Abstract
Background
Recent decades have seen an increase in our understanding of a number of pathophysiological processes associated with type 2 diabetes mellitus (DM). Despite increases in understanding and treatment options, diabetic neuropathy remains a significant problem and is associated with tremendous morbidity and mortality. In this regard, oxidative DNA damage is postulated to play a role in diabetes-mediated neuropathic pathogenesis.
Methods
In this pilot investigation, we studied the extent of chromosomal damage utilizing chromosomal aberration (CA) assay in cultured lymphocytes of patients in 3 subgroups: patients with diabetic neuropathy, patients with type 2 DM and no neuropathy, and a control group.
Results
The patients with diabetic neuropathy showed a statistically significantly higher rate of CA (P<0.001, 0.086 ± 0.04) compared to the DM patients without neuropathy (0.03 ± 0.02). Samples from subjects with diabetic neuropathy were evaluated to check for mutations in the AKR1B1 gene (exon 1). A significant number of mutations appeared after DNA sequencing within the AKR1B1 gene. Of 50 diabetic neuropathy patient samples analyzed, 10 revealed mutations.
Conclusion
Our results suggest that painful diabetic neuropathy is a condition with enhanced genomic instability characterized by increased CA and possible mutations. Exon 1 of the gene AKR1B1 showed significant mutations in patients with painful diabetic neuropathy.
Keywords: Chromosome aberrations, diabetes mellitus, diabetic neuropathies–painful, exons, mutation
INTRODUCTION
Diabetic neuropathy is a significant problem for patients with diabetes mellitus (DM) and is a major cause of morbidity and mortality. The prevalence of diabetic neuropathy in patients with DM varies from 14% to 63%, depending upon the type of population and the criteria used to define diabetic neuropathy.1 Many epidemiological and prospective studies have shown that both increased patient age and duration of diabetes are associated with diabetic neuropathy.2-4 Patients with diabetic neuropathy experience a variety of problems, including paresthesias, allodynia, hyperalgesia, and spontaneous pain that can negatively affect their quality of life.5 Pain is prevalent in 40%-50% of diabetic neuropathy patients.6
Although the mechanisms of painful diabetic neuropathy are not fully understood, observations have led to several postulations.7 Oxidative stress is known to play a crucial role in the cellular and molecular mechanisms of tissue injury in a wide spectrum of disease states.8 Over time, this oxidative DNA damage can change both the structure and function of chromosomes; these changes can lead to chronic disease conditions, such as cancer.9,10
During the last few decades, several techniques have been developed to detect genotoxic effects by assessing the extent of chromosomal damage in patients with DM. Sheth et al11 reported a significant increase in sister chromatid exchange and chromosomal aberration (CA) frequency in patients with type 2 DM compared with those of healthy controls. However, no reports have yet investigated the association between diabetic neuropathy and genotoxic effects.
In this pilot study, we aimed to investigate CA in different subgroups. We compared the rate of CA in patients with diabetic neuropathy to patients with DM without neuropathy and to a control group.
METHODS
Subjects
The study involved a total of 150 subjects. Fifty patients had DM without neuropathy, 50 patients had diabetic neuropathy, and 50 were age- and sex-matched healthy controls. Patients were selected from prescreened DM patients referred from the Vellore Diabetes Centre, Vellore, India. Each subject was on a diabetic diet and had close observation of blood pressure, creatinine, total cholesterol, body mass index, low-density lipoprotein, high-density lipoprotein, and triglycerides. Each subject was evaluated by the same endocrinologist. The onset of painful diabetic neuropathy varied from 1-18 years. Control subjects had normal glucose metabolism and no family history of DM. All patients were nonsmokers and nonalcoholics.
Study Parameters
Each patient's clinical history, including information related to symptoms such as burning, stabbing, crushing, or aching pain was recorded. A family pedigree analysis of DM and cytogenetic studies were evaluated utilizing CA as an endpoint. A biothesiometer was used to measure the vibration perception threshold (VPT) of each subject. The diagnosis of distal, symmetric polyneuropathy was made after careful analysis of history, physical examination, and VPT values. For cytogenetic analysis and the physical and clinical evaluations, informed consent was obtained from all patients, and ethical clearance was obtained from the University Human Ethical Committee, Vellore Institute of Technology, India, and the Institutional Review Board, Yale University. The family histories of all patients were recorded on a spreadsheet. The 2 groups of patients with type 2 DM, those with and without diabetic neuropathy, were compared utilizing a 1-tailed t-test. A P value <0.001 with a confidence interval of 95% was defined as statistically significant. Microsoft Excel and WinSTAT were used for statistical analysis.
