse cardiovascular events | 4 | 792 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.32, 2.54] |
| 46.1 Mean baseline FEV1 >/= 80% of predicted | 1 | 116 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.31 [0.01, 7.49] |
| 46.2 Mean baseline FEV1 61% to 79% of predicted | 3 | 676 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.06 [0.35, 3.24] |
| 47 # Worsening asthma | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 47.1 Mean baseline FEV1 61% to 79% of predicted | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 48 Change in height (cm) as SD scores at 24 +/‐ 4 weeks | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 48.1 Mean baseline FEV1 >/= 80% of predicted | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 49 PC20 Methacholine‐adjusted odds ratio increase from baseline | 1 | Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 49.1 Mean baseline FEV1 61% to 79% of predicted | 1 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] | |
| 50 ACTH induced cortisol < 18 microg/dl at endpoint | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 50.1 Mean baseline FEV1 61% to 79% of predicted | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 51 am cortisol < 5 microg/dl at endpoint | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 51.1 Mean baseline FEV1 61% to 79% of predicted | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 52 Change in % PC20 at endpoint | 1 | 39 | Mean Difference (IV, Fixed, 95% CI) | 0.30 [‐0.68, 1.28] |
| 52.1 Mean baseline FEV1 >/= 80% of predicted | 1 | 39 | Mean Difference (IV, Fixed, 95% CI) | 0.30 [‐0.68, 1.28] |
| 53 PC20 histamine | 1 | Doub'g doses (Fixed, 95% CI) | Totals not selected | |
| 53.1 Mean baseline FEV1 >/= 80% of predicted | 1 | Doub'g doses (Fixed, 95% CI) | 0.0 [0.0, 0.0] |
1.1. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 1 # patients with exacerbations requiring oral steroids.
1.2. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 2 # patients with exacerbations requiring hospitalisation.
1.3. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 3 Serious adverse event including respiratory.
1.7. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 7 # withdrawals due to serious non‐respiratory event.
1.15. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 15 Evening PEF (L/min) at endpoint.
1.23. Analysis.
Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 23 Change in # of symptom‐free nights at endpoint.
1.24. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 24 % symptom‐free nights at 12 +/‐ 4 weeks.
1.33. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 33 Rescue medication‐free days.
1.47. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 47 # Worsening asthma.
1.48. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 48 Change in height (cm) as SD scores at 24 +/‐ 4 weeks.
1.49. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 49 PC20 Methacholine‐adjusted odds ratio increase from baseline.
1.50. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 50 ACTH induced cortisol < 18 microg/dl at endpoint.
1.51. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 51 am cortisol < 5 microg/dl at endpoint.
1.52. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 52 Change in % PC20 at endpoint.
1.53. Analysis.

Comparison 1 Long‐acting beta2 versus placebo: both groups receiving similar dose ICS, Outcome 53 PC20 histamine.
Comparison 2. Additional comparisons for same dose.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 # patients with exacerbations requiring oral steroids by baseline predicted FEV1 | 30 | Risk Ratio (IV, Fixed, 95% CI) | Subtotals only | |
| 1.1 Mean baseline FEV1 >/= 80% of predicted | 8 | 1713 | Risk Ratio (IV, Fixed, 95% CI) | 0.70 [0.57, 0.87] |
| 1.2 Mean baseline FEV1 61% to 79% of predicted | 17 | 3764 | Risk Ratio (IV, Fixed, 95% CI) | 0.83 [0.72, 0.96] |
| 1.3 Mean baseline FEV1 not reported | 5 | 1331 | Risk Ratio (IV, Fixed, 95% CI) | 0.49 [0.21, 1.16] |
| 2 # patients with exacerbations requiring oral steroids children versus adults | 30 | Risk Ratio (IV, Fixed, 95% CI) | Subtotals only | |
| 2.1 Children | 6 | 605 | Risk Ratio (IV, Fixed, 95% CI) | 0.89 [0.58, 1.39] |
| 2.2 Adults | 24 | 6203 | Risk Ratio (IV, Fixed, 95% CI) | 0.77 [0.68, 0.88] |
| 3 # patients with exacerbations requiring oral steroids by dose of ICS in both groups | 30 | 6808 | Peto Odds Ratio (Peto, Fixed, 95% CI) | 0.72 [0.62, 0.83] |
| 3.1 Low dose of ICS (<= 400 mcg/day of BDP‐eq) | 12 | 3398 | Peto Odds Ratio (Peto, Fixed, 95% CI) | 0.63 [0.51, 0.78] |
| 3.2 Moderate dose of ICS (401 to 800 mcg/day of BDP‐eq) | 6 | 1067 | Peto Odds Ratio (Peto, Fixed, 95% CI) | 0.68 [0.48, 0.97] |
| 3.3 High dose of ICS (>800 mcg/day of BDP‐eq) | 7 | 1366 | Peto Odds Ratio (Peto, Fixed, 95% CI) | 0.94 [0.58, 1.54] |
| 3.4 Unspecified dose of ICS or range of dose only mentioned | 5 | 977 | Peto Odds Ratio (Peto, Fixed, 95% CI) | 0.89 [0.65, 1.22] |
| 4 # patients with exacerbations requiring oral steroids by combination inhaler or separate inhaler for LABA | 30 | Risk Ratio (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 Combination inhaler | 13 | 2718 | Risk Ratio (IV, Fixed, 95% CI) | 0.81 [0.50, 1.32] |
| 4.2 Separate inhaler | 16 | 4053 | Risk Ratio (IV, Fixed, 95% CI) | 0.78 [0.69, 0.89] |
| 4.3 Not reported | 1 | 37 | Risk Ratio (IV, Fixed, 95% CI) | 0.21 [0.03, 1.64] |
| 5 # patients with exacerbations requiring oral steroids by whether LABA dose is usual or higher than usual | 30 | Risk Ratio (IV, Fixed, 95% CI) | Subtotals only | |
| 5.1 LABA at usual dose | 27 | 6427 | Risk Ratio (IV, Fixed, 95% CI) | 0.74 [0.65, 0.84] |
| 5.2 LABA at higher than usual dose | 3 | 381 | Risk Ratio (IV, Fixed, 95% CI) | 1.10 [0.79, 1.52] |
| 6 # patients with exacerbations requiring oral steroids by type of LABA | 30 | Risk Ratio (IV, Fixed, 95% CI) | Subtotals only | |
| 6.1 Formoterol | 9 | 2923 | Risk Ratio (IV, Fixed, 95% CI) | 0.74 [0.64, 0.85] |
| 6.2 Salmeterol | 21 | 3885 | Risk Ratio (IV, Fixed, 95% CI) | 0.89 [0.72, 1.10] |
| 7 # patients with exacerbations requiring oral steroids by trial duration | 30 | Risk Ratio (IV, Fixed, 95% CI) | Subtotals only | |
| 7.1 <= 16 weeks | 21 | 3645 | Risk Ratio (IV, Fixed, 95% CI) | 0.91 [0.72, 1.14] |
| 7.2 > 16 weeks | 9 | 3163 | Risk Ratio (IV, Fixed, 95% CI) | 0.74 [0.64, 0.85] |
| 8 # patients with exacerbations requiring oral steroids study unsupported by pharmaceutical industry excluded | 30 | Risk Ratio (IV, Fixed, 95% CI) | Subtotals only | |
| 8.1 Charity funded | 1 | 23 | Risk Ratio (IV, Fixed, 95% CI) | 1.09 [0.28, 4.32] |
| 8.2 Funded by pharmaceutical industry | 29 | 6785 | Risk Ratio (IV, Fixed, 95% CI) | 0.78 [0.69, 0.88] |
| 9 Sensitivity analysis: exacerbations requiring oral steroids: studies with low risk of bias of selection bias (adequate allocation sequence generation) | 26 | 6513 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.69, 0.87] |
| 10 Sensitivity analysis: exacerbations requiring oral steroids: studies with low risk of bias of selection bias (adequate allocation concealment) | 20 | 5042 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.66, 0.91] |
| 11 Sensitivity analysis: exacerbations requiring oral steroids: studies with low risk of detection bias (adequate blinding) | 30 | 6808 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.68, 0.87] |
| 12 Sensitivity analysis: exacerbations requiring oral steroids: studies with low risk of bias of attrition bias (complete follow up of study participants) | 2 | 391 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.10, 3.89] |
| 13 Sensitivity analysis: exacerbations requiring oral steroids by data publication status (data available from published source) | 14 | 3161 | Risk Ratio (IV, Fixed, 95% CI) | 0.77 [0.67, 0.88] |
| 14 Change in FEV1 at endpoint stratifying on age (children versus adults) | 32 | L (Random, 95% CI) | Subtotals only | |
| 14.1 Children | 9 | L (Random, 95% CI) | 0.08 [0.05, 0.11] | |
| 14.2 Adults | 23 | L (Random, 95% CI) | 0.13 [0.10, 0.15] | |
| 15 Change in FEV1 at endpoint stratifying on LABA (formoterol versus salmeterol) | 32 | 9784 | L (Random, 95% CI) | 0.11 [0.09, 0.13] |
| 15.1 Formoterol | 17 | 4057 | L (Random, 95% CI) | 0.09 [0.07, 0.12] |
| 15.2 Salmeterol | 15 | 5727 | L (Random, 95% CI) | 0.14 [0.10, 0.18] |
| 16 Change in FEV1 at endpoint stratifying on baseline FEV1 | 32 | L (Random, 95% CI) | Subtotals only | |
| 16.1 <= 16 weeks | 25 | L (Random, 95% CI) | 0.12 [0.09, 0.14] | |
| 16.2 > 16 weeks | 7 | L (Random, 95% CI) | 0.10 [0.06, 0.13] |
2.1. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 1 # patients with exacerbations requiring oral steroids by baseline predicted FEV1.
2.2. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 2 # patients with exacerbations requiring oral steroids children versus adults.
2.3. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 3 # patients with exacerbations requiring oral steroids by dose of ICS in both groups.
2.4. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 4 # patients with exacerbations requiring oral steroids by combination inhaler or separate inhaler for LABA.
2.6. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 6 # patients with exacerbations requiring oral steroids by type of LABA.
2.7. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 7 # patients with exacerbations requiring oral steroids by trial duration.
2.8. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 8 # patients with exacerbations requiring oral steroids study unsupported by pharmaceutical industry excluded.
2.14. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 14 Change in FEV1 at endpoint stratifying on age (children versus adults).
2.15. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 15 Change in FEV1 at endpoint stratifying on LABA (formoterol versus salmeterol).
2.16. Analysis.

Comparison 2 Additional comparisons for same dose, Outcome 16 Change in FEV1 at endpoint stratifying on baseline FEV1.
Comparison 3. WMD archive.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Change in morning PEF (L/min) at endpoint stratifying on baseline FEV1 | 42 | Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 2 Change in evening PEF (L/min) at endpoint | 25 | Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 3 Change in % symptom‐free days at endpoint | 13 | 2935 | Mean Difference (IV, Random, 95% CI) | 13.34 [9.43, 17.24] |
| 3.1 Mean baseline FEV1 >/= 80% of predicted | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 3.2 Mean baseline FEV1 61% to 79% of predicted | 11 | 2296 | Mean Difference (IV, Random, 95% CI) | 14.98 [11.03, 18.92] |
| 3.3 Mean baseline FEV1 not reported | 2 | 639 | Mean Difference (IV, Random, 95% CI) | 6.57 [1.11, 12.03] |
| 4 Change in mean % rescue free days at 12 +/‐ 4 weeks | 6 | 1698 | Mean Difference (IV, Fixed, 95% CI) | 17.05 [13.75, 20.35] |
| 4.1 Mean baseline FEV1 61% to 79% of predicted | 5 | 1381 | Mean Difference (IV, Fixed, 95% CI) | 17.63 [14.03, 21.23] |
| 4.2 Mean baseline FEV1 not reported | 1 | 317 | Mean Difference (IV, Fixed, 95% CI) | 14.0 [5.77, 22.23] |
| 5 Change in FEV1 at endpoint (L) stratifying on baseline FEV1 | 26 | Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 6 Change in # daytime rescue inhalations (puffs per day) at endpoint | 12 | Mean Difference (IV, Random, 95% CI) | Totals not selected | |
| 7 Change in FEV1 at endpoint (% predicted) stratifying on baseline FEV1 | 7 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 8 Change in # overall daily rescue inhalations at endpoint | 10 | 3088 | Mean Difference (IV, Random, 95% CI) | ‐0.74 [‐1.07, ‐0.42] |
| 8.1 Mean baseline FEV1 >/= 80% of predicted | 2 | 1272 | Mean Difference (IV, Random, 95% CI) | ‐0.17 [‐0.29, ‐0.05] |
| 8.2 Mean baseline FEV1 61% to 79% of predicted | 8 | 1816 | Mean Difference (IV, Random, 95% CI) | ‐1.06 [‐1.76, ‐0.37] |
| 9 Change in quality of life (AQLQ score) at endpoint | 3 | 1354 | Mean Difference (IV, Random, 95% CI) | 0.33 [0.05, 0.60] |
3.1. Analysis.

Comparison 3 WMD archive, Outcome 1 Change in morning PEF (L/min) at endpoint stratifying on baseline FEV1.
3.2. Analysis.

Comparison 3 WMD archive, Outcome 2 Change in evening PEF (L/min) at endpoint.
3.3. Analysis.

Comparison 3 WMD archive, Outcome 3 Change in % symptom‐free days at endpoint.
3.4. Analysis.

Comparison 3 WMD archive, Outcome 4 Change in mean % rescue free days at 12 +/‐ 4 weeks.
3.5. Analysis.

Comparison 3 WMD archive, Outcome 5 Change in FEV1 at endpoint (L) stratifying on baseline FEV1.
3.6. Analysis.

Comparison 3 WMD archive, Outcome 6 Change in # daytime rescue inhalations (puffs per day) at endpoint.
3.7. Analysis.

Comparison 3 WMD archive, Outcome 7 Change in FEV1 at endpoint (% predicted) stratifying on baseline FEV1.
3.8. Analysis.

Comparison 3 WMD archive, Outcome 8 Change in # overall daily rescue inhalations at endpoint.
3.9. Analysis.

Comparison 3 WMD archive, Outcome 9 Change in quality of life (AQLQ score) at endpoint.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Akpinarli 1999.
| Methods | Parallel‐group multicentre study | |
| Participants | Symptomatic asthmatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 32 (ICS + F12 (bid): 16; ICS: 16) WITHDRAWALS: Not described AGE mean (range) or mean (SD): 6 to 14 years GENDER (% male): 47% SEVERITY: Not reported BASELINE % PREDICTED FEV1: Not reported (study categorised as 61% to 79% predicted) BASELINE DOSE OF ICS: 400 to 800 mcg ASTHMA DURATION: Not described ATOPY (%): 68 ELIGIBILITY CRITERIA: Met ATS criteria for asthma; >= 15% increase in FEV1 within the previous year EXCLUSION CRITERIA: Asthma exacerbation or respiratory infection in < month ELIGIBILITY CRITERIA DURING RUN‐IN: Only patients requiring salbutamol more than once a week were randomised |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES reported at 6 weeks RUN‐IN PERIOD: 2 weeks with ICS 400 to 800 mcg/day to document symptoms and beta2 use DOSE OPTIMISATION PERIOD: NONE INTERVENTION PERIOD: 6 weeks TEST GROUP: (ICS + F12) ICS 400 to 800 mcg/day + formoterol 12 mcg bid CONTROL GROUP: (ICS) ICS (400 to 800 mcg/day) + placebo bid DEVICE: MDI + large volume spacer (Volumatic) NUMBER OF DEVICES: 2 COMPLIANCE: assessed by weighing canisters CO‐TREATMENT: Not described |
|
| Outcomes | INTENTION‐TO‐TREAT ANALYSIS: Not described PULMONARY FUNCTION TEST: % of predicted FEV1; morning PEFR (L/min); evening PEFR (L/min); PEF variability (%); PC 20 (mg/ml) SYMPTOM SCORES: score of 0 to 3 (max 9); night‐time symptom score; symptom‐free days or nights FUNCTIONAL STATUS: rescue B2‐agonist use per week (each use consisted of 2 puffs); exacerbation requiring oral steroids; exacerbations requiring admission INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: described WITHDRAWALS: Not described Primary outcome measure not reported |
|
| Notes | Full‐text publication Funded by AstraZeneca Author contacted and unable to confirm methodology or data User‐defined number: 600 (mean ICS dose in LAB2 group in mcg/day of BDP‐equivalent: 400 to 800) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐blind; identical placebo used |
| Incomplete outcome data addressed? All outcomes | Unclear risk | No information available on the statistical handling of missing data |
| Free of selective reporting? | Low risk | Data on OCS‐treated exacerbations available |
Aubier 1999a.
| Methods | Parallel‐group, 55 centres in Germany, Netherlands & France. Three treatment arms: combination FP/SAL, concurrent FP/SAL, FP alone | |
| Participants | Moderately severe asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 503 (Combination FP/SAL: 167; concurrent FP/SAL: 171; FP alone: 165) WITHDRAWALS: Combination FP/SAL: 31; concurrent FP/SAL: 28; FP: 48 AGE: mean (range) or mean (SD): 48 years GENDER: (% male): 54% SEVERITY: Moderate BASELINE % PREDICTED FEV1 (mean): 73 BASELINE DOSE OF ICS: 1500 to 2000 mcg BDP equivalent ASTHMA DURATION: < 1 year: 3%; 1 to 5 years: 23%; 5 to 10 years: 20%; > 10 years: 54% ATOPY (%): 52 ELIGIBILITY CRITERIA: > 12 years; documented history of reversible airways disease; ICS treatment for 12 weeks prior to run‐in; BDP or BUD 1500 to 2000 mcg/d or FP 750 to 1000 mcg/d EXCLUSION CRITERIA: Not reported ELIGIBILITY CRITERIA DURING RUN‐IN: At end of 2‐week run‐in eligible candidates were symptomatic (symptom score >/= 2 on 4 of last 7 consecutive days), mean am PEF > 50% and < 85% of maximum PEF 15 min post SABA, and (FEV1) between 50% and 100% predicted |
|
| Interventions | PROTOCOL: Concurrent ICS and LABA versus ICS alone OUTCOMES: 1, 2, 3, 4 , 5 to 8, 9 to 12 weeks for PEF; 28 weeks for FEV1 RUN‐IN: 2 weeks DOSE OF ICS DURING RUN‐IN: Pre‐study dose of ICS INTERVENTION PERIOD: 28 weeks TEST GROUP: Fluticasone and salmeterol 500/50 mcg bid given via separate inhalers CONTROL GROUP: Fluticasone 500 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 2 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; am PEF predicted; pm PEF SYMPTOM SCORES: Daytime scores FUNCTIONAL STATUS: Not reported INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Stated by treatment group WITHDRAWALS: Stated by treatment group Primary outcome measure* |
|
| Notes | Full‐text publication, additional data from www.ctr.gsk.co.uk Source of funding GSK Confirmation of methodology and data: Not obtained User defined number: 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Double‐blind, double‐dummy design |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Intention‐to‐treat analysis; no description of how population defined for analysis |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
Aubier 1999b.
| Methods | See above | |
| Participants | See above | |
| Interventions | PROTOCOL: Combination ICS and LABA versus ICS via one inhaler All other items listed under interventions identical to Aubier 1999a |
|
| Outcomes | See above | |
| Notes | See above | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | See above |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See above |
| Free of selective reporting? | Unclear risk | See above |
Bailey 2008.
| Methods | Parallel‐group, multicentre study in USA | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: 60 % RUN‐IN PARTICIPANTS RANDOMISED: 90 RANDOMISED: 475 (FP/SAL 239; FP 236) WITHDRAWALS: FP/SAL 67; FP 85 AGE mean (range) or mean (SD): 32 SEVERITY: Not reported BASELINE % PREDICTED FEV1: 78 BASELINE DOSE OF ICS: 400 mcg BDP equivalent ASTHMA DURATION: 19 years ATOPY (%): Not reported ELIGIBILITY CRITERIA: African American by self‐report; 12 to 65 years of age; persistent asthma for at least 6 months; FEV1 60% to 90% predicted; FEV1 reversibility of > 12% post‐SABA; symptomatic while being treated with FP 200 mcg daily or equivalent for at least 1 month prior to screening EXCLUSION CRITERIA: Asthma exacerbation during screening period (worsening of asthma that required treatment with asthma medications other than their ICS and albuterol) ELIGIBILITY CRITERIA DURING RUN‐IN: FEV1 > 60% predicted normal; plus albuterol use on 4 or more days during the 7‐day period prior to the clinical visit; and/or an asthma symptom score > 2 on 4 or more days during the 7‐day period prior to the clinic visit |
|
| Interventions | PROTOCOL: LABA and ICS versus SAME DOSE ICS OUTCOMES: 52 weeks RUN‐IN PERIOD: 4 weeks (FP250 mcg bid) INTERVENTION PERIOD: 52 weeks (plus 4‐week run‐out) TEST GROUP: Combination fluticasone and salmeterol 100/50 mcg bid CONTROL GROUP: Fluticasone 100 mcg bid NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; am PEF; pm PEF SYMPTOM SCORES: Total symptoms FUNCTIONAL STATUS: Exacerbations (undefined)*; rescue medication use INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Full‐text publication, additional data from www.ctr.gsk.co.uk Source of funding GSK Confirmation of methodology and data: Not obtained User defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The primary analysis population was the intent to‐ treat (ITT) population, comprised of all subjects randomized to double‐blind study medication. All data collected prior to early withdrawal was considered evaluable." |
| Free of selective reporting? | Low risk | Exacerbations described in study report; these included OCS‐treated exacerbations, but separate data could not be identified. Request for data submitted. |
Boyd 1995.
| Methods | Parallel‐group, multicentre study (15 centres in the United Kingdom) | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 66% RANDOMISED: 119 (Salm100 + ICS: 55; placebo + ICS: 64) WITHDRAWALS: Salm100 + ICS: 8; placebo + ICS: 14 AGE: mean (range): 47 (18 to 79) GENDER: (% male): 43% BASELINE % PREDICTED FEV1 mean: 66 BASELINE DOSE OF ICS: 1000 to 4000 mcg/day ASTHMA DURATION (years): 15 years ATOPY (%): Not described ELIGIBILITY CRITERIA: >= 15% improvement from baseline in lung function following inhaled salbutamol; at least 2 acute asthma exacerbations in the preceding 18 months EXCLUSION CRITERIA: Concurrent uncontrolled oral disease; having received treatment for an acute respiratory infection in the last 2 weeks or had a FEV1 < 40% predicted |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: reported at 4, 8 and 12 weeks RUN‐IN: 2 weeks DOSE OF ICS DURING RUN‐IN: Usual ICS Intervention period: 12 weeks TEST GROUP: (Salm100 + ICS) Salmeterol 100 mcg bid + ICS CONTROL GROUP: Placebo + ICS DEVICE: Diskhaler NUMBER OF DEVICES: 2 COMPLIANCE: Assessed at each clinic visit CO‐TREATMENT: Salbutamol metered dose inhaler |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; PEF(morning and evening)* SYMPTOM SCORE: Score of 0 to 4 (change); changes in daytime and night‐time score FUNCTIONAL STATUS: Rescue B2‐agonist (number of puffs per 24 hours); nocturnal awakening (change in symptom‐free nights); symptom‐free days change; severe exacerbation (requiring oral steroids) INFLAMMATORY MARKERS: None studied ADVERSE EFFECTS: Reported WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Full‐text publication Funded by Allen & Hanburys Confirmation of methodology and data obtained User‐defined number: 1681 (mean ICS dose in LAB2 group in mcg/day of BDP‐equivalent) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Low risk | Numbered coded inhalers supplied by pharmacy |
| Blinding? All outcomes | Low risk | Identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Not enough information presented to determine how ITT population was defined: "The study was analysed on an intention‐to‐treat basis. All patients randomized to treatment were evaluable." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbation data available |
Buhl 2003a.
| Methods | Parallel‐group, multicentre study (56 centres in 9 countries) | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PATIENTS RANDOMISED: 95% (26 patients were recruited but were not randomised as they deteriorated during run‐in) RANDOMISED 523 (Form 6 mcg + ICS bid: 176; Form 9 mcg + ICS: 176; placebo + ICS: 171) WITHDRAWALS: Form 6 bid + ICS: 14; Form 9 bid + ICS: 14; placebo + ICS: 14 AGE mean (range): 45 (18 to 78) GENDER (% male): 49% BASELINE % PREDICTED FEV1 mean: 77 BASELINE DOSE OF ICS mean: 600 mcg/day ASTHMA DURATION mean (range) in years: 13 (0 to 63) ATOPY (%): Not described ELIGIBILITY CRITERIA: Baseline FEV1 of 60% to 90% normal; >= 12% improvement from baseline in lung function following inhaled salbutamol; at least 2 acute asthma exacerbations in the preceding 18 months EXCLUSION CRITERIA: oral corticosteroids in 4 weeks before run‐in; concurrent respiratory infection in the 4 weeks before run‐in; severe cardiovascular disorder; use of beta blocker; heavy smoking |
|
| Interventions | LABA + ICS TWICE A DAY versus SAME dose of ICS ONCE A DAY OUTCOMES: reported at 4, 8 and 12 weeks RUN‐IN: 4 weeks DOSE OF ICS DURING RUN‐IN: BUD 200 bid INTERVENTION PERIOD: 12 weeks TEST GROUP: (Form 6 + ICS) formoterol 6 mcg bid + ICS bid CONTROL GROUP: Placebo + ICS od DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: Salbutamol metered dose inhaler |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; change in morning PEF*; change in evening PEF SYMPTOM SCORE: Score of 0 to 3 grading daytime and night‐time symptoms; Total daily asthma score = sum of daytime and night‐time scores FUNCTIONAL STATUS: Exacerbations; rescue B2‐agonists use; nocturnal awakening (% nights with awakening; % reliever use‐free days ‐% symptom‐free days; % asthma control days; % asthma control weeks INFLAMMATORY MARKERS: None studied ADVERSE EFFECTS: Reported WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Full‐text publication Funded by AstraZeneca Confirmation of methodology and data obtained User‐defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Low risk | Numbered coded inhalers supplied by pharmacy |
| Blinding? All outcomes | Low risk | Double‐dummy design |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Not enough information presented to determine how ITT population was defined: "All efficacy variables were analysed on an intent‐to‐treat basis and all randomized patients with data were included in the analysis." |
| Free of selective reporting? | Low risk | Exacerbations described in study report; these included OCS‐treated exacerbations, but separate data could not be extracted |
Buhl 2003b.
| Methods | See Buhl 2003a | |
| Participants | See Buhl 2003a | |
| Interventions | LABA + ICS ONCE A DAY vs SAME dose of ICS ONCE A DAY TEST GROUP: (Form 12 + ICS) formoterol 12 mcg bid + ICS CONTROL GROUP: Placebo + ICS Other characteristics the same as above |
|
| Outcomes | See Buhl 2003a | |
| Notes | See Buhl 2003a | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | See above |
| Allocation concealment? | Low risk | See above |
| Blinding? All outcomes | Low risk | See above |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See above |
| Free of selective reporting? | Low risk | See above |
D'Urzo 2001.
| Methods | Parallel‐group, multicentre study (253 centres predominantly general practices) | |
| Participants | Adults and adolescents with asthma % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 911 (Salm50 bid + ICS: 455; placebo + ICS: 456) WITHDRAWALS: Salm50 bid + ICS: 19%; placebo + ICS: 24% AGE mean (range): 46 (17 to 86) GENDER (% male): 46% SEVERITY: Moderate BASELINE FEV1 MEAN (SD): Not reported BASELINE DOSE OF ICS/day: BDP up to 500 mcg: 18% BDP 500 to 1000: 59% BDP > 1000 = 23% ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: History of asthma (ATS criteria); required regular ICS but still required rescue bronchodilator more than twice daily EXCLUSION CRITERIA: Uncontrolled pulmonary or oral disease or psychological condition that in then opinion of investigator precluded their entry into study; concurrent beta blocker therapy |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: measured at 4‐weekly intervals RUN‐IN PERIOD: None DOSE OF ICS DURING RUN‐IN: Not applicable DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 24 weeks TEST GROUP (LABA + SINGLE DOSE ICS): Usual ICS + salmeterol 50 mcg bid CONTROL GROUP: Placebo + usual dose of ICS DEVICE: MDI NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: Not reported |
|
| Outcomes | PULMONARY FUNCTION TEST: Change in clinic PEF SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Rescue medication use day and night; daytime and night‐time symptoms; nocturnal awakenings; serious asthma exacerbation* defined as days in hospital, days of prednisone treatment or ER visit; days requiring increased asthma medication; work or school days lost because of asthma limitation of activities because of asthma INFLAMMATORY MARKERS: Blood eosinophil count measured in subgroup with asthma exacerbation ADVERSE EFFECTS: Heart rate higher in salmeterol group; no other adverse effects reported WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Full‐text publication Supported by GSK Confirmation of methodology and data extraction not obtained User defined number: Not reported (mean ICS dose in LAB2 group in mcg/day of BDP‐equivalent: Not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Unclear risk | Described as double‐blind. Information on masking of treatments not presented |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Described as ITT; composition of ITT population not explicit: "The Intent‐to‐Treat (ITT) population consisted of all randomised subjects except those where verified proof existed that no study medication was taken. The ITT population was used for all analyses and tabulations." |
| Free of selective reporting? | Low risk | Exacerbations described in study report; these included OCS‐treated exacerbations; separate data could not be extracted |
D5896C0001a.
| Methods | Parallel‐group, multicentre 4‐arm trial | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: 44 % RUN‐IN PARTICIPANTS RANDOMISED: 63 RANDOMISED: 618 (BUD/F bid 155; BUD/F high qd 153; BUD/F low qd 153; BUD 153) WITHDRAWALS: Not stated AGE mean (range) or mean (SD): 35 (15) SEVERITY: Not stated BASELINE % PREDICTED FEV1: 76% BASELINE DOSE OF ICS: 375 mcg ASTHMA DURATION: Not stated ATOPY (%): Not stated ELIGIBILITY CRITERIA: > 12 years; documented clinical diagnosis of asthma for 6 months prior to screening; stable; maintenance asthma treatment with inhaled corticosteroids (ICS) for at least 4 weeks; FEV1 60% to 90% predicted EXCLUSION CRITERIA: Not stated ELIGIBILITY CRITERIA DURING RUN‐IN: Not stated |
|
| Interventions | PROTOCOL: LABA and ICS versus SAME DOSE ICS OUTCOMES 12 weeks RUN‐IN PERIOD: 4 to 5 weeks (combination therapy) INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination budesonide and formoterol 200/12 mcg bid (400/24 mcg total) CONTROL GROUP: Budesonide 400 mcg qd NUMBER OF DEVICES: Two (double‐dummy) COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; am PEF; pm PEF SYMPTOM SCORES: Total symptoms FUNCTIONAL STATUS: Rescue medication use INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Stated for adverse events only |
|
| Notes | Trial report available as download from AZ clinical trials website Funded by AZ Confirmation of methodology and data extraction not obtained User defined: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | "To maintain blinding with the twice‐daily dosing regimen, all subjects randomized to receive once‐daily dosing were to take the active treatment in the evening and a matched placebo device (Batch numbers P6492 and P6856) in the morning." |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The efficacy analysis set (EAS), defined as all randomized subjects who took at least 1 dose of double‐blind treatment and for whom the primary efficacy endpoint could be calculated, was used in the primary analysis of efficacy. Sensitivity analyses were performed using the per protocol (PP) analysis set, which excluded subjects with major violations of inclusion or exclusion criteria." |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
D5896C0001b.
| Methods | See above | |
| Participants | See above | |
| Interventions | See above; except for: TEST GROUP: Combination budesonide and formoterol 400/12 mcg qd (evening) |
|
| Outcomes | See above | |
| Notes | See above | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | See above |
| Allocation concealment? | Unclear risk | See above |
| Blinding? All outcomes | Low risk | See above |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See above |
| Free of selective reporting? | Unclear risk | See above |
Fitzgerald 1999.
| Methods | Parallel‐group, multicentre study (15 centres in Canada). Three groups of which 2 considered for this review (group which evaluated regular albuterol use not considered) | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 72 RANDOMISED: 271 (F 12 bid + usual dose ICS: 89; usual dose ICS and on demand albuterol: 91; usual dose ICS + regular albuterol: 91) WITHDRAWALS: F 12 bid + usual dose ICS: 17; usual dose ICS and on demand albuterol: 18 Mean AGE years (SD): 36 (13) GENDER (% male): 44 SEVERITY: Moderate BASELINE FEV1 PREDICTED: 79 BASELINE DOSE OF ICS mean (SD): 732 (280) mcg ASTHMA DURATION (years): Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: Non‐smoking adults with asthma as defined by ATS criteria; treated with ICS 400 to 1200 mcg/day for at least 1 month prior to screening; >= 15% reversibility after bronchodilator; during the last 7 days of run‐in, had used albuterol on at least 5 days awakening on >= 1 night due to asthma symptoms; use of beta agonist >= 10 puffs as weekly mean; competence with turbuhaler; compliance with dairy cards and assessments EXCLUSION: Respiratory infection within 2 months of screening; acute asthma exacerbation requiring an ER visit in the previous 3 months CRITERIA FOR RANDOMISATION DURING RUN‐IN: Had used rescue albuterol on at least 5 of the last 7 days of the run‐in period. Excluded from randomisation if asthma was poorly controlled as defined by 2 or more awakenings per week or a visit 2 premedication FEV1 less than 50% predicted or less than 1 L |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: after initial dose, 3, 6 months and 2 days after end of last dose RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: 400 to 1200 mcg BDP, BUD usual dose of patient DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 24 weeks TEST GROUP (LABA + SINGLE DOSE ICS): Formoterol 12 mcg bid + beclomethasone, budesonide 400 to 1200 mcg/day CONTROL GROUP: Beclomethasone, budesonide 400 to 1200 mcg bid DEVICE: Formoterol ‐ dry powder inhalation capsules; albuterol MDI NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: Not reported |
|
| Outcomes | PULMONARY FUNCTION TEST: Change in morning PEF*; Change in FEV1 SYMPTOM SCORES: Change in daytime and night‐time scores FUNCTIONAL STATUS: Change in rescue medication use day and night (puffs per day or night) INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Reported WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Full‐text publication Supported by Novartis Confirmation of methodology and data extraction not obtained User defined number: (mean ICS dose in LAB2 group in mcg/day of BDP‐equivalent: 730 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy design |
| Incomplete outcome data addressed? All outcomes | Unclear risk | No information provided |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available in trial publication |
Gardiner 1994.
| Methods | Cross‐over, single‐centre study | |
| Participants | Stable asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 10 AGE: median (range): 42 (23 to 64) GENDER: (% males): 60 SEVERITY: Moderate BASELINE % PREDICTED FEV1: Not reported BASELINE DOSE OF ICS (before start of run‐in): 400 to 1000 BDP equivalent ASTHMA DURATION: Not reported ATOPY (%): 70 ELIGIBILITY CRITERIA: Non‐smoking; asthma diagnosed by ATS criteria; 15% reversibility following bronchodilator EXCLUSION CRITERIA: Respiratory infection or asthma exacerbation in 2 months prior to study |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: 2 and 4 months RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Usual ICS (400 to 1000) DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 8 weeks TEST GROUP (LABA + SINGLE DOSE ICS): salmeterol 50 mcg bid CONTROL GROUP: Usual ICS DEVICE: Not stated NUMBER OF DEVICES: 2 COMPLIANCE: Not stated CO‐TREATMENT: Inhaled albuterol as rescue medication but no oral beta agonists, inhaled anticholinergic medication or theophylline |
|
| Outcomes | PULMONARY FUNCTION TEST: PEF SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Not assessed INFLAMMATORY MARKERS: BAL differential cell count; BAL mast cell tryptase & al; serum ECP; respiratory burst; release of PAF before and after allergen inhalation challenge ADVERSE EFFECTS: Not reported WITHDRAWALS: Not reported |
|
| Notes | Full‐text publication Source of funding not reported Confirmation of methodology and data extraction not obtained User defined number: (mean ICS dose in LAB2 group in mcg/day of BDP‐equivalent: Not reported range 400 to 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Not enough information available to determine population analysed |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
GOAL.
| Methods | Parallel‐group, multicentre study (326 centres in Europe, North America, Latin America and Asia Pacific) | |
| Participants | Uncontrolled asthmatic adults % ELIGIBLE OF SCREENED POPULATION: 67 % RUN‐IN PARTICIPANTS RANDOMISED: Not clear RANDOMISED: 3416 (FP/SAL: 1707; FP: 1709). NB ‐ data in this review are taken from the strata of patients randomised who were on ICS prior to study entry (N = FP/SAL: 1133; FP: 1119) WITHDRAWALS: FP/SAL: 162; FP: 215 AGE: mean (range) or mean (SD) 40 (16) GENDER: (% male) 42 SEVERITY: Moderate BASELINE % PREDICTEDFEV1: 77 BASELINE DOSE OF ICS: Divided into 3 strata: 0; 500 mcg/d or less; between 500 and 1000 mcg/d ASTHMA DURATION: 0 to 1 year: FP/SAL: 56; FP: 97‐ 1 to 10 years: FP/SAL: 649; FP: 647 ‐> 10 years: FP/SAL: 1004; FP: 992 ATOPY (%): 58 ELIGIBILITY CRITERIA: 12 to 80 years of age; 6‐month history of asthma; FEV1 reversibility of 15%; smoking history of less than 10 pack‐years; no use of LABA or oral beta‐agonists in previous 2 weeks EXCLUSION CRITERIA: Not reported |
|
| Interventions | LABA + ICS versus SAME DOSE ICS OUTCOMES: End of phase RUN‐IN: 4 weeks DOSE OF ICS DURING RUN‐IN: Usual maintenance dose of ICS (including 0 for participants not treated with ICS) INTERVENTION PERIOD: Two different phases: I = dose step‐up until total asthma control achieved, or until maximum dose of study drug given for 12 weeks; II = constant dose of final dose of study drug until 52 weeks since randomisation had elapsed TEST GROUP: Combination fluticasone and salmeterol 50/100; 50/250 or 50/500 mcg bid CONTROL GROUP: Fluticasone 100, 250 or 500 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1 SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: N achieving total asthma control*; exacerbations (defined as OCS course, ED visit/hospitalisation) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported by treatment group, but not collected since they refer to all phase, all strata participants WITHDRAWALS: Reported by treatment group Primary outcome measure* |
|
| Notes | Full‐text publication Source of funding: GSK Confirmation of methodology and data: Not obtained User defined number: 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The Intent‐To‐Treat Population (ITT) consisting of all subjects who were randomised to treatment and received at least one single dose of study medication, was the primary population for analysis of efficacy and safety." |
| Free of selective reporting? | Low risk | Exacerbations requiring OCS treatment described as a composite in a rate; data could not be extracted and used in meta‐analysis |
Green 2006.
| Methods | Cross‐over, single‐centre study in UK | |
| Participants | N SCREENED: 66 N RANDOMISED: 49 N COMPLETED: 39 GENDER (% male): 52 MEAN AGE: 42 SEVERITY: Not stated BASELINE FEV1 PREDICTED: 74.8% ATOPY (%): 93% INCLUSION CRITERIA: 18 to 75 years, diagnosed with asthma; receiving treatment with 400 mcg/day beclomethasone dipropionate; one or more of 1) > 15% increase in FEV1 post‐SABA; 2) > 20% within‐day variability in PEF assessed twice daily over a 2‐week period; 3) provocative concentration of methacholine causing a 20% fall in FEV1 (PC20) < 8 mg/mL‐1; following run‐in on 200 mcg day BUD, participants were eligible if they had recorded day‐ or night‐time asthma symptoms on their diary cards on at least 4 days in the third or fourth baseline week. EXCLUSION: Current smokers or smoking history of > 10 pack‐years, significant comorbidity, treated with oral corticosteroids, long‐acting ß2‐agonists, leukotriene antagonists or theophylline; asthma exacerbation or lower respiratory tract infection within the 4 weeks prior to trial entry |
|
| Interventions | LABA + ICS versus SAME DOSE ICS OUTCOMES: End of phase TREATMENT PERIOD: 6 weeks RUN‐IN PERIOD: 4 weeks TEST GROUP: Budesonide 100 mg bid + formoterol 12 mg bid CONTROL: Budesonide 100 mg bid NUMBER OF DEVICES: 2 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TESTS: am PEF; FEV1 SYMPTOM SCORES: *VAS; daytime symptoms; nocturnal symptoms FUNCTIONAL STATUS: Quality of life (AQLQ); exacerbations (deterioration in PEF or requirement for OCS. Patients who experienced 2 or more exacerbations were withdrawn from the study) INFLAMMATORY MARKERS: *sputum eosinophils; exhaled nitric oxide; *PC20 ADVERSE EFFECTS: Not reported WITHDRAWALS: Reported (not by treatment group) Primary outcome measure* |
|
| Notes | Full‐text publication Funding source: Not declared (AZ provided active and placebo inhalers) Confirmation of methodology and data: Obtained User defined number: 200 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Randomisation from correspondence with investigator: "I believe that this was generated using a computer statistical package generating a random sequence. I don't know the package that was used and unfortunately the individual has left our organisation but had extensive clinical trials expertise. None of the study investigators were aware of the randomisation schedule until the last patient had completed the cross‐over study" |
| Allocation concealment? | Low risk | Correspondence with investigator: "...this was indeed generated by a third party, namely the pharmacist responsible for dispensing the double blind medication." |
| Blinding? All outcomes | Low risk | Identical placebos |
| Incomplete outcome data addressed? All outcomes | High risk | Completers used for analysis |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations described; could not extract data as proportion of participants with one or more events. |
Houghton 2007.
| Methods | Parallel‐group, single‐centre study | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 70 RANDOMISED: 39 WITHDRAWALS: FP/SAL: 1; FP: 1 AGE mean (range) or mean (SD): 40 SEVERITY: Mild BASELINE % PREDICTED FEV1: 94 BASELINE DOSE OF ICS: FP 200 mcg/day ASTHMA DURATION: Not specified ATOPY (%): Not reported ELIGIBILITY CRITERIA: Physician diagnosed asthma for >/= 6 months; receiving stable total daily dose of ICS (equivalent to 200 to 500 mcg BDP) for at least 4 weeks prior to the study; FEV1 > 80% predicted and demonstration of a > 30% decrease in sRaw in response to 400 mcg of inhaled salbutamol at screening EXCLUSION CRITERIA: Use of parenteral, oral and nebulised steroids 4 weeks prior to study (12 weeks for depot corticosteroids); positive pregnancy test; current smokers or ex‐smokers for < 12 months ELIGIBILITY CRITERIA DURING RUN‐IN: Not symptomatic on every day of run‐in period |
|
| Interventions | PROTOCOL: LABA + ICS versus SAME dose ICS OUTCOMES: 4 weeks RUN‐IN PERIOD: 2 weeks DOSE OPTIMISATION PERIOD: Not reported INTERVENTION PERIOD: 4 weeks TEST GROUP: Combination fluticasone and salmeterol (100/50 mcg bid) CONTROL GROUP: Fluticasone 100 mcg bid NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; PEF; airway resistance* SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Not reported INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Not reported WITHDRAWALS: Reported |
|
| Notes | Funding source: GSK Confirmation of methodology and data: Not obtained User defined: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | "Randomised" |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | "...identical Accuhaler devices..." |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "Statistical analysis was performed on an Intention To Treat basis with all subjects randomised to treatment being included." |
| Free of selective reporting? | Unclear risk | We could not determine whether information on oral steroid‐treated exacerbations was collected |
Hultquist 2000.
| Methods | Parallel‐group, multicentre in 49 clinical centres in 6 countries; 3 groups of which 2 are considered for this review | |
| Participants | Symptomatic asthmatic patients aged 12 to 70 years % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 74% (126 enrolled patients not randomised for reasons as follows: eligibility criteria not fulfilled (110); adverse events (6); lost to follow up (1); other reasons (9)) RANDOMISED: 352 (F 9 bid + BUD 200 bid: 118; BUD 200 bid: 116 BUD + montelukast: 118) WITHDRAWALS: F 9 bid + BUD 200 bid: 10%; BUD 200 bid: 7% Mean AGE years: 38.1 GENDER (% male): 51 SEVERITY: Moderate BASELINE % PREDICTED FEV1: 71 BASELINE DOSE OF ICS: 400 to 1000 mcg per day ASTHMA DURATION (years): 11 years ATOPY (%): Not reported ELIGIBILITY CRITERIA: Aged 12 to 70 years; treated with ICS 400 to 1000 mcg/day for at least 3 months prior to visit 1; FEV1 between 50% to 80 % of pred normal; >= 12% reversibility after bronchodilator; smoking history <= 10 years EXCLUSION CRITERIA: Patients who had other diseases that may interfere with assessments; respiratory infection, COPD or pulmonary dysfunction other than asthma; pregnant or lactating women; use of LABA within 1 month prior to visit 1; previous use of leukotriene antagonist; known intolerance to study drugs or inhaled lactose CRITERIA FOR RANDOMISATION DURING RUN‐IN: During the last 7 days of run‐in, having an asthma score >= 1 on 4 days or awakening on >= 1 night due to asthma symptoms; use of beta agonist >= 10 puffs as weekly mean; competence with turbuhaler; compliance with dairy cards and assessments |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: Not reported RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Not stated DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 8 weeks TEST GROUP (LABA + SINGLE DOSE ICS): Budesonide 200 mcg bid + formoterol 9 mcg bid CONTROL GROUP: Budesonide 200 mcg bid DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; FEV1 SYMPTOM SCORES: Daytime and night‐time score FUNCTIONAL STATUS: Rescue medication use per day; % night‐time awakenings INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Not described WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Abstract and full study report from sponsoring drug company Supported by AstraZeneca Confirmation of methodology and data extraction obtained User defined number: (mean ICS dose in LAB2 group in mcg/day of BDP‐equivalent: 800 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Low risk | Opaque consecutive numbered envelopes containing assignment |
| Blinding? All outcomes | Low risk | Use of identical placebo (double‐dummy) |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Analysis described as modified |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
Ind 2003.
| Methods | Parallel‐group, multicentre study in 100 hospitals and general practices in 6 countries (3 groups of which 2 are considered for this review) | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 58 RANDOMISED: 502 (496 with completed case report forms included in intent‐to‐treat population); FP/SAL: 171; FP250: 160 (additional treatment arm: FP500: 165) WITHDRAWALS: FP/SAL: 27; FP250: 15 AGE mean years (SD): 45 (15.4) GENDER: (% male): 45 SEVERITY: Moderate to severe BASELINE PREDICTED FEV1: 2.3 L/sec % PREDICTED PEF am: 75 BASELINE DOSE OF ICS (median): 1000 ASTHMA DURATION (range in years): 0.2 to 65 ATOPY (%): Information unavailable ELIGIBILITY CRITERIA: Aged 15 to 75; symptomatic on BDP 500 to 800 mcg bid or equivalent via MDI with good technique; 2 documented exacerbations needing hospitalisation or change in treatment with one occurring in last 6 months; PEF less than 85% of post bronchodilator PEF at first clinic visit INCLUSION CRITERIA FOR RANDOMISATION DURING RUN‐IN: Period variation in PEF over 10 days of >= 15% (highest evening PEF minus lowest morning PEF as a percentage of highest value); PEF not exceeding 90% of the post‐bronchodilator PEF at first clinic visit EXCLUSION CRITERIA: Patients receiving regular oral corticosteroid; patients who had serious uncontrolled oral disease; participation was deemed unsuitable by their physician from the study |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: 6, 12 18 and 24 weeks RUN‐IN PERIOD: 4 weeks DOSE OF ICS DURING RUN‐IN: FP 250 mcg bid DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 24 weeks TEST GROUP (LABA + SAME DOSE ICS): Fluticasone propionate 250 mcg and salmeterol 50 mcg bid CONTROL GROUP: FP 250 mcg bid DEVICE: MDI NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: Rescue short‐acting beta2‐agonists (salbutamol MDI) as needed, other asthma drugs as needed except LABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF SYMPTOM SCORES: Night‐time scores 0 to 4; daytime score 0 to 5 FUNCTIONAL STATUS: % symptom‐free days and nights; rescue medication use; exacerbations (defined as: mild (requiring increase in relief medication); moderate (requiring the use of additional corticosteroid); severe (requiring emergency hospital treatment) INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Reported WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Full‐text publication Supported by GSK Confirmation of methodology and data extraction not obtained User defined number: 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical placebo (double‐dummy) |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "intent‐to‐treat population (...) included all patients randomised to treatment with completed case report forms and verifiable data." |
| Free of selective reporting? | Low risk | Moderate exacerbations extracted as proxy for OCS‐treated exacerbations (moderate exacerbations) |
Jenkins 2006a.
| Methods | Parallel‐group, multicentre study (54 centres in 6 countries) | |
| Participants | Symptomatic asthmatic adults and adolescents % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 93% RANDOMISED: 341 (combination BUD/F: 226; BUD: 115) WITHDRAWALS: Not reported for 12 weeks data AGE: mean (range): (12 to 79) GENDER (% male): 40 SEVERITY: Moderate to severe persistent asthma BASELINE % PREDICTED FEV1(mean): 66 BASELINE DOSE OF ICS: 1040 mcg/d ASTHMA DURATION: 8 years ATOPY (%): Not reported ELIGIBILITY CRITERIA: >/= 12 years; diagnosis of asthma (for at least 6 months); FEV1 40% to 85% predicted; >/= 15% reversibility to SABA; use of >/= 750 mcg ICS for 4 months; symptomatic during run‐in EXCLUSION CRITERIA: Deterioration in asthma leading to change in therapy |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: 12 weeks RUN‐IN: 2 weeks DOSE OF ICS DURING RUN‐IN: Regular ICS use from pre‐study INTERVENTION PERIOD: 24 weeks in total (participants in budesonide group switched to combination therapy or separate administration of ICS and LABA after 12 weeks) TEST GROUP: Combination budesonide 320 mcg bid + formoterol 9 mcg bid (+ placebo inhaler) CONTROL GROUP: Budesonide 400 mcg bid + placebo inhaler DEVICE: Turbuhaler NUMBER OF DEVICES: 1 COMPLIANCE: Self‐report (98%) CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1 SYMPTOM SCORES: Total symptoms (0 to 6); symptom‐free days % FUNCTIONAL STATUS: Asthma control days (%); rescue medication free days (%); puffs/day INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Not reported by treatment group Primary outcome measure* |
|
| Notes | Full‐text publication Source of funding AstraZeneca Confirmation of methodology and data not obtained User defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated, blocks of 8 |
| Allocation concealment? | Low risk | Codes "assigned to patients and kept in sealed envelopes until data analysis." |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "Efficacy analysis was performed on all randomized patients (intention to treat population) over 12 weeks." |
| Free of selective reporting? | Unclear risk | Data presented in trial report for mild exacerbations; unclear with OCS‐treated exacerbations collected and reported in the trial |
Jenkins 2006b.
