Abstract
The aim of this study was to describe specific histological findings of the Coxiella burnetii-infected aneurysmal abdominal aortic wall. Tissue samples of the aneurysmal abdominal aortic wall from seven patients with chronic Q fever and 15 patients without evidence of Q fever infection were analysed and compared. Chronic Q fever was diagnosed using serology and tissue PCR analysis. Histological sections were stained using haematoxylin and eosin staining, Elastica van Gieson staining and immunohistochemical staining for macrophages (CD68), T lymphocytes (CD3), T lymphocyte subsets (CD4 and CD8) and B lymphocytes (CD20). Samples were scored by one pathologist, blinded for Q fever status, using a standard score form. Seven tissue samples from patients with chronic Q fever and 15 tissue samples from patients without Q fever were collected. Four of seven chronic Q fever samples showed a necrotizing granulomatous response of the vascular wall, which was characterized by necrotic core of the arteriosclerotic plaque (P = 0.005) and a presence of high numbers of macrophages in the adventitia (P = 0.007) distributed in typical palisading formation (P = 0.005) and surrounded by the presence of high numbers of T lymphocytes located diffusely in media and adventitia. Necrotizing granulomas are a histological finding in the C. burnetii-infected aneurysmal abdominal aortic wall. Chronic Q fever should be included in the list of infectious diseases with necrotizing granulomatous response, such as tuberculosis, cat scratch disease and syphilis.
Keywords: aneurysm, chronic Q fever, granulomatous disease, infection, vascular disease
Q fever is a worldwide zoonotic disease caused by Coxiella burnetii, a Gram-negative and aerobic bacterium, which causes an intracellular infection in monocytes and macrophages (Waag 2007). The bacterium is highly infectious, and humans can be infected by inhalation. Most acute infections are asymptomatic (60%; Maurin & Raoult 1999). Symptomatic patients present with a mild influenza-like illness, atypical pneumonia or hepatitis. Chronic Q fever may develop several months to years after the acute infection (Fenollar et al. 2004; Maurin and Raoult 1999). Patients with a history of cardiac valve surgery, an aneurysm, vascular reconstruction, increasing age and renal insufficiency are at greater risk of developing chronic Q fever (Kampschreur et al. 2012a). Generally, the main clinical manifestation of chronic Q fever is Q fever endocarditis and, in less extent, infection of a vascular graft or aneurysm (vascular chronic Q fever; Parker et al. 2006). Patients with vascular chronic Q fever have a pre-existing aneurysm and/or graft, in which C. burnetii inhabits in macrophages and monocytes in the arteriosclerotic plaque, thrombus or vascular wall, causing a secondary infection of the aneurysm or graft (Waag 2007).
From 2007 to 2010, the Netherlands were confronted with a large Q fever outbreak with over 4000 notified acute Q fever cases. In the Dutch cohort, after primary infection, progression to vascular chronic Q fever in patients with an abdominal aortic aneurysm was high, 30.8% compared with 7.8% progression to Q fever endocarditis in patients with a history of cardiac valve surgery (Kampschreur et al. 2012b; Hagenaars 2014). Patients with vascular chronic Q fever can present with a life-threatening acute complication, such as a symptomatic aneurysm, ruptured aneurysm, mycotic aneurysm, aorto-duodenal fistula or aorto-caval fistula. High morbidity and a mortality rate of up to 25–60% have been described in patients with a mycotic aneurysm due to C. burnetii (Botelho-Nevers et al. 2007).
Diagnosing chronic Q fever is challenging. During primary infection with C. burnetii, IgM and IgG against phase II antigens are first expressed, followed by IgM and IgG against phase I antigens. In chronic infection, IgG against phase I antigens (IgG phase I) is predominant and their level increases and persists over the course of infection (van der Hoek et al. 2011). The diagnosis chronic Q fever can be achieved through detection of C. burnetii in blood or tissue by polymerase chain reaction (PCR) or culture (Schneeberger et al. 2010). The Dutch Q fever Consensus Group developed a guideline in which the certainty of having chronic Q fever is based on a combination of multiple diagnostic tests, such as serology, PCR, culture, as well as histology, clinical parameters and imaging studies (Wegdam-Blans et al. 2012).
Histology plays a minor role in the Dutch consensus guideline for diagnosis of chronic Q fever. In cases where the serology demonstrates an IgG phase I titre of 1:1024 and tissue shows a granulomatous response, the patient is categorized as having probable chronic Q fever (Wegdam-Blans et al. 2012). Histological examinations into Q fever were previously performed on cardiac valves, bone marrow, lung, spleen, placenta, liver, heart and kidney tissues from patients and animals (Lepidi et al. 2003; Wouda & Dercksen 2007; Hansen et al. 2011; Norina et al. 2011; Szablewski et al. 2012). Characteristic features of an acute infection with C. burnetii are granulomatous lesions containing doughnut cells, infiltration with mononuclear cells, neutrophils, eosinophils, lymphocytes and macrophages, presence of histiocytes, focal necrosis, haemorrhages, giant cells and plasma cells (Janigan & Marrie 1983; Lepidi et al. 2003; Wouda & Dercksen 2007; Hansen et al. 2011; Norina et al. 2011; Szablewski et al. 2012). Lepidi et al. (2003) demonstrated that valve tissue damaged by Q fever infection is localized in different parts of the valve, showing mononuclear cell inflammatory infiltrates with lymphocytes and macrophages. The lesions are small and discrete, so can even be confused with degenerative changes or could unintentionally be missed (Turck et al. 1976; Lepidi et al. 2003).
