Abstract
African Americans have exceptionally high rates of hypertension and hypertension related complications. It is commonly reported that the blood pressure lowering efficacy of renin angiotensin system (RAS) inhibitors is attenuated in African Americans due to a greater likelihood of having a low renin profile. Therefore these agents are often not recommended as initial therapy in African Americans with hypertension. However, the high prevalence of comorbid conditions, such as diabetes, cardiovascular and chronic kidney disease makes treatment with RAS inhibitors more compelling. Despite lower circulating renin levels and a less significant fall in blood pressure in response to RAS inhibitors in African Americans, numerous clinical trials support the efficacy of RAS inhibitors to improve clinical outcomes in this population, especially in those with hypertension and risk factors for cardiovascular and related diseases. Here, we discuss the rationale of RAS blockade as part of a comprehensive approach to attenuate the high rates of premature morbidity and mortality associated with hypertension among African Americans.
Keywords: African American, Blood pressure, Ethnicity, Hypertension, Renin, Angiotensin
Core tip: African Americans have exceptionally high rates of hypertension and hypertension related complications. Due to a greater likelihood of having a low plasma renin levels, inhibitors of the renin angiotensin system (RAS) are often not recommended as initial antihypertensive therapy. However, animal models suggest hypertension characterized by low circulating renin levels have a paradoxical increase in tissue RAS activity. Thus treatment with RAS inhibitors may be critical to preventing end organ damage. We describe the rationale of RAS blockade as part of a comprehensive approach to attenuate the high rates of premature morbidity and mortality associated with hypertension among African Americans.
INTRODUCTION
Hypertension is characterized by a persistent and frequently progressive elevation in blood pressure[1]. The level of systolic blood pressure (SBP) and/or diastolic blood pressure (DBP), which connotes a diagnosis of hypertension, may vary depending on the presence or absence of coexisting comorbidities[1,2]. Hypertension is commonly defined as physician diagnosed SBP ≥ 140 mmHg and DBP ≥ 90 mmHg; and pre-hypertension is defined as SBP ≥ 120 mmHg and < 140 mmHg or DBP ≥ 80 mmHg and < 90 mmHg[1]. However, a recent report from the panel members appointed to the Eighth Joint National Committee recommended a SBP goal of < 150 mmHg and DBP goal < 90 mmHg in persons ≥ 60 years of age without diabetes mellitus (DM) or chronic kidney disease (CKD)[3]. The committee also recommended that in the African American hypertensive population, including those with diabetes, initial therapy should begin with a calcium channel blocker or thiazide-type diuretic, but acknowledged that there was modest evidence for renin-angiotensin system (RAS) inhibition as initial or add-on antihypertensive therapy in African Americans with CKD[3]. However, a minority of the committee members did not support a higher SBP goal at age ≥ 60 years or the choices for initial antihypertensive therapy in African Americans. They were particularly concerned that the newly recommended higher blood pressure goal may adversely affect patients aged ≥ 60 years with cardiovascular disease (CVD) risk factors other than DM or CKD[4]. In addition, they interpreted the evidence supporting an increase in the SBP target from < 140 mmHg to < 150 mmHg in persons 60 years of age or older as insufficient and inconsistent with the evidence supporting the panel’s recommendations for an SBP target of < 140 mmHg in younger persons[4]. There is also the risk that as the new guidelines are disseminated that the “take home” messages may miss the nuances such as the new recommended higher goal in persons ≥ 60 years of age excluding persons with DM or CKD, and that with a goal of < 150 mmHg many patients may spend much of their time with their blood pressure (BP) above that level.
In the United States African Americans develop hypertension at an earlier age than whites, have much higher average blood pressure readings, a greater likelihood of refractory hypertension, and greater rates of premature hypertensive complications such as CKD, stroke and heart disease[5-7]. Importantly, African Americans suffer from a three-fold higher death rate from hypertension with cardiovascular complications accounting for the majority of deaths[5]. Data from the National Health and Nutrition Examination Survey (NHANES) indicate/show that although there has been a trend toward improving BP control among African Americans, the overall control of BP remains suboptimal at a national level. During the period from 1999-2004, blood pressure was adequately controlled in only 35% of whites, 29% of African Americans, and 27% of Hispanics[8]. By 2007-2008 blood pressure control rates had increased to 50% and for the first time were now similar across race/ethnicity[9]. While this is a marked improvement, it still represents half of Americans with hypertension having poor blood pressure control. However other data underscore the lingering concern for BP control as a significant problem among African Americans. In the Multi-Ethnic Study of Atherosclerosis (MESA) the percentage of treated but uncontrolled hypertension was significantly higher in African Americans (35%), Chinese (33%), and Hispanics (32%) than in whites (24%)[10]. After adjustment for clinical and socioeconomic factors the relative higher rates of uncontrolled hypertension for Chinese and Hispanic participants largely disappeared, but persisted in the African American population, suggesting an independent effect to account for these observed differences such as other biologic and/or non-biologic factors not assessed in the MESA study[10].
BIOLOGIC FACTORS INFLUENCING HYPERTENSION IN AFRICAN AMERICANS
The reasons for the exceptionally high rates of hypertension and associated end organ damage among African Americans, are not entirely clear but likely include socioeconomic status, lifestyle choices, clinical factors (e.g., increased risks of diabetes and hypertension), environmental, and biologic/genetic factors that may contribute broadly to racial/ethnic differences in not only outcomes but in response to therapeutic intervention[11-17]. Our understanding of the complex factors that predispose African Americans to hypertension and hypertension-related complications is evolving. While RAS inhibition has emerged as an important antihypertensive therapy, the blood pressure lowering efficacy of RAS inhibitors is attenuated in African Americans[18], likely due to an increased prevalence of having a low renin profile. Therefore these agents may be less likely to be recommended as initial therapy in African Americans with hypertension[3].
One major biologic factor that is highly relevant to RAS inhibition is the particularly high prevalence of salt sensitivity in African Americans, which is also associated with a low circulating plasma renin profile[19-26]. Salt sensitivity is defined as an increase in blood pressure in response to sodium or salt intake, and is commonly associated with a low circulating renin profile[24,27,28]. Wilson et al[29] postulated that the slave trade from Africa to the Americas led to extreme volume depletion and cardiovascular collapse during the journey due to diarrheal diseases and limited access to water favoring the survival of persons who were avid sodium retainers and accelerating gene selection for sodium retention. Consistent with this premise, Maseko et al[30] found no relationship between blood pressure and 24-h urinary sodium and potassium excretion rates in nearly 300 city-dwelling black South Africans, suggesting African slave descendants have a higher rate of salt sensitivity than native black Africans. However many of the historical claims which form the basis of the slavery hypothesis for hypertension have been challenged by other authors[29,31]. In question are the key tenets of the theory which implicate salt deficiency in the areas of Africa from which slaves originated, the trauma of the slave trade, and conditions in the Americas as triggers for unnatural genetic selection for renal sodium-retainers[29,32]. According to this theory, these factors collectively evolved into the eventual present-day disproportionately higher blood pressure in African Americans compared with their counterpart whites or African blacks in modern sodium-rich societies[29,31,32]. In the absence of access to records of past salt availability in Africa, slave trade disease states, and dietary salt content in the Americas between the 16th and 19th century, it will be impossible to ever fully confirm or fully refute this hypothesis[33]. However, others have refuted the slave trade hypothesis as contributing to excess rates of hypertension in blacks of African descent[31].
Some specific features of salt-sensitive hypertension have been characterized. Evidence suggests that the sympathetic nervous system may play a role in the modulation of salt sensitivity. In the presence of salt loading, the typical response of the sympathetic nervous system is to decrease norepinephrine, a known sodium retainer, and to increase dopamine which promotes sodium excretion[34,35]. However in salt sensitive hypertensive individuals, particularly African Americans, there appears to be a dysfunctional response of the sympathetic nervous system in the presence of excess salt. In these patients, salt loading is associated with decreased urinary dopamine levels and the absence of any significant decreases in norepinephrine[35,36]. Another factor which is implicated as accounting for differences in salt sensitivity is kallikrein which has been demonstrated to be excreted in lower levels in salt sensitive hypertensive persons, particularly African Americans[37,38]. Whether the fact that African Americans consume less potassium, a known kallikrein releaser, is contributory, remains unclear[24,37]. Evaluation of the relationship between potassium intake, urinary kallikrein levels and salt sensitivity is warranted using large scale clinical trials.