| Methods | See Jenkins 2006a | |
| Participants | See Jenkins 2006a; except for: RANDOMISED: 230 (BUD + F: 115; BUD: 115) |
|
| Interventions | See Jenkins 2006a except for: TEST GROUP: Budesonide 400 mcg bid + formoterol 9 mcg bid via separate inhalers |
|
| Outcomes | See Jenkins 2006a | |
| Notes | See Jenkins 2006a | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Jenkins 2006a |
| Allocation concealment? | Low risk | See Jenkins 2006a |
| Blinding? All outcomes | Low risk | See Jenkins 2006a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See Jenkins 2006a |
| Free of selective reporting? | Unclear risk | See Jenkins 2006a |
Kavaru 2000.
| Methods | Parallel‐group, multicentre (42 centres); 4 treatment arms of which 2 considered in this review | |
| Participants | Asthmatic patients over 12 years % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 68 RANDOMISED: 182 randomised to treatment groups in this review: FP/SAL: 92; FP: 90 WITHDRAWALS: FP/SAL: 15; FP: 22 AGE mean: 39 years GENDER (% male): 56 SEVERITY: Moderate BASELINE FEV1 MEAN %: 64 BASELINE DOSE OF ICS (RANGE): BDP 300 to 500 mcg/day; triamcinolone acetate 600 to 1000 mcg/day; flunisolide 1000 mcg/day; FP 200 mcg/day ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: Asthma (ATS criteria) of at least 6 months duration; required pharmacotherapy for at least 6 months before study; inhaled corticosteroids for at least 1 month without change before study; 15% improvement in FEV1 post‐bronchodilator; female patients negative pregnancy test, surgically sterile, postmenopausal or using birth control EXCLUSION CRITERIA: History of life threatening asthma; hypersensitivity rxn to sympathomimetic drugs or corticosteroids; smoking in year before study or smoking history of > 10 pack‐years; received a course of oral corticosteroids in 6 months before study of use of any other prescription or OTC medication that could affect asthma or interact with other medications; abnormal CXR or EKG; history of diabetes glaucoma, hypertension EXCLUSION CRITERIA FOR RANDOMISATION DURING RUN‐IN: Unstable asthma during run‐in periods, i.e. more than 3 nights with awakenings, during 7 days before randomisation, more than 12 puffs of rescue medication/day for more than 3 days; FEV1 not within 15% of value obtained at beginning of screening |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: Reported weekly weeks 1 to 4 and thereafter 2‐weekly RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Placebo in addition to usual medication DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination fluticasone and salmeterol 100/50 mcg bid CONTROL GROUP: Fluticasone 100 mcg bid DEVICE: Diskhaler NUMBER OF DEVICES: 1 COMPLIANCE: Measured using dose counter on DISKUS device CO‐TREATMENT: Albuterol as needed; no other prophylactic asthma medication permitted |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1* SYMPTOM SCORES: Symptom score (rated daily on 6‐point scale) FUNCTIONAL STATUS: Rescue B2‐agonists (puffs per day); nocturnal awakenings (% of nights with no awakenings); % of days with no asthma symptoms OTHER: Probability of remaining in study over time* INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Described WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Full‐text publication Funded by Glaxo Wellcome Confirmation of methodology and data not obtained User‐defined number: 400 (mean ICS dose in LAB2 group in mcg/day of BDP‐equivalent: FP 200 x 2) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | High risk | "To compensate for differential withdrawal rates among the treatment groups, end‐point analyses were used. End‐point analyses included data from the final visit during treatment for patients completing the study and the last available data for patients who were withdrawn early." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available on request from GSK |
Kemp 1998.
| Methods | Parallel‐group, multicentre (44 centres in USA) | |
| Participants | Symptomatic asthmatic adolescents and adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 87 RANDOMISED: 506 (Salm 50 + ICS: 252; ICS: 254) WITHDRAWALS: Salm 50 + ICS: 25; ICS: 47 AGE: mean (range): 42 (12 to 85) GENDER (% male): 47 SEVERITY: Moderate BASELINE % PREDICTED FEV1: 63 BASELINE DOSE OF ICS: See below ASTHMA DURATION: Not described ATOPY(%): Not described ELIGIBILITY CRITERIA: Average daytime symptom score of 1 on a 0 to 3 point scale over a 2‐week screening period; use of a short‐acting bronchodilator on a daily basis; FEV1 of 40% to 80% predicted; >= 15% improvement from baseline in FEV1 following inhaled albuterol; use of one of the following inhaled corticosteroids on a daily basis at a fixed dose that is within package insert guidelines for a minimum of 6 weeks prior to the screening visit: beclomethasone (300 to 900 mcg/day), flunisolide (1000 to 2000 mcg/day), triamcinolone (600 to 1600 mcg/day) EXCLUSION CRITERIA: Concurrent tobacco use; oral corticosteroid therapy immunotherapy requiring dosage change; inability to withdraw asthma/allergy medication before PFTs at screening or clinic visits throughout the study; cystic fibrosis, COPD, any significant uncontrolled disease state other than asthma; any other significant illness; pregnancy or lactation; contraindication to study medications; unstable asthma requiring albuterol >= 12 puffs/day or 12 puffs for > 3 days/ week; hospitalisation for asthma within 3 months; mechanical ventilation during an asthma exacerbation within 2 years or > 2 albuterol (or equivalent) inhalers/month within 3 months of screening |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: Reported on day 1 and after 4, 8 and 12 weeks RUN‐IN PERIOD: 2 weeks DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: Salmeterol xinafoate 50 ug bid + usual but unspecified doses of ICS CONTROL GROUP: Placebo 2 puffs bid + usual but unspecified doses of ICS DEVICE: Metered dose inhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: Rescue short‐acting beta2‐agonist (albuterol aerosol) as needed |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1 SYMPTOM SCORES: Overall score FUNCTIONAL STATUS: Asthma Quality of Life Questionnaire scores; rescue medication use; awakenings; exacerbations (undefined); symptoms INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Described WITHDRAWALS: Reported Primary outcome measure not reported |
|
| Notes | Full‐text publication Funded by GSK Methodology and data extraction confirmed. User‐defined number: 600 (mean ICS dose in LAB2 group in mcg/day of BDP‐equivalent: between 300 to 900) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated |
| Allocation concealment? | Low risk | Assignment by opaque consecutive numbered envelopes |
| Blinding? All outcomes | Low risk | Identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "intent‐to‐treat population (i.e., all patients exposed to the study drug), and efficacy analyses (including AQLQ analyses) were done on the basis of data from the intent‐to‐treat population minus patients excluded because of significant protocol violations." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available for meta‐analysis |
Koopmans 2006.
| Methods | Parallel‐group trial, single‐centre trial | |
| Participants | Mild to moderately asthmatic adults % ELIGIBLE OF SCREENED POPULATION: 93 % RUN‐IN PARTICIPANTS RANDOMISED: 90 RANDOMISED: 54 (FP/SAL: 27; FP: 27) WITHDRAWALS: FP/SAL: 0; FP: 4 AGE: mean (range): 32 (19 to 59) GENDER: (% male) 33 SEVERITY: Mild to moderate (GINA stage 2 & 3) BASELINE % PREDICTED FEV1 (mean): 92.9 BASELINE DOSE OF ICS: 500 mcg/d ASTHMA DURATION: Not reported ATOPY (%): 100 ELIGIBILITY CRITERIA: Documented, mild to moderate persistent, allergic asthma (GINA II and III); sensitisation to house dust mite and/or cat dander and/or grass pollen, as determined by Radio‐Allergo‐Sorbent‐Test (RAST) and skin prick test; 18 to 60 years; FEV1 > 70 % predicted post‐SABA bronchial hyper‐responsiveness to histamine (PC20 histamine > 8.0 mg/ml at the end of the run‐in period); exacerbation‐free for 3 months prior to inclusion (defined as no requirement for oral steroids and/or antibiotics); no changes to regular asthma medication for 4 weeks before study entry; ability to use Diskus inhaler;reproducible lung function tests EXCLUSION CRITERIA: Comorbidity likely to interfere with the study; lower respiratory tract infection during 4 weeks before entry; use of theophyline, sodium cromoglycate, nedocromil sodium or anti‐leukotrienes during the study or antibiotics 4 weeks prior to the study; current smoking, regularly smoking within 6 months before entry or a smoking history of more than 10 pack‐years; pregnant or lactating females; inability to follow the therapy instructions; participation in another clinical trial within 4 weeks prior to the study |
|
| Interventions | LABA + ICS versus SAME DOSE ICS OUTCOMES: 52 weeks RUN‐IN: 4 weeks DOSE OF ICS DURING RUN‐IN: 500 mcg/d INTERVENTION PERIOD: 52 weeks TEST GROUP: Combination fluticasone and salmeterol 250/50 mcg bid CONTROL GROUP: Fluticasone 250 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: As needed SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; am PEF; pm PEF SYMPTOM SCORES: Morning scores; evening scores FUNCTIONAL STATUS: Rescue medication use INFLAMMATORY MARKERS: PC20; sputum eosinophils*; eosinophil cationic protein concentrations* ADVERSE EFFECTS: Not stated WITHDRAWALS: Stated Primary outcome measure* |
|
| Notes | Full‐text publication Source of funding: GSK Confirmation of methodology and data: Not obtained User defined number: 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "Intention to treat population was all subjects who received treatment" |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
Kuna 2006.
| Methods | Parallel‐group study conducted in 61 centres in Europe, Central America and New Zealand. Three treatment arms (BDF once daily; BDF twice daily; BUD once daily) | |
| Participants | Moderately severe asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not stated % RUN‐IN PARTICIPANTS RANDOMISED 94 RANDOMISED: 617 (ITT population: 616 ‐ once daily BDF: 202; once daily BUD: 207, additional treatment arm not considered by this review: twice daily BDF: 207) WITHDRAWALS BDF: 21; BUD: 23 AGE: mean (range): 45.4 (18 to 80) GENDER (% male): 42 SEVERITY: Moderate BASELINE % PREDICTED FEV1 (mean): 78.8 BASELINE DOSE OF ICS: 365 mcg/d ASTHMA DURATION: 11 years ATOPY (%): Not reported ELIGIBILITY CRITERIA: Diagnosis of asthma (at least 6 months); not optimally controlled despite daily ICS dose of 200 to 500 mg for 1 month before study entry; baseline FEV1 60% to 90% predicted normal; reversibility of FEV1 at least 12% post‐SABA EXCLUSION CRITERIA: Oral corticosteroids within one month; seasonal asthma (asthma exacerbated by seasonal increases in aeroallergens); respiratory infection in 4 weeks before study entry; severe cardiovascular disorder/any other significant disease; beta‐blocker therapy (including eye drops); history of heavy smoking (10 pack‐years) |
|
| Interventions | LABA + ICS versus SAME DOSE ICS alone OUTCOMES: 12 weeks RUN‐IN: 2 weeks DOSE OF ICS DURING RUN‐IN: BUD 200 mcg/d INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination budesonide and formoterol 160/9 mcg/d CONTROL GROUP: Budesonide 200 mcg/d DEVICE: Turbuhaler NUMBER OF DEVICES: 1 (double‐dummy design: use of additional inhaler to control for third treatment group in the study, combination BUD/F 320/18 mcg/d) COMPLIANCE: Self‐reported > 97% CO‐TREATMENT: As needed SABA (terbutaline or preferred SABA) |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1 SYMPTOM SCORES: % symptom‐free days FUNCTIONAL STATUS: Asthma control days; night‐time awakenings due to asthma; % rescue‐free days INFLAMMATORY MARKERS: Not stated ADVERSE EFFECTS: Stated WITHDRAWALS: Stated by treatment group Primary outcome measure* |
|
| Notes | Full‐text publication Source of funding: AstraZeneca Confirmation of methodology and data: Not obtained User defined number: 200 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Stated as intention‐to‐treat analysis for diary cards FEV1 analysed as last observation carried forward: "...treatment value was that obtained at the last clinic visit." |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
Langton Hewer 1995.
| Methods | Parallel‐group, single‐centre study | |
| Participants | Symptomatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported NUMBER RECRUITED NOT RANDOMISED: Not stated RANDOMISED: 23 (usual ICS + Sal 100 bid: 11; usual ICS: 12) WITHDRAWALS: Usual ICS + S: 0; usual ICS: 2 AGE median (range) years: 15 (12 to 17) GENDER (% male): 70 SEVERITY: Severe BASELINE % PREDICTED FEV1: 82 BASELINE DOSE OF ICS (start of run‐in): 400 ASTHMA DURATION: 13 years ATOPY (%): 100% ELIGIBILITY CRITERIA: Severe asthma (not defined but severe enough to be attending residential school for asthma and persistent symptoms) EXCLUSION CRITERIA: Already on LABA CRITERIA FOR RANDOMISATION DURING RUN‐IN: None specified |
|
| Interventions | LABA + ICS versus SAME DOSE (usual dose) of ICS OUTCOMES reported at 8 and 10 weeks RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Same as during study DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 8 weeks TEST GROUP: (Usual ICS + S): Salmeterol 100 mcg bid CONTROL GROUP: Usual ICS and placebo bid DEVICE: Diskhaler NUMBER OF DEVICES: 2 COMPLIANCE: Supervised in school taking medication by investigators CO‐TREATMENT oral steroids, methylxanthines and anticholinergics taken by 20% participants |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; am PEF; pm PEF SYMPTOM SCORES: Morning and evening symptom scores FUNCTIONAL STATUS: Rescue B2‐agonist; symptom‐free days/nights; exacerbation (requiring oral steroids); quality of life score INFLAMMATORY MARKERS: none ADVERSE EFFECTS: Described WITHDRAWALS: Described Primary outcome measure not reported |
|
| Notes | Full‐text publication Funded by charity Confirmation of methodology and data pending User‐defined number: Not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Not stated |
| Free of selective reporting? | Low risk | Data on OCS‐treated exacerbations available for meta‐analysis |
Leblanc 1996.
| Methods | Cross‐over, multicentre study. 4 treatment arms, of which 2 considered for this review | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 367 WITHDRAWALS: 66 (18%) AGE: mean (range) or mean (SD): 40 GENDER: (% male): 45 SEVERITY: Moderate BASELINE % PREDICTED FEV1: 77.1 BASELINE DOSE OF ICS: Not reported ASTHMA DURATION: < 1 years = 10; 1 to 5 years = 89; 6 to 10 years = 71; > 10 years = 197 ATOPY (%): Not reported ELIGIBILITY CRITERIA: >= 18 to 70 years old; demonstrated both FEV1 of at least 60% of their predicted value and an increase in FEV1 of at least 15% after inhalation of 200 ug salbutamol; on 4 of the last 7 days of the pre‐randomisation period, patients had to be either symptomatic or demonstrate a greater than 20% diurnal variation in PEF EXCLUSION CRITERIA: FEV1 < 60% of predicted having withheld inhaler bronchodilators for at least 4 hours previously; have lab or clinical evidence in the opinion of the investigator to suggest a serious or uncontrolled systemic disease; clinically significant abnormalities at Visit 1 lab test; have had a lower respiratory tract infection within previous 1 month; abnormal 12‐lead ECG measurement; experienced an acute asthma exacerbation requiring emergency room treatment within the past 3 months; been hospitalised for any aspect of their reversible airways disease within the past 12 months; required daily maintenance therapy with oral steroids within the past 3 months; required a booster course of oral prednisolone in excess of 10 mg prednisolone or equivalent per day within the previous month; a history of acute sudden deterioration of their asthma symptoms; are pregnant or lactating. Females of childbearing potential may be included in the study providing that in the opinion of the investigator is that they are taking adequate contraceptive precautions; hypersensitive to beta‐receptor agonists; treatment with beta‐receptor antagonists; known to abuse alcohol or drugs; unable to use the peak flow meter properly; unlikely to take their medication in the prescribed manner, complete daily record card properly or attend the clinic on the required occasions; unwilling to sign consent form; in the opinion of the investigator are unsuitable for this clinical trial ELIGIBILITY CRITERIA FOR RANDOMISATION DURING RUN‐IN: Not other criteria other than above reported |
|
| Interventions | Assumed to be single‐dose ICS and LABA versus same dose of ICS OUTCOMES: Reported from 14 daily observations from each 1 month treatment period RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Usual ICS DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: (Salm 50 bid) Salmeterol 50 ug bid CONTROL GROUP: Placebo DEVICE: Not reported NUMBER OF DEVICES: 2 COMPLIANCE: Medications taken recorded by patients in diary card CO‐TREATMENT: Salbutamol as rescue medication; other medication which could be taken concurrently provided they had been initiated at least one month prior to visit 1 and that the dose remain constant throughout the study: inhaled and intranasal corticosteroids, inhaled sodium cromoglycate, antihistamines and immunotherapy (e.g. Pollinex anti‐hay fever injection) |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; diurnal variation in PEF SYMPTOM SCORES: 0 to 10 FUNCTIONAL STATUS: Rescue medication use; symptom‐free days/nights; sleep disturbance INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Not described WITHDRAWALS: Described Primary outcome measure* |
|
| Notes | Full‐text publication Funded by GSK Methodology confirmed but data extraction not confirmed User‐defined number: Not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Low risk | Numbered coded randomisation envelopes supplied by pharmacy |
| Blinding? All outcomes | Low risk | Use of identical placebo |
| Incomplete outcome data addressed? All outcomes | High risk | Completers used as population analysed |
| Free of selective reporting? | Unclear risk | Not clear whether data on OCS‐treated exacerbations collected during study |
Li 1999.
| Methods | Parallel‐group; 3 groups of which 2 are considered for this review | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 70 RANDOMISED: 34 (Sal 50 mcg bid + usual ICS: 16; placebo + usual ICS: 18) WITHDRAWALS: Sal 50 mcg bid + usual ICS: 3; placebo + usual ICS: 2 AGE: mean (range) 35 (20 to 70) GENDER (% male): 53 SEVERITY: Mild BASELINE % PREDICTED FEV1: 84 BASELINE DOSE OF ICS (median): 400 ASTHMA DURATION: Not reported ATOPY (%): 87 ELIGIBILITY CRITERIA: > 20 to 70 years old; non‐smokers; diagnosed asthma treated for at least 12 months with ICS in a dose up to 500 mg of beclomethasone dipropionate or budesonide per day; FEV1 at baseline) >= 60% of its predicted value EXCLUSION CRITERIA: Having suffered from acute respiratory tract infection during the previous 4 weeks; change in asthma medication in < 4 weeks; admission to hospital with airway disease in the < 4 weeks; patients unable to discontinue use of methylxanthines, inhaled anticholinergics and oral steroids CRITERIA FOR RANDOMISATION DURING RUN‐IN: Symptom score of more than 2 on 7 of the last 14 days; required the use of rescue inhaled albuterol on more than 7 of the last 14 days; had a variation of more than 15% in PEF over a 24‐hour period on at least 7 of the last 14 days and some degree of symptoms and rescue medication use during that time |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: measured at 12 weeks RUN‐IN PERIOD: 2 to 6 weeks DOSE OF ICS DURING RUN‐IN: Same as baseline dose of ICS DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: Salmeterol 50 mcg bid + usual ICS CONTROL GROUP: Placebo + usual ICS DEVICE: Dry powder Diskhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: Not described |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; FEV1 SYMPTOM SCORES: score of 0 to 4 (mean/day) FUNCTIONAL STATUS: Rescue medication use; night awakenings OTHER: Methacholine challenge ‐ PD 20 methacholine before and after treatment INFLAMMATORY MARKERS: On BAL and bronchial biopsy; mast cells in BAL; eosinophils in BAL; lymphocytes in BAL; macrophages in BAL and bronchial biopsies ADVERSE EFFECTS: Not reported WITHDRAWALS: Reported Primary outcome not specified |
|
| Notes | Full‐text publication Funded by GSK, Alfred Foundation and the NH&MRC of Australia Confirmation of methodology and data obtained User‐defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated numbers in balanced blocks |
| Allocation concealment? | Low risk | Opaque consecutive numbered envelopes containing assignment |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | High risk | Completers analysed |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available for meta‐analysis |
Malone 2005.
| Methods | Parallel‐group, multicentre (66 centres in North America) | |
| Participants | Steroid‐using asthmatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 48 RANDOMISED: 203 (FP/SAL: 101; FP: 102) WITHDRAWAL: FP/SAL: 19; FP: 16 AGE mean: 8 years GENDER (% male): 64 ASTHMA SEVERITY: Mild‐moderate BASELINE % PREDICTED FEV1 mean: 80 BASELINE DOSE OF ICS (start of run‐in): 166 mcg (FP stratum) ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: 4 to 11 years; ATS defined asthma for at least 2 months; ICS therapy (BDP equivalent 252 to 336 mcg /d) for 1 month prior to entry; participants aged 6 to 11 required to have FEV1% predicted; participants aged 4 to 5 required to have am PEF 50% to 95% predicted; >/= 12% response to beta‐agonist at screening visit or within one year of screening visit EXCLUSION CRITERIA: History of life‐threatening asthma; hospitalisation with asthma twice or more in previous year; significant concurrent disease; oral or parenteral use of steroids in month prior to study entry CRITERIA FOR RANDOMISATION DURING RUN‐IN: am FEV1 50% to 95% predicted; daytime asthma (score at least 1)/use of SABA on 3+ days of last 7 days of run‐in; 70% or greater diary card entry |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: 12 weeks RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Usual maintenance dose INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination salmeterol 50/fluticasone 100 mcg bid CONTROL GROUP: Fluticasone 100 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; clinic PEF; am PEF; pm PEF SYMPTOM SCORES: Symptom scores; symptom‐free days FUNCTIONAL STATUS: OCS‐treated exacerbations; hospitalisations; use of reliever medication; SABA‐free days INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWAL: Stated *Primary outcome: Not identified (safety study) |
|
| Notes | Full‐text publication Funded by GSK User‐defined number: 400 Confirmation of data and methodology obtained |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical placebo used |
| Incomplete outcome data addressed? All outcomes | High risk | Last observation carried forward: "...all available data up to the time of discontinuation were included in the intent to treat population." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available on request from GSK |
Meijer 1995.
| Methods | Parallel‐group, single‐centre study | |
| Participants | Asymptomatic asthmatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 40 (Sal 50 mcg bid + ICS: 20; ICS + placebo: 20) WITHDRAWALS: Sal50 mcg bid + ICS: 0; ICS + placebo: 1 (5%) AGE: mean (SD): 11.4 (2.6) GENDER (% male): 58 SEVERITY: Mild BASELINE % PREDICTED FEV1: 94 BASELINE DOSE OF ICS: Twice daily 200 or 400 mcg beclomethasone dipropionate Rotadisk ASTHMA DURATION: 8.4 years ATOPY (%): 100 ELIGIBILITY CRITERIA: None reported EXCLUSION CRITERIA: None reported CRITERIA FOR RANDOMISATION DURING RUN‐IN: N/A |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: reported at 1, 8, 16 weeks RUN‐IN PERIOD: None DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 16 weeks TEST GROUP: Salmeterol 50 mcg bid + BDP 250 mcg bid CONTROL GROUP: BDP 250 mcg bid + placebo DEVICE: Dry powder inhaler (Diskhaler) NUMBER OF DEVICES: 2 COMPLIANCE: Returned powder disks counted CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1 predicted; PC20 doubling doses (DD); circadian variation (day‐night differences in FEV1) SYMPTOM SCORES: Only individual symptoms reported (yes/no) FUNCTIONAL STATUS: Rescue medication use INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Not reported WITHDRAWALS: Reported Primary outcome not specified |
|
| Notes | Full‐text publication Funded by GSK User‐defined number: 500 Confirmation of data and methodology not obtained |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Use of identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Not enough information presented to determine this |
| Free of selective reporting? | Unclear risk | Not clear whether data on OCS‐treated exacerbations collected during study |
Molimard 2001.
| Methods | Parallel‐group, multicentre study | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 97 (of 7 patients who discontinued before randomisation 3 failed to fulfil the selection criteria, 2 withdrew their consent and 2 were lost to follow up) RANDOMISED: 259 (Form 12 mcg bid: 130; on‐demand salbutamol (ODS): 129) WITHDRAWALS: 30 (Form 12 mcg bid: 12; ODS: 18) AGE: mean: Form 12 mcg bid: 39 years GENDER: (% male): 57 SEVERITY: Moderate BASELINE % PREDICTED FEV1: 73 BASELINE DOSE OF ICS (start of run‐in): Not reported; maximum dose 1000 mcg BDP equivalent ASTHMA DURATION: 15 years ATOPY (%): Not reported ELIGIBILITY CRITERIA: 18 years or over; moderate persistent asthma; taking daily treatment with an ICS, same one for at least 1 month prior to first visit; require daily treatment with inhaled bronchodilators; asthma defined according to criteria of ATS; FEV1 >= 60% of predicted normal value for patient; reversibility test (increase in FEV1 >= 10% of predicted value) had to be documented at first visit within 3 months prior to visit; refrain from taking salbutamol 6 hours before each spirometry EXCLUSION CRITERIA: Known hypersensitivity to sympathetic amines or to lactose; pregnancy or breast‐feeding; women of childbearing potential who did not use a reliable contraceptive method; significant change in the regular asthma medication; asthma exacerbation or respiratory tract infection in the month prior to the first visit; incapacity to use a metered‐dose inhaler correctly or to complete patient diary; concomitant treatments with theophylline, anticholinergic bronchodilators and inhaled or oral B2 agonists other than the trial medications were not allowed CRITERIA FOR RANDOMISATION DURING RUN‐IN: No additional criteria reported |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: measured at 12 weeks RUN‐IN PERIOD: 2 to 6 weeks DOSE OF ICS DURING RUN‐IN: Same as usual DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: (Form12 + ICS) formoterol 12 mcg bid + ICS CONTROL GROUP: (ODS) On‐demand salbutamol + usual ICS (up to 1000 ucg beclomethasone or 800 mcg budesonide or 500 mcg fluticasone per day) DEVICE: Dry powder Diskhaler NUMBER OF DEVICES: 2 (test group) COMPLIANCE: Not reported CO‐TREATMENT: Salbutamol prn |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF* (average of 2 weeks); FEV1; bronchial responsiveness to methacholine (PD20) SYMPTOM SCORES: Score of 0 to 4 (mean/day) FUNCTIONAL STATUS: Rescue medication use; nocturnal awakenings INFLAMMATORY MARKERS: BAL and bronchial biopsy; mast cells in BAL; eosinophils in BAL; lymphocytes in BAL; macrophages in BAL and bronchial biopsies ADVERSE EFFECTS: Not reported WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Full‐text publication Funded by GSK, Alfred Foundation and the NH&MRC of Australia Confirmation of methodology and data not obtained User‐defined number: Not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Low risk | Telephone notification of assignment by co‐ordinating centre |
| Blinding? All outcomes | High risk | Open label |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The analyses were carried out in the intent‐to‐treat population, i.e. in all randomized patients with a post‐baseline efficacy measurement." |
| Free of selective reporting? | Unclear risk | Not clear whether data on OCS‐treated exacerbations collected during study |
Morice 2008a.
| Methods | Parallel‐group, multicentre study (53 centres in South America, Europe, Hong Kong and Taiwan) | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 77 RANDOMISED: 622 (BUD: 207; BUD/F (DPI): 203; BUD/F (MDI): 212) WITHDRAWALS: BUD: 14 BUD/F (DPI): 11; BUD/F (MDI): 14 AGE: mean (range): 9 (6 to 11 years) GENDER (% male): 66 SEVERITY: Not specified BASELINE % PREDICTED FEV1: 89 BASELINE DOSE OF ICS: (Start of run‐in): 470 mcg ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: Age 6 to 11 years; diagnosis of asthma for at least 6 months; PEF > 50% of predicted normal; history daily ICS use (stable dose of 375 to 1000 mcg 30 days prior to enrolment); clinically important exercise‐induced bronchoconstriction for 3 months before enrolment; ability to use DPI, pMDI and peak flow meter EXCLUSION CRITERIA: Not reported CRITERIA FOR RANDOMISATION DURING RUN‐IN: Symptom score 1 to 4; mean morning PEF 50% to 85% post‐SABA |
|
| Interventions | OUTCOMES: 12 weeks RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: 470 DOSE OPTIMISATION PERIOD: Not reported INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination formoterol and budesonide (160/9 mcg) bid via dry powder inhaler + placebo metered dose inhaler CONTROL GROUP: Budesonide 100 mcg bid DEVICE: BUD/F MDI and budesonide: MDI; BUD/F DPI: DPI NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: Terbutaline |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1 SYMPTOM SCORES: Day/night scores FUNCTIONAL STATUS: Paediatric AQLQ INFLAMMATORY MARKERS: NA ADVERSE EFFECTS: Stated WITHDRAWALS: Stated Primary outcome measure* |
|
| Notes | Full‐text publication AZ funded User defined: 200 Confirmation of data and methodology not obtained |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated schedule (blocks of 6) |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The intent‐to‐treat (ITT) population (i.e. all randomised patients with post‐randomisation data) was used for the main efficacy analyses." |
| Free of selective reporting? | Unclear risk | Not clear whether data on OCS‐treated exacerbations collected during study |
Morice 2008b.
| Methods | As above | |
| Participants | As above | |
| Interventions | As above except for: TEST GROUP: Combination formoterol and budesonide (160/9 mcg) bid via metered dose inhaler + placebo dry powder inhaler | |
| Outcomes | As above | |
| Notes | As above | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See above |
| Allocation concealment? | Unclear risk | See above |
| Blinding? All outcomes | Low risk | See above |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See above |
| Free of selective reporting? | Unclear risk | See above |
Nathan 2006.
| Methods | Parallel‐group, multicentre study (45 centres in USA) | |
| Participants | Moderately severe asthmatic adults on ICS % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 48 RANDOMISED: 365 (185 to study groups of interest to the review: FP/SAL: 94 FP: 91) WITHDRAWALS: FP/SAL: 13; FP: 20 AGE: mean (range): 39 (12 to 82) GENDER (% male): 38 SEVERITY: Moderate BASELINE % PREDICTED FEV1 (mean): 69 BASELINE DOSE OF ICS: 470 mcg FP ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: > 12 years; ATS defined asthma for at least 6 months; FEV1 40% to 85% predicted; >/= 15% reversibility post‐SABA; 440 to 660 mcg/d FP or equivalent for at least 1 month prior to visit 1 with no change in regimen EXCLUSION CRITERIA: Not reported CRITERIA FOR RANDOMISATION DURING RUN‐IN: >/= 3 nights with awakenings requiring SABA over last 7 days of run‐in; >/= 3 days where SABA was used 12 times or more over last 7 days of run‐in |
|
| Interventions | LABA + ICS versus SAME DOSE ICS OUTCOMES: 12 weeks RUN‐IN: 2 weeks DOSE OF ICS DURING RUN‐IN: Usual dose of ICS INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination fluticasone and salmeterol 220/42 mcg bid CONTROL GROUP: Fluticasone 220 mcg bid DEVICE: MDI NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1* SYMPTOM SCORES: Daily symptom score; % days with no symptoms FUNCTIONAL STATUS: Night‐time awakenings; % nights with no awakenings; rescue medication use; % days with no rescue medication use INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Due to worsening asthma* Primary outcome measure* |
|
| Notes | Full‐text publication and unpublished data available from http://www.ctr.gsk.co.uk Source of funding: GSK Confirmation of methodology and data: Not obtained User defined number: 880 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices used |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The intent‐to‐treat (ITT) population included all patients who were randomized to treatment and received > 1 dose of study medication." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available on request from GSK |
Noonan 2006a.
| Methods | Parallel‐group, multicentre study (84 centres in USA). Five treatment groups (formoterol and placebo not considered in this review) | |
| Participants | Moderately severe asthmatic adults % ELIGIBLE OF SCREENED POPULATION: 43 RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 233 (BUD/F: 124; BUD: 109) WITHDRAWALS: BUD/F: 27; BUD: 39 AGE: mean (range): 40.7 (12 to 87) GENDER (% male): 35 SEVERITY: Moderate BASELINE % PRED FEV1 (mean): 68 BASELINE DOSE OF ICS: 580 mcg/d ASTHMA DURATION: 22 years ATOPY (%): Not stated ELIGIBILITY CRITERIA: >/= 12 years; documented history of asthma for >/= 6 months according to ATS; moderate to high doses of ICS for more than 4 weeks; FEV1 45% to 85% predicted; FEV1 reversibility of > 12% and > 200 mL EXCLUSION CRITERIA: Hospitalisation within previous 6 months; requirement for oral CS within previous 4 weeks; > 10 pack‐year smoking habit |
|
| Interventions | LABA + ICS versus SAME DOSE ICS OUTCOMES: 12 weeks RUN‐IN: 2 weeks DOSE OF ICS DURING RUN‐IN: BUD 160 mcg bid INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination budesonide and formoterol 320/9 mcg bid + placebo DPI CONTROL GROUP: Budesonide 320 mcg bid DEVICE: Combination BUD/F and BUD: Metered dose inhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1* SYMPTOM SCORES: Daytime symptoms; night‐time symptoms; % symptom‐free days FUNCTIONAL STATUS: % awakening‐free nights; rescue medication use (puffs/d) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Stated WITHDRAWALS: Stated Primary outcome measure* |
|
| Notes | Full‐text publication Source of funding: AstraZeneca Confirmation of methodology and data. OCS and hospitalisation obtained from AZ User defined number: 640 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated randomisation schedule, stratified by baseline ICS dose |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy design |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Population used for analysis comprised participants who received more than one dose of study medication |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available from AZ |
Noonan 2006b.
| Methods | As above | |
| Participants | Moderately severe asthmatic adults % ELIGIBLE OF SCREENED POPULATION: 43 RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 224 (BUD + F: 115; BUD: 109) WITHDRAWALS: BUD + F: 29; BUD: 39 AGE: mean (range): 40.7 (12 to 87) GENDER (% male): 35 SEVERITY: Moderate BASELINE % PREDICTED FEV1(mean): 68 BASELINE DOSE OF ICS: 580 mcg/d ASTHMA DURATION: 22 years ATOPY (%): Not stated As for Noonan 2006a |
|
| Interventions | LABA + ICS versus SAME DOSE ICS OUTCOMES: 12 weeks RUN‐IN: 2 weeks DOSE OF ICS DURING RUN‐IN: BUD 160 mcg bid INTERVENTION PERIOD: 12 weeks TEST GROUP: Budesonide 320 mcg bid + formoterol 9 mcg bid CONTROL GROUP: Budesonide 320 mcg bid DEVICE: BUD: Metered dose inhaler; formoterol DPI NUMBER OF DEVICES: 2 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | As for Noonan 2006a | |
| Notes | As for Noonan 2006a | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | As for Noonan 2006a |
| Allocation concealment? | Unclear risk | As for Noonan 2006a |
| Blinding? All outcomes | Low risk | As for Noonan 2006a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | As for Noonan 2006a |
| Free of selective reporting? | Low risk | As for Noonan 2006a |
Norhaya 1999.
| Methods | Cross‐over study; single‐centre in Malaysia | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 83 (5 patients were withdrawn as they did not meet the selection criteria, 3 were unable to record their symptoms satisfactorily, 1 had no nocturnal symptoms or significant PEF variability and 1 had taken prednisolone in excess of 10 mg per day) RANDOMISED: 25 (20 completed) WITHDRAWALS: 5 AGE: mean (SD): 41.8 years ( 9.5) GENDER (% male): 30 SEVERITY: Moderate BASELINE % PREDICTED FEV1: 68 BASELINE DOSE OF ICS (range): 885 (200 to 1600) ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: 15% improvement from baseline in FEV1 following salbutamol via Diskhaler EXCLUSION CRITERIA: Lower respiratory tract infection within previous 28 days; need for maintenance oral prednisolone > 10 mg/day within previous 28 days; pregnant or lactating women CRITERIA FOR RANDOMISATION DURING RUN‐IN: Night‐time symptom score >= 2/5 or diurnal variation in peak flow >= 20% on at least 3 nights in the 1 week run‐in |
|
| Interventions | LABA + ICS vs SAME dose ICS OUTCOMES: 4 weeks RUN‐IN PERIOD: 1 week DOSE OF ICS DURING RUN‐IN: Usual dose of ICS INTERVENTION PERIOD: 4 weeks per group with 2 week wash‐out in between DOSE OPTIMISATION PERIOD: None TEST GROUP: (Salm 50) salmeterol 50 ug bid + usual, but unspecified dose of ICS CONTROL GROUP: Placebo + usual, but unspecified, dose of ICS DEVICE: Diskhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: sodium cromoglycate, theophylline and short‐acting b2‐agonist (salbutamol) as needed |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1; FVC SYMPTOM SCORES: Score of 0 to 5 daytime; score of 0 to 4 night‐time FUNCTIONAL STATUS: Daytime dose of rescue bronchodilator; night‐time dose of rescue bronchodilator; episode‐free days; exacerbations requiring oral steroids INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Not reported WITHDRAWALS: Described *Primary outcome measure |
|
| Notes | Full‐text publication Funded by GSK Confirmation of methodology and data not obtained; GSK unable to provide confirmation User‐defined number: Not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Identical placebo used |
| Incomplete outcome data addressed? All outcomes | High risk | Completers used for analysis |
| Free of selective reporting? | Low risk | Data available for OCS‐treated exacerbations |
O'Byrne 2001a.
| Methods | Parallel‐group multicentre study (7 groups of which 2 considered here) | |
| Participants | Symptomatic asthmatic teenagers and adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 645 (BUD200: 322; BUD200 + F: 323) WITHDRAWALS: Not reported AGE: mean: 37 years GENDER (% male): 44 SEVERITY: Mild BASELINE % PREDICTED FEV1: 86.4 BASELINE DOSE OF ICS: Not reported (</= 400 mcg/d BUD) ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: >= 12 years of age with mild asthma; taking <= 400 mcg/daily of inhaled budesonide or its equivalent for >= 3 months; FEV1 >= 70% predicted normal after terbutaline CRITERIA FOR RANDOMISATION DURING RUN‐IN: Randomised patients demonstrated a need for 2 or more inhalations per week of rescue medication during the last 2 weeks of run‐in, a >= 15% variability in peak expiratory flows, or a >= 12% increase in FEV1 after terbutaline EXCLUSION CRITERIA: Experience 3 severe exacerbations during the initial 6 months or 5 exacerbations in total; 2 poorly controlled asthma days, defined as days with morning PEF values >= 2 above baseline, or with asthma awakening |
|
| Interventions | LABA + ICS vs SAME dose ICS OUTCOMES: Reported at 52 weeks RUN‐IN PERIOD: 4 weeks DOSE OF ICS DURING RUN‐IN: BDP 100 bid DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 52 weeks TEST GROUP: Budesonide 200 mcg + formoterol 9 mcg via separate inhalers CONTROL GROUP: Budesonide 200 mcg DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; FEV1 SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Percentage of days with symptoms; percentage of asthma awakenings; number of rescue inhalations; rate per patient per year of severe asthma exacerbations INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWAL: Reported *Primary outcome: time to the first severe asthma exacerbation defined as need for treatment with oral corticosteroids or hospital admission or emergency treatment for worsening asthma or a decrease in morning PEF > 25% from baseline |
|
| Notes | Full‐text publication Funded by AstraZeneca Confirmation of methodology and data not obtained User‐defined order: 400 (BUD 200 bid) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Low risk | Opaque consecutive numbered envelopes containing assignment |
| Blinding? All outcomes | Low risk | Identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The study was analyzed using intention–to–treat principles." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available from study report |
O'Byrne 2001b.
| Methods | Parallel‐group multicentre study (7 groups of which 2 considered here) | |
| Participants | Symptomatic asthmatic teenagers and adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 627 (BUD400: 312; BUD400 + F: 315) WITHDRAWALS: Not reported by subgroup AGE mean: 37 years GENDER (% male): 42 SEVERITY: Mild BASELINE % PREDICTED FEV1: 87 BASELINE DOSE OF ICS : </= 400 mcg/d BUD ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: >= 12 years of age with mild asthma; taking <= 400 mcg/daily of inhaled budesonide or its equivalent for >= 3 months; FEV1 >= 70% predicted normal after terbutaline. EXCLUSION CRITERIA: Experience 3 severe exacerbations during the initial 6 months or five exacerbations in total; 2 poorly controlled asthma days, defined as days with morning PEF values >= 2 above baseline, or with asthma awakening CRITERIA FOR RANDOMISATION FOLLOWING RUN‐IN: Randomised patients demonstrated a need for two or more inhalations per week of rescue medication during the last 2 weeks of run‐in, a >= 15% variability in peak expiratory flows, or a >= 12% increase in FEV1 after terbutaline. |
|
| Interventions | LABA + ICS vs SAME dose ICS OUTCOMES: Reported at 52 weeks RUN‐IN PERIOD: 4 weeks DOSE OF ICS DURING RUN‐IN: BUD 100 bid DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 52 weeks TEST GROUP: Budesonide 800 mcg + formoterol 9 mcg via separate inhalers CONTROL GROUP: Budesonide 800 mcg DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; FEV1 SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Percentage of days with symptoms; percentage of asthma awakening; rescue medication use; exacerbations (defined as need for treatment with oral corticosteroids or hospital admission or emergency treatment for worsening asthma or a decrease in morning PEF > 25% from baseline); rate per patient per year of severe asthma exacerbations* INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWAL: Reported Primary outcome measure* |
|
| Notes | Full‐text publication Funded by AstraZeneca Confirmation of methodology and data not obtained User‐defined number: 800 (BUD 400 bid) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See O'Byrne 2001a |
| Allocation concealment? | Low risk | See O'Byrne 2001a |
| Blinding? All outcomes | Low risk | See O'Byrne 2001a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See O'Byrne 2001a |
| Free of selective reporting? | Low risk | See O'Byrne 2001a |
Pauwels 1997a.
| Methods | Parallel‐group, multicentre study (71 centres in North America, Europe, and Middle East); 4 treatment arms | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 77 RANDOMISED: 423 (F12 + BUD 100 bid: 210; BUD 100 bid: 213) WITHDRAWAL: F12 + BUD 100 mcg bid: 62; BUD 100 mcg bid: 82 AGE: mean (range): 42 years (18 to 70) GENDER (% male): 51% SEVERITY: Moderate BASELINE % PREDICTED FEV1 mean: 76 BASELINE DOSE OF ICS mean (range): 822 (100 to 2000) ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: Asthma for at least 6 months; treated with an inhaled corticosteroid for at least 3 months; baseline FEV1 >= 50% predicted; >= 15% improvement following inhalation of 1 mg of terbutaline EXCLUSION CRITERIA: Use of beclomethasone > 2000 ug/day or budesonide by MDI > 1600 ug/day or budesonide by turbuhaler > 800 ug/day or fluticasone > 800 ug/day; >= 3 courses of oral steroids in past 6 months; hospitalisation for asthma in past 6 months CRITERIA FOR RANDOMISATION DURING RUN‐IN: Compliance with 75% to 125% of the recommended dose of budesonide; stable asthma over the preceding 10 days as defined by the absence of the following criteria: diurnal variation of more than 20% in PEF on 2 consecutive days; use of 4 or more inhalations of rescue medication per day on 2 consecutive days;awakening due to asthma on 2 consecutive nights or the need to use oral glucocorticoids. |
|
| Interventions | LABA + ICS vs SAME dose ICS OUTCOMES: 1, 2, 3, 6, 9 and 12 months of treatment RUN‐IN PERIOD: 4 weeks DOSE OF ICS DURING RUN‐IN: BUD 800 bid DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 52 weeks TEST GROUP: Budesonide 100 mcg bid + formoterol 12 mcg bid CONTROL GROUP: Budesonide 100 mcg bid + placebo DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Yes ‐ hidden mechanical counter built into inhaler which could only be seen by investigators CO‐TREATMENT: prn SABA |
|
| Outcomes | OUTCOMES: Reported at 1, 2, 3, 6, 9 and 12 months PULMONARY FUNCTION TEST: FEV1 predicted; am PEF; pm PEF SYMPTOM SCORES: Mean day time and night‐time symptom scores at end of study (4‐point scale: averaged over 10 days) FUNCTIONAL STATUS: Rescue medication use; nocturnal awakening; *severe exacerbation (requiring oral steroids); episode‐free days (mean % of year) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWAL: Reported *Primary outcome measure: rates of severe and mild exacerbations of asthma per patient per year |
|
| Notes | Full‐text publication Funded by Astra Draco, Lund, Sweden Confirmation of methodology and data obtained User‐defined order: 200 (BUD 100 bid) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated randomisation sequence |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The analysis included all randomized patients (intention‐to‐treat approach). Data for patients who withdrew or discontinued therapy were included up to the time of their withdrawal." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available for meta‐analysis |
Pauwels 1997b.
| Methods | Parallel‐group, multicentre study (71 centres in North America, Europe, and Middle East); 4 treatment arms | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 77 RANDOMISED: 429 (F12 bid + BUD 400 bid: 215; BUD 400 mcg bid: 214) WITHDRAWAL: F12 bid + BUD 400 bid: 41; BUD 400 mcg bid: 60 AGE: mean (range): 42 years (17 to 70) GENDER (% male): 48 SEVERITY: Moderate BASELINE % PREDICTED FEV1: 76 BASELINE DOSE OF ICS (start of run‐in): 835 (100 to 2000) ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: Asthma for at least 6 months; treated with an inhaled corticosteroid for at least 3 months; baseline FEV1 >= 50% predicted; >= 15% improvement following inhalation of 1 mg of terbutaline EXCLUSION CRITERIA: Use of beclomethasone > 2000 ug/day or budesonide by MDI > 1600 ug/day or budesonide by turbuhaler > 800 ug/day or fluticasone > 800 ug/day; >= 3 courses of oral steroids in past 6 months; hospitalisation for asthma in past 6 months CRITERIA FOR RANDOMISATION DURING RUN‐IN: Compliance with 75 to 125 % of the recommended dose of budesonide; stable asthma over the preceding 10 days as defined by the absence of the following criteria: diurnal variation of more than 20% in PEF on 2 consecutive days; use of 4 or more inhalations of rescue medication per day on 2 consecutive days;awakening due to asthma on 2 consecutive nights or the need to use oral glucocorticoids. |
|
| Interventions | LABA + ICS vs SAME dose ICS OUTCOMES: Reported at 1, 2, 3, 6, 9 and 12 months RUN‐IN PERIOD: 4 weeks DOSE OF ICS DURING RUN‐IN: BUD 800 bid DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 52 weeks TEST GROUP: Formoterol 12 mcg bid + budesonide 400 mcg bid CONTROL GROUP: Budesonide 400 mcg bid + placebo DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Yes; hidden mechanical counter built into inhaler which could only be seen by investigators CO‐TREATMENT: prn SABA |
|
| Outcomes | OUTCOMES: Reported at 1, 2, 3, 6, 9 and 12 months PULMONARY FUNCTION TEST: FEV1 predicted; am PEF; pm PEF SYMPTOM SCORES: Mean daytime and night‐time symptom scores at end of study (4‐point scale: averaged over 10 days) FUNCTIONAL STATUS: Rescue medication use; nocturnal awakening; *severe exacerbation (requiring oral steroids); episode‐free days (mean % of year) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWAL: Reported *Primary outcome measures: rates of severe and mild exacerbations of asthma per patient per year |
|
| Notes | Full‐text publication Funded by Astra Draco, Lund, Sweden Confirmation of methodology and data obtained User‐defined order: 200 (BUD 100 bid) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Pauwels 1997a |
| Allocation concealment? | Unclear risk | See Pauwels 1997a |
| Blinding? All outcomes | Low risk | See Pauwels 1997a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See Pauwels 1997a |
| Free of selective reporting? | Low risk | See Pauwels 1997a |
Pohunek 2006a.
| Methods | Parallel‐group, multicentre study (80 centres in Europe); 3 treatment groups | |
| Participants | Steroid‐using asthmatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 77 RANDOMISED: 429 (BUD/F: 216; BUD: 213) WITHDRAWAL: BUD/F: 14 BUD: 13 AGE mean (range) 8 (4 to 11) GENDER (% male): 67 ASTHMA SEVERITY: Mild‐moderate BASELINE % PREDICTED FEV1 mean: 92% BASELINE DOSE OF ICS (start of run‐in): 454 mcg/d ASTHMA DURATION: 3 ATOPY(%): Not reported ELIGIBILITY CRITERIA: 4 to 11 years; diagnosis of asthma (ATS) for a minimum period of 6 months; pre‐SABA PEF >/= 50% predicted; ICS treatment for at least 12 weeks before entry into the study, at a constant dose of 375 to 1000 mcg/d during the 30 days prior to enrolment; history an average of >/= 1 clinically important exercise induced bronchoconstriction per week during the 12 weeks months leading up to the study; ability to use Turbuhaler device and peak flow meter EXCLUSION CRITERIA: Oral, parenteral or rectal corticosteroids within 30 days; respiratory infection affecting asthma control within 30 days; any significant coexisting disease/disorder; known/suspected hypersensitivity to study medication or inhaled lactose; inhaled anticholinergics, ß‐blockers (including eye drops), xanthines and other anti‐asthma agents not permitted during the study POST‐RUN‐IN: Total asthma‐symptom score of at least one on a minimum of 4 of last 7 days of the run‐in period; during last 7 days of the run‐in, patients had to have a mean morning PEF of 50% to 85% of the post‐SABA PEF |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 12 weeks RUN‐IN PERIOD: 10 to 14 days DOSE OF ICS DURING RUN‐IN: Usual dose of ICS INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination budesonide/formoterol 200/6 mcg bid CONTROL GROUP: Budesonide 200 mcg bid DEVICE: Turbuhaler NUMBER OF DEVICES: 1 (double‐dummy design used; second inhaler device to deliver) COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1 SYMPTOM SCORES: 24‐hour symptom scores; symptom‐free days FUNCTIONAL STATUS: Rescue SABA use; reliever‐free days; night‐time awakenings; paediatric AQLQ INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWAL: Reported by treatment group Primary outcome measure |
|
| Notes | Full‐text publication Funded by AstraZeneca Confirmation of methodology and data: Obtained User‐defined order: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "Intent to treat analysis was performed using data from all randomized patients." |
| Free of selective reporting? | Unclear risk | Not clear whether data on OCS‐treated exacerbations collected during study |
Pohunek 2006b.
