Abstract
Purpose
To compare a femtosecond laser with a microkeratome for flap creation during laser in situ keratomileusis (LASIK) in terms of flap thickness predictability and visual outcomes.
Patients and methods
This was a prospective, randomized, masked, paired-eye study. Forty-four patients (34 females) who received bilateral LASIK were included. Patients were stratified by ocular dominance, and they then underwent randomization of flap creation using the femtosecond laser on one eye and undergoing the microkeratome procedure on the other one. The visual outcome differences between the corrected distance visual acuity (CDVA) at baseline and the uncorrected distance visual acuity (UDVA) on the first day postoperatively were set as the efficiency index for both groups. All visual acuity outcome results and the deviation of flap thickness were evaluated. P-values <0.05 were considered statistically significant.
Results
The index of efficiency regarding the postoperative visual outcomes in the microkeratome group was lower (P<0.0001). This result was correlated with the difference between intended and achieved flap thickness (P=0.038; r=0.28), and a negative relationship in the regression analysis was confirmed (P<0.04; R2=0.1428). The UDVA in the microkeratome group improved significantly by the end of the first month (P<0.0271) in comparison to the baseline CDVA. The deviation between intended and postoperative flap thickness using either optical coherence pachymetry or Heidelberg Retinal Tomography II confocal microscopy was statistically significant (paired t-test; P<0.001) between the groups. The flap thickness deviation in the microkeratome group was higher. In the femtosecond laser group, the efficiency index was stable postoperatively (P=0.64) The UDVA improved significantly by the end of the first postoperative week (P=0.0043) in comparison to the baseline CDVA. Six months after surgery, improvement in the UDVA was significant in both groups (all P<0.001; one way analysis of variance).
Conclusion
Femtosecond laser was superior to microkeratome-assisted LASIK in terms of flap thickness predictability and the speed of visual acuity recovery. A negative relationship in the regression analysis between increasing flap thickness deviation and visual acuity recovery was confirmed.
Keywords: LASIK, femtosecond laser, flap predictability, microkeratome
Introduction
Femtosecond laser flap creation systems like the Femto LDV (Ziemer Ophthalmic Systems AG, Port, Switzerland) have been successfully used in modern corneal and refractive surgery. Many femtosecond lasers like the LDV are offering advantages in terms of size, mobility, and safety. The Femto LDV operates at high-pulse frequencies exceeding 5 MHz, approximately 1,000 times faster than other femtosecond laser technologies.1 This results in ultrashort laser pulses (200–300 femtoseconds) of low-energy density, which reduces undesirable side effects and results in a smooth corneal stromal bed.1 Some advantages of femtosecond technology over mechanical microkeratomes in flap creation for laser in situ keratomileusis (LASIK) are well documented.2–8 Still, both flap creation techniques can provide high safety and accuracy, and have become by many surgeons, the preferred method for flap creation in refractive surgery; however there is still little data available from paired-eye comparative studies. As these studies are performed on both eyes of the same patient, they represent an excellent tool to compare surgical setups, eliminating potential differences regarding the comparison of two different eyes of two different patients.
In this study, one eye from each patient was randomly selected for Femto LDV-assisted LASIK treatment, while the fellow eye was operated using the Amadeus II microkeratome (Amadeus®; Ziemer Ophthalmic Systems AG). The Amadeus microkeratome operated at a blade oscillation frequency of 11,000 rpm and at a blade advance rate of 1.6 mm/second. The purpose of this study was to evaluate the flap predictability and the visual outcomes during a follow-up period of 6 months, investigating the potential advantages or disadvantages regarding both flap creation technologies.
Patients and methods
Clinical setting
All procedures were performed at the Eye Clinic ORASIS/Swiss Eye Research Foundation in Reinach, Switzerland. Forty-four patients were included in the study; they underwent bilateral same-session LASIK. Ten patients were male and 34 patients were female. The study was approved by the local institutional review board (Kantonale Ethikkommission LDV number 2006/11, Aargau) and adhered to the tenets of the Declaration of Helsinki. All patients provided written informed consent.
