Abstract
Background
Restricting fluids and foods during labour is common practice across many birth settings with some women only being allowed sips of water or ice chips. Restriction of oral intake may be unpleasant for some women, and may adversely influence their experience of labour.
Objectives
To determine the benefits and harms of oral fluid or food restriction during labour.
Search methods
We searched the Cochrane Pregnancy and Childbirth Group’s Trials Register (April 2009).
Selection criteria
Randomised controlled trials (RCTs) and quasi-RCTs of restricting fluids and food for women in labour compared with women free to eat and drink.
Data collection and analysis
Two authors independently assessed the studies for inclusion, assessed risk of bias and carried out data extraction.
Main results
We identified five studies (3130 women). All studies looked at women in active labour and at low risk of potentially requiring a general anaesthetic. One study looked at complete restriction versus giving women the freedom to eat and drink at will; two studies looked at water only versus giving women specific fluids and foods and two studies looked at water only versus giving women carbohydrate drinks.
When comparing any restriction of fluids and food versus women given some nutrition in labour, the meta-analysis was dominated by one study undertaken in a highly medicalised environment. There were no statistically significant differences identified in: caesarean section (average risk ratio (RR) 0.89, 95% confidence interval (CI) 0.63 to 1.25, five studies, 3103 women), operative vaginal births (average RR 0.98, 95% CI 0.88 to 1.10, five studies, 3103 women) and Apgar scores less than seven at five minutes (average RR 1.43, 95% CI 0.77 to 2.68, three studies, 2574 infants), nor in any of the other outcomes assessed. Women’s views were not assessed. The pooled data were insufficient to assess the incidence of Mendelson’s syndrome, an extremely rare outcome. Other comparisons showed similar findings, except one study did report a significant increase in caesarean sections for women taking carbohydrate drinks in labour compared with water only, but these results should be interpreted with caution as the sample size was small.
Authors’ conclusions
Since the evidence shows no benefits or harms, there is no justification for the restriction of fluids and food in labour for women at low risk of complications. No studies looked specifically at women at increased risk of complications, hence there is no evidence to support restrictions in this group of women. Conflicting evidence on carbohydrate solutions means further studies are needed and it is critical in any future studies to assess women’s views.
Medical Subject Headings (MeSH): *Drinking; *Fasting [adverse effects]; *Labor, Obstetric; Beverages [adverse effects]; Dietary Carbohydrates [administration & dosage; adverse effects]; Randomized Controlled Trials as Topic
MeSH check words: Female, Humans, Pregnancy
BACKGROUND
Historically, in some societies, special food and drinks were offered during labour and specific foods were sometimes discouraged (Broach 1988a; McCormick 2002). In the early 1900s, Dr DeLee, an American obstetrician, recommended that women take liquids to preserve their strength during labour (Broach 1988a). However, by the 1940s views had changed, and practitioners believed that eating and drinking in labour might be dangerous. Work by Mendelson in the 1940s showed high morbidity and mortality in women under general anaesthesia for caesarean section who inhaled liquids and particles of food from the stomach (Mendelson 1946). This led to common policies of food and fluid restrictions during labour (Champion 2002). Although many women may not feel like eating in labour (McNabb 2002), some women find this restriction unpleasant and sometimes harrowing (Armstrong 2000; Johnson 1989). The importance of this aspect of the woman’s autonomy, choice and control in labour should also be a consideration (Pengelley 2002).
A survey of labour ward policies in England in 1985 revealed that over a third of consultant maternity units allowed no fluids whatsoever during labour (Garcia 1985). In the United States, a survey in 1988 showed that almost half the hospitals and birth centres had a policy of ‘nil by mouth’ except ice chips (McKay 1988). A later survey of 351 units in England and Wales found that one-third allowed some form of food and drink and more than ninety per cent allowed some form of oral intake, usually water (Michael 1991). More recently, in 2003, a survey in the UK showed a liberalising trend, with 47% of women having ‘access’ to food and drink in labour (Hart 2006).
Description of the condition
Fluid and nutrient needs during labour are not well studied (Micklewright 2002). The need for energy is increased during pregnancy and labour, and metabolic processes generally adjust to address these needs (Pipkin 2001). Many believe that elevated levels of ketone bodies, which accumulate during exercise or starvation (Williamson 1971), and at times during labour are a physiological response with little clinical significance. However, associations between ketone levels and longer labours and maternal psychological stress have been reported (Chang 1993; Foulkes 1985). It is difficult to determine whether ketone production contributes to the longer labour or whether such production is a consequence of longer labour. The presence of ketonuria should be considered a signal for metabolic imbalance, though the effect of the imbalance is not known (Johnson 1989).
Description of the intervention
People take for granted the ability to eat and drink in response to hunger and thirst. Hence, withholding all food and fluids, or withholding all food but allowing only sips of water, from women during labour is an ‘intervention’. This intervention is generally used during active labour when women are in hospital, and many women in early labour will eat and drink at will at home. Some hospitals prescribe specific foods which are ‘allowed’ in labour, but this is also considered an intervention since it limits a women’s autonomy and freedom of choice.
How the intervention might work
The rationale for withholding food and fluid during labour is to decrease the risk of maternal morbidity and mortality from Mendelson’s syndrome if a general anaesthetic is required (Mendelson 1946). It was thought that fasting would ensure small gastric volumes, but recent reviews suggest that there is no evidence to support this belief (Micklewright 2002; O’Sullivan 1994). Interventions to reduce stomach contents or the acidity of the content, both by pharmacological means and by restriction of oral intake, have not proved successful (Gyte 2006; Taylor 1975). Also gastric emptying is delayed during labour, which may contribute to the problem (Davidson 1975). Irrespective of whether a woman has been starved or not during labour, anaesthetic precautions are necessary to reduce the risk of gastric content aspiration. These include using regional rather than general anaesthesia, and using rapid sequence induction with airway protection for general anaesthesia (Am Soc Anesth 1999). With modern techniques, particularly the use of regional analgesia, the risk of gastric content aspiration has become extremely small (McKay 1988).
Intravenous therapy instead of oral hydration is common practice during labour. Historically, practitioners administered high-dose glucose solutions to combat the development of ketones (Ketteringham 1939). More commonly now, intravenous fluids are isotonic or low-dose glucose, as high-dose glucose solutions are associated with increased incidence of neonatal hypoglycaemia (low blood sugar levels) (Grylack 1984; Mendiola 1982). Dextrose-only solutions cause a fall in serum osmolality and sodium concentration (hyponatraemia) (Begum 1999). Regardless of solution type, intravenous therapy predisposes women to immobilisation, stress, increased risk of fluid overload, and does not ensure a nutrient and fluid balance for the demands of labour (Simkin 1986a; Simkin 1986b). The value and safety of routine intravenous fluid therapy has been questioned (Begum 1999).
Why it is important to do this review
Restricting oral food and fluid intake of women in active labour in hospitals is a strongly held obstetric and anaesthetic tradition. Restriction of oral intake is not a common practice in home births or birth centres (Rooks 1989), nor is the practice consistent across hospital sites (Haire 1991). Few, if any, centres have policies that are reflective of women’s preferences (Pengelley 1998). Most are based on historical, but important concerns, related to the risks of gastric content regurgitation and aspiration into the lungs during general anaesthesia, a risk first identified by Mendelson in the 1940s. The incidence is very rare with modern anaesthetic techniques and the use of regional anaesthesia rather then general anaesthesia. However, the syndrome is potentially fatal.
The risks of regurgitation and Mendelson’s syndrome exist, although the incidence of each is very rare. The policy of routine restriction of foods and fluids in labour in many hospitals across the world generally does not reflect women’s preferences or cultural expectations (Broach 1988a; Broach 1988b). It is critical that any policy should be based on evidence of overall benefit to women and babies. This systematic review may assist in resolving the clinical uncertainty, which is currently apparent.
OBJECTIVES
To determine the benefits and harms of oral fluid and food restriction during labour, with or without intravenous hydration.
METHODS
Criteria for considering studies for this review
Types of studies
We included randomised controlled clinical trials (RCTs) and quasi-RCTs, although we identified no quasi-RCTs. Had we identified quasi-RCTs we would have undertaken sensitivity analysis by study quality.
Types of participants
Women in labour.
Types of interventions
We considered studies comparing any two or more of the following regimens for inclusion.
Complete restriction of oral food and fluids (other than ice chips).
Allowing only water.
Allowing only oral carbohydrate based fluids.
Allowing particular oral food and fluid regimens.
Freedom to take oral food and/or fluids at will.
We then assessed each restrictive regimen (interventions one to four above). In each case, we regarded the more restrictive regimen as the experimental intervention.
We excluded studies on intravenous feeding in labour unless being given on a clinical need within a study on oral fluids and food.
Types of outcome measures
We divided outcomes into maternal and fetal categories. For relative outcomes (e.g. ketoacidosis, dehydration, hyponatraemia, hypoglycaemia, maternal satisfaction, etc), we used the definitions chosen by the trial authors to categorise outcomes.
Primary outcomes
Maternal
Caesarean section.
Operative vaginal birth.
Maternal satisfaction.
Fetal
Five-minute Apgar score less than seven.
Hypoglycaemia.
Secondary outcomes
Maternal
Ketoacidosis.
Dehydration.
Hyponatraemia.
Hypoglycaemia.
Duration of labour.
Mobility in labour.
Nausea and vomiting.
Labour augmentation.
Narcotic pain relief.
Epidural analgesia.
Poor expulsive efforts.
Regurgitation during general anaesthesia.
Mendelson’s syndrome.
Maternal mortality.
Postpartum haemorrhage.
Admission to intensive care.
Length of hospital stay; maternal comfort.
Feelings of pain, thirst, hunger.
Breastfeeding success.
Personal control.
Fetal
Fetal distress.
Cord blood pH less than 7.2.
Hyperinsulinisim.
Hyponatraemia.
Intravenous therapy.
Gavage feeding.
Admission to intensive care.
Length of hospital stay.
We are aware that RCTs are unlikely to be able to adequately address rare adverse outcomes, like Mendelson’s syndrome and maternal mortality in this review, and so we have chosen outcomes which we think might be impacted on, for benefit or harm, by restriction of fluids and foods in labour.
Search methods for identification of studies
Electronic searches
We searched the Cochrane Pregnancy and Childbirth Group’s Trials Register by contacting the Trials Search Co-ordinator (April 2009).
The Cochrane Pregnancy and Childbirth Group’s Trials Register is maintained by the Trials Search Co-ordinator and contains trials identified from:
quarterly searches of the Cochrane Central Register of Controlled Trials (CENTRAL);
weekly searches of MEDLINE;
handsearches of 30 journals and the proceedings of major conferences;
weekly current awareness alerts for a further 44 journals plus monthly BioMed Central email alerts.
Details of the search strategies for CENTRAL and MEDLINE, the list of handsearched journals and conference proceedings, and the list of journals reviewed via the current awareness service can be found in the ‘Specialized Register’ section within the editorial information about the Cochrane Pregnancy and Childbirth Group.
Trials identified through the searching activities described above are each assigned to a review topic (or topics). The Trials Search Co-ordinator searches the register for each review using the topic list rather than keywords.
Searching other resources
We searched the reference list for each study identified for any additional studies.
We did not apply any language restrictions.
Data collection and analysis
Selection of studies
Two review authors have independently assessed for inclusion all the potential studies we identified as a result of the search strategy. We have resolved any disagreement through discussion, or if required we have consulted the third author.
Data extraction and management
We designed a form to extract data. At least two review authors have extracted the data using the agreed form. We resolved discrepancies through discussion, or if required we consulted the third author. We entered data into Review Manager software (RevMan 2008) (all or a subsample) and checked for accuracy.
When information regarding any of the above was unclear, we have attempted to contact authors of the original reports to provide further details.
Assessment of risk of bias in included studies
Two review authors have independently assessed risk of bias for each study using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2008). We have resolved any disagreements by discussion or by involving the third author.
