Abstract
Amyotrophic lateral sclerosis (ALS) is a neurodegenerative disease, characterized by the predominant loss of motor neurons (MNs) in primary motor cortex, the brainstem, and the spinal cord, causing premature death in most cases. Minimal delay of pathological development by available medicine has prompted the search for novel therapeutic treatments to cure ALS. Cell-based therapy has been proposed as an ultimate source for regeneration of MNs. Recent completion of non-autologous fetal spinal stem cell transplant to ALS patients brought renewed hope for further human trials to cure the disease. Autologous somatic stem cell-based human trials are now in track to reveal the outcome of the ongoing trials. Furthermore, induced pluripotent stem cell (iPSC)-based ALS disease drug screen and autologous cell transplant options will broaden therapeutic options. In this review paper, we discuss recent accomplishments in cell transplant treatment for ALS and future options with iPSC technology.
Keywords: ALS, cell transplant, stem cell, iPSC, drug screen, neural stem cell
INTRODUCTION
Amyotrophic lateral sclerosis (ALS), also known as Lou Gehrig's disease, is the most fatal progressive neurodegenerative disease in the motor neurons (MNs) [1]. The loss of spinal cord MNs devastates basic, fundamental movements, such as breathing, and typically causes death to patients within 2~5 years after diagnosis. Progressive deterioration of motor function in patients severely disrupts their breathing ability, requiring some form of breathing aid for survival of the patients.
The causes of ALS are complicated and still under investigation. 5~10% of total ALS cases have been genetically identified, and more than dozen genes have been found to be involved [2, 3, 4]. Cu2+/Zn2+ superoxide dismutase (SOD-1), TAR DNA binding protein-43 (TDP-43), and GGGGCC repeat expansions in C9orf72 locus are the major familial ALS mutations in general. SOD-1 and Fused in Sarcoma/Translocated in Sarcorma (FUS) gene mutations are more common in Korean familial ALS [3, 4]. Genetic mutation studies using transgenic mice and immortalized cell-based studies revealed that those mutations increased ER stress, reactive oxygen species (ROS) generation, mitochondrial damage, insulin-like growth factor (IGF1) dependency for survival, neurofilament protein aggregation, and glutamate excitotoxicity, specifically in the motor neurons (MNs). Targeting one of those pathological phenomena has achieved little or no cure against ALS. Currently, the only available FDA approved drug is Riluzole, which antagonizes the glutamate response to reduce the pathology development of ALS [1, 5]. However, only about a three-month life span expansion for ALS patients in the early stages has been reported, and no therapeutic benefit for ALS patients in the late stages has been observed, indicating lack of therapeutic options for the patients [5]. Since, the effect of current medicine is limited to the patient conditions, cell transplant to replace the dying neurons has been suggested. This idea to replenish tissues with stem cells has also been suggested for traumatic spinal injuries, which currently have no available treatment.
There are two major cell origins that are used for cell-based transplant in ALS. One involves mesenchymal stromal cells (MSC), such as bone marrow mesenchymal stromal cells (BMSC), adipocyte stem cells (ASC), bone marrow mobilized CD133+ hematopoietic stem cell (HSC), and CD34+ progenitor from umbilical cord blood cells (CB) [2, 6, 7]. The other involves neural tissue origin neural stem cells, such as fetal origin spinal cord and olfactory ensheathing glial cells (OEC) [6, 8, 9]. There are ongoing debates over which origin of transplant cell has regenerative potential [2, 6, 7].
Recently, induced pluripotent stem cell (iPSCs)-based autologous cell transplant has been tested in various animal models with similar or better symptomatic outcome [10]. Current progress in gene editing and neuronal differentiation from iPSCs can ensure unlimited autologous cell sources with genetically modified manners. Furthermore, small molecule screening to revive the dying MNs with ALS patient specific iPSC disease modeling can provide valuable treatment to early stage ALS patients. In this regard, recent progress in cell-based transplant results, mostly in human trials, and future iPSC-based ALS treatment will be discussed.
CELL-BASED THERAPY FOR ALS
Mesenchymal stromal cell (MSC) transplant
Benefit of MSC transplantation has been well established from non-neurological disorders such as renal disease, cardiac disease, and inflammatory bowl disease (Crohn's disease) [7, 11]. In MSC transplant, multipotent somatic stem cells are isolated from bone marrow, adipocyte, and cord blood cells [12, 13]. MSC showed potential to differentiate into the three germ layers (the ectoderm, endoderm, and mesoderm) in highly specified conditions, both animal and human trials with MSC infusion showed almost no integration of MSC into the host tissue [11].
MSC population can rejuvenate old tissues, not only in blood lineage tissues, but also in cartilage, smooth muscles, and neurons. Researchers found that exogenously added MSC cells, by scavenging toxic inflammatory cytokines and releasing tropic cytokines, were able to relieve the symptoms and replenish the residual parenchymal cells.
