Abstract
Type 2 inflammation underlies allergic diseases such as atopic dermatitis (AD), which is characterized by the accumulation of basophils and group 2 innate lymphoid cells (ILC2s) in inflamed skin lesions. Although murine studies have demonstrated that cutaneous basophil and ILC2 responses are dependent on thymic stromal lymphopoietin (TSLP), whether these cell populations interact to regulate the development of cutaneous type 2 inflammation is poorly defined. Here, we identify that basophils and ILC2s significantly accumulate in inflamed human and murine skin and form clusters not observed in control skin. We demonstrate that murine basophil responses precede ILC2 responses and that basophils are the dominant IL-4-eGFP-expressing cell type in inflamed skin. Further, basophils and IL-4 were necessary for the optimal accumulation of ILC2s and induction of AD-like disease. We show that ILC2s express the IL-4 receptor alpha (IL-4Rα) and proliferate in an IL-4-dependent manner. In addition, basophil-derived IL-4 was required for cutaneous ILC2 responses in vivo and directly regulated ILC2 proliferation ex vivo. Collectively, these data reveal a previously unrecognized role for basophil-derived IL-4 in promoting ILC2 responses during cutaneous inflammation.
Keywords: basophil, group 2 innate lymphoid cell (ILC2), atopic dermatitis, type 2 inflammation
Introduction
Type 2 cytokine responses promote inflammation associated with allergic rhinitis, food allergy, asthma and atopic dermatitis (AD) that is characterized by IL-4, IL-5 and IL-13 production, induction of IgE and recruitment of granulocytes (1-4). The epithelial cell-derived cytokine thymic stromal lymphopoietin (TSLP) inititates type 2 immune responses via effects on multiple cellular targets including CD4+ T cells, dendritic cells (DCs), basophils and group 2 innate lymphoid cells (ILC2s) (5-12). Polymorphisms in TSLP are associated with AD in humans (13, 14) and TSLP expression is elevated in lesional skin and sera of AD patients (15, 16). In mice, TSLP-TSLP receptor (TSLPR) interactions promote the development of AD-like disease (17-20), supporting a role for TSLP in the pathogenesis of human and murine skin inflammation.
Recently, we demonstrated that murine basophils and ILC2s accumulate in inflamed AD-like skin lesions in a TSLP-dependent manner and contribute to type 2 cytokine-associated inflammation (9, 20). Basophils lack expression of cell lineage markers associated with T and B cells, DCs, macrophages, and other granulocytes, but express FcεRIα and CD49b (21). Functionally, basophils express high levels of IL-4 in vivo and promote the accumulation of other innate cells such as eosinophils in the context of chronic allergic dermatitis (9, 21, 22). ILC2s also lack expression of lineage markers but can be identified by the expression of CD25 and IL-33R (3). In contrast to basophils, which predominately express IL-4, ILC2s express IL-5 and IL-13 (23-26). The differential effector cytokine expression profiles of basophils and ILC2s define their specialized functions in vivo (25), but whether functional interactions or cross-regulation occurs between basophils and ILC2s remains unknown.
Here, we demonstrate that basophils and ILC2s accumulate in close proximity to each other in the dermis of inflamed skin lesions isolated from AD patients and in AD-like murine lesions. Quantification of basophil-ILC2 clusters demonstrated a significant accumulation of these clusters in AD-associated skin in comparison to healthy control skin. Temporal analyses revealed that the accumulation of basophils in murine skin precedes that of ILC2s in the context of AD-like inflammation. Further, loss- and gain-of-function studies demonstrated that basophils are required to promote cutaneous ILC2 responses in vivo. Strikingly, employing IL-4-eGFP (4get) reporter mice, we found that basophils were the dominant source of IL-4-eGFP expression in AD-like skin. Both basophils and IL-4 were necessary for the optimal accumulation of ILC2s and induction of AD-like disease. Further examination of ILC2s revealed cell surface expression of the IL-4 receptor alpha (IL-4Rα) and their dependence on IL-4 for proliferation in AD-like inflammation. Basophil-derived IL-4 was required for cutaneous ILC2 responses in vivo and directly regulated ILC2 proliferation ex vivo. Collectively, these studies identify that basophils are early regulators of ILC2 responses in the context of cutaneous type 2 inflammation and AD-like disease.
Materials and Methods
Patients and samples
Human skin samples were assessed as previously described for flow cytometry and basophils were defined as CD123+ FcεRIα+ cells negative for expression of CD3, TCRαβ, CD11c, CD19, CD56 and c-Kit, while ILC2s were defined as CD25+ IL-33R+ cells negative for expression of lineage (CD3, CD5, CD11b, CD11c, CD19, CD56 and FcεRIα) markers (10, 20). Experiments using human skin samples were approved by the University of Pennsylvania IRB (Protocol #814945).
Mice and samples
C57BL/6 wild-type (WT), BALB/c WT, 4get, Rag1−/− and Il4−/− mice were purchased from the Jackson Laboratory. Il4rα−/− mice were purchased from Taconic. BaS-TRECK (BaS) mice were provided by Dr. M. Kubo (Tokyo). All mice were treated with MC903 as previously described (9, 20, 27). Murine skin samples were assessed as previously described for flow cytometry and basophils were defined as CD49b+ FcεRIα+ cells negative for expression of CD3, CD5, CD11c, CD19, NK1.1 and c-Kit, while ILC2s were defined as CD25+ IL-33R+ cells negative for expression of lineage (CD3, CD5, CD11b, CD11c, B220, NK1.1 and FcεRIα) markers (10, 20). Splenic basophils were sort-purified from TSLP cDNA plasmid-treated WT or Il4−/− mice using a BD FACS Aria cell sorter, 3 weeks post-TSLP cDNA plasmid injection as previously described (9). TSLP cDNA plasmid was provided by M.R. Comeau. WT and BaS mice were treated with diphtheria toxin (D.T.) as previously described (9). Basophils (10,000 cells) were suspended in 50 μL of PBS and injected intradermally (i.d.) into naïve WT mice. WT mice were treated with 300 ng of recombinant murine (rm)IL-33 (R&D Systems) daily in 200 μL of PBS intraperitoneally (i.p.) for seven days prior to sort-purification of ILC2s on a BD FACS Arial cell sorter. ILC2s were sort-purified from pooled skin-draining lymph nodes, mesenteric lymph nodes, peritoneal cavity and adipose tissue as previously described (20, 28). Annexin V, 7-AAD, KLRG1 and Ki67 staining of ILC2s was performed as previously described (29-31). Experiments were performed according to the guidelines of the University of Pennsylvania IACUC.
