Abstract
NCCN guidelines recommend genetic testing for all triple-negative breast cancer (TNBC) patients aged ≤60 years. However, due to the lack of prospective information in unselected patients, these guidelines are not uniformly adopted by clinicians and insurance carriers. The aim of this study was to determine the prevalence of BRCA mutations and evaluate the utility of NCCN guidelines in unselected TNBC population. Stage I–IV TNBC patients were enrolled on a prospective registry at academic and community practices. All patients underwent BRCA1/2 testing. Significant family history (SFH) was defined >1 relative with breast cancer at age ≤50 or ≥1 relative with ovarian cancer. Mutation prevalence in the entire cohort and subgroups was calculated. 207 TNBC patients were enrolled between 2011 and 2013. Racial/ethnic distribution: Caucasian (80 %), African–American (14 %), Ashkenazi (1 %). Deleterious BRCA1/2 mutations were identified in 15.4 % (32/207) of patients (BRCA1:11.1 %, BRCA2:4.3 %). SFH reported by 36 % of patients. Mutation prevalence in patients with and without SFH was 31.6 and 6.1 %, respectively. When assessed by age at TNBC diagnosis, the mutation prevalences were 27.6 % (≤50 years), 11.4 % (51–60 years), and 4.9 % (≥61 years). Using SFH or age ≤50 as criteria, 25 and 34 % of mutations, respectively, were missed. Mutation prevalence in patients meeting NCCN guidelines was 18.3 % (32/175) and 0 % (0/32) in patients who did not meet guidelines (p = .0059). In this unselected academic and community population with negligible Ashkenazi representation, we observed an overall BRCA mutation prevalence rate of 15.4 %. BRCA testing based on NCCN guidelines identified all carriers supporting its routine application in clinical practice for TNBC.
Keywords: Triple-negative breast cancer, Germline BRCA mutation, Genetic testing guidelines, NCCN guidelines
Introduction
Inherited mutations in BRCA1 and 2 are associated with an extraordinarily high risk of breast and ovarian cancers [1–3]. Women who harbor deleterious mutations in BRCA1 and 2 are faced with difficult, but potentially life-saving preventive strategies, such as prophylactic surgery and/or chemoprevention with anti-estrogen therapies. Furthermore, in addition to being important for preventive counseling regarding second malignancies, BRCA genotyping information also has the potential to aid in guiding therapy. Preclinical and preliminary clinical studies suggest that germline BRCA mutation-associated breast and ovarian cancers are more sensitive to DNA-damaging therapies, such as the platinum salts and poly(adenosine diposphate-ribose) polymerase (PARP) inhibitors [4–10]. Taken together, these considerations emphasize the importance of knowing the germline BRCA1/2 mutation status.
However, due to low prevalence of BRCA mutations in unselected breast cancer patients and expense associated with testing, routine BRCA germline testing is not recommended for all women with breast cancer [11–13]. Rather, recommendations for genetic testing are based on algorithms that utilize risk factors like family history, ethnicity, and age at diagnosis of breast cancer to identify women deemed appropriate for testing. In addition to these conventional risk factors, the intrinsic phenotype of the breast cancer can also impact the probability of finding BRCA mutation. Compared to other subtypes of breast cancers, the population of women with estrogen receptor (ER), progesterone receptor (PR), and ERBB2 (HER2) negative (Triple-negative) breast cancer is enriched for germline BRCA mutations [14–20]. However, the published literature shows a wide variation in the prevalence of germline BRCA mutations in triple-negative breast cancer (TNBC) patients with reported rates varying from 10–42 % [14–23]. The majority of these prior studies evaluated BRCA mutations in either high-risk cohorts (selected by family history, age, or ethnicity) or were based on subsets of patients from tissue banks/clinical practices explaining this variability in the reported prevalence rates.
In light of this variability, governing organizations have not uniformly incorporated the intrinsic subtype of TNBC as an independent criterion in hereditary breast and/or ovarian cancer syndrome (HBOC) testing guidelines (Table 1). For example, the National Comprehensive Cancer Network (NCCN) guidelines recommend genetic risk assessment of all TNBC patients and HBOC testing for all TNBC patients aged ≤60 years regardless of family history [24]. However, the European Society of Medical Oncology (ESMO) guidelines do not specify triple-negative phenotype as a criterion for BRCA mutation testing, but suggest that consideration of triple-negative phenotype in women younger than 50 years may be a cost effective strategy [12, 23, 25]. The National Institute for Health and Care Excellence (NICE) guidelines also do not specify the triple-negative phenotype as a criterion for testing and recommends testing if BRCA mutation carrier probability is ≥10 % [11]. Lack of prospective information on prevalence of BRCA mutations in unselected TNBC patients is one of the reasons underlying the variability in recommendations and adoption of these recommendations by providers and insurance carriers [18, 26]. In 2011, we initiated a multisite prospective registry of TNBC patients in the Kansas City Metropolitan area (P.R.O.G.E.C.T, PROspective evaluation of GErmline mutations, Cancer outcome and Tissue biomarkers in TNBC). One of the initial objectives of this registry was to determine the prevalence of germline BRCA mutations in an unselected TNBC population. We report the prevalence of germline BRCA mutations in an unselected TNBC cohort and discuss our findings in the context of current NCCN guidelines regarding appropriate patient selection for HBOC testing.
Table 1.
