Abstract
Sipuleucel-T is an autologous cellular immunotherapy used to treat asymptomatic or minimally symptomatic metastatic castration-resistant prostate cancer (mCRPC). Traditional short-term indicators of clinical response commonly used with chemotherapy have not correlated with survival in patients treated with sipuleucel-T. This retrospective study aimed to evaluate laboratory parameters as possible early biomarkers associated with clinical benefit following sipuleucel-T treatment. Patients treated with sipuleucel-T from 3 randomized, controlled, phase III clinical trials in mCRPC were considered: IMPACT (NCT00065442, n = 512), D9901 (NCT00005947, n = 127), and D9902A (NCT01133704, n = 98). Patients from these trials were included in this study if their samples were analyzed by the central laboratory, and if data were available from baseline and ≥1 post-treatment time point (n = 377). We found that sipuleucel-T treatment was associated with a transient increase in serum eosinophil count at week 6 that resolved by week 14 in 28% of patients (105/377). This eosinophil increase correlated with induced immune response, longer prostate cancer-specific survival (HR = 0.713; 95% confidence interval [CI], 0.525–0.970; P = 0.031) and trend in overall survival (HR = 0.753; 95% CI, 0.563–1.008; P = 0.057). Median serum globulin protein levels also increased transiently, which was associated with antigen-specific antibody responses; however, this did not correlate with longer survival. We conclude that transient increases in eosinophils at week 6 may be a useful, objective, short-term indicator of global immune activation and survival benefit with sipuleucel-T in patients with mCRPC. This observation warrants prospective evaluation in future clinical trials.
Keywords: sipuleucel-T, immunotherapy, prostate cancer, eosinophil, survival
Introduction
Prostate cancer is the fifth most common cancer worldwide. In 2012, prostate cancer was diagnosed in more than 400,000 men and accounted for 92,000 deaths in Europe alone (1). In the US, it is the most common new diagnosis of cancer and the second leading cause of cancer death in men (2). Surgery or radiation therapy can cure localized prostate cancer, but approximately a third of the patients relapse. Androgen-deprivation therapy delays, but does not prevent, progression in most patients with recurrent prostate cancer, and many of these patients eventually develop metastatic castration-resistant prostate cancer (mCRPC). mCRPC is the lethal form of prostate cancer and median survival has been shown to be less than 30 months, even in the active treatment arms of recent phase III clinical trials (3–7).
Sipuleucel-T is an autologous cellular immunotherapy designed to stimulate an immune response to prostate cancer. It has a category 1 recommendation in the National Comprehensive Cancer Network (NCCN) 2013 guidelines for the treatment of patients with asymptomatic or minimally symptomatic mCRPC (8). Each dose of sipuleucel-T is manufactured from autologous peripheral blood mononuclear cells (PBMC) that have been activated ex vivo with a fusion protein (PA2024) composed of prostatic acid phosphatase (PAP), an antigen expressed by the vast majority of prostate carcinomas, fused to granulocyte-macrophage colony-stimulating factor (GM-CSF). Treatment with sipuleucel-T consists of 3 infusions at approximately 2-week intervals (9). Sipuleucel-T prolonged median survival by 4.1 months compared with control in patients with mCRPC in the pivotal phase III IMPACT trial (HR = 0.78; 95% confidence interval [CI], 0.61–0.98; P = 0.03) (4), with similar differences in survival observed in 2 other phase III trials (D9901 and D9902A) (10, 11).
Traditionally, short-term measures of prostate cancer progression, such as radiographic changes, changes in serum prostate-specific antigen (PSA), and changes in time to radiographic progression, have been used as proximal indicators of clinical benefit. However, there was discordance between the effect of sipuleucel-T on overall survival (OS) and its effect on these short-term objective indicators of disease progression (4, 10, 11). Similar dissociations between treatment effects on survival and disease progression endpoints have been observed with other immunotherapies (12–16). It has been suggested that this is due to slowing, rather than temporarily reversing, tumor growth rate – a mechanism of action distinct from that of cytotoxic therapies. Radiographic changes evaluated by Response Evaluation Criteria in Solid Tumors (RECIST) and World Health Organization (WHO) criteria are often seen after 2 cycles of standard chemotherapy. In contrast, changes observed after immunotherapy treatment may take weeks or months, and effects of treatment can persist long after the treatment is stopped (17). Early surrogate biomarkers for immunotherapies that correlate with long-term outcomes, including OS, are therefore needed (18).
The goal of an antigen-specific active immunotherapy such as sipuleucel-T is to elicit immune responses to target antigen-expressing tumor cells. Hence, exploration of immune effects is warranted to identify potential early biomarkers. Recent data exploring the immunological mechanism of sipuleucel-T demonstrated that activation of antigen-presenting cells (APCs) was observed after each infusion of sipuleucel-T (19). Additionally, sipuleucel-T-induced antigen-specific immune responses in vivo, as shown by PA2024 antigen-specific T-cell and antibody immune responses in peripheral blood that were sustained for at least 26 weeks after treatment (19). Importantly, an integrated analysis of data from IMPACT, D9901, and D9902A demonstrated that the measures of product potency (magnitude of APC activation and APC counts), and total nucleated cell [TNC] counts, as well as antigen-specific peripheral immune responses, correlated with OS (19). These findings support the concept that the immune activation stimulated by sipuleucel-T contributes to the survival benefit, and suggest that other simpler parameters indicative of immune activation, which can be readily assessed in clinical practice, could be potential surrogate markers of clinical benefit. To identify other potential short-term objective indicators that might serve as early biomarkers of response, we therefore conducted this retrospective analysis of clinical laboratory parameters in patients with mCRPC treated in 3 randomized phase III trials (IMPACT, D9901, and D9902A).
Patients and Methods
Patients and study design
The phase III trials IMPACT (NCT00065442, n = 512), D9901 (NCT00005947, n = 127), and D9902A (NCT01133704, n = 98) were randomized, double-blind, multicenter studies of sipuleucel-T versus control in men with mCRPC, and were similar in design and patient population (Supplementary Fig. S1) (4, 10, 11). In these trials, patients were randomized 2:1 to receive sipuleucel-T or control as a 30- to 60-minute intravenous infusion approximately every 2 weeks for a total of 3 infusions. Patients were followed for 34–36 months.
