Abstract
Background
MicroRNAs (miRNAs) play a pivotal role in coordinating messenger RNA (mRNA) transcription and stability in almost all known biological processes, including the development of the central nervous system. Despite our broad understanding of their involvement, we still have a very sparse understanding of specifically how miRNA contribute to the strict regional and temporal regulation of brain development. Accordingly, in the current study we have examined the contribution of miRNA in the developing rat telencephalon and mesencephalon from just after neural tube closure till birth using a genome-wide microarray strategy.
Results
We identified temporally distinct expression patterns in both the telencephalon and mesencephalon for both miRNAs and their target genes. We demonstrate direct miRNA targeting of several genes involved with the migration, differentiation and maturation of neurons.
Conclusions
Our findings suggest that miRNA have significant implications for the development of neural structure and support important mechanisms that if disrupted, may contribute to or drive neurodevelopmental disorders.
Electronic supplementary material
The online version of this article (doi:10.1186/1471-2164-15-777) contains supplementary material, which is available to authorized users.
Keywords: Cortex, Interactions, MicroRNA, mRNA, Neurogenesis
Background
The developing cerebral cortex is highly compartmentalized and involves precise regulation of cell proliferation, migration and differentiation [1, 2]. The behaviour of cells undergoing these neurodevelopmental processes is governed by spatiotemporal changes in gene expression [3]. Translation and intracellular traffic of these transcripts is influenced by small non-coding miRNA that function as molecular guide sequences for ribonucleoprotein complexes involved in targeted mRNA degradation or translational repression (reviewed [4–7]). By regulating mRNAs post-transcriptional fate, miRNA subsequently play a significant role in organizing complex patterns of gene activity. Thus these small RNA molecules also display distinct expression profiles [8–10], and exhibit both temporal and spatial specificity [8, 11–13]. For example, miR-9 has been demonstrated to play a role in controlling neurogenesis timing via targeting of progenitor-promoting and cell-cycle exit-promoting genes [14]. Accordingly, its expression has been shown to increase during neurodevelopment, but not in the postnatal period [11], and to be regionally restricted to telencephalic, diencephalic and tectal periventricular proliferative zones [12].
Analysis of miRNA and their target mRNA expression in neuronal cell cultures supports a regulatory role for miRNAs in neuronal development [15–18]. Consistent with this role, a number of neural-specific or neural-enriched miRNAs have been identified [19] and are thought to be involved in regulating early brain development (reviewed [20]). For example, miR-134 and miR-137 regulate neuronal maturation through the modulation of their target mRNA. The brain-specific miR-134 regulates dendritic spine development by repressing the translation of an mRNA encoding a protein kinase, Limk1, that controls spine development [21], while the brain-enriched miR-137 has a significant role in neuronal maturation and dendritic morphogenesis via direct targeting of Mib1, a ubiquitin ligase important for neurogenesis and neurodevelopment [22]. This supports existing evidence for the role of miRNAs in neurogenesis [23], morphogenesis [24], neuronal cell specification [25] and oligodendrocyte differentiation and myelination [26].
In the current study we wanted to determine if miRNA and their predicted targets are altered in accordance with their developmental stage and neurodevelopmental regionalization. More specifically we compare developmental miRNA and gene expression in the caudal regions including the mesencephalon and metencephalon that develop relatively early, with more rostral areas such as the telencephalon and diencephalon which develop later [27], as we hypothesize that miRNA are the subject of related spatiotemporal regulation. By matching miRNAs with their inversely-regulated mRNA targets, we suggest that miRNA’s post-transcriptional regulatory influence changes throughout neurodevelopment, with a pattern of expression in the developing telencephalon occurring after that of the mesencephalon in accordance with their respective cytoarchitectural and synaptic maturity.
It is critical for the development and use of rodents as animal models of neuropsychiatric disorders that we have a good understanding of the nature and timing of regional differentiation in the rodent brain. Predicting neurodevelopmental events and recognizing differing windows of vulnerability to environmental insults will help develop clinically relevant experimental models.
Results
Temporal dynamics of miRNA expression in the telencephalon and mesencephalon between E12 and P0
miRNA expression was analysed in the telencephalon and mesencephalon of embryonic (E) (E12, E14, E15, E18, E19) and newborn (postnatal (P) day 0) rats using GeneChip miRNA array matrices (Affymetrix). Data analysis revealed 303 miRNA in the telencephalon and 266 in the mesencephalon with differential expression during brain development. miRNA expression displayed greater variation in the telencephalon (Figure 1A) compared to the mesencephalon (Figure 1B) throughout development (Figure 1C), with 97 miRNA in the telencephalon having substantive temporal variance (SD > 1.3) across development as compared to 38 miRNA in the mesencephalon. There were also a number of miRNA with altered expression specific to the telencephalon. This occurred at E12 (32 miRNA) and E19 (26 miRNA) only. In contrast the mesencephalon had no region-specific miRNA expression at any time during the developmental timepoints examined. Furthermore we also observed stage-associated expression in the telencephalon-specific miRNA, with 13 and 7 expressed only at E12 or E19 respectively (Additional file 1). To gain an appreciation of the biological implications of these telencephalon-specific miRNA during early telencephalon development, putative target genes of the 32 miRNA specific to the telencephalon at E12 and the 26 miRNA specific to the telencephalon at E19 were assessed for Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment using the Gene Annotation Tool to Help Explain Relationships (GATHER) online database. For the telencephalon-specific miRNA at E12 this revealed a number of significantly enriched pathways with relevance to neural connectivity and synaptic plasticity, such as focal adhesion; adherens junction; oxidative phosphorylation; regulation of actin cytoskeleton and ECM-receptor signalling. Similarly, the telencephalon-specific miRNA at E19 were predicted to be enriched in the Mitogen-activated protein (MAP) kinase signalling pathway and Wingless/int (Wnt) signalling pathway (Additional file 2). Further analysis using Ingenuity Pathway Analysis (IPA) software (Ingenuity Systems) revealed roles in important neurodevelopmental processes including: the differentiation of dopamine neurons; neuron and neuroglia differentiation; myelination; synaptogenesis; and axonogenesis (Figure 2).
Figure 1.
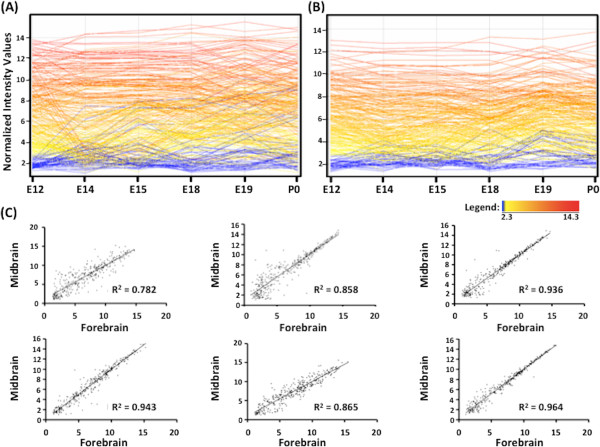
miRNA expression profiles in the developing rat brain. Intensities of differentially expressed miRNA microarray probes across development in the telencephalon (A) and mesencephalon (B). Each colored line represents one probe. Normalized signal intensities range from 2.3 to 14.3 as indicated by color legend. Color legend is relative to E12 such that probes with low expression at E12 (signal intensity of 2.3) are in blue while those with high expression at E12 (signal intensity of 14.3) are in red. (C) Scatter plots illustrating miRNA expression in the mesencephalon and telencephalon at each timepoint. Solid line = line of best fit; R2 = Pearson correlation coefficient.
Figure 2.
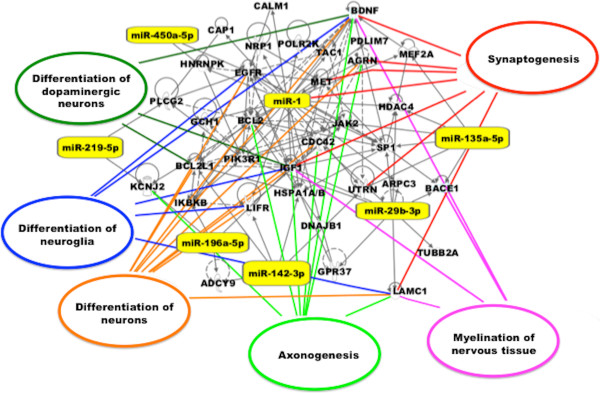
Computational gene interaction predictions: network of E12 telencephalon-specific miRNA involved in the Neurotransmitters and other nervous system signalling pathways. A network of putative gene targets of the 32 telencephalon-specific miRNA at E12 was constructed with the Ingenuity Systems Pathway Analysis (IPA) software. Solid grey lines specify direct relationships whereas dotted grey lines indicate indirect interactions. Genes involved in select neurodevelopmental processes are indicated with solid color lines.