Histology
For all human and murine immunofluorescence (IF) microscopy, paraffin-embedded 5-μm skin sections were incubated with primary antibody at 4°C overnight, followed by incubation with secondary antibodies at 37°C for 30 minutes. For human samples, primary antibodies against 2D7 (1:250, BioLegend, Ab mouse IgG1), IL-33R (1:250, MD Bioproducts, biotin-conjugated mouse IgG1,) or CD3 (1:50, Dako, rabbit IgG) and secondary antibodies to mouse IgG conjugated with PE-Cy5 (1:300), streptavidin-Cy3 (1:300) or rabbit IgG conjugated with Cy2 (1:300), respectively, were used. Murine samples were stained with hemotoxylin and eosin (H&E), or primary antibodies against Mcpt8 (1:500, TUG8, BioLegend, rat IgG2) or IL-33R (1:300, DJ8, MD Bioproducts, biotin-conjugated rat IgG1) and secondary antibodies to rat IgG conjugated with PE-Cy5 (1:300) and streptavidin-Cy3 (1:300), respectively, were used. Sections were placed in mounting medium with DAPI (Vectashield).
Histology score and ear thickness measurements
Histology score was determined by the following formula: total number of lymphocytes per high power field (HPF) + thickness of the epidermis measured in microns from the basement membrane to the top of the stratum corneum divided by 100. The ear thickness was measured on day 7 following treatment by measuring the entire width of the ear at the widest point in microns from the top of one stratum corneum to the top of the stratum corneum on the opposite side of the ear. All measurements were performed using the NIH ImageJ program.
In vitro/ex vivo cultures
Sort-purified basophils from the spleens of TSLP cDNA plasmid-treated WT and Il4−/− mice were cultured for 24 hours at 37°C in supplemented DMEM with rmIL-3 (10 ng/mL, R&D Systems) as previously described (9). CFSE-labeled ILC2s (20,000 cells) from rmIL-33-treated mice were then cultured in 200 μL of WT and Il4−/− basophil-conditioned media for four days at 37°C with rmIL-7 (R&D Systems) as previously described (20), and cell division was assessed by FACS analysis of dye dilution as previously described (32).
Basophil-ILC2, basophil-T cell and ILC2-T cell cluster analysis
Basophil-ILC2 (yellow circle), basophil-T cell (green circles) and ILC2-T cell (red circle) clusters were analyzed by drawing 25 μm colored circles around the cell-cell clusters per HPF.
Statistical analysis
Data are means ± SEM unless indicated otherwise. Statistical significance was determined by unpaired Student’s t-test or one-way ANOVA for multiple comparisons using GraphPad Prism 6 software. Actual P-values are denoted for each analysis.
Results
Basophils and ILC2s are enriched and found in close proximity to each other in the dermis of human AD lesions
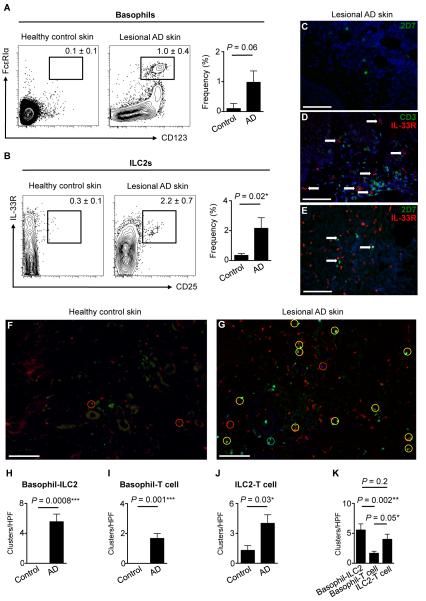
We recently reported that murine basophils and ILC2s are elicited in a TSLP-dependent manner during AD-like inflammation (9, 20). Consistent with this, basophils have been identified in skin lesions from AD patients, and we recently showed that ILC2s are enriched in the inflamed skin of patients (20, 33). However, whether basophils and ILC2s accumulate in the same lesions and whether these cell types co-localize in inflamed human skin has not been examined. To address this, four millimeter (mm) punch biopsies of normal and diseased human skin were obtained from healthy control subjects and AD patients. Strikingly, frequencies of CD123+ FcεRIα+ basophils (Fig. 1A) and lineage-negative (Lin−) CD25+ IL-33R+ ILC2s (Fig. 1B) were markedly elevated in lesional human AD skin, indicating that basophil and ILC2 responses occur simultaneously in the same inflamed lesion.
Figure 1.
Basophils and ILC2s are enriched in human AD skin lesions. (A) Skin basophils in healthy human control and lesional AD skin. Healthy human control skin n = 6, lesional AD skin n = 7. Basophils were gated on and noted as a percentage of CD3-CD19− CD11c− CD56− c-Kit− cells. (B) Skin ILC2s in healthy human control and lesional AD skin. Healthy human control skin n = 4, lesional AD skin n = 3. ILC2s were gated on lineage (Lin)-negative cells. Lin markers included CD3, TCRαβ, CD19, CD11b, CD11c, CD56 and FcεRIα. Cell frequencies are noted as a percentage of Lin− cells. (C) Immunofluorescence (IF) staining of 2D7+ basophils (green), (D) IL-33R+ ILC2s (red, white arrows) and CD3+ T cells (green) and (E) 2D7+ basophils (green) and IL-33R+ ILC2s (red) cells in lesional AD skin. DAPI+ cells are blue. Lesional AD skin n = 14. (F) IF staining of 2D7+ basophils (green), IL-33R+ ILC2s (red) and CD3+ T cells (blue) in healthy control and (G) lesional AD skin. Clusters per high power field (HPF) of (H) basophil-ILC2 (yellow circle), (I) basophil-T cell (green circle) and (J) ILC2-T cell (red circle) clusters in healthy control and lesional AD skin. (K) Basophil-ILC2, basophil-T cell and ILC2-T cell clusters per HPF in lesional AD skin. Scale bars, 100 μm. Healthy human control skin tissue n = 4, lesional AD skin n = 9. Actual P-values are indicated.
To visualize the distribution of cutaneous basophils and ILC2s, immunofluorescence (IF) microscopy was employed. Basophils were identified by staining with an Ab against the basophil-specific granule Ag 2D7 (34). Consistent with the flow cytometric analyses (Fig. 1A), 2D7+ basophils were undetectable in healthy control skin (Supplemental Fig. 1A) but were readily identified in lesional AD skin (Fig. 1C and Supplemental Fig. 1B). To visualize ILC2s and identify non-T cell populations that express the IL-33R, sections were co-stained with anti-CD3 Ab and anti-IL33R Ab. Although IL-33R+ cells and CD3+ T cells were rarely identified in healthy control skin (Supplemental Fig. 1C), both were enriched in lesional AD skin and accumulated in a perivascular distribution in the dermis (Fig. 1D and Supplemental 1D). The majority of IL-33R+ cells were negative for CD3 (Fig. 1D, white arrows), suggesting that these IL-33R+ cells are ILC2s in this context. Finally, 2D7+ basophils and IL-33R+ ILC2s accumulated in close proximity to each other in the papillary dermis in lesional human AD skin compared to healthy control skin (Fig. 1E, 1F, 1G and Supplemental Fig. 1E, 1F). Although our analyses demonstrated that some basophils express both 2D7 and IL-33R (data not shown), the vast majority of basophils were single-positive (2D7+ IL-33R−) by IF (Fig. 1E and Supplemental Fig 1F). Together, analyses of the flow cytometry data and microscopy images indicate that basophils and ILC2s are enriched in the dermis of lesional skin from AD patients and accumulate in close proximity in inflamed lesions.
