Abstract
Background
Environmental surfaces play an important role in the transmission of healthcare-associated pathogens. Because environmental cleaning is often suboptimal, there is a growing demand for safe, rapid, and automated disinfection technologies, which has lead to a wealth of novel disinfection options available on the market. Specifically, automated ultraviolet-C (UV-C) devices have grown in number due to the documented efficacy of UV-C for reducing healthcare-acquired pathogens in hospital rooms. Here, we assessed and compared the impact of pathogen concentration, organic load, distance, and radiant dose on the killing efficacy of two analogous UV-C devices.
Principal Findings
The devices performed equivalently for each impact factor assessed. Irradiation delivered for 41 minutes at 4 feet from the devices consistently reduced C. difficile spores by ∼ 3 log10CFU/cm2, MRSA by>4 log10CFU/cm2, and VRE by >5 log10CFU/cm2. Pathogen concentration did not significantly impact the killing efficacy of the devices. However, both a light and heavy organic load had a significant negative impacted on the killing efficacy of the devices. Additionally, increasing the distance to 10 feet from the devices reduced the killing efficacy to ≤3 log10CFU/cm2 for MRSA and VRE and <2 log10CFU/cm2 for C.difficile spores. Delivery of reduced timed doses of irradiation particularly impacted the ability of the devices to kill C. difficile spores. MRSA and VRE were reduced by >3 log10CFU/cm2 after only 10 minutes of irradiation, while C. difficile spores required 40 minutes of irradiation to achieve a similar reduction.
Conclusions
The UV-C devices were equally effective for killing C. difficile spores, MRSA, and VRE. While neither device would be recommended as a stand-alone disinfection procedure, either device would be a useful adjunctive measure to routine cleaning in healthcare facilities.
Introduction
Environmental surfaces may play an important role in transmission of healthcare-associated pathogens such as Clostridium difficile, methicillin-resistant Staphylococcus aureus (MRSA), and vancomycin-resistant Enterococcus (VRE) [1]–[6]. Pathogens are shed onto environmental surfaces and will remain for several days, or possibly months, if the surfaces are not effectively disinfected [1]–[6]. Unfortunately, several recent studies have demonstrated that environmental cleaning is often suboptimal in healthcare facilities [5]–[8]. Interventions such as education of housekeeping staff or use of fluorescent markers to provide feedback to housekeepers may result in improved cleaning [5]–[8]. Yet, despite the promise of improvement in routine cleaning, there remains a demand for novel, automated technologies that are effective against hard to kill Clostridium difficile spores, but are also safe and rapid. As a consequence, there has been an upsurge in automated disinfection technologies on the market, many of which have yet to be rigorously evaluated.
Novel ultraviolet disinfection devices are currently on the fore-front of burgeoning automated technologies due to the well documented efficacy of ultraviolet-C (UV-C) irradiation for killing bacteria, viruses, and persistent spores [9]–[15]. The mechanism of killing of microorganisms by UV-C is primarily due to inactivation of DNA and RNA through absorption of photons resulting in formation of pyrimidine dimers from thymine and cytosine [9], [12], [15]. We previously demonstrated that an automated room disinfection device that utilizes low pressure mercury lamps for emitting UV-C radiation was effective for significantly reducing C. difficile, MRSA, and VRE contamination in hospital rooms (Tru-D Rapid Room Disinfection, Lumalier, Memphis, TN, USA) [16]. Similarly, Rutala et al. evaluated the Tru-D device and concluded that it was an efficacious and environmentally friendly method for disinfecting surfaces in healthcare facilities [17]. Here, we performed a side-by-side comparative evaluation of a homologous automated UV-C room disinfection device (Pathogon UV Disinfection System, Steris Corporation, Mentor, Ohio, USA) against the previously tested Tru-D device, in the laboratory setting. For each device, the impact of pathogen concentration, organic load, distance, and radiant dose on killing efficacy was assessed.