Bone marrow-derived mesenchymal stromal cell (BMSC) transplant
BMSC can be purified from bone marrow relatively easily by simple adherent culture and cell sorting with CD20+, CD73+, CD90+, CD105+/CD34-, CD45-, and CD19- cells [12]. Researchers found that exogenously added stem cells did not differentiate into MNs, rather tropic benefits from stem cell induced residual MNs survival and regrowth. Neurotropic effect on MSC transplant in ALS animal models is well correlated with current understanding of ALS pathology. In addition, ALS has non-cell autonomous toxicity in the astrocytes, oligodendrocytes, and glial cells [14]. Dysfunction of non-neuronal cells, such as loss of metabolic support, alteration of neuronal-glia, secretion of toxic metabolites, exacerbated MN death in ALS [12, 15]. Recent findings in MSCs' regulation upon Treg, indicating the direct immune modulatory role of MSC in progression of ALS [16].
There have been ongoing human trials with MSC on ALS treatment with completed human trial data (human clinical trials data as time of review paper written, https://clinicaltrials.gov) [7, 8, 17]. In 2010, Mazzini et al. reported phase 1 trial data with MSC transplant in ALS patients in Italy [18]. 10 patients received autologous BMSC directly into the spinal cord after BMSC expansion. Expanded BMSC was re-suspended in patient autologous cerebrospinal fluid (CSF) before injection into the central part of the spinal cord (T4/T5;T5/T6, high thoracic level with median 75*106 cells injection per person). There was no significant adverse effect due to the surgical treatment, either in the brain or in the spinal cord. Two patients showed significant delay of the disease, compared to the previous control data, but most of the patients did not show any significant improvement compared to the control group. It is noteworthy to mention that MSC from the patients or from the control group showed little or no difference in terms of MSC properties in growth factor release and neuro-protection [18].
Recently, a Korean researcher group revealed that MSC from the individual patients could be pre-selected, based on good-behaving biomarkers [2]. The phase 2 trials finished in 2014 with ALS treatment with BM MSC in Korea brought valuable data for BM MSC treatment with ALS. 37 ALS patents received autologous MSC intrathecally (at the level of L2-3, 1×106 cells/kg) twice with one-month interval. Their injection protocol was similar with other studies, in which the patients' CSF was used to dilute the BM MSC for injection. ALS functional rating scale (ALSFRS-R) was used to categorize patient outcome. With their category, responder vs. non-responder, BM MSC information was reanalyzed and confirmed with ALS model animal injection studies. Vascular enthothelial growth factor (VEGF), Angiogenin (ANG), and Transforming growth factor-β (TGF-β) could be biological markers for predicting prognosis MSC transplant, indicating further optimization of ALS treatment [2]. Also, HYNR-CS non-autologous cell therapy products from the same group, which originated from ALS patients BM MSC with superior ability to delay ALS symptom development, obtained conditional approval from Korean Food and Drug Administration for use in ALS patients.
There has been a report that CD133+ peripheral blood cell injection into ALS patients brought about great improvement to the patients [19]. A human trial in Mexico showed significant improvement of ALS patient symptoms with 10 patients, who received 300 µg of filgrastim for 3 days before collecting CD133+ cells from their peripheral blood mononuclear cells. Filgrastim is a recombinant human granulocyte colony-stimulating factor (G-CSF) that stimulates hematopoietic cells proliferation and activation. Autologous CSF was used for CD133+ cell resuspension. 13 patients in the control group were treated with conventional Riluzole, antioxidant, and physical treatment. Compared to the control group's median survival time, the treated group showed more than three times the survival time (19 months vs. 66 months). G-CSF mediated bone marrow mobilization in the ALS patients is not mutually exclusive in ALS symptom relief [20]. However, CD133+ cells transplanted (1×105 cells or more per person into frontal motor cortex) case only showed clinical improvement, indicating that the ALS symptom release was due to the transplanted stem cells. Furthermore, a small-scale safety trial in Spain showed significant reduction in the cellular level of TDP-43 and increase of motor neuron cortex after BMSC treatment, which suggested the possible cell transplant efficacy of BMSC in ALS [21].
Adipocyte stem cell (ASC) transplant
Spine injury treatment with ASC transplant in rodents has been reported [13]. Similar technical application into an ALS mice model also showed delayed ALS symptoms in the model animals [22]. Although the number of preclinical studies was low, it was very feasible to see the clinical outcome from the MSC replacement.
Umbilical cord blood cell transplant
Cord blood transplant studies reported similar outcomes, as expected in the hematopoietic stem cell transplant studies (Table 1) [23]. The neurotropic effects were effective enough to revive the ALS animals, which consisted mostly of SOD1 rodents. Current ongoing human trials will give better understanding of cord blood cells against ALS [17, http://clinicaltrials.org].