1) Sequence generation (checking for possible selection bias)
We have described for each included study the methods used to generate the allocation sequence in sufficient detail to allow an assessment of whether it should have produced comparable groups. We have assessed the methods used as:
adequate (e.g. random number table; computer random-number generator; tossing a coin, minimisation);
inadequate (e.g. odd or even date of birth; hospital or clinic record number); or
unclear.
2) Allocation concealment (checking for possible selection bias)
We have described for each included study the method used to conceal the allocation sequence in sufficient detail and determined whether intervention allocation could have been foreseen in advance of, or during, recruitment.
We have assessed the methods as:
adequate (e.g. telephone or central randomisation; consecutively numbered sealed opaque envelopes);
inadequate (open random allocation; unsealed or non-opaque envelopes, alternation; date of birth);
unclear.
3) Blinding (checking for possible performance bias)
We have described for each included study all the methods used, if any, to blind study participants and personnel from knowledge of which intervention a participant received. We have also provided any information relating to whether the intended blinding was effective. Where blinding was not possible we have assessed whether the lack of blinding was likely to have introduced bias. We have assessed the methods as:
adequate, inadequate or unclear for participants;
adequate, inadequate or unclear for personnel;
adequate, inadequate or unclear for outcome assess.
4) Incomplete outcome data (checking for possible attrition bias through withdrawals, dropouts, protocol deviations)
We have described for each included study the completeness of outcome data for each main outcome, including attrition and exclusions from the analysis. We have stated whether attrition and exclusions were reported, the numbers (compared with the total randomised participants), reasons for attrition/exclusion where reported, and any re-inclusions in analyses which we have undertaken.
We have assessed the methods as:
adequate (e.g. where there was no missing data or where reasons for missing data are balanced across groups);
inadequate (e.g. where missing data are likely to be related to outcomes or are not balanced across groups);
unclear (e.g. where there is insufficient reporting of attrition or exclusions to permit a judgement to me made).
5) Selective reporting bias
We have described for each included study how the possibility of selective outcome reporting bias was examined by us and what we found.
We have assessed the methods as:
adequate (where it is clear that all of the study’s prespecified outcomes and all expected outcomes of interest to the review have been reported);
inadequate (where not all the study’s pre-specified outcomes have been reported; one or more reported primary outcomes were not pre-specified; outcomes of interest are reported incompletely and so cannot be used; study fails to include results of a key outcome that would have been expected to have been reported);
unclear.
6) Other sources of bias
We have described for each included study any important concerns we had about other possible sources of bias. For example, was there a potential source of bias related to the specific study design? Was the trial stopped early due to some data-dependent process? Was there extreme baseline imbalance? Has the study been claimed to be fraudulent?
We have assessed whether each study was free of other problems that could put it at risk of bias:
yes;
no;
unclear.
7) Overall risk of bias
We have made explicit judgements about risk of bias for important outcomes both within and across studies. With reference to (1) to (6) above, we have assessed the likely magnitude and direction of the bias and whether we have considered it is likely to impact on the findings. We have explored the impact of the level of bias through undertaking sensitivity analyses; see ‘Sensitivity analysis’.
Measures of treatment effect
Dichotomous data
For dichotomous data, we have presented results as summary risk ratio (RR) with 95% confidence intervals.
Continuous data
For continuous data, we have used the mean difference (MD) if outcomes were measured in the same way between trials. We have used the standardised mean difference (SMD) to combine trials that measure the same outcome, but used different methods.
Unit of analysis issues
Cluster-randomised trials
We found no cluster-randomised trials, but had we done so we would have included them in the analyses along with individually randomised trials. Their sample sizes would have been adjusted using the methods described in Gates 2005 using an estimate of the intracluster correlation co-efficient (ICC) derived from the trial (if possible), or from another source. We would also have performed the meta-analysis in two parts.
Dealing with missing data
For included studies, we have noted the levels of attrition. We would have explored the impact of including studies with high levels of missing data in the overall assessment of treatment effect by using sensitivity analysis, had the issue arisen.
Where data have not been reported for some outcomes or groups, we have attempted to contact the study authors for further information.
Assessment of heterogeneity
We have assessed statistical heterogeneity in each meta-analysis using the T2, I2 and Chi2 statistics. We have regarded heterogeneity as substantial if T2 was greater than zero and either I2 was greater than 30% or there was a low P-value (less than 0.10) in the Chi2 test for heterogeneity.
Assessment of reporting biases
Where we might have suspected reporting bias (see ‘Selective reporting bias’ above), we would have attempted to contact study authors asking them to provide missing outcome data. Where this was not possible, and we thought the missing data could introduce serious bias, we would have explored the impact of including such studies in the overall assessment of results by a sensitivity analysis. Had there been 10 or more studies in a meta-analysis, we would have investigated reporting biases (such as publication bias) using funnel plots. We would have assessed funnel plot asymmetry visually, and used formal tests for funnel plot asymmetry. For continuous outcomes, we would have used the test proposed by Egger 1997, and for dichotomous outcomes we would have used the tests proposed by Harbord 2006 or Peters 2006. If asymmetry had been detected by any of these tests or had been suggested by a visual assessment, we would have performed exploratory analyses to investigate it.
Intention-to-treat analysis
We have analysed data on all participants with available data in the group to which they were allocated, regardless of whether or not they received the allocated intervention. If in the original reports participants were not analysed in the group to which they were randomised, and there was sufficient information in the trial report, we have attempted to restore them to the correct group.
Incomplete outcome data (attrition and exclusions)
See ‘Assessment of risk of bias in included studies’ section above.
Selective outcome reporting bias
See ‘Assessment of risk of bias in included studies’ section above.
Data synthesis
We have carried out statistical analysis using the Review Manager software (RevMan 2008). We have used fixed-effect meta-analysis for combining data where it was reasonable to assume that studies were estimating the same underlying treatment effect: i.e. where trials were examining the same intervention, and the trials’ populations and methods were judged sufficiently similar. If there was clinical heterogeneity sufficient to expect that the underlying treatment effects differed between trials, or if substantial statistical heterogeneity was detected, we have used random-effects analysis to produce an overall summary, if this was considered clinically meaningful. If an average treatment effect across trials had not been clinically meaningful, we would not have combined the heterogeneous trials. If we used random-effects analyses, we have presented the results as the average treatment effect with its 95% confidence interval.
Subgroup analysis and investigation of heterogeneity
Had there been sufficient data, we would have conducted planned subgroup analyses classifying whole trials by interaction tests as described by Deeks 2001.
We planned to carry out the following subgroup analyses.
Women at low risk of potentially requiring general anaesthesia versus women at high risk of potentially requiring general anaesthesia. We looked at all outcomes for this subgroup analysis as we believed this to be such an important consideration.
Routine administration of intravenous (IV) fluids in the restricted group versus no routine administration of IV fluids in the restricted group. We restricted these analyses to primary outcomes only.
We were unable to undertake the subgroup analysis comparing the administration of IV fluids as the data did not exist. We will restrict any future subgroup analyses to primary outcomes only.
Sensitivity analysis
We have carried out sensitivity analysis to explore the effect of trial quality for primary outcomes in the review. Where there was risk of bias associated with a particular aspect of study quality (e.g. inadequate allocation concealment), we have explored this by sensitivity analysis.
RESULTS
Description of studies
See: Characteristics of included studies; Characteristics of excluded studies; Characteristics of studies awaiting classification.
Results of the search
The search identified 22 publications covering 11 studies. Of these, we have included five studies involving 3130 women in this review (Kubli 2002; O’Sullivan 2009; Scheepers 2002; Scrutton 1999; Tranmer 2005), excluded two studies (Scheepers 2004; Shennan 2005 ) and assigned four studies as awaiting classification; we have attempted to contact these authors (Goodall 1999; Laifer 2000; Yiannouzis 1994; Zhao 1996).
Included studies
All of the included studies involved women considered at low risk of potentially requiring general anaesthesia (for example, for caesarean section). No studies looked specifically at women considered at increased risk of general anaesthesia.
Of the five included studies, only one study looked at restricting women to only ice chips or sips of water compared with giving women the freedom to eat and drink what they wished during labour. The women who were able to choose what they ate and drank in labour did have some guidance antenatally with easy-to-read guidelines on suggested nutrition and fluid intake during labour based on nutritional guidelines for individuals who participate in prolonged, moderate aerobic exercise (Tranmer 2005). Two included studies looked at restricting women to drinking water only compared with giving women specific foods during labour and encouraging them to take some food and fluids in active labour (O’Sullivan 2009; Scrutton 1999). Two studies looked at giving women water only compared with carbohydrate drinks in labour (Kubli 2002; Scheepers 2002).
Excluded studies
We excluded one study because it was a discussion paper (Shennan 2005) and the other because it looked at giving a high glucose load once women reached 8 to 10 cm dilatation (Scheepers 2004).
Risk of bias in included studies
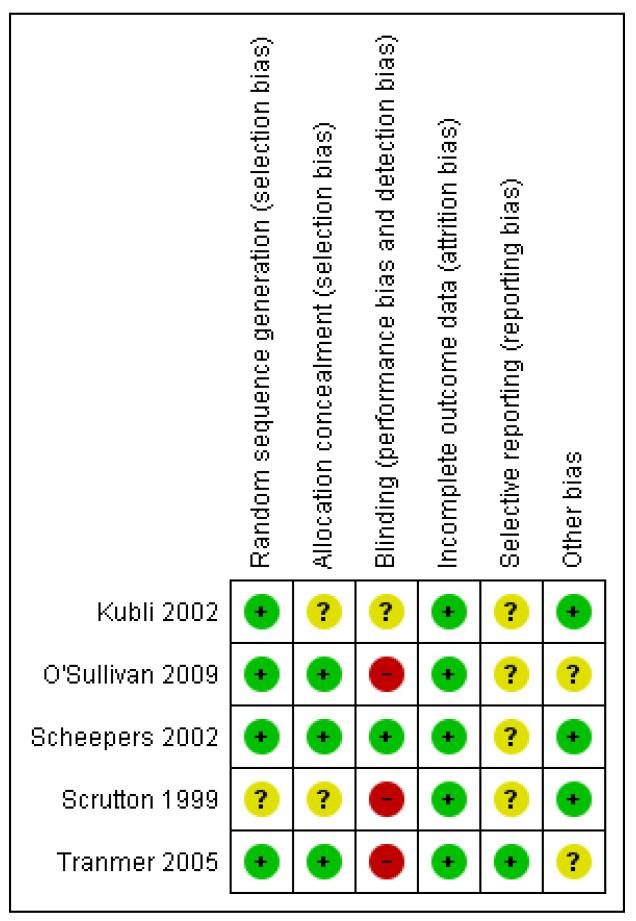
The studies were of reasonable overall quality although some aspects, like selective reporting bias, were unclear. However, the review is dominated by one large trial of reasonable overall quality involving 2443 women (O’Sullivan 2009), while the other four studies together involve just 687 women (Kubli 2002; Scheepers 2002; Scrutton 1999; Tranmer 2005). For the Methodological quality summary, see Figure 1.
Figure 1.
Methodological quality summary: review authors’ judgements about each methodological quality item for each included study.
Allocation
Three studies had adequate sequence generation and allocation concealment (O’Sullivan 2009; Scheepers 2002; Tranmer 2005). One study had adequate sequence generation but allocation concealment was unclear (Kubli 2002) and one study was unclear for both adequate sequence generation but allocation concealment (Scrutton 1999).
Blinding
The only study that blinded the women and the clinicians was the study comparing carbohydrate fluids with coloured water (Scheepers 2002). Studies that looked at women in labour eating and drinking compared with women having ice chips or water only were not able to blind women or clinicians, and hence some outcomes may be subject to bias.
Incomplete outcome data
We considered all studies to have complete outcome data reported. There were a few exclusions but these were very small compared with the overall numbers of women involved (see ‘Characteristics of included studies’).
Selective reporting
The assessment of selective reporting bias was considered adequate for the one study where we were able to assess the trial protocol as reported in the PhD thesis (Tranmer 2005). We assessed the other studies as unclear on selective reporting bias, as we were unable to assess the trial protocols.