Materials and Methods
C. difficile, MRSA, and VRE Strains
Two clinical isolates of C. difficile, MRSA and VRE were studied. The MRSA strains were a pulsed-field gel electrophoresis (PFGE) type USA300 (community-associated) and USA800 (hospital-associated). The VRE strains were a VanA-type isolate (C37) and a VanB-type isolate (C68). The C. difficile strains were VA 17, a restriction endonuclease analysis (REA) type BI strain, and VA 11, an REA type J strain.
Preparation of C. difficile Spores
Spores were prepared by growth on brain-heart infusion agar (Becton Dickinson, Cockeysville, MD) supplemented with yeast extract (5 mg/ml) and L-cysteine (0.1%) at 37°C under anaerobic conditions as previously described [18]. Spores were stored at 4°C in sterile distilled water until use. Prior to testing, spore preps were confirmed by phase contrast microscopy and malachite green staining to be > 99% dormant, bright-phase spores.
Microbiology
For VRE, MRSA, and C. difficile cultures, media included Enterococcosel agar (Becton Dickinson, Cockeysville, MD) containing 20 µg/mL of vancomycin, CHROMagar (CHROMagar, Paris, France) containing 6 µg/mL of cefoxitin, and cycloserine-cefoxitin-brucella agar containing 0.1% taurocholic acid and lysozyme 5 mg/L (CDBA), respectively [19]. Plates containing MRSA or VRE were incubated aerobically at 37°C for 48 hours. C. difficile plates were incubated in a Whitley Workstation MG1000 anaerobic chamber (Microbiology International, Frederick, MD) at 37°C for 48 hours.
The UV-C Disinfection Devices
Figure 1A/B is a photograph of the devices. The Pathogon device (1A) is 28 inches wide, 31 inches long, and stands 67 inches tall. The system is a wheeled mobile unit that is controlled remotely by a Windows-based tablet controller. It is placed in the center of the room and commonly touched surfaces are arranged close to the device for optimal exposure to UV-C radiation (i.e. bedrails pulled up, call buttons placed on the bed, tables placed near the device). The device contains motion and heat sensor that are connected to a safety rated relay, aborting the UV-C cycle if someone enters the room during use. The unit has 24, 45 inch low pressure mercury bulbs. Once the operator has exited the room, a pre-programed germicidal dose is chosen based on the dimensions of the room. UV-C radiation penetrates all areas of the room that receive light, but the highest exposure occurs for areas that are in direct line of exposure to the output of the device; areas that are not in direct line of exposure to UV-C may receive radiation that is reflected from the walls and ceiling or from other surfaces in the room.
Figure 1. Photographs of the Pathogon (A) and Tru-D (B) devices.

The Tru-D device (1B) is 72 inches tall and measures 24 inches at the widest portion of the base. It is a wheeled mobile device that is placed strategically in the center of the room just as described above for the Pathogon device. The operator exits the room, closes the door, and places a door sensor on the frame of the door. Continuous monitoring during operation is not required because the sensor triggers automatic discontinuation of the cycle if the door is opened. A handheld remote is used to select either a vegetative cycle that is effective for killing of non-spore forming organisms or a spore cycle that is effective in killing spores. The unit has 28, 36 inch low pressure mercury bulbs. The device contains eight sensors spaced at equal distances on a ring at the top of the device. The sensors measure the amount of UV-C light reflected back to the device. The device automatically ends the cycle when the area reflecting the lowest level of UV-C back to the sensors (i.e. shaded areas in the room) has received an adequate dose.
