Abstract
Ovarian cancer is the deadliest of all gynecological cancers and the fifth leading cause of death due to cancer in women. This is largely due to late-stage diagnosis, poor prognosis related to advanced-stage disease, and the high recurrence rate associated with development of chemoresistance. Survival statistics have not improved significantly over the last three decades, highlighting the fact that improved therapeutic strategies and early detection require substantial improvements. Here, we review and highlight nanotechnology-based approaches that seek to address this need. The success of Doxil, a PEGylated liposomal nanoencapsulation of doxorubicin, which was approved by the FDA for use on recurrent ovarian cancer, has paved the way for the current wave of nanoparticle formulations in drug discovery and clinical trials. We discuss and summarize new nanoformulations that are currently moving into clinical trials and highlight novel nanotherapeutic strategies that have shown promising results in preclinical in vivo studies. Further, the potential for nanomaterials in diagnostic imaging techniques and the ability to leverage nanotechnology for early detection of ovarian cancer are also discussed.
Keywords: clinical trials, photodynamic therapy, gene therapy
I. OVARIAN CANCER
A. Introduction
Ovarian cancer ranks as the fifth leading cause of death due to cancer in women, yet this disease only accounts for ~5% of all female cancer cases.1 In 2013, it was predicted that approximately 22,240 new cases of ovarian cancer will be diagnosed in the United States.1 Ovarian cancer is the most common cause of death due to gynecological malignancy, with an estimated 14,030 deaths predicted for 2013.1 Survival statistics for ovarian cancer are encouraging if the cancer is detected at an early stage (<III), without spread to the peritoneum and surrounding organs. However, if a patient is diagnosed with significant metastatic disease, the five-year overall survival drops to a dismal 20%. Unfortunately, more than 60% of all ovarian cancer cases are still diagnosed at stage III or later and the overall survival statistics have not improved greatly over the past 30 years.2 These data highlight that early detection of ovarian cancer and improvements in therapeutic intervention for late-stage metastatic cancer are desperately needed to combat this deadly disease.
Greater than 90% of ovarian cancer cases are of epithelial origin. Examples of the much less frequent nonepithelial ovarian cancers include germ cell and stromal derived tumors. It is becoming increasingly clear that the broad classification of epithelial ovarian cancer (EOC) encompasses a number of different diseases that simply share the same localization within the intraperitoneal (IP) cavity.2 These are divided into five histological subgroups: high-grade serous, low-grade serous, mucinous, clear cell, and endometriod ovarian cancer. It is now known that these are very distinct diseases based on histology, genomic analysis, and their tissue of origin (Table 1). Most prominently, the cancer genome atlas (TCGA) recently revealed that >96% of high-grade serous ovarian cancers contain mutations in TP53 and alterations in the BRCA genes (mutation and gene methylation). These are also characterized by high frequency in somatic gene copy number alterations, indicative of defects in homologous recombination repair.3 Conversely, clear cell carcinomas have less genomic instability, but display a wide variety of somatic mutations, some of which are pathway specific, such as those affecting PI3 kinase signaling. While it is beyond the scope of the present review to discuss these in detail, we are aware that novel nanotechnology-based therapies will have to take these differences into consideration, especially when developing pathway specific/targeted drug therapies and biomarker-based diagnostics. To date, most research efforts have focused on the development of novel therapies for serous ovarian cancer, since this is the most frequently occurring histological subtype of ovarian cancer and provides the largest patient pool in clinical studies. We refer the reader to a number of excellent reviews that discuss the different histological subtypes, their genetic markers, and tissues of origin in greater detail.2,4–7
TABLE 1.
| Histologic subtype | Percentage of EOC cases (%) | Most common genetic changes | Tissue of origin |
|---|---|---|---|
| Serous, high grade (type II)* | 70 | TP53 (96%) BRCA1/BRCA2 (22%) |
Fallopian tube fimbria |
| Serous, low grade (type I) | <5 | BRAF/KRAS (50%) ERBB2 (9%) |
Fallopian tube fimbria |
| Clear cell (type I) | 10 | ARIK1A (50%) PIK3CA (50%) PTEN (20%) |
Endometrial/endometriosis |
| Endometrioid (low grade, type I; high grade, type II) | 10 | CTNNB1 (40%) PIK3CA (20%) PTEN (20%) |
Endometrial/endometriosis |
| Mucinous (type I) | <5 | KRAS (75%) | — |
An additional classification of ovarian cancers into two “types” has been proposed, with type II tumors being characterized by TP53 mutations, late-stage diagnosis, and poor outcome.5 It is generally now accepted that the fallopian fimbria is the precursor sites of HGSOC (high-grade serous ovarian carcinoma), possibly due to the implantation of the fimbria epithelial cells on the ovarian surface. However, a body of literature exists that continues to argue that a certain percentage of serous ovarian cancers may arise from the surface epithelium of the ovary. The estimation of mucinous subtype frequency was previously much higher, due to the misdiagnosis of gastrointestinal cancer derived–IP metastatic lesions.5
Germ-line mutations in the BRCA1 and BRCA2 genes that normally assist in DNA breakage repair are associated with increased risk and found in ~22% of high-grade serous ovarian cancer cases.3 The risk of developing clear cell and endometriod carcinomas has largely been associated with endometriosis, while the origin and risk factors for the low-frequency mucinous subtype remains largely unknown.5 It has been known for some time that the risk of developing ovarian cancer is proportional to the number of lifetime ovulations. For example, risk is decreased by pregnancies, contraceptive use, and tubal ligation or hysterectomy.8 Thus, the site of ovulation on the ovarian surface epithelium was previously thought to be the site of the precursor lesion of ovarian cancer. However, since the origin of most serous ovarian cancers has now been demonstrated to be derived from the fallopian tube epithelium, largely by genomic association of BRCA1/BRCA2 mutations,9 it is now believed that the fallopian tube fimbria comes into close contact with the ruptured ovarian surface epithelium during ovulation, which enhances formation of precursor lesions on the ovary.
Unlike other cancer types, ovarian cancer metastasis occurs largely by the transcoelomic route and disease is often diagnosed, especially in the case of high-grade serous ovarian cancer (type II tumors), once the cancer has extensively spread throughout the IP cavity.10,11 Stage III and IV cancers often present with high volumes of ascites, which lead to symptomatic distention and abdominal pain for the patient and this has been associated with poor outcomes.12
B. Current Diagnosis and Biomarkers
Unfortunately, 75% of ovarian cancer diagnoses occur after the carcinoma has reached stage III or IV.8 Often termed “the silent killer,” it was perceived that ovarian cancer does not normally manifest symptoms until spread beyond the ovaries. However, it is becoming evident that early symptoms of ovarian cancer exist but are variable between patients and often misdiagnosed as benign gastrointestinal and gynecological issues. These include bloating, abdominal pain, feeling full after eating, frequent urination, and irregular menstrual bleeding. As such, continuing education of the early signs and symptoms of ovarian cancer are pivotal to minimize the time to an accurate diagnosis. Manual pelvic examinations often suffer from a low sensitivity for early detection, which is improved by transvaginal sonographies. After pelvic examination, gynecological oncologists proceed to biopsy and histological analysis to confirm the presence and stage of the disease. This will often be accompanied by assessment of CA125 antigen levels in the patient serum.
Serum cancer antigen 125 (CA125), a glycoprotein, also known as mucin16 (MUC16), was first discovered by Bast et al., in an epithelial ovarian cancer–derived cell line.13 Unfortunately, CA125 is not a suitable marker for routine annual screening due to the large false-negative and false-positive incidences of the test. Many other benign conditions also exhibit expression of this marker, including endometriosis-induced irritation, pregnancy, and liver disease, potentially leading to false diagnosis. Routine screening of the general population by a combination of transvaginal ultrasound and CA125 blood tests was not recommended by the American Medical Association, since it did not reduce overall ovarian cancer mortality.14 It further highlighted the false-positive nature of the screen, which can result in significant complications for patients that undergo unnecessary abdominal biopsies. However, CA125 is still a valid tool for clinicians and most commonly used to monitor disease-free status and recurrence of the disease following resection and chemotherapeutic intervention.
The lack of a good biomarker has led to an intense search for alternatives. Several new tumor markers for ovarian cancer have been identified in sera of patients, based on proteomics analysis.15 For example, enhanced protein glycosylation has been observed in serum from ovarian cancer patients16–18 and elevation of 11 N-glycans was shown to be slightly more specific and sensitive than CA125.19 Unfortunately, none of these have proven to significantly enhance diagnosis of ovarian cancer on their own. To improve screening using CA125 antigen, several clinical trials are now focused on multiplex analysis, combining CA125 with other markers.20 An example is human epididymis secretory protein 4 (HE4), which is a glycoprotein that is expressed in 50% of ovarian cancer specimens that show negligible expression in CA125, and may therefore reduce the false-negative population pool.21 The risk of ovarian malignancy algorithm (ROMA), which combines CA125 and HE4 levels with menopausal status, has shown mixed results for its improvement over CA125-only screening. However, there appears to be an improvement in diagnosis of post-menopausal women.22,23 Similarly, the FDA-approved OVA1 test, which combines serum markers that were identified by SELDI-TOF-MS (CA125, transthyretin, ApoA1, beta-2 microglobulin, transferrin), did not show significant improved screening for patients when compared with CA125 alone.24,25 Other markers of ovarian cancer that are currently being tested as potential serum indicators of the disease include the glycoprotein mesothelin and the proteinases Kallikrein and Prostatin (PRSS8).26 In addition, ovarian cancer cells express high levels of vascular endothelial growth factor (VEGF) and cell surface proteins, such as the folate receptor, epidermal growth factor receptors (EGFR and HER2), integrins, and claudins. Screening tumor specimens for specific protein expression levels will aid in determining the optimal treatment regime and developing targeted therapies against tumor specific cell surface proteins (see below).
Our ability to better classify the different histological subtypes of ovarian cancer has significantly been improved by recent advances in genomic analysis. 3 The association of BRCA1/BRCA2 mutations with high-grade serous ovarian cancer has prompted increased screening for these markers in patients with familial history of ovarian and breast cancers. With the focus being placed on increasing the sensitivities to detect circulating tumor cells and DNA, our ability to incorporate routine genomic analysis to detect common mutations, gene copy number alterations, and hypermethylation signatures will only aid in our ability to more accurately diagnose ovarian cancer. While it is beyond the scope of this review, nanotechnology applications will likely aid our ability to isolate circulating tumor cells and detect circulating tumor DNA.27
C. Standard Clinical Treatment
The first-line treatment for ovarian cancer is cytoreductive surgery. After optimal tumor debulking, the standard therapy, which has been used since the 1970s, is largely comprised of intravenous (IV) platinum-based chemotherapy over a six- to eightweek treatment course. Initial response to agents such as cisplatin or carboplatin is usually greater than 70%. Unfortunately, a large proportion of patients relapses and develops platinum resistance. In this case, the second line of therapies include taxane or PEGylated liposomal doxorubicin (Doxil; see the Nanotherapeutic section for more detail). For platinum-sensitive cancers, most clinicians rely on the combination of carboplatin and Doxil for recurrent disease, since these have a lower toxicity profile compared with cisplatin and free doxorubicin. Other combination chemotherapies that have been approved for ovarian cancer include gemcitabine-taxol and bleomycin-etoposide-cisplatin. Due to the relative confinement of ovarian cancer and its metastatic lesions to the in IP cavity, gynecological oncologists have administered chemotherapy agents directly via IP route for many years. “Bathing the abdomen” in cisplatin and paclitaxel was shown to significantly increase progression-free survival for advanced stage (>III) ovarian cancer patients28 and a recent long-term follow-up study demonstrated a 17% increase in overall survival.29,30 Compared to IV, the administration of IP therapy is more cumbersome and subjects the patient to more discomfort, due to the positioning of an abdominal catheter. It has also been associated with increased risk of intra-abdominal infections and gastrointestinal side effects.30 However, the major benefits of IP therapy include better tumor targeting and potential decreased systemic toxicity. Novel nanotechnology-based therapeutic and imaging strategies may take advantage of this administrative route.
Due to the high frequency of platinum resistance, much effort is being placed on development of novel therapies. Although it is beyond the scope here to discuss these in detail, we refer the reader to other reviews.7,31 Most of the drugs entering clinical trials for ovarian cancer are based on our increasing knowledge of the genomic and signaling pathway aberrations of the different subtypes of ovarian cancer. For example, PARP (poly ADP ribose polymerase) inhibitors are increasingly being investigated for use in high-grade serous ovarian cancers, due to high-frequency defects in homologous recombination repair. Phase II clinical trials indicate that maintenance therapy with the PARP inhibitor olaparib, following two or more courses of platinum-based chemotherapy, increases progression-free survival.32 Other examples of clinical trials that focus on pathway-specific inhibition are those studying the use of mTOR inhibitors (e.g., temsirolimus), kinase inhibitors (sorafanib), and MAP kinase pathway inhibitors. Knowledge of aberrant genetics in the histological subtypes will likely guide treatment strategies. For example, in low-grade serous ovarian cancer, MAP kinase signaling pathways are an attractive target due to their high frequency in Ras mutations. A recent phase II trial with the MEK1/2 inhibitor AZD6244 (selumetinib) has shown an improvement in outcome, with 65% of patients in the trial showing stabilization of the disease.33 Unfortunately, clinical trials are often made difficult by the small number of type I histological ovarian cancer cases.
Unlike breast cancer, ovarian cancer does not generally present with mutations in the epidermal growth factor receptors EGFR (HER1) or HER2, although there is high expression of wild-type EGFR in about 60% of ovarian cancer cases and 18% of the mucinous subtype have enhanced levels of HER2. Unfortunately, clinical trials have largely reported no significant benefit in use of EGFR and HER2 inhibitors, such as erlotinib and herceptin, on ovarian cancer.7,34 On the other hand, targeting angiogenesis, the vascular endothelial growth factor (VEGF) pathway has been met with more success. Bevacizumab, an inhibitory antibody against VEGF-A, is often used in combination therapy, and has been shown to improve the progression-free survival in two recent phase III clinical trials.35,36 Importantly, it has shown significant benefit for platinum-resistant populations.7 As in other cancers, drug resistance to bevacizumab and toxicity need to be taken into consideration for future treatment strategies for ovarian cancer.
Given the increase in DNA synthesis and repair of proliferating cells, most cancer cells have a need for enhanced folate uptake, a precursor for purine and pyrimidine synthesis. Compounds such as the antifolate pemetrexed, the folate receptor (FR) inhibitor vintafolide, and folate receptor inhibitory antibody farletuzumab have been primarily tried on platinum-resistant ovarian cancer patient population and some encouraging results from phase II trials have been observed.37,38 As expected, patients with folate receptor positive cancers had greater response to these therapies, further arguing for individualized drug treatment strategies for ovarian cancer. It is anticipated that nanotechnology formulations will aid in improving targeting, drug efficacy, and toxicity profiles of these new treatment strategies.
II. NANOTECHNOLOGY IN DIAGNOSIS AND IMAGING
The last decades have seen great advancements in biosensor technology, point-of-care systems, improved imaging technology, and the use of multiplexed assays combined with bioinformatics. In recent years, microelectromechanical systems (MEMS) technology and various nanoparticle platforms have made strides to improve diagnostic techniques, largely by enhancing contrast agents used in imaging techniques and as a means to detect biomarkers. Given the lack of early diagnosis and discrepancy in clinical procedure for detection of ovarian cancer, novel nanotechnology-based tools may provide alternatives that will improve this area. Below, we provide a brief overview of how nanotechnology has been applied to ovarian cancer detection and imaging.
A. Nanotechnology for Biomarker Detection
As mentioned, there are limited established biomarkers for ovarian cancers and currently no routine screening of healthy women. The inadequate performance of current diagnostic methods has led to several investigations to identify new biomarkers and use of multiplex assays of serum protein levels (ROMA/OVA1). Recent reports indicate that in addition to CA125, human epididymis protein 4 (HE4) and mesothelin have some promise for use in multiplex assays to improve reliability and performance. 20,39 While any diagnostic test relies on good biomarkers, nanotechnology can help improve on current diagnostic technologies with capture, purification, and detection techniques to enable rapid, reliable, low-cost, and multiplex screening. There are a plethora of examples of how nanotechnology-based sensors and assays can be applied to cancer diagnostics. For the interested reader, we point to several reviews discussing nanotechnology and biosensing in general.40–43
That being said, demonstrations of nanotechnology and nanoparticles toward ovarian cancer screening/detection have been somewhat limited, and as mentioned above suffer from the same issues associated with use of CA125 as a biomarker for ovarian cancer. Nevertheless, in 2009, a microfluidic platform incorporating quantum dot-labeled CA125 antibodies was demonstrated.44 This assay relies on micropatterned wells, each holding a single agarose particle with immobilized capture antibody. Following antigen capture, a second quantum dot-labeled antibody enables fluorescent visualization. Using individually conjugated agarose beads, a multiplex assay for CA125, HER2, and carcinoembryonic antigen was realized with two orders of magnitude improved sensitivity for carcino embryonic antigen over enzyme-linked immunosorbent assay. Since the initial proof of principle study, this “programmable bio-nano-chip” has been further developed toward a point-of-care system for ovarian cancer screening.45
A recent example of ovarian cancer diagnostics incorporating nanoparticles include screen-printed gold nanoparticle electrodes for label-free impedimetric sensing of CA125, with a lower limit of detection of 6.7 U/ml (for adequate diagnosis, the serum levels should reach approximately 35 U/ml).46 Another example is an electroluminescent sensing platform that incorporates iron oxide nanoparticles labeled with a primary antibody and a dendrimer/luminol modified secondary antibody. This platform was shown capable of detecting CA125 at concentrations as low as 0.03 μU/ml.47 It is anticipated that MEMS systems, nanoparticles, lab-on-chip, and various alternative sensing platforms will be further developed to realize improved ovarian cancer screening and diagnostics, particularly for the multiplex screening approaches that appear necessary.
B. Nanoparticle-Based Imaging Agents
Over the last 10–15 years, a broad assortment of nanoparticle-based enhancement agents for in vitro and in vivo imaging has been developed. Essentially, every nanoparticle platform has been adapted for common imaging modalities in some way or another, and a complete survey is not within the scope of this article. For the interested reader, there are several reviews that include discussion of various nanoparticle systems for imaging.48–55 In this section, we discuss and summarize recent use of nanoparticles toward ovarian cancer imaging (see Table 2). Imaging modalities for cancers in general include computer tomography, positron-emission tomography/single-photon emission computed tomography (SPECT), magnetic resonance imaging (MRI), ultrasound, and optical imaging. For ovarian cancer, the latter three are most common, where transvaginal ultrasound is the preferred imaging modality for clinical diagnostics.
TABLE 2.