To date, comparison of the histology of the vascular wall in aneurysmal patients with or without chronic Q fever has not been described. Therefore, the aim of this study is to investigate specific histological findings of the C. burnetii-infected aneurysmal vascular wall.
Materials and methods
Tissue samples
From November 2011 until November 2012, tissue of the aneurysmal abdominal aortic wall was prospectively collected from patients operated on for an aneurysm correction by elective surgery or emergent surgery. Informed consent was signed by both patient and surgeon. Before the operation, serum was tested for the presence or absence of C. burnetii antibodies. Following surgery, aneurysmal wall tissue was tested using PCR for C. burnetii DNA. Patients with a serological profile indicative of chronic Q fever (IgG phase I of 1:1024 or higher; sensitivity of the test is 97.8%) and a positive tissue PCR (specificity of PCR is 100%) were included in the positive study group (Wegdam-Blans et al. 2012), while patients with a negative serological profile and negative tissue PCR comprised the control group (Huijsmans et al. 2010). In addition, patients with proven chronic Q fever operated before November 2011 of whom PCR-positive tissue had been stored were added to the positive study group. In these cases, informed consent was retrospectively obtained. The institutional review board of the Jeroen Bosch Hospital had approved the study.
Microbiological analysis
Sera were tested for C. burnetii antibodies using immunofluorescence assay (IFA; Focus Diagnostics, Inc., Cypress, CA, USA), assessing the presence of IgM and IgG phase I and II antibodies. Tissue was tested by PCR for the presence of C. burnetii DNA. The NucliSens EasyMAG extraction system (bioMerieux, Boxtel, the Netherlands) was used for extraction of DNA after proteinase K digestion of paraffin sections of fresh biopsies. PCR was performed as previously described (Huijsmans et al. 2010, 2011; Schneeberger et al. 2010).
Routine bacteriologic cultures were performed on all tissues to rule out infection by other bacteria. Additionally, Ziehl-Neelsen staining, Grocott staining and PCR for Bartonella henselae DNA (cat scratch disease) were performed on aneurysmal wall tissue of patients in the positive study group to rule out infection by other organisms causative of a necrotizing granulomatous response (Diederen et al. 2007). A serologic test for Treponema pallidum antibodies was performed to rule out syphilis.
Pathological analysis
Tissues were fixed in 0.01 M buffered (0.005 M disodium hydrogen phosphate anhydrous and 0.005 M sodium dihydrogen phosphate dihydrate, pH 7.0) 10% formalin and processed for paraffin embedding using a Tissue-Tek VIP 5 (Sakura, Torrance, CA, USA). The dehydration programme consisted of 14 steps of 1 h under continuous agitation, pressure, vacuum and heating. At 40 °C, two 10% formalin steps were followed by one 70% (v/v) ethanol step, two 96% ethanol steps, three 100% ethanol steps and two 100% xylene steps. Paraffin embedding was carried out at 60 °C in four 100% paraffin steps.
Three micromolar thick sections were cut from each formalin-fixed paraffin-embedded tissue and mounted on glass slides. One was used for haematoxylin and eosin (HE) staining, one for Elastic van Giesson staining (EvG), and five for immunohistochemistry using the markers CD3, CD4, CD8, CD20 and CD68.
For HE staining, Tissue-Tek PRISMA (Sakura Fineteck Holland BV, Alphen aan de Rijn, the Netherlands) was used. Sections were deparaffinized and re-hydrated again. The sections were stained for 8 min in a haematoxylin solution. After washing and bluing in ammonia water, the counterstain of eosin–phloxine B solution was used for 30–60 s. Afterwards, the slides were dehydrated and covered with film using Tissue-Tek FILM. (Sakura Fineteck Holland BV).
Elastic van Giesson staining was performed using VENTANA NEXUS special stains (Roche Diagnostics GmbH, Woerden, the Netherlands), according to the manufacturers' protocol.
For immunological staining, the following monoclonal markers were used: CD3 for T lymphocytes; (T lymphocytes were differentiated into T-helper cells and cytotoxic T cells using CD4 and CD8 staining respectively; CD20 for B lymphocytes; and CD68 for macrophages (Roche Diagnostics GmbH). The slides were placed in an oven at 60 °C for 15–30 min, to melt the paraffin. Next, the slides were put in an automated machine for immunological staining (Ventana Benchmark XT; Roche Diagnostics GmbH). Liquid cover slip was used to prevent dehydration. Cell conditioning CC1 was used to increase antibody accessibility. A biotine-labelled secondary antibody which detected the primary antibody was added. Next, streptavidin (horseradish conjugated) was added, followed by the substrate di-aminobenzidine, which bind together and form an insoluble brown reaction product. Because of the reported possibility of sampling error (Lepidi et al. 2003), two tissue samples of each patient were processed and examined. Within the same staining session, we conducted a negative control sample and a positive control sample.
Microscopic examination and grading was performed by one pathologist blinded for Q fever status. All samples were examined systematically at a 40× and 100× magnification using a standard score form grading morphology and immunological staining. The presence of B lymphocytes, T lymphocytes and macrophages in the intima, media and adventitia was scored in four categories: not present, mild, moderate and severe. Due to the low number of patients, we combined ′not present′ with ′mildly present′ and ′moderately present′ with ′severely' present. The presence of T lymphocytes was differentiated into T-helper cells (CD4) and cytotoxic T cells (CD8). Ratios of presence (CD4/CD3 and CD8/CD3) were calculated. The localization of B lymphocytes, T lymphocytes and macrophages in the intima, media and adventitia of the vascular wall was scored in one of the four following categories: not present, diffuse, focal or granulomatous response.