Efficacy of the UV-C Devices for the Reduction of Pathogens on Carriers
The Effect of Pathogen Concentration and Organic Load
Initial experiments were conducted to determine whether pathogen concentration (i.e. concentration of organisms per cm2) or organic load influenced the disinfection efficacy of the UV-C devices. For pathogen concentration experiments, ten µl aliquots of two strains of C. difficile spores, MRSA, and VRE were suspended in sterile phosphate buffered saline (PBS), inoculated onto stainless steel carriers, and then spread to cover a 1 cm2 area. Organisms were allowed to desiccate onto the carriers under ambient room conditions. For C. difficile spores, the inoculum applied to the carriers ranged from 2 to >5 log10CFU/cm2. Previous experiments demonstrated that a reduction in vegetative organisms (MRSA and VRE) was observed after initial desiccation onto the carriers, however no further reduction was observed within the duration of treatment time (author's unpublished data). For each vegetative pathogen, the inoculum applied to the slide was adjusted such that 2 to >5 log10CFU/cm2 were recovered from the positive control specimens after desiccation. For organic load experiments, two strains of C. difficile spores, MRSA, and VRE were suspended in either sterile PBS, light organic load (5% fetal calf serum), or heavy organic load (5% fetal calf serum and 5% tryptone) and inoculated onto stainless steel carriers as described above. However, the inoculum was altered such that each carrier yielded 6 log10 CFU at baseline.
The carriers were placed on a laboratory bench top 4 feet from the UV-C device, within the direct field of radiation. Baseline slides were left untreated outside of the room (i.e., positive controls). The room dimensions were approximately 10×10 feet. Based on these dimensions, the UV-C devices were run for 41 minutes, as suggested by the manufacturer of the Pathogon device to deliver a spore-killing dose of UV-C within a 10×10 foot range.
To quantify viable organisms, the carriers were submersed in 10 mL of sterile PBS, vortexed vigorously, and dilutions of the suspensions were plated onto selective media as described in Microbiology. Following 48 hours of incubation, log10 colony forming unit (CFU) reductions were calculated by comparing the log10CFU recovered from carriers post UV-C disinfection to untreated controls. All experiments were repeated three times.
The Effect of Distance and Indirect Irradiation
The killing efficacies of the UV-C devices were evaluated at increasing distances and shaded from the direct field of radiation. Carriers were prepared and processed as described above in The Effect of Pathogen Concentration and Organic Load, however, the organisms were suspended in PBS and altered such that each carrier yielded 6 log10 CFU at baseline. Additionally, carriers were placed 6 inches, 4 feet and 10 feet within the direct field of radiation, and also 4 feet shaded from direct radiation. The UV-C devices were run for 41 minutes.
The Effect of Radiant Dose
The effect of radiant dose on the killing efficacies of the UV-C devices was determined for vegetative organism (MRSA and VRE) and spores (C. difficile). Carriers were prepared and processed as described above in The Effect of Pathogen Concentration and Organic Load, however, the organism were suspended in PBS and altered such that each carrier yielded 6 log10 CFU at baseline. Carriers were placed 4 feet from the device in the direct field of UV-C and irradiated for either 10, 20, or 40 minutes.
Statistical Analysis
Data were analyzed using STATA 9.0 software (StataCorp, College Station, TX). Continuous data were analyzed using paired t tests.
