Key Points
Exosome complex components are endogenous suppressors of erythroid cell maturation.
GATA-1 and Foxo3 transcriptionally repress exosome complex components, thus abrogating the erythroid maturation blockade.
Abstract
Complex genetic networks control hematopoietic stem cell differentiation into progenitors that give rise to billions of erythrocytes daily. Previously, we described a role for the master regulator of erythropoiesis, GATA-1, in inducing genes encoding components of the autophagy machinery. In this context, the Forkhead transcription factor, Foxo3, amplified GATA-1–mediated transcriptional activation. To determine the scope of the GATA-1/Foxo3 cooperativity, and to develop functional insights, we analyzed the GATA-1/Foxo3-dependent transcriptome in erythroid cells. GATA-1/Foxo3 repressed expression of Exosc8, a pivotal component of the exosome complex, which mediates RNA surveillance and epigenetic regulation. Strikingly, downregulating Exosc8, or additional exosome complex components, in primary erythroid precursor cells induced erythroid cell maturation. Our results demonstrate a new mode of controlling erythropoiesis in which multiple components of the exosome complex are endogenous suppressors of the erythroid developmental program.
Introduction
Hematopoietic stem cells give rise to lineage-committed progenitors that differentiate into erythroid precursors termed erythroblasts. Erythroblasts undergo sequential maturation steps, culminating in enucleation to yield reticulocytes and subsequently erythrocytes.1,2 Despite sophisticated knowledge of the underlying mechanisms, major questions remain including: how are the genetic networks established/maintained, how is the massive cellular remodeling orchestrated, and do endogenous factors restrict the traversal from one maturation step to the next, in opposition to factors that promote the process?
A restricted cohort of transcription factors, including GATA-1, KLF1, and Scl/TAL1,3,4 are responsible for establishing and/or maintaining the expression of genes that endow the red cell with its unique identity. GATA-1 interacts with the coregulator FOG-1, which mediates activation and repression of most, but not all, GATA-1 target genes.5-7 GATA-1 functions with Scl/TAL1 and the non-DNA–binding proteins LDB1 and LMO2 in a pentameric complex to regulate select target genes.8-11 GATA-1 target genes can have distinct coregulator requirements at endogenous loci.7 We demonstrated that the Forkhead transcription factor Foxo3 amplifies GATA-1–mediated transcriptional activation of genes encoding autophagy machinery components.12
The Forkhead transcription factor Foxo3 regulates oxidative stress response genes in erythroblasts.13 GATA-1 directly upregulates Foxo3 expression, which amplifies GATA-1 activity to regulate autophagy gene expression.12 Herein, we describe genomic studies to establish the scope of the GATA-1/Foxo3 coregulated genetic network in erythroid cells. We aimed to distinguish between models in which Foxo3 amplifies GATA-1 activity at canonical GATA-1 target genes encoding fundamental erythroid cell components or at more specialized genes. This analysis revealed a broader scope of the mechanism and pinpointed genes encoding components of the exosome complex, which were GATA-1/Foxo3-repressed.
The exosome complex mediates degradation of aberrant pre-mRNAs and mRNAs and is required for the processing/degradation of pre-rRNAs, pre-snRNAs, and pre-tRNAs.14-16 Beyond RNA processing/degradation, the complex regulates transcription start site usage, maintains a heterochromatin mark (trimethylation of lysine 9 on histone H3), and regulates noncoding RNA expression.17-22 The exosome complex and chromatin insulator components cooccupy boundary elements and promoters in Drosophila.23
Although the exosome complex is implicated in diverse molecular processes, whether it controls essential biological processes is largely unexplored. Our loss-of-function analysis revealed a mechanism in which the core exosome components Exosc8 (Rrp43) and Exosc9 (Rrp45) and catalytic subunit Dis3 (Rrp44) create a blockade to primary erythroid cell maturation. Disruption of this blockade induced erythroid maturation to yield late-stage erythroblasts and reticulocytes. The exosome complex had not been linked to hematopoiesis or erythroid biology. The identification of exosome complex components as endogenous suppressors of erythroid maturation reveals a novel avenue for controlling erythroid maturation.
Methods
Cell culture
G1E-ER-GATA-1 cells were cultured with or without 1 μM estradiol (Steraloids, Inc.) as described.24 Fetal liver erythroid precursors were cultured in StemPro-34 (Gibco) supplemented with 10% nutrient supplement (Gibco), 2 mM l-glutamine (Cellgro), 1% penicillin-streptomycin (Cellgro), 100 μM monothioglycerol (Sigma-Aldrich), 1 μM dexamethasone (Sigma-Aldrich), 0.5 U/mL erythropoietin, and 1% conditioned medium from a kit ligand-producing CHO cell line for expansion. For differentiation, cells were cultured in ES IMDM (glutamine-free) (Hyclone) containing 10% fetal bovine serum (FBS) (Gemini), 10% plasma derived serum (PDS; Animal Technologies), 5% protein free hybridoma medium II (PFHM II; Gibco), 2mM l-glutamine (Cellgro), 1% penicillin-streptomycin (Cellgro), 100 μM monothioglycerol (Sigma-Aldrich), and 5 U/mL of erythropoietin. Cells were cultured in a humidified incubator at 37°C (5% carbon dioxide).
Quantitative real-time reverse transcriptase polymerase chain reaction
cDNA was analyzed in reactions (20 μL) containing 2 μL of cDNA, appropriate primers, and 10 μL of SYBR green master mix (Applied Biosystems). Product accumulation was monitored by SYBR green fluorescence using a StepOnePlus or Viia 7 real-time PCR instrument (Applied Biosystems). A standard curve of serial dilutions of cDNA samples was used to determine relative expression. mRNA levels were normalized to 18S rRNA.
Protein analysis
Equal numbers of cells boiled for 10 minutes in sodium dodecyl sulfate (SDS) sample buffer were resolved by SDS-polyacrylamide gel electrophoresis and analyzed by semiquantitative western blotting with ECL Plus (Pierce) with antibodies described in the supplemental Methods, available on the Blood Web site.
Quantitative chromatin immunoprecipitation (ChIP) assay
ChIP was conducted as described12 with antibodies indicated in the supplemental Methods. Samples were analyzed by real-time PCR (ABI StepOnePlus) as described.24 GATA-1 ChIP-seq profiles in primary human erythroblasts were generated from our published dataset (Gene Expression Omnibus, GSE32491).
Primary erythroid precursor cell isolation
Primary erythroid precursors were isolated from E14.5 fetal livers using the EasySep negative selection Mouse Hematopoietic Progenitor Cell Enrichment Kit (StemCell Technologies). Fetal liver cells were resuspended at 5 × 107 cells/mL in phosphate-buffered saline (PBS) containing 2% FBS, 2.5 mM ethylenediamine tetraacetic acid (EDTA), and 10 mM glucose, and EasySep Mouse Hematopoietic Progenitor Cell Enrichment Cocktail was added at 50 μL/mL supplemented with 2.5 μg/mL biotin-conjugated CD71 antibody (eBioscience). After 15 minutes on ice, the cells were washed by centrifugation for 5 minutes at 1200 rpm at 4°C and resuspended at 5 × 107 cells/mL in PBS containing 2% FBS, 2.5 mM EDTA, and 10 mM glucose, and EasySep Biotin Selection Cocktail was added at 100 μl/mL. After 15 minutes at 4°C, EasySep Mouse Progenitor Magnetic Microparticles were added at 50 μL/mL. After 10 minutes at 4°C, cells were resuspended to 2.5 mL and incubated with a magnet for 3 minutes. Unbound cells were analyzed.
siRNA/shRNA-mediated knockdown
Dharmacon siGENOME Smartpools against mouse Foxo3 and Exosc8 were used with nontargeting siRNA pool as a control. siRNA (240 pmol) was transfected into 3 × 106 of G1E-ER-GATA-1 cells using the Lonza Nucleofector Kit R with an Amaxa Nucleofector II (Lonza). siRNA was transfected twice at 0 and 24 hours. G1E-ER-GATA-1 cells were treated with estradiol 6 hours after the first nucleofection for 42 hours (Foxo3) or 12 hours after the second nucleofection for 12 hours (Exosc8).
MiR-30 context Exosc8 (Rrp43), Exosc9 (Rrp45), and Dis3 (Rrp44) shRNAs were cloned into MSCV-PIG vector (kindly provided by Dr Mitchell Weiss) using Bgl II and Xho I restriction sites. 1 × 105 erythroid precursors were spinfected with 100 μL of retrovirus supernatant and 8 μg/mL polybrene in 400 μL of expansion media at 1200g for 90 minutes at 30°C. shRNA sequences are described in the supplemental Methods.
Flow cytometry
PBS-washed cells (1 × 106) were stained with 0.8 μg of anti-mouse Ter119-APC and anti-mouse CD71-PE (eBioscience) at 4°C for 30 minutes in the dark. Stained cells were washed 3 times with 2% bovine serum albumin in PBS. For Exosc8 knockdowns, samples were analyzed using a BD LSR II (BD Biosciences). For Exosc9 knockdowns (with Exosc8 knockdown as a control), Ter119 and CD71 staining was analyzed using a BD FACSAria II (BD Biosciences). shRNA-expressing R1, R2, R3, and R4/5 cells were sorted from the total population using the green fluorescent protein marker coexpressed with the shRNA and the Ter119 and CD71 expression profile. DAPI (Sigma-Aldrich) staining discriminated dead cells.
