Abstract
Background
Obstructive sleep apnea is associated with hypertension, inflammation, and increased cardiovascular risk. Continuous positive airway pressure (CPAP) reduces blood pressure, but adherence is often suboptimal, and the benefit beyond management of conventional risk factors is uncertain. Since intermittent hypoxemia may underlie cardiovascular sequelae of sleep apnea, we evaluated the effects of nocturnal supplemental oxygen and CPAP on markers of cardiovascular risk.
Methods
We conducted a randomized, controlled trial in which patients with cardiovascular disease or multiple cardiovascular risk factors were recruited from cardiology practices. Patients were screened for obstructive sleep apnea with the use of the Berlin questionnaire, and home sleep testing was used to establish the diagnosis. Participants with an apnea–hypopnea index of 15 to 50 events per hour were randomly assigned to receive education on sleep hygiene and healthy lifestyle alone (the control group) or, in addition to education, either CPAP or nocturnal supplemental oxygen. Cardiovascular risk was assessed at baseline and after 12 weeks of the study treatment. The primary outcome was 24-hour mean arterial pressure.
Results
Of 318 patients who underwent randomization, 281 (88%) could be evaluated for ambulatory blood pressure at both baseline and follow-up. On average, the 24-hour mean arterial pressure at 12 weeks was lower in the group receiving CPAP than in the control group (−2.4 mm Hg; 95% confidence interval [CI], −4.7 to −0.1; P = 0.04) or the group receiving supplemental oxygen (−2.8 mm Hg; 95% CI, −5.1 to −0.5; P = 0.02). There was no significant difference in the 24-hour mean arterial pressure between the control group and the group receiving oxygen. A sensitivity analysis performed with the use of multiple imputation approaches to assess the effect of missing data did not change the results of the primary analysis.
Conclusions
In patients with cardiovascular disease or multiple cardiovascular risk factors, the treatment of obstructive sleep apnea with CPAP, but not nocturnal supplemental oxygen, resulted in a significant reduction in blood pressure. (Funded by the National Heart, Lung, and Blood Institute and others; HeartBEAT ClinicalTrials.gov number, NCT01086800.)
Obstructive sleep apnea is a highly prevalent, chronic illness in adults, affecting an estimated 9% of middle-aged women and 24% of middle-aged men, with 4% and 9%, respectively, having moderate-to-severe obstructive sleep apnea.1 Cohort studies have shown that obstructive sleep apnea is a risk factor for hypertension, coronary heart disease, stroke, and death.2–9 The mechanisms underlying these associations are thought to include sympathetic activation, oxidative stress, and inflammation.10 Although reports from uncontrolled, clinic-based studies have indicated that cardiovascular risk is reduced among patients with obstructive sleep apnea who are treated with continuous positive airway pressure (CPAP) as compared with those who decline treatment,8,9,11,12 adequately powered randomized trials have not yet been conducted to evaluate the effect of CPAP on myocardial infarction, stroke, or death. One randomized trial designed to evaluate cardiovascular risk reduction with CPAP was recently published, but it was limited by inadequate statistical power.13
A meta-analysis of small, unregistered, single-center clinical trials showed a reduction of approximately 2 mm Hg in mean arterial pressure after CPAP treatment,14 and two multicenter clinical trials involving patients with newly diagnosed obstructive sleep apnea showed modest improvements in blood pressure with CPAP.15,16 In one of these studies, involving patients with untreated hypertension, the 24-hour mean arterial pressure was reduced by 1.5 mm Hg (95% confidence interval [CI], 0.4 to 2.7) over a 12-week period of CPAP treatment.15 The other study, involving patients with hypertension who were not sleepy during the day, showed a reduction in blood pressure (measured at an office visit while the patient was seated) of approximately 2 mm Hg over a 12-month follow-up period16; fewer than half the patients were receiving antihypertensive therapy, and their mean blood pressure at baseline was approximately 141/85 mm Hg. Because the effect of CPAP on blood pressure in patients with obstructive sleep apnea and hypertension is much smaller than that of valsartan,17 it remains uncertain whether the specific treatment of obstructive sleep apnea provides an additional benefit beyond that achieved with adequate pharmacotherapy alone.
Moreover, the effectiveness of CPAP in reducing elevated blood pressure or preventing the development of hypertension appears to be correlated with hours of adherence,13,15,16,18,19 with effects in one study noted in only those participants who used CPAP for more than 5.6 hours per night.16 Since epidemiologic data have shown that most persons with obstructive sleep apnea do not report excessive sleepiness,20 and since adherence to CPAP therapy is often poor in patients who are not sleepy during the day,21 less intrusive alternatives to CPAP are needed for the reduction of cardiovascular risk. Although obstructive sleep apnea probably increases the risk of cardiovascular disease through a number of mechanisms, repetitive cycles of hypoxemia and reoxygenation are thought to play a central role by augmenting sympathetic nervous system activity, systemic inflammation, and oxidative stress.22–27 Consequently, it is believed that nocturnal supplemental oxygen may serve as a potential alternative to CPAP in the reduction of cardiovascular risk. Although studies of supplemental oxygen treatment in patients with obstructive sleep apnea have shown that it can eliminate or markedly reduce nocturnal hypoxemia, in most of these studies, the apnea–hypopnea index (AHI) was not reduced when hypopnea was identified on the basis of a reduction in airflow, without regard to oxygen desaturation, and daytime functioning was not improved.28,29 In one small study in which CPAP and supplemental oxygen were compared with a sham CPAP control, only CPAP was found to significantly reduce 24-hour blood pressure; although supplemental oxygen appeared to be more effective than the control, the difference was not significant, and it was less effective than CPAP.30
In the present study, we sought to determine the effectiveness of both CPAP and supplemental oxygen as compared with usual care for reducing markers of cardiovascular risk in patients recruited from cardiology practices, where the standard of care was likely to be high. The primary outcome measure was 24-hour mean arterial blood pressure. Patients were also assessed for systemic inflammation, reactive hyperemia, fasting glycemia, and dyslipidemia, and adherence to therapy was compared across the active treatment groups.
