Abstract
Aims/hypothesis
Targeted metabolomic and transcriptomic approaches were used to evaluate the relationship between skeletal muscle metabolite signatures, gene expression profiles and clinical outcomes in response to various exercise training interventions. We hypothesised that changes in mitochondrial metabolic intermediates would predict improvements in clinical risk factors, thereby offering novel insights into potential mechanisms.
Methods
Subjects at risk of metabolic disease were randomised to six months of inactivity or one of five aerobic and/or resistance training programmes (n = 112). Pre/post-intervention assessments included cardiorespiratory fitness (V̇O2peak), serum triacylglycerols (TGs) and insulin sensitivity (SI). In this secondary analysis, muscle biopsy specimens were used for targeted mass spectrometry-based analysis of metabolic intermediates and measurement of mRNA expression of genes involved in metabolism.
Results
Exercise regimens with the largest energy expenditure produced robust increases in muscle concentrations of even-chain acylcarnitines (median 37–488%), which correlated positively with increased expression of genes involved in muscle uptake and oxidation of fatty acids. Along with free carnitine, the aforementioned acylcarnitine metabolites were related to improvements in V̇O2peak, TGs and SI (R = 0.20–0.31, p < 0.05). Muscle concentrations of the tricarboxylic acid cycle intermediates succinate and succinylcarnitine (R = 0.39 and 0.24, p < 0.05) emerged as the strongest correlates of SI.
Conclusions/interpretation
The metabolic signatures of exercise-trained skeletal muscle reflected reprogramming of mitochondrial function and intermediary metabolism and correlated with changes in cardiometabolic fitness. Succinate metabolism and the succinate dehydrogenase complex emerged as a potential regulatory node that intersects with whole-body insulin sensitivity. This study identifies new avenues for mechanistic research aimed at understanding the health benefits of physical activity.
Trial registration
ClinicalTrials.gov NCT00200993 and NCT00275145
Keywords: Acylcarnitines, Branched-chain amino acids, Metabolomics, Physical activity, Skeletal muscle, Succinate
Introduction
Exercise training provides a wealth of health, cardiovascular and metabolic benefits with skeletal muscle serving as the major organ responsible for these metabolic adaptations [1–4]. At a cellular level, aerobic physical activity enhances both the quantity and quality of muscle mitochondria, which in turn augments oxidative capacity and exercise tolerance [5]. Although muscle mitochondrial biogenesis and remodelling are generally presumed to contribute to exercise-induced improvements in glucose and lipid control, the molecular basis of this link remains poorly understood.
The advent of ‘omics’ technology affords new opportunities to shed light on mechanisms of disease as well as those underlying the health benefits of physical activity. In recent years, both unbiased and targeted metabolomics approaches haves proven useful for gaining new insights into the pathogenesis of chronic metabolic disorders, including obesity, type 2 diabetes and cardiovascular disease (reviewed in [6, 7]). Metabolic profiling allows identification of metabolite signatures that track specific disease states and intervention outcomes. For example, comprehensive analysis of circulating metabolic intermediates linked exercise training-induced improvements in insulin sensitivity to increased levels of plasma glycine and proline and reductions in plasma acylcarnitines [8]. These metabolic signatures can be used to guide mechanistic studies and/or pinpoint potential therapeutic targets [6, 7]. Herein, we sought to apply a similar, yet tissue-specific, approach to better understand the salutary metabolic consequences of habitual physical exercise training.
The present study used mass spectrometry to inventory changes in muscle quantities of several mitochondrial intermediary metabolites in response to exercise training. We hypothesised that training-induced adaptations in skeletal muscle would be evident at the metabolite level and questioned whether changes in mitochondrial metabolic intermediates would predict improvements in clinically relevant outcomes. The study was conducted in a group of sedentary, overweight-to-obese men and women enrolled in the Studies of Targeted Risk Reduction Interventions through Defined Exercise (STRRIDE) trials[3].
Methods
Participants
This investigation included 112 participants who were part of the first and second STRRIDE trials conducted in 1998–2003 and 2004–2009, respectively. For these trials, participants were between ages 18 and 70 years, inactive (not participating in regular exercise), overweight to mildly obese (BMI 25–35 kg/m2) and dyslipidaemic (LDL-cholesterol level of 3.34–4.92 mmol/l [130–190 mg/dl] or HDL-cholesterol < 1.04 mmol/l [40 mg/dl] for men and < 1.17 mmol/l [45 mg/dl] for women). Participants were excluded if they had diagnosed diabetes mellitus or fasting glucose > 6.99 mmol/l (126 mg/dl), hypertension (BP > 160/90 mmHg), known cardiovascular disease, musculoskeletal conditions that prohibited exercise training, used tobacco or medications known to affect carbohydrate or lipid metabolism or followed a dietary regimen designed to induce weight loss [3]. For this secondary analysis, participants were selected randomly from those who completed the intervention and had skeletal muscle available for measurement of metabolic intermediates.
Exercise training
Participants were randomised to continued inactivity or to one of the following supervised aerobic exercise training groups: low-amount moderate-intensity exercise (5,021 kJ/week [1,200 kcal/week] at 40–55% V̇O2peak); low-amount vigorous-intensity exercise (5,021 kJ/week [1,200 kcal/week] at 65–80% V̇O2peak); high-amount vigorous-intensity exercise (8,368 kJ/week [2,000 kcal/week] at 65–80% V̇O2peak); resistance training (RT; three days/week, three sets/day, 8–12 repetitions/set of upper and lower body exercises using Cybex machines); and the linear combination of low-amount vigorous-intensity exercise and RT all with continuous progression and performed primarily under observation in a local fitness centre [4, 9]. At baseline, a maximal exercise test was used to determine participants’ V̇O2peak, maximum heart rate and heart-rate range corresponding to the prescribed intensity. To minimise musculoskeletal injury, participants completed a ramp period where they exercised within the prescribed heart-rate range while weekly exercise amounts (minutes) were progressed. At the end of the ramping phase, maximal and submaximal exercise tests verified that the heart-rate range corresponded to the correct percentage V̇O2peak; after this, participants exercised at the assigned exercise training prescription for a total of six months. Thus, progression occurred by using heart-rate ranges such that as the fitness of the participants improved, to maintain the same heart rate, the work-rate increased. Exercise training compliance was verified by recorded activity with heart-rate monitors (Polar Electro, Woodbury, NY, USA) and visual supervision.
Metabolic, cardiovascular risk factor and dietary assessments
As described previously [2], at baseline and the conclusion of training, participants completed a maximal cardiopulmonary treadmill test to determine cardiorespiratory fitness (V̇O2peak).
A frequently sampled intravenous glucose tolerance test was performed in all participants, after they had been fasted for 12 h, at baseline and at the conclusion of training (16–24 h after the last exercise bout) or inactivity. Insulin (Access Immunoassay System; Beckman Coulter, Fullerton, CA, USA) and glucose (YSI model 2300 Stat Plus; Yellow Springs Instruments, Yellow Springs, OH, USA) were measured and an insulin sensitivity index (SI) was derived based on Bergman’s Minimal Model [3, 10]. Triacylglycerol (TG) measurements were performed on fasted plasma samples using nuclear magnetic resonance (Liposcience, Cary, IN, USA [4]).
Participants were counselled to maintain dietary stability and verification was performed with 3 day food records and 24 h dietary recall interviews at baseline, mid-intervention and at training conclusion.
Skeletal muscle metabolites
Before and at the conclusion of the intervention training (16–24 h after the last exercise bout), vastus lateralis needle biopsies were performed in fasted participants using a modified Bergstrom technique [11]. Samples were snap frozen and stored at −80°C. Tissue was diluted 20-fold (mass : volume) in ice-cold water, minced with surgical scissors and homogenised for 30 s using a rotor-stator tissue disruptor.
