Abstract
Background. Chloroquine-azithromycin is being evaluated as combination therapy for malaria. It may provide added benefit in treating or preventing bacterial infections that occur in children with malaria.
Objective. We aim to evaluate the effect of treating clinical malaria with chloroquine-azithromycin on the incidence of respiratory-tract and gastrointestinal-tract infections compared to treatment with chloroquine monotherapy.
Methods. We compared the incidence density and time to first events of respiratory-tract and gastrointestinal-tract infections among children assigned to receive chloroquine-azithromycin or chloroquine for all symptomatic malaria episodes over the course of 1 year in a randomized longitudinal trial in Blantyre, Malawi.
Results. The incidence density ratios of total respiratory-tract infections and gastrointestinal-tract infections comparing chloroquine-azithromycin to chloroquine monotherapy were 0.67 (95% confidence interval [CI], .48, .94) and 0.74 (95% CI, .55, .99), respectively. The time to first lower-respiratory-tract and gastrointestinal-tract infections were significantly longer in the chloroquine-azithromycin arm compared to the chloroquine arm (P = .04 and P = .02, respectively).
Conclusions. Children treated routinely with chloroquine-azithromycin had fewer respiratory and gastrointestinal-tract infections than those treated with chloroquine alone. This antimalarial combination has the potential to reduce the burden of bacterial infections among children in malaria-endemic countries.
Keywords: azithromycin, chloroquine, gastrointestinal tract infection, malaria treatment, Malawi, respiratory tract infection, secondary benefit
(See the editorial commentary by Holtz and Tarr on pages 514–6.)
In sub–Saharan Africa, bacterial and parasitic infections are major contributors to the high rates of childhood morbidity and mortality. In these settings, children with fever are frequently treated with an antimalarial drug based on the clinical presumptive diagnosis of malaria or a positive malaria diagnostic test. The use of azithromycin in combination with another antimalarial medication in this context is appealing. Azithromycin has modest antimalarial properties as well as activity against the common bacterial pathogens associated with childhood infections in malaria-endemic countries. Chloroquine and azithromycin have been coformulated and the combination is undergoing testing because of evidence that the combination is effective against chloroquine-resistant malaria in addition to its benefits in treating and preventing bacterial infections [1, 2].
The strongest existing evidence to support azithromycin's ability to protect against future bacterial illnesses comes from studies of the delivery of azithromycin through mass drug administration for the treatment of trachoma. Children from villages where azithromycin was administered have been shown to have lower rates of respiratory infection, diarrhea, impetigo, and fever in children after the treatment campaign compared to children in villages which did not receive the azithromycin intervention [3–6]. Mass azithromycin administration has even been associated with a measurable decrease in child mortality [7, 8]. Many of these results emerge from studies with limited generalizability because they compared intervention villages where trachoma was prevalent and mass drug administration was implemented to villages where trachoma was rare and there were no new public health programs [3, 4] or they compared children who did or did not receive the azithromycin during the mass drug campaign [7]. Both types of comparison fail to control for other factors that could affect the risk of bacterial disease. One of these studies did not identify a control group [6]. Because trachoma is linked to poor basic hygiene and lower socioeconomic status [9], baseline factors that influence risk of both trachoma and bacterial disease may have differed between the treated and untreated communities and individuals. In addition, the presence of programs, personnel and infrastructure associated with mass drug administration may have influenced outcomes.
In Malawi, chloroquine-susceptible malaria became ubiquitous several years after the cessation of chloroquine use in 1993 and chloroquine has since demonstrated excellent clinical efficacy against malaria illness [10, 11]. We recently completed a clinical trial in Malawi, comparing chloroquine monotherapy to chloroquine plus artesunate, atovaquone-proguanil, or azithromycin for the treatment of uncomplicated malaria in children and demonstrated that chloroquine alone or in combination had sustained high antimalarial efficacy when used for repeated episodes of malaria over the course of a year [12]. The equivalent antimalarial efficacy of all treatment arms offered a unique opportunity to examine the potential secondary benefits of including azithromycin, when it is combined with a highly effective antimalarial medication. We did not include the other combination therapy arms in the control group because artesunate and atovaquone-proguanil have antimicrobial properties. We conducted active and passive case detection of all illnesses occurring during 12 months of study surveillance and used the information captured about nonmalarial illnesses to assess the ability of azithromycin to prevent key infections. Because observations occurred as part of a randomized clinical trial, any differences in outcomes can likely be attributed to the intervention.