Guidelines addressing HBOC testing
| Name of guideline | Incorporation of TNBC subtype | Comments |
|---|---|---|
| National Comprehensive Cancer Network [24] | Yes | HBOC testing recommended for women with TNBC diagnosed at ≤60 years |
| European Society for Medical Oncology [12] | No | Suggests that BRCA testing in women with TNBC diagnosed at ≤50 year may be a cost effectiveness strategy |
| National Institute for Health and Care Excellence [11] | No | Recommends testing if the combined BRCA1 and BRCA2 mutation carrier probability is ≥10 % |
| American Society of Clinical Oncology [30] | No | HBOC testing recommended based on family history and age at diagnosis |
| American Society of Breast Surgeons [31] | Yes | HBOC testing recommended for women with TNBC diagnosed at ≤60 years |
| National Society of Genetic Counselors [32] | Suggested | BRCA testing should be discussed in patients with TNBC |
| U.S. Preventative Services Task Force [33] | No | Guidelines are not applicable to patients with cancer |
TNBC triple-negative breast cancer, HBOC hereditary breast and/or ovarian cancer syndrome
Methods
Patients
Eligible patients with stage I–IV TNBC presenting for treatment at an academic center and five community practice locations in the Kansas City metro area were approached for participation in a registry protocol approved by the University of Kansas Medical Center Human Subjects Committee, the designated Institutional Review Board, as required by 45 CFR 46 and 21 CFR 56. TNBC was defined as negative ER, PR, and HER2/neu status. The clinicians treating the patients made the determination of TNBC status and were encouraged to follow the 2010, American Society of Clinical Oncology and the College of American Pathologists (ASCO/CAP) guidelines for ER/PR and HER2/neu negativity (immunohistochemical nuclear staining of less than 1 % for ER and PR and IHC of 0 or 1+ on and/or lack of gene amplification on FISH testing) [27].
All patients signed a written informed consent. Patients were eligible to participate if they were within five years of diagnosis of TNBC regardless of age at diagnosis of TNBC and family history of breast/ovarian cancer. Appropriate treatment of the TNBC as directed by individual patient’s clinicians was recommended.
Detailed family history, demographic, and clinical information were collected. Both Medicare and NCCN guidelines recommend HBOC testing for breast cancer patients with ≥1 close blood relative (first- second- or third-degree relative) with breast cancer at age ≤50 years or ≥1 close blood relative with epithelial ovarian cancer at any age. Thus, we defined significant family history (SFH) as ≥1 close blood relative (first- second- or third-degree relative) with breast cancer at age ≤50 years or ≥1 close blood relative with epithelial ovarian cancer at any age. Positive family history (FH) was defined as a report of any blood relative with breast or ovarian cancer at any age. Limited family structure was defined as ≤2 two first or second-degree female relatives or female relatives surviving ≥45 years in either lineage (definition used by Medicare and NCCN HBOC testing guidelines). The 2013, NCCN guidelines were applied to all patients. NCCN guidelines recommend HBOC testing for all patients with TNBC diagnosed at age ≤60 and for patients with TNBC diagnosed at age >60 in the presence of appropriate FH of other cancers (www.nccn.org).
Testing for germline BRCA mutations: All patients underwent comprehensive BRACAnalysis® (Myriad Genetic Laboratories, Inc.). Patients who had already completed BRCA testing prior to enrollment were also eligible to participate regardless of the BRCA testing results. For patients who completed BRCA testing after enrollment, the study covered the cost of testing in the event of lack of insurance coverage. Fifty-four percent (112/207) of patients underwent large genomic rearrangement (BART) testing. Appropriate genetic counseling as per standard clinical guidelines was recommended.
Statistical analysis
Study data were collected and managed using Research Electronic Data Capture (REDCap) version 5.3.4 hosted at The University of Kansas Medical Center [28]. REDCap is a secure, web-based application designed to support data capture for research studies. Data analyses were conducted using R version 3.0.0 (R Core Team, 2013) with the RStudio Integrated Development Environment version 0.97.449 (2009–2012, RStudio, Inc.).
Descriptive statistics were generated, including median, minimum and maximum values for age at TNBC diagnosis, and frequencies and relative frequencies (percentages) for categorical measures. The Kruskal–Wallis test was used to compare the distributions of age at diagnosis across BRCA mutation groups (no mutation, BRCA1, or BRCA2) followed by pairwise comparisons using the Wilcoxon rank sum test. Bivariate analyses of categorical measures used Pearson’s Chi-square test, or alternatively Fisher’s exact test when some table cells had expected counts less than five. Proportions and corresponding 95 % Wald confidence intervals (CIs) were generated. To assess the impacts of age at TNBC diagnosis and SFH on the probability of a TNBC subject carrying a deleterious BRCA1 or BRCA2 mutation, unconditional logistic regression was used. Age at diagnosis was treated as continuous, and we tested for a possible interaction between these two explanatory measures in our model. Model assessment included visual inspection of observed versus expected plots as well as the Hosmer–Lemeshow goodness-of-fit test. Predicted probability curves and corresponding point-wise 95 % confidence bands from the resulting model were presented.