A targeted, mass-spectrometry-based platform was used to measure a total of 15 amino acids, 45 acylcarnitines and seven organic acids as previously described [12, 13]. For analyses of amino acids and acylcarnitines, samples were precipitated with methanol, esterified and analysed with tandem mass spectrometry (MS/MS). Organic acids were measured with gas chromatography– mass spectrometry (GC/MS) [13].
Gene expression
Gene expression arrays were performed in half (n = 57) of the 112 participants, evenly distributed among groups and sexes. Total RNA was extracted from skeletal muscle biopsies using Trizol (Invitrogen, Carlsbad, CA, USA) and 30–50 mg of starting material. Two-round amplification of total RNA was performed using Affymetrix kits (Affymetrix, Santa Clara, CA, USA). Thirty microgrammes of biotinylated cRNA from the second amplification round was hybridised to Affymetrix U133 Plus 2.0 microarrays. Data processing of resultant CEL (cell intensity) files is described below. As previously described, quality controls included adequate amplifications, thresholds for appropriate scaling factors and controls for RNA integrity (GAPDH 3′/5′ and HSACO7 3′/5′ ratios) [14, 15]. If a sample did not meet all quality control values after processing through microarray, it was reprocessed from the original total RNA sample, after the integrity of the RNA sample was verified by agarose gel electrophoresis and imaging. For probe set analysis, probe set means were generated from the PLIER (probe logarithmic intensity error) algorithm (typically six iterations) in Expression Console (Affymetrix). PLIER is a model-based signal estimator beneficial to multi-array estimations.
Data analysis
Per cent changes were calculated for metabolic intermediates and clinical changes as follows: ([post-intervention variable − pre-intervention variable]/pre-intervention variable) × 100. Gene expression changes were calculated as log2 post-intervention expression minus log2 pre-intervention expression. Non-parametric ANOVA (Kruskal–Wallis) with pairwise post hoc tests were used to compare the five intervention groups with the inactive group. Parametric ANOVAs with Dunnett’s post-hoc testing were also performed. Spearman correlations were used to relate changes in metabolites to changes in clinical variables (V̇O2peak, SI, TG) and gene expression. Lack of normality was based on visualisation of distributions. Except for pairwise post hoc tests where p < 0.01 was accepted as significant, p < 0.05 was considered significant. No additional corrections for multiple statistical tests were used.
Results
Exercise training increased skeletal muscle content of mitochondrial metabolic intermediates
As previously reported, exercise training produced robust changes in V̇O2peak, SI, and TGs [2–4, 16]. Table 1 illustrates the baseline and post-intervention values for the subset of STRRIDE participants included in the current analysis. Figure 1 shows the average change in skeletal muscle metabolites measured in the fasted state, grouped according to the intervention regimen. Training-induced changes in the individual metabolites in muscle were most pronounced in the high-amount/vigorous and RT plus low-amount/vigorous-intensity groups while changes were minimal in the RT group. These results reflect the targeted nature of our analysis, which focused heavily on mitochondrial intermediates. As compared with the inactive control group, high-amount/vigorous-intensity training produced marked statistically significant increases (60–312%) in skeletal muscle concentrations of several even-chain acylcarnitines, primarily medium- and long-chain species generated as byproducts of β-oxidation (C6, C4DC, C8, C6DC, C10:2, C10, C12:1, C12, C14:2, C14:1, C14, C16:2, C16:1, C18:1) (Fig. 1; p < 0.01 for all post-hoc tests). ). Similarly, RT plus low-amount/vigorous-intensity training also increased several short- and medium-chain acylcarnitine species C2, C4/C4i, C4OH, C6, C4DC, C6DC, C10:1, C10, C7DC, C10OH/C8DC, C12:1, C12, C12OH/C10DC, C14:2, C14:1, C14, C14oh/C12DC, C16:2, C16:1, C16, C16:1OH/C14:1DC, C18:2, C18:1, C18:1OH/C16:1DC) in the range of 40–488% (Fig. 1; p < 0.01 for all post-hoc tests). By contrast, the glycolytic metabolites, lactate and pyruvate, and most organic acid intermediates of the tricarboxylic acid (TCA) cycle were unaffected by exercise training, with the exception of succinate, which appeared to increase dose dependently, with an 85% increase in the high-amount/vigorous-intensity group (Fig. 1, p < 0.01 for all post-hoc tests). Table 2 shows results from parametric testing.
Table 1.
Cardiorespiratory fitness (V̇O2peak), SI and TGs at baseline and post-intervention
|
|
|
|
|
|
|
|
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | All participants (n = 112) | Inactive (n = 20) | Low/Moderate (n = 15) | Low/Vigorous (n = 20) | High/Vigorous (n = 17) | AT/RT (n = 20) | RT (n = 20) | |||||||
|
|
|
|
|
|
|
|
||||||||
| Pre | Post | Pre | Post | Pre | Post | Pre | Post | Pre | Post | Pre | Post | Pre | Post | |
| V̇O2peak (ml O2/[kg × min]) | 28.0 (5.8) | 30.4 (7.1) | 27.7 (6.4) | 27.5 (6.6) | 27.8 (5.7) | 29.5a (7.3) | 27.9 (6.8) | 30.2* (6.9) | 29.1 (5.3) | 33.8 a (7.2) | 27.9 (6.3) | 32.4* (8.1) | 27.8 (5.0) | 28.7 (5.3) |
| SI (mU / [l × min]) | 4.0 (2.8) | 5.2 (4.3) | 3.7 (2.7) | 3.4 (1.8) | 3.2 (2.7) | 4.1 a (2.9) | 4.0 (2.9) | 5.0 (3.8) | 4.5 (4.1) | 5.2 (3.6) | 4.2 (2.8) | 9.9* (7.2) | 4.2 (2.0) | 4.5 (2.9) |
| TG (mmol/l) | 1.57 (0.67) | 1.43 (0.67) | 1.85 (0.84) | 1.73 (0.94) | 1.66 (0.76) | 1.40 (0.44) | 1.39 (0.55) | 1.40 (0.74) | 1.57 (0.61) | 1.36* (0.51) | 1.47 (0.49) | 1.27 (0.57) | 1.50 (0.72) | 1.43 (0.65) |
Data are presented as mean (SD)
Low/Moderate: low-amount moderate-intensity aerobic exercise; Low/Vigorous: low-amount vigorous-intensity aerobic exercise; High/Vigorous: high-amount vigorous-intensity aerobic exercise; AT/RT: combination of low/vigorous aerobic exercise and RT
p<0.05 for change from baseline using paired t test
Fig. 1.
Muscle acylcarnitines and organic acids: per cent change from baseline and relation to cardiorespiratory fitness (V̇O2peak) and insulin sensitivity change. Bars represent skeletal muscle metabolite median per cent change for each intervention arm: Inactive; low-amount moderate-intensity aerobic exercise (Low/Moderate); low-amount vigorous-intensity aerobic exercise (Low/Vigorous); high-amount vigorous-intensity aerobic exercise (High/Vigorous); combination of low/vigorous aerobic exercise and resistance training (Low Vigorous/RT); resistance training only (RT). *p<0.01 vs Inactive (non-parametric ANOVAs with post hoc comparisons). Spearman correlations were used to relate per cent changes in skeletal muscle metabolites and per cent changes in cardiorespiratory fitness (V̇O2peak), insulin sensitivity, and TGs. For acylcarnitine relationships with V̇O2peak (positive): dashed line, p<0.05; solid line, p≤0.01. For acylcarnitine relationships with insulin sensitivity (positive): †p<0.05; ‡p≤0.01. For acylcarnitine relationships with TGs (negative): §p<0.05 §§p≤0.01
Table 2.