To determine those miRNA that were differentially regulated with respect to developmental age, all miRNA were ranked based on their temporal expression variance through E12 to P0. miRNA undergoing substantial changes throughout development (SD > 1.3) were selected for downstream analysis (Additional file 3). The 97 miRNA identified were found to have significantly lower expression (F = 5.271, n = 12, p < 0.05) in the telencephalon compared to the mesencephalon at embryonic day 14 (Figure 3A). Supervised hierarchical clustering of these miRNA was characterized by distinct expression clusters, two of which showed significantly lower expression (p < 0.001) in the telencephalon compared to the mesencephalon at embryonic day 12 (Figure 3B & C). Prominent among this group were miRNAs that are enriched in neurons and associated with developmental regulation (let-7i, let-7f, let-7b, miR-98), cell cycle regulation (miR-137, mIR-128) and neural activity (miR-132, miR-212). Bioinformatic analysis of these 97 miRNA through IPA (Ingenuity Systems) identified 9652 putative miRNA target genes. Gene ontology (GO) function and KEGG pathway enrichments were performed by mapping the predicted target genes from the developmentally regulated miRNAs using the GATHER online database. For these miRNAs and their predicted targets, 22 GO functions and 3 KEGG pathways were significant (Bayes factor >6). The top ontologies included: regulation of cell cycle; development; morphogenesis; regulation of transcription; and neurogenesis (Additional file 4).
Figure 3.
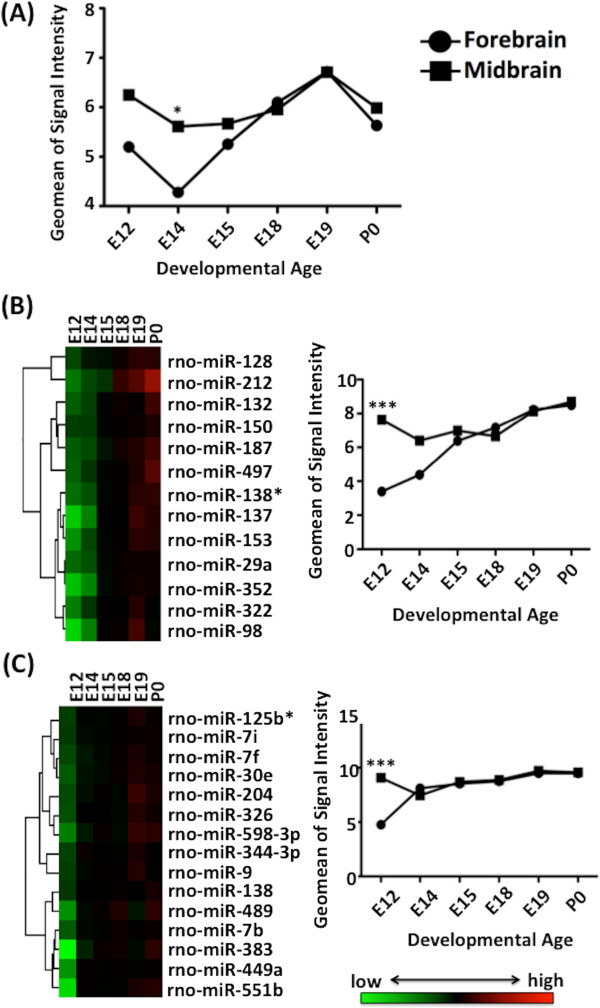
miRNA expression in the developing rat brain. (A) Expression of the 97 miRNA showing strong developmental regulation in the telencephalon and mesencephalon throughout development (for display purposes expression was derived by calculating the geomean of signal intensity values for all dynamic miRNAs at each developmental timepoint). All data were analysed for significance by one-way ANOVA, followed by Tukey-Kramer’s post hoc t-testing (* = p < 0.05). (B & C) Relative expression levels for the 97 miRNA showing strong development regulation were hierarchically clustered (supervised, correlation uncentred, average linkage; Cluster 3.0). Colors indicate relative signal intensities. (Java Treeview; http://jtreeview.sourceforge.net). Two major clusters are shown in six columns corresponding to the six time points. Comparison of expression of telencephalon and mesencephalon miRNA for each cluster is shown adjacent to the heatmap. All data were analysed for significance by one-way ANOVA, followed by Tukey-Kramer’s post hoc t-test. *** = p < 0.001.
In summary, while we observed substantial overlap between miRNA expressed in the telencephalon and mesencephalon, there was a number of functionally significant miRNA expressed exclusively in the telencephalon. From longitudinal analysis in each brain region, it was also apparent that telencephalon miRNA expression was subject to more temporal variation compared to the mesencephalon. Genes predicted to respond to this developmental variation in telencephalon cortical miRNA expression were enriched in pathways associated with neural development and function.
Developmental regulation of mRNA expression in the telencephalon and mesencephalon between E12 and P0
mRNA expression was analysed in the telencephalon and mesencephalon of embryonic (E12, E14, E15, E18, E19) and newborn (P0) rats using RatRef-12 Expression BeadChips (Illumina). Data analysis revealed a total of 16,691 genes with differential expression during neural development (E12-P0), comprised of 14,371 genes in the telencephalon and 13,906 genes in the mesencephalon. To determine those genes that were differentially regulated with respect to developmental age, all mRNA were ranked based on their temporal expression variance between E12 to P0. Those that demonstrated the greatest variation throughout development (SD >3.2) were selected for bioinformatic evaluation (Additional file 5). These 100 genes with high temporal expression dynamics were further assessed for their involvement in biological pathways. GO function and KEGG pathway enrichment of these genes revealed 43 significant (p < 0.05) GO functions and 1 KEGG pathway (Additional file 6). Network analysis using IPA (Ingenuity Systems) for predicting potential interactions between the differentially expressed genes generated three networks with the highest IPA analysis scores (47, 25 and 21) including Cell-To-Cell Signalling and Interaction; Nervous System Development and Function; Cellular Growth and Proliferation; and Cellular Development (Table 1). While these biological processes accorded well with those predicted by the developmentally regulated miRNA, we wanted to explore the possibility that these 100 dynamically expressed genes were most likely responding to changes in miRNA expression and driving developmentally significant changes in cell morphology and physiology, by bioinformatically associating the putative interactions with observed changes in predicted relationships or miRNA-mRNA interactions.
Table 1.
Network analysis using IPA
| Associated Network Functions | Score |
|---|---|
| Cell-To-Cell Signalling and Interaction, Nervous System Development and Function, Cell Death | 47 |
| Cellular Growth and Proliferation, Haematological System Development and Function, Haematopoiesis | 25 |
| Cellular Development, Cell Signalling, Organ Development | 21 |
| Cell Morphology, Cellular Function and Maintenance, Cell Cycle | 19 |
| Psychological Disorders, Cell Death, Gene Expression | 13 |
Network analysis using IPA (Ingenuity Systems) for predicting potential interactions between the differentially expressed genes potentially regulated by differentially expressed miRNA.
miRNA-mRNA interactions
Given that miRNAs predominantly function to negatively regulate gene expression, we examined the biological significance of inverse expression patterns between differentially expressed miRNA and differentially expressed genes in the telencephalon during development. We used Ingenuity’s miRNA Target Filter® to analyze all possible miRNA and target-gene interactions from the miRNA and gene expression array data sets. The results were filtered based on the confidence of interaction and an inverse miRNA to target mRNA relationship. These gene sets were then functionally annotated using the Database for Annotation, Visualization, and Integrated Discovery bioinformatics resources 6.7 (DAVID) online database, revealing significant over-representation in neural pathways throughout development. This included axon guidance, neuroactive ligand-receptor interaction, MAP kinase signalling, Wnt signalling, long-term depression and calcium signalling (Additional file 7).