To quantify basophil-ILC2 clusters in the skin, 2D7+ cell-IL-33R+ cell groupings were counted in both healthy control and lesional AD skin (Fig. 1F, 1G, 1H). Enumeration of these groupings identified a marked and significant increase of basophil-ILC2 clusters in the inflamed skin (Fig. 1H). Additional analyses of CD3+ T cells and their clustering with basophils and ILC2s identified a significant increase of both basophil-T cell clusters (Fig. 1I) as well as ILC2-T cell clusters (Fig. 1J). A three-way comparison of basophil-ILC2, basophil-T cell and ILC2-T cell clusters demonstrated that basophil-ILC2 and ILC2-T cell clusters are more numerous than basophil-T cell clusters (Fig. 1K). Taken together, these findings provoke the hypothesis that basophils, ILC2s and T cells interact in vivo.
Cutaneous basophil responses precede ILC2 responses in AD-like inflammation
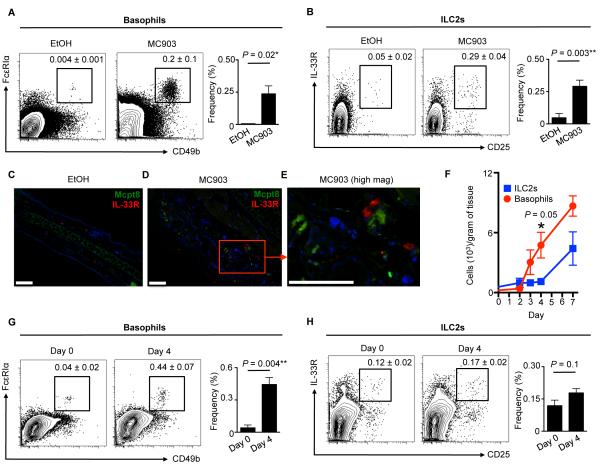
To test whether murine basophils and ILC2s also accumulate in murine AD-like skin lesions, a previously established mouse model of AD-like disease was employed (19, 20) in which wild-type (WT) mice were treated topically with vehicle (ethanol, EtOH) or a vitamin D analog (MC903) (20). On day 7 of treatment, flow cytometric analysis revealed that CD49b+ FcεRIα+ basophils (Fig. 2A) and Lin− CD25+ IL-33R+ ILC2s (Fig. 2B) were significantly enriched in AD-like skin lesions compared to control skin. Similar to results obtained using human samples, IF microscopy revealed that basophils (identified by staining with anti-Mcpt8 Ab) could not be detected and ILC2s (identified by staining with anti-IL-33R Ab) were rare in control EtOH-treated healthy murine skin (Fig. 2C). In contrast, Mcpt8+ basophils and IL-33R+ innate cells co-localized in clusters within the papillary dermis in AD-like lesional skin from MC903-treated lymphocyte-deficient Rag1−/− mice (Fig. 2D, 2E). Collectively, these findings suggest that basophils and ILC2s reside in close proximity in inflamed human and murine skin.
Figure 2.
Basophils and ILC2s are enriched in murine AD-like skin lesions. (A) Skin basophils and (B) ILC2s in control vehicle (ethanol, EtOH) or MC903-treated wild-type (WT) mice. Cell frequencies are noted as a percentage of Lin− cells. (C) IF staining of Mcpt8+ basophils (green) and IL-33R+ ILC2s (red) cells in EtOH-treated and (D) MC903-treated Rag1−/− mice at low and (E) high magnification. DAPI+ cells are blue. Scale bars, 100 μm. All mice were treated topically daily for 7 days. (F) Skin basophils and ILC2s in MC903-treated WT mice on days 0, 2, 3, 4 and 7. (G) Skin basophils and (H) ILC2s in MC903-treated WT mice on days 0 and 4. Cell frequencies are noted as a percentage of Lin− cells. Data are representative of three independent experiments; n = 3 mice per group per experiment. Actual P-values are indicated.
While basophils and ILC2s accumulate in close proximity to each other in the inflamed skin of humans and mice, whether functional interactions or cross-regulation occurs between these cell populations is unknown. To examine this, we performed temporal analysis of cutaneous basophil and ILC2 responses following elicitation of AD-like disease. By day 4 post-MC903 treatment, cutaneous basophil responses were markedly elevated (Fig. 2F, 2G). In contrast, frequencies and absolute numbers of ILC2s were not significantly increased in MC903-treated mice at this early time point relative to controls (Fig. 2F, 2H) and did not increase in the skin until day 7 post-treatment (Fig. 2F), indicating that cutaneous basophil responses precede ILC2 responses.
ILC2 responses are dependent on basophils in the context of skin inflammation
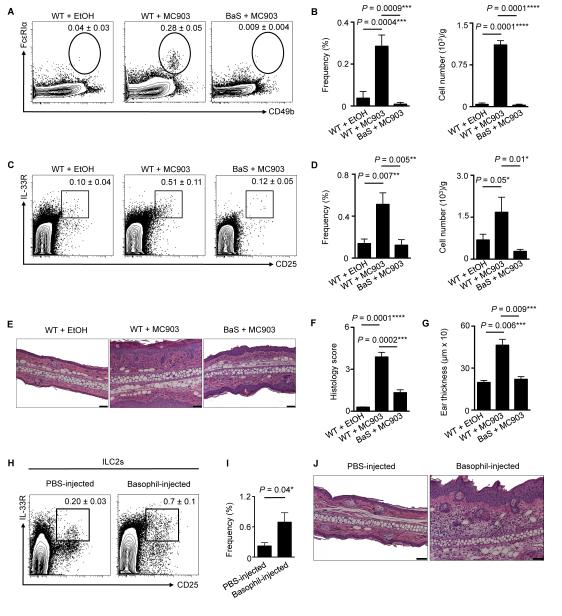
To test whether early cutaneous basophil responses influence ILC2 responses in vivo, we employed BaS-TRECK (BaS) mice, which harbor basophils that selectively express the simian diphtheria toxin receptor (DTR) (35). Treatment of BaS mice with diphtheria toxin (D.T.) results in selective depletion of cutaneous basophils during inflammation (9, 10). Treatment of BaS mice with both MC903 and D.T. resulted in effective depletion of basophils in the inflamed skin (Fig. 3A, 3B), which was associated with a reduction in skin ILC2 responses (Fig. 3C, 3D), CD4+ T cell responses (Supplemental Fig. 2A, 2B), histological findings of epidermal hyperplasia (acanthosis), thickening of the stratum corneum (hyperkeratosis) and dermal inflammation (Fig. 3E, 3F), and ear thickness (Fig. 3G). These data suggest that basophil responses precede, and are necessary for, ILC2 responses in the context of skin inflammation.
Figure 3.