Skeletal muscle metabolite changes and relations to per cent change in V̇O2peak, SI and TGs
|
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Group | Inactive (n = 20) | Low/Mod erate (n = 15) | Low/Vigo rous (n = 20) | High/Vig orous (n = 17) | AT/RT (n = 20) | RT (n = 20) | Overall p value (ANOVA) | R value | ||
|
|
||||||||||
| V̇O2peak (n = 109) | SI (n = 103) | TG (n = 111) | ||||||||
|
|
||||||||||
| Acylcarnitines | ||||||||||
| C0 | −6.0 (35.4) | −7.2 (35.3) | 14.8 (88.7) | 16.7 (46.4) | 14.9 (45.5) | −9.0 (23.0) | 0.35 | 0.08 | 0.21‡‡ | −0.13 |
| C2 | −11.4 (55.4) | 6.6 (6101) | −0.6 (61.2) | 36.0 (95.4) | 64.9 (101.6) * | 28.1 (72.9) | 0.03† | 0.13 | 0.16 | −0.12 |
| C3 | 26.2 (151.3) | 0.6 (39.1) | −6.3 (46.7) | 1.8 (51.1) | 21.9 (80.0) | −14.4 (28.8) | 0.56 | 0.02 | 0.13 | −0.12 |
| C4/Ci4 | −1.7 (83.2) | 125.7 (198.5) | 61.4 (124.0) | 151.6 (279.5) | 195.4 (190.5) * | 72.4 (179.4) | 0.02† | 0.22‡ | 0.10 | −0.23‡ |
| C5:1 | 43.7 (90.5) | 1.4 (72.6) | −15.2 (54.4) | 4.7 (64.2) | 21.4 (70.5) | 7.7 (54.1) | 0.16 | −0.18 | 0.08 | −0.06 |
| C5 | −13.7 (59.6) | 44.1 (84.1) | 12.9 (80.0) | 32.7 (74.7) | 88.6 (230.5) | 5.9 (78.7) | 0.12 | 0.00 | 0.09 | −0.19‡ |
| C4OH | 17.2 (76.9) | 59.3 (131.5) | 56.0 (119.0) | 212.3 (432.4)* | 186.4 (202.3) | 105.5 (185.5) | 0.04† | 0.21‡ | 0.15 | −0.24‡‡ |
| C6 | 34.6 (151.5) | 255.8 (405.5) | 207.1 (329.6) | 620.4 (937.6)* | 552.2 (580.6) † | 147.6 (293.6) | 0.002† | 0.24‡‡ | 0.10 | −0.24‡‡ |
| C5OH/C3DC | −12.3 (34.1) | 26.9 (88.0) | 2.3 (41.5) | 21.8 (56.5) | 30.1 (79.3) | 0.9 (38.3) | 0.17 | 0.06 | 0.12 | −0.108 |
| C4DC/Ci4DC | −15.9 (34.8) | 7.1 (56.2) | 18.7 (76.5) | 41.6 (89.7) | 27.4 (61.5) | −0.8 (39.2) | 0.08 | 0.09 | 0.24‡‡ | −0.155 |
| C8:1 | 5.13 (45.2) | 29.4 (94.7) | 47.3 (116.7) | 69.8 (112.6) | 49.7 (65.1) | 20.7 (67.6) | 0.28 | 0.03 | 0.23‡ | −0.111 |
| C8 | 42.2 (144.9) | 156.1 (224.7) | 127.9 (221.3) | 677.6 (1,411.3)* | 382.9 (422.4) | 69.8 (174.1) | 0.01† | 0.23‡‡ | 0.14 | −0.24‡‡ |
| C5DC | −1.32 (52.0) | −4.3 (38.9) | 75.2 (225.0) | 53.2 (104.9) | 23.9 (54.0) | 35.2 (182.5) | 0.43 | 0.09 | 0.23‡ | −0.10 |
| C8:1OH/C6:1DC | 71.3 (305.7) | 7.7 (97.8) | 41.2 (96.0) | 100.1 (219.9) | 51.1 (74.3) | 122.3 (466.3) | 0.82 | 0.09 | 0.19 | 0.04 |
| C6DC | 4.2 (69.3) | 41.2 (66.5) | 67.9 (145.1) | 117.9 (135.3)* | 131.1 (112.2)* | 46.8 (114.8) | 0.006† | 0.24‡‡ | 0.18 | −0.16 |
| C10:3 | 32.0 (174.3) | −1.6 (98.4) | 75.1 (175.4) | 66.4 (268.3) | 75.4 (181.7) | 5.8 (65.5) | 0.61 | 0.03 | 0.08 | −0.04 |
| C10:2 | 13.8 (173.1) | 116.0 (224.0) | 44.2 (138.8) | 229.0 (413.4) | 389.9 (893.7)* | −19.1 (34.8) | 0.04† | 0.20‡ | 0.22‡ | −0.21‡ |
| C10:1 | 4.7 (64.1) | 97.9 (121.4) | 162.6 (252.7) | 432.4 (914.9) | 245.7 (282.3) | 135.2 (536.7) | 0.11 | 0.23‡ | 0.30‡‡ | −0.24‡‡ |
| C10 | 16.4 (109.3) | 242.8 (288.5) | 128.9 (210.2) | 682.0 (1174.7)* | 397.6 (427.1) | 41.0 (153.0) | 0.001† | 0.30‡‡ | 0.21‡ | −0.23‡‡ |
| C7DC | −10.2 (48.9) | 13.5 (123.4) | 46.8 (110.2) | 112.5 (217.0) | 181.5 (220.5)* | 14.2 (74.8) | 0.001† | 0.09 | 0.19 | 0.06 |
| C8:1DC | −29.3 (71.2) | 75.2 (140.0) | 109.1 (360.0) | 1322.5 (4501.2) | 141.6 (252.6) | 10.7 (98.1) | 0.23 | 0.20‡ | 0.30‡‡ | −0.04 |
| C10OH/C8DC | 39.8 (110.5) | 48.8 (136.2) | 125.0 (205.4) | 239.9 (312.4)* | 209.1 (223.9)* | 42.4 (120.6) | 0.004† | 0.17 | 0.16 | −0.06 |
| C12:1 | 38.4 (117.3) | 110.6 (138.5) | 128.9 (214.1) | 803.6 (2421.3) | 501.3 (646.5) | 61.8 (152.4) | 0.12 | 0.24‡‡ | 0.24‡‡ | −0.22‡ |
| C12 | 55.3 (139.5) | 222.1 (225.4) | 195.2 (311.6) | 1,389.1 (3,059.7)* | 469.6 (499.6) | 86.1 (216.2) | 0.02† | 0.24‡‡ | 0.25‡‡ | −0.23‡ |
| C12OH/C10DC | 97.2 (263.6) | 172.8 (262.9) | 148.2 (400.1) | 180.2 (310.4) | 393.7 (528.8) | 68.4 (139.3) | 0.06 | 0.20‡ | 0.22‡ | −0.23‡ |
| C14:2 | 34.1 (136.1) | 388.5 (633.3) | 640.8 (1,869.2) | 1,170.0 (3,233.2) | 652.0 (748.3) | 98.6 (209.5) | 0.23 | 0.25‡‡ | 0.24‡ | −0.23‡ |
| C14:1 | 52.5 (202.2) | 376.4 (362.3) | 244.6 (391.1) | 1,564.8 (3,523.8)* | 694.2 (862.3) | 87.7 (220.8) | 0.02† | 0.24‡‡ | 0.22‡ | −0.21‡ |
| C14 | 54.7 (143.3) | 286.0 (296.9) | 210.0 (332.7) | 547.8 (966.8)* | 539.9 (658.2)* | 87.8 (271.0) | 0.008† | 0.24‡‡ | 0.28‡‡ | −0.25‡‡ |
| C14:1OH | 37.9 (111.7) | 140.1 (217.8) | 63.5 (128.2) | 260.4 (505.6) | 252.5 (404.3) | 57.6 (126.1) | 0.05† | 0.18 | 0.23‡ | −0.21‡ |
| C14OH/C12DC | 39.2 (146.4) | 268.7 (403.8) | 140.9 (257.1) | 369.7 (673.4)* | 198.4 (236.4) | 35.2 (77.3) | 0.03† | 0.15 | 0.28‡‡ | −0.16 |
| C16:2 | 47.1 (169.6) | 389.5 (524.1) | 136.2 (250.7) | 1,375.3 (2,466.4)* | 622.7 (811.6) | 118.8 (310.4) | 0.003† | 0.20‡ | 0.23‡ | −0.21‡ |
| C16:1 | 33.5 (130.7) | 297.7 (336.5) | 250.4 (393.2) | 1,494.7 (4,300.9) | 587.7 (751.9) | 103.1 (254.3) | 0.13 | 0.21‡ | 0.24‡‡ | −0.19‡ |
| C16 | 41.6 (113.7) | 201.2 (253.0) | 105.8 (148.2) | 385.2 (685.1)* | 321.5 (428.0)* | 58.1 (159.1) | 0.01† | 0.17 | 0.28‡‡ | −0.21‡ |