We further investigated the putative roles of mir-98, mir-212 and mir-137 in the development of the telencephalon. All three miRNA were determined to be dynamically regulated with more than a three-fold increase in telencephalon expression between E12 and E19. These neuronally important miRNAs have critical roles in neurodevelopment and are associated with neuropathology (see Discussion). Concordantly, their predicted gene targets were also highly significant in our study. Validation of microarray expression for these three miRNA using quantitative real-time reverse transcription PCR (Q-PCR) (Figure 4) confirmed significantly lower expression in the telencephalon compared to the mesencephalon at E12. To further investigate their biological relevance, IPA software was used to identify putative differentially expressed target genes of these dynamically regulated miRNA. Analysis of this gene dataset using the DAVID online database revealed significant (p < 0.05) over-representation of neurodevelopmental functions in the mammalian forebrain cortex, including axonogenesis; neuron projection development; synaptic transmission; forebrain development; regionalization; and learning and behaviour (Additional file 8).
Figure 4.
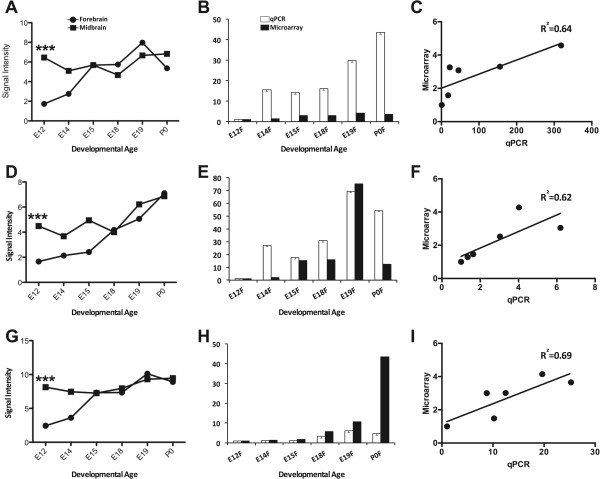
Select miRNA expression in the developing rat brain. Total RNA was isolated from rat telencephalon and mesencephalon at E12, E14, E15, E18, E19 and P0, amplified, labelled and hybridised to Affymetrix Genechip miRNA 1.0 microarrays. Data were normalised using cubic spline and analysed using GeneSpring GX11. Figures show the normalised signal intensity values for miR-98 (A) and miR-212 (D) and miR-137 (G) at each developmental stage in the mesencephalon and telencephalon (*** = p < 0.001). Findings were validated by Q-PCR and data expressed as a fold-change. Mean value of E12 was set to 1-fold induction, and mean values of all other ages were related to E12. MiRNA expression as determined by microarray appears as black bars and Q-PCR results appear as white bars. For Q-PCR, relative miRNA expression in the telencephalon was determined by the difference between their individual cycle threshold (Ct) value and that produced in the same sample for the U6 snRNA (∆Ct). Error bars represent standard deviation of the means. Microarray expression data was validated by RT-PCR for miR-98 (B), miR-212 (E) and miR-137 (H) with correlation coefficients of 0.64 (C), 0.62 (F) and 0.69 (I) respectively.
Target gene silencing in vitro
Our expression analysis of miRNA and their cognate mRNA targets suggested that there are a tremendous number of interactions that underlie the spatiotemporal segmentation in mammalian brain development. However, to further support the functional association between neural development-associated target genes and altered expression in this group of miRNAs, the respective miRNA recognition elements (MRE) from a selection of six functionally significant target genes, Clasp2, Rab15, Cbll1, Wnt7a, Gpr88 and Plxnb2, were cloned into the 3’ untranslated region (UTR) of a luciferase reporter gene construct and co-transfected into a recipient cell line with synthetic miRNA mimics corresponding to those altered in vivo. The degree of reporter gene activity and the impact of miRNAs were then ascertained by measuring the relative luciferase activity (Figure 5). MiRNAs interact with their respective MREs in the 3’UTR of transcripts and each gene may contain binding sites for multiple miRNA. Therefore observing miRNA-mediated effects using a reporter gene construct allows subtle changes in gene expression to be observed. Most of the reporter constructs behaved in accordance with expectation and were repressed 8-25% (p < 0.05) in the presence of synthetic miRNA in comparison to a scrambled-duplex co-transfected control. These data indicate that transfected miRNAs bound to their target MREs and repressed the expression of luciferase, with the most responsive targets derived from the 3’-UTR of Wnt7a and Gpr88. These were both targeted by miR-137, which appeared to have the greatest overall effect, whereas miR-212 had the least effect on these target gene constructs. The only exception was the Plxnb2 construct, which displayed increased expression in the presence of synthetic miR-137. Collectively, these reporter assays supported the existence of relationships between genes reported to be associated with neural development and developmentally regulated miRNAs.
Figure 5.
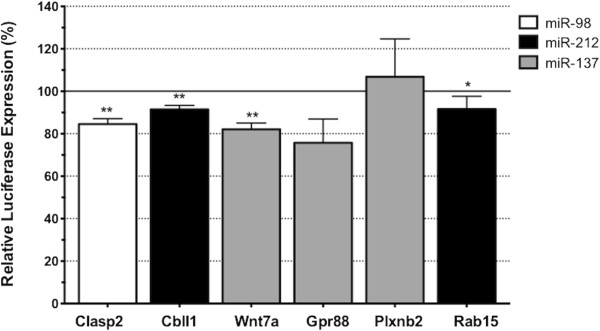
Regulation of neuronal development-associated reporter gene constructs by miRNA. Chart shows relative activity of reporter gene constructs (x-axis) in response to co-transfected miRNAs. Relative luciferase activity for each reporter/miRNA combination was expressed as a percentage of the response to scrambled controls. Bars are mean ± SEM. (** = p < 0.01, * = p < 0.05).
Discussion
miRNA and post-transcriptional regulation of neurodevelopment
The trajectory of human brain development closely mirrors that seen in the rodent [28] and as such the rat offers an excellent model to investigate the coordination and timing of miRNA expression across fetal development. In the current study we examined the expression profiles of miRNA within the mesencephalon and telencephalon from E12 until birth. We hypothesized that the developing telencephalon would exhibit a significantly different temporal miRNA profile in relation to the mesencephalon and this was supported by the observation that 87% of expressed miRNA underwent significant change across development, with clear differences in patterning between the two regions. The highest level of change in miRNA expression occurred early in development (E12-E15) and plateaued thereafter, with differences between the telencephalon and mesencephalon decreasing noticeably during that time period. In accordance with our hypothesis, telencephalon miRNA expression was significantly lower than the mesencephalon at E12 and consistent with the delayed development of this region. We also observed 32 miRNA with predicted function in neurodevelopmental processes, exclusively expressed in the telencephalon during early brain development (E12).
We then employed a bioinformatics approach to further understand the differential developmental trajectories of these two brain regions. Temporally regulated miRNAs and genes were examined using a combination of miRNA and gene expression profiling and computational target prediction. Hierarchical clustering of miRNA during telencephalon development revealed two distinct clusters of miRNA. These clusters had significantly higher expression in the mesencephalon at E12 whilst expression in the telencephalon remained significantly lower (Figure 3B & C). The miRNA contained in these clusters are predicted to target temporally-regulated genes involved in brain signalling pathways important for neurodevelopment and neuronal functioning including: dopamine receptor signalling; GABA receptor signalling; glutamate receptor signalling and neuroactive ligand-receptor interactions. As expression of the neuroactive ligand-receptor interactions pathway is demonstrated to correlate with synaptic maturity [29], this data is consistent both with the telencephalon being less differentiated at this time point and more importantly implicates a critical role for miRNA in this process. This also indicates that at such an early timepoint, the developing telencephalon may be largely unaffected by environmental insults compared to the mesencephalon. This may be particularly relevant for certain developmental animal models of disease. For instance the widely used maternal immune activation model of schizophrenia employs the artificial viral construct poly I:C which induces a strictly time-limited inflammatory response of approximately two days in the maternal animal [30]. This may be why when it is commonly administered at E9 in mice (equivalent to E10 in a rat), the downstream effects may be restricted to the developing mesencephalon rather than later maturing regions such as the telencephalon [31].
miRNA-mRNA interactions
While there are some exceptions, miRNA function is usually associated with repression of target gene expression, such that when a miRNA is expressed at high levels, its target mRNA is reduced [32–34]. Indeed, several reports have identified inverse expression patterns between differentially expressed miRNA and differentially expressed mRNA during development [17, 18]. In this study we found the putative down-regulated gene targets of up-regulated miRNA in the telencephalon between E12 and E14 are predicted to be involved in many important brain signalling pathways including axon guidance, neuroactive ligand-receptor interaction, calcium signalling and long-term depression.