Skin ILC2 responses are dependent on basophils. (A) Skin basophils and associated (B) frequencies and total cell numbers in WT mice treated with EtOH or MC903 and BaS-TRECK (BaS) mice treated with MC903. (C) Skin ILC2s and associated (D) frequencies and total cell numbers and (E) histopathology, (F) histology score and (G) ear thickness on day 7 in WT mice treated with EtOH or MC903 and BaS mice treated with MC903. (H) Skin ILC2s and associated (I) frequencies and (J) histopathology on day 4 following intradermal (i.d.) injection of PBS or sort-purified basophils from TSLP cDNA-injected WT mice. Scale bars, 50 μm. Cell frequencies are noted as a percentage of Lin− cells. Data are representative of three independent experiments; n = 3-4 mice per group per experiment. Actual P-values are indicated.
To test whether basophils are sufficient to promote skin-associated ILC2 responses and AD-like inflammation, gain-of-function studies were performed. Basophils were sort-purified from WT mice and adoptively transferred by intradermal (i.d.) injection into the skin of naïve WT mice. Flow cytometric analysis of the skin of basophil-injected mice revealed elevated frequencies of ILC2s (Fig. 3H, 3I) and the induction of AD-like disease, histologically characterized by a mixed dermal inflammatory infiltrate associated with acanthosis and hyperkeratosis (Fig. 3J). These findings indicate that basophils are sufficient for the induction of ILC2 responses and AD-like skin inflammation.
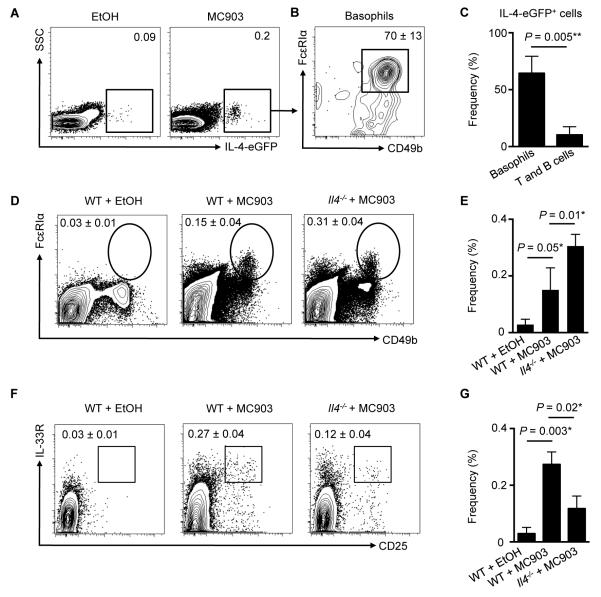
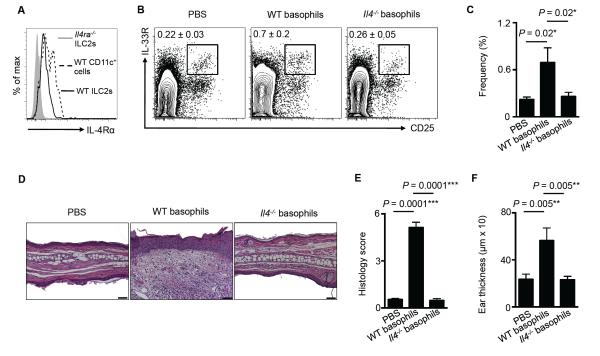
Multiple studies indicate that basophils promote type 2 inflammation via production of IL-4 (25, 36-38). Employing IL-4-eGFP reporter (4get) mice, we observed elevated IL-4-eGFP+ cells following MC903 treatment (Fig. 4A), and ~70% of IL-4-eGFP+ cells were basophils (Fig. 4B), while only ~12% were T (CD3+ CD5+) and B (CD19+) cells (data not shown, Fig. 4C). These data provoked the hypothesis that IL-4 may be a key factor through which basophils influence ILC2 responses in the context of skin inflammation. To directly test this, WT or Il4−/− mice were treated topically daily with MC903, and the skin was examined by flow cytometry on day 7. Basophil accumulation was unimpaired in Il4−/− mice compared to WT mice (Fig. 4D, 4E), but skin ILC2 responses were significantly reduced following induction of AD-like inflammation (Fig. 4F, 4G). Flow cytometric analysis revealed that cutaneous ILC2s from WT mice expressed the IL-4Rα (Fig. 5A), suggesting that ILC2s are capable of responding to basophil-derived IL-4. Finally, to test whether basophil-derived IL-4 was required to induce cutaneous ILC2 responses, basophils were sort-purified from WT or Il4−/− mice and injected i.d. into the skin of naïve WT mice. On day 4 following transfer of basophils, WT basophils induced local skin ILC2 responses while IL-4-deficient basophils did not (Fig. 5B, 5C). In association with decreased ILC2 responses in the skin, WT mice injected with Il4−/− basophils exhibited reduced inflammation as determined by histology (Fig. 5D, 5E) and ear thickness (Fig. 5F). Collectively, these findings indicate that skin ILC2 responses and AD-like disease are dependent on basophil-derived IL-4.
Figure 4.
Skin ILC2 responses are dependent on IL-4. (A) Skin IL-4-eGFP+ cells and (B) basophils as a percentage of IL-4-eGFP+ cells in MC903-treated 4get mice. (C) Frequencies of skin basophils and T and B cells as a percentage of IL-4-eGFP+ cells. (D) Skin basophils and associated (E) frequencies on day 7 in WT mice treated with EtOH or MC903 and Il4−/− mice treated with MC903. (F) Skin ILC2s and associated (G) frequencies on day 7 in WT mice treated with EtOH or MC903 and Il4−/− mice treated with MC903. Data are representative of three independent experiments; n = 3-4 mice per group per experiment. Actual P-values are indicated.
Figure 5.
Basophil-derived IL-4 is necessary for the induction of skin ILC2 responses and AD-like disease. (A) Expression of IL-4Rα on ILC2s from WT and Il4ra−/− mice and CD11c+ dendritic cells (DCs) from WT mice. Data are representative of five individual mice. (B) Skin ILC2s in WT mice on day 4 following i.d. injection of PBS, or sort-purified basophils from TSLP cDNA-injected WT or Il4−/− mice and associated (C) frequencies, (D) histopathology, (E) histology score and (F) ear thickness. Scale bars, 50 μm. Cell frequencies are noted as a percentage of Lin− cells. Data are representative of three independent experiments; n = 4 mice per group per experiment. Actual P-values shown.