Summary of nanoparticle agents used for ovarian cancer imaging
| Imaging modality | Nanoparticle | Targeting | Drug | Specimen | Cell line | Refs |
|---|---|---|---|---|---|---|
| Optical | Quantum dot | CA125-antibody | N | Xenograft | HO8910 | 71 |
| Quantum dot | EPR | N | Xenograft | HEYA8 | 70 | |
| Quantum dot | Her2-antibody | N | In vitro | SKOV-3 | 72 | |
| Quantum dot | MUC1-aptamer | Y | Xenograft | A2780 | 73 | |
| Polymethacrylic acid (VIS) | Her2-antibody | In vitro | SKOV-3 | 184 | ||
| Lipoprotein (NIR) | Folic acid | Xenograft | — | 185 | ||
| PEG-PLA (NIR) | EPR | Xenograft | A2780 | 68 | ||
| PEG-lipid (NIR) | EPR | Xenograft | SKOV-3 | 69 | ||
| NaYF4:Yb3+/Er3+ @ SiO2 | — | In vitro | SKOV-3 | 186 | ||
| Magnetic resonance | Dendrimer | Biotin | Xenograft | SHIN3 | 62 | |
| Liposome | Folic acid | Xenograft | IGROV-1, OVCAR-3 | 63 | ||
| Iron oxide | Folic acid | In vitro | SKOV-3 | 64 | ||
| Iron oxide/Gd | EPR | Xenograft | SKOV-3 | 65 | ||
| Photo acoustic | Gold nanorods | EPR | Xenograft | 2008, HEY, SKOV-3 | 75 | |
| Ultrasound | Liposome microbubbles | — | In vivo | N/A | 56, 57 |
1. Ultrasound (US) Imaging
US imaging is based on a high-frequency sound wave (>20 kHz), where a transducer is placed against the tissue and an image is reconstructed from the sound wave reflected by the internal organs. It is the most common imaging modality for ovarian cancer in the clinic, much of which can be attributed to its relatively low cost, simple/safe operation, and fast acquisition. Contrast agents for US imaging need to change the acoustic properties of the tissue, where particles with gas-filled cores (perfluorocarbon, air, etc.) are the most common approach to realize this. Albumin-stabilized perflutren particles (OPTISON) and lipid microspheres with perflutren (Definity) are two examples of commercial US imaging agents. Such microparticles have been demonstrated for transvaginal sonography to monitor changes in ovarian tumor microvasculature. 56,57 However, the contrast enhancement is related to particle size, and micron-scaled particles have shown better performance than nanoparticles, which limits the usefulness of reducing the size of such systems. Nevertheless, submicron polymeric particles containing gas bubbles are of interest since after IV administration they can accumulate in tumors to a higher degree than microparticles (through the EPR effect). Also, adding targeting ligands to these smaller particles can negate some of the loss of contrast.58–60 Currently, new technological advancements in ultrasound imaging, such as 3D and color Doppler sonography, combined with microbubble contrast appear promising for improved ovarian cancer imaging and diagnostics,61 where nanoparticle imaging agents may have a future role.
2. Magnetic Resonance Imaging (MRI)
MRI is a powerful, noninvasive in vivo imaging technique with high resolution (10–100 μm) and unlimited imaging depth. The MRI signal is based on the relaxation of hydrogen nuclei (in water) after being exposed to a pulsed radiofrequency signal. Contrast in MRI can be T1 weighted (differences in the spin-lattice relaxation of tissues), or T2 weighted (difference in spin-spin relaxation). Contrast agents are administered to influence the T1 or T2 relaxation parameters and enhance the imaging quality. Numerous nanoparticle constructs have been developed to provide contrast enhancement where several have been applied to cancer imaging. Iron oxide (IO) nanoparticles are frequently used for T2 imaging, whereas liposomes, micelles, dendrimers, and polymeric nanoparticles incorporating paramagnetic species, such as Gd3+, are common for T1 imaging.
An early example of MRI nanoparticles for in vivo imaging of ovarian cancer xenografts is biotin-conjugated dendrimers with Gd-chelates.62 In a similar fashion, a folate-modified liposome containing Gd3+-DOTA has been demonstrated for targeted in vivo imaging.63 Folic acid grafted to hyperbranched polyglycerol has also been used to modify the surface of iron oxide nanoparticles and has been demonstrated for targeted T2 imaging of SKOV-3 ovarian cells using phantoms (in vitro).64 Recently, a small 5 nm IO particle with embedded gadolinium and a zwitterionic surface coating was demonstrated for T1 in vivo imaging of SKOV-3 xenografts. These particles accumulated in tumors via the EPR effect and had good contrast with a circulation half-life of ~50 min with renal clearance.65 Nanoparticles with embedded gadolinium can possibly negate some of the toxicity concerns associated with ion leeching from standard chelates and may see clinical use in the future. Another interesting development is the combination of MRI and SPECT. Recently, an IO nanoparticle with a dextran coating was used to conjugate an 111In-labeled mesothelin antibody for targeted MRI/SPECT dual imaging of A431-K5 cell xenografts.66 Considering the same cell receptor is overexpressed in ovarian cancer cells, these or similar particles could potentially be used for dual imaging and diagnostics of ovarian tumors.
3. Optical Imaging (OI)
OI is gaining popularity for cancer in vivo imaging and, similar to ultrasound imaging, it is noninvasive, comparatively inexpensive, and fast. OI also has superior lateral resolution (<1 mm), though ultraviolet-visible wavelengths (250–700 nm) have poor tissue permeability and imaging is compromised by tissue scattering and background noise. There are similar issues associated with the near-infrared (NIR) window (700–900 nm); however, these wavelengths offer improved in vivo imaging over the other “visible” wavelengths with reduced scattering, deeper tissue penetration, and limited autofluorescence. NIR fluorescent probes for optical imaging should ideally have a large Stokes shift, high molar adsorption coefficient, good quantum yield, and photostability. A host of NIR dyes are available (or are being developed) for tissue imaging and have been reviewed by Luo et al.67 As can be expected, such NIR dyes have been incorporated in a host of polymeric nanoparticles, liposomes, dendrimers, etc. For example, a NIR dye (DiR) was loaded into polyethylene glycol-polylactic acid copolymer nanoparticles, and tumor accumulation and biodistribution were evaluated on mice xenografts of ovarian (A2780) and colon (HT29) cell lines. In vivo imaging was possible up to 48 h after injection with associated reticuloendothelial system (RES) clearance with accumulation in liver and spleen. They noted a higher tumor accumulation and slower RES clearance of 111 nm particles compared with 166 nm particles.68 In another study, PEGylated micelles encapsulating a NIR dye (ICG) along with drugs (paclitaxel and tanespimycin) were injected in mice with SKOV-3 ovarian xenografts. These nanoparticles, accumulated in tumors via the EPR effect, displayed improved therapeutic effect over free drug and allowed imaging over 48 h.69
The emergence of inorganic semiconducting nanoparticles, i.e., “quantum dots, QDs,” have great potential for optical imaging where QDs have improved optical qualities compared with most dyes such as reduced photobleaching, broad excitation range, narrow emission window, good quantum yield, and large Stokes shifts. QDs are frequently used in vitro and have been used for imaging of several ovarian cancer cell lines.70–72 Quantum dots (with emission at approximately 500 nm) have also been conjugated with both a targeting aptamer (MUC1) and doxorubicin for dual imaging and treatment of A2780 ovarian cancer xenografts.73 One major concern regarding in vivo use of QDs in general is their stability and potential toxicity. Recent developments in quantum dot synthesis and their application toward deep tissue imaging has focused on using less toxic materials (see review by Cassette et al.),74 which may lead to breakthroughs using QDs for clinical in vivo cancer imaging.
4. Photoacoustic (PA) Imaging
Another “optical” technique that is complementary to US is photoacoustic (PA) imaging. In PA imaging, local thermal heating using NIR light induces a sound wave that is used to build an image. PA imaging has good spatial resolution (up to 50–500 μm) and reasonable tissue penetration (5 cm). Local heating can be achieved by exiting endogenous molecules such as hemoglobin or via administered small molecules and nanomaterials. Gold nanorods, for instance, can be optically stimulated with NIR light to induce enhanced local heating effects and provide contrast. In a recent paper, gold nanorods were demonstrated for in vivo PA imaging of ovarian cancer xenografts in mice. The authors found that a nanorod aspect ratio of 3.5 was optimal for in vivo imaging, where maximum signal was seen 3 h postinjection with signal enhancement effects for up to two days. The authors also demonstrated surface enhanced Raman in vivo imaging with these particles.75 The combination of optical/photoacoustic imaging with ultrasound imaging is interesting and has the potential to make the translation into a clinical setting with their complementary qualities and similar acquisition modes.
III. NANOTECHNOLOGY IN THERAPEUTICS
A. Chemotherapeutics
The unique physical and chemical properties of nanoparticles have been exploited to enhance delivery of chemotherapeutics. Due to leaky vasculature and enhanced uptake mechanisms of cancer cells, nanoparticle formulations can take advantage of the enhanced permeability and retention effect (EPR) to accumulate in tumors without any specific targeting (passive targeting).76 Compared to systemic administration, nanoparticles with encapsulated chemotherapeutics can facilitate increased dose delivery to the tumor environment. Beyond the EPR effect, decorating the particles with cancer cell–specific ligands offers an additional route to deliver a high drug dose in a targeted fashion. Importantly for the patient, encapsulation and site-specific delivery can reduce the toxicity profile of a number of therapeutics already accepted for use in the clinic. The vast array of different nanostructures and chemistries available enables design of functionalized particles that incorporate diagnostic, imaging, and drug-delivery properties, now termed “theranostics.” Below, we describe the most common nanoparticle platforms and provide examples of their use in ovarian cancer therapeutics, focusing on systems moving toward clinical translation (summarized in Table 3).
TABLE 3.
Clinical trials for ovarian cancer using nanoformulations
| Compound Name | Formulation | Active agent | Status for ovarian cancer | Trial No., Refs. |
|---|---|---|---|---|
| Doxil | PEGylated liposomal doxorubicin | Doxorubicin | FDA approved | NCT00945139, NCT00862355, NCT00248248 |
| - | Liposomal topotecan | Topotecan | Phase I | NCT00765973 |
| OSI-211 | Liposomal lurotecan | Lurotecan | Phase II | NCT00010179 |
| Abraxane, ABI-007 | Nanoparticle bound albumin paclitaxel | Paclitaxel | Phase II Approved for breast, small cell and pancreatic cancers |
NCT00466986, NCT00407563, 96–98, 187 |
| Xyotax | Paclitaxel poliglumex | Paclitaxel | Phase II | NCT00060359, NCT00017017, 99, 101 |
| Nanotax | Nanoparticle suspension | Paclitaxel | Phase I | NCT00666991 |
| NKTR-102 | Etirinotecan pegol | Topoisomerase I inhibitor | Phase II | NCT00806156103, 104 |
| CRLX101 | Cyclodextrin-PEG Camptothecin |
Camptothecin | Phase I | NCT00333502, NCT01652079, 105 |
| ProLindac (AP5346) | HPMA-oxaliplatin analogue | Oxaliplatin analogue | Phase II | 106, 107 |
| AP5280 | HPMA-carboplatinate analogue | Carboplatin analogue | Phase II | 107, 108, 188 |
1. Liposomes
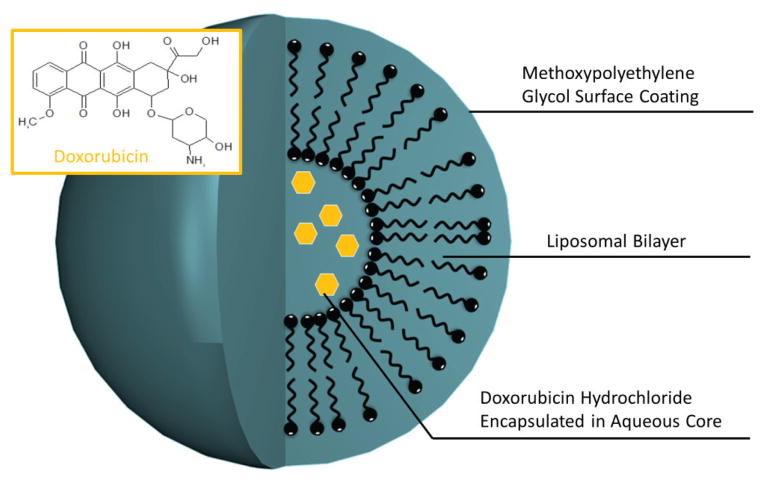
Early on, liposomal encapsulation was recognized to effectively enhance solubility and plasma retention time for a number of compounds. Nanoliposomal formulations offer hope in increasing the biocompatibility, retention time, and accumulation of chemotherapeutic agents. The quintessential example of this is Doxil, a PEGylated liposomal doxorubicin formulation that has been FDA approved for use in recurrent and platinum-resistant cancers (see Fig. 1).
FIG. 1.
Schematic of PEGylated liposomal doxorubicin (Doxil) structure. Doxorubicin (inset) loaded core within a liposomal formulation protected by methyoxypolyethylene glycol.
Doxil is now often used in combination therapy with gemcitabine in platinum-resistant patients. Nanoparticle encapsulation was shown to enhance accumulation in patient tumors and increase plasma half-life compared with free doxorubicin, with circulating liposomes staying intact after several days following administration.77 Phase II trials also demonstrated a minimal toxicity profile of Doxil when compared with studies of free doxorubicin.78 In phase III clinical trials, use of Doxil showed comparable benefit to the standard therapy of treatment populations with topotecan and gemcitabine for patients with recurrent or refractory ovarian cancer.79–82 Doxil displayed a progression-free and overall survival advantage in a subpopulation with platinum-resistant cancers.79,80 The CALYPSO trial further demonstrated that use of Doxil in combination with carboplatin does not appear to provide an advantage over standard therapy of paclitaxel with carboplatin in platinum-sensitive cancers. However, increases in progression-free survival were observed in the Doxil/carboplatin arm of the study in a subpopulation of sia.86–88 A caveat to the prolonged plasma half-life of nanoencapsulated drug formulations is the potential for long-term side effects. Although the EPR effect ensures that most of the nanoparticle will accumulate in tumor tissue, once this dosage has been reached there is a potential for the remaining drug in circulation to enter other tissues. To decrease the chance of these side effect, the CARL-trial (controlled application and removal of liposomal chemotherapeutics) investigated the use of double-filtration plasmapheresis for extracorporeal elimination of Doxil from the circulation.76 The investigators were able to successfully remove >60% of total plasma Doxil (~45% of the initial drug dose) in intact liposomal form, from plasma of ovarian cancer patients.
Since the successful implementation of Doxil in the clinical setting, a variety of nanoformulations for anticancer treatment have been investigated (Table 3). A phase I clinical trial of liposomal topotecan is partially platinum-resistant patients.83 The benefit of Doxil combination therapy on platinum-resistant cancers was further highlighted by a study that compared Doxil and trabectedin to Doxil monotherapy, with improved overall and progression-free survival observed in both platinum-resistant and partially resistant subpopulations.84,85 Since Doxil has been incorporated as a standard therapy for ovarian cancer, many current clinical trials are comparing novel therapeutics to this agent.
Acute Doxil toxicity includes hair loss, nausea, vomiting, diarrhea, constipation, tiredness, and orange discoloration of sweat and urine, and in certain cases severe skin irritations, such as erythrodysesthesia. While the acute toxicity profile of Doxil appears to be milder than that of free drug and other standard therapies, there is some concern that prolonged maintenance therapy may lead to cardiac problems, such as hypertension, renal dysfunction, and neoplaongoing for use in ovarian cancer (NCT00765973). Topotecan, a topoisomerase I inhibitor indicated for the treatment of recurrent ovarian metastatic carcinoma, has been shown to have efficacy equivalent to both paclitaxel and Doxil for relapsed ovarian cancer.89 The liposomal formulation seeks to protect and selectively deliver topotecan to the tumor site to improve efficacy. One modification for increased nanoparticle uptake is the targeting of the liposome to HER2. In vivo studies in breast cancer xenografts indicate there is an increased uptake of targeted liposomal topotecan over untargeted analogues or free drug.90 PEGylation has also been considered for use with liposomal topotecan. Unfortunately, the increased retention for PEGylated liposomal doxorubicin has not been observed with topotecan.91 Rather, an unfavorable accelerated blood clearance phenomenon is seen in rats.92 An alternative treatment to topotecan also in clinical trials for use in recurrent ovarian cancer is a liposomal formulation of lurtotecan, OSI-211 (NCT00010179). This liposomal formulation had substantially different exposure profiles than nonencapsulated compound, achieving prolonged exposures clinically. Despite this increased exposure, daily dosing of inhibitor was still needed for optimal activity of the agent. While this had tolerable side effects, the activity was not significant in pretreated populations and the formulation has not yet garnered approval.93
2. Micelle-Like Structures and Polymer Aggregates
These structures typically contain a more hydrophobic component that helps solubilize/encapsulate therapeutic compounds, while a hydrophilic component provides stability of the assembly in aqueous environments and offers conjugation sites for eventual targeting ligands. Alternatively, the drug can be conjugated directly to a polymer backbone before assembly of the nanostructure (where the drug also can induce the assembly/aggregation). The line between associated polymer chains in a nanoscale aggregate compared with a solid-core polymeric nanoparticle with encapsulated drug is not always clear cut; however, most polymer systems used in clinical trials to date we deem to belong to this category.
One exception would be nanoparticle bound albumin paclitaxel (nab-paclitaxel or commercial Abraxane), which has been approved by the FDA for the treatment of metastatic breast cancer, small cell lung cancer, and most recently pancreatic adenocarcinoma. 94 Here, the albumin stabilizes ~130 nm paclitaxel particles and has an advantage over the current formulations of paclitaxel that involve synthetic solvents for delivery, which require premedications and can cause hypersensitivity reactions.95 The first reported usage of Abraxane in ovarian cancer occurred in 2006 to continue taxane treatment after a paclitaxel-induced hypersensitivity reaction in 60-year-old patients. The solvent-free formulation allowed for completion of three cycles of treatment without incident.96 Since then, nab-paclitaxel has undergone phase II clinical trials for both platinum-sensitive and -resistant ovarian and peritoneal cancers. Weekly infusion of Abraxane resulted in significant clinical response and a progression-free survival of 4.5 months in a refractory-dominated platinum-resistant population.97 In the platinum-sensitive population, there was a higher overall response rate of 64%. Both studies indicated noteworthy activity and tolerable toxicities, calling for further phase III studies.98
Another solvent-free nanoformulation is paclitaxel poliglumex (Xyotax), which is a linear poly-L-glutamic acid-paclitaxel conjugate. The biodegradable polymer is conjugated via an ester linkage to the paclitaxel carboxylic acid side chains, resulting in a 37% by weight drug compound that can degrade into L-glutamic acid and enter normal cellular metabolism. In animal studies, this proved to be more effective than free drug and had limited systemic exposure. Phase I and II clinical studies show an encouraging therapeutic efficiency without the need for premedications.99 A phase II clinical trial for recurrent EOC indicated a modest activity level for Xyotax when administered every 21 days as a second- or third-line therapy, but it may have more applicability as a maintenance chemotherapeutic. 100 When delivered every three weeks in conjunction with carboplatin, Xyotax was shown to be feasible as a first-line therapy. Multiple cycles of therapy were tolerable, with a decrease in alopecia, shorter infusion times, and less hypersensitivity than paclitaxel combination therapy.101 Nanotax, a proprietary nanoparticle suspension of paclitaxel, is also currently undergoing phase I trials (NCT00666991) for use in ovarian cancer.