Patients’ data were included in this retrospective analysis if: 1) they received sipuleucel-T in any of the above 3 phase III trials; 2) their blood was analyzed by the central laboratory (data from local laboratories were excluded to ensure consistency); and 3) baseline complete blood count (CBC) plus at least 1 post-treatment CBC between study weeks 2–16 were available.
Each of the phase III studies was conducted in accordance with applicable regulations of the US Food and Drug Administration (FDA) and Good Clinical Practice guidelines of the International Conference on Harmonisation (ICH), and was approved by the institutional review board for each study center. All patients gave written informed consent.
Product parameters
Each dose of sipuleucel-T was manufactured and assessed to ensure that pre-specified parameters for APC activation, APC number, and TNC count were met as previously described (4, 9–11, 20, 21). APCs were defined by flow cytometric assessment as large cells expressing CD54, and APC activation was defined as the fold-increase in surface expression of CD54 following co-culture with PA2024 (21).
Blood cell counts, immune responses, and clinical chemistry
CBCs, including differential leukocyte counts, were performed at baseline and weeks 2–34. A patient was considered to have an elevated eosinophil count if they met one or both of the following criteria that were defined prior to this post-hoc data analysis: 1) their eosinophil count was normal at baseline and, at any assessment from study weeks 2–16, their eosinophil count increased to greater than the upper limit of normal (ULN; >ULN; >0.57 × 103 eosinophils/μL); or 2) their eosinophil count increased to within the top quartile of maximum change from baseline (>Q3; increase by >0.36 × 103/μL; to capture patients with eosinophil counts that remained <ULN but who had measurable increases in eosinophil count).
Serum levels of alanine aminotransferase (ALT), aspartate transaminase (AST), total bilirubin, creatinine, and blood urea nitrogen (BUN) were assessed at baseline and study weeks 6, 14, 26, and 34 in the IMPACT trial, and at baseline and weeks 8 and 16 in studies D9901 and D9902A. A comprehensive metabolic panel (including serum total protein and albumin) was performed at baseline and study weeks 6, 14, and 26 in patients in the IMPACT study only. Serum protein electrophoresis (SPEP) was performed with a Sebia automated electrophoresis system (Norcross, GA, USA). SPEP was performed on samples from sipuleucel-T patients treated in the IMPACT trial for whom both a baseline and post-treatment sample were available and whose total protein was found to increase to within the upper 10th percentile following treatment (n = 13).
ELISAs were performed to measure levels of PA2024 and PAP-specific antibody (IgG/IgM) in a subset of patients in the IMPACT study for whom samples were available, with sera collected at baseline, and weeks 6, 14, and 26, as previously described (19). Antigen-specific T-cell responses were assessed by interferon-gamma (IFNγ) enzyme-linked immunosorbent spot (ELISPOT) performed on PBMCs obtained at baseline and weeks 6, 14, and 26, as described previously (19). PA2024 and PAP-stimulated T-cell proliferation was assessed based on incorporation of tritiated thymidine (3H-thymidine) in cells from patients’ PBMCs at these same time points following exposure to antigen as previously described (22). Results are expressed as a stimulation index (SI), defined as the ratio of 3H-thymidine incorporation in the presence of antigen versus in media alone (19). Cytokine concentrations in patients’ sera obtained prior to treatment and at week 6 were measured with multiplex assays from Meso Scale Discovery (Rockville, MD, USA) (19).
Statistical analyses
The Wilcoxon test was used to compare differences in baseline parameters between the elevated eosinophil count groups (yes or no) for continuous variables, while Fisher’s exact text was used for categorical variable. Spearman rank correlation was used to determine the relationships between changes observed from baseline for eosinophil counts with immune response parameters, cumulative product parameters for sipuleucel-T (i.e., the sum of values across all 3 infusions for APC activation, APC number, and TNC count), and change from baseline for cytokines in patient sera samples. Similar analyses were performed for change from baseline for serum globulin protein (calculated as total protein minus albumin protein). Cox regression analyses, stratified by study, were used to assess if elevated eosinophil counts (yes or no) in patients treated with sipuleucel-T were associated with OS or prostate cancer-specific survival (PCSS), and to assess associations between the level of globulin protein (continuous variable) with OS and PCSS. Similar analyses comparing OS in each elevated eosinophil count group (yes or no) with the control group were performed using Cox regression analyses, as were analyses comparing each quartile of maximal change from baseline eosinophil count in patients treated with sipuleucel-T versus the control group. A multivariate Cox regression model was used to assess overall survival with the following variables: eosinophil increase (yes or no), prior docetaxel (yes or no), baseline hemoglobin (continuous variable), and Halabi predicted survival (continuous variable). Fisher’s exact test was used to compare the incidence of select adverse events between the elevated eosinophil count groups (yes or no).
