Abstract
Our previous work has shown that non-thermal plasma treatment of demineralized dentin significantly (p<0.05) improved adhesive/dentin bonding strength for dental composite restoration as compared with the untreated controls. This study is to achieve mechanistic understanding of the plasma treatment effects on dentin surface through investigating the plasma treated dentin surfaces and their interaction with adhesive monomer, 2-Hydroxyethyl methacrylate (HEMA). The plasma treated dentin surfaces from human third molars were evaluated by water contact angle measurements and scanning electron microscopy (SEM). It was found that plasma-treated dentin surface with subsequent HEMA immersion (Plasma/HEMA Treated) had much lower water contact angle compared with only plasma-treated (Plasma Treated) or only HEMA immersed (HEMA Treated) dentin surfaces. With prolong water droplet deposition time, water droplets spread out completely on the Plasma/HEMA Treated dentin surfaces. SEM images of Plasma/HEMA Treated dentin surfaces verified that dentin tubules were opened-up and filled with HEMA monomers. Extracted type I collagen fibrils, which was used as simulation of the exposed dentinal collagen fibrils after acid etching step, were plasma treated and analyzed with Fourier transform infrared spectroscopy (FT-IR) and circular dichroism (CD) spectra. FT-IR spectra of the Plasma/HEMA Treated collage fibrils showed broadened amide I peak at 1660 cm−1 and amide II at 1550 cm−1, which indicate secondary structure changes of the collagen fibrils. CD spectra indicated that 67.4% collagen helix structures were denatured after plasma treatment. These experimental results demonstrate that non-thermal argon plasma treatment was very effective in loosing collagen structure and enhancing adhesive monomer penetration, which are beneficial to thicker hybrid layer and longer resin tag formation, and consequently enhance adhesive/dentin interface bonding.
Keywords: Plasma treatment, demineralized dentin surface, adhesive penetration, adhesive/dentin bonding: denatured collagen
1. Introduction
Polymer-based dental resin composites are replacing dental amalgam and becoming the most popular dental filling materials. Resin composites are widely accepted by patients due to aesthetic reasons and decrease of the potential health risk caused by mercury release from amalgams [1]. However, compared with dental amalgam, resin composite generally has shorter longevity [2, 3]. Extensive studies indicated that adhesive-dentin interface failure is a common issue reducing longevity of dental composite restoration [4–7]. There are two commercial adhesive systems—the self-etch and total-etch [8], in which the total-etch adhesive system is the most commonly used in dentist offices since it is deemed to provide stronger bonding on enamel than self-etch system [9]. However, the bond strength on dentin surface is still not very satisfactory with total-etching adhesive system. In dental composite restoration process, to remove smear layers, acid is used to etch dentin surface before total-etch adhesive application. The resulting dentin surface after acid etching is composed of collagen fibril network. Infiltration and polymerization of adhesive monomers into the exposed collagen network and dentin tubules forms micromechanical interlocking at the adhesive-dentin interface [10, 11]. Because of the unsatisfactory longevity and higher failure rate of dental composite restoration, numerous research works have been done to enhance adhesive infiltration in the exposed collagen fibril network and open dentin tubules in order to improve the adhesive/dentin interface bonding [12, 13].
Non-thermal plasma technique has attracted increasing attention in the biomedical field [14, 15]. In dentistry research, non-thermal plasma technique has been applied for dental cavity treatment [16], teeth whitening [17], oral bacteria deactivation [18]. Our group has reported the improvement of composite restoration in term of bonding strength using non-thermal plasma techniques [19–21]. It was observed that non-thermal plasma treatment of demineralized dentin surfaces could significantly improve adhesive-dentin interfacial bonding. However, the effects of plasma treatment on dentin substrate have not been well understood.
The micro-tensile test results reported in our previous study have shown that plasma treatment of demineralized dentin surfaces significantly improved the adhesive/dentin bonding strength with a 64% increase as compared with the untreated dentin controls [19]. Our very recent study using the same tooth control further verified that plasma treatment could enhance the adhesive/dentin interface bonding by 30.8% and 45.1% of mean increases (p<0.05) as compared with the untreated same tooth controls obtained using micro-tensile test specimens with cross-sectional areas of 1 × 1mm2 and 1 × 2mm2 respectively [21]. Surface analysis and characterization of the adhesive/denting interface did provide information about the possible underlying mechanism of why plasma treatment significantly improved the adhesive/dentin interface bonding [19–21]. However, the mechanistic understanding of the plasma treatment effects is still lacking. In literature, study of the adhesive/dentin interface have also shown controversial results regarding whether chemical or physical bonds play a more important role on interaction between dentin substrate and adhesive monomers [22–24]. In this study, the plasma treatment effects on dentin surfaces were further investigated and evaluated through characterizing the surface properties and collagen structure changes in order to understand the interactions of adhesive monomers with plasma-treated demineralized dentin surfaces and thus the resulting adhesive/dentin interface bonding.
2. Materials and Methods
2.1 Materials
Extracted human third molars without caries were collected under a project reviewed by the Institutional Review Board. All tissue samples were non-patient identified and all samples were handled and disposed according to the protocols suggested by Environmental Health and Safety Department at University of Missouri. The teeth were stored in the pH7.4 phosphate buffered saline solution (PBS) with 0.02% sodium azide to inhibit bacteria growth. Distilled 2-Hydroxyethyl methacrylate (HEMA) was obtained from Esstech (Essington, PA, USA) which was used directly. Type I collagen was supplied by Elastin Products Company (Owensville, MO, USA), which was extracted from calf skin and lyophilized. Analytical grade argon was purchased from Airgas Company (Holts Summit, MO, USA). All other reagents were of analytical grade and were used without further purification.