A polymer conjugate that is a possible prodrug for platinum-resistant ovarian cancer is etirinotecan pegol (NKTR-102, Nektar). Here, the topoisomerase I inhibitor irinotecan is bound to a proprietary PEG nanoparticle via a biodegradable linker. This formulation is inactive until broken down via cellular mechanisms, releasing the active inhibitor. In a mouse model, it was shown to have superior activity than free irinotecan.102 Phase I clinical studies in a small cohort showed antitumor activity in heavily pretreated patients.103 This system was highly active in phase II trials against platinum-resistant ovarian cancer, administered every three to four weeks, resulting in 5.4 months progression-free survival and continuing on to phase III studies for both ovarian and breast cancer.104 Another topoisomerase inhibitor that benefits from nanoformulation is camptothecin, which has been incorporated into a cyclodextrin-PEG nanoparticle (see scheme in Fig. 2). This nanocomplex demonstrated superior therapeutic efficiency to free irinotecan by facilitating retention and sustaining an active supply of inhibitor at the tumor target. Phase I trials showed improved tolerability and efficacy versus free camptothecin.105
FIG. 2.
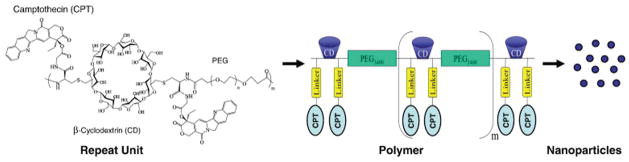
Schematic diagram of CRLX101, a nanopharmaceutical comprised of camptothecin conjugated to a linear, cy clodextrin-poly(ethylene glycol) (CD-PEG) copolymer and formulated into nanoparticles. Reprinted from Ref. 105. Copyright 2011, with permission from Elsevier.
HPMA (N-(2-hydroxypropyl)methacrylamide) is similar to PEG, a well-established biocompatible polymer used as a platform for drug delivery. Clinical trials of two different carboplatin mimetics, an oxaliplatin analogue (ProLindac, AP5346) and a carboplatinate analogue (AP5280), bound to HPMA via an acid-sensitive linker have been initiated. ProLindac had excellent preclinical results and was more effective than cisplatin, with a 16-fold higher platinum accumulation in the tumor than oxaliplatin. Phase I studies showed excellent tolerability but did not exhibit a marked increase in efficiency over oxaliplatin.106 In phase II trials for single-agent efficacy, two thirds of late-stage heavily pretreated ovarian cancer patients exhibited disease stabilization, opening avenues for further combination clinical studies. The AP5280 HPMA carboplatinate analogue has also undergone phase I and II trials, showing favorable toxicities.107,108
3. Nanoparticle Systems in Preclinical Development
Above, we have focused on the current efforts for translational applications of nanoparticles for ovarian cancer therapy. Notably, these are primarily consisting of liposomal or polymer-aggregate/micelle–based structures and are non-targeting. Below, we provide examples of nanoparticle systems and therapeutics in the preclinical stage, which may be of benefit for future ovarian cancer therapeutics. This includes the use of targeted nanoparticles to improve tumor specificity and drug accumulation. Our intention is not to discuss in detail all preclinical chemotherapeutic nanoformulations that have been demonstrated, but rather we choose to highlight some representative examples. Table 4 provides a broader summary of various preclinical targeted and non-targeted nanoparticle systems demonstrated on ovarian cancer cells.
TABLE 4.
Examples of preclinical studies of chemotherapy-loaded nanoparticle systems for ovarian cancer treatment
| Composition | Targeting moiety | Target | Active agent | Status | Refs. |
|---|---|---|---|---|---|
| Biodegradable micelle | — | — | Doxorubicin | Xenograft | 110 |
| PEG-glutamic acid-phenylalanine micelle | — | — | Cisplatin, paclitaxel | Xenograft | 109 |
| PEG- poly(epsilon-caprolactone) micelle | — | — | Paclitaxel, cylopamine, gossypol | Xenograft | 111 |
| PEG-cholic acid-lysine Telodendrimer | — | — | Paclitaxel | In vitro, xenograft | 116, 117 |
| PAMAM dendrimer | — | — | Cisplatin | Xenograft | 112 |
| PLGA Conjugate | — | — | Docetaxel | Xenograft | 189 |
| PLGA Conjugate | — | — | Curcumin | In vitro | 190 |
| HPMA Conjugate | — | — | Gemcitabine, Paclitaxel | In vitro | 191 |
| MPEG-PCL micelle | — | — | Quercetin | In vitro | 192 |
| Nanogel | — | — | Paclitaxel | Xenograft | 193 |
| Polymeric nanoparticle | — | — | Paclitaxel, C-6 Ceramide | Xenograft | 118 |
| Cholesterol nanoparticle | — | — | Platinum (II) complex | Xenograft | 194 |
| Iron oxide nanoparticle | Magnetic | — | Cisplatin | In vitro | 119 |
| PEG-PE micelle | Monoclonal antibody | Nucleosome | Doxorubicin | In vitro | 195 |
| Quantum dot | Aptamer | Mucin 1 | Doxorubicin | Xenograft | 73 |
| PLGA-lecithin-PEG nanoparticle | Folate | Folate binding protein | Paclitaxel | Xenograft | 196 |
| Liposome | Folate | Folate binding protein | Carboplatin | Xenograft | 197 |
| Nanogel | Folate | Folate binding pro | Paclitaxel, cisplatin | Xenograft | 122 |
| Poly(L-histidine) micelle | Folate | Folate Binding Protein | Doxorubicin | Xenograft | 125, 126, 198 |
| Nanogel | LHRH | LHRH receptor | Paclitaxel, cisplatin | Xenograft | 123 |
| Liposome | LHRHa | LHRH receptor | Docetaxel | Xenograft | 120 |
| PLA nanoparticle | Peptide | Follicle-stimulating hormone receptor | Paclitaxel | Xenograft | 199, 200 |
| Polyacrylamide | Peptide (F3) | Nucleolin protein | Cisplatin | Xenograft | 201 |
| PEG micelle | Peptide (OA02) | Integrin (α-3) | Paclitaxel | Xenograft | 121 |
| Liposome | Peptide (RGD) | Integrin | Paclitaxel | Xenograft | 202 |
| PLA nanoparticle | Peptide | HER-2 | Paclitaxel | In vitro, xenograft | 203–205 |
| mPEG-PLGA-PLL nanoparticle | EGF | EGFR | Cisplatin | Xenograft | 206 |
| Liposome | Transferrin | TfR | Cisplatin | In vitro | 207 |
a. Non-Targeted Nanoparticles
Polymeric nanostructures can be designed to provide additional control of drug release at the tumor site due to changes in the intracellular microenvironment, including pH and enzyme activity. In a recent study, a triblock ethylene glycol-glutamic acid-phenylalanine assembled to form a hydrophobic core covered in a cross-linked shell and protective PEG corona. These micelles were coloaded with both cisplatin and paclitaxel, which have a synergistic cytotoxicity. In the presence of proteolytic enzymes such as cathepsin B, the polymer degrades and releases both drugs. In an in vivo xenograft of multidrug-resistant ovarian cancer tumors, this combination therapy excelled compared to co-administration of free drugs or micelles containing only one therapeutic agent.109
Another example of a biodegradable copolymer micelle, not yet in clinical trials, has been designed for IP sustained release of doxorubicin to stabilize chemotherapeutics and improve pharmacokinetics. This novel nanoparticle release system is composed of 30 nm micelles, formed from a poly(ethylene glycol)–poly(ε-caprolactone)–poly(ethylene glycol) copolymer that aggregates at physiological temperatures to form a nonflowing gel. This encapsulation significantly improved antitumor effects and demonstrated anti-adhesion effects, preventing rapid absorption that can cause harmful postsurgical adhesions and reduce the duration of drug exposure. A gradual degradation of the gel then allows for sustained drug release followed by complete clearance from the cavity.110 A similar micelle platform for IP delivery utilized the simultaneous loading of paclitaxel along with two different inhibitors, cylopamine and gossypol. This three-pronged therapeutic approach dramatically reduced tumor volumes, inhibiting growth and prolonging survival after IP injection in a mouse xenograft.111
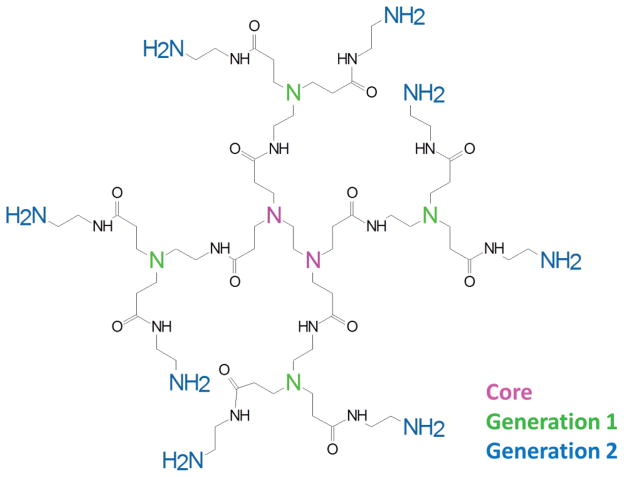
Traditional dendrimers, such as polyamidoamine (PAMAM) (Fig. 3), have shown promise as delivery systems of pharmaceutical compounds. For instance, the drug cisplatin was loaded into PAMAM dendrimers of varying size diameters ranging from 2.7 to 5.9 nm, depending on generation. Loading efficiencies and cisplatin concentrations could be tuned accordingly and showed activity comparable to free cisplatin when tested in an in vivo murine model. The dendrimeric loading also exhibited a lower toxicity, subsequently increasing the maximum tolerated dose, which resulted in improved tumor reduction.112 Controlled release from PAMAM dendrimers has been investigated for a variety of cancers, using pH-sensitive linkers, steric hindrance, and different linking chemistries to control drug release to a desired environment.113–115 An amphiphilic dendritic copolymer telodendrimer for ovarian cancer therapy has been pioneered by the Lam group from the University of California Davis Cancer Center. This system utilizes a dendritic composition of PEG, cholic acid, and lysine to form a drug-loaded core/shell structure with a high loading capacity for aqueous paclitaxel. While this formulation exhibited similar antitumor activity to the clinical Taxol and nanoformulation Abraxane in vitro, it had preferential tumor uptake and deep tissue penetration in a mouse model. This resulted in superior antitumor effects compared to the FDA-approved complexes.116 A radiolabeled biodistribution confirmed this preferential uptake at the tumor site as well as slower pharmacokinetics than Taxol, thus establishing it as a promising carrier to move forward into clinical testing.117
FIG. 3.
Example of a second-generation PAMAM dendrimer structure
While not used in clinical trials against ovarian cancer per se, an interesting polymeric nanoparticle system was demonstrated to effectively reduced tumor burden in xenografts through controlled release of coloaded paclitaxel and ceramide. The particle design separated the two drug components by encapsulating paclitaxel in a pH-responsive poly(beta-amino ester), which was internalized into slow-releasing PLGA [poly(d,l-lactice-co-glycolide)] containing ceramide.118
b. Targeted Nanoparticles
Naturally, many of the nanoparticle systems in clinical trials are destined to be developed further to incorporate active targeting. In ovarian cancer, highly expressed cell-surface proteins include the folate receptor, EGF receptor (EGFR, HER2), luteinizing hormone receptor, claudins, mucins, and integrins. While the most popular strategies include targeting of these by peptides and antibodies, we make a note that novel magnetic targeting is also on the horizon for tumor localization. For example, iron oxide nanoparticles loaded with cisplatin were magnetically localized to ovarian cancer cell lines in vitro, resulting in a 110-fold increase in cytotoxicity.119 The major obstacle to practical clinical implementation of this technique, however, is controlling a magnetic field strong and sharp enough for targeting in tissue.
Several in vivo xenograft studies reveal promising results using targeted liposomal formulations for ovarian cancer. Docetaxel-loaded cholesterol liposomes were targeted using a lutenizing hormone-releasing hormone analogue, LHRHa. Within 60 min of administration, the targeted liposome accumulated nine times more docetaxel at the ovarian tumor compared with the free compound and significantly decreased unwanted accumulation in the liver and spleen.120 Another targeting moiety explored for micelle carriers is the “OA02” peptide, having a high affinity for the α-3 integrin receptor overexpressed on tumor cell surfaces. This ligand improved localization and intracellular uptake of PEG micelles. When loaded with paclitaxel, the complex exhibited a greater antitumor efficacy in vivo versus the clinical formulation Taxol and the untargeted PEG complex, demonstrating the value of targeting. This not only offers hope clinically for efficacy, but its decreased systemic toxicity might relieve some of the negative chemotherapeutic symptoms.121
A folate-targeted nanogel, utilizing a cross-linked diblock copolymer, was designed by Nukolova et al. for ovarian cancer therapy. This complex, loaded with cisplatin or doxorubicin, demonstrated tumor-specific delivery and impressive antitumor activity in a murine ovarian cancer model.122 This group achieved similarly promising results targeting a cisplatin-loaded nanogel to lutenizing hormone-releasing hormone. This marker is overexpressed, not only in ovarian but also breast and prostate cancers, opening the door for use in multiple cancer types.123 In nanogel synthesis schemes, the 3D bulk material is first engineered, followed by the introduction of the therapeutic agent. This limited manipulation of the drug helps maintain pharmaceutical integrity.
Another example where folate is used as a targeting ligand is for doxorubicin-loaded poly(L-histidine) micelles. This system was designed for controlled release in acidic pH, and untargeted micelles increased the plasma half-life by five times to that of free doxorubicin. The micelles preferentially accumulated at the tumor site in an in vivo mouse xenograft of human ovarian carcinoma A2780, which led to a high dose of drug in the solid tumor and high treatment efficacy.124 The same group later designed a similar micelle, modified with folate allowing for receptor-mediated endocytosis and improved drug uptake. In mice, these targeted mixed micelles inhibited the growth of multidrug-resistant tumors with minimal weight loss to the animal.125 Tuning the pH sensitivity to the early endosomal pH range of 6.0 provided for an even higher activity, suppressing tumor growth in mice for at least 50 days.126
B. Novel Therapies
As described above, encapsulation and targeting of chemotherapy agents can significantly improve the response of tumors to chemotherapeutics. These nanotechnology-based therapeutic strategies still largely depend on delivering agents that are considered standard therapy for ovarian cancer. This is a natural progression, since it improves the efficacy of drugs that have already been FDA approved for use in ovarian cancer. However, following several treatment courses, patients often develop chemoresistance to these agents, which is particularly prevalent with platinum-based therapies. Below, we describe nanotechnology-based therapeutic strategies that may provide valid alternatives to chemotherapeutics in the treatment of ovarian cancer.
1. Photodynamic Therapy (PDT)
PDT is a treatment modality involving the interaction of light, a photosensitizer (PS), and tissue oxygen to generate cytotoxic reactive oxygen species that cause cell necrosis and/or apoptosis. It is a two-step process that requires administration of the photosensitizer, usually by injection, where after 6 to 24 h the tumor site is illuminated with light to activate accumulated PS.127 In the United States, PDT was first approved by the FDA in 1995 for the treatment of esophageal cancer using Photofrin (a porphyrin-based PS).128 Since then, clinical use of PDT has been expanded to several cancer types and other PS compounds.129 Some of the advantages of PDT include the following: it has the low toxicities of photosensitizing compounds compared with chemotherapy drugs, it is a minimally invasive technique, it uses non-ionizing radiation at relatively low energy doses, and it offers localized treatment (i.e., where light is administered). The mild conditions enable PDT to be used for repeated treatments with reduced risk of side effects and damaging of nearby tissue. Challenges associated with PDT include tissue penetration; thus, similar to optical imaging, photosensitizers activated by red or NIR wavelengths are preferred to improve the efficacy. Nonetheless, the delivery of light to deeper-laying tissue has prevented widespread use. Light administration using catheters and fiber optics offers a possible solution where innovations such as radial emitting fiber optics may play a role.130 Even though PSs tend to have less systemic toxicity than chemotherapeutics, one challenge is their inherent lack of tissue specificity. Photofrin, for example, has been shown to accumulate preferentially in tumors, yet it has a very low clearance rate, resulting in accumulation in other tissues, which can lead to photosensitivity. Patients treated with Photofrin must avoid direct sunlight for up to six weeks after treatment since residual photosensitizer found in skin and eye tissue can become photoactivated and generate unwanted cytotoxicity in healthy tissues.131 Also, many potential PS agents are hydrophobic and significant effort has been placed into the development of suitable nanoparticle delivery vehicles for PDT. See the review by Master et al. for examples of photodynamic therapy using nanoparticles.132
Although PDT has mainly been used for easy accessible tumors, the interest of nanoparticle-mediated PDT for ovarian cancers is gaining traction. An early demonstration was the use of hypericin-loaded polylactic-acid nanoparticles to demonstrate photodynamic treatment of ovarian cancer cells in vitro.133 This was later followed up by an in vivo study using the particles in an epithelial ovarian cancer cell line (NuTu-19) in rats with improved accumulation in micrometastases compared with free drug.134 More recently, a magnetically targeted PEG nanoformulation of a chlorine PS (Ce6) has been demonstrated to have therapeutic efficiency for ovarian cancer in a murine model.135 The increase in penetration depth using NIR light and efficient PS agents, such as Ce6, may have potential for ovarian cancer treatment, considering one challenge to PDT is successful irradiation within the IP cavity. A demonstration of NIR in combination with upconverting nanoparticles, co-administered with Ce6, was demonstrated in a breast cancer mouse model and showed increased tissue penetration with good clearance without noticeable toxicity.136
Targeted novel nanoparticles for PDT are also being developed. For example, virus capsid encapsulating a porphyrin PS, and surface modified with a nucleolin-targeting aptamer has been demonstrated to specifically target breast cancer cells in vitro.137 Since the nucleolin receptor is overexpressed in many ovarian cancer cell lines, similar targeting systems can be envisioned. Nanoparticles may be used not only as carriers for photosensitive agents, but also as agents themselves. Functionalized fullerene, a carbon nanocage, has the ability to generate reactive oxygen species on illumination by visible light. Such particles were demonstrated for IP PDT using a colon adenocarcinoma mouse model.138 Fullerenes have also shown efficacy as PS by both intravenous injection and local exposure against fibrosarcoma and bacterial infection.139,140
Although PDT has to date mainly been used for melanoma and other cancers that are easily accessible to irradiate by light, the development of targeted nanoparticle delivery systems and improved photosensitizers and light administration techniques provide opportunities for PDT in ovarian cancer therapy in the future.