METHODS
Clinical Trial Study Design
This is a secondary analysis of data collected from a randomized, open-label clinical trial designed to evaluate the treatment efficacy of chloroquine monotherapy versus chloroquine in combination with artesunate, atovaquone-proguanil, or azithromycin for repeated episodes of uncomplicated malaria. Children with an additional potential source of fever, including evidence of respiratory or gastrointestinal infection at the time of the initial malaria episode, were not enrolled. Children with known human immunodeficiency virus infection were excluded from the study because the standard use of trimethoprim-sulfamethoxazole prophylaxis, with antimalarial activity, would interfere with efficacy assessment. At every episode of symptomatic malaria infection, participants were treated with the regimen to which they were initially randomized. The treatment dose of chloroquine was 10 mg/kg on days 0 and 1 and 5 mg/kg on day 2, and the dose of azithromycin was 30 mg/kg daily for 3 days. Details of the methods of this study and the results (which showed no difference in antimalarial efficacy between the treatment arms) were reported previously [12]. Participants were evaluated every month and at unscheduled visits any time they felt unwell. At each visit, hemoglobin concentration was measured and participants were assessed for malaria and other illnesses. Adverse events were recorded at every visit as they were identified.
Study Population
This analysis included 160 participants randomized to the chloroquine-azithromycin treatment group and 160 participants randomized to chloroquine monotherapy as the comparator group. All data from the time of enrollment until study completion were included from participants who were randomized.
Outcome Variables
The primary outcome variables for this analysis were respiratory-tract infection and gastrointestinal-tract infection events recorded during the 12-month postrandomization follow-up period. Study participants visited the clinic every month for active surveillance and were also encouraged to attend the clinic any time they were ill. Respiratory-tract infection and gastrointestinal-tract infection data were obtained from adverse event documentation. Diagnoses were made based on clinical signs and symptoms by study clinicians and physicians, including participant complaints and physical examination; treatment was prescribed according to the standard treatment guidelines in Malawi. Respiratory-tract infection events were subcategorized based on the diagnosis and anatomical site affected. Lower-respiratory-tract infection included pneumonia, bronchopneumonia, and bronchitis. Upper-respiratory-tract infection included only pharyngitis and tonsillitis. Acute otitis media, chronic otitis media, and ear infection were categorized as otitis media. Clinical diagnoses of mild illnesses, such as rhinitis and viral upper-respiratory-tract infection, were not included. Gastrointestinal-tract infections events included diarrhea, dysentery, and gastroenteritis (vomiting and diarrhea). Weight-for-age and height-for-age were calculated based on the World Health Organization (WHO) child growth standards.
Statistical Analysis
Incidence rates for total respiratory-tract infections and total gastrointestinal-tract infections were calculated as the ratio of total events per total person-years for each treatment group, where total person-years were calculated as the sum of the difference in days between enrollment and the final study visit or early termination. Participants who permanently left the study area, withdrew consent, or missed more than 2 consecutive monthly visits were discontinued from the study, but their observation period up until the time of censoring was included in the analyses. A generalized linear model was used to fit a Poisson, or quasi-Poisson if the data were overdispersed, distribution for the estimation of rates and rate ratios by analysis of maximum likelihood for respiratory-tract infections and gastrointestinal-tract infections. The analysis was repeated separately for the subcategories of upper-respiratory-tract infection, lower-respiratory-tract infection, otitis media, diarrhea, dysentery, and gastroenteritis events. Time to first respiratory-tract infection or gastrointestinal-tract infections events and time to first event of each subcategory were modeled using Kaplan–Meier methodology and compared using the log-rank test. Median times to event are presented because data were censored at last visit. To assess for effect modification, we fit models including the baseline variables for age, weight-for-age Z score, and hemoglobin. Numbers needed to treat estimates were calculated as the ratio of the absolute risk differences and corresponding confidence intervals.
Analyses were performed in SAS 9.2 (SAS Institute, Cary, NC) and in R version 2.15.2 (R Core Team, Vienna, Austria). P values are based on 2-sided tests and are unadjusted for multiple comparisons. P values ≤ .05 were considered statistically significant.
Ethical Considerations
All parents or guardians provided written informed consent prior to the initiation of any study-related procedures. The study protocol was reviewed and approved by the University of Malawi College of Medicine Research and Ethics Committee and the Institutional Review Board of the University of Maryland, Baltimore. The study was registered on ClinicalTrials.gov (NCT00379821).
