Abstract
Background:
The aim of the present study was to analyse the prognostic value of microRNA-21 (miRNA-21) in patients with stage II colon cancer aiming at a risk index for this group of patients.
Methods:
A population-based cohort of 554 patients was included. MicroRNA-21 was analysed by qPCR based on tumour tissue. An index was created using the coefficients obtained from a collective multiple Cox regression. The entire procedure was cross-validated (10-fold). The performance of the index was quantified by time-dependent receiver operating characteristics curves.
Results:
High miRNA-21 expression was associated with an unfavourable recurrence-free cancer-specific survival (RF-CSS), hazard ratio 1.35 (95% confidence interval, 1.03–1.76) (P=0.028). The generated RF-CSS index divided the traditional high-risk patients into subgroups with 5-year RF-CSS rates of 87% and 73%, respectively (P<0.001). The overall survival (OS) index identified three different subgroups (P<0.001). Cross-validated 5-year OS rates were 88%, 68%, and 50%, respectively.
Conclusions:
This population-based study supports miRNA-21 as an additional prognostic biomarker in patients with stage II colon cancer. Furthermore, the introduction of a risk index may guide the use of postoperative adjuvant treatment in a more appropriate way compared with current practice.
Keywords: colon cancer, microRNA-21, prognostic markers
The 5-year overall survival (OS) rates of patients with stage II colon cancer (surgical treatment only) is 70–80%, with the majority of the patients dying from causes other than cancer (Zaniboni and Labianca, 2004; Niedzwiecki et al, 2011). However, with an estimated worldwide incidence of 1.2 million new cases of colorectal cancer (CRC) each year (Ferlay et al, 2010), the fraction of uncured patients with stage II colon cancer becomes a considerable problem.
Efficiency of adjuvant fluorouracil-based therapy was documented in the 1990s for patients operated for stage III colon cancer (Moertel et al, 1995; O'Connell et al, 1997). A disease-free survival benefit from adding oxaliplatin was later shown for stage III disease, whereas the efficiency in stage II colon cancer remained less clear (Andre et al, 2004, 2009; Kuebler et al, 2007). Selecting patients with stage II colon cancer for adjuvant chemotherapy is therefore based on an individual evaluation supported by a panel of pathologic characteristics related to the risk of disease recurrence such as pT4 tumours, bowel obstruction, localised perforation, lymphatic/vascular invasion, perineural invasion, poorly differentiated histology, and inadequate lymph node assessment (Schmoll et al, 2012). However, the predictive value of these criteria, in selecting high-risk patients likely to benefit from adjuvant chemotherapy, has recently been questioned (O'Connor et al, 2011).
MicroRNAs (miRNAs) are short non-coding RNAs with the ability to modulate protein expression by degrading/repressing their target mRNAs (Filipowicz et al, 2008). Our understanding of miRNAs and their role in cancer is rapidly growing (Esquela-Kerscher and Slack, 2006). MicroRNA-21, with chromosomal location 17q23.2, is upregulated in virtually all malignant neoplasms and with oncogenic functions related to all the hallmarks of a malignant cell (Zhu et al, 2008; Slaby et al, 2009).
Accordingly, upregulation of miRNA-21 has been demonstrated in CRC tissue compared with normal colorectal tissue (Slaby et al, 2007, 2009; Kulda et al, 2010), and a role in the CRC carcinogenesis has been documented as well (Slaby et al, 2007, 2009) with increased miRNA-21 expression detected already at the precancerous adenoma stage (Yamamichi et al, 2009). Several reports have later on documented a relationship between miRNA-21 expression and survival in patients with colon cancer (Schetter et al, 2008; Kulda et al, 2010; Shibuya et al, 2010; Nielsen et al, 2011; Kjaer-Frifeldt et al, 2012).
The aim of the present study was to elucidate the prognostic value of miRNA-21 analysed by qPCR and combine the results with traditional prognostic markers for the calculation of an individual risk index in patients with stage II colon cancer from a population-based cohort.
Materials and methods
This study is reported in accordance with the REMARK (McShane et al, 2005) and BRISQUE (Moore et al, 2011) criteria.
Patient population
The patient population, follow-up, and sources of data have previously been described in detail Kjaer-Frifeldt et al (2012). In brief, a population-based cohort of patients surgically resected for stage II colon cancer was identified in the Danish Colorectal Cancer Group database, in which surgical and pathologic data are collected prospectively. The entire cohort from 2003 constituted 764 patients. Participation in the study was high (∼93%) providing a representative cohort of the population (n=708). Exclusion criteria were as follows: patients who died within 30 days after the operation (n=66), missing tissue samples (n=23), incorrectly staged patients (n=14), prior adjuvant chemotherapy (n=16), and insufficient tissue samples for analyses (n=7), leaving a cohort of 582 patients to be considered for analyses. A further 24 patients were excluded in relation to the qPCR analysis owing to insufficient amount of RNA, and finally, 4 patients were excluded owing to missing values of one or more tumour characteristics, leaving a final study cohort of 554 patients. Verified disease recurrence and/or death from colon cancer was recorded for 116 patients. Death by any cause was recorded for 256 patients. The study was approved by the Regional Scientific Ethical Committee for Southern Denmark (S-20090049) and the Danish Data Protection Agency, according to Danish law.