For cell cycle analysis, cells were resuspended at 5 × 105/mL in medium containing 20 µg/mL Hoechst 33342 (Invitrogen), incubated at 37°C for 30 minutes, and adjusted to 2 × 106 cells/mL. For analysis of flow-sorted R3 cells and cells treated with hydroxyurea (HU), 0.5 to 1 million cells were washed in PBS before being resuspended in 300 μL of cold PBS and fixed by adding 900 μL of 70% cold ethanol drop-wise. Cells were incubated at 4°C overnight, washed twice in PBS, and stained overnight in 100 μL of 2 μg/mL DAPI in PBS. Stained cells were resuspended in 500 μL PBS. DNA content was measured using a BD LSR II (BD Biosciences) and Modfit LT 3.2.1 (Verity Software).
Transcriptional profiling
Amino Allyl RNA was synthesized from mRNA, labeled, and hybridized to 8 × 60K Mouse Whole Genome arrays (Agilent) (3 biological replicates). Arrays were read using a G-2505C DNA Microarray Scanner with Surescan High Resolution (Agilent). EDGE3 web-based 1-color microarray analysis software25 was used for data analysis. The data are available at the Gene Expression Omnibus (GEO) under accession numbers GSE49174 (G1E-ER-GATA-1), GSE60136 (EXOSC8), and GSE60137 (FOXO3).
Generation of 3D exosome complex structures
Protein-structure coordinate files for the human exosome complex26 were downloaded from the Research Collaboratory for Structural Bioinformatics Protein Data Bank (www.RCSB.org, accession number 2NN6). Images were generated using PyMOL (www.PyMOL.org).
Statistics
JMP (version 11, SAS Institute Inc., Cary, NC) was used for statistical analysis. Student t test to determine significance was used to compare experimental samples to controls. Asterisks indicate significance relative to the control. EDGE3 analysis software25 and the National Institutes of Health DAVID tool (http://david.abcc.ncifcrf.gov/) were used for statistical analysis of microarray data and gene ontology analysis, respectively.
Results
GATA-1/Foxo3 cooperativity in erythroid cell biology
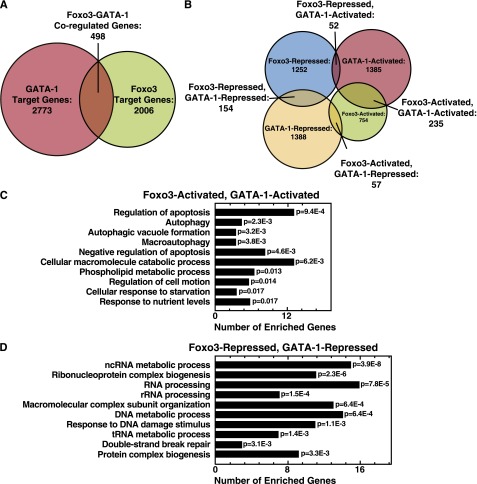
Foxo3 amplifies GATA-1 activity to induce autophagy gene expression in erythroid cells.12 To determine the extent of the GATA-1/Foxo3-regulated genetic network in erythroid cells, we profiled the transcriptome of Foxo3-knockdown vs control cells in the context of active GATA-1. Comparison of Foxo3-regulated genes to our GATA-1 target gene dataset in G1E-ER-GATA-1 cells7 revealed 498 of the 2006 Foxo3-regulated genes (∼25%) as GATA-1-regulated, indicating that GATA-1/Foxo3 coregulate a cohort of genes in erythroid cells beyond the autophagy genes (Figure 1A).
Figure 1.
GATA-1/Foxo3-dependent genetic network. (A) Venn diagrams depicting genes regulated uniquely or coregulated by GATA-1 and Foxo3. (B) Venn diagrams demonstrating relationships between GATA-1– and Foxo3-activated and -repressed genes. (C) Gene ontology analysis of GATA-1/Foxo3-coactivated genes. (D) Gene ontology analysis of GATA-1– and Foxo3-corepressed genes. The top 10 gene ontology categories are displayed and ordered by P value. Redundant gene ontology categories were curated and removed.
We classified GATA-1– or Foxo3-regulated genes as activated or repressed. GATA-1 and Foxo3 regulated 78% of the coregulated genes in the same direction (Figure 1B). GATA-1/Foxo3 coactivated and corepressed 235 and 154 genes, respectively (Figure 1B). Gene ontology (GO) analysis was conducted to assess whether these distinct modes have hallmark functional signatures. The top 5 GO terms ascribed to the coactivated genes implicate regulation of apoptosis and autophagy (Figure 1C) as indicated by our prior work.12 Genes involved in “non-coding RNA metabolic process” or “RNA processing” categories were enriched in the corepressed cohort (Figure 1D). Genes within these categories (listed in supplemental Table 1) were not predictable based on prior analyses of GATA factor function or hematopoiesis.
Linking the GATA-1/Foxo3 genetic network to RNA surveillance machinery
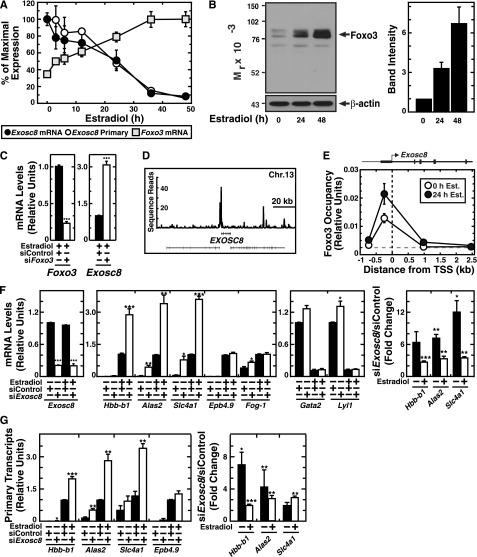
Two of the highly GATA-1/Foxo3-repressed genes classified in the “non-coding RNA metabolic process” category were Exosc8 (Rrp43) and Exosc5 (Rrp46), essential core components of the exosome complex.15 Using real-time RT-PCR to confirm the microarray data, GATA-1 repressed Exosc8 expression 25-fold, and knocking down Foxo3 de-repressed Exosc8 expression threefold (Figure 2A,C). In primary human erythroblasts, GATA-1 occupied the EXOSC8 promoter (Figure 2D) and Foxo3 occupied the Exosc8 promoter (Figure 2E), suggesting direct transcriptional regulation. The kinetics of GATA-1–mediated Exosc8 repression are slower than prototypical GATA-1–repressed target genes and resemble the slow induction of the direct GATA-1 target Hbb-b1, which has been attributed to a GATA-1 requirement to upregulate FOG-1.27 The kinetics of Foxo3 mRNA induction (maximal after 36 hours β-estradiol treatment) were similar to the slow kinetics of Exosc8 repression (Figure 2A). Foxo3 protein was induced gradually over 48 hours of β-estradiol treatment (Figure 2B). These data suggest that GATA-1 upregulates Foxo3 as a prelude to functioning combinatorially with Foxo3 to confer maximal Exosc8 repression.
Figure 2.
GATA-1/Foxo3-mediated repression of Exosc8 expression. (A) Real-time RT-PCR analysis of Foxo3, Exosc8 mRNA and primary transcripts upon GATA-1 activation in G1E-ER-GATA-1 cells (mean ± SE; 3 independent experiments). Values were normalized to 18S rRNA expression. (B) Semiquantitative western blot analysis of Foxo3 in G1E-ER-GATA-1 cells after 0, 24, or 48 hours estradiol treatment (left). The representative image is shown from 3 independent experiments. Quantitation of band intensity (mean ± SE; 3 independent experiments) (right). (C) Real-time RT-PCR analysis of Foxo3 and Exosc8 mRNA upon Foxo3 knockdown in G1E-ER-GATA-1 cells after 42 hours activation of GATA-1 (mean ± standard error [SE]; 4 independent experiments). Values were normalized to 18S rRNA expression and expression is shown relative to the control. (D) ChIP-seq profile of GATA-1 occupancy at EXOSC8 in primary human erythroblasts. (E) ChIP analysis of Foxo3 occupancy at the Exosc8 promoter in untreated and estradiol-treated (24 hours) G1E-ER-GATA-1 cells (mean ± SE; 4 independent experiments). (F) Real-time RT-PCR analysis of mRNA levels in control vs Exosc8 knockdown G1E-ER-GATA-1 cells (mean ± SE; 3 independent experiments). Values were normalized to 18S rRNA expression. mRNA levels are shown relative to the control siRNA with estradiol treatment of activated genes (left) and without estradiol treatment of repressed genes (middle). Fold changes upon Exosc8 knockdown relative to control (right). (G) Real-time RT-PCR analysis of primary transcripts in control vs Exosc8 knockdown G1E-ER-GATA-1 cells (mean ± SE; 3 independent experiments). Values were normalized to 18S rRNA expression, and the expression is shown relative to estradiol-treated control siRNA. Fold changes are also depicted upon Exosc8 knockdown relative to control (right). *P < .05, **P < .01, ***P < .001.