Study protocol
The study included patients with the following types of refractive errors: myopia from −0.75 to −7.0; hyperopia; and astigmatism <+4.0 D. Central corneal thickness was measured using corneal topography (Orbscan II version 3.0; Bausch & Lomb Zyoptix) and was at least 500 μm or more. Additional inclusion criteria were a minimum age of 21 years, normal corneal topographical findings with no suspicion of corneal ectatic diseases and a stable refraction of ±0.5 D within the last 2 years before surgery. Patients were excluded if they had prior cataract or refractive surgery, abnormal corneal topographical findings, dry-eye syndrome, and/or amblyopia. All patients were obliged to attend the scheduled postoperative follow-up program. Two patients who failed to comply with the follow-up program were excluded from the study. Demographic data and operation settings are listed in Table 1.
Table 1.
Demographic data, as well as clinical and surgical settings
| Clinical settings | |
| Number of patients: | 44 patients (originally 46 patients; two excluded) |
| Number eyes treated: | 88 eyes (44 treated with LDV and Amadeus II, respectively) |
| Number of dominant eyes: | 58 OD, 28 OS, two eyes with no dominance |
| Number of dominant eyes treated with LDV: | 29 OD, 14 OS, one nondominant eye |
| Number of dominant eyes treated with SIS: | 29 OD, 14 OS, one nondominant eye |
| Age of patients: | 21–59 years old, median 37.5 years old |
| Patients’ sex: | 27 male patients and 17 female patients |
| Refraction error: | Eleven myopic eyes, 69 with myopic astigmatism Two hyperopic eyes, four with hyperopic astigmatism Two eyes with mixed astigmatism |
| Central corneal thickness: | 500–630 μm; mean: 552.39±29.11 μm; median: 553 μm |
| Operating room settings | |
| Excimer laser: | Bausch & Lomb Technolas® 217z |
| Flap creating techniques: | Amadeus II – Superpass Blade (11,000 rpm; advance rate: 1.6 mm/second) Femtosecond Da Vinci (pulse duration: 250 fs) |
| Hinge: | All located nasally |
| Eye tracker: | For all accounted treatments |
| Humidity: | 32%–60%; mean, 47.08%±6.8%; median, 48% |
| Temperature: | 17°C–21°C; mean, 19.54°C±0.9°C; median, 19.5°C |
| Surgical settings (Amadeus®-SIS group) | |
| Pupil size: | 4.2–7.5 mm; mean, 5.97±0.66 mm |
| Flap size: | 8.5–9.5 mm, mean, 8.86±0.46 mm |
| Flap thickness: | 110–140 μm; mean, 139.31±4.5 μm; median, 140 μm |
| Optical zone of treatment: | 6.0–7.8 mm; mean, 6.54±0.45 mm; median, 6.5 mm |
| Surgical settings (Da Vinci group) | |
| Pupil size: | 3.7–7.0 mm; mean, 5.88±0.74 mm |
| Flap size: | 8.5–9.5 mm; mean, 8.95±0.26 mm |
| Flap thickness: | 110–140 μm; mean, 114.20±7.6 μm; median, 110 μm |
| Optical zone of treatment: | 6.0–7.5 mm; mean, 6.53±0.43 mm; median, 6.5 mm |
Abbreviations: OD, oculus dexter; OS, oculus sinister.