Samples
Histologic samples from the resected colon tumours followed routine formaldehyde fixation and paraffin embedding (FFPE) and were stored at room temperature. One FFPE tissue block containing the deepest invasive front was used from each patient to assess the pathologic characteristics (Kjaer-Frifeldt et al, 2012). A 6-μm-thick tissue section was used for miRNA analyses.
qPCR analyses of miRNA-21
MicroRNA quantification of 554 samples was carried out by Exiqon A/S (Vedbaek, Denmark).
Initially, 10 ng RNA was reverse transcribed (RT) in 10 μl reactions, using the miRCURY LNA Universal RT miRNA PCR, Polyadenylation and cDNA Synthesis Kit (Exiqon). cDNA was diluted 100 × and assayed in 10 μl PCR reactions according to the miRCURY LNA protocol. MicroRNA-21, miRNA-16, miRNA-103, and negative controls were assayed by qPCR on the microRNA Ready-to-Use PCR, Pick-n-Mix panel (Exiqon, Vedbaek, Denmark). Amplification was performed in a LightCycler 480 Real-Time PCR System (Roche Diagnostics, Basel, Switzerland) in 384 well plates. Amplification curves were analyzed using the Roche LC software (Roche Diagnostics), both for determination of crossing point (Cp) and for melting-curve analysis.
A prespecified quality control removed reactions with several melting points, reactions with melting points not within assay specification, with amplification efficacy below 1.6, or with values within 5 Cp of the negative control. Furthermore, an RNA spike-in control (Sp6) was added in the RT reaction to evaluate both the RT reaction and the following qPCR reaction. All samples passed the quality control, and additionally, the results showed similar Cp values across all the samples indicating successful RT–qPCR analyses. The analyses were performed over three working periods and the results demonstrated comparable sample quality (were similar in miRNA content), indicating that they had been processed reproducibly. The initial batch was analysed in duplicate. The variation in the system was very low (coefficient of variation (%), miRNA-16 (2.0%), miRNA-21 (1.9%), and miRNA-103 (1.2%)) and the following batches were consequently only analysed once.
The average of miRNA-16 and miRNA-103 was used as a normalisation factor (mean 27.7, s.d. 1.21). An alternative normalisation procedure using ‘global' miRNA content was also considered. This ‘global' miRNA pool consisted of an additional eight miRNAs with a presumed relationship to CRC carcinogenesis. This strategy resulted in a normalisation factor with a higher degree of diversity (mean 28.3, s.d. 1.29) and was consequently abandoned. The presented values for normalised miRNA-21 expression constitute a ratio and are thus without a dimension.
Statistics
Median values were compared using the Wilcoxon's rank-sum test. Overall survival was defined as time from operation until death from any course (n=256). Recurrence-free cancer-specific survival (RF-CSS) was defined as time from operation until documented tumour recurrence or death from colon cancer. Censored in this analysis were patients dying from other causes than colon cancer and patients with later diagnoses of other malignancies.
Predictors were prechosen according to clinical consensus and guidelines: T-category, age, gender, localization, +/− perforation, differentiation, +/− nerve involvement, +/−venous involvement, > or < 12 lymph nodes retrieved and microsatellite instability (MSI). We added miR21 quantified by qPCR, as a new possible predictor. Hence, we investigated the association of these 11 predictors with outcome, CS-RFS and OS. We adopted the methodology from the area of high-dimensional genomic data developed by Simon et al (2011). We selected features by simple Cox regression, that is, predictors with a Wald test P-value <0.1. As the total number of predictors is fairly small, we judged that the risk of overfitting was minimal, and we used a less strict P-level for selection. This may lead to some noise in the model, but we would rather accept this than the risk of overlooking an important predictor. For each outcome, a multiple Cox regression including the selected predictors was performed, and the resulting coefficients were combined with the predictors to form a prognostic index for each outcome. The patient population was a priori stratified into risk sets by the quartiles of the respective indices. The resulting stratification was evaluated using Kaplan–Meier curves, which were compared using the log-rank test. Risk sets with similar prognoses were combined. The performance of each raw index was evaluated by subjecting the entire procedure, including the feature selection, to a 10-fold cross-validation from which we obtained cross-validated Kaplan–Meier curves (Simon et al, 2011). We compared the cross-validated Kaplan–Meier curves, using the cross-validated log-rank test statistics (Simon et al, 2011), thus repeating the cross-validation process for 1000 permutation samples of the survival–time/censoring relationship. Additionally, we produced time-dependent receiver operating characteristics (ROC) curves (Heagerty et al, 2000) (http://cran.rproject.org/web/packages/survivalROC/index.html, February 2013) for the raw and the cross-validated indices. The ability of the indices to predict outcome was assessed by comparing the area under the curve (AUC) for the respective ROC curves. No formal testing was carried out for the ROC curves. For the cross-validated ROC curves, we used the rank of the index to predict survival rather than the index itself. This strategy was chosen, as within the same cross-validated risk set the index values vary depending on the set of predictors from which they originate. For example, the scale of an index changes markedly whether age is in the model or not. The ROC curve is a two-dimensional graph in which the true-positive rate is plotted on one axis and the false-positive rate on the other. Thus, a higher AUC indicates a better model performance with AUC=0.5 indicating random performance.