RESULTS
Baseline Characteristics
Baseline characteristics for the 2 treatment groups were similar (Table 1). The average age in both groups was 33 months, and females comprised approximately half of the study population. The study participants had slightly lower weight and height for age than expected based on WHO growth standards, but the 2 groups had similar nutritional status at enrollment. Mean hemoglobin concentrations were low and all participants had uncomplicated malaria at enrollment into the clinical trial.
Table 1.
Baseline Characteristics of Participants Randomized to Receive Chloroquine Plus Azithromycin or Chloroquine Alone for Treatment of Malaria
| Baseline Variable | CQ-AZ (n = 160) | CQ (n = 160) | Difference (95% CI) |
|---|---|---|---|
| Mean age (months) | 33.0 ± 12.0 | 33.0 ± 13.2 | 0 (−3.0, 3.0) |
| Females (%) | 73 (49) | 77 (51) | 0.02 (−.1, .1) |
| Mean weight for age (Z score) | −1.0 ± 0.1 | −1.1 ± 0.1 | −0.1 (−.3, .1) |
| Mean height for age (Z score) | −1.5 ± 0.1 | −1.6 ± 0.1 | −0.1 (−.3, .1) |
| Mean initial hemoglobin (g/dL) | 9.9 ± 0.1 | 9.6 ± 0.1 | −0.4 (−.7, 0) |
Abbreviations: CI, confidence interval; CQ, chloroquine monotherapy group; CQ-AZ, chloroquine-azithromycin group.
Follow-up Characteristics
The total duration of follow-up was 121.7 and 113.3 person-years for the chloroquine-azithromycin and chloroquine alone groups, respectively. The median follow-up time was similar between the groups. Early termination was predominantly due to migration out of the study area. Follow-up rates and reasons for loss to follow-up were similar in both groups (Table 2).
Table 2.
Participant Follow-up Characteristics by Treatment Group
| Summary Variable | Treatment Group |
P Value | |
|---|---|---|---|
| CQ-AZ (n = 160) | CQ (n = 160) | ||
| Subsequent malaria treatment episodes (n, %) | |||
| 1 | 141 (88) | 151 (94) | .08 |
| 2 or more | 19 (12) | 9 (6) | |
| Early termination (n, %) | 60 (38) | 72 (45) | .14 |
| Reasons for early termination (n, %) | |||
| Migration | 39 (65) | 47 (65) | |
| Lost to follow-up | 14 (23) | 11 (15) | |
| Parental consent withdrawal |
5 (8) | 11 (15) | |
| Investigator withdrawal | 1 (2) | 1 (1) | |
| Serious adverse event/death |
1 (2) | 2 (3) | |
| Mean person-years follow-up | 0.75 | 0.69 | .15 |
Abbreviations: CQ, chloroquine monotherapy; CQ-AZ, chloroquine-azithromycin group.
Respiratory-Tract and Gastrointestinal-Tract Infections Incidence Density
Respiratory-tract infections and gastrointestinal-tract infection were common in this population. In the chloroquine-azithromycin and chloroquine alone groups, 30% and 35%, respectively had at least 1 respiratory-tract infection during the course of the study (P = .43). Forty percent and 31% of children in the chloroquine-azithromycin and chloroquine alone groups, respectively, had at least 1 gastrointestinal-tract infection during the course of the study (P = .13).
The overall incidence density of respiratory-tract infections was one-third lower in the chloroquine-azithromycin group compared to the chloroquine alone arm (rate ratio 0.67, P = .02). When analyzing anatomic sites of respiratory-tract infections separately, only the incidence of lower-respiratory-tract infections was statistically significantly reduced in the chloroquine-azithromycin group compared to the control group (Table 3).
Table 3.
Episodes, Incidence Densities, and Rate Ratios of Respiratory-Tract Infections, by Treatment Group
| CQ-AZ |
CQ |
Rate Ratio (95% CI) | P Value | |||
|---|---|---|---|---|---|---|
| Count | Rate | Count | Rate | |||
| All RTI events | 65 | 0.53 | 90 | 0.79 | 0.67 (.48, .94) | .02 |
| Lower RTI events | 47 | 0.39 | 65 | 0.57 | 0.67 (.45, .99) | .05 |
| Upper RTI events | 3 | 0.02 | 4 | 0.04 | 0.70 (.10, 4.13) | .92 |
| Otitis media events | 15 | 0.12 | 21 | 0.19 | 0.66 (.32, 1.35) | .29 |
Rate calculated as event per total person-years of follow-up (CQ-AZ = 121.7, CQ = 113.3). Total person-years calculated as total time from enrollment until censoring at termination from the study. Rate ratio is event rate for chloroquine-azithromycin over the rate for chloroquine monotherapy.