Exosc8 regulates a cohort of GATA-1 target genes
To investigate the consequences of GATA-1–mediated Exosc8 repression, we conducted loss-of-function analysis in G1E-ER-GATA-1 cells. Knocking down Exosc8 by ∼80% enhanced expression of several GATA-1–activated genes three- to 12-fold (Hbb-b1, Alas2, and Slc4a1), whereas other GATA-1–activated genes (Epb4.9 and Fog-1) were largely unaffected. GATA-1–mediated repression of Gata2 and Lyl1 was also unaltered (Figure 2F).
Impairing exosome complex mRNA degradation function by disrupting a core subunit could underlie mRNA accumulation. To determine whether mRNA alterations upon Exosc8 downregulation resulted solely from altered mRNA degradation, we quantitated primary unprocessed transcripts for Exosc8-responsive, GATA-1–activated genes, using the Exosc8-unresponsive gene Epb4.9 as a control. Hbb-b1, Alas2, and Slc4a1 primary transcripts increased two- to sevenfold, whereas Epb4.9 primary transcripts were insensitive (Figure 2G). Thus, Exosc8 suppression of a select cohort of erythroid mRNAs does not involve an exclusive mRNA degradation mechanism.
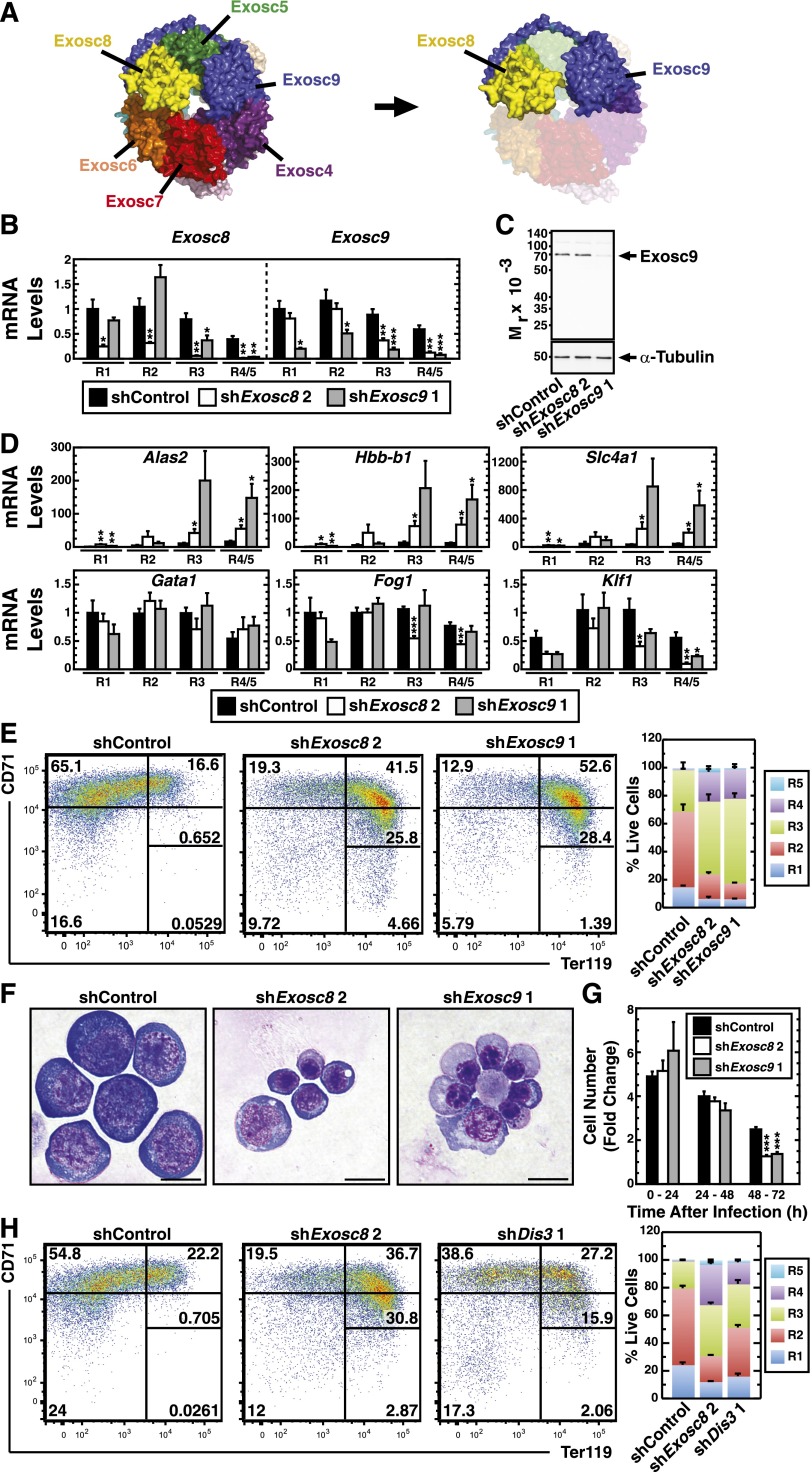
Exosome complex structure and expression during primary erythroblast maturation
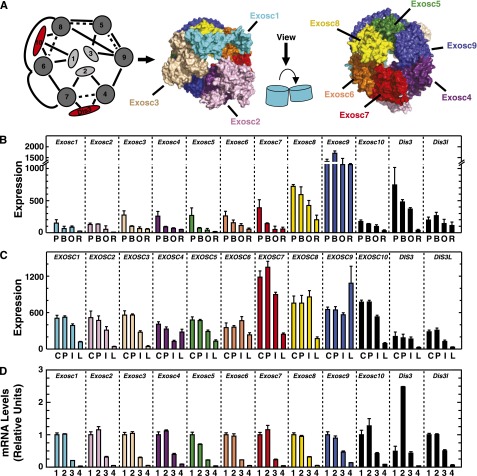
The exosome complex consists of 9 core subunits (Exosc1-9). Exosc1 (Csl4), Exosc2 (Rrp4), and Exosc3 (Rrp40) are RNA binding proteins.26,28 Exosc4/Exosc9 (Rrp41/Rrp45), Exosc6/Exosc7 (Mtr3/Rrp42), and yeast, but not human, Exosc5/Exosc8 (Rrp46/Rrp43) form stable dimers.26 Human EXOSC6/EXOSC7/EXOSC8 form a stable trimer (Figure 3A).26,29,30 Exosc1 interacts with Exosc6, Exosc2 interacts with Exosc4 and Exosc7, and Exosc3 interacts with Exosc5 and Exosc9 to stabilize subunit interactions within the complex26 (Figure 3A). Three catalytic subunits, Exosc10 (Rrp6), Dis3 (Rrp44), and Dis3l (human homolog of yeast Dis3, not found in yeast) bind the core exosome.26,28 Exosc10 and Dis3l are 3′ to 5′ exoribonucleases,31-33 whereas Dis3 exhibits endoribonuclease and exoribonuclease activity.31 Dis3 is mainly associated with the nuclear exosome complex and mediates rRNA and PROMT (promoter upstream transcript) degradation.32,34 Cytoplasmic Dis3l selectively degrades mRNAs.32,34 Exosc10 binds Exosc1, Exosc6, and Exosc828 (Figure 3A) and improves exosome complex activity to process substrates using the long RNA-binding pathway by positioning Exosc1 correctly to bind RNA.28 Exosc10 is enriched in the nucleolus, indicating a role in pre-rRNA processing,34 and a small percentage is cytoplasmic.32 Dis3 or Dis3l binds the exosome core opposite of Exosc10. The Dis3 (or Dis3l) N-terminus wedges between Exosc4 and Exosc7 and approaches the Exosc2 N-terminus (Figure 3A).28
Figure 3.
Repression of exosome-complex component expression during erythroid maturation. (A) Schematic diagram and crystal structure of the human exosome complex.26 Solid line, direct interactions; dashed line, indirect interactions; red, associated components with catalytic activity. (B) Expression profile of mRNA levels for exosome complex components in primary mouse bone marrow erythroblasts during distinct maturation stages mined from the Erythron DB.29 P, proerythroblast; B, basophilic erythroblast; O, orthochromatic erythroblast; R, reticulocyte. (C) Expression profile of mRNAs encoding exosome-complex components during primary human erythroid differentiation mined from the Human Erythroblast Maturation (HEM) Database.30 C, colony-forming unit-erythroid (CFU-E); P, proerythroblast; I, intermediate-stage erythroblast; L, late-stage erythroblast. (D) Expression profile of mRNAs encoding exosome-complex components during ex vivo differentiation of primary mouse fetal liver–derived erythroid precursor cells. 1, R1 population: progenitor cells; 2, R2 population: proerythroblasts and early basophilic erythroblasts; 3, R3 population: early and late basophilic erythroblasts. 4, R4/5 population: polychromatophilic, orthochromatic erythroblasts, and reticulocytes.
To assess whether Exosc8 expression in G1E-ER-GATA-1 cells reflects its behavior in primary cells, we mined genomic databases29,30 and determined the expression of exosome components in primary erythroblasts. During definitive erythropoiesis in the mouse (Figure 3B) and human (Figure 3C), expression of exosome complex components, including Exosc8, decreased. RT-PCR analysis revealed decreased exosome complex component expression during ex vivo differentiation of mouse fetal liver erythroid precursors (Figure 3D). The direct Exosc8 repression by GATA-1/Foxo3, and Exosc8 repression upon erythroid maturation suggests important exosome complex functions in erythroid biology.