Altered expression of miRs-98, -212 and -137 was particularly noteworthy, as these three neuronally important miRNAs were much more abundant in developing mesencephalon than telencephalon at the earliest embryonic age showing more than a three-fold increase in telencephalon expression between E12 and E19. All three miRNA have critical roles in neurodevelopment and are associated with neurological disorders such as Alzheimer’s disease and schizophrenia. We confirmed a functional link between these three miRNA and suppression of expressed target genes by cloning the MREs from Clasp2, Rab15, Cbll1, Wnt7a, Gpr88 and Plxnb2 downstream of a luciferase reporter gene. These reporter constructs were then co-transfected with synthetic analogues of their cognate miRNA into a recipient cell line, such that the extent of reporter gene expression and therefore the influence of miRNA could be ascertained through the measurement of relative luciferase activity (Figure 5). Most of these constructs were significantly repressed in the presence of synthetic miRNA. In particular, regulation of 3’-UTR elements from Wnt7a (miR-137) and Gpr88 (miR-137) was particularly strong and, along with Clasp2 (miR-98) and Rab15 (miR-212) provides support for the role of post-transcriptional regulation in axon and dendritic growth, maturation and function [35–38]. Pathway analysis of these target genes indicated that these miRNA may exert significant influence during neurodevelopment through the regulation of pathways including: axon guidance; neuroactive ligand-receptor interaction; Wnt; Erythroblastic Leukemia Viral Oncogene Homolog (ErbB); and MAP kinase signalling.
In the telencephalon, expression levels of miR-98 increased over four-fold between E12 and E19, a similar finding to that of Sempere and colleagues [15], who found mir-98 expression levels progressively accumulated upon neuronal differentiation in both mouse and human embryonic carcinoma cells. Experiments by Thomson and colleagues [39] similarly found levels of miR-98 were significantly increased in mice at E14.5 compared to levels in embryonic stem cells. Mir-98 is a member of the let-7 family, highly conserved across species in sequence and function and involved in the developmental timing of cell fates (reviewed [40]). In support of a role for miR-98 in telencephalon development, mir-98 overexpression was found to significantly decrease the activity of a luciferase reporter gene fused to the cytoplasmic linker associated protein 2 (Clasp2) 3’-UTR. Clasp2 plays a crucial role in chromosome segregation during mitosis [41] and is a key regulator of neuronal morphogenesis, neuronal polarity and synapse formation and activity [37].
Similarly, the results suggest a role for miR-212 in brain development with expression levels increased more than four-fold in the telencephalon between E12 and birth. MiR-212 arises from the miR-212/132 cluster, which is highly conserved in vertebrates. MiR-212 and miR-132 are tandem miRNAs with similar target specificity due to their identical seed sequences. In the brain, miR-212 and miR-132 play vital roles in the formation and plasticity of neuronal connections, and long-term synapse activation (reviewed [42, 43]). Dysregulation of miR-212 is associated with drug addiction [44], neuropathological disorders [43] and schizophrenia [45]. Our luciferase reporter assays support miR-212 involvement in neurodevelopment via regulation of Rab15, member RAS oncogene family (Rab15) and Cbl proto-oncogene-like 1, E3 ubiquitin protein ligase (Cbll1). Rab15 belongs to the Rab family of low molecular weight (LMW) GTP-binding proteins, crucial in ensuring the spatiotemporal regulation of membrane traffic [46]. Rab15 is enriched in neural tissue and plays a role in regulating neurotransmitter release [47]. Cbll1 is a RING finger type E3 ubiquitin ligase and an important regulator of cell proliferation [48]. In Drosophila, Cbll1 is required for proper function of cell surface proteins and for endoderm and mesoderm morphogenesis [49]. Consistent with the increase of miR-212 expression between E12 and E19, Cbll1 function is demonstrated to be most vital in the early stages of embryogenesis, with its contribution declining in later stages [49].
Another developmentally regulated miRNA is miR-137, with expression levels increasing more than four-fold in the telencephalon between E12 and E19. Mir-137 is enriched in neurons and involved in neuronal maturation, regulation of dendritic development and phenotypic maturation of new neurons [22]. Recent studies demonstrate increasing expression of miR-137 during neuronal differentiation, leading to reduced neural stem cell proliferation, accelerated neural differentiation and decreased dendritic development [22, 50]. Dysregulation of miR-137 has been associated with intellectual disability and Alzheimer’s Disease [51, 52] and the MIR137 gene has also recently found to be highly associated with schizophrenia in a genome wide screen and associated with cognitive function in patients with the disorder [53, 54]. This study confirmed the potency of miR-137 to down-regulate G-protein coupled receptor 88 (Gpr88) and wingless-type MMTV integration site family, member 7A (Wnt7a) using a reporter gene strategy. Gpr88 is a brain-enriched G protein–coupled receptor with a role in modulating the striatal dopaminergic system [55] and in regulating medium spiny neuron excitability [38]. Wnt7a signalling is critical to regulate dendritic spine growth and synaptic strength [35], mediating synapse density and numbers, and hippocampal network structure [56]. During development Wnt signalling is required for cell proliferation and differentiation, cell polarity generation and embryonic patterning.
As various brain regions develop at different times there is likely to be differing windows of vulnerability to environmental insults. Appropriate spatiotemporal expression of miRNA is vital during brain development and dysregulation during sensitive stages is likely to have a profound impact on the coordinated expression of developmentally significant genes associated with the pathophysiology of neuropsychiatric disorders. This is supported by the alterations of miRNA expression observed in the neuropathology of Alzheimer’s disease [51], Huntington’s disease [57, 58], Parkinson’s disease [59–61] and schizophrenia [62, 63]. Understanding the role of miRNA in brain ontogeny could identify critical windows of developmental significance that are susceptible to environmental insults which contribute to the development of neurobehavioural or neuropsychiatric syndromes.
Conclusions
This study represents the first global characterization of both miRNA and mRNA expression between the telencephalon and mesencephalon at various stages during development in rodent brain. Comparing the telencephalon and mesencephalon affords a molecular basis for understanding the nature and timing of regional differentiation. Although these results may have been hypothesised based on current evidence suggesting that the mesencephalon develops earlier than the telencephalon, the present data now provide direct molecular support that differential regional and temporal miRNA expression within the brain is part of this process. Knowledge of the temporal and spatial expression profiles of miRNA and their target genes is an important initial step in elucidating their functions. Together these results show that miRNA expression is frequently region- and time-specific with an important influence in functionally relevant pathways in the developing brain.
Methods
RNA preparation and analysis of integrity
Telencephalon and mesencephalon were dissected from brains of embryonic (E) (E12, E14, E15, E18, E19) and newborn (P0) rats. Each time point represents a pooled litter of rat embryos (n = 8-12). Animals were handled in accordance with the University of Queensland's animal care and ethics committee approval, in compliance with the current Australian Law. Total RNA was isolated from the telencephalon and mesencephalon using TRIzol reagent as described previously [64]. RNA concentration, integrity and purity were assessed using the Agilent 2100 Bioanalyser (Agilent Technologies). RNA Integrity Numbers (RIN) were automatically calculated with the provided system software [65]. All samples showed RIN values superior to 8.5.
MicroRNA expression profiling
For miRNA profiling, total RNA (700-1000 ng) was labelled using a FlashTag Biotin HSR RNA labelling kit according to manufacturer’s instructions (Genisphere). Labelled RNA was hybridised to the Affymetrix Genechip miRNA 1.0 microarrays (miRBase version 11.0) for 16 hours at 48°C and 60 rpm in a hybridisation oven (Affymetrix) and washed and stained with the Fluidics Station 450 (Affymetrix). Hybridisation, wash and stain reagents were from the Hyb, Wash and Stain kit (Affymetrix). Arrays were scanned with Affymetrix GeneChip Scanner 3000 7G. Differential miRNA expression was analysed using GeneSpring GX 11.0 (Agilent Technologies) and was assessed as having a minimum 2-fold difference in either direction between two time-points. To determine those miRNA that were differentially regulated with respect to developmental age, the standard deviation of each miRNA across development (E12-P0) was calculated. MiRNA with a standard deviation of >1.3 were considered to be developmentally regulated.
mRNA expression profiling
Total RNA was prepared for microarray using the RNeasy Mini Kit (Qiagen) and amplified with the Illumina TotalPrep RNA Amplification Kit (Ambion). Microarrays were performed using the Illumina Rat-Ref-12_V1 Expression BeadChip, following the manufacturer's instructions, and scanned using the Illumina BeadArray Reader and BeadScan software (Ambion, Scoresby, VIC, Australia). Bead summary data were normalized in GenomeStudio v3 (Illumina) using Cubic Spline. Differential mRNA expression was analysed using GeneSpring GX 11.0 (Agilent Technologies) and was assessed as having a minimum 2-fold difference in either direction between any two time-points.