Abbreviations: CI, confidence interval; CQ: chloroquine monotherapy group; CQ-AZ, chloroquine-azithromycin group; RTI, respiratory-tract infection.
The overall incidence density of gastrointestinal-tract infections was 26% lower in the chloroquine-azithromycin group compared to chloroquine alone group (rate ratio 0.74, P = .04). In the analysis of specific clinical diagnoses of gastrointestinal-tract infections, the incidence of diarrhea was statistically significantly lower in the chloroquine-azithromycin compared to the chloroquine alone group (Table 4).
Table 4.
Episodes, Incidence Densities, and Unadjusted Rate Ratios of Gastrointestinal-Tract Infections, by Treatment Group
| CQ-AZ |
CQ |
Rate Ratio (95% CI) | P Value | |||
|---|---|---|---|---|---|---|
| Count | Rate | Count | Rate | |||
| All GITI events | 88 | 0.74 | 111 | 0.98 | 0.74 (.55, .99) | .04 |
| Diarrhea events | 33 | 0.27 | 49 | 0.43 | 0.63 (.39, .99) | .05 |
| Dysentery events | 14 | 0.12 | 14 | 0.12 | 0.93 (.41, 2.11) | 1.0 |
| Gastroenteritis events | 41 | 0.34 | 48 | 0.42 | 0.80 (.51, 1.23) | .33 |
Rate calculated as event per total person-years (CQ-AZ = 121.7, CQ = 113.3). Total person-years calculated as total time from enrollment until censoring at termination from the study. Rate ratio is event rate for chloroquine-azithromycin over the rate for chloroquine monotherapy.
Abbreviations: CI, confidence interval; CQ, chloroquine monotherapy group; CQ-AZ, chloroquine-azithromycin group; GITI, gastrointestinal-tract infection.
In the subgroup analysis, there was no evidence of effect modification by age and weight-for-age Z score for the total respiratory-tract infection and gastrointestinal-tract infection rate ratios. However, there was evidence of a modest effect modification by hemoglobin for total gastrointestinal-tract infections where hemoglobin was categorized as severe anemia (5.0–6.9 mg/dL), mild to moderate anemia (7.0–11.0 mg/dL), and no anemia (>11.0 mg/dL). Only participants with severe anemia showed a significantly lower rate of gastrointestinal-tract infections in the chloroquine-azithromycin group, with a rate ratio of 0.16 (95% confidence interval [CI], .04, .61).
Time to First Event
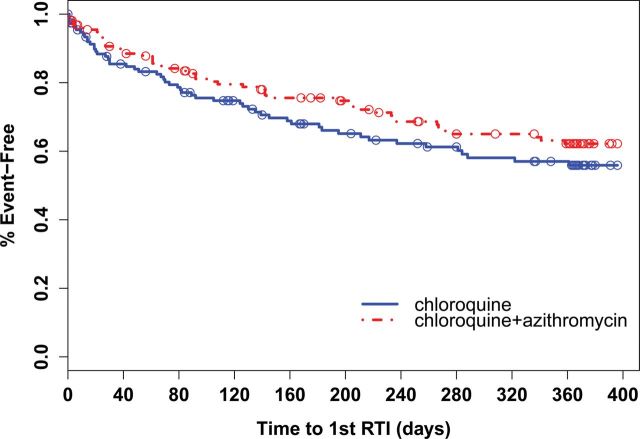
Times to first respiratory-tract infection were similar between treatment groups (log-rank, P = .22) with median times of 0.64 and 0.41 years in the chloroquine-azithromycin and chloroquine alone groups, respectively (Figure 1). When analyzing by subcategory, time to first lower-respiratory-tract infection was statistically significantly longer in the chloroquine-azithromycin arm compared to the chloroquine alone group (median time 0.88 vs 0.46 years, P = .04).
Figure 1.
Kaplan–Meier analysis comparing time to first respiratory-tract infection (RTI) between treatment groups. Open circles indicate time points where a participant terminated the study (censored). Log-rank test P = .22.