Methods
Study Design
The Heart Biomarker Evaluation in Apnea Treatment (HeartBEAT) study was a four-site, randomized, parallel-group clinical trial designed to assess the effects of CPAP and supplemental oxygen therapy as compared with usual care in reducing cardiovascular risk among high-risk patients with obstructive sleep apnea. To assess the value these treatments may offer beyond that of conventional risk-factor management, the study targeted patients whose care was managed by cardiologists, and education on habits promoting sleep hygiene (e.g., appropriate timing and duration of sleep) and a healthy lifestyle was provided to all participants. Cardiovascular risk factors were measured at baseline and after 12 weeks of treatment.
The research protocol was approved by the institutional review board of each participating institution. The study adhered to the protocol, which is available with the full text of this article at NEJM.org. All participants provided written informed consent. A data and safety monitoring board reviewed the protocol, monitored the progress of the study, and conducted a single interim analysis. The authors designed the study, oversaw data collection, performed the analyses, prepared the manuscript for publication, and made the decision to submit the manuscript for publication without input from or review by Philips Respironics, which donated equipment used in the study. All the authors vouch for the accuracy and completeness of the data.
Eligibility
Patients 45 to 75 years of age who were enrolled in cardiology practices at the four participating medical centers were screened for obstructive sleep apnea with the use of the Berlin questionnaire, which consists of three categories of questions related to the risk of sleep apnea: snoring and cessation of breathing, daytime sleepiness, and obesity or hypertension. Patients are considered to be at high risk if their scores are positive in two or more categories.31 Eligible patients had scores of 2 or 3 on the Berlin questionnaire and had established coronary heart disease or multiple cardiovascular risk factors. At screening, participants also completed the Epworth Sleepiness Scale, a self-reported measure of daytime sleepiness with scores ranging from 0 to 24 and higher scores indicating more sleepiness. Detailed inclusion and exclusion criteria are provided in the Supplementary Appendix, available at NEJM.org.
Sleep tests of eligible participants were conducted at their homes with the use of a portable sleep monitor (Embletta Gold, Embla Systems), which assessed airflow by means of a nasal cannula pressure transducer and an oronasal thermal sensor, thoracic and abdominal movement by means of inductance plethysmography, and oxygen saturation by means of pulse oximetry with a finger-tip sensor. Body position was also monitored, and three-lead electrocardiography was used to monitor the heart. The studies were scored by a single certified scorer in accordance with guidelines from the American Academy of Sleep Medicine, which specify a reduction in airflow accompanied by a reduction in oxygen saturation of 3% or more as indicative of hypopnea.32 Participants were eligible for randomization if their AHI was 15 or more events (occurrences of apnea or hypopnea) per hour. Participants with severe obstructive sleep apnea, defined as an AHI above 50 or an oxygen saturation of less than 85% for more than 10% of the recording, were excluded from further participation, as were those with a central apnea index above 5.
Group Assignments and Interventions
After baseline evaluation, a stratified permuted block design was used to randomly assign participants to one of three interventions: healthy lifestyle and sleep education (HLSE) alone (control), CPAP with HLSE, or supplemental oxygen with HLSE. All participants received standardized education in habits that promote improved sleep quality and reduce cardiovascular risk, including advice on diet and exercise. This information was based on guidelines from the American Heart Association. Participants in the CPAP group received a CPAP device with automatic adjustment (REMstar Auto CPAP, Philips Respironics), set at a pressure range of 4 to 20 cm of water for 7 days and then reset to the best fixed pressure for each patient, defined as the 90th percentile of the pressure distribution generated through automatic adjustment during those 7 days (see the Supplementary Appendix). For the group receiving supplemental oxygen, a stationary oxygen concentrator (EverFlo, Philips Respironics) was used to provide nightly treatment with oxygen at a rate of 2 liters per minute through a nasal cannula.
Outcomes
Outcomes were measured at baseline and 12 weeks after randomization. The primary outcome was 24-hour mean arterial pressure (measured with the 90207 Ambulatory Blood Pressure Monitor, Spacelabs Healthcare). Mean pressure was calculated at each reading as one third of systolic pressure plus two thirds of diastolic pressure. The 24-hour mean pressure was calculated as a weighted average of the mean pressure during wakefulness and sleep, with weights determined by the percentage of reported time spent in each state, as recorded in a sleep diary. Nocturnal nondipping blood pressure was defined as a mean nocturnal blood pressure higher than 90% of the mean daytime value. Venipuncture was performed in the morning after a 12-hour fast to measure serum levels of glucose, insulin, low-density lipoprotein cholesterol, high-density lipoprotein cholesterol, total cholesterol, triglycerides, N-terminal pro–brain (B-type) natriuretic peptide (BNP), and high-sensitivity C-reactive protein. Endothelial function was assessed on the basis of the reactive hyperemia index, as measured with the use of the EndoPAT device (Itamar Medical) after an overnight fast. Additional details are provided in the Supplementary Appendix.