Exosc8 suppresses primary erythroblast maturation
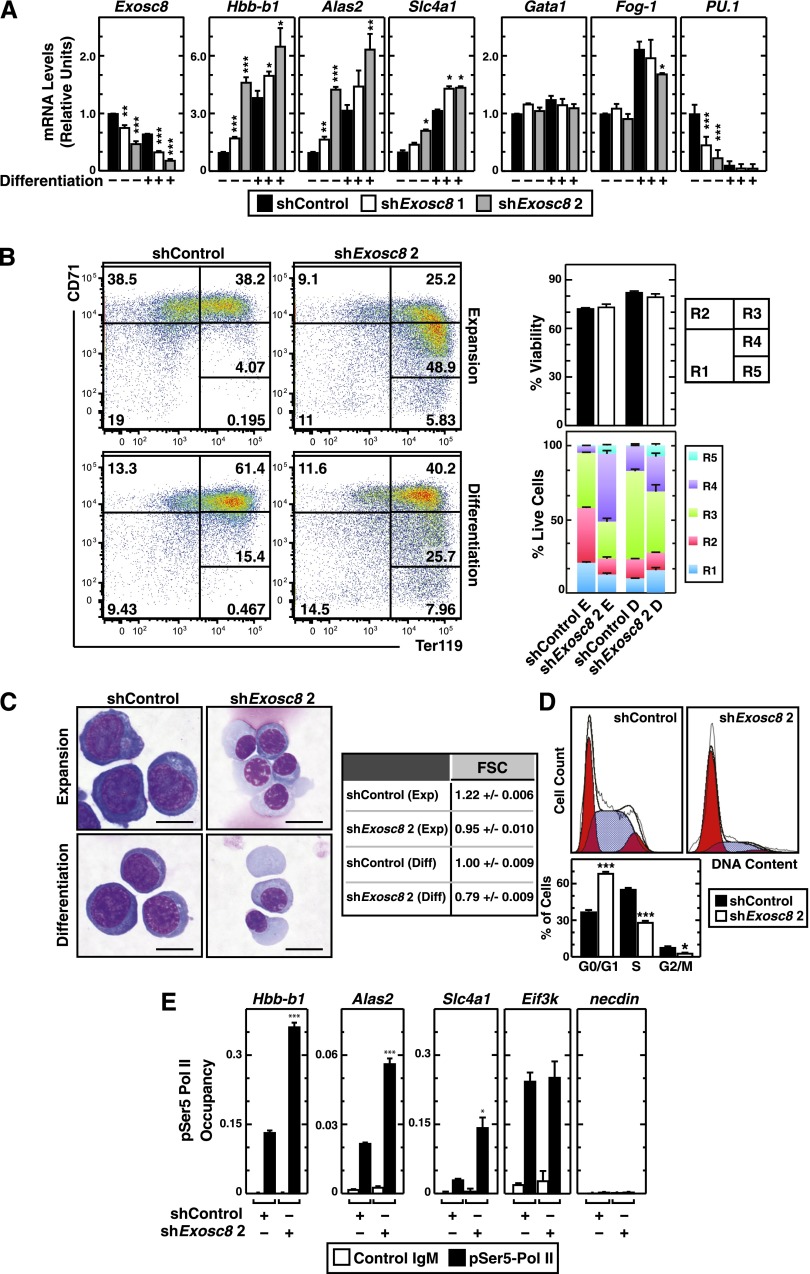
We tested whether Exosc8 suppresses a cohort of erythroid-specific RNAs in primary murine erythroid precursor cells isolated from E14.5 fetal livers. Cells were infected with 2 distinct Exosc8 shRNA-expressing retroviruses (with luciferase shRNA as a control), expanded for 3 days, and differentiated for 1 day. The Exosc8 shRNAs induced qualitatively similar results. Exosc8 shRNA upregulated Hbb-b1, Alas2, and Slc4a1 expression, whereas GATA-1 and Fog-1 expressions were largely unaffected during expansion conditions (Figure 4A). Expression of PU.1, which opposes GATA-1 activity in certain contexts,3 decreased in the Exosc8-knockdown cells under expansion conditions (Figure 4A). These data suggest that GATA-1–mediated repression of Exosc8 during erythropoiesis contributes to expression of GATA-1 target genes important for erythroid maturation.
Figure 4.
Exosc8-dependent barrier to erythroid maturation. (A) Real-time RT-PCR analysis of Exosc8 and selected GATA-1 target gene mRNA levels in control vs Exosc8 knockdown primary murine erythroid precursor cells cultured under expansion (–) or differentiation conditions (+) (mean ± SE; 3 independent experiments). Values were normalized to 18S rRNA expression and the expression is shown relative to control shRNA under expansion conditions. (B) Flow cytometric quantitation of erythroid developmental stage by CD71 and Ter119 staining upon Exosc8 knockdown in primary erythroid precursor cells. Representative flow cytometry data, with the R1-R5 gates denoted from 3 independent experiments. The percentage of live cells from each condition and the cell populations in R1-R5 stages (mean ± SE; 3 independent experiments). E, expansion; D, differentiation. (C) Representative images of Wright-Giemsa staining in control vs Exosc8 shRNA–infected primary erythroid precursor cells cultured in expansion or differentiation media (scale bar = 10 µm) and quantitation of cell size by measuring forward scatter using flow cytometry. (D) Flow cytometric cell-cycle analysis of primary erythroid precursor cells infected with retrovirus expressing control or Exosc8 2 shRNA. Representative cell-cycle profile is shown from 3 independent experiments. The percentage of the cell population in each cell-cycle stage is from 3 independent experiments (mean ± SE). Blue, S phase; red, G0/G1 or G2/M phase. (E) Quantitative ChIP analysis of serine 5-phosphorylated RNA Polymerase II occupancy at Exosc8-regulated GATA-1 target and control genes in control and Exosc8-knockdown primary murine erythroid precursor cells (mean ± SE; 3 independent experiments). *P < .05, **P < .01, ***P < .001.
Exosc8 knockdown cells were smaller and red, indicating hemoglobinization, vs control cells, even when differentiation was not induced. We assessed maturation of control and Exosc8-knockdown cells using flow cytometry with erythroid markers (Ter119 and CD7135) and Wright-Giemsa staining. In expansion culture, most cells represented R2 (proerythroblasts and early basophilic erythroblasts) and R3 (early and late basophilic erythroblasts) populations in the control (Figure 4B). The Exosc8 knockdown induced a shift from R2 to R3 and R4 (polychromatophilic and orthochromatic erythroblasts) and even to R5 (late orthochromatic erythroblasts and reticulocytes) populations, without influencing viability, in every condition tested (Figure 4B). Exosc8-knockdown cells exhibited features of mature erythroblasts and reticulocytes. The knockdown induced accumulation of cells with more condensed nuclei, and the cytoplasmic color became slate blue, indicating hemoglobinization (Figure 4C). Forward scatter measurements confirmed the smaller size of Exosc8-knockdown cells (Figure 4C). Exosc8 knockdown induced cell-cycle exit, increasing G0/G1 populations approximately twofold and decreasing S and G2/M phase cells approximately twofold, as expected for erythroid maturation (Figure 4D).36-38
The Exosc8 knockdown would be expected to impair exosome complex activity to degrade certain mRNAs, thereby posttranscriptionally upregulating mRNAs. However, the Exosc8 knockdown also increased primary transcripts (Figure 2G), suggesting a mechanism not restricted to mRNA degradation. We explored the possibility that elevated primary transcripts upon Exosc8 knockdown reflects increased transcription. The exosome complex can function via epigenetic regulation39 and noncoding RNA surveillance,40 which impact transcription. We tested whether Exosc8 regulates serine 5-phosphorylated RNA Polymerase II (pSer5-Pol II) occupancy at the affected promoters. Quantitative ChIP analysis in murine erythroid precursors revealed that the Exosc8 knockdown elevated pSer5-Pol II occupancy at Hbb-b1, Alas2, and Slc4a1 promoters, but not at the active Eif3k promoter and the repressed Necdin promoter, indicating that Exosc8 represses transcription of these erythroid genes (Figure 4E). In aggregate, these data demonstrate that Exosc8 suppresses maturation of primary erythroid precursor cells.
The exosome complex as an endogenous suppressor of erythroid maturation
To test whether Exosc8 suppresses erythroid maturation by functioning within or independent of the exosome complex, we assessed whether another exosome component suppresses maturation. Exosc9 was prioritized because of its integral position within the complex. Exosc9 binds Exosc4 and interacts with Exosc8 via its C-terminal tail, which interfaces with Exosc5 and wraps around Exosc5 and Exosc8 (Figure 5A).26 We infected fetal liver erythroid precursors with 2 distinct Exosc9 shRNA-expressing retroviruses and expanded the cells for 3 days. Knockdown of Exosc9 upregulated Slc4a1, Alas2, and Hbb-b1 mRNA, whereas mRNAs for the erythroid transcription factors Gata1 and Fog1 were unaffected (supplemental Figure 1A).
Figure 5.