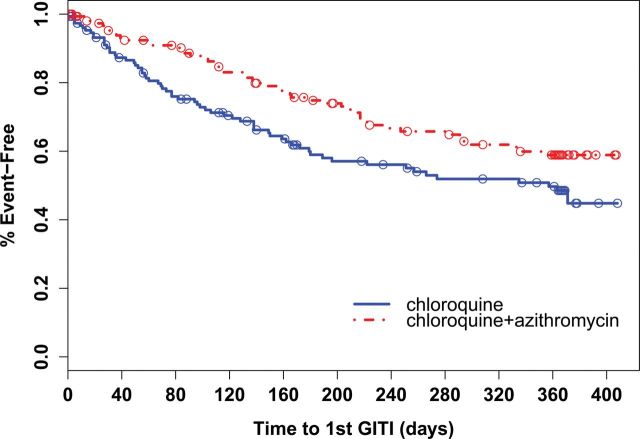
Time to first gastrointestinal-tract infection event was statistically significantly longer in the chloroquine-azithromycin group compared to the chloroquine alone group (median time 0.58 vs 0.38 years, P = .02) (Figure 2). Of the gastrointestinal-tract infections infection subtypes, only the time to first diarrheal event was significantly longer in the chloroquine-azithromycin group compared to the chloroquine alone group (median time 0.97 vs 0.70 years, P = .03).
Figure 2.
Kaplan–Meier analysis comparing time to first gastrointestinal-tract infection (GITI) between treatment groups. Open circles indicate time points where a participant terminated the study (censored). Log-rank test P = .02.
Number Needed to Treat Analysis
The absolute risk differences for respiratory-tract infection and gastrointestinal-tract infections between the chloroquine-azithromycin and chloroquine alone groups were 0.16 (95% CI, .05, .27) and 0.14 (95% CI, .07, .22), respectively. This corresponds to a number needed to treat of 7 for both respiratory-tract infections and gastrointestinal-tract infections. For every 7 children treated for malaria with chloroquine-azithromycin, on average 1 case of respiratory-tract infection and 1 case of gastrointestinal-tract infections could be prevented when compared to treating with chloroquine alone.
DISCUSSION
Adding azithromycin to the treatment of malaria prevented 33% of respiratory-tract infection and 26% of gastrointestinal-tract infections in Malawi. Both respiratory- and gastrointestinal-tract infections were significantly reduced, and the protective efficacy was most significant among infections, such as lower-respiratory-tract infections, that were likely to be bacterial in origin. To our knowledge, this is the most definitive evidence that the use of azithromycin has a detectable, long-lasting benefit in the protection against new infections. Because respiratory-tract infections and gastrointestinal-tract infections are so common in this population, the impact of the addition of azithromycin to malaria treatment is high: every 7 treatment episodes with chloroquine-azithromycin will prevent 1 respiratory-tract infection and 1 gastrointestinal-tract infection.
The significant findings in our study appear to be driven by prevention of lower-respiratory-tract infections and diarrhea, which were the most commonly observed infections. The lack of statistical significance for upper-respiratory-tract infections and nondiarrheal gastrointestinal-tract infections may be due to their low incidence or because azithromycin did not have an impact on prevention of those specific infections. The latter explanation is less likely, given the spectrum of activity of azithromycin and the common pathogens in diseases such as otitis media, pharyngitis, and dysentery. Nonetheless, all the point estimates were uniformly in favor of lower infection rates in the chloroquine-azithromycin group compared to the chloroquine monotherapy group.
This study was done as part of a clinical trial in which all study participants were from the same health center catchment area and randomly assigned to treatment with or without azithromycin, reducing bias that may have been present in previous nonrandomized evaluations of azithromycin's benefits in African children. Another important strength of our study is the expert and comprehensive clinical evaluation and diagnosis of the endpoints of interest. Previous studies have relied on guardian report and field worker assessment so that the diagnoses were likely less specific than in our study, where trained clinicians and physicians were responsible for determining the diagnosis. In addition, the follow-up period of 1 year allowed for the opportunity to detect extended efficacy or rebound infections.