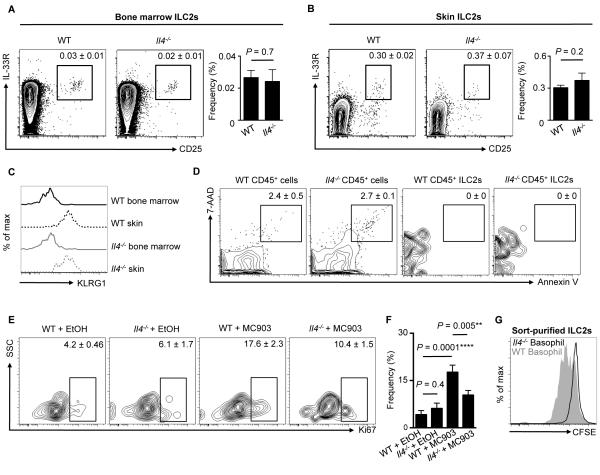
Although IL-4 critically promotes basophil-mediated cutaneous ILC2 responses and AD-like disease (Fig. 5), how IL-4 influences ILC2 function remains unclear. To test this, we employed Il4−/− mice that lack endogenous IL-4, and examined ILC2 development, survival and proliferation. Examination of the bone marrow (BM) and the skin of WT versus Il4−/− mice in the steady state revealed no significant differences in the frequencies of ILC2s between WT and Il4−/− mice (Fig 6A, 6B). In addition, consistent with a progenitor phenotype, ILC2s in the BM of both WT and Il4−/− mice lacked expression of KLRG1 (30), while skin ILC2s expressed KLRG1, indicating a mature phenotype (Fig. 6C). Taken together, these studies suggest that there are no defects in ILC progenitors in the BM or the skin between WT and Il4−/− mice. To test whether there was differential survival of ILC2s in WT versus Il4−/− mice, ILC2s in the skin were stained with 7-AAD and Annexin V to assess apoptosis. In contrast to small populations of 7-AAD+ Annexin V+ cells in the CD45+ pan-leukocyte compartment in both WT and Il4−/− mice, both 7-AAD and Annexin V were not detected on ILC2s (Fig. 6D). These studies indicate that there is no enhancement of ILC2 apoptosis in Il4−/− mice.
Figure 6.
Basophil-derived IL-4 promotes ILC2 proliferation. (A) Bone marrow (BM) and (B) skin ILC2 frequencies in WT and Il4−/− mice. Cell frequencies are noted as a percentage of Lin− cells. (C) KLRG1 expression on BM ILC2s from WT (solid black line) and Il4−/− (solid gray line) mice and skin ILC2s from WT (dashed black line) and Il4−/− (dashed gray line) mice. (D) Annexin V and 7-AAD expression on all CD45+ cells and CD45+ ILC2s from the skin of WT and Il4−/− mice. Cell frequencies are noted as a percentage of Lin− cells. (E) Ki67 expression on skin ILC2s from WT and Il4−/− mice treated with EtOH or MC903 and associated (F) frequencies. Cell frequencies are noted as a percentage of ILC2s. Data are representative of two independent experiments; n = 3-4 mice per group per experiment. (G) CFSE dilution of sort-purified CFSE-labeled ILC2s stimulated with WT (solid gray histogram) and Il4−/− (black line) TSLP-elicited basophil supernatants. Data are representative of two independent experiments; n = 3 mice per group per experiment. Actual P-values shown.
To examine whether there were any differences in ILC2 proliferation, both WT and Il4−/− mice were treated with topical MC903 for seven days, and proliferation was assessed by Ki67 staining. Examination of the skin revealed proliferation of ILC2s in WT mice that was significantly diminished in Il4−/− compared to WT mice (Fig. 6E, 6F), suggesting that IL-4 supports ILC2 proliferation. Finally, to test whether basophil-derived IL-4 is necessary for the proliferation of ILC2s, TSLP-elicited basophils were sort-purified from WT and Il4−/− mice and stimulated in vitro with rmIL-3. Sort-purified CFSE-labeled ILC2s were then cultured in supernatants from either WT or Il4−/− basophils for four days. Strikingly, supernatants from WT basophils promoted the proliferation of ILC2s as measured by CFSE dilution while supernatants from Il4−/− basophils did not (Fig. 6G). Collectively, these studies indicate that, although there are no detectable defects in ILC2 progenitor capacity or cell survival in the steady state, basophil-derived IL-4 is critical for ILC2 proliferation in the context of skin inflammation.
Discussion
Recent studies demonstrated that ILC2s critically promote AD-like inflammation and significantly accumulate in lesional skin of AD patients (20, 39, 40). Additionally, basophils have also been shown to contribute to type 2 inflammation in the skin and accumulate in skin lesions of AD patients (9, 33). We have previously shown that TSLP elicits both basophils (9) and ILC2s (20) early in the context of AD-like inflammation. Despite these associations, whether functional interactions or cross-regulation occurs between these cell lineages has never been tested.
The present study provides three conceptual advances that broaden our understanding of the function and regulation of basophils and ILC2s. First, we show that both basophil and ILC2 populations accumulate in close proximity in human and murine inflamed skin. Second, we demonstrate a previously unrecognized role for basophils in promoting ILC2 responses in the skin. Third, we identify that basophil-derived IL-4 is necessary for the induction of ILC2 responses, optimal proliferation of ILC2s and AD-like inflammation. Collectively, these findings indicate that coordinated interactions between basophils and ILC2s regulate skin inflammation.
Basophils have been implicated in both human AD and shown to be initiators of chronic allergic dermatitis in mice (22, 33, 41). Further, we have shown that TSLP-elicited basophils promote local TH2 cell responses in the context of AD-like disease and promote epicutaneous sensitization to food allergens (9, 10, 41). However, the mechanisms by which basophils contribute to type 2 cytokine-associated inflammation in the skin has been poorly defined. In the current study, we identify a previously unrecognized basophil-ILC2 axis in the elicitation of skin inflammation.
Emerging evidence indicates that ILC2s can interact with multiple cell populations to influence type 2 inflammation (3). We have previously shown that adoptive transfer of ILC2s can induce local TH2 cell responses and AD-like disease (20). Recent studies have also demonstrated that this process can be mediated by ILC2-derived IL-13 influencing DC migration and/or contact-dependent stimulation of T cells by MHC class II or other factors (32, 42). Taken together, these studies highlight a role for ILC2s in promoting innate and adaptive immune responses. Further, Roediger et al. employed two-photon intravital microscopy in the skin to show that ILC2s suppress the function of other granulocytes such as mast cells in vivo (43). However, whether other upstream innate cellular mechanisms influence ILC2 function in the context of AD was not known. We now show that basophils can directly influence ILC2 function through the production of IL-4. Therefore, although ILC2s can be activated directly by epithelial cell-derived cytokines (3), it appears that hematopoietic cell-derived cytokines can also influence ILC2 function in the context of inflammation.
It has been appreciated that IL-4 is highly expressed in lesional AD skin and contributes to the pathogenesis of AD (44-46). Further, other common gamma chain (γc) cytokines, such as IL-2 and IL-7, have shown to be critical for the development and survival of ILCs (43, 47). However, we found that IL-4 was dispensable for these processes, but essential to promote ILC2 proliferation both in vitro and in vivo. This is consistent with the role of IL-4 in supporting the growth and proliferation multiple other cell types such as T cells and B cells (48, 49). However, our studies do not preclude other mechanisms by which basophils and IL-4 can directly promote ILC2 function. For example, basophil-derived IL-4 may influence the responsiveness of ILC2s to epithelial cell-derived cytokines such as IL-25, IL-33 and TSLP (3), or influence the activation and/or recruitment of ILC2s into inflamed skin. Motomura et al. recently demonstrated a role for basophils in regulating ILC2 activation in the context of protease-induced airway inflammation (48). These findings, coupled with the present study suggest that the capacity of basophils to promote ILC2 responses may be a conserved pathway at multiple barrier surfaces.
