Abstract
Background
The relationship between Klebsiella pneumoniae and nosocomial and community-acquired infections is well known, and K. pneumoniae resistance to most antibiotics is increasing worldwide. In contrast, tigecycline remains active against many bacterial strains, and serves as a last resort for treating multi-drug resistant bacterial infections. That tigecycline nonsusceptibility among K. pneumoniae isolates has been reported worldwide is worrying. However, the mechanisms of tigecycline resistance in K. pneumoniae are less well known. We report the genome sequence and genomic characterization of tigecycline-resistant K. pneumoniae strain 5422 isolated from the bile samples of a patient with cholangiocarcinoma.
Results
We sequenced the K. pneumoniae strain 5422 genome using next-generation sequencing technologies. Sequence data assembly revealed a 5,432,440-bp draft genome and 57.1% G + C content, which contained 5397 coding sequences. The genome has extensive similarity to other sequenced K. pneumoniae genomes, but also has several resistance-nodulation-cell division (RND) efflux pump genes that may be related to tigecycline resistance.
Conclusions
K. pneumoniae strain 5422 is resistant to multiple antibiotics. The genome sequence of the isolate and comparative analysis with other K. pneumoniae strains presented in this paper are important for better understanding of K. pneumoniae multi-drug resistance. The RND efflux pump genes identified in the genome indicate the presence of an antibiotic resistance mechanism prior to antibiotics overuse. The availability of the genome sequence forms the basis for further comparative analyses and studies addressing the evolution of the K. pneumoniae drug resistance mechanism and the K. pneumoniae transcriptome.
Keywords: Tigecycline-resistant, Klebsiella pneumoniae, Cholangiocarcinoma, Next-generation sequencing, Comparative genomics
Background
Klebsiella pneumoniae is a Gram-negative opportunistic pathogen from the family Enterobacteriaceae. The increasing resistance detected in clinical isolates has become a matter of significant concern, and K. pneumoniae is an ESKAPE (Enterococcus faecium, Staphylococcus aureus, K. pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species) pathogen [1,2]. Infections caused by K. pneumoniae have been identified worldwide. It is worth noting that it is a major cause of nosocomial and community-acquired infections [3,4]. Moreover, a distinct invasive syndrome that causes liver abscesses has been detected in increasing numbers in Asia in the past two decades, and this syndrome is emerging as a global disease [5,6]. Furthermore, K. pneumoniae harboring extended-spectrum β-lactamases, and more recently, carbapenemase, which confers resistance to multiple antibiotics, has become a significant clinical concern worldwide [7,8]. Mortality among patients infected with extremely resistant K. pneumoniae is high, perhaps due to the limited therapeutic options remaining [9].
Tigecycline, a novel class of glycylcyclines, is a minocycline derivative synthesized to overcome the major tetracycline resistance mechanisms and to extend its spectrum of activity to multidrug-resistant (MDR) bacteria [10]. Tigecycline has enhanced antimicrobial activity compared to tetracycline, and can overcome efflux pump systems and ribosome protection mechanisms, retaining activity against a broad range of both Gram-positive and Gram-negative bacteria [11-13]. K. pneumoniae resistance to most antibiotics is increasing globally. Nevertheless, tigecycline remains active against many bacterial strains, and serves as last resort for treating MDR bacterial infections [14]. However, tigecycline resistance has emerged recently and been widely reported in Enterobacteriaceae isolates. It is worth noting that tigecycline non-susceptibility among K. pneumoniae isolates has been reported from different continents and ranges between 0% and 50% [15-17].
Previous studies have suggested that tigecycline resistance in Enterobacteriaceae is attributed to resistance-nodulation-cell division (RND)-type efflux pumps and transcriptional regulators of the efflux pump systems [15]. However, reports on the tigecycline resistance mechanisms in K. pneumoniae are rare. We hypothesized that a wide range of genes is involved in tigecycline resistance and contribute to decreased tigecycline susceptibility in K. pneumoniae, and performed whole-genome sequencing (WGS) to investigate this. We report the genome sequence of the tigecycline-resistant K. pneumoniae strain 5422 isolated from the bile samples of a patient with cholangiocarcinoma.