Table 1.
Representative cell transplant trials in ALS

HSSC, Human fetus spinal cord stem cell; OEC, Olfactory ensheathing cell; hiPSC/iNPC, Human iPSC-derived neural progenitor cells; hES, Human embryonic stem cell; NA, Not applicable.
NEURONAL LINEAGE STEM CELL TRANSPLANT
Neural stem cell-based transplant
Previous trials with undifferentiated stem cell (BMSC, human embryonic stem cells, and hematopoietic stem cells) transplant did not fully visualize the potential of stem cell therapy. Conditions in the adult spine are not favorable for transplanted stem cells to differentiate into motor neurons. Although the researchers observed paracrine effect to relieve the ALS symptoms, they looked at other sources of stem cells to replenish the dying MNs in the spinal cord. Compared to MSCs, neural stem cells from the spinal cord not only showed beneficial growth factor release but also became MNs and part of the spinal neuro circuit, showing proper synapse formation [7, 8, 10].
Use of human neural stem cell transplant in ALS model animals showed promising results, leading to studies in human trials [10, 24]. Human fetal spinal cord derived neural stem cells (HSSC) could be the ultimate replacement for the dying ALS MNs. Due to preclinical success from the human neural stem cell transplant studies in the ALS models, phase 1 human trial with HSSC transplant started in 2009 [8]. The surgical procedure was identical to the ones in previous neural tissue transplant trials with 5*105~1*106 cells per person [25, 26].
In 2014, the first report regarding cervical injection of neural stem cells in 6 ALS patients was released [8]. The release data from Feldman et al. was combined clinical data from a safety trial with lumbar intra-spinal injection by Glass et al. [8, 25]. Feldman et al. showed two important clinical observations of HSSC transplant in ALS. First, repeated intra-spinal injection of HSSC into the same patients showed to be beneficial for treating ALS without causing any severe side effects. Second, some of the patients without bulbar palsy and early transplant during the time of ALS disease development showed great reduction of pathology development, indicating a potential benefit [8].
Olfactory ensheathing glial cell (OEC) transplant
Olfactory ensheathing cells are known as olfactory Schwann cells, which ensheath the non-myelinated olfactory neurons. They assist in axonal growth, path finding, and synapse formation during regeneration of neuron. Their ability to connect the central nervous system (CNS) with the peripheral nervous system (PNS) and their multi-potent stem cell properties make OEC an excellent candidate for ALS cell transplant. Successful preclinical studies with OEC cells have been shown in clinical tests [7, 17].
Two major human clinical trials have been reported in China [9, 27]. Initial safety trials with 14 patients with OEC transplant did not show any adverse symptoms from the intra-spinal injection of fetus-OEC cells (2*106 cells per person into bilateral corona radiata), but showed minor improvement of the patients' survival rate [27]. For the follow-up multiple treatment of OEC transplant, the authors found increment of functional improvement, indicating the safety and feasibility of multiple injection toward the fragile ALS spine [9].
NOVEL APPROACH WITH INDUCED PLURIPOTENT STEM CELL (iPSC) TECHNOLOGY
Nobel prize-winning induced pluripotent stem cell (iPSCs) technology endowed us with two major tools in the fight against ALS. First, it allowed us to generate patient derived iPSC to study ALS. Less than 10% of the ALS cases showed inherited problems. Furthermore, various genetic backgrounds among the familial ALS cases have hindered a detailed pathology study to find a possible cure for the disease with conventional disease models, such as transgenic rodent models or ones using immortalized artificially overexpressing cell lines [28]. However, iPSC disease models use the patients' own samples and provide patient oriented disease pathology. Second, iPSC technology can provide an autologous source of cell transplant for ALS patients. hES and iPSC have proven and necessary properties to become motor neurons both in vitro and in vivo in an unlimited way. iPSC can provide more accessibility of transplant sources and property to become desired neurons compared to somatic sources of stem cells.
OVERVIEW OF iPSC
The pluripotency of iPSCs parallels that of hES [29, 30]. For example, iPSCs can differentiate into 3 germ layer lineages, such as the ectoderm, endoderm, and mesoderm. But, iPSCs can have intrinsic differences, such as epigenetic memories depending on the origin of the transformed cells. It has been reported that iPSC can be differentiated into cell origin tissues much easier, compared to hES [31]. However, target tissue differentiation from iPSC is basically identical to hES-derived differentiation.
Whether mice iPSCs are equivalent to mice ES cells was addressed by making transgenic mice, which satisfied the golden standard definition of stemness [32]. However, there were critics questioning the quality issues of human ES and iPSCs [33], since hES and hiPSC were more like epiblast-looking rather than mice ES cells. The golden standard definition of stemness entailed generating a germline competent transgenic organism, which could not be applied in human cases. Recently, studies from pig iPSC and transgenic pig generation using Yamanaka 4 factors supported the stemness of hES and hiPSC indirectly [32, 34]. Some of the epiblast features in human iPSC and hES may be too trivial to become fully competent, since epiblast-looking, non-rodent porcine iPSC could be fully regenerated into a transgenic pig.