Results
Leukocyte counts
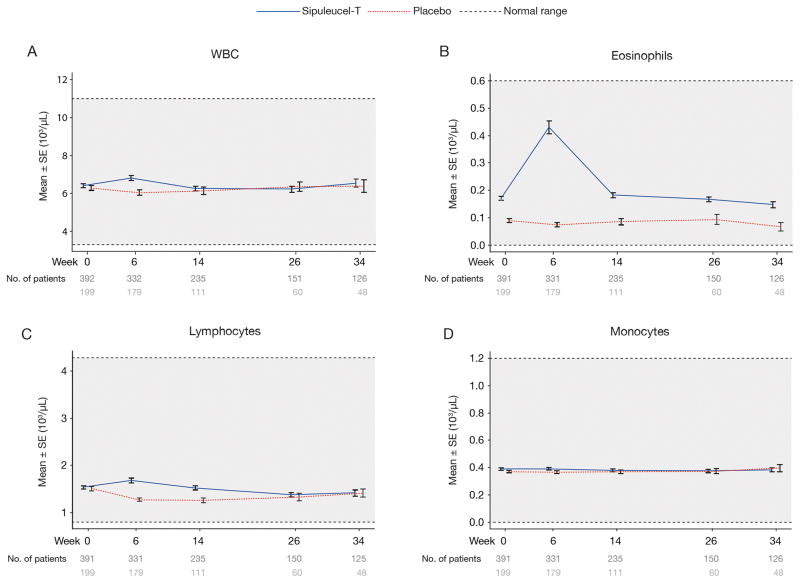
Integrated data from 737 patients treated in the phase III trials (IMPACT [n = 512; 341 sipuleucel-T, 171 control], D9901 [n = 127; 82 sipuleucel-T, 45 control], and D9902A [n = 98; 65 sipuleucel-T, 33 control]) show that mean values for total leukocyte and leukocyte subtype counts remained within normal limits at all time-points in the sipuleucel-T and control treatment groups. The mean values for all leukocyte subtype counts remained well above the lower limit of normal during treatment. However a small, non-statistically significant, increase in mean total leukocyte counts was observed at study week 6 in patients treated with sipuleucel-T, but not control, that resolved by week 14 (Fig. 1A). On review of the leukocyte subsets, while a small increase in basophil counts was observed in these patients (data not shown), this increase in leukocyte counts was primarily accounted for by an increase in mean eosinophil count at week 6, and occurred in the sipuleucel-T but not the control treatment group (Fig. 1B). At week 6, mean lymphocyte counts were also slightly increased from baseline in patients who received sipuleucel-T and slightly decreased in those who received control; these differences resolved by week 26 (Fig. 1C). Mean monocyte counts did not vary during the study or between the treatment groups (Fig. 1D).
Figure 1. Changes in leukocyte counts of patients treated with sipuleucel-T or control in randomized phase III trials.
Shown are the mean and standard error of (A) WBC, (B) eosinophils, (C) lymphocytes, and (D) monocytes of patients treated with sipuleucel-T (blue) or control (red). The number of patients included is indicated for each data point.
WBC, white blood cells.
Data from a total of 377 patients who received sipuleucel-T, and had a baseline eosinophil count and at least one post-baseline count performed at the central laboratory, were available to further characterize the elevation of eosinophil counts: D9901, n = 27; D9902A, n = 39; IMPACT, n = 311 (where noted, some data were available for only a subset of these patients). The demographics and disease characteristics of this subpopulation were similar to the overall population from the phase III trials (Supplementary Table S1). Of the patients pooled from the phase III studies, 28% experienced elevated eosinophil counts (as defined in the Methods section) during treatment with sipuleucel-T (Table 1). These patients experienced an increase from baseline in mean overall leukocyte and lymphocyte counts at week 6, which was not observed in sipuleucel-T-treated patients without elevated eosinophil counts or the control group (Supplementary Fig. S2). A small increase from baseline in mean eosinophil counts was also observed in sipuleucel-T-treated patients who did not meet the elevated eosinophil criteria, which was not observed in the control group. However, this was substantially lower than in patients who met the elevated eosinophil criteria. There were no notable differences in monocyte counts between groups. Patients with elevated eosinophil counts were significantly more likely to have had prior orchiectomy, no previous treatment with docetaxel, higher hemoglobin levels, and longer predicted survival (based on the Halabi nomogram prediction using baseline criteria (23)) than those who did not experience increases in eosinophil counts (Table 2). The following adverse events experienced within 1 day of infusion, which occurred in 10% or more of the patients receiving sipuleucel-T, were all more frequently observed in patients who experienced elevated eosinophil counts than those who did not: pyrexia (33% vs. 21%; P = 0.023), nausea (19% vs. 11%; P = 0.040), and headache (18% vs. 10%; P = 0.032).
Table 1.
Incidence of elevated eosinophil counts following treatment with sipuleucel-T in phase III trials (IMPACT, D9901, and D9902A) of patients with mCRPCa
| Patients | Definition of elevated eosinophil count
|
|||
|---|---|---|---|---|
| >ULN only n (%) | >Q3 only n (%) | >Q3 and >ULN n (%) | >Q3 and/or >ULN n (%) | |
| IMPACT (n = 311) | 10 (3.2) | 12 (3.9) | 65 (20.9) | 87 (28.0) |
| D9901 (n = 27) | 1 (3.7) | 4 (14.8) | 3 (11.1) | 8 (29.6) |
| D9902A (n = 39) | 1 (2.6) | 1 (2.6) | 8 (20.5) | 10 (25.6) |
| Pooled data (n = 377) | 12 (3.2) | 17 (4.5) | 76 (20.2) | 105 (27.9) |
Abbreviations: Q3, third quartile for increase from baseline in eosinophil count (increase by >0.36 × 103/μL); ULN, upper limit of normal (>0.57 × 103 eosinophils/μL).
Eosinophil counts were compared to baseline; elevated eosinophil count response could occur at any time point from study week 2–16. In the control group (n=196), only four patients met elevated eosinophil count criteria at any time point.
Table 2.