2. Gene Therapy
Small-molecule therapy has its limitations largely due to nonspecific side effects and associated toxicity. With increasing knowledge of tumor-specific gene expression signatures, there is a push to specifically target single proteins, with the hope of altering cancer signaling pathways in a more controlled manner. Specifically, delivering vectors for targeted expression of tumor suppressors or silencing expression of oncogenes by RNA interference (RNAi) are avenues that many investigators are pursuing for potential anticancer therapy. There are many caveats associated with direct systemic administration of genetic material. For example, in the case of small interfering (siRNA), free oligonucleotides are very unstable, easily degraded, unable to enter the cell efficiently, and may be associated with toxic and immune-stimulatory side effects.141 Vector-based delivery of genetic material is associated with similar caveats, which have been partly addressed by the development of adenoviral gene delivery. The use of nanoparticle systems for oligonucleotide and plasmid delivery has proven to be an effective and potentially superior alternative. A number of different chemistries and structures have been explored for this purpose. As with drug delivery, these are chosen largely based on biocompatibility, and are charge modified (cationic) to enable effective nucleotide binding. 142–144 Similar to targeted nanoparticle approaches for chemotherapeutic delivery, many gene delivery techniques are utilizing functionalized nanoparticles to specifically reach ovarian tumor cells.145 There has been a push toward clinical translation and a number of nanoparticle systems are currently being developed for delivery of plasmids and siRNA, with toxicity and bioavailability studies being carried out in clinical trials.
a. Expression Vector Delivery of Tumor Suppressors
A major focus of DNA plasmid delivery to cancer cells is the forced expression of tumor suppressor genes (see summary in Table 5). Given the high frequency of TP53 mutations observed in a number of cancers, this tumor repressor gene is one of the major targets for nanoparticle-based gene delivery currently proceeding into clinical trials. Of note is a recent phase I clinical trial on “SGT-53,” which is an antitransferrin receptor scFV antibody conjugated to liposomal nanocarriers for TP53 gene delivery. This study showed encouraging results, demonstrating stabilization of the disease in patients with advanced solid tumors of different origin and a minimal toxicity profile.146 Since TP53 mutations are observed in almost 100% of high-grade serous ovarian cancers, targeting this molecular pathway by gene therapy approaches may prove beneficial in this population. This was first investigated in clinical trials in the late 1990s using adenoviral delivery of the TP53 gene (Ad-p53). Unfortunately, adenoviral-based gene therapy offered little benefit to patients and this failure was in part attributed to the fact that the virus was recognized by patient antibodies and was unable to efficiently reach the tumors.147 Utilizing a nanoparticle formulation for TP53 gene delivery may provide more advantageous outcomes. However, thus far no clinical trials are underway to test SGT-53 on high-grade serous ovarian cancer. Few clinical trials have specifically focused on nanoparticle delivery of genetic material for ovarian cancer. In the early 2000s, IP delivery of liposomal human adenovirus type 5 early region 1A (E1A), which downregulates HER2, was studied in phase I and II trials in combination treatment with IV administered paclitaxel on platinum-resistant ovarian cancer patients, with a low reported toxicity profile. However, long-term follow-up of this study has not been reported.148 A recently published phase I clinical study utilized a PEG-poly(ethylene imine) (PEI)-cholesterol lipopolymer for IP delivery of a plasmid encoding for interleukin 12 (IL-12; “EGEN-001”), with the intent of eliciting a localized immune response against the tumor in ovarian cancer patients cotreated with IV carboplatin. All patients displayed tolerable abdominal toxicity to the plasmid and responded favorably with enhanced interferon-γ and tumor necrosis factor-a production, two cytokines that are stimulated by interleukin-12.149 Patients with persistent or recurrent ovarian cancer are currently being recruited for phase II clinical trials for treatment with IP EGEN-001 (NCT01118052) or in combination with IP Doxil (NCT01489371).
TABLE 5.
Examples of nanoparticle-based gene therapy approaches for ovarian cancer
| Gene target Tumor suppressors | NP composition | Target | Refs | |
|---|---|---|---|---|
| Human adenovirus type 5 early region 1A (E1A) Trial drug: “tgDCC-E1” |
Plasmid vector | liposome | — | Phase I and II: NCT00102622, 148, 208 |
| Plasmid vector | PEG-PEI-cholesterol Lipopolymer | — | Phase I: NCT01489371 Phase I: NCT00473954 Phase II: NCT01118052, 149 |
|
| HSulf-1 | Plasmid vector | Heparin-polyethyleneimine nanogels | — | Xenograft, 150 |
| FILIP1L | Plasmid vector | Heparin-polyethyleneimine nanogels | — | Xenograft, 151 |
| Diphtheria toxin | Plasmid vector (ovarian specific HE4 and MSLN promoters) | Poly(β-amino ester) polymer-DNA complex | — | Xenograft, 209 |
|
Oncogenes HIF1α |
siRNA |
Liposome |
— |
Xenograft, 210 |
| PARP1 | siRNA | Liposome | — | Xenograft, 157 |
| Src | siRNA | Chitosan | — | Xenograft, 154 |
| Claudin-3 | Codelivery of both shRNA plasmid vectors | PLGA nanoparticles | — | Xenograft, 155 |
| CD44 and FAK | Codelivery of both shRNA plasmid vectors | PLGA nanoparticles | — | Xenograft, 156 |
| EphA2 Trial Drug: “siRNA-EphA2-DOPC” |
siRNA | Mesoporous silicon loaded with DOPC liposomes | — | Xenograft, 159, 164 Phase I: NCT01591356 |
| EphA2 miR-520d-3p (targets EphB2) |
siRNA with codelivery of miRNA | mesoporous silicon loaded with DOPC liposomes | — | Xenograft, 158 |
| Jagged1 | siRNA | — | Xenograft, 169 | |
| EGFR | siRNA | EphA2 | Cell lines, 165 | |
| STAT3 and FAK | siRNA | HDL nanoparticles | SR-B1 | Xenograft, 166 |
| ID4 | siRNA | Peptide nanocomplex | Neuropilin-1 | Xenograft, 150 |
| PLXDC1 | siRNA | Chitosan | Integrin | Xenograft, 168 |
| — | Non-targeting siRNA | Polyethylenimine nanoparticles | TLR5 | Xenograft, 170, 171 |
In a number of preclinical studies, nanoparticles have been utilized to deliver expression vectors of tumor suppressor genes with hope of eliciting antitumor activity following successful uptake and expression by ovarian cancer cells. Examples include heparin-polyethyleneimine nanogels delivering DNA plasmid for expression of heparin sulfate 6-O-endosulfatase 1 (HSulf-1)150 and filamin A interacting protein 1-like (FILIP1L).151 Expression of these proteins is often downregulated in ovarian cancer. IP injection of nanoparticle-containing expression plasmids for these genes effectively reduced ovarian cancer xenografts in nude mice, inducing significant apoptosis, decreasing proliferation and halting angiogenesis in the tumors. The effect of inducing HSulf-1 expression was further enhanced by cotreatment with cisplatin.150
b. Inhibition of Oncogenes by RNAi
Nanoparticle formulations have proved to be effective methods to deliver small interfering (siRNA) or short hairpin RNA (shRNA) to cancer cells with the aim of silencing gene expression of oncogenes. The first clinical trial on nanoparticle-based siRNA delivery was published in 2010, showing effective knockdown of the M2 subunit of ribonucleotide reductase (RRM2) in melanoma patient tumors following IV administration [RNAi/oligonucleotide nanoparticle delivery (RONDEL)].152 This PEGylated cyclodextrin-based polymer nanoparticle formulation (Cyclosert) incorporates transferrin protein to target the transferrin receptor. This trial revealed that siRNA could effectively be targeted to tumor tissue and affect RRM2 expression. Similarly, liposomal nanocarriers incorporating siRNA toward VEGF and kinesin spindle protein (ALN-VSP) showed promising results in a phase II trial with high tumor targeting, as assessed by protein expression knockdown, low toxicity profiles, and reports of regression of liver metastases in patients with endometrial cancer.153
Although no specific RNAi nanoparticle delivery strategies are currently in clinical trials for ovarian cancer, several promising preclinical studies make the case for developing nanoparticle formulations for RNAi-based therapy in ovarian cancer. A list of example studies is shown in Table 5; nanoparticle RNAi delivery has resulted in effective gene knockdown of HIF1α, PARP1, Claudin-3, Src, CD44, and focal adhesion kinase, resulting in significant decreases in tumor burden in ovarian cancer xenograft models.154–157
Several studies from MD Anderson Cancer Center have focused on the tyrosine kinase ephrin receptor EphA2,158,159 which is observed to be highly expressed in ovarian cancer and associated with increased angiogenesis and metastatic invasion.160–162 Nanoparticle-mediated siRNA delivery to knock down EphA2 expression showed significant reduction in in vivo tumor burden in mice ovarian cancer xenografts.158,159,163 One example is the use of mesoporous silicon particles loaded with EphA2 siRNA-containing dioleoyl phosphatidylcholine (DOPC) liposomes.164 This formulation allows for slow release of the liposomal compartment from the silicon particle and enable sustained delivery of the siRNA liposome into cells, leading to prolonged knockdown of EphA2. This knockdown also enhanced tumor sensitivity to docetaxel in chemotherapy-resistant HeyA8 ovarian tumors, and prevented tumor growth in animals treated with both siRNA-nanoparticle and docetaxel.159 A phase I study is due to be initiated at MD Anderson Cancer Center to test the safety of this nanoparticle formulation via the IV route on patients with advanced, recurrent cancers, including ovarian cancer (NCT01591356). Recently, the same group reported improved antitumor efficacy in xenografts when the microRNA miR-520d-3p was codelivered with EphA2 siRNA in the DOPC liposomes/mesoporous silicon nanoparticles. miR-520d-3p expression is associated with favorable outcome in ovarian cancer patients and its target is another ephrin receptor, EphB2.158
To improve ovarian cancer cell targeting, Dickerson et al. developed an EGFR siRNA loaded hydrogel nanoparticle, functionalized with a peptide that binds EphA2 receptor. The hydrogel particles efficiently decreased EGFR expression and enhanced sensitivity to doxetacel in EpHA2-positive cell lines.165 Even though EGFR targeting by small molecules has not shown any benefit for ovarian cancer in clinical trials, functionalizing nanoparticles with EphA2-specific peptides may enhance nanoparticle localization to ovarian cancer cells and improve the modulation of EGF signaling. In a similar approach, Shahzad et al. took advantage of the high expression of the scavenger receptor type B1 (SR-B1) on tumor cells, which binds high-density lipoprotein to maintain rapid growth. Encapsulating siRNA against protumorigenic genes STAT3 (signal transducer and activator of transcription 3) and focal adhesion kinase in reconstituted high-density lipoprotein nanoparticles, the investigators found enhanced tumor targeting and silencing of protein expression in orthotopic mouse models of ovarian and colon cancer.166
Recently, Ren et al. identified the transcriptional regulator ID4 (inhibitor of DNA binding 4) as an important regulator of ovarian cancer proliferation.150 To decrease expression of ID4, the investigators electrostatically bound siRNA against ID4 using a tandem peptide sequence. The peptide incorporates an arginine domain for siRNA binding and the cyclic nanopeptide LyP-1 (CGNKRTRGC) as the tumor-penetrating domain. This approach is based on the iRGD peptide that has been demonstrated to enhance vascular penetration and specifically target tumors in a neuropilin-1 dependent manner.167 IV injections of the ID4siRNA nanocomplex were highly effective at decreasing subcutaneous tumor xenografts, and IP treatment was able to abolish tumor growth in an orthotopic xenograft model of ovarian cancer.150 Similarly, arginylglycylaspartic acid (RGD) peptide linkages in combination with chitosan nanoparticles have been used to target integrin receptors to specifically deliver siRNA against multiple genes important in ovarian cancer proliferation and shown to decrease tumor growth in ovarian cancer mouse xenograft models.168
Nanoparticle-based gene therapy has also been exploited to influence the tumor microenvironment. For instance, chitosan was used to deliver siRNA against the Notch ligand Jagged1, expression of which is increased in some ovarian tumor cells and in the tumor microenvironment. Investigators showed that IV-administered chitosan nanoparticles were able to downregulate both human tumor cell–derived and murine stromal-derived Jagged1 expression, effectively decreasing proliferation of the xenografts and tumor microenvironment–derived angiogenesis, respectively.169 Similarly, Cubillos-Ruiz et al. took advantage of the preferential uptake of polyethylenimine-based (PEI-based) nanoparticles by dendritic cells in the tumor microenvironment. The particles were loaded with nontargeting siRNA sequences that elicited an immune response via toll-like receptor 5, effectively stimulating immune system–mediated clearance of ovarian cancer xenografts in vivo.170,171
Most nanoparticle gene/RNAi delivery strategies developed for therapy are only just moving through phase I and II clinical trials, and their validity as novel therapeutic strategies against ovarian cancer are eagerly anticipated as these trials mature. The moderate toxicity profile and the high efficacy in preclinical animal studies are very encouraging. The infinite flexibility of nanoparticle chemistries will allow for further enhancement of particle designs, such as functionalization and combining coloading of siRNA/plasmid DNA with chemotherapeutic agents to enhance targeted killing of tumor cells.
IV. EMERGING THERAPIES AND FUTURE DIRECTIONS
Throughout this review we have mentioned drug administration to the IP cavity and would like to discuss this strategy in more detail. The relatively confined anatomical localization of ovarian cancer and its metastatic lesions within the IP cavity make this route attractive for delivery of therapeutics in a localized confined manner. Clinicians have administered standard chemotherapies directly via the IP route for decades and demonstrated a therapeutic benefit.28,29 However, it is still debated whether or not IP therapy significantly reduces systemic side effects compared with the IV route, since patients can still exhibit systemic toxicity on IP administration. This is likely due to the leakiness in vasculature observed in higher-stage cancers, which is also associated with significant ascites fluid build-up within the abdomen. However, nanoparticle delivery techniques may improve the toxicity profile via the IP route. Early studies on IP delivery of Doxil showed promising results and a decreased systemic toxicity profile.172 The intrinsic nature of nanoparticles to preferentially localize in tumor tissues and cells within the tumor microenvironment may help decrease systemic toxicity following IP administration. A recent proof of concept study has argued for direct IP administration of nanoparticle-encapsulated chemotherapies (paclitaxel-loaded expansile nanoparticles) at the time of cytoreductive surgery, and has shown improved outcome in ovarian cancer xenograft studies.173 IP administration may also be advantageous for effective folate receptor targeting to ovarian cancer in vivo, since a recent report suggests that IV administration of folate-conjugated polycationic amphiphilic cyclodextrin-DNA nanocomplexes accumulate in lung and liver rather than in the targeted tissue.170 A combination of IP administration and targeting of nanoparticles by functionalization with antibodies, peptides, and aptamers to tumor-specific proteins may aid in decreasing systemic side effects and enhance the payload of delivered chemotherapeutic (or alternate therapy) to the tumors. Researchers developing nanoparticle formulations for ovarian cancer therapeutics are increasingly taking the IP delivery route into consideration.
Beyond their use as drug carriers and imaging agents, an interesting prospect for nanomaterials in cancer therapy is their role as a “drug” themselves. One example is nanocrystalline cerium oxide particles (nanoceria), which can change between oxidation states III and IV depending on environment, and have been compared with biological antioxidants.174,175 Nanoceria particles were recently administrated IP in a ovarian cancer model and shown to reduce angiogenesis at doses as low as 0.1 mg/kg.176 The therapeutic effect behind the nanoceria was not made clear, but the particles inhibited VEGF-induced proliferation, capillary tube formation, and matrix metalloproteinase-2 (MMP2) activation. The authors hinted that nanoceria could be involved in modulation of VEGF-mediated, redox-sensitive signaling events. High doses of nanoceria have been administered without noticeable toxicity (up to 250 mg/kg), which make these nanoparticle alternatives to potentially toxic, small-molecule antiangiogenic agents.177–179 Other examples include iron oxide nanoparticles. A recent study showed that IP-administered iron oxide could effectively kill tumor cells in a murine ovarian cancer xenograft model by hyperthermia, following application of an alternating magnetic field.180 Interestingly, rather than being directly taken up by the cancer cells, the authors present evidence that peritoneal phagocytes engulf and deliver the iron oxide particles to the tumor tissue. This result implies that nanoparticles administered via IP injection can also localize to the tumor environment regardless of surface coating or targeting moieties, and warrant further investigation. Nanoparticles also have the ability to deliver alternate cargo beyond standard chemotherapies, photosensitizers, and gene therapy constructs. Examples include the delivery of radio-isotopes for radionuclide therapy,181 delivery of nitric oxide donors for eradication of cancer cells,182 and using the intrinsic properties of nanoparticles for drug release, such as near-infrared triggered doxorubicin release from hollow gold nanoparticles.183
As is evident from the recent articles and clinical trials discussed above, nanoparticle systems are gaining traction in ovarian cancer therapeutics. It is clear that nanoparticle-based technology will be an integral part of improving tumor targeting, drug efficacy, and toxicity profiles of new emerging strategies. While current translational efforts are on nontargeted liposome or polymer aggregate formulations, recent findings using targeted nanomaterials and codelivery systems show therapeutic potential and are likely to enter clinical trials in the near future. Likewise, novel treatment strategies based on nanoparticle delivery systems, such as RNAi and gene therapy, herald their use in forthcoming ovarian cancer therapies.
Acknowledgments
Nadine Hempel recognizes support from the National Cancer Institute under NIH Grant No. R00CA143229.