Evidence that the exosome complex creates a barrier to erythroid maturation. (A) Crystal structure of the human exosome complex26 demonstrating the interaction between Exosc8 and Exosc9. (B) Real-time RT-PCR analysis of Exosc8 and Exosc9 mRNA levels in control vs Exosc8 or Exosc9 knockdown in expanding primary murine erythroid precursor cells sorted into distinct, R1, R2, R3, and R4/5 cell populations (mean ± SE; 5 independent experiments). Values were normalized to 18S rRNA level and the expression relative to the control R1 shRNA condition. (C) Semiquantitative western blot analysis of Exosc9 protein in expanding primary murine erythroid precursor cells 24 hours postinfection with Exosc8- or Exosc9-specific shRNA retroviruses. Representative image from 3 independent experiments. (D) Real-time RT-PCR analysis of selected GATA-1 target genes and erythroid transcription factors mRNA levels in control vs Exosc8 or Exosc9 knockdown in expanding primary murine erythroid precursor cells sorted into distinct, R1, R2, R3, and R4/5 cell populations (mean ± SE; 5 independent experiments). Values were normalized to 18S rRNA level and the expression relative to the control R1 shRNA condition. (E) Flow cytometric quantitation of erythroid maturation stage by CD71 and Ter119 staining upon Exosc8 or Exosc9 knockdown in primary erythroid precursor cells. Representative flow cytometry data, with the R1-R5 gates denoted from 4 independent experiments. The percentage of the cell populations in R1-R5 stages (mean ± SE; 4 independent experiments). (F) Representative images of Wright-Giemsa–stained cells infected with control vs Exosc8 or Exosc9 shRNA-expressing virus. Cells were cultured under expansion conditions (scale bar = 10 µm). (G) Cell number fold change during the 72-hour expansion of primary murine erythroid precursor cells postinfection with control, Exosc8, or Exosc9 shRNA-expressing retroviruses (mean ± SE; 8 independent experiments). (H) Flow cytometric quantitation of erythroid maturation stage by CD71 and Ter119 staining upon Exosc8 or Dis3 knockdown in primary erythroid precursor cells. Representative flow cytometry data, with the R1-R5 gates denoted. The percentage of the cell populations in R1-R5 stages (mean ± SE; 6 independent experiments. *P < .05, **P < .01, ***P < .001.
To address whether the increased erythroid gene mRNA levels upon Exosc8, or Exosc9 knockdown reflects increased mature cells, we infected fetal liver erythroid precursors with retroviruses expressing shRNAs targeting Exosc8, Exosc9, or luciferase and sorted cells into R1, R2, R3, and R4/5 populations. Analysis of sorted cells revealed reduced Exosc8 and Exosc9 mRNA in each population (Figure 5B). Exosc9 shRNA, but not Exosc8 shRNA, reduced Exosc9 protein 24 hours postinfection (Figure 5C). Exosc8 or Exosc9 knockdowns upregulated Alas2, Slc4a1, and Hbb-b1 expression in specific erythroid precursors (Figure 5D), indicating that transcript upregulation is not a consequence of altered maturational status in the population. Exosc8 and Exosc9 knockdown did not increase Gata1, Fog1, and Klf1 mRNA levels in the erythroid precursors, inconsistent with an indirect mode of altering gene expression (Figure 5D).
CD71 and Ter119 staining revealed a shift from R2 (pro-erythroblasts and early basophilic erythroblasts) to R3 (early and late basophilic erythroblasts) and R4 (polychromatophilic and orthochromatic erythroblasts) upon Exosc9 knockdown (Figure 5E). Wright-Giemsa staining demonstrated enhanced maturation in the Exosc9 knockdown cells (Figure 5F); a larger proportion of cells were orthrochromatic in the Exosc9 vs Exosc8 knockdown (supplemental Table 2). Exosc8 and Exosc9 knockdowns reduced proliferation (Figure 5G).
To assess the importance of the exosome complex catalytic function, we infected fetal liver erythroid precursors with 2 distinct shRNAs targeting Dis3. Knockdown of Dis3 induced Alas2, Slc4a1, and Hbb-b1 mRNAs without altering Gata1, and Fog1 mRNAs (supplemental Figure 1B). Dis3 knockdown induced a shift from R2 to R3 and R4 cells (Figure 5H). Thus, a catalytic subunit is important for establishing the blockade. Although we cannot unequivocally rule out the possibility that the 3 subunits of the exosome complex block erythroid maturation through exosome complex-independent actions, the common phenotypes strongly suggest that the exosome complex suppresses erythroid maturation.
To further dissect how downregulating Exosc8 promotes erythroid maturation, we compared the transcriptomes of Exosc8-knockdown primary murine erythroid precursors and control cells. We also compared Exosc8-regulated genes with GATA-1– or Foxo3-regulated genes. We classified GATA-1– and Exocs8-regulated genes as repressed or activated. The most frequent mode of coregulation was Exosc8-repressed and GATA-1–activated (Figure 6A). The differentially regulated transcripts included those important for erythroid cell maturation (heme biosynthesis [Alas2], antiapoptosis [Bcl-xL], and the red cell cytoskeleton [Gypa]). The top 3 GO terms for GATA-1–activated/Exosc8–repressed genes were “cell cycle arrest,” regulation of apoptosis,” and “cellular protein catabolic process” (Figure 6B). Analysis of the Exosc8/Foxo3-regulated gene cohort revealed that, like the GATA-1/Exosc8-regulated genes, the most frequent mode of regulation was Exosc8-repressed and Foxo3-activated (Figure 6C). Exosc8 and Foxo3 regulate genes involved in “apoptosis” and “macromolecular metabolism” in maturing erythroid cells (Figure 6D).
Figure 6.
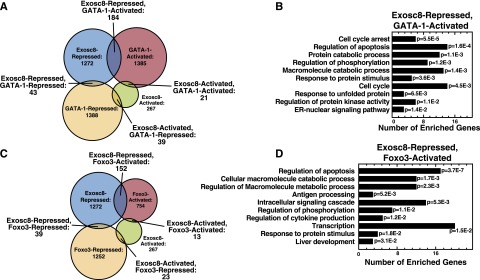
GATA-1/Foxo3/Exosc8 coregulate genes important for erythroid maturation. (A) Classification of GATA-1– and Exosc8-regulated genes based on activation or repression. (B) Gene ontology analysis of GATA-1–activated, Exosc8-repressed genes. Redundant Gene Ontology categories were curated and removed. (C) Classification of Foxo3- and Exosc8-regulated genes based on activation or repression. (D) Gene ontology analysis of Foxo3-activated, Exosc8-repressed genes. Redundant Gene Ontology categories were curated and removed.
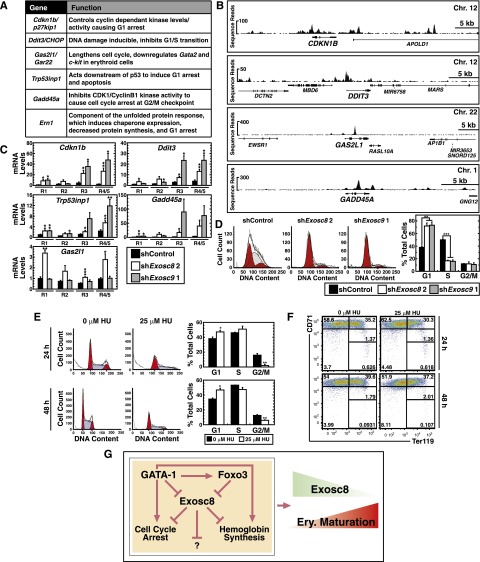
Given the G0/G1 accumulation of Exosc8 knockdown cells (Figure 4D), we explored the GATA-1–activated, Exosc8-repressed genes involved in cell-cycle arrest (Cdkn1b [p27kip1], Ddit3 [CHOP], Gas2l1 [Gar22], Trp53inp1, Gadd45a, and Ern1) (Figure 7A). Cdkn1b controls cyclin-dependent kinases that bind cyclin D and cyclin E, arresting cells in G1.41,42 Ddit3 (CHOP) is a DNA damage–inducible G1/S transition inhibitor.43,44 Trp53inp1 functions downstream of p53 and p73 to induce G1 arrest and to enhance apoptosis.45,46 Ern1, a component of the unfolded protein response, promotes G1 arrest.47-49 The thyroid receptor effector Gas2l1 (Gar22) enhances Gata2 and c-Kit repression during erythroid maturation.50 Gas2l1 is highly expressed in cells arrested in G1,51 and its overexpression lengthens the erythroid cell cycle, especially the S phase.50 Gadd45a responds to genotoxic stress by interacting with CDK1/cyclin B1 to induce G2/M arrest52-54; no role for Gadd45a in erythropoiesis has been reported (Figure 7A).
Figure 7.