Statistical Analysis
For outcomes measured on a continuous scale, the primary analysis at 12 weeks was based on an analysis-of-covariance model with adjustment for the baseline value and stratification variables (study site and the presence or absence of coronary artery disease). A Wald test was used to compare the adjusted mean value for each outcome at 12 weeks between any two of the three groups, and the adjusted mean difference (and 95% confidence interval) for each pairwise comparison was estimated. Residual analysis indicated that the normality assumption was violated for all biochemical outcomes. Therefore, values were log-transformed, and the adjusted geometric mean ratio between groups was estimated. Because of the persistence of outliers despite the use of log transformation, a robust regression model was used to analyze values for C-reactive protein and N-terminal pro-BNP.33 A logistic-regression model was used to model the log-odds rate of nondipping blood pressure at 12 weeks. A family-wise type I error rate of 0.15 was chosen a priori to permit the detection of reasonable effect sizes with a sample that could be feasibly recruited within the time frame allowed by the grant, with a Bonferroni-corrected significance level of 0.05 for each pairwise comparison. Multiple imputation approaches were used to assess the sensitivity of the results from the primary analysis to missing data (see the Supplementary Appendix).
Results
Study Population
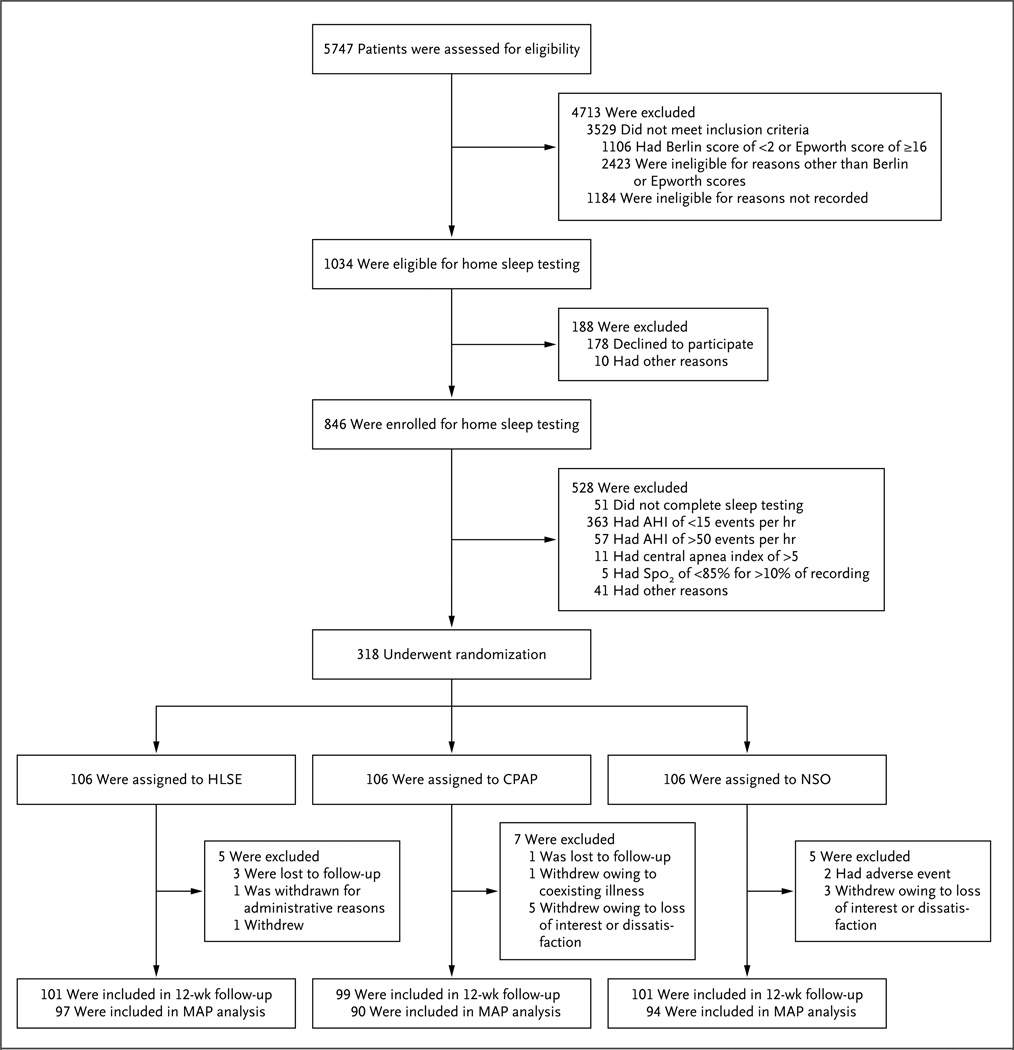
Recruitment began in February 2010 and ended in September 2011, with the final follow-up visit completed in January 2012. Given a higher-than-expected rate of study completion, 318 participants underwent randomization and 106 were assigned to each of the three study groups. A total of 301 participants (95%) completed the 12-week study (Fig. 1). All participants received the assigned intervention. Among the 301 participants who completed the study, 281 (93%) underwent 24-hour blood-pressure monitoring at both baseline and 12 weeks. The 37 participants excluded from the primary analysis because of missing data for the monitoring of 24-hour blood pressure were significantly heavier than the other participants, and their reported usual sleep duration was significantly shorter. The characteristics of these participants were otherwise similar to those of the other participants, and their exclusion did not create any imbalances across the three groups.
Figure 1. Numbers of Patients Who Were Screened, Randomly Assigned to a Study Group, and Included in Follow-up Analyses.
AHI denotes apnea–hypopnea index, CPAP continuous positive airway pressure, HLSE healthy lifestyle and sleep education, MAP mean arterial pressure, and NSO nocturnal supplemental oxygen. The Berlin questionnaire consists of three categories of questions related to the risk of sleep apnea: snoring and cessation of breathing, daytime sleepiness, and obesity or hypertension. Patients are considered to be at high risk if their scores are positive in two or more categories. The Epworth Sleepiness Scale is a measure of daytime sleepiness, with higher scores indicating more sleepiness.