Methods
Strain information and growth conditions
Previously, we isolated strain 5422 from a bile sample obtained from a 54-year-old woman with bile duct cancer on February 25, 2012. After enrichment in Mueller-Hinton broth, the strain was identified as K. pneumoniae following the combination of its 16S rRNA gene sequencing and biochemical reaction results (VITEK 2 compact, bioMérieux, France). The strain exhibited high resistance to ciprofloxacin, cefotaxime, cefoxitin, ampicillin/sulbactam, sulfamethoxazole, tigecycline, tetracycline, and piperacillin, and was susceptible to imipenem, meropenem, and gentamicin. Multi-locus sequence typing revealed that it belonged to ST37 (unpublished data). The strain we reported here is available in the State Key Laboratory for Diagonosis and Treatment of Infectious Diseases, Zhejiang University.
Genomic DNA extraction
Late log-phase cells were harvested and lysed with EDTA, lysozyme, and detergent treatment, followed by proteinase K and RNase digestion. Genomic DNA was extracted using a DNeasy Blood & Tissue Kit (Qiagen, Germany) according to the manufacturer’s recommended protocol. Genomic DNA yield, purity, and concentration was evaluated using 0.7% agarose gel electrophoresis with λ-Hind III digest DNA Marker and measured using a NanoDrop 1000 Spectrophotometer (Thermo Fisher Scientific, USA). The genomic DNA was stored at-20°C.
Genome sequencing and annotation
Whole-genome shotgun sequencing of K. pneumoniae strain 5422 was performed using a standard run of IlluminaHiSeq2000 sequencing by generating paired-end libraries (500-bp insert size) with a 2 × 100 pair-end sequencing strategy according to the manufacturer’s instructions. Clean reads were assembled into scaffolds using Velvet version 1.2.07 [18], and then we used PAGIT (Post-Assembly Genome Improvement Toolkit) [19] to extend the initial contiguous sequences (contigs) and correct sequencing errors. We identified tRNAs and rRNAs using tRNAscan-SE [20] and RNAmmer [21], respectively. Open reading frames (ORFs) were identified using Glimmer version 3.0 [22]. The genome was annotated using the RAST (Rapid Annotation using Subsystem Technology) server [23]. The classification of some predicted genes and pathways was analyzed using the COGs (Clusters of Orthologous Groups of proteins) [24] and KEGG (Kyoto Encyclopedia of Genes and Genomes) [25] databases. Stretches of amino acids containing the efflux pump genes were searched using BLAST (Basic Local Alignment Search Tool, http://blast.ncbi.nlm.nih.gov/Blast.cgi); protein-coding sequences were further BLAST-searched against the Antibiotic Resistance Database (ARDB) [26]. To find genes with hypothetical or putative functions, we aligned genes against the National Center for Biotechnology Information (NCBI) nucleotide sequence database (downloaded September 20, 2013) using NCBI BLASTn: we accepted only hits with identity of ≥ 0.95, coverage ≥ 0.9, and putative or hypothetical reference gene annotation.
Initial comparative genomic and phylogenetic analysis
For comparative analysis, we downloaded the reference genome sequences of the closest genetic relatives of K. pneumoniae strain 5422 and representative strains from the NCBI website: K. pneumoniae LCT-KP182 (ATRN00000000), K. pneumoniae LCT-KP289 (ATRO00000000), K. pneumoniae subsp. pneumoniae LZ (AJVY00000000), K. pneumoniae ATCC BAA-2146 (AOCV00000000), K. pneumoniae 12 3578 (PRJNA199972), K. pneumoniae NB60 (AZAP00000000), K. pneumoniae subsp. pneumoniae 1084 (CP003785), K. pneumoniae subsp. pneumoniae NTUH-K2044 (AP006725), K. pneumoniae subsp. pneumoniae WGLW5 (AMLO00000000), K. pneumoniae ATCC 25955 (AQQH00000000), and K. pneumoniae G5-2 (AQQI00000000). Whole-genome alignments, single-nucleotide polymorphism (SNP) identification, and phylogenetic tree construction were performed using snpTree version 1.1, a server for online automatic SNP analysis of assembled genomes (http://cge.cbs.dtu.dk/services/snpTree-1.1/) [27].
Quality assurance
The 16S rDNA gene from the draft genome was used to check for contamination. Further VITEK biochemical identification data confirmed that the strain 5422 belonged to K. pneumoniae. Bioinformatics assessment of potential contamination of the genomic library by allochthonous microorganisms was achieved using the BLAST non-redundant database.