Among the non-biologic factors which could possibly explain the inequity in the occurrence of hypertension among the races, obesity remains a tempting option given its similar trend of increased prevalence in African Americans. Excess adiposity, a reflection of lifestyle habits, has a reported 51% greater prevalence in this population. In addition, there has been an association reported between obesity, insulin resistance and other adipokine-mediated pathways with the occurrence of salt-sensitivity[35]. However, NHANES data from the 1988-1994 time period show no significant difference in obesity between white men and black men (20.3% and 21.1% respectively) while there was a 46% greater prevalence of hypertension in black men over white men during that same period[39]. Also, Okosun et al[40] have shown that the risk of African American race for high blood pressure remains after adjusting for abdominal adiposity in NHANES III data. Therefore the evidence does not support obesity as the sole contributor to the disparity in prevalence of hypertension among blacks although it is not excluded as a contributory factor.
RENIN-ANGIOTENSIN SYSTEM IN SALT SENSITIVE HYPERTENSION
The disproportionate burden of both hypertension and its sequelae in African Americans underscores the significance of optimizing approaches to blood pressure control in order to prevent and/or attenuate the high rate of hypertensive complications. A comprehensive understanding of the role of RAS blockade as part of a strategy for blood pressure control and attenuation of end organ disease is critical to selecting therapeutic agents which might reverse hypertension related premature morbidity and mortality. The RAS system is an important modulator of blood pressure and vascular function/disease.
As noted above, in addition to an increase in the prevalence of salt sensitivity, an increased prevalence of reduced plasma renin levels have been noted in hypertensive African Americans and Caribbean Hispanics[41-43]. Given the documented role of RAS in the progression of vascular disease, an attenuated risk of hypertension-related end-organ damage might also be expected in patients with low-renin hypertension. Paradoxically, however, many such individuals experience high rates of hypertension-related end-organ complications suggesting RAS may still be important at the tissue level in patients with reduced plasma renin levels.
Much of our understanding of the role of the RAS in blood-pressure regulation at a tissue level derives from Dahl salt-sensitive and salt-resistant rat studies as a model of human salt-sensitive, low renin hypertension that is more commonly noted in African Americans[44]. Consumption of a high-salt diet by Dahl salt-sensitive rats results in hypertension and early onset of renal injury and dysfunction. This is associated with reduced plasma renin activity and angiotensinogen (ATG) concentrations, but accompanied by paradoxical elevations of renal tissue angiotensin (Ag) II, tissue Ag II receptor 1 (AT1) receptor expression and urinary ATG excretion as well as oxidative stress and activation of NAD(P)H oxidase in the kidney and cardiovascular tissues[45,46]. These observations support a dissociation of low circulating RAS from the upregulated intrarenal and tissue RAS in this model. Despite the low circulating renin level, RAS blockade in Dahl salt-sensitive rats fed a high-salt diet reversed endothelial dysfunction, attenuated proteinuria and reduced cardio-renal injury even though it did not normalize the blood pressure supporting a blood pressure independent RAS related effect[47]. These findings suggest that the intrarenal and tissue RAS may be more important in the pathogenesis of salt sensitive hypertension and hypertensive nephropathy than the circulating RAS, which may be more reflective of the regulation of sodium balance, vascular resistance and arterial blood pressure.
The efficacy of RAS inhibition has also been shown to be widely effective in many other animal models of hypertension, including but not limited to hypertension in obese Zucker rats animals with renal mass reduction[48,49] and rodents with hypertensive nephropathy induced by Ag II infusion[50]. Osteopontin (OPN), which is a secreted matrix glycoprotein that is expressed in Ag II-injured tissues, is an important modulator of several of the Ag II-induced mechanisms of hypertensive nephropathy. Global deletion of OPN in hypertensive, albuminuric mice promoted Ag II-induced monocyte chemoattractant protein-1, NADPH oxidase subunits (NOX2, gp47phox and NOX4) and plasminogen activator inhibitor-1, compared to Ag II-infused wild-type mice[50]. Also, inhibition of OPN expression may account for a mechanism by which Ag II blockade attenuated renal injury following renal ablation[51], consistent with OPN modulating the effects of Ag I converting enzyme (ACE) inhibitor therapy in hypertensive nephropathy. Finally, Gonzalez-Villalobos et al[52] recently demonstrated that the absence of kidney ACE substantially blunts the hypertension induced by Ag II infusion or nitric oxide synthesis inhibition. Moreover, in mice that lack kidney ACE the renal responses to high serum Ag II such as intrarenal Ag II accumulation, sodium and water retention, and activation of transporter activating kinases Ste20-related proline alanine-rich kinase and oxidative stress response kinase were effectively prevented. These findings led them to conclude that renal ACE activity is required to increase local Ag II to stimulate sodium transport and induce hypertension[52]. These findings are consistent with the importance of inhibition of intrarenal RAS to attenuate hypertension and its sequelae.
In summary, hypertension has been reported in animal models to induce glomerular hypertension and glomerular hyperfiltration, oxidative stress, inflammation, endothelial damage due to enhanced traffic of plasma proteins and/or increased translational and shear forces and other that may lead to worsening vascular disease, CKD and worsening blood pressure[53,54]. Ag II is one of the more extensively studied mediators of vascular function. Ag II upregulates transforming growth factor-β1, tumor necrosis factor-α, nuclear factor-κB, OPN, several adhesion molecules and chemoattractants, and more recently, interactions with adiponectin and select microRNAs which together conspire to promote renal inflammation and fibrosis[55,56]. The documented role of RAS in the progression of end organ damage has positioned it as a prime therapeutic target in high-risk patients. RAS blockade can reduce blood pressure, reverse endothelial dysfunction, attenuate proteinuria, and reduce renal injury independent of blood pressure changes[47]. The paradoxical increase in tissue RAS in salt sensitive, low renin hypertension makes RAS blockade an important therapeutic option for treating African Americans and other patients with hypertension and a circulating low renin profile[44].
GENETIC POLYMORPHISMS, THE RAS, AND HYPERTENSION IN AFRICAN AMERICANS
At the level of the kidney the pool of intra-renal RAS is upregulated in CKD independently of systemic RAS. This pathological upregulation of the intra-renal RAS is marked by simultaneous increases in the AT1 expression and the number of the Ag II-producing cells, many of which are macrophages and serve as ectopic sources of angiotensin[57]. In addition, the kidney not only contains ATG, angiotensin converting enzyme, and renin, but is a recipient of their physiological and pathophysiological actions[58,59]. In CKD, AT1 receptor activation by Ag II raises superoxide production via upregulation of NAD(P)H oxidase, and inhibits Nrf2 expression, which is the master regulator of genes encoding many antioxidant and cytoprotective enzymes and related molecules[60-62]. This may be an important mechanism of action through which intra-renal RAS promotes, oxidative stress, inflammation and subsequent tissue damage and dysfunction in animals, and likely humans with CKD and/or hypertension.
Several genetic variations (e.g., promoter region variants of the ATG gene) have been identified which may contribute to ethnic disparities in salt-sensitive hypertension and response to RAS blockade. Tiago et al[63] reported a marked influence of homozygosity for the -20A allele (n = 399) of the ATG on the relationship between body mass index and systolic blood pressure (r = 0.23; P < 0.0001) in over 1000 South Africans of African ancestry. More specific to the response to RAS inhibition, the African-American Study of Kidney Disease and Hypertension (AASK) study showed that African Americans who were homozygous for the ACE polymorphism 12269G > A experienced a more rapid reduction in blood pressure following ACE inhibition than those who were heterozygous for this variant (P < 0.001), but blood pressure response to calcium channel blockers did not vary by ACE polymorphism variants[64]. Similarly, ATG promoter region variants among a cohort of South Africans of African ancestry influenced the blood pressure response to an Angiotensin converting enzyme inhibitor (ACEI), but not to a calcium channel blocker[65]. Recent genome-wide admixture mapping studies have demonstrated genetic variation in the regions of MYH9 and APOL 1 on chromosome 22 that have been estimated to explain over 50% of the difference in the rates of non-diabetic end-stage renal disease (ESRD) between white and black Americans[13,66-69], but to date no reports have linked these gene variants to response to RAS inhibition therapy. Limited data exist for the study of ACE polymorphism variants in animal models of high BP. One report suggested a locus for the inducible, but not a constitutive, nitric oxide synthase cosegregated with blood pressure in the Dahl salt-sensitive rat[70], while microsatellite of ACE was reported to be associated with the development of salt-sensitive hypertension in the stroke-prone spontaneously hypertensive rat[71].