The baseline characteristics of the participants who were evaluated at the 12-week follow-up visit were similar across groups, with significant differences only in daytime sleepiness and in the proportion of participants who were receiving alpha-adrenergic blockers (Table 1). Approximately half the participants had established coronary heart disease, and 88% had hypertension. Blood pressure was generally well controlled at baseline; the mean (±SD) number of antihypertensive medications that patients were receiving was 2.4±1.4, and the mean 24-hour arterial pressure was 124/71±15/8 mm Hg. The degree of obstructive sleep apnea among the study participants was moderate to severe by design, although excessive daytime sleepiness was uncommon. The group receiving CPAP and the group receiving supplemental oxygen had similar reductions in nocturnal hypoxemia, with each group having a reduction of at least 64% in the frequency of 3% desaturation events and a decline of at least 2.6% in the percentage of sleep time during which oxygen saturation was below 90% (P<0.001 for the comparison of each of these groups with the control group); there was no significant difference in the rates of either event between the CPAP group and the supplemental-oxygen group. The mean duration of use of supplemental oxygen was 4.8±2.4 hours per night, which was significantly greater than the duration of CPAP use, which was 3.5±2.7 hours per night (P = 0.001).
Table 1.
Baseline Characteristics of the Study Population.*
| Characteristic | HLSE (N = 97) |
CPAP (N = 90) |
NSO (N = 94) |
|---|---|---|---|
| Age — yr | 63.1±7.7 | 63.5±7.0 | 62.9±7.3 |
| Male sex — no. (%) | 76 (78) | 68 (76) | 65 (69) |
| Race — no./total no. (%)† | |||
| White | 79/95 (83) | 75/89 (84) | 69/93 (74) |
| Black | 10/95 (11) | 6/89 (7) | 19/93 (20) |
| Other or unknown | 6/95 (6) | 8/89 (9) | 5/93 (5) |
| Hispanic ethnic group — no./total no. (%)† | 3/97 (3) | 2/89 (2) | 0/93 (0) |
| Body-mass index‡ | 33.7±5.8 | 33.0±5.0 | 34.7±5.7 |
| Coexisting condition — no./total no. (%) | |||
| Coronary artery disease | 54/97 (56) | 46/90 (51) | 51/94 (54) |
| Diabetes mellitus | 44/96 (46) | 42/90 (47) | 40/93 (43) |
| Hypertension | 89/96 (93) | 76/90 (84) | 82/93 (88) |
| Medication use — no. (%) | |||
| ACE inhibitor or ARB | 69 (71) | 61 (68) | 65 (69) |
| Beta-adrenergic blocker | 68 (70) | 53 (59) | 69 (73) |
| Alpha-adrenergic blocker | 2 (2) | 0 (0) | 6 (6) |
| Diuretic | 36 (37) | 33 (37) | 39 (41) |
| Calcium-channel blocker | 28 (29) | 28 (31) | 32 (34) |
| Lipid-lowering medication | 85 (88) | 82 (91) | 83 (88) |
| Medication for diabetes | 40 (41) | 34 (38) | 36 (38) |
| Smoking status — no. (%) | |||
| Current smoker | 14 (14) | 4 (4) | 10 (11) |
| Former smoker | 50 (52) | 52 (58) | 45 (48) |
| Apnea–hypopnea index — no. of events/hr | 25.5±8.8 | 25.4±8.7 | 24.0±8.1 |
| SpO2 <90% — % of recording time | |||
| Median | 6.0 | 3.8 | 5.7 |
| Interquartile range | 2.4–11.7 | 1.6–8.7 | 2.1–13.2 |
| Epworth Sleepiness Scale score§ | 8.8±3.6 | 8.0±3.8 | 9.6±3.6 |
| Reported usual sleep duration — hr | 7.2±1.4 | 7.3±1.7 | 7.3±1.5 |
Plus–minus values are means ±SD. There was a significant difference across groups for the use of alpha-adrenergic blockers and for the score on the Epworth Sleepiness Scale (P = 0.02); there were no significant differences for any of the other baseline characteristics (P>0.05). ACE denotes angiotensin-converting enzyme, ARB angiotensin-receptor blocker, CPAP continuous positive airway pressure, HLSE healthy lifestyle and sleep education, NSO nocturnal supplemental oxygen, and SpO2 oxygen saturation level as measured by pulse oximetry.
Race and ethnic group were self-reported.
The body-mass index is the weight in kilograms divided by the square of the height in meters.
Scores on the Epworth Sleepiness Scale range from 0 to 24, with higher scores indicating more daytime sleepiness.
Outcomes
The adjusted 24-hour mean arterial pressure at 12 weeks was significantly lower in the group receiving CPAP than in either the control group or the group receiving supplemental oxygen (Table 2). No significant difference in this measure was detected between the group receiving supplemental oxygen and the control group. The results were almost identical when multiple imputation analyses were used to account for missing data (CPAP vs. education alone, −2.4 mm Hg; P = 0.04; CPAP vs. supplemental oxygen, −2.8 mm Hg; P = 0.02; supplemental oxygen vs. education alone, 0.3 mm Hg; P = 0.77). The magnitude of the effect of CPAP as compared with that of education alone or supplemental oxygen was somewhat larger for nighttime blood pressure than for daytime blood pressure (Table 3). Although there was no significant treatment effect on the odds of nondipping nocturnal blood pressure, the odds in the group receiving CPAP were 40% lower than the odds in the control group (odds ratio, 0.60; 95% confidence interval [CI], 0.32 to 1.11; P = 0.10), and the odds in the group receiving supplemental oxygen were 15% lower than the odds in the control group (odds ratio, 0.85; 95% CI, 0.46 to 1.56; P = 0.60). A secondary analysis with further adjustment for age, sex, race, baseline body-mass index, AHI, and use or nonuse of each class of antihypertensive medication had no appreciable influence on the effect estimates.