The baseline rate of lower-respiratory-tract infections in the chloroquine control arm of 0.39 per child year is very similar to the global estimate of 0.33 episodes per child-year for children in developing countries [13]. In contrast, the rate of gastrointestinal-tract infections in the chloroquine alone group was more than 3-fold lower than estimates for this age group in developing countries (0.7 vs 2.7 episodes per child-year) [14]. The measured rates of both respiratory-tract infections and gastrointestinal-tract infections were both lower than the baseline rates reported from the children enrolled in the mass treatment program for trachoma in Tanzania. The lower rates of infection in this study are likely due to the urban setting of the study, in which children live in permanent housing, have minimal exposure to indoor smoke, and have good access to clean water [15]. Nevertheless, our study confirms the magnitude of the benefit of azithromycin measured in studies performed in Tanzania [3, 4].
The precise mechanism by which azithromycin prevents respiratory- and gastrointestinal-tract infections is not known. It may directly eradicate pathogenic bacteria in the nasopharynx and gastrointestinal tract. Although azithromycin has activity against Streptococcus pneumoniae and group A Streptococcus, recent studies of azithromycin for mass drug administration have not shown that it had an effect on S. pneumoniae colonization rates [5, 16]. A short course of azithromycin also was not associated with prolonged decreases in colonization with enteric pathogens [17]. However, the immunomodulatory effects of azithromycin have been shown to improve outcomes for children with cystic fibrosis and noncystic fibrosis bronchiectasis [18, 19]. Further investigation is required to evaluate the mechanism of azithromycin's protection against respiratory and gastrointestinal symptoms.
The most significant limitation of this study is lack of microbiological data. Identifying the etiology of respiratory-tract infections and gastrointestinal-tract infections in any context is challenging because many basic culture or highly sensitive detection methods yield multiple pathogens. Such investigations are not part of routine evaluation of illnesses in resource-limited settings. Many, if not most, of the respiratory and gastrointestinal illnesses were likely due to common viral etiologies, such as influenza, respiratory syncytial virus, and rotavirus. Such misclassification would have diluted our ability to detect differences between the 2 groups. The fact that statistically significant differences were still detectable suggests that azithromycin would have protected against more than a third or a quarter of infections if we had limited our analyses to bacterial infections using more specific case definitions. Another limitation of the study was the high rate of loss to follow-up among the participants. We routinely encounter this in our urban setting where the population is highly mobile. The rates of loss to follow-up were similar in both groups and the baseline characteristics of participants who did and did not complete the study were equivalent. It is unlikely that incomplete follow-up led to a bias in the results.
The role of combining chloroquine plus azithromycin in the treatment and prevention of both malaria and other infections is being explored in other field trials. Currently, a multicenter phase III clinical trial is evaluating a fixed-dose combination of chloroquine plus azithromycin for the intermittent preventive treatment of malaria during pregnancy [20]. The expected advantage of using chloroquine-azithromycin in pregnancy is that azithromycin will also treat common sexually transmitted diseases, including Neisseria gonorrhea, Chlamydia trachomatis, chancroid, and syphilis [2]. The treatment and prevention of sexually transmitted disease in pregnant women are especially important because the infections are associated with adverse pregnancy outcomes, including preterm delivery, low birth weight, growth restriction, and fetal and neonatal mortality [21]. The results of this study in Malawi further support the use of chloroquine-azithromycin for the treatment of current malaria and bacterial infections as well as for the posttreatment prophylactic effect.
Chloroquine-azithromycin may play a role in the treatment or prevention of malaria in children in the future, particularly if emerging artemisinin resistance becomes more widespread [22, 23]. Due to widespread chloroquine-resistant malaria and the relatively weak antimalarial activity of azithromycin, multicenter trials have only demonstrated the efficacy of chloroquine plus azithromycin compared to standard antimalarial treatment among semi-immune adults in settings outside of Malawi [24]. The combination demonstrated outstanding efficacy in our clinical trial in Malawi because of the return of chloroquine-susceptible malaria in Malawi following the removal of chloroquine from use in 1993 [10, 11]. As chloroquine use decreases throughout sub–Saharan Africa because it is being replaced by now widely available artemisinin-based combination therapy, there is increasing evidence that chloroquine-susceptible malaria may predominate throughout the region in the near future [25]. If the reintroduction of chloroquine use occurs, it will have to be used in combination with another drug to prevent the reemergence of resistance. The selection of the appropriate partner drug to chloroquine will be based primarily on the ability of the partner drug to act synergistically to treat the malaria infection and prevent the reemergence of resistance, but the secondary benefits should also be taken into account. If this combination not only treats the acute malaria infection but also prevents future bacterial infections, the public health benefit of treating multiple leading causes of childhood deaths simultaneously would be significant.