Patients were examined preoperatively and at the first day, first week, and first month, third month, and sixth month after surgery. All baseline examinations included slit-lamp examination, manifest refraction, objective refractometry (Topcon, Tokyo, Japan), corneal topography (Orbscan® II version 3.0; Bausch & Lomb Zyoptix; Bausch & Lomb Incorporated, Bridgewater, NJ, USA), pupillometry (Procyon Instruments Ltd, London, UK), and wavefront aberrometry (ZyWave, Bausch & Lomb Zyoptix; Bausch & Lomb Incorporated, Bridgewater, NJ, USA). The visual outcomes were measured in Snellen optotypes and then converted in LogMAR analogs.9 The visual outcome differences between the corrected distance visual acuity (CDVA) at baseline and the uncorrected distance visual acuity (UDVA) at the first day postoperatively were set as the efficiency index for both groups. Trained optometrists who were unaware of the eye’s group assignment measured the manifest refraction. Corneal morphology and corneal flap thickness were assessed intraoperatively directly after the flap was repositioned in place with the aid of optical coherence pachymetry (OCP) (Technolas Perfect Vision GmbH) and at the first week postoperatively with the aid of confocal microscopy (Heidelberg Retinal Tomograph II; Heidelberg Engineering GmbH, Heidelberg, Germany). Measures of flap thickness deviation were made only at the center of the cornea. In this way, we could be sure of and objective to the measurement, since the center is, in our opinion, the best anatomical landmark to make comparisons among OCP and confocal microscopy findings.
Randomization
Patients were stratified by ocular dominance. One eye of each patient was randomly chosen to undergo flap creation using the Femto LDV, while the fellow eye underwent microkeratome.
Surgical technique
All patients were subjected to bilateral, same-session LASIK using an excimer laser with an eye tracker (Technolas® 217P Excimer; Technolas Perfect Vision GmbH), performed by an experienced refractive surgeon. The best average aberrometry measurements were entered into the excimer laser for iris recognition without any manual adjustments. All patients were treated with a tissue-saving ablation profile, and the targeted outcome was set to emmetropia in all cases. The hinge was created in all eyes nasally. In the operating room, humidity and temperature settings were similar during each surgery.
Postoperative management
All patients received a protective contact lens directly after surgery, and the patients were evaluated at the slit-lamp after 20 minutes. Topical therapy included topical combined corticosteroids with antibiotic eye drops (Tobradex) four times daily for 2 weeks, and hourly preservative-free artificial tears (Hyabak®) during the first week postoperatively.
Statistical analysis
For statistical analysis, MedCalc® version 12.7.5.0 was used. According to the distribution of data, parametrical or nonparametrical tests were used. P-values <0.05 were considered significant and were included.
Results
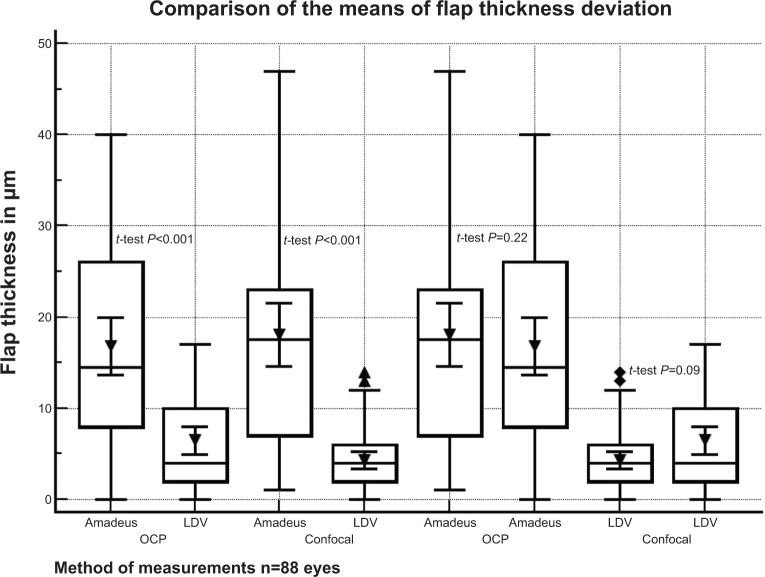
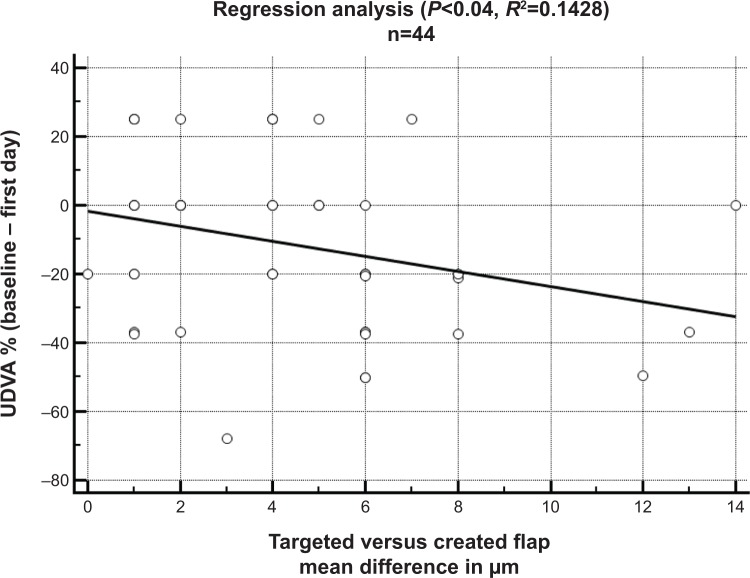