| C16:1OH/C14:1DC | 60.1 (170.7) | 162.8 (181.8) | 111.7 (197.0) | 226.5 (551.0) | 250.8 (293.8) | 1.1 (92.3) | 0.05 | 0.17 | 0.31‡ | −0.26‡‡ |
| C16OH/C14DC | 78.6 (210.3) | 178.1 (440.1) | 120.8 (213.3) | 204.3 (314.4) | 191.7 (222.0) | 153.8 (574.7) | 0.9 | 0.06 | 0.21‡ | −0.22‡ |
| C18:2 | 11.2 (73.9) | 218.7 (302.9) | 196.6 (407.1) | 494.41 (1,199.0) | 268.9 (409.9) | 70.2 (150.2 | 0.12 | 0.19 | 0.29‡‡ | −0.24‡‡ |
| C18:1 | 29.8 (105.6) | 235.5 (327.6) | 110.8 (191.1) | 589.1 (1,480.0) | 270.2 (382.1) | 61.5 (150.4) | 0.08 | 0.21 | 0.26‡‡ | −0.24‡‡ |
| C18 | 47.1 (138.3) | 171.8 (238.7) | 150.7 (284.5) | 273.0 (469.9) | 204.4 (343.0) | 47.2 (155.0) | 0.14 | 0.13 | 0.24‡ | −0.26‡‡ |
| C18:2OH | 16.2 (79.8) | 101.6 (193.4) | 323.1 (1,063.8) | 140.9 (347.2) | 178.8 (269.1) | 174.3 (236.0) | 0.59 | 0.02 | 0.16 | −0.136 |
| C18:1OH/C16:1DC | 17.4 (125.8) | 151.3 (318.2) | 132.3 (246.7) | 1,396.9 (4,377.5) | 182.1 (238.2) | 48.4 (108.3) | 0.16 | 0.20‡ | 0.21‡ | −0.148 |
| C18OH/C16DC | 25.4 (96.1) | 450.2 (1,497.3) | 60.0 (172.2) | 149.0 (205.3) | 318.4 (717.6) | 2.9 (115.4) | 0.25 | 0.15 | 0.16 | −0.105 |
| C20:4 | 34.1 (123.3) | 141.3 (276.7) | 131.5 (213.7) | 64.0 (142.1) | 233.3 (499.3) | 21.0 (83.1) | 0.15 | 0.15 | 0.20‡ | −0.085 |
| C20 | 55.4 (152.1) | 123.2 (206.3) | 86.6 (160.2) | 351.7 (714.6) | 231.2 (377.9) | 61.6 (200.7) | 0.12 | 0.07 | 0.18 | −0.095 |
| C18:1DC | 85.5 (270.6) | 80.2 (350.2) | 48.2 (167.1) | 62.6 (128.6) | 147.8 (317.1) | 109.7 (280.5) | 0.91 | 0.14 | 0.08 | −0.152 |
| C20OH/C18DC | 8.6 (108.5) | 1,122.7 (2,462.4)* | 54.0 (170.7) | 116.8 (124.0) | 129.2 (251.5) | 70.1 (221.3) | 0.01† | 0.11 | 0.19 | 0.003 |
| C22 | 33.2 (154.0) | 139.7 (528.1) | 177.7 (311.3) | 258.5 (703.4) | 118.3 (337.1) | 9.0 (117.9) | 0.61 | −0.01 | −0.06 | 0.083 |
| Organic acids | ||||||||||
| Lactate | 10.6 (28.8) | 16.3 (35.1) | −4.4 (59.3) | 3.7 (67.1) | 8.3 (50.4) | 18.1 (491) | 0.76 | −0.17 | −0.00 | −0.124 |
| Pyruvate | 2.6 (46.1) | 29.8 (111.2) | −11.5 (65.0) | 8.6 (61.0) | 10.9 (77.3) | 54.8 (96.1) | 0.13 | −0.12 | −0.06 | −0.084 |
| Succinate | 1.4 (50.8) | 38.5 (74.4) | 61.3 (82.4) | 84.5 (122.0)* | 33.2 (48.3) | 14.2 (63.4) | 0.02† | 0.20‡ | 0.39‡‡ | −0.20‡ |
| Fumarate | 23.7 (67.5) | 33.2 (68.8) | 44.5 (75.2) | 85.6 (108.1) | 68.2 (95.3) | 34.4 (69.5) | 0.20 | 0.15 | 0.25‡‡ | −0.18 |
| Malate | −1.2 (28.8) | 10.7 (33.6) | 6.3 (40.8) | 14.8 (47.6) | 22.1 (45.3) | 9.6 (43.1) | 0.59 | 0.04 | 0.19 | −0.20‡ |
| α-Ketoglutarate | 6.1 (54.8) | 16.2 (76.6) | 20.2 (75.2) | 49.9 (100.0) | 19.0 (58.8) | 39.0 (78.0) | 0.52 | 0.05 | 0.21‡ | −0.27‡‡ |
| Citrate | −2.0 (32.5) | 7.6 (41.0) | 10.1 (53.4) | 33.5 (64.8) | 10.8 (63.2) | −16.8 (40.2) | 0.10 | −0.08 | 0.15 | −0.26‡‡ |
| Amino acids | ||||||||||
| Glycine | 0.30 (25.2) | −11.0 (25.7) | −1.6 (46.2) | −1.0 (35.3) | 1.7 (34.2) | −6.1 (35.2) | 0.90 | −0.02 | 0.15 | −0.101 |
| Alanine | 2.8 (26.1) | −1.8 (33.4) | 10.8 (50.1) | 11.6 (40.5) | 11.1 (36.0) | −4.7 (25.7) | 0.60 | 0.10 | 0.26‡‡ | −0.22‡ |
| Serine | 0.4 (29.3) | 7.6 (47.1) | 12.5 (37.8) | 12.0 (41.2) | 7.5 (29.2) | −4.5 (28.7) | 0.63 | 0.04 | 0.21‡ | −0.061 |
| Proline | 3.8 (27.7) | −14.5 (28.8) | −7.35 (32.1) | −0.9 (36.9) | 7.3 (46.0) | −0.1 (47.5) | 0.58 | −0.06 | 0.04 | −0.22‡ |
| Valine | 1.2 (22.4) | −5.3 (26.7) | 3.05 (36.0) | 6.9 (33.6) | 12.6 (40.6) | −8.8 (23.3) | 0.32 | −0.10 | 0.14 | −0.091 |
| Leucine/isoleucine | −4.3 (24.6) | −6.1 (27.7) | 0.55 (37.7) | 7.8 (33.1) | 14.6 (42.6) | −6.3 (21.4) | 0.26 | 0.03 | 0.15 | −0.161 |
| Methionine | −3.5 (21.0) | −10.0 (25.1) | 7.9 (42.7) | 2.6 (29.6) | 8.5 (28.2) | −7.7 (23.5) | 0.26 | −0.06 | 0.21‡ | −0.135 |
| Histidine | 4.01 (33.0) | −2.5 (25.8) | 5.2 (44.0) | 6.1 (36.4) | 10.6 (40.6) | −4.8 (23.7) | 0.77 | −0.13 | 0.13 | −0.089 |
| Phenylalanine | −5.2 (16.3) | −8.1 (27.0) | 3.1 (41.2) | 10.9 (33.8) | 6.7 (27.5) | −5.6 (22.6) | 0.31 | −0.03 | 0.12 | −0.126 |
| Tyrosine | 0.3 (28.4) | −7.6 (29.1) | −4.9 (38.5) | 4.6 (40.1) | 2.7 (30.0) | −3.1 (29.4) | 0.89 | −0.07 | 0.07 | −0.140 |
| Aspartate/asparagine | 38.9 (116.1) | 9.0 (94.9) | 28.5 (86.5) | 43.9 (66.6) | 57.5 (89.6) | 18.4 (86.3) | 0.65 | 0.08 | 0.12 | −0.150 |
| Glutamine/glutamate | 2.0 (38.0) | −6.3 (36.7) | 7.9 (51.9) | 18.9 (51.4) | 19.2 (43.0) | 9.6 (43.5) | 0.54 | 0.03 | 0.22‡ | −0.123 |
| Ornithine | 15.2 (55.5) | 4.7 (39.0) | −4.7 (38.7) | −3.1 (32.2) | 4.4 (41.8) | −6.1 (48.5) | 0.67 | −0.09 | 0.22‡ | −0.018 |
| Citrulline | −2.6 (55.0) | −9.4 (33.3) | −10.2 (56.3) | 6.0 (57.5) | −1.8 (29.5) | −13.7 (35.9) | 0.83 | −0.01 | 0.10 | −0.044 |
| Arginine | −1.4 (39.7) | −4.4 (38.2) | 5.6 (67.0) | −5.2 (30.4) | −5.2 (34.4) | −12.1 (31.4) | 0.87 | −0.10 | 0.25‡‡ | −0.019 |
Metabolite data are shown as mean (SD) per cent changes
p<0.05, Intervention group change vs Inactive group (ANOVA with Dunnett’s post hoc testing)
Overall ANOVA p<0.05 (see Fig. 1 for median per cent changes and nonparametric ANOVA significance testing)
