Abstract
The delivery of exogenous agents can enable noninvasive disease monitoring, but existing low-dose approaches require complex infrastructure. In this paper, we describe a microdose-scale injectable formulation of nanoparticles that interrogate the activity of thrombin, a key regulator of clotting, and produce urinary reporters of disease state. We establish a customized single molecule detection assay that enables urinary discrimination of thromboembolic disease in mice using doses of the nanoparticulate diagnostic agents that fall under regulatory guidelines for “microdosing.”
1. Introduction
Biomarkers are endogenously produced indicators of biological state and encompass metabolites, proteins, nucleic acids, and other biological compounds.1 An ideal biomarker provides specific information about a disease for screening, diagnosis, or prognosis. Unfortunately, biomarkers are often limited by lack of specificity or sensitivity, interpatient variability, or poorly understood disease-biomarker biology.2 A promising strategy to bypass these limitations is to introduce exogenous compounds that exploit pathological differences in metabolism, biodistribution, or excretion to produce a unique, specific, and detectable biomarker. An example is [18F]fluoro-2-deoxyglucose, which is metabolized identically to glucose and enables visualization of diseases with increased glucose metabolism (e.g., cancer, Alzheimer’s disease) by positron emission tomography (PET).3
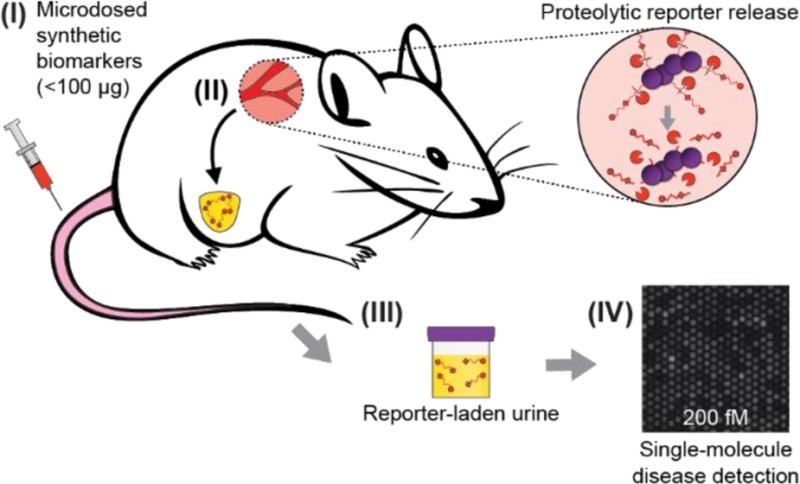
In a similar vein, we have developed “synthetic biomarkers”, nanoparticle agents that sample disease-associated proteases and produce a urinary signal.4−6 Upon intravenous (IV) injection, synthetic biomarkers passively home to sites of disease, where reporters are liberated by proteolytic cleavage of a disease-tuned peptide substrate and subsequently filter into the urine as reporters of disease state (Figure 1A). Although synthetic biomarkers have the potential to improve disease diagnosis and monitoring, a challenge to their adoption is the use of administered synthetic compounds, which require rigorous evaluation by the U.S. Food and Drug Administration (FDA) before approval and use in humans. By comparison, the FDA approval time frame for new radioisotope agents can be minimized by using minute doses that are still detectable owing to PET’s remarkable sensitivity and are therefore regulated as “microdoses” (≤100 μg), greatly compressing the time frame and simplifying the translational path to the clinic.7
Figure 1.