Neural differentiation is the first specified stem cell target tissue [35]. Due to spontaneous differentiation from the early method, most of the current neural differentiation utilized various chemicals and growth factors for neural induction [35, 36, 37, 38]. Neural rosette-based induction separated central nervous system (CNS) neurons and peripheral nervous system (PNS) neurons to specify further subtypes of neurons. Both purity and maturity of diverse neuronal inductions allowed the researchers to use iPSC for neuronal disease modeling and cell transplant therapy.
iPSC-BASED DRUG SCREEN FOR ALS
Using Yamanaka's iPSC technology, several trials were able to generate ALS patient specific iPSC [30, 39]. The initial effort was to generate a disease model to find novel cures for ALS [40]. Recently, two prominent studies reported novel therapeutic options [41, 42]. SOD-1 mutations containing patient iPSCs were used in the studies.
Kiskinis et al. used human SOD1+/A4V dominant mutation iPSC derived MNs and compared them with isogenic control MNs. They found that the expression of mitochondria functional and ER stress regulating genes was greatly reduced in the ALS patientderived iPSC MNs [42]. Previous human pathology studies with GGGGCC repeat expansions in C9orf72 locus patients' MNs also showed a similar molecular signature, indicating common ALS pathology pathway with a different genetic background [43]. Human origin based common ALS pathway studies may be the closest step to finding the most optimal therapy for ALS.
Chen et al. approached ALS with a different pathological aspect using the aforementioned SOD1 mutation [41]. They found that the ALS patient iPSC-derived MNs accumulated neurofilaments. Defected SOD1+/D90A blocked the expression of neurofilament-L (NF-L) by binding to the 3'UTR region of NF-L. Reduction of NF-L caused NF aggregation in the MNs, by improper oligormerization with subtypes of NFs, such as NF-H and NFM. MN expression of normal NF-L reversed the ALS symptom, indicating potential clinical intervention for ALS [41].
A novel ALS regimen through the combination of mice and human stem cell-derived MNs have been reported [44]. Yang M et al. initiated a screen with SOD1-mutated mice ES derived MNs, and then confirmed the efficacy with human ALS iPSC-derived MNs [44]. One of their candidates was Kenpaullone, and showed significant increase in MN survival. Kenpaullone was characterized as GSK-3 inhibitor, however other GSK-3 inhibitor such as CHIR99021 couldn't recapitulate MNs survival. Yang M et al. also found that Kenpaullone had inhibitory role in mitogen-activated protein kinase kinase kinase kinase 4 (MAP4K4, aka HGK,) mediated cell death pathway, suggesting the novel ALS treatment option in near future.
iPSC-BASED CELL THERAPY UPON ALS
Current progress of regeneration medicine using iPSC allows us to envision a potential, autologous pluripotent stem cell transplant. Compared to somatic stem cells, such as bone marrow-derived mesenchymal stem cells (BMSC), adipocyte stem cells (ASC), and even fetus derived neural stem cells, iPSC has superior potential to become mature motor neurons. Moreover unlimited expansion from autologous iPSC can potentially allow for unlimited opportunities for cell transplant for needed patients. A series of successful differentiation from iPSCs into target tissues have been reported, even in highly specified neuron, muscle, cardiac muscle, hepatocytes, and blood lineages cells with relevant animal transplant studies [30, 45, 46].
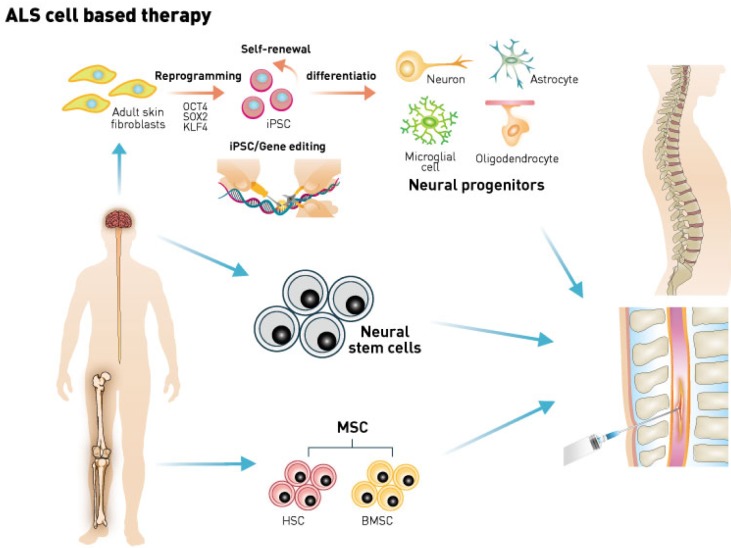
Recently, iPSC-derived neural progenitor cells (iNPC) showed compatible properties as human fetal-derived neural progenitor cells (fNPC), indicating the substitution of non-autologous fetal origin cell with patients' own iPSC derived cells [47]. Previous studies with iPSC neural progenitor differentiated cells did not provoke the host immune system in non-human primate brain injection models without any signs of tumorigenesis [48]. Furthermore, iNPC survived host neural circuit with synapse formation, providing proof-of-concept for future human transplant [48]. It is realistic to provide autologous iPSC-based cell transplant to suffering ALS patients. Combination of gene editing technology to fix the mutations in ALS patient-derived iPSC can be utilized to avoid transplanted cell survival issues of autologous MNs [30, 49] (Fig. 1).