In the microkeratome (Amadeus II) group, the efficiency index at the first day, and until the first week after surgery, was reduced (P<0.0001). A significant improvement of the UDVA in comparison to the CDVA at baseline was first observed by the end of the first month (P=0.0271). The deviation between intended flap thickness and the postoperative flap thickness measurement according to OCP was 16.80±10.52 μm for the microkeratome group and 6.47±5.19 μm for the femtosecond laser group. In confocal microscopy, the deviation was 18.08 μm ±11.78 μm for the microkeratome group and 4.28 μm ±3.24 μm for the femtosecond laser group. With respect to paired t-tests, both comparisons of the means between the femtosecond laser and the microkeratome group using either OCP or confocal microscopy were statistically significant (P<0.001), while the difference between the type of measurement (OCP or confocal microscopy) for either group was not (Figure 1). The efficiency index in the microkeratome group correlated with the difference between the intended flap thickness and the postoperative flap thickness measurement (P<0.038, r=0.28). A negative relationship was confirmed in the regression analysis (P<0.04, R2=0.1428) regarding the difference in the μm between the intended and achieved flap thickness and the postoperative efficiency index (Figure 2).
Figure 1.
Difference between intended and actual flap thickness in eyes treated with femtosecond laser (Femto LDV) or microkeratome (Amadeus II).
Notes: The microkeratome group had significantly more deviation in terms of flap thickness (P<0.001), as measured with OCP and confocal microscopy, while the difference between the type of measurement (OCP or confocal microscopy) for either group was not significant.
Abbreviations: OCP, optical coherence pachymetry; n, number.
Figure 2.
Regression analysis with a negative relationship in terms of flap thickness deviation and the loss of visual acuity between CDVA at baseline and the UDVA percentage.
Notes: P<0.04; R2=0.14.
Abbreviations: UDVA, uncorrected distance visual acuity; n, number; CDVA, corrected distance visual acuity.
When expressing the visual outcomes as a logarithmic expression of the visual outcome (LogMAR) analogs,9 the microkeratome group lost 1.13±1.09 lines at the first day and 0.3±0.96 lines by the end of the first week. By the end of the first postoperative month, patients regained an average of 0.25±1.01 lines in comparison to the baseline measurements (P<0.001). In the femtosecond laser group, the efficiency index was stable (P=0.64). The UDVA was significantly increased at the end of the first week (P=0.0043; mean: −0.002±0.13 LogMAR) and at the end of the first postoperative month when compared to the baseline CDVA (P=0.001; mean: −0.041±0.13). Overall, the efficiency index for the LDV group was higher than that for the microkeratome group. The average results of UDVA for both groups are displayed in Table 2, and the average LogMAR results for both groups over the 6-month follow-up period are depicted in Figure 3. The difference in flap target thickness deviation and predictability between the femtosecond laser group and the microkeratome group was statistically significant (P<0.001), as shown in Figure 1. All eyes in the microkeratome group showed a minimal interface fluid accumulation, while the eyes in the femtosecond laser group did not. The sex of the patient, the dominance of the eye treated, the optical zone, the flap size, or the pupil size for either group did not correlate with the UDVA results in this study. Likewise, target spherical equivalent and root mean square were also not significantly different between groups.