Statistical analyses
Differences between two groups were calculated in spss 18.0 (SPSS Inc., Chicago, IL, USA), using the Fisher's exact test in case of categorical data. P-values < 0.05 were considered statistically significant.
Results
A total of 22 aneurysmal patients were included in this study, seven patients with chronic Q fever and 15 patients without serological evidence of Q fever infection. Serology for C. burnetii showed IgG phase I of 1:1024 or higher in all seven patients in the positive study group. The C. burnetii real-time PCR analyses of the tissues of the seven chronic Q fever patients were positive with cycle threshold values of 22.2 ± 4.0 (mean ± SD; range 18.2–29.0), while the B. henselae PCR, Ziehl-Neelsen staining and Grocott staining tested negative on all tissues. Serologic tests for T. pallidum antibodies ruled out syphilis in all cases. Baseline characteristics showed no significant differences in gender, sex and co-morbidity, see Table 1. The number of acute complications did not significantly differ between the two groups.
Table 1.
Baseline characteristics of 22 patients with an aneurysm. Separately described are baseline characteristics of chronic Q fever patients and patients negative for Q fever infection
| Patients with chronic Q fever N = 7 N (%) | Negative control group N = 15 N (%) | P-value | |
|---|---|---|---|
| Male gender | 7 (100) | 12 (80) | 0.203 |
| Mean age (years) | 74 ± 12 | 70 ± 12 | 0.269 |
| Co-morbidity | |||
| Diabetes mellitus | 0 (0) | 2 (13) | 0.311 |
| Cerebrovascular incidents | 2 (29) | 3 (20) | 0.655 |
| Cardiac attacks | 2 (29) | 5 (33) | 0.823 |
| Pulmonary diseases | 1 (14) | 0 (0) | 0.134 |
| Malignancy | 1 (14) | 3 (20) | 0.746 |
| Kidney dysfunction | 3 (43) | 3 (20) | 0.262 |
| Number of acute aneurysms | 4 (57) | 4 (27) | 0.166 |
N, number of patients; P-value, calculated probability; definition acute aneurysm, symptomatic aneurysm, ruptured aneurysm, mycotic aneurysm, aorto-duodenal fistula and aorto-caval fistula.
The morphological features in intima, media and adventitia of the aneurysmal vascular wall of patients in both groups are summarized in Table 2. Necrotic core of the arteriosclerotic plaque was present in seven of seven chronic Q fever patients and in five of 15 Q fever-negative patients (P = 0.005). In two of seven tissues positive for Q fever, a giant cell in a necrotizing granuloma was seen (Figure 1), while this was not observed in any of the 15 Q fever-negative patients (P = 0.091).
Table 2.
Morphological features, comparing abdominal aneurysmal wall of patients with chronic Q fever with patients negative for Q fever infection
| Morphological feature | Chronic Q fever+ (N = 7) N | Q fever− (N = 15) N | P-value |
|---|---|---|---|
| Intima atheroma | 6 | 11 | 0.477 |
| Intima calcifications | |||
| Not present | 0 | 2 | 0.474 |
| Mild | 3 | 5 | |
| Moderate | 3 | 4 | |
| Severe | 1 | 4 | |
| Intima fibrosis | |||
| Not present | 1 | 1 | 0.400 |
| Mild | 2 | 6 | |
| Moderate | 3 | 5 | |
| Severe | 1 | 3 | |
| Necrotic core of the arteriosclerotic plaque | 7 | 5 | 0.005 |
| (Luminal) trombosis | 6 | 15 | 0.318 |
| Giant cell | 2 | 0 | 0.091 |
| Media destruction | |||
| Not present | 0 | 0 | 0.462 |
| Mild | 0 | 2 | |
| Moderate | 1 | 3 | |
| Severe | 6 | 10 | |
| Adventitia inflammation | |||
| Not present | 0 | 2 | 0.262 |
| Mild | 2 | 5 | |
| Moderate | 3 | 5 | |
| Severe | 2 | 3 | |
| Adventitia fibrosis | |||
| Not present | 0 | 2 | 0.101 |
| Mild | 0 | 6 | |
| Moderate | 2 | 3 | |
| Severe | 5 | 4 | |
Chronic Q fever+, group of patients with chronic Q fever (corresponding serological profile and PCR positive on tissue); Q fever−, group of patients without Q fever (negative serological profile and PCR negative on tissue); NS, not significant.
Bold values were significant.
Figure 1.
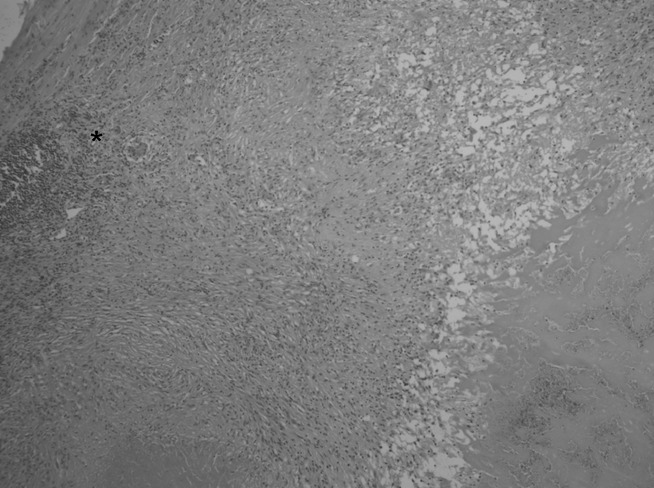
Haematoxylin and eosin staining of the aneurysmal abdominal aortic wall of a chronic Q fever patient displaying a giant cell in a necrotizing granuloma.