Other potential sources of bias
Three studies were assessed as free of other sources of bias (Kubli 2002; Scheepers 2002; Scrutton 1999). For the other studies we assessed this aspect as unclear.
Effects of interventions
1. Any restriction of oral fluid and food versus some eating and drinking (five studies, 3130 women)
Five studies assessed this comparison (Kubli 2002; O’Sullivan 2009; Scheepers 2002; Scrutton 1999; Tranmer 2005). The overall quality of the studies was reasonable with three studies having adequate randomisation sequence generation, allocation concealment and no exclusions of loss to follow up (O’Sullivan 2009; Scheepers 2002; Tranmer 2005). The large size of the O’Sullivan study in comparison with the size of the other studies means that for any outcome that included data from the O’Sullivan study will be dominated by this data (O’Sullivan 2009). This study was undertaken in a highly medicalised environment where women entering the study were considered to have no known obstetric or medical complication that would increase the likelihood of an operative birth, yet 30% of women in the trial had a caesarean section. Other interventions, including oxytocin augmentation, IV fluids, epidurals, caesarean sections and instrumental births, also showed high rates in this study (Table 1).
Table 1.
Interventions during labour in women at low risk in O’Sullivan study
| Intervention | Water only | Food in labour |
|---|---|---|
| Oxytocin augmentation | 56% (673/1207) | 53% (647/1219) |
| IV fluids in labour | 69% (838/1207) | 67% (820/1219) |
| Epidural | 67% (813/1207) | 66% (804/1219) |
| Caesarean section | 30% (363/1207) | 30% (362/1219) |
| Operative vaginal birth | 27% (310/1207) | 27% (324/1219) |
IV: intravenous
Since in this comparison we were pooling a range of restrictive practices and a range on comparison forms of care, we considered we had clinical heterogeneity. We, therefore, used the random-effects model of meta-analyses as we did not expect a single common (or ‘fixed’) effect underling every study in the meta-analysis. Rather, we assumed that the individual studies were estimating different treatment effects and that the different effects had a distribution with some central value and some degree of variability.
Primary outcomes
Reporting all the meta-analyses as random effects, we identified no significant differences in:
caesarean section (average risk ratio (RR) 0.89, 95% confidence interval (CI) 0.63 to 1.25, five studies, 3103 women (T2 = 0.07, Chi2P = 0.05, I2 = 57%), graph 1.1);
operative vaginal birth (average RR 0.98, 95% CI 0.88 to 1.10, five studies, 3103 women (T2 = 0.00, Chi2P = 0.67, I2 = 0%), graph 1.2);
Apgar scores less than seven at five minutes (average RR 1.43, 95% CI 0.77 to 2.68, four studies, 2902 infants (T2 = 0.00, Chi2 P = 0.64, I2 = 0%), graph 1.4).
None of the studies assessed maternal satisfaction or infant hypoglycaemia.
Secondary outcomes
Reporting all the meta-analyses as random effects, we identified no significant differences in:
maternal ketosis (average RR 0.99, 95% CI 0.66 to 1.49, one study, 328 women, Graph 1.6);
duration of labour (average mean difference (MD) −0.29, 95% CI −1.55 to 0.97, three studies, 476 women (T2 = 0.72, Chi2 P = 0.09, I2 = 58%), graph 1.10);
maternal nausea (average RR 0.80, 95% CI 0.54 to 1.18, one study, 255 women, graph 1.12);
maternal vomiting (average RR 0.90, 95% CI 0.90 to 1.31, three studies, 2574 women, (T2 = 0.06, Chi2 P = 0.13, I2 = 50%), graph 1.13);
augmentation of labour (average RR 1.02, 95% CI 0.95 to 1.09, five studies, 3103 women (T2 = 0.00, Chi2 P = 0.39, I2 = 3%), graph 1.14);
narcotic pain relief (average RR 0.94, 95% CI 0.74 to 1.21, three studies, 349 women (T2 = 0.04, Chi2 P = 0.0002, I2 = 88%), graph 1.15);
epidural analgesia (average RR 0.98, 95% CI 0.91 to 1.05, five studies, 3103 women (T2 = 0.00, Chi2 P = 0.22, I2 =30%), graph 1.16);
infant admission to intensive care (average RR 1.03, 95% CI 0.73 to 1.45, one study, 2426 women, graph 1.34).
Regurgitation during anaesthesia and Mendelson’s syndrome were not estimable (graph 1.18 and graph 1.19) and other secondary outcomes were not assessed in the included studies.
Although we have chosen to use a random-effects model throughout this comparison, for some outcomes there appeared to be no statistical heterogeneity (e.g. for 1.2 Operative vaginal delivery and 1.4 Apgar score less than seven at five minutes, for both I2 = 0% and Tau2 = 0).
2. Complete restriction of oral fluid and food (other than ice chips) versus freedom to eat and drink (one study, 330 women)
There was only one study that looked at complete restrictions to fluid and food (ice chips only or sips of water only) intake compared with women having the freedom to eat and drink at will. Although there were discussions antenatally about suggested nutrient and fluid intake during labour based on nutritional guidelines for individuals who participate in exercise (prolonged, moderate, aerobic), the women were encouraged to bring to the hospital their own food and drink choices and caregivers did not actively encourage or discourage oral intake in the group able to eat and drink (Tranmer 2005).
The quality of the study was good overall, with adequate sequence generation, allocation concealment and no exclusions nor loss of data (see Figure 1).
Primary outcomes
There were no significant differences identified in:
caesarean sections (RR 0.77, 95% CI 0.51 to 1.16, one study, 328 women, graph 2.1);
operative vaginal births (RR 0.99, 95% CI 0.72 to 1.35, one study, 328 women, graph 2.2).
There were no infants with Apgar scores less than seven at five minutes, and the other primary outcomes of maternal satisfaction and neonatal hypoglycaemia were not assessed in the included study.
Secondary outcomes
There were no significant differences identified in:
maternal ketosis (RR 0.99, 95% CI 0.66 to 1.49, one study, 328 women, graph 2.6);
duration of labour (RR MD −0.80, 95% CI −2.13 to 0.53, one study, 328 women, graph 2.10);
maternal nausea (RR 0.80, 95% CI 0.54 to 1.18, one study, 328 women, graph 2.12);
augmentation of labour (RR 00.98, 95% CI 0.81 to 1.18, one study, 328 women, graph 2.14);
epidural (RR 0.92, 95% Ci 0.81 to 1.04, one study, 328 women, graph 2.16).
Mendelson’s syndrome was not estimable, and the other secondary outcomes were not assessed in this one included study.
3. Water only versus freedom to eat and drink (no studies)
There were no studies identified looking at this comparison.
4. Oral carbohydrate based fluids versus freedom to eat and drink (no studies)
There were no studies identified looking at this comparison.
5. Specific oral fluid and food versus freedom to eat and drink (no studies)
There were no studies identified looking at this comparison.
6. Complete restriction of oral fluid and food (other than ice chips versus specific oral foods and fluids (no studies)
There were no studies identified looking at this comparison.
7. Water only versus particular oral fluid and food (two studies, 2520 women)
Two studies looked at the comparison of restricting women to taking only water during active labour compared with advising and encouraging women to take low-residue foods in labour (O’Sullivan 2009; Scrutton 1999). Neither study was able to blind women or clinicians and this may have impacted on the assessment of some outcomes. The large O’Sullivan study (2426 women) had adequate randomisation sequence generation, allocation concealment and had no significant exclusions or loss of data (O’Sullivan 2009). The other small study (94 women) was of unclear quality as there was insufficient information available to assess adequately (Scrutton 1999).
Primary outcomes
There were no significant differences identified in:
caesarean section (RR 1.02, 95% CI 0.91 to 1.15, two studies, 2514 women (T2 = 0.00, Chi2 P = 0.41, I2 = 0%), graph 7.1);
operative vaginal birth (RR 0.96, 95% CI 0.84 to 1.10, two studies, 2514 women (T2 = 0.00, Chi2 P = 0.68, I2 = 0%), graph 7.2);
Apgar scores on less than seven at five minutes (RR 1.39, 95% CI 0.73 to 2.63, two studies (although only one was estimable), 2514 women, graph 7.4).
The other primary outcomes of maternal satisfaction and neonatal hypoglycaemia were not assessed in these included studies.
Secondary outcomes
There were no significant differences identified in:
duration of labour (MD −1.10, 95% CI −2.66 to 0.46, one study, 88 women, graph 7.10);
maternal vomiting (average RR 0.76, 95% CI 0.41 to 1.41, two studies, 2514 women, random-effects (T2 = 0.15, Chi2 P = 0.08, I2 = 68%), graph 7.13);
augmentation of labour (average RR 0.97, 95% CI 0.80 to 1.19, two studies, 2514 women, random-effects (T2 = 0.01, Chi2 P = 0.08, I2 = 67%), graph 7.14);
narcotic pain relief (RR 1.00, 95% CI 0.91 to 1.09, one study, 88 women, graph 7.15);
epidural analgesia (RR 1.02, 95% CI 0.97 to 1.08, two studies, 2514 women (T2 = 0.00, Chi2 P = 0.99, I2 = 0%), graph 7.16);
infant admission to intensive care (RR 1.03, 95% CI 0.73 to 1.45, one study, 2426 women, graph 7.34).
Regurgitation during anaesthesia and Mendelson’s syndrome were not estimable (graph 7.18 and graph 7.19) and other secondary outcomes were not assessed in the included studies.
8. Oral carbohydrate based fluids versus specific oral fluid and food (no studies)
There were no studies identified looking at this comparison.
9. Complete restriction of fluid and food (other than ice chips) versus oral carbohydrate based fluids (no studies)
There were no studies identified looking at this comparison.
10. Water only versus oral carbohydrate based fluids (two studies, 263 women)
Two studies looked at restricting women to water only compared with giving them carbohydrate drinks (Kubli 2002; Scheepers 2002). We assessed the larger study (203 women of mixed risk) to be of good quality with only the selective reporting bias being unclear (Scheepers 2002). We assessed the smaller study (60 women of low risk) as unclear for allocation concealment, blinding and selective reporting boas (Kubli 2002).
Primary outcomes
There were no significant differences identified in:
caesarean section (average RR 0.66, 95% CI 0.17 to 2.53, two studies, 261 women, random-effects (T2 = 0.74, Chi2 P = 0.03, I2 = 79%), graph 10.1);
operative vaginal birth (RR 1.17, 95% CI 0.80 to 1.71, two studies, 261 women, T2 = 0.02, Chi2 P = 0.30, I2 = 8%, graph 10.2);
Apgar scores less than seven at five minutes (RR 3.0, 95% CI 0.13 to 70.83, one study, 60 women, graph 10.4).
The other primary outcomes of maternal satisfaction and neonatal hypoglycaemia were not assessed in these studies.
Secondary outcomes
There were no significant differences identified in:
duration of labour (MD 0.95, 95% CI −0.42 to 2.32, one study, 60 women, graph 10.10);
maternal vomiting (RR 1.27. 95% CI 0.69 to 2.33, one study, 60 women, graph 10.13);
augmentation of labour (RR 1.07, 95% CI 0.75 to 1.52, 2 studies, 261 women (T2 = 0.00, Chi2 P = 0.36, I2 = 0%), graph 10.14);
narcotic pain relief (average RR 0.86, 95% CI 0.36 to 2.06, two studies, 261 women, random-effects (T2 = 0.38, Chi2 P = 0.00001, I2 = 95%), graph 10.15);
epidural analgesia (average RR 0.80, 95% CI 0.44 to 1.43, two studies, 261 women, random-effects (T2 = 0.14, Chi2 P = 0.04, I2 = 76%), graph 10.16).
The other secondary outcomes were not assessed in the included studies.
DISCUSSION
Summary of main results
This review includes five studies involving 3130 women. There are four comparisons and 36 meta-analyses.