Initial findings
Genome characteristics and phylogenetic analysis
Filtered 520.8 M clean reads were assembled into scaffolds, and corresponding 99-fold coverage of the genome was generated. The draft genome sequence of K. pneumoniae strain 5422 was 5,432,440 bp in size and had a G + C content of 57.1% in 133 contigs, with N50 spanning 105,586 bp. Figure 1A depicts the overall genome profile. Annotation of this assembly identified 5,397 coding sequences (CDSs), 65 tRNAs (excluding 0 pseudo tRNAs), and incomplete rRNA operons (three small subunit rRNAs, four large subunit rRNAs). We assigned putative function or hypothetical proteins to 1,478 protein-coding genes. We categorized 4,218 genes into COGs functional groups (including putative or hypothetical genes, Figure 1B). For COGs distribution, R (general function prediction only; 874 ORFs), E (amino acid transport and metabolism; 747 ORFs), G (carbohydrate metabolism and transport; 679 ORFs), and P (inorganic ion transport and metabolism; 525 ORFs) were abundant categories (>10% of total COGs matched counts).
Figure 1.
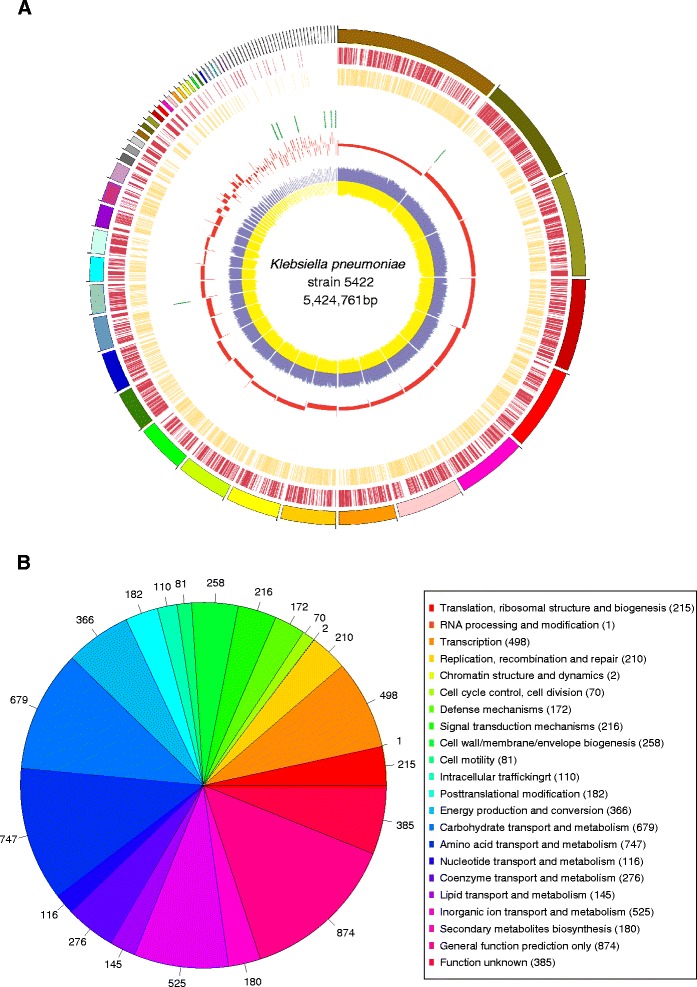
Genome map and phylogenetic analysis. A. Circular map of the genome generated using Circos. Circles from outside to inside: 1, contigs were arrange in clockwise direction from large to small; 2, CDS on forward strand; 3, CDS on reverse strand; 4, tRNA genes; 5, rRNA genes; 6, GC-skew (window size of 10 kb); and 7, blue indicates C content and yellow indicates G content (step size 500 bp). B. COGs distribution of K. pneumoniae strain 5422.