In addition to IL-4, basophils produce a variety of factors such as pro-inflammatory eicosanoids that can influence the function of other immune cells which promote type 2 inflammation (21). Therefore, other endogenous factors derived from basophils may act in concert with IL-4 to modulate ILC2 function. In support of this, recent studies have shown that both murine and human ILC2s directly respond to leukotrienes (50, 51). Moreover, both IL-33 and TSLP are known to differentially regulate the production of cytokines and chemokines from basophils (9). However, how the differential regulation of basophils influences ILC2 function and type 2 inflammation remains to be fully defined. Notwithstanding this, this study demonstrates that TSLP-elicited basophils and IL-4 production promote cutaneous ILC2 responses and proliferation, suggesting that selective targeting of TSLP and TSLP-dependent basophils could prove to be a beneficial strategy in the treatment of AD and related allergic diseases.
Supplementary Material
Acknowledgements
We thank members of the Artis laboratory for discussions and reading of the manuscript, M.R. Comeau (Amgen) for providing TSLP cDNA plasmid, C.J. Miller, J. Sobanko, X. Zhu, T. Nunnciato, N. Manogue, and A.S. Payne for providing healthy human control samples through the Skin Disease Research Center (SDRC) Core B, A.S. Van Voorhees and W.D. James for clinical support, and J. Molleston and S. Cherry for microscopy resources. We also thank Mary Connolly Braun, the Arguild Foundation and the Laffey-McHugh Foundation for generous support of these studies.
Footnotes
Disclosures The authors have no financial conflicts of interest.
Publisher's Disclaimer: This work was supported by the NIH (AI061570, AI087990, AI074878, AI106697, AI095466, AI095608, AI102942 and AI097333 to D.A.; T32-AR007465-28, 5-P30-AR-057217, KL2-RR024132 and K08-AR065577-01 to B.S.K; AI085828 and K22-AI110573 to M.C.S; T-32-A106516 and F30-AI112023 to J.R.B.; T32-AI007532 to L.A.M., F32-AI098365 to E.D.T.W.) and the Burroughs Wellcome Fund. B.S.K. and K.W. were generously supported by Mary Connolly Braun, the Arguild Foundation and the Laffey-McHugh Foundation. K.W. was supported by a Benjamin Franklin Society Research Grant. T.C.F. was supported by the Cancer Research Institute Student Training and Research in Tumor Immunology Grant. M.N. was supported by the Swiss National Science Foundation Prospective Research Fellowship. This work was also supported by the National Center for Research Resources and the National Center for Advancing Translational Sciences, NIH (KL2TR000139) and the Skin Disease Research Center, NIH (through grant SDRC 5-P30-AR-057217).
References
- 1.Paul WE, Zhu J. How are T(H)2-type immune responses initiated and amplified? Nat Rev Immunol. 2010;10:225–235. doi: 10.1038/nri2735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kaplan MH. Th9 cells: differentiation and disease. Immunol Rev. 2013;252:104–115. doi: 10.1111/imr.12028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kim BS, Wojno ED, Artis D. Innate lymphoid cells and allergic inflammation. Curr Opin Immunol. 2013 doi: 10.1016/j.coi.2013.07.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Licona-Limon P, Kim LK, Palm NW, Flavell RA. TH2, allergy and group 2 innate lymphoid cells. Nat Immunol. 2013;14:536–542. doi: 10.1038/ni.2617. [DOI] [PubMed] [Google Scholar]
- 5.Watanabe N, Hanabuchi S, Soumelis V, Yuan W, Ho S, de Waal Malefyt R, Liu YJ. Human thymic stromal lymphopoietin promotes dendritic cell-mediated CD4+ T cell homeostatic expansion. Nat Immunol. 2004;5:426–434. doi: 10.1038/ni1048. [DOI] [PubMed] [Google Scholar]
- 6.Ito T, Wang YH, Duramad O, Hori T, Delespesse GJ, Watanabe N, Qin FX, Yao Z, Cao W, Liu YJ. TSLP-activated dendritic cells induce an inflammatory T helper type 2 cell response through OX40 ligand. J Exp Med. 2005;202:1213–1223. doi: 10.1084/jem.20051135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang YH, Ito T, Homey B, Watanabe N, Martin R, Barnes CJ, McIntyre BW, Gilliet M, Kumar R, Yao Z, Liu YJ. Maintenance and polarization of human TH2 central memory T cells by thymic stromal lymphopoietin-activated dendritic cells. Immunity. 2006;24:827–838. doi: 10.1016/j.immuni.2006.03.019. [DOI] [PubMed] [Google Scholar]
- 8.Omori M, Ziegler S. Induction of IL-4 expression in CD4(+) T cells by thymic stromal lymphopoietin. Journal of immunology. 2007;178:1396–1404. doi: 10.4049/jimmunol.178.3.1396. [DOI] [PubMed] [Google Scholar]
- 9.Siracusa MC, Saenz SA, Hill DA, Kim BS, Headley MB, Doering TA, Wherry EJ, Jessup HK, Siegel LA, Kambayashi T, Dudek EC, Kubo M, Cianferoni A, Spergel JM, Ziegler SF, Comeau MR, Artis D. TSLP promotes interleukin-3-independent basophil haematopoiesis and type 2 inflammation. Nature. 2011;477:229–233. doi: 10.1038/nature10329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Noti M, Wojno ED, Kim BS, Siracusa MC, Giacomin PR, Nair MG, Benitez AJ, Ruymann KR, Muir AB, Hill DA, Chikwava KR, Moghaddam AE, Sattentau QJ, Alex A, Zhou C, Yearley JH, Menard-Katcher P, Kubo M, Obata-Ninomiya K, Karasuyama H, Comeau MR, Brown-Whitehorn T, de Waal Malefyt R, Sleiman PM, Hakonarson H, Cianferoni A, Falk GW, Wang ML, Spergel JM, Artis D. Thymic stromal lymphopoietin-elicited basophil responses promote eosinophilic esophagitis. Nat Med. 2013;19:1005–1013. doi: 10.1038/nm.3281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Halim TY, Krauss RH, Sun AC, Takei F. Lung natural helper cells are a critical source of Th2 cell-type cytokines in protease allergen-induced airway inflammation. Immunity. 2012;36:451–463. doi: 10.1016/j.immuni.2011.12.020. [DOI] [PubMed] [Google Scholar]
- 12.Mjosberg J, Bernink J, Golebski K, Karrich JJ, Peters CP, Blom B, te Velde AA, Fokkens WJ, van Drunen CM, Spits H. The transcription factor GATA3 is essential for the function of human type 2 innate lymphoid cells. Immunity. 2012;37:649–659. doi: 10.1016/j.immuni.2012.08.015. [DOI] [PubMed] [Google Scholar]