For women considered at low risk of potentially needing general anaesthesia
We identified no benefits or risks of restricting oral fluids and foods in labour, in women at low risk of potentially requiring general anaesthesia in this review. In particular, in none of the primary outcomes of caesarean section, operative vaginal birth and five-minute Apgar score less than seven did we see any statistically significant difference between the groups. Maternal satisfaction and hypoglycaemia, both also primary outcomes in this review, were not reported in any of the included studies. No women included in this review suffered from regurgitation during general anaesthesia or Mendelson’s syndrome, a very rare complication in modern anaesthesia. We were not able to undertake a systematic review of such adverse outcomes, but plan to do so in a future update. There were no significant differences identified in any of the outcomes assessed in the review for women at low risk of potentially needing general anaesthesia in any of the comparisons made, though sometimes this may be related to the small numbers of women involved in the assessment of some individual outcomes. However, one large study dominated the findings in this meta-analysis (O’Sullivan 2009) and this hospital had high medical intervention rates. In particular, this study (O’Sullivan 2009) included only women with no known obstetric or medical complication that would increase the likelihood of an operative birth, yet 30% of women in the study had a caesarean section. It may be that in less medicalised environments there might be differences in outcomes identified. Also, women were told that “…eating was not recommended in labour…but were actively encouraged to do so if randomised to the feeding arm.” Since most women do not want conflict with their caregivers, this may have influenced their behaviour.
Only one study looked at restricting oral intake in labour with women who had freedom to eat and drink at will during labour. No benefits or risks of restricting women in this way were identified for women at low risk of needing a general anaesthetic.
Looking at restricting women to water only compared with giving them carbohydrate drinks during labour, identified different findings for the two studies (Kubli 2002; Scheepers 2002). One study, involving 203 women, showed a reduction in caesarean section for women taking water only (RR 0.34, 95% CI 0.15 to 0.77, 201 women) and the authors postulated that this may be due to a re-distribution of blood flow to the gastrointestinal tract from the myometrium (Scheepers 2002). However, the other study found no difference in the incidence of caesarean sections (RR 1.33, 95% CI 0.53 to 3.38, 60 women). It is unclear where this difference arises from and could merely be a result of small numbers of women included in the studies. It would be important to investigate this further as some carbohydrate drinks may increase the risk of caesarean section. It would be worth comparing the use of carbohydrate drinks compared with freedom to eat and drink at will during labour to see if this really is a problem.
For women at increased risk of potentially needing general anaesthesia
There were no studies looking at this population of women. Again, the better general anaesthetic techniques and the increased use of regional anaesthesia mean that the incidence of Mendelson’s syndrome and regurgitation during general anaesthesia are very rare in this population of women as well.
Overall completeness and applicability of evidence
When considering any restriction of fluids and food during labour compared with some fluids and food available to women, the pooled data are dominated by one large study involving 2443 women at low risk of complications (O’Sullivan 2009). However, in this study there were very high intervention rates, particularly for women considered at low risk of complications (over 50% oxytocin augmentation, just under 70% getting intravenous fluids and epidurals in labour, 30% caesarean sections, 27% operative vaginal births). In addition, 20% of the women in the water-only arm ate during labour and 29% of the women in the food and fluids arm choose not to eat in labour (Table 1). This clearly reflects the wide variation in women’s wishes for food and fluids during labour.
Quality of the evidence
The overall quality of the evidence was reasonable (Figure 1).
Potential biases in the review process
The possibility of introducing bias was present at every stage of reviewing process. We attempted to minimise bias in a number of ways; two review authors assessed eligibility for inclusion, carried out data extraction and assessed risk of bias. Each worked independently. Nevertheless, the process of assessing risk of bias, for example, is not an exact science and includes many personal judgements.
Agreements and disagreements with other studies or reviews
Although restricting fluids and foods during labour has been standard policy in many countries over many years (McCormick 2002; Pengelley 1998), current policies are tending to be less restrictive (NICE 2007). This possibly reflects an acknowledgement of the lack of evidence to restrict women’s access to fluids and food during labour (NICE 2007) and some women’s unpleasant and harrowing experiences of such restrictions (Armstrong 2000; Johnson 1989). Although NICE suggested that women should be informed that isotonic drinks may be more beneficial than water, our evidence suggests otherwise (NICE 2007). Where NICE looked at one study (Kubli 2002) that assessed the impact of isotonic drinks on blood levels of biochemical substances, we assessed the impact on clinical outcomes and included all studies assessing carbohydrate solutions, with one study showing an increase in caesarean sections with carbohydrate solutions compared with water. There clearly needs to be more research on this aspect.
AUTHORS’ CONCLUSIONS
Implications for practice
The evidence identified no benefits or harms associated with restricting women’s access to fluids and foods during labour for women at low risk of potentially requiring a general anaesthetic, although the studies did not assess women’s views or feelings of control. Hence, women should have the autonomy and freedom to choose whether to eat or drink in labour, or not. Women should be able to consume what they desire and in doing so experience no adverse impact on labour, maternal or fetal outcomes. Most women seem to naturally reduce their intake as labour progresses and becomes more intense. We found no evidence from randomized trials on which to base practice regarding food or fluids in labour for women at increased risk of complications
Implications for research
Further research would be helpful to determine the impact of differing fluids and foods taken during labour, such as carbohydrate solutions. While Mendelson’s syndrome is an important adverse outcome, it is very rare and not the most appropriate outcome measure for studies exploring the effectiveness of food and fluid intake during labour. A better approach to the problem of this very rare but important complication of regurgitation during general anaesthesia and Mendelson’s syndrome, would be to study interventions that have been shown to be effective at reducing the acidity and volume of stomach contents for elective caesarean section (Paranjothy 2004) to see if such interventions are effective if implemented during labour once it has been decided that a general anaesthetic is required. In addition, research is needed to determine the most effective nutrient and hydration strategies for women in labour within the context of common medical and obstetrical practices, such as epidurals and oxytocin stimulation.
PLAIN LANGUAGE SUMMARY.
Eating and drinking in labour
In some cultures, food and drinks are consumed during labour for nourishment and comfort to help meet the demands of labour. However, in many birth settings, oral intake is restricted in response to work by Mendelson in the 1940s. Mendelson reported that during general anaesthesia, there was an increased risk of the stomach contents entering the lungs. The acid nature of the stomach liquid and the presence of food particles were particularly dangerous, and potentially could lead to severe lung disease or death. Since the 1940s, obstetrical anaesthesia has changed considerably, with better general anaesthetic techniques and a greater use of regional anaesthesia. These advances, and the reports by women that they found the restrictions unpleasant, have led to research looking at these restrictions. In addition, poor nutritional balance may be associated with longer and more painful labours, and fasting does not guarantee an empty stomach or less acidity. This review looked at any restriction of fluids and food in labour compared with women able to eat and drink. The review identified five studies involving 3130 women. Most studies had looked at specific foods being recommended, though one study let women to choose what they wished to eat and drink. The review identified no benefits or harms of restricting foods and fluids during labour in women at low risk of needing anaesthesia. There were no studies identified on women at increased risk of needing anaesthesia. None of the studies looked at women’s views of restricting fluids and foods during labour. Thus, given these findings, women should be free to eat and drink in labour, or not, as they wish.
ACKNOWLEDGEMENTS
We would like to thank Professor GJ Hofmeyr for his support and assistance. Also we would like to thank Lei Dou who helped with some of the data extraction.
As part of the pre-publication editorial process, this review has been commented on by two peers (an editor and referee who is external to the editorial team), a member of the Pregnancy and Childbirth Group’s international panel of consumers and the Group’s Statistical Adviser.
SOURCES OF SUPPORT
Internal sources
University of the Witwatersrand (MS), South Africa.
The University of Liverpool, UK.
External sources
World Health Organization Long-term Institutional Development Grant (MS), Switzerland.
National Institute for Health Research, UK.
NIHR NHS Cochrane Collaboration Programme Grant Scheme award for NHS-prioritised centrally-managed, pregnancy and childbirth systematic reviews: CPGS02
CHARACTERISTICS OF STUDIES
Characteristics of included studies [ordered by study ID]
| Methods | RCT of individual women. | |
| Participants |
Inclusion: women at low risk of complications in early labour (> 37 weeks; singleton; cephalic; < 5 cm dilatation) N = 60. Exclusion: obstetric or medical complication; increased likelihood OVB or CS; mothers requesting IM meperidine |
|
| Interventions |
Intervention: water only, as much as desired. Comparison: isotonic sports drink (dextrose,maltolactose, glucose, 28 kcal/dL), women encouraged to drink 500 ml in 1st hour and further 500 ml very 3-4 hours. Small quantities of water were also available if desired |
|
| Outcomes | Vomiting; duration of labour; oxytocin augmentation; mode of birth; Apgar scores; umbilical artery and venous blood gasses. Also: plasma b-hydroxybutryate; NEFAs; glucose measured early and at end of 1st stage of labour; residual gastric volume (assessed using real-time ultrasound) |
|
| Notes |
|
|
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer random number generation. |
| Allocation concealment (selection bias) | Unclear risk | “Sealed envelopes were opened after recruitment to the study” but no mention if numbered or if opaque |
| Blinding (performance bias and detection bias) All outcomes |
Unclear risk | No information on blinding. |
| Incomplete outcome data (attrition bias) All outcomes |
Low risk | No women withdrew from the study. |
| Selective reporting (reporting bias) | Unclear risk | Not apparent but we did not assess the protocol so cannot be sure |
| Other bias | Low risk | No evidence of other bias. |
| Methods | RCT of individual women. | |
| Participants |
Inclusion: women in labour at low risk of complications (no known obstetric or medical complication that would increase the likelihood of operative birth; nulliparous; singleton; cephalic; > 36 weeks; no diabetes; but included induction and augmentation); also women were < 6 cm N = 2443 women randomised, 2426 women analysed. Exclusion: multiparous; women with a known obstetric or medical complication that could have increased the likelihood of an operative birth, were in severe pain, intended to use parenteral opioids for analgesia in labour; unable to understand English (and no interpreter available) |
|
| Interventions |
Intervention: water and ice chips only Comparison: specific foods and fluids encouraged (women advised to consume low fat, low residue diet at will during labour). Foods advised were: bread, biscuits, vegetables, fruits, low fat yoghurt, soup, isotonic drinks and fruit juice |
|
| Outcomes | Primary: spontaneous vaginal birth. Secondary: included: duration of labour; use of IV oxytocin for augmentation; CS;OVB; food intake in 6 hours before labour (snacks/light meal/largemeal); food intake in labour (no intake/water only/calorific drinks/solids) |
|
| Notes |
|
|
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | “…computer randomisation…” |
| Allocation concealment (selection bias) | Low risk | “Entry of a woman’s initials, hospital number, and date of birth on to a dedicated computer on the labour ward automatically generated the allocation group together with a study number, which was then recorded on the outcomes sheet. These data could, if necessary, be verified against the computer randomisation at a later date.” |
| Blinding (performance bias and detection bias) All outcomes |
High risk | “The attending obstetricians and midwives made all the relevant decisions about the woman’s obstetric management but obviously could not be blinded to trial allocation. The people deciding on obstetric interventions were generally unaware of the trial intervention allocation and had no vested interest in the study.” |
| Incomplete outcome data (attrition bias) All outcomes |
Low risk |
|
| Selective reporting (reporting bias) | Unclear risk | Paper specifies some of the outcomes to be assessed and we did not assess the protocol |
| Other bias | Unclear risk | 20% of women in the water only group ate in labour. This may have caused some bias in the outcome estimations, but shows in part women’s views of restricting food and fluids during labour 29% of women chose not to eat any food during labour. This would be considered part of women’s freedom of choice whether to eat or not during labour |
| Methods | RCT of individual women. | |
| Participants |
Inclusion: nulliparous women in early labour (singleton; cephalic; 2-4 cm dilatation) N = 203. Exclusion: elective caesarean sections; multiple pregnancy; diabetes, and women who were considered by the attending clinician to have a direct risk for a caesarean section |
|
| Interventions |
Intervention: flavoured water (artificial aroma, aspartame, acesulfame), as much as desired Comparison: carbohydrate drink (per 100 mL: 12.6 g carbohydrates: 9.8% polysach/Na: 50 mg, Osm: 280 mOsm/L), as much as desired |
|
| Outcomes | Duration of the active labour (i.e. from randomisation to birth), pain medication; augmentation; the mode of birth, fetal presentation; birthweight, fetal arterial cord pH and Apgar scores | |
| Notes |
|
|
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | ‘Computerised list’. |
| Allocation concealment (selection bias) | Low risk | ‘Drawing consecutive envelopes, which included numbers of a computerised list corresponding to numbered bottles in identical packages’ |
| Blinding (performance bias and detection bias) All outcomes |
Low risk | ‘In identical packages’ ‘both solutions had an identical taste and colour and were prepared by the producer of the carbohydrates.’ |
| Incomplete outcome data (attrition bias) All outcomes |
Low risk | Data were missing in two women and so less than 1% which should not affect outcomes |
| Selective reporting (reporting bias) | Unclear risk | Not apparent, but we did not assess the protocol so cannot be sure |
| Other bias | Low risk | No evidence of other bias. |
| Methods | RCT of individual women, stratified by parity and induction. | |
| Participants |
Inclusion: women at low risk of complications in early labour (> 37 weeks; singleton; cephalic; < 5 cm dilatation) N = 94. Exclusion: obstetric or medical complication increasing the likelihood of instrumental delivery or caesarean; requesting IM pethidine for analgesia |
|
| Interventions |
Intervention: water only. Comparison: low residue food (women were allowed to select from a low-residue diet throughout the course of labour) |
|
| Outcomes | Vomiting; duration of first and second stage of labour; oxytocin requirements; mode of birth; Apgar scores; umbilical artery and venous blood gases Also: plasma ß-hydroxybutyrate; nonesterified fatty acids; glucose; insulin and lactate; gastric volume |
|
| Notes |
|
|
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No information provided. |
| Allocation concealment (selection bias) | Unclear risk | Sealed, pre-randomised envelopes. |
| Blinding (performance bias and detection bias) All outcomes |