Cytogenetic Analysis
The cytogenetic studies were performed on all subjects to determine the frequency of CA. Phytohemagglutinin-stimulated peripheral blood lymphocytes were used for chromosome preparations, and cytogenetic analysis was carried out in Giemsa solution (Sigma)-stained metaphases. Direct microscopic analysis was used to score at least 50 well-spread metaphase plates. Well-spread metaphases were photographed under an oil immersion objective lens (at 100× magnification) of a Leica DM2000 microscope with a MetaSystems camera; the photomicrographs of banded spreads were karyotyped using automatic Ikaros software (MetaSystems). The gene of the AKR1B1 (exon 1) was studied to evaluate mutations in patients with diabetic neuropathy.
RESULTS
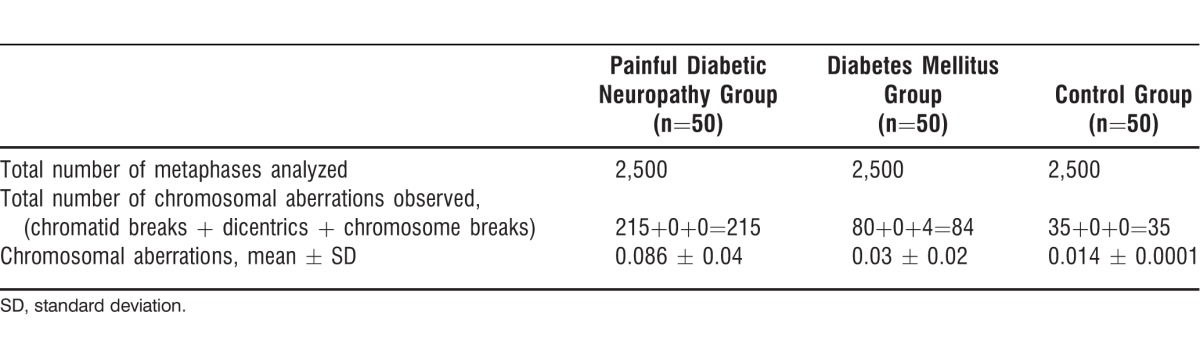
The clinical and general characteristics and rate of CA of patients with diabetic neuropathy are presented in Table 1. The age range was 37-75 years in the patients with diabetic neuropathy (57.86 ± 9.36 years), and their average duration of DM was 9.02 ± 6.78 years. Of these 50 patients, 31 (62%) were males and 19 (38%) were females. Six (12%) patients were consanguineously related. Duration of DM ranged from 6 months to 30 years. Seventeen (34%) patients had a family history of type 2 DM and diabetic neuropathy, and 33 (66%) patients had no family history of DM. Numbness and pain in the feet were observed in 25 (50%) patients, and inflammation and gangrene were observed in 22 (44%) patients. The CA analysis for all 3 groups is summarized in Table 2. Patients with diabetic neuropathy showed a statistically significantly higher rate of CA (P<0.001, 0.086 ± 0.04) than patients with DM alone (0.03 ± 0.02). The frequency of CA per cell was higher (0.11 ± 0.03) in the patients with diabetic neuropathy who had inflammation and gangrene than in the patients with numbness (0.06 ± 0.04) and amputations (0.06 ± 0.02). These results show that the increase in CA was directly related to the severity of the diabetic neuropathy.
Table 1.