2.2. Fabrication of dentin samples
Extracted unerupted human third molars were used by cutting off the root and enamel using an Isomet 5000 diamond saw (Buehler, Lake Bluff, IL, USA) to expose the dentin. The teeth were further cut into dentin slices with 0.3–0.5mm thickness. 600-grit silicon carbide abrasive paper was used to polish the dentin surface. The polished surface was demineralized for 15s using Scotchbond phosphoric acid gel (3M ESPE Dental Products, St Paul, MN, USA) and then thoroughly washed with water spray. Excess water was gently blown away to dry dentin surface with oil-free compressed air.
2.3. Plasma treatment on dentin surface
The plasma treatment method using a plasma brush has been described in our previous works [19, 21]. The plasma brush was operated at a current level of 6 mA (equivalent to a power level of 2–3W) using Spellman HV power supply SL60 (Spellman, New York, USA). Argon was used as gas source with flow rate of 3,000 standard cubic centimeters per minute (sccm). Plasma treatment time was set for 30s. Fifteen dentin slices were randomly assigned to five groups for water contact angle measurement. The first group designated as Dentin Control, was used for water contact angle measurement without any treatment. The second group Designated as Plasma Treated was treated with plasma brush and rinsed with water before water contact angle measurement. The third group slices designed as HEMA Treated were immersed in HEMA for 2 min and then rinsed with water. The fourth group designed as Plasma/HEMA Treated was treated with plasma brush and then immersed into HEMA monomer for 2 min. Then water spray was used to thoroughly washed dentin slices. The last group was used for HEMA Absorption experiments. Dentin slices were immersed into HEMA and taken out at 30 s, 2min, 1 h and 24 h and washed with water spray. The water contact angles were then measured.
2.4. Water contact angle analysis
Excess water on dentin surface was gently blow-dried with oil-free compressed air for about 10s before water contact angle measurement. Water contact angles were determined with a goniometer equipped with a special optical system and a charge-coupled device (CCD) camera. A drop of water (approximately 0.5µl) was placed on dentin surface and the image was immediately sent via the CCD camera to the computer for analysis. Images were captured at 5s and 90s after drop deposition by Windows Live Movie Maker. Image J software was used to determine the contact angle of the water droplet. 4–5 drops of water were applied on dentin surface to get means and standard deviation of water contact angle.
2.4. SEM analysis
Surface morphology of the typical dentin slices in Dentin Control, HEMA Adsorption and Plasma/HEMA Treated groups were examined using scanning electron microscopy (SEM) (Quanta 600, FEI, OR, USA). After drying in vacuum at room temperature overnight, the specimens were mounted on aluminum stubs and coated with 5 nm of platinum for SEM examination at 10 KV.
2.5. Fourier transforms infrared (FT-IR) analyses
Type I collagen was used as an analog of dentin collagen fibril, which was treated with HEMA immersion and plasma treatment. In order to fully remove physically adsorbed HEMA on collagen, acetone was used to thoroughly wash collagen immersed with HEMA. Acetone was selected because it is a good solvent for HEMA but not for collagen, which cannot be dissolved by acetone. After washing thoroughly with acetone, the collagen was dried in vacuum. FT-IR spectra were obtained by KBr pellets technique which were taken on a Cary 660 FT-IR spectrometer (Agilent Technologies, USA) in the absorption mode and averaged from 32 scans.
2.6. Circular dichroism (CD) analysis
Approximately 0.3 mg collagen was exposed to plasma brush for 30s just like dentin slices. Plasma-treated and untreated collagen were then dissolved in 5ml acetic acid (0.1M) and stirring for 3h at room temperature respectively. The thermally denatured collagen was prepared by placing untreated collagen solution in an oven at 95°C for 20min. CD spectra was collected using AVIV Model 62DS spectrometer (Lakewood, NJ, USA) at 25°C over the wavelength interval 190–260nm. Data were averaged from 3 scans. A bandwidth of 1 nm and averaging time of 3s were used in the measurements. The light length of the sample cell was 1mm. A reference spectrum of 0.1M acetic acid was recorded and subtracted from the sample spectra. CD spectra were expressed as molar residual ellipticity and normalized to the protein concentration. They were analyzed using CONTIN algorithm and the Reference set 7 at Dichroweb [25, 26].