All statistical calculations were carried out using STATA 11.2 (Stata Corp., College State TX, USA) and R version 2.15.1 (package survivalROC). P-values <0.05 were considered significant, and all tests were two-sided.
Results
Patient characteristics
Patient and pathologic characteristics according to miRNA-21 expression are shown in Table 1. A slightly but significantly lower miRNA-21 expression was detected in patients with right-sided tumours (P=0.048), patients with MSI tumours (P=0.002), and tumours with a high malignancy grade (P=0.04).
Table 1. Patient and pathologic characteristics according to miRNA-21 expression.
| Parameter | Number (n=554) (%) | miRNA-21 median (95% CI) | P-value |
|---|---|---|---|
|
Gender | |||
| Male | 237 (43) | 5.23 (5.16–5.35) | 0.24 |
| Female |
317 (57) |
5.19 (5.10–5.27) |
|
|
Age, median 74 (years) | |||
| ⩾74 | 279 (50) | 5.18 (5.10–5.24) | 0.68 |
| <74 |
275 (50) |
5.23 (5.15–5.32) |
|
|
T-category | |||
| T4 | 72 (13) | 5.27 (5.10–5.44) | 0.39 |
| T3 |
482 (87) |
5.20 (5.13–5.26) |
|
|
Malignancy grade | |||
| Higha | 107 (19) | 5.03 (4.92–5.19) | 0.04 |
| Medium+low |
447 (81) |
5.23 (5.18–5.32) |
|
|
Localisation | |||
| Right | 285 (51) | 5.18 (5.07–5.23) | <0.05 |
| Left |
269 (49) |
5.26 (5.16–5.38) |
|
|
Perforation | |||
| Yes | 48 (9) | 5.24 (4.98–5.44) | 0.48 |
| No |
506 (91) |
5.20 (5.15–5.27) |
|
|
Lymph nodes | |||
| ⩾12 | 249 (45) | 5.24 (5.14–5.38) | 0.13 |
| <12 |
305 (55) |
5.18 (5.10–5.24) |
|
|
Neuronal invasion | |||
| Yes | 47 (8) | 5.32 (5.03–5.58) | 0.47 |
| No |
507 (92) |
5.20 (5.15–5.27) |
|
|
Vascular invasion | |||
| Yes | 67 (12) | 5.18 (5.01–5.33) | 0.54 |
| No |
487 (88) |
5.21 (5.15–5.28) |
|
|
MSI status | |||
| MSI | 159 (29) | 5.05 (4.97–5.17) | <0.01 |
| MSS | 395 (71) | 5.27 (5.20–5.35) | |
Abbreviations: CI=confidence interval; miRNA=microRNA; MSI=microsatellite instability; MSS=microsatellite stable. Bold numerals are used to emphasize significant P-values.
Including mucinous and signet cell tumours.
Risk index by RF-CSS
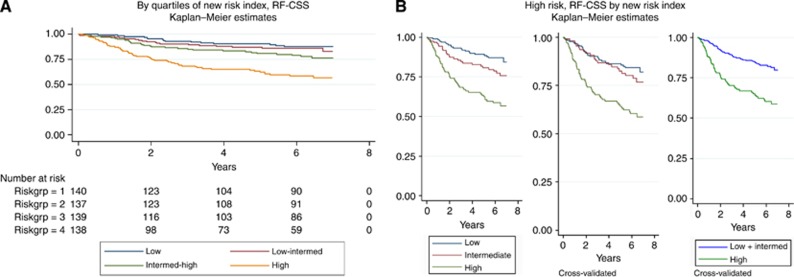
Predictors for the RF-CSS risk index were chosen by Cox regression analyses using P<0.1 as the cutoff value (Table 2). This resulted in six parameters (miRNA-21, T-category, perforation, malignancy grade, neuronal invasion, and vascular invasion) to be included. MicroRNA-21 expression was included as a continuous variable, whereas the remaining parameters were categorical. The model was fitted and the coefficients and values of the included parameters were used to define a risk index (Table 2). The quartiles of the index were used to differentiate the RF-CSS as shown in Figure 1A. The curves for the two lowest quartiles demonstrated a minimal diversity and the corresponding groups were combined. The cross-validated curves showed that the separation into three groups was biased, and the final stratification was therefore fitted into two groups, separating the highest quartile from the rest as shown in Figure 1B. This index partitioned the patients with 23% at high risk and a 5-year RF-CSS of 64%, and 77% at low risk with a 5-year RF-CSS of 86%. The separation was obtained at an index value of 2.22 (Table 2). In comparison, if grouped by traditional risk parameters (as specified in Materials and Methods), 77% of the patients would have been categorised as high risk with a 5-year RF-CSS of 78% and only 23% would have been categorised as low risk with a 5-year RF-CSS of 87%. The separation was statistically significant with P<0.001 in a cross-validated log-rank test.