The presence of B lymphocytes, T lymphocytes and macrophages in the intima, media and adventitia of the vascular wall is demonstrated in Table 3. More macrophages were present in the adventitia (P = 0.007) of the vascular wall of patients with chronic Q fever, and more T lymphocytes were present in media (P = 0.021) and adventitia (P = 0.038) compared with aneurysmal tissue of patients with a negative Q fever status. Additional T-lymphocyte analyses revealed a mean ratio of 12% (SD ± 6) for cytotoxic T cells (CD 8) and a mean ratio of 30% (±28) for T-helper cells (CD 4) in the vascular wall of patients with chronic Q fever. Dividing the results for vascular wall with a necrotizing granulomatous response and without necrotizing granulomatous response, no significant difference is seen for the presence of cytotoxic T cells (P = 0.334), but a significant difference is seen for the presence of T-helper cells. The mean CD4 ratio is 4.0 (SD ± 1.7) in patients with necrotizing granulomatous response and 56.8 (SD ± 20.8) without a necrotizing granulomatous response (P = 0.024).
Table 3.
Differences in presence of B lymphocytes, T lymphocytes and macrophages in the intima, media and adventitia of the abdominal aneurysmal wall of patients with chronic Q fever compared with patients negative for Q fever infection
| Presence of B lymphocytes, T lymphocytes and macrophages | Chronic Q fever+ (N = 7) N | Q fever− (N = 15) N | P-value |
|---|---|---|---|
| Intima B lymphocytes | |||
| Not present or mild | 7 | 15 | – |
| Moderate or severe | 0 | 0 | |
| Media B lymphocytes | |||
| Not present or mild | 7 | 15 | – |
| Moderate or severe | 0 | 0 | |
| Adventitia B lymphocytes | |||
| Not present or mild | 3 | 5 | 0.510 |
| Moderate or severe | 4 | 10 | |
| Intima T lymphocytes | |||
| Not present or mild | 6 | 14 | 0.545 |
| Moderate or severe | 1 | 1 | |
| Media T lymphocytes | |||
| Not present or mild | 3 | 14 | 0.021 |
| Moderate or severe | 4 | 1 | |
| Adventitia T lymphocytes | |||
| Not present or mild | 0 | 7 | 0.038 |
| Moderate or severe | 7 | 8 | |
| Intima macrophages | |||
| Not present or mild | 3 | 6 | 0.628 |
| Moderate or severe | 4 | 9 | |
| Media macrophages | |||
| Not present or mild | 3 | 9 | 0.384 |
| Moderate or severe | 4 | 6 | |
| Adventitia macrophages | |||
| Not present or mild | 1 | 12 | 0.007 |
| Moderate or severe | 6 | 3 | |
Chronic Q fever+: group of patients with chronic Q fever (corresponding serological profile and PCR positive on tissue); Q fever−: group of patients without Q fever (negative serological profile and PCR negative on tissue).
P-value calculated using Fisher's exact test.
Bold values were significant.
The localization of the B lymphocytes, T lymphocytes and macrophages in the intima, media and adventitia of the vascular wall was scored (Table 4). Differences were observed in the characteristic line up of palisading macrophages in the adventitia (Figure 2). Four of seven patients in the positive study group showed a characteristic necrotizing granulomatous response vs. none of the patients in the control group (P = 0.005). Secondly, a significant difference was seen in the distribution of T lymphocytes in the adventitia. In patients with chronic Q fever infection, a diffuse distribution of T lymphocytes among the macrophages was present, compared with a more focal position in the control group (P-value = 0.031; Figure 3).
Table 4.
Localization of B lymphocytes, T lymphocytes and macrophages in intima, media and adventitia in aneurysmal vascular wall in patients with chronic Q fever compared with patients negative for Q fever infection
| Localization of B lymphocytes, T lymphocytes and macrophages | Chronic Q fever+ (N = 7) N | Q fever− (N = 15) N | P-value |
|---|---|---|---|
| Intima B lymphocytes | |||
| Not present | 7 | 13 | 0.516 |
| Diffuse | 0 | 2 | |
| Focal | 0 | 0 | |
| Media B lymphocytes | |||
| Not present | 4 | 7 | 0.491 |
| Diffuse | 2 | 7 | |
| Focal | 1 | 1 | |
| Adventitia B lymphocytes | |||
| Not present | 0 | 2 | 0.500 |
| Diffuse | 1 | 0 | |
| Focal | 6 | 13 | |
| Intima T lymphocytes | |||
| Not present | 3 | 11 | 0.182 |
| Diffuse | 4 | 4 | |
| Focal | 0 | 0 | |
| Media T lymphocytes | |||
| Not present | 0 | 3 | 0.650 |
| Diffuse | 7 | 12 | |
| Focal | 0 | 0 | |
| Adventitia T lymphocytes | |||
| Not present | 0 | 2 | 0.031 |
| Diffuse | 3 | 0 | |
| Focal | 4 | 13 | |
| Intima macrophages | |||
| Not present | 2 | 1 | 0.655 |
| Diffuse | 5 | 14 | |
| Focal | 0 | 0 | |
| Media macrophages | |||
| Not present | 1 | 2 | 0.278 |
| Diffuse | 4 | 13 | |
| Focal | 1 | 0 | |
| Palisade | 1 | 0 | |
| Adventitia macrophages | |||
| Not present | 0 | 2 | 0.005 |
| Diffuse | 2 | 12 | |
| Focal | 1 | 1 | |
| Palisade | 4 | 0 | |
Chronic Q fever+, group of patients with chronic Q fever (corresponding serological profile and PCR positive on tissue); Q fever−, group of patients chronic Q fever (negative serological profile and PCR negative on tissue); NS, not significant.