| Methods | As for Pohunek 2006a | |
| Participants | Steroid‐using asthmatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 77 RANDOMISED: 414 (BUD + F: 201; BUD: 213) WITHDRAWAL: BUD: 13; BUD + F: 11 Baseline characteristics and eligibility criteria as for Pohunek 2006a |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 12 weeks RUN‐IN PERIOD: 10 to 14 days DOSE OF ICS DURING RUN‐IN: Usual dose of ICS INTERVENTION PERIOD: 12 weeks TEST GROUP: Formoterol 4.5 mcg + budesonide 80 mcg bid (separate inhaler devices) CONTROL GROUP: Budesonide 200 mcg bid DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | As for Pohunek 2006a | |
| Notes | As for Pohunek 2006a | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | See Pohunek 2006a |
| Allocation concealment? | Unclear risk | See Pohunek 2006a |
| Blinding? All outcomes | Low risk | See Pohunek 2006a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See Pohunek 2006a |
| Free of selective reporting? | Unclear risk | See Pohunek 2006a |
Price 2002.
| Methods | Parallel‐group, multicentre (72 centres in 14 countries). Three treatment groups (of which 2 are considered here) | |
| Participants | Symptomatic asthmatic patients aged > 12 years % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 88 (of a total of 750 patients 87 were discontinued before randomisation; 15 due to adverse event, 51 failed eligibility criteria and 21 discontinued for other reasons) RANDOMISED: 663 (F12 bid + BUD 400 bid: 332; BUD 400 bid: 331) WITHDRAWALS: F12 bid + BUD 400 bid: 19; BUD 400 bid: 18 AGE mean years: 38.5 GENDER (% male): 42 SEVERITY: Mild to moderate BASELINE PEF % PREDICTED (SD): 74 (13) BASELINE DOSE OF ICS Mean: 358 ASTHMA DURATION : (%): < 1 year 8; 1 to 5 years: 25; > 5 years: 67 ATOPY (%): Not reported ELIGIBILITY CRITERIA: > 12 years; asthma diagnosed > 3 months; treated with ICS < 400 mcg/day at constant dose for at least 1 month prior to entry; asthma symptoms on at least 3 days per week EXCLUSION CRITERIA: Severe or recent unstable asthma; PEF < 50% predicted; oral corticosteroids, nebulised therapy, leukotriene antagonist or LABA within 4 weeks of study entry; upper respiratory infection, COPD CRITERIA FOR RANDOMISATION DURING RUN‐IN: To randomise into part 1: asthma symptoms on 3 of previous 7 days; >= reversibility after bronchodilator of > 12% or (% of predicted normal); diurnal variation of > 20% on at least one day during run‐in period |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: 4 weeks RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Usual ICS DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 4 weeks TEST GROUP (LABA + SINGLE DOSE ICS): Budesonide 400 mcg bid + formoterol 9 mcg bid CONTROL GROUP: Budesonide 400 mcg bid DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Missed doses recorded by patients in diary CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: Change in morning PEF; change in FEV1 SYMPTOM SCORES: Change in day and night‐time score FUNCTIONAL STATUS: Time to asthma control i.e. 3 consecutive nights with a symptom score of 0*; rescue medication use day and night (inhalations per day or night); daytime and night‐time symptoms; nights per week with sleep disturbance INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Not described Primary outcome measure* |
|
| Notes | Full‐text publication Supported by AstraZeneca Confirmation of methodology obtained User defined number (mean ICS dose in LAB2 group in mcg/day of BDP‐equivalent): 800 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Low risk | Numbered coded solutions supplied by pharmacy |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "Efficacy was analysed using an intent‐to‐treat approach using all available data." |
| Free of selective reporting? | Low risk | Exacerbations reported in full‐text article; OCS‐treated exacerbations could not be extracted |
Reddel 2007.
| Methods | Parallel‐group study. Dose of ICS titrated after 8 weeks. | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 82 (FP/SAL: 41; FP: 41) WITHDRAWALS: Not described AGE mean: 47 years SEVERITY: Not reported BASELINE % PREDICTED FEV1: 86 BASELINE DOSE OF ICS: 1000 mcg bid BDP equivalent ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: 18 to 80 years; using combination therapy (FPSAL 500/50 bid) for 4 weeks or more prior to study entry EXCLUSION CRITERIA: Not reported ELIGIBILITY CRITERIA DURING RUN‐IN: Not reported |
|
| Interventions | LABA plus ICS versus SAME DOSE ICS OUTCOMES 8 weeks RUN‐IN PERIOD: Not stated DOSE OPTIMISATION PERIOD: Not stated INTERVENTION PERIOD: 8 weeks TEST GROUP: Combination fluticasone and salmeterol 500/50 mcg bid CONTROL GROUP: Fluticasone 500 mcg bid NUMBER OF DEVICES: 1 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1 SYMPTOM SCORES: Asthma‐free days FUNCTIONAL STATUS: Exacerbations; asthma quality of life questionnaire INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Not stated WITHDRAWALS: Not stated |
|
| Notes | Conference abstract Funding source: GSK Confirmation of methodology and data: pending User defined: 2000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised, no other information reported |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Unclear risk | Information not available |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Information not available |
| Free of selective reporting? | Unclear risk | Information not available |
Russell 1995.
| Methods | Parallel‐group, multicentre study (78 centres) | |
| Participants | Symptomatic asthmatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 208 (Salm50 + ICS: 99; placebo + ICS: 109) WITHDRAWALS: Salm50 + ICS: 22%; placebo + ICS: 16.8% AGE: mean (SD): 10.2 (2.7) GENDER: (% male): 60 SEVERITY: Moderate BASELINE MEAN % PREDICTED FEV1: 78 BASELINE DOSE OF ICS: 750 mcg ASTHMA DURATION (%): < 1 year: 3; 1 to 5 years: 20; > 5 years: 77 ATOPY (%): 77 ELIGIBILITY CRITERIA DURING RUN‐IN: Morning PEF‐PP (percent predicted) <= 90 on 4 or more days of the last 10 days of the baseline period; either recorded symptoms on at least 7 of 14 days of the baseline period for which they used at least one salbutamol blister per episode; recorded a diurnal variation in PEF of >= 15% on at least 7 occasions during baseline period EXCLUSION CRITERIA: Received a course of oral corticosteroids; change in prophylactic therapy during the previous 2 weeks |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: Reported at 4, 8 and 12 weeks RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Continued on usual ICS of at least 400 mcg/day BDP DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: (Salm 50 + ICS) salmeterol 50 mg bid + ICS 400 to 2400 mg/day (average: 750 mcg/day) CONTROL GROUP: (Placebo + ICS) placebo + ICS 400 to 2400 mg/day (average 750 mcg/day) DEVICE: Diskhaler NUMBER OF DEVICES: 2 COMPLIANCE: Evaluated using patient kept record booklets CO‐TREATMENT: Salbutamol as needed and any other prophylactic asthma medication via Diskhaler |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF percent predicted*; pm PEF percent predicted SYMPTOM SCORES: Symptoms were recorded daily as either being present or absent, wheeze or cough during day or night FUNCTIONAL STATUS: Proportion symptom‐free days; proportion symptom‐free nights; rescue medication use INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Described WITHDRAWALS: Described Primary outcome measure* |
|
| Notes | Full‐text publication Funded by Allen & Hanburys Confirmation of methodology and data obtained. User‐defined number: 750 (750 mcg/day) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Low risk | Numbered coded envelopes supplied by pharmacy |
| Blinding? All outcomes | Low risk | Identical placebo |
| Incomplete outcome data addressed? All outcomes | High risk | "Total population used, this comprised of all subjects who received at least one puff of medication and recorded at least one day of valid diary or clinic data during the treatment period. Where a subject withdrew before completion of the study, data recorded after this withdrawal data was excluded." |
| Free of selective reporting? | Low risk | Data on OCS‐exacerbations available for meta‐analysis |
SAM40008.
| Methods | Parallel‐group, multicentre study (34 centres in Europe and New Zealand). Dose of ICS tapered at 6 weeks intervals from 500 to 250 to 100 mcg bid. Endpoint data reported at 6 weeks (stable 500 mcg bid dosing regimen) | |
| Participants | Moderately severe asthmatic adults maintained on high dose ICS % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 186 (FP/SAL: 93; FP: 93) WITHDRAWALS: Not reported for initial dosing phase AGE: mean (range) or mean (SD): 49 (15.5) GENDER: (% male): 47 SEVERITY: Moderate to severe BASELINE % PREDICTED FEV1 (mean): Not reported BASELINE DOSE OF ICS: Usual dose of ICS ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: >/= 18 years; documented history of asthma; 1500 to 2000 mcg/d BUD or equivalent (not FP) for 3 months prior to baseline EXCLUSION CRITERIA: Not reported |
|
| Interventions | LABA and ICS versus SAME DOSE ICS OUTCOMES: TIMING 6, 12, 18 and 26 weeks (outcome data taken from end of first stable dose phase 6 weeks) RUN‐IN: Not reported DOSE OF ICS DURING RUN‐IN: Not clear INTERVENTION PERIOD: 26 weeks (data taken at 6 weeks) TEST GROUP: Combination fluticasone and salmeterol 500/50 mcg bid CONTROL GROUP: Fluticasone 500 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1 SYMPTOM SCORES: Proportion of symptom‐free days FUNCTIONAL STATUS: Rescue medication use; exacerbations (hospital admission data available from Bateman 2008; minimum acceptable dose of ICS* INFLAMMATORY MARKERS: Not assessed ADVERSE EFFECTS: Reported (but not collected) WITHDRAWALS: Reported (but not collected) Primary outcome measure* |
|
| Notes | Full unpublished data set available from http://www.ctr.gsk.co.uk Source of funding GSK Confirmation of methodology and data: Not obtained User defined number: 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The Intent‐To‐Treat (ITT) Population, representing all subjects randomised to treatment who had taken at least one dose of study medication and had at least one post randomisation diary assessment, was the primary population for all safety and efficacy endpoints." |
| Free of selective reporting? | Low risk | Exacerbations described in trial report available; OCS‐treated exacerbations could not be used since data were reported across phases of the study |
SAM40012.
| Methods | Parallel‐group, multicentre study in Europe and Middle East | |
| Participants | Steroid‐using asthmatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 362 (FP/SAL: 181; FP: 181) WITHDRAWAL: FP/SAL: 3; FP: 10 AGE mean: 8 years GENDER (% male): 68 ASTHMA SEVERITY: Moderate BASELINE % PREDICTED FEV1 mean: Not reported BASELINE DOSE OF ICS (start of run‐in): Not reported ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: 400 to 500 mcg BDP equivalent; documented history of asthma EXCLUSION CRITERIA: Not reported ELIGIBILITY CRITERIA DURING RUN‐IN: Symptom score >/= 2 on 3 of last 7 days of run‐in |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: Reported at 6 months RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Not clear DOSE OPTIMISATION PERIOD: None reported INTERVENTION PERIOD: 24 weeks TEST GROUP: Combination salmeterol 50/fluticasone 100 mcg bid CONTROL GROUP: Fluticasone 100 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | OUTCOMES: Reported at 6 months PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1 SYMPTOM SCORES: Symptom‐free days FUNCTIONAL STATUS: Use of reliever medication; exacerbations (undefined) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWAL: Reported Primary outcome measure* |
|
| Notes | Full unpublished data set available from http://www.ctr.gsk.co.uk Source of funding: GSK Confirmation of methodology and data: Not obtained User defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "To be 'evaluable', subjects had to meet the entry and randomisation criteria, receive at least one dose of study medication and have completed at least one day's post‐randomisation diary information." |
| Free of selective reporting? | Low risk | Exacerbations described in trial report available; OCS‐treated exacerbations could not be identified from the data available |
SAS40024.
| Methods | Parallel‐group, 53 centres in USA | |
| Participants | Moderately asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 192 (FP/SAL: 102; FP: 90) WITHDRAWALS: FP/SAL: 4; FP: 3 AGE: mean (range) or mean (SD): 29.3 (11.2) GENDER (% male): 39 SEVERITY: Moderate BASELINE % PREDICTED FEV1 (mean): Not reported BASELINE DOSE OF ICS: FP 500 mcg/d or equivalent ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: 12 to 50 years; diagnosis of asthma for at least 6 months; treatment with FP500 mcg/d equivalent; use of SABA in 6 weeks prior to screening; 65% to 90% predicted; ability to perform stepped treadmill exercises; fall in FEV1 by 20% post‐exercise at screening and 2 to 4 weeks post open label treatment with FP250 EXCLUSION CRITERIA: Not reported |
|
| Interventions | PROTOCOL: Combination FP/SAL versus SAME DOSE FP OUTCOMES: TIMING 4 weeks RUN‐IN: 2 to 4 weeks DOSE OF ICS DURING RUN‐IN: FP 250 mcg bid INTERVENTION PERIOD: 4 weeks TEST GROUP: Combination fluticasone and salmeterol 250/50 mcg bid CONTROL GROUP: Fluticasone 250 mcg bid DEVICE: MDI NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1 post‐exercise*; FEV1; am PEF SYMPTOM SCORES: % symptom‐free days FUNCTIONAL STATUS: Not reported INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported by treatment group WITHDRAWALS: Reported by treatment group Primary outcome measure* |
|
| Notes | Unpublished data set available from http://www.ctr.gsk.co.uk Source of funding: GSK Confirmation of methodology and data: Obtained User defined number: 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhalers |
| Incomplete outcome data addressed? All outcomes | Low risk | "The ITT population consisted of all subjects who were randomized to study drug. All data collected on these subjects, including subjects who discontinued the study, was included." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbation data available on request from GSK |
SAS40036.
| Methods | Parallel‐group, multicentre study (85 centres in USA). Four treatment groups: FP/SAL; SAL; FP; MON (SAL & MON not considered in this review) | |
| Participants | Moderately severe asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 331 (FP/SAL: 172; FP: 159) WITHDRAWALS: FP/SAL: 29; FP: 59 AGE mean (SD): 41 (14) GENDER: (% male): 41 SEVERITY: Moderate BASELINE % PREDICTED FEV1 (mean): 69% to 70% BASELINE DOSE OF ICS: Participants had 2‐week run‐in on current ICS therapy followed by combination treatment for 4 weeks ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: > 15 years; ATS defined asthma; treatment with a fixed dose of ICS for at least 4 weeks prior to screening visit; FEV1 40% to 85% predicted; FEV1 > 12% post‐SABA EXCLUSION CRITERIA: Not reported |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 16 weeks RUN‐IN: 2 weeks (plus 4 weeks open label treatment with FP/SAL 100/50 mcg bid) DOSE OF ICS DURING RUN‐IN: Usual ICS INTERVENTION PERIOD: 16 weeks TEST GROUP: Combination fluticasone and salmeterol 100/50 mcg bid CONTROL GROUP: Fluticasone 100 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; FEV1 SYMPTOM SCORES: % symptom‐free days FUNCTIONAL STATUS: % rescue‐free days INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported by treatment group WITHDRAWALS: Reported by treatment group Primary outcome measure* |
|
| Notes | Unpublished data set from http://www.ctr.gsk.co.uk Source of funding GSK Confirmation of methodology and data: obtained User defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The Intent‐to‐Treat (ITT) population consisted of all subjects who were randomized to treatment and formed the basis for all safety and efficacy measures." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available on request from GSK |
SAS40037.
| Methods | Parallel‐group, multicentre study (87 centres in USA). Four treatment groups: FP/SAL; SAL; FP; MON (SAL and MON not considered in this review) | |
| Participants | Moderately severe asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 322 (FP/SAL: 161; FP: 161) WITHDRAWALS: FP/SAL: 38; FP: 54 AGE mean (SD): 41 (14.5) GENDER (% male): 39 SEVERITY: Moderate BASELINE % PREDICTED FEV1 (mean): Not reported BASELINE DOSE OF ICS: Not reported ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: > 15 years of age; ATS defined asthma; treatment with a fixed dose of ICS for at least 4 weeks prior to screening visit; FEV1 40% to 85% predicted; FEV1 > 12% post‐SABA EXCLUSION CRITERIA: Life‐threatening asthma; hospitalised with asthma in previous 6 months; concurrent respiratory disease, intermittent or seasonal asthma only‐RTI, or use of antibiotics within 14 days of visit 1 |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 16 weeks RUN‐IN: 2 weeks (plus 4 weeks open label treatment with FP/SAL 100/50 mcg bid) DOSE OF ICS DURING RUN‐IN: Usual ICS INTERVENTION PERIOD: 16 weeks TEST GROUP: Combination fluticasone and salmeterol 100/50 mcg bid CONTROL GROUP: Fluticasone 100 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1 SYMPTOM SCORES: % symptom‐free days FUNCTIONAL STATUS: % rescue‐free days INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported by treatment group WITHDRAWALS: Reported by treatment group Primary outcome measure* |
|
| Notes | Unpublished data set from http://www.ctr.gsk.co.uk Source of funding: GSK Confirmation of methodology and data: Obtained User defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The Intent‐to‐Treat (ITT) population consisted of all subjects who were randomized to treatment and formed the basis for all safety and efficacy measures." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available on request from GSK |
SD 037 0344a.
| Methods | Parallel‐group, multicentre study in Central and South America, Southern Europe, Eastern Europe, South Africa and Asia). Three treatment groups. | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 639 (formoterol HFA: 216; formoterol DPI: 213; placebo: 210) WITHDRAWALS: Not stated AGE mean (range) or mean (SD): 35 (17) SEVERITY: Not reported BASELINE % PREDICTED FEV1: Not reported BASELINE DOSE OF ICS: Not reported ASTHMA DURATION: 14 years ATOPY (%): Not reported ELIGIBILITY CRITERIA: 12 years and older; treatment with 200 to 1000 mg/day of inhaled steroids for previous 3 months and stable dose for 30 days prior to run‐in period. EXCLUSION CRITERIA: Not reported ELIGIBILITY CRITERIA DURING RUN‐IN: Total asthma symptom score (night‐time plus daytime) of > 1 on at least 4 of the last 7 days of the run‐in period |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 12 weeks RUN‐IN PERIOD: 2 weeks DOSE OPTIMISATION PERIOD: Not stipulated INTERVENTION PERIOD: 12 weeks TEST GROUP: Formoterol 9 mcg via HFA pMDI plus usual ICS therapy CONTROL GROUP: Placebo in addition to usual ICS therapy NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1 SYMPTOM SCORES: Day and night‐time symptoms FUNCTIONAL STATUS: Use of relief medication; exacerbations (undefined) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Not reported |
|
| Notes | Unpublished data downloaded from AZ website (www.astrazenecaclinicaltrials.com) Funded by AstraZeneca Confirmation of data and methodology: Not obtained User defined order: NA |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "All efficacy analyses were performed on both the Per Protocol (PP) and Intention to Treat (ITT) populations, but the primary presentation of efficacy was based on the PP population". ITT and PP populations similar |
| Free of selective reporting? | Low risk | Exacerbations described in trial report; OCS‐treated exacerbations not available for meta‐analysis |
SD 037 0344b.
| Methods | As for SD 037 0344a | |
| Participants | As for SD 037 0344a | |
| Interventions | As for SD 037 0344a except for: TEST GROUP: Formoterol 9 mcg via Turbuhaler plus usual ICS therapy |
|
| Outcomes | As for SD 037 0344a | |
| Notes | As for SD 037 0344a | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | As for SD 037 0344a |
| Allocation concealment? | Unclear risk | As for SD 037 0344a |
| Blinding? All outcomes | Low risk | As for SD 037 0344a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | As for SD 037 0344a |
| Free of selective reporting? | Low risk | As for SD 037 0344a |
SD 039 0349.
| Methods | Parallel‐group, multicentre study in Western and Northern Europe | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 239 (BUD + F: 115; BUD: 124) WITHDRAWALS: BUD + F: 17; BUD: 16 AGE mean (range) or mean (SD): 46.7 SEVERITY: Not reported BASELINE % PREDICTED FEV1: 73.8 BASELINE DOSE OF ICS: 960 ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: Diagnosis of asthma; FEV1 predicted 50% to 90%; reversibility in FEV1 > 15 % of basal value; fixed‐dose ICS for 30 days prior to visit 1; daily inhaled dose had to be > 800 mcg inhaled budesonide Turbuhaler, > 500 mcg FP, > 1000 mcg BDP any formulation, or budesonide pMDI EXCLUSION CRITERIA: Use of oral, parenteral or rectal GCS within 30 days prior to visit 1; seasonal asthma; females who were pregnant or planning a pregnancy during the study; tobacco smokers or previous smokers, if they had a history of smoking > 10 pack‐years; use of any blocker therapy (including eye‐drops) ELIGIBILITY CRITERIA DURING RUN‐IN: Ability to use a peak flow meter; complete a daily diary card; morning PEF data recorded on at least 7 of the last 10 days of the run‐in period |
|
| Interventions | LABA and ICS versus SAME dose ICS OUTCOMES 12 weeks RUN‐IN PERIOD: 2 weeks DOSE OPTIMISATION PERIOD: NA INTERVENTION PERIOD: 12 weeks TEST GROUP: Budesonide 200 mcg bid and formoterol 9 mcg bid CONTROL GROUP: Budesonide 200 mcg bid NUMBER OF DEVICES: 2 COMPLIANCE: Not assessed CO‐TREATMENT prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; FEV1; pm PEF SYMPTOM SCORES: Awakenings FUNCTIONAL STATUS: Rescue medication use; participants withdrawn if required oral steroids INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Not reported WITHDRAWALS: Reported |
|
| Notes | Unpublished data downloaded from AZ website (www.astrazenecaclinicaltrials.com) Funded by AstraZeneca Confirmation of data and methodology: Not obtained User defined order: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The primary analysis was an intention‐to‐treat analysis, including all randomised patients who had received at least one dose of study medication." |
| Free of selective reporting? | High risk | Exacerbation data not reported (OCS requirement criterion for study withdrawal) |
SD 039 0714.
| Methods | Parallel‐group, multicentre study | |
| Participants | Steroid‐using symptomatic asthmatic adolescents % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 60% RANDOMISED: 271 (F6/BUD 200 mcg bid: 136; BUD 200 mcg bid: 135) WITHDRAWAL: F6/BUD 200 mcg bid: 25; BUD 200 mcg bid: 27 AGE: mean (range): 14 (11 to 17) GENDER (% male): 42 ASTHMA SEVERITY: Moderate BASELINE % PREDICTED FEV1 mean: 75 BASELINE DOSE OF ICS (start of run‐in): Not reported ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: ICS 375 to 1000 mcg BDP equivalent; FEV1 40% to 90% predicted normal; >= 12% improvement following inhalation of 1 mg of terbutaline EXCLUSION CRITERIA: Not reported CRITERIA FOR RANDOMISATION DURING RUN‐IN: Symptomatic |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: Reported at 1, 2 and 3 months RUN‐IN PERIOD: 2 weeks to document stability DOSE OF ICS DURING RUN‐IN: Not clear DOSE OPTIMISATION PERIOD: None reported INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination budesonide and formoterol 200/6 mcg bid CONTROL GROUP: Budesonide 200 mcg bid DEVICE: Turbuhaler NUMBER OF DEVICES: 1 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; am PEF*; pm PEF SYMPTOM SCORES: Recorded but not reported FUNCTIONAL STATUS: Rescue medication use (recorded but not reported); nocturnal awakening (recorded but not reported); episode‐free days (recorded but not reported) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWAL: Reported Primary outcome measure* |
|
| Notes | Unpublished data downloaded from AZ website (www.astrazenecaclinicaltrials.com) Funded by AstraZeneca Confirmation of data and methodology: Obtained User defined: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The statistical analysis was based on the intention to treat (ITT) population." |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
SD 039 0718.
| Methods | Parallel‐group; multicentre study (52 centres in USA) | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 60 RANDOMISED: 273 (BUD/F: 128; BUD: 145) WITHDRAWALS: BUD/F: 36; BUD: 51 AGE mean (range) or mean (SD): 10.4 (2.6) SEVERITY: Not stated BASELINE % PREDICTED FEV1: 82 BASELINE DOSE OF ICS: 235 mcg/d ASTHMA DURATION: 7 years ATOPY (%): Not stated ELIGIBILITY CRITERIA: 6 to 15 years; low to medium dose of ICS; FEV1 predicted > 50%; reversibility criteria age dependent: > 12 years 14% and 0.2L; < 12 years: 12% EXCLUSION CRITERIA: Not reported ELIGIBILITY CRITERIA DURING RUN‐IN: Symptoms and lung function not otherwise described |
|
| Interventions | LABA + ICS versus SAME DOSE ICS OUTCOMES: 12 weeks RUN‐IN PERIOD: 1 to 2 weeks DOSE OPTIMISATION PERIOD: Not applicable INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination budesonide/formoterol (100/9 mcg) bid via metered dose inhaler CONTROL GROUP: Budesonide 100 mcg bid via metered dose inhaler NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1 SYMPTOM SCORES: NA FUNCTIONAL STATUS: NA INFLAMMATORY MARKERS: NA ADVERSE EFFECTS: Stated WITHDRAWALS: Stated |
|
| Notes | Unpublished data from AZ clinical trials website Funded by AstraZeneca Confirmation of data and methodology: Obtained User defined: 200 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The efficacy analysis set (EAS) was defined as all randomized subjects who took at least 1 dose of study medication and contributed at least 1 PEF value to the primary endpoint." No information given on whether EAS population included last observation. |
| Free of selective reporting? | Unclear risk | OCS‐treated exacerbations were not reported in the study publication. Data request has been made to study sponsors for this information. |
SD 039 0719.
| Methods | Parallel‐group, multicentre study | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not stated % RUN‐IN PARTICIPANTS RANDOMISED: 74 RANDOMISED: 186 (BUD/F: 123; BUD: 63) WITHDRAWALS: BUD/F: 13; BUD: 10 AGE mean (range) or mean (SD): 9 (1.7) SEVERITY: Not stated BASELINE % PREDICTED FEV1: 84% BASELINE DOSE OF ICS: 307 ASTHMA DURATION: 6 years ATOPY (%): Not stated ELIGIBILITY CRITERIA: 6 to 12 years; inhaled steroid‐dependent asthma; FEV1 > 50% of predicted; history of PEF or FEV1 reversibility 12%; subjects without history of reversibility must have demonstrated FEV1 reversibility as above at any time before Visit 2 EXCLUSION CRITERIA: Not reported ELIGIBILITY CRITERIA DURING RUN‐IN: Not reported |
|
| Interventions | LABA and ICS versus SAME dose ICS OUTCOMES: 26 weeks RUN‐IN PERIOD: 1 week DOSE OPTIMISATION PERIOD: NA INTERVENTION PERIOD: 26 weeks TEST GROUP: Combination budesonide and formoterol 160/4.5 mcg per actuation, 2 puffs bid via MDI CONTROL GROUP: Budesonide 160 mcg per actuation, 2 puffs bid via MDI NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; am PEF SYMPTOM SCORES: NA FUNCTIONAL STATUS: Paediatric AQLQ INFLAMMATORY MARKERS: NA ADVERSE EFFECTS: Adverse events* WITHDRAWALS: Stated per treatment group Primary outcome measure* |
|
| Notes | Downloaded from AZ clinical trials website (accessed 4 January 2008) Funded by AstraZeneca Confirmation of data: Provided by AZ in April 2008 User‐defined number: 640 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | High risk | Open label |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The safety analysis set was defined as all randomized subjects who took at least 1 dose of study medication. The safety analysis set was used for the analyses of efficacy, health economic, and safety variables." |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
SD 039 0725a.
| Methods | Parallel‐group; multicentre study | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: 35 % RUN‐IN PARTICIPANTS RANDOMISED: 79 RANDOMISED: 521 (BUD/F bid: 184; BUD/F qd: 168; BUD: 169) WITHDRAWALS: BUD/F bid: 21; BUD/F qd: 37; BUD: 33 AGE mean (range) or mean (SD): 10.3 (2.5) SEVERITY: Not stated BASELINE % PREDICTED FEV1: 78.3 (8.56) BASELINE DOSE OF ICS: 245.3 ASTHMA DURATION: 6.8 ATOPY (%): Not reported ELIGIBILITY CRITERIA: 6 to 15 years; diagnosis of asthma for at least 6 months; maintenance inhaled corticosteroids treatment for at least 4 weeks prior to screening; FEV1 predicted 60% to 90% predicted; reversibility of FEV1 of 12% or more and > 0.20 L from baseline; children > 11 years were required to demonstrate reversibility of > 12% only EXCLUSION CRITERIA: Not stated ELIGIBILITY CRITERIA DURING RUN‐IN: Stable asthma symptoms |
|
| Interventions | Combination ICS and LABA versus SAME DOSE ICS OUTCOMES: 12 weeks RUN‐IN PERIOD: 4 to 5 weeks DOSE OPTIMISATION PERIOD: Not reported INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination budesonide and formoterol 80/9 mcg bid via MDI CONTROL GROUP: Budesonide 160 mcg qd via MDI NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; am PEF; pm PEF* SYMPTOM SCORES: Day and nocturnal symptoms FUNCTIONAL STATUS: AQLQ INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Reported by treatment group |
|
| Notes | Funding source: AZ Confirmation of methodology and data obtained from AZ in April 2008 Unpublished data downloaded from: http://www.astrazenecaclinicaltrials.com User defined: 160 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The efficacy analysis set (EAS), defined as all randomized subjects who took at least 1 dose of double‐blind treatment and who contributed at least 1 evening PEF diary entry after receiving double‐blind medication, was used in the primary analysis." |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
SD 039 0725b.
| Methods | As for SD 039 0725a | |
| Participants | As for SD 039 0725a | |
| Interventions | As for SD 039 0725a except for test group: Combination budesonide and formoterol 160/9 mcg qd via MDI |
|
| Outcomes | As for SD 039 0725a | |
| Notes | As for SD 039 0725a | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | As for SD 039 0725a |
| Allocation concealment? | Unclear risk | As for SD 039 0725a |
| Blinding? All outcomes | Low risk | As for SD 039 0725a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | As for SD 039 0725a |
| Free of selective reporting? | Unclear risk | As for SD 039 0725a |
SD 039 0726a.
| Methods | Parallel‐group, multicentre trial (151 centres in USA) | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: 28 % RUN‐IN PARTICIPANTS RANDOMISED: 63 RANDOMISED: 446 (BUD/F 400 qd: 147; BUD/F 200 bid: 154; BUD 400 qd: 145) WITHDRAWALS: BUD/F 200 bid: 14.3%; BUD/F 400 qd: 17%; BUD/F 200 qd: 12.4%; BUD 400 qd: 19.3% AGE mean: 38 SEVERITY: Not reported BASELINE % PREDICTED FEV1: 75.3% BASELINE DOSE OF ICS: 382 ASTHMA DURATION: 19.7 ATOPY (%): Not reported ELIGIBILITY CRITERIA: > 16 years; documented clinical diagnosis of asthma for at least 6 months prior to screening; stable condition; maintenance asthma treatment with a low to medium dose ICS for at least 4 weeks prior to the screening; FEV1 60% to 90% predicted EXCLUSION CRITERIA: Not reported ELIGIBILITY CRITERIA DURING RUN‐IN: Stable during run‐in period |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 12 weeks RUN‐IN PERIOD: 4 to 5 weeks DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination budesonide and formoterol (400/12 mcg) qd CONTROL GROUP: Budesonide 400 mcg qd NUMBER OF DEVICES: 2 (double‐dummy design; LABA co‐delivered with ICS in one inhaler) COMPLIANCE: Not assessed CO‐TREATMENT prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1 SYMPTOM SCORES: Day symptoms; night symptoms FUNCTIONAL STATUS: Quality of life (AQLQ) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Reported |
|
| Notes | Unpublished trial data from www.astrazenecaclinicaltrials.com Funding source: AZ Confirmation of data and methodology: Not obtained User defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; other information not available |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The efficacy analysis set (EAS), defined as all randomized subjects who took at least 1 dose of double‐blind treatment and who contributed at least 1 evening PEF diary entry after receiving randomized study medication, was used in the primary analysis. Sensitivity analyses of evening PEF were performed using the per protocol (PP) analysis set." |
| Free of selective reporting? | Unclear risk | Not clear whether OCS‐treated exacerbations collected in the study |
SD 039 0726b.
| Methods | See SD 039 0726a | |
| Participants | See SD 039 0726a | |
| Interventions | As for SD 039 0726a except for: TEST GROUP: Combination budesonide and formoterol (200/6 mcg) bid |
|
| Outcomes | See SD 039 0726a | |
| Notes | See SD 039 0726a | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | See SD 039 0726a |
| Allocation concealment? | Unclear risk | See SD 039 0726a |
| Blinding? All outcomes | Low risk | See SD 039 0726a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See SD 039 0726a |
| Free of selective reporting? | Unclear risk | See SD 039 0726a |
SD 039 0728.
| Methods | Parallel‐group, multicentre study (77 centres in USA). Three treatment arms, of which 2 are considered here. | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: 62 % RUN‐IN PARTICIPANTS RANDOMISED: 88 RANDOMISED: 576 (BUD/F 640/18 bid: 443; BUD: 133) WITHDRAWALS: Not reported AGE mean (SD): 40 (16.5) SEVERITY: Moderate to severe asthma BASELINE % PREDICTED FEV1: 73 BASELINE DOSE OF ICS: 500 mcg/d ASTHMA DURATION: 22.7 years ATOPY (%) Not reported ELIGIBILITY CRITERIA: > 12 years of age; documented clinical diagnosis of moderate‐to‐severe asthma for at least 6 months prior to screening; stable condition; maintenance asthma treatment with a stable dose of inhaled corticosteroids (ICS) for at least 4 weeks; FEV1 > 45% of predicted normal EXCLUSION CRITERIA: Not reported ELIGIBILITY CRITERIA DURING RUN‐IN: Not reported |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 52 weeks RUN‐IN PERIOD: 2 weeks DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 52 weeks TEST GROUP: Combination budesonide and formoterol 800/24 mcg bid CONTROL GROUP: Budesonide 800 mcg bid NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1 SYMPTOM SCORES: Not measured FUNCTIONAL STATUS: Days without symptoms; exacerbations (defined as requirement for OCS, ED visit and hospitalisation); rescue medication use INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Reported due to adverse events |
|
| Notes | Unpublished data set available from www.astrazenecaclinicaltrials.com Funding source: AZ Data and methodology: Not obtained User defined: 1600 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; other information not available |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy design |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "...all randomized subjects who received at least 1 dose of randomized study drug, the post‐dose analysis set, consisting of all subjects who had clinic visit safety assessments measured 1‐2 hours after randomized treatment at all visits, was also used in the analysis of some safety data." |
| Free of selective reporting? | Low risk | Exacerbations including OCS‐treated events reported as composite endpoint. Separate data could not be extracted. |
SFA100314.
| Methods | Parallel‐group, multicentre study (51 centres in USA) | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 248 (FP/SAL: 124; FP: 124) WITHDRAWALS: FP/SAL: 13/124; FP: 22/124 AGE mean (range) or mean (SD): 11 SEVERITY: Not reported BASELINE % PREDICTED FEV1: Not reported BASELINE DOSE OF ICS: 400 mcg/day (BDP equivalent) ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: Age 4 to 17 years; diagnosed with persistent asthma; experienced activity‐induced bronchospasm EXCLUSION CRITERIA: Not reported ELIGIBILITY CRITERIA DURING RUN‐IN : Not reported |
|
| Interventions | PROTOCOL: LABA + ICS versus SAME dose ICS OUTCOMES: 4 weeks RUN‐IN PERIOD: 7 to 14 days DOSE OPTIMISATION PERIOD: Not reported INTERVENTION PERIOD: 4 weeks TEST GROUP: Combination fluticasone and salmeterol 100/50 mcg bid CONTROL GROUP: Fluticasone 100 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1 AUC SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Oral‐steroid treated exacerbations INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Reported |
|
| Notes | Funding source: GSK Confirmation of methodology and data obtained from GSK in August 2008 Unpublished data downloaded from: www.ctr.gsk.co.uk User defined: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The ITT population consisted of all subjects who were randomized to study drug." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbation data available from GSK on request |
SFA100316.
| Methods | Parallel‐group, multicentre study (49 centres in USA) | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 231 (FP/SAL: 113; FP: 118) WITHDRAWALS: FP/SAL: 7/113; FP 10/118 AGE mean (range) or mean (SD): 11.6 SEVERITY: Not reported BASELINE % PREDICTED FEV1: Not reported BASELINE DOSE OF ICS: FP 100 mcg ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: Diagnosed with persistent asthma for 3 months or longer; experience worsened asthma symptoms during physical activity; using or used an inhaled steroid for the last 4 weeks or longer (such as Aerobid, Azmacort, Flovent, Pulmicort, QVAR, or Vanceril). EXCLUSION CRITERIA: Use of oral steroids as either liquids, pills or injections to treat asthma within the last 3 months; intermittent, seasonal, or exercise‐induced asthma, and not persistent asthma; admitted to a hospital within the last 6 months due to asthma symptoms; poorly controlled medical conditions that may make study participation unsafe or inappropriate in the opinion of the study physician (such as cystic fibrosis, congenital heart disease, insulin dependent diabetes, glaucoma, drug allergies, etc.) ELIGIBILITY CRITERIA DURING RUN‐IN: Not reported |
|
| Interventions | PROTOCOL: LABA + ICS versus SAME dose ICS OUTCOMES: 4 weeks RUN‐IN PERIOD: 7 to 14 days DOSE OPTIMISATION PERIOD: Not reported INTERVENTION PERIOD: 4 weeks TEST GROUP: Combination fluticasone and salmeterol 100/50 mcg bid CONTROL GROUP: Fluticasone 100 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1 AUC SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Oral‐steroid treated exacerbations INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Reported |
|
| Notes | Funding source: GSK Confirmation of methodology and data obtained from GSK in August 2008 Unpublished data downloaded from: www.ctr.gsk.co.uk User defined: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The ITT population consisted of all subjects who were randomized to study drug." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbation data available from GSK on request |
SFCF4026.
| Methods | Parallel‐group, multicentre study (124 centres in France). Three treatment groups (FP/SAL 250/50; FP/SAL 100/50; FP250) | |
| Participants | Moderately severe well‐controlled asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 318 (FP/SAL: 159; FP: 159) WITHDRAWALS: FP/SAL: 18; FP: 30 AGE: mean (range) or mean (SD): 45 (16) GENDER (% male): 50 SEVERITY: Moderately severe BASELINE % PREDICTED FEV1 (mean): Not reported BASELINE DOSE OF ICS: 1000 mcg/d BDP ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: >/= 18 years of age; documented history of asthma for at least 6 months; treatment with high dose BDP and LABA for 4 weeks; symptoms < 2 days per week; use of rescue medication < 2 days and < 4 occasions per week; PEF > 80% every day during run‐in EXCLUSION CRITERIA: Significant smoking history; RTI in 4 weeks prior to randomisation; exacerbation in 4 weeks prior to baseline; use of depot steroid in 12 weeks prior to visit 1; change in asthma medication |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 24 weeks RUN‐IN: 8 weeks DOSE OF ICS DURING RUN‐IN: 500 mcg/d (combination FP/SAL 250/50 mcg bid) INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination fluticasone and salmeterol 250/50 mcg bid CONTROL GROUP: Fluticasone 250 mcg bid DEVICE: Diskus NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1 SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Exacerbations (not defined) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Reported Primary outcome measure* |
|
| Notes | Full unpublished data set available from http://www.ctr.gsk.co.uk Source of funding GSK Confirmation of methodology and data: Not obtained User defined number: 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "Full Analysis Set (FAS) population consisted of all subjects who received at least one dose of study medication and for whom the assessment data for at least one assessment criterion was available." |
| Free of selective reporting? | Low risk | Exacerbations reported as rates; severity of exacerbations not adequately defined for this review |
Shapiro 2000.
| Methods | Parallel‐group, multicentre study (42 centres). Four treatment arms, of which 2 are considered for this review | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 72% (of 484 patients screened 135 not eligible) RANDOMISED: 168 (Salm50 + ICS: 84; placebo + ICS: 84) WITHDRAWALS: Salm50 + ICS: 13; placebo + ICS: 22 AGE mean (range): 39 (12 to 69) GENDER: (% male): 51 SEVERITY: Moderate BASELINE FEV1 MEAN (SD): 67 BASELINE DOSE OF ICS mcg/day: BDP 462 to 672 mcg/day; triamcinolone acetate 1100 to 1600 mcg/day; flunisolide 1250 to 2000 mcg/day; FP 440 mcg/day) ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: Asthma (ATS criteria) of at least 6 months duration; required pharmacotherapy for at least 6 months before study and inhaled corticosteroids for at least 12 weeks before study; 15% improvement in FEV1 post‐bronchodilator; female patients negative pregnancy test, surgically sterile, postmenopausal or using birth control EXCLUSION CRITERIA: History of life threatening asthma; hypersensitivity rxn to sympathomimetic drugs or corticosteroids; smoking in year before study or smoking history of > 10 pack‐years; received a course of oral corticosteroids in 6 months before study of use of any other prescription or OTC medication that could affect asthma or interact with other medications; abnormal CXR or EKG; history of diabetes glaucoma, hypertension CRITERIA FOR RANDOMISATION DURING RUN‐IN: Unstable asthma during run‐in periods, i.e. more than 3 nights with awakenings during 7 days before randomisation, more than 12 puffs of rescue medication/day for more than 3 days, FEV1 not within 15% of value obtained at beginning of screening |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: Reported weekly weeks 1 to 4 and thereafter 2‐weekly RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Not reported DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: Combination fluticasone and salmeterol 250/50 mcg bid CONTROL GROUP: Fluticasone 250 mcg bid DEVICE: Diskhaler NUMBER OF DEVICES: 1 COMPLIANCE: Measured with dose counter CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF; pm PEF; FEV1* SYMPTOM SCORES: Asthma symptom score FUNCTIONAL STATUS: Rescue medication use; % nights with no awakenings; % days with no asthma symptoms INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Described WITHDRAWALS: Described Primary outcome measure* |
|
| Notes | Full‐text publication Funded by GSK Confirmation of methodology not obtained User‐defined number: 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices used |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "The population used for all efficacy analyses was the intent‐to‐treat population minus 13 patients at one site who were excluded because their data did not meet study standards." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available on request from GSK |
Simons 1997.
| Methods | Cross‐over, single‐centre study | |
| Participants | Asymptomatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 16 WITHDRAWALS: 2 (13%) AGE mean (range): 13.1 (12 to 16 years) GENDER (% male): 44 SEVERITY: Not described BASELINE % PREDICTED FEV1: 93.4 BASELINE DOSE OF ICS: 100 to 200 mcg BDP bid ASTHMA DURATION: 5.9 +/‐ 3.4 years ATOPY (%): 100 ELIGIBILITY CRITERIA: 12 to 18 years old; well‐controlled chronic asthma; diagnosed according to American Thoracic Society criteria; able to perform treadmill running tests; do pulmonary function tests satisfactorily; use a Nebulizer Chronolog correctly EXCLUSION CRITERIA: Any significant medical conditions other than mild asthma, allergic rhinitis, or eczema; respiratory tract infection, or an acute asthma exacerbation within the previous month; prednisone treatment, and emergency department visit or hospitalisation within 3 months; life‐threatening asthma episode or an adverse reaction to any B2‐adrenergic agonist, or used salmeterol previously CRITERIA FOR RANDOMISATION DURING RUN‐IN: NA |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES measured at: day 1 and 28 RUN‐IN PERIOD: Not specified DOSE OF ICS DURING RUN‐IN: Not reported DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 4 weeks WASH OUT PERIOD: 14 days TEST GROUP: Salmeterol 50 mcg once daily + BDP 100 to 200 mcg bid CONTROL GROUP: BDP 100 to 200 mcg bid + placebo DEVICE: Metered‐dose inhaler and Nebulizer Chronolog device NUMBER OF DEVICES: 2 COMPLIANCE: Medication usage recorded in patient diary. A device inserted into MDI recorded date, hour and minute of each inhalation. CO‐TREATMENT: prn SABA (200 ug up to 3 times daily) except that albuterol was not permitted 8 hours before each exercise test. If subjects had allergic rhinitis, they were permitted to use pseudoephedrine (Sudafed) 1 to 3 times daily as needed, except on the days when exercise tests were scheduled |
|
| Outcomes | PULMONARY FUNCTION TEST: Exercise challenge (max % fall in FEV1 from pre‐exercise baseline) SYMPTOM SCORES: Symptoms FUNCTIONAL STATUS: Rescue medication use; exacerbations requiring oral steroids INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Described Primary outcome: Not specified |
|
| Notes | Full‐text publication Funded by GSK Confirmation of data and methodology obtained User defined number: 300 (1/2 with BDP 100 bid; 1/2 with BDP 200 bid) |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Use of identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | No indication how withdrawals handled in the analysis |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available for meta‐analysis |
SMS40012.
| Methods | Parallel‐group, multicentre study (56 centres in France) | |
| Participants | Asthmatic adults with mild airway obstruction % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 188 (SAL: 93; PLA: 95) WITHDRAWALS: SAL: 15; PLA: 16 AGE: mean (range) or mean (SD): 40 GENDER (% male): 37 SEVERITY: Mild BASELINE % PREDICTED FEV1 (mean): 91 BASELINE DOSE OF ICS: 800 to 1200 mcg/day BDP equivalent ASTHMA DURATION: Not reported ATOPY (%): Not reported ELIGIBILITY CRITERIA: > 18 years; requirement for 800 to 1200 mcg/d BDP equivalent; SABA treatment in previous 3 months EXCLUSION CRITERIA: More than 1 day with PEF variation > 20%; hospitalisation with asthma in previous year; respiratory tract infection in previous month; oral steroid treatment in previous 3 months; treatment with anti‐leukotriene agent, theophylline, anticholinergic, LABA, fixed‐dose SABA CRITERIA FOR RANDOMISATION DURING RUN‐IN: am PEF > 80% predicted; SABA requirement < 3 x daily |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 24 weeks RUN‐IN: 2 weeks DOSE OF ICS DURING RUN‐IN: Not clear INTERVENTION PERIOD: 24 weeks TEST GROUP: Salmeterol 50 mcg bid in addition to usual maintenance steroid dose CONTROL GROUP: Placebo in addition to usual maintenance steroid dose DEVICE: Not reported NUMBER OF DEVICES: 2 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1; FVC SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Rescue medication use; time to treatment failure INFLAMMATORY MARKERS: ECP ADVERSE EFFECTS: Reported by treatment group WITHDRAWALS: Reported by treatment group Primary outcome measure* |
|
| Notes | Unpublished data set available from http://www.ctr.gsk.co.uk Source of funding GSK Confirmation of methodology and data: Not obtained User defined number: Unclear |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "ITT population was defined as all randomised subjects who had received at least one dose of study medication and who had at least one efficacy criterion." |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
Stelmach 2007.
| Methods | Parallel‐group single‐centre study in Poland | |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 58 (BUD/F: 29; BUD: 29) WITHDRAWALS: BUD/F: 0; BUD: 0 AGE mean (range) or mean (SD): 10 years SEVERITY: Moderate BASELINE % PREDICTED FEV1: 94% BASELINE DOSE OF ICS: < 400 mcg/d BDP equivalent ASTHMA DURATION: 4 years ATOPY (%): 100 ELIGIBILITY CRITERIA: 6 to 18 years; history of asthma requiring up to 400 mcg mcg daily BDP equivalent EXCLUSION CRITERIA: Upper RTI in previous 3 weeks; sinus disease requiring antibiotics within 4 weeks; oral steroids within 4 weeks of study entry; immunotherapy ELIGIBILITY CRITERIA DURING RUN‐IN: Not reported |
|
| Interventions | ICS and LABA versus SAME DOSE ICS OUTCOMES: 8 weeks RUN‐IN PERIOD: 4 weeks DOSE OPTIMISATION PERIOD: NA INTERVENTION PERIOD: 8 weeks TEST GROUP: Budesonide 200 mcg + formoterol 9 mcg via Turbuhaler CONTROL GROUP: Budesonide 200 mcg daily via Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1 predicted; FEF25‐75; SRaw SYMPTOM SCORES: Not reported FUNCTIONAL STATUS: Not reported INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Not reported WITHDRAWALS: Reported |
|
| Notes | Full‐text article Funded by grant from Lodz University, Poland Confirmation of data and methodology: Not obtained User defined: 200 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated randomisation list |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Double‐dummy design |
| Incomplete outcome data addressed? All outcomes | Low risk | All completed |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
Tal 2002.
| Methods | Parallel‐group, multicentre study (48 centres in 7 countries) | |
| Participants | Asymptomatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 286 (F + BDP: 148; BDP: 138) WITHDRAWALS: F/BDP: 9; BDP: 9 AGE: mean (range): 11 (4 to 17) GENDER (% male): 62 SEVERITY: Mild BASELINE % PREDICTED FEV1: 75 BASELINE DOSE OF ICS: 548 ASTHMA DURATION: 6.8 years ATOPY (%): Not reported ELIGIBILITY CRITERIA: 4 to 17 years old; asthma diagnosed minimum 6 months; FEV1 40% to 90%predicted and > 15% reversibility in FEV1 within 15 minutes of bronchodilator; constant dose ICS for prior 6 weeks (> 400 mcg budesonide turbuhaler, > 600 mcg budesonide via MDI, > 375 mcg fluticasone propionate or > 600 mcg CFC beclomethasone dipropionate) EXCLUSION CRITERIA: Unstable asthma (defined as use of oral, parenteral or rectal corticosteroids within 30 days of study commencement); respiratory tract infection within previous 4 weeks; if they had known hypersensitivity to study medications or inhaled lactose; use of inhaled ICS other than study medication not allowed CRITERIA FOR RANDOMISATION DURING RUN‐IN: No other additional criteria |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES measured at: 4,8 and 12 weeks RUN‐IN PERIOD: 2 to 4 weeks DOSE OF ICS DURING RUN‐IN: BUD 200 bid DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: Formoterol 12 mcg bid + BDP 200 mcg bid CONTROL GROUP: BDP 200 mcg bid and placebo DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA. If subjects had allergic rhinitis, they were permitted to use nasal corticosteroids; treatment with other asthma medication not permitted |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1 predicted SYMPTOM SCORES: Daily and nocturnal on 4‐point scale FUNCTIONAL STATUS: Rescue medication use; night‐time awakening; symptom‐free days INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Described Primary outcome measure* |
|
| Notes | Full‐text publication Source of funding: AstraZeneca Confirmation of data and methodology obtained User defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Low risk | "Individual treatment code envelopes were provided for each subject." |
| Blinding? All outcomes | Low risk | Double‐dummy design |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "An intention‐to‐treat analysis was used with all available data." |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
Teper 2005.