Figure 2A illustrates the subsystem distribution and general information on the potential functional distribution of K. pneumoniae strain 5422. Genes responsible for carbohydrates (825 ORFs); amino acids and derivatives (520 ORFs); and cofactors, vitamins, prosthetic groups, and pigments (325 ORFs) were abundant among the SEED subsystem categories. Based on the raw reads and assembled genomes from published K. pneumoniae WGS data sets, we conducted phylogenetic analysis on tree topology and the SNP positions of the reference genome to identify the most closely related organism. The phylogenetic tree based on whole-genome SNPs showed that the closest ancestor to K. pneumoniae strain 5422 was K. pneumoniae ATCC BAA-2146 (Figure 2B), which is the first US isolate found to encode New Delhi metallo-β-lactamase 1 (NDM-1), eight β-lactamases, and 15 additional antibiotic-resistance enzymes [28,29].
Figure 2.
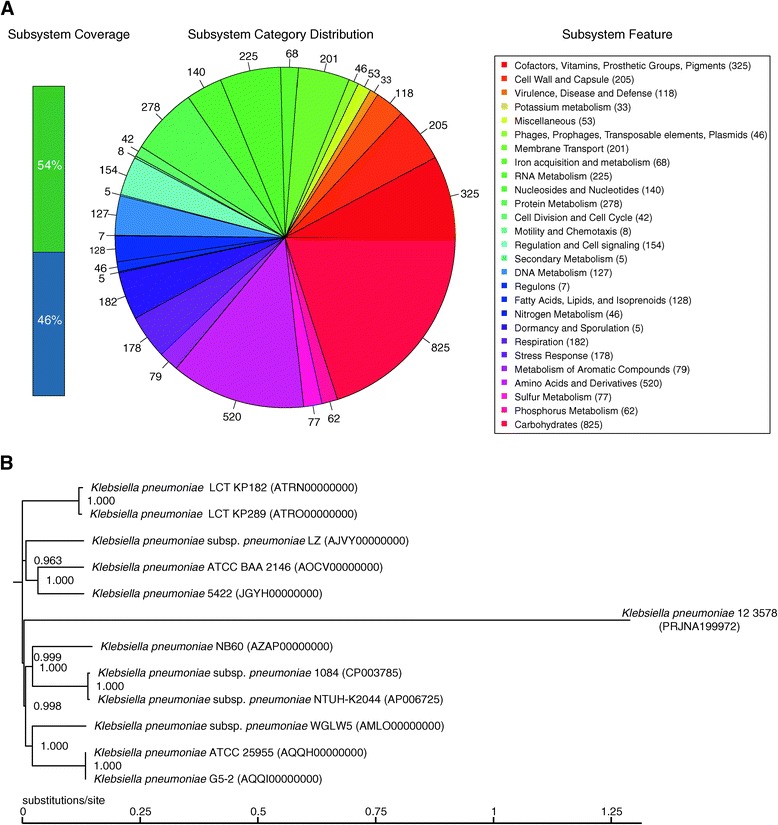
Subsystem distribution and phylogenetic analysis. A. Distribution of genes assigned to SEED subsystems (based on the RAST annotation server). B. Phylogenetic relationships (based on WGS and SNPs) of 12 K. pneumoniae strains and their genomic distance analysis. The snpTree server output used assembled genomes as input data.
RND efflux pumps in K. pneumoniae strain 5422
The RND family members are important mediators of MDR in Gram-negative bacteria. The AcrAB-TolC system in Escherichia coli and the MexAB-OprM complexes in P. aeruginosa are extremely well characterized, and the three-dimensional structures of various components have been resolved [30]. In the genome of K. pneumoniae strain 5422, 16 genes were indicated as probable efflux pumps or translational regulators based on their sequence similarity to known RND efflux pump genes (Table 1).
Table 1.