Results
Study population
Two hundred eleven (211) patients with stage I–IV TNBC were enrolled from March 2011 to June 2013. There was a high patient uptake for participation with 211 out of 227 (93 %) TNBC patients seen during this time frame at the participating sites enrolled. Since BRCA testing results were pending for four patients, the total sample size for analysis was 207 subjects (Supplementary Figure 1). Table 2 describes the demographic and baseline clinical information for the entire study population. Fifty-eight percent (121/207) of patients were accrued at the academic location and 42 % (86/207) at community locations. Fifty-one percent (105/207) of patients were accrued within 12 months from diagnosis, 27 % (55/207) were accrued 13–36 months from diagnosis and 22 % (47/207) accrued >36 months from diagnosis
Table 2.
Baseline characteristics
| Characteristics | N = 207 |
|---|---|
| Median age (years) at diagnosis (range) | 55 (25–85) |
| Accrual site | |
| Academic | 121 (58 %) |
| Community | 86 (42 %) |
| Menopausal status at diagnosis | |
| Pre/perimenopausal | 85 (41 %) |
| Postmenopausal | 122 (59 %) |
| Race/Ethnicitya | |
| Caucasian | 168 (81 %) |
| African American | 30 (14 %) |
| Asian | 2 (1.0 %) |
| Indian American | 1 (0.5 %) |
| Other | 6 (3.0 %) |
| Ashkenazi | 2 (1.0 %) |
| Hispanic | 5 (2.4 %) |
| Lymph node statusb | |
| Negative | 140 (69 %) |
| Positive | 64 (31 %) |
| Stage | |
| I | 71 (34 %) |
| II | 105 (51 %) |
| III | 24 (12 %) |
| IV | 7 (3.3 %) |
| ER and PR | |
| 0 % | 189 (91 %) |
| 1–5 % | 18 (9 %) |
| Any family history of breast/ovarian cancer | |
| Yes | 128 (62 %) |
| No | 79 (38 %) |
| Significant family history of breast/ovarian cancerc | |
| Yes | 76 (37 %) |
| No | 131 (63 %) |
| Limited Family Structured | |
| Yes | 30 (16 %) |
| No | 158 (86 %) |
Categories for race/ethnicity are not mutually exclusive, therefore percentages add up to a value >100 %
Lymph node information was missing for 3 of the 207 patients
Significant family history was defined as ≥ 1 close blood relative (first- second- or third-degree relative) with breast cancer at age ≤50 years or ≥1 close blood relative with epithelial ovarian cancer at any age
Detailed family tree was not available for 19 of the 207 patients
The immunohistochemical nuclear staining for ER and PR was 0 % in 91 % of the study population and was between 1 and 5 % for the remaining 9 % of the study population. Eighty-one-percent of the population was Caucasian, 14 % African–American, 2.4 % Hispanic, and 1 % Ashkenazi Jewish. This ethnic/racial distribution closely reflects the current population distribution in the Midwestern United States with some under-representation of the Hispanic population (expected 4.4 % vs. observed 2 %) [29]. Sixty-two percent (128/207) of patients reported any FH of breast or ovarian cancer, 37 % (76/207) reported SFH of breast or ovarian cancer, and 16 % of patients were noted to have limited family structure.
BRCA testing information was available prior to enrollment for 37 % (77/207) of patients and the remaining 63 % (130/207) underwent testing after enrolling in the study. The median time from diagnosis of TNBC to completion of BRCA testing for the study cohort was 3.5 months (range 1–98 months).
Mutation prevalence
Deleterious BRCA1 mutations were identified in 11.1 % (23/207) and deleterious BRCA2 mutations in 4.3 % (9/207) of patients giving an overall prevalence rate of 15.4 % (32/207). No patients harbored both deleterious BRCA1 and BRCA2 mutations. Variants of uncertain significance were identified in 3.4 % (7/207) of patients. Large genomic rearrangement testing was not available for subjects who had completed BRCA testing prior to enrollment and in subjects enrolled during the first 12 months of the study thus, is available for only 54 % (112/207) of the study cohort. Large rearrangement mutation in BRCA1 was identified in two African–American patients. The above mentioned 11.1 % BRCA1 mutations include the two large rearrangement mutations.
BRCA mutation and age at TNBC diagnosis
Patients with a deleterious BRCA1 mutation were younger at TNBC diagnosis compared to patients without a deleterious BRCA mutation (median age 40.2 vs. 55.7 years p <0.0001). No differences in age at TNBC diagnosis were detected between patients with a deleterious BRCA2 mutation and patients without a deleterious BRCA mutation (median age 51.0 vs. 55.7 years, p = 0.14).
Mutation prevalence and other factors
Table 3 describes the mutation prevalence by age at diagnosis, FH, accrual location, and NCCN guidelines. Mutation prevalence was higher in younger patients and patients with any FH. When assessed by age at TNBC diagnosis, the BRCA mutation prevalence in patients ≤50 years, 51–60 years, and ≥61 years was 27.6, 11.4, and 4.9 %, respectively (p = 0.0007). BRCA mutation prevalence in patients with and without any FH was 21.1 and 6.3 %, respectively (p = 0.0043), and in patients with and without a SFH was 31.6 and 6.1 %, respectively (p =<0.0001). Mutation prevalence in patients with limited family structure was 10 % (3/30). There was a trend toward higher mutation prevalence in the patients accrued at the academic site compared to community sites (prevalence of 19.0 vs. 10.5 % p = 0.094). However, patients accrued at the academic site were younger and more likely to report a SFH (data not shown). BRCA mutation prevalence in patients who underwent testing prior to and after enrollment was 29 and 8 %, respectively (p = 0.001). However, patients tested before enrollments were younger and more likely to report SFH compared to patients tested after enrollment (data not shown).