Table 2. RF-CSS results from the Cox regression analysis and the parameters included in the risk index model (n=554).
|
Cox regression, simple analysis | |||
|---|---|---|---|
| Parameter | HR | 95% CI | P-value |
| miR-21 qPCR |
1.348 |
1.032–1.760 |
0.028 |
| Age |
1.014 |
0.997–1.032 |
0.112 |
| Gender |
1.142 |
0.791–1.648 |
0.478 |
| T4 |
3.733 |
2.491–5.594 |
<0.001 |
| Localisation |
1.069 |
0.742–1.538 |
0.721 |
| Perforation |
3.214 |
2.002–5.160 |
<0.001 |
| Malignancy grade |
1.459 |
0.953–2.232 |
0.082 |
| Neuronal invasion |
2.100 |
1.270–3.471 |
0.004 |
| Vascular invasion |
2.041 |
1.283–3.247 |
0.003 |
| Lymph nodes |
1.212 |
0.846–1.753 |
0.288 |
| MSI status | 1.420 | 0.912–2.211 | 0.121 |
|
Risk index model | ||||
|---|---|---|---|---|
| Parameter | Coefficient | Rounding off | Risk set | Indexa (min–max) |
| miR-21 qPCR |
0.324 |
0.32 |
|
|
| T4 |
1.081 |
1.08 |
Low |
0.45–2.22 |
| Perforation |
0.917 |
0.92 |
|
|
| Malignancy grade |
0.417 |
0.42 |
High |
2.23–4.51 |
| Neuronal invasion |
0.416 |
0.42 |
|
|
| Vascular invasion | 0.474 | 0.47 | ||
Abbreviations: CI=confidence interval; HR=hazard ratio; MSI=microsatellite instability; qPCR=quantitative polymerase chain reaction; RF-CSS=recurrence-free cancer-specific survival. Bold numerals are used to emphasize significant P-values.
The index cutoff is based on the preplanned separation of the index according to quartiles (2.22/2.23 thus mark the separation between high risk and intermediate low as shown in Figure 1B).
Figure 1.
Recurrence-free cancer-specific survival (RF-CSS) by (A) quartiles of new risk index (N=554) and by (B) a priori and final stratification of high-risk patients (N=169 (low), N=122 (intermediate), and N=138 (high)) by the new risk index (upper quartile) vs low risk (lower and two middle quartiles combined) (final cross-validation P<0.001).
Risk index by OS
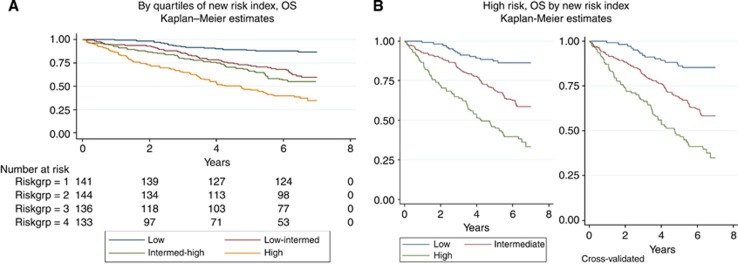
Predictors for the OS risk index were chosen similarly resulting in five included parameters (age, gender, T-category, perforation, and vascular invasion) (Table 3). The quartiles of the rounded index were used to differentiate OS (Figure 2A). The curves for the two middle quartiles were combined for reasons addressed above as shown in Figure 2B. The coefficients and cutoff values are shown in Table 3. This index identified 23% of the patients at high risk, 49% at intermediate risk, and 28% at low risk with a 5-year cross-validated OS of 51% (95% CI, 42%–59%), 68% (62%–73%), and 86% (80%–91%), respectively. In comparison, if grouped by traditional risk parameters, 77% of the patients would have been rated as high risk and 23% as low risk with a 5-year OS of 67% and 74%, respectively. The high-risk group was characterized by index values above 4.4, P<0.001 (overall cross-validated log-rank test).
Table 3. OS: results from the Cox regression analyses and the parameters included in the risk index model (n=554).
|
Cox regression, simple analysis | |||
|---|---|---|---|
| Parameter | HR | 95% CI | P-value |
| miR-21 qPCR |
1.075 |
0.889–1.301 |
0.455 |
| Age |
1.052 |
1.037–1.067 |
<0.001 |
| Gender |
1.380 |
1.058–1.802 |
0.018 |
| T4 |
1.979 |
1.404–2.790 |
<0.001 |
| Localisation |
0.933 |
0.715–1.218 |
0.611 |
| Perforation |
1.610 |
1.045–2.481 |
0.031 |
| Malignancy grade |
1.223 |
0.881–1.698 |
0.229 |
| Neuronal invasion |
1.201 |
0.765–1.883 |
0.426 |
| Vascular invasion |
1.640 |
1.137–2.366 |
0.008 |
| Lymph nodes |
0.961 |
0.735–1.256 |
0.772 |
| MSI status | 0.931 | 0.695–1.249 | 0.635 |
|
Risk index model | ||||
|---|---|---|---|---|
| Parameter | Coefficient | Rounding off | Risk set | Indexa (min–max) |
| Age |
0.054 |
0.05 |
Low |
1.65–3.50 |
| Gender |
0.407 |
0.41 |
|
|
| T4 |
0.711 |
0.71 |
Intermediate |
3.51–4.40 |
| Perforation |
0.495 |
0.50 |
|
|
| Vascular invasion | 0.425 | 0.43 | High | 4.41–6.22 |
Abbreviations: CI=confidence interval; HR=hazard ratio; MSI=microsatellite instability; OS=overall survival; qPCR=quantitative polymerase chain reaction. Bold numerals are used to emphasize significant P-values.
The index cutoff is based on the preplanned separation of the index according to quartiles (3.50/3.51 and 4.40/4.41 thus mark the separation between low- risk and intermediate, and intermediate and high risk as shown in Figure 2B).