| Methods | Parallel‐group, single‐centre study | |
| Participants | Mild‐moderate asthmatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 82 (FP/SAL: 43; FP: 39) WITHDRAWAL: Not reported AGE mean: 10 years GENDER (male%): 59 ASTHMA SEVERITY: Mild to moderate BASELINE % PREDICTED FEV1: 95 BASELINE DOSE OF ICS (start of run‐in): Not reported ASTHMA DURATION: Not reported ATOPY(%): Not reported ELIGIBILITY CRITERIA: ATS diagnosed mild or moderate asthma; age 6 to 14 years participants; FEV1 > 70 % predicted; methacholine PC20 < 2 mcg/ml EXCLUSION CRITERIA: Not reported CRITERIA FOR RANDOMISATION DURING RUN‐IN: Not reported |
|
| Interventions | LABA + ICS versus SAME dose ICS OUTCOMES: 12 months RUN‐IN PERIOD: Unclear DOSE OF ICS DURING RUN‐IN: Not reported DOSE OPTIMISATION PERIOD: None reported INTERVENTION PERIOD: 52 weeks TEST GROUP: Combination fluticasone and salmeterol 125/25 bid CONTROL GROUP: Fluticasone 125 mcg bid DEVICE: MDI (+ aerochamber) NUMBER OF DEVICES: 1 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1 % predicted SYMPTOM SCORES: % symptom‐free days; % symptom‐free nights FUNCTIONAL STATUS: % SABA‐free days INFLAMMATORY MARKERS: PC20 ADVERSE EFFECTS: Reported WITHDRAWAL: Not reported Primary outcome: Not clear |
|
| Notes | Unpublished conference abstract Source of funding: Not reported Confirmation of data and methodology: Not obtained User defined number: 500 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information available |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Unclear risk | Described as double‐blind; no other information available |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Information not available |
| Free of selective reporting? | Unclear risk | Not clear whether the study collected information on exacerbations treated with OCS |
van der Molen 1997.
| Methods | Parallel‐group, multicentre trial (6 centres in the Netherlands; 10 centres in Canada) | |
| Participants | Asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 239 (ICS + F 48: 125; ICS: 114) WITHDRAWALS: ICS + F 48: 18; ICS: 13 AGE: mean: 42.8 GENDER (% male): 49 SEVERITY: Moderate BASELINE FEV1 MEAN (SD): 67.1 BASELINE DOSE OF ICS (mcg/d: n) <= 400 mcg: 45 401 to 800: 47 801 to 1600: 99 ASTHMA DURATION: 20.6 years ATOPY (%): 68 ELIGIBILITY CRITERIA: Asthma according to the definition of the ATS; regular use of any dose of inhaled corticosteroids; use of >= 5 inhalations of short‐acting beta2 agonist/week before entry visit; > 15% reversibility in baseline FEV1 after 2 inhalations of terbutaline or equivalent EXCLUSION CRITERIA: Use of oral steroids at any time in the last month; smoking history of > 20 pack‐years; FEV1 < 40% predicted; exacerbation of asthma symptoms in the last month; use of cromoglycate, theophylline or anticholinergics CRITERIA FOR RANDOMISATION DURING RUN‐IN: No additional criteria |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: Reported at 4, 12 and 24 weeks RUN‐IN PERIOD: 4 weeks DOSE OF ICS DURING RUN‐IN: Usual dose of ICS WASH‐OUT PERIOD: 4 weeks DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 24 weeks TEST GROUP: Formoterol 24 mcg bid + usual dose of ICS 400 to 1600/d (mean: 980 /day) CONTROL GROUP: Placebo + usual dose of ICS (400 to 1600/d) (mean: 1030/day) DEVICE: Turbohaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA. Cromoglycate, theophylline and anticholinergic drugs were not permitted. The dose of inhaled corticosteroids remained constant throughout |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; am PEF*; pm PEF SYMPTOM SCORES: Symptom score (score 0 to 3)* FUNCTIONAL STATUS: Blood pressure and pulse rate; rescue medication use; asthma exacerbations (number of courses of oral prednisolone) INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Described Primary outcome measure* |
|
| Notes | Full‐text publication Funded by Astra Draco Confirmation of methodology and data extraction: Obtained User‐defined number: 980 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | High risk | "...missing data were substituted according to the last value extended principle, were used to illustrate the lung function and asthma symptoms during the entire study." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations reported in full‐text article |
Verberne 1998.
| Methods | Parallel‐group, multicentre study (9 centres). Three groups of which 2 are considered in this review | |
| Participants | Asthmatic children % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 117 (BDP400 + Salm: 60; BDP400: 57) WITHDRAWALS: BDP400 + Salm: 5; BDP400: 4 AGE: mean (SD): 11 (2.6) years GENDER (% male): 65 SEVERITY: Mild BASELINE % PREDICTED FEV1: 88 BASELINE DOSE OF ICS (SD): 489 (153) ASTHMA DURATION mean (SD): 8.1 (3.2) ATOPY(%): 88 ELIGIBILITY CRITERIA: FEV1 between 55% and 90% predicted or a FEV1/FVC ratio of 50% to 75%; >= 10% improvement in FEV1 after inhalation of salbutamol; airway hyper‐responsiveness to methacholine (PD20); ability to reproduce lung function test; history of stable asthma for >= 1 month without exacerbation or respiratory tract infection; use of inhaled steroids between 200 and 800 mg/day for at least 3 months prior to the beginning of the study; EXCLUSION CRITERIA: Operations for congenital heart disease, oesophageal atresia, congenital or acquired anatomical malformation of the lungs or airways, dyskinetic cilia syndrome; bronchiectasis; bronchopulmonary dysplasia; diabetes; renal disease; other serious conditions which may influence the possibility of continuation of the study; were using oral corticosteroids continuously or inhaled corticosteroids at a dose of more than 800 mcg daily; were using B‐blocking agents or had used cromoglycate or nedocromil sodium within the previous 2 weeks; were allergic to B‐agonists; were pregnant or lactating, or females of childbearing age who in the opinion of the supervising physician were not taking adequate contraceptive precautions; an ongoing hyposensitising programme; inability to follow therapy instructions, inability to inhale medications adequately or inability to use peak flow meter. During study: non‐compliance with respect to study medication, completing the diary cards, clinic visits; withdrawal at own or investigators discretion; total number of course of oral corticosteroids more than allowed in study CRITERIA FOR RANDOMISATION DURING RUN‐IN: No additional criteria |
|
| Interventions | LABA + ICS vs SAME dose ICS OUTCOMES: Reported at 6, 12, 18, 24, 30, 36, 42, 48 and 54 weeks RUN‐IN PERIOD: 6 weeks DOSE OF ICS DURING RUN‐IN: BDP 200 bid INTERVENTION PERIOD: 54 weeks DOSE OPTIMISATION PERIOD: None TEST GROUP: (Salm50 + BDP200) salmeterol 50 mcg bid and beclomethasone 200 mcg bid CONTROL GROUP: (BDP 200 + placebo) beclomethasone 200 mcg bid + placebo DEVICE: Rotadisks in combination with a Diskhaler NUMBER OF DEVICES: 2 COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1; am PEF; pm PEF; FVC SYMPTOM SCORES: Asthma symptoms like wheezing, dyspnoea, exercise induced asthma and cough were scored in the morning and evening using a scale from 1 to 3 FUNCTIONAL STATUS: Rescue medication use; exacerbation (requiring oral steroids); height, body weight, heart rate, systolic and diastolic blood pressure were measured INFLAMMATORY MARKERS: Total IgE ADVERSE EFFECTS: Reported WITHDRAWALS: Reported *Primary outcome: airway calibre measured as FEV1 and airway responsiveness to methacholine |
|
| Notes | Full‐text publication Funded by GSK Confirmation of methodology and data obtained User‐defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Low risk | Telephone notification of assignment by co‐ordinating centre |
| Blinding? All outcomes | Low risk | Identical placebo |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "Where patients failed to complete their daily record cards for more than 7 d in any 14‐d period such assessments were not included in the analysis. Otherwise, when there were missing days in the record, pro rata adjustment was made to give a 2‐week assessment." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available for meta‐analysis |
Wallin 2003.
| Methods | Parallel‐group, multicentre study. Three treatment arms of which 2 are considered for this review | |
| Participants | Asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 37 (FP 200 bid + Sal 50 bid: 18; FP 200 bid: 19) WITHDRAWALS: FP 200 bid + Sal 50 bid: 4; FP 200 bid: 3 AGE mean: 43 GENDER: (% male): 50 SEVERITY: Not stated BASELINE FEV1 % PRED: 85 BASELINE DOSE OF ICS BDP equivalent (range): 600 to 1200 mcg/d ASTHMA DURATION years: 17 ATOPY (%): 62 ELIGIBILITY CRITERIA: Free of respiratory tract infection for 4 weeks before study CRITERIA FOR RANDOMISATION DURING RUN‐IN: Despite use of BUD/BDP 800 to 1200 mcg/day or FP 400 to 500 mcg/day patients were included if they had one or more of the following symptoms: symptoms on 6 or more days, symptoms on 4 or more nights; need for rescue bronchodilator on 6 or more nights, greater than 20% variation between AM and PM PEF on 4 or more days. One or more of the following pulmonary function criteria: at least 15% improvement in FEV1 after bronchodilator, 15% increase in PEF post bronchodilator compared to mean PEF on previous week; more than 20% variation between am and pm PEF on at least 4 consecutive days, PC20 methacholine < 4 mg/ml EXCLUSION CRITERIA: None specified |
|
| Interventions | LABA + ICS vs SAME dose of ICS OUTCOMES: Before and after 12 weeks treatment RUN‐IN PERIOD: 2 to 4 weeks DOSE OF ICS DURING RUN‐IN (mean): 876 DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: (FP200 + Sal 50 bid) fluticasone propionate 200 mcg bid + salmeterol 50 mcg bid CONTROL GROUP: (FP200 bid) fluticasone propionate 200 mcg bid DEVICE: Diskhaler (dry powder inhaler) NUMBER OF DEVICES: Not reported COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF predicted; pm PEF predicted; FEV1 SYMPTOM SCORES: None reported FUNCTIONAL STATUS: Exacerbations (requiring OCS treatment) INFLAMMATORY MARKERS: Submucosal mast cells; submucosal eosinophils; adhesion molecules and cytokines ADVERSE EFFECTS: Not reported by group WITHDRAWALS: Reported |
|
| Notes | Full‐text publication Funded by GSK Confirmation of methodology and data not obtained User‐defined number: 800 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Information not available |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available for meta‐analysis |
Weiler 2005.
| Methods | Parallel‐group, multicentre study (53 centres in USA) | |
| Participants | Moderately asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: Not reported RANDOMISED: 192 (FP/SAL: 102; FP: 90) WITHDRAWALS: FP/SAL: 4; FP: 3 AGE: mean (SD): 29.3 (11.2) GENDER (% male): 39 SEVERITY: Moderate BASELINE % PREDICTED FEV1 (mean): 78 BASELINE DOSE OF ICS: FP 500 mcg/day ASTHMA DURATION: 59% > 15 years ATOPY (%): Not reported ELIGIBILITY CRITERIA: 12 to 50 years; diagnosis of asthma for at least 6 months; treatment with FP 500 mcg/d equivalent; use of SABA in 6 weeks prior to screening 65% to 90% predicted; ability to perform stepped treadmill exercises; fall in FEV1 by 20% post‐exercise at screening and 2 to 4 weeks post open label treatment with FP250 EXCLUSION CRITERIA: Not reported |
|
| Interventions | PROTOCOL: LABA + ICS versus SAME dose ICS alone OUTCOMES: 4 weeks RUN‐IN: 2 to 4 weeks DOSE OF ICS DURING RUN‐IN: FP 250 mcg bid INTERVENTION PERIOD: 4 weeks TEST GROUP: Combination fluticasone and salmeterol 250/50 mcg bid CONTROL GROUP: Fluticasone 250 mcg bid DEVICE: MDI NUMBER OF DEVICES: 1 COMPLIANCE: Not assessed CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: FEV1 post exercise*; FEV1; am PEF SYMPTOM SCORES: % symptom‐free days FUNCTIONAL STATUS: OCS‐treated exacerbations INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported by treatment group WITHDRAWALS: Reported by treatment group Primary outcome measure* |
|
| Notes | Unpublished data available from http://www.ctr.gsk.co.uk Source of funding: GSK Confirmation of methodology and data: Obtained User defined number: 1000 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Appendix 2 |
| Allocation concealment? | Low risk | See Appendix 2 |
| Blinding? All outcomes | Low risk | Identical inhaler devices used |
| Incomplete outcome data addressed? All outcomes | Low risk | "The ITT population consisted of all subjects who were randomized to study drug. All data collected on these subjects, including subjects who discontinued the study, was included." |
| Free of selective reporting? | Low risk | OCS‐treated exacerbations available on request from GSK |
Zetterstrom 2001a.
| Methods | Parallel‐group, multicentre study (59 clinical centres in 6 countries). Three treatment arms. Two groups will be considered here and since the same control group is being used for both comparisons half the control group will be applied to each. | |
| Participants | Symptomatic asthmatic adults % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 89 RANDOMISED: 247 (F + Bud: 123; Bud: 124) WITHDRAWALS: F + Bud: 20; Bud: 16 Mean AGE years (range): 47.5 (18 to 78) GENDER (% male): 52 SEVERITY: Moderate BASELINE % PREDICTED FEV1: 73.4 BASELINE DOSE OF ICS: 954 ASTHMA DURATION (years): 18 ATOPY (%): Not reported ELIGIBILITY CRITERIA: Aged >= 18; treated with ICS >= 500 mcg/day for at least 1 month prior to entry; FEV1 between 50% to 90% of predicted normal; >= 15% reversibility after bronchodilator EXCLUSION CRITERIA: Oral corticosteroids within 30 days of study entry; smoking history <= 10 years; respiratory infection, seasonal asthma, severe cardiovascular disorder beta blocker therapy; pregnant or failure to use acceptable contraceptives in women of childbearing potential |
|
| Interventions | LABA + ICS versus SAME dose of ICS OUTCOMES: Measured at 4‐weekly intervals RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Usual ICS DOSE OPTIMISATION PERIOD None INTERVENTION PERIOD: 12 weeks TEST GROUP (LABA + SINGLE DOSE ICS COMBINATION INHALER): Combination budesonide 200 mcg bid + formoterol 6 mcg bid CONTROL GROUP: Budesonide 200 mcg bid DEVICE: Turbuhaler NUMBER OF DEVICES: 1 (study had double‐dummy design as BUD/F compared as combination and concomitant delivery) COMPLIANCE: Not reported CO‐TREATMENT: Not reported |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1 SYMPTOM SCORES: Change from baseline in total asthma symptom score (daytime and night‐time score graded 1 to 3 (1 mild; 2 moderate; 3 severe)) FUNCTIONAL STATUS: Rescue medication use; night‐time awakenings; symptom‐free days; asthma control days INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: No medication related side effect WITHDRAWALS: Described Primary outcome measure* |
|
| Notes | Full‐text publication Supported by AstraZeneca Confirmation of methodology and data extraction not obtained User defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Computer‐generated random numbers |
| Allocation concealment? | Low risk | Opaque consecutive numbered envelopes containing assignment |
| Blinding? All outcomes | Low risk | Use of identical placebo (double‐dummy) |
| Incomplete outcome data addressed? All outcomes | Unclear risk | "All efficacy variables were analysed on an intention‐to‐treat basis, using data from all patients who had taken more than 1 dose of study medication." |
| Free of selective reporting? | Low risk | Exacerbations reported including events requiring OCS‐treatment; could not be extracted for meta‐analysis |
Zetterstrom 2001b.
| Methods | See Zetterstrom 2001a | |
| Participants | As for Zetterstrom 2001a, except for RANDOMISED: 247 (F + BUD: 123; BUD: 124) WITHDRAWALS: F + BUD: 17; BUD: 16 |
|
| Interventions | As for Zetterstrom 2001a, except for TEST GROUP (LABA + SINGLE DOSE ICS SEPARATE INHALERS): budesonide 200 mcg bid + formoterol 6 mcg bid NUMBER OF DEVICES: 2 |
|
| Outcomes | See Zetterstrom 2001a | |
| Notes | Full‐text publication Supported by AstraZeneca Confirmation of methodology and data extraction not obtained User defined number: 400 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | See Zetterstrom 2001a |
| Allocation concealment? | Low risk | See Zetterstrom 2001a |
| Blinding? All outcomes | Low risk | See Zetterstrom 2001a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See Zetterstrom 2001a |
| Free of selective reporting? | Low risk | See Zetterstrom 2001a |
Zimmerman 2004a.
| Methods | Parallel‐group, multicentre study (27 centres in Canada). Three treatment arms comparing LABA/ICS with 2 doses of LABA and ICS alone. Two groups will be considered here and since the same control group is being used for both comparisons half the control group will be applied to each. | |
| Participants | Children aged >= 6 to 11 years % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 68 RANDOMISED: 196 (F + usual ICS bid: 95; usual ICS: 101) WITHDRAWALS: F + usual ICS: 7; usual ICS: 16 Mean AGE years (range): 9 (6 to 11) GENDER (% male): 63 SEVERITY: Moderate BASELINE % PREDICTED FEV1: 77.4 BASELINE DOSE OF ICS: 445 ASTHMA DURATION ( years): 5.7 ATOPY (%): Not reported ELIGIBILITY CRITERIA: Aged >= 12 years; clinical diagnosis of asthma according to ATS criteria for at least 12 months; treated with ICS for at least 3 month prior to entry; FEV1 between 50% to 90 % predicted normal; >= 15% reversibility after bronchodilator; asthma symptoms suggestive that additional therapy might be needed; able to use peak flow meter and turbuhaler, answer questions form the Pediatric Asthma Quality of Life Questionnaire and parent or guardian had to complete a daily diary card EXCLUSION CRITERIA: Oral corticosteroids or anti‐leukotrienes within 30 days of study entry, astemizole within 120 days, sodium cromoglycate or ketotifen within 7 days, salmeterol or formoterol within 72 hours or xanthines or antihistamines within 48 hours; nasal corticosteroids and immunotherapy permitted provided dose had been constant for at least 30 days and 90 days respectively prior to study entry; smoking history RANDOMISATION CRITERIA FOLLOWING RUN‐IN: Post‐bronchodilator reversibility of at least 12% of the pre‐bronchodilator value or at least 9% of predicted normal or diurnal variability or at least 15% on any 5 of the last 10 days of run‐in; 75% to 124% compliance with prescribed dose as assessed by diary card; symptoms during the last 10 days of run‐in (defined as having one or more of the following: 4 or more inhalations of rescue medication; daytime symptoms on 4 or more days, or night‐time awakening on 1 or more nights) |
|
| Interventions | LABA + Usual ICS versus usual dose of ICS OUTCOMES: Measured at trial entry and after 4, 8 AND 12 week intervals RUN‐IN PERIOD: 2 weeks DOSE OF ICS DURING RUN‐IN: Usual ICS DOSE OPTIMISATION PERIOD: None INTERVENTION PERIOD: 12 weeks TEST GROUP: Usual dose ICS + formoterol 12 mcg bid CONTROL GROUP: Usual dose ICS + placebo bid DEVICE: Turbuhaler NUMBER OF DEVICES: 2 COMPLIANCE: Measured during run‐in CO‐TREATMENT: prn SABA |
|
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; FEV1 (Note: mean value during treatment for 12 weeks reported rather than value at endpoint) SYMPTOM SCORES: Total asthma symptom score FUNCTIONAL STATUS: Rescue medication use; paediatric asthma quality of life score INFLAMMATORY MARKERS: Not described ADVERSE EFFECTS: Described WITHDRAWALS: Described Primary outcome measure* |
|
| Notes | Full‐text publication Supported by: Not stated Confirmation of methodology and data extraction not obtained User defined number (mean ICS dose in LABA group in mcg/day of BDP‐equivalent): 444 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Described as randomised; no other information presented |
| Allocation concealment? | Unclear risk | Information not available |
| Blinding? All outcomes | Low risk | Identical inhaler devices used |
| Incomplete outcome data addressed? All outcomes | Unclear risk | Information not available |
| Free of selective reporting? | Low risk | Exacerbations described as those requiring OCS‐treatment and those requiring increased inhaled steroid. Separate OCS‐treated exacerbation data could not be extracted |
Zimmerman 2004b.
| Methods | See Zimmerman 2004a | |
| Participants | As for Zimmerman 2004a, except for: RANDOMISED: 207 (F + usual ICS: 106; usual ICS: 101) WITHDRAWALS: F + usual ICS: 7; usual ICS: 16 |
|
| Interventions | As for Zimmerman 2004a, except for: TEST GROUP: Usual dose ICS + formoterol 6 mcg bid |
|
| Outcomes | See Zimmerman 2004a | |
| Notes | Full‐text publication Supported by: Not stated Confirmation of methodology and data extraction not obtained User defined number: (mean ICS dose in LABA group in mcg/day of BDP‐equivalent): 456 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | See Zimmerman 2004a |
| Allocation concealment? | Unclear risk | See Zimmerman 2004a |
| Blinding? All outcomes | Low risk | See Zimmerman 2004a |
| Incomplete outcome data addressed? All outcomes | Unclear risk | See Zimmerman 2004a |
| Free of selective reporting? | Low risk | See Zimmerman 2004a |
AQLQ = Asthma quality of life questionnaire
ATS = American Thoracic Society
AZ = AstraZeneca
BDP = beclomethasone
bid = twice a day
BUD = budesonide
COPD = chronic obstructive pulmonary disease
CS = corticosteroids
d = day
DPI = Dry powder inhaler
ED = emergency department
F = formoterol
FEV1 = forced expiratory volume in one second
Form = formoterol
FP = fluticasone
GSK = GlaxoSmithKline
ICS = inhaled corticosteroids
LAB2 = long‐acting ß2 agonist
mcg = microgram
MDI = metered dose inhaler
NA = not applicable
OCS = Oral corticosteroids
PEFR = peak expiratory flow rate
PP = per protocol
PRN = as needed
qd = four times a day
RTI = respiratory tract infection
SABA = short‐acting ß2 agonist
SAL = salmeterol
Salm = salmeterol
SD = standard deviation
SL = salmeterol
URTI = upper respiratory tract infection
VAS = visual analogue scale
vs = versus
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Aalbers 2004 | No group with inhaled corticosteroids alone |
| Adinoff 1998 | No consistent use of inhaled corticosteroids in either the intervention or control groups . Co‐intervention with other non‐steroidal anti‐asthmatic drugs not stable during the intervention period. |
| Ankerst 2003 | No group with inhaled corticosteroids alone |
| Anonymous 2003 | No group with inhaled corticosteroids alone |
| Arvidsson 1991 | No group with inhaled corticosteroids alone. |
| Aziz 1998 | Duration of intervention < 30 days |
| Aziz 1999a | Intervention duration < 30 days |
| Aziz 1999b | Outcome measure did not reflect asthma control |
| Aziz 2000 | Duration of intervention < 30 days |
| Bacci 2002 | No consistent co‐intervention with ICS |
| Baker 1998 | Duplicate references |
| Baki 1998 | No consistent intervention with ICS |
| Baraniuk 1999 | Compared LABA and ICS to increased dose of ICS |
| Bateman 1998 | The treatment and intervention groups compared the same classes of medications either in combination or with different delivery devices |
| Bateman 2003a | Increased dose of ICS in control group |
| Bateman 2003b | Control not ICS alone |
| Behling 1999 | Duration < 30 days |
| Bensch 2002 | Not a RCT |
| Berger 2001 | Duplicate references |
| Berggren 2001 | Intervention not regular but prn inhaled long‐acting beta2‐agonists. PM LABA versus PM SABA. Duplicate references. |
| Bergmann 2004 | Compared LABA and ICS to increase dose of ICS |
| Bernstein 2002 | Not a RCT |
| Bessmertny 2002 | Intervention not LAB2 agonists |
| Bijl‐Hofland 2001 | No consistent co‐treatment with ICS |
| Bjermer 2000 | Control not inhaled glucocorticoids alone but montelukast LABA not compared to ICS alone |
| Bjermer 2002 | Duplicate references |
| Bjermer 2003 | No group with ICS alone |
| Bloom 2003 | Compared LABA and ICS to increased dose ICS |
| Boonsawat 2003 | Outcome measures not asthma control |
| Booth 1993 | No consistent co‐intervention with ICS |
| Boskovska 2001 | Not a RCT |
| Bouchard 2000 | Comparison of LABA + ICS with higher dose ICS |
| Boulet 2003 | Increased dose of ICS in control group |
| Bouros 1999 | Increased dose of ICS in control group |
| Brambilla 1994 | Control intervention not ICS but rather slow‐release oral bela2‐agonists |
| Brambilla 2003 | Duration of intervention < 30 days |
| Braniuk 1999 | Not a RCT |
| Brenner 1988 | Intervention not regular inhaled long‐acting beta2‐agonists. Control intervention not ICS alone. |
| Britton 1992 | No group with inhaled corticosteroids alone (control is regular SAB2). No consistent intervention with inhaled glucocorticoids in all subjects. |
| Britton 1998 | The treatment and intervention groups compared the same classes of medications either in combination or with different delivery devices. Duplicate references. |
| Brogden 1991 | Not a RCT |
| Buchvald 2003 | No group with inhaled corticosteroids alone |
| Busse 1999 | No group with inhaled corticosteroids alone (control is LTRA). No consistent intervention with inhaled glucocorticoids in all subjects. |
| Busse 2003a | Increased dose of ICS in control group |
| Busse 2003b | Increased dose of ICS in control group |
| Byrnes 2000 | No group with inhaled corticosteroids alone ( control is LAB2 at a different dose and SAB2 as maintenance Tx) |
| Calhoun 2001 | No group with inhaled corticosteroids alone. (Control intervention is anti‐leukotrienes). Duplicate references. |
| Calverley 2002 | Pat ients not asthmatics |
| Castle 1993 | Not a RCT |
| Cazzola 2000 | Patients not asthmatics |
| Chan 2001 | Intervention not regular inhaled long‐acting beta2‐agonists. Control intervention not ICS alone (but oral prednisolone). Setting acute asthma ED. Duplicate references. |
| Chapman 1999 | Tx and i ntervention compared LAB2 and ICS but in combined versus concurrent devices |
| Cheer 2003 | Duplicate references |
| Cloosterman 2001 | No consistent co‐intervention with ICS. No group with inhaled corticosteroids alone. (Control is regular short‐acting beta2‐antagonist). |
| Condemi 1999 | Increased dose of ICS in control group |
| Condemi 2001 | No group with inhaled corticosteroids alone. (Control is another LAB2). Duplicate references. |
| Cook 2001 | Duplicate references |
| Corren 2007 | Study participants interrupted ICS therapy during run‐in |
| Crompton 1999 | No group with inhaled corticosteroids alone. (Control is oral bambuterol). |
| Currie 2002 | Control is increased dose of ICS |
| Currie 2003a | Duration of intervention < 30 days. Co‐intervention with non permitted Rx. |
| Currie 2003b | Co ‐intervention with non‐ permitted treatment. Duration of intervention < 30 days. |
| Currie 2003c | Duration < 1 month |
| D'Alonzo 1994 | No consistent co‐intervention with ICS ‐ approximately 1/4 of participants were taking regular inhaled corticosteroids at baseline. Control intervention was a short‐acting beta2 agonist. |
| Dahl 1989 | Intervention not inhaled LAB2 |
| Dahl 1991 | No consistent co‐treatment with ICS |
| Dal Negro 2001a | Not a RCT |
| Dal Negro 2001b | The treatment and intervention groups compared the same medications either in combination or with different delivery devices |
| Dal Negro 2002a | Not a RCT |
| Dal Negro 2002b | Control not ICS alone |
| Davis 2001 | Not a RCT |
| Del Rio‐Navarro 2001a | Outcome measures do not reflect asthma control (but rather serum potassium, CPK‐MB and ECG) |
| Del‐Rio‐Navarro 2001b | Outcome measures do not reflect asthma control (but rather saliva flow and IgA) |
| Dempsey 2000a | Control intervention not inhaled glucocorticoids alone. No consistent intervention with inhaled glucocorticoids in all subjects. |
| Dempsey 2000b | Not a RCT |
| Dente 2001a | Duplicate references |
| Dente 2001b | Not a RCT |
| Dicpinigaitis 2002 | Intervention not regular inhaled long‐acting beta2 agonist |
| Didier 1997 | Control intervention is not ICS: this is a randomised, open, parallel‐group, multicentre study comparing salmeterol with an oral bronchodilator, terbutaline |
| Djordjevic 1999 | Not a RCT |
| Dorinsky 2001a | Not a RCT |
| Dorinsky 2001b | Duplicate references |
| Eliraz 2001 | Both the treatment and control group compared ICS with LAB2 with different inhaler devices |
| Eliraz 2002a | Not a RCT |
| Eliraz 2002b | Not a RCT |
| Ericsson 2001a | Duplicate references |
| Ericsson2001b | Not a RCT |
| Everden 2002 | The treatment and intervention groups compared the same medications either in combination or with different delivery devices |
| Faurschou 1994 | Duration of intervention < 30 days. Intervention not regular inhaled long‐acting beta2‐agonists. |
| Faurschou 1996 | Control intervention not ICS alone (but regular SAB2) |
| Fish 2000 | Duplicate references |
| Fish 2001 | Control intervention not ICS (but rather anti‐leukotrienes) |
| Fitzpatrick 1990 | Duration of intervention < 30 days: the treatment period was only 2 weeks. No consistent intervention with ICS in all patients: 19/20 participants were taking regular ICS and 6 were taking oral steroids at baseline. Both treatment groups received different doses of long‐acting beta2‐agonists. |
| Fowler 2002 | Increased dose of ICS in control group |
| Fuglsang 1995 | Duration < 30 days |
| Gabrijelcic 2004 | Outcomes not related to asthma control |
| Giannini 1996 | Duration < 30 days |
| Giannini 1998a | Duration < 30 days. Duplicate references. |
| Giannini 1998b | Duration < 30 days |
| Giannini 1999 | Duration < 30 days |
| Giannini 2000 | Duration < 30 days. Intervention is not LAB2 but 1 dose of salbutamol. Control intervention is not ICS alone (but placebo). |
| Giannini 2001 | Duration of intervention < 30 days |
| Giannini 2002a | No consistent intervention with inhaled glucocorticoids in all subjects |
| Giannini 2002b | Not a RCT |
| Gizycki 2000 | No consistent intervention with inhaled glucocorticoids in all subjects. Duplicate references. |
| Gold 2001 | Control intervention not inhaled glucocorticoids alone |
| Green 2002 | No consistent intervention with inhaled glucocorticoids in all subjects |
| Greening 1994 | Increased dose of ICS in control group |
| Grosclaude 2003 | No group with inhaled corticosteroids alone |
| Grzelewska‐Rzymowska 2003 | No treatment with LABA |
| Gustafsson 1994 | Tx and intervention compared ICS + LAB2 combination therapy using 2 different devices |
| Hasani 2003 | No consistent intervention with inhaled glucocorticoids in all subjects |
| Haughney 2002 | Not a RCT |
| Heuck 1999 | Not a RCT |
| Heuck 2000 | Increased dose of ICS in control group |
| Hyland 1995 | Not a RCT |
| Ind 2002a | No ICS alone |
| Ind 2002b | No ICS alone |
| Ind 2003a | Increased dose of ICS in control group |
| Isabelle 2001 | Not a RCT |
| Jeffery 2002 | No group with inhaled corticosteroids alone. Intervention not regular inhaled long‐acting beta2‐agonists. |
| Jenkins 1995 | No group with inhaled corticosteroids alone. (LAB2 delivered with new propellant HFA134a). |
| Jenkins 2000 | Increased dose of ICS in control group |
| Jenkins 2002 | The treatment and intervention groups compared the same medications either in combination or with different delivery devices |
| Johansson 2001 | Increased dose of ICS in control group |
| Jones 1994 | No consistent intervention with ICS ‐ < 1/3 of participants were taking regular ICS at entry |
| Juniper 1995 | No consistent co‐intervention with ICS ‐ 80% were taking regular ICS at entry. No subgroup analyses available. |
| Juniper 1999 | Duplicate of Pauwel's study (NEJM 1997;337:1405‐11) |
| Kaik 2002 | No ICS alone |
| Kalberg 1998 | Increased dose of ICS in control group |
| Kalra 1996 | Duration < 30 days |
| Karaman 2007 | No prior ICS exposure |
| Kardos 2001 | Tx and intervention compared ICS + LAB2 in a fixed versus flexible schedule |
| Keith 2001 | Allocation to treatment group determined by pre‐study asthma therapy |
| Kelsen 1999 | Increased dose of ICS in control group |
| Kerwin 2001 | Duplicate references |
| Ketchell 2002 | Duration < 30 days |
| Kidney 1995 | No consistent intervention with inhaled glucocorticoids in all subjects |
| Kips 2000 | Increased dose of ICS in control group |
| Kirby 2000 | Subjects not asthmatics |
| Knobil 2000 | Control intervention not inhaled glucocorticoids alone |
| Knorr 2001 | Intervention is not LAB2 (but rather an anti‐leukotriene agent: montelukast) |
| Kraft 2003 | No consistent co‐treatment with ICS |
| LaForce 1994 | Not a RCT |
| Lai 1995 | Control intervention was not ICS alone but regular short‐acting beta2‐agonists instead of placebo. Duration < 30 days (2 weeks). Co‐intervention with non‐permitted drugs: oral steroids. |
| Lalloo 2000 | Duplicate references |
| Lalloo 2001a | Duplicate references |
| Lalloo 2001b | Duplicate references |
| Lalloo 2001c | Not a RCT |
| Lalloo 2003 | Increased dose of ICS in control group |
| Lange 2001 | Inadequate duration |
| Lazarus 2001 | No consistent co‐intervention with ICS ‐ intervention is monotherapy with LAB2 |
| Lee 2003 | Duration of control period less than 4 weeks |
| Lemanske 2001 | Complicated protocol. No data provided for comparison groups of interest. No consistent intervention with inhaled glucocorticoids in all subjects. |
| Lenney 1995 | Not a RCT |
| Leuppi 2003 | No consistent co‐treatment with ICS |
| LHSRG 2000 | Subjects not asthmatics (but rather have COPD) |
| Lindqvist 2001 | No consistent co‐treatment with ICS |
| Lindqvist 2003 | No consistent co‐treatment with ICS |
| Lipworth | Not a RCT |
| Lipworth 1996 | Not a RCT |
| Lipworth 1998 | Duration < 30 days |
| Lipworth 1999a | Duration < 30 days |
| Lipworth 1999b | Duration < 30 days |
| Lipworth 2000a | Duration < 30 days |
| Lipworth 2000b | Duration < 30 days |
| Lockey 1999 | No consistent co‐treatment with ICS |
| Lowhagen 2002 | Intervention not regular inhaled long‐acting beta2‐agonists |
| Lundback 2002 | No group with ICS alone |
| Lyseng‐Williamson 2003 | Outcomes not related to asthma control ‐ pharmacoeconomic review |
| Lötvall 2002 | The treatment and intervention groups compared the same medications either in combination or with different delivery devices |
| Magadle 2001 | Duration < 30 days. Duplicate references. |
| Malmqvist‐Granlund 2000 | Not a RCT |
| Malolepszy 2001 | Outcome of LABA in acute asthma rather than asthma control |
| Malolepszy 2002 | Control intervention not ICS (but oral theophylline). Duplicate references. |
| Martinat 2003 | No group with inhaled corticosteroids alone |
| Matz 2001 | Duplicate publication of 2 RCT s, namely that of Condemi JJ (Ann Allergy Asthma Immunol 1999;82:383‐9) and of Kalberg CJ (J Allergy Clin Immunol 1998;101 (Suppl):S6 |
| McCarthy 2000 | Control intervention not inhaled glucocorticoids alone |
| McCarthy 2001a | Not a RCT |
| McCarthy 2001b | Not a RCT |
| McCarthy 2002 | Not a RCT |
| McCarthy 2003 | No ICS alone group |
| Mcivor 1998 | No consistent co‐treatment with a stable dose of ICS (tapering) |
| Michel 2000 | Compared LABA with increased doses of ICS rather than the same dose. Intervention duration < 30 days. |
| Midgren 1992 | No group with inhaled corticosteroids alone |
| Miraglia del Giudice 2007 | No prior ICS exposure |
| Mitchell 2000 | Duration < 30 days. Duplicate references. |
| Mitchell 2003 | Control group had increased dose of ICS |
| Murray 1998 | No consistent intervention with inhaled glucocorticoids in all subjects. Duplicate references. |
| Murray 1999 | Increased dose of ICS in control group |
| Nathan 1995 | No consistent co‐intervention with ICS in all patients: only 1/4 of participants were taking regular ICS at entry. The usual dose of inhaled corticosteroids taken by participants was not stated in the manuscript. The control intervention was not ICS but a short‐acting beta2‐agonist. |
| Nathan 1999a | Not a RCT |
| Nathan 1999b | Not a RCT |
| Nathan 2001 | Not a RCT |
| Nelson 1999 | Not a RCT |
| Nelson 2000a | Not a RCT |
| Nelson 2000b | Duplicate references |
| Nelson 2001 | Control intervention not ICS alone (but LTRA ‐ zafirlukast) |
| Newnham 1995 | No consistent co‐treatment with ICS |
| Nielsen 1999 | Not a RCT |
| Nightingale 2002 | Treatment and intervention groups compared the same medications either in combination or with different delivery devices |
| Nsouli 2001 | No group with inhaled corticosteroids alone. Duplicate references. |
| O'Brian 2001 | Duration < 30 days |
| Odeback 1998 | Duplicate references |
| Olsson 2002 | Comparison of adjustable maintenance treatment with LABA + ICS rather than ICS alone |
| Ortega‐Cisnero 1998 | Increased dose of ICS in control group |
| Overbeck 2003 | Patients were steroid naive |
| Ozkaya 1999 | Not a RCT |
| Palmer 1992 | No group with inhaled corticosteroids alone ( treatment groups received different doses of long‐acting beta2‐antagonists) |
| Palmqvist 2001 | Both the treatment and control groups compared ICS and LAB2 with different drugs and inhaler devices (concurrent versus combined therapy) |
| Paterson 1999 | Treatment and intervention groups compared the same medications either in combination or with different delivery devices |
| Pauwels 1998 | Intervention not LAB2 but another ICS |
| Pearlman 1992 | No consistent co‐intervention with ICS ‐ < 1/2 the participants were taking regular inhaled corticosteroids at entry. No group with inhaled corticosteroids alone. (Control was short‐acting beta2‐agonists). |
| Pearlman 1994 | No consistent co‐treatment with ICS 26% |
| Pearlman 1999a | Not a RCT |
| Pearlman 1999b | Not a RCT |
| Pearlman 2001 | Not a RCT |
| Pearlman 2002 | No group with inhaled corticosteroids alone. (Control is anti‐leukotriene montelukast as maintenance). Duplicate references. |
| Peters 2000 | No group with inhaled corticosteroids alone. (Control is oral steroids, SAB2 and anticholinergics). In hospital setting. |
| Pieters 1999b | Duplicate references |
| Pieters 2001 | Duplicate references |
| Pinnas 1998 | No consistent intervention with inhaled glucocorticoids in all subjects. Duplicate references. |
| Pizzichini 1996 | Duration < 30 days. Outcomes measures did not reflect asthma control. |
| Pljaskic‐Kamenov 2000 | Cannot determine prior ICS exposure |
| Price 2003 | No ICS alone |
| Pujet 1995 | Intervention is not LAB2 (but theophylline) |
| Pyke 2001 | Comparison of LABA and ICS in separate versus combination devices. No ICS alone. Duplicate references. |
| Rance 2002 | Abstract |
| Rickard 1999 | Outcomes measures did not reflect asthma control |
| Rickard 2001 | Control intervention not inhaled glucocorticoids alone |
| Rijssenbeek‐Nouwens 2002 | Intervention is not LAB2 (but anti‐allergic casing) |
| Ringbaek 1996 | No group with inhaled corticosteroids alone. (Control is oral SAB2 as maintenance). |
| Ringdal 1997 | Not a RCT |
| Ringdal 2002 | Abstract |
| Ringdal 2003 | Control intervention no inhaled glucocorticoids alone. Outcomes measures did not reflect asthma control. |
| Rocca‐Serra 2002 | Intervention not regular long acting beta2 agonists. Duration < 30 days. |
| Rooklin 2001 | Not a RCT |
| Rosenhall 2001a | Duplicate references |
| Rosenhall 2001b | Duplicate references |
| Rosenhall 2001c | Duplicate references |
| Rosenhall 2002 | Treatment and intervention groups compared the same medications either in combination or with different delivery devices. Abstract. |
| Rosenhall 2003 | Treatment and intervention groups compared the same medications either in combination or with different delivery devices |
| Rosenhall 56 | Not a RCT |
| Rosenthal 1999 | No consistent co‐intervention with ICS. Control intervention not ICS alone but SAB2 on demand. |
| Rumbak 1998 | Study of step‐down ICS treatment |
| Sahn 2002 | Duplicate references |
| SAM30007 | Dose of ICS stepped down after 6 weeks, but if participants were unstable their medication was also changed |
| SAM40004 | Mixed population at baseline |
| Schreurs 1996 | No consistent co‐intervention with ICS. 90% used regular ICS at entry. Control intervention not inhaled glucocorticoids alone (but a different dose of LAB2). |
| Sears 2003 | Not a RCT |
| Serrier 2003 | Treatment and intervention groups compared the same medications either in combination or with different delivery devices. Abstract. Duplicate references. |
| Shapiro 2001 | Intervention is not LAB2 |
| Sheth 2002 | Outcomes measures did not reflect asthma control |
| Shrewsbury 2002 | Duplicate references |
| Sienra‐Monge 2001 | The treatment and intervention groups compared the same medications either in combination or with different delivery devices |
| Simons 1997b | No consistent co‐intervention with inhaled corticosteroids. Treatment groups compared ICS to long‐acting beta2‐agonist alone. |
| Sims 2003 | Duration < 30 days |
| Staehr 1995 | Control intervention not ICS (but SAB2 maintenance) |
| Stahl 2003 | No regular LABA rather prn LABA versus SABA |
| Stallberg 2003 | No group with inhaled glucocorticoids alone |
| Stanford 2002 | Outcomes measures did not reflect asthma control |
| Stankovic 2000 | Not a RCT |
| Stelmach 2001 | Duplicate references. No consistent intervention with inhaled glucocorticoids in all subjects. |
| Stelmach 2002a | No co‐intervention with ICS. Duplicate references. |
| Stelmach 2002b | No co‐intervention with ICS |
| Stelmach 2008 | No prior ICS exposure |
| Stojkovic‐Andjelkovi 2001 | Not a RCT |
| Stoloff 2002 | Not a RCT |
| Tan 1997 | Outcomes measures did not reflect asthma control |
| Tattersfield 1999 | Intervention is not daily LAB2 (but rather on‐demand LAB2) |
| Tattersfield 2001 | Not a RCT |
| Tolley 2002 | Not a RCT |
| Tonelli 2001 | No consistent intervention with inhaled glucocorticoids in all subjects |
| Trautmann 2001 | Not a RCT |
| Turner 1998 | No consistent co‐intervention with ICS alone. Intervention duration < 30 days. |
| Ullman 1990 | Duration < 30 days |
| Van den Berg 2000 | No consistent co‐intervention with LAB2‐both groups received LAB2 but compared delivery devices. Duplicate references. |
| van der Woude 2001 | Duplicate references. The treatment and intervention groups compared the same medications either in combination or with different delivery devices. |
| Van der Woude 2004 | No ICS alone |
| Van Der Woude 2004 | No consistent intervention with ICS alone |
| Van Noord 1999 | Increased dose of ICS in control group |
| van Noord 2001 | Tx and intervention compared ICS + LAB2 in 2 different combination devices |
| Van Schayck 2002 | Duplicate references |
| Vastagh 2003 | No LABA |
| Verberne 1997 | No consistent co‐intervention with ICS ‐ approximately 20% were taking regular ICS at entry |
| Vermetten 1999 | Not a RCT |
| Vestbo 2000 | Patients are not asthmatics (but rather have COPD) |
| Vickers 2000 | The intervention is not LAB2 but placebo. No consistent co‐intervention with ICS. Ongoing study ‐ protocol only published. |
| Vilsvik 2001 | Intervention duration < 30 days |
| Virchow 2002 | Duplicate references |
| Von Berg 1989 | Duration < 30 days |
| Von Berg 2003 | No concurrent ICS |
| Wallaert 1999 | No group with inhaled corticosteroids alone. (Control is another LAB2). |
| Wallin 1990 | No group with inhaled corticosteroids alone. (Control is regular SAB2). No consistent intervention with inhaled glucocorticoids in all subjects. Outcomes measures did not reflect asthma control. |
| Wallin 1998 | Not a RCT |
| Wallin 1999 | No consistent co‐treatment with ICS |
| Weinberger 2004 | No LABA |
| Weinstein 1998 | No consistent co‐intervention with ICS ‐ only 57% were on ICS |
| Weinstein 2001 | Not a RCT |
| Wempe 1992 | No consistent co‐treatment with ICS |
| White 2001 | Duplicate references |
| Wilcke 1998 | Duration < 30 days |
| Wilding 1997 | Not a RCT |
| Wilson 1999 | Duplicate references. |
| Wilson 2000 | Duplicate references |
| Wilson 2001a | Control intervention is not ICS alone (but rather ICS with an anti‐leukotriene agent ‐ montelukast) |
| Wilson 2001b | Not a RCT |
| Wong 1992 | Duration < 30 days |
| Woolcock 1995 | Not a RCT |
| Woolcock 1996 | Increased dose of ICS in control group |
| Yates 1995 | No consistent co‐treatment with ICS. Duration < 30 days. |
| Yates 1996 | Duration < 30 days. Outcomes measures did not reflect asthma control. |
| Youngchaiyud 1995 | Intervention not LAB2 (but theophylline) |
| Yurdakul 2002 | Control not regular long‐acting beta2‐agonists alone. Outcomes measures did not reflect asthma control. |
| Zarkovic 1998 | No consistent co‐intervention with ICS. No group with inhaled corticosteroids alone. (Control is placebo). |
COPD = chronic obstructive pulmonary disease ED = emergency department ICS = inhaled corticosteroids LABA/LAB2 = long‐acting ß2 agonist LTRA = leukotriene receptor antagonist NEJM = New England Journal of Medicine RCT = randomised controlled trial Rx = prescription SABA/SAB2 = short‐acting ß2 agonist Tx = treatment
Characteristics of studies awaiting assessment [ordered by study ID]
Bateman 2001.
| Methods | Parallel‐group, multicentre study |
| Participants | % ELIGIBLE OF SCREENED POPULATION: Not reported % RUN‐IN PARTICIPANTS RANDOMISED: 69 RANDOMISED: 497 (FP/SAL MDI: 165; FP/SAL DPI: 167; FP: 165) WITHDRAWALS: FP/SAL MDI: 20; FP/SAL DPI: 22; FP: 25) AGE mean (range): 40 (11 to 79) SEVERITY: Mild to moderate BASELINE % PREDICTED FEV1: 75 BASELINE DOSE OF ICS: BDP equivalent 4 to 500 mcg/d ASTHMA DURATION: 0 to 1 years: 7% 1 to 5 years: 23% 5 to 10 years: 20 > 10 years: 50 SMOKING STATUS: Current: 11%; ex‐smoker: 21% ATOPY (%): Not reported ELIGIBILITY CRITERIA: aged ?12; documented clinical history of reversible airways obstruction; treatment with BDP equivalent 400 to 500 mcg/day or FP at a dose of 200 to 250 mcg/day for > 4 weeks prior to Visit 1 EXCLUSION CRITERIA: Long‐acting or oral ?2‐agonists within 2 weeks of run‐in; change in asthma medication, taken oral, depot or parenteral corticosteroids or taken a combination therapy (including ?2‐agonist or inhaled corticosteroid); lower respiratory tract infection within 4 weeks of the study. Participants were withdrawn at randomisation visit if they required more salbutamol than maximum in prescribing information sheet during run‐in, failed to withhold short‐acting ?2‐agonists in 6 hours prior to visit or had an FEV1 ? 50% or ? 100% of the predicted normal. ELIGIBILITY CRITERIA DURING RUN‐IN: Mean morning PEFR during the last 7 consecutive days of the run‐in period of > 50% and < 85% of PEFR measured 15 minutes after administration of 400 mcg of salbutamol at randomisation visit; cumulative total symptom score (daytime plus night‐time) in diary of ? 8 for the last 7 consecutive days of the run‐in period |
| Interventions | PROTOCOL: LABA + ICS versus SAME DOSE ICS OUTCOMES: 12 weeks RUN‐IN PERIOD: 2 weeks DOSE OPTIMISATION PERIOD: NA INTERVENTION PERIOD: 12 weeks TEST GROUP: i) Combination fluticasone and salmeterol 100/50 mcg bid via HFA metered dose inhaler (+ placebo Diskus) ii) Combination fluticasone and salmeterol 100/50 mcg bid via Diskus (+ placebo metered dose inhaler) CONTROL GROUP: Fluticasone 100 mcg bid via CFC metered dose inhaler (+ placebo Diskus) NUMBER OF DEVICES: 1 (additional inhaler given as dummy device) COMPLIANCE: Not reported CO‐TREATMENT: prn SABA |
| Outcomes | PULMONARY FUNCTION TEST: am PEF*; pm PEF; pm PEF predicted; FEV1 SYMPTOM SCORES: Symptom‐free days FUNCTIONAL STATUS: Rescue medication use INFLAMMATORY MARKERS: Not reported ADVERSE EFFECTS: Reported WITHDRAWALS: Reported |
| Notes | — |
Yancey 1997.
| Methods | |
| Participants | |
| Interventions | |
| Outcomes | |
| Notes |
BDP = beclomethasone FEV1 = forced expiratory volume in one second FP = fluticasone ICS = inhaled corticosteroids mcg = microgram PEFR = peak expiratory flow rate SAL = salmeterol
Differences between protocol and review
Given the large size of the review other comparisons originally stated in the protocol (published in 1999) are now assessed in separate reviews; this includes comparing the addition of LABA to inhaled corticosteroids to increased doses of inhaled corticosteroids (Ducharme 2010) and tapering doses of inhaled corticosteroids (Gibson 2005). A similar comparison focusing only on steroid‐naive patients is the object of another Cochrane Review (Ni Chroinin 2009a) and a recent review focused on the role of LABA in paediatrics.(Ni Chroinin 2009b) Finally, the question of serious asthma‐related events with the use of long‐acting beta‐agonists has been addressed in a series of linked reviews (Cates 2008a; Cates 2009a; Cates 2009b).