TREATMENT TRIALS OF RAS INHIBITION IN AFRICAN AMERICANS
Most clinical trials of RAS inhibition as primary antihypertensive therapy in African Americans have been directed toward patients with diabetes, CKD, and/or high CVD risk. A summary of select trials of RAS inhibition as primary antihypertensive therapy in African Americans follows.
Diabetes
The Collaborative Study Group was the first major study to examine the efficacy of ACEI in slowing the progression of CKD in 409 participants with type 1 diabetes[72], and while it demonstrated efficacy in comparison to usual care, the study included only 15 African Americans. Two subsequent major studies of RAS inhibition in persons with diabetic nephropathy, most of whom had hypertension, were the irbesartan (IDNT) and losartan (RENAAL) trials. These two trials both showed efficacy for ARB therapy and included higher proportions of ethnic minorities than most earlier studies with 13% African Americans and 5% Hispanics in the former and 15% African Americans and 18% Hispanics in the latter[73,74]. Although not powered to perform subgroup analyses according to ethnicity, these studies strongly suggest that the positive outcomes of RAS inhibition extended to all study participants. Moreover, a post-hoc analysis of RENAAL found no ethnic differences in the relationship of baseline albuminuria or 6-mo antiproteinuric response to therapy to ESRD risk, or the overall renoprotective effect of ARB therapy (1513 participants followed for 3.4 years with final SBP of 141 mmHg)[75].
CKD
The AASK is the largest prospective CKD study to focus on African Americans to date[76,77]. The AASK trial (n = 1094) was a randomized controlled study that examined the effects of three classes of initial antihypertensive therapy (ACEI, β-blocker or calcium channel blocker) and two levels of blood pressure control: intensive (≤ 120/80 mmHg) and standard (approximately 135-140/85-90 mmHg) on the progression of renal function and clinical outcomes in a high-risk cohort with hypertension-related CKD[78]. Diuretics were not among the three randomized classes of antihypertensive agents as it was assumed the majority of study participants would require diuretic therapy due to their impaired renal function and associated volume retention and therefore the majority of study participants would require diuretics, allowing the design to most closely emulate clinical practice. Indeed, nearly 90% of AASK participants required adjunct diuretic therapy to achieve target blood pressure levels. While calcium channel blockers were the most commonly prescribed antihypertensive for African Americans with CKD due to their blood pressure lowering efficacy, the calcium channel blocker arm of AASK was terminated early because of increased rates of adverse clinical events[76]. AASK demonstrated that clinical cardio-renal outcomes in African Americans: were improved with ACEI in comparison to β-blocker or calcium channel blocker, with diuretics and other agents added as needed[79]. While outcomes did not initially differ between intensive (≤ 120/80 mmHg) and standard (approximately 135-140/85-90 mmHg) BP targets[79], longer term follow up (8.8 to 12.2 years) in the intensive control group (mean blood pressure was 130/78 mmHg) compared to standard care (141/86 mmHg) revealed a benefit in patients with baseline protein-to-creatinine ratio > 0.22 (equivalent to baseline protein excretion of > 300 mg/d), but not the overall cohort[80].
Importantly, AASK demonstrated that blood pressure can be controlled in African Americans with CKD and that combined clinical outcomes (cardiovascular and renal) can be improved by using not only a beta blocker or calcium channel blocker but an ACEI as initial therapy to reach a usual or strict blood-pressure target, with diuretics in most, and other agents added as needed[71], and that ACEI therapy led to the best clinical outcomes[79]. This contrasts previous[7] and more recent suggestions[3] that RAS inhibition is of limited benefit in African Americans. The notion of the overall efficacy of RAS inhibition is further supported by a recent meta-analysis of 25 randomized controlled trials (n = 45758) by Balamuthusamy et al[81] who found improved or equivalent CVD outcomes in patients with diabetic or non-diabetic CKD and proteinuria treated with RAS blockade (ACEI/ARB) in comparison to placebo and control (β-blocker, calcium-channel blockers and other antihypertensive-based therapy). While the ethnic composition of the meta-analysis was not provided, the preponderance of evidence supports the important role for RAS blockade in treating patients with CKD and proteinuria, including African Americans. Secondary analysis of Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT) trial, detailed below, showed that in patients with reduced renal function, RAS inhibition with lisinopril was equally as effective as amlodipine and chlorthalidone in reducing the rate of development of ESRD[82].
HIGH CARDIOVASCULAR RISK
The Heart Outcomes Prevention Evaluation trial assessed the effectiveness of RAS inhibition in nearly 10000 high-risk patients (55 years of age or older) who had evidence of vascular disease or diabetes plus one other cardiovascular risk factor, but no evidence of over health failure[83]. They found ramipril significantly reduced the rates of death, myocardial infarction, and stroke[83]. Unfortunately, the racial/ethnic composition of the study cohort was not described. Another key hypertension trial, which included ACEI therapy and a large percentage of minority participants, was the ALLHAT. ALLHAT enrolled nearly 34000 high-risk hypertensive patients, of whom 32% were black and 16% were Hispanic[84]. ALLHAT analysis found that first-line therapy with chlorthalidone, amlodipine or lisinopril were similar in efficacy for preventing cardiovascular events[85]. First-line therapy with alpha blockade was not as effective and the alpha blocker arm was discontinued early[86]. Subgroup analysis by race/ethnicity revealed no significant differences by class of therapy from that of the overall trial results. Unfortunately the second line drug for both amlodipine and lisinopril could not be a diuretic or a complementary RAS inhibitor or calcium channel blocker, respectively, so the design limited the ability of ALLHAT to test common clinical practice and practice guidelines. This is especially important for African Americans with high blood pressure who are more likely to require 2 or 3 drugs to achieve blood pressure goal and especially important for RAS inhibition which has been most effective in combination with a diuretic when a second agent is needed[79]. Many authorities still favor initial therapy with RAS blockade, especially in patients with hypertension complicated by diabetes, CKD, or CVD where diuretics are commonly included in treatment[1,2,87]. Secondary analysis of ALLHAT showed that in patients with reduced renal function, RAS inhibition with lisinopril was equally as effective as amlodipine and chlorthalidone in reducing the rate of development of ESRD[82].
SPECIAL CONSIDERATIONS FOR THE USE OF ACEI IN TREATING HYPERTENSION IN AFRICAN AMERICANS
Consideration is necessary of the well-recognized common effects noted with some agents that inhibit the RAS system. ACEI related adverse events are relatively common in African Americans and Chinese[88-90]. Of note, the rate of angioedema in blacks is three times that of non-blacks[88,89], and the rate of ACEI discontinuation due to cough is also very high[91]. Possible mechanisms which could account for the increased incidence of ACEI-related adverse effects in African Americans are angiotensin-converting enzyme and bradykinin gene polymorphisms as have been demonstrated in East Asians[92]. Also notable is that the initiation of RAS inhibition therapy can lead to an acute reduction in renal function regardless of racial/ethnic background, especially in patients with advanced CKD. In most instances this reduction in glomerular filtration rate is a potentially reversible physiologic hemodynamic effect and a modest initial fall in renal function may be a predictor of long-term renoprotection[93,94]. However, close care is required to avoid complications such as hyperkalemia or hypotension, which may occur in some patients with deteriorating renal function and would warrant discontinuation.