Baseline demographics and disease characteristics in patients with mCRPC with and without elevated eosinophil counts after sipuleucel-T in phase III trials (pooled data)
| Parameter | Elevated eosinophil count
|
P-valuea | |
|---|---|---|---|
| Yes (n = 105) | No (n = 272) | ||
| Median age (range), years | 70 (49–91) | 72 (49–89) | 0.360 |
| Median weight (range), lbs | 191 (131–384) | 194 (131–350) | 0.164 |
| Race, n (%) | |||
| Caucasian | 92 (87.6) | 242 (89.0) | 0.609 |
| Black or African American | 6 (5.7) | 19 (7.0) | |
| Asian | 1 (1.0) | 1 (0.4) | |
| Hispanic | 4 (3.8) | 8 (2.9) | |
| Other | 2 (1.9) | 2 (0.7) | |
| ECOG PS = 0, n (%) | 89 (84.8) | 220 (80.9) | 0.456 |
| Gleason sum, n (%) | |||
| ≤6 | 15 (14.3) | 39 (14.3) | 1.000 |
| 7 | 63 (60.0) | 165 (60.7) | |
| ≥8 | 26 (24.8) | 68 (25.0) | |
| Unknown | 1 (1.0) | 0 (0) | |
| Localization of disease, n (%) | |||
| Bone only | 53 (50.5) | 139 (51.1) | 0.773 |
| Soft tissue only | 10 (9.5) | 20 (7.4) | |
| Both bone and soft tissue | 42 (40.0) | 113 (41.5) | |
| Bisphosphonate use, n (%) | 40 (38.1) | 119 (43.8) | 0.353 |
| No. of bone metastases, n (%) | |||
| 0–5 | 50 (47.6) | 110 (40.4) | 0.271 |
| 6–10 | 11 (10.5) | 44 (16.2) | |
| >10 | 43 (41.0) | 117 (43.0) | |
| Unknown | 1 (1.0) | 1 (0.4) | |
| Time from diagnosis to randomization, median (range), years | 7.1 (0.8–22.7) | 6.8 (0.8–24.5) | 0.496 |
| Prior hormone therapy, n (%) | 105 (100.0) | 272 (100.0) | 1.000 |
| Orchiectomy | 19 (18.1) | 25 (9.2) | 0.020 |
| Prior chemotherapy, n (%) | |||
| Any | 11 (10.5) | 51 (18.8) | 0.062 |
| Docetaxel | 6 (5.7) | 41 (15.1) | 0.014 |
| Radical prostatectomy | 38 (36.2) | 94 (34.6) | 0.810 |
| Previous radiotherapy | 56 (53.3) | 142 (52.2) | 0.908 |
| Median serum laboratory value | |||
| PSA (range), ng/mL | 43.0 (5.2–649.9) | 56.1 (5.3–8005.6) | 0.123 |
| LDH (range), U/L | 189.0 (84.0–428.0) | 194.0 (101.0–1730.0) | 0.240 |
| Hemoglobin (range), g/dL | 13.0 (11.1–17.9) | 12.8 (8.4–16.2) | 0.002 |
| Alkphos (range), U/L | 93.0 (42.0–694.0) | 101.5 (18.0–3900.0) | 0.222 |
| Median Halabi score (range), months | 21.6 (9.6–32.0) | 20.1 (4.8–32.0) | 0.010 |
Abbreviations: Alkphos, alkaline phosphatase; ECOG PS, Eastern Cooperative Oncology Group performance status; LDH, lactate dehydrogenase.
Fisher’s exact test was used to evaluate categorical data and the Wilcoxon test was used for continuous data.
Correlations between elevated eosinophil counts, immune responses, and survival
In a subset of patients from the IMPACT trial for whom immune response data were available, none of the patients in either arm had evidence of anti-PA2024 immunity at baseline. Following sipuleucel-T treatment, both cellular and serum antibody responses against PA2024 were detected in sipuleucel-T treated patients. As previously reported, a responder frequency analysis, based upon post-treatment antibody titers being greater than the 95th percentile of the baseline response, showed that 68% (102/151) of sipuleucel-T-treated patients had post-treatment anti-PA2024 responses compared with 3% (2/70) of control patients (19).
Among patients evaluable for elevated eosinophils (n = 377), increases in eosinophil counts were highly associated with product lot release parameters, including cumulative numbers of APCs (r = 0.330; P < 0.001) and TNC counts (r = 0.261; P < 0.001). Cumulative APC activation was not associated with observed changes in eosinophil counts (r = 0.024; P = 0.638). Analyses of immune responses and eosinophil counts showed that antigen-specific T-cell effector response (as assessed by IFNγ ELISPOT) and T-cell proliferation responses, detectable at any time point post treatment, positively correlated with maximal eosinophil change (r = 0.222; P = 0.027 and r = 0.215; P = 0.0325, respectively; Supplementary Figure S3). While there was not a statistically significant correlation between maximal eosinophil change and antibody response at any time point (r = 0.069; P = 0.410), there was a positive correlation with eosinophil change and ELISA antibody titer at week 6 (r = 0.194; P = 0.034; Supplementary Figure S3).
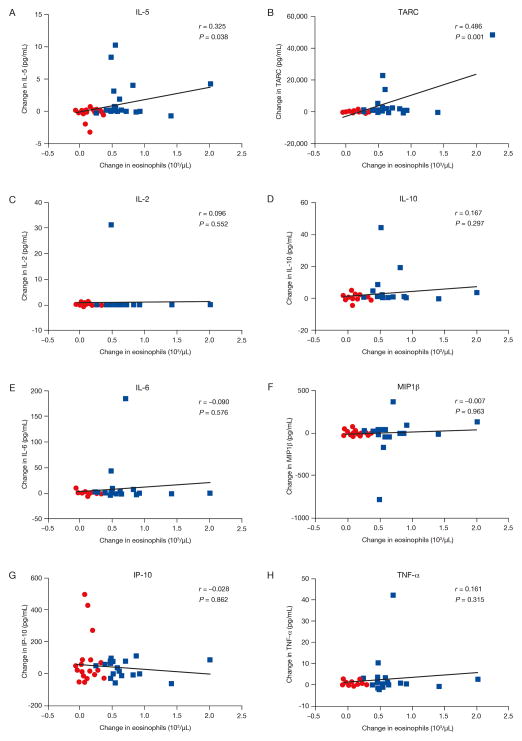
The detection of elevated eosinophil counts was also associated with other signals of immune activation present in the patients’ sera. Specifically, elevated eosinophil counts positively associated with increases in interleukin (IL)-5 (r = 0.325; P = 0.038) and thymus activation-regulated chemokine (TARC, r = 0.486; P = 0.001), also known as CCL17, detected in serum at week 6 relative to baseline (Fig. 2). No correlation was observed with other cytokines measured at week 6 relative to pre-treatment, including IL-2 (r = 0.096; P = 0.552), IL-6 (r = −0.090; P = 0.576), IL-10 (r = 0.167; P = 0.297), IFNγ-induced protein (IP)-10 (r = −0.028; P = 0.862), macrophage inflammatory protein 1β (r = −0.007; P = 0.963), IFNγ (r = −0.242; P = 0.425), or tumor necrosis factor (TNF) α (r = 0.161; P = 0.315).