Spearman correlations significant at p<0.05
Spearman correlations significant at p≤0.01
Low/Moderate: low-amount moderate-intensity aerobic exercise; Low/Vigorous: low-amount vigorous-intensity aerobic exercise; High/Vigorous: high-amount vigorous-intensity aerobic exercise; AT/RT: combination of low/vigorous aerobic exercise and RT
Training-induced changes in muscle metabolites correlated with clinical outcomes
Whereas average changes in clinical outcomes and muscle metabolite levels are depicted by group in Tables 1 and 2, it is important to consider that not all participants within a group responded uniformly. For example, Fig. 2 shows that exercise-induced changes in insulin sensitivity varied among individual participants, not only between but also within each intervention group. While individual responses to training vary, the underlying mechanisms for adaptations are likely uniform among individuals such that responses and adaptations should be proportional. This principle forms the basis of our analytical approach wherein we exploited the dynamic range of individual changes to identify clinical outcomes and metabolic intermediates that best track with one another. As predicted, changes in V̇O2peak and TGs were inversely correlated (R = −0.26, p < 0.005). Surprisingly, however, neither of these outcomes was associated with improvements in SI (SI vs V̇O2peak: R = 0.19, p < 0.12; SI vs TGs: R = −0.19, p < 0.11). By contrast, enhanced aerobic fitness, as assessed by V̇O2peak, correlated positively with training-induced increases in skeletal muscle content of most fatty acid-derived (even-chain) acylcarnitines as well as the TCA cycle intermediate, succinate (Table 2, RFig. 1), with correlation coefficients ( values) ranging from 0.2 to 0.3 (p < 0.05). Lowering of plasma TGs was related to increases in many medium- and long-chain fatty acid-derived acylcarnitines, the amino acids alanine and proline and the TCA cycle intermediates citrate, succinate, fumarate, malate and α-ketoglutarate (R = 0.2–0.3, p < 0.05) (Table 2).
Fig. 2.
Variability in changes in insulin sensitivity. Each bar represents the per cent change in insulin sensitivity for an individual participant; changes are ranked from lowest to highest. (a) Individual changes clustered by intervention arm (Inactive; low-amount moderate-intensity aerobic exercise (Low/Moderate); low-amount vigorous-intensity aerobic exercise (Low/Vigorous); high-amount vigorous-intensity aerobic exercise (High/Vigorous); combination of low/vigorous aerobic exercise and resistance training (Low Vigorous/RT); resistance training only (RT). (b) Individual changes irrespective of group
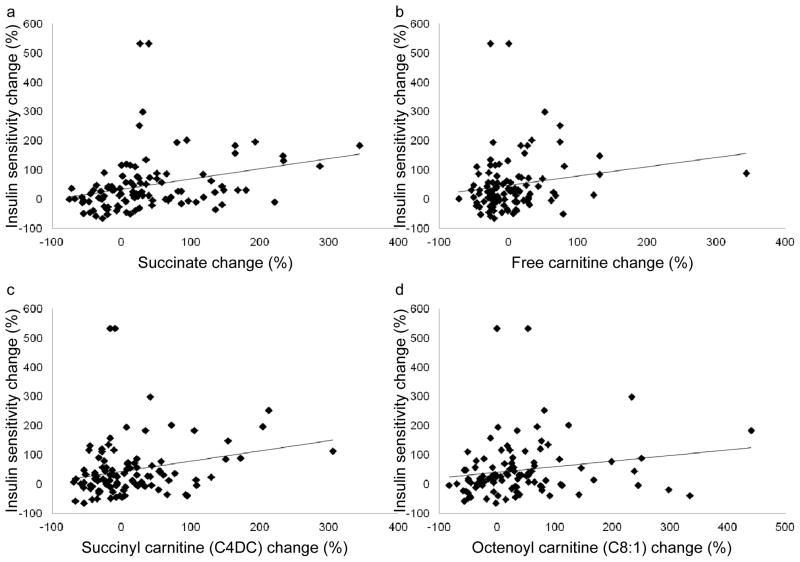
Improvements in SI were related to increases in free carnitine and many of the lipid-derived acylcarnitines (R = 0.2–0.3, p < 0.05; Fig. 3; Table 2), Of note, the relationship between change in insulin sensitivity and free carnitine remained statistically significant even when excluding the outlier with a large (343%) response in free carnitine (R = 0.2, p < 0.05). Stronger associations were observed between changes in SI and the TCA cycle intermediates succinate, fumarate and α-ketoglutarate (R = 0.2–0.4, p < 0.05) (Table 2), as well as alanine, serine, methionine, glutamine/glutamate, ornithine, arginine and the C4-DC acylcarnitine species, which derives in part from succinyl-CoA.
Fig. 3.
Relationship between changes in insulin sensitivity and skeletal muscle metabolites. Scatter plots depicting per cent changes in insulin sensitivity and skeletal muscle succinate (a), free carnitine (b), succinyl carnitine (c) and octenoyl carnitine (d). See Table 2 for Spearman correlations
Exercise-induced changes in muscle metabolites depended on intensity of the training regimen
We found that the relationship between changes in muscle metabolite levels and clinical outcomes appeared to depend on the intensity of the training regimen, as much stronger and some directionally distinct associations emerged when the analyses were performed separately for the vigorous- (65–80% V̇O2peak) compared with the moderate-intensity intervention groups (40–55% V̇O2peak) (see electronic supplementary material [ESM] Table 1). For example, for those performing vigorous-intensity training, associations were stronger between SI and free carnitine, several TCA cycle intermediates, as well as amino acids such as glycine, alanine, proline, leucine/isoleucine, methionine, histidine, glutamine/glutamate, ornithine, citrulline and arginine (R = 0.3–0.5, p < 0.05 for all). Conversely, the same analysis abolished the relation between SI and most acylcarnitines, with the notable exception of C4-DC, succinylcarnitine (R = 0.38, p < 0.005). The relationships between change in SI and succinate, free carnitine, C4-DC/succinylcarnitine and leucine/isoleucine in the vigorous group are depicted in ESM Fig. 1.