Notes
Acknowledgments. We appreciate the assistance of the clinical study team in Malawi (Patricia Mawindo, Nelson Chimbiya, Ernest Thomson, Matthews Mwanamanga, Rhoda Masonga, Godfrey Mvula, Redson Kuyenda, Albert Malenga, Joseph Kanyangalika, Esther Mwagomba, Cristobel Kamodzi, Lexa Mlenga, Josephine Chilongo, Meraby Fusani, Christopher Chiotcha, Charles Kadziwe, and Eva Huwa) as well as the other Blantyre Malaria Project administrative staff. We would like to thank Dr Peter Moons, the Independent Safety Monitor for the study, and the other members of the data safety and monitoring board. We are especially grateful to the children and their parents who participated in this trial and have supported our work at the Ndirande research clinic for over a decade.
Financial support. This work was supported by the National Institute of Allergy and Infectious Diseases (U01AI044824 to C. V. P. and K23AI059316 to M. K. L.). M. K. L. and C. V. P. were supported by the Doris Duke Charitable Foundation, and C. V. P. is supported by the Howard Hughes Medical Institute. Neither the funders nor Pfizer had a role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. These results have not been reported previously.
Potential conflicts of interest. M. K. L. received an investigator-initiated research grant from Pfizer Global Pharmaceuticals from 2006 to 2010. Azithromycin was donated by Pfizer, Inc. All other authors report no potential conflicts.
All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Dunne MW, Singh N, Shukla M, et al. A multicenter study of azithromycin, alone and in combination with chloroquine, for the treatment of acute uncomplicated Plasmodium falciparum malaria in India. J Infect Dis. 2005;191:1582–8. doi: 10.1086/429343. [DOI] [PubMed] [Google Scholar]
- 2.Chico RM, Pittrof R, Greenwood B, Chandramohan D. Azithromycin-chloroquine and the intermittent preventive treatment of malaria in pregnancy. Malar J. 2008;7:255. doi: 10.1186/1475-2875-7-255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Coles CL, Seidman JC, Levens J, Mkocha H, Munoz B, West S. Association of mass treatment with azithromycin in trachoma-endemic communities with short-term reduced risk of diarrhea in young children. Am J Trop Med Hyg. 2011;85:691–6. doi: 10.4269/ajtmh.2011.11-0046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Coles CL, Levens J, Seidman JC, Mkocha H, Munoz B, West S. Mass distribution of azithromycin for trachoma control is associated with short-term reduction in risk of acute lower respiratory infection in young children. Pediatr Infect Dis J. 2012;31:341–6. doi: 10.1097/INF.0b013e31824155c9. [DOI] [PubMed] [Google Scholar]
- 5.Coles CL, Mabula K, Seidman JC, et al. Mass distribution of azithromycin for trachoma control is associated with increased risk of azithromycin-resistant Streptococcus pneumoniae carriage in young children 6 months after treatment. Clin Infect Dis. 2013;56:1519–26. doi: 10.1093/cid/cit137. [DOI] [PubMed] [Google Scholar]
- 6.Fry AM, Jha HC, Lietman TM, et al. Adverse and beneficial secondary effects of mass treatment with azithromycin to eliminate blindness due to trachoma in Nepal. Clin Infect Dis. 2002;35:395–402. doi: 10.1086/341414. [DOI] [PubMed] [Google Scholar]
- 7.Keenan JD, Ayele B, Gebre T, et al. Childhood mortality in a cohort treated with mass azithromycin for trachoma. Clin Infect Dis. 2011;52:883–8. doi: 10.1093/cid/cir069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Porco TC, Gebre T, Ayele B, et al. Effect of mass distribution of azithromycin for trachoma control on overall mortality in Ethiopian children: a randomized trial. JAMA. 2009;302:962–8. doi: 10.1001/jama.2009.1266. [DOI] [PubMed] [Google Scholar]
- 9.Hu VH, Harding-Esch EM, Burton MJ, Bailey RL, Kadimpeul J, Mabey DC. Epidemiology and control of trachoma: systematic review. Trop Med Int Health. 2010;15:673–91. doi: 10.1111/j.1365-3156.2010.02521.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kublin JG, Cortese JF, Njunju EM, et al. Reemergence of chloroquine-sensitive Plasmodium falciparum malaria after cessation of chloroquine use in Malawi. J Infect Dis. 2003;187:1870–5. doi: 10.1086/375419. [DOI] [PubMed] [Google Scholar]