Results
Efficacy of the UV-C Devices for the Reduction of Pathogens on Carriers
The Effect of Pathogen Concentration and Organic Load
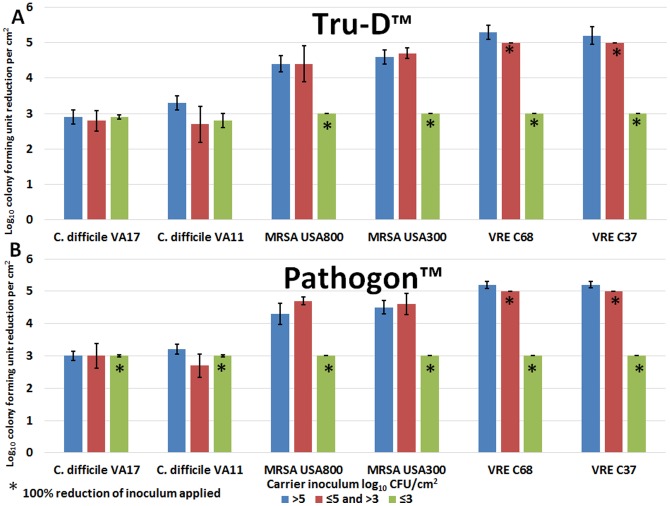
Figures 2A and 2B show the mean log10CFU/cm2 reductions of two strains of C. difficile, MRSA, and VRE on carriers after the use of the Tru-D and Pathogon devices, respectively. There was no significant differences between the log10CFU reductions of the two strains of each pathogen tested. Therefore, in subsequent experiments, the two strains were calculated collectively in the mean. The concentration of pathogens on a surface (≤3 to>5 log10CFU) did not have an impact on the killing efficacy of the UV-C devices. Furthermore, the UV-C devices were equally effective for reducing pathogens. There was no significant difference observed in the log10CFU reductions achieved by the Tru-D or Pathogon device for the two strains of each pathogen assessed (Tru-D vs. Pathogon: P = 0.57 (C.difficile), P = 1.0 (MRSA) and P = 0.97 (VRE)). Irradiation delivered by the Tru-D and Pathogon devices for 41 minutes (spore killing dose) consistently reduced C. difficile spores by ∼ 3 log10CFU/cm2, MRSA by >4 log10CFU/cm2, and VRE by >5 log10CFU/cm2.
Figure 2. The effect of pathogen concentration on the efficacy of the UV-C devices.
The log10CFU reduction/cm2 of two strains of C. difficile spores, MRSA, and VRE inoculated onto carriers. Carriers contained either >5, ≤5 and >3, or ≤3 log10CFU of each pathogen. The carriers were irradiated for 41 minutes at a distance of 4 feet from the Tru-D (2A) or Pathogon (2B) device. The means of the data from experiments conducted in triplicate are presented. Error bars indicate standard error.
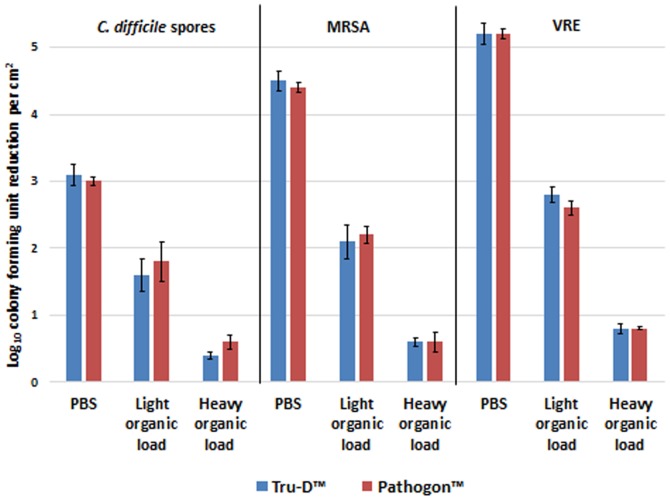
Figure 3 shows the effects of a light and heavy organic load on the killing efficacy of the Tru-D and Pathogon device. Both the light (5% fetal calf serum) and heavy organic load (5% fetal calf serum, 5% tryptone) had a significant deleterious impact on the efficacy of the devices. The light organic load decreased the log reductions achieved by the devices to <2 log10CFU/cm2 for C.difficile spores, <2.5 log10CFU/cm2 for MRSA, and <3 log10CFU/cm2 for VRE. The heavy organic load had a more dramatic effect, decreasing the log reduction to <1 log10CFU/cm2 for each pathogen assessed.
Figure 3. The effect of organic load on the efficacy of the UV-C devices.
The log10CFU reduction/cm2 of C. difficile spores, MRSA, and VRE suspended in phosphate-buffered saline (PBS), light organic load (5% fetal calf serum), or heavy organic load (5% fetal calf serum, 5% tryptone). Carriers contained 6 log10CFU of each pathogen. The carriers were irradiated for 41 minutes at a distance of 4 feet from the Tru-D or Pathogon device. The means of the data from experiments conducted in triplicate are presented. Error bars indicate standard error.