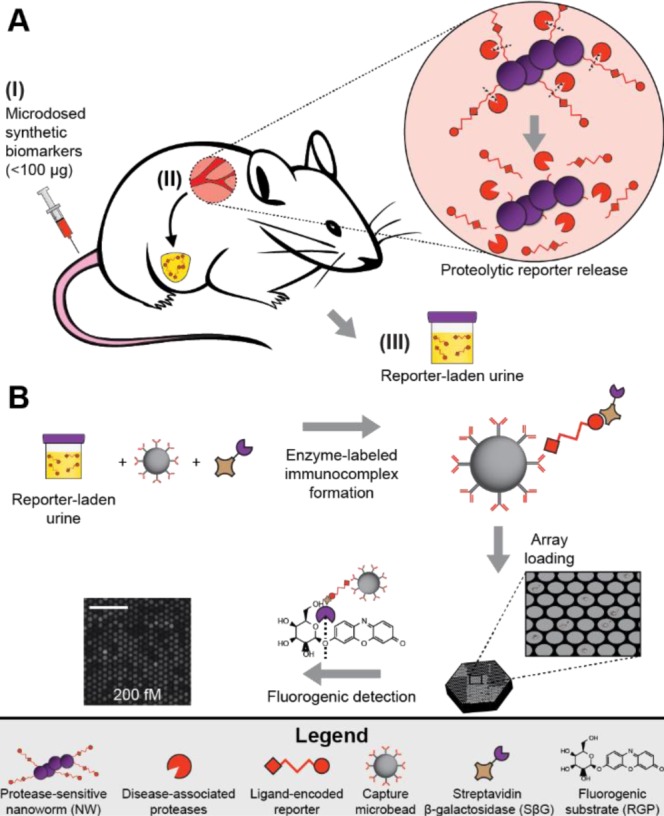
(A) Injected synthetic biomarkers (I) release reporters upon interaction with disease-associated proteases (II). Ligand-encoded reporters liberated from carrier nanoparticles are small enough to be concentrated into the urine (III). (B) The SiMoA assay uses capture antibody-coated beads and SβG to form reporter–sandwich complexes. Beads are loaded into arrays of ∼50 000 wells and sealed with fluorogenic substrate RGP to detect single sandwich complexes. Scale is 50 μm.
Inspired by this approach, we combined our existing single molecule array (SiMoA) technology with synthetic biomarkers to enable disease detection at microdose levels. SiMoA technology has been applied to the subfemtomolar detection of PSA, HIV p24 protein, genomic DNA and cytokines based on counting of single molecules.8,9 SiMoA uses antibody-coated capture beads to facilitate the formation of enzyme-labeled sandwich complexes in the presence of low concentrations of analyte; upon singulation of beads into ∼50 000 50 fL fiber optic wells, the signal generated by fluorogenic substrate cleavage from even a single enzyme-labeled sandwich complex may be detected. Using reformulated disease reporters with stable poly(ethylene glycol) linkers, we modified the SiMoA technology to capture and directly detect synthetic compounds rather than biologically derived molecules without relying on a sandwich immunoassay by using prebiotinylated analytes that reduce processing time, reagent use, and sources of noise introduced by secondary recognition agents.
These new tools allow study of stability, pharmacokinetics, and diagnostic performance at microdose levels, without requiring use of radioactive or stable isotope reformulations of our reporters as required in previous types of microdose studies.3,7 Here, we describe the use of microdosed thrombin-sensitive synthetic biomarkers in mice coupled to our customized single molecule detection platform (Figure 1B) to detect thrombosis, a disease with poor biomarkers owing to an inability to directly detect thrombin activity and a reliance on measuring upstream or downstream byproducts of clotting.5,10 Our approach obviates costly, specialized radioisotopes and sophisticated sample preparation typical of microdose studies.
2. Experimental Section
2.1. Reporter/Peptide Synthesis
Thrombin-sensitive peptides were synthesized by CPC Scientific, with the sequence Biotin-PEG5kDa-Lys(5FAM)-Gly-Gly-DPhe-Pro-Arg-Ser-Gly-Gly-Gly-Cys, where PEG5kDa is 5 kDa poly(ethylene glycol). Standard (nonprotease-sensitive) ligand-encoded reporters R1–2 were synthesized by derivatizing biotin-poly(ethylene glycol) 5 kDa-amine (Laysan) with NHS-fluorescein (R1; Sigma) or NHS-Alexa Fluor 488 (R2; Invitrogen).
2.2. Protease-Sensitive R1-Nanoworm (NW) Synthesis
Dextran-cross-linked nanoworms (NWs) were synthesized as previously described.11,12 Aminated NWs were activated with N-succinimidyl iodoacetate (SIA; Pierce) in a 500-fold molar excess to NWs. Activated NWs were purified and reacted with sulfhydryl-terminated protease-sensitive peptide reporters (95-fold excess; thrombin-sensitive R1 is terminated with a cysteine) and mPEG20kDa-SH (20-fold excess) for 1 h, resulting in thrombin-sensitive peptide valency of 50–70 per NW. R1-NW concentrations refer to the concentration of thrombin-sensitive R1-substrate peptides on the NW surface, with molecular weight of NW carrier included (see Supporting Information for calculations).