3. Results
3.1 Water contact angle measurements
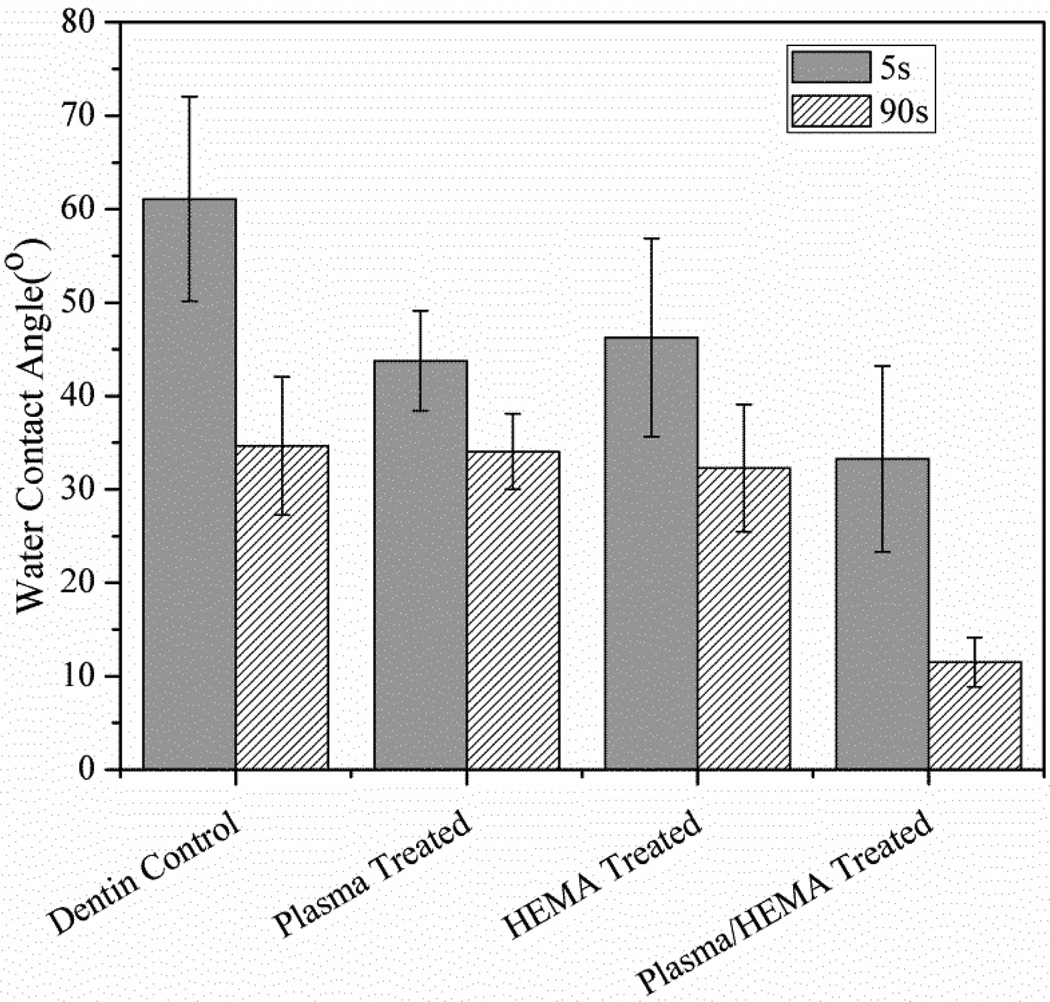
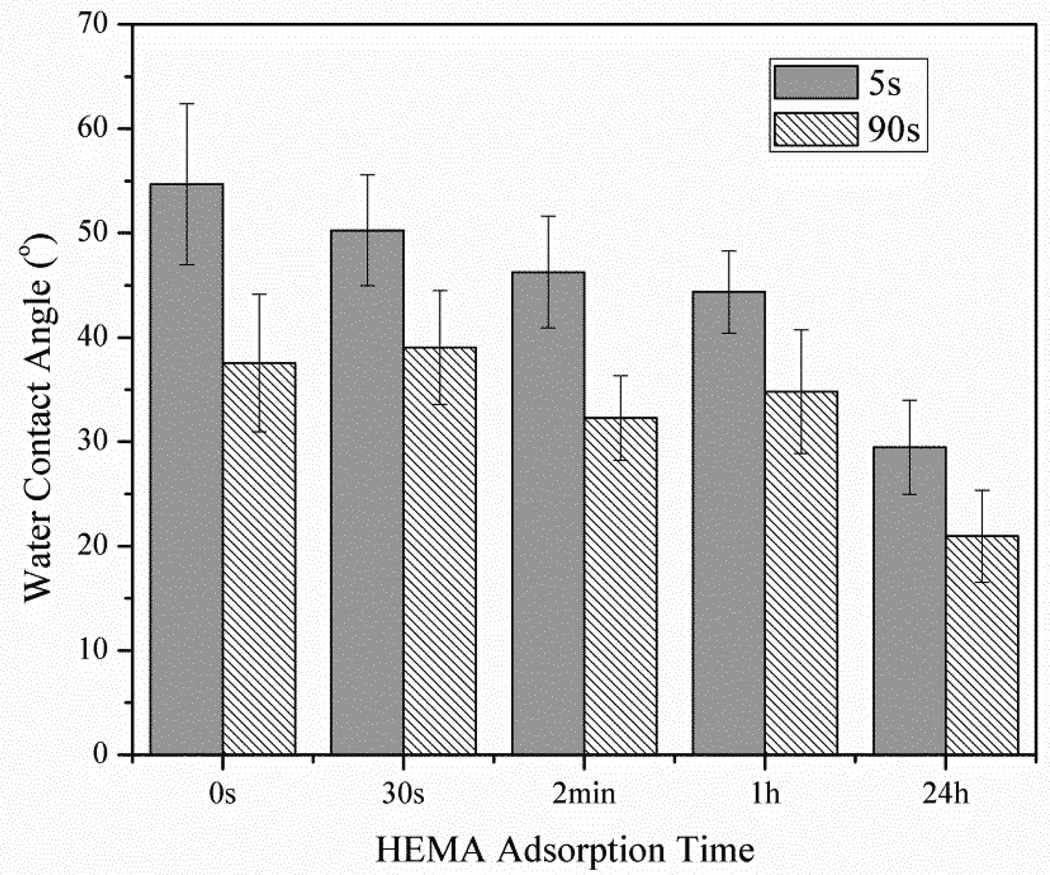
Fig. 1 shows average water contact angle on dentin slices in Dentin Control, Plasma Treated, HEMA Treated and Plasma/HEMA Treated groups. As shown in Fig. 1, the highest value of water contact angle was obtained with untreated dentin surface. Only plasma treatment or 2 min of HEMA immersion resulted in a slight decrease of water contact angles (around 25%) at 5 s of water drop deposition. However, after 90s of drop deposition, there was no significant difference for water contact angle between untreated dentin and plasma-treated or 2min of HEMA immersed dentin slices (p=0.187). Fig. 2 shows the average water contact angle on dentin slices with different HEMA adsorption times. After prolonged HEMA immersion time to 24h, significant decrease of water contact angle started to be observed on dentin surface at 90 s of water drop deposition. Plasma treatment plus HEMA immersion showed the lowest values for contact angles. After 90 s of the water drop deposition, the drops were observed to spread out on dentin surfaces. Less than 10° of water contact angle was measured. Compared with untreated dentin surface, dentin slices in Plasma/HEMA Treated groups showed 68% decrease of contact angle. The difference of water contact angle between Dentin Control and Plasma/HEMA Treated groups was statistically significant (p<0.001).