Figure 2. Change in eosinophil counts versus change in serum cytokines from baseline to week 6 following treatment with sipuleucel-T.
Patients from the IMPACT trial who received sipuleucel-T, and from whom serum samples were available from pre-treatment and week 6 time points (n = 41), were evaluated for changes in (A) IL-5, (B) TARC, (C) IL-2, (D) IL-10, (E) IL-6, (F) MIP1β, (G) IP-10, and (H) TNFα with respect to the changes in eosinophil counts. Blue squares represent patients with elevated eosinophils and red circles represent those without elevated eosinophils. Best-fit regression lines are shown on each graph.
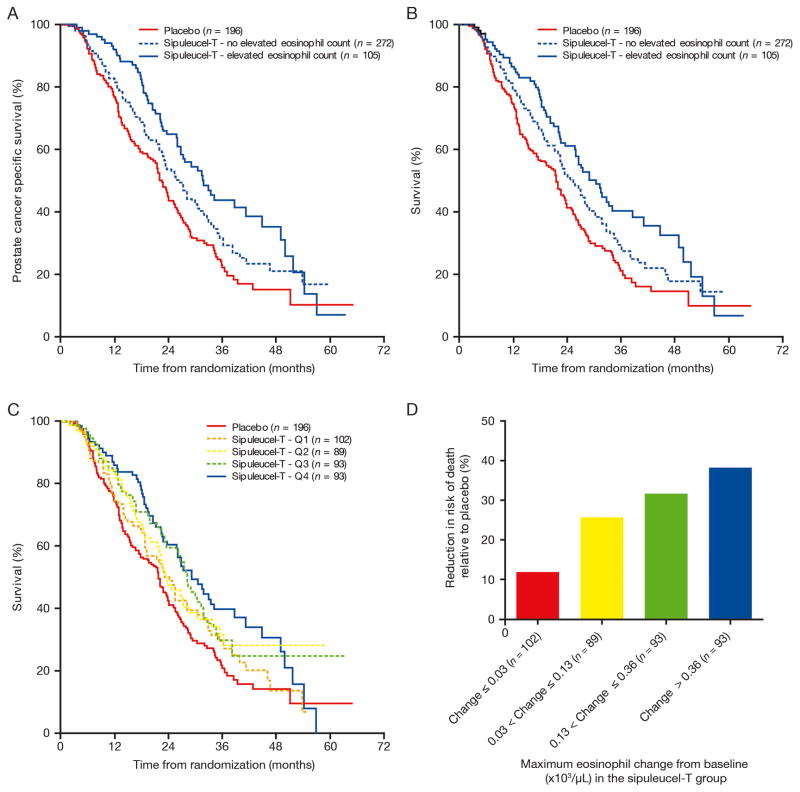
PCSS and OS were assessed in patients with evaluable eosinophil counts (n = 377; Fig. 3A and B). Patients with elevated eosinophil counts had a significantly improved PCSS compared with those without elevated counts (HR = 0.713; 95% CI, 0.525–0.970; P = 0.031; Fig. 3A). A trend toward improved OS was also observed in patients with elevated eosinophil counts compared with those who did not (HR = 0.753; 95% CI, 0.563–1.008; P = 0.057; Fig. 3B). Eosinophil increases were not significantly associated with longer survival in a multivariate analysis (HR = 0.859; 95% CI, 0.635–1.163; P = 0.326), however this analysis included both pre-treatment predictive factors (prior chemotherapy, baseline hemoglobin, baseline predicted survival) and post-treatment pharmacodynamics information (eosinophil increase), potentially confounding this analysis. When compared with patients who received control (limited to those with centrally reviewed CBCs at baseline and within study days 14–112; n = 196), there was a significant increase in OS for sipuleucel-T-treated patients with elevated eosinophil counts (HR = 0.595; 95% CI, 0.442–0.801; P < 0.001), with a greater magnitude of benefit than those without elevated eosinophil counts (HR = 0.788; 95% CI, 0.631–0.984; P = 0.035; Fig. 3A and B). Of note, both subgroups showed a significant improvement in OS versus the control population. Similar results were observed when evaluating PCSS (Fig. 3A). An analysis of patients subdivided into quartiles for maximal change in eosinophil count showed that, while a treatment effect was consistently observed across the subgroups, there was a trend toward improved magnitude of benefit from sipuleucel-T treatment for patients with greater increases in eosinophils. The relative reduction in risk of death with sipuleucel-T was 38.4% (HR = 0.616; 95% CI, 0.453–0.838) in the highest quartile versus 11.8% (HR = 0.882; 95% CI, 0.660–1.177) in the lowest quartile (Fig. 3C and D).
Figure 3. Kaplan-Meier estimates of overall survival in patients with or without elevated eosinophil counts.
Shown is the (A) PCSS and (B) OS in a pooled analysis of patients who received sipuleucel-T in the 3 phase III trials (n = 377) for patients who experienced elevated eosinophil counts (blue) or those who did not (dotted blue) compared with control (red). (C) OS by the maximal change from baseline in eosinophil count quartile in patients who received sipuleucel-T is shown as is (D) the associated percent reduction in risk of death relative to control.