Summary of Clinical Characteristics, General Characteristics, and Chromosomal Aberration Frequency
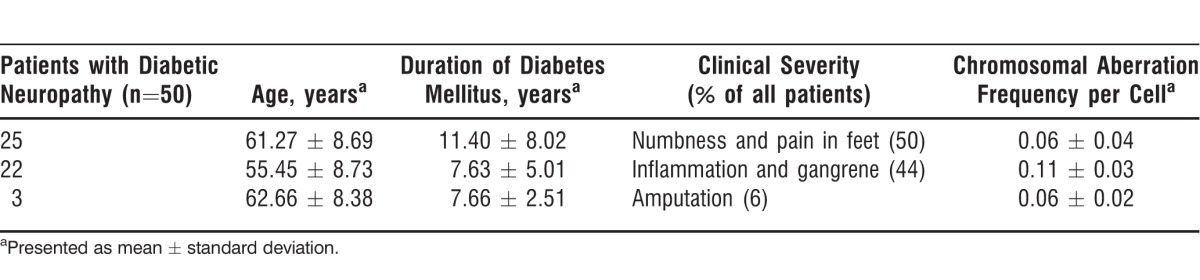
Table 2.
Chromosomal Aberration Analysis
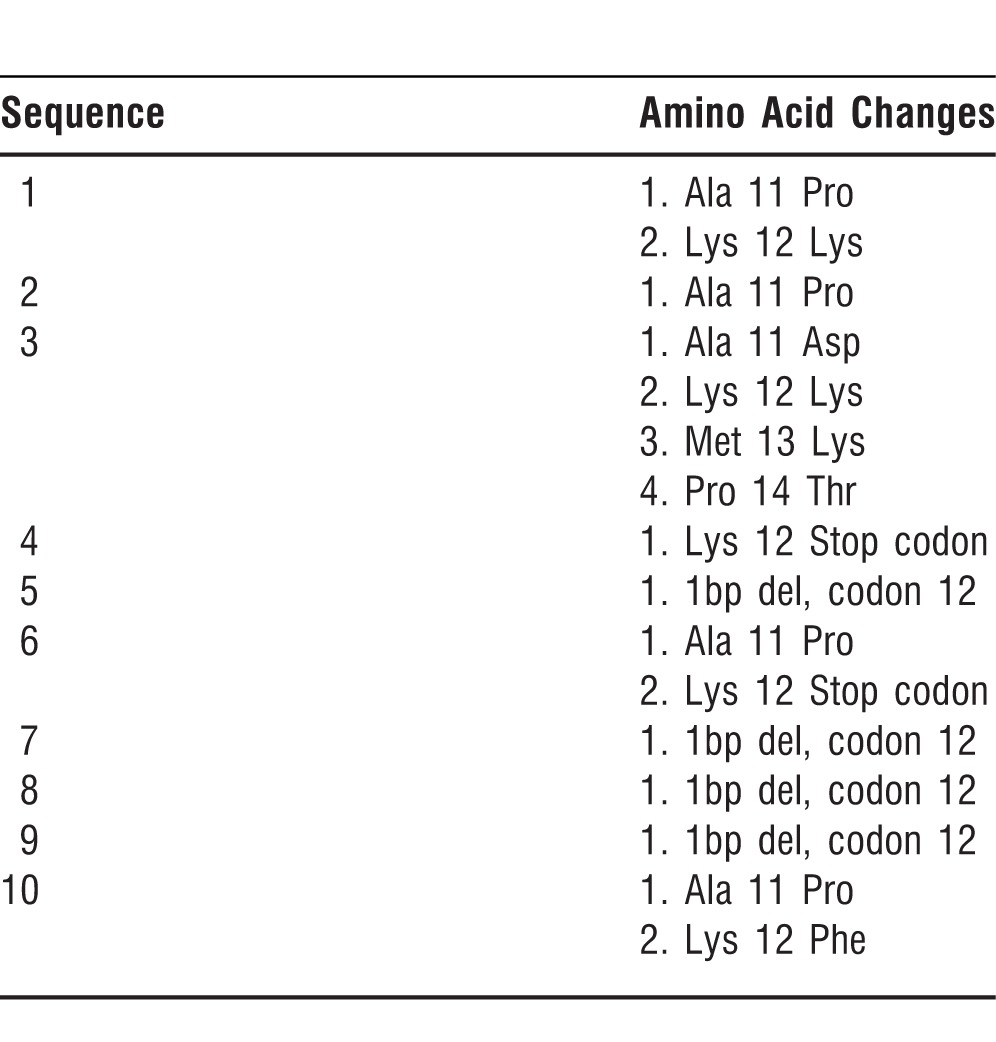
The AKR1B1 gene (exon 1) was evaluated to check mutations in samples of subjects with diabetic neuropathy (n=50). A significant number of mutations were apparent after DNA sequencing within the AKR1B1 gene (Table 3). Exon mutation was seen in samples from 10 of 50 patients with diabetic neuropathy. This type of exon mutation is rare and has never been reported on exon 1 in painful diabetic neuropathy.