Summary of CDSs annotated to RND efflux pump genes
| Aligned protein | Query sequence length | Coverage (%) | Hit length | Identity (%) | Description |
|---|---|---|---|---|---|
| AcrA | 240 | 64.17 | 146 | 60.08 | Probable RND efflux membrane fusion protein |
| OqxB | 217 | 20.66 | 141 | 64.98 | Probable RND efflux system inner membrane transporter CmeB |
| OqxB | 181 | 18.53 | 110 | 60.77 | Probable RND multi-drug efflux transporter |
| OqxB | 160 | 15.27 | 105 | 65.62 | Probable RND efflux system inner membrane transporter CmeB |
| AcrA | 397 | 100 | 397 | 100 | Membrane fusion protein of RND family multi-drug efflux pump |
| OqxR | 483 | 100 | 480 | 99.38 | Transcriptional regulator |
| OqxB | 1050 | 100 | 1050 | 100 | RND multi-drug efflux transporter |
| OqxA | 391 | 100 | 391 | 100 | RND multi-drug efflux transporter |
| RarA | 369 | 100 | 366 | 99.19 | Bacterial regulatory helix-turn-helix proteins, AraC family |
| OqxB | 161 | 15.33 | 101 | 62.73 | Probable RND multi-drug efflux transporter |
| AcrA | 328 | 83.89 | 248 | 75.61 | Probable RND efflux system membrane fusion protein CmeA |
| OqxB | 217 | 20.67 | 143 | 65.9 | Probable RND efflux system inner membrane transporter CmeB |
| TolC | 1520 | 100 | 1474 | 96.97 | Outer membrane efflux protein |
| OqxB | 326 | 31.05 | 200 | 61.35 | Probable RND efflux system inner membrane transporter CmeB |
Future directions
The rapid progress of WGS has permitted detailed investigation of genetic differences between bacterial isolates with different phenotypic characteristics. Whole-genome studies of K. pneumoniae have mainly focused on comparing either distinct antibiotic-susceptible and MDR strains or related isolates from different patients. Therefore, large-scale genomic sequencing and comparative genome analysis of tigecycline-resistant, tigecycline non-susceptible, and tigecycline-susceptible clinical isolates will identify the differences in the genomic content of this species and yield evolutionary information on the development of tigecycline resistance through mutations. Moreover, further studies involving extensive high-throughput mRNA sequencing (RNA-Seq) experiments to significantly improve annotation and to provide exceptionally robust analysis of RNA expression under selective antibiotic pressure are warranted.
Ethics approval
This research was approved by the Research Ethics Committee of the First Affiliated Hospital, School of Medicine, Zhejiang University, and informed consent was obtained from the patient.
Availability of supporting data
This Whole Genome Shotgun project has been deposited at DDBJ/EMBL/GenBank under the accession JGYH00000000. The version described in this paper is version JGYH01000000.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (Grant No. 81361138021 and 81301461), Zhejiang Provincial Natural Science Foundation of China (Grant No. LQ13H190002) and the Scientific Research Foundation of Zhejiang Provincial Health Bureau (Grant No. 2012KYB083).
Footnotes
Beiwen Zheng, and Ang Li, are contributed equally.
Competing interests
The authors have no competing interests.
Authors’ contributions
BWZ, XJH, LNZ, JRJ and MY performed microbiology and molecular biology experiments. XWJ and JY generated and analyzed the sequencing data. BWZ and AL participated all discussions of data analysis and write the manuscript. BWZ, YHX and LJL were involved in overall experimental design. All authors read and approved the final manuscript.
Contributor Information
Beiwen Zheng, Email: zhengbw@zju.edu.cn.
Ang Li, Email: dd152003@gmail.com.
Xiawei Jiang, Email: xiaweijiang@gmail.com.
Xinjun Hu, Email: hxj5129@163.com.
Jian Yao, Email: lifeyaojian@gmail.com.
Lina Zhao, Email: lnzhao@live.cn.
Jinru Ji, Email: susu0572@126.com.
Min Ye, Email: maicye@126.com.
Yonghong Xiao, Email: xiao-yonghong@163.com.
Lanjuan Li, Email: lilj@zju.edu.cn.