Figure 2.
Overall survival (OS) by (A) quartiles of new risk index (N=554) and by (B) high-risk patients by new risk index (N=102 (low), N=216 (intermediate), and N=111 (high)) with the two middle quartiles combined (overall cross-validated log-rank P<0.001).
Index performance, ROC curves
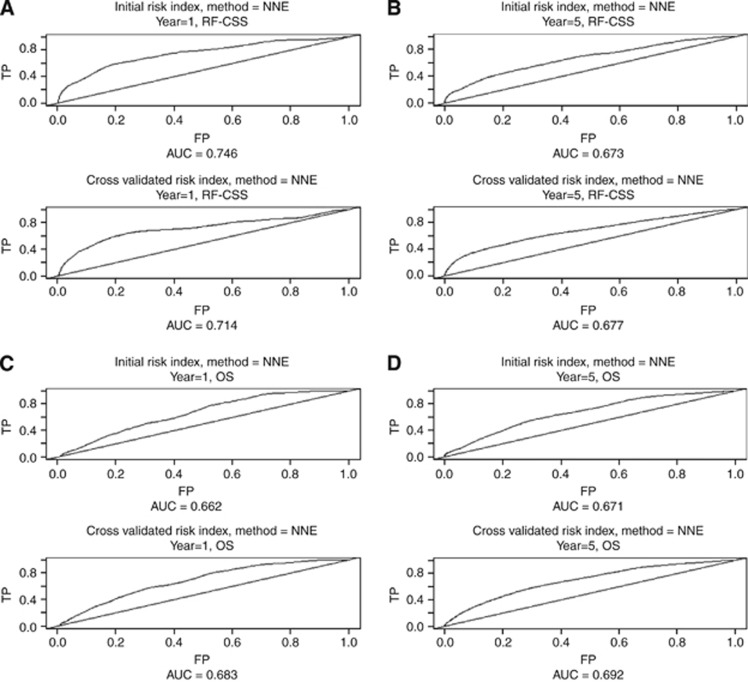
Time-dependent ROC curves were calculated for the ‘raw' as well as the cross-validated index (Figure 3). The AUC (and the cross-validated AUC) for 1- and 5-year RF-CSS were 0.75 (0.67) and 0.71 (0.68), respectively. The AUC (and the cross-validated AUC) for 1- and 5-year OS were 0.66 (0.68) and 0.67 (0.69), respectively.
Figure 3.
Time-dependent ROC curves for the initial as well as the cross-validated index according to recurrence-free cancer-specific survival (RF-CSS) and overall survival (OS): (A) year 1 (RF-CSS), (B) year 5 (RF-CSS), (C) year 1 (OS) and (D) year 5 (OS), respectively.
Discussion
The present study investigated the proposed candidature of miRNA-21 as a prognostic biomarker in an untreated population-based cohort of patients with stage II colon cancer and argues for an alternative approach for handling the impact of existing prognostic biomarkers.
MicroRNA-21 was analysed by qPCR based on FFPE tumour tissue sections. Previous reports have demonstrated stable miRNA expression by qPCR profiling in FFPE tissue samples despite large differences in fixation and storing time (Xi et al, 2007; Bovell et al, 2012). Analyses on the FFPE tumour tissue in the present study were not possible on approximately 4% of the samples. However, analysing FFPE samples holds the advantage of a large-scale biomarker validation using a generally accepted method, although a validated method for normalisation of data in this setting is still warranted. Chang et al (2010) recommended several suitable reference genes for normalisation of RT–qPCR data in CRC and miRNA-16 was the single most suitable reference gene. Normalisation in the present study was initially tested by two different approaches and the average of miRNA-16 and miRNA-103 was chosen, as this normalisation parameter demonstrated the lowest variation. RNUs were not considered for normalisation owing to concerns about their stability in FFPE tissue samples. Their longer nucleotide sequences might be challenged differently than miRNAs depending on the quality of the FFPE samples. Furthermore, reports have argued for a relationship between RNUs and clinicopathologic characteristics, including prognosis, potentially leading to the introduction of bias if used as reference genes in this setting (Gee et al, 2011).
The miRNA-21 expression was a significant biomarker according to the RF-CSS analysis in a highly representative, untreated, population-based cohort of 554 patients with stage II colon cancer. No prognostic impact was demonstrated regarding OS. These results are in accordance with previous studies. In 2008 Schetter et al (2008) demonstrated an independent prognostic value, in two independent cohorts, of miRNA-21 (119 patients with stage II and III colon cancer) analysed by qPCR using RNU6B for normalisation. In 2010, Kulda et al analysed miRNA-21 by qPCR in 44 patients with stage I–IV CRC using total RNA for normalisation, whereas Nielsen et al (2011) used ISH analysis and image-guided quantification of miRNA-21 in samples from 129 patients with stage II colon cancer. In 2010, Shibuya et al reported on 156 patients with stage I–IV CRC, in which miRNA-21 was analysed by qPCR using RNU6B for normalisation. Finally, in 2012 Kjaer-Frifeldt et al published a study originating from the same population-based cohort as the present study, in which miRNA-21 was analysed by ISH and image-guided quantification in samples from 520 patients. The relationship between high miRNA-21 expression and poor prognosis is the common denominator of all these studies despite their methodologic differences. In fact, quite unusual for a putative prognostic biomarker, no studies have demonstrated the opposite relationship between miRNA-21 and prognosis.