Fig. 1.
Schematic view of current ongoing human cell based transplant approaches in ALS. Human hematopoietic lineage cell derived transplant and fetal origin neural tissues or neural stem cell transplant showed minimal reduction of symptoms however no significant harming on the patients. iPSC based autologous transplant in animal have been reported with great reduction of ALS symptoms, indicating possible human trials with iPSC derived neuronal stem cell or motor neurons.
CONCLUSION
Series of promising human trials with various sources of stem cells have been reported. Although the best results included the expansion of patient life span by about three-fold compared to the control group, it is still a devastating disease. The fight toward degenerative ALS should be more aggressive, instead of relying on small success in cell transplant trials.
Recently, we have devised other ways of fighting against ALS. Compared to previous stem cell sources, iPSC have two distinct advantages in combating ALS. We can study ALS from patient samples to find patient specific therapeutic regimen with a conventional drug screen, focusing on individual conditions. Second, we can use iPSC-derived neural progenitor cells for direct cell transplant after proper mutations fixation with known gene editing technology. Hopefully, we can someday finish the journey to overcome ALS and even other types of devastating spinal cord injuries.
ACKNOWLEDGEMENTS
This work was supported by the faculty research fund of Sejong University in 2014.
References
- 1.Bensimon G, Lacomblez L, Meininger V ALS/Riluzole Study Group. A controlled trial of riluzole in amyotrophic lateral sclerosis. N Engl J Med. 1994;330:585–591. doi: 10.1056/NEJM199403033300901. [DOI] [PubMed] [Google Scholar]
- 2.Kim HY, Kim H, Oh KW, Oh SI, Koh SH, Baik W, Noh MY, Kim KS, Kim SH. Biological markers of mesenchymal stromal cells as predictors of response to autologous stem cell transplantation in patients with amyotrophic lateral sclerosis; an investigator-initiated trial and in vivo study. Stem Cells. 2014 doi: 10.1002/stem.1770. (in press) [DOI] [PubMed] [Google Scholar]
- 3.Robberecht W, Philips T. The changing scene of amyotrophic lateral sclerosis. Nat Rev Neurosci. 2013;14:248–264. doi: 10.1038/nrn3430. [DOI] [PubMed] [Google Scholar]
- 4.Kwon MJ, Baek W, Ki CS, Kim HY, Koh SH, Kim JW, Kim SH. Screening of the SOD1, FUS, TARDBP, ANG, and OPTN mutations in Korean patients with familial and sporadic ALS. Neurobiol Aging. 2012;33:1017.e17–1017.e23. doi: 10.1016/j.neurobiolaging.2011.12.003. [DOI] [PubMed] [Google Scholar]
- 5.Bensimon G, Lacomblez L, Delumeau JC, Bejuit R, Truffinet P, Meininger V Riluzole/ALS Study Group II. A study of riluzole in the treatment of advanced stage or elderly patients with amyotrophic lateral sclerosis. J Neurol. 2002;249:609–615. doi: 10.1007/s004150200071. [DOI] [PubMed] [Google Scholar]
- 6.Mariano ED, Batista CM, Barbosa BJ, Marie SK, Teixeira MJ, Morgalla M, Tatagiba M, Li J, Lepski G. Current perspectives in stem cell therapy for spinal cord repair in humans: a review of work from the past 10 years. Arq Neuropsiquiatr. 2014;72:451–456. doi: 10.1590/0004-282x20140051. [DOI] [PubMed] [Google Scholar]
- 7.Lunn JS, Sakowski SA, Feldman EL. Concise review: Stem cell therapies for amyotrophic lateral sclerosis: recent advances and prospects for the future. Stem Cells. 2014;32:1099–1109. doi: 10.1002/stem.1628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Feldman EL, Boulis NM, Hur J, Johe K, Rutkove SB, Federici T, Polak M, Bordeau J, Sakowski SA, Glass JD. Intraspinal neural stem cell transplantation in amyotrophic lateral sclerosis: phase 1 trial outcomes. Ann Neurol. 2014;75:363–373. doi: 10.1002/ana.24113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen L, Chen D, Xi H, Wang Q, Liu Y, Zhang F, Wang H, Ren Y, Xiao J, Wang Y, Huang H. Olfactory ensheathing cell neurorestorotherapy for amyotrophic lateral sclerosis patients: benefits from multiple transplantations. Cell Transplant. 2012;21(Suppl 1):S65–S77. doi: 10.3727/096368912X633789. [DOI] [PubMed] [Google Scholar]