We have incorporated a new method to assess the risk of bias in line with recommendations described in the Cochrane Handbook for Systematic Reviews of Interventions (Cochrane Handbook).
Contributions of authors
Francine Ducharme revised the protocol, supervised the literature search, created the methodology and data extraction forms, reviewed full‐text publications for relevance, participated in the selection of trials, methodology assessment and data extraction, corresponded with authors and/or the pharmaceutical companies to identify other possibly relevant trials, verify methodology and data extraction and request additional information, supervised the analysis, interpretation, and wrote the 2010 version of the review.
Muireann Ni Chroinin reviewed the literature searches from 2002 to 2004, identified and reviewed the full‐text publication of all citations of potential or potentially eligible RCTs, extracted the methodology and data, entered the description of studies and data entry in RevMan and analysed the data. Muireann interpreted results of the meta‐analysis and wrote the 2005 review.
Toby Lasserson assessed titles and abstracts for the update in 2010, assessed studies for inclusion, corresponded with trialists and study sponsors, extracted data, entered data and wrote up results.
Dr Ilana Greenstone conceived the protocol, requested the literature search, identified and reviewed the full‐text publication of all citations of potential or potentially eligible RCTs from 1999 to 2001, drafted the correspondence to authors and/or the pharmaceutical companies to solicit their collaboration in this review and to identify other possibly relevant trials, participated in extraction of the methodology and data, entering the description of studies and data entry in RevMan, interpreted results of the meta‐analysis and approved the review.
Four research assistants participated in some aspects of the review. Helen Magdalinos (May to July 2001), Alya Danish (November 2001 to March 2002), Vincent Masse (June to August 2004) participated in the entry of data, references, characteristics of included and excluded studies, and revision of the table of comparisons. Under the guidance of Nils Chaillet, Marilyse Julien (2009) performed and assisted in the interpretation of the meta‐regression in 2009.
Although not listed as an author, the data for the primary outcome were verified by Chris Cates.
Sources of support
Internal sources
Canadian Cochrane Network, McGill University, Canada.
External sources
Francine Ducharme was supported by a National Scientist Award from the Fonds de la Santé du Québec, Canada.
Declarations of interest
In the past five years, Francine Ducharme received some research funding from GSK and Merck & Co, USA and gave CME conferences supported by Merck Frosst. M Ni Chroinin, IR Greenstone, A Danish, H Magalinos, V Masse, M Julien and T Lasserson report no conflict of interest.
New search for studies and content updated (conclusions changed)
References
References to studies included in this review
Akpinarli 1999 {published data only}
- Akpinarli A, Tuncer A, Saraclar Y, Sekerel BE, Kalayci O. Effect of formoterol on clinical parameters and lung functions in patients with bronchial asthma: a randomised controlled trial. Archives of Disease in Childhood 1999;81:45‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Aubier 1999a {published data only}
- Aubier M, Pieters WR, Schlosser NJ, Steinmetz KO. Salmeterol/fluticasone propionate (50/500 mug) in combination in a Diskus(TM) inhaler (Seretide(TM)) is effective and safe in the treatment of steroid‐dependent asthma. Respiratory Medicine 1999;93(12):876‐84. [DOI] [PubMed] [Google Scholar]
- Pieters W, Ringdal N, Aubier M, Chapman KR, Huskisson SC. A new inhaler combination containing salmeterol and fluticasone propionate is well‐tolerated in long‐term use. European Respiratory Journal. 1998; Vol. 12, issue Suppl 29:P164.
- Pieters WR, Lundback B, Sondhi S, Price MJ. Cost effectiveness analysis of salmeterol/fluticasone propionate 50/500mcg vs fluticasone propionate 500mcg in patients with corticosteroid‐dependent asthma. Pharmacoeconomics 1999;16(Suppl 2):29‐34. [Google Scholar]
- Pieters WR, Sondhi S, Price MJ, Thwaites RM, Nyth A. The cost effectiveness of salmeterol/fluticasone propionate 50/500 microgram combination inhaler versus fluticasone propionate 500 microgram in patients with chronic asthma. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999:P2458.
- Pieters WR, Steinmetz KO, Aubier M, Johnson L, Gomez E, Bogolubov M. Effectiveness of a new salmeterol/fluticasone propionate (50/500µg) combination inhaler in patients with reversible airways obstruction. European Respiratory Journal 1998; Vol. 12, issue Suppl 28:35s.
- Pieters WR, Wilson KK, Smith HCE, Tamminga JJ. Cost‐effectiveness of fluticasone propionate/salmeterol combination product and fluticasone propionate/montelukast in asthma. Annual Thoracic Society 97th International Conference; San Francisco CA , May 18‐23. 2001.
- SFCB30019. A multicentre randomized, double‐blind, double‐dummy, parallel‐group comparison of the salmeterol/fluticasone propionate combination product (50/500mcg strength) BD via one DISKUS/Accuhaler inhaler with salmeterol 50mcg BD via one DISKUS/Accuhaler and fluticasone propionate 500mcg BD via another DISKUS/Accuhaler and with fluticasone propionate 500mcg BD via one DISKUS/Accuhaler in adolescents and adults with reversible airways obstruction. http://www.ctr.gsk.co.uk 2004.
- Schlosser NJ, Steinmetz KO, Aubier M, Gomez E, Wixon C. Evaluation of long‐term safety of salmeterol/fluticasone propionate (50/500µg) combination inhaler in patients with reversible airways obstruction. European Respiratory Journal 1998; Vol. 12, issue Suppl 28:35s.
Aubier 1999b {published data only}
- Aubier M, Pieters WR, Schlosser NJ, Steinmetz KO. Salmeterol/fluticasone propionate (50/500 mug) in combination in a Diskus(TM) inhaler (Seretide(TM)) is effective and safe in the treatment of steroid‐dependent asthma. Respiratory Medicine 1999;93(12):876‐84. [DOI] [PubMed] [Google Scholar]
- Pieters W, Ringdal N, Aubier M, Chapman KR, Huskisson SC. A new inhaler combination containing salmeterol and fluticasone propionate is well‐tolerated in long‐term use. European Respiratory Journal. 1998; Vol. 12, issue Suppl 29:P164.
- Pieters WR, Lundback B, Sondhi S, Price MJ. Cost effectiveness analysis of salmeterol/fluticasone propionate 50/500mcg vs fluticasone propionate 500mcg in patients with corticosteroid‐dependent asthma. Pharmacoeconomics 1999;16(Suppl 2):29‐34. [Google Scholar]
- Pieters WR, Sondhi S, Price MJ, Thwaites RM, Nyth A. The cost effectiveness of salmeterol/fluticasone propionate 50/500 microgram combination inhaler versus fluticasone propionate 500 microgram in patients with chronic asthma. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999:P2458.
- Pieters WR, Steinmetz KO, Aubier M, Johnson L, Gomez E, Bogolubov M. Effectiveness of a new salmeterol/fluticasone propionate (50/500µg) combination inhaler in patients with reversible airways obstruction. European Respiratory Journal 1998; Vol. 12, issue Suppl 28:35s.
- Pieters WR, Wilson KK, Smith HCE, Tamminga JJ. Cost‐effectiveness of fluticasone propionate/salmeterol combination product and fluticasone propionate/montelukast in asthma. Annual Thoracic Society 97th International Conference; San Francisco CA , May 18‐23. 2001.
- SFCB30019. A multicentre randomized, double‐blind, double‐dummy, parallel‐group comparison of the salmeterol/fluticasone propionate combination product (50/500mcg strength) BD via one DISKUS/Accuhaler inhaler with salmeterol 50mcg BD via one DISKUS/Accuhaler and fluticasone propionate 500mcg BD via another DISKUS/Accuhaler and with fluticasone propionate 500mcg BD via one DISKUS/Accuhaler in adolescents and adults with reversible airways obstruction. http://www.ctr.gsk.co.uk 2004.
- Schlosser NJ, Steinmetz KO, Aubier M, Gomez E, Wixon C. Evaluation of long‐term safety of salmeterol/fluticasone propionate (50/500µg) combination inhaler in patients with reversible airways obstruction. European Respiratory Journal 1998; Vol. 12, issue Suppl 28:35s.
Bailey 2008 {unpublished data only}
- Bailey W, Castro M, Matz J, White M, Dransfield M, Yancey S, et al. Asthma exacerbations in African Americans treated for 1 year with combination fluticasone propionate and salmeterol or fluticasone propionate alone. Current Medical Research and Opinion 2008;24(6):1669‐82. [DOI] [PubMed] [Google Scholar]
- Glaxo Smith Kline (SFA103153). A multicenter, randomized, double‐blind, parallel group, 52‐week comparison of asthma control and measures of airway inflammation in subjects of African descent receiving fluticasone propionate/salmeterol 100/50mcg DISKUS™ BID or fluticasone propionate 100mcg DISKUS™ BID alone. http://www.gsk.ctr.co.uk 2007 (accessed 30 April 2008).
Boyd 1995 {published data only}
- Boyd G. Salmeterol xinafoate in asthmatic patients under consideration for maintenance oral corticosteroid therapy. European Respiratory Journal 1995;8:1494‐8. [PubMed] [Google Scholar]
Buhl 2003a {published data only}
- Buhl R, Creemers JPHM, Vondra V, Martelli NA. Improved and maintained asthma control with once‐daily budesonide/formoterol single inhaler in mild‐to‐moderate persistent asthma. European Respiratory Journal. 2001; Vol. 18, issue Suppl 33:21s.
- Buhl R, Creemers JPHM, Vondra V, Martelli NA. Once daily Symbicort (budesonide/eformoterol in a single inhaler) is effective in moderate‐persistent asthma. Thorax 2001;56(Suppl 3):iii62. [Google Scholar]
- Buhl R, Creemers JPHM, Vondra V, Martelli NA. Once‐daily budesonide/formoterol via a single inhaler is effective in mild‐to‐moderate persistent asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:21s.
- Buhl R, Creemers JPHM, Vondra V, Martelli NA, Naya IP, Eksstrom T. Once daily budesonide /formoterol in a single inhaler in adults with moderate persistent asthma. Respiratory Medicine 2003;97(4):323‐30. [DOI] [PubMed] [Google Scholar]
- Buhl R, Zetterstrom O, Mellem H, Perpina M, Hedman J, O'Neill S, et al. Improved asthma control with budesonide/formoterol via a single inhaler compared with budesonide alone, in moderate persistent asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:48s. [DOI] [PubMed]
- Buhl R, Creemers JPHM, Vondra V, Martelli NA. Symbicort® (budesonide and formoterol in a single inhaler) administered once daily is effective in mild to moderate asthma. Annual Thoracic Society 97th International Conference; San Francisco CA , May 18‐23. 2001.
- SD 039 0666. Symbicort high dose once daily in mild to moderate asthmatic patients. http://www.astrazenecaclinicaltrials.com 2005.
Buhl 2003b {published data only}
- Buhl R, Creemers JPHM, Vondra V, Martelli NA. Improved and maintained asthma control with once‐daily budesonide/formoterol single inhaler in mild‐to‐moderate persistent asthma. European Respiratory Journal. 2001; Vol. 18, issue Suppl 33:21s.
- Buhl R, Creemers JPHM, Vondra V, Martelli NA. Once daily Symbicort (budesonide/eformoterol in a single inhaler) is effective in moderate‐persistent asthma. Thorax 2001;56(Suppl 3):iii 62. [Google Scholar]
- Buhl R, Creemers JPHM, Vondra V, Martelli NA. Once‐daily budesonide/formoterol via a single inhaler is effective in mild‐to‐moderate persistent asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:21s.
- Buhl R, Creemers JPHM, Vondra V, Martelli NA, Naya IP, Eksstrom T. Once daily budesonide /formoterol in a single inhaler in adults with moderate persistent asthma. Respiratory Medicine 2003;97:323‐30. [DOI] [PubMed] [Google Scholar]
- Buhl R, Zetterstrom O, Mellem H, Perpina M, Hedman J, O'Neill S, et al. Improved asthma control with budesonide/formoterol via a single inhaler compared with budesonide alone, in moderate persistent asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:48s. [DOI] [PubMed]
- Buhl R, Creemers JPHM, Vondra V, Martelli NA. Symbicort® (budesonide and formoterol in a single inhaler) administered once daily is effective in mild to moderate asthma. Annual Thoracic Society 97th International Conference; San Francisco CA , May 18‐23. 2001.
- SD 039 0666. Symbicort high dose once daily in mild to moderate asthmatic patients. http://www.astrazenecaclinicaltrials.com 2005.
D'Urzo 2001 {published data only}
- D'Urzo AD, Chapman KR, Cartier A, Hargreave FE, Fitzgeerald M, Tesarowski D. Effectiveness and safety of salmeterol in non‐specialist practice settings. Chest 2001;119:714‐9. [DOI] [PubMed] [Google Scholar]
- SLGQ94 (521/180) (GSK). A multicenter, randomized, double‐blind, parallel‐group trial to evaluate the long‐term efficacy and safety of inhaled salmeterol 50μg BID compared to short‐acting β2‐agonists as‐needed in adult patients with asthma. http://ctr.gsk.co.uk 2007 (accessed 20 June 2008).
D5896C0001a {unpublished data only}
- AstraZeneca (D5896C00001). A randomized, double‐blind, active‐controlled, parallel‐group, single‐dummy, multicenter, 12 week study to assess the efficacy and safety of SYMBICORT® pMDI 160/4.5 μg x 2 actuations once‐daily (qd) compared to SYMBICORT pMDI 80/4.5 μg x 2 actuations qd, SYMBICORT pMDI80/4.5 μg x 2 actuations twice‐daily (bid) and to budesonide pMDI 160 μgx 2 actuations qd in asthmatic subjects 12 years of age and older. http://www.astrazenecaclinicaltrials.com 2005 (accessed 30 April 2008).
D5896C0001b {unpublished data only}
- AstraZeneca (D5896C00001). A randomized, double‐blind, active‐controlled, parallel‐group, single‐dummy, multicenter, 12 week study to assess the efficacy and safety of SYMBICORT® pMDI 160/4.5 μg x 2 actuations once‐daily (qd) compared to SYMBICORT pMDI 80/4.5 μg x 2 actuations qd, SYMBICORT pMDI80/4.5 μg x 2 actuations twice‐daily (bid) and to budesonide pMDI 160 μgx 2 actuations qd in asthmatic subjects 12 years of age and older. http://www.astrazenecaclinicaltrials.com 2005 (accessed 30 April 2008).
Fitzgerald 1999 {published data only}
- FitzGerald JM, Chapman KR, Della Cioppa G, Stubbing D, Fairbarn MS, Till MD, et al. Sustained bronchoprotection, bronchodilatation, and symptom control during regular formoterol use in asthma of moderate or greater severity. The Canadian FO/OD1 Study Group. Journal of Allergy & Clinical Immunology 1999;103(3 pt 1):427‐35. [DOI] [PubMed] [Google Scholar]
Gardiner 1994 {published data only}
- Gardiner PV, Ward C, Booth H, Allison A, Hendrick DJ, Walters EH. Bronchoalveolar lavage inflammatory indices in asthmatics. American Journal of Respiratory & Critical Care Medicine 1994;150(4):1006‐11. [DOI] [PubMed] [Google Scholar]
GOAL {published and unpublished data}
- Anonymous. GSK asthma trial suggests total control is possible. Pharmaceutical Journal 2004;273(7322):594. [Google Scholar]
- Arthurs R. Gaining optimal asthma control. Practice Nurse 2004;Suppl:3‐8. [Google Scholar]
- Bateman E, Boushey H, Bousquet J, Busse W, Clark T, Pauwels R, et al. Achievement for guideline based asthma control with salmeterol/fluticasone propionate compared with fluticasone propionate alone; results of Goal study. Triennial World Asthma Meeting, Thailand (16‐19 February). 2004.
- Bateman E, Boushey H, Bousquet J, Busse W, Clark T, Pauwels R, et al. Achieving and maintaining guideline defined asthma control with salmeterol/fluticasone propionate versus fluticasone propionate alone: the results of the GOAL study. American Journal of Respiratory & Critical Care Medicine 2004;169(7):A87. [DOI] [PubMed] [Google Scholar]
- Bateman E, Pauwels R, Boushey H, Bousquet J, Busse W, Clark T, et al. Aiming for total control of asthma significantly improves asthma‐related quality of life: salmeterol/fluticasone propionate versus fluticasone propionate alone. American Journal of Respiratory & Critical Care Medicine 2004;169(7):A87. [Google Scholar]
- Bateman ED, Boushey HA, Bousquet J, Busse WW, Clark TJ, Pauwels RA, et al. GOAL Investigators Group. Can guideline‐defined asthma control be achieved? The Gaining Optimal Asthma ControL study. American Journal of Respiratory & Critical Care Medicine 2004;170(8):836‐44. [DOI] [PubMed] [Google Scholar]
- Bateman ED, Boushey HA, Bousquet J, Busse WW, Clark TJ, Pauwels RA, et al. GOAL Investigators Group. Can guideline‐defined asthma control be achieved? The Gaining Optimal Asthma ControL study. American Respiratory & Critical Care Medicine. 2004:Online data supplement. [DOI] [PubMed]
- Bateman ED, Bousquet J, Keech ML, Busse WW, Clark TJ, Pedersen SE. The correlation between asthma control and health status: the GOAL study. European Resspiratory Journal 2006:Epub ahead of print. [DOI] [PubMed]
- Bateman ED, Edin HM, Sondhi S, Gul N. Asthma‐related quality of life in the GOAL study: baseline results. European Respiratory Journal 2002;20(Suppl 38):46s. [Google Scholar]
- Bons J, Cordier JF, Godard P, Prud’Homme A, Celli I, Bousquet J. Aiming for total control of asthma: the GOAL study design. Revue Française d’Allergologie et d’Immunologie Clinique 2004;44(367):TT13. [Google Scholar]
- Boushey H, Bateman E, Bousquet J, Busse W, Clark T, Pauwels R, et al. Achieving total control of asthma with salmeterol/fluticasone propionate versus fluticasone propionate alone: Goal study results. Triennial World Asthma Meeting, Thailand (16‐19 February). 2004.
- Boushey H, Bateman E, Bousquet J, Busse W, Clark T, Pauwels R, et al. Improvements in asthma outcomes following 1 year of treatment with salmeterol/fluticasone or fluticasone alone when stepped up to achieve guideline‐defined total control. Journal Allergy and Clinical Immunology 2004;113(2 (Suppl 1)):114s‐5s. [Google Scholar]
- Boushey HA, Pedersen S, Bateman E, Clark T, Busse W, Bousquet J, et al. Improved exacerbation rates and asthma control in current and former smokers treated with salmeterol/fluticasone propionate: results of the GOAL study. Journal of Allergy & Clinical Immunology 2005;115(Suppl 2):S59. [Google Scholar]
- Bousquet J. Is asthma control achievable?. European Respiratory Review 2004;13(92):102‐4. [Google Scholar]
- Bousquet J, Bateman E, Boushey H, Busse W, Clark T, Pauwels R, et al. The effect of oral corticosteroids and high‐dose combination therapy on achieving control of refractory asthma. Journal Allergy and Clinical Immunology 2004;113(2 (Suppl 1)):113s. [Google Scholar]
- Bousquet J, Bons J, Godard P, Cordier JF, Desfougeres JL, Prud’Homme A. Aiming for total control of asthma: the GOAL study results. Revue Française d’Allergologie et d’Immunologie Clinique 2004;44(367):TT16. [Google Scholar]
- Briggs AH, Bousquet J, Wallace MV, Busse WW, Clark TJH, Pedersen SE, et al. Cost‐effectiveness of asthma control: an economic appraisal of the GOAL study. Allergy 2006;61(5):531‐6. [DOI] [PubMed] [Google Scholar]
- Busse W, Bateman E, Boushey H, Bousquet J, Clark T, Pauwels R, et al. Achieving GINA/NIH guideline‐based asthma control with salmeterol/fluticasone compared with fluticasone alone: the results of the GOAL study. Journal of Allergy and Clinical Immunology 2004;113(2 (Suppl 1)):114s. [Google Scholar]
- Busse W, Bateman E, Boushey H, Bousquet J, Clark T, Pauwels R, et al. Aiming to achieve total control of asthma with salmeterol/fluticasone propionate and fluticasone propionate alone is well tolerated: Goal 1 year safety data. Triennial World Asthma Meeting, Thailand (16‐19 February). 2004.
- Clark T, Bateman E, Boushey H, Bousquet J, Busse W, Pauwels R, Pedersen S. Salmeterol/fluticasone and fluticasone alone are well tolerated over 1 year of treatment stepped‐up to achieve total control: safety results of the GOAL study. Journal Allergy and Clinical Immunology 2004;113(2 ( Suppl 1)):115s. [Google Scholar]
- Clark T, Bateman E, Boushey H, Bousquet J, Busse W, Pauwels R, et al. Time course of achievement of individual clinical goals of asthma treatment: the results of the GOAL study. American Journal of Respiratory & Critical Care Medicine 2004;169(7):A318. [Google Scholar]
- Clark TJ, Bousquet J, Bateman ED, James MH. GOAL (gaining optimal asthma control): a study to assess asthma control. European Respiratory Journal 2001;18(Suppl 33):175‐6s. [Google Scholar]
- Clark TJH, Bateman ED on behalf of the GOAL Steering Committee. Aiming for Total Control of asthma in ICS‐free patients improves traditional outcomes: results of the Gaining Optimal Asthma controL (GOAL) study [abstract]. 23rd European Academy of Allergology and Clinical Immunology Meeting Abstract Book. June 12‐16. Amsterdam, The Netherlands. 2004; Vol. 204:669.
- Clark TJH, Bateman ED, James MH, on behalf of the GOAL Steering Committee. Assessing asthma control using a composite measure based on GINA/NIH guidelines: an analysis of GOAL baseline data. European Respiratory Journal 2002;20(Suppl 38):47s. [Google Scholar]
- Cordier JF, Bousquet J, Boucot I, Prud’Homme A, Godard P. Which dose for achieving total control of asthma? The results of the GOAL study. Revue Française d’Allergologie et d’Immunologie Clinique 2004;44(367):TT14. [Google Scholar]
- Godard P, Prud’Homme A, Cordier JF, Sohier B, Bousquet J. Time course of achievement of asthma control: the results of the GOAL study. Revue Française d’Allergologie et d’Immunologie Clinique 2004;44(367):TT15. [Google Scholar]
- Juniper EF, Bateman ED, Sondhi S, Gul N. Asthma Control Questionnaire (ACQ) differentiates between levels of clinical control in a large scale trial. American Journal of Respiratory & Critical Care Medicine 2003;167(7):A37. [Google Scholar]
- Pauwels R, Bateman E, Boushey H, Bousquet J, Busse W, Clark T, et al. Addition of oral corticosteroids to combination therapy has little impact on achieving total control of asthma. 4th Triennial World Asthma Meeting Abstract Book, Bangkok, Thailand, February 16‐19. 2004:135.
- Pauwels R, Bateman E, Boushey H, Bousquet J, Busse W, Clark T, et al. Aiming for total control of asthma reduces the risk of exacerbations: a comparison of salmeterol/fluticasone propionate versus fluticasone propionate alone. American Journal of Respiratory and Critical Care Medicine 2004;169(7):A87. [Google Scholar]
- Pauwels R, Bateman E, Boushey H, Bousquet J, Busse W, Clark T, et al. Can total control of asthma be achieved?: the results of the GOAL study. Journal of Allergy and Clinical Immunology 2004;113(2 (Suppl 1)):114s. [Google Scholar]
- Pedersen SE. Is guideline‐defined asthma control achievable? The Gaining Optimal Asthma ControL (GOAL) Study [Er guideline‐defineret astmakontrol opnaelig?]. Ugeskrift for Laeger 2005;167(38):3595‐7. [PubMed] [Google Scholar]
- Pedersen SE, Bateman ED on behalf of the GOAL Steering Committee. Aiming for Total Control of asthma in patients taking inhaled corticosteroids improves traditional outcomes: Results of the Gaining Optimal Asthma controL (GOAL) study. [abstract]. 3rd European Academy of Allergology and Clinical Immunology Meeting Abstract Book. June 12‐16. Amsterdam, The Netherlands. 2004; Vol. 204:670.
- Pederson S, Bateman E, Boushey H, Bousquet J, Busse W, Clark T, et al. Aiming for guideline defined total control of asthma improves one‐year asthma outcomes: results of Goal study. Triennial World Asthma Meeting, Thailand (16‐19 February). 2004.
- SAM40027. Gaining Optimal Asthma ControL (GOAL): A multi‐centre, stratified, randomised, double‐blind, parallel‐group, step‐up comparison of the level of asthma control achieved with salmeterol/fluticasone propionate combination DISKUS (ACCUHALER) dry powder inhaler compared with fluticasone propionate DISKUS (ACCUHALER) alone in adults and adolescents. http://www.ctr.gsk.co.uk 2004.
Green 2006 {published data only}
- Green RH, Brightling CE, McKenna S, Hargadon B, Neale N, Parker D, et al. Comparison of asthma treatment given in addition to inhaled corticosteroids on airway inflammation and responsiveness. European Respiratory Journal 2006;27(6):1144‐51. [DOI] [PubMed] [Google Scholar]
- Green RH, Brightling CE, McKenna S, Hargadon B, Parker D, Pavord ID. A placebo controlled comparison of formoterol, montelukast or higher dose of inhaled budesonide in subjects with symptomatic asthma despite treatment with low dose inhaled budesonide. American Thoracic Society 99th International Conference. 2003:B036 Poster H82.
Houghton 2007 {published data only}
- Houghton CM, Lawson N, Borrill ZL, Wixon CL, Yoxall S, Langley SJ, et al. Comparison of the effects of salmeterol/fluticasone propionate with fluticasone propionate on airway physiology in adults with mild persistent asthma. Respiratory Research 2007;8(1):52. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hultquist 2000 {unpublished data only}
- AstraZeneca (SD 004 0216). Oxis turbuhaler® (formoterol), Accolate® (zafirlukast) or placebo as add on treatment to Pulmicort Turbuhaler® (budesonide) in asthmatic patients on inhaled steroids. http://www.astrazenecaclinicaltrials.com 2000 (accessed 30 April 2008).
- Hultquist C. Unpublished data. Personal communication.
Ind 2003 {published data only}
- Ind P, Haughney J, Price D, Rosen J‐P, Kennelly J. Four months adjustable or fixed BD dosing with budesonide /Formoterol in a single inhaler reduces symptom severity. Thorax 2002; Vol. 57, issue Suppl 3:iii88.
- Ind PW, Dal Negro R, Colman N, Fletcher CP, Browning DC, James MH. Inhaled fluticasone propionate and salmeterol in moderate adult asthma I: lung function and symptoms. American Journal of Respiratory and Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A416.
- Ind PW, Dal Negro R, Colman N, Fletcher CP, Browning DC, James MH. Inhaled fluticasone propionate and salmeterol in moderate adult asthma II: exacerbations. American Journal of Respiratory and Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A415.
- Ind PW, Dal Negro R, Colman NC, Fletcher CP, Browning D, James MH. Addition of salmeterol to fluticasone propionate treatment in moderate to severe asthma. Respiratory Medicine 2003;97(5):555‐62. [DOI] [PubMed] [Google Scholar]
- SLGQ97. A multi‐centre double‐blind, parallel group study to evaluate the relative clinical benefits of three treatment interventions: i) salmeterol xinafoate 50 mcg bd plus fluticasone propionate 250 mcg bd; ii) fluticasone propionate 500 mcg bd; iii) fluticasone propionate 250 mcg bd, in adult asthmatic subjects poorly controlled on current inhaled corticosteroids.. http://ctr.gsk.co.uk 2005.
Jenkins 2006a {published and unpublished data}
- Jenkins C, Kolarikova R, Kuna P, Caillaud D, Sanchis J, Popp W, et al. Efficacy and safety of high‐dose budesonide/formoterol (Symbicort(R)) compared with budesonide administered either concomitantly with formoterol or alone in patients with persistent symptomatic asthma. Respirology 2006;11(3):276‐86. [DOI] [PubMed] [Google Scholar]
- SD 039 0689. Efficacy and safety of Symbicort® (budesonide/ formoterol) 1280/36 mcg daily delivered dose compared to Pulmicort® (budesonide) 1600 mcg metered dose and Pulmicort (budesonide) 1600 mcg metered dose plus Oxis® (formoterol) 36 mcg delivered dose all delivered via Turbuhaler® in steroid‐using asthmatic adolescents and adults. A double‐blind, double‐dummy, randomized, parallel group, phase III, multicentre study. http://www.astrazeneca.com 2005.
Jenkins 2006b {published data only}
- Jenkins C, Kolarikova R, Kuna P, Caillaud D, Sanchis J, Popp W, et al. Efficacy and safety of high‐dose budesonide/formoterol (Symbicort(R)) compared with budesonide administered either concomitantly with formoterol or alone in patients with persistent symptomatic asthma. Respirology 2006;11(3):276‐86. [DOI] [PubMed] [Google Scholar]
- SD 039 0689. Efficacy and safety of Symbicort® (budesonide/ formoterol) 1280/36 mcg daily delivered dose compared to Pulmicort® (budesonide) 1600 mcg metered dose and Pulmicort (budesonide) 1600 mcg metered dose plus Oxis® (formoterol) 36 mcg delivered dose all delivered via Turbuhaler® in steroid‐using asthmatic adolescents and adults. A double‐blind, double‐dummy, randomized, parallel group, phase III, multicentre study. http://www.astrazeneca.com 2005.
Kavaru 2000 {published and unpublished data}
- Edin HM, Payne E, Herrle MR, Schoaf L, Mather DB, Scott CA, et al. Salmeterol/fluticasone propionate combination via HFA MDI improves quality of life in asthma patients. Journal of Allergy and Clinical Immunology 2001; Vol. 107, issue 2:S246.
- Edin HM, Prillaman B, Baitinger LA, House K, Shah TP. Improved ability to perform strenuous activities after treatment with fluticasone propionate‐salmeterol combination. American Journal of Respiratory & Critical Care Medicine 2002; Vol. 165, issue Suppl 8:A112.
- Edwards T, Gross G, Mitchell D, Chervinsky P, Woodring A, Baitinger L, et al. The salmeterol xinafoate/fluticasone propionate dry powder combination product via diskus® inhaler improves asthma control compared to salmeterol xinafoate or fluticasone propionate dry powder alone. American Journal of Respiratory and Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A414.
- Johansson G, Price MJ, Sondhi S. Cost‐effectiveness analysis of salmeterol/fluticasone propionate 50/100mug vs fluticasone propionate 100mug in adults and adolescents with asthma III. Pharmacoeconomics 1999;16(Suppl 2):15‐21. [Google Scholar]
- Kavuru M, Melamed J, Gross G, Laforce C, House K, Prillaman B, et al. Salmeterol and fluticasone propionate combined in a new powder inhalation device for the treatment of asthma: a randomised, double blind,placebo controlled trial. Journal of Allergy and Clinical Immunology 2000;105(6):1108‐16. [DOI] [PubMed] [Google Scholar]
- Nathan RA, Dorinsky P, Rosenzweig JR, Shah T, Edin H, Prillaman B. Improved ability to perform strenuous activities after treatment with fluticasone propionate/salmeterol combination in patients with persistent asthma. Journal of Asthma 2003;40(7):815‐22. [DOI] [PubMed] [Google Scholar]
- SFCA3002. A randomized, double‐blind, parallel‐group trial evaluating safety and efficacy of salmeterol 50mcg BID and fluticasone propionate 100mcg BID individually and in combination and placebo in subjects with asthma. http://www.clinicalstudyresults.org 2005.
Kemp 1998 {published data only}
- Kemp JP, Cook DA, Incaudo GA, Corren J, Kalberg C, Emmett A, et al. Salmeterol improves quality of life in patients with asthma requiring inhaled corticosteroids. Journal of Allergy & Clinical Immunology 1998;101:188‐95. [DOI] [PubMed] [Google Scholar]
Koopmans 2006 {published data only}
- Koopmans JG, Lutter R, Jansen HM, Zee JS. Adding salmeterol to an inhaled corticosteroid: long term effects on bronchial inflammation in asthma. Thorax 2006;61(4):306‐12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SAS30013. A study to compare the long term effects on airway inflammation of Seretide versus Flixotide in adult subjects with asthma. http://www.ctr.gsk.co.uk 2004.
Kuna 2006 {published data only}
- AstraZeneca (SD 039 0665). Symbicort low dose once daily in mild to moderate asthmatic patients. http://www.astrazenecaclinicaltrials.com 2000 (accessed 30 April 2008).
- Kuna P, Creemers JPHM, Vondra V, Black PN, Lindqvist A, Nihlen U, et al. Once‐daily dosing with budesonide/formoterol compared with twice‐daily budesonide/formoterol and once‐daily budesonide in adults with mild to moderate asthma. Respiratory Medicine 2006;100(12):2151‐9. [DOI] [PubMed] [Google Scholar]
Langton Hewer 1995 {published data only}
- Langton Hewer S, Hobbs J, French D, Lenney W. Pilgrims progress: the effect of salmeterol in older children with chronic severe asthma. Respiratory Medicine 1995;89:435‐40. [DOI] [PubMed] [Google Scholar]
Leblanc 1996 {published data only}
- Leblanc P, Knight A, Kreisman H, Borkhoff CM, Johnston PR. A placebo‐controlled, crossover comparison of salmeterol and salbutamol in patients with asthma. American Journal of Respiratory & Critical Care Medicine 1996;154:324‐8. [DOI] [PubMed] [Google Scholar]
Li 1999 {published data only}
- Li X, Ward C, Thien F, Bish R, Bamford T, Bao X, et al. An antiinflammatory effect of salmeterol, a long‐acting B2‐agonist, assessed in airway biopsies and bronchoalveolar lavage in asthma. American Journal of Respiratory & Critical Care Medicine 1999;160:1493‐9. [DOI] [PubMed] [Google Scholar]
- Reid DW, Ward C, Wang N, Zheng L, Bish R, Orsida B, et al. Possible anti‐inflammatory effect of salmeterol against interleukin‐8 and neutrophil activation in asthma in vivo. European Respiratory Journal 2003;21(6):994‐9. [DOI] [PubMed] [Google Scholar]
Malone 2005 {published and unpublished data}
- Dorinsky P, Emmett A, Sutton L. Reduced risk for asthma exacerbations in pediatric patients receiving salmeterol plus inhaled corticosteroids (ICS) vs ICS alone [Abstract]. European Respiratory Journal 2004;24(Suppl 48):308s. [Google Scholar]
- House K, Dorinsky PM, Stauffer J, Schoaf L, Ellsworth A. The safety of fluticasone propionate/salmeterol Diskus(R) in pediatric patients ages 4 ‐11 with asthma [Abstract]. Chest 2004;126(Suppl 4):911S. [Google Scholar]
- Malone R, LaForce C, Nimmagadda S, Schoaf L, House K, Ellsworth A, et al. The safety of twice‐daily treatment with fluticasone propionate and salmeterol in pediatric patients with persistent asthma. Annals of Allergy, Asthma, and Immunology 2005;95(1):66‐71. [DOI] [PubMed] [Google Scholar]
- SAS30031(GSK). A randomized, double‐blind, 12‐week trial evaluating the safety of the fluticasone propionate/salmeterol DISKUS combination product 100/50mcg BID versus fluticasone propionate DISKUS 100mcg BID in symptomatic pediatric subjects (4‐11 years) with asthma. http://www.ctr.gsk.co.uk 2005 (accessed 30 April 2008).
- Scott C, Wu W, Ellsworth A, Crim C. Efficacy and safety of fluticasone propionate/salmeterol DISKUS and fluticasone propionate DISKUS and HFA in children [Abstract]. European Respiratory Journal 2005;26(Suppl 49):Abstract No. 1057. [Google Scholar]
Meijer 1995 {published data only}
- Meijer GG, Postma DS, Mulder PG, Aalderen WM. Long‐term circadian effects of salmeterol in asthmatic children treated with inhaled corticosteroids. American Journal of Respiratory & Critical Care Medicine 1995;152:1887‐92. [DOI] [PubMed] [Google Scholar]
Molimard 2001 {published data only}
- Molimard M, Bourcereau J, Gros V, Bourdeix I, Leynadier F, Duroux P. Comparison between formoterol 12 ug bid and on‐demand salbutamol in moderate persistent asthma. Respiratory Medicine 2001;95(1):64‐70. [DOI] [PubMed] [Google Scholar]
Morice 2008a {published and unpublished data}
- AstraZeneca. A 12‐week randomised, double‐blind, parallel‐group, multicentre phase‐III study to compare the efficacy and safety of Symbicort® pMDI (budesonide/formoterol 80/4.5 mcg 2 actuations b.i.d., delivered dose) with that of Pulmicort® pMDI (budesonide 100 mcg 2 actuations b.i.d., metered dose) and Symbicort Turbuhaler® (budesonide/formoterol 80/4.5 mcg 2 actuations b.i.d., delivered dose) in children with asthma. http://www.astrazenecaclinicaltrials.com 2007 (accessed 4 January 2008).
- Morice AH, Peterson S, Beckman O, Kukova Z. Efficacy and safety of a new pressurised metered‐dose inhaler formulation of budesonide/formoterol in children with asthma: A superiority and therapeutic equivalence study. Pulmonology Pharmacology Therapeutics 2008;21(1):152‐9. [DOI] [PubMed] [Google Scholar]
Morice 2008b {published data only}
- AstraZeneca. A 12‐week randomised, double‐blind, parallel‐group, multicentre phase‐III study to compare the efficacy and safety of Symbicort®pMDI (budesonide/formoterol 80/4.5 mcg 2 actuations b.i.d., delivered dose) with that of Pulmicort®pMDI (budesonide 100 mcg 2 actuations b.i.d., metered dose) and Symbicort Turbuhaler® (budesonide/formoterol 80/4.5 mcg 2 actuations b.i.d., delivered dose) in children with asthma. http://www.astrazenecaclinicaltrials.com 2007 (accessed 4 January 2008).
- Morice AH, Peterson S, Beckman O, Kukova Z. Efficacy and safety of a new pressurised metered‐dose inhaler formulation of budesonide/formoterol in children with asthma: a superiority and therapeutic equivalence study. Pulmonology Pharmacology Therapeutics 2008; Vol. 21, issue 1:152‐9. [DOI] [PubMed]
Nathan 2006 {published and unpublished data}
- Edin HM, Payne E, Herrle MR, Schoaf L, Mather DB, Scott CA, et al. Salmeterol/fluticasone propionate combination via HFA MDI improves quality of life. Journal of Allergy & Clinical Immunology 2001;107(2):S246. [Google Scholar]
- Nathan RA, Mitchell D, Condemi J, Heller A, Schoaf L, Herrle M, et al. Cardiovascular and hypothalamic‐pituitary‐adrenal axis safety of fluticasone propionate/salmeterol HFA MDI in adolescent and adult patients with asthma. American Journal for Respiratory & Critical Care Medicine 2001;163(5):A863. [Google Scholar]
- Nathan RA, Rooklin A, Schoaf L, Scott C, Ellsworth A, House K, et al. Efficacy and tolerability of fluticasone propionate/salmeterol administered twice daily via hydrofluoroalkane 134a metered‐dose inhaler in adolescent and adult patients with persistent asthma: a randomized, double‐blind, placebo‐controlled, 12‐week study. Clinical Therapeutics 2006;28(1):73‐85. [DOI] [PubMed] [Google Scholar]
- Pearlman DS, Kent E, Lanz MJ, Peden D, Baitinger L, Herrle M, et al. Fluticasone propionate/salmeterol HFA MDI has a rapid onset of effect in asthmatics treated with short or long‐acting beta‐agonists (BA) or inhaled corticosteroids (ICS). American Journal of Respiratory & Critical Care Medicine 2001;163(5):A865. [Google Scholar]
- Rooklin A, Elkayam D, Weiler J, Windom H, Schoaf L, Scott C, et al. The fluticasone propionate/salmeterol HFA MDI is significantly more efficacious in treating asthma than placebo HFA MDI, fluticasone propionate CFC MDI or salmeterol CFC MDI. Journal of Allergy and Clinical Immunology 2001;107(2):100s. [Google Scholar]
- SAS30004. A randomized, double‐blind, placebo‐controlled, parallel‐group 12‐week trial evaluating the safety and efficacy of the salmeterol/fluticasone propionate combination in GR106642X MDI, 50/250mcg BID, and salmeterol in propellant 11/12 MDI, 50mcg BID, fluticasone propionate in propellant 11/12 MDI, 250mcg BID, and placebo propellant GR106642X MDI in adult and adolescent subjects with asthma. http://ctr.gsk.co.uk 2005.
Noonan 2006a {published data only}
- Noonan M, Rosenwasser LJ, Martin P, O'Brien CD, O'Dowd L. Efficacy and safety of budesonide and formoterol in one pressurised metered‐dose inhaler in adults and adolescents with moderate to severe asthma: a randomised clinical trial. Drugs 2006;66(17):2235‐54. [DOI] [PubMed] [Google Scholar]
- SD 039 0717 (AstraZeneca). A twelve‐week, randomized, double‐blind, double‐dummy, placebo‐controlled trial of Symbicort® (160/4.5 μg) versus its mono‐products (budesonide and formoterol) in adolescents (≥12 years of age) and adults with asthma. http://www.astrazenecaclinicaltrials.com 2004 (accessed 13 June 2008).
Noonan 2006b {published data only}
- Noonan M, Rosenwasser LJ, Martin P, O'Brien CD, O'Dowd L. Efficacy and safety of budesonide and formoterol in one pressurised metered‐dose inhaler in adults and adolescents with moderate to severe asthma: a randomised clinical trial. Drugs 2006;66(17):2235‐54. [DOI] [PubMed] [Google Scholar]
- SD 039 0717 (AstraZeneca). A twelve‐week, randomized, double‐blind, double‐dummy, placebo‐controlled trial of Symbicort® (160/4.5 μg) versus its mono‐products (budesonide and formoterol) in adolescents (≥12 years of age) and adults with asthma. http://www.astrazenecaclinicaltrials.com 2004 (accessed 13 June 2008).
Norhaya 1999 {published data only}
- Norhaya MR, Yap TM, Zainudin BMZ. Addition of inhaled salmeterol to inhaled corticosteroids in patients with poorly controlled nocturnal asthma. Repirology 1999;4(1):77‐81. [DOI] [PubMed] [Google Scholar]
O'Byrne 2001a {published data only}
- Barnes PJ, O'Byrne PM, Rodriguez‐Roisin R, Runnerstrom E, Sandstrom T, Svensson K, et al. Treatment of mild persistent asthma with low doses of inhaled budesonide alone or in combination with formoterol. For the Oxis and Pulmicort Turbuhaler in the Management of Asthma (OPTIMA) international study group. Thorax 2000; Vol. 55, issue Suppl 3:s5.
- Barnes PJ, O'Byrne PM, Rodriguez‐Roisin R, Runnerstrom E, Sandstrom T, Svensson K, et al. Oxis and Pulmicort turbuhaler in the management of asthma OPTIMA international study group. Treatment of mild persistent asthma with low doses of inhaled budesonide alone or in combination with formoterol. Thorax 2000;55(Suppl 3):A4. [Google Scholar]
- Jönsson BG, Berggren FE, Svensson K, O'Byrne PM. Budesonide and formoterol in mild persistent asthma compared with doubling the dose of budesonide ‐ a cost‐effectiveness analysis. European Respiratory Journal. 2001; Vol. 18, issue Suppl 33:517s.
- Jönsson BG, Berggren FE, Svensson K, O'Byrne PM. Economic results of adding formoterol to budesonide in mild persistent asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:331s.
- O'Byrne PM, Barnes PJ, Rodriguez‐Roisin R, Runnerstrom E, Sandstrom T, Svensson K, et al. Low dose inhaled budesonide and formoterol in mild persistent asthma: The OPTIMA randomized trial. American Journal of Respiratory & Critical Care Medicine 2001;164(8 pt 1):1392‐7. [DOI] [PubMed] [Google Scholar]
- O'Byrne PM, Barnes PJ, Rodriguez‐Roisin R, Sandtröm T, Tattersfield AE, Runnerström EM, et al. Addition of formoterol Turbuhaler® to budesonide Tubuhaler® is safe and well tolerated in the long‐term treatment of mild asthma: results from the OPTIMA trial. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:330s.
O'Byrne 2001b {published data only}
- Barnes PJ, O'Byrne PM, Rodriguez‐Roisin R, Runnerstrom E, Sandstrom T, Svensson K, et al. Oxis and Pulmicort turbuhaler in the management of asthma OPTIMA international study group. Treatment of mild persistent asthma with low doses of inhaled budesonide alone or in combination with formoterol. Thorax 2000;55(Suppl 3):A4. [Google Scholar]
- Barnes PJ, O'Byrne PM, Rodriguez‐Roisin R, Runnerstrom E, Sandstrom T, Svensson K, et al. Treatment of mild persistent asthma with low doses of inhaled budesonide alone or in combination with formoterol. For the Oxis and Pulmicort Turbuhaler in the Management of Asthma (OPTIMA) international study group. Thorax 2000; Vol. 55, issue Suppl 3:s5.
- Jönsson BG, Berggren FE, Svensson K, O'Byrne PM. Budesonide and formoterol in mild persistent asthma compared with doubling the dose of budesonide ‐ a cost‐effectiveness analysis. European Respiratory Journal. 2001; Vol. 18, issue Suppl 33:517s.
- Jönsson BG, Berggren FE, Svensson K, O'Byrne PM. Economic results of adding formoterol to budesonide in mild persistent asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:331s.
- O'Byrne PM, Barnes PJ, Rodriguez‐Roisin R, Runnerstrom E, Sandstrom T, Svensson K, et al. Low dose inhaled budesonide and formoterol in mild persistent asthma: the OPTIMA randomized trial. American Journal of Respiratory & Critical Care Medicine 2001;164(8 pt 1):1392‐7. [DOI] [PubMed] [Google Scholar]
- O'Byrne PM, Barnes PJ, Rodriguez‐Roisin R, Sandtröm T, Tattersfield AE, Runnerström EM, et al. Addition of formoterol Turbuhaler® to budesonide Tubuhaler® is safe and well tolerated in the long‐term treatment of mild asthma: results from the OPTIMA trial. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:330s.
Pauwels 1997a {published data only}
- Pauwels R. Additive effects of inhaled formoterol and budesonide in reducing asthma exacerbations. Allergy 1998;53:20‐3. [DOI] [PubMed] [Google Scholar]
- Pauwels RA, Lofdahl CG, Postma DA, Tattersfield AE, O'Byrne P, Barnes PJ, et al. Effect of inhaled formoterol and budesonide on exacerbations of asthma. New England Journal of Medicine 1997;337(20):1405‐11. [DOI] [PubMed] [Google Scholar]
Pauwels 1997b {published data only}
- Pauwels R. Additive effects of inhaled formoterol and budesonide in reducing asthma exacerbations. Allergy 1998;53:20‐3. [DOI] [PubMed] [Google Scholar]
- Pauwels RA, Lofdahl CG, Postma DA, Tattersfield AE, O'Byrne P, Barnes PJ, et al. Effect of inhaled formoterol and budesonide on exacerbations of asthma. New England Journal of Medicine 1997;337(20):1405‐11. [DOI] [PubMed] [Google Scholar]
Pohunek 2006a {published data only}
- Pohunek P, Kuna P, Boeck K. Budesonide/formoterol improves lung function compared with budesonide alone in children with asthma [Abstract]. European Respiratory Journal 2004;24(Suppl 48):379s. [Google Scholar]
- Pohunek P, Kuna P, Jorup C, Boeck K. Budesonide/formoterol improves lung function compared with budesonide alone in children with asthma. Pediatric Allergy and Immunology 2006;17(6):458‐65. [DOI] [PubMed] [Google Scholar]
- Pohunek P, Matulka M, Rybnicek O, Kopriva F, Honomichlova H, Svobodova T. Dose‐related efficacy and safety of formoterol (Oxis) Turbuhaler compared with salmeterol Diskhaler in children with asthma. Pediatric Allergy & Immunology 2004;15(1):32‐9. [DOI] [PubMed] [Google Scholar]
Pohunek 2006b {published data only}
- Pohunek P, Kuna P, Boeck K. Budesonide/formoterol improves lung function compared with budesonide alone in children with asthma [Abstract]. European Respiratory Journal 2004;24(Suppl 48):379s. [Google Scholar]
- Pohunek P, Kuna P, Jorup C, Boeck K. Budesonide/formoterol improves lung function compared with budesonide alone in children with asthma. Pediatric Allergy and Immunology 2006;17(6):458‐65. [DOI] [PubMed] [Google Scholar]
- Pohunek P, Matulka M, Rybnicek O, Kopriva F, Honomichlova H, Svobodova T. Dose‐related efficacy and safety of formoterol (Oxis) Turbuhaler compared with salmeterol Diskhaler in children with asthma. Pediatric Allergy & Immunology 2004;15(1):32‐9. [DOI] [PubMed] [Google Scholar]
Price 2002 {published data only}
- Price D, Dutchman D, Mawson A, Bodalia B, Duggan S, Todd P, FLOW (Eformoterol in the management of mild asthma‐eformoterol Turboinhaler with budesonide Turbohaler) research group. Early asthma control and maintenance with eformoterol following reduction of inhaled corticosteroid dose. Thorax 2002;57(9):791‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Price MJ, Briggs AH. Development of an economic model to assess the cost effectiveness of asthma management strategies. Pharmacoeconomics 2002; Vol. 20, issue 3:183‐94. [DOI] [PubMed]
- Price MJ, Sondhi S, Yan S, Nyth A, House K. Salmeterol/fluticasone propionate combination inhaler is more cost effective than fluticasone propionate in patients with asthma. European Respiratory Society 1999 Annual Congress; Oct 9‐13; Madrid, Spain. 1999.