2.3. Enzyme-Linked Immunosorbent Assay (ELISA)
ELISAs for modified R1–2 were performed as previously described.5,6 Briefly, fluorescein antibody (αR1; Genetex) or Alexa Fluor 488 antibody (αR2; Life Technologies) were adsorbed to 96-well Bacti plates (Thermo Scientific) at 0.4–0.8 μg/mL. Following washing with PBS with 0.05% (v/v) Tween 20, plates were blocked with PBS with 1% (w/v) bovine serum albumin (BSA; Sigma). Urine samples (diluted 1:102–104) and R1–2 standards were applied. Following washing, antibody–reporter–neutravidin sandwich complexes were formed by the addition of 0.4 μg/mL NeutrAvidin-horseradish peroxidase (Pierce). Sandwich complexes were detected by the oxidation of chromogenic substrate 3,3′,5,5′-tetramethylbenzidine (Pierce) at 450 nm and reporter concentration was calculated using the linear response region of reporter standards.
2.4. Single Molecule Array (SiMoA) Assay
Carboxylic acid functionalized paramagnetic beads (2.7 μm, Agilent Technologies) were covalently coupled with capture antibody (same as for ELISA) via 1-ethyl-3-(3-(dimethylamino)propyl)carbodiimide hydrochloride (EDC; Thermo Scientific) coupling protocol. Antibody-functionalized beads were blocked with PBS with 1% (w/v) BSA and roughly 200 000 beads were incubated with reporter standards or urine (diluted 102–104). Following addition of streptavidin-β-galactosidase to complete the sandwich complex, fiber optic arrays with 50 000 chemically etched wells were loaded with washed beads by centrifugation. Individual wells were sealed with fluorescent substrate resorufin-β-d-galactopyranoside (RDG; Life Technologies) and local fluorescence generated by substrate cleavage was imaged by custom equipment.9
2.5. Enzyme Kinetics
Michaelis–Menten enzyme kinetics were observed using an intramolecularly quenched reporter with the structure 5FAM-Gly-Gly-DPhe-Pro-Arg-Ser-Gly-Gly-Gly-Lys(CPQ2)-Lys-PEG2-Cys (CPC Scientific), which fluoresces upon proteolytic substrate cleavage. Intramolecularly quenched reporter was incubated with recombinant thrombin (2 nM, Haematologic Technologies) and substrate cleavage velocity was observed via fluorimeter. To quantify release kinetics of R1 from thrombin-sensitive R1-NWs, we added physiologic levels of recombinant thrombin (15 nM),13 purified released reporters by filtration (30 kDa Mr Amicon Ultra filters; Millipore), and quantified proteolytically released reporters by ELISA or SiMoA.
2.6. R1 Renal Clearance
MIT’s committee on animal care approved all animal studies (protocol 0411-036-14), which were performed with 6–10 week female Swiss Webster mice (Taconic). Excretion dose–response experiments were performed by intravenous injection of free R1 (not protease-sensitive, in 200 μL of PBS, n = 3 mice) followed by collection of all urine from 0 to 60 min postinjection. Log–log linear fit was performed using average urine concentration from n = 3 mice at five injected doses. Urine was weighed to calculate volume and frozen at −80 °C until analysis by ELISA (200–20 000 fmol) or SiMoA (0.2–20 fmol). Urinary recovery was calculated as total reporter excreted (the product of urine concentration with urine volume) normalized to injected dose.
2.7. Disease Detection with R1-NWs and R2
Thrombin-sensitive R1-NWs and renal clearance control R2 were intravenously (IV) co-injected in equimolar amounts at 0.2–200 pmol in 200 μL of PBS into female Swiss Webster mice (n = 5–10 per concentration) and all urine 0–30 min postinjection was collected to obtain control clearance values. A total of 3–5 days later (to allow synthetic biomarker clearance), mice were again co-injected with R1-NWs and R2 along with 20 U/g thromboplastin (Biopharm Laboratories) to induce intravascular coagulation as previously described5,14 and all urine 0–30 min postinjection was collected and pooled. R1 and R2 concentration was quantified by ELISA (20–200 pmol) or SiMoA (0.2–2 pmol).