Figure 1.
Average water contact angle on slices in Dentin Control, Plasma Treated, HEMA Treated and Plasma/HEMA Treated groups. 5s and 90s represent water droplets deposition time.
Figure 2.
Water contact angle on dentin slices with different HEMA absorption times. 5s and 90s represent water droplets deposition time.
3.2 SEM examination
Representative SEM images of dentin surfaces with different treatment are presented in Fig. 3. Since dentin was demineralized with acid, the tubules were clearly observed without any smear plugs (Fig. 3A, D). After immersion in HEMA monomer for 24h, HEMA was adsorbed on the dentin surface and the edge of tubules (Fig. 3B, 3E). In Plasma/HEMA Treated group, more HEMA was penetrated into tubules instead of adsorbed on dentin surface after only 2min immersion (Fig. 3C, 3F).
Figure 3.
SEM images of dentin surface (A, D), dentin with HEMA absorption for 24h (B, E) and plasma-treated dentin with HEMA immersion for 2min (C, F).
3.4 FT-IR Spectra
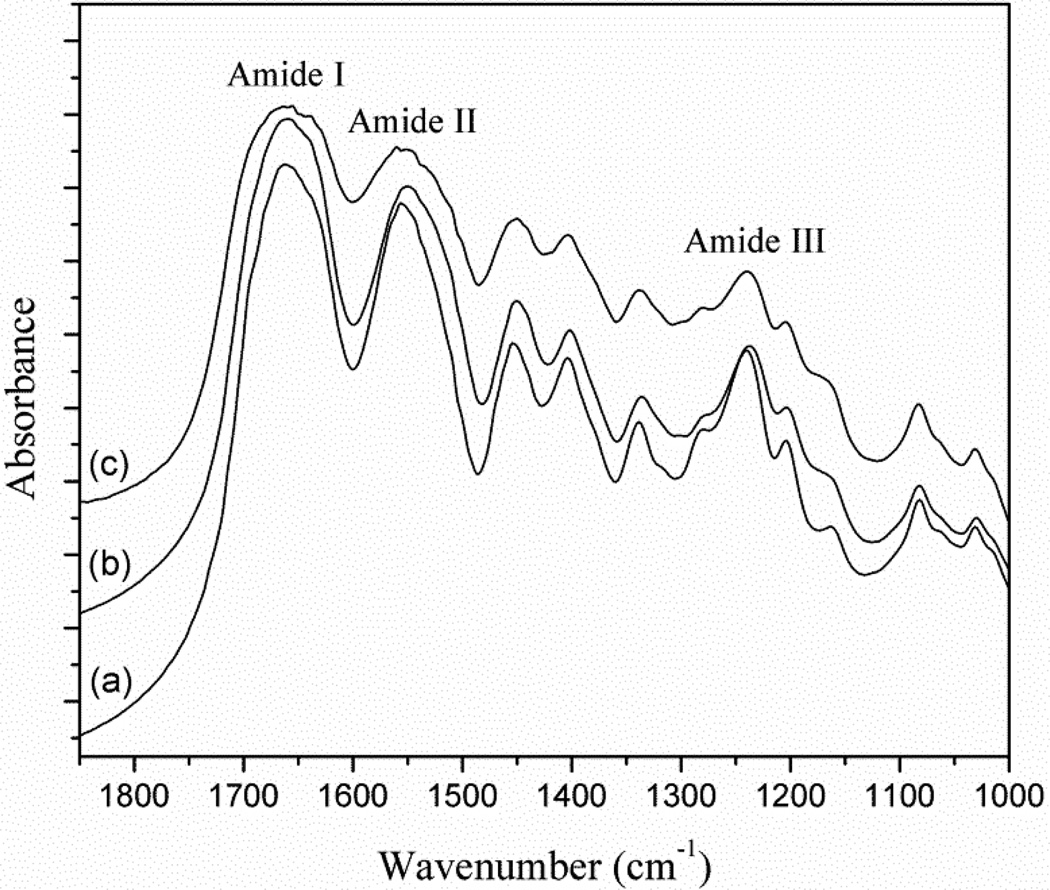
Fig. 4 shows the FTIR spectra of pure collagen (a), collagen immersed into HEMA (b) and plasma-treated collagen immersed into HEMA (c), respectively. The bands at 1660 cm−1, 1550 cm−1 and 1235 cm−1 corresponded to the amide I, II and III respectively. The main band of HEMA is the C=O group which should be at 1730cm−1. In Fig. 4b and 4c, no new characteristic absorptions band of HEMA in collagen was found after rinsing with acetone thoroughly. In Fig. 4c, it was observed that after plasma treatment the absorption band of amide I at 1660 cm−1 and amide II at 1550 cm−1 became broader.
Figure 4.
FT-IR spectra of (a) pure collagen, (b) collagen immersed into HEMA for 2 min and rinsed with acetone thoroughly, and (c) collagen treated with plasma for 30 s and immersed into HEMA for 2 min and then rinsed with acetone thoroughly.