Exercise-induced changes in muscle metabolites correlated with transcriptional induction of fatty acid metabolism genes
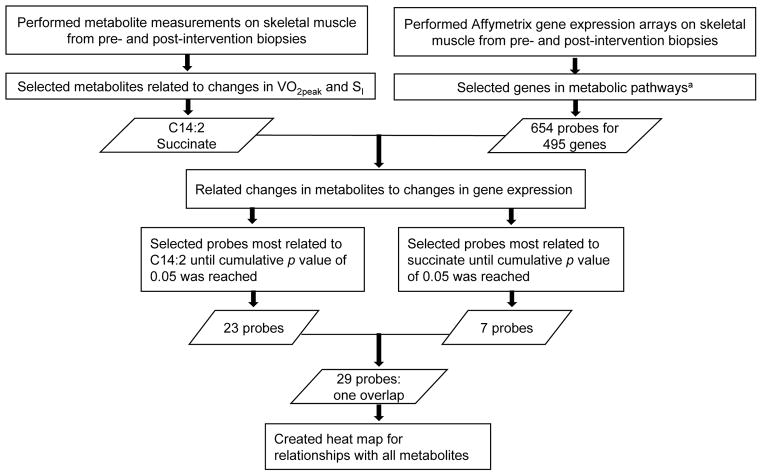
We hypothesised that fasting levels of fatty acid-derived acylcarnitines might reflect muscle abundance of the enzymes that produce these metabolites. Also, we hypothesised that changes in succinate might be related to enzymes in the succinate dehydrogenase node (Fig. 4). To test these hypotheses, we extracted data from our Affymetrix microarray analysis to examine training-induced regulation of a targeted set of 654 probes—corresponding to 495 genes—with functions in the following pathways: TCA cycle, β-oxidation, amino acid metabolism, ketone synthesis and degradation, lipid metabolism and trafficking, oxidative phosphorylation, branched-chain amino acid (BCAA) catabolism, succinate metabolism and glycine metabolism. The last three of these pathways were highlighted based on prior relationships that we observed between these metabolites and insulin action [8, 17]. We related changes in expression of genes associated with these pathways to changes in the metabolites. For those metabolites with changes most closely related to changes in V̇O2peak (C14:2) and SI (succinate), we selected the gene expression changes most related to the changes in metabolites. Gene selection proceeded from the gene related with the smallest p value to those with successively larger p values until a cumulative sum p value of 0.05 was reached. These 30 genes/probes were combined, arranged according to pathway involvement and a heat map showing R > 0.3 and R < −0.3 was created. This process is depicted in Fig. 5.
Fig. 4.
Representation of the succinate dehydrogenase node. Metabolic intermediates are centralised around succinate as a nodal point for the convergence of the TCA cycle, BCAA catabolism, β-oxidation, haem biosynthesis and ketone catabolism. Changes in metabolic intermediates associated with changes in insulin sensitivity are shown in orange. ETS, electron transport system; Suc-CoA, succinyl-CoA; SucL, succinate–CoA ligase; αKDH, α-ketoglutarate dehydrogenase; α-KG, α-ketoglutarate, PCC, propionyl-CoA carboxylase; C4DC-carnitine, succinyl carnitine; SCOT, succinyl-CoA : 3-ketoacid-CoA transferase; BHBDH, β-hydroxybutyrate dehydrogenase; KT, 3- ketoacyl-CoA thiolase. LC-AcCarn, long chain acylcarnitine; Lc-AcCoA, long chain acyl-CoA; AcAc-CoA, acetylacetyl-Coa; MMCM, methylmalonyl-CoA mutase
Fig. 5.
Flow chart for associations of gene expression with metabolites. aPathways: TCA cycle, β-oxidation, amino acid metabolism, ketone synthesis and degradation, lipid metabolism and trafficking, oxidative phosphorylation, BCAA catabolism, succinate metabolism, glycine metabolism
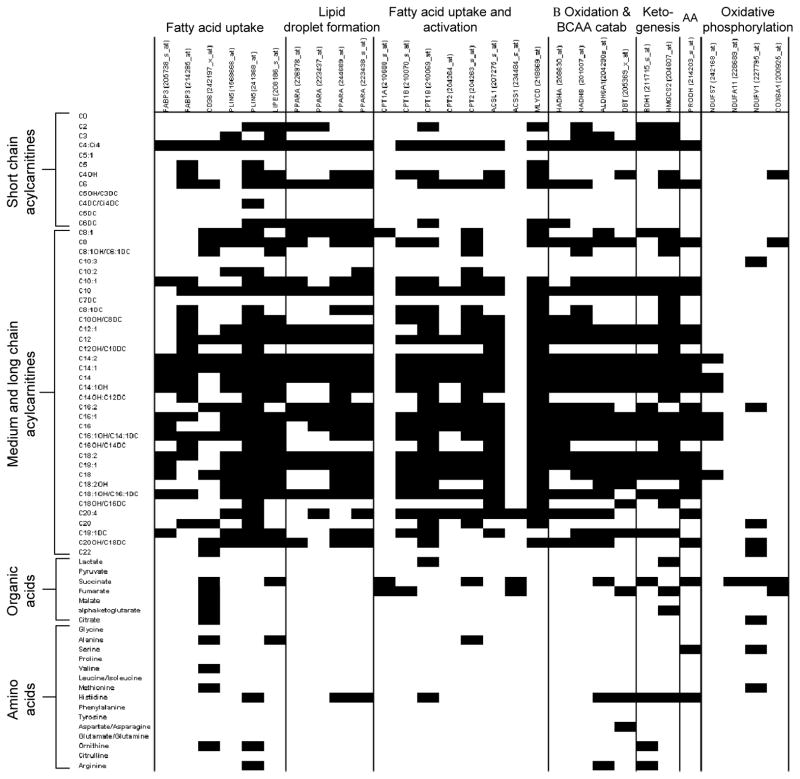
The heat map presented in Fig. 6 shows that exercise-induced increases in muscle acylcarnitines correlated positively with the induction of several lipid metabolic genes (R = 0.3–0.7, p < 0.05). Confidence in the reliability and specificity of this result comes from the finding that these gene/metabolite associations were specific for fatty acid-derived metabolites, as changes in most amino and organic acids were unrelated to lipid-related genes selected for this analysis (Fig. 6). Exercise-induced increases in muscle succinate were correlated with upregulation of a similar set of genes involved in lipid metabolism as well as genes involved in oxidative phosphorylation (Fig. 6).
Fig. 6.
Relationship between changes in skeletal muscle metabolites and expression of genes involved in fatty acid metabolism. Spearman correlations were performed between changes in skeletal muscle metabolites (first column) and changes in skeletal muscle gene expression (first row). Black indicates significant positive relationship (p<0.001)
Discussion
To our knowledge, this is the first investigation to apply targeted, semi-quantitative metabolic profiling to examine metabolic signatures of exercise training in human skeletal muscle and how these relate to both clinical and gene expression responses to exercise training. We studied individuals at risk of cardiometabolic disease to identify metabolic correlates of training-induced improvements in clinical outcomes.