Table 2.
Analytical results of the paired samples t-test of the UDVA percentage difference between all follow-up intervals
| Time | Femtosecond LDV
|
Amadeus II
|
||
|---|---|---|---|---|
| UDVA % | P<0.05 | UDVA % | P<0.05 | |
| Baseline | 0% | 0 | 0% | 0 |
| First day | −2.4%±27.0% | P=0.64 | −21.32%±20.5% | P<0.0001 |
| First week | +28.0%±38.4% | P=0.0043 | +23.0%±20.5% | P<0.0001 |
| First month | +7.2%±19.8% | P=0.0091 | +15.7%±25.0% | P=0.32 |
| Third month | +6.8%±17.3% | P=0.90 | +7.3%±18.5% | P=0.35 |
| Sixth month | +6.6%±15.3% | P=0.82 | +2.6%±11.3% | P=0.11 |
Abbreviations: UDVA, uncorrected distance visual acuity.
Figure 3.
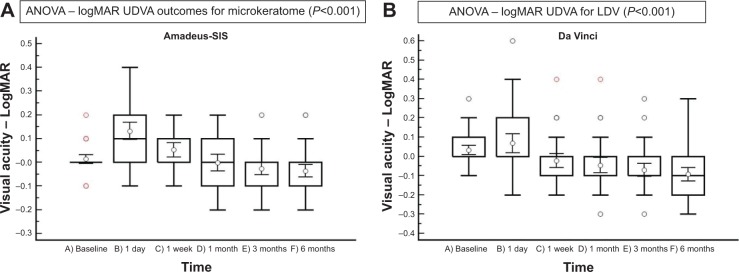
Logarithmic expression of UDVA (LogMAR analog) in the microkeratome (Amadeus II) and femtosecond laser (Femto LDV) group during the 6-month follow-up period.
Notes: (A) Amadeus II; (B) Femto LDV. Time is represented as 1 day, 1 week, 1 month, 3 months, or 6 months postoperatively.
Abbreviations: ANOVA, analysis of variance; UDVA, uncorrected distance visual acuity.
Discussion
This prospective, randomized, masked, paired-eye study investigated the visual outcomes after LASIK using two different devices for flap creation: the femtosecond laser LDV; and the Amadeus II. Overall results were excellent for both groups, with a UDVA of 20/20 or better at the first postoperative month. Overall, the efficiency index for the LDV group was higher than that of the microkeratome group, since the visual recovery in the microkeratome group was significantly delayed in comparison to the femtosecond laser group. Significant differences in visual acuity were evident between the two groups until the end of the first postoperative week. We could assume that this difference was due to a minimal interface fluid accumulation induced by the head or razor advancing system of the microkeratome and the oscillation of the microkeratome during flap creation. Complications, such as fluid accumulation or even debris at the interface, are frequent and well documented for both flap creation technologies.10,11,17–23 The fluid accumulation in the microkeratome group resolved within a few days, and visual acuity recovery was noted as expected.
Over the last few years, various studies regarding the accuracy of flap thickness creation with either the microkeratome or the femtosecond technology have surfaced.12–16 Measuring flap thickness deviation and comparing the two different flap creation systems is challenging and difficult. Recently, Kanellopoulos and Asimellis13 measured flap thickness deviation in a three-dimensional fashion including central, paracentral, and peripheral measurements. The comparison between the two different femtosecond lasers (FS60 and FS200) and a mechanical microkeratome (M2) showed a smaller flap thickness and a reduced variability in the Intralase femtosecond in comparison to the microkeratome group. In this study, flap thickness deviation was measured only in the center of the cornea; since we used confocal microscopy and OCP for the measurement, the corneal center was the best and safest option, in our opinion, to use as an anatomical landmark to compare our findings. Our results demonstrated that flap creation with the femtosecond laser was more accurate and predictable. All flaps created using the femtosecond laser deviated significantly less from the intended flap thickness, as evaluated by confocal microscopy and OCP in comparison to the microkeratome group. Furthermore, the increased precision of the femtosecond laser seemed to have a direct effect on UDVA, since less deviation in terms of flap thickness from the intended value correlated with the UDVA postoperatively. A negative relationship was confirmed in the regression analysis regarding the impact of this difference between the intended and achieved flap thickness on the decrease of postoperative visual acuity.