3.5 Collagen structure
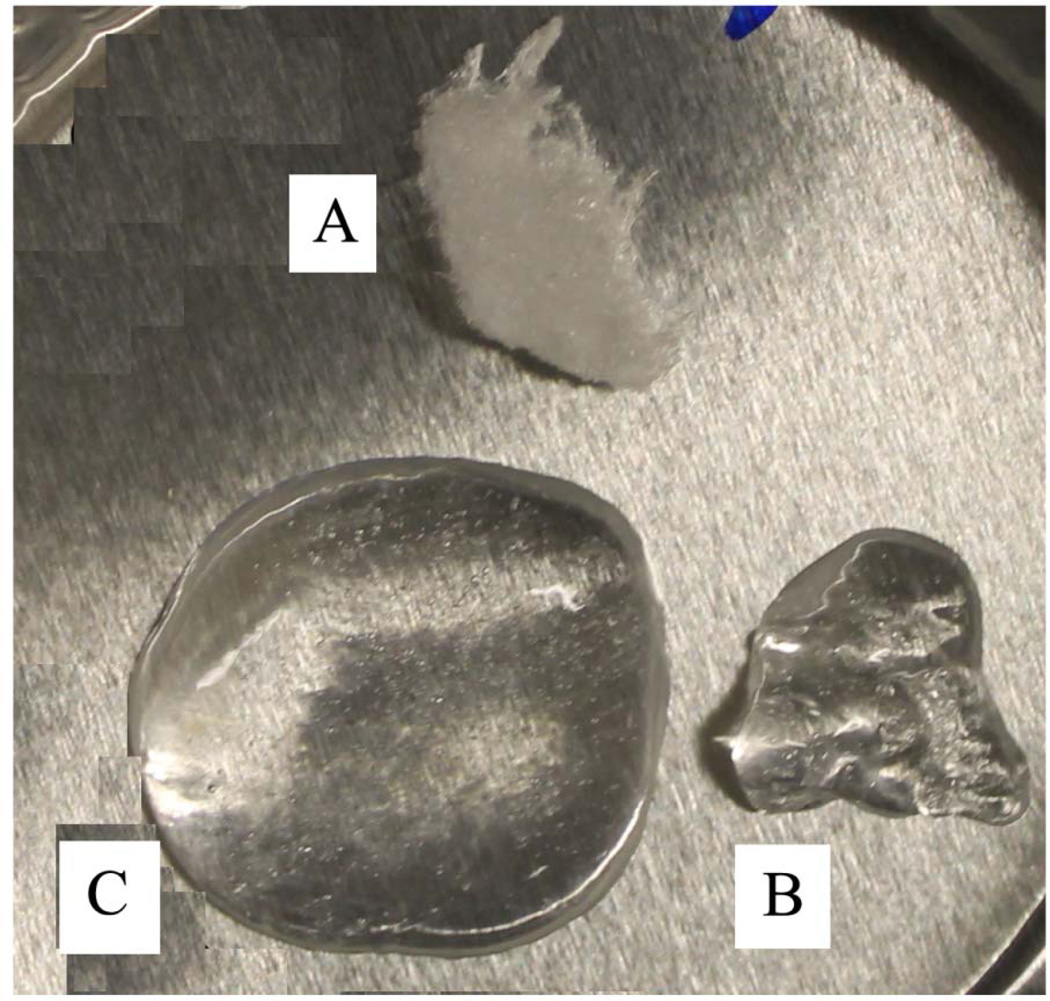
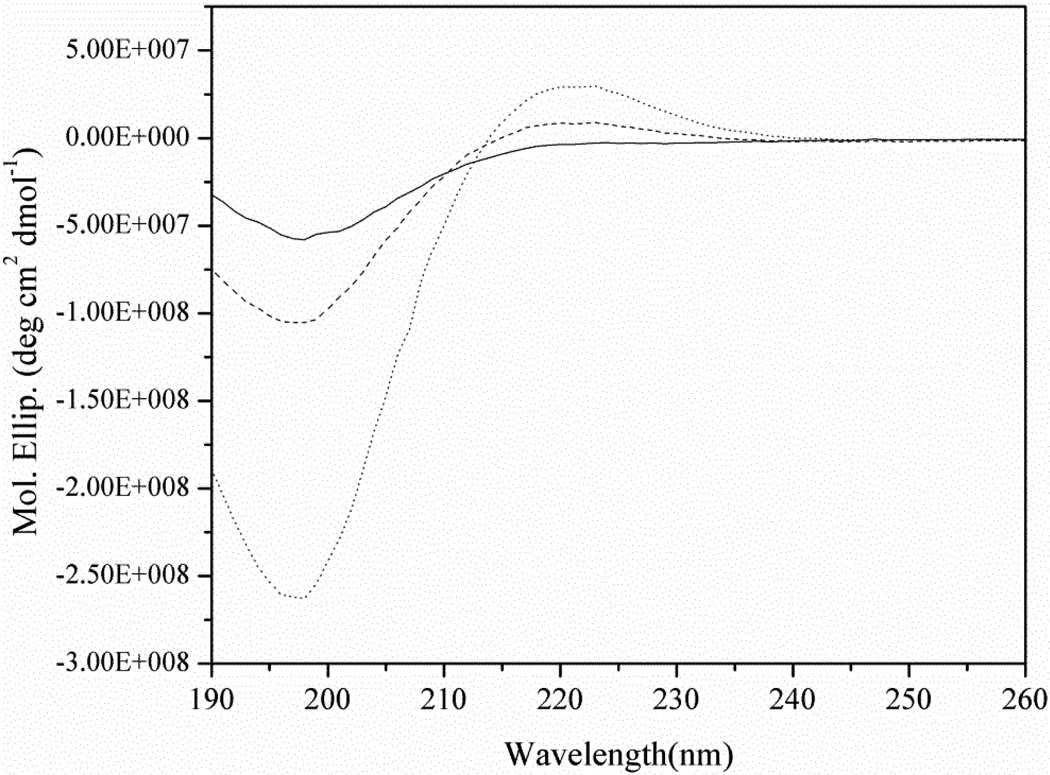
Water adsorption was investigated on untreated and plasma-treated collagen because plasma-treated collagen is first exposed to oral environment with high humidity. In dental composite restoration process, moreover, the water rewet process is performed before adhesive application. As shown in Fig. 5, Fig. 5 shows that compared with untreated collagen, plasma-treated collagen could adsorb more water, swelling and increase in volume. The CD spectra of untreated collagen, thermally denatured collagen and plasma-treated collagen have been measured and were shown in Fig. 6. The general shape and peaks of the spectrum of untreated and denatured collagen were in agreement with literature data [27]. The spectrum of the plasma-treated collagen displays positive and negative peaks with lower intensity compared to the untreated soluble collagen. As summarized in Table 1, analysis using CONTIN algorithm at Dichroweb indicated that 84.4% helix was thermally denatured by incubating at 95 °C for 20 min and 67.4% helix was denatured by 30s plasma treatment.