P-value is calculated using Fisher's exact test between the different groups per item, lowest P-value is noted.
Bold values were significant.
Figure 2.
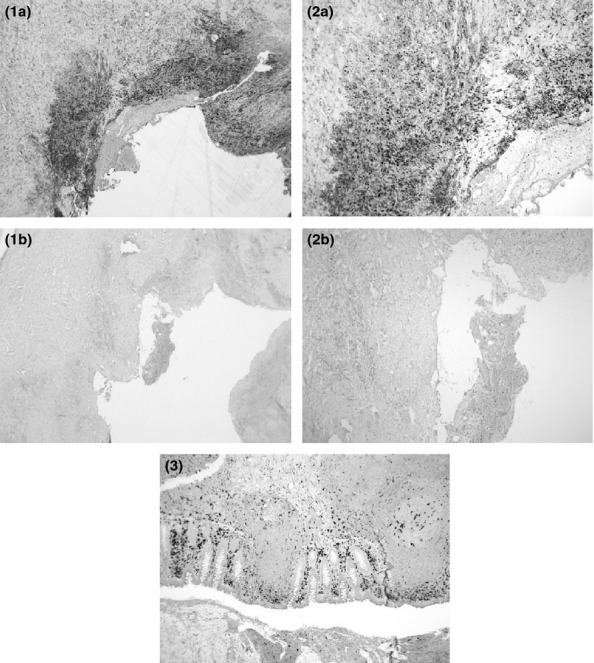
Immunohistochemical staining for CD68 of the aneurysmal abdominal aortic wall of a chronic Q fever patient displaying characteristic palisading line up of macrophages (a) and the negative control sample. (b) An overview (1) and a magnified illustration (2) are given. A positive control sample for CD68 staining (3) is added (contains tissue of appendix).
Figure 3.
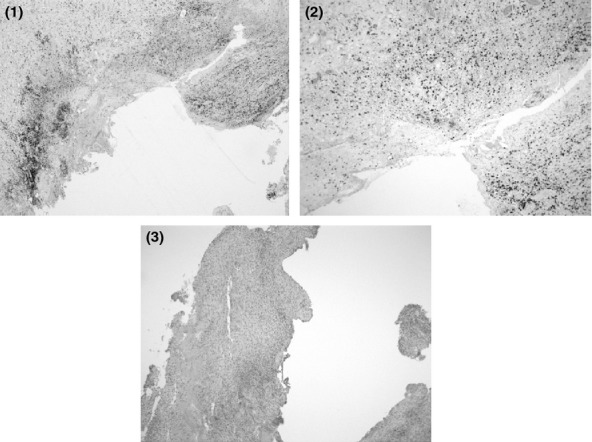
Immunohistochemical staining for CD3 of the aneurysmal abdominal aortic wall of a chronic Q fever patient displaying diffuse accentuation of T lymphocytes in the media and adventitia: an overview (1) and a magnified illustration (2). Additionally, an image of the negative control sample (same patient) is added (3).
Discussion
To date, no studies investigating the histological characteristics of the abdominal aortic aneurysm wall of patients with chronic Q fever. In this study we have documented these characteristics and have demonstrated a necrotizing granulomatous response of the vascular wall in more than half of the examined tissues.