The optimism for enhanced efficacy of RAS inhibition by using a combination of ARB/ACE inhibitor has recently dampened. The ONTARGET trial followed 25000 participants (11% Aboriginal/African) with diabetes with end-organ damage (13% with microalbuminuria) or vascular disease for 4.5 years, randomized to Ramipril group (ACE inhibitor), Telmisartan group (ARB), or both[95]. They found no difference in the composite outcome of cardiovascular events including death or hospitalization for heart failure between groups, and at trend toward increased cardiovascular events in the group receiving combination ARB/ACEI[95].
TARGET BLOOD PRESSURE IN AFRICAN AMERICANS
The 2007 European Society of Hypertension (ESH) and the European Society of Cardiology (ESC) Guidelines recommended two distinct BP targets: 140/90 mmHg in low-moderate risk hypertensive individuals and 130/80 mmHg in high-risk hypertensive persons (e.g., those with diabetes, cerebrovascular, cardiovascular, or renal disease)[96]. The 2013 ESH and the ESC Guidelines recommend a blood pressure target of 140/90 mmHg regardless of risk, with a less stringent SBP goal of between 150 and 140 mmHg in the elderly[2]. They found no evidence to support a lower blood pressure goal (130/80 mmHg) in patients with diabetes or a history of cardiovascular or renal disease[2]. This was not specific to any racial/ethnic group. This lack of support for a lower blood pressure goal is further supported by the results of the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial[97]. The ACCORD trial, which included 19.3% black participants with type 2 DM, used both ACEI and ARB as part of antihypertensive therapeutic approach and found no benefit in regards to major cardiovascular events with a SBP target of < 120 vs < 140 mmHg . This is also consistent with the findings mentioned earlier from the AASK trial, which found no differences in clinical outcomes between intensive (≤ 120/80 mmHg) and standard (approximately 135-140/85-90 mmHg) blood pressure targets[79]. However, the trend toward improved outcomes at a lower blood pressure in patients with elevated baseline protein-to-creatinine ratio > 0.22 (equivalent to baseline protein excretion of > 300 mg/d)[80], suggests further studies are needed to assess the benefit of a lower target blood pressure in higher risk groups such as African Americans with target organ damage. In fact, based on this and other secondary analyses, the International Society on Hypertension in Blacks consensus statement suggested a BP target of < 130/80 mmHg in hypertensive blacks with target organ damage[87], although others suggest the data to support such a recommendation is still insufficient[98].
One of the obstacles to attaining target blood pressure goals in African Americans is the issue of medication adherence. Low medication adherence rates and higher rates of uncontrolled blood pressures are more common in African Americans[33,99,100]. Keys to the effectiveness of ACEI therapy is adherence to both pharmacologic and non-pharmacologic therapy particularly in African Americans whose lower adherence rates have been attributable to both patient-related and physician-related factors including medication cost, insurance issues and access to healthcare. Aggressive measures are required to target interventions such as patient education focused on patient misconceptions regarding hypertension, home visits by trained community health workers, culturally appropriate storytelling, home blood pressure monitoring and behavioral counseling-all of which have been associated with improved medication adherence and decreased blood pressure measurements in blacks[100,101].
CONCLUSION
Health care providers currently consider a patient’s age, gender and ethnic background when making clinical decisions based on the evidence from clinical research findings. As in the non-African American hypertensive patient, first excluding primary renal or secondary causes of hypertension, then establishing a comprehensive treatment approach, is paramount to slowing the progression of end organ damage (Figure 1). Aggressive treatment of the primary etiology, addressing select lifestyle and socio-cultural issues, and the use of two or more antihypertensive agents for control of blood pressure is typically required in African American patients[102]. The existing data highly supports the inclusion of RAS blockade agents as initial therapy for African Americans with hypertension. In fact, the data from animal models of salt sensate, low renin hypertension suggest RAS blockade may be even more imperative in treating African Americans with hypertension than the general population. These agents appear to confer additional end organ protection beyond that offered by other antihypertensive agents in this patient subgroup. Importantly, there is no evidence of reduced efficacy for ACEI or ARB therapy on clinical outcomes in African Americans[103].
Figure 1.
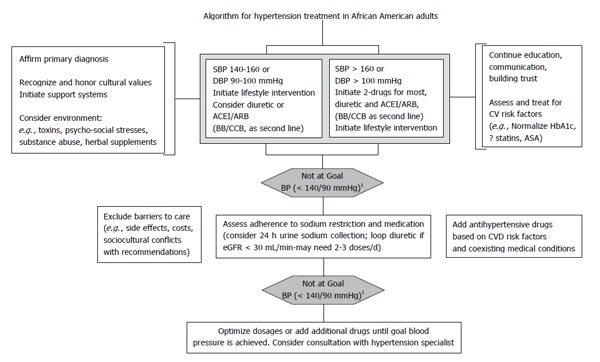
Algorithm for hypertension treatment in African American adults (adapted from ref. [86]). 1For persons > 60 years of age consider < 150/100 mmHg if initial SBP is > 160 mmHg[2]. ACEI: Angiotensin converting enzyme inhibitor; ARB: Angiotensin receptor blocker; BB: Beta blocker; CCB: Calcium channel blocker; SBP: Systolic blood pressure; CVD: Cardiovascular disease; DBP: Diastolic blood pressure; eGFR: Estimated glomerular filtration rate; CV: Cardiovascular; ASA: Aspirin; ?: Possibly the use of statins or aspirin or possible use of statins or aspirin; BP: Blood pressure.
In conclusion, the overall treatment plan should be guided by individual patient response, coexisting risk factors and potential sociocultural considerations such as cost of medications and insurance coverage, which affect adherence to both pharmacologic and non-pharmacologic interventions[14]. In all racial groups, blood pressure target goals in uncomplicated hypertension based on clinical trial data is < 140/90 mmHg with debate over a lower target (< 130/80 mmHg) in cases with end organ damage due to the data mostly being surrogate markers with a lack of consistent hard outcome data. Further elucidation of the optimal treatment for hypertension may be provided by the ongoing NIH-funded Systolic Blood Pressure Intervention Trial trial which will include a diverse patient population in regards to gender, race/ethnicity and comorbidities in over 7500 persons over 55 years of age[104].