The exosome complex suppresses cell-cycle arrest genes during erythroid maturation. (A) Name and function41-43,45,47,52,61 of genes in “cell cycle arrest” category derived from GO term analysis from GATA-1–activated and Exosc8-repressed genes. (B) ChIP-seq profiles of GATA-1 occupancy at cell-cycle regulatory genes in primary human erythroblasts. All genes were orientated left to right. (C) Real-time RT-PCR analysis of genes in “cell cycle arrest” category upon Exosc8 or Exosc9 knockdown in primary murine erythroid precursor cells under expansion conditions, sorted into distinct, R1, R2, R3, and R4/5 cell populations (mean ± SE; 5 independent experiments). Values were normalized to 18S rRNA and the expression relative to the control R1 population. (D) Flow cytometric cell cycle analysis of primary erythroid precursor cells, within the R3 population, infected with retrovirus expressing control, Exosc8, or Exosc9 shRNA. Representative cell-cycle profile from 2 independent experiments. The percentage of the cell population in each cell cycle stage (mean ± SE; 2 independent experiments). Shaded, S phase; red, G0/G1 or G2/M phase. *P < .05, **P < .01, ***P < .001. (E) Flow cytometric cell cycle analysis of primary erythroid precursor cells treated with 25 μM HU for either 24 or 48 hours. Representative cell-cycle profile. The percentage of the cell population in each cell cycle stage (mean ± SE; 3 independent experiments). Blue, S phase; red, G0/G1 or G2/M phase. (F) Flow cytometric quantitation of erythroid maturation stage by CD71 and Ter119 staining upon 25 μM HU treatment of 24 or 48 hours in primary erythroid precursor cells. Representative flow cytometry data, with the R1-R5 gates denoted (3 independent experiments). (G) Model of GATA-1/Foxo3 function to overcome the exosome complex–dependent erythroid maturation barricade, which involves multiple alterations in erythroid cell function.
In primary human erythroblasts, GATA-1 occupied CDKN1B, GAS2L1, and GADD45A introns and CDKN1B, DDIT3, GAS2L1, and GADD45A promoters, suggesting direct GATA-1 regulation of these genes (Figure 7B). Exosc8 knockdown in murine erythroid precursors sorted into R1, R2, R3, and R4/5 populations elevated Cdkn1b, Ddit3, Gas2l1, Trp53inp1, and Gadd45a mRNAs in individual precursor populations (Figure 7C). Thus, upregulation of these mRNAs is not a consequence of altered cellularity. Exosc9 knockdown upregulated the same mRNAs (Figure 7C).
We considered 2 models to explain the cell-cycle arrest resulting from Exosc8 knockdown (Figure 4D). The arrest might result from Exosc8 action in a specific erythroid precursor cell type or from accumulation of noncycling R4 and R5 cells. We conducted cell-cycle analysis with sorted, fetal liver R3 (basophilic erythroblast) cells expressing Exosc8-, Exosc9-, or luciferase shRNA. Exosc8 and Exosc9 knockdown significantly reduced the S-phase and increased G1 cells. Because Exosc8 and Exosc9 regulate the cell cycle in a specific erythroid precursor cell type, the cell-cycle change did not result from altered cellularity (Figure 7D).
Considering the erythroid and cell cycle–regulatory mRNAs suppressed by the exosome complex, the maturation blockade involves a multicomponent mechanism. To determine whether cell-cycle arrest in G1 is sufficient for maturation, we induced G1 arrest in fetal liver erythroid precursors with HU. HU inhibits DNA synthesis, causing cells to accumulate in late G1.55,56 Treatment of erythroid precursors 24 hours postisolation with 25 μM HU for 24 or 48 hours increased G1 and reduced G2/M cells (Figure 7E) without inducing maturation (Figure 7F). Although the Exosc8/Exosc9 knockdown phenotype involves premature cell-cycle arrest, mimicking the arrest with HU did not induce maturation when the exosome complex is unaltered. This is not surprising given the important erythroid functions of other exosome component–regulated mRNAs.
GATA-1 directly repressed Exosc8 expression during maturation, thereby indirectly upregulating Exosc8 targets. Because GATA-1 also directly activated Exosc8-repressed genes, this suggests that GATA-1/Foxo3 abrogate the Exosc8-mediated suppression, activating genes that promote erythroid maturation, including those mediating cell-cycle arrest and hemoglobin synthesis (Figure 7G).
Discussion
Herein, we describe a novel genetic network regulated combinatorially by 2 proteins that control erythropoiesis. In addition to GATA-1/Foxo3-activated autophagy genes,12 this network includes GATA-1/Foxo3-repression of Exosc8. We established a mechanism in which GATA-1/Foxo3 erythroid maturation activity is opposed by exosome complex components. By downregulating these components, GATA-1/Foxo3 overcome an erythroid maturation barrier.
Reducing Exosc8 or Exosc9 levels upregulated mRNAs encoding proteins that mediate multiple aspects of erythroid maturation/function. Although Exosc8/Exosc9 repress maturation of primary erythroid precursor cells, they do not regulate Gata1, Fog1, and Klf1 expression. Exosc8 suppresses select erythroid mRNAs, as well as primary transcripts and pSer5-Pol Il occupancy at erythroid promoters. In Drosophila, exosome complex components colocalize with chromatin insulator factors at boundary elements and also localize to promoters.23 In yeast, the exosome complex localizes to nucleoli and subnuclear regions enriched in actively transcribed genes.57-59 The exosome complex regulates alternative transcription start site selection and may directly affect heterochromatin formation and gene silencing through posttranscriptional regulation.17,18,21,22
GATA-1/Exosc8 coregulate genes involved in cell-cycle arrest, especially those that establish G1 arrest (Figure 7A). Although G1 arrest is one of the hallmarks of late-stage erythroblast differentiation,36,60 this is insufficient to induce late-stage erythroid differentiation (Figure 7E-F). Enforced cell proliferation of late-stage bone marrow erythroblasts leads to decreased hemoglobin production, mitochondria retention, and apoptosis.38 Cdkn1b (p27kip1) (GATA-1–activated/Exosc8-repressed) controls cyclin-dependent kinases (CDK2, CDK4, and CDK6), which bind cyclin D and cyclin E, inducing G1 arrest in multiple cell types.37,41,42,61-63 Cdkn1b appears to be important for erythroid maturation, because it is induced by GATA-136 and accumulates upon terminal erythroid maturation.60 Ectopic Cdkn1b expression promotes erythroid maturation of K562 erythroleukemia cells.41 Although the other GATA-1–activated/Exosc8-repressed G1 arrest genes had not been linked to erythropoiesis, based on their functions in other systems, these genes might contribute to G1 arrest during erythroid maturation.
GATA-1 directly upregulates Foxo3 and functions in concert with Foxo3 to repress Exosc8 expression. Exosc8, functioning in the exosome complex, establishes an erythroid maturation blockade by suppressing GATA-1–activated genes that contribute to essential erythroid cell processes including hemoglobin synthesis and cell-cycle arrest. Reduced expression of exosome complex components, either physiologically through GATA-1/Foxo3-mediated corepression, or experimentally, eliminates this blockade, thus facilitating erythroid maturation (Figure 7G). Because Foxo3 and GATA-2 control HSCs and neural stem/progenitor cells,64-72 we propose that GATA-2-Foxo3 cooperativity may be an important determinant of HSC function.
In summary, we describe unexpected biological functions of the exosome complex and new mechanistic insights into erythropoiesis. We predict that key nodal points in the respective mechanisms are vulnerable to perturbations that cause and/or contribute to red cell disorders, and this work establishes a new framework for controlling erythroid maturation.
Acknowledgments
The authors thank M. J. Weiss for providing retroviral vector, control shRNA virus plasmid, MSCV-PIG vector, and detailed protocols for erythroid precursor cell culture.
This work was supported by National Institutes of Health grants (National Heart, Lung, and Blood Institute) HL116365 (E.H.B., S.G.) and (National Institute of Diabetes and Digestive and Kidney Diseases) DK50107 (E.H.B.); Cancer Center Support Grant P30 CA014520; American Heart Association predoctoral fellowships (Y.K., S.C.M., A.W.D., N.J.P.); and National Institutes of Health, National Institute of General Medical Sciences grant T32 GM081061 (A.W.D.).
Footnotes
The online version of this article contains a data supplement.
There is an Inside Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: Y.-A.K., S.C.M., A.W.D., and E.H.B. designed experiments, analyzed data, and wrote the manuscript; Y.-A.K., S.C.M., A.W.D., C.A.O., and J.N.O. performed experiments; N.J.P. analyzed data; D.Y. performed hematology analysis; and E.E.B., C.-J.C., S.G., and G.C. supplied reagents.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Emery H. Bresnick, 1111 Highland Ave, 4009 WIMR, Madison, WI, 53705; e-mail: ehbresni@wisc.edu.