The current panel of prognostic biomarkers used for the selection of patients with high-risk stage II colon cancer for adjuvant chemotherapy has been questioned (O'Connor et al, 2011). This inspired us to test the clinical relevance of a risk index in this setting, whereas simultaneously integrating the prognostic value of the molecular-based biomarker miRNA-21 into the already existing panel of biomarkers. Individually, these biomarkers may not be powerful enough to identify clinically relevant risk groups in this group of patients. Consequently, too many patients with stage II colon cancer are categorised as high risk and treated with chemotherapy leaving them the adverse events but not the benefit of adjuvant therapy. Assessing the combined impact of the included biomarkers might provide a clinically more operative stratification.
Have we succeeded? The genomic research inspired method we used to develop the indices ensures that they are robust and unbiased with respect to stratification of the patient population. The RF-CSS risk index, which was based only on tumour-related parameters, identified a group of patients with a significantly higher risk of disease recurrence than the remaining cohort. The OS risk index, on the other hand, generated a low-, intermediate-, and high-risk group and included non-tumour-related parameters as well, which may not be surprising, as the majority of the patients die of reasons other than cancer.
First of all, it is evident that a group of the patients is at low risk of disease recurrence or death of cancer. These patients are not likely to benefit from any postoperative intervention. Second, the OS risk index identified an intermediate group with a risk profile that may be comparable to that of otherwise healthy individuals at a similar age. An ‘observation-only' approach seems reasonable for this group, too. Finally, the RF-CSS high-risk patients demonstrate a worse prognosis than would normally be expected, which is translated into a distinct OS high-risk group as well. Consequently, introducing a risk index for stratifying patients with stage II colon cancer may lead to the identification of a considerably smaller group than the traditional approach would have been categorised as high risk, sparing a large fraction of the patients of adjuvant chemotherapy with doubtful efficacy.
The cross-validated AUC for the RF-CSS index at 1- and 5-year survival was 0.714 and 0.667, respectively (Figure 3A and B), suggesting a slight decrease in the performance of the index over time, which may be explained by the rather low risk of relapse after 5 years. Thus, our proposed risk index classifies patients correctly (high or low risk) with a probability of approximately 70%. Even though this is comparable with recent results in breast cancer applying the same statistics (Volinia and Croce, 2013), one must still acknowledge that there are issues of prognostic importance not accounted for by the index, which calls for a continued search for prognostic biomarkers in this setting.
It is worth noting that although the analyses of miRNA-21 by the ISH technique, as previously applied to this cohort (Kjaer-Frifeldt et al, 2012), led to the same conclusions as the ones presented in this paper, this was not achieved through the identification of the same patients at risk of recurrence. A linear regression analyses comparing the two continuous variables resulted in a correlation coefficient (r) of 0.09 (P=0.053). This may not be surprising given the two very different techniques, but it stresses the need for further analyses of miRNA-21 before a uniform recommendation can be provided for its clinical application in the current setting.
It is worth mentioning that although miRNA-21 may add to the identification of patients with high-risk stage II colon cancer, it may simultaneously pose another question regarding therapy. Studies have suggested that high miRNA-21 expression causes downregulation of human mutS homolog 2 resulting in a deficient mismatch repair system and 5-FU resistance in CRC (Schetter et al, 2008; Valeri et al, 2010). The possible predictive value of miRNA-21 in patients with colon cancer calls for clarification in a clinical context.
Referring patients with stage II colon cancer for adjuvant chemotherapy is a complicated issue. The benefit from adjuvant chemotherapy is small at best and detrimental at worst. The present study provides new evidence that allows a better choice on a rational basis. The results support miRNA-21 as a prognostic biomarker in patients with stage II colon cancer. Furthermore, the introduction of a risk index holds the potential of sparing a considerable fraction of the patients from unnecessary treatments, thus impacting current clinical practice.
Acknowledgments
We thank Birgit Roed Sørensen and Stine Jørgensen for the technical assistance and Karin Larsen for the linguistic editing. This study was supported by The Cancer Foundation, The Danish Council for Independent Research and the Danish Colorectal Cancer Group. The supporters had no influence on any part of the study.
Thorarinn Blondal and Søren Morgenthaler Echwald held full-time employments at the time of analysis for an entity (name of entity: Exiqon A/S) having a commercial interest in the subject matter under consideration in the manuscript.