Table 2.
Effect of Treatment on 24-Hour Blood Pressure.*
| Variable | CPAP (N = 90) |
NSO (N = 94) |
HLSE (N = 97) |
CPAP vs. HLSE | NSO vs. HLSE | CPAP vs. NSO |
|---|---|---|---|---|---|---|
| 24-Hr mean arterial blood pressure | ||||||
| Baseline | 89.5±8.6 | 88.6±10.0 | 87.7±9.3 | |||
| 12 Wk | 87.8±8.1 | 90.2±11.1 | 89.0±11.2 | −2.4 (P = 0.04) | 0.4 (P = 0.71) | −2.8 (P = 0.02) |
| 24-Hr mean systolic blood pressure | ||||||
| Baseline | 124.7±13.5 | 125.3±16.9 | 123.6±14.3 | |||
| 12 Wk | 123.4±12.8 | 126.9±16.5 | 124.7±16.4 | −1.9 (P = 0.25) | 1.2 (P = 0.45) | −3.1 (P = 0.06) |
| 24-Hr mean diastolic blood pressure | ||||||
| Baseline | 72.0±7.7 | 70.8±8.3 | 69.6±8.6 | |||
| 12 Wk | 69.8±7.5 | 71.7±9.8 | 70.9±10.1 | −2.8 (P = 0.005) | −0.1 (P = 0.95) | −2.8 (P = 0.006) |
Plus–minus values are means ±SD. The between-group differences are the mean differences at 12 weeks, adjusted for study site, presence or absence of coronary artery disease, and blood pressure as measured at baseline. CPAP denotes continuous positive airway pressure, NSO nocturnal supplemental oxygen, and HLSE healthy lifestyle and sleep education.
Table 3.
Effect of Treatment on Nighttime and Daytime Blood Pressure.*
| Variable | CPAP (N = 93) |
NSO (N = 95) |
HLSE (N = 97)† |
CPAP vs. HLSE | NSO vs. HLSE | CPAP vs. NSO |
|---|---|---|---|---|---|---|
| Nighttime mean arterial blood pressure | ||||||
| Baseline | 83.0±9.9 | 82.1±10.4 | 80.7±9.8 | |||
| 12 Wk | 80.9±9.3 | 83.9±12.1 | 82.9±12.9 | −3.5 (P = 0.005) | −0.0 (P = 0.97) | −3.5 (P = 0.005) |
| Nighttime mean systolic blood pressure | ||||||
| Baseline | 116.6±15.7 | 117.0±17.8 | 115.5±15.7 | |||
| 12 Wk | 114.7±14.9 | 119.3±17.8 | 117.4±19.3 | −3.3 (P = 0.11) | 0.9 (P = 0.60) | −4.2 (P = 0.02) |
| Nighttime mean diastolic blood pressure | ||||||
| Baseline | 66.2±8.4 | 64.7±8.2 | 63.4±8.5 | |||
| 12 Wk | 64.1±8.1 | 66.1±10.5 | 65.6±10.9 | −3.7 (P = 0.001) | −0.5 (P = 0.61) | −3.2 (P = 0.003) |
| Daytime mean arterial blood pressure | ||||||
| Baseline | 92.4±8.9 | 91.8±10.6 | 90.7±9.9 | |||
| 12 Wk | 90.5±8.5 | 92.8±11.3 | 91.6±11.4 | −2.1 (P = 0.09) | 0.5 (P = 0.66) | −2.6 (P = 0.04) |
| Daytime mean systolic blood pressure | ||||||
| Baseline | 128.0±13.6 | 128.7±17.1 | 127.2±14.6 | |||
| 12 Wk | 126.8±12.9 | 130.2±17.0 | 128.0±16.0 | −1.7 (P = 0.33) | 1.2 (P = 0.50) | −2.9 (P = 0.10) |
| Daytime mean diastolic blood pressure | ||||||
| Baseline | 74.6±8.2 | 72.4±9.1 | 72.4±9.2 | |||
| 12 Wk | 72.4±7.9 | 74.2±9.9 | 73.4±10.6 | −2.5 (P = 0.02) | 0.1 (P = 0.91) | −2.6 (P = 0.02) |
Plus–minus values are means ±SD. The between-group differences are the mean differences at 12 weeks, adjusted for study site, presence or absence of coronary artery disease, and blood pressure as measured at baseline. CPAP denotes continuous positive airway pressure, NSO nocturnal supplemental oxygen, and HLSE healthy lifestyle and sleep education.
For the measurement of daytime blood pressure, the number of participants in the group receiving HLSE was 99.
Analysis of the reactive hyperemia index at 12 weeks revealed no evidence of significant differences among the three groups. CPAP was associated with a significantly lower adjusted level of C-reactive protein at 12 weeks than was education alone (geometric mean ratio, 0.80; 95% CI, 0.65 to 0.97; P = 0.03); no significant difference in levels of other laboratory values was detected (Tables S1 and S2 in the Supplementary Appendix).
There was no significant association between hours of CPAP use and 24-hour mean arterial pressure, although nocturnal systolic blood pressure fell significantly for each additional hour of CPAP use per night (−0.93 mm Hg per hour; 95% CI, −1.77 to −0.10; P = 0.03). In addition, the odds of nondipping nocturnal blood pressure fell sharply with each additional hour of CPAP use (odds ratio, 0.76 per hour; 95% CI, 0.63 to 0.92; P = 0.006). No significant differences among groups were observed in the rate of nonserious or serious adverse events (see the Supplementary Appendix).