Figure 5.
Photograph of (A) untreated collagen, (B) untreated collagen with water adsorption, and (C) plasma-treated collagen with water adsorption
Figure 6.
CD spectra of collagen (dotted line), plasma-treated collagen (dash line) and thermal denatured collagen (solid line) dissolved in 0.1M acetic acid at 25°C. All the spectra shown were obtained after subtraction of baseline spectrum for the acetic acid.
Table 1.
Summary of secondary structure analysis data of collagen, denatured collagen and plasma-treated collagen using CONTIN algorithm at Dichroweb
| Samples | Helix (%) | Unordered (%) | Helix segment per 100 residues |
Ave helix length per segment |
|---|---|---|---|---|
| Collagen | 100 | 0 | 25.0 | 4.00 |
| Denatured collagen | 16 | 84 | 3.88 | 4.07 |
| Plasma-treated collagen | 33 | 67 | 8.95 | 4.00 |
Discussion
It is known that intrinsic dentin characteristics such as the number of tubules per mm2, water content of dentin and tooth condition could affect the adhesive/dentin interface bonding strength [28]. Comparisons of the adhesive-dentin bond strength of plasma-treated specimens with that of their untreated same-tooth controls were performed in our previous work. 30.8% and 45.1% of mean increases of bonding strengths were shown in plasma-treated group with a cross-sectional area of 1×1mm2 and 1×2mm2 respectively, which are statistically significant higher than their same-tooth untreated control groups(p<0.05) [21]. In our previous studies [19–21], we have proposed the hypothesis about plasma effects on collagen fibrils on demineralized dentin surface. Plasma species consist of high-energy electrons, ionic species, electronically excited neutrals and free radicals, which interact with dried collagen fibrils and might change the conformation of the collagen fibrils. After rewetting with water, the collagen fibers partially de-aggregate and allow the dental adhesive to easily adsorbed into collagen and interact with the underlying fibers to achieve higher bonding strength. We have also suggested that the significantly improved adhesive/dentin interface bonding could be attributed to the free radicals or peroxide groups introduced to the exposed collagen fibrils on dentin surface by plasma treatment. Both the surface free radicals and peroxide groups could initiate the polymerization and thus curing of resin monomers and form covalent chemical bonds between the resin and the collagen fibrils [20].
In this work, HEMA was employed to investigate the adhesive and plasma-treated demineralized dentin surface interaction. HEMA is widely used as monomer in dental adhesive because it can enhance monomer diffusion, improve resin entanglement with collagen fibrils and facilitate the formation of hybrid layers and resin tags [29]. The present results reveal that only 30s plasma treatment led to completely change of HEMA distribution on dentin surface. As shown in SEM images (Fig. 3), after plasma treatment HEMA infiltrated into tubules instead of adsorbed on dentin surface. HEMA in dentin tubules adsorbed water and decreased water contact angle with less than 10° was found after 90s of water drop deposition in Plasma/HEMA Treated groups.
In order to identify whether chemical bonding or physical adsorption play a more dominant role in the interaction between HEMA and dentin collagen fibrils, FTIR was conducted on collagen, collagen immersed with HEMA and plasma-treated collagen immersed with HEMA. Through acetone rinse, physically adsorbed HEMA was removed. If HEMA was chemically bonded to collagen, characteristic absorbance peak of HEMA would be observed on FTIR spectra. However, the peak of C=O at 1730cm−1 from HEMA was not observed in Fig. 4. This indicates chemical bonding between HEMA and collagen is not remarkable. Physical adsorption is supposed to be the dominant interaction between HEMA and dentin collagen fibrils.
It should be noted that three major findings were obtained in this experimental study: i) After plasma treatment, more HEMA was penetrated into tubules ; ii) The amide I band of collagen at 1660 cm−1 became much broader after plasma treatment. It is well-recognized that amide I band is the most useful information in infrared spectra for the secondary structure of proteins [31, 32]. The helix state was shown at 1660cm−1 while the bands around 1630, 1650 and 1675 cm−1 have been reported as to imide residues partly to β-sheet, random coils and β-turns, respectively[33]. The much broader amide I band at 1660 cm−1 suggested plasma treatment led to loss in the triple helical structure. iii) The collagen swelled more and absorbed more water after plasma treatment. It is known that triple helix structure of collagen could be denatured by heat[34], electrospinning[35], chemical agents[36] et al. It is reasonable to presume that collagen structure arrangement has been partially broken down after plasma treatment. The loosened collagen structure could absorb more water and thus make the collagen fibers swell more as observed in Fig. 5. All the results strongly indicated plasma treatment change the secondary structural of collagen. CD spectra confirmed collagen structure change after plasma treatment, which fully verified our pervious hypothesis. Collagen structure was well-recognized as a helical conformation which was made up of three polypeptide strands through N-H…C=O hydrogen bonds [37]. When electronically activated in plasma state, argon plasmas consist of many high energy species which could break hydrogen bonds, loose structure and expose hydrophilic amino acid inside the collagen helix structure. It helps HEMA to penetrate into tubules and collagen structures which lead to thick hybrid layer and longer resin tags, which has been observed and demonstrated in our previous work [19–21].