The Effect of Distance and Indirect Irradiation
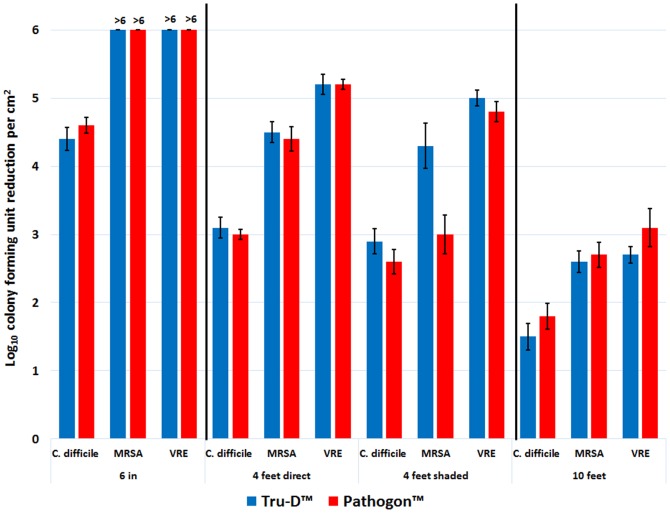
The germicidal efficacy of UV-C light as a function of distance follows an inverse relationship, as shown in Figure 4. The Tru-D and Pathogon devices achieved analogous log reductions for each distance assessed. Six inches away from the device, vegetative organisms (MRSA and VRE) were completely eliminated (≥6 log10CFU/cm2) and C. difficile spores were reduced by >4 log10CFU/cm2. As the distance from the device was increased to 4 feet, the log reduction decreased to ≤5 log10CFU/cm2 for vegetative organisms and ≤3 log10CFU/cm2 for C. difficile spores. Shading the organisms from the direct field of radiation did not have a significant impact on the killing efficacy of the devices. Ten feet from the devices, the log reductions decreased further to ≤3 log10CFU/cm2 for vegetative organisms and <2 log10CFU/cm2 for C.difficile spores.
Figure 4. The effect of distance and indirect irradiation on the efficacy of the UV-C devices.
The log10CFU reduction/cm2 of C.difficile spores, MRSA, and VRE at increasing distances and shaded from the direct field of radiation delivered by the UV-C device. Carriers contained 6 log10CFU of each pathogen. The carriers were irradiated for 41 minutes at a distance of 6 in, 4 feet, 4 feet shaded, and 10 ft from the Tru-D or Pathogon device. The means of the data from experiments conducted in triplicate are presented. Error bars indicate standard error.
The Effect of Radiant Dose
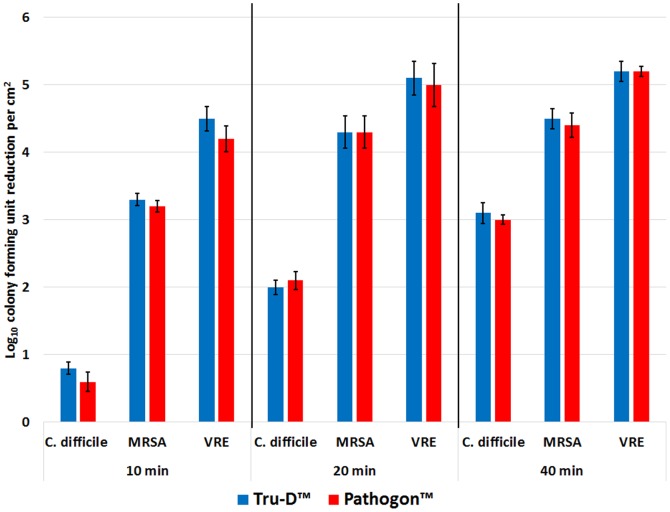
Figure 5 shows the effect of radiant dose on the killing efficacy of the UV-C devices. There was no significant difference between the killing efficacies of the Tru-D or Pathogon device for each of the timed doses of irradiation delivered. Killing achieved by the UV-C devices was directly proportional to the dose of irradiation delivered. MRSA and VRE were reduced by >3 log10CFU/cm2 after only 10 minutes of irradiation, while the hardier C. difficile spores required 40 minutes of irradiation to achieve a >3 log10CFU/cm2 reduction.