Discussion
The present study shows that among patients with previously undiagnosed moderate-to-severe obstructive sleep apnea, treatment with CPAP resulted in reduced 24-hour mean arterial pressure, even in a clinical setting in which cardiovascular risk factors, including blood pressure, were well managed and despite the exclusion of patients with the most severe obstructive sleep apnea, who might be expected to derive the greatest benefit from treatment. Although the average decrease in blood pressure resulting from CPAP was modest as compared with the effect of antihypertensive medications, the treatment effect was observed in patients who were already receiving these medications. The reduction in blood pressure was similar to that in prior studies of the treatment of obstructive sleep apnea14–17 and was of a magnitude associated with a meaningful reduction in cardiovascular risk. A meta-analysis of 61 observational studies that evaluated the relation of blood pressure to mortality from vascular disease showed that a reduction of 2 mm Hg in systolic blood pressure would reduce mortality from stroke by 10% and mortality from ischemic heart disease or other vascular causes by 7%, with reductions in diastolic blood pressure having effects that were approximately twice as large.34 A meta-analysis of 147 randomized trials showed effects of similar magnitude for stroke and events related to coronary heart disease, even in patients with blood pressure as low at 110/70 mm Hg before treatment.35 The effect of CPAP appears to be greatest at night, perhaps reflecting the prevention of postapneic increases in blood pressure and indicating that CPAP may thereby lower the cardiovascular risk associated with the absence of a normal nocturnal reduction in blood pressure.36
Since patients were recruited from general cardiology practices rather than from referral centers for sleep disorders, most participants had only mild symptoms of obstructive sleep apnea, and adherence to treatment with CPAP was somewhat lower (i.e. fewer hours of nightly use) than that reported in several European studies.13,15–18 The beneficial effect of CPAP, despite modest average adherence levels, may be relevant to public policy, given the common use of adherence data to restrict reimbursement for CPAP to patients whose adherence exceeds a specified threshold. However, the study was not powered to determine a critical adherence threshold for the beneficial effect of CPAP, and further clarification of the dose–response relationship is needed.
Although observational studies have generally shown that obstructive sleep apnea is associated with inflammation and with impaired fasting glycemia or overt diabetes mellitus, intervention studies have yielded inconsistent results.37,38 In the present study, the CPAP group had a 20% reduction in the mean level of C-reactive protein at 12 weeks, as compared with the control group, although this finding should be interpreted with caution, since no significant treatment effects were observed for other biochemical measures. It is possible that the recruitment of patients from cardiology practices, which provided effective risk-factor management at baseline, limited our ability to detect further improvement in biochemical measures.
In contrast to the beneficial effects of CPAP on blood pressure, the use of supplemental oxygen was not associated with a reduction in blood pressure, even though adherence was greater among patients receiving supplemental oxygen. This finding was unexpected, since intermittent hypoxemia induces sustained elevations in blood pressure in both rodents23 and humans.39 Moreover, in dog models of obstructive sleep apnea, obstructive apneas result in sustained hypertension, whereas arousals of a similar frequency caused by sound do not,40 and supplemental oxygen markedly attenuates the immediate hypertensive response to obstructive apneas.24 Since CPAP and supplemental oxygen had similar effects on nocturnal hypoxemia, the lack of a beneficial effect of supplemental oxygen could indicate that other factors (e.g., arousal, hypercapnia, and large swings in intrathoracic pressure) play a role in hypertension resulting from obstructive sleep apnea; however, in the absence of more detailed physiological measures, our ability to draw mechanistic inferences from these data is limited.
In conclusion, the treatment of obstructive sleep apnea with CPAP, but not with nocturnal supplemental oxygen, results in a significant reduction in blood pressure, even in patients with well-controlled hypertension at baseline. Although there may be other clinical reasons that a treating physician should consider using supplemental oxygen in specific patients, this study offers no support for the common but largely untested clinical practice of providing supplemental oxygen as salvage therapy in patients with obstructive sleep apnea for whom CPAP is problematic.
Supplementary Material
Acknowledgments
Supported by grants from the National Heart, Lung, and Blood Institute (RC2 HL101417, 1R01HL109493, and R21HL108226) and by a grant from the National Center for Research Resources (UL1 RR024989).
We thank the members of the data safety and monitoring board (Wayne Cascio, M.D., chair; Vernon M. Chinchilli, Ph.D.; William Cotts, M.D.; Patrick Strollo, M.D.; and Terri Weaver, R.N., Ph.D.) for providing expert and timely guidance and oversight, and the study staff (Joan Aylor, Amanda Barbeau, Denise P. Clarke, Daniel Cooper, Kelly Devine, Anna Maria Kibler, Melissa Minotti, Rawan Nawabit, Gaynor A. Stephens, Ashley Wagner, Cynthia Williams, and Christina L. Zenobi) for their invaluable assistance.
Footnotes
Disclosure forms provided by the authors are available with the full text of this article at NEJM.org.