- 10.Lee HJ, Kim KS, Ahn J, Bae HM, Lim I, Kim SU. Human motor neurons generated from neural stem cells delay clinical onset and prolong life in ALS mouse model. PLoS One. 2014;9:e97518. doi: 10.1371/journal.pone.0097518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Salem HK, Thiemermann C. Mesenchymal stromal cells: current understanding and clinical status. Stem Cells. 2010;28:585–596. doi: 10.1002/stem.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Forostyak S, Jendelova P, Sykova E. The role of mesenchymal stromal cells in spinal cord injury, regenerative medicine and possible clinical applications. Biochimie. 2013;95:2257–2270. doi: 10.1016/j.biochi.2013.08.004. [DOI] [PubMed] [Google Scholar]
- 13.Zhou Z, Chen Y, Zhang H, Min S, Yu B, He B, Jin A. Comparison of mesenchymal stromal cells from human bone marrow and adipose tissue for the treatment of spinal cord injury. Cytotherapy. 2013;15:434–448. doi: 10.1016/j.jcyt.2012.11.015. [DOI] [PubMed] [Google Scholar]
- 14.Ilieva H, Polymenidou M, Cleveland DW. Non-cell autonomous toxicity in neurodegenerative disorders: ALS and beyond. J Cell Biol. 2009;187:761–772. doi: 10.1083/jcb.200908164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lunn JS, Sakowski SA, Federici T, Glass JD, Boulis NM, Feldman EL. Stem cell technology for the study and treatment of motor neuron diseases. Regen Med. 2011;6:201–213. doi: 10.2217/rme.11.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kwon MS, Noh MY, Oh KW, Cho KA, Kang BY, Kim KS, Kim YS, Kim SH. The immunomodulatory effects of human mesenchymal stem cells on peripheral blood mononuclear cells in ALS patients. J Neurochem. 2014 doi: 10.1111/jnc.12814. (in press) [DOI] [PubMed] [Google Scholar]
- 17.Faravelli I, Riboldi G, Nizzardo M, Simone C, Zanetta C, Bresolin N, Comi GP, Corti S. Stem cell transplantation for amyotrophic lateral sclerosis: therapeutic potential and perspectives on clinical translation. Cell Mol Life Sci. 2014;71:3257–3268. doi: 10.1007/s00018-014-1613-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mazzini L, Ferrero I, Luparello V, Rustichelli D, Gunetti M, Mareschi K, Testa L, Stecco A, Tarletti R, Miglioretti M, Fava E, Nasuelli N, Cisari C, Massara M, Vercelli R, Oggioni GD, Carriero A, Cantello R, Monaco F, Fagioli F. Mesenchymal stem cell transplantation in amyotrophic lateral sclerosis: A Phase I clinical trial. Exp Neurol. 2010;223:229–237. doi: 10.1016/j.expneurol.2009.08.007. [DOI] [PubMed] [Google Scholar]
- 19.Martinez HR, Gonzalez-Garza MT, Moreno-Cuevas JE, Caro E, Gutierrez-Jimenez E, Segura JJ. Stem-cell transplantation into the frontal motor cortex in amyotrophic lateral sclerosis patients. Cytotherapy. 2009;11:26–34. doi: 10.1080/14653240802644651. [DOI] [PubMed] [Google Scholar]
- 20.Tarella C, Rutella S, Gualandi F, Melazzini M, Scimè R, Petrini M, Moglia C, Ulla M, Omedé P, Bella VL, Corbo M, Silani V, Siciliano G, Mora G, Caponnetto C, Sabatelli M, Chiò A STEMALS STUDY GROUP. Consistent bone marrow-derived cell mobilization following repeated short courses of granulocyte-colony-stimulating factor in patients with amyotrophic lateral sclerosis: results from a multicenter prospective trial. Cytotherapy. 2010;12:50–59. doi: 10.3109/14653240903300682. [DOI] [PubMed] [Google Scholar]
- 21.Blanquer M, Moraleda JM, Iniesta F, Gómez-Espuch J, Meca-Lallana J, Villaverde R, Pérez-Espejo MÁ, Ruíz-López FJ, García Santos JM, Bleda P, Izura V, Sáez M, De Mingo P, Vivancos L, Carles R, Jiménez J, Hernández J, Guardiola J, Del Rio ST, Antúnez C, De la Rosa P, Majado MJ, Sánchez-Salinas A, López J, Martínez-Lage JF, Martínez S. Neurotrophic bone marrow cellular nests prevent spinal motoneuron degeneration in amyotrophic lateral sclerosis patients: a pilot safety study. Stem Cells. 2012;30:1277–1285. doi: 10.1002/stem.1080. [DOI] [PubMed] [Google Scholar]