Figure 5. The effect of radiant dose on the efficacy of the UV-C devices.
The log10CFU reduction/cm2 of C.difficile spores, MRSA, and VRE after receiving increasing timed doses of irradiation delivered by the UV-C devices. Carriers contained 6 log10CFU of each pathogen. The carriers were irradiated for 10, 20, 40 minutes at a distance of 4 feet from the Tru-D or Pathogon device. The means of the data from experiments conducted in triplicate are presented. Error bars indicate standard error.
Discussion
We found that the Tru-D and Pathogon devices were equally effective for killing C. difficile spores, MRSA, and VRE in a laboratory setting. Surfaces in a real-world setting contain variable levels of contamination, and in our experience, yield between 4 to <1 log10CFU when cultured (author's unpublished data). Here, we determined that the concentration of pathogens on a surface did not have a significant impact on the killing efficacy of the UV-C devices. Conversely, organic load did significantly reduce the killing efficacy of both devices. These findings are inconsistent with previously published data showing that organic load did not impact the killing efficacy of the Tru-D device [16]. However, the organic load used in the current study was much more concentrated (5% fetal calf serum or 5% fetal calf serum plus 5% tryptone) than in the previously published study (1% bovine serum albumin). The current study demonstrates that as the matrix of organic load increased from light to heavy, the killing efficacy of the devices decreased, suggesting that UV-C light does not penetrate heavy soils, but may break through lighter organic loads.
Other factors known to impact the delivery of lethal doses of UV-C irradiation are distance from the device and time of radiant exposure [16]–[17]. The efficacy of the Tru-D and Pathogon devices significantly decreased as distance from the devices increased. We have previously demonstrated that shading from the direct field of irradiation inhibited the lethal effects of the Tru-D device (assessed at 10 feet from the device) [16]. Conversely, here we found that at 4 feet from the devices, shading did not have a significant impact on the killing efficacy of UV-C. For killing of C. difficile spores, time of UV-C exposure was of particular importance. While vegetative organisms were reduced by 3 log10CFU after only 10 minutes of exposure, it took 40 minutes to achieve the same level of reduction for C. difficile spores. These results suggest that the Tru-D and Pathogon devices are similarly effective at delivering lethal doses of UV-C irradiation under analogous conditions. And as previously demonstrated for the Tru-D device, the Pathogon device may be a promising new environmental disinfection technology that could be a useful adjunct to routine cleaning measures in healthcare facilities.
UV-C devices have important advantages over other disinfection strategies that are effective against C. difficile spores. Sodium hypochlorite has corrosive effects on various materials, may irritate the eyes and respiratory tracts of cleaning staff and patients, and the efficacy is dependent on correct application by housekeeping staff [20]. Hydrogen peroxide vapor and hydrogen peroxide dry-mist have been shown to be highly effective in elimination of C. difficile spores [20]–[22]. However, these systems are relatively expensive to operate, a dedicated staff is required, and up to several hours may be required to complete room disinfection [20]–[22]. In contrast, after the initial purchase of the UV-C device, the cost of operating and maintaining them is minimal (i.e., electricity and annual bulb replacement of ∼ $20 each), a dedicated staff is not essential, and a 3 log10CFU reduction in C. difficile spores can be achieved in less than an hour. Additionally, UV-C may be less damaging to surfaces than bleach and does not produce emissions that are harmful or irritating to operators.