Footnotes
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
References
- Andre T, Boni C, Mounedji-Boudiaf L, Navarro M, Tabernero J, Hickish T, Topham C, Zaninelli M, Clingan P, Bridgewater J, Tabah-Fisch I, De GA. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med. 2004;350:2343–2351. doi: 10.1056/NEJMoa032709. [DOI] [PubMed] [Google Scholar]
- Andre T, Boni C, Navarro M, Tabernero J, Hickish T, Topham C, Bonetti A, Clingan P, Bridgewater J, Rivera F, De GA. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J Clin Oncol. 2009;27:3109–3116. doi: 10.1200/JCO.2008.20.6771. [DOI] [PubMed] [Google Scholar]
- Bovell L, Shanmugam C, Katkoori VR, Zhang B, Vogtmann E, Grizzle WE, Manne U. miRNAs are stable in colorectal cancer archival tissue blocks. Front Biosci (Elite Ed) 2012;4:1937–1940. doi: 10.2741/514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang KH, Mestdagh P, Vandesompele J, Kerin MJ, Miller N. MicroRNA expression profiling to identify and validate reference genes for relative quantification in colorectal cancer. BMC Cancer. 2010;10:173. doi: 10.1186/1471-2407-10-173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esquela-Kerscher A, Slack FJ. Oncomirs - microRNAs with a role in cancer. Nat Rev Cancer. 2006;6:259–269. doi: 10.1038/nrc1840. [DOI] [PubMed] [Google Scholar]
- Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893–2917. doi: 10.1002/ijc.25516. [DOI] [PubMed] [Google Scholar]
- Filipowicz W, Bhattacharyya SN, Sonenberg N. Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight. Nat Rev Genet. 2008;9:102–114. doi: 10.1038/nrg2290. [DOI] [PubMed] [Google Scholar]
- Gee HE, Buffa FM, Camps C, Ramachandran A, Leek R, Taylor M, Patil M, Sheldon H, Betts G, Homer J, West C, Ragoussis J, Harris AL. The small-nucleolar RNAs commonly used for microRNA normalisation correlate with tumour pathology and prognosis. Br J Cancer. 2011;104:1168–1177. doi: 10.1038/sj.bjc.6606076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heagerty PJ, Lumley T, Pepe MS. Time-dependent ROC curves for censored survival data and a diagnostic marker. Biometrics. 2000;56:337–344. doi: 10.1111/j.0006-341x.2000.00337.x. [DOI] [PubMed] [Google Scholar]
- Kjaer-Frifeldt S, Hansen TF, Nielsen BS, Joergensen S, Lindebjerg J, Soerensen FB, Depont CR, Jakobsen A. The prognostic importance of miR-21 in stage II colon cancer: a population-based study. Br J Cancer. 2012;107:1169–1174. doi: 10.1038/bjc.2012.365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuebler JP, Wieand HS, O'Connell MJ, Smith RE, Colangelo LH, Yothers G, Petrelli NJ, Findlay MP, Seay TE, Atkins JN, Zapas JL, Goodwin JW, Fehrenbacher L, Ramanathan RK, Conley BA, Flynn PJ, Soori G, Colman LK, Levine EA, Lanier KS, Wolmark N. Oxaliplatin combined with weekly bolus fluorouracil and leucovorin as surgical adjuvant chemotherapy for stage II and III colon cancer: results from NSABP C-07. J Clin Oncol. 2007;25:2198–2204. doi: 10.1200/JCO.2006.08.2974. [DOI] [PubMed] [Google Scholar]
- Kulda V, Pesta M, Topolcan O, Liska V, Treska V, Sutnar A, Rupert K, Ludvikova M, Babuska V, Holubec L, Jr., Cerny R. Relevance of miR-21 and miR-143 expression in tissue samples of colorectal carcinoma and its liver metastases. Cancer Genet Cytogenet. 2010;200:154–160. doi: 10.1016/j.cancergencyto.2010.04.015. [DOI] [PubMed] [Google Scholar]
- McShane LM, Altman DG, Sauerbrei W, Taube SE, Gion M, Clark GM. REporting recommendations for tumour MARKer prognostic studies (REMARK) Br J Cancer. 2005;93:387–391. doi: 10.1038/sj.bjc.6602678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moertel CG, Fleming TR, Macdonald JS, Haller DG, Laurie JA, Tangen CM, Ungerleider JS, Emerson WA, Tormey DC, Glick JH, Veeder MH, Mailliard JA. Fluorouracil plus levamisole as effective adjuvant therapy after resection of stage III colon carcinoma: a final report. Ann Intern Med. 1995;122:321–326. doi: 10.7326/0003-4819-122-5-199503010-00001. [DOI] [PubMed] [Google Scholar]
- Moore HM, Kelly AB, Jewell SD, McShane LM, Clark DP, Greenspan R, Hayes DF, Hainaut P, Kim P, Mansfield EA, Potapova O, Riegman P, Rubinstein Y, Seijo E, Somiari S, Watson P, Weier HU, Zhu C, Vaught J. Biospecimen reporting for improved study quality (BRISQ) Cancer Cytopathol. 2011;119:92–101. doi: 10.1002/cncy.20147. [DOI] [PubMed] [Google Scholar]