Footnotes
Supported by UL1TR000124, P30AG021684, P20-MD000182 and DK065455, National Institutes of Health
P- Reviewer: Efstathiou S, Ozdemir S, Shimada Y S- Editor: Wen LL L- Editor: A E- Editor: Liu SQ
References
- 1.Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL, Jones DW, Materson BJ, Oparil S, Wright JT, et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension. 2003;42:1206–1252. doi: 10.1161/01.HYP.0000107251.49515.c2. [DOI] [PubMed] [Google Scholar]
- 2.ESH/ESC Task Force for the Management of Arterial Hypertension. 2013 Practice guidelines for the management of arterial hypertension of the European Society of Hypertension (ESH) and the European Society of Cardiology (ESC): ESH/ESC Task Force for the Management of Arterial Hypertension. J Hypertens. 2013;31:1925–1938. doi: 10.1097/HJH.0b013e328364ca4c. [DOI] [PubMed] [Google Scholar]
- 3.James PA, Oparil S, Carter BL, Cushman WC, Dennison-Himmelfarb C, Handler J, Lackland DT, LeFevre ML, MacKenzie TD, Ogedegbe O, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8) JAMA. 2014;311:507–520. doi: 10.1001/jama.2013.284427. [DOI] [PubMed] [Google Scholar]
- 4.Wright JT, Fine LJ, Lackland DT, Ogedegbe G, Dennison Himmelfarb CR. Evidence supporting a systolic blood pressure goal of less than 150 mm Hg in patients aged 60 years or older: the minority view. Ann Intern Med. 2014;160:499–503. doi: 10.7326/M13-2981. [DOI] [PubMed] [Google Scholar]
- 5.Go AS, Mozaffarian D, Roger VL, Benjamin EJ, Berry JD, Borden WB, Bravata DM, Dai S, Ford ES, Fox CS, et al. Heart disease and stroke statistics--2013 update: a report from the American Heart Association. Circulation. 2013;127:e6–e245. doi: 10.1161/CIR.0b013e31828124ad. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bibbins-Domingo K, Pletcher MJ, Lin F, Vittinghoff E, Gardin JM, Arynchyn A, Lewis CE, Williams OD, Hulley SB. Racial differences in incident heart failure among young adults. N Engl J Med. 2009;360:1179–1190. doi: 10.1056/NEJMoa0807265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Calhoun DA, Booth JN, Oparil S, Irvin MR, Shimbo D, Lackland DT, Howard G, Safford MM, Muntner P. Refractory hypertension: determination of prevalence, risk factors, and comorbidities in a large, population-based cohort. Hypertension. 2014;63:451–458. doi: 10.1161/HYPERTENSIONAHA.113.02026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ong KL, Cheung BM, Man YB, Lau CP, Lam KS. Prevalence, awareness, treatment, and control of hypertension among United States adults 1999-2004. Hypertension. 2007;49:69–75. doi: 10.1161/01.HYP.0000252676.46043.18. [DOI] [PubMed] [Google Scholar]
- 9.Egan BM, Zhao Y, Axon RN. US trends in prevalence, awareness, treatment, and control of hypertension, 1988-2008. JAMA. 2010;303:2043–2050. doi: 10.1001/jama.2010.650. [DOI] [PubMed] [Google Scholar]
- 10.Kramer H, Han C, Post W, Goff D, Diez-Roux A, Cooper R, Jinagouda S, Shea S. Racial/ethnic differences in hypertension and hypertension treatment and control in the multi-ethnic study of atherosclerosis (MESA) Am J Hypertens. 2004;17:963–970. doi: 10.1016/j.amjhyper.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 11.Tarver-Carr ME, Powe NR, Eberhardt MS, LaVeist TA, Kington RS, Coresh J, Brancati FL. Excess risk of chronic kidney disease among African-American versus white subjects in the United States: a population-based study of potential explanatory factors. J Am Soc Nephrol. 2002;13:2363–2370. doi: 10.1097/01.asn.0000026493.18542.6a. [DOI] [PubMed] [Google Scholar]
- 12.Norris K, Nissenson AR. Race, gender, and socioeconomic disparities in CKD in the United States. J Am Soc Nephrol. 2008;19:1261–1270. doi: 10.1681/ASN.2008030276. [DOI] [PubMed] [Google Scholar]
- 13.Freedman BI, Murea M. Target organ damage in African American hypertension: role of APOL1. Curr Hypertens Rep. 2012;14:21–28. doi: 10.1007/s11906-011-0237-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Martins D, Norris K. Hypertension treatment in African Americans: physiology is less important than sociology. Cleve Clin J Med. 2004;71:735–743. doi: 10.3949/ccjm.71.9.735. [DOI] [PubMed] [Google Scholar]
- 15.Norris K, Francis C. Gender and ethnic differences and considerations in cardiovascular risk assessment and prevention in African Americans. Practical Strategies Pre Heart Dis: 2004. pp. 415–440. [Google Scholar]
- 16.Suthanthiran M, Li B, Song JO, Ding R, Sharma VK, Schwartz JE, August P. Transforming growth factor-beta 1 hyperexpression in African-American hypertensives: A novel mediator of hypertension and/or target organ damage. Proc Natl Acad Sci USA. 2000;97:3479–3484. doi: 10.1073/pnas.050420897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ferdinand KC, Townsend RR. Hypertension in the US Black population: risk factors, complications, and potential impact of central aortic pressure on effective treatment. Cardiovasc Drugs Ther. 2012;26:157–165. doi: 10.1007/s10557-011-6367-8. [DOI] [PubMed] [Google Scholar]
- 18.Materson BJ, Reda DJ, Cushman WC, Massie BM, Freis ED, Kochar MS, Hamburger RJ, Fye C, Lakshman R, Gottdiener J. Single-drug therapy for hypertension in men. A comparison of six antihypertensive agents with placebo. The Department of Veterans Affairs Cooperative Study Group on Antihypertensive Agents. N Engl J Med. 1993;328:914–921. doi: 10.1056/NEJM199304013281303. [DOI] [PubMed] [Google Scholar]
- 19.Grim CE, Miller JZ, Luft FC, Christian JC, Weinberger MH. Genetic influences on renin, aldosterone, and the renal excretion of sodium and potassium following volume expansion and contraction in normal man. Hypertension. 1979;1:583–590. doi: 10.1161/01.hyp.1.6.583. [DOI] [PubMed] [Google Scholar]
- 20.Grim CE, Robinson M. Blood pressure variation in blacks: genetic factors. Semin Nephrol. 1996;16:83–93. [PubMed] [Google Scholar]
- 21.Wilson DK, Bayer L, Krishnamoorthy JS, Ampey-Thornhill G, Nicholson SC, Sica DA. The prevalence of salt sensitivity in an African-American adolescent population. Ethn Dis. 1999;9:350–358. [PubMed] [Google Scholar]
- 22.Schmidlin O, Forman A, Leone A, Sebastian A, Morris RC. Salt sensitivity in blacks: evidence that the initial pressor effect of NaCl involves inhibition of vasodilatation by asymmetrical dimethylarginine. Hypertension. 2011;58:380–385. doi: 10.1161/HYPERTENSIONAHA.111.170175. [DOI] [PubMed] [Google Scholar]
- 23.Flack JM, Grimm RH, Staffileno BA, Dnsc P, Yunis C, Hedquist L, Dudley A. New salt-sensitivity metrics: variability-adjusted blood pressure change and the urinary sodium-to-creatinine ratio. Ethn Dis. 2002;12:10–19. [PubMed] [Google Scholar]
- 24.Richardson SI, Freedman BI, Ellison DH, Rodriguez CJ. Salt sensitivity: a review with a focus on non-Hispanic blacks and Hispanics. J Am Soc Hypertens. 2013;7:170–179. doi: 10.1016/j.jash.2013.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Weinberger MH. Salt sensitivity of blood pressure in humans. Hypertension. 1996;27:481–490. doi: 10.1161/01.hyp.27.3.481. [DOI] [PubMed] [Google Scholar]
- 26.Weinberger MH. Hypertension in African Americans: the role of sodium chloride and extracellular fluid volume. Semin Nephrol. 1996;16:110–116. [PubMed] [Google Scholar]