Several studies demonstrated the comparisons of these two flap creation technologies in terms of safety, flap thickness, flap predictability, UDVA, and stromal residual bed morphology.22–28 Durrie and Kezirian2 reported that femtosecond laser (IntraLase) was superior to microkeratome (Hansatome) for flap creation during LASIK, providing lower postoperative astigmatism and trefoil. Rosa et al24 compared the flap thickness after microkeratome-assisted flap creation and femtosecond laser-assisted flap creation (IntraLase 60 kHz) and concluded that flap predictability was superior in the femtosecond laser group. Although the visual outcomes we attained were excellent overall in both groups by the end of the first postoperative month, the superiority of the femtosecond laser technology was evident regarding the predictability and reproducibility of the flap thickness and the speed of visual recovery after the surgery. The LDV was extremely fast and safe, and it provided excellent visual outcomes; the Amadeus microkeratome also produced remarkable results with the exception of the slower UDVA visual recovery. The microkeratome technology still represents a reliable method for customized LASIK procedures.6,27,29,30 Nevertheless, the LDV femtosecond laser provides superior results by more closely creating the desired flap thickness, thus accelerating the visual recovery in comparison to microkeratome.7,24,29,30
Conclusion
Although microkeratome technology still provides reliable results and improvements in UDVA, it cannot outperform femtosecond technology. Flap creation with femtosecond technology is superior with respect to flap thickness predictability, reproducibility, and faster UDVA rehabilitation for the patient. Flap thickness deviations could possible play a role in delaying the visual acuity recovery of the patients.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Wirthlin AC. The ultrastable and compact Femto LDV. Cataract and Refractive Surgery Today Europe. 2007;2(3):65–66. [Google Scholar]
- 2.Durrie DS, Kezirian GM. Femtosecond laser versus mechanical keratome flaps in wavefront-guided laser in situ keratomileusis: prospective contralateral eye study. J Cataract Refract Surg. 2005;31(1):120–126. doi: 10.1016/j.jcrs.2004.09.046. [DOI] [PubMed] [Google Scholar]
- 3.Binder PS. One thousand consecutive IntraLase laser in situ keratomileusis flaps. J Cataract Refract Surg. 2006;32(6):962–969. doi: 10.1016/j.jcrs.2006.02.043. [DOI] [PubMed] [Google Scholar]
- 4.Kezirian GM, Stonecipher KG. Comparison of the IntraLase femtosecond laser and mechanical keratomes for laser in situ keratomileusis. J Cataract Refract Surg. 2004;30(4):804–811. doi: 10.1016/j.jcrs.2003.10.026. [DOI] [PubMed] [Google Scholar]
- 5.Tran DB, Sarayba MA, Bor Z, et al. Randomized prospective clinical study comparing induced aberrations with IntraLase and Hansatome flap creation in fellow eyes: potential impact on wavefront-guided laser in situ keratomileusis. J Cataract Refract Surg. 2005;31(1):97–105. doi: 10.1016/j.jcrs.2004.10.037. [DOI] [PubMed] [Google Scholar]
- 6.Ahn H, Kim JK, Kim CK, et al. Comparison of laser in situ keratomileusis flaps created by 3 femtosecond lasers and a microkeratome. J Cataract Refract Surg. 2011;37(2):349–357. doi: 10.1016/j.jcrs.2010.08.042. [DOI] [PubMed] [Google Scholar]