- 13.Gao PS, Rafaels NM, Mu D, Hand T, Murray T, Boguniewicz M, Hata T, Schneider L, Hanifin JM, Gallo RL, Gao L, Beaty TH, Beck LA, Leung DY, Barnes KC. Genetic variants in thymic stromal lymphopoietin are associated with atopic dermatitis and eczema herpeticum. J Allergy Clin Immunol. 2010;125:1403–1407. e1404. doi: 10.1016/j.jaci.2010.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Margolis DJ, Kim B, Apter AJ, Gupta J, Hoffstad O, Papadopoulos M, Mitra N. Thymic Stromal Lymphopoietin Variation, Filaggrin Loss of Function, and the Persistence of Atopic Dermatitis. JAMA Dermatol. 2014 doi: 10.1001/jamadermatol.2013.7954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Soumelis V, Reche PA, Kanzler H, Yuan W, Edward G, Homey B, Gilliet M, Ho S, Antonenko S, Lauerma A, Smith K, Gorman D, Zurawski S, Abrams J, Menon S, McClanahan T, de Waal-Malefyt Rd R, Bazan F, Kastelein RA, Liu YJ. Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nat Immunol. 2002;3:673–680. doi: 10.1038/ni805. [DOI] [PubMed] [Google Scholar]
- 16.Lee EB, Kim KW, Hong JY, Jee HM, Sohn MH, Kim KE. Increased serum thymic stromal lymphopoietin in children with atopic dermatitis. Pediatr Allergy Immunol. 2010;21:e457–460. doi: 10.1111/j.1399-3038.2009.00919.x. [DOI] [PubMed] [Google Scholar]
- 17.Yoo J, Omori M, Gyarmati D, Zhou B, Aye T, Brewer A, Comeau MR, Campbell DJ, Ziegler SF. Spontaneous atopic dermatitis in mice expressing an inducible thymic stromal lymphopoietin transgene specifically in the skin. J Exp Med. 2005;202:541–549. doi: 10.1084/jem.20041503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.He R, Oyoshi MK, Garibyan L, Kumar L, Ziegler SF, Geha RS. TSLP acts on infiltrating effector T cells to drive allergic skin inflammation. Proc Natl Acad Sci U S A. 2008;105:11875–11880. doi: 10.1073/pnas.0801532105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Li M, Hener P, Zhang Z, Ganti KP, Metzger D, Chambon P. Induction of thymic stromal lymphopoietin expression in keratinocytes is necessary for generating an atopic dermatitis upon application of the active vitamin D3 analogue MC903 on mouse skin. J Invest Dermatol. 2009;129:498–502. doi: 10.1038/jid.2008.232. [DOI] [PubMed] [Google Scholar]
- 20.Kim BS, Siracusa MC, Saenz SA, Noti M, Monticelli LA, Sonnenberg GF, Hepworth MR, Van Voorhees AS, Comeau MR, Artis D. TSLP Elicits IL-33-Independent Innate Lymphoid Cell Responses to Promote Skin Inflammation. Sci Transl Med. 2013;5:170ra116. doi: 10.1126/scitranslmed.3005374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Siracusa MC, Kim BS, Spergel JM, Artis D. Basophils and allergic inflammation. The Journal of allergy and clinical immunology. 2013;132:789–801. doi: 10.1016/j.jaci.2013.07.046. quiz 788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Obata K, Mukai K, Tsujimura Y, Ishiwata K, Kawano Y, Minegishi Y, Watanabe N, Karasuyama H. Basophils are essential initiators of a novel type of chronic allergic inflammation. Blood. 2007;110:913–920. doi: 10.1182/blood-2007-01-068718. [DOI] [PubMed] [Google Scholar]
- 23.Neill DR, Wong SH, Bellosi A, Flynn RJ, Daly M, Langford TK, Bucks C, Kane CM, Fallon PG, Pannell R, Jolin HE, McKenzie AN. Nuocytes represent a new innate effector leukocyte that mediates type-2 immunity. Nature. 2010;464:1367–1370. doi: 10.1038/nature08900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Moro K, Yamada T, Tanabe M, Takeuchi T, Ikawa T, Kawamoto H, Furusawa J, Ohtani M, Fujii H, Koyasu S. Innate production of T(H)2 cytokines by adipose tissue-associated c-Kit(+)Sca-1(+) lymphoid cells. Nature. 2010;463:540–544. doi: 10.1038/nature08636. [DOI] [PubMed] [Google Scholar]
- 25.Liang HE, Reinhardt RL, Bando JK, Sullivan BM, Ho IC, Locksley RM. Divergent expression patterns of IL-4 and IL-13 define unique functions in allergic immunity. Nat Immunol. 2012;13:58–66. doi: 10.1038/ni.2182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Price AE, Liang HE, Sullivan BM, Reinhardt RL, Eisley CJ, Erle DJ, Locksley RM. Systemically dispersed innate IL-13-expressing cells in type 2 immunity. Proc Natl Acad Sci U S A. 2010;107:11489–11494. doi: 10.1073/pnas.1003988107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Siracusa MC, Saenz SA, Tait Wojno ED, Kim BS, Osborne LC, Ziegler CG, Benitez AJ, Ruymann KR, Farber DL, Sleiman PM, Hakonarson H, Cianferoni A, Wang ML, Spergel JM, Comeau MR, Artis D. Thymic stromal lymphopoietin-mediated extramedullary hematopoiesis promotes allergic inflammation. Immunity. 2013;39:1158–1170. doi: 10.1016/j.immuni.2013.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Saenz SA, Siracusa MC, Monticelli LA, Ziegler CG, Kim BS, Brestoff JR, Peterson LW, Wherry EJ, Goldrath AW, Bhandoola A, Artis D. IL-25 simultaneously elicits distinct populations of innate lymphoid cells and multipotent progenitor type 2 (MPPtype2) cells. J Exp Med. 2013;210:1823–1837. doi: 10.1084/jem.20122332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Turner JE, Morrison PJ, Wilhelm C, Wilson M, Ahlfors H, Renauld JC, Panzer U, Helmby H, Stockinger B. IL-9-mediated survival of type 2 innate lymphoid cells promotes damage control in helminth-induced lung inflammation. J Exp Med. 2013;210:2951–2965. doi: 10.1084/jem.20130071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hoyler T, Klose CS, Souabni A, Turqueti-Neves A, Pfeifer D, Rawlins EL, Voehringer D, Busslinger M, Diefenbach A. The transcription factor GATA-3 controls cell fate and maintenance of type 2 innate lymphoid cells. Immunity. 2012;37:634–648. doi: 10.1016/j.immuni.2012.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Spencer SP, Wilhelm C, Yang Q, Hall JA, Bouladoux N, Boyd A, Nutman TB, Urban JF, Jr., Wang J, Ramalingam TR, Bhandoola A, Wynn TA, Belkaid Y. Adaptation of innate lymphoid cells to a micronutrient deficiency promotes type 2 barrier immunity. Science. 2014;343:432–437. doi: 10.1126/science.1247606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mirchandani AS, Besnard AG, Yip E, Scott C, Bain CC, Cerovic V, Salmond RJ, Liew FY. Type 2 innate lymphoid cells drive CD4+ Th2 cell responses. Journal of immunology. 2014;192:2442–2448. doi: 10.4049/jimmunol.1300974. [DOI] [PubMed] [Google Scholar]