- 27.Weinberger MH, Miller JZ, Luft FC, Grim CE, Fineberg NS. Definitions and characteristics of sodium sensitivity and blood pressure resistance. Hypertension. 1986;8:II127–II134. doi: 10.1161/01.hyp.8.6_pt_2.ii127. [DOI] [PubMed] [Google Scholar]
- 28.Wedler B, Wiersbitzki M, Gruska S, Wolf E, Luft FC. Definitions and characteristics of salt-sensitivity and resistance of blood pressure: should the diagnosis depend on diastolic blood pressure? Clin Exp Hypertens A. 1992;14:1037–1049. doi: 10.3109/10641969209038191. [DOI] [PubMed] [Google Scholar]
- 29.Wilson TW, Grim CE. Biohistory of slavery and blood pressure differences in blacks today. A hypothesis. Hypertension. 1991;17:I122–I128. doi: 10.1161/01.hyp.17.1_suppl.i122. [DOI] [PubMed] [Google Scholar]
- 30.Maseko MJ, Majane HO, Milne J, Norton GR, Woodiwiss AJ. Salt intake in an urban, developing South African community. Cardiovasc J S Afr. 2006;17:186–191. [PubMed] [Google Scholar]
- 31.Curtin PD. The slavery hypothesis for hypertension among African Americans: the historical evidence. Am J Public Health. 1992;82:1681–1686. doi: 10.2105/ajph.82.12.1681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Diamond J. The saltshaker’s curse. Natural History. 1991;10:20–26. [Google Scholar]
- 33.Fuchs FD. Why do black Americans have higher prevalence of hypertension?: an enigma still unsolved. Hypertension. 2011;57:379–380. doi: 10.1161/HYPERTENSIONAHA.110.163196. [DOI] [PubMed] [Google Scholar]
- 34.Ely DL. Overview of dietary sodium effects on and interactions with cardiovascular and neuroendocrine functions. Am J Clin Nutr. 1997;65:594S–605S. doi: 10.1093/ajcn/65.2.594S. [DOI] [PubMed] [Google Scholar]
- 35.Luft FC, Weinberger MH. Heterogeneous responses to changes in dietary salt intake: the salt-sensitivity paradigm. Am J Clin Nutr. 1997;65:612S–617S. doi: 10.1093/ajcn/65.2.612S. [DOI] [PubMed] [Google Scholar]
- 36.Gill JR, Grossman E, Goldstein DS. High urinary dopa and low urinary dopamine-to-dopa ratio in salt-sensitive hypertension. Hypertension. 1991;18:614–621. doi: 10.1161/01.hyp.18.5.614. [DOI] [PubMed] [Google Scholar]
- 37.Katori M, Majima M. Are all individuals equally sensitive in the blood pressure to high salt intake? (Review article) Acta Physiol Hung. 2008;95:247–265. doi: 10.1556/APhysiol.95.2008.3.2. [DOI] [PubMed] [Google Scholar]
- 38.Katori M, Majima M. Roles of the renal kallikrein-kinin system in salt-sensitive hypertension. Hypertension. 2004;44:e12. doi: 10.1161/01.HYP.0000146401.40304.51. [DOI] [PubMed] [Google Scholar]
- 39.Ford ES, Zhao G, Li C, Pearson WS, Mokdad AH. Trends in obesity and abdominal obesity among hypertensive and nonhypertensive adults in the United States. Am J Hypertens. 2008;21:1124–1128. doi: 10.1038/ajh.2008.246. [DOI] [PubMed] [Google Scholar]
- 40.Okosun IS, Choi S, Dent MM, Jobin T, Dever GE. Abdominal obesity defined as a larger than expected waist girth is associated with racial/ethnic differences in risk of hypertension. J Hum Hypertens. 2001;15:307–312. doi: 10.1038/sj.jhh.1001179. [DOI] [PubMed] [Google Scholar]
- 41.Luft FC, Miller JZ, Grim CE, Fineberg NS, Christian JC, Daugherty SA, Weinberger MH. Salt sensitivity and resistance of blood pressure. Age and race as factors in physiological responses. Hypertension. 1991;17:I102–I108. doi: 10.1161/01.hyp.17.1_suppl.i102. [DOI] [PubMed] [Google Scholar]
- 42.Laffer CL, Elijovich F. Essential hypertension of Caribbean Hispanics: sodium, renin, and response to therapy. J Clin Hypertens (Greenwich) 2002;4:266–273. doi: 10.1111/j.1524-6175.2002.00973.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Luft FC, Grim CE, Fineberg N, Weinberger MC. Effects of volume expansion and contraction in normotensive whites, blacks, and subjects of different ages. Circulation. 1979;59:643–650. doi: 10.1161/01.cir.59.4.643. [DOI] [PubMed] [Google Scholar]
- 44.Norris KC, Tareen N, Martins D, Vaziri ND. Implications of ethnicity for the treatment of hypertensive kidney disease, with an emphasis on African Americans. Nat Clin Pract Nephrol. 2008;4:538–549. doi: 10.1038/ncpneph0909. [DOI] [PubMed] [Google Scholar]
- 45.Chandramohan G, Bai Y, Norris K, Rodriguez-Iturbe B, Vaziri ND. Effects of dietary salt on intrarenal angiotensin system, NAD(P)H oxidase, COX-2, MCP-1 and PAI-1 expressions and NF-kappaB activity in salt-sensitive and -resistant rat kidneys. Am J Nephrol. 2008;28:158–167. doi: 10.1159/000110021. [DOI] [PubMed] [Google Scholar]
- 46.Kobori H, Nishiyama A, Abe Y, Navar LG. Enhancement of intrarenal angiotensinogen in Dahl salt-sensitive rats on high salt diet. Hypertension. 2003;41:592–597. doi: 10.1161/01.HYP.0000056768.03657.B4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hayakawa H, Coffee K, Raij L. Endothelial dysfunction and cardiorenal injury in experimental salt-sensitive hypertension: effects of antihypertensive therapy. Circulation. 1997;96:2407–2413. doi: 10.1161/01.cir.96.7.2407. [DOI] [PubMed] [Google Scholar]
- 48.Xu ZG, Lanting L, Vaziri ND, Li Z, Sepassi L, Rodriguez-Iturbe B, Natarajan R. Upregulation of angiotensin II type 1 receptor, inflammatory mediators, and enzymes of arachidonate metabolism in obese Zucker rat kidney: reversal by angiotensin II type 1 receptor blockade. Circulation. 2005;111:1962–1969. doi: 10.1161/01.CIR.0000161831.07637.63. [DOI] [PubMed] [Google Scholar]
- 49.Vaziri ND, Xu ZG, Shahkarami A, Huang KT, Rodríguez-Iturbe B, Natarajan R. Role of AT-1 receptor in regulation of vascular MCP-1, IL-6, PAI-1, MAP kinase, and matrix expressions in obesity. Kidney Int. 2005;68:2787–2793. doi: 10.1111/j.1523-1755.2005.00750.x. [DOI] [PubMed] [Google Scholar]
- 50.Wolak T, Kim H, Ren Y, Kim J, Vaziri ND, Nicholas SB. Osteopontin modulates angiotensin II-induced inflammation, oxidative stress, and fibrosis of the kidney. Kidney Int. 2009;76:32–43. doi: 10.1038/ki.2009.90. [DOI] [PubMed] [Google Scholar]
- 51.Yu XQ, Wu LL, Huang XR, Yang N, Gilbert RE, Cooper ME, Johnson RJ, Lai KN, Lan HY. Osteopontin expression in progressive renal injury in remnant kidney: role of angiotensin II. Kidney Int. 2000;58:1469–1480. doi: 10.1046/j.1523-1755.2000.00309.x. [DOI] [PubMed] [Google Scholar]
- 52.Gonzalez-Villalobos RA, Janjoulia T, Fletcher NK, Giani JF, Nguyen MT, Riquier-Brison AD, Seth DM, Fuchs S, Eladari D, Picard N, et al. The absence of intrarenal ACE protects against hypertension. J Clin Invest. 2013;123:2011–2023. doi: 10.1172/JCI65460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sangalli F, Carrara F, Gaspari F, Corna D, Zoja C, Botti L, Remuzzi G, Remuzzi A. Effect of ACE inhibition on glomerular permselectivity and tubular albumin concentration in the renal ablation model. Am J Physiol Renal Physiol. 2011;300:F1291–F1300. doi: 10.1152/ajprenal.00656.2010. [DOI] [PubMed] [Google Scholar]
- 54.Keane WF. Proteinuria: its clinical importance and role in progressive renal disease. Am J Kidney Dis. 2000;35:S97–105. doi: 10.1016/s0272-6386(00)70237-x. [DOI] [PubMed] [Google Scholar]
- 55.Eskildsen TV, Jeppesen PL, Schneider M, Nossent AY, Sandberg MB, Hansen PB, Jensen CH, Hansen ML, Marcussen N, Rasmussen LM, et al. Angiotensin II Regulates microRNA-132/-212 in Hypertensive Rats and Humans. Int J Mol Sci. 2013;14:11190–11207. doi: 10.3390/ijms140611190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Fang F, Liu GC, Kim C, Yassa R, Zhou J, Scholey JW. Adiponectin attenuates angiotensin II-induced oxidative stress in renal tubular cells through AMPK and cAMP-Epac signal transduction pathways. Am J Physiol Renal Physiol. 2013;304:F1366–F1374. doi: 10.1152/ajprenal.00137.2012. [DOI] [PubMed] [Google Scholar]