Serum protein and clinical chemistry analyses
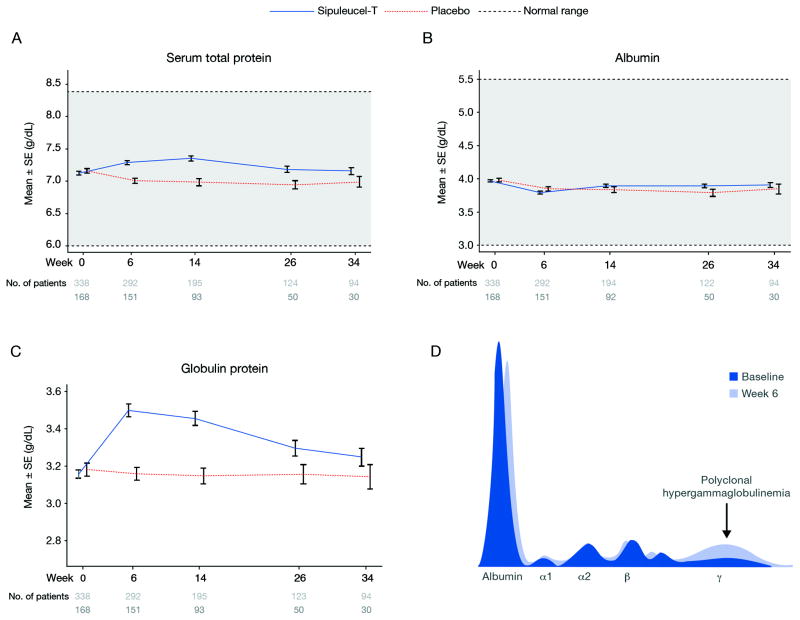
In further exploratory analyses, serum clinical chemistry measurements (ALT, AST, total bilirubin, total protein, creatinine, BUN, calcium, magnesium, potassium, and sodium) were assessed for change from baseline to weeks 6–34 in patients who received sipuleucel-T. Mean values for serum calcium, magnesium, potassium, and sodium levels were measured and are reported only in patients in the IMPACT trial. All of the mean values remained within normal limits, were stable, and did not vary with respect to treatment group in patients in the phase III trials combined (data not shown) with the exception of serum total protein, which was elevated from weeks 6–34 in patients (n = 338) who received sipuleucel-T relative to those who received control (n = 168; Fig. 4A). Serum albumin levels did not vary with sipuleucel-T treatment, and remained stable during treatment and follow-up (Fig. 4B). The increase in total serum protein was accounted for by an increase in serum globulin proteins that occurred by week 6 and declined between weeks 6–34 (Fig. 4C). Increases of ≥0.5 g/dL were observed in 41.5% (132/318) of patients treated with sipuleucel-T versus 10.8% (17/157) of those in the control group (P < 0.001). Analyses of antigen-specific immune responses and globulin protein showed a significant correlation between maximal serum globulin increases and antibody responses (r = 0.176; P = 0.032). There was no correlation observed for T-cell proliferation response (r = 0.018; P = 0.861) and a negative correlation for T-cell IFNγ-secreting responses (r = −0.222; P = 0.027). Changes in serum globulin levels also correlated with increases in TARC expression (r = 0.310; P = 0.048; data not shown). No direct correlation was observed between the level of globulin protein and OS (HR = 0.93; 95% CI, 0.64–1.35; P = 0.72) or PCSS (HR = 0.92; 95% CI, 0.63–1.35; P = 0.67) in a pooled analysis of patients from the phase III trials.
Figure 4.
Serum protein levels in 13 sipuleucel-T treated patients in the phase III IMPACT trial. Shown are the mean and standard error of (A) serum total protein, (B) albumin, or (C) globulin protein of patients treated with sipuleucel-T (blue) or control (red) from the 3 randomized phase III trials. The number of patients included is indicated for each data point. Serum protein electrophoresis was performed to characterize serum protein from 13 patients in the IMPACT trial with the highest post-baseline serum globulin protein levels; data from a representative patient at baseline and week 6 are presented (D).
Serum proteins from the 13 patients who exhibited the greatest increases in serum protein with sipuleucel-T were further characterized by SPEP. In all of these patients, the observed increase in globulin proteins could be accounted for by an increase in polyclonal gamma globulin (Fig. 4D). In sera from 3 of the 13 analyzed by SPEP, small monoclonal globulin components with concentrations of 0.1, 0.2, and 0.5 g/dL, respectively, were present at baseline. In these 3 patients, the concentrations of the monoclonal components did not change during treatment, but those of polyclonal gamma globulins increased (data not shown).
Discussion
Our retrospective analyses from the 3 phase III trials of sipuleucel-T indicate that eosinophil counts and serum globulin protein levels, both potentially non-antigen-specific measures of immune activation following sipuleucel-T treatment, may be potential biomarkers of treatment effect. Transient increases in eosinophil counts following sipuleucel-T treatment correlated with increased PCSS as well as with antigen-specific immune responses elicited with sipuleucel-T treatment. Although increased serum globulin protein levels following sipuleucel-T did not correlate with OS or PCSS, they did correlate with antigen-specific antibody immune responses.
Elevated eosinophil counts were associated with favorable disease characteristics (longer predicted survival based on the Halabi nomogram, higher hemoglobin, no prior docetaxel chemotherapy). Therefore, it is conceivable that the generation of eosinophils was a consequence of patients who were more ’immunocompetent’ (i.e., better able to mount immune responses) potentially as a result of having a lower tumor burden, or not having received previous immunosuppressive chemotherapy and corticosteroids, and hence more likely to benefit from an immune-based therapy such as sipuleucel-T. This hypothesis is consistent with findings from a recent analysis showing that patients with better baseline prognostic factors, specifically those with lower baseline PSA values, derived the greatest magnitude of benefit from sipuleucel-T treatment, with a 13-month improvement in median OS for those with PSA values < 22.1 ng/mL (24). This hypothesis is also consistent with data from other studies suggesting that measures of better baseline hematologic function, including higher hemoglobin concentration, were predictive of treatment benefit from the MVA-5T4 tumor vaccine in separate trials of patients with renal cell cancer or prostate cancer (25, 26). Other studies have similarly demonstrated that higher absolute lymphocyte counts, or higher lymphocyte counts relative to monocyte counts, are prognostic for patients with different kinds of malignancies (27–29). Future studies will help to evaluate whether increased eosinophil counts is an expected immune effect of treating immune-competent individuals with sipuleucel-T and/or whether it is similarly associated with improved long-term outcome regardless of disease stage. In either scenario, however, our findings do not suggest that the absence of increased eosinophil counts is necessarily a negative predictor for prolonged survival.