3. Results and Discussion
3.1. Synthetic Biomarker Design
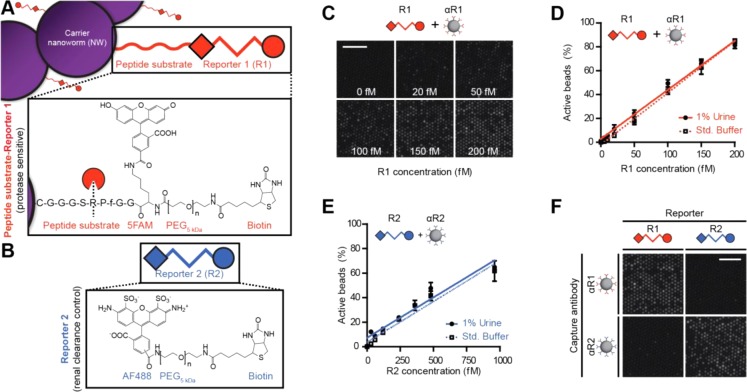
Our thrombin-sensitive synthetic biomarkers consist of iron oxide nanoworms (NWs) decorated with reporters bound to the NW surface by proteolytically degradable peptide substrates. Extensively characterized as biocompatible and long-circulating, the NWs are synthesized by the reaction of Fe(II) and Fe(III) salts in the presence of dextran11,12 and serve as carriers that render urinary reporter accumulation contingent upon thrombin-mediated degradation of the linking peptide substrate. To enable femtomolar-scale detection from the urine via sandwich complex assay, we designed our ligand-encoded reporter R1 with a capture ligand (fluorescein; for reporter immobilization), a detection ligand (biotin; for enzyme-mediated amplification), and a nondegradable poly(ethylene glycol) (PEG) 5 kDa spacer to enable favorable pharmacokinetics and simultaneous binding of both ligands (Figure 2A). The use of biotin enables direct detection of captured reporters without needing to form sandwich immunocomplexes, thus simplifying the assay. To account for differences in renal clearance, we designed a second orthogonal reporter R2, which passively filters into the urine and is not protease sensitive, with the structure Alexa Fluor 488-PEG5kDa-biotin (Figure 2B).
Figure 2.
(A) Protease-sensitive synthetic biomarkers release reporter 1 (R1; structure fluorescein-PEG5kDa-Biotin) from carrier NWs upon proteolytic cleavage of thrombin substrate. (B) Renal clearance control R2 is protease insensitive and uses Alexa Fluor 488 as capture ligand. (C) SiMoA assay with αR1-coated beads and increasing concentrations of R1 resulted in higher proportions of active fluorescent wells. (D) SiMoA assay for R1 demonstrated a linear relationship between active beads and R1 concentration from 5 to 200 fM and was unaffected by incubation with 1% control mouse urine. (E) The R2 SiMoA assay resulted in a similar linear relationship from 50 to 1000 fM. (F) Combination of R1 or R2 with αR1- or αR2-coated beads results in signal only from appropriately paired reporters and capture antibodies. Scale bar is 50 μm.
3.2. ELISA and SiMoA Assay
By conventional ELISA, the reporters are detected at low picomolar concentrations.5,6 For ultrasensitive reporter quantification, we modified our existing single molecule array technology, which relies on reporter capture by antibody-coated beads.8,9 In the digital sandwich assay approach used here, capture beads are incubated with analyte and, subsequently, with streptavidin β-galactosidase (SβG) as an enzyme label that enables direct detection of bead-bound reporters. At very low analyte concentrations where many more beads are present than analyte molecules, the distribution of sandwich complexes may be described by Poisson statistics and is linearly proportional to concentration (Supporting Information Figures S1A and B). Beads are then loaded by centrifugation into an array of ∼50 000 chemically etched wells, incubated with fluorogenic substrate resorufin-β-d-galactopyranoside (RGP), and sealed in individual 50 fL wells where a single enzyme label can generate detectable fluorescence.
SiMoA assay using αR1-coated beads resulted in an increase of fluorescent wells proportional to R1 concentration (Figure 2C). Quantitative imaging demonstrated a linear relationship between R1 concentration and active wells over a range of 5–200 fM that was unaffected by 1% control mouse urine; similar results were obtained with the R2 assay (Figures 2D,E; R2 linear range 50–1000 fM). To confirm R1–2 assay specificity, we applied saturating concentrations of each reporter to capture beads containing the other capture agent (e.g., R1 with αR2 and vice versa), resulting in no detectable off-target binding (Figures 2F, Supporting Information Figure S1C). These initial results indicated that the SiMoA assays were nearly 1000× more sensitive than the corresponding ELISAs and demonstrate the versatility of SiMoA for the sensitive detection of rationally designed synthetic compounds.