References
- 1.Hattangadi SM, Wong P, Zhang L, Flygare J, Lodish HF. From stem cell to red cell: regulation of erythropoiesis at multiple levels by multiple proteins, RNAs, and chromatin modifications. Blood. 2011;118(24):6258–6268. doi: 10.1182/blood-2011-07-356006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.McGrath K, Palis J. Ontogeny of erythropoiesis in the mammalian embryo. Curr Top Dev Biol. 2008;82:1–22. doi: 10.1016/S0070-2153(07)00001-4. [DOI] [PubMed] [Google Scholar]
- 3.Cantor AB, Orkin SH. Transcriptional regulation of erythropoiesis: an affair involving multiple partners. Oncogene. 2002;21(21):3368–3376. doi: 10.1038/sj.onc.1205326. [DOI] [PubMed] [Google Scholar]
- 4.Kim S-I, Bresnick EH. Transcriptional control of erythropoiesis: emerging mechanisms and principles. Oncogene. 2007;26(47):6777–6794. doi: 10.1038/sj.onc.1210761. [DOI] [PubMed] [Google Scholar]
- 5.Crispino JD, Lodish MB, MacKay JP, Orkin SH. Use of altered specificity mutants to probe a specific protein-protein interaction in differentiation: the GATA-1:FOG complex. Mol Cell. 1999;3(2):219–228. doi: 10.1016/s1097-2765(00)80312-3. [DOI] [PubMed] [Google Scholar]
- 6.Tsang AP, Visvader JE, Turner CA, et al. FOG, a multitype zinc finger protein, acts as a cofactor for transcription factor GATA-1 in erythroid and megakaryocytic differentiation. Cell. 1997;90(1):109–119. doi: 10.1016/s0092-8674(00)80318-9. [DOI] [PubMed] [Google Scholar]
- 7.DeVilbiss AW, Boyer ME, Bresnick EH. Establishing a hematopoietic genetic network through locus-specific integration of chromatin regulators. Proc Natl Acad Sci USA. 2013;110(36):E3398–E3407. doi: 10.1073/pnas.1302771110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wadman IA, Osada H, Grütz GG, et al. The LIM-only protein Lmo2 is a bridging molecule assembling an erythroid, DNA-binding complex which includes the TAL1, E47, GATA-1 and Ldb1/NLI proteins. EMBO J. 1997;16(11):3145–3157. doi: 10.1093/emboj/16.11.3145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fujiwara T, Lee HY, Sanalkumar R, Bresnick EH. Building multifunctionality into a complex containing master regulators of hematopoiesis. Proc Natl Acad Sci USA. 2010;107(47):20429–20434. doi: 10.1073/pnas.1007804107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xu Z, Huang S, Chang LS, Agulnick AD, Brandt SJ. Identification of a TAL1 target gene reveals a positive role for the LIM domain-binding protein Ldb1 in erythroid gene expression and differentiation. Mol Cell Biol. 2003;23(21):7585–7599. doi: 10.1128/MCB.23.21.7585-7599.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lahlil R, Lécuyer E, Herblot S, Hoang T. SCL assembles a multifactorial complex that determines glycophorin A expression. Mol Cell Biol. 2004;24(4):1439–1452. doi: 10.1128/MCB.24.4.1439-1452.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kang YA, Sanalkumar R, O’Geen H, et al. Autophagy driven by a master regulator of hematopoiesis. Mol Cell Biol. 2012;32(1):226–239. doi: 10.1128/MCB.06166-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Marinkovic D, Zhang X, Yalcin S, et al. Foxo3 is required for the regulation of oxidative stress in erythropoiesis. J Clin Invest. 2007;117(8):2133–2144. doi: 10.1172/JCI31807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lykke-Andersen S, Brodersen DE, Jensen TH. Origins and activities of the eukaryotic exosome. J Cell Sci. 2009;122(Pt 10):1487–1494. doi: 10.1242/jcs.047399. [DOI] [PubMed] [Google Scholar]
- 15.Belostotsky D. Exosome complex and pervasive transcription in eukaryotic genomes. Curr Opin Cell Biol. 2009;21(3):352–358. doi: 10.1016/j.ceb.2009.04.011. [DOI] [PubMed] [Google Scholar]
- 16.Vanacova S, Stefl R. The exosome and RNA quality control in the nucleus. EMBO Rep. 2007;8(7):651–657. doi: 10.1038/sj.embor.7401005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jenks MH, O’Rourke TW, Reines D. Properties of an intergenic terminator and start site switch that regulate IMD2 transcription in yeast. Mol Cell Biol. 2008;28(12):3883–3893. doi: 10.1128/MCB.00380-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kuehner JN, Brow DA. Regulation of a eukaryotic gene by GTP-dependent start site selection and transcription attenuation. Mol Cell. 2008;31(2):201–211. doi: 10.1016/j.molcel.2008.05.018. [DOI] [PubMed] [Google Scholar]
- 19.Thiebaut M, Colin J, Neil H, et al. Futile cycle of transcription initiation and termination modulates the response to nucleotide shortage in S. cerevisiae. Mol Cell. 2008;31(5):671–682. doi: 10.1016/j.molcel.2008.08.010. [DOI] [PubMed] [Google Scholar]
- 20.Bühler M, Haas W, Gygi SP, Moazed D. RNAi-dependent and -independent RNA turnover mechanisms contribute to heterochromatic gene silencing. Cell. 2007;129(4):707–721. doi: 10.1016/j.cell.2007.03.038. [DOI] [PubMed] [Google Scholar]
- 21.Nicolas E, Yamada T, Cam HP, Fitzgerald PC, Kobayashi R, Grewal SI. Distinct roles of HDAC complexes in promoter silencing, antisense suppression and DNA damage protection. Nat Struct Mol Biol. 2007;14(5):372–380. doi: 10.1038/nsmb1239. [DOI] [PubMed] [Google Scholar]
- 22.Zofall M, Yamanaka S, Reyes-Turcu FE, Zhang K, Rubin C, Grewal SI. RNA elimination machinery targeting meiotic mRNAs promotes facultative heterochromatin formation. Science. 2012;335(6064):96–100. doi: 10.1126/science.1211651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lim SJ, Boyle PJ, Chinen M, Dale RK, Lei EP. Genome-wide localization of exosome components to active promoters and chromatin insulators in Drosophila. Nucleic Acids Res. 2013;41(5):2963–2980. doi: 10.1093/nar/gkt037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fujiwara T, O’Geen H, Keles S, et al. Discovering hematopoietic mechanisms through genome-wide analysis of GATA factor chromatin occupancy. Mol Cell. 2009;36(4):667–681. doi: 10.1016/j.molcel.2009.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vollrath AL, Smith AA, Craven M, Bradfield CA. EDGE(3): a web-based solution for management and analysis of Agilent two color microarray experiments. BMC Bioinformatics. 2009;10:280. doi: 10.1186/1471-2105-10-280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu Q, Greimann JC, Lima CD. Reconstitution, activities, and structure of the eukaryotic RNA exosome. Cell. 2006;127(6):1223–1237. doi: 10.1016/j.cell.2006.10.037. [DOI] [PubMed] [Google Scholar]
- 27.Welch JJ, Watts JA, Vakoc CR, et al. Global regulation of erythroid gene expression by transcription factor GATA-1. Blood. 2004;104(10):3136–3147. doi: 10.1182/blood-2004-04-1603. [DOI] [PubMed] [Google Scholar]
- 28.Makino DL, Baumgärtner M, Conti E. Crystal structure of an RNA-bound 11-subunit eukaryotic exosome complex. Nature. 2013;495(7439):70–75. doi: 10.1038/nature11870. [DOI] [PubMed] [Google Scholar]
- 29.Kingsley PD, Greenfest-Allen E, Frame JM, et al. Ontogeny of erythroid gene expression. Blood. 2013;121(6):e5–e13. doi: 10.1182/blood-2012-04-422394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Merryweather-Clarke AT, Atzberger A, Soneji S, et al. Global gene expression analysis of human erythroid progenitors. Blood. 2011;117(13):e96–e108. doi: 10.1182/blood-2010-07-290825. [DOI] [PubMed] [Google Scholar]
- 31.Lebreton A, Tomecki R, Dziembowski A, Séraphin B. Endonucleolytic RNA cleavage by a eukaryotic exosome. Nature. 2008;456(7224):993–996. doi: 10.1038/nature07480. [DOI] [PubMed] [Google Scholar]
- 32.Tomecki R, Kristiansen MS, Lykke-Andersen S, et al. The human core exosome interacts with differentially localized processive RNases: hDIS3 and hDIS3L. EMBO J. 2010;29(14):2342–2357. doi: 10.1038/emboj.2010.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Tomecki R, Drazkowska K, Dziembowski A. Mechanisms of RNA degradation by the eukaryotic exosome. ChemBioChem. 2010;11(7):938–945. doi: 10.1002/cbic.201000025. [DOI] [PubMed] [Google Scholar]
- 34.Schneider C, Tollervey D. Threading the barrel of the RNA exosome. Trends Biochem Sci. 2013;38(10):485–493. doi: 10.1016/j.tibs.2013.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhang J, Socolovsky M, Gross AW, Lodish HF. Role of Ras signaling in erythroid differentiation of mouse fetal liver cells: functional analysis by a flow cytometry-based novel culture system. Blood. 2003;102(12):3938–3946. doi: 10.1182/blood-2003-05-1479. [DOI] [PubMed] [Google Scholar]
- 36.Rylski M, Welch JJ, Chen YY, et al. GATA-1-mediated proliferation arrest during erythroid maturation. Mol Cell Biol. 2003;23(14):5031–5042. doi: 10.1128/MCB.23.14.5031-5042.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Taniguchi T, Endo H, Chikatsu N, et al. Expression of p21(Cip1/Waf1/Sdi1) and p27(Kip1) cyclin-dependent kinase inhibitors during human hematopoiesis. Blood. 1999;93(12):4167–4178. [PubMed] [Google Scholar]