ABBREVIATIONS
- CA 125
serum cancer antigen 125
- DNA
deoxyribonucleic acid
- DOPC
dioleoyl phosphatidylcholine
- EGFR
epidermal growth factor receptor
- EOC
epithelial ovarian cancer
- EPR
enhanced permeability and retention
- FDA
Food and Drug Administration
- HE4
human epididymis protein 4
- HER2
human epidermal growth factor receptor 2
- HPMA
N-(2-hydroxypropyl)methacrylamide
- IP
intraperitoneal
- IV
intravenous
- MRI
magnetic resonance imaging
- NIR
near-infrared
- PA
photoacoustic
- PAMAM
polyamidoamine
- PARP
poly ADP ribose polymerase
- PDT
photodynamic therapy
- PEG
polyethylene glycol
- PLGA
poly(d,l-lactice-co-glycolide)
- PS
photosensitizer
- QDs
quantum dots
- RNA
ribonucleic acid
- RNAi
RNA interference
- siRNA
small interfering RNA
- SPECT
single-photon emission computed tomography
- US
ultrasound
- VEGF
vascular endothelial growth factor
References
- 1.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. doi: 10.3322/caac.21166. [DOI] [PubMed] [Google Scholar]
- 2.Vaughan S, Coward JI, Bast RC, Jr, Berchuck A, Berek JS, Brenton JD, Coukos G, Crum CC, Drapkin R, Etemadmoghadam D, Friedlander M, Gabra H, Kaye SB, Lord CJ, Lengyel E, Levine DA, McNeish IA, Menon U, Mills GB, Nephew KP, Oza AM, Sood AK, Stonach EA, Walczak H, Bowtell DD, Balkwill FR. Rethinking ovarian cancer: recommendations for improving outcomes. Nat Rev Cancer. 2011;11:719–25. doi: 10.1038/nrc3144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cancer Genome Atlas Research Network. Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609–15. doi: 10.1038/nature10166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Berns EM, Bowtell DD. The changing view of high-grade serous ovarian cancer. Cancer Res. 2012;72:2701–4. doi: 10.1158/0008-5472.CAN-11-3911. [DOI] [PubMed] [Google Scholar]
- 5.Kurman RJ, Shih Ie M. Molecular pathogenesis and extraovarian origin of epithelial ovarian cancer—shifting the paradigm. Hum Pathol. 2011;42:918–31. doi: 10.1016/j.humpath.2011.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Prat J. Ovarian carcinomas: five distinct diseases with different origins, genetic alterations, and clinicopathological features. Virchows Archiv. 2012;460:237–49. doi: 10.1007/s00428-012-1203-5. [DOI] [PubMed] [Google Scholar]
- 7.Banerjee S, Kaye SB. New strategies in the treatment of ovarian cancer: current clinical perspectives and future potential. Clin Cancer Res. 2013;19:961–8. doi: 10.1158/1078-0432.CCR-12-2243. [DOI] [PubMed] [Google Scholar]
- 8.Jelovac D, Armstrong DK. Recent progress in the diagnosis and treatment of ovarian cancer. CA Cancer J Clin. 2011;61:183–203. doi: 10.3322/caac.20113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Piek JM, Verheijen RH, Kenemans P, Massuger LF, Bulten H, van Diest PJ. BRCA1/2-related ovarian cancers are of tubal origin: a hypothesis. Gynecol Oncol. 2003;90:491. doi: 10.1016/s0090-8258(03)00365-2. [DOI] [PubMed] [Google Scholar]
- 10.Tan DS, Agarwal R, Kaye SB. Mechanisms of transcoelomic metastasis in ovarian cancer. Lancet Oncol. 2006;7:925–34. doi: 10.1016/S1470-2045(06)70939-1. [DOI] [PubMed] [Google Scholar]
- 11.Landen CN, Jr, Birrer MJ, Sood AK. Early events in the pathogenesis of epithelial ovarian cancer. J Clin Oncol. 2008;26:995–1005. doi: 10.1200/JCO.2006.07.9970. [DOI] [PubMed] [Google Scholar]
- 12.Davidson B. Ovarian carcinoma and serous effusions. changing views regarding tumor progression and review of current literature. Anal Cell Pathol. 2001;23:107–28. doi: 10.1155/2001/418547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bast RC, Jr, Feeney M, Lazarus H, Nadler LM, Colvin RB, Knapp RC. Reactivity of a monoclonal antibody with human ovarian carcinoma. J Clin Invest. 1981;68:1331–7. doi: 10.1172/JCI110380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Buys SS, Partridge E, Black A, Johnson CC, Lamerato L, Isaacs C, Reding DJ, Greenlee RT, Yokochi LA, Kessel B, Crawford ED, Church TR, Andriole GL, Weissfeld JL, Fouad MN, Chia D, O’Brien B, Ragard LR, Clapp JD, Rathmell JM, Riley TL, Hartge P, Pinsky PF, Zhu CS, Izmirlian G, Kramer BS, Miller AB, Xu JL, Prorok PC, Gohagan JK, Berg CD PLCO Project Team. Effect of screening on ovarian cancer mortality: the Prostate, Lung, Colorectal and Ovarian (PLCO) Cancer Screening Randomized Controlled Trial. JAMA: the journal of the American Medical Association. 2011;305:2295–303. doi: 10.1001/jama.2011.766. [DOI] [PubMed] [Google Scholar]
- 15.Husseinzadeh N. Status of tumor markers in epithelial ovarian cancer has there been any progress? a review. Gynecol Oncol. 2011;120:152–7. doi: 10.1016/j.ygyno.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 16.Qian Y, Wang Y, Zhang X, Zhou L, Zhang Z, Xu J, Ruan Y, Ren S, Xu C, Gu J. Quantitative analysis of serum IgG galactosylation assists differential diagnosis of ovarian cancer. J Proteome Res. 2013;12:4046–55. doi: 10.1021/pr4003992. [DOI] [PubMed] [Google Scholar]
- 17.Li B, An HJ, Kirmiz C, Lebrilla CB, Lam KS, Miyamoto S. Glycoproteomic analyses of ovarian cancer cell lines and sera from ovarian cancer patients show distinct glycosylation changes in individual proteins. J Proteome Res. 2008;7:3776–88. doi: 10.1021/pr800297u. [DOI] [PubMed] [Google Scholar]
- 18.Saldova R, Royle L, Radcliffe CM, Abd Hamid UM, Evans R, Arnold JN, Banks RE, Hutson R, Harvey DJ, Antrobus R, Petrescu SM, Dwek RA, Rudd PM. Ovarian cancer is associated with changes in glycosylation in both acute-phase proteins and IgG. Glycobiology. 2007;17:1344–56. doi: 10.1093/glycob/cwm100. [DOI] [PubMed] [Google Scholar]
- 19.Biskup K, Braicu EI, Sehouli J, Fotopoulou C, Tauber R, Berger M, Blanchard V. Serum glycome profiling: a biomarker for diagnosis of ovarian cancer. J Proteome Res. 2013;12:4056–63. doi: 10.1021/pr400405x. [DOI] [PubMed] [Google Scholar]
- 20.Nolen BM, Lokshin AE. Biomarker testing for ovarian cancer: clinical utility of multiplex assays. Mol Diagn Ther. 2013;17:139–46. doi: 10.1007/s40291-013-0027-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Moore RG, McMeekin DS, Brown AK, DiSilvestro P, Miller MC, Allard WJ, Gajewski W, Kurman R, Bast RC, Jr, Skates SJ. A novel multiple marker bioassay utilizing HE4 and CA125 for the prediction of ovarian cancer in patients with a pelvic mass. Gynecol Oncol. 2009;112:40–6. doi: 10.1016/j.ygyno.2008.08.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jacob F, Meier M, Caduff R, Goldstein D, Pochechueva T, Hacker N, Fink D, Heizelmann-Schwarz V. No benefit from combining HE4 and CA125 as ovarian tumor markers in a clinical setting. Gynecol Oncol. 2011;121:487–91. doi: 10.1016/j.ygyno.2011.02.022. [DOI] [PubMed] [Google Scholar]
- 23.Montagnana M, Danese E, Ruzzenente O, Bresciani V, Nuzzo T, Gelati M, Salvagno GL, Franchi M, Lippi G, Guidi GC. The ROMA (risk of ovarian malignancy algorithm) for estimating the risk of epithelial ovarian cancer in women presenting with pelvic mass: is it really useful? Clin Chem Lab Med. 2011;49:521–5. doi: 10.1515/CCLM.2011.075. [DOI] [PubMed] [Google Scholar]
- 24.Zhang Z, Chan DW. The road from discovery to clinical diagnostics: lessons learned from the first FDA-cleared in vitro diagnostic multivariate index assay of proteomic biomarkers. Cancer Epidemiol Biomarkers Prev. 2010;19:2995–9. doi: 10.1158/1055-9965.EPI-10-0580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Moore LE, Pfeiffer RM, Zhang Z, Lu KH, Fung ET, Bast RC., Jr Proteomic biomarkers in combination with CA 125 for detection of epithelial ovarian cancer using prediagnostic serum samples from the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial. Cancer. 2012;118:91–100. doi: 10.1002/cncr.26241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sarojini S, Tamir A, Lim H, Li S, Zhang S, Goy A, Pecora A, Suh KS. Early detection biomarkers for ovarian cancer. J Oncol. 2012;2012:709049. doi: 10.1155/2012/709049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Scarberry KE, Dickerson EB, McDonald JF, Zhang ZJ. Magnetic nanoparticle-peptide conjugates for in vitro and in vivo targeting and extraction of cancer cells. J Am Chem Soc. 2008;130:10258–62. doi: 10.1021/ja801969b. [DOI] [PubMed] [Google Scholar]
- 28.Armstrong DK, Bundy B, Wenzel L, Huang HQ, Baergen R, Lele S, Copeland LJ, Walker JL, Burger RA Gyencologic Oncology Group. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. New Engl J Med. 2006;354:34–43. doi: 10.1056/NEJMoa052985. [DOI] [PubMed] [Google Scholar]
- 29.Tewari D, Java J, Salari D, Armstrong M, Markman K, Herzog T, Monk B, Chan J. Long-term survival advantage of intraperitoneal chemotherapy treatment in advanced ovarian cancer: an analysis of a Gynecologic Oncology Group ancillary data study (abstract GOG114/172). SGO Annual Meeting on Women’s Cancer; Los Angeles. 2013. [Google Scholar]
- 30.Jaaback K, Johnson N, Lawrie TA. Intraperitoneal chemotherapy for the initial management of primary epithelial ovarian cancer. Cochrane Database Syst Rev. 2011:CD005340. doi: 10.1002/14651858.CD005340.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rescigno P, Cerillo I, Ruocco R, Condello C, De Placido S, Pensabene M. New hypothesis on pathogenesis of ovarian cancer lead to future tailored approaches. BioMed Res Int. 2013;2013:852839. doi: 10.1155/2013/852839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ledermann J, Harter P, Gourley C, Friedlander M, Vergote I, Rustin G, Scott C, Meier W, Shapira-Frommer R, Safra T, Matei D, Macpherson E, Watkins C, Carmichael J, Matulonis U. Olaparib maintenance therapy in platinum-sensitive relapsed ovarian cancer. New Engl J Med. 2012;366:1382–92. doi: 10.1056/NEJMoa1105535. [DOI] [PubMed] [Google Scholar]
- 33.Farley J, Brady WE, Vathipadiekal V, Lankes HA, Coleman R, Morgan MA, Mannel R, Yamada SD, Mutch D, Rodgers WH, Birrer M, Gersherson DM. Selumetinib in women with recurrent low-grade serous carcinoma of the ovary or peritoneum: an open-label, single-arm, phase 2 study. Lancet Oncol. 2013;14:134–40. doi: 10.1016/S1470-2045(12)70572-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bookman MA, Darcy KM, Clarke-Pearson D, Boothby RA, Horowitz IR. Evaluation of monoclonal humanized anti-HER2 antibody, trastuzumab, in patients with recurrent or refractory ovarian or primary peritoneal carcinoma with overexpression of HER2: a phase II trial of the Gynecologic Oncology Group. J Clin Oncol. 2003;21:283–90. doi: 10.1200/JCO.2003.10.104. [DOI] [PubMed] [Google Scholar]
- 35.Burger RA, Brady MF, Bookman MA, Fleming GF, Monk BJ, Huang H, Mannel RS, Homesley HD, Fowler J, Greer BE, Boente M, Birrer MJ, Liang SX Gynecologic Oncology Group. Incorporation of bevacizumab in the primary treatment of ovarian cancer. New Engl J Med. 2011;365:2473–83. doi: 10.1056/NEJMoa1104390. [DOI] [PubMed] [Google Scholar]
- 36.Perren TJ, Swart AM, Pfisterer J, Ledermann JA, Pujade-Lauraine E, Kristensen G, Carey MS, Beale P, Cervantes A, Kurzeder C, du Bois A, Sehouli J, Kimmig R, Stähle A, Collinson F, Essapen S, Gourley C, Lortholary A, Selle F, Mirza MR, Leminen A, Plante M, Stark D, Qian W, Parmar MK, Oza AM ICON7 Investigators. A phase 3 trial of bevacizumab in ovarian cancer. New Engl J Med. 2011;365:2484–96. doi: 10.1056/NEJMoa1103799. [DOI] [PubMed] [Google Scholar]
- 37.Miller DS, Blessing JA, Krasner CN, Mannel RS, Hanjani P, Pearl ML, Waggoner SE, Boardman CH. Phase II evaluation of pemetrexed in the treatment of recurrent or persistent platinum-resistant ovarian or primary peritoneal carcinoma: a study of the Gynecologic Oncology Group. J Clin Oncol. 2009;27:2686–91. doi: 10.1200/JCO.2008.19.2963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Vergote I, Calvert H, Kania M, Kaiser C, Zimmermann AH, Sehouli J. A randomised, double-blind, phase II study of two doses of pemetrexed in the treatment of platinum-resistant, epithelial ovarian or primary peritoneal cancer. Eur J Cancer. 2009;45:1415–23. doi: 10.1016/j.ejca.2008.12.013. [DOI] [PubMed] [Google Scholar]
- 39.Sarojini S, Tamir A, Lim H, Li S, Zhang S, Goy A, Pecora A, Suh KS. Early detection biomarkers for ovarian cancer. J Oncol. 2012;2012:709049. doi: 10.1155/2012/709049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kimmel DW, LeBlanc G, Meschievitz ME, Cliffel DE. Electrochemical sensors and biosensors. Anal Chem. 2011;84:685–707. doi: 10.1021/ac202878q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Perfezou M, Turner A, Merkoci A. Cancer detection using nanoparticle-based sensors. Chem Soc Rev. 2012;41:2606–22. doi: 10.1039/c1cs15134g. [DOI] [PubMed] [Google Scholar]
- 42.Swierczewska M, Liu G, Lee S, Chen X. High-sensitivity nanosensors for biomarker detection. Chem Soc Rev. 2012;41:2641–55. doi: 10.1039/c1cs15238f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bellan LM, Wu D, Langer RS. Current trends in nanobiosensor technology. Rev Nanomed Nanobiotechnol. 2011;3:229–46. doi: 10.1002/wnan.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jokerst JV, Raamanathan A, Christodoulides N, Floriano PN, Pollard AA, Simmons GW, Wong J, Gage C, Furmaga WB, Redding SW, McDevitt JT. Nano-bio-chips for high performance multiplexed protein detection: determinations of cancer biomarkers in serum and saliva using quantum dot bioconjugate labels. Biosens Bioelectron. 2009;24:3622–9. doi: 10.1016/j.bios.2009.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Raamanathan A, Simmons GW, Christodoulides N, Floriano PN, Furmaga WB, Redding SW, Lu KH, Bast RC, Jr, McDevitt JT. Programmable bio-nano-chip systems for serum CA125 quantification: toward ovarian cancer diagnostics at the point-of-care. Cancer Prev Res (Phila) 2012;5:706–16. doi: 10.1158/1940-6207.CAPR-11-0508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ravalli A, dos Santos GP, Ferroni M, Faglia G, Yamanaka H, Marrazza G. New label free CA125 detection based on gold nanostructured screen-printed electrode. Sens Actuators B. 2013;179:194–200. [Google Scholar]
- 47.Li J, Xu Q, Fu C, Zhang Y. A dramatically enhanced electrochemiluminescence assay for CA125 based on dendrimer multiply labeled luminol on Fe3O4 nanoparticles. Sens Actuators B. 2013;185:146–53. [Google Scholar]
- 48.Hong H, Zhang Y, Sun J, Cai W. Molecular imaging and therapy of cancer with radiolabeled nanoparticles. Nano Today. 2009;4:399–413. doi: 10.1016/j.nantod.2009.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hahn MA, Singh AK, Sharma P, Brown SC, Moudgil BM. Nanoparticles as contrast agents for in-vivo bioimaging: current status and future perspectives. Anal Bioanal Chem. 2011;399:3–27. doi: 10.1007/s00216-010-4207-5. [DOI] [PubMed] [Google Scholar]
- 50.Wu X, Wu M, Zhao JX. Recent development of silica nanoparticles as delivery vectors for cancer imaging and therapy. Nanomedicine. 2014;10:297–312. doi: 10.1016/j.nano.2013.08.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Maldonado CR, Salassa L, Gomez-Blanco N, Mareque-Rivas JC. Nano-functionalization of metal complexes for molecular imaging and anticancer therapy. Coord Chem Rev. 2013;257:2668–88. [Google Scholar]
- 52.Petersen AL, Hansen AE, Gabizon A, Andresen TL. Liposome imaging agents in personalized medicine. Adv Drug Deliv Rev. 2012;64:1417–35. doi: 10.1016/j.addr.2012.09.003. [DOI] [PubMed] [Google Scholar]