References
- 1.Young T, Palta M, Dempsey J, Skatrud J, Weber S, Badr S. The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med. 1993;328:1230–1235. doi: 10.1056/NEJM199304293281704. [DOI] [PubMed] [Google Scholar]
- 2.Nieto FJ, Young TB, Lind BK, et al. Association of sleep-disordered breathing, sleep apnea, and hypertension in a large community-based study: Sleep Heart Health Study. JAMA. 2000;283:1829–1836. doi: 10.1001/jama.283.14.1829. [Erratum, JAMA 2002;288:1985.] [DOI] [PubMed] [Google Scholar]
- 3.Peppard PE, Young T, Palta M, Skatrud J. Prospective study of the association between sleep-disordered breathing and hypertension. N Engl J Med. 2000;342:1378–1384. doi: 10.1056/NEJM200005113421901. [DOI] [PubMed] [Google Scholar]
- 4.Gottlieb DJ, Yenokyan G, Newman AB, et al. Prospective study of obstructive sleep apnea and incident coronary heart disease and heart failure: the Sleep Heart Health Study. Circulation. 2010;122:352–360. doi: 10.1161/CIRCULATIONAHA.109.901801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yaggi HK, Concato J, Kernan WN, Lichtman JH, Brass LM, Mohsenin V. Obstructive sleep apnea as a risk factor for stroke and death. N Engl J Med. 2005;353:2034–2041. doi: 10.1056/NEJMoa043104. [DOI] [PubMed] [Google Scholar]
- 6.Redline S, Yenokyan G, Gottlieb DJ, et al. Obstructive sleep apnea-hypopnea and incident stroke: the Sleep Heart Health Study. Am J Respir Crit Care Med. 2010;182:269–277. doi: 10.1164/rccm.200911-1746OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mooe T, Franklin KA, Holmström K, Rabben T, Wiklund U. Sleep-disordered breathing and coronary artery disease: long-term prognosis. Am J Respir Crit Care Med. 2001;164:1910–1913. doi: 10.1164/ajrccm.164.10.2101072. [DOI] [PubMed] [Google Scholar]
- 8.Peker Y, Hedner J, Norum J, Kraiczi H, Carlson J. Increased incidence of cardiovascular disease in middle-aged men with obstructive sleep apnea: a 7-year follow-up. Am J Respir Crit Care Med. 2002;166:159–165. doi: 10.1164/rccm.2105124. [DOI] [PubMed] [Google Scholar]
- 9.Marin JM, Carrizo SJ, Vicente E, Agusti AG. Long-term cardiovascular outcomes in men with obstructive sleep apnoea-hypopnoea with or without treatment with continuous positive airway pressure: an observational study. Lancet. 2005;365:1046–1053. doi: 10.1016/S0140-6736(05)71141-7. [DOI] [PubMed] [Google Scholar]
- 10.Somers VK, White DP, Amin R, et al. Sleep apnea and cardiovascular disease: an American Heart Association/American College of Cardiology Foundation Scientific Statement from the American Heart Association Council for High Blood Pressure Research Professional Education Committee, Council on Clinical Cardiology, Stroke Council, and Council on Cardiovascular Nursing: in collaboration with the National Heart, Lung, and Blood Institute National Center on Sleep Disorders Research (National Institutes of Health) Circulation. 2008;118:1080–1111. doi: 10.1161/CIRCULATIONAHA.107.189375. [Erratum, Circulation 2009;119(12):e380.] [DOI] [PubMed] [Google Scholar]
- 11.Milleron O, Pillière R, Foucher A, et al. Benefits of obstructive sleep apnoea treatment in coronary artery disease: a long-term follow-up study. Eur Heart J. 2004;25:728–734. doi: 10.1016/j.ehj.2004.02.008. [DOI] [PubMed] [Google Scholar]
- 12.Doherty LS, Kiely JL, Swan V, McNicholas WT. Long-term effects of nasal continuous positive airway pressure therapy on cardiovascular outcomes in sleep apnea syndrome. Chest. 2005;127:2076–2084. doi: 10.1378/chest.127.6.2076. [DOI] [PubMed] [Google Scholar]
- 13.Barbé F, Durán-Cantolla J, Sánchez-de-la-Torre M, et al. Effect of continuous positive airway pressure on the incidence of hypertension and cardiovascular events in nonsleepy patients with obstructive sleep apnea: a randomized controlled trial. JAMA. 2012;307:2161–2168. doi: 10.1001/jama.2012.4366. [DOI] [PubMed] [Google Scholar]
- 14.Bazzano LA, Khan Z, Reynolds K, He J. Effect of nocturnal nasal continuous positive airway pressure on blood pressure in obstructive sleep apnea. Hypertension. 2007;50:417–423. doi: 10.1161/HYPERTENSIONAHA.106.085175. [DOI] [PubMed] [Google Scholar]
- 15.Durán-Cantolla J, Aizpuru F, Montserrat JM, et al. Continuous positive airway pressure as treatment for systemic hypertension in people with obstructive sleep apnoea: randomised controlled trial. BMJ. 2010;341:c5991. doi: 10.1136/bmj.c5991. [DOI] [PubMed] [Google Scholar]
- 16.Barbé F, Durán-Cantolla J, Capote F, et al. Long-term effect of continuous positive airway pressure in hypertensive patients with sleep apnea. Am J Respir Crit Care Med. 2010;181:718–726. doi: 10.1164/rccm.200901-0050OC. [DOI] [PubMed] [Google Scholar]
- 17.Pépin JL, Tamisier R, Barone-Rochette G, Launois SH, Lévy P, Baguet JP. Comparison of continuous positive airway pressure and valsartan in hypertensive patients with sleep apnea. Am J Respir Crit Care Med. 2010;182:954–960. doi: 10.1164/rccm.200912-1803OC. [DOI] [PubMed] [Google Scholar]
- 18.Lozano L, Tovar JL, Sampol G, et al. Continuous positive airway pressure treatment in sleep apnea patients with resistant hypertension: a randomized, controlled trial. J Hypertens. 2010;28:2161–2168. doi: 10.1097/HJH.0b013e32833b9c63. [DOI] [PubMed] [Google Scholar]