Table 3.
Mutation prevalence and clinical and demographic characteristics
| Characteristics | N 207 | Patients with deleterious mutations | Deleterious mutation prevalence % (95 % CI) | p valuea |
|---|---|---|---|---|
| Age at TNBC Diagnosis | ||||
| ≤50 | 76 (37 %) | 21 | 27.6 % (18–38 %) | 0.0007 |
| 51–60 | 70 (34 %) | 8 | 11.4 % (4–19 %) | |
| ≥61 | 61 (29 %) | 3 | 4.9 % ( <1–10 %) | |
| Any family history of breast/ovarian cancer | ||||
| Yes | 128 (26 %) | 27 | 21.1 % (14–28 %) | 0.0043 |
| No | 79 (38 %) | 5 | 6.3 % (1–12 %) | |
| Significant family historyb of breast/ovarian cancer | ||||
| Yes | 76 (37 %) | 24 | 31.6 % (21–42 %) | <0.0001 |
| No | 131 (63 %) | 8 | 6.1 % (2–10 %) | |
| Accrual location | ||||
| Academic | 121 (58 %) | 23 | 19.0 % (12–26 %) | 0.0938 |
| Community | 86 (42 %) | 9 | 10.5 % (4–17 %) | |
| ER and PR | ||||
| 0 % | 189 (91 %) | 28 | 14.8 % (10–20 %) | 0.4909 |
| 1–5 % | 18 (9 %) | 4 | 22.2 % (3–41 %) | |
| Met NCCN guideline for HBOC testing | ||||
| Yes | 175 (85 %) | 32 | 18.3 % (13–24 %) | 0.0059 |
| No | 32 (15 %) | 0c | – | |
TNBC triple-negative breast cancer, HBOC hereditary breast and/or ovarian cancer syndrome
Pearson’s Chi square test, or Fisher’s exact test when table cells had expected counts less than five
Significant family history was defined as ≥1 close blood relative (first- second- or third-degree relative) with breast cancer at age ≤50 years or ≥1 close blood relative with epithelial ovarian cancer at any age
The three patients with TNBC diagnosis at age >60 years met NCCN guidelines for testing based on additional personal/family history of cancer
Age at TNBC diagnosis, family history, and probability of BRCA mutation
In the multivariable model, a priori effects of age at diagnosis and SFH were investigated. Both age at diagnosis and SFH were highly significant predictors of mutation status (p <0.0001 for both). A test for a significant interaction between these effects resulted in p = 0.1927; thus only these main effects were included in the model. Consistent with the visual model diagnostics, no significant lack-of-fit by the Hosmer–Lemeshow goodness-of-fit test was detected (p = 0.09). The resulting modeled probability of carrying a BRCA mutation versus age at diagnosis of TNBC stratified by SFH is presented in Fig. 1. For a patient with a SFH and a TNBC diagnosis at age 51, our model estimated a 29.5 % (95 % CI 19.5–42.0 %) probability of BRCA mutation compared to 5.3 % (95 % CI 2.4–11.0 %) probability if TNBC diagnosis at age 51 and absence of SFH.
Fig. 1.
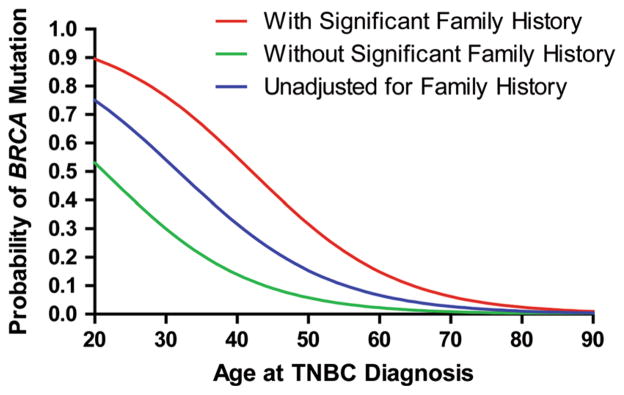
Probability of carrying BRCA mutation by age at TNBC diagnosis and significant family history
Performance of established BRCA testing criteria/guidelines in capturing BRCA mutation carriers
Twenty-five percent (8/32) and 34 % (11/32) of the deleterious BRCA mutations would have been missed using SFH or age ≤50, respectively, as criteria for testing. When both SFH and age ≤50 were used, 12.5 % (4/32) of mutation carriers were missed. When both age ≤50 and any FH were used, 6.3 % (2/32) mutations were still missed.
Insurance coverage for comprehensive BRACAnalysis® and BART was denied for 24 % (49/207) and 32 % (36/112) of the patients, respectively. Of patients with lack of insurance coverage for BRCA testing, 86 % meet NCCN HBOC testing guidelines. Overall, 16 % (5/32) of the deleterious BRCA mutation carriers would have been missed due to lack of adequate insurance coverage.
Eighty-five percent (175/207) of patients met the 2013 NCCN guidelines for HBOC testing. Mutation prevalence in patients who met the NCCN guidelines was 18.3 % (32/175) and 0 % (0/32) in patients who did not meet the NCCN guidelines. Thus, NCCN guidelines captured all mutation carriers in this study population.