Target gene and pathway analyses
miRNAs that were determined to be differentially expressed were further analysed for putative target genes under their influence using Ingenuity® microRNA target filter [Ingenuity® Systems (http://www.ingenuity.com)]. Pathways and gene ontology enrichment analysis of the target genes was performed using the DAVID (http://david.abcc.ncifcrf.gov/) [66, 67] and GATHER (http://gather.genome.duke.edu/) [68] online databases. To further investigate biologically relevant targeting, differential miRNA and mRNA expression data was analyzed through the use of Ingenuity® microRNA target filter to generate lists of interactions between genes and miRNAs. Results were filtered based on a high confidence of interaction and an inverse miRNA to target mRNA expression relationship. The identified genes were then subjected to pathways and gene ontology enrichment analysis.
Quantitative real-time reverse transcription PCR
Validation of differentially expressed miRNA was performed by Q-PCR on a subset of the differentially expressed miRNA. Briefly, 500 ng of sample RNA was treated with DNase-I (Invitrogen), and multiplex reverse transcription was performed with Superscript II reverse transcriptase (Invitrogen), a 3 nmol/L mix of miRNA sequence specific primers and primers for U6 small nuclear RNA. The primer sequences for the amplification of specific primers were as follows: rno-miR-98 - 5′-GTAAAACGACGGCCAGTAACAATA-3′ (forward), 5′-TGAGGTAGTAAGTTG-3′ (reverse); rno-miR-212 - 5′-TAA + CAGTCTCCAG-3′ (forward), 5′-GTAAAACGACGGCCAGTCTACGCG-3′ (reverse); rno-miR-137 - 5′-TT + ATT + GCTTAAGAATA-3′ (forward), 5′-GTAAAACGACGGCCAGTCTACGCG-3′ (forward); U6 snRNA 5′-TGCTTCGGCAGCACATATAC-3′ (forward), 5′-AGGGGCCATGCTAATCTTCT-3′ (reverse). Triplicate reactions were set up in a 96-well format with the epMotion 5070 automated pipetting system (Eppendorf, Hamburg, Germany) and carried out with the Applied Biosystems (Foster City, California) 7500 real-time PCR machine. Relative quantification was assessed using the formula 2-ΔCT. The delta Ct was calculated by subtracting the Ct of the endogenous control (geometric mean of U6 small nuclear RNA (snRNA)) from the Ct of the miRNA.
Reporter construction
To generate reporter vectors bearing binding sites for the three miRNA examined in detail, miR-98, miR-212 and miR-137, oligonucleotides encoding target gene miRNA recognition elements (MREs) were annealed to form SpeI and HindIII restricted overhangs of a ligatable cassette compatible with SpeI and HindIII digested pMIR-REPORT vector (Ambion, Austin, TX) as described previously [69, 70]. Recombinant reporter constructs were amplified in chemicompetent DH5alpha Escherichia coli bacteria cells (Bioline, Sydney) and purified using endotoxin free minipreps (Promega, Madison, WI) before being verified by DNA sequencing (AGRF, St. Lucia, QLD, Australia) and quantitated using a NanoDrop 2000 (Thermo Scientific, Delaware, ME, USA).
Cell culture
Human embryonic kidney (HEK)-293 cell cultures were maintained as confluent monolayers at 37°C with 5% CO2 and 90% humidity in Dulbecco’s modified Eagles medium (DMEM) with 10% (vol/vol-1) fetal calf serum, 20 mM HEPES (4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid), 0.15% (wt/vol-1) sodium bicarbonate and 2 mM L-glutamine.
Transfection
For the luciferase assay, HEK-293 cells were seeded into a 96-well plate at a density of 20,000 cells per well and transfected 24 h later using Lipofectamine 2000 (Invitrogen). In each case transfections were performed according to the manufacturer’s instructions, with 100nM synthetic miRNA (Invitrogen). Validation of predicted target genes was accomplished by co-transfecting HEK293 cells with synthetic miRNA and recombinant firefly luciferase reporter gene constructs containing 3’-UTR sequences substituted from the target gene. Each reaction was performed in quadruplicate and replicated at least once. To validate the putative MREs, the HEK293 cell line, which is of embryonic origin and easily transfected, was an ideal candidate for the investigation of the functional regulation of miRNA. Candidate genes were selected based on: their inverse expression respective to their targeting miRNA; their role in neurodevelopment and association with neuropathology; a high MRE-binding specificity score using the miRanda online software [71] (Clasp2 – 142; Rab15 – 158; Cbll1 – 161; Wnt7a – 149; Gpr88 – 150; Plxnb2 – 161).
Reporter gene assay
Twenty-four hours after transfection, cells were harvested in 100 ml Passive Lysis Buffer (Promega, Madison, WI) and 10 ml lysate was used to measure the relative luciferase activity using the dual-luciferase reporter assay kit (Promega, Madison, WI). Reporter gene silencing in response to miRNA co-transfection was monitored with respect to a pRL-TK control plasmid expressing renilla luciferase using a Synergy plate reader with dual reagent injector (Biotek). To control for the nonspecific effects associated with transfection, the controls were co-transfected with mutant miRNAs or mutant anti-miRs (Invitrogen).
Statistical analysis
Statistical significance analysis was assessed using one-way or two-way analysis of variance (ANOVA) with Benjamini Hochberg multiple comparisons testing (Prism software version 5.0; GraphPad Software Inc., San Diego, CA). A value of p < 0.05 was considered significant. The Tukey-HSD test with a level of significance set at p < 0.05 was used for post hoc comparisons. Unsupervised hierarchical clustering was performed in Cluster (Stanford University, Palo Alto, CA, USA) [72]. Data were log transformed and median centred by genes. Genes and arrays were clustered, correlation uncentred, by average linkage clustering and visualized through Java Treeview V.1.1.6r2 (http://jtreeview.sourceforge.net) [73]. Statistical comparison of luciferase data sets was performed by two-tailed paired Student’s t-test.
Electronic supplementary material
Additional file 1: A table listing miRNA expressed in the telencephalon but not the mesencephalon at E12 and at E19. Also listed are miRNA that are specific to the telencephalon at E12 only and at E19 only. (XLSX 50 KB)
Additional file 2: A table listing putative pathways regulated by the E12 and E19 telencephalon-specific miRNA. Enriched pathways of putative target genes of the 32 miRNA specific to the telencephalon at E12 and the 26 miRNA specific to the telencephalon at E19 were identified using the GATHER online database. Putative gene targets were identified using IPA. Only those pathways and ontologies with a Bayes factor >6 were considered significant. (XLSX 42 KB)
Additional file 3: A table listing the 97 dynamically regulated miRNA. Also listed are the standard deviation values for each miRNA in both the forebrain and midbrain. (XLSX 49 KB)
Additional file 4: A table listing enriched ontologies of putative target genes of the 97 dynamically regulated miRNA identified using the GATHER online database. Putative gene targets were identified using IPA. Only those pathways and ontologies with a Bayes factor >6 were considered significant. (XLSX 9 KB)
Additional file 5: A table listing the 100 dynamically regulated mRNA. Also listed are the standard deviation values for each mRNA in both the forebrain and midbrain. (XLSX 46 KB)
Additional file 6: A table listing enriched ontologies of the 100 dynamically regulated miRNA identified using the GATHER online database. Only those pathways and ontologies with a Bayes factor >6 were considered significant. (XLSX 10 KB)
Additional file 7: A table listing enriched pathways of differentially expressed miRNA targeting differentially expressed RNA identified using the DAVID online database. Only those pathways with a p-value <0.05 were considered significant. (XLSX 14 KB)
Additional file 8: A table listing predicted ontologies enriched with target genes of miRs-98, -212 andX - 137. Enriched ontologies of putative target genes of miR-98, miR-212 and miR-137 were identified using the DAVID online database. Putative gene targets were identified using an intersection of the miRanda, IPA and TargetScan output. n, number of input genes in pathway.%, percentage of total target genes analysed that are represented in this pathway. (XLSX 11 KB)
Acknowledgements
The authors declare no biomedical financial interests or potential conflicts of interest. This study was supported by the Schizophrenia Research Institute, utilising infrastructure funding from NSW Ministry of Health and an M.C. Ainsworth Research Fellowship in Epigenetics (MC); and a National Health and Medical Research Council project grant (631057).