A granulomatous response is an expression of a chronic infection or immunological reaction. Chronic infections associated with granulomas are caused by bacteria, such as Mycobacterium tuberculosis, T. pallidum, B. henselae, Mycobacterium leprae, Mycobacterium lepromatosis, and are reported in different fungal diseases, such as histoplasmosis, cryptococcosis, coccidioidomycosis and blastomycosis (Woodard et al. 1982; Su et al. 1992). Granulomas based on an immunological response can be seen in rheumatic diseases, granuloma annulare, Churg Strauss granulomatosis, sarcoidosis, Crohn's disease, berylliosis, IgG 4, Lupus erythematosis and Wegener's granulomatosis (Su et al. 1992; Barzilai et al. 2005; Brinster et al. 2009; Deonarain et al. 2009; Trinidad-Hernandez & Duncan 2012). We show here that chronic Q fever should be included in the list of diseases that can present with a necrotizing granulomatous response. The causative mechanism for the development of granuloma in chronic Q fever infection is not yet established. Coxiella burnetii is a strictly intracellular Gram-negative bacterium that infects and multiplies in macrophages and monocytes located in body tissue (Baca et al. 1985; Raoult et al. 1999; Porter et al. 2011). Coxiella burnetii might induce granulomas by the mechanism similar to ones induced by M. tuberculosis, which is also an intracellular bacterium. Due to M. tuberculosis infection, granuloma formation is initiated when the bacterium is phagocytozed by macrophages. As the granuloma matures, the centre becomes necrotic. The bacterium can survive for years in granulomas and at a later stage can spread in aerosols and cause illness (Chan & Flynn 2004; van der Wel et al. 2007; Pieters 2008; Helming & Gordon 2009). A second explanation is that C. burnetii can cause an immunological response, in which the formation of granulomas is evoked by tumour necrosis factor (TNF) and other interleukins (Algood et al. 2005; Koo et al. 2010). A relationship has been demonstrated between production of TNF and interleukin-10 (IL-10) and the risk of developing chronic Q fever (Ghigo et al. 2001; Honstettre et al. 2003). A high level of IL-10 induces the replication of C. burnetii in monocytes and is associated with a chronic outcome for the disease (Ghigo et al. 2001). Whether immunological responses in the patient or C. burnetii itself causes the formation of granulomas is still unclear. An important pathological difference between granuloma caused by bacteria and those resulting from a non-specific immunological responses is the presence of a necrotic core in the granuloma. A necrotic core appears most likely in bacteria-induced granulomas (Mukhopadhyay et al. 2012). We found necrosis in the tissue of the vascular wall in patients with chronic Q fever significantly more often than in controls, which makes the bacterial hypothesis more likely. Thus the hypothesis suggested by our observations is that the bacteria in the vessel wall are responsible for the granuloma formation, but to prove this hypothesis, the presence of the bacterium needs to be demonstrated in the granulomatous vascular issue from patients with vascular chronic Q fever.
A typical necrotizing granulomatous response was seen in the abdominal aortic aneurysm wall tissue in four of seven chronic Q fever patients but not in the control group (P = 0.005). Previously, one French report was published in which tissue of chronic Q fever patients with endocarditis was compared with tissue of patients without chronic Q fever (Lepidi et al. 2003). This study did find mononuclear cell inflammatory infiltrates with lymphocytes, macrophages, fibrosis and calcifications, but did not find the characteristic necrotizing granulomatous response. Other reports noted that specific histological features in tissue of chronic Q fever patients are rare or small and can easily be confused with degenerative lesions (Turck et al. 1976; Lepidi et al. 2003; Norina et al. 2011). There are some reports in which the formation of granulomatous lesions is described, but only in tissue infected with C. burnetii as a result of acute Q fever and not in patients or animals with chronic Q fever (Janigan & Marrie 1983; Lepidi et al. 2003; Wouda & Dercksen 2007; Norina et al. 2011; Szablewski et al. 2012). This report is the first to demonstrate a necrotizing granulomatous response in tissue of the vascular wall in patients with vascular chronic Q fever. The additional analyses concerning the distribution of T lymphocytes in the vascular wall showed comparable results with previous literature in patients without necrotizing granulomatous response, but a remarkably low ratio of T-helper cells in the necrotizing granulomatous infiltrate (Galle et al. 2005). Normally more T-helper cells are present in granulomas, but apparently not in chronic Q fever associated granulomas. Further research is needed as to how this change in T helper response arises.
Granulomatous tissue inflammation proven by histological examination is included as one of the diagnostic criteria of the Dutch Consensus Guideline for Diagnosing Chronic Q fever (Wegdam-Blans et al. 2012). The Dutch Consensus Guideline categorizes patients with a serologic profile matching chronic Q fever (IgG phase I ≥ 1:1024) and granulomatous tissue inflammation as probable chronic Q fever cases. We would like to suggest an alteration in the guideline recommending that patients with a granulomatous tissue inflammation in abdominal aortic aneurysm wall tissue and a serological profile matching chronic Q fever, in the absence of PCR positivity, positive imaging studies and evidence of other causative microorganisms, should be categorized as proven chronic Q fever cases. Furthermore, in any case that vascular tissue shows necrotizing granulomatous response, the diagnosis chronic Q fever should be considered.
A limitation of the study is still the low number of tissue samples included. Although the largest group ever described, a restricted statistical significance could be given (only in P-values and no 95% CI), and more samples should be evaluated to calculate an accurate diagnostic performance of histopathology in patients with vascular chronic Q fever.
Conclusion
Necrotizing granulomas are a histological finding in the C. burnetii-infected aneurysmal abdominal aortic wall. Chronic Q fever should be included in the list of infectious diseases with necrotizing granulomatous response, such as tuberculosis, cat scratch disease and syphilis.
Acknowledgments
This study was financially supported by the Netherlands Organization for Health Research and Development (ZonMw, Grant No. 205520001). The sponsor had no involvement in the study design, data collection, data analysis or interpretation of the study results. We would like to thank Jan Damen, laboratory technician, Jeroen Bosch Hospital, ‘s- Hertogenbosch, for his participation in data collection and preparation. We thank Dr. Ruud van Loenhout for his participation in data collection, and we thank Prof. Frans Moll for his role as supervisor.
Conflict of interest
The authors declare no conflict of interest.