- 22.Kim KS, Lee HJ, An J, Kim YB, Ra JC, Lim I, Kim SU. Transplantation of human adipose tissue-derived stem cells delays clinical onset and prolongs life span in ALS mouse model. Cell Transplant. 2013 doi: 10.3727/096368913X673450. (in press) [DOI] [PubMed] [Google Scholar]
- 23.Ende N, Weinstein F, Chen R, Ende M. Human umbilical cord blood effect on sod mice (amyotrophic lateral sclerosis) Life Sci. 2000;67:53–59. doi: 10.1016/s0024-3205(00)00602-0. [DOI] [PubMed] [Google Scholar]
- 24.Yan J, Xu L, Welsh AM, Hatfield G, Hazel T, Johe K, Koliatsos VE. Extensive neuronal differentiation of human neural stem cell grafts in adult rat spinal cord. PLoS Med. 2007;4:e39. doi: 10.1371/journal.pmed.0040039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Glass JD, Boulis NM, Johe K, Rutkove SB, Federici T, Polak M, Kelly C, Feldman EL. Lumbar intraspinal injection of neural stem cells in patients with amyotrophic lateral sclerosis: results of a phase I trial in 12 patients. Stem Cells. 2012;30:1144–1151. doi: 10.1002/stem.1079. [DOI] [PubMed] [Google Scholar]
- 26.Riley J, Federici T, Polak M, Kelly C, Glass J, Raore B, Taub J, Kesner V, Feldman EL, Boulis NM. Intraspinal stem cell transplantation in amyotrophic lateral sclerosis: a phase I safety trial, technical note, and lumbar safety outcomes. Neurosurgery. 2012;71:405–416. doi: 10.1227/NEU.0b013e31825ca05f. [DOI] [PubMed] [Google Scholar]
- 27.Huang H, Chen L, Xi H, Wang H, Zhang J, Zhang F, Liu Y. Fetal olfactory ensheathing cells transplantation in amyotrophic lateral sclerosis patients: a controlled pilot study. Clin Transplant. 2008;22:710–718. doi: 10.1111/j.1399-0012.2008.00865.x. [DOI] [PubMed] [Google Scholar]
- 28.Seok J, Warren HS, Cuenca AG, Mindrinos MN, Baker HV, Xu W, Richards DR, McDonald-Smith GP, Gao H, Hennessy L, Finnerty CC, López CM, Honari S, Moore EE, Minei JP, Cuschieri J, Bankey PE, Johnson JL, Sperry J, Nathens AB, Billiar TR, West MA, Jeschke MG, Klein MB, Gamelli RL, Gibran NS, Brownstein BH, Miller-Graziano C, Calvano SE, Mason PH, Cobb JP, Rahme LG, Lowry SF, Maier RV, Moldawer LL, Herndon DN, Davis RW, Xiao W, Tompkins RG Inflammation and Host Response to Injury, Large Scale Collaborative Research Program. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc Natl Acad Sci U S A. 2013;110:3507–3512. doi: 10.1073/pnas.1222878110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 30.Kim C. Disease modeling and cell based therapy with iPSC: future therapeutic option with fast and safe application. Blood Res. 2014;49:7–14. doi: 10.5045/br.2014.49.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vitaloni M, Pulecio J, Bilic J, Kuebler B, Laricchia-Robbio L, Izpisua Belmonte JC. MicroRNAs contribute to induced pluripotent stem cell somatic donor memory. J Biol Chem. 2014;289:2084–2098. doi: 10.1074/jbc.M113.538702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Okita K, Ichisaka T, Yamanaka S. Generation of germline-competent induced pluripotent stem cells. Nature. 2007;448:313–317. doi: 10.1038/nature05934. [DOI] [PubMed] [Google Scholar]
- 33.Tesar PJ, Chenoweth JG, Brook FA, Davies TJ, Evans EP, Mack DL, Gardner RL, McKay RD. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature. 2007;448:196–199. doi: 10.1038/nature05972. [DOI] [PubMed] [Google Scholar]
- 34.West FD, Terlouw SL, Kwon DJ, Mumaw JL, Dhara SK, Hasneen K, Dobrinsky JR, Stice SL. Porcine induced pluripotent stem cells produce chimeric offspring. Stem Cells Dev. 2010;19:1211–1220. doi: 10.1089/scd.2009.0458. [DOI] [PubMed] [Google Scholar]
- 35.Tabar V, Studer L. Pluripotent stem cells in regenerative medicine: challenges and recent progress. Nat Rev Genet. 2014;15:82–92. doi: 10.1038/nrg3563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang SC, Wernig M, Duncan ID, Brüstle O, Thomson JA. In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat Biotechnol. 2001;19:1129–1133. doi: 10.1038/nbt1201-1129. [DOI] [PubMed] [Google Scholar]