Reddel 2007 {unpublished data only}
- Reddel HK, Peyters MJ, Wark PA, Sand IB, Jenkins CR. Comparison of the efficacy of Seretide and Flixotide when down‐titrating the inhaled corticosteroid dose. Respirology 2007;12(Suppl 1):A40. [Google Scholar]
Russell 1995 {published data only}
- Russell G, Williams DAJ, Weller P, Price JF. Salmeterol xinafoate on children on high dose inhaled steroids. Annals of Allergy, Asthma, and Immunology 1995;75:423‐8. [PubMed] [Google Scholar]
- SALMP/AH91/D89 (GSK). A phase III, multi‐centre, double‐blind, placebo controlled, parallel group study assessing the efficacy and safety of inhaled salmeterol xinafoate (Serevent™) 50 micrograms BD via the Diskhaler™ when added to the existing treatment of moderate to severe asthmatic children. http://ctr.gsk.co.uk 2006 (accessed 20 June 2008).
SAM40008 {unpublished data only}
- SAM40008. A multicentre, randomised, double‐blind, parallel group comparison of the efficacy of Seretide bd and fluticasone propionate bd (both via diskus/accuhaler inhaler) when tapering the inhaled corticosteroid dose in asthmatic adults. http://www.ctr.gsk.co.uk 2004.
SAM40012 {unpublished data only}
- Dorinsky P, Emmett A, Sutton L. Reduced risk for asthma exacerbations in pediatric patients receiving salmeterol plus inhaled corticosteroids (ICS) versus ICS alone [Abstract]. European Respiratory Journal 2004;24(Suppl 48):308s. [Google Scholar]
- SAM40012. A multicentre, randomised, double‐blind, double‐dummy, parallel group comparison of three treatments: 1) salmeterol/fluticasone propionate (SFC) (50/100mcg strength) bd via DISKUS/ACCUHALER inhaler, 2) fluticasone propionate 200mcg bd via DISKUS/ACCUHALER inhaler, 3) fluticasone propionate 100mcg bd via DISKUS/ACCUHALER inhaler in children aged 4‐11 years with asthma. http://www.ctr.gsk.co.uk 2005.
SAS40024 {unpublished data only}
- Dorinsky P, Jones S, Kalberg C, Emmett A, Rickard K. Sustained protection against activity‐induced bronchospasm (AIB) during chronic treatment with the fluticasone propionate/salmeterol combination (FSC). American Journal of Respiratory and Critical Care Medicine 2002; Vol. 165, issue Suppl 8:A568.
- Dorinsky P, Kalberg C, Emmett A, Rickard K. Sustained protection against activity‐induced bronchospasm during chronic treatment with fluticasone/salmeterol combination. European Respiratory Journal 2002;20(Suppl 38):308s. [Google Scholar]
- SAS40024. A randomized, double‐blind, parallel‐group study evaluating the protective effects of the fluticasone propionate/salmeterol combination product (FSC, 100/50mcg BID via diskus) against bronchospasms induced by activity as measured by exercise challenge testing in adolescent and adult subjects who require chronic inhaled corticosteroid therapy for the treatment of persistent asthma. http://www.ctr.gsk.co.uk 2005.
SAS40036 {unpublished data only}
- Keonig S, Waitkus‐Edwards K, Yancey S, Prillaman B, Dorinsky P. Loss of asthma control when patients receiving fluticasone propionate/salmeterol 100/50 mcg Diskus® are stepped‐down to fluticasone propionate, salmeterol or montelukast alone. Journal of Allergy and Clinical Immunology 2004;113(2 (Suppl 1)):S94. [DOI: 10.1016/j.jaci.2003.12.325] [DOI] [Google Scholar]
- SAS40036. A multicenter, randomized, double‐blind, double‐dummy, parallel group, 16‐week comparison of asthma control in adolescents and adults receiving either fluticasone propionate/salmeterol Diskus combination product 100/50mcg BID, fluticasone propionate Diskus 100mcg BID, salmeterol xinafoate Diskus 50mcg BID, or oral montelukast 10mg QD. http://www.ctr.gsk.co.uk 2005.
SAS40037 {unpublished data only}
- SAS40037. A multicenter, randomized, double‐blind, double‐dummy, parallel group, 16‐week comparison of asthma control in adolescents and adults receiving either fluticasone propionate/salmeterol Diskus (combination product 100/50mcg BID, fluticasone propionate Diskus 100mcg BID, salmeterol xinafoate Diskus 50mcg BID, or oral montelukast 10mg QD. http://www.ctr.gsk.co.uk 2005.
SD 037 0344a {unpublished data only}
- AstraZeneca (SD 037 0344). A 3‐month, multi‐centre, double‐blind, double‐dummy, randomised, parallel group, phase III study to investigate the efficacy and safety of formoterol HFA pMDI compared with placebo and Oxis® Turbuhaler® in subjects with asthma. http://www.astrazenecaclinicaltrials.com 2003 (accessed 30 April 2008).
SD 037 0344b {unpublished data only}
- AstraZeneca (SD 037 0344). A 3‐month, multi‐centre, double‐blind, double‐dummy, randomised, parallel group, phase III study to investigate the efficacy and safety of Formoterol HFA pMDI compared with placebo and Oxis® Turbuhaler® in subjects with asthma. http://www.astrazenecaclinicaltrials.com 2003 (accessed 30 April 2008).
SD 039 0349 {unpublished data only}
- AstraZeneca (SD 039 0349). Efficacy and safety of a fixed combination of budesonide/formoterol Turbuhaler® in inhaled steroid‐using asthmatic adults. http://www.astrazenecaclinicaltrials.com 1999 (accessed 30 April 2008).
SD 039 0714 {unpublished data only}
- AstraZeneca Pharmaceuticals. Efficacy and safety of budesonide/formoterol Turbuhaler® (160/4.5 mcg b.i.d. delivered dose) compared to budesonide Turbuhaler® (200 mcg b.i.d. metered dose) in steroid‐using asthmatic adolescent patients. A double‐blind, double‐dummy, randomised, parallel group, phase III, multicentre study. www.astrazenecaclinicaltrials.com 2005 (accessed 21 February 2006).
SD 039 0718 {unpublished data only}
- AstraZeneca Pharmaceuticals (SD 039 0718). A twelve‐week, randomized, double‐blind, double‐dummy trial of Symbicort® (40/4.5 mcg) versus its mono‐products (budesonide and formoterol) in asthmatic children aged six to fifteen years. http://www.astrazenecaclinicaltrials.com 2005 (accessed 4 January 2008).
SD 039 0719 {unpublished data only}
- AstraZeneca Pharmaceuticals (SD 039 0719). A six‐month, randomized, open‐label safety study of Symbicort® (160/4.5 mcg) compared to Pulmicort Turbuhaler® in asthmatic children aged 6 to 11 years. http://www.astrazenecaclinicaltrials.com 2005 (accessed 4 January 2008).
SD 039 0725a {unpublished data only}
- AstraZeneca Pharmaceuticals (SD 039 0725). A twelve‐week, randomized, double‐blind, double‐dummy, active‐controlled study of Symbicort® pMDI administered once daily in children and adolescents 6 to 15 years of age with asthma. http://www.astrazenecaclinicaltrials.com 2005 (accessed 4 January 2008).
SD 039 0725b {unpublished data only}
- AstraZeneca Pharmaceuticals (SD 039 0725). A twelve‐week, randomized, double‐blind, double‐dummy, active‐controlled study of Symbicort® pMDI administered once daily in children and adolescents 6 to 15 years of age with asthma. http://www.astrazenecaclinicaltrials.com 2005 (accessed 4 January 2008).
SD 039 0726a {unpublished data only}
- Ambrose H, Lawrance R, Goldman M. Beta‐adrenergic receptor Gly16Arg variation: effect on response to budesonide/formoterol or budesonide (post‐formoterol) in asthma patients. http://meeting.chestjournal.org. 2007 (accessed 27 June 2008).
- Berger WE, Bleecker ER, O'Dowd L, Miller CJ. Asthma control with once‐daily budesonide/formoterol (BUD/FM) pressurized metered‐dose inhaler. http://www.abstracts2view.com 2007 (accessed 26 June 2008).
- Bleecker ER, Berger WE, O'Dowd L, Miller CJ. Safety of once‐daily budesonide (BUD) and formoterol (FM) administered via one pressurized metered‐dose inhaler (pMDI) in patients with asthma. http://www.abstracts2view.com 2007 (accessed 27 June 2008).
- SD 039 0726 (AstraZeneca). A twelve‐week, randomized, double‐blind, double‐dummy, placebo‐ and active‐controlled study of SYMBICORT® pMDI administered once daily in adults and adolescents with asthma. www.astrazenecaclinicaltrials.com 2005 (accessed 26 June 2008).
SD 039 0726b {unpublished data only}
- Ambrose H, Lawrance R, Goldman M. Beta‐adrenergic receptor Gly16Arg variation: Effect on response to budesonide/formoterol or budesonide (post‐formoterol) in asthma patients. http://meeting.chestjournal.org. 2007 (accessed 27 June 2008).
- Berger WE, Bleecker ER, O'Dowd L, Miller CJ. Asthma control with once‐daily budesonide/formoterol (BUD/FM) pressurized metered‐dose inhaler. http://www.abstracts2view.com 2007 (accessed 26 June 2008).
- Bleecker ER, Berger WE, O'Dowd L, Miller CJ. Safety of once‐daily budesonide (BUD) and formoterol (FM) administered via one pressurized metered‐dose inhaler (pMDI) in patients with asthma. http://www.abstracts2view.com 2007 (accessed 27 June 2008).
- SD 039 0726 (AstraZeneca). A twelve‐week, randomized, double‐blind, double‐dummy, placebo‐ and active‐controlled study of SYMBICORT® pMDI administered once daily in adults and adolescents with asthma. www.astrazenecaclinicaltrials.com 2005 (accessed 26 June 2008).
SD 039 0728 {unpublished data only}
- O'Brien CD, Peters SP, Prenner BM, Martin P. Long‐term safety of budesonide/formoterol pressurized metered‐dose inhaler (BUD/FM pMDI) in asthma patients: adverse events and asthma exacerbations. http://www.abstracts2view.com 2007 (accessed 26 June 2008).
- O'Brien CD, Peters SP, Prenner BM, Martin P. Resource use with budesonide/formoterol pressurized metered‐dose inhaler (BUD/FM pMDI) versus BUD pMDI in asthma patients. www.astrazenecaclinicaltrials.com 2007 (accessed 26 June 2008).
- Peters SP, Prenner BM, Martin P, O'Brien CD. Long‐term effects on lung function of budesonide (BUD) and formoterol (FM) in one pressurized metered‐dose inhaler (BUD/FM pMDI) and BUD pMDI in patients with asthma. http://www.abstracts2view.com 2007 (accessed 26 June 2008).
- Prenner BM, Peters SP, Martin P, O'Brien CD. Long‐term control of asthma symptoms with budesonide/formoterol pressurized metered‐dose inhaler (BUD/FM pMDI) versus BUD pMDI. http://www.abstracts2view.com 2007 (accessed 26 June 2008).
- Prenner BM, Peters SP, Martin P, O'Brien CD. Safety pharmacodynamics (PD) of budesonide/formoterol (BUD/FM) pMDI in asthma patients. http://www.abstracts2view.com 2007 (accessed 26 June 2008).
- SD 039 0728 (AstraZeneca). A 52‐week, randomized, double‐blind, single‐dummy, parallel‐group,multicenter Phase III study comparing the long‐term safety of SYMBICORT® pMDI 160/4.5 μg x 4 actuations twice daily to SYMBICORT® pMDI 160/4.5 μg x 2 actuations twice daily and budesonide HFA pMDI 160 μg x 4 actuations twice daily in adult and adolescent subjects with asthma. www.astrazenecaclinicaltrials.com 2006 (accessed 26 June 2008).
SFA100314 {unpublished data only}
- GlaxoSmithKline (SFA100314). A stratified, multicenter, randomized, double‐blind, parallel group, 4‐week comparison of fluticasone propionate/salmeterol DISKUS combination product 100/50mcg BID versus fluticasone propionate DISKUS 100mcg BID in pediatric and in adolescent subjects with activity‐induced bronchospasm. http://www.ctr.gsk.co.uk 2007 (accessed 16 May 2008).
SFA100316 {unpublished data only}
- GlaxoSmithKline (SFA100316). A stratified, multicenter, randomized, double‐blind, parallel group, 4‐week comparison of fluticasone propionate/salmeterol DISKUS combination product 100/50mcg BID versus fluticasone propionate DISKUS 100mcg BID in pediatric and in adolescent subjects with activity‐induced bronchospasm. http://ctr.gsk.co.uk 2006 (accessed 30 April 2008).
SFCF4026 {unpublished data only}
- SFCF4026. Maintenance of asthma control in adults: comparison of three therapeutic strategies in patients whose asthma is controlled by a medium dose of inhaled corticosteroid and a long‐acting inhaled beta2‐agonist. http://www.ctr.gsk.co.uk 2005.
Shapiro 2000 {published data only}
- SFCA3003. A randomized, double‐blind, parallel‐group trial evaluating safety and efficacy of salmeterol 50mcg BID and fluticasone propionate 250mcg BID individually and in combination and placebo in subjects with asthma. http://ctr.gsk.co.uk 2004 (accessed 4 June 2008).
- Shapiro G, Lumry W, Wolfe J, Given J, White MV, Woodring A, et al. Combined salmeterol 50mcg and fluticasone propionate 350mcg in the Diskus device for the treatment of asthma. American Journal of Respiratory & Critical Care Medicine 2000;161:527‐34. [DOI] [PubMed] [Google Scholar]
Simons 1997 {published data only}
- Simons FE, Gerstner TV, Cheang MS. Tolerance to the bronchoprotective effect of salmeterol in adolescents with exercise‐induced asthma using concurrent inhaled glucocorticoid treatment. Pediatrics 1997;99(5):655‐9. [DOI] [PubMed] [Google Scholar]
SMS40012 {unpublished data only}
- SLMF 4002 (SMS40012). Efficacy and safety of salmeterol in patients with asthma controlled with inhaled corticosteroids. http://www.ctr.gsk.co.uk 2005.
Stelmach 2007 {published data only (unpublished sought but not used)}
- Stelmach I, Grzelewskia T, Bobrowska‐Korzeniowska M, Stelmach P, Kuna P. A randomized, double‐blind trial of the effect of anti‐asthma treatment on lung function in children with asthma. Pulmonary Pharmacology & Therapeutics 2007;20:691‐700. [DOI] [PubMed] [Google Scholar]
Tal 2002 {published data only}
- AstraZeneca (SD 039 0353). Efficacy and safety of budesonide/formoterol Turbuhaler® in a fixed combination in steroid‐using asthmatic children. http://www.astrazenecaclinicaltrials.com 1999 (accessed 30 April 2008).
- Tal A, Simon G, Vermeulen JH. Symbicort® (budesonide and formoterol in a single inhaler) is effective and well tolerated in children with asthma. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
- Tal A, Simon G, Vermeulen JH, Petru V, Cobos N, Everard ML, et al. Budesonide/formoterol in a single inhaler versus inhaled corticosteroids alone in the treatment of asthma. Pediatric Pulmonology 2002;34(5):342‐50. [DOI] [PubMed] [Google Scholar]
- Tal A, Simon G, Vermeulen JH, Petru V, Cobos N, Everard ML, et al. Rapid and sustained improvements in lung function and symptom control with budesonide/formoterol in adolescent asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:494s.
- Tal A, Simon G, Vermeulen JH, Petru V, Cobos N, Everard ML, et al. Symbicort (budesonide and formoterol in a single inhaler) improves lung function in children with asthma. International Paediatric Respiratory and Allergy Congress, April 1‐4; Prague. 2001:85.
- Tal A, Simon G, Vermeulen JH, Petru V, Cobos N, Everard ML, et al. Symbicort (budesonide and formoterol in a single inhaler) is more effective that budesonide alone in children with asthma. International Paediatric Respiratory and Allergy Congress April 1‐4; Prague. 2001:84‐5.
- Vermeulen JH, Simon G, Tal A. Symbicort® (budesonide and formoterol in a single inhaler) improves lung function in asthmatic children aged 4‐17 years. Annual Thoracic Society 97th International Conference; San Francisco CA, May 18‐23. 2001.
Teper 2005 {published data only}
- Teper AM, Zaragoza SM, Lubovich S, Rodriguez VA, Venalago C, Kofman CD, et al. Effect of fluticasone propionate (FP) with or without salmeterol (S) on bronchial reactivity (BR) in children with mild to moderate persistent asthma [Abstract]. American Thoracic Society 2005 International Conference; May 20‐25; San Diego, California. 2005:[C47] [Poster: A5].
van der Molen 1997 {published data only}
- Molen T, Postma DS, Kraan J, Chapman K, Grossman R, Turner MO, et al. No influence of six months treatment with formoterol on airway hyperresponsiveness in asthma subjects using inhaled corticosteroids. American Journal of Respiratory & Critical Care Medicine 1998;157(3 Suppl):A400. [Google Scholar]
- Molen T, Postma DS, Turner MO, Meyboom‐de Jong B, Malo JL, Chapman K, et al. Effects of the long acting beta agonist formoterol on asthma control in asthmatic patients using inhaled corticosteroids. Thorax 1997;52:535‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molen T, Sears MR, Graaff CS, Postma DS, Meyboom‐de Jong B. Quality of life during formoterol treatment: comparison between asthma‐specific and generic questionnaires. European Respiratory Journal 1998;12(1):30‐4. [DOI] [PubMed] [Google Scholar]
Verberne 1998 {published data only}
- SLGB4014 (SLPT15). Placebo controlled study during one year comparing the addition of salmeterol with an increase of the dose of the inhaled corticosteroid in asthmatic children already on treatment with inhaled corticosteroids. http://ctr.gsk.co.uk 2006 (accessed 20 June 2008).
- Verberne AAPH, Frost C, Duiverman EJ, Grol MH, Kerribijn KF. Addition of salmeterol versus doubling the dose of beclomethasone in children with asthma. American Journal of Respiratory & Critical Care Medicine 1998;158:213‐19. [DOI] [PubMed] [Google Scholar]
Wallin 2003 {published data only}
- Sue‐Chu M, Wallin A, Wilson S, Ward J, Sandstrom T, Djukanovic R, et al. Bronchial biopsy study in asthmatics treated with low and high dose fluticasone propionate (FP) compared to low dose FP combined with salmeterol. European Respiratory Society 1999 Annual Congress Oct 9‐13; Madrid, Spain. 1999.
- Wallin A, Sandstrom T, Cioppa GD, Holgate S, Wilson S. The effects of regular inhaled formoterol and budesonide on preformed Th‐2 cytokines in mild asthmatics. Respiratory Medicine 2002;96(12):1021‐5. [DOI] [PubMed] [Google Scholar]
- Wallin A, Sue‐Chu M, Bjermer L, Ward J, Sandstrom T, Lindberg A, et al. Effect of inhaled fluticasone with and without salmeterol on airway inflammation in asthma. Journal of Allergy & Clinical Immunology 2003;112(1):72‐8. [DOI] [PubMed] [Google Scholar]
Weiler 2005 {published and unpublished data}
- SAS40025. A randomized, double‐blind, parallel‐group study evaluating the protective effects of the fluticasone propionate/salmeterol combination product (FSC 250/50mcg BID via diskus) against bronchospasms induced by activity as measured by exercise challenge testing in adolescent and adult subjects who require chronic inhaled corticosteroid therapy for the treatment of persistent asthma. http://www.clinicalstudyresults.org 2005 (accessed 4 November 2008).
- Weiler JM, Nathan RA, Rupp NT, Kalberg CJ, Emmett A, Dorinsky P. Effect of fluticasone/salmeterol via a single device on exercise‐induced bronchospasm in patients with persistent asthma. Annals of Allergy Asthma and Immunology 2005;94(1):65‐72. [DOI] [PubMed] [Google Scholar]
Zetterstrom 2001a {published data only}
- Zetterstrom O, Buhl R, Mellem H. Efficacy and safety of Symbicort® (budesonide and formoterol in a single inhaler) in adults with asthma. Annual Thoracic Society 97th International Conference; San Francisco CA, May 18‐23. 2001.
- Zetterstrom O, Buhl R, Mellem H, Perpina M, Hedman J, O'Neill S, et al. Improved asthma control with budesonide/formoterol in a single inhaler, compared with budesonide alone. European Respiratory Journal 2001;18(2):262‐8. [DOI] [PubMed] [Google Scholar]
- Zetterstrom O, Buhl R, Mellem H, Perpina M, Hedman J, O'Neill S, et al. The new single inhaler product containing both budesonide/formoterol improves asthma control in adults. European Respiratory Journal 2000; Vol. 16, issue Suppl 31:455s. [DOI] [PubMed]
- Zetterström O, Buhl R, Mellem H, Perpiñá M, Hedman J, O'Neill S, et al. Efficacy and safety of a new single inhaler product containing both budesonide and formoterol in adult asthma. European Respiratory Journal 2000; Vol. 16, issue 31:455s.
Zetterstrom 2001b {published data only}
- Zetterstrom O, Buhl R, Mellem H. Efficacy and safety of Symbicort® (budesonide and formoterol in a single inhaler) in adults with asthma. Annual Thoracic Society 97th International Conference; San Francisco CA, May 18‐23. 2001.
- Zetterstrom O, Buhl R, Mellem H, Perpina M, Hedman J, O'Neill S, et al. Improved asthma control with budesonide/formoterol in a single inhaler, compared with budesonide alone. European Respiratory Journal 2001;18(2):262‐8. [DOI] [PubMed] [Google Scholar]
- Zetterstrom O, Buhl R, Mellem H, Perpina M, Hedman J, O'Neill S, et al. The new single inhaler product containing both budesonide/formoterol improves asthma control in adults. European Respiratory Journal 2000;16(Suppl 31):455s. [DOI] [PubMed] [Google Scholar]
- Zetterström O, Buhl R, Mellem H, Perpiñá M, Hedman J, O'Neill S, et al. Efficacy and safety of a new single inhaler product containing both budesonide and formoterol in adult asthma. European Respiratory Journal 2000; Vol. 16, issue 31:455s.
Zimmerman 2004a {published data only}
- Zimmerman B, D'Urzo A, Berube D. Efficacy and safety of formoterol turbuhaler(R) when added to inhaled corticosteroid treatment in children with asthma. Pediatric Pulmonology 2004;37(2):122‐7. [DOI] [PubMed] [Google Scholar]
- Zimmerman B, D'Urzo A, Berube D. Efficacy and tolerability of formoterol Turbuhaler in 6‐11 year old children with asthma, not adequately controlled with inhaled corticosteroids. European Respiratory Society Annual Congress. 2002:P2734.
- Zimmerman B, D'Urzo A, Berube D. Efficacy and tolerability of formoterol turbuhaler(r) compared with placebo, in children (6‐11 yr) with asthma poorly controlled with inhaled corticosteroids [abstract]. American Journal of Respiratory and Critical Care Medicine. 2002; Vol. 165, issue 8 Suppl:A746.
Zimmerman 2004b {published data only}
- Zimmerman B, D'Urzo A, Berube D. Efficacy and safety of formoterol turbuhaler(R) when added to inhaled corticosteroid treatment in children with asthma. Pediatric Pulmonology 2004;37(2):122‐7. [DOI] [PubMed] [Google Scholar]
- Zimmerman B, D'Urzo A, Berube D. Efficacy and tolerability of formoterol Turbuhaler in 6‐11 year old children with asthma, not adequately controlled with inhaled corticosteroids. European Respiratory Society Annual Congress. 2002:P2734.
- Zimmerman B, D'Urzo A, Berube D. Efficacy and tolerability of formoterol turbuhaler(r) compared with placebo, in children (6‐11 yr) with asthma poorly controlled with inhaled corticosteroids [abstract]. American Journal of Respiratory and Critical Care Medicine. 2002; Vol. 165, issue 8 Suppl:A746.
References to studies excluded from this review
Aalbers 2004 {published data only}
- Aalbers R, Backer V, Kava TT, Omenaas ER, Sandstrom T, Jorup C, et al. Adjustable maintenance dosing with budesonide/formoterol compared with fixed‐dose salmeterol/fluticasone in moderate to severe asthma. Current Medical Research & Opinion 2004;20(2):225‐40. [DOI] [PubMed] [Google Scholar]
- Aalbers R, Backer V, Kava TT, Welte T, Omenaas ER, Bergqvist PB, et al. Adjustable dosing with budesonide/formoterol reduces the rate of asthma exacerbations compared with fixed dosing salmeterol/fluticasone. European Respiratory Society. 2003:p‐2‐20.
- Aalbers R, Backer V, Kava TT, Welte T, Omenaas ER, Bergqvist PB, et al. Improvements in FEV1 are greater with budesonide/formoterol than with salmeterol/fluticasone. European Respiratory Society Annual Congress. 2003:P‐2‐19.
- Aalbers R, Backer V, Kava TT, Welte T, Omenaas ER, Bergqvist PBF, et al. Is well controlled asthma weeks a useful measure? Fewer exacerbations in patients treated with budesonide/formoterol than salmeterol/fluticasone. European Respiratory Society. 2003:P‐2‐18.
Adinoff 1998 {published data only}
- Adinoff AD, Schwartz HJ, Rickard KA, Yancey SW, Swearingen BE. Salmeterol compared with current therapies in chronic asthma. Journal of Family Practice 1998;47(4):278‐84. [PubMed] [Google Scholar]
Ankerst 2003 {published data only}
- Ankerst J, Persson G, Weibull E. Cardiovascular effects of a high dose of the budesonide/formoterol single inhaler in asthmatic patients. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:53s.
- Ankerst J, Persson G, Weibull E. Tolerability of a high dose of budesonide/formoterol in a single inhaler in patients with asthma. Pulmonary Pharmacology & Therapeutics 2003;16:147‐51. [DOI] [PubMed] [Google Scholar]
Anonymous 2003 {published data only}
- Anonymous. Flexible dosing of combination inhaler cuts asthma exacerbations. Pharmaceutical Journal 2003;271(7271):535. [Google Scholar]
Arvidsson 1991 {published data only}
- Arvidsson P, Larsson S, Lofdahl CG, Melander B, Svedmyr N, Wahlander L. Inhaled formoterol during one year in asthma: a comparison with salbutamol. European Respiratory Journal 1991;4(10):1168‐73. [PubMed] [Google Scholar]
Aziz 1998 {published data only}
- Aziz I, Hall IP, McFarlane LC, Lipworth BJ. Beta2‐adrenoceptor regulation and bronchodilator sensitivity after regular treatment with formoterol in subjects with stable asthma. Journal of Allergy & Clinical Immunology 1998;101(3):337‐41. [DOI] [PubMed] [Google Scholar]
Aziz 1999a {published data only}
- Aziz I, Lipworth BJ. A bolus of inhaled budesonide rapidly reverses airway subsensitivity and beta2‐adrenoceptor down‐regulation after regular inhaled formoterol. Chest 1999;115(3):623‐8. [DOI] [PubMed] [Google Scholar]
Aziz 1999b {published data only}
- Aziz I, Lipworth BJ. In vivo effect of albuterol on methacholine‐contracted bronchi in conjunction with salmeterol and formoterol. Journal of Allergy & Clinical Immunology 1999;103(5 pt 1):816‐22. [DOI] [PubMed] [Google Scholar]
Aziz 2000 {published data only}
- Aziz I, Wilson AM, Lipworth BJ. Effects of formoterol (FM) and budesonide (BUD) alone or in combination (FM+BUD) on inflammatory markers in asthmatic patients. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999:2854.
- Aziz I, Wilson AM, Lipworth BJ. Effects of once‐daily formoterol and budesonide given alone or in combination on surrogate inflammatory markers in asthmatic adults. Chest 2000;118(4):1049‐58. [DOI] [PubMed] [Google Scholar]
Bacci 2002 {published data only}
- Bacci E, Franco A, Bartoli ML, Carnevali S, Cianchetti S, Dente FL, et al. Comparison of anti‐inflammatory and clinical effects of beclomethasone dipropionate and salmeterol in moderate asthma. European Respiratory Journal 2002;20(1):66‐72. [DOI] [PubMed] [Google Scholar]
Baker 1998 {published data only}
- Baker J, Yancey S, Kalberg C, Petrocella V, Emmett A, Bowers B, et al. Added salmeterol versus increased‐dose fluticasone in patients symptomatic on low‐dose fluticasone. American Journal of Respiratory and Critical Care Medicine 1998;157(Suppl 3):A406. [Google Scholar]
Baki 1998 {published data only}
- Baki A, Karaguzel G. Short‐term effects of budesonide, nedocromil sodium and salmeterol on bronchial hyperresponsiveness in childhood asthma. Acta Paediatrica Japonica 1998;40(3):247‐51. [DOI] [PubMed] [Google Scholar]
Baraniuk 1999 {published data only}
- Baraniuk J, Murray JJ, Nathan RA, Berger WE, Johnson M, Edwards LD, et al. Fluticasone alone or in combination with salmeterol vs triamcinolone in asthma. Chest 1999;116(3):625‐32. [DOI] [PubMed] [Google Scholar]
- Cook D, Srebro SH, Rogenes PR, Rickard K, Edwards L, Johnson MC. A comparison of the safety and efficacy of fluticasone, triamcinolone, and fluticasone plus salmeterol in patients with mild to moderate asthma. American Journal of Respiratory & Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A416.
- Johnson MC, Srebro SH, Rogenes PR, Rickard K, Edwards L. A comparison of physician‐rated and patient‐rated outcomes in a study with fluticasone, triamcinolone, and fluticasone plus salmeterol. American Journal of Respiratory & Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A414.
Bateman 1998 {published data only}
- Bateman ED, Britton M, Carrillo J, Almeida J, Wixon C. Salmeterol/fluticasone combination inhaler. A new, effective and well tolerated treatment for asthma. Clinical Drug Investigation 1998;16(3):193‐201. [DOI] [PubMed] [Google Scholar]
Bateman 2003a {published data only}
- Bateman ED, Bantje TA, Gomes M, Toumbis M, Huber R, Eliraz A, et al. Symbicort (budesonide and formoterol in a single inhaler) is a more effective treatment than fluticasone in asthma patients. Annual Thoracic Society 97th International Conference. 2001.
- Bateman ED, Bantje TA, Gomes MJ, Toumbis MG, Huber RM, Naya I, et al. Combination therapy with a single inhaler budesonide/formoterol compared with high dose fluticasone propionate alone in patients with moderate persistent asthma. American Journal of Respiratory Medicine 2003;2(3):275‐81. [DOI] [PubMed] [Google Scholar]
- Bateman ED, Bantje TA, Joao Gomes M, Toumbis M, Huber R, Eliraz A. Early and sustained benefits of budesonide and formoterol in a single inhaler vs fluticasone in moderate asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:157s.
- Bateman ED, Bantje TA, Joao Gomes M, Toumbis M, Huber R, Eliraz A. Early and sustained benefits of budesonide and formoterol in a single inhaler vs fluticasone in moderate asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:157s.
- Bateman ED, Bantje TA, Joao Gomes M, Toumbis M, Huber R, Eliraz A, et al. Symbicort (budesonide/eformoterol) Turbohaler controls asthma more effectively than fluticasone Diskus. Thorax 2001; Vol. 56, issue Suppl 3:iii 63.
- Ericsson K, Bantje TA, Huber H, Borg S. Symbicort® Turbuhaler® is more cost effective than fluticasone Diskus™ in the treatment of asthma. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
- Ericsson K, Bantje TA, Huber R, Borg S, Anderson F. Cost‐effectiveness of budesonide and formoterol in a single inhaler compared to fluticasone in the treatment of asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:157s.
- Ericsson K, Bantje TA, Huber R, Borg S, Andersson F. Symbicort Turbohaler is more effective than fluticasone Diskus in the treatment of asthma. Thorax 2001; Vol. 56, issue Suppl 3:iii63.
Bateman 2003b {published data only}
- Bateman ED, Akveld M, Ho M. Greater responder rate to fluticasone propionate/salmeterol combination over montelukast plus fluticasone in asthma. American Thoracic Society 99th International Conference. 2003:B036 Poster H90.
Behling 1999 {published data only}
- Behling B, Matthys H. Comparison of efficacy and safety of formoterol with terbutaline in children with mild to moderate asthma. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999:369.
Bensch 2002 {published data only}
- Bensch G, Berger WE, Blokhnn M, Socolovshy AL, Thomson MH, Till MD, et al. One year efficacy and safety of inhaled formoterol dry powder in children with persistent asthma. Annals of Allergy, Asthma, and Immunology 2002;89:180‐90. [DOI] [PubMed] [Google Scholar]
Berger 2001 {published data only}
- Berger W, Bensch G, Blokhin BM, Socolovsky AL, Thompson MH, Till D. Addition of formoterol (Foradil®) improves lung function and symptoms in children with persistent asthma not controlled by inhaled corticosteroids. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Berggren 2001 {published data only}
- Berggren F, Ekstrom T. A cost‐effectiveness study comparing the as‐needed use of formoterol (Oxis) and terbutaline (Bricanyl) in patients with moderate to severe asthma. Respiratory Medicine 2001;95(9):753‐8. [DOI] [PubMed] [Google Scholar]
Bergmann 2004 {published data only}
- Bergmann KC, Lindemann L, Braun R, Steinkamp G. Salmeterol/fluticasone propionate (50/250 mug) combination is superior to double dose fluticasone (500 mug) for the treatment of symptomatic moderate asthma: a prospective, double‐blind trial. Swiss Medical Weekly 2004;134(3‐4):50‐8. [DOI] [PubMed] [Google Scholar]
Bernstein 2002 {published data only}
Bessmertny 2002 {published data only}
- Bessmertny O, DiGregorio RV, Cohen H, Becker E, Looney D, Golden J, et al. A randomized clinical trial of nebulized magnesium sulfate in addition to albuterol in the treatment of acute mild‐to‐moderate asthma exacerbations in adults. Annals of Emergency Medicine 2002;39(6):585‐91. [DOI] [PubMed] [Google Scholar]
Bijl‐Hofland 2001 {published data only}
- Bijl‐Hofland ID, Cloosterman SG, Folgering HT, Elshout FJ, Weel C, Schayck CP. Inhaled corticosteroids, combined with long‐acting beta(2)‐agonists, improve the perception of bronchoconstriction in asthma. American Journal of Respiratory & Critical Care Medicine 2001;164(5):764‐9. [DOI] [PubMed] [Google Scholar]
Bjermer 2000 {published data only}
- Bjermer L, Bisgaard H, Bousquet J, Fabbri LM, Greening A, Haahtela T, et al. Montelukast or salmeterol combined with an inhaled steroid in adult asthma: design and rationale of a randomized, double‐blind comparative study (the IMPACT Investigation of Montelukast as a Partner Agent for Complementary Therapy‐trial). Respiratory Medicine 2000;94(6):612‐21. [DOI] [PubMed] [Google Scholar]
Bjermer 2002 {published data only}
- Bjermer L, Greening A, Haahtela T, Bousquet J, Holgate ST, Picado C, et al. Addition of montelukast or salmeterol to fluticasone in patients with uncontrolled asthma: results of the IMPACT trial. Chest 2002; San Diego, CA. 2002:434.
Bjermer 2003 {published data only}
- Bjermer L, Bisgaard H, Bousquet J, Fabbri LM, Greening AP, Haahtela T, at al. Montelukast and fluticasone compared with salmeterol and fluticasone in protecting against asthma exacerbation in adults: one year, double blind, randomised, comparative trial . BMJ 2003;327(7420):891. [DOI] [PMC free article] [PubMed] [Google Scholar]
Bloom 2003 {published data only}
- Bloom J, Calhoun W, Koenig S, Yancey S, Reilly D, Edwards L, et al. Fluticasone propionate/salmeterol 100/50mcg is inhaled steroid sparing in patients who require fluticasone propionate 250mcg for asthma stability. American Thoracic Society 99th International Conference. 2003:D034 Poster C33.
Boonsawat 2003 {published data only}
- Boonsawat W, Charoenratanakul S, Pothirat C, Sawanyawisuth K, Seearamroongruang T, Bengtsson T, et al. Formoterol (OXIS) Turbuhaler as a rescue therapy compared with salbutamol pMDI plus spacer in patients with acute severe asthma. Respiratory Medicine 2003;97(9):1067‐74. [DOI] [PubMed] [Google Scholar]
Booth 1993 {published data only}
- Booth H, Fishwick K, Harkawat R, Devereux G, Hendrick DJ, Walters EH. Changes in methacholine induced bronchoconstriction with the long acting beta 2 agonist salmeterol in mild to moderate asthmatic patients. Thorax 1993;48(11):1121‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Boskovska 2001 {published data only}
- Boskovska MI, Dokic D, Busletic‐Bozinovska K, Arbutina S, Goseva Z. Concomitant use of low‐dose inhaled corticosteroids and a long‐acting bronchodilator vis a vis doubling the dose of inhaled corticosteroid in asthma patients. European Respiratory Journal 2001, issue Suppl 33:98s.
Bouchard 2000 {published data only}
- Bouchard J, Arkinstall W, Tesarowski D. Efficacy of salmeterol and fluticasone propionate (FP) combination therapy versus FP alone in mild/moderate asthma. American Journal of Respiratory and Critical Care Medicine. 2000; Vol. 161, issue Suppl 3:A197.
Boulet 2003 {unpublished data only}
- Boulet LP, Chapman K, Roberts J, Watson EG. Efficacy of salmeterol/fluticasone propionate HFA MDI versus high dose fluticasone propionate HFA MDI in adolescent and adult asthma. European Respiratory Society Meeting. 2003.
Bouros 1999 {published data only}
- Bouros D, Bachlitzanakis N, Kottakis J, Pfister P, Polychronopoulos V, Papadakis E, et al. Foromoterol and beclomethasone versus higher dose beclomethasone as maintenance therapy in adult asthma. European Respiratory Journal 1999;14:627‐32. [DOI] [PubMed] [Google Scholar]
Brambilla 1994 {published data only}
- Brambilla C, Chastang C, Georges D, Bertin L. Salmeterol compared with slow‐release terbutaline in nocturnal asthma. A multicenter, randomized, double‐blind, double‐dummy, sequential clinical trial. French Multicenter Study Group. Allergy 1994;49(6):421‐6. [DOI] [PubMed] [Google Scholar]
Brambilla 2003 {published data only}
- Brambilla C, Gros V, Bourdeix I. Formoterol 12 mcg bid administered via single dry powder inhaler in adults with asthma suboptimally controlled with salmeterol or on demand salbutamol. Clinical Therapeutics 2003;25(7):2022‐36. [DOI] [PubMed] [Google Scholar]
Braniuk 1999 {published data only}
- Braniuk J, Murray JJ, Nathan RA, Berger WE, Johnson M, Edwards LD, et al. Fluticasone alone or in combination with salmeterol vs triamcinolone in asthma. Chest 1999;116(3):625‐32. [DOI] [PubMed] [Google Scholar]
Brenner 1988 {published data only}
- Brenner M, Berkowitz R, Marshall N, Strunk RC. Need for theophylline in severe steroid‐requiring asthmatics. Clinical Allergy 1988;18(2):143‐50. [DOI] [PubMed] [Google Scholar]
Britton 1992 {published data only}
- Britton MG, Earnshaw JS, Palmer JBD. A twelve month comparison of salmeterol with salbutamol in asthmatic patients. European Respiratory Journal 1992;5(9):1062‐7. [PubMed] [Google Scholar]
Britton 1998 {published data only}
- Britton MG, Carrillo T, Almeida J, Wixon C. Combined Serevent™ and fluticasone propionate (50/100µg strength) bd via one Diskus™ (Accuhaler™) inhaler compared with salmeterol 50µg and fluticasone propionate 100µg bd via two separate Diskus inhalers. American Journal of Respiratory & Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A415.
Brogden 1991 {published data only}
- Brogden RN, Faulds D. Salmeterol xinafoate. A review of its pharmacological properties and therapeutic potential in reversible obstructive airways disease. Drugs 1991;42(5):895‐912. [DOI] [PubMed] [Google Scholar]
Buchvald 2003 {published data only}
- Buchvald F, Bisgaard H. Comparisons of the complementary effect on exhaled nitric oside of salmeterol vs montelukast in asthmatic children taking regular budesonide. Annals of Allergy, Asthma & Immunology 2003;91(3):309‐13. [DOI] [PubMed] [Google Scholar]
Busse 1999 {published data only}
- Busse W, Nelson H, Wolfe J, Kalberg C, Yancey SW, Rickard KA. Comparison of inhaled salmeterol and oral zafirlukast in patients with asthma. Journal of Allergy & Clinical Immunology 1999;103(6):1075‐80. [DOI] [PubMed] [Google Scholar]
Busse 2003a {published data only}
- Busse W, Koenig SM, Oppenheimer J, Sahn SA, Yancey SW, Reilly D, et al. Steroid‐sparing effects of fluticasone propionate 100mcg and salmeterol 50mcg administered twice daily in a single product in patients previously controlled with fluticasone propionate 250mcg administered twice daily. Journal of Allergy & Clinical Immunology 2003;111(2):57‐65. [DOI] [PubMed] [Google Scholar]
- Sahn S, Yancey S, Reilly D, Edwards L, Rickard K, Dorinsky P. The fluticasone propionate/salmeterol (FSC) combination product 100/50 mcg BID is steroid sparing in patients who require FP250 mcg BID for asthma stability . Chest 2002; San Diego, CA. 2002.
Busse 2003b {published data only}
- Busse W, Koenig SM, Oppenheimer J, Sahn SA, Yancey SW, Reilly D, et al. Steroid‐sparing effects of fluticasone propionate 100mcg and salmeterol 50mcg administered twice daily in a single product in patients previously controlled with fluticasone propionate 250mcg administered twice daily. Journal of Allergy & Clinical Immunology 2003;111(2):57‐65. [DOI] [PubMed] [Google Scholar]
Byrnes 2000 {published data only}
- Byrnes C, Shrewsbury S, Barnes PJ, Bush A. Salmeterol in paediatric asthma. Thorax 2000;55(9):780‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Calhoun 2001 {published data only}
- Calhoun WJ, Nelson HS, Nathan RA, Pepsin PJ, Kalberg C, Emmett A, et al. Comparison of fluticasone propionate‐salmeterol combination therapy and montelukast in patients who are symptomatic on short‐acting 2‐agonists alone. American Journal of Respiratory & Critical Care Medicine 2001;164(5):759‐63. [DOI] [PubMed] [Google Scholar]
Calverley 2002 {published data only}
- Calverley PMA, Pauwels RA, Vestbo J, Jones PW, Pride NB, Gulsvik A, et al. Salmeterol/fluticasone propionate combination for one year provides greater clinical benefit than its individual components in COPD. American Journal of Respiratory & Critical Care Medicine 2002; Vol. 165, issue Suppl 8:A226.
Castle 1993 {published data only}
- Castle W, Fuller R, Hall J, Palmer J. Serevent nationwide surveillance study: comparison of salmeterol with salbutamol in asthmatic patients who require regular bronchodilator treatment. BMJ 1993;306(6884):1034‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Cazzola 2000 {published data only}
- Cazzola M, Lorenzo G, Perna F, Calderaro F, Testi R, Centanni S. Additive effects of salmeterol and fluticasone or theophylline in COPD. Chest 2000;118(6):1576‐81. [DOI] [PubMed] [Google Scholar]
Chan 2001 {published data only}
- Chan JS, Cowie RL, Lazarenko GC, Little C, Scott S, Ford GT. Comparison of intramuscular betamethasone and oral prednisone in the prevention of relapse of acute asthma. Canadian Respiratory Journal. 2001;8(3):147‐52. [DOI] [PubMed] [Google Scholar]
Chapman 1999 {published data only}
- Chapman KR, Ringdal N, Backer V, Palmqvist M, Saarelainen S, Briggs M. Salmeterol and fluticasone propionate (50/250 mug) administered via combination Diskus inhaler: as effective as when given via separate Diskus inhalers. Canadian Respiratory Journal 1999;6(1):45‐51. [DOI] [PubMed] [Google Scholar]
Cheer 2003 {published data only}
- Cheer SM, Warner GT, Easthope SE. Formoterol delivered by Turbuhaler: in pediatric asthma. Paediatric Drugs 2003;5(1):63‐8. [DOI] [PubMed] [Google Scholar]
Cloosterman 2001 {published data only}
- Cloosterman SG, Bijl‐Hofland ID, Herwaarden CL, Akkermans RP, Elshout FJ, Folgering HT, et al. A placebo‐controlled clinical trial of regular monotherapy with short‐acting and long‐acting beta(2)‐agonists in allergic asthmatic patients. Chest 2001;119(5):1306‐15. [DOI] [PubMed] [Google Scholar]
Condemi 1999 {published data only}
- Condemi JJ, Goldstein S, Kalberg C, Yancey S, Emmett A, Rickard K. The addition of salmeterol to fluticasone propionate versus increasing the dose of fluticasone propionate in patients with persistent asthma. Annals of Allergy, Asthma, and Immunology 1999;82:383‐9. [DOI] [PubMed] [Google Scholar]
Condemi 2001 {published data only}
- Condemi JJ. Comparison of the efficacy of formoterol and salmeterol in patients with reversible obstructive airway disease: a multicenter, randomized, open‐label trial. Clinical Therapeutics 2001;23(9):1529‐41. [DOI] [PubMed] [Google Scholar]
Cook 2001 {published data only}
- Cook CK, Prillaman BA, House KW, Rickard KA, Shah TP. Concurrent use of salmeterol/fluticasone propionate Diskus® powder combination product and fluticasone propionate aqueous nasal spray does not adversely affect HPA‐axis function. Annals of Allergy, Asthma, and Immunology 2001;86:98. [Google Scholar]
Corren 2007 {published and unpublished data}
- AstraZeneca (SD 039 0617). A twelve week, randomized, double‐blind, double‐dummy, placebo‐controlled trial of SYMBICORT® pMDI (80/4.5 mcg) versus its mono‐products (budesonide and formoterol) in children and adults with asthma. www.astrazenecaclinicaltrials.com 2007 (accessed 15 May 2008).
- Corren J, Korenblat PE, Miller CJ, O'Brien CD, Mezzanotte WS. Twelve‐week, randomized, placebo‐controlled, multicenter study of the efficacy and tolerability of budesonide and formoterol in one metered‐dose inhaler compared with budesonide alone and formoterol alone in adolescents and adults with asthma. Clinical Therapeutics 2007;29(5):823‐43. [DOI] [PubMed] [Google Scholar]
Crompton 1999 {published data only}
- Crompton GK, Ayres JG, Basran G, Schiraldi G, Brusasco V, Eivindson A, et al. Comparison of oral bambuterol and inhaled salmeterol. American Journal of Respiratory & Critical Care Medicine 1999;159(3):824‐8. [DOI] [PubMed] [Google Scholar]
Currie 2002 {published data only}
- Currie GP, Stenback S, Lipworth BJ. Dissociation in effects of fluticasone but not fluticasone/salmeterol on lung function and airway hyperresponsiveness in mild persistent asthma. European Respiratory Society Annual Congress. 2002:P2391.
Currie 2003a {published data only}
- Currie GP, Lee DK, Haggart K, Bates CE, Lipworth BJ. Effects of montelukast on surrogate inflammatory markers in corticosteroid‐treated patients with asthma. American Journal of Respiratory and Critical Care Medicine 2003;167(9):1232‐8. [DOI] [PubMed] [Google Scholar]
Currie 2003b {published data only}
- Currie GP, Bates CE, Lee DK, Jackson CM, Lipworth BJ. Effects of fluticasone plus salmeterol versus twice the dose of fluticasone in asthmatic patients. European Journal of Clinical Pharmacology 2003;59(1):11‐15. [DOI] [PubMed] [Google Scholar]
Currie 2003c {published data only}
- Currie GP, Stenback S, Lipworth BJ. Effects of fluticasone vs. fluticasone/salmeterol on airway calibre and airway hyperresponsiveness in mild persistent asthma. British Journal of Clinical Pharmacology 2003;56(1):11‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
D'Alonzo 1994 {published data only}
- D'Alonzo GE, Nathan RA, Henochowicz, Morris RJ, Ratner P, Rennard SI. Salmetrol xinafoate as maintenance therapy compared with albuterol in patients with asthma. JAMA 1994;271:1412‐16. [PubMed] [Google Scholar]
Dahl 1989 {published data only}
- Dahl R, Pedersen B, Hagglof B. Nocturnal asthma: effect of treatment with oral sustained‐release terbutaline, inhaled budesonide, and the two in combination. Journal of Allergy & Clinical Immunology. 1989;83(4):811‐5. [DOI] [PubMed] [Google Scholar]
Dahl 1991 {published data only}
- Dahl R, Earnshaw JS, Palmer JBD. Salmeterol: a four week study of a long‐acting beta‐adrenoceptor agonist for the treatment of reversible airways disease. European Respiratory Journal 1991;4(10):1178‐84. [PubMed] [Google Scholar]
Dal Negro 2001a {published data only}
- Dal Negro R, Micheletto C, Tognella S, Trevisan F, Pomari C. Short‐term bronchodilation following salmeterol 50mcg and combined salmeterol + fluticasone p(50/250mcg) via Diskus: a randomized, double blind cross‐over study in reversible airway obstruction. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Dal Negro 2001b {published data only}
- Dal Negro RW, Micheletto C, Pomari C, Trevisan F, Tognella S. The combination salmeterol (S) + fluticasone propionate (F) in mild‐to‐moderate asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:426s.
Dal Negro 2002a {published data only}
- Dal Negro R, Micheletto C, Trevison F, Tognells S, Pomari C, Spencer S. Salmeterol & fluticasone 50µg/250µg bid vs salmeterol 50µg bid and vs placebo in the long‐term treatment of COPD. American Journal of Respiratory & Critical Care Medicine 2002, issue Suppl 8:A228.
Dal Negro 2002b {published data only}
- Dal Negro RW, Micheletto C, Tognella S, Trevisan F, Pomari C. Is the sequence of salmeterol (SM) and fluticasone p. (FP) dry powder inhalation crucial in persistent mild asthma?. European Respiratory Society Annual Congress. 2002:P396.