Induction of muscle content of acylcarnitines emerged as the strongest metabolic biomarker of the trained state. These metabolites are generated by a family of mitochondria-localised acyltransferase enzymes that convert acyl-CoA intermediates of fat and amino acid catabolism to their cognate carnitine esters. When substrate supply exceeds metabolic flux at specific catabolic enzymes, the resulting bottleneck leads to acyl-CoA accumulation and a concomitant rise in acylcarnitine production. In the present study, acylcarnitine concentrations measured in the fasted state (when fatty acid supply is plentiful) changed in concert with expression of nuclear-encoded genes that regulate fatty acid uptake and activation, lipid droplet metabolism and fatty acid transport into mitochondria. Most convincing was the finding that changes in muscle levels of lipid-derived acylcarnitines correlated strongly (R = 0.3–0.7) with changes in abundance of mRNA of mitochondrial enzymes in the pathway for producing these metabolites. Thus, exercise-induced reprogramming of mitochondrial metabolism was clearly evident at the metabolite level. Moreover, increased muscle concentrations of even-chain acylcarnitines, as well as several TCA cycle intermediates, were related to improved cardiorespiratory fitness (V̇O2peak) and lowering of circulating TGs, suggesting that enhanced mitochondrial quantity and/or quality contributed to improved lipid clearance.
A distinguishing and important feature of this study was the comparison of metabolic responses across six distinct interventions differing in amount and intensity of exercise. In some but not all cases, changes in muscle metabolite levels were dose responsive with respect to energy expenditure. Interestingly, the associations between metabolites and whole-body cardiometabolic fitness were modified according to the aerobic intensity (40–55% V̇O2peak vs 65–80% V̇O2peak) of the exercise programmes. For example, although vigorous-intensity aerobic training (65–80% V̇O2peak) produced robust increases in several even-chain acylcarnitine species, this response was unrelated to changes in SI in the vigorous-intensity groups. Changes in aerobic fitness (V̇O2peak) were likewise unrelated to SI, consistent with findings of our previous study [3, 18]. Taken together, these results imply that cardiorespiratory capacity and mitochondrial potential for β-oxidation were not primary drivers of improved insulin sensitivity in these cohorts. By contrast, increases in the fat-derived acylcarnitines were much more modest in response to moderate-intensity exercise training, but in this group several of the even-chain species correlated positively with SI. Although our power to interpret these analyses is clearly limited by the small sample size, the results suggest that moderate- vs vigorous-intensity activity might influence the metabolic network and SI via distinct biological mechanisms.
At first glance, the results of the current study appear to conflict with previous reports identifying medium- and long-chain acylcarnitines as metabolic markers of insulin resistance [19–23]. However, emerging evidence argues against the notion that acylcarnitines act as direct mediators of insulin resistance and instead suggests that the carnitine system defends against mitochondrial stress by buffering the local acyl-CoA pool [24]. Unlike their acyl-CoA precursors, the membrane-permeant acylcarnitines can be shuttled between cellular compartments [24]. Thus, elevated acylcarnitines in the settings of obesity and diabetes could reflect an attempt to reroute carbon intermediates from mitochondria to other cellular or extracellular sites. Likewise, in the context of overnight fasting, lipid supply to inactive muscles exceeds metabolic demand. Therefore, we propose that the training-induced rise in fasting levels of long- and medium-chain acylcarnitines coincides with a more robust carnitine buffering system and enhanced capacity to cope with transient fluctuations in metabolic load.
Also notable was the finding that muscle concentrations of free carnitine, succinate and succinylcarnitine, along with several TCA cycle intermediates, emerged as strong, positive correlates of SI, particularly in the vigorous-intensity exercise groups. Although exercise training did not produce consistent increases in muscle levels of free carnitine and succinylcarnitine, participants in whom these metabolites increased tended to show greater improvement in SI (Fig. 3, ESM Fig. 1). Changes in free carnitine correlated strongly with changes in succinylcarnitine (R = 0.71, p < 0.001) and other short-chain carnitine esters, but not with changes in long-chain acylcarnitines. These findings align with recent reports showing that L-carnitine supplementation increased muscle production and efflux of short-chain acylcarnitines in accordance with improved glucose tolerance [20, 24]. Further evidence that mitochondrial acylcarnitine production and/or efflux benefits glucose homeostasis comes from studies showing that mice with muscle-specific deletion of carnitine acetyltransferase (CrAT), the enzyme that generates short-chain acylcarnitines, develop whole-body glucose intolerance [24]. CrAT acts on acyl-CoA species ranging from two to five carbons and is a potential source of the C4-DC carnitine species, which derives from both succinyl-CoA and methylmalonyl-CoA. In the present study, C4-DC correlated strongly with succinate, which derives from succinyl-CoA via succinate dehydrogenase (SDH). We therefore surmise that C4-DC could be reporting on succinyl-CoA. Intriguingly, network reconstruction of the muscle metabolites most strongly associated with improved SI pointed to the SDH complex as a potential regulatory hub that intersects with insulin action (Fig. 4). The lack of strong succinate signature at the gene expression level suggests that the relationship between succinate and SI stems from changes in enzyme/protein activity, local energetics and/or metabolic flux rather than transcriptional regulation.
In addition to the aforementioned succinate and TCA cycle intermediates, improved insulin sensitivity was directly related to skeletal muscle concentrations of several amino acids including alanine, serine, methionine, glutamate/glutamine, ornithine and arginine. These associations were again most pronounced in the vigorous-intensity exercise group. Also, in this group, improvement in insulin sensitivity was related to increases in the BCAAs leucine and isoleucine. This positive relationship was somewhat surprising given that numerous studies in both rodents and humans have linked elevations in circulating BCAAs to insulin resistance and type 2 diabetes [12, 17, 25]. However, it is worth noting that in the previous studies obesity-related increases in peripheral blood BCAA levels were accompanied by a corresponding rise in the odd-chain acylcarnitine byproducts of amino acid catabolism [12, 17, 25]. By contrast, in the muscle tissue from participants in the STRRIDE studies, changes in the C3 and C5 acylcarnitines did not track with their amino acid precursors. Thus, in the context of exercise training, especially during the period following the exercise stimulus (STRRIDE muscle samples were collected 16–24 h after the last exercise training bout), amino acids might be preferentially diverted towards muscle protein synthesis rather than mitochondrial energy production. Another interesting observation was that changes in muscle concentrations of leucine and alanine showed remarkably strong associations with changes in free carnitine and C4-DC (R = 0.64–0.85, p < 0.0001), thereby linking amino acid metabolism to the aforementioned SDH node.
In summary, this investigation identified a subset of skeletal muscle metabolic intermediates that track with exercise-induced improvements in cardiometabolic fitness, including exercise capacity, circulating triacylglycerols and insulin sensitivity. It is important to emphasise that these measurements were made in the overnight fasted state when fatty acids and ketones, and perhaps amino acids, predominate as preferred mitochondrial substrates, and 16–24 h after the last exercise bout, when skeletal muscle metabolic anabolic activity is still relatively high and post-exercise lipid oxidation might continue. Still, despite robust training-induced increases in muscle content of lipid-derived acylcarnitines, these metabolites did not appear to exert a strong influence on insulin sensitivity. Instead, our analysis identified a more compelling connection between SI and succinate metabolism. SDH represents a unique metabolic node where the TCA cycle and electron transport chain converge with the pathways of ketone and amino acid catabolism. Also, SDH is thought to be an important control site for mitochondrial reactive oxygen species emission, which has been shown to influence insulin action in studies using transgenic mouse models [26]. Although the mechanistic relevance of these findings remains uncertain at this stage, the results offer new and provocative clues for further study.
Supplementary Material
Acknowledgments
We thank the rest of the STRRIDE research team at East Carolina University and Duke University.
Funding
This work was supported by the National Heart, Lung, and Blood Institute (National Institutes of Health) R01HL-57354 (WEK), National Institute on Aging (National Institutes of Health) P30 AGO28716-01 (WEK, KMH), R01AG-028930 and R01DK-089312 (DMM) and National Institute of Arthritis and Musculoskeletal and Skin Diseases (National Institutes of Health) K23AR054904 (KMH).