- 37.Reubinoff BE, Itsykson P, Turetsky T, Pera MF, Reinhartz E, Itzik A, Ben-Hur T. Neural progenitors from human embryonic stem cells. Nat Biotechnol. 2001;19:1134–1140. doi: 10.1038/nbt1201-1134. [DOI] [PubMed] [Google Scholar]
- 38.Chambers SM, Fasano CA, Papapetrou EP, Tomishima M, Sadelain M, Studer L. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nat Biotechnol. 2009;27:275–280. doi: 10.1038/nbt.1529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ross CA, Akimov SS. Human-induced pluripotent stem cells: potential for neurodegenerative diseases. Hum Mol Genet. 2014;23:R17–R26. doi: 10.1093/hmg/ddu204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang H, Doering LC. Induced pluripotent stem cells to model and treat neurogenetic disorders. Neural Plast. 2012;2012:346053. doi: 10.1155/2012/346053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Chen H, Qian K, Du Z, Cao J, Petersen A, Liu H, Blackbourn LW, 4th, Huang CL, Errigo A, Yin Y, Lu J, Ayala M, Zhang SC. Modeling ALS with iPSCs reveals that mutant SOD1 misregulates neurofilament balance in motor neurons. Cell Stem Cell. 2014;14:796–809. doi: 10.1016/j.stem.2014.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kiskinis E, Sandoe J, Williams LA, Boulting GL, Moccia R, Wainger BJ, Han S, Peng T, Thams S, Mikkilineni S, Mellin C, Merkle FT, Davis-Dusenbery BN, Ziller M, Oakley D, Ichida J, Di Costanzo S, Atwater N, Maeder ML, Goodwin MJ, Nemesh J, Handsaker RE, Paull D, Noggle S, McCarroll SA, Joung JK, Woolf CJ, Brown RH, Eggan K. Pathways disrupted in human ALS motor neurons identified through genetic correction of mutant SOD1. Cell Stem Cell. 2014;14:781–795. doi: 10.1016/j.stem.2014.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mori K, Weng SM, Arzberger T, May S, Rentzsch K, Kremmer E, Schmid B, Kretzschmar HA, Cruts M, Van Broeckhoven C, Haass C, Edbauer D. The C9orf72 GGGGCC repeat is translated into aggregating dipeptide-repeat proteins in FTLD/ALS. Science. 2013;339:1335–1338. doi: 10.1126/science.1232927. [DOI] [PubMed] [Google Scholar]
- 44.Yang YM, Gupta SK, Kim KJ, Powers BE, Cerqueira A, Wainger BJ, Ngo HD, Rosowski KA, Schein PA, Ackeifi CA, Arvanites AC, Davidow LS, Woolf CJ, Rubin LL. A small molecule screen in stem-cell-derived motor neurons identifies a kinase inhibitor as a candidate therapeutic for ALS. Cell Stem Cell. 2013;12:713–726. doi: 10.1016/j.stem.2013.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kaufman DS. Toward clinical therapies using hematopoietic cells derived from human pluripotent stem cells. Blood. 2009;114:3513–3523. doi: 10.1182/blood-2009-03-191304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ledran MH, Krassowska A, Armstrong L, Dimmick I, Renström J, Lang R, Yung S, Santibanez-Coref M, Dzierzak E, Stojkovic M, Oostendorp RA, Forrester L, Lako M. Efficient hematopoietic differentiation of human embryonic stem cells on stromal cells derived from hematopoietic niches. Cell Stem Cell. 2008;3:85–98. doi: 10.1016/j.stem.2008.06.001. [DOI] [PubMed] [Google Scholar]
- 47.Sareen D, Gowing G, Sahabian A, Staggenborg K, Paradis R, Avalos P, Latter J, Ornelas L, Garcia L, Svendsen CN. Human induced pluripotent stem cells are a novel source of neural progenitor cells (iNPCs) that migrate and integrate in the rodent spinal cord. J Comp Neurol. 2014;522:2707–2728. doi: 10.1002/cne.23578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Emborg ME, Liu Y, Xi J, Zhang X, Yin Y, Lu J, Joers V, Swanson C, Holden JE, Zhang SC. Induced pluripotent stem cell-derived neural cells survive and mature in the nonhuman primate brain. Cell Rep. 2013;3:646–650. doi: 10.1016/j.celrep.2013.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kim H, Kim JS. A guide to genome engineering with programmable nucleases. Nat Rev Genet. 2014;15:321–334. doi: 10.1038/nrg3686. [DOI] [PubMed] [Google Scholar]