The Tru-D and Pathogon devices do have some potential limitations. First, because spores require a minimum of 40 minutes of irradiation to achieve significant reductions, it may not be feasible to use the devices in circumstances where rapid turn-over of rooms is required. Second, surface properties and organic debris may potentially inhibit lethal doses of UV-C from killing pathogens. For example, UV-C does not penetrate porous surfaces such as sheets, upholstery and curtains [15]. In our current study, lethal doses of UV-C irradiation were significantly or completely inhibited by organic matrices. Finally, the efficacy of the UV-C devices was reduced at sites further from the devices. Therefore, it is recommended that commonly touched surfaces (e.g., bedside table, call button, telephone) be arranged close to the device for optimal exposure to UV-C radiation.
Both UV-C disinfection systems performed similarly in a laboratory setting, however, each system has certain advantages and limitations. The Tru-D device is unique in that it uses UV-C light reflected from the walls, ceilings, floors, and items in the room to calculate the amount of irradiation required to deliver a programmed lethal dose for either vegetative or spore-forming pathogens. This feature is advantageous because it delivers a customized dose of irradiation to each room based on the areas of the room that are hardest for light to penetrate. However, this advantage translates into longer cycle times for rooms that inhibit the reflection of light, often increasing the run time to greater than 50 minutes for a 10×10 foot room. On the other hand, the Pathogon device delivers a pre-programmed dose of irradiation that is configured based on the size of the room and type of pathogen contamination suspected, therefore, cycle time does not fluctuate. However, shaded areas of the room may not receive sufficient lethal doses of irradiation. In our opinion, the Pathogon control interface is more user-friendly, however, the next generation of the Tru-D device has been updated with an iPad interface. Last, the Pathogon device is significantly less expensive than the Tru-D device, however after the initial cost, both units require equivalent care and maintenance.
Our study does have some limitations. This study was not designed to address the impact of the UV-C devices on native pathogens found on surfaces in hospital rooms. Nevertheless, the two devices performed equivalently in a laboratory setting, and because the Tru-D device has been previously reported as effective for significantly reducing C. difficile, MRSA, and VRE contamination in hospital rooms it can be inferred that the Pathogon device may perform similarly. Further studies are needed to determine whether reductions achieved by these devices translates to reduced rates of infection.
Acknowledgments
We thank Steris Corporation, Mentor, OH for providing the Pathogon unit used in this study free of charge. Steris Corporation did not provide funding for the study, however C.W.F contributed to the study design and editing of the manuscript.
Data Availability
The authors confirm that all data underlying the findings are fully available without restriction. All relevant data are within the paper.
Funding Statement
This work was supported by a Merit Review grant from the Department of Veterans Affairs to C.J.D. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Goodman ER, Platt R, Bass R, Onderdonk AB, Yokoe DS, et al. (2008) Impact of an environmental cleaning intervention on the presence of methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci on surfaces in intensive care unit rooms. Infect Control Hosp Epidemiol 29: 593–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Weber DJ, Anderson D, Rutala WA (2013) The role of the surface environment in healthcare associated infections. Curr Opin Infect Dis 26(4): 338–44. [DOI] [PubMed] [Google Scholar]
- 3. Sitzlar B, Deshpande A, Fertelli D, Kundrapu S, Sethi AK, et al. (2013) An environmental disinfection odyssey: evaluation of sequential interventions to improve disinfection of Clostridium difficile isolation rooms. Infect Control Hosp Epidemiol 34(5): 459–65. [DOI] [PubMed] [Google Scholar]