3.3. Enzymatic Reporter Release and Renal Clearance Kinetics
It is important that enzymatic kinetics are sufficiently fast at low synthetic biomarker concentrations to enable microdosing to be useful. Furthermore, linear dose–response of reporter excretion is required for unbiased disease detection. Nonlinearities in metabolism or excretion are a common microdosing mode of failure, often due to low-capacity, high-affinity drug binding reservoirs or dose-dependent metabolism. To demonstrate susceptibility of our synthetic biomarkers to proteolytic cleavage, we incubated a fluorescent reporting variant of our thrombin-sensitive NWs with thrombin, demonstrating thrombin-mediated peptidolysis with Km = 3.55 μM and Vmax = 2.26 nM s–1 (Figure 3A; [E]0 = 2 nM). Quasi-steady-state Michaelis–Menten enzyme kinetics approximate enzymatic velocity (v) as linear with substrate concentration at low concentrations (for [S] ≪ Km, v ≈ Vmax[S]/Km). Incubation of thrombin-sensitive NWs with physiological concentrations of thrombin ([E]0 = 15 nM; catalytically active thrombin during clotting is typically ∼10–30 nM)13 at 37 °C for 60 min demonstrated approximately linear scaling of released reporter concentrations with NW concentration over four logs (Figure 3B).
Figure 3.
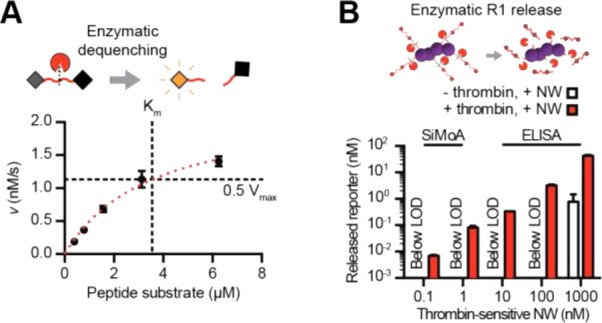
(A) In vitro incubation of a fluorescent reporter substrate with thrombin gave Km = 3.55 μM and Vmax = 2.26 nM s–1. (B) Incubation of thrombin-sensitive NWs with thrombin resulted in an approximately linear relationship between released reporter concentration and input concentration.
To determine renal clearance efficiencies of our reporters, we quantified R1 concentration in urine excreted 0–60 min following IV injection of R1 and observed a linear relationship between injection and urine concentration (Figure 4A, log–log linear fit of mean value R2 = 1.00, n = 3 mice for each of five concentrations). To examine total reporter excretion, we compared urinary reporter recovery (total reporter excreted over total reporter injected) and found that percent excretion did not decrease over five logs of input dose (Figure 4B).
Figure 4.
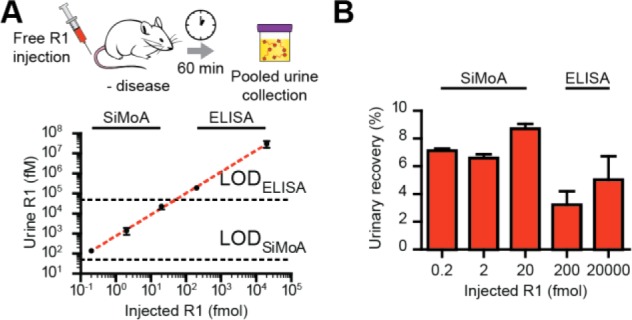
(A and B) IV injection of R1 and measurement of pooled urine concentration after 60 min demonstrated a linear dose–response relationship in urine concentration and no decrease in urine excretion percentage with decreasing dose.
3.4. Disease Detection Using Microdoses of Synthetic Biomarkers
Favorable synthetic biomarker pharmacokinetics at low concentrations support the use of microdosed synthetic biomarkers to detect disease using an induced model of pulmonary embolism in mice. As demonstrated by our group and others in the hematology literature, IV administration of thromboplastin results in the formation of blood clots via the extrinsic clotting cascade that embolize to the lungs to recapitulate this life-threatening condition.5,14,15
Previous studies by our group used 0.2–1.0 nmol of injected NWs to detect disease;4−6 here, we examined the ability of our reformulated synthetic biomarkers dosed at 200, 20, 2, and 0.2 pmol (1–1000× lower amounts than previously demonstrated) to diagnose thrombosis in vivo when paired with our new SiMoA assay. As anticipated, dosage of thrombin-sensitive R1-NWs and renal clearance control R2 at 200 pmol demonstrated greater R1 urinary concentration in the presence of disease (P = 0.0027), with R2 remaining relatively unaffected (P = 0.30, Figure 5A). These trends were successfully recapitulated at 0.2 pmol, 1000-fold lower and below microdose levels, demonstrating increased thrombin-sensitive R1 clearance (P = 0.017) in response to disease with urinary clearance control R2 remaining constant (P = 0.15, Figure 5B). On average, fold increase in urinary accumulation of R1 in response to clotting did not change with respect to concentration (slope of fit does not deviate significantly from zero with P = 0.22, Figure 5C). Collectively, these results indicate that our customized SiMoA assay could enable microdosed disease detection using synthetic biomarkers reformulated for low-dose applications.