Davis 2001 {published data only}
- Davis ES, Bowers B, Pepsin P, Kalberg C, Dorinsky P. The impact of fluticasone propionate/salmeterol combination product compared to oral montelukast on asthma related quality of life. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Del Rio‐Navarro 2001a {published data only}
- Del‐Rio‐Navarro BE, Sienra‐Monge JJL, Alvarez‐Amador M, Reyes‐Ruiz N, Arevalo‐Salas A, Berber A. Serum potassium levels, CPK‐MB and ECG in children suffering asthma treated with beclomethasone or beclomethasone‐salmeterol. Allergologia et Immunopathologia 2001;29(1):16‐21. [DOI] [PubMed] [Google Scholar]
Del‐Rio‐Navarro 2001b {published data only}
- Rio BE, Corona L, Fregosa R, Berber A, Magana J, Sienra‐Monge JL. Effect of salmeterol and salmeterol plus beclomethasone on the saliva flow and saliva IgA levels on patients with chronic moderate persistent asthma (CMPA). Journal of Allergy and Clinical Immunology 2001; Vol. 107, issue 2:s11. [DOI] [PubMed]
- Del‐Rio‐Navarro BE, Coron‐Hernandez L, Fragoso‐Rios R, Berber A, Torres‐Alcantara S, Cuairan‐Ruidiaz V, et al. Effect of salmeterol and salmeterol plus beclomethasone on saliva flow and IgA in patients with moderate‐persistent chronic asthma. Annals of Allergy, Asthma and Immunology 2001;87(5):420‐3. [DOI] [PubMed] [Google Scholar]
Dempsey 2000a {published data only}
- Dempsey OJ, Wilson AM, Sims EJ, Lipworth BJ. Additive anti‐inflammatory effects of montelukast but not salmeterol in asthmatics suboptimally controlled on inhaled steroids. American Journal of Respiratory and Critical Care Medicine 2000; Vol. 161, issue Suppl 3:A198.
Dempsey 2000b {published data only}
- Dempsey OJ, Wilson AM, Sims EJ, Mistry C, Lipworth BJ. Additive bronchoprotective and bronchodilator effects with single doses of salmeterol and montelukast in asthmatic patients receiving inhaled corticosteroids. Chest 2000;117(4):950‐3. [DOI] [PubMed] [Google Scholar]
Dente 2001a {published data only}
- Dente FL, Scuotri L, Bacci E, Franco A, Giannini D, Taccola M, et al. Effects of combined treatment ‐ fluticasone plus salmeterol ‐ on allergen‐induced asthmatic responses. American Journal of Respiratory and Critical Care Medicine. 2001; Vol. 463, issue Suppl 5:A419.
Dente 2001b {published data only}
- Dente FL, Scuotri L, Bacci E, DeSanctis M, Franco A, Giannini D, et al. Combined treatment with fluticasone plus salmeterol protects against allergen‐induced asthmatic responses better than each drug alone. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:349s.
Dicpinigaitis 2002 {published data only}
- Dicpinigaitis PV, Dobkin JB, Reichel J. Antitussive effect of the leukotriene receptor antagonist zafirlukast in subjects with cough‐variant asthma. Journal of Asthma 2002;39(4):291‐7. [DOI] [PubMed] [Google Scholar]
Didier 1997 {published data only}
- Didier A, Campos Oriola R. A two‐month comparison of salmeterol/beclomethasone and slow‐release terbutaline/budesonide in moderate asthma management. Clinical Drug Investigation 1997;14(1):1‐11. [Google Scholar]
Djordjevic 1999 {published data only}
- Djordjevic D, Zickovic D, Stankovic I, Pejcic T, Ducic J, Rancic M, et al. Comparative study of three months treatment in combination of salmeterol and beclomethasone dipropionate (BDP) with doubling the dose of BDP in mild asthma. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999:p849.
Dorinsky 2001a {published data only}
- Dorinsky PM, Kalberg C, Pepsin P, Emmett A, Rickard K. Greater onset of improvement in clinical efficacy measures with first line use of the fluticasone/salmeterol combination product compared to montelukast. Annual Thoracic Society 97th International Conference; San Francisco CA May 18‐23. 2001.
Dorinsky 2001b {published data only}
- Dorinsky P, Kalberg C, Pepsin P, Emmett A, Bowers B, Rickard K. The fluticasone/salmeterol combination product is superior to montelukast as first‐line asthma control. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:263s.
Eliraz 2001 {published data only}
- Eliraz A, Ramirez‐Rivera A, Ferranti P, Holzer R, Garcia JM, Turcotte C, et al. Similar efficacy following four weeks treatment of asthmatics with formoterol 12 mcg BD delivered by two different dry powder inhalers; differences in inhaler handling. International Journal of Clinical Practice 2001;55(3):164‐70. [PubMed] [Google Scholar]
Eliraz 2002a {published data only}
- Eliraz A, Fritscher CC, Perez CMR, Boonsawat W, Nang AN, Bardin P, et al. Symbicort® (budesonide/formoterol) achieves more rapid control of asthma that fluticasone in patients with mild asthma. American Journal of Respiratory & Critical Care Medicine 2002; Vol. 165, issue Suppl 8:A567.
Eliraz 2002b {published data only}
- Eliraz A, Fritscher CC, Perez CMR, Boonsawat W, Nang AN, Bardin P, et al. Budesonide and formoterol in a single inhaler quickly gains asthma control compared with fluticasone propionate in mild asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:48s.
Ericsson 2001a {published data only}
- Ericsson K, Bantje TA, Huber H, Borg S. Symbicort® Turbuhaler® is more cost effective than fluticasone Diskus™ in the treatment of asthma. Annual Thoracic Society 97th International Conference; San Francisco CA , May 18‐23. 2001.
Ericsson2001b {published data only}
- Ericsson K, Bantje TA, Huber R, Borg S, Anderson F. Cost‐effectiveness of budesonide and formoterol in a single inhaler compared to fluticasone in the treatment of asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:157s.
Everden 2002 {published data only}
- Everden P, Lloyd A, Hutchinson J, Plumb J. Cost‐effectiveness of eformoterol Turbohaler(R) versus salmeterol Accuhaler(R) in children with symptomatic asthma. Respiratory Medicine 2002;96(4):250‐8. [DOI] [PubMed] [Google Scholar]
Faurschou 1994 {published data only}
- Faurschou P, Engel AM, Haanaes OC. Salmeterol in two different doses in the treatment of nocturnal bronchial asthma poorly controlled by other therapies. Allergy 1994;49(40):827‐32. [DOI] [PubMed] [Google Scholar]
Faurschou 1996 {published data only}
- Faurschou P, Steffensen I, Jacques L. Effect of addition of inhaled salmeterol to the treatment of moderate‐to‐severe asthmatics uncontrolled on high‐dose inhaled steroids. European Respiratory Journal 1996;9(9):1885‐90. [DOI] [PubMed] [Google Scholar]
Fish 2000 {published data only}
- Fish J, Boone R, Emmett A, Yancey S, Knobil K, Rickard K. Salmeterol added to inhaled corticosteroids (ICS) provides greater asthma control compared to montelukast. American Journal of Respiratory & Critical Care Medicine 2000; Vol. 161, issue Suppl 3:A203.
Fish 2001 {published data only}
- Fish JE, Israel E, Murray JJ, Emmett A, Boone R, Yancey SW, et al. Salmeterol powder provides significantly better benefit than montelukast in asthmatic patients receiving concomitant inhaled corticosteroid therapy. Chest 2001;120(2):423‐30. [DOI] [PubMed] [Google Scholar]
Fitzpatrick 1990 {published data only}
- Fitzpatrick MF, Mackay T, Driver H, Douglas NJ. Salmeterol in nocturnal asthma: a double‐blind, placebo controlled trial of long‐acting inhaled beta2 agonist. BMJ 1990;301:1365‐8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Fowler 2002 {published data only}
- Fowler SJ, Currie PC, Lipworth BJ. Step down therapy with low dose fluticasone‐salmeterol combination or medium dose hydrofluoroalkane 134a‐beclomethasone alone. Journal of Allergy & Clinical Immunology 2002;109(6):929‐35. [DOI] [PubMed] [Google Scholar]
Fuglsang 1995 {published data only}
- Fuglsang G, Agertoft L, Vikre‐Jorgensen J, Pedersen S. Influence of budesonide on the response to inhaled terbutaline in children with mild asthma. Pediatric Allergy and Immunology 1995;6(2):103‐8. [DOI] [PubMed] [Google Scholar]
Gabrijelcic 2004 {published data only}
- Gabrijelcic J, Casas A, Rabinovich RA, Roca J, Barbera JA, Chung KF, et al. Formoterol protects against platelet‐activating factor‐induced effects in asthma. European Respiratory Journal. 2004;23(1):71‐5. [DOI] [PubMed] [Google Scholar]
Giannini 1996 {published data only}
- Giannini D, Carletti A, Dente FL, Testi R, Bacci D, Bancalari L, et al. Effect of inhaled beclomethasone dipropionate (BDP) on tolerance to salmeterol (S) in allergen induced bronchoconstriction. European Respiratory Journal 1996; Vol. 9, issue Suppl 23:272s.
Giannini 1998a {published data only}
- Giannini D, Franco A, Bacci E, Conti I, Dente FL, Kotopulos C, et al. Long‐term treatment with salmeterol and inhaled corticosteroids does not induce tolerance to the protective effect of salmeterol on allergen challenge. American Journal of Respiratory & Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A414.
Giannini 1998b {published data only}
- Giannini D, Franco A, Bacci E, Conti I, Dente FL, Kotopulos C, et al. One‐week regular treatment with salmeterol induces tolerance to the protective effect of salmeterol on allergen challenge only in subjects not regularly treated with salmeterol and inhaled corticosteroid. European Respiratory Journal 1998;12(Suppl 28):156s. [Google Scholar]
Giannini 1999 {published data only}
- Giannini D, Bacci E, Dente FL, Franco A, Vagaggini B, Testi R, et al. Inhaled beclomethasone dipropionate reverts tolerance to the protective effect of salmeterol on allergen challenge. Chest 1999;115(3):629‐34. [DOI] [PubMed] [Google Scholar]
Giannini 2000 {published data only}
- Giannini D, Franco A, Bacci E, Dente FL, Taccola M, Vagaggini B, et al. The protective effect of salbutamol inhaled using different devices on methacholine bronchoconstriction. Chest 2000;117(5):1319‐23. [DOI] [PubMed] [Google Scholar]
Giannini 2001 {published data only}
- Giannini D, Tonelli M, Franco A, Bacci E, Conti I, Dente FL, et al. Tolerance to the protective effect of salmeterol on allergen challenge in mild untreated asthmatics and in moderate asthmatics on inhaled corticosteroid treatment.. European Respiratory Journal. 2001; Vol. 18, issue Suppl 33:103s.
Giannini 2002a {published data only}
- Giannini D, Tonelli M, Franco A, Bacci E, Conti I, Dente FL, et al. Tolerance to the protective effect of salmeterol + fluticasone combination (50/250 µg) on allergen challenge in mild untreated asthmatics. American Journal of Respiratory and Critical Care Medicine 2002; Vol. 165, issue Suppl 8:A566.
Giannini 2002b {published data only}
- Giannini D, Franco A, Bacci E, Conti I, Dente FL, Kotopulos C, et al. One‐week regular treatment with salmeterol induces tolerance to the protective effect of salmeterol on allergen challenge only in subjects not regularly treated with salmeterol and inhaled corticosteroid. European Respiratory Journal 1998;12(Suppl 28):156s. [Google Scholar]
Gizycki 2000 {published data only}
- Gizycki MJ, Venge P, Dahl R, Jeffery PK. Comparison of the effects of six weeks treatment with fluticasone or salmeterol on the late phase response (LPR) in mild asthma ‐ a bronchial biopsy study. American Journal of Respiratory and Critical Care Medicine 2000; Vol. 161, issue Suppl 3:A203.
Gold 2001 {published data only}
- Gold M, Jõgi R, Mulder PGH, Akveld MLM. Salmeterol/fluticasone propionate combination 50/100µg bid is more effective than fluticasone propionate 100µg bid plus montelukast 10 mg once daily in reducing exacerbations. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:262s.
Green 2002 {published data only}
- Green RH, Brightling CE, McKenna S, Hargadon B, Parker D, Wardlaw AJ, et al. A placebo controlled comparison of formoterol, montelukast or higher dose of inhaled corticosteroids in subjects with symptomatic asthma despite treatment with low dose inhaled corticosteroids. Thorax 2002; Vol. 57, issue Suppl III:iii 11.
Greening 1994 {published data only}
- Greening AP, Ind PW, Northfield M, Shaw G. Added salmeterol versus higher‐dose corticosteroid in asthma patients with symptoms on existing inhaled corticosteroid. Lancet 1994;344:219‐24. [DOI] [PubMed] [Google Scholar]
Grosclaude 2003 {published data only}
- Grosclaude M, Cerruti JL, Delannay B, Herent M, Spilthooren F, Desfougeres JL. The fixed fluticasone/salmeterol gives better control of asthma than the association of beclomethasone dipropionate‐montelukast. Allergie et Immunologie 2003;35(9):356‐62. [PubMed] [Google Scholar]
Grzelewska‐Rzymowska 2003 {published data only}
- Grzelewska‐Rzymowska I, Malolepszy J, Molina M, Sladek K, Zarkovic J, Siergiejko Z. Equivalent asthma control and systemic safety of inhaled budesonide delivered via HFA‐134a or CFC propellant in a broad range of doses. Respiratory Medicine 2003;97(Suppl D):S10‐S19. [DOI] [PubMed] [Google Scholar]
Gustafsson 1994 {published data only}
- Gustafsson PM, Von BA, Jenkins MM. Salmeterol 50 mug twice daily in the treatment of mild‐to‐moderate asthma in childhood ‐ a comparison of two inhalation devices. European Journal of Clinical Research 1994;5:63‐73. [Google Scholar]
Hasani 2003 {published data only}
- Hasani A, Toms N, O'Connor J, Dilworth JP, Agnew JE. Effect of salmeterol xinafoate on lung mucociliary clearance in patients with asthma. Respiratory Medicine 2003;97(6):667‐71. [DOI] [PubMed] [Google Scholar]
Haughney 2002 {published data only}
- Haughney J, Price D, Rosen JP, Kennelly J. Adjustable maintenance treatment with budesonide/formoterol combination rapidly improves and maintains quality of life in asthma patients. European Respiratory Society Annual Congress. 2002:P379.
Heuck 1999 {published data only}
- Heuck C, Heickendorff L, Wolthers OD, Sygehus S. Short term growth and collagen turnover in asthmatics treated inhaled formoterol and budesonide. European Respiratory Society; Oct 9‐13; Madrid, Spain. 1999:364.
Heuck 2000 {published data only}
- Heuck C, Heickendorff L, Wolthers OD. A randomized controlled trial of short term growth and collagen turnover in asthmatics with inhaled formoterol and budesonide. Archives of Disease in Childhood 2000;83:334‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hyland 1995 {published data only}
- Hyland ME, Crocker GR. Validation of an asthma quality of life diary in a clinical trial. Thorax 1995;50(7):724‐30. [DOI] [PMC free article] [PubMed] [Google Scholar]
Ind 2002a {published data only}
- Ind P, Haughney J, Price D, Rosen JP, Kennelly J. Managed adjustable dosing of budesonide/formoterol combination provides equivalent asthma control to fixed dosing at a lower overall dose. European Respiratory Society Annual Congress. 2002:P2450.
Ind 2002b {published data only}
- Ind PW, Villasante C, Shiner RJ, Pietinalho A, Boszormenyi NG, Soliman S, et al. Safety of formoterol by Turbuhaler as reliever medication compared with terbutaline in moderate asthma. European Respiratory Journal 2002;20(4):859‐66. [DOI] [PubMed] [Google Scholar]
Ind 2003a {published data only}
- Haughney J, Price D, Rosen JP, Kennelly J. Adjustable maintenance treatment with budesonide/formoterol combination rapidly improves and maintains quality of life in asthma patients. European Respiratory Society Annual Congress. 2002:P379.
- Ind PW, Price D, Haughney J, Rosen JP. Adjustable dosing with budesonide/formoterol in a single inhaler (Symbicort(r)) provides similarly effective treatment of asthma compared with fixed dosing but at a lower overall dose. American Thoracic Society 99th International Conference. 2003:D034 Poster C38.
Isabelle 2001 {published data only}
- Isabelle P, Bjamer D, Neuparth N, Desfougeres JL. Efficacy and safety of salmeterol/fluticasone combination 50/100mug bd via two different powder devices in children. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Jeffery 2002 {published data only}
- Jeffery PK, Venge P, Gizycki MJ, Egerod I, Dahl R, Faurschou P. Effects of salmeterol on mucosal inflammation in asthma: a placebo‐controlled study. European Respiratory Journal 2002;20(6):1378‐85. [DOI] [PubMed] [Google Scholar]
Jenkins 1995 {published data only}
- Jenkins M. Clinical evaluation of CFC‐free metered dose inhalers. Journal of Aerosol Medicine: Deposition, Clearance, and Effects in the Lung 1995;8(Suppl 1):s41‐s47. [DOI] [PubMed] [Google Scholar]
Jenkins 2000 {published data only}
- Becker I, Kielborn A, Price MJ, Volmer T, Lloyd AC. Cost‐effectiveness of salmeterol/fluticasone combination product and budesonide in asthma patients in Germany. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999:854.
- Jenkins C, Woolcock A, James M. Superior overall control of moderate to severe asthma with salmeterol/fluticasone propionate (FP) combination (50/250 mcg bd) compared with three‐fold‐higher dose of budesonide (800mcg bd). European Respiratory Journal. 2000; Vol. 16, issue Suppl 31:456s.
- Jenkins C, Woolcock AJ, Saarelainen P, Lundbaack B, James MH. Salmeterol/fluticasone propionate combination therapy 50/250mcgs twice daily is more effective than budesonide 800 twice daily in treating moderate to severe asthma. Respiratory Medicine 2000;94:715‐23. [DOI] [PubMed] [Google Scholar]
- Lundback B, Jenkins C, Price MJ, Thwaites RM. Cost‐effectiveness of salmeterol/fluticasone propionate combination product 50/250 microgram twice daily and budesonide 800 microgram twice daily in the treatment of adults and adolescents with asthma. Respiratory Medicine 2000;94(7):724‐32. [DOI] [PubMed] [Google Scholar]
- Lundback B, Ronmark E, Jonsson AC, et al. Treatment effectiveness and exacerbations during one year with Seretide compared to fluticasone propionate and salmeterol in mild to moderate asthma. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:176s.
- Parnaby A, Lloyd A, Browning D, McCarthy TP. A comparison of the cost‐effectiveness of salmeterol/fluticasone combination inhaler and budesonide in the management of asthma. Thorax 2000; Vol. 55, issue Suppl 3:A64.
Jenkins 2002 {published data only}
- Jenkins C. Combination therapy with fluticasone and salmeterol for symptomatic asthma produces similar benefits when given by Accuhaler in a single or two separate devices over 24 weeks. Respirology 2002; Vol. 7, issue Suppl:A20. [P22]
Johansson 2001 {published data only}
- Johansson G, McIvor RA, D'Ambrosio FP, Gratziou C, James MH. Comparison of salmeterol/fluticasone propionate combination with budesonide in patients with mild‐to‐moderate asthma. Clinical Drug Investigation 2001;21(9):633‐42. [Google Scholar]
Jones 1994 {published data only}
- Jones KP. Salmeterol xinafoate in the treatment of mild to moderate asthma in primary care. Thorax 1994;49:971‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Juniper 1995 {published data only}
- Juniper EF, Johnston PR, Borkhoff CM, Guyatt GH, Boulet LP, Haukioja A. Quality of life in asthma clinical trials: comparison of salmeterol and salbutamol. American Journal of Respiratory & Critical Care Medicine 1995;151(1):66‐70. [DOI] [PubMed] [Google Scholar]
Juniper 1999 {published data only}
- Juniper EF, Svenson K, O'Byrne PM, Barnes PJ, Bauer CA, Lofdahl CG, et al. Asthma quality of life during 1 year of treatment with budesonide with or without formoterol. European Respiratory Journal 1999;14:1038‐43. [DOI] [PubMed] [Google Scholar]
Kaik 2002 {published data only}
- Kaik G, Kottakis I, Anagnostopoulou O, Sichletidis L, Bachlitzanakis N, D'Amato M, et al. Sequential flexible therapy with formoterol (Foradil(r)) plus budesonide (Miflonide(r)) versus a fixed combination of salmeterol and fluticasone (Seretide(r)) in asthma self‐management. European Respiratory Society Annual Congress. 2002.
Kalberg 1998 {published data only}
- Kalberg CJ, Nelson H, Yancey S, Petrocella V, Emmett AH, Rickard KA, et al. A comparison of added salmeterol versus increased‐dose fluticasone in patients symptomatic on low‐dose fluticasone [Abstract]. Journal of Allergy & Clinical Immunology 1998;101(Suppl):S6. [Google Scholar]
- Knobil K, Kalberg C, Emmett A, Rickard K. Adding salmeterol is more effective than increasing the dose of fluticasone for patients with asthma who are symptomatic on low dose fluticasone. European Respiratory Journal. 1998; Vol. 12, issue Suppl 29:19s, P160.
Kalra 1996 {published data only}
- Kalra S, Swystun VA, Bhagat R, Cockcroft DW. Inhaled corticosteroids do not prevent the development of tolerance to the bronchoprotective effect of salmeterol. Chest 1996;109(4):953‐6. [DOI] [PubMed] [Google Scholar]
Karaman 2007 {published data only (unpublished sought but not used)}
- Karaman O, Arli O, Uzuner N, Islekel H, Babayigit A, Olmez D, et al. The effectiveness of asthma therapy alternatives and evaluating the effectiveness of asthma therapy by interleukin‐13 and interferon gamma levels in children. Allergy & Asthma Proceedings 2007;28(2):204‐9. [DOI] [PubMed] [Google Scholar]
Kardos 2001 {published data only}
- Kardos P, Bruggenjurgen B, Martin A, Meyer‐Sabellek W, Richter K, Vogelmeier C, et al. Treatment of bronchial asthma using a new adjustable combination treatment plan: Asthma Control Plan (ATACO). Pneumologie 2001;55(5):253‐7. [DOI] [PubMed] [Google Scholar]
Keith 2001 {published data only}
- Keith P, D' Urzo A, Stepner N, et al. Fluticasone/salmeterol combination (FSC) is safe and provides effective long‐term (52 week) control in the management of patients with persistent asthma (PA). European Respiratory Journal 2001; Vol. 18, issue Suppl 33:176s.
Kelsen 1999 {published data only}
- Kelsen SG, Church NL, Gillman SA, Lanier BQ, Emmett AH, Rickard KA, et al. Salmeterol added to inhaled corticosteroids therapy is superior to doubling the dose of inhaled corticosteroids: a randomized clinical trial. Journal of Asthma 1999;36(8):703‐15. [DOI] [PubMed] [Google Scholar]
Kerwin 2001 {published data only}
- Kerwin E, Srebro S, Church N, Emmett A, Rickard K, Knobil K. Salmeterol added to low‐dose fluticasone propionate (fp) improves pulmonary function and albuterol use more rapidly than adding montelukast. Annals of Allergy, Asthma & Immunology 2001;86:99. [Google Scholar]
Ketchell 2002 {published data only}
- Ketchell RI, Jensen MW, Spina D, O'Connor BJ. Dose‐related effects of formoterol on airway responsiveness to adenosine 5'‐monophosphate and histamine. European Respiratory Journal 2002;19(4):611‐16. [DOI] [PubMed] [Google Scholar]
Kidney 1995 {published data only}
Kips 2000 {published data only}
- Kips JC, O'Connor BJ, Inman MD, Svenson K, Pauwels RA, O'Byrne PM. A long‐term study of the antiinflammatory effect of low‐dose budesonide plus formoterol versus high‐dose budesonide in asthma. American Journal Respiratory Critical Care Medicine 2000;161:996‐1001. [DOI] [PubMed] [Google Scholar]
Kirby 2000 {published data only}
- Kirby S, Falcoz C, Daniel MJ, Milleri S, Squassante L, Ziviani L, et al. Salmeterol and fluticasone propionate given as a combination: lack of systemic pharmacodynamic and pharmacokinetic interactions. European Journal of Clinical Pharmacology 2000;56(11):781‐91. [DOI] [PubMed] [Google Scholar]
Knobil 2000 {published data only}
- Knobil K, Dorinsky P, Yancey S, Emmett A, Rickard K. Salmeterol is superior to montelukast as add‐on therapy to inhaled corticosteroids. European Respiratory Journal 2000; Vol. 16, issue Suppl 31:457s.
Knorr 2001 {published data only}
- Knorr B, Franchi LM, Bisgaard H, Vermeulen JH, LeSouef P, Santanello N, et al. Montelukast, a leukotriene receptor antagonist, for the treatment of persistent asthma in children aged 2 to 5 years. Pediatrics 2001;108(3):E48. [DOI] [PubMed] [Google Scholar]
Kraft 2003 {published data only}
- Kraft M, Martin RJ, Lazarus SC, Lemanske RF, Szefler SJ. Airway tissue mast cells in persistent asthma: predictor of treatment failure when patients discontinue inhaled corticosteroids. Chest 2003;124(1):42‐50. [DOI] [PubMed] [Google Scholar]
LaForce 1994 {published data only}
- LaForce C, Liddle RF, Yancey SW. Salmeterol response in asthmatic patients using inhaled corticosteroids and in those not using inhaled corticosteroids. Annals of Allergy. 1994; Vol. 72:100.
Lai 1995 {published data only}
- Lai CKW, Chan CHS, Ho SS, Hui ACF, Lai KN. Inhaled salmeterol and albuterol in asthmatic patients receiving high‐dose inhaled corticosteroids. Chest 1995;108:36‐40. [DOI] [PubMed] [Google Scholar]
Lalloo 2000 {published data only}
- Lalloo UG, Bantje TA, Kozma D, Krofta K, Ankerst J, Johansen B, et al. Low‐dose Symbicort (budesonide/formoterol) is more effective than double‐dose inhaled corticosteroid in mild asthma. Allergy Clinical Immunology Int 2000; Vol. Suppl 2:122.
Lalloo 2001a {published data only}
- Lalloo UG, Malolepszy J, Kozma D, Krofta K, Ankerst J, Johansen B, et al. Symbicort® (budesonide and formoterol in a single inhaler) is more effective than increasing the dose of inhaled corticosteroids in mild asthma. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Lalloo 2001b {published data only}
- Lalloo UG, Malolepszy J, Kozma D, Krofta K, Ankerst J, Johansen B, et al. Budesonide and formoterol in a single inhaler is more effective than a higher dose of inhaled corticosteroid in mild‐moderate persistent asthma. European Respiratory Journal 2001;18(Suppl 33):159s. [Google Scholar]
Lalloo 2001c {published data only}
- Lalloo UG, Malolepsky J, Kozma D, Krofta K, Ankerst J, Johansen B, et al. Budesonide and formoterol in a single inhaler controls exacerbations more effectively than a higher dose of inhaled corticosteroids alone, in mild‐moderate persistent asthma. European Respiratory Journal 2001;18(Suppl 33):43s. [Google Scholar]
Lalloo 2003 {published data only}
- Lalloo UG, Malolepszy D, Kozma K, Krofta J, Ankerst B, Johansen NC, et al. Budesonide and formoterol in a single inhaler improves asthma control compared with increasing the dose of corticosteroid in adults with mild to moderate asthma. Chest 2003;123(5):1480‐7. [DOI] [PubMed] [Google Scholar]
Lange 2001 {published data only}
- Lange ML, House KW, Scott CA, Shah TP, Akveld MLM. The salmeterol/fluticasone propionate combination 50/100µg bid is effective as initial maintenance therapy in mild and moderate asthmatics. European Respiratory Journal. 2001; Vol. 18, issue Suppl 33:176s.
Lazarus 2001 {published data only}
- Lazarus SC, Boushey HA, Fahy JV, Chinchilli VM, Lemanske RF Jr, Sorkness CA, et al. Long‐acting beta2 agonist monotherapy vs continued therapy with inhaled corticosteroids in patients with persistent asthma: a randomized controlled trial. JAMA 2001;285(20):2583‐93. [DOI] [PubMed] [Google Scholar]
Lee 2003 {published data only}
- Lee DK, Jackson CM, Currie GP, Cockburn WJ, Lipworth BJ. Comparison of combination inhalers vs inhaled corticosteroids alone in moderate persistent asthma. British Journal of Clinical Pharmacology 2003;56(5):494‐500. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lemanske 2001 {published data only}
- Lemanske RF Jr, Sorkness CA, Mauger EA, Lazarus SC, Boushey HA, Fahy JV, et al. Inhaled corticosteroid reduction and elimination in patients with persistent asthma receiving salmeterol: a randomized controlled trial. JAMA 2001;285(20):2594‐603. [DOI] [PubMed] [Google Scholar]
Lenney 1995 {published data only}
- Lenney W, Pedersen S, Boner AL, Ebbutt A, Jenkins MM. Efficacy and safety of salmeterol in childhood asthma. European Journal of Pediatrics 1995;154:983‐90. [DOI] [PubMed] [Google Scholar]
Leuppi 2003 {published data only}
- Leuppi FD, Salzberg M, Meyer L, Bucher SE, Nief M, Brutsche MH, et al. An individualized, adjustable maintenance regimen of budesonide/formoterol provides effective asthma symptom control at a lower overall dose than fixed dosing. Swiss Medical Weekly 2003;133(21‐2):302‐9. [DOI] [PubMed] [Google Scholar]
LHSRG 2000 {published data only}
- The Lung Health Study Research Group. Effect of inhaled triamcinolone on the decline in pulmonary function in chronic obstructive pulmonary disease. New England Journal of Medicine 2000;343(26):1902‐9. [DOI] [PubMed] [Google Scholar]
Lindqvist 2001 {published data only}
- Lindqvist AE, Karjalainen EM, Laitinen LA, Kava T, Altraja A, Pulkkinen M, et al. Salmeterol (SLM), fluticasone propionate (FP) or disodium cromoglycate (DSCG) in the treatment of newly diagnosed asthma. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Lindqvist 2003 {published data only}
- Lindqvist A, Karjalainen EM, Laitinen L A, Kava T, Altraja A, Pulkkinen M, et al. Salmeterol resolves airway obstruction but does not possess anti‐eosinophil efficacy in newly diagnosed asthma: a randomized, double‐blind, parallel group biopsy study comparing the effects of salmeterol, fluticasone propionate, and disodium cromoglycate . Journal of Allergy & Clinical Immunology 2003;112(1):23‐8. [DOI] [PubMed] [Google Scholar]
Lipworth {published data only}
- Lipworth BJ. Does genetic polymorphism of b2‐adrenoceptors determine airway sensitivity to regular long‐acting b2‐agonist therapy?. National Research Register (UK). [N0405016375]
Lipworth 1996 {published data only}
- Lipworth BJ, Hall IP, Aziz I, Tan KS, Wheatley A. Beta2‐adrenoceptor polymorphism and bronchoprotective sensitivity with regular short‐ and long‐acting beta2‐agonist therapy. Clinical Science 1996;3:253‐9. [PubMed] [Google Scholar]
Lipworth 1998 {published data only}
- Lipworth B, Tan S, Devlin M, Aiken T, Baker R, Hendrick D. Effects of treatment with formoterol on bronchoprotection against methacholine. American Journal of Medicine 1998;104(5):431‐8. [DOI] [PubMed] [Google Scholar]
Lipworth 1999a {published data only}
- Lipworth BJ, Aziz I. A high dose of albuterol does not overcome bronchoprotective subsensitivity in asthmatic subjects receiving regular salmeterol or formoterol. Journal of Allergy & Clinical Immunology 1999;103(1 part 1):88‐92. [DOI] [PubMed] [Google Scholar]
Lipworth 1999b {published data only}
- Lipworth BJ, Hall IP, Aziz I, Tan KS, Wheatley A. Beta2‐adrenoceptor polymorphism and bronchoprotective sensitivity with regular short‐ and long‐acting beta2‐agonist therapy. Clinical Science 96;3:253‐9. [PubMed] [Google Scholar]
Lipworth 2000a {published data only}
- Lipworth BJ, Dempsey OJ, Aziz I. Functional antagonism with formoterol and salmeterol in asthmatic patients expressing the homozygous glycine‐16 beta(2)‐adrenoceptor polymorphism. Chest 2000;118(2):321‐8. [DOI] [PubMed] [Google Scholar]
Lipworth 2000b {published data only}
- Lipworth BJ, Dempsey OJ, Aziz I, Wilson AM. Effects of adding a leukotriene antagonist or a long‐acting beta(2)‐agonist in asthmatic patients with the glycine‐16 beta(2)‐adrenoceptor genotype. American Journal of Medicine 2000;109(2):114‐21. [DOI] [PubMed] [Google Scholar]
Lockey 1999 {published data only}
- Lockey RF, DuBuske LM, Friedman B, Petrocella V, Cox F, Rickard K. Nocturnal asthma: effect of salmeterol on quality of life and clinical outcomes. Chest 1999;115(3):666‐73. [DOI] [PubMed] [Google Scholar]
Lowhagen 2002 {published data only}
- Lowhagen O, Wever AM, Lusuardi M, Moscato G, Backer WA, Gandola L, at al. The inflammatory marker serum eosinophil cationic protein (ECP) compared with PEF as a tool to decide inhaled corticosteroid dose in asthmatic patients. Respiratory Medicine 2002;96(2):95‐101. [DOI] [PubMed] [Google Scholar]
Lundback 2002 {published data only}
- Lundback B, Ronmark E, Jonsson AC, Larsson LG, Petavy F, James MH. Airway hyperresponsiveness and treatment effectiveness during a one year study of the combination of salmeterol and fluticasone propionate (FP) compared with FP and salmeterol alone in mild to moderate asthma. European Respiratory Society Annual Congress. 2002:P2397.
Lyseng‐Williamson 2003 {published data only}
- Lyseng‐Williamson KA, Plosker GL. Inhaled salmeterol/fluticasone propionate combination: a pharmacoeconomic review of its use in the management of asthma. Pharmacoeconomics 2003;21(13):951‐89. [DOI] [PubMed] [Google Scholar]
Lötvall 2002 {published data only}
- Lötvall J, Woude HJ, Palmqvist M, Arvidsson P, Beckman O, Boorsma M, et al. More rapid onset of action of budesonide/formoterol (Symbicort®) than salmeterol/fluticasone (Seretide™). American Journal of Respiratory & Critical Care Medicine 2002; Vol. 165, issue Suppl 8:A567.
Magadle 2001 {published data only}
- Magadle R, Berar‐Yanay N, Weiner P. Long‐acting bronchodilators in premenstrual exacerbation of asthma. Respiratory Medicine 2001;95(9):740‐3. [DOI] [PubMed] [Google Scholar]
Malmqvist‐Granlund 2000 {published data only}
- Malmqvist‐Granlund K, Asking L, Lindbald T, Rollwage U, Steckel H. An in vitro comparison of budesonide/formoterol and fluticasone/salmeterol in dry powder inhalers. European Respiratory Journal 2000; Vol. 16, issue Suppl 31:455s.
Malolepszy 2001 {published data only}
- Malolepszy J, Boszormenyi Nagy G, Selroos O, Larsson P, Brander R. Safety of formoterol turbuhaler at cumulative dose of 90 mug in patients with acute bronchial obstruction. European Respiratory Journal 2001;18(6):928‐34. [DOI] [PubMed] [Google Scholar]
Malolepszy 2002 {published data only}
- Malolepszy J. Efficacy and tolerability of oral theophylline slow‐release versus inhaled formoterol in moderate asthma poorly controlled on low‐dose steroids. Atemwegs‐ und Lungenkrankheiten 2002;28(2):78‐87. [Google Scholar]
Martinat 2003 {published data only}
- Martinat Y, Desfougeres JL. Fixed‐dose fluticasone‐salmeterol combination: at least effective and better tolerated than open‐dose combinations. Revue De Pneumologie Clinique 2003;59(3):139‐48. [PubMed] [Google Scholar]
Matz 2001 {published data only}
- Matz J, Emmett A, Rickard K, Kalberg C. Addition of salmeterol to low‐dose fluticasone versus higher‐dose fluticasone: an analysis of asthma exacerbations. Journal of Allergy and Clinical Immunology 2001;107(5):783‐9. [DOI] [PubMed] [Google Scholar]
McCarthy 2000 {published data only}
- McCarthy TP, Boone R, Yancey S, Rickard K. Salmeterol compared to montelukast as adjunctive therapy to inhaled corticosteroids. Thorax 2000; Vol. 55, issue Suppl 3:A63.
McCarthy 2001a {published data only}
- McCarthy TP, Russell D, Baxter LE, et al. A comparison of the efficacy of salmeterol/fluticasone propionate combination (SF) with beclometasone dipropionate (BDP) delivered via metered dose inhaler (MDI) in patients not well controlled on bronchodilators alone. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:53‐54s.
McCarthy 2001b {published data only}
- McCarthy TP, Greening AP, Holgate SK, Whitehead C, Rice L. The efficacy of salmeterol/fluticasone propionate combination (SFC) metered dose inhaler compared with beclomethasone dipropionate (BDP) in patients not well controlled at step 1 of the British guidelines on asthma management (BGAM). Thorax 2001; Vol. 56, issue Suppl 3:iii62.
McCarthy 2002 {published data only}
- McCarthy TP, Grening AP, Holgate SK, Whitehead C, Rice L. Salmeterol/fluticasone propionate combination (SFC) is more effective that beclometasone dipropionate (BDP) in patients not well controlled on bronchodilators alone. American Journal of Respiratory & Critical Care Medicine 2002; Vol. 165, issue Suppl 8:A566.
McCarthy 2003 {published data only}
- McCarthy TP, Woodcock A A, Pavord I D, Allen DJ, Parker D, Rice L. A comparison of the anti‐inflammatory and clinical effects of salmeterol 25mcg/fluticasone propionate 50mcg combination (SFC 50) with fluticasone propionate (FP) plus montelukast (M) in patients with mild to moderate asthma. American Thoracic Society 99th International Conference. 2003:B036 Poster H89.
Mcivor 1998 {published data only}
- Mcivor RA, Pizzichini E, Turner MO, Hussack P, Hargreave FE, Sears MR. Potential masking effects of salmeterol on airway inflammation in asthma. American Journal of Respiratory & Critical Care Medicine 1998;158(3):924‐30. [DOI] [PubMed] [Google Scholar]
Michel 2000 {published data only}
- Michel O, Olbrecht J, Moulard D, Sergysels R. Effect of anti‐asthmatic drugs on the response to inhaled endotoxin. Annals of Allergy, Asthma, & Immunology 2000;85(4):305‐10. [DOI] [PubMed] [Google Scholar]
Midgren 1992 {published data only}
- Midgren‐B, Melander‐B, Persson‐G. Formoterol, a new long‐acting beta 2 agonist, inhaled twice daily, in stable asthmatic subjects. Chest 1992;101(4):1019‐22. [DOI] [PubMed] [Google Scholar]
Miraglia del Giudice 2007 {published data only}
- Miraglia del Giudice M, Capristo M, Amelio R, Rocco A, Fusco N, Brunese FP. Combined fluticasone propionate/salmeterol (Diskus) vs fluticasone propionate (Diskus) in the control of persistent asthma in children. American Thoracic Society 99th International Conference. 2003:A117 Poster D78.
- Miraglia del Giudice M, Piacentini GL, Capasso M, Capristo C, Maiello N, Boner AL, et al. Formoterol, montelukast, and budesonide in asthmatic children: effect on lung function and exhaled nitric oxide. Respiratory Medicine 2007;101(8):1809‐13. [DOI] [PubMed] [Google Scholar]
Mitchell 2000 {published data only}
- Mitchell C, Jenkins C, Scicchitano R, Rubinfeld A. Adding formoterol is more effective and safer than doubling the dose of inhaled steroids in moderately severe asthma. American Journal of Respiratory & Critical Care Medicine 2000; Vol. 161, issue Suppl 3:A197.
Mitchell 2003 {published data only}
- Mitchell C, Jenkins C, Scicchitano R, Rubinfeld A, Kottakis J. Formoterol (Foradil) and medium‐high doses of inhaled corticosteroids are more effective than high doses of corticosteroids in moderate‐to‐severe asthma. Pulmonary Pharmacology & Therapeutics 2003;16(5):229‐306. [DOI] [PubMed] [Google Scholar]
Murray 1998 {published data only}
- Murray JJ, Hagaman DD, Dworski R, Keane B, Sheller JR. Inhibition by salmeterol and beclomethasone of late phase response to segmental antigen challenge in asthmatics. American Journal of Respiratory & Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A872.
Murray 1999 {published data only}
- Murray JJ, Church NL, Anderson WH, Bernstein DI, Wenzel SE, Emmett A, et al. Concurrent use of salmeterol with inhaled corticosteroids is more effective than inhaled corticosteroid dose increases. Allergy & Asthma Proceedings 1999;20(3):173‐80. [DOI] [PubMed] [Google Scholar]
Nathan 1995 {published data only}
- Nathan RA, Seltzer JM, Kemp JP, Chervinsky P, Alexander WJ, Liddle R, et al. Safety of salmeterol in the maintenance treatment of asthma. Annals of Allergy, Asthma and Immunology 1995;75:243‐8. [PubMed] [Google Scholar]
Nathan 1999a {published data only}
- Nathan RA, Pinnas JL, Schwartz HJ, Grossman J, Yancey SW, Emmett AH, et al. A six‐month, placebo‐controlled comparison of the safety and efficacy of salmeterol or beclomethasone for persistent asthma. Annals of Allergy, Asthma, & Immunology 1999;82:521‐9. [DOI] [PubMed] [Google Scholar]
Nathan 1999b {published data only}
- Nathan R, Woodring A, Baitinger L, Prillaman B, Faris M, House K, et al. The salmeterol/fluticasone propionate Diskus combination decreases the incidence of exacerbations compared to treatment with salmeterol or fluticasone propionate alone. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999:848.
Nathan 2001 {published data only}
- Nathan RA, Mitchell D, Condemi J, Heller A, Schoaf L, Herrle M, et al. Cardiovascular and hypothalamic‐pituitary‐adrenal axis safety of fluticasone propionate/salmeterol HFA MDI in adolescent and adult patients with asthma. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Nelson 1999 {published data only}
- Nelson HS, Berkowitz RB, Tinkelman DA, Emmett AH, Rickard KA, Yancey SW. Lack of sub‐sensitivity to albuterol after treatment with salmeterol in patients with asthma. American Journal of Respiratory and Critical Care Medicine 1999;159(5 part 1):1556‐61. [DOI] [PubMed] [Google Scholar]
Nelson 2000a {published data only}
- Nelson H, Chervinsky P, Greos L, Pelskow W, Baitinger L, Scott C, et al. The salmeterol/fluticasone propionate combination product improves asthma control compared with the individual products in asthmatics treated with prn short‐acting beta2‐agonists alone. American Journal of Respiratory & Critical Care Medicine 2000; Vol. 161, issue Suppl 3:A196.
- Nelson HS, Baitinger L, Scott C, House K, Payne E, Shah T. Salmeterol/fluticasone propionate (50/100µg dose) non‐CFC metered dose inhaler is safe and effective in in patients with asthma using short‐acting ß²‐agonists alone. European Respiratory Journal 2000; Vol. 16, issue 31:53s.
- Nelson HS, Busse WW, Kerwin E, Church N, Emmett A, Rickard K, et al. Fluticasone propionate/salmeterol combination provides more effective asthma control than low‐dose inhaled corticosteroid plus montelukast. Journal of Allergy and Clinical Immunology 2000;106(6):1088‐95. [DOI] [PubMed] [Google Scholar]
Nelson 2000b {published data only}
- Nelson HS, Chervinsky P, Greos L, Pleskow W, Baitinger L, Scott C, et al. The salmeterol/fluticasone propionate combination product improves asthma control compared with the individual products in asthmatics treated with PRN short‐acting beta2‐agonists alone. American Journal of Respiratory & Critical Care Medicine 2000; Vol. 161, issue Suppl 3:A196.
Nelson 2001 {published data only}
- Nelson HS, Nathan RA, Kalberg C, Yancey SW, Rickard KA. Comparison of inhaled salmeterol and oral zafirlukast in asthmatic patients using concomitant inhaled corticosteroids. Medscape General Medicine 2001; Vol. 3, issue 4:3. [PubMed]
Newnham 1995 {published data only}
- Newnham DM, Grove A, McDevitt DG, Lipworth BJ. Subsensitivity of bronchodilator and systemic beta 2 adrenoceptor responses after regular twice daily treatment with eformoterol dry powder in asthmatic patients. Thorax 1995;50(5):497‐504. [DOI] [PMC free article] [PubMed] [Google Scholar]
Nielsen 1999 {published data only}
- Nielsen LP, Pedersen B, Faurschou P, Madsen F, Wilcke JTR, Dahl R. Salmeterol reduces the need for inhaled corticosteroids in steroid‐dependent asthmatics. Respiratory Medicine 1999;93:863‐8. [DOI] [PubMed] [Google Scholar]
Nightingale 2002 {published data only}
- Nightingale JA, Rogers DF, Barnes PJ. Comparison of the effects of salmeterol and formoterol in patients with severe asthma. Chest 2002;121(5):1401‐6. [DOI] [PubMed] [Google Scholar]
Nsouli 2001 {published data only}
- Nsouli SM, McNutt WJ. The additive effects of montelukast and salmeterol in moderate asthmatics who are uncontrolled on a low dose on inhaled corticosteroids. Annals of Allergy, Asthma & Immunology 2001;86:81. [Google Scholar]
O'Brian 2001 {published data only}
- O'Brian J, Carlos‐Palma A, Bogolubov M, Davies P, Payne E. Benefits of fluticasone propionate/salmeterol [fp/s] HFA‐MDI are apparent on the first day of dosing. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Odeback 1998 {published data only}
- Odeback P. Is the addition of salmeterol more effective than doubling the dose of budesonide in mild asthma?. American Journal of Respiratory & Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A417.
Olsson 2002 {published data only}
- Olsson P, Stallberg B, Ekstrom T, Lindarck N, Jorgensen LA. Adjustable maintenance treatment of asthma with budesonide and formoterol in a single inhaler. European Respiratory Society Annual Congress. 2002:P2451.
Ortega‐Cisnero 1998 {published data only}
- Ortega‐Cisnero M, Maldonado‐Alaniz ML, Rosas Vargas MA, Sierra‐Monge JJL. Salmeterol and inhaled beclomethasone versus high dose inhaled beclomethasone in the control of pediatric patients with moderate asthma. Annals of Allergy, Asthma and Immunology 1998;80:131. [Google Scholar]
Overbeck 2003 {published data only}
- Overbeck SE, Mulder PG, Baelemans SM, Hoogsteden HC, Prins JB. Comparison of budesonide plus formoterol with budesonide alone on airway inflammation in mild asthmatics. American Thoracic Society 99th International Conference. 2003:D034 Poster C36.
Ozkaya 1999 {published data only}
- Ozkaya O, Turktas I, Cengizlier R. Low dose budesonide plus formoterol versus high dose budesonide in children with bronchial asthma. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999.
Palmer 1992 {published data only}
- Palmer JBD, Stuart AM, Shepherd GL, Viskum K. Inhaled salmeterol in the treatment of patients with moderate to severe reversible obstructive airways disease ‐ a 3 month comparison of the efficacy and safety of twice‐daily salmeterol (100 mcg) with salmeterol (50 mcg). Respiratory Medicine 1992;86:409‐17. [DOI] [PubMed] [Google Scholar]
Palmqvist 2001 {published data only}
- Palmqvist M, Arvidsson P, Beckman O, Peterson S, Lotvall J. Onset of bronchodilation of budesonide/formoterol vs salmeterol/fluticasone in single inhalers. Pulmonary Pharmacology & Therapeutics 2001;14(1):29‐34. [DOI] [PubMed] [Google Scholar]
Paterson 1999 {published data only}
- Paterson MC, Wilson AM, Dempsey OJ, Sims EJ, Lipworth BJ. The effect of combination therapy with salmeterol and montelukast in asthmatic patients receiving inhaled corticosteroids. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999:P3490.
Pauwels 1998 {published data only}
- Pauwels RA, Yernault JC, Demedts MG, Geusens P. Safety and efficacy of fluticasone and beclomethasone in moderate to severe asthma. American Journal of Respiratory & Critical Care Medicine 1998;157(3 pt 1):827‐32. [DOI] [PubMed] [Google Scholar]
Pearlman 1992 {published data only}
- Pearlman DA, Chervinsky P, LaForce C, Seltzer JM, Southern DL, Kemp JP, et al. A comparison of salmeterol with albuterol in the treatment of mild to moderate asthma. New England Journal of Medicine 1992;327:1420‐5. [DOI] [PubMed] [Google Scholar]
Pearlman 1994 {published data only}
- Pearlman DS, Liddle R. Controlling asthma symptoms: salmeterol compared with salbutamol in large‐scale multicentre studies. European Respiratory Review 1994;4(21):301‐5. [Google Scholar]
Pearlman 1999a {published data only}
- Pearlman DS, Stricker W, Weistein S, Gross G, Chervinsky P, Woodring A, et al. Inhaled salmeterol and fluticasone: a study comparing monotherapy and combination therapy in asthma. Annals of Allergy, Asthma, & Immunology 1999;82:257‐65. [DOI] [PubMed] [Google Scholar]
Pearlman 1999b {published data only}
- Pearlman DS, Stricker W, Weistein S, Gross G, Chervinsky P, Woodring A, et al. Inhaled salmeterol and fluticasone: a study comparing monotherapy and combination therapy in asthma. Annals of Allergy, Asthma, & Immunology 1999;82:257‐65. [DOI] [PubMed] [Google Scholar]
Pearlman 2001 {published data only}
- Pearlman DS, Kent E, Lanz MJ, Peden D, Baitinger L, Herrle M, et al. Fluticasone propionate/salmeterol HFA‐MDI has a rapid onset of effect in asthmatics treated with short or long‐acting beta2‐agonists (ba) or inhaled corticosteroids. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Pearlman 2002 {published data only}
- Pearlman DS, White MV, Lieberman AK, Pepsin PJ, Kalberg C, Emmett A, et al. Fluticasone propionate/salmeterol combination compared with montelukast for the treatment of persistent asthma. Annals of Allergy, Asthma and Immunology 2002;88(2):227‐35. [DOI] [PubMed] [Google Scholar]
Peters 2000 {published data only}
- Peters JI, Shelledy DC, Jones AP Jr, Lawson RW, Davis CP, LeGrand TS. A randomized, placebo‐controlled study to evaluate the role of salmeterol in the in‐hospital management of asthma. Chest 2000;118(2):313‐20. [DOI] [PubMed] [Google Scholar]
Pieters 1999b {published data only}
- Pieters WR, Lundback B, Sondhi S, Price MJ. Cost effectiveness analysis of salmeterol/fluticasone propionate 50/500mcg vs fluticasone propionate 500mcg in patients with corticosteroid‐dependent asthma. Pharmacoeconomics 1999;16(Suppl 2):29‐34. [Google Scholar]
Pieters 2001 {published data only}
- Pieters WR, Wilson KK, Smith HCE, Tamminga JJ. Cost‐effectiveness of fluticasone propionate/salmeterol combination product and fluticasone propionate/montelukast in asthma. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Pinnas 1998 {published data only}
- Pinnas JL, Schwartz H, Yancey SW, Rickard K. Six month comparison of beclomethasone versus salmeterol or placebo in adults with asthma. American Journal of Respiratory & Critical Care Medicine 1998; Vol. 157, issue Suppl 3:A417.