References
- 1.Boucher HW, Talbot GH, Bradley JS, Edwards JE, Gilbert D, Rice LB, Scheld M, Spellberg B, Bartlett J. Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin Infect Dis. 2009;48:1–12. doi: 10.1086/595011. [DOI] [PubMed] [Google Scholar]
- 2.Rice LB. Federal funding for the study of antimicrobial resistance in nosocomial pathogens: no ESKAPE. J Infect Dis. 2008;197:1079–1081. doi: 10.1086/533452. [DOI] [PubMed] [Google Scholar]
- 3.Moore R, O'Shea D, Geoghegan T, Mallon PW, Sheehan G. Community-acquired Klebsiella pneumoniae liver abscess: an emerging infection in Ireland and Europe. Infection. 2013;41:681–686. doi: 10.1007/s15010-013-0408-0. [DOI] [PubMed] [Google Scholar]
- 4.Henderson KL, Muller-Pebody B, Johnson AP, Wade A, Sharland M, Gilbert R. Community-acquired, healthcare-associated and hospital-acquired bloodstream infection definitions in children: a systematic review demonstrating inconsistent criteria. J Hosp Infect. 2013;85:94–105. doi: 10.1016/j.jhin.2013.07.003. [DOI] [PubMed] [Google Scholar]
- 5.Siu LK, Yeh KM, Lin JC, Fung CP, Chang FY. Klebsiella pneumoniae liver abscess: a new invasive syndrome. Lancet Infect Dis. 2012;12:881–887. doi: 10.1016/S1473-3099(12)70205-0. [DOI] [PubMed] [Google Scholar]
- 6.Lin JC, Koh TH, Lee N, Fung CP, Chang FY, Tsai YK, Ip M, Siu LK. Genotypes and virulence in serotype K2 Klebsiella pneumoniae from liver abscess and non-infectious carriers in Hong Kong, Singapore and Taiwan. Gut Pathog. 2014;6:21. doi: 10.1186/1757-4749-6-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nordmann P, Cuzon G, Naas T. The real threat of Klebsiella pneumoniae carbapenemase-producing bacteria. Lancet Infect Dis. 2009;9:228–236. doi: 10.1016/S1473-3099(09)70054-4. [DOI] [PubMed] [Google Scholar]
- 8.Richter SN, Frasson I, Franchin E, Bergo C, Lavezzo E, Barzon L, Cavallaro A, Palu G. KPC-mediated resistance in Klebsiella pneumoniae in two hospitals in Padua, Italy, June 2009-December 2011: massive spreading of a KPC-3-encoding plasmid and involvement of non-intensive care units. Gut Pathog. 2012;4:7. doi: 10.1186/1757-4749-4-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Munoz-Price LS, Poirel L, Bonomo RA, Schwaber MJ, Daikos GL, Cormican M, Cornaglia G, Garau J, Gniadkowski M, Hayden MK, Kumarasamy K, Livermore DM, Maya JJ, Nordmann P, Patel JB, Paterson DL, Pitout J, Vilegas MV, Wang H, Woodford N, Quinn JP. Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. Lancet Infect Dis. 2013;13:785–796. doi: 10.1016/S1473-3099(13)70190-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Petersen PJ, Jacobus NV, Weiss WJ, Sum PE, Testa RT. In vitro and in vivo antibacterial activities of a novel glycylcycline, the 9-t-butylglycylamido derivative of minocycline (GAR-936) Antimicrob Agents Chemother. 1999;43:738–744. doi: 10.1128/aac.43.4.738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jenner L, Starosta AL, Terry DS, Mikolajka A, Filonava L, Yusupov M, Blanchard SC, Wilson DN, Yusupova G. Structural basis for potent inhibitory activity of the antibiotic tigecycline during protein synthesis. Proc Natl Acad Sci U S A. 2013;110:3812–3816. doi: 10.1073/pnas.1216691110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brust K, Evans A, Plemmons R. Tigecycline in treatment of multidrug-resistant Gram-negative bacillus urinary tract infections: a systematic review. J Antimicrob Chemother. 2014;ᅟ:ᅟ. doi: 10.1093/jac/dku189. [DOI] [PubMed] [Google Scholar]
- 13.Kanj SS, Whitelaw A, Dowzicky MJ. In vitro activity of tigecycline and comparators against Gram-positive and Gram-negative isolates collected from the Middle East and Africa between 2004 and 2011. Int J Antimicrob Agents. 2014;43:170–178. doi: 10.1016/j.ijantimicag.2013.10.011. [DOI] [PubMed] [Google Scholar]
- 14.Lin YT, Wang FD, Chan YJ, Fu YC, Fung CP. Clinical and microbiological characteristics of tigecycline non-susceptible Klebsiella pneumoniae bacteremia in Taiwan. BMC Infect Dis. 2014;14:1. doi: 10.1186/1471-2334-14-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sun Y, Cai Y, Liu X, Bai N, Liang B, Wang R. The emergence of clinical resistance to tigecycline. Int J Antimicrob Agents. 2013;41:110–116. doi: 10.1016/j.ijantimicag.2012.09.005. [DOI] [PubMed] [Google Scholar]