Abbreviations
- miRNA
microRNA
- mRNA
Messenger RNA
- E
Embryonic
- P
Postnatal
- KEGG
Kyoto Encyclopedia of Genes and Genomes
- GATHER
Gene Annotation Tool to Help Explain Relationships
- MAP
Mitogen-activated protein
- Wnt
Wingless/int
- IPA
Ingenuity pathway analysis
- GO
Gene ontology
- DAVID
Database for annotation, visualization, and integrated discovery
- Q-PCR
Quantitative real-time reverse transcription PCR
- MRE
miRNA recognition elements
- UTR
Untranslated region
- RIN
RNA Integrity Numbers
- HEK
Human embryonic kidney
- SD
Standard deviation.
Footnotes
Competing interests
The author(s) declare that they have no competing interests.
Authors’ contributions
DE and MC concept and design. SH, EM, DE and MC designed and conceived experiments. SH, BG, EM and AC performed experiments. SH analysed and interpreted all data and wrote the manuscript with input from all authors. MC, DE and FW oversaw the work. All authors read and approved the final manuscript.
Contributor Information
Sharon L Hollins, Email: sharon.hollins@uon.edu.au.
Belinda J Goldie, Email: belinda.goldie@uon.edu.au.
Adam P Carroll, Email: adam.carroll@uon.edu.au.
Elizabeth A Mason, Email: e.mason@uq.edu.au.
Frederick R Walker, Email: rohan.walker@newcastle.edu.au.
Darryl W Eyles, Email: d.eyles@uq.edu.au.
Murray J Cairns, Email: murray.cairns@newcastle.edu.au.
References
- 1.Eagleson KL, Lillien L, Chan AV, Levitt P. Mechanisms specifying area fate in cortex include cell-cycle-dependent decisions and the capacity of progenitors to express phenotype memory. Development. 1997;124:1623–1630. doi: 10.1242/dev.124.8.1623. [DOI] [PubMed] [Google Scholar]
- 2.Florio M, Leto K, Muzio L, Tinterri A, Badaloni A, Croci L, Zordan P, Barili V, Albieri I, Guillemot F, Rossi F, Consalez GG. Neurogenin 2 regulates progenitor cell-cycle progression and Purkinje cell dendritogenesis in cerebellar development. Development. 2012;139:2308–2320. doi: 10.1242/dev.075861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stead JDH, Neal C, Meng F, Wang Y, Evans S, Vazquez DM, Watson SJ, Akil H. Transcriptional profiling of the developing rat brain reveals that the most dramatic regional differentiation in gene expression occurs postpartum. J Neurosci. 2006;26:345–353. doi: 10.1523/JNEUROSCI.2755-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cai Y, Yu X, Hu S, Yu J. A brief review on the mechanisms of miRNA regulation. Genomics Proteomics Bioinformatics. 2009;7:147–154. doi: 10.1016/S1672-0229(08)60044-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fabian MR, Sonenberg N, Filipowicz W. Regulation of mRNA translation and stability by microRNAs. Annu Rev Biochem. 2010;79:351–379. doi: 10.1146/annurev-biochem-060308-103103. [DOI] [PubMed] [Google Scholar]
- 6.Goldie BJ, Cairns MJ. Post-transcriptional trafficking and regulation of neuronal gene expression. Mol Neurobiol. 2012;45:99–108. doi: 10.1007/s12035-011-8222-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Carroll AP, Tooney PA, Cairns MJ. Context-specific microRNA function in developmental complexity. J Mol Cell Biol. 2013;5:73–84. doi: 10.1093/jmcb/mjt004. [DOI] [PubMed] [Google Scholar]
- 8.Miska EA, Alvarez-Saavedra E, Townsend M, Yoshii A, Sestan N, Rakic P, Constantine-Paton M, Horvitz HR. Microarray analysis of microRNA expression in the developing mammalian brain. Genome Biol. 2004;5:R68. doi: 10.1186/gb-2004-5-9-r68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Smirnova L, Gräfe A, Seiler A, Schumacher S, Nitsch R, Wulczyn FG. Regulation of miRNA expression during neural cell specification. Eur J Neurosci. 2005;21:1469–1477. doi: 10.1111/j.1460-9568.2005.03978.x. [DOI] [PubMed] [Google Scholar]
- 10.Smith B, Treadwell J, Zhang D, Ly D, McKinnell I, Walker PR, Sikorska M. Large-scale expression analysis reveals distinct microRNA profiles at different stages of human neurodevelopment. PLoS One. 2010;5:e11109. doi: 10.1371/journal.pone.0011109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Krichevsky AM, King KS, Donahue CP, Khrapko K, Kosik KS. A microRNA array reveals extensive regulation of microRNAs during brain development. RNA. 2003;9:1274–1281. doi: 10.1261/rna.5980303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kapsimali M, Kloosterman WP, de Bruijn E, Rosa F, Plasterk RHA, Wilson SW. MicroRNAs show a wide diversity of expression profiles in the developing and mature central nervous system. Genome Biol. 2007;8:R173. doi: 10.1186/gb-2007-8-8-r173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mukhopadhyay P, Brock G, Appana S, Webb C, Greene RM, Pisano MM. MicroRNA gene expression signatures in the developing neural tube. Birth Defects Res (Part A) Clin Mol Teratol. 2011;91:744–762. doi: 10.1002/bdra.20819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Coolen M, Thieffry D, Drivenes Ø, Becker TS, Bally-Cuif L. miR-9 controls the timing of neurogenesis through the direct inhibition of antagonistic factors. Dev Cell. 2012;22:1052–1064. doi: 10.1016/j.devcel.2012.03.003. [DOI] [PubMed] [Google Scholar]
- 15.Sempere LF, Freemantle S, Pitha-Rowe I, Moss E, Dmitrovsky E, Ambros V. Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol. 2004;5:R13. doi: 10.1186/gb-2004-5-3-r13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Beveridge NJ, Tooney PA, Carroll AP, Tran N, Cairns MJ. Down-regulation of miR-17 family expression in response to retinoic acid induced neuronal differentiation. Cell Signal. 2009;21:1837–1845. doi: 10.1016/j.cellsig.2009.07.019. [DOI] [PubMed] [Google Scholar]
- 17.Manakov SA, Grant SGN, Enright AJ. Reciprocal regulation of microRNA and mRNA profiles in neuronal development and synapse formation. BMC Genomics. 2009;10:419. doi: 10.1186/1471-2164-10-419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nielsen JA, Lau P, Maric D, Barker JL, Hudson LD. Integrating microRNA and mRNA expression profiles of neuronal progenitors to identify regulatory networks underlying the onset of cortical neurogenesis. BMC Neurosci. 2009;10:98. doi: 10.1186/1471-2202-10-98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hua Y-J, Tang Z-Y, Tu K, Zhu L, Li Y-X, Xie L, Xiao H-S. Identification and target prediction of miRNAs specifically expressed in rat neural tissue. BMC Genomics. 2009;10:214. doi: 10.1186/1471-2164-10-214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Olde Loohuis NFM, Kos A, Martens GJM, Van Bokhoven H, Nadif Kasri N, Aschrafi A. MicroRNA networks direct neuronal development and plasticity. Cell Mol Life Sci. 2011;69:89–102. doi: 10.1007/s00018-011-0788-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schratt GM, Tuebing F, Nigh EA, Kane CG, Sabatini ME, Kiebler M, Greenberg ME. A brain-specific microRNA regulates dendritic spine development. Nature. 2006;439:283–289. doi: 10.1038/nature04367. [DOI] [PubMed] [Google Scholar]
- 22.Smrt RD, Szulwach KE, Pfeiffer RL, Li X, Guo W, Pathania M, Teng Z-Q, Luo Y, Peng J, Bordey A, Jin P, Zhao X. MicroRNA miR-137 regulates neuronal maturation by targeting ubiquitin ligase mind bomb-1. Stem Cells. 2010;28:1060–1070. doi: 10.1002/stem.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Maiorano NA, Mallamaci A. Promotion of embryonic cortico-cerebral neuronogenesis by miR-124. Neural Dev. 2009;4:1–16. doi: 10.1186/1749-8104-4-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Giraldez AJ, Cinalli RM, Glasner ME, Enright AJ, Thomson JM, Baskerville S, Hammond SM, Bartel DP, Schier AF. MicroRNAs regulate brain morphogenesis in zebrafish. Science (80-) 2005;308:833–838. doi: 10.1126/science.1109020. [DOI] [PubMed] [Google Scholar]