Pizzichini 1996 {published data only}
- Pizzichini MM, Kidney JC, Wong BJ, Morris MM, Efthimiadis A, Dolovich J, et al. Effect of salmeterol compared with beclomethasone on allergen‐induced asthmatic and inflammatory responses. European Respiratory Journal 1996;9(3):449‐55. [DOI] [PubMed] [Google Scholar]
Pljaskic‐Kamenov 2000 {published data only}
- Pljaskic‐Kamenov SS, Filipovic MD, Kamenov BA. Comparison of addition of salmeterol xinafoate to budesonide with budesonide alone on symptoms and quality of life in asthmatic children. European Respiratory Journal 2000; Vol. 16, issue Suppl 31:518s.
Price 2003 {published data only}
- Price D, Haughney J, Rosen J P, Morrison K. Switching to Symbicort(r) from beclomethasone dipropionate (BDP) with or without salmeterol significantly improved symptom severity in patients with moderate asthma. American Thoracic Society 99th International Conference. 2003; Vol. D034 Poster C40.
Pujet 1995 {published data only}
- Pujet JC, Evano CI. A randomized double‐blind study comparing inhaled beclomethasone with long‐acting theophylline for the first‐line treatment of moderate asthma. Semaine Des Hopitaux 1995;71(27‐8):865‐72. [Google Scholar]
Pyke 2001 {published data only}
- Pyke SD. Synergy with salmeterol and fluticasone propionate after administration from a single inhaler (Seretide(tm)). European Respiratory Journal. 2001; Vol. 18, issue Suppl 33:176s.
Rance 2002 {published data only}
- Rance L, Musin L. Asthma management costs in Canada are lower with combination fluticasone propionate/salmeterol (250/50mcg BID) in as single inhaler than with budesonide 800mcg BID plus eformoterol 12 mcg BID via separate inhalers. Chest 2002; San Diego, CA. 2002:S6.
Rickard 1999 {published data only}
- Rickard KA, Yancey S, Emmett AH, Kalberg CJ. Salmeterol compared to zafirlukast when added to inhaled corticosteroid therapy in patients with persistent asthma. European Respiratory Society; 1999 Oct 9‐13; Madrid, Spain. 1999:P839.
Rickard 2001 {published data only}
- Rickard K, Dorinsky PM, Knobil K, Pepsin P, Akveld ML. The salmeterol/fluticasone propionate combination 50/100µg bid is more effective than oral montelukast 10mg of as a first line therapy in mild and moderate asthmatics. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:262s.
Rijssenbeek‐Nouwens 2002 {published data only}
- Rijssenbeek‐Nouwens LH, Oosting AJ, Monchy JG, Bregman I, Postma DS, Bruin‐Weller MS. The effect of anti‐allergic mattress encasings on house dust mite‐induced early‐ and late‐airway reactions in asthmatic patients. A double‐blind, placebo‐controlled study . Clinical & Experimental Allergy 2002;32(1):117‐25. [DOI] [PubMed] [Google Scholar]
Ringbaek 1996 {published data only}
- Ringbaek TJ, Soes‐Petersen U, Christensen M, Iversen ET, Rasmussen FV. Salmeterol improves the control of disease in patients with moderate asthma. A comparative study of inhaled salmeterol 50 mg and salbutamol depot tablets 8 mg, both administered twice daily]. Ugeskrift for Laeger 1996;158(27):3940‐3. [PubMed] [Google Scholar]
Ringdal 1997 {published data only}
- Ringdal N, Eliraz A, Pruzinec R, Weber HH, Mulder PG, Akveld M, et al. The salmeterol/fluticasone combination is more effective than fluticasone plus oral montelukast in asthma. Respiratory Medicine 97;3:234‐4. [DOI] [PubMed] [Google Scholar]
Ringdal 2002 {published data only}
- Alonso JF, Badiola C, Kielhorn A. Economic evaluation of salmeterol/fluticasone combination vs budesonide plus formoterol in Spain. European Respiratory Journal 2001;18(Suppl 33):49s. [Google Scholar]
- Chuchalin AG, Chovan L, Ringdal N, Whitehead PJ. Advair™ seretide™ (250/50 mcg bid) shows nocturnal benefit over budesonide 800mu‐g + formoterol 12mu‐g bid in moderate to severe asthma. Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
- Jenkins C, Wilson J, Rutherford C, Perry AS, Whitehead PJ. Asthma management costs are lower with combination fluticasone/salmeterol (25/50 mcg BD) in a single inhaler than with budesonide (800 mcg BD) plus eformoterol (12 mcg BD) via separate inhalers. Respirology 2002; Vol. 7, issue Suppl:A20. [P23]
- Martin AA, Whitehead PJ, McCarthy TP. Asthma costs with salmeterol/fluticasone combination 50/250mcg bd compared to budesonide 800mcg bd plus formoterol 12mcg bd. American Thoracic Society 99th International Conference. 2003:D034 Poster C43.
- Ringdal N, Chuchalin A, Chovan L, Tudoric N, Maggi E, Whitehead PJ. Evaluation of different inhaled combination therapies (EDICT): a randomised, double‐blind comparison of Seretide (50/250 microgram bd Diskus vs. formoterol (12 microgram bd) and budesonide (800 microgram bd) given concurrently (both via Turbuhaler) in patients with moderate‐to‐severe asthma. Respiratory Medicine 2002;96(11):851‐61. [DOI] [PubMed] [Google Scholar]
- Ringdal N, Chuchalin AG, Chovan L, Whitehead PJ. A comparison of Advair™/Seretide™ (salmeterol 50 mcg/fluticasone propionate 250 mcg bid) with formoterol 12 mcg + budesonide 800 mcg bid in moderate‐severe asthma. American Journal of Respiratory & Critical Care Medicine 2000; Vol. 161, issue 3 part 2 (Supp1):A196.
- Ringdal NR, Chovan L, Chuchalin AG, Whitehead PJ. Advair™/Seretide™ (250/50mu‐g bid) shows exacerbation benefit over budesonide plus formoterol bid in moderate‐severe asthma. Annual Thoracic Society 97th International Conference; San Francisco CA, May 18‐23. 2001.
Ringdal 2003 {published data only}
- Ringdal N, Eliraz A, Pruzinec R, Weber HH, Mulder PG, Akveld M, et al. The salmeterol/fluticasone combination is more effective than fluticasone plus oral montelukast in asthma. Respiratory Medicine 97;3:234‐4. [DOI] [PubMed] [Google Scholar]
Rocca‐Serra 2002 {published data only}
- Rocca‐Serra JP, Vicaut E, Lefrancois G, Umile A. Efficacy and tolerability of a new non‐extrafine formulation of beclomethasone HFA‐134a in patients with asthma: comparison with beclomethasone CFC. Clinical Drug Investigation 2002;22(10):653‐65. [Google Scholar]
Rooklin 2001 {published data only}
- Rooklin A, Elkayam D, Weiler J, Windom H, Schoaf L, Scott C, et al. The fluticasone propionate/salmeterol HFA MDI is significantly more efficacious in treating asthma than placebo HFA MDI, fluticasone propionate CFC MDI or salmeterol CFC MDI. Journal of Allergy and Clinical Immunology 2001; Vol. 107, issue 2:S100.
Rosenhall 2001a {published data only}
- Rosenhall L, Stahl E, Heinig JH, Lindquist A, Leegard J, Bergqvist PBF. Health‐related quality of life and asthma control in patients using Symbicort® (budesonide and formoterol in a single inhaler). Annual Thoracic Society 97th International Conference; San Francisco CA ,May 18‐23. 2001.
Rosenhall 2001b {published data only}
- Rosenhall L, Heinig JH, Lindqvist A, Leegard J, Bergqvist PB. Budesonide and formoterol in a single inhaler is safe and effective in the treatment of asthma. European Respiratory Journal 2001;18(33):159s. [Google Scholar]
Rosenhall 2001c {published data only}
- Rosenhall L, Stahl E, Heinig JH, Lindqvist A, Leegard J, Bergqvist PB. Health‐related quality of life and asthma control in patients treated with budesonide and formoterol in a single inhaler. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:46s.
Rosenhall 2002 {published data only}
- Rosenhall L, Heinig JH, Lindqvist A, Leegaard J, Stahl E, Bergqvist PB. Budesonide/formoterol (Symbicort) is well tolerated and effective in patients with moderate persistent asthma. International Journal of Clinical Practice 2002;56(6):427‐33. [PubMed] [Google Scholar]
Rosenhall 2003 {published data only}
- Rosenhall L, Elvstrand A, Tilling B, Vinge I, Jemsby P, Stahl E, et al. One‐year safety and efficacy of budesonide/formoterol in a single inhaler (Symbicort Turbuhaler ) for the treatment of asthma. Respiratory Medicine 2003;97(6):702‐8. [DOI] [PubMed] [Google Scholar]
Rosenhall 56 {published data only}
- Rosenhall L, Heinig JH, Lindqvist A, Leegard J, Bergqvist PB. Symbicort (budesonide/eformoterol in a single inhaler) is safe and effective in the treatment of asthma. Thorax 56; Vol. Suppl 3:iii 63.
Rosenthal 1999 {published data only}
- Rosenthal RR, Busse WW, Kemp JP, Baker JW, Kalberg C, Emmett A, Rickard KA. Effect of long‐term salmeterol therapy compared with as‐needed albuterol use on airway hyperresponsiveness. Chest 1999;116(3):595‐602. [DOI] [PubMed] [Google Scholar]
Rumbak 1998 {published data only}
- Rumbak M, Self T, Kelso T, Eberle L, Abou‐Shala N, Learned S, et al. Moderate to high dose inhaled corticosteroids in adult asthmatics: does salmeterol facilitate step down therapy?. European Respiratory Journal 1998;12(Suppl 29):19s. [Google Scholar]
Sahn 2002 {published data only}
- Sahn S, Yancey S, Reilly D, Edwards L, Rickard K, Dorinsky P. The fluticasone propionate/salmeterol (FSC) combination product 100/50 mcg BID is steroid sparing in patients who require FP250 mcg BID for asthma stability. Chest 2002 (conference); San Diego, CA. 2002.
SAM30007 {unpublished data only}
- SAM30007. A multicentre, randomised, double‐blind, controlled, parallel‐group, comparative investigation of the corticosteroid‐saving potential of the combination therapy fluticasone propionate and salmeterol (SERETIDE) compared with fluticasone propionate alone, given to adult asthmatic subjects, when reducing the inhaled corticosteroid dose from an initially high level of 500μg bd. www.ctr.gsk.co.uk 2005 (accessed 5 June 2008).
SAM40004 {published data only}
- Beckett P, Hewitt L, Woodcock A, Smith J, Seghal N, Rice L, et al. Improvement in airway hyper‐responsiveness (AHR) and lung function with salmeterol/fluticasone propionate combination (SFC) in persistent asthma [abstract]. American Thoracic Society 99th International Conference. 2003:D034 Poster C27.
- SAM40004. A multi‐centre, randomised, double‐blind, placebo‐controlled parallel group study to compare the effect on airway inflammation and remodelling of treatment with salmeterol/fluticasone propionate combination product (50/100μg strength) bd via the Accuhaler inhaler, or fluticasone propionate 100μg bd via the Accuhaler inhaler or placebo via the Accuhaler inhaler for 16 weeks, followed by double‐blind treatment for 52 weeks with the salmeterol/fluticasone propionate combination product (50/100μg strength) bd via the Accuhaler inhaler or fluticasone propionate 100μg bd via the Accuhaler inhaler, in adults with reversible airways obstruction (SIRIAS ‐ Seretide in Inflammation and Remodelling In Asthma Study). http://www.gsk.ctr.co.uk 2006 (accessed 30 April 2008).
Schreurs 1996 {published data only}
- Schreurs AJ, Sinninghe Damste HE, Graaff CS, Greefhorst AP. A dose‐response study with formoterol Turbuhaler as maintenance therapy in asthmatic patients. European Respiratory Journal 1996;9:1678‐83. [DOI] [PubMed] [Google Scholar]
Sears 2003 {published data only}
- Sears MR, McIvor A, Becker A, Fitzgearld JM, Boulet LP, Ernsy P, et al. Budesonide/formoterol adjustable maintenance dosing effectively improves asthma symptom severity: a multicentre Canadian study. European Respiratory Journal 2003;22(Suppl 45):P1695. [Google Scholar]
Serrier 2003 {published data only}
- Serrier P, Roche N, Pello JY, Larguier JS, Mezzi K. Asthma control achieved with inhaled corticosteroids and long‐acting beta2‐agonists in a free or fixed combination: results of the ALISE survey. Presse Medicale 2003;32(11):493‐7. [PubMed] [Google Scholar]
Shapiro 2001 {published data only}
- Shapiro GG, Mendelson LM, Pearlman DS. Once‐daily budesonide inhalation powder (Pulmicort Turbuhaler) maintains pulmonary function and symptoms of asthmatic children previously receiving inhaled corticosteroids. Annals of Allergy, Asthma, and Immunology 2001;86(6):633‐40. [DOI] [PubMed] [Google Scholar]
Sheth 2002 {published data only}
- Sheth K, Borker R, Emmett A, Rickard K, Dorinsky P. Cost‐effectiveness comparison of salmeterol/fluticasone propionate versus montelukast in the treatment of adults with persistent asthma. Pharmacoeconomics 2002;20(13):909‐18. [DOI] [PubMed] [Google Scholar]
Shrewsbury 2002 {published data only}
- Shrewsbury SB, Pyke S. MIASMA: asthma exacerbation reduction with salmeterol [Letter]. Chest 2002;121(3):1002‐4. [DOI] [PubMed] [Google Scholar]
Sienra‐Monge 2001 {published data only}
- Sienra‐Monge JJL, Rio BE, Alvarez ME, Magana AJ. Comparison of quality of life and pulmonary function on moderate asthmatic children treated with beclomethasone and beclomethasone plus salmeterol. Journal of Allergy and Clinical Immunology 2001; Vol. 107, issue 2:s263.
Simons 1997b {published data only}
- Simons FER. A comparison of beclomethasone, salmeterol and placebo in children with asthma. New England Journal of Medicine 1997;337:1659‐65. [DOI] [PubMed] [Google Scholar]
Sims 2003 {published data only}
- Sims EJ, Jackson CM, Lipworth BJ. Add‐on therapy with montelukast or formoterol in patients with the glycine‐16 beta2‐receptor genotype. British Journal of Clinical Pharmacology 2003;56(1):104‐11. [DOI] [PMC free article] [PubMed] [Google Scholar]
Staehr 1995 {published data only}
- Staehr P, Vestbo I. Salmeterol improves control in asthmatic patients treated in general practice. A comparative study of salmeterol (Serevent) and salbutamol. Ugeskr‐Laeger 1995;157(1):36‐40. [PubMed] [Google Scholar]
Stahl 2003 {published data only}
- Stahl E, Postma DS, Svensson K, Tattersfield AE, Eivindson A, Schreurs A, et al. Formoterol used as needed improves health‐related quality of life in asthmatic patients uncontrolled with inhaled corticosteroids. Respiratory Medicine 2003;97(9):1061‐6. [DOI] [PubMed] [Google Scholar]
Stallberg 2003 {published data only}
- Stallberg B, Olsson P, Jorgensen LA, Lindarck N, Ekstrom T. Budesonide/formoterol adjustable maintenance dosing reduces asthma exacerbations versus fixed dosing. International Journal of Clinical Practice 2003;57(8):656‐61. [PubMed] [Google Scholar]
Stanford 2002 {published data only}
- Stanford RH, Borker R, Dorinsky P, Pepsin P, Kalberg C, Emmett A. The costs and efficacy of fluticasone propionate/salmeterol combination versus montelukast in the treatment of adults with persistent asthma. Chest 2002; San Diego, CA. 2002:422.
Stankovic 2000 {published data only}
- Stankovic IJ, Djordjevic DV, Pejcic TA, Zivkovic DS, Rancic MR, et al. Formoterol and beclomethasone dipropionate versus higher dose beclomethasone dipropionate in asthma patients. European Respiratory Journal 2000;16(Suppl 31):455s. [Google Scholar]
Stelmach 2001 {published data only}
- Stelmach I, Grzelewski T, Majak P, Majak J, Bobrowska M, Jerzynska J, et al. The effect of triamcinolone, montelukast and formoterol on serum levels of il‐4, IgE and clin.ical parameters in children with asthma. Polski Merkuriusz Lekarski 2001;11(63):247‐51. [PubMed] [Google Scholar]
Stelmach 2002a {published data only}
- Stelmach I, Jerzynska J, Majak P, Grzelewski T, Gorski P, Stelmach W, Kuna P. Effect of triamcinolone acetonide, montelukast, nedocromil sodium, formoterol on serum levels of sICAM‐1, sIL‐2R and clinical parameters of asthma in children. Polski Merkuriusz Lekarsk 2002;12(68):99‐103. [PubMed] [Google Scholar]
Stelmach 2002b {published data only}
- Stelmach I, Jerzynska J, Kuna P. A randomized, double‐blind trial of the effect of glucocorticoid, antileukotriene and [beta]‐agonist treatment on IL‐10 serum levels in children with asthma. Clinical & Experimental Allergy 2002;32(2):264‐9. [DOI] [PubMed] [Google Scholar]
Stelmach 2008 {published data only (unpublished sought but not used)}
- Stelmach I, Grzelewski T, Jerzynska J, Kuna P. A randomized, double‐blind trial on the effect of treatment with montelukast, budesonide, montelukast with budesonide, formoterol with budesonide on lung function and clinical symptoms in children with asthma [Abstract]. Journal of Allergy & Clinical Immunology 2005;115(Suppl 2):S151. [Google Scholar]
- Stelmach I, Grzelewski T, Majak P, Jerzynska J, Stelmach W, Kuna P. Effect of different antiasthmatic treatments on exercise‐induced bronchoconstriction in children with asthma. Journal of Allergy and Clinical Immunology 2007; Epub ahead of print (accessed 17 January 2008). [DOI] [PubMed]
Stojkovic‐Andjelkovi 2001 {published data only}
- Stojkovic‐Andjelkovic AK, Pajovic DM, Protrka OJ, Ugrinovic BS, Obradovic SM, Pavicevic MD. Effect of combination fluticasone propionate and salmeterol diskhaler in treatment of moderate to severe asthma: comparison of initial high dose, constant medium dose and placebo. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:123s.
Stoloff 2002 {published data only}
- Stoloff S, Poinsett‐Holmes K, Dorinsky PM. Combination therapy with inhaled long‐acting beta(2)‐agonists and inhaled corticosteroids: a paradigm shift in asthma management. Pharmacotherapy 2002;22(2):212‐6. [DOI] [PubMed] [Google Scholar]
Tan 1997 {published data only}
- Tan S, Hall IP, Dewar J, Dow E, Lipworth B. Association between beta 2‐adrenoceptor polymorphism and susceptibility to bronchodilator desensitisation in moderately severe stable asthmatics. Lancet 1997;350(9083):995‐9. [DOI] [PubMed] [Google Scholar]
Tattersfield 1999 {published data only}
- Tattersfield AE, Postma DS, Barnes PJ, Svensson K, Bauer CA, O'Byrne PM, et al. Exacerbations of asthma: a descriptive study of 425 severe exacerbations. The FACET International Study Group. American Journal of Respiratory & Critical Care Medicine 1999;160(2):594‐9. [DOI] [PubMed] [Google Scholar]
Tattersfield 2001 {published data only}
- Tattersfield AE, Lofdahl CG, Postma DS, Eivindson A, Schreurs AG, Rasidakis A, et al. Comparions of formoterol and terbutaline for as‐needed treatment of asthma: a randomised trial. Lancet 2001;357(9252):257‐61. [DOI] [PubMed] [Google Scholar]
Tolley 2002 {published data only}
- Tolley K, Martin A, Rice L, McCarthy TP. Salmeterol/fluticasone propionate combination (SFC) demonstrates improved health outcomes and good cost effectiveness compared with beclometasone dipropionate. American Journal of Respiratory & Critical Care Medicine 2002;165(Suppl 8):A112. [Google Scholar]
Tonelli 2001 {published data only}
- Tonelli M, Giannini D, Franco A, Carnevali S, Bartoli ML, Cianchetti S, et al. Symptoms, induced sputum eosinophil percentages and treatment with salmeterol or fluticasone in mild asthmatics. European Respiratory Journal 2001, issue Suppl 33:336s.
Trautmann 2001 {published data only}
- Trautmann M. Treatment with salmeterol/fluticasone propionate (50/250g) inhaler improves lung function, asthma symptoms and quality of life in a large group of patients with mild to moderate asthma. American Journal of Respiratory & Critical Care Medicine 2001; Vol. 163, issue 5:A864.
Turner 1998 {published data only}
- Turner MO, Johnston PR, Pizzichini E, Pizzichini MM, Hussack PA, Hargreave FE. Anti‐inflammatory effects of salmeterol compared with beclomethasone in eosinophilic mild exacerbations of asthma: a randomized, placebo controlled trial. Canadian Respiratory Journal 1998;5(4):261‐8. [DOI] [PubMed] [Google Scholar]
Ullman 1990 {published data only}
- Ullman A, Hedner J, Svedmyr N. Inhaled salmeterol and salbutamol in asthmatic patients. An evaluation of asthma symptoms and the possible development of tachyphylaxis. American Review of Respiratory Disease 1990;142(3):571‐5. [DOI] [PubMed] [Google Scholar]
Van den Berg 2000 {published data only}
- Berg NJ, Ossip MS, Hederos CA, Anttila H, Ribeiro BL, Davies PI. Salmeterol/fluticasone propionate (50/100 ug) in combination in Diskus inhaler (Seretide) is effective and safe in children with asthma. Pediatric Pulmonology 2000;30:97‐105. [DOI] [PubMed] [Google Scholar]
van der Woude 2001 {published data only}
- Woude HJ, Winter TH, Boorsma M, Bergqvist PBF, Aalbers R. More rapid relief of methacholine‐induced bronchoconstriction with budesonide/formoterol than with salmeterol/fluticasone. European Respiratory Journal 2001; Vol. 18, issue Suppl 33:53s.
Van der Woude 2004 {published data only}
- Woude HJ, Boorsma M, Bergqvist PBF, Winter TH, Aalbers RH. Budesonide/formoterol in a single inhaler rapidly relieves methacholine‐induced moderate‐to‐severe bronchoconstriction. Pulmonary Pharmacology & Therapeutics 2004;17(2):89‐95. [DOI] [PubMed] [Google Scholar]
Van Der Woude 2004 {published data only}
- Woude HJ, Boorsma M, Bergqvist PB, Winter TH, Aalbers R. Budesonide/formoterol in a single inhaler rapidly relieves methacholine‐induced moderate‐to‐severe bronchoconstriction. Pulmonary Pharmacology & Therapeutics 2004;17(2):89‐95. [DOI] [PubMed] [Google Scholar]
Van Noord 1999 {published data only}
- Schreurs AJ, Noord JA, Mulder PG. Fluticasone propionate (FP) and salmeterol xinafoate (SLM) in patients with mild to moderate asthma. European Respiratory Journal. 1998; Vol. 12, issue Suppl 29:19s, F159.
- Noord JA, Schreurs AJM, Mol SJM, Mulder PGH. Addition of salmeterol versus doubling the dose of fluticasone propionate in patients with mild to moderate asthma. Thorax 1999;54:207‐12. [DOI] [PMC free article] [PubMed] [Google Scholar]
van Noord 2001 {published data only}
- SFCB3023. A multicentre, randomised, double‐blind, double‐dummy, parallel‐group, three‐month comparison of the salmeterol/fluticasone propionate combination product (2x25/250mcg strength) bd via the pressurised metered dose inhaler with salmeterol/fluticasone propionate combination product (1x50/500mcg strength) bd via the Diskus/Accuhaler™ inhaler and with fluticasone propionate (2x 250mcg strength) alone bd via the pressurised metered dose inhaler in adolescents and adults with reversible airways obstruction. http://www.ctr.gsk.co.uk 2004.
- Noord JA, Lill H, Carrillo Diaz T, Greefhorst AP, Davies P. Clinical equivalence of a salmeterol/fluticasone propionate combination product delivered via a chlorofluorocarbon‐free metered‐dose inhaler with the Diskus in patients with moderate to severe asthma. Clinical Drug Investigation 2001;21(4):243‐55. [Google Scholar]
- Noord JA, Lill H, Carrillo T, Davies P. Clinical equivalence of salmeterol/fluticasone propionate combination 50/500 bid delivered via metered dose inhaler (MDI) or diskus™ in patients with reversible airways obstruction. American Journal of Respiratory & Critical Care Medicine 2000; Vol. 161, issue Suppl 3:A197.
Van Schayck 2002 {published data only}
- Schayck CP, Cloosterman SG, Bijl‐Hofland ID, Hoogen H, Folgering HT, Weel C. Is the increase in bronchial responsiveness or FEV1 shortly after cessation of beta2‐agonists reflecting a real deterioration of the disease in allergic asthmatic patients? A comparison between short‐acting and long‐acting beta2‐agonists. Respiratory Medicine 2002;96(3):155‐62. [DOI] [PubMed] [Google Scholar]
Vastagh 2003 {published data only}
- Vastagh E, Kuna P, Calistru P, Bogdan MA. Efficacy and safety of inhaled budesonide delivered once or twice daily via HFA‐134a in mild to moderate persistent asthma in adult patients. Comparison with budesonide CFC. Respiratory Medicine 2003;97(Suppl D):S20‐S28. [DOI] [PubMed] [Google Scholar]
Verberne 1997 {published data only}
- Verberne AA, Frost C, Roorda RJ, Laag H, Kerrebijn KF. One year treatment with salmeterol compared with beclomethasone in children with asthma. American Journal of Respiratory & Critical Care Medicine 1997;156:688‐95. [DOI] [PubMed] [Google Scholar]
Vermetten 1999 {published data only}
- Vermetten AM, Boermans JM, Luiten WDVF, Mulder PGH, Vermue NA. Comparison of salmeterol with beclomethasone in adult patients with mild persistent asthma who are already on low‐dose inhaled steroids. Journal of Asthma 1999;36(1):97‐106. [DOI] [PubMed] [Google Scholar]
Vestbo 2000 {published data only}
- Vestbo J, Sorensen T, Lange P, Brix A, Torre P, Viskum K. Long‐term effect of inhaled budesonide in patients with mild to moderate chronic obstructive lung disease. Ugeskr‐Laeger 2000;162(4):493‐7. [PubMed] [Google Scholar]
Vickers 2000 {published data only}
- Vickers M. Assessment of long‐term efficacy of early introduction of inhaled steroids in asthma. National Health Technology Assessment (NCCHTA).
Vilsvik 2001 {published data only}
- Vilsvik J, Ankerst J, Palmqvist M, Persson G, Schaanning J, Schwabe G, et al. Protection against cold air and exercise‐induced bronchoconstriction while on regular treatment with Oxis. Respiratory Medicine 2001;95(6):484‐90. [DOI] [PubMed] [Google Scholar]
Virchow 2002 {published data only}
- Virchow JC. Salmeterol powder provides significantly better benefit than montelukast in asthmatic patients receiving concomitant inhaled corticosteroid therapy. Chest 2002;121(6):2083‐4. [DOI] [PubMed] [Google Scholar]
Von Berg 1989 {published data only}
- von‐Berg A, Berdel D. Formoterol and salbutamol metered aerosols: comparison of a new and an established beta‐2‐agonist for their bronchodilating efficacy in the treatment of childhood bronchial asthma. Pediatric Pulmonology 1989;7(2):89‐93. [DOI] [PubMed] [Google Scholar]
Von Berg 2003 {published data only}
- Berg A, Papageorgiou Saxoni F, Wille S, Carrillo T, Kattamis C, Helms PJ. Efficacy and tolerability of formoterol Turbuhaler in children. International Journal of Clinical Practice 2003;57(10):852‐6. [PubMed] [Google Scholar]
Wallaert 1999 {published data only}
- Wallaert B, Brun P, Ostinelli J, Murciano D, Champel F, Blaive B, et al. A comparison of two long‐acting beta‐agonists, oral bambuterol and inhaled salmeterol, in the treatment of moderate to severe asthmatic patients with nocturnal symptoms. The French Bambuterol Study Group. Respiratory Medicine 1999;93(1):33‐8. [DOI] [PubMed] [Google Scholar]
Wallin 1990 {published data only}
- Wallin A, Melander B, Rosenhall L, Sandstrom T, Wahlander L. Formoterol, a new long acting beta 2 agonist for inhalation twice daily, compared with salbutamol in the treatment of asthma. Thorax 1990;45(4):259‐61. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wallin 1998 {published data only}
- Wallin A, Sandstrom T, Soderberg M, Howarth P, Djukanovic R, Wilson S, et al. Effects of formoterol, budesonide and placebo treatment on asthmatic airway inflammation. Annals of Allergy, Asthma, and Immunology 1998; Vol. 80:88.
Wallin 1999 {published data only}
- Wallin A, Sandstrom T, Soderberg M, Howarth P, Lundback B, Della‐Cioppa G, et al. The effects of regular inhaled formoterol, budesonide, and placebo on mucosal inflammation and clinical indices in mild asthma. American Journal of Respiratory & Critical Care Medicine 1999;159(1):79‐86. [DOI] [PubMed] [Google Scholar]
Weinberger 2004 {published data only}
- Weinberger M. Innovative therapies for asthma: Anti‐IgE ‐ the future?. Paediatric Respiratory Reviews 2004;5(Suppl A):S115‐18. [DOI] [PubMed] [Google Scholar]
Weinstein 1998 {published data only}
- Weinstein SF, Pearlman DS, Bronsky EA, Byrne A, Arledge T, Liddle R, et al. Efficacy of salmeterol xinafoate powder in children with chronic persistent asthma. Annals of Allergy, Asthma and Immunology 1998;81:51‐8. [DOI] [PubMed] [Google Scholar]
Weinstein 2001 {published data only}
- Weinstein SF, Pearlman DS, Condemi JJ, Herrle MR, Scott CA, Payne JE, et al. Superior efficacy of the fluticasone propionate/salmeterol (88/42mcg) HFA‐MDI combination product versus the individual components in asthmatics previously treated with either short‐ or long‐acting beta2‐agonists or inhaled corticosteroids. Journal of Allergy and Clinical Immunology 2001;107(2):S102. [Google Scholar]
Wempe 1992 {published data only}
- Wempe JB, Tammeling EP, Koeter GH, Hakansson L, Venge P, Postma DS. Blood eosinophil numbers and activity during 24 hours: effects of treatment with budesonide and bambuterol. Journal of Allergy and Clinical Immunology 1992;90(5):757‐65. [DOI] [PubMed] [Google Scholar]
White 2001 {published data only}
- White M, Scott C, Herrle MR, Pearlman D, Payne E, House K, et al. Salmeterol/fluticasone propionate (42/88mcg) HFA‐MDI improves asthma control in asthmatics previously treated with short‐ or long‐acting beta2‐agonists or inhaled corticosteroids. Annals of Allergy, Asthma & Immunology 2001; Vol. 86:81.
Wilcke 1998 {published data only}
- Wilcke JT, Iversen ET, Kok‐Jensen A. Effect of salmeterol is independent of previously inhaled salbutamol: a clinical controlled study. Lung 1998;176(2):133‐9. [DOI] [PubMed] [Google Scholar]
Wilding 1997 {published data only}
- Wilding P, Clark M, Thompson Coon J, Lewis S, Rushton L, Bennett J, et al. Effect of long term treatment with salmeterol on asthma control: a double‐blind, randomised crossover study. BMJ 1997;314:1441‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Wilson 1999 {published data only}
- Wilson AM, Dempsey OJ, Sims EJ, Lipworth BJ. A comparison of salmeterol and montelukast as second‐line therapy in asthmatic patients not controlled on inhaled corticosteroids. Thorax 1999; Vol. 54, issue Suppl 3:A66, P189.
Wilson 2000 {published data only}
- Wilson SJ, Ward JA, Djukanovic R, Wallin A, Sue‐Chu M, Sandstrom T, et al. Effects of high and low dose inhaled fluticasone propionate (FP) compared to low dose FP combined with salmeterol (SAL) on airway inflammation in asthma. American Journal of Respiratory & Critical Care Medicine 2000; Vol. 161, issue Suppl 3:A196.
Wilson 2001a {published data only}
- Wilson AM, Dempsey OJ, Sims EJ, Lipworth BJ. Evaluation of salmeterol or montelukast as second‐line therapy for asthma not controlled with inhaled corticosteroids. Chest 2001;119(4):1021‐6. [DOI] [PubMed] [Google Scholar]
Wilson 2001b {published data only}
- Wilson SJ, Wallin A, Della‐Cioppa G, Sandstrom T, Holgate ST. Effects of budesonide and formoterol on NF‐kappaB, adhesion molecules, and cytokines in asthma. American Journal of Respiratory & Critical Care Medicine 2001;164(6):1047‐52. [DOI] [PubMed] [Google Scholar]
Wong 1992 {published data only}
- Wong BJ, Dolovich J, Ramsdale EH, O'Byrne P, Gontovnick L, Denburg JA, et al. Formoterol compared with beclomethasone and placebo on allergen‐induced asthmatic responses. American Review of Respiratory Disease 1992;146(5 pt 1):1156‐60. [DOI] [PubMed] [Google Scholar]
Woolcock 1995 {published data only}
- Woolcock A. Continuing patient care with metered‐dose inhalers. Journal of Aerosol Medicine: Deposition, Clearance, and Effects in the Lung 1995;8(Suppl 2):s5‐s10. [DOI] [PubMed] [Google Scholar]
Woolcock 1996 {published data only}
- Woolcock A, Lunback B, Ringdal N, Jacques LA. Comparison of addition of salmeterol to inhaled steroids with doubling the dose of inhaled steroids. American Journal of Respiratory & Critical Care Medicine 1996;153:1481‐8. [DOI] [PubMed] [Google Scholar]
Yates 1995 {published data only}
- Yates DH, Sussman HS, Shaw MJ, Barnes PJ, Chung KF. Regular formoterol treatment in mild asthma: effect on bronchial responsiveness during and after treatment. American Journal of Respiratory & Critical Care Medicine 1995;152(41):1170‐4. [DOI] [PubMed] [Google Scholar]
Yates 1996 {published data only}
- Yates DH, Kharitonov SA, Barnes PJ. An inhaled glucocorticoid does not prevent tolerance to the bronchoprotective effect of a long‐acting inhaled beta 2‐agonist. American Journal of Respiratory & Critical Care Medicine 1996;154(6 pt 1):1603‐7. [DOI] [PubMed] [Google Scholar]
Youngchaiyud 1995 {published data only}
- Youngchaiyud P, Permpikul C, Suthamsmai T, Wong E. A double‐blind comparison of inhaled budesonide, long‐acting theophylline, and their combination in treatment of nocturnal asthma. Allergy 1995;50:28‐33. [DOI] [PubMed] [Google Scholar]
Yurdakul 2002 {published data only}
- Yurdakul AS, Calisir HC, Tunctan B, Ogretensoy M. Comparison of second controller medications in addition to inhaled corticosteroid in patients with moderate asthma. Respiratory Medicine 2002;96(5):322‐9. [DOI] [PubMed] [Google Scholar]
Zarkovic 1998 {published data only}
- Zarkovic J, Gotz MH, Holgate ST, Taak NK. Effect of long‐term regular salmeterol treatment in children with moderate asthma. Clinical Drug Investigation 1998;15(3):169‐75. [Google Scholar]
References to studies awaiting assessment
Bateman 2001 {published data only}
- Bateman ED, Beasley R, Silins V, Bogolubov M. Comparison of salmeterol/fluticasone propionate combination 50/100 bid delivered via metered dose inhaler or diskus in patients with reversible airways obstruction. American Journal of Respiratory & Critical Care Medicine 2000;161(Suppl 3):A197. [Google Scholar]
- Bateman ED, Silins V, Bogolubov M. Clinical equivalence of salmeterol/fluticasone propionate in combination (50/100 mcg twice daily) when administered via a chlorofluorocarbon‐free metered dose inhaler or dry powder inhaler to patients with mild‐to‐moderate asthma. Respiratory Medicine 2001;95(2):136‐46. [DOI] [PubMed] [Google Scholar]
- SFCB3022. A multicentre, randomised, double‐blind, double‐dummy, parallel‐group, three‐month comparison of the salmeterol/fluticasone propionate combination product (2x25/50mcg strength) bd via the pressurised metered dose inhaler with salmeterol/fluticasone propionate combination product (1x50/100mcg strength) bd via the Diskus/Accuhaler™ inhaler and with fluticasone propionate (2x 50mcg strength) alone bd via the pressurised metered dose inhaler in adolescents and adults with reversible airways obstruction. www.ctr.gsk.co.uk 2004 (accessed 12 June 2008).
Yancey 1997 {published data only}
- Yancey SW, Rickard KA, Emmett A, Cox F. The response to salmeterol or theophylline in asthmatics either receiving or not‐receiving inhaled corticosteroids. Annals of Allergy, Asthma and Immunology 1997; Vol. 78:110.
References to ongoing studies
Aziz {published data only}
- Aziz I. Comparison of anti inflammatory effect of once daily inhaled formoterol and once daily inhaled budesonide on inflammatory markers in asthmatic patients. Clinical Pharmacology, Ninewells Hospital and Medical School, University of Dundee, Dundee, DD1 9SY, Scotland, UK.
Barnes a {published data only}
- Barnes NC. A randomized parallel 3 arm study to assess asthma control, lung function, costs and quality of life in patients with asthma treated with formoterol twice daily and either budesonide once or twice daily from a novel multidisc dry powder inhaler. Respiratory Medicine, London Chest Hospital, Bonner Road, London, E2 9JX, United Kingdom.
Barnes b {published data only}
- Barnes PJ. Effect of low dose formoterol and budesonide on exhaled breath in asthma. Royal Brompton and Harefield NHS Trust.
Bush {published data only}
- Bush A. Efficacy and safety of budesonide/formoterol Turbuhaler , compared to budesonide turbuhaler metered dose, in steroid using asthmatic adolescent patients, double blind, double dummy parallel group, Phase III, multicentre study. Department of Paediatrics, Imperial College School of Medicine, Brompton Campus, Dovehouse Street, London, SW3 6LY, United Kingdom). Royal Brompton and Harefield NHS Trust.
Currie {published data only}
- Currie GP. Are there additional anti‐inflammatory effects of leukotriene receptor antagonists in persistent asthmatics receiving inhaled steroid alone and combined inhaled steroid/long acting B2‐agonists?. Academic Publications.
Goves {published data only}
- Goves JR. Eformoterol in the management of mild asthma ‐ eformoterol Turbohaler R with budesonide Turbohaler R . Department of Paediatrics, Level 4 John Radcliffe Hospital, Headley Way, Headington, Oxford, OX3 9DU.
Hill {published data only}
- Hill J. The ASSURE Study ‐ the effectiveness and safety of an individualised Symbicort Turbohaler maintenance dosing regimen (Symbicort Asthma Control Plan) versus Symbicort Turbohaler given as standard regular twice daily therapy. Northern General Hospital NHS Trust.
Kharitonov {published data only}
- Kharitonov SA. Effect of inhaled corticosteroids (budesonide 100 micrograms/400 micrograms vs placebo) and formoterol on localisation of glucocorticoid receptor and exhaled markers in asthma. Royal Brompton and Harefield NHS Trust.
Lipworth 2001 {unpublished data only}
- Lipworth BJ. Relative lung bioavailability with fluticasone via dry powder inhaler versus: (a) fluticasone/salmeterol combination vis dry powder inhaler; (b) fluticasone/salmeterol combination via HFA‐pMDI plus spacer, in patients with moderate persistent asthma. Academic Publications 2001.
Millar {published data only}
- Millar AB. A randomised double‐blind multi‐centre study to evaluate the effect of adding alternative agents to inhaled steroids in adult asthmatics. Lung Research Unit, Southmead Hospital, Southmead Road, Westbury‐on‐Trym, Bristol, BS10 5NB, UK.
Ruggins {published data only}
- Ruggins N. Pragmatic trial of add‐on therapy in paediatric asthma. Southern Derbyshire Acute Hospitals NHS Trust, Consultant Paediatrician, Children's Hospital, Uttoxeter Road, Derby, DE22 3NE, England 2003.
Thomson {published data only}
- Thomson NC. Bundesonide/formoterol fixed combination in adults with asthma. Respiratory Medicine, Gartnavel General Hospital, Great Western Road, Glasgow, G12 0YN, Scotland.
Young {published data only}
- Young KM. Budesonide/formoterol fixed combination in adults with asthma. Cambridge Consortium ‐ Addenbrookes.
Additional references
Adams 2008
- Adams NP, Bestall JC, Lasserson TJ, Jones P, Cates CJ. Fluticasone versus placebo for chronic asthma in adults and children. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD003135.pub4] [DOI] [Google Scholar]
Bateman 2008
Bisgaard 2003
- Bisgaard H. Effect of long acting beta 2 agonists on exacerbation rates of asthma in children. Pediatric Pulmonology 2003;36:391‐8. [DOI] [PubMed] [Google Scholar]
BTS 2007
- British Thoracic Society. British Guideline on the Management of Asthma. www.brit‐thoracic.org.uk 2007.
Canadian Paediatic Asthma Guideline 2005
- Becker A, Bérubé D, Chad Z, Dolovich M, Ducharme F, D'Urzo T, et al. Canadian Pediatric Asthma Consensus Guidelines, 2003 (updated to December 2004). Canadian Medical Association Journal 2005; Vol. 173, issue Suppl 6. [DOI] [PMC free article] [PubMed]
Cates 2002
- Cates CJ. Simpson's paradox and calculation of number needed to treat from meta‐analysis. BioMed Central 2002;BMC Medical Research Methodology 2:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Cates 2008a
- Cates CJ, Cates MJ. Regular treatment with salmeterol for chronic asthma: serious adverse events. Cochrane Database of Systematic Reviews 2008, Issue 3. [DOI: 10.1002/14651858.CD006363.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Cates 2008b
- Cates CJ, Cates MJ, Lasserson TJ. Regular treatment with formoterol for chronic asthma: serious adverse events. Cochrane Database of Systematic Reviews 2008, Issue 4. [DOI: 10.1002/14651858.CD006923.pub2] [DOI] [PubMed] [Google Scholar]
Cates 2009a
- Cates CJ, Lasserson T, Cates MJ. Regular treatment with formoterol and inhaled steroids for chronic asthma: serious adverse events. Cochrane Database of Systematic Reviews 2009, Issue 2. [DOI: 10.1002/14651858.CD006924.pub2] [DOI] [PubMed] [Google Scholar]
Cates 2009b
- Cates CJ, Cates MJ, Lasserson T. Regular treatment with salmeterol and inhaled steroids for chronic asthma: serious adverse events. Cochrane Database of Systematic Reviews 2009, Issue 3. [DOI: 10.1002/14651858.CD006922.pub2] [DOI] [PubMed] [Google Scholar]
Cochrane Handbook
- Higgins JPT, Green S (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.0 [updated February 2008]. The Cochrane Collaboration. .Available from www.cochrane‐handbook.org.
D'Alonzo 1997
- D'Alonzo GE, Tolep KA. Salmeterol in the treatment of chronic asthma. American Family Physician 1997;56(2):558‐62. [PubMed] [Google Scholar]
Deeks 2001
- Deeks JJ, Altman DG, Bradburn MJ. Statistical methods for examining heterogeneity and combining results from several studies in meta‐analysis. In: Egger M, Smith GD, Altman DG editor(s). Systematic reviews in health care: meta‐analysis in context. London: BMJ Publishing, 2001:285‐312. [Google Scholar]
DerSimonian 1986
- DerSimonian R, Laird N. Meta‐analysis in clinical trials. Controlled Clinical Trials 1986;7:177‐88. [DOI] [PubMed] [Google Scholar]
Ducharme 2006
- Ducharme FM, Lasserson TJ, Cates CJ. Long‐acting beta2‐agonists versus anti‐leukotrienes as add‐on therapy to inhaled corticosteroids for chronic asthma. Cochrane Database of Systematic Reviews 2006, Issue 4. [DOI: 10.1002/14651858.CD003137.pub3] [DOI] [PubMed] [Google Scholar]
Ducharme 2010
- Ducharme FM, Ni Chroinin M, Greenstone I, Lasserson TJ. Combination of inhaled long‐acting beta2‐agonists and inhaled steroids versus higher dose of inhaled steroids in children and adults with persistent asthma. Cochrane Database of Systematic Reviews 2010, Issue 1. [DOI: 10.1002/14651858.CD005533.pub2] [DOI] [Google Scholar]
Egger 1997
- Egger M, Smith GD, Schneider M, Minder C. Bias in meta‐analysis detected by a simple, graphical test. BMJ 1997;315:629‐34. [DOI] [PMC free article] [PubMed] [Google Scholar]
Ernst 2006
- Ernst P, McIvor A, Ducharme FM, Boulet L‐P, FitzGerald M, Chapman K, et al. Long‐acting inhaled beta‐agonist bronchodilators are safe and effective in conjunction with inhaled corticosteroids. Annals of Internal Medicine 2006;145(9):693‐4. [DOI] [PubMed] [Google Scholar]
Gibson 2005
- Gibson PG, Powell H, Ducharme F. Long‐acting beta2‐agonists as an inhaled corticosteroid‐sparing agent for chronic asthma in adults and children. Cochrane Database of Systematic Reviews 2005, Issue 4. [DOI: 10.1002/14651858.CD005076.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
GINA 2007
- Global Initiative for Asthma. Global Strategy for Asthma Management and Prevention (NIH publication). http://www.ginasthma.com 2007.
Gleser 1996
- Gleser LJ, Olkin I. Models for estimating the number of unpublished studies. Statistics in Medicine 1996;15:2493‐507. [DOI] [PubMed] [Google Scholar]
Greenland 1985
- Greenland S, Robins JM. Estimation of a common effect parameter from sparse follow‐up data. Biometrics 1985;41:55‐68. [PubMed] [Google Scholar]
Higgins 2003
- Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta‐analyses. BMJ 2003;327(557):560. [DOI] [PMC free article] [PubMed] [Google Scholar]
Manning 2008
- Manning P, Gibson PG, Lasserson TJ. Ciclesonide versus placebo for chronic asthma in adults and children. Cochrane Database of Systematic Reviews 2008, Issue 2. [DOI: 10.1002/14651858.CD006217.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
NAC 2006
- National Asthma Council. Australia. Asthma Management Handbook (http://www.nationalasthma.org.au/publications/amh/amhcont.htm). 5th Edition. Melbourne: National Asthma Campaign, 2006. [Google Scholar]
NAEPP 2007
- National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR 3) Guidelines for the Diagnosis and Management of Asthma. In: NIH Publication. Bethesda, MD: National Heart, Lung and Blood Institute (available at: http://www.nhlbi.nih.gov/guidelines/asthma/index.htm). Bethesda, MD: National Heart, Lung and Blood Institute, 2007. [Google Scholar]
Nelson 1995
- Nelson HS. Beta‐adrenergic bronchodilators. New England Journal of Medicine 1995;333(8):499‐506. [DOI] [PubMed] [Google Scholar]
Nelson 2003
- Nelson HS, Chapman KR, Pyke SD, Johnson M, Pritchard JN. Enhanced synergy between fluticasone propionate and salmeterol inhaled from a single inhaler versus separate inhalers. Journal of Allergy and Clinical Immunology 2003;112(1):29‐36. [DOI] [PubMed] [Google Scholar]
Ni Chroinin 2009a
- Ni Chroinin M, Greenstone I, Lasserson TJ, Ducharme FM. Addition of inhaled long acting beta2‐agonists to inhaled steroids as first line therapy for persistent asthma in steroid naive adults. Cochrane Database of Systematic Reviews 2009, Issue 4. [Art. No.: CD005307. DOI: 10.1002/14651858.CD005307.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ni Chroinin 2009b
- Ni Chroinin M, Lasserson TJ, Greenstone I, Ducharme FM. Addition of long‐acting beta‐agonists to inhaled corticosteroids for chronic asthma in children. Cochrane Database of Systematic Reviews 2009, Issue 3. [DOI: 10.1002/14651858.CD007949] [DOI] [PMC free article] [PubMed] [Google Scholar]
RevMan 2008 [Computer program]
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan). Version 5.0. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2008.
Salpeter 2006
- Salpeter SR, Buckley NS, Ormiston TM, Salpeter EE. Meta‐analysis: effect of long‐acting ß‐agonists on severe asthma exacerbations and asthma‐related deaths. Annals of Internal Medicine 2006;144(12):904‐12. [DOI] [PubMed] [Google Scholar]
SMART
- FDA. Smart study site. www.fda.gov/bbs/topics/ANSWERS/2003/ANS01192.html (accessed 18 May 2009).
Storms 2003
- Storms W. Clinical trials: are these your patients?. Journal of Allergy & Clinical Immunology 2003;5(Suppl):S107‐S111. [DOI] [PubMed] [Google Scholar]
Walters 2007
- Walters EH, Gibson PG, Lasserson TJ, Walters JAE. Long‐acting beta2‐agonists for chronic asthma in adults and children where background therapy contains varied or no inhaled corticosteroid. Cochrane Database of Systematic Reviews 2007, Issue 1. [DOI: 10.1002/14651858.CD001385.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Warner 1998
- Warner JO, Naspitz CK. Third International Pediatric Consensus statement on the management of childhood asthma. International Pediatric Asthma Consensus Group. Pediatric Pulmonology 1998;25(1):1‐17. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Ni Chroinin 2005
- Ni Chroinin M, Greenstone IIG, Ducharme F. Long‐acting beta2‐agonists versus placebo in addition to inhaled corticosteroids for chronic asthma in adults and children. Cochrane Database of Systematic Reviews 2005, Issue 4. [DOI: 10.1002/14651858.CD005535] [DOI] [PubMed] [Google Scholar]