- 53.Cho EC, Glaus C, Chen J, Welch MJ, Xia Y. Inorganic nanoparticle-based contrast agents for molecular imaging. Trends Mol Med. 2010;16:561–73. doi: 10.1016/j.molmed.2010.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Janib SM, Moses AS, MacKay JA. Imaging and drug delivery using theranostic nanoparticles. Adv Drug Deliv Rev. 2010;62:1052–63. doi: 10.1016/j.addr.2010.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mura S, Couvreur P. Nanotheranostics for personalized medicine. Adv Drug Deliv Rev. 2012;64:1394–416. doi: 10.1016/j.addr.2012.06.006. [DOI] [PubMed] [Google Scholar]
- 56.Fleischer AC, Lyshchik A, Jones HW, Jr, Crispens M, Loveless M, Andreotti RF, Williams PK, Fishman DA. Contrast-enhanced transvaginal sonography of benign versus malignant ovarian masses: Preliminary findings. J Ultrasound Med. 2008;27:1011–8. doi: 10.7863/jum.2008.27.7.1011. [DOI] [PubMed] [Google Scholar]
- 57.Fleischer AC, Lyshchik A, Andreotti RF, Hwang M, Jones HW, Fishman DA. Advances in sonographic detection of ovarian cancer: depiction of tumor neovascularity with microbubbles. Am J Roentgenol. 2010;194:343–8. doi: 10.2214/AJR.09.3446. [DOI] [PubMed] [Google Scholar]
- 58.Wheatley MA, Lewandowski J. Nano-sized ultrasound contrast agent: salting-out method. Mol Imaging. 2010;9:96–107. [PubMed] [Google Scholar]
- 59.Rapoport N, Gao Z, Kennedy A. Multifunctional nanoparticles for combining ultrasonic tumor imaging and targeted chemotherapy. J Natl Cancer Inst. 2007;99:1095–106. doi: 10.1093/jnci/djm043. [DOI] [PubMed] [Google Scholar]
- 60.Rapoport NY, Kennedy AM, Shea JE, Scaife CL, Nam KH. Controlled and targeted tumor chemotherapy by ultrasound-activated nanoemulsions/microbubbles. J Control Release. 2009;138:268–76. doi: 10.1016/j.jconrel.2009.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Fleischer AC, Lyshchik A, Hirari M, Moore RD, Abramson RG, Fishman DA. Early detection of ovarian cancer with conventional and contrast-enhanced transvaginal sonography: recent advances and potential improvements. J Oncol. 2012;2012:11. doi: 10.1155/2012/302858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Xu H, Regino CAS, Koyama Y, Hama Y, Gunn AJ, Bernardo M, Kobayashi H, Choyke PL, Brechbiel MW. Preparation and preliminary evaluation of a biotin-targeted, lectin-targeted dendrimer-based probe for dual-modality magnetic resonance and fluorescence imaging. Bioconj Chem. 2007;18:1474–82. doi: 10.1021/bc0701085. [DOI] [PubMed] [Google Scholar]
- 63.Kamaly N, Kalber T, Thanou M, Bell JD, Miller AD. Folate receptor targeted bimodal liposomes for tumor magnetic resonance imaging. Bioconj Chem. 2009;20:648–55. doi: 10.1021/bc8002259. [DOI] [PubMed] [Google Scholar]
- 64.Wang L, Neoh KG, Kang E-T, Shuter B. Multifunctional polyglycerol-grafted Fe3O4@ SiO2 nanoparticles for targeting ovarian cancer cells. Biomaterials. 2011;32:2166–73. doi: 10.1016/j.biomaterials.2010.11.042. [DOI] [PubMed] [Google Scholar]
- 65.Zhou Z, Wang L, Chi X, Bao J, Yang L, Zhao W, Chen Z, Wang X, Chen X, Gao J. Engineered iron-oxide-based nanoparticles as enhanced T1 contrast agents for efficient tumor imaging. ACS Nano. 2013;7:3287–96. doi: 10.1021/nn305991e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Misri R, Meier D, Yung AC, Kozlowski P, Häfeli UO. Development and evaluation of a dual-modality (MRI/SPECT) molecular imaging bioprobe. Nanomedicine. 2012;8:1007–16. doi: 10.1016/j.nano.2011.10.013. [DOI] [PubMed] [Google Scholar]
- 67.Luo S, Zhang E, Su Y, Cheng T, Shi C. A review of NIR dyes in cancer targeting and imaging. Biomaterials. 2011;32:7127–38. doi: 10.1016/j.biomaterials.2011.06.024. [DOI] [PubMed] [Google Scholar]
- 68.Schädlich A, Caysa H, Mueller T, Tenambergen F, Rose C, Göpferich A, Kuntsche J, Mäder K. Tumor accumulation of NIR fluorescent PEG–PLA nanoparticles: impact of particle size and human xenograft tumor model. ACS Nano. 2011;5:8710–20. doi: 10.1021/nn2026353. [DOI] [PubMed] [Google Scholar]
- 69.Katragadda U, Fan W, Wang Y, Teng Q, Tan C. Combined delivery of paclitaxel and tanespimycin via micellar nanocarriers: pharmacokinetics, efficacy and metabolomic analysis. PLoS ONE. 2013;8:e58619. doi: 10.1371/journal.pone.0058619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Nathwani BB, Jaffari M, Juriani AR, Mathur AB, Messer KE. Fabrication and characterization of silk-fibroin-coated quantum dots. IEEE Trans Nano Biosci. 2009;8:72–7. doi: 10.1109/TNB.2009.2017295. [DOI] [PubMed] [Google Scholar]
- 71.Wang H-Z, Wang H-Y, Liang R-Q, Ruan K-C. Detection of Tumor Marker CA125 in Ovarian Carcinoma Using Quantum Dots. Acta Biochimica et Biophysica Sinica. 2004;36:681–6. doi: 10.1093/abbs/36.10.681. [DOI] [PubMed] [Google Scholar]
- 72.Zdobnova TA, Dorofeev SG, Tananaev PN, Zlomanov VP, Stremovskiy OA, Lebedenko EN, Balalaeva IV, Deyev SM, Petrov RV. Imaging of human ovarian cancer SKOV-3 cells by quantum dot bioconjugates. Dokl Biochem Biophys. 2010;430:41–4. doi: 10.1134/s1607672910010126. [DOI] [PubMed] [Google Scholar]
- 73.Savla R, Taratula O, Garbuzenko O, Minko T. Tumor targeted quantum dot-mucin 1 aptamer-doxorubicin conjugate for imaging and treatment of cancer. J Control Release. 2011;153:16–22. doi: 10.1016/j.jconrel.2011.02.015. [DOI] [PubMed] [Google Scholar]
- 74.Cassette E, Helle M, Bezdetnaya L, Marchal F, Dubertret B, Pons T. Design of new quantum dot materials for deep tissue infrared imaging. Adv Drug Deliv Rev. 2013;65:719–31. doi: 10.1016/j.addr.2012.08.016. [DOI] [PubMed] [Google Scholar]
- 75.Jokerst JV, Cole AJ, Van de Sompel D, Gambhir SS. Gold nanorods for ovarian cancer detection with photoacoustic imaging and resection guidance via raman imaging in living mice. ACS Nano. 2012;6:10366–77. doi: 10.1021/nn304347g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Maeda H, Wu J, Sawa T, Matsumura Y, Hori K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release. 2000;65:271–84. doi: 10.1016/s0168-3659(99)00248-5. [DOI] [PubMed] [Google Scholar]
- 77.Gabizon A, Catane R, Uziely B, Kaufman B, Safra T, Cohen R, Martin F, Huang A, Barenholz Y. Prolonged circulation time and enhanced accumulation in malignant exudates of doxorubicin encapsulated in polyethylene-glycol coated liposomes. Cancer Res. 1994;54:987–92. [PubMed] [Google Scholar]
- 78.Muggia FM, Hainsworth JD, Jeffers S, Miller P, Groshen S, Tan M, Roman L, Uziely B, Muderspach L, Garcia A, Burnett A, Greco FA, Morrow CP, Paradiso LJ, Liang LJ. Phase II study of liposomal doxorubicin in refractory ovarian cancer: antitumor activity and toxicity modification by liposomal encapsulation. J Clin Oncol. 1997;15:987–93. doi: 10.1200/JCO.1997.15.3.987. [DOI] [PubMed] [Google Scholar]
- 79.Gordon AN, Tonda M, Sun S, Rackoff W. Long-term survival advantage for women treated with pegylated liposomal doxorubicin compared with topotecan in a phase 3 randomized study of recurrent and refractory epithelial ovarian cancer. Gynecol Oncol. 2004;95:1–8. doi: 10.1016/j.ygyno.2004.07.011. [DOI] [PubMed] [Google Scholar]
- 80.Gordon AN, Fleagle JT, Guthrie D, Parkin DE, Gore ME, Lacave AJ. Recurrent epithelial ovarian carcinoma: a randomized phase III study of pegylated liposomal doxorubicin versus topotecan. J Clin Oncol. 2001;19:3312–22. doi: 10.1200/JCO.2001.19.14.3312. [DOI] [PubMed] [Google Scholar]
- 81.Mutch DG, Orlando M, Goss T, Teneriello MG, Gordon AN, McMeekin SD, Wang Y, Scribner DR, Jr, Marciniack M, Naumann RW, Secord AA. Randomized phase III trial of gemcitabine compared with pegylated liposomal doxorubicin in patients with platinum-resistant ovarian cancer. J Clin Oncol. 2007;25:2811–8. doi: 10.1200/JCO.2006.09.6735. [DOI] [PubMed] [Google Scholar]
- 82.Ferrandina G, Ludovisi M, Lorusso D, Pignata S, Breda E, Savarese A, Del Medico P, Scaltriti L, Katsaros D, Priolo D, Scambia G. Phase III trial of gemcitabine compared with pegylated liposomal doxorubicin in progressive or recurrent ovarian cancer. J Clin Oncol. 2008;26:890–6. doi: 10.1200/JCO.2007.13.6606. [DOI] [PubMed] [Google Scholar]
- 83.Wagner U, Marth C, Largillier R, Kaern J, Brown C, Heywood M, Bonaventura T, Vergote I, Piccirillo MC, Fossati R, Gebski V, Lauraine EP. Final overall survival results of phase III GCIG CALYPSO trial of pegylated liposomal doxorubicin and carboplatin vs paclitaxel and carboplatin in platinum-sensitive ovarian cancer patients. Br J Cancer. 2012;107:588–91. doi: 10.1038/bjc.2012.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Monk BJ, Herzog TJ, Kaye SB, Krasner CN, Vermorken JB, Muggia FM, Pujade-Lauranine E, Lisyanskaya AS, Makhson AN, Rolski J, Gorbounova VA, Ghatage P, Bidzinski M, Shen K, Ngan HY, Vergote IB, Nam JH, Park YC, Lebedinsky CA, Poveda AM. Trabectedin plus pegylated liposomal Doxorubicin in recurrent ovarian cancer. J Clin Oncol. 2010;28:3107–14. doi: 10.1200/JCO.2009.25.4037. [DOI] [PubMed] [Google Scholar]
- 85.Poveda A, Vergote I, Tjulandin S, Kong B, Roy M, Chan S, Filipczyk-Cisarz E, Hagberg H, Kaye SB, Colombo N, Lebedinsky C, Parekh T, Gómez J, Park YC, Alfaro V, Monk BJ. Trabectedin plus pegylated liposomal doxorubicin in relapsed ovarian cancer: outcomes in the partially platinum-sensitive (platinum-free interval 6–12 months) subpopulation of OVA-301 phase III randomized trial. Ann Oncol. 2011;22:39–48. doi: 10.1093/annonc/mdq352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Andreopoulou E, Gaiotti D, Kim E, Downey A, Mirchandani D, Hamilton A, Jacobs A, Curtin J, Muggia F. Pegylated liposomal doxorubicin HCL (PLD; Caelyx/Doxil): experience with long-term maintenance in responding patients with recurrent epithelial ovarian cancer. Ann Oncol. 2007;18:716–21. doi: 10.1093/annonc/mdl484. [DOI] [PubMed] [Google Scholar]
- 87.Martin-Carbonero L, Palacios R, Valencia E, Saballs P, Sirera G, Santos I, Baldobi F, Alegre M, Goyenechea A, Pedreira J, Gonzalez del Castillo J, Martinez-Lacasa J, Ocampo A, Alsina M, Santos J, Podzamczer D, Gonzalez-Lahoz J Caelyx/Kaposi’s Sarcoma Spanish Group. Long-term prognosis of HIV-infected patients with Kaposi sarcoma treated with pegylated liposomal doxorubicin. Clin Infect Dis. 2008;47:410–7. doi: 10.1086/589865. [DOI] [PubMed] [Google Scholar]
- 88.Muggia F, Cannon T, Safra T, Curtin J. Delayed neoplastic and renal complications in women receiving long-term chemotherapy for recurrent ovarian cancer. J Natl Cancer Inst. 2011;103:160–1. doi: 10.1093/jnci/djq484. [DOI] [PubMed] [Google Scholar]
- 89.Herzog TJ. Update on the role of topotecan in the treatment of recurrent ovarian cancer. Oncologist. 2002;7 (Suppl 5):3–10. doi: 10.1634/theoncologist.7-suppl_5-3. [DOI] [PubMed] [Google Scholar]
- 90.Drummond DC, Noble CO, Guo Z, Hayes ME, Connolly-Ingram C, Gabriel BS, Hann B, Liu B, Park JW, Hong K, Benz CC, Marks JD, Kirpotin DB. Development of a highly stable and tar-getable nanoliposomal formulation of topotecan. J Control Release. 2010;141:13–21. doi: 10.1016/j.jconrel.2009.08.006. [DOI] [PubMed] [Google Scholar]
- 91.Dadashzadeh S, Vali AM, Rezaie M. The effect of PEG coating on in vitro cytotoxicity and in vivo disposition of topotecan loaded liposomes in rats. Int J Pharm. 2008;353:251–9. doi: 10.1016/j.ijpharm.2007.11.030. [DOI] [PubMed] [Google Scholar]
- 92.Ma Y, Yang Q, Wang L, Zhou X, Zhao Y, Deng Y. Repeated injections of PEGylated liposomal topotecan induces accelerated blood clearance phenomenon in rats. Eur J Pharm Sci. 2012;45:539–45. doi: 10.1016/j.ejps.2011.11.014. [DOI] [PubMed] [Google Scholar]
- 93.Dark GG, Calvert AH, Grimshaw R, Poole C, Swenerton K, Kaye S, Coleman R, Jayson G, Le T, Ellard S, Trudeau M, Vasey P, Hamilton M, Cameron T, Barrett E, Walsh W, Mcintosh L, Eisenhauer EA. Randomized trial of two intravenous schedules of the topoisomerase I inhibitor liposomal lurtotecan in women with relapsed epithelial ovarian cancer: a trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 2005;23:1859–66. doi: 10.1200/JCO.2005.02.028. [DOI] [PubMed] [Google Scholar]
- 94.Corporation C. Abraxane. Celgene Corporation; Summit NY: [Google Scholar]
- 95.Mirtsching B, Cosgriff T, Harker G, Keaton M, Chidiac T, Min M. A phase II study of weekly nanoparticle albumin-bound paclitaxel with or without trastuzumab in metastatic breast cancer. Clin Breast Cancer. 2011;11:121–8. doi: 10.1016/j.clbc.2011.03.007. [DOI] [PubMed] [Google Scholar]
- 96.Micha JP, Goldstein BH, Birk CL, Rettenmaier MA, Brown JV., 3rd Abraxane in the treatment of ovarian cancer: the absence of hypersensitivity reactions. Gynecol Oncol. 2006;100:437–8. doi: 10.1016/j.ygyno.2005.09.012. [DOI] [PubMed] [Google Scholar]
- 97.Coleman RL, Brady WE, McMeekin DS, Rose PG, Soper JT, Lentz SS, Hoffman JS, Shahin MS. A phase II evaluation of nanoparticle, albumin-bound (nab) paclitaxel in the treatment of recurrent or persistent platinum-resistant ovarian, fallopian tube, or primary peritoneal cancer: a Gynecologic Oncology Group study. Gynecol Oncol. 2011;122:111–5. doi: 10.1016/j.ygyno.2011.03.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Teneriello MG, Tseng PC, Crozier M, Encarnacion C, Hancock K, Messing MJ, Boehm KA, Williams A, Asmar L. Phase II evaluation of nanoparticle albumin-bound paclitaxel in platinum-sensitive patients with recurrent ovarian, peritoneal, or fallopian tube cancer. J Clin Oncol. 2009;27:1426–31. doi: 10.1200/JCO.2008.18.9548. [DOI] [PubMed] [Google Scholar]
- 99.Singer JW. Paclitaxel poliglumex (XYOTAX, CT-2103): a macromolecular taxane. J Control Release. 2005;109:120–6. doi: 10.1016/j.jconrel.2005.09.033. [DOI] [PubMed] [Google Scholar]
- 100.Sabbatini P, Sill MW, O’Malley D, Adler L, Secord AA Gynecologic Oncology Group S. A phase II trial of paclitaxel poliglumex in recurrent or persistent ovarian or primary peritoneal cancer (EOC): a Gynecologic Oncology Group Study. Gynecol Oncol. 2008;111:455–60. doi: 10.1016/j.ygyno.2008.07.049. [DOI] [PubMed] [Google Scholar]
- 101.Morgan MA, Darcy KM, Rose PG, DeGeest K, Bookman MA, Aikins JK, Sill MW, Mannel RS, Allievi C, Egorin MJ Gynecologic Oncology Group. Paclitaxel poliglumex and carboplatin as first-line therapy in ovarian, peritoneal or fallopian tube cancer: a phase I and feasibility trial of the Gynecologic Oncology Group. Gynecol Oncol. 2008;110:329–35. doi: 10.1016/j.ygyno.2008.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Hoch UML, Maslyar D, Von Hoff D. NKTR-102 demonstrates nonclinical and phase 1 clinical anti-tumor activity in ovarian cancer. Hall 141, Gynecologic Cancer, Poster Board 161. Nektar Therapeutics; Abstract 8015, ESMO 34 Conference; September 22 2009, 2:00 pm; San Carlos, CA. [Google Scholar]
- 103.Awada A, Garcia AA, Chan S, Jerusalem GH, Coleman RE, Huizing MT, Mehdi A, O’Reilly SM, Hamm JT, Barrett-Lee PJ, Cocquyt V, Sideras K, Young DE, Zhao C, Chia YL, Hoch U, Hannah AL, Perez EA NKTR-102 Study Group. Two schedules of etirinotecan pegol (NKTR-102) in patients with previously treated metastatic breast cancer: a randomised phase 2 study. Lancet Oncol. 2013;14:1216–25. doi: 10.1016/S1470-2045(13)70429-7. [DOI] [PubMed] [Google Scholar]
- 104.Iqbal S, Tsao-Wei DD, Quinn DI, Gitlitz BJ, Groshen S, Aparicio A, Lenz HJ, El-Khoueiry A, Pinski J, Garcia AA. Phase I clinical trial of pegylated liposomal Doxorubicin and docetaxel in patients with advanced solid tumors. Am J Clin Oncol. 2011;34:27–31. doi: 10.1097/COC.0b013e3181cae766. [DOI] [PubMed] [Google Scholar]
- 105.Svenson S, Wolfgang M, Hwang J, Ryan J, Eliasof S. Preclinical to clinical development of the novel camptothecin nanopharmaceutical CRLX101. J Control Release. 2011;153:49–55. doi: 10.1016/j.jconrel.2011.03.007. [DOI] [PubMed] [Google Scholar]
- 106.Nowotnik DP, Cvitkovic E. ProLindac (AP5346): a review of the development of an HPMA DACH platinum polymer therapeutic. Adv Drug Deliv Rev. 2009;61:1214–9. doi: 10.1016/j.addr.2009.06.004. [DOI] [PubMed] [Google Scholar]