Discussion
In this prospective academic and community-based TNBC cohort with negligible Ashkenazi representation, we observed an overall deleterious BRCA mutation prevalence rate of 15.4 %. Our study has several strengths including a sizable cohort of patients who were unselected for age/FH, a good representation of academic and community practices and availability of BRCA1 and BRCA2 mutation testing regardless of insurance coverage. Furthermore, detailed collection of FH allowed us to interrogate the performance and validity of various clinical guidelines in capturing BRCA mutations. We found that utilization of the NCCN testing guidelines detected all deleterious BRCA mutations in our study cohort.
Previously published literature shows a wide variation in the prevalence of germline BRCA mutations in TNBC patients with reported rates varying from 10 to 42 % [14, 15, 17–20]. Some previous studies that reported a higher prevalence (24–34 %) of BRCA mutation in TNBC patients were either done in predominantly Ashkenazi Jewish populations or were focused on young TNBC patients presenting to cancer genetics clinics [14, 16, 17]. Our cohort comprised only two Ashkenazi patients and was not selected based on age and/or FH. Our findings of higher prevalence in younger patients and those with SFH are in agreement with these prior reports. Our findings of mutation rates of 10.5 % in community and 19 % in academic subpopulations are also in agreement with prior reports from community and academic practices [18, 19]. The higher mutation prevalence observed in the academic compared to community practices is likely driven by patient characteristics. This was supported by our observation of academic cohort being younger with more FH compared to the community cohort. This study also provides important insights into the current state of financial insurance coverage for HBOC testing in TNBC. Insurance coverage for BRCA testing was denied for a significant proportion (26 %) of eligible patients, and 16 % percent of the deleterious BRCA mutations in our study cohort would have been missed if patients without insurance coverage were not tested. Thus, financial constraints and insurance coverage remain a challenge for appropriate utilization of BRCA testing in the clinical setting. The majority (91 %) of our cohort meet the current ASCO/CAP ER/PR negativity criterion (<1 %). Our observation of 22 % BRCA mutation prevalence in the subgroup of patients with low (1–5 %) ER/PR is interesting and warrants further study. If confirmed in other studies, perhaps more liberal ER/PR IHC criterion may have to be applied for TNBC definition when making recommendations for BRCA testing.
The detailed collection of demographics and FH allowed us to stratify the prevalence of BRCA mutations based on age and FH and evaluate various BRCA testing guidelines used in clinical settings. As expected, the probability of carrying a BRCA mutation decreased with age of diagnosis. Modeling based on age and FH suggested that even in the older patients presence of FH significantly increased the risk of BRCA mutation. Even though FH and age were important predictors, one-quarter and one-third of the deleterious BRCA mutations would have been missed using SFH or age ≤50, respectively, as criteria alone for BRCA testing. When both SFH and age ≤50 were used, 12.5 % of mutation carriers were still missed. The 2013, NCCN guidelines were the only criteria that identified all BRCA1/2 mutation carriers in our cohort.
Although this is one of the largest studies addressing the prevalence of BRCA mutations in TNBC patients unselected for FH and age of TNBC diagnosis, we acknowledge that the sample size of 207 subjects is modest and represents a limitation of this study. Another potential limitation of our study is an under-representation of Ashkenazi Jewish patients. However, the prevalence of BRCA mutations in TNBC patients with Ashkenazi Jewish ethnicity has previously been reported, and BRCA testing is routinely recommended for all patients of Ashkenazi Jewish ancestry with breast cancer [16, 17]. The mutation prevalence of 15 % in a predominantly non-Ashkenazi Jewish TNBC population is a relevant clinical finding, and we would argue that this constitutes a strength and not weakness of our study. Another limitation of our study is that large genomic rearrangement testing was not available for the entire study cohort. Fifty-four percent of our study population underwent large genomic rearrangement testing and only two women were noted to have a deleterious BRCA1 mutation. A recent study by Hartman and colleagues also reported a very low prevalence (0.5 %) of BRCA large genomic rearrangement in 199 TNBC patients [19]. Thus, lack of large rearrangement testing for the entire cohort may have caused some underestimation of BRCA mutation prevalence, and efforts are currently underway to complete large rearrangement testing for the whole study cohort. We did not assess prevalence of mutations in other genes (like CHEK2 and PALB1) implicated in HBOC or somatic changes leading to DNA damage repair deficits but these studies are underway. These additional analyses will offer further insight into the underlying defects leading to homologous recombination deficiency in TNBC. In conclusion, in a cohort of TNBC patients who were unselected for age and FH, we observed an overall deleterious BRCA mutation prevalence rate of 15.4 % with a prevalence of 18.3 % in patients who met the NCCN guidelines for HBOC testing. BRCA mutation testing based on current NCCN guidelines identified all carriers in this registry supporting its routine application in clinical practice for patients with TNBC.
Supplementary Material
Acknowledgments
This work was supported by The University of Kansas Department of Internal Medicine Research Career Award, KU Cancer Center’s CCSG [P30 CA168524] Biospecimen Repository; and Myriad Genetic Laboratories, Inc.
Abbreviations
- NCCN
National Comprehensive Cancer Network
- HBOC
Hereditary breast and/or ovarian cancer
- TNBC
Triple-negative breast cancer
- ER
Estrogen receptor
- PR
Progesterone receptor
- SFH
Significant family history
- PARP
Poly(adenosine diposphate-ribose) polymerase
- NICE
National Institute for Health and Care Excellence
- ASCO/CAP
American Society of Clinical Oncologists and College of American Pathologists
Footnotes
Electronic supplementary material The online version of this article (doi:10.1007/s10549-014-2980-0) contains supplementary material, which is available to authorized users.