References
- Algood HM, Lin PL, Flynn JL. Tumor necrosis factor and chemokine interactions in the formation and maintenance of granulomas in tuberculosis. Clin. Infect. Dis. 2005;41:S189–S193. doi: 10.1086/429994. [DOI] [PubMed] [Google Scholar]
- Baca OG, Scott TO, Akporiaye ET, DeBlassie R, Crissman HA. Cell cycle distribution patterns and generation times of L929 fibroblast cells persistently infected with Coxiella burnetii. Infect. Immun. 1985;47:366–369. doi: 10.1128/iai.47.2.366-369.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barzilai A, Huszar M, Shpiro D, Nass D, Trau H. Pseudo-rheumatoid nodules in adults. A juxta-articular form of nodular granuloma annulare. Am. J. Dermatopathol. 2005;27:1–5. doi: 10.1097/01.dad.0000148873.13318.87. [DOI] [PubMed] [Google Scholar]
- Botelho-Nevers E, Fournier PE, Richet H, et al. Coxiella burnetii infection of aortic aneurysms or vascular grafts: report of 30 new cases and evaluation of outcome. Eur. J. Clin. Microbiol. Infect. Dis. 2007;26:635–640. doi: 10.1007/s10096-007-0357-6. [DOI] [PubMed] [Google Scholar]
- Brinster DR, Grizzard JD, Dash A. Lupus aortitis leading to aneurysmal dilatation in the aortic root and ascending aorta. Heart Surg. Forum. 2009;12:105–108. doi: 10.1532/HSF98.20081123. [DOI] [PubMed] [Google Scholar]
- Chan J, Flynn J. The immunological aspects of latency in tuberculosis. Clin. Immunol. 2004;110:2–12. doi: 10.1016/s1521-6616(03)00210-9. [DOI] [PubMed] [Google Scholar]
- Deonarain J, Ramdial PK, Sing Y, Calonje E, Singh B. Subcutaneous palisading granulomatous pseudocysts of Echinococcus granulosus origin. J. Cutan. Pathol. 2009;36:240–245. doi: 10.1111/j.1600-0560.2008.01017.x. [DOI] [PubMed] [Google Scholar]
- Diederen BM, Vermeulen MJ, Verbakel H, van der Zee A, Bergmans A, Peeters MF. Evaluation of an internally controlled real-time polymerase chain reaction assay targeting the groEL gene for the detection of Bartonella spp. DNA in patients with suspected cat-scratch disease. Eur. J. Clin. Microbiol. Infect. Dis. 2007;26:629–633. doi: 10.1007/s10096-007-0353-x. [DOI] [PubMed] [Google Scholar]
- Fenollar F, Fournier PE, Raoult D. Molecular detection of Coxiella burnetii in the sera of patients with Q fever endocarditis or vascular infection. J. Clin. Microbiol. 2004;42:4919–4924. doi: 10.1128/JCM.42.11.4919-4924.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galle C, Schandené L, Stordeur P, et al. Predominance of type 1 CD4+ T cells in human abdominal aortic aneurysm. Clin. Exp. Immunol. 2005;142:519–527. doi: 10.1111/j.1365-2249.2005.02938.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghigo E, Capo C, Raoult D, Mege JL. Interleukin-10 stimulates Coxiella burnetii replication in human monocytes through tumor necrosis factor down-modulation: role in microbicidal defect of Q fever. Infect. Immun. 2001;69:2345–2352. doi: 10.1128/IAI.69.4.2345-2352.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagenaars JCJP. Estimated prevalence of chronic Q fever among Coxiella burnetii seropositive patients with an abdominal aortic/iliac aneurysm or aorto-iliac reconstruction after a large Dutch Q fever outbreak. J. Infect. 2014 doi: 10.1016/j.jinf.2014.03.009. doi: 10.1016/j.jinf.2014.03.009. [DOI] [PubMed] [Google Scholar]
- Hansen MS, Rodolakis A, Cochonneau D, et al. Coxiella burnetii associated placental lesions and infection level in parturient cows. Vet. J. 2011;190:135–139. doi: 10.1016/j.tvjl.2010.12.021. [DOI] [PubMed] [Google Scholar]
- Helming L, Gordon S. Molecular mediators of macrophage fusion. Trends Cell Biol. 2009;19:514–522. doi: 10.1016/j.tcb.2009.07.005. [DOI] [PubMed] [Google Scholar]
- van der Hoek W, Versteeg B, Meekelenkamp JC, et al. Follow-up of 686 patients with acute Q fever and detection of chronic infection. Clin. Infect. Dis. 2011;52:1431–1436. doi: 10.1093/cid/cir234. [DOI] [PubMed] [Google Scholar]
- Honstettre A, Imbert G, Ghigo E, et al. Dysregulation of cytokines in acute Q fever: role of interleukin -10 and tumor necrosis factor in chronic evolution of Q fever. J. Infect. Dis. 2003;187:956–962. doi: 10.1086/368129. [DOI] [PubMed] [Google Scholar]
- Huijsmans CJ, Damen J, van der Linden JC, Savelkoul PH, Hermans MH. Comparative analysis of four methods to extract DNA from paraffin-embedded tissues: effect on downstream molecular applications. BMC Res. Notes. 2010;14:239. doi: 10.1186/1756-0500-3-239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huijsmans CJ, Schellekens JJ, Wever PC, et al. Single-nucleotide-polymorphism genotyping of Coxiella burnetii during a Q fever outbreak in The Netherlands. Appl. Environ. Microbiol. 2011;77:2051–2057. doi: 10.1128/AEM.02293-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janigan DT, Marrie TJ. An inflammatory pseudotumor of the lung in Q fever pneumonia. N. Engl. J. Med. 1983;308:86–87. doi: 10.1056/NEJM198301133080207. [DOI] [PubMed] [Google Scholar]