- 38.Xu Y, Swartz KL, Siu KT, Bhattacharyya M, Minella AC. Fbw7-dependent cyclin E regulation ensures terminal maturation of bone marrow erythroid cells by restraining oxidative metabolism. Oncogene. 2014;33(24):3161–3171. doi: 10.1038/onc.2013.289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Oya E, Kato H, Chikashige Y, Tsutsumi C, Hiraoka Y, Murakami Y. Mediator directs co-transcriptional heterochromatin assembly by RNA interference-dependent and -independent pathways. PLoS Genet. 2013;9(8):e1003677. doi: 10.1371/journal.pgen.1003677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wolin SL, Sim S, Chen X. Nuclear noncoding RNA surveillance: is the end in sight? Trends Genet. 2012;28(7):306–313. doi: 10.1016/j.tig.2012.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Muñoz-Alonso MJ, Acosta JC, Richard C, Delgado MD, Sedivy J, León J. p21Cip1 and p27Kip1 induce distinct cell cycle effects and differentiation programs in myeloid leukemia cells. J Biol Chem. 2005;280(18):18120–18129. doi: 10.1074/jbc.M500758200. [DOI] [PubMed] [Google Scholar]
- 42.Tane S, Ikenishi A, Okayama H, Iwamoto N, Nakayama KI, Takeuchi T. CDK inhibitors, p21(Cip1) and p27(Kip1), participate in cell cycle exit of mammalian cardiomyocytes. Biochem Biophys Res Commun. 2014;443(3):1105–1109. doi: 10.1016/j.bbrc.2013.12.109. [DOI] [PubMed] [Google Scholar]
- 43.Barone MV, Crozat A, Tabaee A, Philipson L, Ron D. CHOP (GADD153) and its oncogenic variant, TLS-CHOP, have opposing effects on the induction of G1/S arrest. Genes Dev. 1994;8(4):453–464. doi: 10.1101/gad.8.4.453. [DOI] [PubMed] [Google Scholar]
- 44.Zinszner H, Kuroda M, Wang X, et al. CHOP is implicated in programmed cell death in response to impaired function of the endoplasmic reticulum. Genes Dev. 1998;12(7):982–995. doi: 10.1101/gad.12.7.982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tomasini R, Seux M, Nowak J, et al. TP53INP1 is a novel p73 target gene that induces cell cycle arrest and cell death by modulating p73 transcriptional activity. Oncogene. 2005;24(55):8093–8104. doi: 10.1038/sj.onc.1208951. [DOI] [PubMed] [Google Scholar]
- 46.Tomasini R, Samir AA, Carrier A, et al. TP53INP1s and homeodomain-interacting protein kinase-2 (HIPK2) are partners in regulating p53 activity. J Biol Chem. 2003;278(39):37722–37729. doi: 10.1074/jbc.M301979200. [DOI] [PubMed] [Google Scholar]
- 47.Brewer JW, Hendershot LM, Sherr CJ, Diehl JA. Mammalian unfolded protein response inhibits cyclin D1 translation and cell-cycle progression. Proc Natl Acad Sci USA. 1999;96(15):8505–8510. doi: 10.1073/pnas.96.15.8505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang XZ, Harding HP, Zhang Y, Jolicoeur EM, Kuroda M, Ron D. Cloning of mammalian Ire1 reveals diversity in the ER stress responses. EMBO J. 1998;17(19):5708–5717. doi: 10.1093/emboj/17.19.5708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Thorpe JA, Schwarze SR. IRE1alpha controls cyclin A1 expression and promotes cell proliferation through XBP-1. Cell Stress Chaperones. 2010;15(5):497–508. doi: 10.1007/s12192-009-0163-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gamper I, Koh KR, Ruau D, et al. GAR22: a novel target gene of thyroid hormone receptor causes growth inhibition in human erythroid cells. Exp Hematol. 2009;37(5):539–548 e534. doi: 10.1016/j.exphem.2009.02.002. [DOI] [PubMed] [Google Scholar]
- 51.Brancolini C, Bottega S, Schneider C. Gas2, a growth arrest-specific protein, is a component of the microfilament network system. J Cell Biol. 1992;117(6):1251–1261. doi: 10.1083/jcb.117.6.1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wang XW, Zhan Q, Coursen JD, et al. GADD45 induction of a G2/M cell cycle checkpoint. Proc Natl Acad Sci USA. 1999;96(7):3706–3711. doi: 10.1073/pnas.96.7.3706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rosemary Siafakas A, Richardson DR. Growth arrest and DNA damage-45 alpha (GADD45alpha). Int J Biochem Cell Biol. 2009;41(5):986–989. doi: 10.1016/j.biocel.2008.06.018. [DOI] [PubMed] [Google Scholar]
- 54.Vairapandi M, Balliet AG, Hoffman B, Liebermann DA. GADD45b and GADD45g are cdc2/cyclinB1 kinase inhibitors with a role in S and G2/M cell cycle checkpoints induced by genotoxic stress. J Cell Physiol. 2002;192(3):327–338. doi: 10.1002/jcp.10140. [DOI] [PubMed] [Google Scholar]
- 55.Krakoff IH, Brown NC, Reichard P. Inhibition of ribonucleoside diphosphate reductase by hydroxyurea. Cancer Res. 1968;28(8):1559–1565. [PubMed] [Google Scholar]
- 56.Pledger WJ, Stiles CD, Antoniades HN, Scher CD. An ordered sequence of events is required before BALB/c-3T3 cells become committed to DNA synthesis. Proc Natl Acad Sci USA. 1978;75(6):2839–2843. doi: 10.1073/pnas.75.6.2839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Andrulis ED, Werner J, Nazarian A, Erdjument-Bromage H, Tempst P, Lis JT. The RNA processing exosome is linked to elongating RNA polymerase II in Drosophila. Nature. 2002;420(6917):837–841. doi: 10.1038/nature01181. [DOI] [PubMed] [Google Scholar]
- 58.Hieronymus H, Yu MC, Silver PA. Genome-wide mRNA surveillance is coupled to mRNA export. Genes Dev. 2004;18(21):2652–2662. doi: 10.1101/gad.1241204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dez C, Houseley J, Tollervey D. Surveillance of nuclear-restricted pre-ribosomes within a subnucleolar region of Saccharomyces cerevisiae. EMBO J. 2006;25(7):1534–1546. doi: 10.1038/sj.emboj.7601035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hsieh FF, Barnett LA, Green WF, et al. Cell cycle exit during terminal erythroid differentiation is associated with accumulation of p27(Kip1) and inactivation of cdk2 kinase. Blood. 2000;96(8):2746–2754. [PubMed] [Google Scholar]
- 61.Toyoshima H, Hunter T. p27, a novel inhibitor of G1 cyclin-Cdk protein kinase activity, is related to p21. Cell. 1994;78(1):67–74. doi: 10.1016/0092-8674(94)90573-8. [DOI] [PubMed] [Google Scholar]
- 62.Nakayama K, Ishida N, Shirane M, et al. Mice lacking p27(Kip1) display increased body size, multiple organ hyperplasia, retinal dysplasia, and pituitary tumors. Cell. 1996;85(5):707–720. doi: 10.1016/s0092-8674(00)81237-4. [DOI] [PubMed] [Google Scholar]
- 63.Reynisdóttir I, Polyak K, Iavarone A, Massagué J. Kip/Cip and Ink4 Cdk inhibitors cooperate to induce cell cycle arrest in response to TGF-beta. Genes Dev. 1995;9(15):1831–1845. doi: 10.1101/gad.9.15.1831. [DOI] [PubMed] [Google Scholar]
- 64.Miyamoto K, Araki KY, Naka K, et al. Foxo3a is essential for maintenance of the hematopoietic stem cell pool. Cell Stem Cell. 2007;1(1):101–112. doi: 10.1016/j.stem.2007.02.001. [DOI] [PubMed] [Google Scholar]
- 65.Tothova Z, Kollipara R, Huntly BJ, et al. FoxOs are critical mediators of hematopoietic stem cell resistance to physiologic oxidative stress. Cell. 2007;128(2):325–339. doi: 10.1016/j.cell.2007.01.003. [DOI] [PubMed] [Google Scholar]
- 66.Tsai FY, Keller G, Kuo FC, et al. An early haematopoietic defect in mice lacking the transcription factor GATA-2. Nature. 1994;371(6494):221–226. doi: 10.1038/371221a0. [DOI] [PubMed] [Google Scholar]
- 67.Tsai FY, Orkin SH. Transcription factor GATA-2 is required for proliferation/survival of early hematopoietic cells and mast cell formation, but not for erythroid and myeloid terminal differentiation. Blood. 1997;89(10):3636–3643. [PubMed] [Google Scholar]
- 68.Warr MR, Binnewies M, Flach J, et al. FOXO3A directs a protective autophagy program in haematopoietic stem cells. Nature. 2013;494(7437):323–327. doi: 10.1038/nature11895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Paik JH, Ding Z, Narurkar R, et al. FoxOs cooperatively regulate diverse pathways governing neural stem cell homeostasis. Cell Stem Cell. 2009;5(5):540–553. doi: 10.1016/j.stem.2009.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Renault VM, Rafalski VA, Morgan AA, et al. FoxO3 regulates neural stem cell homeostasis. Cell Stem Cell. 2009;5(5):527–539. doi: 10.1016/j.stem.2009.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.El Wakil A, Francius C, Wolff A, Pleau-Varet J, Nardelli J. The GATA2 transcription factor negatively regulates the proliferation of neuronal progenitors. Development. 2006;133(11):2155–2165. doi: 10.1242/dev.02377. [DOI] [PubMed] [Google Scholar]
- 72.Nardelli J, Thiesson D, Fujiwara Y, Tsai FY, Orkin SH. Expression and genetic interaction of transcription factors GATA-2 and GATA-3 during development of the mouse central nervous system. Dev Biol. 1999;210(2):305–321. doi: 10.1006/dbio.1999.9278. [DOI] [PubMed] [Google Scholar]