Figure 5.
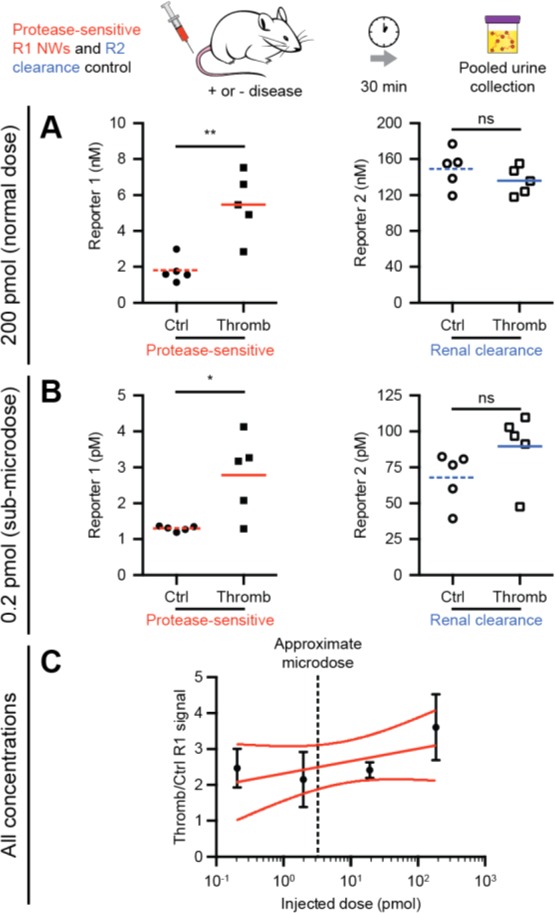
(A) Injection of 200 pmol thrombin-sensing R1-NWs and renal clearance control R2 demonstrated detection of disease by a significant increase in urine R1 concentration (P = 0.0027) but no significant change in control reporter R2 concentration (P = 0.30). (B) Injection of 1000-fold lower amounts of thrombin-sensing R1-NWs and control R2 demonstrated a significant increase in disease-sensitive R1 release (P = 0.017) but no significant change in control reporter R2 (P = 0.15). (C) Normalization of thrombin-sensitive R1-NW release in diseased mice to control mice revealed an average 2.35-fold increase in R1 signal over 3 orders of magnitude injected dose with a best fit line slope that did not deviate significantly from zero (P = 0.22).
4. Conclusion
In this paper, we described a custom single molecule assay that enables detection of synthetic ligand-encoded reporters. While previous SiMoA technologies have required sandwich immunocomplex formation for detection, we demonstrate femtomolar quantification of directly labeled targets that decreases processing time, reagent use, and obviates use of secondary recognition agents. When combined with a microdosed disease-tailored nanoparticle formulation, this modified SiMoA assay enables noninvasive urinary discrimination of thrombosis, a disease with poor existing biomarkers. Currently available diagnostics for thrombosis typically rely on sampling blood or urine for byproducts either upstream or downstream of the clotting cascade and efforts to directly detect thrombin activity rely on blood samples or do not provide a clinically useful urinary readout.10,16,17
Looking forward, our synthetic biomarker technology could be useful in conditions such as deep vein thrombosis (DVT) where the determination of clotting kinetics in stable vs extending thrombi would provide clinically actionable information. Others have shown the utility of thrombin-sensitive formulations in differentiating stable vs unstable atherosclerotic plaques, but these approaches have relied on fluorescent detection;17 our platform could conceivably provide urinary monitoring without a need for imaging. Finally, the ability to detect disease with up to 1000× lower doses of synthetic biomarkers could facilitate the development of implants that slowly elute microdoses of protease sensors that enable near-continuous urinary monitoring of thrombotic events.