- 57.Vaziri ND, Bai Y, Ni Z, Quiroz Y, Pandian R, Rodriguez-Iturbe B. Intra-renal angiotensin II/AT1 receptor, oxidative stress, inflammation, and progressive injury in renal mass reduction. J Pharmacol Exp Ther. 2007;323:85–93. doi: 10.1124/jpet.107.123638. [DOI] [PubMed] [Google Scholar]
- 58.Vío CP, Jeanneret VA. Local induction of angiotensin-converting enzyme in the kidney as a mechanism of progressive renal diseases. Kidney Int Suppl. 2003;(86):S57–S63. doi: 10.1046/j.1523-1755.64.s86.11.x. [DOI] [PubMed] [Google Scholar]
- 59.Kobori H, Nangaku M, Navar LG, Nishiyama A. The intrarenal renin-angiotensin system: from physiology to the pathobiology of hypertension and kidney disease. Pharmacol Rev. 2007;59:251–287. doi: 10.1124/pr.59.3.3. [DOI] [PubMed] [Google Scholar]
- 60.Kim HJ, Sato T, Rodríguez-Iturbe B, Vaziri ND. Role of intrarenal angiotensin system activation, oxidative stress, inflammation, and impaired nuclear factor-erythroid-2-related factor 2 activity in the progression of focal glomerulosclerosis. J Pharmacol Exp Ther. 2011;337:583–590. doi: 10.1124/jpet.110.175828. [DOI] [PubMed] [Google Scholar]
- 61.Rodríguez-Iturbe B, Vaziri ND, Herrera-Acosta J, Johnson RJ. Oxidative stress, renal infiltration of immune cells, and salt-sensitive hypertension: all for one and one for all. Am J Physiol Renal Physiol. 2004;286:F606–F616. doi: 10.1152/ajprenal.00269.2003. [DOI] [PubMed] [Google Scholar]
- 62.Vaziri ND, Rodríguez-Iturbe B. Mechanisms of disease: oxidative stress and inflammation in the pathogenesis of hypertension. Nat Clin Pract Nephrol. 2006;2:582–593. doi: 10.1038/ncpneph0283. [DOI] [PubMed] [Google Scholar]
- 63.Tiago AD, Samani NJ, Candy GP, Brooksbank R, Libhaber EN, Sareli P, Woodiwiss AJ, Norton GR. Angiotensinogen gene promoter region variant modifies body size-ambulatory blood pressure relations in hypertension. Circulation. 2002;106:1483–1487. doi: 10.1161/01.cir.0000029093.93362.fc. [DOI] [PubMed] [Google Scholar]
- 64.Bhatnagar V, O’Connor DT, Schork NJ, Salem RM, Nievergelt CM, Rana BK, Smith DW, Bakris GL, Middleton JP, Norris KC, et al. Angiotensin-converting enzyme gene polymorphism predicts the time-course of blood pressure response to angiotensin converting enzyme inhibition in the AASK trial. J Hypertens. 2007;25:2082–2092. doi: 10.1097/HJH.0b013e3282b9720e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Woodiwiss AJ, Nkeh B, Samani NJ, Badenhorst D, Maseko M, Tiago AD, Candy GP, Libhaber E, Sareli P, Brooksbank R, et al. Functional variants of the angiotensinogen gene determine antihypertensive responses to angiotensin-converting enzyme inhibitors in subjects of African origin. J Hypertens. 2006;24:1057–1064. doi: 10.1097/01.hjh.0000226195.59428.57. [DOI] [PubMed] [Google Scholar]
- 66.Genovese G, Friedman DJ, Ross MD, Lecordier L, Uzureau P, Freedman BI, Bowden DW, Langefeld CD, Oleksyk TK, Uscinski Knob AL, et al. Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science. 2010;329:841–845. doi: 10.1126/science.1193032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Parsa A, Kao WH, Xie D, Astor BC, Li M, Hsu CY, Feldman HI, Parekh RS, Kusek JW, Greene TH, et al. APOL1 risk variants, race, and progression of chronic kidney disease. N Engl J Med. 2013;369:2183–2196. doi: 10.1056/NEJMoa1310345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lipkowitz MS, Freedman BI, Langefeld CD, Comeau ME, Bowden DW, Kao WH, Astor BC, Bottinger EP, Iyengar SK, Klotman PE, et al. Apolipoprotein L1 gene variants associate with hypertension-attributed nephropathy and the rate of kidney function decline in African Americans. Kidney Int. 2013;83:114–120. doi: 10.1038/ki.2012.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ko WY, Rajan P, Gomez F, Scheinfeldt L, An P, Winkler CA, Froment A, Nyambo TB, Omar SA, Wambebe C, et al. Identifying Darwinian selection acting on different human APOL1 variants among diverse African populations. Am J Hum Genet. 2013;93:54–66. doi: 10.1016/j.ajhg.2013.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Deng AY, Rapp JP. Locus for the inducible, but not a constitutive, nitric oxide synthase cosegregates with blood pressure in the Dahl salt-sensitive rat. J Clin Invest. 1995;95:2170–2177. doi: 10.1172/JCI117906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Nara Y, Nabika T, Ikeda K, Sawamura M, Endo J, Yamori Y. Blood pressure cosegregates with a microsatellite of angiotensin I converting enzyme (ACE) in F2 generation from a cross between original normotensive Wistar-Kyoto rat (WKY) and stroke-prone spontaneously hypertensive rat (SHRSP) Biochem Biophys Res Commun. 1991;181:941–946. doi: 10.1016/0006-291x(91)92027-h. [DOI] [PubMed] [Google Scholar]
- 72.Lewis EJ, Hunsicker LG, Bain RP, Rohde RD. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med. 1993;329:1456–1462. doi: 10.1056/NEJM199311113292004. [DOI] [PubMed] [Google Scholar]
- 73.Lewis EJ, Hunsicker LG, Clarke WR, Berl T, Pohl MA, Lewis JB, Ritz E, Atkins RC, Rohde R, Raz I. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N Engl J Med. 2001;345:851–860. doi: 10.1056/NEJMoa011303. [DOI] [PubMed] [Google Scholar]
- 74.Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, Remuzzi G, Snapinn SM, Zhang Z, Shahinfar S. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:861–869. doi: 10.1056/NEJMoa011161. [DOI] [PubMed] [Google Scholar]
- 75.de Zeeuw D, Ramjit D, Zhang Z, Ribeiro AB, Kurokawa K, Lash JP, Chan J, Remuzzi G, Brenner BM, Shahinfar S. Renal risk and renoprotection among ethnic groups with type 2 diabetic nephropathy: a post hoc analysis of RENAAL. Kidney Int. 2006;69:1675–1682. doi: 10.1038/sj.ki.5000326. [DOI] [PubMed] [Google Scholar]
- 76.Agodoa LY, Appel L, Bakris GL, Beck G, Bourgoignie J, Briggs JP, Charleston J, Cheek D, Cleveland W, Douglas JG, et al. Effect of ramipril vs amlodipine on renal outcomes in hypertensive nephrosclerosis: a randomized controlled trial. JAMA. 2001;285:2719–2728. doi: 10.1001/jama.285.21.2719. [DOI] [PubMed] [Google Scholar]
- 77.Whelton PK, Lee JY, Kusek JW, Charleston J, DeBruge J, Douglas M, Faulkner M, Greene PG, Jones CA, Kiefer S, et al. Recruitment experience in the African American Study of Kidney Disease and Hypertension (AASK) Pilot Study. Control Clin Trials. 1996;17:17S–33S. doi: 10.1016/s0197-2456(96)00087-6. [DOI] [PubMed] [Google Scholar]
- 78.Wright JT, Bakris G, Greene T, Agodoa LY, Appel LJ, Charleston J, Cheek D, Douglas-Baltimore JG, Gassman J, Glassock R, et al. Effect of blood pressure lowering and antihypertensive drug class on progression of hypertensive kidney disease: results from the AASK trial. JAMA. 2002;288:2421–2431. doi: 10.1001/jama.288.19.2421. [DOI] [PubMed] [Google Scholar]
- 79.Wright JT, Agodoa L, Contreras G, Greene T, Douglas JG, Lash J, Randall O, Rogers N, Smith MC, Massry S. Successful blood pressure control in the African American Study of Kidney Disease and Hypertension. Arch Intern Med. 2002;162:1636–1643. doi: 10.1001/archinte.162.14.1636. [DOI] [PubMed] [Google Scholar]