Conflict of interest Priyanka Sharma currently conducts research sponsored by Myriad Genetic Laboratories, Inc. Jennifer Klemp and Larry Geier are members of the speakers’ bureau and receive honoraria from Myriad Genetic Laboratories, Inc. All remaining authors have declared no conflicts of interest.
Ethics Statement All patients signed a written informed consent on a registry protocol approved by the University of Kansas Medical Center Human Subjects Committee, the designated Institutional Review Board, as required by 45 CFR 46 and 21 CFR 56.
Contributor Information
Priyanka Sharma, Email: psharma2@kumc.edu, Division of Hematology/Oncology, Department of Internal Medicine, University of Kansas Medical Center, 2330 Shawnee Mission Parkway, MS5003, Westwood, KS 66205, USA.
Jennifer R. Klemp, Division of Hematology/Oncology, Department of Internal Medicine, University of Kansas Medical Center, 2330 Shawnee Mission Parkway, MS5003, Westwood, KS 66205, USA
Bruce F. Kimler, Department of Radiation Oncology, University of Kansas Medical Center, Kansas City, KS, USA
Jonathan D. Mahnken, Department of Biostatistics, University of Kansas Medical Center, Kansas City, KS, USA. The University of Kansas Cancer Center, Kansas City, KS, USA
Larry J. Geier, Division of Hematology/Oncology, Department of Internal Medicine, University of Kansas Medical Center, 2330 Shawnee Mission Parkway, MS5003, Westwood, KS 66205, USA
Qamar J. Khan, Division of Hematology/Oncology, Department of Internal Medicine, University of Kansas Medical Center, 2330 Shawnee Mission Parkway, MS5003, Westwood, KS 66205, USA
Manana Elia, Division of Hematology/Oncology, Department of Internal Medicine, University of Kansas Medical Center, 2330 Shawnee Mission Parkway, MS5003, Westwood, KS 66205, USA.
Carol S. Connor, Department of Surgery, University of Kansas Medical Center, Kansas City, KS, USA
Marilee K. McGinness, Department of Surgery, University of Kansas Medical Center, Kansas City, KS, USA
Joshua M. W. Mammen, Department of Surgery, University of Kansas Medical Center, Kansas City, KS, USA
Jamie L. Wagner, Department of Surgery, University of Kansas Medical Center, Kansas City, KS, USA
Claire Ward, Division of Hematology/Oncology, Department of Internal Medicine, University of Kansas Medical Center, 2330 Shawnee Mission Parkway, MS5003, Westwood, KS 66205, USA.
Lori Ranallo, The University of Kansas Cancer Center, Kansas City, KS, USA.
Catherine J. Knight, The University of Kansas Cancer Center, Kansas City, KS, USA
Shane R. Stecklein, Department of Pathology & Laboratory Medicine, University of Kansas Medical Center, Kansas City, KS, USA
Roy A. Jensen, The University of Kansas Cancer Center, Kansas City, KS, USA. Department of Pathology & Laboratory Medicine, University of Kansas Medical Center, Kansas City, KS, USA
Carol J. Fabian, Division of Hematology/Oncology, Department of Internal Medicine, University of Kansas Medical Center, 2330 Shawnee Mission Parkway, MS5003, Westwood, KS 66205, USA
Andrew K. Godwin, The University of Kansas Cancer Center, Kansas City, KS, USA. Department of Pathology & Laboratory Medicine, University of Kansas Medical Center, Kansas City, KS, USA
References
- 1.Antoniou A, Pharoah PD, Narod S, Risch HA, Eyfjord JE, Hopper JL, Loman N, Olsson H, Johannsson O, Borg A, Pasini B, Radice P, Manoukian S, Eccles DM, Tang N, Olah E, Anton-Culver H, Warner E, Lubinski J, Gronwald J, Gorski B, Tulinus H, Thorlacius S, Eerola H, Nevalinna H, Syrjakowski K, Kallioniemi OP, Thompson D, Evans C, Peto J, Lalloo F, Evans DG, Easton DF. Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case series unselected for family history: a combined analysis of 22 studies. Am J Hum Genet. 2003;72:1117–1130. doi: 10.1086/375033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen S, Parmigiani G. Meta-analysis of BRCA1 and BRCA2 penetrance. J Clin Oncol. 2007;25:1329–1333. doi: 10.1200/JCO.2006.09.1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mavaddat N, Peock S, Frost D, Ellis S, Platte R, Fineberg E, Evans DG, Izatt L, Eeles RA, Adlard J, Davidson R, Eccles D, Cole T, Cook J, Brewer C, Tischkowits M, Douglas F, Hodgson S, Walker L, Porteous ME, Morrison PJ, Sid LE, Kennedy MJ, Houghton C, Donaldson A, Rogers MT, Dorkins H, Miedzybrodzka Z, Gregory H, Eason J, Barwell J, McCann E, Murray A, Antoniou AC, Easton DF EMBRACE . Cancer risks for BRCA1 and BRCA2 mutation carriers: results from prospective analysis of EMBRACE. J Natl Cancer Inst. 2013;105:812–822. doi: 10.1093/jnci/djt095. [DOI] [PubMed] [Google Scholar]