Table 3.
Amino Acid Changes in Exon 1 of the AKR1B1 Gene Observed in Patients with Diabetic Neuropathy
DISCUSSION
The detection of CA in peripheral blood lymphocytes has been used to monitor genomic instability in human populations exposed to mutagenic agents. However, no studies are available on the genotoxicity of patients with diabetic neuropathy. Pradeepa et al12 reported that among urban South Indian type 2 DM subjects, the prevalence of diabetic neuropathy is 26.1% and is significantly associated with age, glycosylated hemoglobin, and the duration of DM.
The natural course of painful diabetic neuropathy is variable. The prevalence of pain varies from 10%-20% in subjects with DM and from 40%-50% in those with diabetic neuropathy.6 In our study, the prevalence of pain was varied in the patients with DM. In patients with diabetic neuropathy, numbness and pain in the feet were observed in 25 patients (50%) and inflammation and gangrene in 22 patients (44%).
Contradicting results associated with sex and CA are presented in the literature. Boehm et al reported an enhanced number of structural CA in the peripheral lymphocytes of women with type 2 DM.13 These findings were specific to younger age groups and associated with increased mortality. Another investigation, however, showed a significantly decreased mortality rate in patients with DM.14 The present study data clearly indicate that patients with diabetic neuropathy have a significantly increased level of CA (0.086 ± 0.04) compared to controls (0.014 ± 0.0001).
Regarding cytogenetic testing, we obtained statistically significant data that could have important clinical implications for patients with DM. We observed a significantly increased rate of CA in patients with diabetic neuropathy compared to patients with DM without diabetic neuropathy. AKR1B1 is the gene from the enzyme aldose reductase and ketose reductase. Exon mutation was seen in samples from 10 of 50 diabetic neuropathy patients. This type of exon mutation is rare and has never been reported on exon 1 in painful diabetic neuropathy. The fact that this observation was seen in the present study represents evidence of a link between the AKR1B1 gene and patients with diabetic neuropathy. The precise mechanism involved in mediation or modulation of diabetic pathogenesis from the AKR1B1 gene warrants future investigation.
CONCLUSION
Our results suggest that painful diabetic neuropathy is a condition with enhanced genomic instability characterized by increased CA and possible mutations. Exon 1 of the gene AKR1B1 showed significant mutations in patients with painful diabetic neuropathy. The discovery of specific mutations at the genetic level could lead to the development of novel pharmacotherapeutic agents or of gene therapy for the treatment of patients with painful diabetic neuropathy. Because no therapies are yet proven, future studies are warranted to further understand the relationships and mechanisms involved in the mediation or modulation of diabetic neuropathy.
ACKNOWLEDGMENTS
The authors would like to acknowledge Kalaiselvi A, Tamizhselvan G, Alex George, Badarinath ARS, Girish Rajendran, MSc, Vijay Kodumudi, and Gopal Kodumudi for their help in the preparation of the manuscript. The authors would like to thank Innomed UK Ltd, Crawley, UK for the financial grant and Vellore Institute of Technology University for providing facilities to carry out this study.
Footnotes
The authors have no financial or proprietary interest in the subject matter of this article.
This article meets the Accreditation Council for Graduate Medical Education and the American Board of Medical Specialties Maintenance of Certification competencies for Patient Care, Medical Knowledge, and Practice-Based Learning and Improvement.