- Niedzwiecki D, Bertagnolli MM, Warren RS, Compton CC, Kemeny NE, Benson ABI, Eckhardt SG, Alberts S, Porjosh GN, Kerr DJ, Fields A, Rougier P, Pipas JM, Schwartz JH, Atkins J, O'Rourke M, Perry MC, Goldberg RM, Mayer RJ, Colacchio TA. Documenting the natural history of patients with resected stage II adenocarcinoma of the colon after random assignment to adjuvant treatment with edrecolomab or observation: results from CALGB 9581. J Clin Oncol. 2011;29:3146–3152. doi: 10.1200/JCO.2010.32.5357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen BS, Jorgensen S, Fog JU, Sokilde R, Christensen IJ, Hansen U, Brunner N, Baker A, Moller S, Nielsen HJ. High levels of microRNA-21 in the stroma of colorectal cancers predict short disease-free survival in stage II colon cancer patients. Clin Exp Metast. 2011;28:27–38. doi: 10.1007/s10585-010-9355-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Connell MJ, Mailliard JA, Kahn MJ, Macdonald JS, Haller DG, Mayer RJ, Wieand HS. Controlled trial of fluorouracil and low-dose leucovorin given for 6 months as postoperative adjuvant therapy for colon cancer. J Clin Oncol. 1997;15:246–250. doi: 10.1200/JCO.1997.15.1.246. [DOI] [PubMed] [Google Scholar]
- O'Connor ES, Greenblatt DY, LoConte NK, Gangnon RE, Liou JI, Heise CP, Smith MA. Adjuvant chemotherapy for stage II colon cancer with poor prognostic features. J Clin Oncol. 2011;29:3381–3388. doi: 10.1200/JCO.2010.34.3426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schetter AJ, Leung SY, Sohn JJ, Zanetti KA, Bowman ED, Yanaihara N, Yuen ST, Chan TL, Kwong DL, Au GK, Liu CG, Calin GA, Croce CM, Harris CC. MicroRNA expression profiles associated with prognosis and therapeutic outcome in colon adenocarcinoma. JAMA. 2008;299:425–436. doi: 10.1001/jama.299.4.425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmoll HJ, Van CE, Stein A, Valentini V, Glimelius B, Haustermans K, Nordlinger B, van d V, Balmana J, Regula J, Nagtegaal ID, Beets-Tan RG, Arnold D, Ciardiello F, Hoff P, Kerr D, Kohne CH, Labianca R, Price T, Scheithauer W, Sobrero A, Tabernero J, Aderka D, Barroso S, Bodoky G, Douillard JY, El GH, Gallardo J, Garin A, Glynne-Jones R, Jordan K, Meshcheryakov A, Papamichail D, Pfeiffer P, Souglakos I, Turhal S, Cervantes A. ESMO Consensus Guidelines for management of patients with colon and rectal cancer. A personalized approach to clinical decision making. Ann Oncol. 2012;23:2479–2516. doi: 10.1093/annonc/mds236. [DOI] [PubMed] [Google Scholar]
- Shibuya H, Iinuma H, Shimada R, Horiuchi A, Watanabe T. Clinicopathological and prognostic value of microRNA-21 and microRNA-155 in colorectal cancer. Oncology. 2010;79:313–320. doi: 10.1159/000323283. [DOI] [PubMed] [Google Scholar]
- Simon RM, Subramanian J, Li MC, Menezes S. Using cross-validation to evaluate predictive accuracy of survival risk classifiers based on high-dimensional data. Brief Bioinform. 2011;12:203–214. doi: 10.1093/bib/bbr001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slaby O, Svoboda M, Fabian P, Smerdova T, Knoflickova D, Bednarikova M, Nenutil R, Vyzula R. Altered expression of miR-21, miR-31, miR-143 and miR-145 is related to clinicopathologic features of colorectal cancer. Oncology. 2007;72:397–402. doi: 10.1159/000113489. [DOI] [PubMed] [Google Scholar]
- Slaby O, Svoboda M, Michalek J, Vyzula R. MicroRNAs in colorectal cancer: translation of molecular biology into clinical application. Mol Cancer. 2009;8:102. doi: 10.1186/1476-4598-8-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valeri N, Gasparini P, Braconi C, Paone A, Lovat F, Fabbri M, Sumani KM, Alder H, Amadori D, Patel T, Nuovo GJ, Fishel R, Croce CM. MicroRNA-21 induces resistance to 5-fluorouracil by down-regulating human DNA MutS homolog 2 (hMSH2) Proc Natl Acad Sci USA. 2010;107:21098–21103. doi: 10.1073/pnas.1015541107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Volinia S, Croce CM. Prognostic microRNA/mRNA signature from the integrated analysis of patients with invasive breast cancer. Proc Natl Acad Sci USA. 2013;110:7413–7417. doi: 10.1073/pnas.1304977110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xi Y, Nakajima G, Gavin E, Morris CG, Kudo K, Hayashi K, Ju J. Systematic analysis of microRNA expression of RNA extracted from fresh frozen and formalin-fixed paraffin-embedded samples. RNA. 2007;13:1668–1674. doi: 10.1261/rna.642907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamichi N, Shimomura R, Inada K, Sakurai K, Haraguchi T, Ozaki Y, Fujita S, Mizutani T, Furukawa C, Fujishiro M, Ichinose M, Shiogama K, Tsutsumi Y, Omata M, Iba H. Locked nucleic acid in situ hybridization analysis of miR-21 expression during colorectal cancer development. Clin Cancer Res. 2009;15:4009–4016. doi: 10.1158/1078-0432.CCR-08-3257. [DOI] [PubMed] [Google Scholar]
- Zaniboni A, Labianca R. Adjuvant therapy for stage II colon cancer: an elephant in the living room. Ann Oncol. 2004;15:1310–1318. doi: 10.1093/annonc/mdh342. [DOI] [PubMed] [Google Scholar]
- Zhu S, Wu H, Wu F, Nie D, Sheng S, Mo YY. MicroRNA-21 targets tumor suppressor genes in invasion and metastasis. Cell Res. 2008;18:350–359. doi: 10.1038/cr.2008.24. [DOI] [PubMed] [Google Scholar]