- 80.Appel LJ, Wright JT, Greene T, Agodoa LY, Astor BC, Bakris GL, Cleveland WH, Charleston J, Contreras G, Faulkner ML, et al. Intensive blood-pressure control in hypertensive chronic kidney disease. N Engl J Med. 2010;363:918–929. doi: 10.1056/NEJMoa0910975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Balamuthusamy S, Srinivasan L, Verma M, Adigopula S, Jalandhara N, Hathiwala S, Smith E. Renin angiotensin system blockade and cardiovascular outcomes in patients with chronic kidney disease and proteinuria: a meta-analysis. Am Heart J. 2008;155:791–805. doi: 10.1016/j.ahj.2008.01.031. [DOI] [PubMed] [Google Scholar]
- 82.Rahman M, Pressel S, Davis BR, Nwachuku C, Wright JT, Whelton PK, Barzilay J, Batuman V, Eckfeldt JH, Farber M, et al. Renal outcomes in high-risk hypertensive patients treated with an angiotensin-converting enzyme inhibitor or a calcium channel blocker vs a diuretic: a report from the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT) Arch Intern Med. 2005;165:936–946. doi: 10.1001/archinte.165.8.936. [DOI] [PubMed] [Google Scholar]
- 83.Yusuf S, Sleight P, Pogue J, Bosch J, Davies R, Dagenais G. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342:145–153. doi: 10.1056/NEJM200001203420301. [DOI] [PubMed] [Google Scholar]
- 84.ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group, The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT) JAMA. 2002;288:2981–2997. doi: 10.1001/jama.288.23.2981. [DOI] [PubMed] [Google Scholar]
- 85.Wright JT, Dunn JK, Cutler JA, Davis BR, Cushman WC, Ford CE, Haywood LJ, Leenen FH, Margolis KL, Papademetriou V, et al. Outcomes in hypertensive black and nonblack patients treated with chlorthalidone, amlodipine, and lisinopril. JAMA. 2005;293:1595–1608. doi: 10.1001/jama.293.13.1595. [DOI] [PubMed] [Google Scholar]
- 86.Messerli FH, Grossman E. Doxazosin arm of the ALLHAT study discontinued: how equal are antihypertensive drugs? Antihypertensive and Lipid Lowering Treatment to Prevent Heart Attack Trial. Curr Hypertens Rep. 2000;2:241–242. doi: 10.1007/s11906-000-0005-3. [DOI] [PubMed] [Google Scholar]
- 87.Flack JM, Sica DA, Bakris G, Brown AL, Ferdinand KC, Grimm RH, Hall WD, Jones WE, Kountz DS, Lea JP, et al. Management of high blood pressure in Blacks: an update of the International Society on Hypertension in Blacks consensus statement. Hypertension. 2010;56:780–800. doi: 10.1161/HYPERTENSIONAHA.110.152892. [DOI] [PubMed] [Google Scholar]
- 88.Leenen FH, Nwachuku CE, Black HR, Cushman WC, Davis BR, Simpson LM, Alderman MH, Atlas SA, Basile JN, Cuyjet AB, et al. Clinical events in high-risk hypertensive patients randomly assigned to calcium channel blocker versus angiotensin-converting enzyme inhibitor in the antihypertensive and lipid-lowering treatment to prevent heart attack trial. Hypertension. 2006;48:374–384. doi: 10.1161/01.HYP.0000231662.77359.de. [DOI] [PubMed] [Google Scholar]
- 89.Gibbs CR, Lip GY, Beevers DG. Angioedema due to ACE inhibitors: increased risk in patients of African origin. Br J Clin Pharmacol. 1999;48:861–865. doi: 10.1046/j.1365-2125.1999.00093.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Woo KS, Nicholls MG. High prevalence of persistent cough with angiotensin converting enzyme inhibitors in Chinese. Br J Clin Pharmacol. 1995;40:141–144. [PMC free article] [PubMed] [Google Scholar]
- 91.Elliott WJ. Higher incidence of discontinuation of angiotensin converting enzyme inhibitors due to cough in black subjects. Clin Pharmacol Ther. 1996;60:582–588. doi: 10.1016/S0009-9236(96)90155-1. [DOI] [PubMed] [Google Scholar]
- 92.Nishio K, Kashiki S, Tachibana H, Kobayashi Y. Angiotensin-converting enzyme and bradykinin gene polymorphisms and cough: A meta-analysis. World J Cardiol. 2011;3:329–336. doi: 10.4330/wjc.v3.i10.329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Holtkamp FA, de Zeeuw D, Thomas MC, Cooper ME, de Graeff PA, Hillege HJ, Parving HH, Brenner BM, Shahinfar S, Lambers Heerspink HJ. An acute fall in estimated glomerular filtration rate during treatment with losartan predicts a slower decrease in long-term renal function. Kidney Int. 2011;80:282–287. doi: 10.1038/ki.2011.79. [DOI] [PubMed] [Google Scholar]
- 94.Weir MR. Acute fall in glomerular filtration rate with renin-angiotensin system inhibition: a biomeasure of therapeutic success? Kidney Int. 2011;80:235–237. doi: 10.1038/ki.2011.132. [DOI] [PubMed] [Google Scholar]
- 95.Yusuf S, Teo KK, Pogue J, Dyal L, Copland I, Schumacher H, Dagenais G, Sleight P, Anderson C. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med. 2008;358:1547–1559. doi: 10.1056/NEJMoa0801317. [DOI] [PubMed] [Google Scholar]
- 96.Mancia G, De Backer G, Dominiczak A, Cifkova R, Fagard R, Germano G, Grassi G, Heagerty AM, Kjeldsen SE, Laurent S, et al. 2007 Guidelines for the Management of Arterial Hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC) J Hypertens. 2007;25:1105–1187. doi: 10.1097/HJH.0b013e3281fc975a. [DOI] [PubMed] [Google Scholar]
- 97.Cushman WC, Evans GW, Byington RP, Goff DC, Grimm RH, Cutler JA, Simons-Morton DG, Basile JN, Corson MA, Probstfield JL, et al. Effects of intensive blood-pressure control in type 2 diabetes mellitus. N Engl J Med. 2010;362:1575–1585. doi: 10.1056/NEJMoa1001286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wright JT, Agodoa LY, Appel L, Cushman WC, Taylor AL, Obegdegbe GG, Osei K, Reed J. New recommendations for treating hypertension in black patients: evidence and/or consensus? Hypertension. 2010;56:801–803. doi: 10.1161/HYPERTENSIONAHA.110.159566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Krousel-Wood MA, Muntner P, Islam T, Morisky DE, Webber LS. Barriers to and determinants of medication adherence in hypertension management: perspective of the cohort study of medication adherence among older adults. Med Clin North Am. 2009;93:753–769. doi: 10.1016/j.mcna.2009.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Odedosu T, Schoenthaler A, Vieira DL, Agyemang C, Ogedegbe G. Overcoming barriers to hypertension control in African Americans. Cleve Clin J Med. 2012;79:46–56. doi: 10.3949/ccjm.79a.11068. [DOI] [PubMed] [Google Scholar]
- 101.Turner BJ, Hollenbeak C, Weiner MG, Ten Have T, Roberts C. Barriers to adherence and hypertension control in a racially diverse representative sample of elderly primary care patients. Pharmacoepidemiol Drug Saf. 2009;18:672–681. doi: 10.1002/pds.1766. [DOI] [PubMed] [Google Scholar]
- 102.Egan BM, Zhao Y, Li J, Brzezinski WA, Todoran TM, Brook RD, Calhoun DA. Prevalence of Optimal Treatment Regimens in Patients With Apparent Treatment-Resistant Hypertension Based on Office Blood Pressure in a Community-Based Practice Network. Hypertension. 2013;62:691–697. doi: 10.1161/HYPERTENSIONAHA.113.01448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Martins D, Agodoa L, Norris KC. Hypertensive chronic kidney disease in African Americans: strategies for improving care. Cleve Clin J Med. 2012;79:726–734. doi: 10.3949/ccjm.79a.11109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.NIH. NIH launches multicenter clinical trial to test blood pressure strategy. 2009. Available from: http: //www.nih.gov/news/health/oct2009/nhlbi-29.htm. [Google Scholar]