Previous efforts to diagnose disease using microdosed compounds typically rely on radiolabeled drugs and complex detection techniques like accelerator mass spectrometry (AMS) or PET;18 here, we show radiolabel-free detection of rationally designed, proteolytically released fragments in urine using a single molecule-sensitive assay platform. In contrast to unstable radiolabels, the synthetic biomarker injectable is stable at 4 °C and the reporter-laden urine may be stored frozen long-term prior to analysis. Our platform enables detection sensitivities greater than traditional MALDI/MS or LC/ESI-MS/MS systems, while allowing rapid, multiplexed reporter detection and sensitivities roughly comparable to AMS without the need for time-consuming and difficult sample preparation.19 Consequently, the synthetic and analytical infrastructures are greatly simplified compared to existing low dose strategies for disease diagnosis.
To successfully microdose synthetic biomarkers, scaling between mouse and human should be considered. Although interspecies protease kinetics are similar (Supporting Information Table S1), dosing disparities are due to blood volume (scales linearly with weight20) and renal filtration (slower in humans21). Here, the lowest functional synthetic biomarker dose was 0.2 pmol (3.9 ng). To maintain synthetic biomarker plasma concentration, the corresponding human dose is predicted to be ∼8.7 μg, well under the 100 μg microdose threshold (see Supporting Information). Interspecies differences in renal function (inulin’s half-life is ∼16 times longer in humans than in mice) and the expected lower urine reporter concentrations may be resolved by the high sensitivity of the SiMoA.
The single molecule-sensitive SiMoA platform enables the use of multiplexable microdose-scale protease sensitive exogenous agents to detect disease, without the need for radiochemical synthesis, unstable reagents, or costly imaging platforms typical of microdose studies. These advances enable simplified, low cost (a single SiMoA assay is a few U.S. dollars), and safe diagnosis of thrombosis. Furthermore, they demonstrate the feasibility of more generalizable sandwich complex-based approaches to analysis that use the same diagnostics platform across broad concentration scales for different applications, from microdosing to standard clinical or point-of-care settings. By targeting different aberrantly regulated proteases with modified nanoparticles, the platform should be applicable to a broad variety of diseases including cancers, fibrosis, and inflammatory disorders.
Acknowledgments
We thank Prof. D. Wood and Dr. H. Fleming for guidance. A.D.W. thanks the NSF Graduate Research Fellowship Program for support. G.A.K. acknowledges support from the Ruth L. Kirchstein NRSA (F32CA159496-02) and holds a Career Award at the Scientific Interface from the Burroughs Wellcome Fund. This work was supported in part by the Koch Institute Support (core) Grant P30-CA14051 from the National Cancer Institute as well as a grant from the Koch Institute Frontier Research Program through the Koch Institute Frontier Research Fund. S.N.B. is an HHMI investigator.
Supporting Information Available
Text S1, giving detailed experimental protocols and calculations; Figure S1, expanding upon SiMoA assay validation; Figure S2, demonstrating first-order exponential clearance kinetics of urinary reporters; Figure S3, displaying all in vivo disease detection data; Table S1, comparing hemostatic coagulation indices between mouse and humans. This material is available free of charge via the Internet at http://pubs.acs.org.
Author Contributions
‡ A.D.W. and S.T.G. contributed equally.
The authors declare no competing financial interest.
Funding Statement
National Institutes of Health, United States
Supplementary Material
References
- Hanash S. M.; Pitteri S. J.; Faca V. M. Nature 2008, 4527187571–9. [DOI] [PubMed] [Google Scholar]