- 25.Conaco C, Otto S, Han J-J, Mandel G. Reciprocal actions of REST and a microRNA promote neuronal identity. Proc Natl Acad Sci U S A. 2006;103:2422–2427. doi: 10.1073/pnas.0511041103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dugas JC, Cuellar TL, Scholze A, Ason B, Ibrahim A, Emery B, Zamanian JL, Foo LC, McManus MT, Barres BA. Dicer1 and miR-219 Are required for normal oligodendrocyte differentiation and myelination. Neuron. 2010;65:597–611. doi: 10.1016/j.neuron.2010.01.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rice D, Barone S., Jr Critical periods of vulnerability for the developing nervous system: evidence from humans and animal models. Environ Health Perspect. 2000;108(Suppl):511–533. doi: 10.1289/ehp.00108s3511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Clancy B, Finlay BL, Darlington RB, Anand KJS. Extrapolating brain development from experimental species to humans. Neurotoxicology. 2007;28:931–937. doi: 10.1016/j.neuro.2007.01.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Paul A, Cai Y, Atwal GS, Huang ZJ. Developmental coordination of gene expression between synaptic partners during GABAergic circuit assembly in cerebellar cortex. Front Neural Circuits. 2012;6(June):37. doi: 10.3389/fncir.2012.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Meyer U, Nyffeler M, Engler A, Urwyler A, Schedlowski M, Knuesel I, Yee BK, Feldon J. The time of prenatal immune challenge determines the specificity of inflammation-mediated brain and behavioral pathology. J Neurosci. 2006;26:4752–4762. doi: 10.1523/JNEUROSCI.0099-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Eyles D, Feldon J, Meyer U. Schizophrenia: do all roads lead to dopamine or is this where they start? Evidence from two epidemiologically informed developmental rodent models. Transl Psychiatry. 2012;2:e81. doi: 10.1038/tp.2012.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Farh KK-H, Grimson A, Jan C, Lewis BP, Johnston WK, Lim LP, Burge CB, Bartel DP. The widespread impact of mammalian MicroRNAs on mRNA repression and evolution. Science (80-) 2005;310:1817–1821. doi: 10.1126/science.1121158. [DOI] [PubMed] [Google Scholar]
- 33.Stark A, Brennecke J, Bushati N, Russell RB, Cohen SM. Animal MicroRNAs confer robustness to gene expression and have a significant impact on 3’UTR evolution. Cell. 2005;123:1133–1146. doi: 10.1016/j.cell.2005.11.023. [DOI] [PubMed] [Google Scholar]
- 34.Guo H, Ingolia NT, Weissman JS, Bartel DP. Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature. 2010;466:835–840. doi: 10.1038/nature09267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ciani L, Boyle KA, Dickins E, Sahores M, Anane D, Lopes DM. Wnt7a signaling promotes dendritic spine growth and synaptic strength through Ca2+/Calmodulin-dependent protein kinase II. Proc Natl Acad Sci U S A. 2011;108:10732–10737. doi: 10.1073/pnas.1018132108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nishimura N, Van Huyen Pham T, Hartomo TB, Lee MJ, Hasegawa D, Takeda H, Kawasaki K, Kosaka Y, Yamamoto T, Morikawa S, Yamamoto N, Kubokawa I, Mori T, Yanai T, Hayakawa A, Takeshima Y, Nishio H, Matsuo M. Rab15 expression correlates with retinoic acid-induced differentiation of neuroblastoma cells. Oncol Rep. 2011;26:145–151. doi: 10.3892/or.2011.1255. [DOI] [PubMed] [Google Scholar]
- 37.Beffert U, Dillon GM, Sullivan JM, Stuart CE, Gilbert JP, Kambouris JA, Ho A. Microtubule plus-end tracking protein CLASP2 regulates neuronal polarity and synaptic function. J Neurosci. 2012;32:13906–13916. doi: 10.1523/JNEUROSCI.2108-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Quintana A, Sanz E, Wang W, Storey GP, Güler AD, Wanat MJ, Roller BA, La Torre A, Amieux PS, McKnight GS, Bamford NS, Palmiter RD. Lack of GPR88 enhances medium spiny neuron activity and alters motor- and cue-dependent behaviors. Nat Neurosci. 2012;15:1547–1555. doi: 10.1038/nn.3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thomson JM, Newman M, Parker JS, Morin-kensicki EM, Wright T, Hammond SM. Extensive post-transcriptional regulation of microRNAs and its implications for cancer. Genes Dev. 2006;20:2202–2207. doi: 10.1101/gad.1444406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Roush S, Slack FJ. The let-7 family of microRNAs. Trends Cell Biol. 2008;18:505–516. doi: 10.1016/j.tcb.2008.07.007. [DOI] [PubMed] [Google Scholar]
- 41.Logarinho E, Maffini S, Barisic M, Marques A, Toso A, Meraldi P, Maiato H. CLASPs prevent irreversible multipolarity by ensuring spindle-pole resistance to traction forces during chromosome alignment. Nat Cell Biol. 2012;14:295–303. doi: 10.1038/ncb2423. [DOI] [PubMed] [Google Scholar]
- 42.Tognini P, Pizzorusso T. MicroRNA212/132 family: molecular transducer of neuronal function and plasticity. Int J Biochem Cell Biol. 2012;44:6–10. doi: 10.1016/j.biocel.2011.10.015. [DOI] [PubMed] [Google Scholar]
- 43.Wanet A, Tacheny A, Arnould T, Renard P. miR-212/132 expression and functions: within and beyond the neuronal compartment. Nucleic Acids Res. 2012;40:4742–4753. doi: 10.1093/nar/gks151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hollander JA, Im H, Amelio AL, Kocerha J, Bali P, Lu Q, Willoughby D, Wahlestedt C, Conkright MD, Paul J. Striatal microRNA controls cocaine intake through CREB signaling. Nature. 2010;466:197–202. doi: 10.1038/nature09202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Perkins DO, Jeffries CD, Jarskog LF, Thomson JM, Woods K, Newman MA, Parker JS, Jin J, Hammond SM. microRNA expression in the prefrontal cortex of individuals with schizophrenia and schizoaffective disorder. Genome Biol. 2007;8:R27. doi: 10.1186/gb-2007-8-2-r27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Takai Y, Sasaki T, Matozaki T. Small GTP-binding proteins. Physiol Rev. 2001;81:153–208. doi: 10.1152/physrev.2001.81.1.153. [DOI] [PubMed] [Google Scholar]
- 47.Elferink LA, Anzai K, Scheller RH. rab15, a novel low molecular weight GTP-binding protein specifically expressed in rat brain. J Biol Chem. 1992;267:22693. [PubMed] [Google Scholar]
- 48.Figueroa A, Kotani H, Toda Y, Mazan-mamczarz K, Mueller E, Otto A, Disch L, Norman M, Ramdasi RM, Keshtgar M, Gorospe M, Fujita Y. Novel roles of hakai in cell proliferation and oncogenesis. Mol Biol Cell. 2009;20:3533–3542. doi: 10.1091/mbc.E08-08-0845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kaido M, Wada H, Shindo M, Hayashi S. Essential requirement for RING finger E3 ubiquitin ligase Hakai in early embryonic development of Drosophila. Genes Cells. 2009;14:1067–1077. doi: 10.1111/j.1365-2443.2009.01335.x. [DOI] [PubMed] [Google Scholar]
- 50.Sun G, Ye P, Murai K, Lang M-F, Li S, Zhang H, Li W, Fu C, Yin J, Wang A, Ma X, Shi Y. miR-137 forms a regulatory loop with nuclear receptor TLX and LSD1 in neural stem cells. Nat Commun. 2011;2:529. doi: 10.1038/ncomms1532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Geekiyanage H, Chan C. MicroRNA-137/181c regulates serine palmitoyltransferase and in turn amyloid β, novel targets in sporadic alzheimer’s disease. J Neurosci. 2011;31:14820–14830. doi: 10.1523/JNEUROSCI.3883-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Willemsen MH, Vallès A, Kirkels LAMH, Mastebroek M, Olde Loohuis N, Kos A, Wissink-Lindhout WM, de Brouwer APM, Nillesen WM, Pfundt R, Holder-Espinasse M, Vallée L, Andrieux J, Coppens-Hofman MC, Rensen H, Hamel BCJ, van Bokhoven H, Aschrafi A, Kleefstra T. Chromosome 1p21.3 microdeletions comprising DPYD and MIR137 are associated with intellectual disability. J Med Genet. 2011;48:810–819. doi: 10.1136/jmedgenet-2011-100294. [DOI] [PubMed] [Google Scholar]