- 4.Byrski T, Gronwald J, Huzarski T, Grzbowska E, Budryk M, Stawicka M, Mierzwa T, Szwiec M, Wisniowski R, Siolek M, Dent R, Lubinski J, Narod S. Pathologic complete response rates in young women with BRCA1-positive breast cancers after neoadjuvant chemotherapy. J Clin Oncol. 2010;28:375–379. doi: 10.1200/JCO.2008.20.7019. [DOI] [PubMed] [Google Scholar]
- 5.Esteller M. Epigenetic lesions causing genetic lesions in human cancer: promoter hypermethylation of DNA repair genes. Eur J Cancer. 2000;36:2294–2300. doi: 10.1016/S0959-8049(00)00303-8. [DOI] [PubMed] [Google Scholar]
- 6.Ledermann J, Harter P, Gourley C, Friedlander M, Vergote I, Rustin GJS, Scott CL, Meier W, Shapira-Frommer R, Safra T, Matei D, Fielding A, Macpherson E, Dougherty B, Jurgensmeier JM, Orr M, Matulonis U. Olaparib maintenance therapy in patients with platinum-sensitive relapsed serous ovarian cancer (SOC) and a BRCA mutation (BRCAm) J Clin Oncol. 2013;31:5505. [Google Scholar]
- 7.Sandhu SK, Schelman WR, Wilding G, Moreno V, Baird RD, Miranda S, Hylansa L, Riisnaes R, Foster M, Omlin A, Kreischer N, Thway K, Gevensleben H, Sun L, Loughney J, Chatterjee M, Toniatti C, Carpenter CL, Lannone R, Kaye SB, Bono JS, Wenham RM. The poly(ADP-ribose) polymerase inhibitor niraparib (MK4827) in BRCA mutation carriers and patients with sporadic cancer: a phase 1 dose-escalation trial. Lancet Oncol. 2013;14:882–892. doi: 10.1016/S1470-2045(13)70240-7. [DOI] [PubMed] [Google Scholar]
- 8.Silver DP, Richardson AL, Eklund AC, Wang ZC, Szallasi Z, Li Q, Juul N, Leong CO, Calogrias D, Buraimoh A, Fatima A, Gelman RS, Ryan PD, Tung NM, De Nicolo A, Ganesan S, Miron A, Colin C, Sgroi DC, Ellisen LW, Winer EP, Gerber JE. Efficacy of neoadjuvant Cisplatin in triple-negative breast cancer. J Clin Oncol. 2010;28:1145–1153. doi: 10.1200/JCO.2009.22.4725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tutt A, Robson M, Garber JE, Domchek SM, Audeh MW, Weitzel JN, Friedlander M, Arun B, Loman N, Schmutzler RK, Wardley A, Mitchell G, Earl H, Wickens M, Carmichael J. Oral poly(ADP-ribose) polymerase inhibitor olaparib in patients with BRCA1 or BRCA2 mutations and advanced breast cancer: a proof-of-concept trial. Lancet. 2010;376:235–244. doi: 10.1016/S0140-6736(10)60892-6. [DOI] [PubMed] [Google Scholar]
- 10.Wei M, Grushko TA, Dignam J, Hagos F, Nanda R, Sveen L, Xu J, Fackenthal J, Tretiakova M, Das S, Olopade OI. BRCA1 promoter methylation in sporadic breast cancer is associated with reduced BRCA1 copy number and chromosome 17 aneusomy. Cancer Res. 2005;65:10692–10699. doi: 10.1158/0008-5472.CAN-05-1277. [DOI] [PubMed] [Google Scholar]
- 11.NICE clinical guideline. 164: Familial breast cancer. National Institutes for Health and Care Excellence; 2013. [Accessed 10 April 2014]. http://guidance.nice.org.uk/CG164. [PubMed] [Google Scholar]
- 12.Balmana J, Diez O, Rubio IT, Cardoso F. BRCA in breast cancer: ESMO Clinical Practice Guidelines. Ann Oncol. 2011;22(Suppl 6):vi31–vi34. doi: 10.1093/annonc/mdr373. [DOI] [PubMed] [Google Scholar]
- 13.Fackenthal JD, Olopade OI. Breast cancer risk associated with BRCA1 and BRCA2 in diverse populations. Nat Rev Cancer. 2007;12:937–948. doi: 10.1038/nrc2054. [DOI] [PubMed] [Google Scholar]
- 14.Atchley DP, Albarracin CT, Lopez A, Valero V, Amos CI, Gonzalez-Alguno AM, Hortobagyi GN, Arun BK. Clinical and pathologic characteristics of patients with BRCA-positive and BRCA-negative breast cancer. J Clin Oncol. 2008;26:4282–4288. doi: 10.1200/JCO.2008.16.6231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Collins LC, Martyniak A, Kandel MJ, Stadler ZK, Masciari S, Miron A, Richardson AL, Schnitt SJ, Garber JE. Basal cytokeratin and epidermal growth factor receptor expression are not predictive of BRCA1 mutation status in women with triple-negative breast cancers. Am J Surg Pathol. 2009;33:1093–1097. doi: 10.1097/PAS.0b013e31819c1c93. [DOI] [PubMed] [Google Scholar]
- 16.Comen E, Davids M, Kirchhoff T, Hudis C, Offit K, Robson M. Relative contributions of BRCA1 and BRCA2 mutations to “triple-negative” breast cancer in Ashkenazi Women. Breast Cancer Res Treat. 2011;129:185–190. doi: 10.1007/s10549-011-1433-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Foulkes WD, Stefansson IM, Chappuis PO, Bégin LR, Goffin JR, Wong N, Trudel M, Akslen LA. Germline BRCA1 mutations and a basal epithelial phenotype in breast cancer. J Natl Cancer Inst. 2003;95:1482–1485. doi: 10.1093/jnci/djg050. [DOI] [PubMed] [Google Scholar]