- 107.Duncan R. Development of HPMA copolymer-anticancer conjugates: clinical experience and lessons learnt. Adv Drug Deliv Rev. 2009;61:1131–48. doi: 10.1016/j.addr.2009.05.007. [DOI] [PubMed] [Google Scholar]
- 108.Lin X, Zhang Q, Rice JR, Stewart DR, Nowotnik DP, Howell SB. Improved targeting of platinum chemotherapeutics. the antitumour activity of the HPMA copolymer platinum agent AP5280 in murine tumour models. Eur J Cancer. 2004;40:291–7. doi: 10.1016/j.ejca.2003.09.022. [DOI] [PubMed] [Google Scholar]
- 109.Desale SS, Cohen SM, Zhao Y, Kabanov AV, Bronich TK. Biodegradable hybrid polymer micelles for combination drug therapy in ovarian cancer. J Control Release. 2013;171:339–48. doi: 10.1016/j.jconrel.2013.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Gong C, Yang B, Qian Z, Zhao X, Wu Q, Qi X, Wang Y, Guo G, Kan B, Luo F, Wei Y. Improving intraperitoneal chemotherapeutic effect and preventing postsurgical adhesions simultaneously with biodegradable micelles. Nanomedicine. 2012;8:963–73. doi: 10.1016/j.nano.2011.10.010. [DOI] [PubMed] [Google Scholar]
- 111.Cho H, Lai TC, Kwon GS. Poly(ethylene glycol)-block-poly(epsilon-caprolactone) micelles for combination drug delivery: evaluation of paclitaxel, cyclopamine and gossypol in intraperitoneal xenograft models of ovarian cancer. J Control Release. 2013;166:1–9. doi: 10.1016/j.jconrel.2012.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Kirkpatrick GJ, Plumb JA, Sutcliffe OB, Flint DJ, Wheate NJ. Evaluation of anionic half generation 3.5–6.5 poly(amidoamine) dendrimers as delivery vehicles for the active component of the anticancer drug cisplatin. J Inorg Biochem. 2011;105:1115–22. doi: 10.1016/j.jinorgbio.2011.05.017. [DOI] [PubMed] [Google Scholar]
- 113.Kurtoglu YE, Mishra MK, Kannan S, Kannan RM. Drug release characteristics of PAMAM dendrimer-drug conjugates with different linkers. Int J Pharm. 2010;384:189–94. doi: 10.1016/j.ijpharm.2009.10.017. [DOI] [PubMed] [Google Scholar]
- 114.Zhu S, Hong M, Tang G, Qian L, Lin J, Jiang Y, Pei Y. Partly PEGylated polyamidoamine dendrimer for tumor-selective targeting of doxorubicin: the effects of PEGylation degree and drug conjugation style. Biomaterials. 2010;31:1360–71. doi: 10.1016/j.biomaterials.2009.10.044. [DOI] [PubMed] [Google Scholar]
- 115.Zhu S, Hong M, Zhang L, Tang G, Jiang Y, Pei Y. PEGylated PAMAM dendrimer-doxorubicin conjugates: in vitro evaluation and in vivo tumor accumulation. Pharm Res. 2010;27:161–74. doi: 10.1007/s11095-009-9992-1. [DOI] [PubMed] [Google Scholar]
- 116.Xiao K, Luo J, Fowler WL, Li Y, Lee JS, Xing L, Cheng RH, Wang L, Lam KS. A self-assembling nanoparticle for paclitaxel delivery in ovarian cancer. Biomaterials. 2009;30:6006–16. doi: 10.1016/j.biomaterials.2009.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Xiao W, Luo J, Jain T, Riggs JW, Tseng HP, Henderson PT, Cherry SR, Rowland D, Lam KS. Biodistribution and pharmacokinetics of a telodendrimer micellar paclitaxel nanoformulation in a mouse xenograft model of ovarian cancer. Int J Nanomed. 2012;7:1587–97. doi: 10.2147/IJN.S29306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.van Vlerken LE, Duan Z, Little SR, Seiden MV, Amiji MM. Augmentation of therapeutic efficacy in drug-resistant tumor models using ceramide coadministration in temporal-controlled polymer-blend nanoparticle delivery systems. AAPS J. 2010;12:171–80. doi: 10.1208/s12248-010-9174-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Wagstaff AJ, Brown SD, Holden MR, Craig GE, Plumb JA, Brown RE, Schreiter N, Chrzanowski W, Wheate NJ. Cisplatin drug delivery using gold-coated iron oxide nanoparticles for enhanced tumour targeting with external magnetic fields. Inorg Chim Acta. 2012;393:328–33. [Google Scholar]
- 120.Qin Y, Song Q-G, Zhang Z-R, Liu J, Fu Y, He Q, Liu J. Ovarian tumor targeting of docetaxel-loaded liposomes mediated by luteinizing hormone-releasing hormone analogues. Arzneimittelforschung. 2008;58:529–34. doi: 10.1055/s-0031-1296553. [DOI] [PubMed] [Google Scholar]
- 121.Xiao K, Li Y, Lee JS, Gonik AM, Dong T, Fung G, Sanchez E, Xing L, Cheng HR, Luo J, Lam KS. “OA02” peptide facilitates the precise targeting of paclitaxel-loaded micellar nanoparticles to ovarian cancer in vivo. Cancer Res. 2012;72:2100–10. doi: 10.1158/0008-5472.CAN-11-3883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Nukolova NV, Oberoi HS, Cohen SM, Kabanov AV, Bronich TK. Folate-decorated nanogels for targeted therapy of ovarian cancer. Biomaterials. 2011;32:5417–26. doi: 10.1016/j.biomaterials.2011.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Nukolova NV, Oberoi HS, Zhao Y, Chekhonin VP, Kabanov AV, Bronich TK. LHRH-targeted nanogels as a delivery system for Cisplatin to ovarian cancer. Mol Pharm. 2013;10:3913–21. doi: 10.1021/mp4003688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Gao ZG, Lee DH, Kim DI, Bae YH. Doxorubicin loaded pH-sensitive micelle targeting acidic extracellular pH of human ovarian A2780 tumor in mice. J Drug Target. 2005;13:391–7. doi: 10.1080/10611860500376741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Kim D, Lee ES, Park K, Kwon IC, Bae YH. Doxorubicin loaded pH-sensitive micelle: antitumoral efficacy against ovarian A2780/DOXR tumor. Pharm Res. 2008;25:2074–82. doi: 10.1007/s11095-008-9603-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Kim D, Gao ZG, Lee ES, Bae YH. In vivo evaluation of doxorubicin-loaded polymeric micelles targeting folate receptors and early endosomal pH in drug-resistant ovarian cancer. Mol Pharm. 2009;6:1353–62. doi: 10.1021/mp900021q. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Dolmans DEJGJ, Fukumura D, Jain RK. Photodynamic therapy for cancer. Nat Rev Cancer. 2003;3:380–7. doi: 10.1038/nrc1071. [DOI] [PubMed] [Google Scholar]
- 128.Usuda J, Kato H, Okunaka T, Furukawa K, Tsutsui H, Yamada K, Suga Y, Honda H, Nagatsuka Y, Ohira T, Tsuboi M, Hirano T. Photodynamic therapy (PDT) for lung cancers. J Thorac Oncol. 2006;1:489–93. [PubMed] [Google Scholar]
- 129.Allison RR, Sibata CH. Oncologic photodynamic therapy photosensitizers: a clinical review. Photodiagn Photodyn Ther. 2010;7:61–75. doi: 10.1016/j.pdpdt.2010.02.001. [DOI] [PubMed] [Google Scholar]
- 130.Stolyarov AM, Wei L, Shapira O, Sorin F, Chua SL, Joannopoulos JD, Fink Y. Microfluidic directional emission control of an azimuthally polarized radial fibre laser. Nat Photon. 2012;6:229–33. [Google Scholar]
- 131.Davila ML. Photodynamic therapy. Gastrointest Endosc Clin N Am. 2011;21:67–79. doi: 10.1016/j.giec.2010.09.002. [DOI] [PubMed] [Google Scholar]
- 132.Master A, Livingston M, Sen Gupta A. Photodynamic nanomedicine in the treatment of solid tumors: perspectives and challenges. J Control Release. 2013;168:88–102. doi: 10.1016/j.jconrel.2013.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Zeisser-Labouebe M, Lange N, Gurny R, Delie F. Hypericin-loaded nanoparticles for the photodynamic treatment of ovarian cancer. Int J Pharm. 2006;326:174–81. doi: 10.1016/j.ijpharm.2006.07.012. [DOI] [PubMed] [Google Scholar]
- 134.Zeisser-Labouèbe M, Delie F, Gurny R, Lange N. Benefits of nanoencapsulation for the hypercin-mediated photodetection of ovarian micrometastases. Eur J Pharm Biopharma. 2009;71:207–13. doi: 10.1016/j.ejpb.2008.10.005. [DOI] [PubMed] [Google Scholar]
- 135.Li Z, Wang C, Cheng L, Gong H, Yin S, Gong Q, Li Y, Liu Z. PEG-functionalized iron oxide nanoclusters loaded with chlorin e6 for targeted, NIR light induced, photodynamic therapy. Biomaterials. 2013;34:9160–70. doi: 10.1016/j.biomaterials.2013.08.041. [DOI] [PubMed] [Google Scholar]
- 136.Wang C, Tao H, Cheng L, Liu Z. Near-infrared light induced in vivo photodynamic therapy of cancer based on upconversion nanoparticles. Biomaterials. 2011;32:6145–54. doi: 10.1016/j.biomaterials.2011.05.007. [DOI] [PubMed] [Google Scholar]
- 137.Cohen BA, Bergkvist M. Targeted in vitro photodynamic therapy via aptamer-labeled, porphyrin-loaded virus capsids. J Photochem Photobiol B. 2013;121:67–74. doi: 10.1016/j.jphotobiol.2013.02.013. [DOI] [PubMed] [Google Scholar]
- 138.Mroz P, Xia Y, Asanuma D, Konopko A, Zhiyentayev T, Huang YY, Sharma SK, Dai T, Khan UJ, Wharton T, Hamblin MR. Intraperitoneal photodynamic therapy mediated by a fullerene in a mouse model of abdominal dissemination of colon adenocarcinoma. Nanomedicine. 2011;7:965–74. doi: 10.1016/j.nano.2011.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Liu J, Ohta S, Sonoda A, Yamada M, Yamamoto M, Nitta N, Murata K, Tabata Y. Preparation of PEG-conjugated fullerene containing Gd3+ ions for photodynamic therapy. J Control Release. 2007;117:104–10. doi: 10.1016/j.jconrel.2006.10.008. [DOI] [PubMed] [Google Scholar]
- 140.Tabata Y, Murakami Y, Ikada Y. Photodynamic effect of polyethylene glycol-modified fullerene on tumor. Jpn J Cancer Res. 1997;88:1108–16. doi: 10.1111/j.1349-7006.1997.tb00336.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Judge AD, Sood V, Shaw JR, Fang D, McClintock K, MacLachlan I. Sequence-dependent stimulation of the mammalian innate immune response by synthetic siRNA. Nat Biotechnol. 2005;23:457–62. doi: 10.1038/nbt1081. [DOI] [PubMed] [Google Scholar]
- 142.Mansouri S, Cuie Y, Winnik F, Shi Q, Lavigne P, Benderdour M, Beaumont E, Fernandes JC. Characterization of folate-chitosan-DNA nanoparticles for gene therapy. Biomaterials. 2006;27:2060–5. doi: 10.1016/j.biomaterials.2005.09.020. [DOI] [PubMed] [Google Scholar]
- 143.Kim DH, Rossi JJ. Strategies for silencing human disease using RNA interference. Nat Rev Genet. 2007;8:173–84. doi: 10.1038/nrg2006. [DOI] [PubMed] [Google Scholar]
- 144.Heidel JD, Schluep T. Cyclodextrin-containing polymers: versatile platforms of drug delivery materials. J Drug Deliv. 2012;2012:262731. doi: 10.1155/2012/262731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Reddy JA, Abburi C, Hofland H, Howard SJ, Vlahov I, Wils P, Leamon CP. Folate-targeted, cationic liposome-mediated gene transfer into disseminated peritoneal tumors. Gene Ther. 2002;9:1542–50. doi: 10.1038/sj.gt.3301833. [DOI] [PubMed] [Google Scholar]
- 146.Senzer N, Nemunaitis J, Nemunaitis D, Bedell C, Edelman G, Barve M, Nunan R, Pirollo KF, Rait A, Chang EH. Phase I study of a systemically delivered p53 nanoparticle in advanced solid tumors. Mol Ther. 2013;21:1096–103. doi: 10.1038/mt.2013.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Zeimet AG, Marth C. Why did p53 gene therapy fail in ovarian cancer? Lancet Oncol. 2003;4:415–22. doi: 10.1016/s1470-2045(03)01139-2. [DOI] [PubMed] [Google Scholar]
- 148.Madhusudan S, Tamir A, Bates N, Flanagan E, Gore ME, Barton DP, Harper P, Secki M, Thomas H, Lemoine NR, Charnock M, Habib NA, Lechler R, Nicholls J, Pignatelli M, Ganesan TS. A multicenter phase I gene therapy clinical trial involving intraperitoneal administration of E1A-lipid complex in patients with recurrent epithelial ovarian cancer overexpressing HER-2/neu oncogene. Clin Cancer Res. 2004;10:2986–96. doi: 10.1158/1078-0432.ccr-03-0291. [DOI] [PubMed] [Google Scholar]
- 149.Anwer K, Kelly FJ, Chu C, Fewell JG, Lewis D, Alvarez RD. Phase I trial of a formulated IL-12 plasmid in combination with carboplatin and docetaxel chemotherapy in the treatment of platinum-sensitive recurrent ovarian cancer. Gynecol Oncol. 2013;131:169–73. doi: 10.1016/j.ygyno.2013.07.081. [DOI] [PubMed] [Google Scholar]
- 150.Ren Y, Cheung HW, von Maltzhan G, Agrawal A, Cowley GS, Weir BA, Boehm JS, Tamayo P, Karst AM, Liu JF, Hirsch MS, Mesirov JP, Drapkin R, Root DE, Lo J, Fogal V, Ruoslahti E, Hahn WC, Bhatia SN. Targeted tumor-penetrating siRNA nanocomplexes for credentialing the ovarian cancer oncogene ID4. Sci Transl Med. 2012;4:147ra12. doi: 10.1126/scitranslmed.3003778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Xie C, Gou ML, Yi T, Deng H, Li ZY, Liu P, Qi XR, He X, Wei Y, Zhao X. Efficient inhibition of ovarian cancer by truncation mutant of FILIP1L gene delivered by novel biodegradable cationic heparin-polyethyleneimine nanogels. Human Gene Ther. 2011;22:1413–22. doi: 10.1089/hum.2011.047. [DOI] [PubMed] [Google Scholar]
- 152.Davis ME, Zuckerman JE, Choi CH, Seligson D, Tolcher A, Alabi CA, Yen Y, Heidel JD, Ribas A. Evidence of RNAi in humans from systemically administered siRNA via targeted nanoparticles. Nature. 2010;464:1067–70. doi: 10.1038/nature08956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Tabernero J, Shapiro GI, LoRusso PM, Cervantes A, Schwartz GK, Weiss GJ, Paz-Ares L, cho DC, Infante JR, Alsina M, Gounder MM, Falzone R, Harrop J, White AC, Toudjarska I, Bumcrot D, Meyers RE, Hinkle G, Svrzikapa N, Hutabarat RM, Clausen VA, Cehelsky J, Nochur SV, Gamba-Vitalo C, Vaishnaw AK, Sah DW, Gollob JA, Burris HA., 3rd First-in-humans trial of an RNA interference therapeutic targeting VEGF and KSP in cancer patients with liver involvement. Cancer Discov. 2013;3:406–17. doi: 10.1158/2159-8290.CD-12-0429. [DOI] [PubMed] [Google Scholar]
- 154.Kim HS, Han HD, Armaiz-Pena GN, Stone RL, Nam EJ, Lee JW, Shahzad MM, Nick AM, Lee SJ, Roh JW, Nishimura M, Mangala LS, Bottsford-Miller J, Gallick GE, Lopez-Berestein G, Sood AK. Functional roles of Src and Fgr in ovarian carcinoma. Clin Cancer Res. 2011;17:1713–21. doi: 10.1158/1078-0432.CCR-10-2081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Sun C, Yi T, Song X, Li S, Qi X, Chen X, Lin H, He X, Li Z, Wei Y, Zhao X. Efficient inhibition of ovarian cancer by short hairpin RNA targeting claudin-3. Oncol Rep. 2011;26:193–200. doi: 10.3892/or.2011.1275. [DOI] [PubMed] [Google Scholar]
- 156.Zou L, Song X, Yi T, Li S, Deng H, Chen X, Li Z, Bai Y, Zhong Q, Wei Y, Zhao X. Administration of PLGA nanoparticles carrying shRNA against focal adhesion kinase and CD44 results in enhanced antitumor effects against ovarian cancer. Cancer Gene Ther. 2013;20:242–50. doi: 10.1038/cgt.2013.12. [DOI] [PubMed] [Google Scholar]
- 157.Goldberg MS, Xing D, Ren Y, Orsulic S, Bhatia SN, Sharp PA. Nanoparticle-mediated delivery of siRNA targeting Parp1 extends survival of mice bearing tumors derived from Brca1-deficient ovarian cancer cells. Proc Natl Acad Sci U S A. 2011;108:745–50. doi: 10.1073/pnas.1016538108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Nishimura M, Jung EJ, Shah MY, Lu C, Spizzo R, Shimizu M, Han HD, Ivan C, Rossi S, Zhang X, Nicoloso MS, Wu SY, Almeida MI, Bottsford-Miller J, Pecot CV, Zand B, Matsuo K, Shahzad MM, Jennings NB, Rodriguez-Aguayo C, Lopez-Berestein G, Sood AK, Calin GA. Therapeutic synergy between microRNA and siRNA in ovarian cancer treatment. Cancer Discov. 2013;3:1302–15. doi: 10.1158/2159-8290.CD-13-0159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Shen H, Rodriguez-Aguayo C, Xu R, Gonzalez-Villasana V, Mai J, Huang Y, Zhang G, Guo X, Bai L, Qin G, Deng X, Li Q, Erm DR, Aslan B, Liu X, Sakamoto J, Chavez-Reyes A, Han HD, Sood AK, Ferrari M, Lopez-Berestein G. Enhancing chemotherapy response with sustained EphA2 silencing using multistage vector delivery. Clin Cancer Res. 2013;19:1806–15. doi: 10.1158/1078-0432.CCR-12-2764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Pasquale EB. Eph receptor signalling casts a wide net on cell behaviour. Nat Rev Mol Cell Biol. 2005;6:462–75. doi: 10.1038/nrm1662. [DOI] [PubMed] [Google Scholar]
