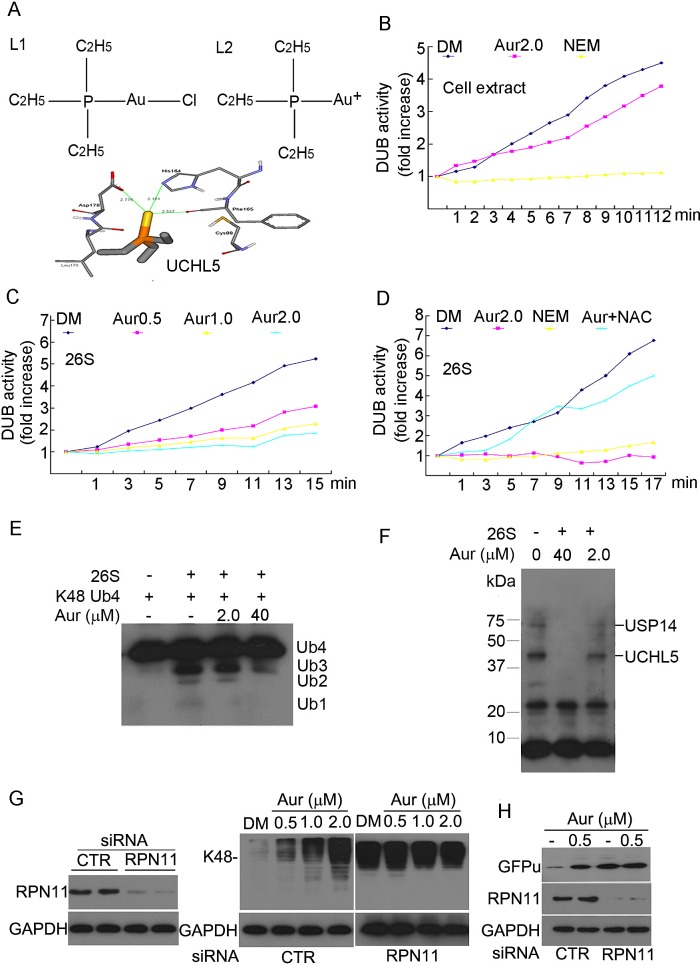
Figure 3. Aur inhibits the 19S proteasome DUB activity rather than 20S proteasome peptidases.
(A) Computational molecular docking of Au+ with UCHL5 of 19S proteasomes. The hydrolysed form of chloro (triethylphosphine) gold or Aur, (triethylphosphine) gold cation (L2, left), and its binding mode at the active site of UCHL5 were shown (right). (B) Effect of Aur on DUB activities in cell lysate. Cell lysate was treated with Aur (2μM) or NEM (N-ethylmaleimide, 2 mM), then the DUB activity at different times was recorded by using the Ub-AMC substrate. The experiment was repeated three times, yielding the similar results. (C) Inhibition of the DUB activity in 26S proteasomes. Purified 26S proteasomes were treated with increasing doses of Aur, then DUB activity was kinetically detected as in (B). (D) NAC rescues Aur-mediated DUB inhibition. Purified 26S proteasomes were treated with Aur (2 μM), Aur+NAC (100 μM), or NEM (2 mM) for 15 min, then DUB activity was detected. (E) Ubiquitin chain disassembly assay. K48-linked ubiquitin tetramers were disassembled by the 26S proteasomes after treatment with Aur (2.0, 40 μM). (F) Active-site–directed labeling of proteasomal DUBs. Purified 26S proteasomes were treated with Aur (2.0, 40 μM) for 10 min and then labeled with HA-UbVS and fractionated via SDS-PAGE. The covalently bound HA-UbVS was detected by western blot for the HA tag. (G) The effect of 26S proteasome disassembly by siRNA-mediated knockdown of RPN11 on Aur induced Ub-prs accumulation. HepG2 cells were transfected with specific siRNA against RPN11 for 48 h, and then treated with Aur (0.5, 1.0, 2.0 μM) for 6 h. Scrambled siRNA was used as control. K48-linked polyubiquitin and RPN11 protein was detected by western blot analyses. GAPDH was used as a loading control. (H) GFPu accumulation with RPN11 siRNA silencing and Aur treatment. GFPu-HEK293 cells were transfected with control siRNA or RPN11 siRNA for 48 h, and then treated with 0.5 μM Aur for 6 h. GFPu and RPN11 protein was detected by western blot analyses.