- 11.Laufer MK, Thesing PC, Eddington ND, et al. Return of chloroquine antimalarial efficacy in Malawi. N Engl J Med. 2006;355:1959–66. doi: 10.1056/NEJMoa062032. [DOI] [PubMed] [Google Scholar]
- 12.Laufer MK, Thesing PC, Dzinjalamala FK, et al. A longitudinal trial comparing chloroquine as monotherapy or in combination with artesunate, azithromycin or atovaquone-proguanil to treat malaria. PLOS One. 2012;7:e42284. doi: 10.1371/journal.pone.0042284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rudan I, Boschi-Pinto C, Biloglav Z, Mulholland K, Campbell H. Epidemiology and etiology of childhood pneumonia. Bull World Health Organ. 2008;86:408–16. doi: 10.2471/BLT.07.048769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fischer Walker CL, Perin J, Aryee MJ, Boschi-Pinto C, Black RE. Diarrhea incidence in low- and middle-income countries in 1990 and 2010: a systematic review. BMC Public Health. 2012;12:220. doi: 10.1186/1471-2458-12-220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kalilani-Phiri L, Thesing PC, Nyirenda OM, et al. Timing of malaria infection during pregnancy has characteristic maternal, infant and placental outcomes. PLOS One. 2013;8:e74643. doi: 10.1371/journal.pone.0074643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Skalet AH, Cevallos V, Ayele B, et al. Antibiotic selection pressure and macrolide resistance in nasopharyngeal Streptococcus pneumoniae: a cluster-randomized clinical trial. PLOS Med. 2010;7:e1000377. doi: 10.1371/journal.pmed.1000377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Matute AJ, Schurink CA, Krijnen RM, Florijn A, Rozenberg-Arska M, Hoepelman IM. Double-blind, placebo-controlled study comparing the effect of azithromycin with clarithromycin on oropharyngeal and bowel microflora in volunteers. Eur J Clin Microbiol Infect Dis. 2002;21:427–31. doi: 10.1007/s10096-002-0728-y. [DOI] [PubMed] [Google Scholar]
- 18.Southern KW, Barker PM, Solis-Moya A, Patel L. Macrolide antibiotics for cystic fibrosis. Cochrane Database Syst Rev. 2012;11 doi: 10.1002/14651858.CD002203.pub4. CD002203. doi:10.1002/14651858.CD002203.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wong C, Jayaram L, Karalus N, et al. Azithromycin for prevention of exacerbations in non-cystic fibrosis bronchiectasis (EMBRACE): a randomised, double-blind, placebo-controlled trial. Lancet. 2012;380:660–7. doi: 10.1016/S0140-6736(12)60953-2. [DOI] [PubMed] [Google Scholar]
- 20.Chandra RS, Orazem J, Ubben D, Duparc S, Robbins J, Vandenbroucke P. Creative solutions to extraordinary challenges in clinical trials: methodology of a phase III trial of azithromycin and chloroquine fixed-dose combination in pregnant women in Africa. Malar J. 2013;12:122. doi: 10.1186/1475-2875-12-122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mullick S, Watson-Jones D, Beksinska M, Mabey D. Sexually transmitted infections in pregnancy: prevalence, impact on pregnancy outcomes, and approach to treatment in developing countries. Sex Transm Infect. 2005;81:294–302. doi: 10.1136/sti.2002.004077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Noedl H, Se Y, Schaecher K, Smith BL, Socheat D, Fukuda MM. Evidence of artemisinin-resistant malaria in western Cambodia. N Engl J Med. 2008;359:2619–20. doi: 10.1056/NEJMc0805011. [DOI] [PubMed] [Google Scholar]
- 23.Dondorp AM, Nosten F, Yi P, et al. Artemisinin resistance in Plasmodium falciparum malaria. N Engl J Med. 2009;361:455–67. doi: 10.1056/NEJMoa0808859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.van Eijk AM, Terlouw DJ. Azithromycin for treating uncomplicated malaria. Cochrane Database Syst Rev. 2011 doi: 10.1002/14651858.CD006688.pub2. CD006688. doi:10.1002/14651858.CD006688.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Frosch AE, Venkatesan M, Laufer MK. Patterns of chloroquine use and resistance in sub–Saharan Africa: a systematic review of household survey and molecular data. Malar J. 2011;10:116. doi: 10.1186/1475-2875-10-116. [DOI] [PMC free article] [PubMed] [Google Scholar]