- 7.Muñoz G, Albarrán-Diego C, Ferrer-Blasco T, García-Lázaro S, Cerviño-Expósito A. Long-term comparison of corneal aberration changes after laser in situ keratomileusis: mechanical microkeratome versus femtosecond laser flap creation. J Cataract Refract Surg. 2010;36(11):1934–1944. doi: 10.1016/j.jcrs.2010.06.062. [DOI] [PubMed] [Google Scholar]
- 8.Moshirfar M, Gardiner JP, Schliesser JA, et al. Laser in situ keratomileusis flap complications using mechanical microkeratome versus femtosecond laser: retrospective comparison. J Cataract Refract Surg. 2010;36(11):1925–1933. doi: 10.1016/j.jcrs.2010.05.027. [DOI] [PubMed] [Google Scholar]
- 9.Holladay JT. Visual acuity measurements. J Cataract Refract Surg. 2004;30(2):287–290. doi: 10.1016/j.jcrs.2004.01.014. [DOI] [PubMed] [Google Scholar]
- 10.Heichel J, Wilhelm F, Kunert KS, Schlueter R, Stuhltraeger U, Hammer T. Influence of microkeratome parameters on the stromal bed and flap edge quality in laser in situ keratomileusis. Clin Ophthalmol. 2014;8:61–69. doi: 10.2147/OPTH.S51200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bardet E, Touboul D, Kerautret J, Léger F, Binder PS, Colin J. Interface fluid syndrome after bioptics. J Refract Surg. 2011;27(5):383–386. doi: 10.3928/1081597X-20101111-01. [DOI] [PubMed] [Google Scholar]
- 12.Pietilä J, Huhtala A, Mäkinen P, Uusitalo H. Flap characteristics, predictability, and safety of the Ziemer FEMTO LDV femtosecond laser with the disposable suction ring for LASIK. Eye (Lond) 2014;28(1):66–71. doi: 10.1038/eye.2013.244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kanellopoulos AJ, Asimellis G. Three-dimensional LASIK flap thickness variability: topographic central, paracentral and peripheral assessment, in flaps created by a mechanical microkeratome (M2) and two different femtosecond lasers (FS60 and FS200) Clin Ophthalmol. 2013;7:675–683. doi: 10.2147/OPTH.S40762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Reinstein DZ, Archer TJ, Gobbe M. Accuracy and reproducibility of cap thickness in small incision lenticule extraction. J Refract Surg. 2013;29(12):810–815. doi: 10.3928/1081597X-20131023-02. [DOI] [PubMed] [Google Scholar]
- 15.Reinstein DZ, Carp GI, Archer TJ, Gobbe M. Transitioning from mechanical microkeratome to femtosecond laser flap creation: visual outcomes of an experienced and a novice LASIK surgeon. J Cataract Refract Surg. 2012;38(10):1788–1795. doi: 10.1016/j.jcrs.2012.05.038. [DOI] [PubMed] [Google Scholar]
- 16.Hasimoto AR, Gomes MF, de Siqueira MA, Moreira H. Femtosecond laser versus mechanical microkeratome for LASIK flap creation. Arq Bras Oftalmol. 2013;76(6):335–338. doi: 10.1590/s0004-27492013000600002. Portuguese. [DOI] [PubMed] [Google Scholar]
- 17.Portellinha W, Kuchenbuk M, Nakano K, Oliveira M. Interface fluid and diffuse corneal edema after laser in situ keratomileusis. J Refract Surg. 2001;17(2 Suppl):S192–S195. doi: 10.3928/1081-597X-20010302-08. [DOI] [PubMed] [Google Scholar]
- 18.Ramírez M, Hernández-Quintela E, Naranjo-Tackman R. A comparative confocal microscopy analysis after LASIK with the IntraLase femtosecond laser vs Hansatome microkeratome. J Refract Surg. 2007;23(3):305–307. doi: 10.3928/1081-597X-20070301-15. [DOI] [PubMed] [Google Scholar]