REFERENCES
- 1.Boyko EJ. The epidemiology of diabetic neuropathy. In: Veves A, editor. Clinical Management of Diabetic Neuropathy. Totowa, NJ: Humana Press;; 1998. pp. 1–12. In. ed. [Google Scholar]
- 2.Tesfaye S, Stevens LK, Stephenson JM, et al. Prevalence of diabetic peripheral neuropathy and its relation to glycaemic control and potential risk factors: the EURODIAB IDDM Complications Study. Diabetologia. 1996 Nov;39(11):1377–1384. doi: 10.1007/s001250050586. [DOI] [PubMed] [Google Scholar]
- 3.Adler AI, Boyko EJ, Ahroni JH, Stensel V, Forsberg RC, Smith DG. Risk factors for diabetic peripheral sensory neuropathy. Results of the Seattle Prospective Diabetic Foot Study. Diabetes Care. 1997 Jul;20(7):1162–1167. doi: 10.2337/diacare.20.7.1162. [DOI] [PubMed] [Google Scholar]
- 4.Maser RE, Usher DC, DeCherney GS. Little association of lipid parameters and large sensory nerve fiber function in diabetes mellitus. J Diabetes Complications. 1996 Jan-Feb;10(1):54–59. doi: 10.1016/1056-8727(94)00055-7. [DOI] [PubMed] [Google Scholar]
- 5.Calcutt NA. Potential mechanisms of neuropathic pain in diabetes. Int Rev Neurobiol. 2002;50:205–228. doi: 10.1016/s0074-7742(02)50078-7. [DOI] [PubMed] [Google Scholar]
- 6.Veves A, Backonja M, Malik RA. Painful diabetic neuropathy: epidemiology, natural history, early diagnosis, and treatment options. Pain Med. 2008 Sep;9(6):660–674. doi: 10.1111/j.1526-4637.2007.00347.x. [DOI] [PubMed] [Google Scholar]
- 7.Boulton AJ, Malik RA, Arezzo JC, Sosenko JM. Diabetic somatic neuropathies. Diabetes Care. 2004 Jun;27(6):1458–1486. doi: 10.2337/diacare.27.6.1458. [DOI] [PubMed] [Google Scholar]
- 8.Domínguez C, Ruiz E, Gussinye M, Carrascosa A. Oxidative stress at onset and in early stages of type 1 diabetes in children and adolescents. Diabetes Care. 1998 Oct;21(10):1736–1742. doi: 10.2337/diacare.21.10.1736. [DOI] [PubMed] [Google Scholar]
- 9.Shigenaga MK, Hagen TM, Ames BN. Oxidative damage and mitochondrial decay in aging. Proc Natl Acad Sci U S A. 1994 Nov 8;91(23):10771–10778. doi: 10.1073/pnas.91.23.10771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Loft S, Poulsen HE. Cancer risk and oxidative DNA damage in man. J Mol Med (Berl) 1996 Jun;7475(6)(1):297–312. 67–68. doi: 10.1007/BF00207507. Erratum in: J Mol Med. 1997 Jan; [DOI] [PubMed] [Google Scholar]
- 11.Sheth FJ, Patel P, Vaidya ADB, Vaidya R, Sheth J. Increased frequency of sister chromatid exchanges in patients with type II diabetes. Curr Sci. 2006 Jan 25;90(2):236–240. [Google Scholar]
- 12.Pradeepa R, Rema M, Vignesh J, Deepa M, Deepa R, Mohan V. Prevalence and risk factors for diabetic neuropathy in an urban south Indian population: the Chennai Urban Rural Epidemiology Study (CURES-55) Diabet Med. 2008 Apr;25(4):407–412. doi: 10.1111/j.1464-5491.2008.02397.x. Epub 2008 Feb 19. [DOI] [PubMed] [Google Scholar]
- 13.Boehm BO, Möller P, Högel J, et al. Lymphocytes of type 2 diabetic women carry a high load of stable chromosomal aberrations: a novel risk factor for disease-related early death. Diabetes. 2008 Nov;57(11):2950–2957. doi: 10.2337/db08-0274. Epub 2008 Jul 23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.He J, Gu D, Wu X, et al. Major causes of death among men and women in China. N Engl J Med. 2005 Sep 15;353(11):1124–1134. doi: 10.1056/NEJMsa050467. [DOI] [PubMed] [Google Scholar]