Conclusion
It can be concluded that plasma treatment is effective on modifying dentin surfaces and improving monomer penetration into the exposed collagen fibrils and dentin tubules. Plasma treatment could loose collagen helix structure and help monomer penetrate into dentin tubules and collagen networks, form thicker hybrid layer and longer resin tags, which is beneficial to enhancing adhesive-dentin interfacial bonding. Investigation of the interaction between resin monomers and plasma-treated collagen fibers results in a better understanding of plasma treatment effect on improving adhesive-dentin bonding.
Acknowledgements
This study was supported, in part, by the US National Institute of Health (NIH), grant numbers 5R01DE021431 and 5R44DE019041. The authors would thank Drs. Thomas E. Coyle DDS, John A. Johnson DDS, and Timothy T. Coyle DDS, MD from Coyle & Johnson Oral and Maxillofacial Surgery for providing the extracted human teeth.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Lutz F, Krejci I, Oddera M. Advanced adhesive restorations: the post-amalgam age. Pract Periodontics Aesthet Dent. 1996;8:385. [PubMed] [Google Scholar]
- 2.Demarco FF, Corrêa MB, Cenci MS, Moraes RR, Opdam NJM. Longevity of posterior composite restorations: not only a matter of materials. Dent Mater. 2012;28:87–101. doi: 10.1016/j.dental.2011.09.003. [DOI] [PubMed] [Google Scholar]
- 3.Antony K, Genser D, Hiebinger C, Windisch F. Longevity of dental amalgam in comparison to composite materials. GMS Health Technol Assess. 2008;4:Doc12. [PMC free article] [PubMed] [Google Scholar]
- 4.Hannig M, Friedrichs C. Comparative in vivo and in vitro investigation of interfacial bond variability. Oper Dent. 2001;26:3–11. [PubMed] [Google Scholar]
- 5.Tam L, Pilliar R. Effects of dentin surface treatments on the fracture toughness and tensile bond strength of a dentin-composite adhesive interface. J Dent Res. 1994;73:1530–1538. doi: 10.1177/00220345940730090801. [DOI] [PubMed] [Google Scholar]
- 6.Spencer P, Ye Q, Park J, Parthasarathy R, Marangos O, Misra A, et al. Dentin/adhesive interface in teeth. Structural Interfaces and Attachments in Biology. 2013:133–151. [Google Scholar]
- 7.Mollica F, De Santis R, Ambrosio L, Nicolais L, Prisco D, Rengo S. Mechanical and leakage behaviour of the dentin–adhesive interface. J Mater Sci Mater Med. 2004;15:485–492. doi: 10.1023/b:jmsm.0000021125.40282.ba. [DOI] [PubMed] [Google Scholar]
- 8.Van Landuyt KL, Snauwaert J, De Munck J, Peumans M, Yoshida Y, Poitevin A, et al. Systematic review of the chemical composition of contemporary dental adhesives. Biomaterials. 2007;28:3757–3785. doi: 10.1016/j.biomaterials.2007.04.044. [DOI] [PubMed] [Google Scholar]
- 9.Bouillaguet S, Gysi P, Wataha JC, Ciucchi B, Cattani M, Godin C, et al. Bond strength of composite to dentin using conventional, one-step, and self-etching adhesive systems. J Dent. 2001;29:55–61. doi: 10.1016/s0300-5712(00)00049-x. [DOI] [PubMed] [Google Scholar]
- 10.Pashley DH, Carvalho RM. Dentine permeability and dentine adhesion. J Dent. 1997;25:355–372. doi: 10.1016/s0300-5712(96)00057-7. [DOI] [PubMed] [Google Scholar]
- 11.Swift JREJ, Perdigao J, Heymann HO. Bonding to enamel and dentin: a brief history and state of the art, 1995. Quintessence Int. 1995;26:95–110. [PubMed] [Google Scholar]
- 12.Shin TP, Yao X, Huenergardt R, Walker MP, Wang Y. Morphological and chemical characterization of bonding hydrophobic adhesive to dentin using ethanol wet bonding technique. Dent Mater. 2009;25:1050–1057. doi: 10.1016/j.dental.2009.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Eick J, Robinson S, Cobb C, Chappell R, Spencer P. The dentinal surface: its influence on dentinal adhesion. 2. Quintessence Int. 1992;23:43–51. [PubMed] [Google Scholar]
- 14.Fridman G, Friedman G, Gutsol A, Shekhter AB, Vasilets VN, Fridman A. Applied plasma medicine. Plasma Processes Polym. 2008;5:503–533. [Google Scholar]
- 15.Kong MG, Kroesen G, Morfill G, Nosenko T, Shimizu T, Van Dijk J, et al. Plasma medicine: an introductory review. New J Phys. 2009;11:115012(35pp). [Google Scholar]
- 16.Sladek REJ, Stoffels E, Walraven R, Tielbeek PJA, Koolhoven RA. Plasma treatment of dental cavities: a feasibility study. IEEE Trans Plasma Sci. 2004;32:1540–1543. [Google Scholar]
- 17.Sun P, Pan J, Tian Y, Bai N, Wu H, Wang L, et al. Tooth whitening with hydrogen peroxide assisted by a direct-current cold atmospheric-pressure air plasma microjet. IEEE Trans Plasma Sci. 2010;38:1892–1896. [Google Scholar]