- 4. Boyce JM, Potter-Bynoe G, Chenevert C, King T (1997) Environmental contamination due to methicillin-resistant Staphylococcus aureus (MRSA): possible infection control implications. Infect Control Hosp Epidemiol 18: 622–7. [PubMed] [Google Scholar]
- 5. Eckstein BC, Adams DA, Eckstein EC, Rao A, Sethi AK, et al. (2007) Reduction of Clostridium difficile and vancomycin-resistant Enterococcus contamination of environmental surfaces after an intervention to improve cleaning methods. BMC Infect. Dis 7: 61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hayden MK, Bonten JM, Blom DW, Lyle EA, van de Vijver D, et al. (2006) Reduction in acquisition of vancomycin-resistant Enterococcus after enforcement of routine environmental cleaning measures. Clin Infect Dis 42: 1552–60. [DOI] [PubMed] [Google Scholar]
- 7. Hacek DM, Ogle AM, Fisher A, Robicsek A, Peterson LR (2010) Significant impact of terminal room cleaning with bleach on reducing nosocomial Clostridium difficile . Am J Infect Control 38(5): 350–3. [DOI] [PubMed] [Google Scholar]
- 8. Carling PC, Parry MF, Bruno-Murtha LA, Dick B (2010) Improving environmental hygiene in 27 intensive care units to decrease multidrug-resistant bacterial transmission. Crit Care Med 38: 1212–4. [DOI] [PubMed] [Google Scholar]
- 9. Conner-Kerr TA, Sullivan PK, Gaillard J, Jones RM (1998) The effects of ultraviolet radiation on antibiotic-resistant bacteria in vitro. . Ostomy Wound Manage 44: 50–6. [PubMed] [Google Scholar]
- 10. Griego VM, Spence KD (1977) Inactivation of Bacillus thuringiensis spores by ultraviolet and visible light. Appl Env Microbiol 35: 906–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hercik F (1936) Action of ultraviolet light on spores and vegetative forms of Bacillus megatherium sp. J Gen Physiol 20: 589–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Setlow P (2006) Spores of Bacillus subtilis: their resistance to and killing by radiation, heat and chemicals. J Appl Microbiol 101: 514–25. [DOI] [PubMed] [Google Scholar]
- 13. Setlow P (2001) Resistance of spores of Bacillus species to ultraviolet light. Environ Mol Mutagen 38: 97–104. [DOI] [PubMed] [Google Scholar]
- 14. Thai TP, Keast DH, Campbell KE, Woodbury MG, Houghton PE (2005) Effect of ultraviolet light C on bacterial colonization in chronic wounds. Ostomy Wound Manage 51: 32–45. [PubMed] [Google Scholar]
- 15. Owens MU, Deal DR, Shoemaker MO, Knudson GB, Meszaros JE, et al. (2005) High-dose ultraviolet C light inactivates spores of Bacillus subtilis var. niger and Bacillus anthracis Sterne on non-reflective surfaces. Appl Biosafety 10: 240–7. [Google Scholar]
- 16. Nerandzic MM, Cadnum JL, Pultz MJ, Donskey CJ (2010) Evaluation of an automated ultraviolet radiation device for decontamination of Clostridium difficile and other healthcare-associated pathogens in hospital rooms. BMC Infect Dis 10: 197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Rutala WA, Gergen MF, Weber DJ (2010) Room decontamination with UV radiation. Infect Control Hosp Epidemiol 31(10): 1025–9. [DOI] [PubMed] [Google Scholar]
- 18. Sorg JA, Sonenshein AL (2008) Bile salts and glycine as cogerminants for Clostridium difficile spores. J Bacteriol 190(7): 2505–2512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Nerandzic MM, Donskey CJ (2009) Effective and reduced-cost modified selective medium for isolation of Clostridium difficile . J Clin Microbiol 47(2): 397–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Barbut F, Menuet D, Verachten M, Girou E (2009) Comparison of the efficacy of a hydrogen peroxide dry-mist disinfection system and sodium hypochlorite solution for reduction of Clostridium difficile spores. Infect Control Hosp Epidemiol 30: 507–14. [DOI] [PubMed] [Google Scholar]
- 21. Boyce JM, Havill NL, Otter JA, McDonald LC, Adams NM, et al. (2008) Impact of hydrogen peroxide vapor room decontamination on Clostridium difficile environmental contamination and transmission in a healthcare setting. Infect Control Hosp Epidemiol 29: 723–9. [DOI] [PubMed] [Google Scholar]
- 22. Boyce JM (2009) New approaches to decontamination of rooms after patients are discharged. Infect Control Hosp Epidemiol 30: 515–7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The authors confirm that all data underlying the findings are fully available without restriction. All relevant data are within the paper.