- 33.Ito Y, Satoh T, Takayama K, Miyagishi C, Walls AF, Yokozeki H. Basophil recruitment and activation in inflammatory skin diseases. Allergy. 2011;66:1107–1113. doi: 10.1111/j.1398-9995.2011.02570.x. [DOI] [PubMed] [Google Scholar]
- 34.Kepley CL, Craig SS, Schwartz LB. Identification and partial characterization of a unique marker for human basophils. Journal of immunology. 1995;154:6548–6555. [PubMed] [Google Scholar]
- 35.Sawaguchi M, Tanaka S, Nakatani Y, Harada Y, Mukai K, Matsunaga Y, Ishiwata K, Oboki K, Kambayashi T, Watanabe N, Karasuyama H, Nakae S, Inoue H, Kubo M. Role of mast cells and basophils in IgE responses and in allergic airway hyperresponsiveness. Journal of immunology. 2012;188:1809–1818. doi: 10.4049/jimmunol.1101746. [DOI] [PubMed] [Google Scholar]
- 36.van Panhuys N, Prout M, Forbes E, Min B, Paul WE, Le Gros G. Basophils are the major producers of IL-4 during primary helminth infection. Journal of immunology. 2011;186:2719–2728. doi: 10.4049/jimmunol.1000940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ohnmacht C, Schwartz C, Panzer M, Schiedewitz I, Naumann R, Voehringer D. Basophils orchestrate chronic allergic dermatitis and protective immunity against helminths. Immunity. 2010;33:364–374. doi: 10.1016/j.immuni.2010.08.011. [DOI] [PubMed] [Google Scholar]
- 38.Ohnmacht C, Voehringer D. Basophils protect against reinfection with hookworms independently of mast cells and memory Th2 cells. Journal of immunology. 2010;184:344–350. doi: 10.4049/jimmunol.0901841. [DOI] [PubMed] [Google Scholar]
- 39.Salimi M, Barlow JL, Saunders SP, Xue L, Gutowska-Owsiak D, Wang X, Huang LC, Johnson D, Scanlon ST, McKenzie AN, Fallon PG, Ogg GS. A role for IL-25 and IL-33-driven type-2 innate lymphoid cells in atopic dermatitis. J Exp Med. 2013;210:2939–2950. doi: 10.1084/jem.20130351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Imai Y, Yasuda K, Sakaguchi Y, Haneda T, Mizutani H, Yoshimoto T, Nakanishi K, Yamanishi K. Skin-specific expression of IL-33 activates group 2 innate lymphoid cells and elicits atopic dermatitis-like inflammation in mice. Proc Natl Acad Sci U S A. 2013;110:13921–13926. doi: 10.1073/pnas.1307321110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Noti M, Kim BS, Siracusa MC, Rak GD, Kubo M, Moghaddam AE, Sattentau QA, Comeau MR, Spergel JM, Artis D. Exposure to food allergens through inflamed skin promotes intestinal food allergy through the thymic stromal lymphopoietin-basophil axis. The Journal of allergy and clinical immunology. 2014;133:1390–1399. e1396. doi: 10.1016/j.jaci.2014.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Halim TY, Steer CA, Matha L, Gold MJ, Martinez-Gonzalez I, McNagny KM, McKenzie AN, Takei F. Group 2 innate lymphoid cells are critical for the initiation of adaptive T helper 2 cell-mediated allergic lung inflammation. Immunity. 2014;40:425–435. doi: 10.1016/j.immuni.2014.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Roediger B, Kyle R, Yip KH, Sumaria N, Guy TV, Kim BS, Mitchell AJ, Tay SS, Jain R, Forbes-Blom E, Chen X, Tong PL, Bolton HA, Artis D, Paul WE, Fazekas de St Groth B, Grimbaldeston MA, Le Gros G, Weninger W. Cutaneous immunosurveillance and regulation of inflammation by group 2 innate lymphoid cells. Nat Immunol. 2013;14:564–573. doi: 10.1038/ni.2584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Renz H, Jujo K, Bradley KL, Domenico J, Gelfand EW, Leung DY. Enhanced IL-4 production and IL-4 receptor expression in atopic dermatitis and their modulation by interferon-gamma. J Invest Dermatol. 1992;99:403–408. doi: 10.1111/1523-1747.ep12616114. [DOI] [PubMed] [Google Scholar]
- 45.Gao PS, Rafaels NM, Mu D, Hand T, Murray T, Boguniewicz M, Hata T, Schneider L, Hanifin JM, Gallo RL, Gao L, Beaty TH, Beck LA, Leung DY, Barnes KC. Genetic variants in thymic stromal lymphopoietin are associated with atopic dermatitis and eczema herpeticum. The Journal of allergy and clinical immunology. 2010;125:1403–1407. e1404. doi: 10.1016/j.jaci.2010.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kayserova J, Sismova K, Zentsova-Jaresova I, Katina S, Vernerova E, Polouckova A, Capkova S, Malinova V, Striz I, Sediva A. A prospective study in children with a severe form of atopic dermatitis: clinical outcome in relation to cytokine gene polymorphisms. J Investig Allergol Clin Immunol. 2012;22:92–101. [PubMed] [Google Scholar]
- 47.Schmutz S, Bosco N, Chappaz S, Boyman O, Acha-Orbea H, Ceredig R, Rolink AG, Finke D. Cutting edge: IL-7 regulates the peripheral pool of adult ROR gamma+ lymphoid tissue inducer cells. Journal of immunology. 2009;183:2217–2221. doi: 10.4049/jimmunol.0802911. [DOI] [PubMed] [Google Scholar]
- 48.Motomura Y, Morita H, Moro K, Nakae S, Artis D, Endo TA, Kuroki Y, Ohara O, Koyasu S, Kubo M. Basophil-Derived Interleukin-4 Controls the Function of Natural Helper Cells, a Member of ILC2s, in Lung Inflammation. Immunity. 40:758–771. doi: 10.1016/j.immuni.2014.04.013. [DOI] [PubMed] [Google Scholar]
- 49.Li-Weber M, Krammer PH. Regulation of IL4 gene expression by T cells and therapeutic perspectives. Nat Rev Immunol. 2003;3:534–543. doi: 10.1038/nri1128. [DOI] [PubMed] [Google Scholar]
- 50.Doherty TA, Khorram N, Lund S, Mehta AK, Croft M, Broide DH. Lung type 2 innate lymphoid cells express cysteinyl leukotriene receptor 1, which regulates TH2 cytokine production. The Journal of allergy and clinical immunology. 2013;132:205–213. doi: 10.1016/j.jaci.2013.03.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Barnig C, Cernadas M, Dutile S, Liu X, Perrella MA, Kazani S, Wechsler ME, Israel E, Levy BD. Lipoxin A4 regulates natural killer cell and type 2 innate lymphoid cell activation in asthma. Sci Transl Med. 2013;5:174ra126. doi: 10.1126/scitranslmed.3004812. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.