The transient increase in eosinophils following infusion of sipuleucel-T is most likely a consequence of global immune activation. In this way, changes in eosinophil counts may be a pharmacodynamic biomarker indicating a biological response, rather than serve as an independent predictor of overall survival. However, a growing body of evidence suggests that eosinophils play a role in antitumor immune responses, possibly through T-cell recruitment and polarization, or via direct antitumor activity (30, 31). Eosinophils may also play a role in dendritic cell activation/recruitment and generation of antibody responses (30), and eosinophilia has also previously been observed in acute and chronic graft-versus-host disease, further suggesting these cells may be involved, directly or indirectly, in tissue-destructive immune reactions (32). Thus, it is possible that the increased eosinophil counts observed following sipuleucel-T treatment may contribute to the improvement in OS. Further studies are necessary to evaluate the potential role of eosinophils in the sipuleucel-T treatment effect.
Due to the limited sample size, the findings from the correlative analyses of eosinophil increases and immune parameters are hypothesis-generating observations. Nonetheless, it is interesting that elevated eosinophils correlated with both antigen-specific IFNγ ELISPOT and proliferative responses. While maximal eosinophil changes did not correlate with antibody responses at any time point, a correlation was found for antibody titer at week 6. It will be interesting to evaluate correlations between elevated eosinophils and antibody responses using more sensitive methods of antibody detection as have been employed in more recent trials of sipuleucel-T. It is perhaps not surprising that elevated eosinophil levels correlated with increases in the TH2-type cytokines IL-5 and TARC, given that IL-5, in particular, is a known growth and maturation factor for eosinophils (and B cells), and is expressed by eosinophils (33). Additionally, TARC functions as a key chemoattractant for eosinophils (34). The contemporaneous generation of antigen-specific antibody responses and transient eosinophil elevations at week 6 (2 weeks following the final sipuleucel-T infusion) do indicate the presence of a productive antitumor TH2-type signature initially after treatment. Curiously, the anti-PAP antibodies and/or this TH2-profile observed in sera following sipuleucel-T treatment have not been seen with other vaccine strategies targeting PAP (35, 36). TH1 cytokine profiles, which are known to be associated with cellular antitumor immunity, did not correlate with transient eosinophil increases. Indeed, TH1 cytokines do not directly promote eosinophil production, which may explain this lack of association. Given the small sample size available for this analysis, it is unclear whether the data suggest a shift from a TH2- to TH1-type response over time, or if it is simply a random finding. It should be noted that the collective immune response data and cytokine profiles with sipuleucel-T treatment show that both TH1- and TH2-type profiles are evident during and after treatment (at both early and late time points) (19), suggesting a broader immune activation that is not skewed purely towards either a TH1 or a TH2 profile.
It is interesting that the elevated globulin proteins following sipuleucel-T treatment appeared to be due to an increase in polyclonal gamma globulins. One potential explanation for this effect could be that sipuleucel-T induced antigen spreading or the development of antibody responses to additional (non-PAP or PA2024) tumor antigens, as has been observed with sipuleucel-T in an analysis of the IMPACT trial and also with other vaccines (37–39). This observation may also have been directly related to the increase in IL-5 and the consequent activation of B cells with increased immunoglobulin secretion, as has been reported (33). Although elevations in serum globulin did not correlate with OS in this analysis, it is possible that non-antibody globulin protein subsets may have confounded the analysis. Evaluation of quantitative immunoglobulin levels following sipuleucel-T treatment as a potential predictor of response, as well as the specific immunoglobulin targets, warrants further investigation.
Mean cell counts for other leukocyte subtypes remained within normal limits at all time-points during and after treatment. This result was not surprising, as there has been no evidence of immune-cell depletion in patients who undergo repeated leukapheresis procedures (40–42). These results effectively serve as ‘controls’, demonstrating the selectivity of the response observed in the eosinophil subset and changes in gamma globulins. Additionally, as expected, no electrolyte changes, or serum markers to suggest renal/hepatic toxicity, were observed.
Potential biomarkers to predict the OS benefit of sipuleucel-T currently include baseline PSA, product parameters such as APC count and degree of APC activation, and antigen-specific immune responses measured following treatment (19, 24). However, aside from PSA, none of these laboratory tests are currently routinely available to clinicians. Both eosinophil counts and serum protein analyses would be particularly useful in this regard, as they are also easily monitored and routinely obtained when managing patients with mCRPC. Of note, the association with treatment benefit (survival) and transient increases in eosinophil counts following sipuleucel-T treatment observed in this study was not as strong as the association with antigen-specific immune responses as previously reported (18). This is perhaps not surprising, as the generation of antigen-specific T cells is the intended mechanism of action of this therapy. Nonetheless, the timing of when antigen-specific T cells should optimally be measured is uncertain, and could vary from individual to individual. Our findings here suggest that more immediate global measures of immune activation might also be developed as prognostic indicators. This could be particularly useful for other immune-active therapies without readily identifiable antigenic targets, such as following the use of whole cell vaccines or immune checkpoint inhibitor therapies.
With further characterization and validation in prospective studies, transient changes in eosinophil counts may serve as an objective, early marker of response to sipuleucel-T. In particular, studies combining sipuleucel-T with enzalutamide (NCT01981122), abiraterone (NCT01487863), standard androgen deprivation (NCT01431391), or a DNA vaccine targeting the same PAP antigen (NCT01706458), are currently ongoing or were recently completed. These studies will also explore the relationship between eosinophil counts and other measures of immune response and disease progression. These additional studies, and other prospective studies in patients with different stages of disease, will be important to define whether changes in eosinophil counts are generally associated with sipuleucel-T treatment and treatment outcome.
Supplementary Material
Acknowledgments
Financial support: Dendreon Corporation
All authors contributed to the analysis and interpretation of the data, writing and editing of the manuscript, and approved the final version for submission. Medical writing assistance was provided by Gardiner-Caldwell Communications, which was funded by Dendreon Corporation.
Footnotes
Potential conflicts of interest
DGM has served as a consultant to Dendreon Corporation and his institution receives funding for clinical research from Dendreon; TAG has served as a consultant to Dendreon and his institution receives funding for clinical research from Dendreon; CSH has served as a consultant to Dendreon and her institution receives funding for clinical research from Dendreon; PWK has served as a consultant to Dendreon; EJS has received funding for research and honoraria/travel reimbursement for consulting from Dendreon; MHW has received funding for research from Dendreon; RBS, TD and NS are full-time employees of Dendreon Corporation; RD has received honoraria as a consultant for Dendreon and has received funding for research from Dendreon.