- Lutz A. M.; Willmann J. K.; Cochran F. V.; Ray P.; Gambhir S. S. PLoS Med. 2008, 58e170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagner C. C.; Langer O. Adv. Drug Delivery Rev. 2011, 637539–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwong G. A.; von Maltzahn G.; Murugappan G.; Abudayyeh O.; Mo S.; Papayannopoulos I. A.; Sverdlov D. Y.; Liu S. B.; Warren A. D.; Popov Y.; Schuppan D.; Bhatia S. N. Nat. Biotechnol. 2013, 31163–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin K. Y.; Kwong G. A.; Warren A. D.; Wood D. K.; Bhatia S. N. ACS Nano 2013, 7109001–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warren A. D.; Kwong G. A.; Wood D. K.; Lin K. Y.; Bhatia S. N. Proc. Natl. Acad. Sci. U.S.A. 2014, 111103671–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kummar S.; Doroshow J. H.; Tomaszewski J. E.; Calvert A. H.; Lobbezoo M.; Giaccone G.; Eur. J. Cancer 2009, 455741–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang L.; Song L.; Fournier D. R.; Kan C. W.; Patel P. P.; Ferrell E. P.; Pink B. A.; Minnehan K. A.; Hanlon D. W.; Duffy D. C.; Wilson D. H. J. Virol. Methods 2013, 1881–2153–60. [DOI] [PubMed] [Google Scholar]
- a Rissin D. M.; Kan C. W.; Campbell T. G.; Howes S. C.; Fournier D. R.; Song L.; Piech T.; Patel P. P.; Chang L.; Rivnak A. J.; Ferrell E. P.; Randall J. D.; Provuncher G. K.; Walt D. R.; Duffy D. C. Nat. Biotechnol. 2010, 286595–9. [DOI] [PMC free article] [PubMed] [Google Scholar]; b Rissin D. M.; Fournier D. R.; Piech T.; Kan C. W.; Campbell T. G.; Song L.; Chang L.; Rivnak A. J.; Patel P. P.; Provuncher G. K.; Ferrell E. P.; Howes S. C.; Pink B. A.; Minnehan K. A.; Wilson D. H.; Duffy D. C. Anal. Chem. 2011, 8362279–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- a Brill-Edwards P.; Lee A. Thromb. Haemostasis 1999, 822688–94. [PubMed] [Google Scholar]; b Ginsberg J. S.; Wells P. S.; Kearon C.; Anderson D.; Crowther M.; Weitz J. I.; Bormanis J.; Brill-Edwards P.; Turpie A. G.; MacKinnon B.; Gent M.; Hirsh J. Ann. Int. Med. 1998, 129121006–11. [DOI] [PubMed] [Google Scholar]; c Bounameaux H.; de Moerloose P.; Perrier A.; Reber G. Thromb. Haemostasis 1994, 7111–6. [PubMed] [Google Scholar]; d Kockum C. Thromb Res. 1980, 194–5639–46. [DOI] [PubMed] [Google Scholar]; e Becker R. C.; Spencer F. A. J. Thromb. Thrombolysis 1998, 53215–229. [DOI] [PubMed] [Google Scholar]
- Park J. H.; von Maltzahn G.; Zhang L.; Schwartz M. P.; Ruoslahti E.; Bhatia S. N.; Sailor M. J. Adv. Mater. 2008, 2091630–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park J. H.; von Maltzahn G.; Zhang L.; Derfus A. M.; Simberg D.; Harris T. J.; Ruoslahti E.; Bhatia S. N.; Sailor M. J. Small 2009, 56694–700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- a Brummel K. E.; Paradis S. G.; Butenas S.; Mann K. G. Blood 2002, 1001148–52. [DOI] [PubMed] [Google Scholar]; b Mann K. G.; Butenas S.; Brummel K. Arterioscler., Thromb., Vasc. Biol. 2003, 23117–25. [DOI] [PubMed] [Google Scholar]
- Weiss E. J.; Hamilton J. R.; Lease K. E.; Coughlin S. R. Blood 2002, 10093240–4. [DOI] [PubMed] [Google Scholar]
- Smyth S. S.; Reis E. D.; Väänänen H.; Zhang W.; Coller B. S. Blood 2001, 9841055–62. [DOI] [PubMed] [Google Scholar]
- a Pelzer H.; Schwarz A.; Heimburger N. Thromb. Haemostasis 1988, 591101–6. [PubMed] [Google Scholar]; b Pelzer H.; Schwarz A.; Stüber W. Thromb Haemost 1991, 652153–9. [PubMed] [Google Scholar]; c Lau H. K.; Rosenberg R. D. J. Biol. Chem. 1980, 255125885–93. [PubMed] [Google Scholar]
- Olson E. S.; Whitney M. A.; Friedman B.; Aguilera T. A.; Crisp J. L.; Baik F. M.; Jiang T.; Baird S. M.; Tsimikas S.; Tsien R. Y.; Nguyen Q. T. Integr Biol. 2012, 46595–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Josephson L.; Rudin M. J. Nucl. Med. 2013, 543329–32. [DOI] [PubMed] [Google Scholar]
- Vogel J. S.; Love A. H. Methods Enzymol. 2005, 402, 402–22. [DOI] [PubMed] [Google Scholar]; Yamane N.; Tozuka Z.; Sugiyama Y.; Tanimoto T.; Yamazaki A.; Kumagai Y. J. Chromatogr., B: Anal. Technol. Biomed. Life Sci. 2007, 8581–2118–28. [DOI] [PubMed] [Google Scholar]
- Sharma V.; McNeill J. H. Br. J. Pharmacol. 2009, 1576907–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prescott L. F.; McAuslane J. A.; Freestone S. Eur. J. Clin. Pharmacol. 1991, 406619–24. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.