- 53.Ripke S, Sanders AR, Kendler KS, Levinson DF, Sklar P, Holmans PA, Lin D-Y, Duan J, Ophoff RA, Andreassen OA, Scolnick E, Cichon S, St Clair D, Corvin A, Gurling H, Werge T, Rujescu D, Blackwood DHR, Pato CN, Malhotra AK, Purcell S, Dudbridge F, Neale BM, Rossin L, Visscher PM, Posthuma D, Ruderfer DM, Fanous A, Stefansson H, Steinberg S, et al. Genome-wide association study identifies five new schizophrenia loci. Nat Genet. 2011;43:969–976. doi: 10.1038/ng.940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Green MJ, Cairns MJ, Wu J, Dragovic M, Jablensky A, Tooney PA, Scott RJ, Carr VJ. Genome-wide supported variant MIR137 and severe negative symptoms predict membership of an impaired cognitive subtype of schizophrenia. Mol Psychiatry. 2012;2013:774–780. doi: 10.1038/mp.2012.84. [DOI] [PubMed] [Google Scholar]
- 55.Logue SF, Grauer SM, Paulsen J, Graf R, Taylor N, Sung MA, Zhang L, Hughes Z, Pulito VL, Liu F, Rosenzweig-Lipson S, Brandon NJ, Marquis KL, Bates B, Pausch M. The orphan GPCR, GPR88, modulates function of the striatal dopamine system: a possible therapeutic target for psychiatric disorders? Mol Cell Neurosci. 2009;42:438–447. doi: 10.1016/j.mcn.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 56.Gogolla N, Galimberti I, Deguchi Y, Caroni P. Wnt signaling mediates experience-related regulation of synapse numbers and mossy fiber connectivities in the adult hippocampus. Neuron. 2009;62:510–525. doi: 10.1016/j.neuron.2009.04.022. [DOI] [PubMed] [Google Scholar]
- 57.Gaughwin PM, Ciesla M, Lahiri N, Tabrizi SJ, Brundin P, Björkqvist M. Hsa-miR-34b is a plasma-stable microRNA that is elevated in pre-manifest Huntington’s disease. Hum Mol Genet. 2011;20:2225–2237. doi: 10.1093/hmg/ddr111. [DOI] [PubMed] [Google Scholar]
- 58.Lee S-T, Chu K, Im W-S, Yoon H-J, Im J-Y, Park J-E, Park K-H, Jung K-H, Lee SK, Kim M, Roh J-K. Altered microRNA regulation in Huntington’s disease models. Exp Neurol. 2011;227:172–179. doi: 10.1016/j.expneurol.2010.10.012. [DOI] [PubMed] [Google Scholar]
- 59.Kim J, Inoue K, Ishii J, Vanti WB, Voronov SV, Murchison E, Hannon G, Abeliovich A. A microRNA feedback circuit in midbrain dopamine neurons. Science (80-) 2007;317:1220–1224. doi: 10.1126/science.1140481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Margis R, Margis R, Rieder CRM. Identification of blood microRNAs associated to Parkinsonĭs disease. J Biotechnol. 2011;152:96–101. doi: 10.1016/j.jbiotec.2011.01.023. [DOI] [PubMed] [Google Scholar]
- 61.Miñones-Moyano E, Porta S, Escaramís G, Rabionet R, Iraola S, Kagerbauer B, Espinosa-Parrilla Y, Ferrer I, Estivill X, Martí E. MicroRNA profiling of Parkinson’s disease brains identifies early downregulation of miR-34b/c which modulate mitochondrial function. Hum Mol Genet. 2011;20:3067–3078. doi: 10.1093/hmg/ddr210. [DOI] [PubMed] [Google Scholar]
- 62.Beveridge NJ, Gardiner E, Carroll AP, Tooney PA, Cairns MJ. Schizophrenia is associated with an increase in cortical microRNA biogenesis. Mol Psychiatry. 2010;15:1176–1189. doi: 10.1038/mp.2009.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Santarelli DM, Beveridge NJ, Tooney PA, Cairns MJ. Upregulation of dicer and microRNA expression in the dorsolateral prefrontal cortex Brodmann area 46 in schizophrenia. Biol Psychiatry. 2011;69:180–187. doi: 10.1016/j.biopsych.2010.09.030. [DOI] [PubMed] [Google Scholar]
- 64.Wu JQ, Wang X, Beveridge NJ, Tooney PA, Scott RJ, Carr VJ, Cairns MJ. Transcriptome sequencing revealed significant alteration of cortical promoter usage and splicing in schizophrenia. PLoS One. 2012;7:e36351. doi: 10.1371/journal.pone.0036351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Schroeder A, Mueller O, Stocker S, Salowsky R, Leiber M, Gassmann M, Lightfoot S, Menzel W, Granzow M, Ragg T. The RIN: an RNA integrity number for assigning integrity values to RNA measurements. BMC Mol Biol. 2006;7:3. doi: 10.1186/1471-2199-7-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Huang DW, Sherman BT, Lempicki RA. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc. 2009;4:44–57. doi: 10.1038/nprot.2008.211. [DOI] [PubMed] [Google Scholar]
- 67.Huang DW, Sherman BT, Lempicki RA. Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res. 2009;37:1–13. doi: 10.1093/nar/gkn923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Chang JT, Nevins JR. GATHER: a systems approach to interpreting genomic signatures. Bioinformatics. 2006;22:2926–2933. doi: 10.1093/bioinformatics/btl483. [DOI] [PubMed] [Google Scholar]
- 69.Carroll AP, Tooney PA, Cairns MJ. Design and interpretation of microRNA-reporter gene activity. Anal Biochem. 2013;437:164–171. doi: 10.1016/j.ab.2013.02.022. [DOI] [PubMed] [Google Scholar]
- 70.Carroll AP, Tran N, Tooney PA, Cairns MJ. Alternative mRNA fates identified in microRNA-associated transcriptome analysis. BMC Genomics. 2012;13:561. doi: 10.1186/1471-2164-13-561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.John B, Enright AJ, Aravin A, Tuschl T, Sander C, Marks DS. Human MicroRNA targets. PLoS Biol. 2004;2:e363. doi: 10.1371/journal.pbio.0020363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Eisen MB, Spellman PT, Brown PO, Botstein D. Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci U S A. 1998;95:14863–14868. doi: 10.1073/pnas.95.25.14863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Saldanha AJ. Java Treeview-extensible visualization of microarray data. Bioinformatics. 2004;20:3246–3248. doi: 10.1093/bioinformatics/bth349. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: A table listing miRNA expressed in the telencephalon but not the mesencephalon at E12 and at E19. Also listed are miRNA that are specific to the telencephalon at E12 only and at E19 only. (XLSX 50 KB)
Additional file 2: A table listing putative pathways regulated by the E12 and E19 telencephalon-specific miRNA. Enriched pathways of putative target genes of the 32 miRNA specific to the telencephalon at E12 and the 26 miRNA specific to the telencephalon at E19 were identified using the GATHER online database. Putative gene targets were identified using IPA. Only those pathways and ontologies with a Bayes factor >6 were considered significant. (XLSX 42 KB)
Additional file 3: A table listing the 97 dynamically regulated miRNA. Also listed are the standard deviation values for each miRNA in both the forebrain and midbrain. (XLSX 49 KB)
Additional file 4: A table listing enriched ontologies of putative target genes of the 97 dynamically regulated miRNA identified using the GATHER online database. Putative gene targets were identified using IPA. Only those pathways and ontologies with a Bayes factor >6 were considered significant. (XLSX 9 KB)
Additional file 5: A table listing the 100 dynamically regulated mRNA. Also listed are the standard deviation values for each mRNA in both the forebrain and midbrain. (XLSX 46 KB)
Additional file 6: A table listing enriched ontologies of the 100 dynamically regulated miRNA identified using the GATHER online database. Only those pathways and ontologies with a Bayes factor >6 were considered significant. (XLSX 10 KB)
Additional file 7: A table listing enriched pathways of differentially expressed miRNA targeting differentially expressed RNA identified using the DAVID online database. Only those pathways with a p-value <0.05 were considered significant. (XLSX 14 KB)
Additional file 8: A table listing predicted ontologies enriched with target genes of miRs-98, -212 andX - 137. Enriched ontologies of putative target genes of miR-98, miR-212 and miR-137 were identified using the DAVID online database. Putative gene targets were identified using an intersection of the miRanda, IPA and TargetScan output. n, number of input genes in pathway.%, percentage of total target genes analysed that are represented in this pathway. (XLSX 11 KB)