- 19.Marin JM, Agusti A, Villar I, et al. Association between treated and untreated obstructive sleep apnea and risk of hypertension. JAMA. 2012;307:2169–2176. doi: 10.1001/jama.2012.3418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kapur VK, Baldwin CM, Resnick HE, Gottlieb DJ, Nieto FJ. Sleepiness in patients with moderate to severe sleep-disordered breathing. Sleep. 2005;28:472–477. doi: 10.1093/sleep/28.4.472. [DOI] [PubMed] [Google Scholar]
- 21.McArdle N, Devereux G, Heidarnejad H, Engleman HM, Mackay TW, Douglas NJ. Long-term use of CPAP therapy for sleep apnea/hypopnea syndrome. Am J Respir Crit Care Med. 1999;159:1108–1114. doi: 10.1164/ajrccm.159.4.9807111. [DOI] [PubMed] [Google Scholar]
- 22.Somers VK, Mark AL, Zavala DC, Abboud FM. Contrasting effects of hypoxia and hypercapnia on ventilation and sympathetic activity in humans. J Appl Physiol. 1989;67:2101–2106. doi: 10.1152/jappl.1989.67.5.2101. (1985) [DOI] [PubMed] [Google Scholar]
- 23.Fletcher EC, Lesske J, Culman J, Miller CC, Unger T. Sympathetic denervation blocks blood pressure elevation in episodic hypoxia. Hypertension. 1992;20:612–619. doi: 10.1161/01.hyp.20.5.612. [DOI] [PubMed] [Google Scholar]
- 24.Schneider H, Schaub CD, Chen CA, et al. Neural and local effects of hypoxia on cardiovascular responses to obstructive apnea. J Appl Physiol. 2000;88:1093–1102. doi: 10.1152/jappl.2000.88.3.1093. (1985) [DOI] [PubMed] [Google Scholar]
- 25.Ryan S, McNicholas WT. Intermittent hypoxia and activation of inflammatory molecular pathways in OSAS. Arch Physiol Biochem. 2008;114:261–266. doi: 10.1080/13813450802307337. [DOI] [PubMed] [Google Scholar]
- 26.Greenberg H, Ye X, Wilson D, Htoo AK, Hendersen T, Liu SF. Chronic intermittent hypoxia activates nuclear factor-kappaB in cardiovascular tissues in vivo. Biochem Biophys Res Commun. 2006;343:591–596. doi: 10.1016/j.bbrc.2006.03.015. [DOI] [PubMed] [Google Scholar]
- 27.Jelic S, Le Jemtel TH. Inflammation, oxidative stress, and the vascular endothelium in obstructive sleep apnea. Trends Cardiovasc Med. 2008;18:253–260. doi: 10.1016/j.tcm.2008.11.008. [DOI] [PubMed] [Google Scholar]
- 28.Phillips BA, Schmitt FA, Berry DTR, Lamb DG, Amin M, Cook YR. Treatment of obstructive sleep apnea: a preliminary report comparing nasal CPAP to nasal oxygen in patients with mild OSA. Chest. 1990;98:325–330. doi: 10.1378/chest.98.2.325. [DOI] [PubMed] [Google Scholar]
- 29.Loredo JS, Ancoli-Israel S, Kim E-J, Lim WJ, Dimsdale JE. Effect of continuous positive airway pressure versus supplemental oxygen on sleep quality in obstructive sleep apnea: a placebo-CPAP-controlled study. Sleep. 2006;29:564–571. doi: 10.1093/sleep/29.4.564. [DOI] [PubMed] [Google Scholar]
- 30.Norman D, Loredo JS, Nelesen RA, et al. Effects of continuous positive airway pressure versus supplemental oxygen on 24-hour ambulatory blood pressure. Hypertension. 2006;47:840–845. doi: 10.1161/01.HYP.0000217128.41284.78. [DOI] [PubMed] [Google Scholar]
- 31.Netzer NC, Stoohs RA, Netzer CM, Clark K, Strohl KP. Using the Berlin Questionnaire to identify patients at risk for the sleep apnea syndrome. Ann Intern Med. 1999;131:485–491. doi: 10.7326/0003-4819-131-7-199910050-00002. [DOI] [PubMed] [Google Scholar]
- 32.Iber C, Ancoli-Israel S, Chesson A, Quan SF. The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications. Westchester, IL: American Academy of Sleep Medicine; 2007. [Google Scholar]
- 33.Maronna RA, Yohai VJ. Robust regression with both continuous and categorical predictors. J Stat Plan Infer. 2000;89:197–214. [Google Scholar]
- 34.Lewington S, Clarke R, Qizilbash N, Peto R, Collins R. Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Lancet. 2002;360:1903–1913. doi: 10.1016/s0140-6736(02)11911-8. [Erratum, Lancet 2003;361:1060.] [DOI] [PubMed] [Google Scholar]
- 35.Law MR, Morris JK, Wald NJ. Use of blood pressure lowering drugs in the prevention of cardiovascular disease: meta-analysis of 147 randomised trials in the context of expectations from prospective epidemiological studies. BMJ. 2009;338:b1665. doi: 10.1136/bmj.b1665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Friedman O, Logan AG. Can nocturnal hypertension predict cardiovascular risk? Integr Blood Press Control. 2009;2:25–37. doi: 10.2147/ibpc.s4364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Punjabi NM. Do sleep disorders and associated treatments impact glucose metabolism? Drugs. 2009;69(Suppl 2):13–27. doi: 10.2165/11531150-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 38.Kohler M, Ayers L, Pepperell JC, et al. Effects of continuous positive airway pressure on systemic inflammation in patients with moderate to severe obstructive sleep apnoea: a randomised controlled trial. Thorax. 2009;64:67–73. doi: 10.1136/thx.2008.097931. [DOI] [PubMed] [Google Scholar]
- 39.Tamisier R, Pépin JL, Rémy J, et al. 14 Nights of intermittent hypoxia elevate daytime blood pressure and sympathetic activity in healthy humans. Eur Respir J. 2011;37:119–128. doi: 10.1183/09031936.00204209. [DOI] [PubMed] [Google Scholar]
- 40.Brooks D, Horner RL, Kozar LF, Render-Teixeira CL, Phillipson EA. Obstructive sleep apnea as a cause of systemic hypertension: evidence from a canine model. J Clin Invest. 1997;99:106–109. doi: 10.1172/JCI119120. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.