- 16.Nigo M, Cevallos CS, Woods K, Flores VM, Francis G, Perlman DC, Revuelta M, Mildvan D, Waldron M, Gomez T, Koshy S, Jodlowski T, Riley W, Ruhe JJ. Nested case–control study of the emergence of tigecycline resistance in multidrug-resistant Klebsiella pneumoniae. Antimicrob Agents Chemother. 2013;57:5743–5746. doi: 10.1128/AAC.00827-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Huang TD, Berhin C, Bogaerts P, Glupczynski Y. In vitro susceptibility of multidrug-resistant Enterobacteriaceae clinical isolates to tigecycline. J Antimicrob Chemother. 2012;67:2696–2699. doi: 10.1093/jac/dks288. [DOI] [PubMed] [Google Scholar]
- 18.Zerbino DR, Birney E. Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res. 2008;18:821–829. doi: 10.1101/gr.074492.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Swain MT, Tsai IJ, Assefa SA, Newbold C, Berriman M, Otto TD. A Post-Assembly Genome-Improvement Toolkit (PAGIT) to obtain annotated genomes from contigs. Nat Protoc. 2012;7:1260–1284. doi: 10.1038/nprot.2012.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lowe TM, Eddy SR. tRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res. 1997;25:0955–0964. doi: 10.1093/nar/25.5.0955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lagesen K, Hallin P, Rødland EA, Stærfeldt H-H, Rognes T, Ussery DW. RNAmmer: consistent and rapid annotation of ribosomal RNA genes. Nucleic Acids Res. 2007;35:3100–3108. doi: 10.1093/nar/gkm160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Delcher AL, Bratke KA, Powers EC, Salzberg SL. Identifying bacterial genes and endosymbiont DNA with Glimmer. Bioinformatics. 2007;23:673–679. doi: 10.1093/bioinformatics/btm009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Aziz RK, Bartels D, Best AA, De Jongh M, Disz T, Edwards RA, Formsma K, Gerdes S, Glass EM, Kubal M, Meyer F, Olsen GJ, Olson R, Osterman AL, Overbeek RA, McNeil LK, Paarmann D, Paczian T, Parrello B, Pusch GD, Reich C, Stevens R, Vassieva O, Vonstein V, Wilke A, Zagnitko O. The RAST Server: rapid annotations using subsystems technology. BMC Genomics. 2008;9:75. doi: 10.1186/1471-2164-9-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tatusov RL, Fedorova ND, Jackson JD, Jacobs AR, Kiryutin B, Koonin EV, Krylov DM, Mazumder R, Mekhedov SL, Nikolskaya AN, Rao BS, Smirnov S, Sverdlov AV, Vasudevan S, Wolf YI, Yin JJ, Natale DA. The COG database: an updated version includes eukaryotes. BMC Bioinformatics. 2003;4:41. doi: 10.1186/1471-2105-4-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mitra S, Rupek P, Richter DC, Urich T, Gilbert JA, Meyer F, Wilke A, Huson DH. Functional analysis of metagenomes and metatranscriptomes using SEED and KEGG. BMC Bioinformatics. 2011;12 Suppl 1:S21. doi: 10.1186/1471-2105-12-S1-S21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu B, Pop M. ARDB–Antibiotic Resistance Genes Database. Nucleic Acids Res. 2009;37:D443–D447. doi: 10.1093/nar/gkn656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Leekitcharoenphon P, Kaas RS, Thomsen MC, Friis C, Rasmussen S, Aarestrup FM. snpTree--a web-server to identify and construct SNP trees from whole genome sequence data. BMC Genomics. 2012;13 Suppl 7:S6. doi: 10.1186/1471-2164-13-S7-S6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Broberg CA, Palacios M, Miller VL. Whole-genome draft sequences of three multidrug-resistant klebsiella pneumoniae strains available from the American type culture collection. Genome Announc. 2013;1:e00312–e00313. doi: 10.1128/genomeA.00312-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hudson CM, Bent ZW, Meagher RJ, Williams KP. Resistance determinants and mobile genetic elements of an NDM-1-encoding Klebsiella pneumoniae strain. PLoS One. 2014;9:e99209. doi: 10.1371/journal.pone.0099209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Alvarez-Ortega C, Olivares J, Martinez JL. RND multidrug efflux pumps: What are they good for? Front Microbiol. 2013;4:7. doi: 10.3389/fmicb.2013.00007. [DOI] [PMC free article] [PubMed] [Google Scholar]