High risk | Blinding of participants and clinician was not feasible; information on blinding of outcome assessor was not provided |
| Incomplete outcome data (attrition bias) All outcomes |
Low risk | Two women withdrew from the trial (one from each group). Four (two from each group) were excluded because they reached the second stage of labour within one hour of entry to the study. Their data could not be re-included but were not considered large enough (6%) to impact differentially on outcomes |
| Selective reporting (reporting bias) | Unclear risk | Not apparent, but we did not assess the protocol so cannot be sure |
| Other bias | Low risk | Two groups were similar with respect to age, parity, induction and cervical dilatation at the time of randomisation |
| Methods | RCT of individual women at low risk of complications. | |
| Participants |
Inclusion: women at low risk of complications (> 30 weeks; singleton; no recorded maternal or fetal complication) N = 330. Exclusion: planned CS; maternal illness such as oral intake would be restricted during labour and IV therapy required (e.g. diabetes); fetal compromise such that there was high risk of CS (e.g. severe FGR, fetal anomalies) |
|
| Interventions |
Intervention: ice chips and sips water (women were permitted ice chips, popsicles, or sips of fluid during active labour.Women received no specific or written instructions on oral intake during labour and were permitted) Comparison: unrestricted access to their choice of food and fluids during labour (women received a booklet containing easy-to-read guideline on suggested nutrient and fluid intake during labour based on nutritional guidelines for individuals who participate in prolonged, moderate, aerobic exercise. Women in the intervention group were encouraged to eat easily digestible foods or fluids in frequent and small amounts and to bring their own selection of desired food and drinks. Although the investigators suggested certain foods and fluids, women were free to consume what they desired) |
|
| Outcomes | Primary: incidence of dystocia (mean rate of dilatation of < 0.5 cm/hr during a period of at least 4 hours after 3 cm cervical dilatation) Secondary: perception of thirst, hunger, nausea, and fatigue; labour length, the incidence of medical interventions during labour, fluid and nutrient balance, and the incidence of maternal and newborn complications |
|
| Notes |
|
|
| Risk of bias | ||
| Bias | Authors’ judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | “…computer-generated group assignment designations” |
| Allocation concealment (selection bias) | Low risk | “..sequentially numbered opaque envelopes…” |
| Blinding (performance bias and detection bias) All outcomes |
High risk | Participants and clinicians cannot be blinded but outcome assessor could be; no information provided |
| Incomplete outcome data (attrition bias) All outcomes |
Low risk | Two women withdrew after randomisation. Primary outcomes and clinical estimates, none fromrestricted group and twowomen lost from freedom to E&D group For data from questionnaires; 22% (72/330) lost - more than 20%loss so some risk of bias. Losses were 24% in ice chips group and 19% in free to E&D group Overall for clinical outcomes, loss of data probably not significant. |
| Selective reporting (reporting bias) | Low risk | All pre-specified outcomes were reported and we assessed the protocol as set out in the author’s thesis (Tranmer 2005). |
| Other bias | Unclear risk | Oral intake was restricted when women developed intrapartum complications or had epidural analgesia. Epidural analgesia was used in 120/165 (73%) of women in the water only group and 129/163 (79%) women in the freedomto eat group. It is unclear whether this impacted on outcomes Of the 78% who returned the questionnaire, 56% (63/119) of women in the unrestricted access group reported they ate or drank some source of carbohydrates, in comparison to only 13% (15/120) in the ice-chips/sips of water only group |
CS: caesarean section
E&D: eating and drinking
FGR: fetal growth restriction
IM: intramuscular
ITT: intention to treat
IV: intravenous
NEFAs: non-esterified fatty acids
OVB: operative vaginal births
RCT: randomised controlled trial
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
| Scheepers 2004 | The study was not looking at the same participants as the review. The participants in the study were women at 8-10 cm dilatation given high glucose load |
| Shennan 2005 | This is a discussion paper. |
Characteristics of studies awaiting assessment [ordered by study ID]
| Methods | RCT of individual women. |
| Participants | Women in labour at low risk of caesarean. |
| Interventions |
Intervention: sips of water. Comparison: feeding in labour. |
| Outcomes | Mode of birth; blood loss; augmentation of labour; analgesia requirement; duration of labour; Apgar scores; admission to SCBU; infant jaundice; initiation of breastfeeding |
| Notes | We are attempting to contact this author, but as yet we have had no success |
| Methods | RCT of individual women. |
| Participants | Women at low risk of obstetric and medical complications in spontaneous labour at term |
| Interventions |
Intervention: iced water + IV hydration. Comparison: clear oral liquids. |
| Outcomes | Length of labour; length of second stage. |
| Notes | Conference abstract only. We are attempting to contact the authors but as yet with no success. It is unclear what ‘clear liquids’ mean and whether these contain sugars or not |
| Methods | RCT of individual women. |
| Participants | All women admitted to delivery suite who were of 34 weeks’ gestation or more, with a singleton fetus, cephalic presentation, who had not had a previous caesarean section and were not at greater risk of caesarean section than the general pregnant population N = 297. |
| Interventions |
Intervention: water only (Yiannouzis 2009 - personal communication) (N = 143) Comparison: women offered a low fat diet during labour. Food was as much as women desired, as often as they wished for the duration of labour. (N = 154) |
| Outcomes | Syntocinon augmentation; pain relief; length of labour; type of birth; vomiting; Apgar scores and admission to SCBU or NICU |
| Notes | Therewas no statistical difference identified between the two groups in the outcomes of labour except in the occurrence of vomiting and length of labour. Women in the low fat diet group were twice as likely to vomit as those in the control group and their labours were longer Most women did not crave large amounts of food during labour. Some women commented that vomiting is less unpleasant if the stomach is not empty. The increased length of labour may not have been perceived as totally negative as women applauded the availability of choice and the improved social context of labour when food was offered We have written and spoken with the author, but the detailed data are no longer available |
| Methods | Quasi-RCT of individual women. |
| Participants | Healthy women in labour for the first time. No exclusions. |
| Interventions |
Intervention: routine diet in labour Comparison: high energy liquid diet (Heng Nai Jin). One bottle in each of the three stages of labour (first, second and third) |
| Outcomes | Length of the three stages of labour and total duration of labour |
| Notes | We had this paper translated from Chinese. However, there were no data presented |
IV: intravenous
NICU: neonatal intensive care unit
RCT: randomised controlled trial
SCBU: special care baby unit
DATA AND ANALYSES
Comparison 1.
Any restriction of oral fluid and food versus some fluid and food
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Caesarean section | 5 | 3103 | Risk Ratio (M-H, Random, 95% CI) | 0.89 [0.63, 1.25] |
| 1.1 Women at low risk of caesarean/complications | 5 | 3103 | Risk Ratio (M-H, Random, 95% CI) | 0.89 [0.63, 1.25] |
| 1.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 1.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Operative vaginal birth | 5 | 3103 | Risk Ratio (M-H, Random, 95% CI) | 0.98 [0.88, 1.10] |
| 2.1 Women at low risk of caesarean/complications | 5 | 3103 | Risk Ratio (M-H, Random, 95% CI) | 0.98 [0.88, 1.10] |
| 2.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 2.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Maternal satisfaction | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 3.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 3.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 3.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Apgar < 7 at 5 min | 4 | 2902 | Risk Ratio (M-H, Random, 95% CI) | 1.43 [0.77, 2.68] |
| 4.1 Women at low risk of caesarean/complications | 4 | 2902 | Risk Ratio (M-H, Random, 95% CI) | 1.43 [0.77, 2.68] |
| 4.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 4.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Neonatal hypogylcaemia | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 5.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 5.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 5.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Maternal ketoacidosis | 1 | 328 | Risk Ratio (M-H, Random, 95% CI) | 0.99 [0.66, 1.49] |
| 6.1 Women at low risk of caesarean/complications | 1 | 328 | Risk Ratio (M-H, Random, 95% CI) | 0.99 [0.66, 1.49] |
| 6.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 6.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Maternal dehydration | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 7.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 7.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 7.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Maternal hyponatraemia | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 8.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 8.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 8.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 9 Maternal hypoglycaemia | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 9.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 9.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 9.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 10 Duration of labour (hours) | 3 | 476 | Mean Difference (IV, Random, 95% CI) | −0.29 [−1.55, 0.97] |
| 10.1 Women at low risk of caesarean/complications | 3 | 476 | Mean Difference (IV, Random, 95% CI) | −0.29 [−1.55, 0.97] |
| 10.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 10.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 11 Mobility in labour | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 11.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 11.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 11.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 12 Maternal nausea | 1 | 255 | Risk Ratio (M-H, Random, 95% CI) | 0.80 [0.54, 1.18] |
| 12.1 Women at low risk of caesarean/complications | 1 | 255 | Risk Ratio (M-H, Random, 95% CI) | 0.80 [0.54, 1.18] |
| 12.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 12.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 13 Maternal vomiting | 3 | 2574 | Risk Ratio (M-H, Random, 95% CI) | 0.90 [0.62, 1.31] |
| 13.1 Women at low risk of caesarean/complications | 3 | 2574 | Risk Ratio (M-H, Random, 95% CI) | 0.90 [0.62, 1.31] |
| 13.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 13.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 14 Augmentation of labour | 5 | 3103 | Risk Ratio (M-H, Random, 95% CI) | 1.02 [0.95, 1.09] |
| 14.1 Women at low risk of caesarean/complications | 5 | 3103 | Risk Ratio (M-H, Random, 95% CI) | 1.02 [0.95, 1.09] |
| 14.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 14.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 15 Narcotic pain relief | 3 | 349 | Risk Ratio (M-H, Random, 95% CI) | 0.94 [0.74, 1.21] |
| 15.1 Women at low risk of caesarean/complications | 3 | 349 | Risk Ratio (M-H, Random, 95% CI) | 0.94 [0.74, 1.21] |
| 15.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 15.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 16 Epidural analgesia | 5 | 3103 | Risk Ratio (M-H, Random, 95% CI) | 0.98 [0.91, 1.05] |
| 16.1 Women at low risk of caesarean/complications | 5 | 3103 | Risk Ratio (M-H, Random, 95% CI) | 0.98 [0.91, 1.05] |
| 16.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 16.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 17 Poor maternal expulsive efforts | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 17.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 17.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 17.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 18 Regurgitation during general anaesthesia | 1 | 2426 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 18.1 Women at low risk of caesarean/complications | 1 | 2426 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 18.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 18.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 19 Mendelson’s syndrome | 2 | 2754 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 19.1 Women at low risk of caesarean/complications | 2 | 2754 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 19.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 19.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 20 Maternal mortality | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21 Postpartum haemorrhage (> 1000 ml) | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 21.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 21.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 21.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 22 Maternal admission to intensive care | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 22.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 22.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 22.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 23 Length of maternal hospital stay | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 23.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 23.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 23.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 24 Maternal comfort | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 24.1 Women at low risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 24.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 24.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 25 Maternal feelings of pain, thirst or hunger | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 25.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 25.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 25.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 26 Breastfeeding success | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 26.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 26.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 26.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 27 Maternal feelings of control | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 27.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 27.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 27.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 28 Fetal distress | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 28.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 28.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 28.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 29 Cord blood pH < 7.2 | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 29.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 29.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 29.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 30 Infant hyperinsulinism | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 30.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 30.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 30.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 31 Infant hyponatraemia | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 31.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 31.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 31.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 32 Infant intravenous therapy | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 32.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 32.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 32.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 33 Infant gavage feeding | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 33.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 33.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 33.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 34 Infant admission to intensive care | 1 | 2426 | Risk Ratio (M-H, Random, 95% CI) | 1.03 [0.73, 1.45] |
| 34.1 Women at low risk of caesarean/complications | 1 | 2426 | Risk Ratio (M-H, Random, 95% CI) | 1.03 [0.73, 1.45] |
| 34.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 34.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 35 Length of infant hospital stay | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0 |
| 35.1 Women at low risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 35.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 35.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Random, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 2.