- 161.Thaker PH, Deavers M, Celestino J, Thornton A, Fletcher MS, Landen CN, Kinch MS, Kiener PA, Sood AK. EphA2 expression is associated with aggressive features in ovarian carcinoma. Clin Cancer Res. 2004;10:5145–50. doi: 10.1158/1078-0432.CCR-03-0589. [DOI] [PubMed] [Google Scholar]
- 162.Lin YG, Han LY, Kamat AA, Merritt WM, Landen CN, Deavers MT, Fletcher MS, Urbauer DL, Kinch MS, Sood AK. EphA2 overexpression is associated with angiogenesis in ovarian cancer. Cancer. 2007;109:332–40. doi: 10.1002/cncr.22415. [DOI] [PubMed] [Google Scholar]
- 163.Hasan N, Mann A, Ferrari M, Tanaka T. Mesoporous silicon particles for sustained gene silencing. Methods Mol Biol. 2013;1049:481–93. doi: 10.1007/978-1-62703-547-7_36. [DOI] [PubMed] [Google Scholar]
- 164.Tanaka T, Mangala LS, Vivas-Mejia PE, Nieves-Alicea R, Mann AP, Mora E, Han HD, Shahzad MM, Liu X, Bhavane R, Gu J, Fakhoury JR, Chiappini C, Lu C, Matsuo K, Godin B, Stone RL, Nick AM, Lopez-Berestein G, Sood AK, Ferrari M. Sustained small interfering RNA delivery by mesoporous silicon particles. Cancer Research. 2010;70:3687–96. doi: 10.1158/0008-5472.CAN-09-3931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Dickerson EB, Blackburn WH, Smith MH, Kapa LB, Lyon LA, McDonald JF. Chemosensitization of cancer cells by siRNA using targeted nanogel delivery. BMC Cancer. 2010;10:10. doi: 10.1186/1471-2407-10-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Shahzad MM, Mangala LS, Han HD, Lu C, Bottsford-Miller J, Nishimura M, Mora EM, Lee JW, Stone RL, Pecot CV, Thanapprapasr D, Roh JW, Gaur P, Nair MP, Park YY, Sabnis N, Deavers MT, Lee JS, Ellis LM, Lopez-Berestein G, McConathy WJ, Prokai L, Lacko AG, Sood AK. Targeted delivery of small interfering RNA using reconstituted high-density lipoprotein nanoparticles. Neoplasia. 2011;13:309–19. doi: 10.1593/neo.101372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Sugahara KN, Teesalu T, Karmali PP, Kotamraju VR, Agemy L, Greenwald DR, Ruoslahti E. Coadministration of a tumor-penetrating peptide enhances the efficacy of cancer drugs. Science. 2010;328:1031–5. doi: 10.1126/science.1183057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Han HD, Mangala LS, Lee JW, Shahzad MM, Kim HS, Shen D, Nam EJ, Mora EM, Stone RL, Lu C, Lee SJ, Roh JW, Nick AM, Lopez-Berestein G, Sood AK. Targeted gene silencing using RGD-labeled chitosan nanoparticles. Clin Cancer Res. 2010;16:3910–22. doi: 10.1158/1078-0432.CCR-10-0005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Steg AD, Katre AA, Goodman B, Han HD, Nick AM, Stone RL, Coleman RL, Alvarez RD, Lopez-Berestein G, Sood AK, Landen CN. Targeting the notch ligand JAGGED1 in both tumor cells and stroma in ovarian cancer. Clin Cancer Res. 2011;17:5674–85. doi: 10.1158/1078-0432.CCR-11-0432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Cubillos-Ruiz JR, Engle X, Scarlett UK, Martinez D, Barber A, Elgueta R, Wang L, Nesbeth Y, Durant Y, Gewirtz AT, Sentman CL, Kedl R, Conejo-Garcia JR. Polyethylenimine-based siRNA nanocomplexes reprogram tumor-associated dendritic cells via TLR5 to elicit therapeutic antitumor immunity. J Clin Invest. 2009;119:2231–44. doi: 10.1172/JCI37716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Cubillos-Ruiz JR, Baird JR, Tesone AJ, Rutkowski MR, Scarlett UK, Camposeco-Jacobs AL, Anadon-Arnillas J, Harwood NM, Korc M, Fiering SN, Sempere LF, Conejo-Garcia JR. Reprogramming tumor-associated dendritic cells in vivo using miRNA mimetics triggers protective immunity against ovarian cancer. Cancer Res. 2012;72:1683–93. doi: 10.1158/0008-5472.CAN-11-3160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Delgado G, Potkul RK, Treat JA, Lewandowski GS, Barter JF, Forst D, Rahman A. A phase I/II study of intraperitoneally administered doxorubicin entrapped in cardiolipin liposomes in patients with ovarian cancer. Am J Obstet Gynecol. 1989;160:812–7. doi: 10.1016/0002-9378(89)90296-2. discussion 817–9. [DOI] [PubMed] [Google Scholar]
- 173.Gilmore D, Schulz M, Liu R, Zubris KA, Padera RF, Catalano PJ, Grinstaff MW, Colson YL. Cytoreductive surgery and intra-operative administration of paclitaxel-loaded expansile nanoparticles delay tumor recurrence in ovarian carcinoma. Ann Surg Oncol. 2013;20:1684–93. doi: 10.1245/s10434-012-2696-5. [DOI] [PubMed] [Google Scholar]
- 174.Das M, Patil S, Bhargava N, Kang J-F, Riedel LM, Seal S, Hickman JJ. Auto-catalytic ceria nanoparticles offer neuroprotection to adult rat spinal cord neurons. Biomaterials. 2007;28:1918–25. doi: 10.1016/j.biomaterials.2006.11.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Heckert EG, Karakoti AS, Seal S, Self WT. The role of cerium redox state in the SOD mimetic activity of nanoceria. Biomaterials. 2008;29:2705–9. doi: 10.1016/j.biomaterials.2008.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Giri S, Karakoti A, Graham RP, Maguire JL, Reilly CM, Seal S, Rattan R, Shridhar V. Nanoceria: a rare-earth nanoparticle as a novel anti-angiogenic therapeutic agent in ovarian cancer. PLoS ONE. 2013;8(1):e54578. doi: 10.1371/journal.pone.0054578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Randall LM, Monk BJ. Bevacizumab toxicities and their management in ovarian cancer. Gynecol Oncol. 2010;117:497–504. doi: 10.1016/j.ygyno.2010.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Lecarpentier E, Ouaffi L, Mir O, Berveiller P, Maurel M, Pujade-Lauraine E, Bouillot JL, Veyrie N. Bevacizumab-induced small bowel perforation in a patient with breast cancer without intraabdominal metastases. Invest New Drugs. 2011;29:1500–3. doi: 10.1007/s10637-010-9505-4. [DOI] [PubMed] [Google Scholar]
- 179.Bansal N, Hoffman M. Bladder perforation in a patient with recurrent epithelial ovarian cancer after treatment with bevacizumab. Gynecol Oncol. 2011;120:313–4. doi: 10.1016/j.ygyno.2010.10.002. [DOI] [PubMed] [Google Scholar]
- 180.Toraya-Brown S, Sheen MR, Baird JR, Barry S, Demidenko E, Turk MJ, Hoopes PJ, Conejo-Garcia JR, Fiering S. Phagocytes mediate targeting of iron oxide nanoparticles to tumors for cancer therapy. Integr Biol (Camb) 2013;5:159–71. doi: 10.1039/c2ib20180a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Di Pasqua AJ, Yuan H, Chung Y, Kim JK, Huckle JE, Li C, Sadgrove M, Tran TH, Jay M, Lu X. Neutron-activatable holmium-containing mesoporous silica nanoparticles as a potential radionuclide therapeutic agent for ovarian cancer. J Nucl Med. 2013;54:111–6. doi: 10.2967/jnumed.112.106609. [DOI] [PubMed] [Google Scholar]
- 182.Stevens EV, Carpenter AW, Shin JH, Liu J, Der CJ, Schoenfisch MH. Nitric oxide-releasing silica nanoparticle inhibition of ovarian cancer cell growth. Mol Pharm. 2010;7:775–85. doi: 10.1021/mp9002865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.You J, Zhang R, Zhang G, Zhong M, Liu Y, Van Pelt CS, Liang D, Wei W, Sood AK, Li C. Photothermal-chemotherapy with doxorubicin-loaded hollow gold nanospheres: a platform for near-infrared light-trigged drug release. J Control Release. 2012;158:319–28. doi: 10.1016/j.jconrel.2011.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Hun X, Zhang Z, Tiao L. Anti-Her-2 monoclonal antibody conjugated polymer fluorescent nanoparticles probe for ovarian cancer imaging. Anal Chim Acta. 2008;625:201–6. doi: 10.1016/j.aca.2008.07.038. [DOI] [PubMed] [Google Scholar]
- 185.Corbin IR, Ng KK, Ding L, Jurisicova A, Zheng G. Near-infrared fluorescent imaging of metastatic ovarian cancer using folate receptor-targeted high-density lipoprotein nanocarriers. Nanomedicine (Lond) 2013;8:875–90. doi: 10.2217/nnm.12.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Dai Y, Ma Pa, Cheng Z, Kang X, Zhang X, Hou Z, Li C, Yang D, Zhai X, Lin J. Up-Conversion Cell imaging and pH-induced thermally controlled drug release from NaYF4:Yb3+/Er3+@ hydrogel core–shell hybrid microspheres. ACS Nano. 2012;6:3327–38. doi: 10.1021/nn300303q. [DOI] [PubMed] [Google Scholar]
- 187.Ibrahim NK, Desai N, Legha S, Soon-Shiong P, Theriault RL, Rivera E, Esmaeli B, Ring SE, Bedikian A, Hortobagyi GN, Ellerhorst JA. Phase I and pharmacokinetic study of ABI-007, a Cremophor-free, protein-stabilized, nanoparticle formulation of paclitaxel. Clin Cancer Res. 2002;8:1038–44. [PubMed] [Google Scholar]
- 188.Rademaker-Lakhai JM, Terret C, Howell SB, Baud CM, De Boer RF, Pluim D, Beijnen JH, Schellens JH, Droz JP. A Phase I and pharmacological study of the platinum polymer AP5280 given as an intravenous infusion once every 3 weeks in patients with solid tumors. Clin Cancer Res. 2004;10:3386–95. doi: 10.1158/1078-0432.CCR-03-0315. [DOI] [PubMed] [Google Scholar]
- 189.Chu KS, Hasan W, Rawal S, Walsh MD, Enlow EM, Luft JC, Bridges AS, Kuijer JL, Napier ME, Zamboni WC, DeSimone JM. Plasma, tumor and tissue pharmacokinetics of Docetaxel delivered via nanoparticles of different sizes and shapes in mice bearing SKOV-3 human ovarian carcinoma xenograft. Nanomedicine. 2013;9:686–93. doi: 10.1016/j.nano.2012.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Yallapu MM, Maher DM, Sundram V, Bell MC, Jaggi M, Chauhan SC. Curcumin induces chemo/radio-sensitization in ovarian cancer cells and curcumin nanoparticles inhibit ovarian cancer cell growth. J Ovar Res. 2010;3:11. doi: 10.1186/1757-2215-3-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Larson N, Yang J, Ray A, Cheney DL, Ghandehari H, Kopecek J. Biodegradable multiblock poly(N-2-hydroxypropyl)methacrylamide gemcitabine and paclitaxel conjugates for ovarian cancer cell combination treatment. Int J Pharm. 2013;454:435–43. doi: 10.1016/j.ijpharm.2013.06.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Gao X, Wang B, Wei X, Men K, Zheng F, Zhou Y, Zheng Y, Gou M, Huang M, Guo G, Huang N, Qian Z, Wei Y. Anticancer effect and mechanism of polymer micelle-encapsulated quercetin on ovarian cancer. Nanoscale. 2012;4:7021–30. doi: 10.1039/c2nr32181e. [DOI] [PubMed] [Google Scholar]
- 193.Colson YL, Liu R, Southard EB, Schulz MD, Wade JE, Griset AP, Zubris KA, Padera RF, Grinstaff MW. The performance of expansile nanoparticles in a murine model of peritoneal carcinomatosis. Biomaterials. 2011;32:832–40. doi: 10.1016/j.biomaterials.2010.09.059. [DOI] [PubMed] [Google Scholar]
- 194.Sengupta P, Basu S, Soni S, Pandey A, Roy B, Oh MS, Chin KT, Paraskar AS, Sarangi S, Connor Y, Sabbisetti VS, Kipparam J, Kulkarni A, Muto K, Amarasiriwardena C, Jayawardene I, Lupoli N, Dinulescu DM, Bonventre JV, Mashelkar RA, Sengupta S. Cholesterol-tethered platinum II-based supramolecular nanoparticle increases antitumor efficacy and reduces nephrotoxicity. Proc Natl Acad Sci U S A. 2012;109:11294–9. doi: 10.1073/pnas.1203129109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Perche F, Patel NR, Torchilin VP. Accumulation and toxicity of antibody-targeted doxorubicin-loaded PEG-PE micelles in ovarian cancer cell spheroid model. J Control Release. 2012;164:95–102. doi: 10.1016/j.jconrel.2012.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Werner ME, Karve S, Sukumar R, Cummings ND, Copp JA, Chen RC, Zhang T, Wang AZ. Folate-targeted nanoparticle delivery of chemo- and radiotherapeutics for the treatment of ovarian cancer peritoneal metastasis. Biomaterials. 2011;32:8548–54. doi: 10.1016/j.biomaterials.2011.07.067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Chaudhury A, Das S, Bunte RM, Chiu GN. Potent therapeutic activity of folate receptor-targeted liposomal carboplatin in the localized treatment of intraperitoneally grown human ovarian tumor xenograft. Int J Nanomed. 2012;7:739–51. doi: 10.2147/IJN.S26172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Kim D, Lee ES, Oh KT, Gao ZG, Bae YH. Doxorubicin-loaded polymeric micelle overcomes multidrug resistance of cancer by double-targeting folate receptor and early endosomal pH. Small. 2008;4:2043–50. doi: 10.1002/smll.200701275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Zhang XY, Chen J, Zheng YF, Gao XL, Kang Y, Liu JC, Cheng MJ, Sun H, Xu CJ. Follicle-stimulating hormone peptide can facilitate paclitaxel nanoparticles to target ovarian carcinoma in vivo. Cancer Res. 2009;69:6506–14. doi: 10.1158/0008-5472.CAN-08-4721. [DOI] [PubMed] [Google Scholar]
- 200.Zhang X, Chen J, Kang Y, Hong S, Zheng Y, Sun H, Xu C. Targeted paclitaxel nanoparticles modified with follicle-stimulating hormone beta 81–95 peptide show effective antitumor activity against ovarian carcinoma. Int J Pharm. 2013;453:498–505. doi: 10.1016/j.ijpharm.2013.06.038. [DOI] [PubMed] [Google Scholar]
- 201.Winer I, Wang S, Lee Y-EK, Fan W, Gong Y, Burgos-Ojeda D, Spahlinger G, Kipelman R, Buckanovich RJ. F3-targeted cisplatin-hydrogel nanoparticles as an effective therapeutic that targets both murine and human ovarian tumor endothelial cells in vivo. Cancer Res. 2010;70:8674–83. doi: 10.1158/0008-5472.CAN-10-1917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Zhao H, Wang J-C, Sun Q-S, Luo C-L, Zhang Q. RGD-based strategies for improving antitumor activity of paclitaxel-loaded liposomes in nude mice xenografted with human ovarian cancer. J Drug Target. 2009;17:10–8. doi: 10.1080/10611860802368966. [DOI] [PubMed] [Google Scholar]
- 203.Cirstoiu-Hapca A, Bossy-Nobs L, Buchegger F, Gurny R, Delie F. Differential tumor cell targeting of anti-HER2 (Herceptin) and anti-CD20 (Mabthera) coupled nanoparticles. Int J Pharm. 2007;331:190–6. doi: 10.1016/j.ijpharm.2006.12.002. [DOI] [PubMed] [Google Scholar]
- 204.Cirstoiu-Hapca A, Buchegger F, Bossy L, Kosinski M, Gurny R, Delie F. Nanomedicines for active targeting: physico-chemical characterization of paclitaxel-loaded anti-HER2 immunonanoparticles and in vitro functional studies on target cells. Eur J Pharm Sci. 2009;38:230–7. doi: 10.1016/j.ejps.2009.07.006. [DOI] [PubMed] [Google Scholar]
- 205.Cirstoiu-Hapca A, Buchegger F, Lange N, Bossy L, Gurny R, Delie F. Benefit of anti-HER2-coated paclitaxel-loaded immuno-nanoparticles in the treatment of disseminated ovarian cancer: therapeutic efficacy and biodistribution in mice. J Control Release. 2010;144:324–31. doi: 10.1016/j.jconrel.2010.02.026. [DOI] [PubMed] [Google Scholar]
- 206.Wang Y, Liu P, Qiu L, Sun Y, Zhu M, Gu L, Di W, Duan Y. Toxicity and therapy of cisplatin-loaded EGF modified mPEG-PLGA-PLL nanoparticles for SKOV3 cancer in mice. Biomaterials. 2013;34:4068–77. doi: 10.1016/j.biomaterials.2012.12.033. [DOI] [PubMed] [Google Scholar]
- 207.Krieger ML, Eckstein N, Schneider V, Koch M, Royer H-D, Jaehde U, Bendas G. Overcoming cisplatin resistance of ovarian cancer cells by targeted liposomes in vitro. Int J Pharm. 2010;389:10–7. doi: 10.1016/j.ijpharm.2009.12.061. [DOI] [PubMed] [Google Scholar]
- 208.Xing X, Zhang S, Chang JY, Tucker SD, Chen H, Huang L, Hung MC. Safety study and characterization of E1A-liposome complex gene-delivery protocol in an ovarian cancer model. Gene Ther. 1998;5:1538–44. doi: 10.1038/sj.gt.3300771. [DOI] [PubMed] [Google Scholar]
- 209.Huang YH, Zugates GT, Peng W, Holtz D, Dunton C, Green JJ, Hossain N, Chernick MR, Padera RF, Jr, Langer R, Anderson DG, Sawicki JA. Nanoparticle-delivered suicide gene therapy effectively reduces ovarian tumor burden in mice. Cancer Res. 2009;69:6184–91. doi: 10.1158/0008-5472.CAN-09-0061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Wang Y, Saad M, Pakunlu RI, Khandare JJ, Garbuzenko OB, Vetcher AA, Soldatenkov VA, Pozharov VP, Minko T. Nonviral nanoscale-based delivery of antisense oligonucleotides targeted to hypoxia-inducible factor 1 alpha enhances the efficacy of chemotherapy in drug-resistant tumor. Clin Cancer Res. 2008;14:3607–16. doi: 10.1158/1078-0432.CCR-07-2020. [DOI] [PubMed] [Google Scholar]