Abbreviations
- BCAA
Branched-chain amino acid
- CrAT
Carnitine acetyltransferase
- SI
Insulin sensitivity index
- SDH
Succinate dehydrogenase
- STRRIDE
Studies of Targeted Risk Reduction Interventions through Defined Exercise
- TCA
Tricarboxylic acid
- TG
Triacylglycerol
- V̇O2peak
Cardiopulmonary fitness as determined by a maximal treadmill test
Footnotes
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Contribution statement
KMH, DMM, and WEK participated in conceptual design, data analysis and data interpretation and drafting and editing of the manuscript. EPH participated in conceptual design, data interpretation and editing of the manuscript. TRK, MJH, HA, NB, LAB, RDS and ORI participated in data collection, data interpretation and editing of the manuscript. All authors approved the final version. KMH is the guarantor of this work.
References
- 1.Physical Activity Guidelines Advisory Committee. Physical Activity Guidelines Advisory Committee Report, 2008. Washington, DC: US Department of Health and Human Services; 2008. [Google Scholar]
- 2.Duscha BD, Slentz CA, Johnson JL, et al. Effects of exercise training amount and intensity on peak oxygen consumption in middle-age men and women at risk for cardiovascular disease. Chest. 2005;128:2788–2793. doi: 10.1378/chest.128.4.2788. [DOI] [PubMed] [Google Scholar]
- 3.Houmard JA, Tanner CJ, Slentz CA, Duscha BD, McCartney JS, Kraus WE. Effect of the volume and intensity of exercise training on insulin sensitivity. J Appl Physiol. 2004;96:101–106. doi: 10.1152/japplphysiol.00707.2003. [DOI] [PubMed] [Google Scholar]
- 4.Kraus WE, Houmard JA, Duscha BD, et al. Effects of the amount and intensity of exercise on plasma lipoproteins. N Engl J Med. 2002;347:1483–1492. doi: 10.1056/NEJMoa020194. [DOI] [PubMed] [Google Scholar]
- 5.Jacobs RA, Lundby C. Mitochondria express enhanced quality as well as quantity in association with aerobic fitness across recreationally active individuals up to elite athletes. J Appl Physiol. 2013;114:344–350. doi: 10.1152/japplphysiol.01081.2012. [DOI] [PubMed] [Google Scholar]
- 6.Bain JR, Stevens RD, Wenner BR, Ilkayeva O, Muoio DM, Newgard CB. Metabolomics applied to diabetes research: moving from information to knowledge. Diabetes. 2009;58:2429–2443. doi: 10.2337/db09-0580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shah SH, Kraus WE, Newgard CB. Metabolomic profiling for the identification of novel biomarkers and mechanisms related to common cardiovascular diseases: form and function. Circulation. 2012;126:1110–1120. doi: 10.1161/CIRCULATIONAHA.111.060368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huffman KM, Slentz CA, Bateman LA, et al. Exercise-induced changes in metabolic intermediates, hormones, and inflammatory markers associated with improvements in insulin sensitivity. Diabetes Care. 2011;34:174–176. doi: 10.2337/dc10-0709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Willis LH, Slentz CA, Bateman LA, et al. Effects of aerobic and/or resistance training on body mass and fat mass in overweight or obese adults. J Appl Physiol. 2012;113:1831–1837. doi: 10.1152/japplphysiol.01370.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bergman RN. Orchestration of glucose homeostasis: from a small acorn to the California oak. Diabetes. 2007;56:1489–1501. doi: 10.2337/db07-9903. [DOI] [PubMed] [Google Scholar]
- 11.Duscha BD, Kraus WE, Keteyian SJ, et al. Capillary density of skeletal muscle: a contributing mechanism for exercise intolerance in class II–III chronic heart failure independent of other peripheral alterations. J Am Coll Cardiol. 1999;33:1956–1963. doi: 10.1016/s0735-1097(99)00101-1. [DOI] [PubMed] [Google Scholar]
- 12.Newgard CB, An J, Bain JR, et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab. 2009;9:311–326. doi: 10.1016/j.cmet.2009.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Haqq AM, Lien LF, Boan J, et al. The Study of the Effects of Diet on Metabolism and Nutrition (STEDMAN) weight loss project: rationale and design. Contemp Clin Trials. 2005;26:616–625. doi: 10.1016/j.cct.2005.09.003. [DOI] [PubMed] [Google Scholar]
- 14.Tumor Analysis Best Practices Working Group. Expression profiling--best practices for data generation and interpretation in clinical trials. Nat Rev Genet. 2004;5:229–237. doi: 10.1038/nrg1297. [DOI] [PubMed] [Google Scholar]
- 15.Seo J, Bakay M, Chen YW, Hilmer S, Shneiderman B, Hoffman EP. Interactively optimizing signal-to-noise ratios in expression profiling: project-specific algorithm selection and detection p-value weighting in Affymetrix microarrays. Bioinformatics. 2004;20:2534–2544. doi: 10.1093/bioinformatics/bth280. [DOI] [PubMed] [Google Scholar]
- 16.Slentz CA, Aiken LB, Houmard JA, et al. Inactivity, exercise, and visceral fat. STRRIDE: a randomized, controlled study of exercise intensity and amount. J Appl Physiol. 2005;99:1613–1618. doi: 10.1152/japplphysiol.00124.2005. [DOI] [PubMed] [Google Scholar]
- 17.Huffman KM, Shah SH, Stevens RD, et al. Relationships between circulating metabolic intermediates and insulin action in overweight to obese, inactive men and women. Diabetes Care. 2009;32:1678–1683. doi: 10.2337/dc08-2075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Huffman KM, Slentz CA, Johnson JL, et al. Impact of hormone replacement therapy on exercise training-induced improvements in insulin action in sedentary overweight adults. Metabolism. 2008;57:888–895. doi: 10.1016/j.metabol.2008.01.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Boyle KE, Canham JP, Consitt LA, et al. A high-fat diet elicits differential responses in genes coordinating oxidative metabolism in skeletal muscle of lean and obese individuals. J Clin Endocrinol Metab. 2011;96:775–781. doi: 10.1210/jc.2010-2253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Noland RC, Koves TR, Seiler SE, et al. Carnitine insufficiency caused by aging and overnutrition compromises mitochondrial performance and metabolic control. J Biol Chem. 2009;284:22840–22852. doi: 10.1074/jbc.M109.032888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Koves TR, Li P, An J, et al. Peroxisome proliferator-activated receptor-gamma co-activator 1alpha-mediated metabolic remodeling of skeletal myocytes mimics exercise training and reverses lipid-induced mitochondrial inefficiency. J Biol Chem. 2005;280:33588–33598. doi: 10.1074/jbc.M507621200. [DOI] [PubMed] [Google Scholar]
- 22.Koves TR, Ussher JR, Noland RC, et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 2008;7:45–56. doi: 10.1016/j.cmet.2007.10.013. [DOI] [PubMed] [Google Scholar]
- 23.Chao LC, Wroblewski K, Zhang Z, et al. Insulin resistance and altered systemic glucose metabolism in mice lacking Nur77. Diabetes. 2009;58:2788–2796. doi: 10.2337/db09-0763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Muoio DM, Noland RC, Kovalik JP, et al. Muscle-specific deletion of carnitine acetyltransferase compromises glucose tolerance and metabolic flexibility. Cell Metab. 2012;15:764–777. doi: 10.1016/j.cmet.2012.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wang TJ, Larson MG, Vasan RS, et al. Metabolite profiles and the risk of developing diabetes. Nat Med. 2011;17:448–453. doi: 10.1038/nm.2307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Anderson EJ, Lustig ME, Boyle KE, et al. Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. J Clin Invest. 2009 doi: 10.1172/JCI37048. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.