Complete restriction of oral fluid and food (other than ice chips) versus freedom to eat and drink
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Caesarean section | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.77 [0.51, 1.16] |
| 1.1 Women at low risk of caesarean/complications | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.77 [0.51, 1.16] |
| 1.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 1.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Operative vaginal birth | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.99 [0.72, 1.35] |
| 2.1 Women at low risk of caesarean/complications | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.99 [0.72, 1.35] |
| 2.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Maternal satisfaction | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Apgar < 7 at 5 min | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.1 Women at low risk of caesarean/complications | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Neonatal hypogylcaemia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Maternal ketoacidosis | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.99 [0.66, 1.49] |
| 6.1 Women at low risk of caesarean/complications | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.99 [0.66, 1.49] |
| 6.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Maternal dehydration | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Maternal hypoglycaemia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9 Maternal hyponatraemia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10 Duration of labour (hours) | 1 | 328 | Mean Difference (IV, Fixed, 95% CI) | −0.80 [−2.13, 0.53] |
| 10.1 Women at low risk of caesarean/complications | 1 | 328 | Mean Difference (IV, Fixed, 95% CI) | −0.80 [−2.13, 0.53] |
| 10.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11 Mobility in labour | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12 Maternal nausea | 1 | 255 | Risk Ratio (M-H, Fixed, 95% CI) | 0.80 [0.54, 1.18] |
| 12.1 Women at low risk of caesarean/complications | 1 | 255 | Risk Ratio (M-H, Fixed, 95% CI) | 0.80 [0.54, 1.18] |
| 12.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 13 Maternal vomiting | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 13.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 13.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 13.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 14 Augmentation of labour | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.98 [0.81, 1.18] |
| 14.1 Women at low risk of caesarean/complications | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.98 [0.81, 1.18] |
| 14.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 14.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 15 Narcotic pain relief | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 15.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 15.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 15.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 16 Epidural analgesia | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.92 [0.81, 1.04] |
| 16.1 Women at low risk of caesarean/complications | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.92 [0.81, 1.04] |
| 16.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 16.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17 Poor maternal expulsive efforts | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18 Regurgitation during general anaesthesia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19 Mendelson’s syndrome | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19.1 Women at low risk of caesarean/complications | 1 | 328 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20 Maternal mortality | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21 Postpartum haemorrhage (> 1000 ml) | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22 Maternal admission to intensive care | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23 Length of maternal hospital stay | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24 Maternal comfort | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24.1 Women at low risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25 Maternal feelings of pain, thirst or hunger | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26 Breastfeeding success | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27 Maternal feelings of control | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28 Fetal distress | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29 Cord blood pH < 7.2 | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30 Infant hyperinsulinism | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31 Infant intravenous therapy | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32 Infant intravenous therapy | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33 Infant gavage feeding | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34 Infant admission to intensive care | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35 Length of infant hospital stay | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35.1 Women at low risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 7.
Water only versus specific oral fluid and food
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Caesarean section | 2 | 2514 | Risk Ratio (M-H, Fixed, 95% CI) | 1.02 [0.91, 1.15] |
| 1.1 Women at low risk of caesarean/complications | 2 | 2514 | Risk Ratio (M-H, Fixed, 95% CI) | 1.02 [0.91, 1.15] |
| 1.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 1.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Operative vaginal birth | 2 | 2514 | Risk Ratio (M-H, Fixed, 95% CI) | 0.96 [0.84, 1.10] |
| 2.1 Women at low risk of caesarean/complications | 2 | 2514 | Risk Ratio (M-H, Fixed, 95% CI) | 0.96 [0.84, 1.10] |
| 2.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Maternal satisfaction | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Apgar < 7 at 5 min | 2 | 2514 | Risk Ratio (M-H, Fixed, 95% CI) | 1.39 [0.73, 2.63] |
| 4.1 Women at low risk of caesarean/complications | 2 | 2514 | Risk Ratio (M-H, Fixed, 95% CI) | 1.39 [0.73, 2.63] |
| 4.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Neonatal hypogylcaemia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Maternal ketoacidosis | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Maternal dehydration | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Maternal hyponatraemia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9 Maternal hypoglycaemia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10 Duration of labour (hours) | 1 | 88 | Mean Difference (IV, Fixed, 95% CI) | −1.10 [−2.66, 0.46] |
| 10.1 Women at low risk of caesarean/complications | 1 | 88 | Mean Difference (IV, Fixed, 95% CI) | −1.10 [−2.66, 0.46] |
| 10.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11 Mobility in labour | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12 Maternal nausea | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 13 Maternal vomiting | 2 | 2514 | Risk Ratio (M-H, Random, 95% CI) | 0.76 [0.41, 1.41] |
| 13.1 Women at low risk of caesarean/complications | 2 | 2514 | Risk Ratio (M-H, Random, 95% CI) | 0.76 [0.41, 1.41] |
| 13.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 13.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 14 Augmentation of labour | 2 | 2514 | Risk Ratio (M-H, Random, 95% CI) | 0.97 [0.80, 1.19] |
| 14.1 Women at low risk of caesarean/complications | 2 | 2514 | Risk Ratio (M-H, Random, 95% CI) | 0.97 [0.80, 1.19] |
| 14.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 14.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 15 Narcotic pain relief | 1 | 88 | Risk Ratio (M-H, Fixed, 95% CI) | 1.00 [0.91, 1.09] |
| 15.1 Women at low risk of caesarean/complications | 1 | 88 | Risk Ratio (M-H, Fixed, 95% CI) | 1.00 [0.91, 1.09] |
| 15.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 15.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 16 Epidural analgesia | 2 | 2514 | Risk Ratio (M-H, Fixed, 95% CI) | 1.02 [0.97, 1.08] |
| 16.1 Women at low risk of caesarean/complications | 2 | 2514 | Risk Ratio (M-H, Fixed, 95% CI) | 1.02 [0.97, 1.08] |
| 16.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 16.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17 Poor maternal expulsive efforts | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18 Regurgitation during general anaesthesia | 1 | 2426 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18.1 Women at low risk of caesarean/complications | 1 | 2426 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19 Mendelson’s syndrome | 1 | 2426 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19.1 Women at low risk of caesarean/complications | 1 | 2426 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20 Maternal mortality | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21 Postpartum haemorrhage (> 1000 ml) | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22 Maternal admission to intensive care | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23 Length of maternal hospital stay | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24 Maternal comfort | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24.1 Women at low risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25 Maternal feelings of pain, thirst or hunger | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26 Breastfeeding success | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27 Maternal feelings of control | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28 Fetal distress | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29 Cord blood pH < 7.2 | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30 Infant hyperinsulinism | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31 Infant intravenous therapy | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32 Infant intravenous therapy | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33 Infant gavage feeding | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34 Infant admission to intensive care | 1 | 2426 | Risk Ratio (M-H, Fixed, 95% CI) | 1.03 [0.73, 1.45] |
| 34.1 Women at low risk of caesarean/complications | 1 | 2426 | Risk Ratio (M-H, Fixed, 95% CI) | 1.03 [0.73, 1.45] |
| 34.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35 Length of infant hospital stay | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35.1 Women at low risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
Comparison 10.
Water only versus oral carbohydrate based fluids
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Caesarean section | 2 | 261 | Risk Ratio (M-H, Random, 95% CI) | 0.66 [0.17, 2.53] |
| 1.1 Women at low risk of caesarean/complications | 2 | 261 | Risk Ratio (M-H, Random, 95% CI) | 0.66 [0.17, 2.53] |
| 1.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 1.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 2 Operative vaginal birth | 2 | 261 | Risk Ratio (M-H, Fixed, 95% CI) | 1.17 [0.80, 1.71] |
| 2.1 Women at low risk of caesarean/complications | 2 | 261 | Risk Ratio (M-H, Fixed, 95% CI) | 1.17 [0.80, 1.71] |
| 2.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Maternal satisfaction | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Apgar < 7 at 5 min | 1 | 60 | Risk Ratio (M-H, Fixed, 95% CI) | 3.0 [0.13, 70.83] |
| 4.1 Women at low risk of caesarean/complications | 1 | 60 | Risk Ratio (M-H, Fixed, 95% CI) | 3.0 [0.13, 70.83] |
| 4.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Neonatal hypoglycaemia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6 Maternal ketoacidosis | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7 Maternal dehydration | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Maternal hyponatraemia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9 Maternal hypoglycaemia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 9.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10 Duration of labour (hours) | 1 | 60 | Mean Difference (IV, Fixed, 95% CI) | 0.95 [−0.42, 2.32] |
| 10.1 Women at low risk of caesarean/complications | 1 | 60 | Mean Difference (IV, Fixed, 95% CI) | 0.95 [−0.42, 2.32] |
| 10.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11 Mobility in labour | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12 Maternal nausea | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 12.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 13 Maternal vomiting | 1 | 60 | Risk Ratio (M-H, Fixed, 95% CI) | 1.27 [0.69, 2.33] |
| 13.1 Women at low risk of caesarean/complications | 1 | 60 | Risk Ratio (M-H, Fixed, 95% CI) | 1.27 [0.69, 2.33] |
| 13.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 13.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 14 Augmentation of labour | 2 | 261 | Risk Ratio (M-H, Fixed, 95% CI) | 1.07 [0.75, 1.52] |
| 14.1 Women at low risk of caesarean/complications | 2 | 261 | Risk Ratio (M-H, Fixed, 95% CI) | 1.07 [0.75, 1.52] |
| 14.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 14.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 15 Narcotic pain relief | 2 | 261 | Risk Ratio (M-H, Random, 95% CI) | 0.86 [0.36, 2.06] |
| 15.1 Women at low risk of caesarean/complications | 2 | 261 | Risk Ratio (M-H, Random, 95% CI) | 0.86 [0.36, 2.06] |
| 15.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 15.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 16 Epidural analgesia | 2 | 261 | Risk Ratio (M-H, Random, 95% CI) | 0.80 [0.44, 1.43] |
| 16.1 Women at low risk of caesarean/complications | 2 | 261 | Risk Ratio (M-H, Random, 95% CI) | 0.80 [0.44, 1.43] |
| 16.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 16.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 17 Poor maternal expulsive efforts | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18 Regurgitation during general anaesthesia | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19 Mendelson’s syndrome | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 19.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20 Maternal mortality | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 20.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21 Postpartum haemorrhage (> 1000 ml) | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 21.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22 Maternal admission to intensive care | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 22.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23 Length of maternal hospital stay | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 23.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24 Maternal comfort | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24.1 Women at low risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 24.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25 Maternal feelings of pain, thirst or hunger | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 25.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26 Breastfeeding success | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 26.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27 Maternal feelings of control | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 27.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28 Fetal distress | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 28.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29 Cord blood pH < 7.2 | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 29.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30 Infant hyperinsulinism | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 30.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31 Infant intravenous therapy | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 31.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32 Infant intravenous therapy | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 32.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33 Infant gavage feeding | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 33.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34 Infant admission to intensive care | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34.1 Women at low risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34.2 Women at increased risk of caesarean/complications | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 34.3 Women with no defined risk status | 0 | 0 | Risk Ratio (M-H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35 Length of infant hospital stay | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35.1 Women at low risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35.2 Women at increased risk of caesarean/complications | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 35.3 Women with no defined risk status | 0 | 0 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
FEEDBACK
Nazziwa, 14 February 2012
Summary
The objective of this review was to evaluate the effects of oral intake during labour on obstetric and neonatal outcomes. The authors’ conclusions were that oral fluids and food during labour do not effect neonatal or obstetric outcomes. However, a fully dilated cervix is a major contributor to spontaneous delivery, and the quicker the cervix dilates the shorter the labour. The kind of food laboring mothers need is food that contributes to the process of cervical dilation, thus shortening labor, that gives them energy, and that hydrates them and prepares them for the afterbirth period. The foods used in the studies in this review were those rich in carbohydrates, thus providing energy and hydration to the patients. They lacked nutritional benefits that might help with cervical dilation process. This may have caused the balance between the two groups, hence the observation of no difference between the groups.