- 18.Yang B, Chen J, Yu Q, Li H, Lin M, Mustapha A, et al. Oral bacterial deactivation using a low-temperature atmospheric argon plasma brush. J Dent. 2011;39:48–56. doi: 10.1016/j.jdent.2010.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ritts AC, Li H, Yu Q, Xu C, Yao X, Hong L, et al. Dentin surface treatment using a non-thermal argon plasma brush for interfacial bonding improvement in composite restoration. Eur J Oral Sci. 2010;118:510–516. doi: 10.1111/j.1600-0722.2010.00761.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yu QS, Li H, Ritts AC, Yang B, Chen M, Hong L, et al. Non-thermal atmospheric plasma treatment for deactivation of oral bacteria and improvement of dental composite restoration. In: Machala Z, Hensel K, Akishev Y, editors. Plasma for bio-decontamination, medicine and food security. Netherlands: Springer; 2012. pp. 215–228. [Google Scholar]
- 21.Dong X, Ritts AC, Staller C, Yu Q, Chen M, Wang Y. Evaluation of plasma treatment effects on improving adhesive–dentin bonding by using the same tooth controls and varying cross-sectional surface areas. Eur J Oral Sci. 2013;121:355–362. doi: 10.1111/eos.12052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Spencer P, Byerley T, Eick J, Witt J. Chemical characterization of the dentin/adhesive interface by Fourier transform infrared photoacoustic spectroscopy. Dent Mater. 1992;8:10–15. doi: 10.1016/0109-5641(92)90046-f. [DOI] [PubMed] [Google Scholar]
- 23.Ubaldini ALM, Baesso ML, Sehn E, Sato F, Benetti AR, Pascotto RC. Fourier transform infrared photoacoustic spectroscopy study of physicochemical interaction between human dentin and etch-&-rinse adhesives in a simulated moist bond technique. J Biomed Opt. 2012;17 doi: 10.1117/1.JBO.17.6.065002. 065002-1. [DOI] [PubMed] [Google Scholar]
- 24.Yoshida Y, Van Meerbeek B, Nakayama Y, Snauwaert J, Hellemans L, Lambrechts P, et al. Evidence of chemical bonding at biomaterial-hard tissue interfaces. J Dent Res. 2000;79:709–714. doi: 10.1177/00220345000790020301. [DOI] [PubMed] [Google Scholar]
- 25.Van Stokkum IHM, Spoelder HJW, Bloemendal M, Van Grondelle R, Groen FCA. Estimation of protein secondary structure and error analysis from circular dichroism spectra. Anal Biochem. 1990;191:110–118. doi: 10.1016/0003-2697(90)90396-q. [DOI] [PubMed] [Google Scholar]
- 26.Whitmore L, Wallace BA. Protein secondary structure analyses from circular dichroism spectroscopy: methods and reference databases. Biopolymers. 2008;89:392–400. doi: 10.1002/bip.20853. [DOI] [PubMed] [Google Scholar]
- 27.Holmgren SK, Taylor KM, Bretscher LE, Raines RT. Code for collagen's stability deciphered. Nature. 1998;392:666–667. doi: 10.1038/33573. [DOI] [PubMed] [Google Scholar]
- 28.Marshall GW, Marshall SJ, Kinney JH, Balooch M. The dentin substrate: structure and properties related to bonding. J Dent. 1997;25:441–458. doi: 10.1016/s0300-5712(96)00065-6. [DOI] [PubMed] [Google Scholar]
- 29.Nakabayashi N, Takarada K. Effect of HEMA on bonding to dentin. Dent Mater. 1992;8:125–130. doi: 10.1016/0109-5641(92)90067-m. [DOI] [PubMed] [Google Scholar]
- 30.Lehmann A, Rueppell A, Schindler A, Zylla IM, Seifert HJ, Nothdurft F, et al. Modification of enamel and dentin surfaces by non-thermal atmospheric plasma. Plasma Processes Polym. 2013;10:262–270. [Google Scholar]
- 31.Surewicz WK, Mantsch HH. New insight into protein secondary structure from resolution-enhanced infrared spectra. Biochim Biophys Acta. 1988;952:115–130. doi: 10.1016/0167-4838(88)90107-0. [DOI] [PubMed] [Google Scholar]
- 32.Muyonga J, Cole C, Duodu K. Fourier transform infrared (FTIR) spectroscopic study of acid soluble collagen and gelatin from skins and bones of young and adult Nile perch (Lates niloticus) Food Chem. 2004;86:325–332. [Google Scholar]
- 33.Payne K, Veis A. Fourier transform IR spectroscopy of collagen and gelatin solutions: deconvolution of the amide I band for conformational studies. Biopolymers. 1988;27:1749–1760. doi: 10.1002/bip.360271105. [DOI] [PubMed] [Google Scholar]
- 34.Leikina E, Mertts M, Kuznetsova N, Leikin S. Type I collagen is thermally unstable at body temperature. Proc Natl Acad Sci. 2002;99:1314–1318. doi: 10.1073/pnas.032307099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang L, Fitié CFC, van der Werf KO, Bennink ML, Dijkstra PJ, Feijen J. Mechanical properties of single electrospun collagen type I fibers. Biomaterials. 2008;29:955–962. doi: 10.1016/j.biomaterials.2007.10.058. [DOI] [PubMed] [Google Scholar]
- 36.Usha R, Ramasami T. The effects of urea and n-propanol on collagen denaturation: using DSC, circular dicroism and viscosity. Thermochim Acta. 2004;409:201–206. [Google Scholar]
- 37.Shoulders MD, Raines RT. Collagen structure and stability. Annu Rev Biochem. 2009;78:929–958. doi: 10.1146/annurev.biochem.77.032207.120833. [DOI] [PMC free article] [PubMed] [Google Scholar]