- 18.Gonzalez-Angulo AM, Timms KM, Liu S, Chen H, Litton JK, Potter J, Lanchbury JS, Stemke-Hale K, Hennessy BT, Arun BK, Hortobagyi GN, Do KA, Mills BG, Meric-Bernstam F. Incidence and outcome of BRCA mutations in unselected patients with triple receptor-negative breast cancer. Clin Cancer Res. 2011;17:1082–1089. doi: 10.1158/1078-0432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hartman AR, Kaldate RR, Sailer LM, Painter L, Grier CE, Endsley RR, Griffin M, Hamilton SA, Frye CA, Silberman MA, Wenstrup RJ, Sandbach JF. Prevalence of BRCA mutations in an unselected population of triple-negative breast cancer. Cancer. 2012;118:2787–2795. doi: 10.1002/cncr.26576. [DOI] [PubMed] [Google Scholar]
- 20.Cancer Genome Atlas Network . Comprehensive molecular portraits of human breast tumours. Nature. 2012;490:61–70. doi: 10.1038/nature11412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Young S, Pilarski R, Donenberg T, Shapiro C, Hammond LS, Miller J, Brooks KA, Cohen S, Tenenholz B, Desai D, Zandvakill I, Royer R, Li S, Narod SA. The prevalence of BRCA1 mutations among young women with triple-negative breast cancer. BMC Cancer. 2009;9:86. doi: 10.1186/1471-2407-9-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Meyer P, Landgraf K, Hogel B, Eiermann W, Ataseven B. BRCA2 mutations and triple-negative breast cancer. PLoS One. 2012;7:e38361. doi: 10.1371/journal.pone.0038361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Robertson L, Hanson H, Seal S, Warren-Perry M, Hughes D, Howell I, Turnbull C, Houlston R, Shanley S, Butler S, Evans DG, Ross G, Eccles D, Tutt A, Rahman N. BRCA1 testing should be offered to individuals with triple-negative breast cancer diagnosed below 50 years. Br J Cancer. 2012;106:1234–1238. doi: 10.1038/bjc.2012.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines) Genetic/Familial High-Risk Assessment: Breast and Ovarian. National Comprehensive Cancer Network; [Accessed 10 April 2014]. (ed 4.2013) http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. [Google Scholar]
- 25.Kwon JS, Gutierrez-Barrera AM, Young D, Sun CC, Daniels MS, Lu KH, Arun B. Expanding the criteria for BRCA mutation testing in breast cancer survivors. J Clin Oncol. 2010;27:4214–4220. doi: 10.1200/JCO.2010.28.0719. [DOI] [PubMed] [Google Scholar]
- 26.Wang G, Beattie MS, Ponce NA, Phillips KA. Eligibility criteria in private and public coverage policies for BRCA genetic testing and genetic counseling. Genet Med. 2011;13:1045–1050. doi: 10.1097/GIM.0b013e31822a8113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S, Fitzgibbons PL, Francis G, Goldstein NS, Hayes M, Hicks DG, Lester S, Love R, Mangu PB, McShane L, Miller K, Osborne CK, Paik S, Perlmutter J, Rhodes A, Sasano H, Schwartz JN, Sweep FC, Taube S, Torlakovic EE, Valenstein P, Viale G, Visscher D, Wheeler T, Williams RB, Wittliff JL, Wolff AC. American society of clinical oncology/college of american pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol. 2010;28:2784–2795. doi: 10.1200/JOP.777003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)–a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377–381. doi: 10.1016/j.jbi.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.U. S. Census Bureau. [Accessed 10 April 2014];Census Data, 2010 Census Briefs. 2010 http://www.census.gov/2010census/data/
- 30.Robson ME, Storm CD, Weitzel J, Wollins DS, Offit K. American society of clinical oncology policy statement update: genetic and genomic testing for cancer susceptibility. J Clin Oncol. 2010;5:893–901. doi: 10.1200/JCO.2009.27.0660. [DOI] [PubMed] [Google Scholar]
- 31.American Society of Breast Surgeons. Position Statement on BRCA Genetic Testing for Patients With and Without Breast Cancer. American Society of Breast Surgeons; 2012. [Accessed 11 April 2014]. https://www.breastsurgeons.org/statements/PDF_Statements/BRCA_Testing.pdf. [Google Scholar]
- 32.Berliner JL, Fay AM, Cummings SA, Burnett B, Tillmans T. NSGC practice guideline: risk assessment and genetic counseling for hereditary breast and ovarian cancer. J Genet Couns. 2013;22:155–163. doi: 10.1007/s10897-012-9547-1. [DOI] [PubMed] [Google Scholar]
- 33.US Preventive Task Force . Genetic risk assessment and BRCA mutation testing for breast and ovarian cancer susceptibility: recommendation. Ann Intern Med. 2005;143:355–361. doi: 10.7326/0003-4819-143-5-200509060-00011. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