The review needed to include foods rich in oxytocin for improvement in rate of dilation, carbohydrates for energy; potassium to reduce the incidence of vomiting, and foods that contribute to milk production. One food that is rich in these essential components is the date fruit1. The botanical name of the date palm is Phoenix dactylifera, a member of the family Palmae (Arecaceae). If such foods had been included in the review then there would have been an effect on some of the outcome variables, such as labour duration and cervical dilation rate.
References
1. Al-Kuran O, Al-Mehaisen L, Bawadi H, Beitawi S, Amarin Z. The effect of late pregnancy consumption of date fruit on labour and delivery. J Obstet Gynaecol. 2011;31(1):29-31.
[Feedback received from Aisha Nazziwa, 14 February 2012]
Reply
Thank you for your feedback. You are proposing an interesting hypothesis. In fact, the objective of this review was not to evaluate the effects of oral intake in labour on obstetric and neonatal outcomes but “To determine the benefits and harms of oral fluid and food restriction during labour…”, so we considered the restriction of what women might normally consume during labour as the intervention. We concluded that “The evidence identified no benefits or harms…” so we were careful not to say that ‘oral foods and fluids do not affect neonatal and obstetric outcomes’, but that we found no evidence to this effect; that is, we found ‘no evidence of effect’ and not ‘evidence of no effect’. We can only include in the systematic review trials that have been undertaken, and we identified none which tested your hypothesis on the consumption of dates. If we were in the future to identify such a trial, we would include it, if appropriate; for example, in Comparison 6 ‘Complete restriction of oral fluid and food (other than ice chips) versus specific oral food and fluids’, In addition, for some women it is not the length of labour which is important but their ability to work with their body to give birth to their baby, and restricting food and fluids in labour may interfere with how women feel about the birth.
Contributors
Mandisa Singata, Joan Tranmer and Gill Gyte
Osborn, 6 March 2012
Summary
Aspiration in association with obstetric anaesthesia is a condition with significant maternal morbidity and risk of maternal mortality. This review includes a relatively small number of cases of the rare but serious complications of aspiration. It is therefore possibly open to debate for the authors to state in the abstract conclusion: “there is no justification for the restriction of fluids and food in labour for women at low risk of complications”.
The next sentence states that as there are no studies looking specifically at women with increased risk of complications: “there is no evidence to support restrictions in this group of women”. This conclusion could potentially be misconstrued as supporting women in the high risk group eating in labour. As there are no specific studies comparing diet in labour in high risk women, the conclusion for high risk women, could have been: “as there are no studies looking specifically at women with increased risk of complication, there is no evidence to support not restricting food in this group of women”.
Conflict of interest statement:
I certify that I have no affiliations with or involvement in any organization or entity with a financial interest in the subject matter of my feedback.
[Feedback received from Kym Osborn, 6 March 2012]
Reply
Thank you for your feedback. We agree that in the absence of evidence regarding the risk for women with high risk of complications, the conclusion of the review should be neutral, so we have modified our wording to make this clearer: “We found no evidence from randomized trials on which to base practice regarding food or fluids in labour for women at increased risk of complications”.
Contributors
Mandisa Singata, Joan Tranmer and Gill Gyte
HISTORY
Protocol first published: Issue 4, 2002
Review first published: Issue 1, 2010
| Date | Event | Description |
|---|---|---|
| 10 October 2012 | Feedback has been incorporated | Feedback received from Aisha Nazziwa and Kym Osborn - see Feedback 1 and Feedback 2. |
| 10 February 2012 | Amended | Contact details updated. |
| 19 November 2008 | Amended | Converted to new review format. |
WHAT’S NEW
Last assessed as up-to-date: 1 October 2009.
| Date | Event | Description |
|---|---|---|
| 16 October 2012 | Feedback has been incorporated | Authors responded to feedback - see Feedback 1 and Feedback 2. |
DIFFERENCES BETWEEN PROTOCOL AND REVIEW
The protocol said an additional search would be undertaken of the Cochrane Controlled Trials Register using specific search terms.
#1 (ORAL near FEED*)
#2 (ORAL near FLUID*)
#3 (ORAL near HYDRAT*)
#4 (ORAL near INTAKE)
#5 EAT*
#6 DRINK*
#7 FOOD
#8 LABOR
#9 LABOUR
#10 LABOR*:ME
#11 ((((((#1 or #2) or #3) or #4) or #5) or #6) or #7)
#12 ((#8 or #9) or #10)
#13 (#11 and #12)
However, as this was already covered in the search undertaken by the Pregnancy and Childbirth Group office, we did not undertake the additional search and we have removed the reference to it.
We have modified the wording in the methods sections for ‘Assessment of heterogeneity’, ‘Assessment of reporting bias’ and ‘Data synthesis’ to update them with the new methods being used by the group, developed in conjunction with the group’s statistician, Simon Gates, and Richard Riley. We have used these new methods in the review.
In contrast to the published protocol, we included ‘allowing particular oral food and fluid regimens’ in our types of interventions.
We have added ‘maternal mortality’ as a secondary outcome.
Footnotes
DECLARATIONS OF INTEREST: One of the review authors (JE Tranmer) is principal author of one of the trials that was considered for inclusion. She did not participate in the decisions regarding data from this trial, which were considered by the other two review authors.
References to studies included in this review
- Kubli 2002 {published data only} .Kubli M, Scrutton M, O’Sullivan G, Seed P. Evaluation of isotonic ‘sports drinks’ in labour [abstract] International Journal of Obstetric Anesthesia. 2000;9:195. [Google Scholar]; Kubli M, Scrutton MJ, OSullivan G, Seed P. Evaluation of isotonic ‘sports drinks’ in labor [abstract] Anesthesiology. 2000;92(Suppl):A9. [Google Scholar]; *; Kubli M, Scrutton MJ, Seed PT, O’Sullivan G. An evaluation of isotonic “sport drinks” during labor. Anesthesia & Analgesia. 2002;94(2):404–8. doi: 10.1097/00000539-200202000-00033. [DOI] [PubMed] [Google Scholar]
- O’Sullivan 2009 {published data only} .Hart D. Does feeding in labour influence obstetric outcome? A randomised controlled trial. 2003. Personal communication.; Liu B, O’Sullivan G, Hart D, Waterstone M. Does eating during labour influence obstetric outcome [abstract] International Journal of Obstetric Anesthesia. 2007;16(Suppl 1):S11. [Google Scholar]; Liu B, O’Sullivan G, Hart D, Waterstone M, Shennan AH. A randomised controlled trial of feeding in labour [abstract] BJOG: an international journal of obstetrics and gynaecology. 2007;114(8):1036. [Google Scholar]; Liu B, O’Sullivan G, Hart D, Waterstone M, Shennan AH. Does eating during labour affect obstetric and neonatal outcome? - a randomised controlled trial [abstract] Journal of Obstetrics and Gynaecology. 2007;27(Suppl 1):S18–9. [Google Scholar]; O’Sullivan G, Liu B, Hart D, Seed P, Shennan A. Effect of food intake during labour on obstetric outcome: randomised controlled trial. BMJ. 2009;338:b784. doi: 10.1136/bmj.b784. [DOI: 10.1136/bmj.b784] [DOI] [PMC free article] [PubMed] [Google Scholar]; O’Sullivan G, Liu B, Shennan A, Hart D. Does eating in labor influence obstetric outcome: a randomized controlled trial in 2400 primiparous women? [abstract] Anesthesiology. 2006;104(Suppl 1):13. [Google Scholar]; *; Shennan A. [accessed 25 February 2004];Maternal and Fetal Research Unit (MFRU). Feeding in labour trial: does feeding during labour influence the obstetric outcome? Maternal and Fetal Research Unit. 2004 www.mfru.org.uk/FIL.htm
- Scheepers 2002 {published data only} .Scheepers HCJ, Thans MCJ, de Jong PA, Essed GGM, Le Cessie S, Kanhai HHH. A double-blind, randomised, placebo controlled study on the influence of carbohydrate solution intake during labour. BJOG: an international journal of obstetrics and gynaecology. 2002;109:178–81. doi: 10.1111/j.1471-0528.2002.t01-1-01062.x. [DOI] [PubMed] [Google Scholar]
- Scrutton 1999 {published data only} .Scrutton M, Lowy C, O’Sullivan G. Eating in labour: an assessment of the risks and benefits [abstract] International Journal of Obstetric Anesthesia. 1996;5:214–5. [Google Scholar]; *; Scrutton MJ, Metcalfe GA, Lowy C, Seed PT, O’Sullivan G. Eating in labour: a randomised controlled trial assessing the risks and benefits. Anaesthesia. 1999;54(4):329–34. doi: 10.1046/j.1365-2044.1999.00750.x. [DOI] [PubMed] [Google Scholar]
- Tranmer 2005 {published data only} .Tramner JE. Nutritional support during labour: a randomized clinical trial of patient controlled oral intake during labour [thesis] University of Toronto; Toronto, Canada: 1999. [Google Scholar]; *; Tranmer JE, Hodnett ED, Hannah ME, Stevens BJ. The effect of unrestricted oral carbohydrate intake on labor progress. Journal of Obstetric, Gynecologic & Neonatal Nursing. 2005;34(3):319–28. doi: 10.1177/0884217505276155. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
- Scheepers 2004 {published data only} .Scheepers HC, Thans MC, de Jong PA, Essed GG, Kanhai HH. The effects of oral carbohydrate administration on fetal acid base balance. Journal of Perinatal Medicine. 2002;30(5):400–4. doi: 10.1515/JPM.2002.062. [DOI] [PubMed] [Google Scholar]; *; Scheepers HCJ, de Jong PA, Essed GGM, Kanhai HHH. Carbohydrate solution intake during labour just before the start of the second stage: a double-blind study on metabolic effects and clinical outcome. BJOG: an international journal of obstetrics and gynaecology. 2004;111:1382–7. doi: 10.1111/j.1471-0528.2004.00277.x. [DOI] [PubMed] [Google Scholar]
- Shennan 2005 {published data only} .Shennan A, Thallon A, Hart D, O’Sullivan G. Caesarean section and calorific intake in labour; the influence of carbohydrate solution intake during labour. BJOG: an international journal of obstetrics and gynaecology. 2005;Vol. 112(issue 10):1454. doi: 10.1111/j.1471-0528.2005.00668.x. [DOI] [PubMed] [Google Scholar]
References to studies awaiting assessment
- Goodall 1999 {published data only} .Goodall U. RCT measuring the effects of eating and drinking in labour for low risk women. 1999. Personal communication.