- Kampschreur LM, Hagenaars JCJP, Wielders CCH, et al. Screening for Coxiella burnetii seroprevalance in chronic Q fever high-risk groups reveals magnitude of Dutch Q fever outbreak. Emerg. Infect. Dis. 2012a;18:563–570. [Google Scholar]
- Kampschreur LM, Oosterheert JJ, Hoepelman AI, et al. Prevalence of chronic Q fever in patients with a history of cardiac valve surgery in an area where Coxiella burnetii is epidemic. Clin. Vaccine Immunol. 2012b;19:1165–1169. doi: 10.1128/CVI.00185-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koo S, Marty FM, Baden LR. Infectious complications associated with immunomodulating biologic agents. Infect. Dis. Clin. North Am. 2010;24:285–306. doi: 10.1016/j.idc.2010.01.006. [DOI] [PubMed] [Google Scholar]
- Lepidi H, Houpikian P, Zhongxing L, Raoult D. Cardiac valves in patients with Q fever endocarditis: microbiological, molecular and histologic studies. J. Infect. Dis. 2003;187:1097–1106. doi: 10.1086/368219. [DOI] [PubMed] [Google Scholar]
- Maurin M, Raoult D. Q fever. Clin. Microbiol. Rev. 1999;12:518–553. doi: 10.1128/cmr.12.4.518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukhopadhyay S, Farver CF, Vaszar LT, et al. Causes of pulmonary granulomas: a retrospective study of 500 cases from seven countries. J. Clin. Pathol. 2012;65:51–57. doi: 10.1136/jclinpath-2011-200336. [DOI] [PubMed] [Google Scholar]
- Norina l, Sabri Y, Goh MY, et al. Immunohistological localisation of Coxiella burnetii in various organs of naturally Q fever infected goats. Pertanika J. Trop. Agric. Sci. 2011;34:167–173. [Google Scholar]
- Parker NR, Barralet JH, Bell AM. Q fever. Lancet. 2006;367:679–688. doi: 10.1016/S0140-6736(06)68266-4. [DOI] [PubMed] [Google Scholar]
- Pieters J. Mycobacterium tuberculosis and the macrophage: maintaining a balance. Cell Host Microbe. 2008;3:399–407. doi: 10.1016/j.chom.2008.05.006. [DOI] [PubMed] [Google Scholar]
- Porter SR, Czaplicki G, Mainil J, Guatteo R, Saegerman C. Q fever: current state of knowledge and perspectives of research of a neglected zoonosis. Int. J. Microbiol. 2011;2011:248418. doi: 10.1155/2011/248418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raoult D, Houpikian P, Dupont HT, Riss JM, Arditi-Djiane J, Brouqui P. Treatment of Q fever endocarditis. Arch. Intern. Med. 1999;159:167–173. doi: 10.1001/archinte.159.2.167. [DOI] [PubMed] [Google Scholar]
- Schneeberger PM, Hermans MH, van Hannen EJ, Schellekens JJ, Leenders AC, Wever PC. Real-time PCR with serum samples is indispensable for early diagnosis of acute Q fever. Clin. Vaccine Immunol. 2010;17:286–290. doi: 10.1128/CVI.00454-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su WPD, Kuechle MK, Peters MS, Muller SA. Palisading granulomas caused by infectious disease. Am. J. Dermatopathol. 1992;14:211. doi: 10.1097/00000372-199206000-00005. [DOI] [PubMed] [Google Scholar]
- Szablewski V, Costes V, Rousset T, Mania E, El Aoufi N. Q fever: bone marrow characteristic granuloma. Ann. Pathol. 2012;32:263–266. doi: 10.1016/j.annpat.2012.05.003. [DOI] [PubMed] [Google Scholar]
- Trinidad-Hernandez M, Duncan AA. Contained ruptured paravisceral aortic aneurysm related to immunoglobulin G4 aortitis. Ann. Vasc. Surg. 2012;26:108. doi: 10.1016/j.avsg.2011.07.004. [DOI] [PubMed] [Google Scholar]
- Turck WP, Howitt G, Turnberg LA, et al. Chronic Q fever. Q. J. Med. 1976;45:193–217. [PubMed] [Google Scholar]
- Waag DM. Coxiella burnetii: host and bacterial responses to infection. Vaccine. 2007;25:7288–7295. doi: 10.1016/j.vaccine.2007.08.002. [DOI] [PubMed] [Google Scholar]
- Wegdam-Blans MCA, Kampschreur LM, Nabuurs-Franssen MH, et al. Chronic Q fever: review of the literature and a proposal of new diagnostic criteria. J. Infect. 2012;64:247–259. doi: 10.1016/j.jinf.2011.12.014. [DOI] [PubMed] [Google Scholar]
- van der Wel N, Hava D, Houben D, et al. M. tuberculosis and M. leprae translocate from the phagolysosome to the cytosol in myeloid cells. Cell. 2007;129:1287–1298. doi: 10.1016/j.cell.2007.05.059. [DOI] [PubMed] [Google Scholar]
- Woodard BH, Rosenberg SI, Farnham R, Adams DO. Incidence and nature of primary granulomatous inflammation in surgically removed material. Am. J. Surg. Pathol. 1982;6:119–129. doi: 10.1097/00000478-198203000-00004. [DOI] [PubMed] [Google Scholar]
- Wouda W, Dercksen DP. Abortion and stillbirth among dairy goats as a consequence of Coxiella burnetii. Tijdschr. Diergeneeskd. 2007;132:908–911. [PubMed] [Google Scholar]