- 19.Hoffman RS, Fine IH, Packer M. Persistent interface fluid syndrome. J Cataract Refract Surg. 2008;34(8):1405–1408. doi: 10.1016/j.jcrs.2008.03.042. [DOI] [PubMed] [Google Scholar]
- 20.Dawson DG, Schmack I, Holley GP, Waring GO, Grossniklaus HE, Edelhauser HF. Interface fluid syndrome in human eye bank corneas after LASIK: causes and pathogenesis. Ophthalmology. 2007;114(10):1848–1859. doi: 10.1016/j.ophtha.2007.01.029. [DOI] [PubMed] [Google Scholar]
- 21.Knorz MC. Complications of refractive excimer laser surgery. Ophthalmologe. 2006;103(3):192–198. doi: 10.1007/s00347-006-1314-y. German. [DOI] [PubMed] [Google Scholar]
- 22.Eisemann J, Carkeet A, Swann PG. Large interface particles from LASIK surgery. Clin Exp Optom. 2006;89(4):253–256. doi: 10.1111/j.1444-0938.2006.00046.x. [DOI] [PubMed] [Google Scholar]
- 23.Tanna M, Schallhorn SC, Hettinger KA. Femtosecond laser versus mechanical microkeratome: a retrospective comparison of visual outcomes at 3 months. J Refract Surg. 2009;25(Suppl 7):S668–S671. doi: 10.3928/1081597X-20090611-08. [DOI] [PubMed] [Google Scholar]
- 24.Rosa AM, Neto Murta J, Quadrado MJ, et al. Femtosecond laser versus mechanical microkeratomes for flap creation in laser in situ keratomileusis and effect of postoperative measurement interval on estimated femtosecond flap thickness. J Cataract Refract Surg. 2009;35(5):833–838. doi: 10.1016/j.jcrs.2008.12.038. [DOI] [PubMed] [Google Scholar]
- 25.Krueger RR, Dupps WJ. Biomechanical effects of femtosecond and microkeratome-based flap creation: prospective contralateral examination of two patients. J Refract Surg. 2007;23(8):800–807. doi: 10.3928/1081-597X-20071001-10. [DOI] [PubMed] [Google Scholar]
- 26.Patel SV, Maguire LJ, McLaren JW, Hodge DO, Bourne WM. Femtosecond laser versus mechanical microkeratome for LASIK: a randomized controlled study. Ophthalmology. 2007;114(8):1482–1490. doi: 10.1016/j.ophtha.2006.10.057. [DOI] [PubMed] [Google Scholar]
- 27.Kohnen T, Klaproth OK, Derhartunian V, Kook D. Results of 308 consecutive femtosecond laser cuts for LASIK. Ophthalmologe. 2010;107(5):439–445. doi: 10.1007/s00347-009-2029-7. German. [DOI] [PubMed] [Google Scholar]
- 28.Kouassi FX, Blaizeau M, Buestel C, et al. Comparison of Lasik with femtosecond laser versus Lasik with mechanical microkeratome: predictability of flap depth, corneal biomechanical effects and optical aberrations. J Fr Ophtalmol. 2012;35(1):2–8. doi: 10.1016/j.jfo.2011.03.013. French. [DOI] [PubMed] [Google Scholar]
- 29.Guber I, Moetteli L, Magnin L, Majo F. Moving from a mechanical microkeratome to a femtosecond laser for LASIK to correct astigmatic patients: clinical outcomes of a retrospective, consecutive, comparative study. Klin Monbl Augenheilkd. 2013;230(4):337–341. doi: 10.1055/s-0032-1328384. [DOI] [PubMed] [Google Scholar]
- 30.Chen S, Feng Y, Stojanovic A, Jankov MR, Wang Q. IntraLase femtosecond laser vs mechanical microkeratomes in LASIK for myopia: a systematic review and meta-analysis. J Refract Surg. 2012;28(1):15–24. doi: 10.3928/1081597X-20111228-02. [DOI] [PubMed] [Google Scholar]