- Laifer 2000 {published data only} .Laifer SA, Siddiqui DS, Do TP, Collins JE, Stiller RJ, Moffat SL, et al. A prospective randomized controlled trial of oral intake of liquids during the first stage of labor [abstract] Anesthesiology. 2000;92(Suppl):A53. [Google Scholar]
- Yiannouzis 1994 {published and unpublished data} .Yiannouzis K, Parnell C. The Midwifery Research Database: MIRIAD, A sourcebook of information about research in midwifery. 1st edition. Books for Midwives Press; Cheshire: 1994. Randomized controlled trial measuring the effects on labour of offering a light, low fat diet; pp. 196–8. Vol. [Google Scholar]
- Zhao 1996 {published data only} .Zhao H, Zhu S. The role of heng nai jin, a high energy liquid diet, in labor process. Chung-Kuo i Hsueh Ko Hsueh Yuan Hsueh Pao. 1996;18(4):310–1. [PubMed] [Google Scholar]
Additional references
- Am Soc Anesth 1999 .American Society of Anaesthesiologists Task Force Practice guidelines for preoperative fasting and the use of pharmacologic agents to reduce the risk of pulmonary aspiration: application to healthy patients undergoing elective procedures: a report by the American Society of Anesthesiologist Task Force on Preoperative Fasting. Anesthesiology. 1999;90(3):896–905. doi: 10.1097/00000542-199903000-00034. [DOI] [PubMed] [Google Scholar]
- Armstrong 2000 .Armstrong TS, Johnston IG. Which women want food during labour? Results of an audit in a Scottish DGH. Health Bulletin. 2000;58(2):141–4. [PubMed] [Google Scholar]
- Begum 1999 .Begum M. Fluid management in labour. In: Sengupta BS, Chattopadhyay SK, Thornton JG, Sengupta PS, editors. Obstetrics for Postgraduates and Practitioners. 1st Edition BI Churchill Livingstone Pvt Ltd; New Delhi: 1999. pp. 442–51. [Google Scholar]
- Broach 1988a .Broach J, Newton N. Food and beverages in labor. Part 1: cross-cultural and historic practices. Birt. h;1988:15–81. doi: 10.1111/j.1523-536x.1988.tb00811.x. [DOI] [PubMed] [Google Scholar]
- Broach 1988b .Broach J, Newton N. Food and beverages in labour. Part II: the effects of cessation of oral intake during labour. Birth. 1988;15:88–92. doi: 10.1111/j.1523-536x.1988.tb00813.x. [DOI] [PubMed] [Google Scholar]
- Champion 2002 .Champion P, McCormick C. Eating and drinking in labour. 1st Edition Books for Midwives; Oxford: 2002. [Google Scholar]
- Chang 1993 .Chang S. The psychological and physiological effects of ketonuria during labour [thesis] University of Michigan; Michigan: 1993. [Google Scholar]
- Davidson 1975 .Davidson JS. Gastric emptying in labor. Lancet. 1975;ii:227–8. doi: 10.1016/s0140-6736(75)90695-9. [DOI] [PubMed] [Google Scholar]
- Deeks 2001 .Deeks JJ, Altman DG, Bradburn MJ. Statistical methods for examining heterogeneity and combining results from several studies in meta-analysis. In: Egger M, Davey Smith G, Altman DG, editors. Systematic reviews in health care: meta-analysis in context. BMJ Books; London: 2001. [Google Scholar]
- Egger 1997 .Egger M, Smith GD, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–34. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foulkes 1985 .Foulkes J, Dumoulin J. The effects of ketonuria in labour. British Journal of Clinical Practice. 1985;91:59–62. [PubMed] [Google Scholar]
- Garcia 1985 .Garcia J, Garforth S, Ayers S. Midwifes confined? Labour ward policies and routines. Proceedings of the 1985 Conference on Research and the Midwife; Manchester, UK. 1985 November; University of Manchester; 1986. pp. 2–30. [Google Scholar]
- Gates 2005 .Gates S, the Editorial Team . Pregnancy and Childbirth Group. Issue 2. About The Cochrane Collaboration (Collaborative Review Groups (CRGs); 2005. Methodological Guidelines. [Google Scholar]
- Grylack 1984 .Grylack LJ, Chu SS, Scanlon JW. Use of intravenous fluids before caesarean section: effects on perinatal glucose, insulin and sodium homoeostasis. Obstetrics & Gynecology. 1984;63(5):654–8. [PubMed] [Google Scholar]
- Gyte 2006 .Gyte GML, Richens Y. Routine prophylactic drugs in normal labour for reducing gastric aspiration and its effects. Cochrane Database of Systematic Reviews. 2006;(Issue 3) doi: 10.1002/14651858.CD005298.pub2. [DOI: 10.1002/14651858.CD005298.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haire 1991 .Haire DB, Elsberry DD. Maternity care and outcomes in a high risk service: the North Central Bronx Hospital experience. Birth. 1991;18(1):33–7. doi: 10.1111/j.1523-536x.1991.tb00051.x. [DOI] [PubMed] [Google Scholar]
- Harbord 2006 .Harbord RM, Egger M, Sterne JA. A modified test for small-study effects in meta-analyses of controlled trials with binary endpoints. Statistics in Medicine. 2006;25:3443–57. doi: 10.1002/sim.2380. [DOI] [PubMed] [Google Scholar]
- Hart 2006 .Hart D. Eating an drinking during labour. In: Hall Moran V, Dykes F, editors. Maternal and infant nutrition and nurture: controversies and challenges. Quay Books; London: 2006. pp. 102–27. [Google Scholar]
- Higgins 2008 .Higgins JPT, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions. Version 5.0.1. The Cochrane Collaboration; [updated September 2008]. 2008. www.cochrane-handbook.org Available from. [Google Scholar]
- Johnson 1989 .Johnson C, Keirse MJNC, Enkin M, Chalmers I. Nutrition and hydration in labour. In: Chalmers I, Enkin M, Keirse MJNC, editors. Effective care in pregnancy and childbirth. Oxford University Press; Oxford: 1989. pp. 827–32. [Google Scholar]
- Ketteringham 1939 .Ketteringham BS, Austin BR. Induced hyperglycemia in delivery. American Journal of Obstetrics and Gynecology. 1939;37(6):1000–8. [Google Scholar]
- McCormick 2002 .McCormick C, Champion P. Cultural and historical perspectives on eating and drinking in labour. In: Champion P, McCormick C, editors. Eating and drinking in labour. 1st Edition Books for Midwives; Oxford: 2002. pp. 10–28. [Google Scholar]
- McKay 1988 .McKay S, Mahan C. Modifying the stomach contents of labouring women: why, how, with what success, and at what risk? How can aspiration of vomitus in obstetrics be prevented? Birth. 1988;15(4):213–21. [Google Scholar]
- McNabb 2002 .McNabb M. Changes in maternal food appetite and metabolism in labour and its effect on outcomes in active labor. In: Champion P, McCormack C, editors. Eating and drinking in labour. Books for Midwives; Oxford: 2002. pp. 46–110. [Google Scholar]
- Mendelson 1946 .Mendelson CL. The aspiration of stomach contents into the lungs during obstetric anaesthesia. American Journal of Obstetrics and Gynecology. 1946;52:191–205. doi: 10.1016/s0002-9378(16)39829-5. [DOI] [PubMed] [Google Scholar]
- Mendiola 1982 .Mendiola J, Grylack LJ, Scanlon JW. Effects of intrapartum maternal glucose infusion on the normal fetus and newborn. Anesthesia and Analgesia. 1982;61(1):32–5. [PubMed] [Google Scholar]
- Michael 1991 .Michael S, Reilly C, Caunt J. Policies for oral intake during labour: a survey of maternity units in England and Wales. Anaesthesia. 1991;46:1071–3. doi: 10.1111/j.1365-2044.1991.tb09928.x. [DOI] [PubMed] [Google Scholar]
- Micklewright 2002 .Micklewright A, Champion P. Labouring over food: the dietician’s view. In: Champion P, McCormick C, editors. Eating and drinking in labour. 1st Edition Books for Midwives; Oxford: 2002. pp. 29–45. [Google Scholar]
- NICE 2007 .National Collaborating Centre for Women’s and Children’s Health . Clinical Guideline 6. National Institute for Health and Clinical Excellence; London: [accessed 14 March 2009]. 2007. Intrapartum care: care for the healthy women and their babies during childbirth.http://www.nice.org.uk/guidance/index.jsp?action=download&o=36275 [Google Scholar]
- O’Sullivan 1994 .O’Sullivan G. The stomach: fact and fantasy: eating and drinking during labour. International Anesthesiology Clinics. 1994;32:31–44. [PubMed] [Google Scholar]
- Paranjothy 2004 .Paranjothy S, Liu E, Brown H, Thomas J. Drugs at caesarean section for preventing nausea, vomiting and aspiration pneumonitis. Cochrane Database of Systematic Reviews. 2004;(Issue 4) [DOI: 10.1002/14651858.CD004943] [Google Scholar]
- Pengelley 1998 .Pengelley L, Gyte G. Eating and drinking in labour (V) An update of the NCT briefing paper. Practising Midwife. 1998;1:26–8. [PubMed] [Google Scholar]
- Pengelley 2002 .Pengelley L. Eating and drinking in labour: the consumer’s view. In: Champion P, McCormick C, editors. Eating and drinking in labour. 1st Edition Books for Midwives; Oxford: 2002. pp. 111–23. [Google Scholar]
- Peters 2006 .Peters JL, Sutton AJ, Jones DR, Abrams KR, Rushton L. Comparison of two methods to detect publication bias in meta-analysis. JAMA. 2006;295:676–80. doi: 10.1001/jama.295.6.676. [DOI] [PubMed] [Google Scholar]
- Pipkin 2001 .Pipkin FB. Maternal physiology in pregnancy. In: Chamberlain G, Steer PJ, editors. Turnbull’s Obstetrics. 3rd Edition Churchill Livingstone; Edinburgh: 2001. pp. 71–91. [Google Scholar]
- RevMan 2008 .The Nordic Cochrane Centre. The Cochrane Collaboration . Review Manager (RevMan). 5.0. The Nordic Cochrane Centre, The Cochrane Collaboration; Copenhagen: 2008. [Google Scholar]
- Rooks 1989 .Rooks J, Weatherby N, Ernst E, Stapleton S, Rosen D, Rosenfield A. Outcomes of care in birthcentres: the National Birth Centre Study. New England Journal of Medicine. 1989;321:1804–11. doi: 10.1056/NEJM198912283212606. [DOI] [PubMed] [Google Scholar]
- Simkin 1986a .Simkin P. Stress, pain, and catecholamines in labor: Part 1. A Review. Birth. 1986;13:227–33. doi: 10.1111/j.1523-536x.1986.tb01053.x. [DOI] [PubMed] [Google Scholar]
- Simkin 1986b .Simkin P. Stress, pain, and catecholamines in labour: Part 2. Stress associated with childbirth events: a pilot survey of new mothers. Birth. 1986;13:234–40. doi: 10.1111/j.1523-536x.1986.tb01054.x. [DOI] [PubMed] [Google Scholar]
- Taylor 1975 .Taylor G. Acid pulmonary aspiration syndrome after antacids. British Journal of Anaesthesia. 1975;47:615–7. doi: 10.1093/bja/47.5.615. [DOI] [PubMed] [Google Scholar]
- Williamson 1971 .Williamson DH. Physiological ketosis, or why ketone bodies? Postgraduate Medical Journal. 1971;47(Suppl):371–5. [PubMed] [Google Scholar]
- * Indicates the major publication for the study