References
- 1.Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, Rosso S, Coebergh JW, Comber H, et al. Cancer incidence and mortality patterns in Europe: estimates for 40 countries in 2012. Eur J Cancer. 2013;49:1374–403. doi: 10.1016/j.ejca.2012.12.027. [DOI] [PubMed] [Google Scholar]
- 2.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62:10–29. doi: 10.3322/caac.20138. [DOI] [PubMed] [Google Scholar]
- 3.Parker C, Nilsson S, Heinrich D, Helle SI, O’Sullivan JM, Fosså SD, et al. Alpha emitter Radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369:213–23. doi: 10.1056/NEJMoa1213755. [DOI] [PubMed] [Google Scholar]
- 4.Kantoff PW, Higano CS, Shore ND, Berger ER, Small EJ, Penson DF, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411–22. doi: 10.1056/NEJMoa1001294. [DOI] [PubMed] [Google Scholar]
- 5.Petrylak DP, Tangen CM, Hussain MH, Lara PN, Jr, Jones JA, Taplin ME, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med. 2004;351:1513–20. doi: 10.1056/NEJMoa041318. [DOI] [PubMed] [Google Scholar]
- 6.Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–12. doi: 10.1056/NEJMoa040720. [DOI] [PubMed] [Google Scholar]
- 7.Ryan CJ, Smith MR, de Bono JS, Molina A, Logothetis CJ, de Souza P, et al. Abiraterone in metastatic prostate cancer without previous chemotherapy. N Engl J Med. 2013;368:138–48. doi: 10.1056/NEJMoa1209096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.National Comprehensive Cancer Network. [Accessed September 2013];NCCN Clinical Practice Guidelines in Oncology: Prostate Cancer (Version 4.2013) Available from: http://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf.
- 9.Provenge® (sipuleucel-T) 2011 [package insert] [cited 2013 March 5]. Available from: http://www.provenge.com/pdf/prescribing-information.pdf.
- 10.Small EJ, Schellhammer PF, Higano CS, Redfern CH, Nemunaitis JJ, Valone FH, et al. Placebo-controlled phase III trial of immunologic therapy with sipuleucel-T (APC8015) in patients with metastatic, asymptomatic hormone refractory prostate cancer. J Clin Oncol. 2006;24:3089–94. doi: 10.1200/JCO.2005.04.5252. [DOI] [PubMed] [Google Scholar]
- 11.Higano CS, Schellhammer PF, Small EJ, Burch PA, Nemunaitis J, Yuh L, et al. Integrated data from 2 randomized, double-blind, placebo-controlled, phase 3 trials of active cellular immunotherapy with sipuleucel-T in advanced prostate cancer. Cancer. 2009;115:3670–9. doi: 10.1002/cncr.24429. [DOI] [PubMed] [Google Scholar]
- 12.Gulley JL, Drake CG. Immunotherapy for prostate cancer: recent advances, lessons learned, and areas for further research. Clin Cancer Res. 2011;17:3884–91. doi: 10.1158/1078-0432.CCR-10-2656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kantoff PW, Schuetz TJ, Blumenstein BA, Glode LM, Bilhartz DL, Wyand M, et al. Overall survival analysis of a phase II randomized controlled trial of a Poxviral-based PSA-targeted immunotherapy in metastatic castration-resistant prostate cancer. J Clin Oncol. 2010;28:1099–105. doi: 10.1200/JCO.2009.25.0597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Stein WD, Gulley JL, Schlom J, Madan RA, Dahut W, Figg WD, et al. Tumor regression and growth rates determined in five intramural NCI prostate cancer trials: the growth rate constant as an indicator of therapeutic efficacy. Clin Cancer Res. 2011;17:907–17. doi: 10.1158/1078-0432.CCR-10-1762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Madan RA, Gulley JL, Fojo T, Dahut WL. Therapeutic cancer vaccines in prostate cancer: the paradox of improved survival without changes in time to progression. Oncologist. 2010;15:969–75. doi: 10.1634/theoncologist.2010-0129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tuma RS. Immunotherapies in clinical trials: do they demand different evaluation tools? J Natl Cancer Inst. 2011;103:780–1. doi: 10.1093/jnci/djr184. [DOI] [PubMed] [Google Scholar]
- 17.Saenger YM, Wolchok JD. The heterogeneity of the kinetics of response to ipilimumab in metastatic melanoma: patient cases. Cancer Immun. 2008;8:1. [PMC free article] [PubMed] [Google Scholar]
- 18.Sharma P, Wagner K, Wolchok JD, Allison JP. Novel cancer immunotherapy agents with survival benefit: recent successes and next steps. Nat Rev Cancer. 2011;11:805–12. doi: 10.1038/nrc3153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sheikh NA, Petrylak D, Kantoff PW, Dela Rosa C, Stewart FP, Kuan LY, et al. Sipuleucel-T immune parameters correlate with survival: an analysis of the randomized phase 3 clinical trials in men with castration-resistant prostate cancer. Cancer Immunol Immunother. 2013;62:137–47. doi: 10.1007/s00262-012-1317-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Burch PA, Breen JK, Buckner JC, Gastineau DA, Kaur JA, Laus RL, et al. Priming tissue-specific cellular immunity in a phase I trial of autologous dendritic cells for prostate cancer. Clin Cancer Res. 2000;6:2175–82. [PubMed] [Google Scholar]
- 21.Sheikh NA, Jones LA. CD54 is a surrogate marker of antigen presenting cell activation. Cancer Immunol Immunother. 2008;57:1381–90. doi: 10.1007/s00262-008-0474-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Corradin G, Etlinger HM, Chiller JM. Lymphocyte specificity to protein antigens. I. Characterization of the antigen-induced in vitro T cell-dependent proliferative response with lymph node cells from primed mice. J Immunol. 1977;119:1048–53. [PubMed] [Google Scholar]
- 23.Halabi S, Small EJ, Kantoff PW, Kattan MW, Kaplan EB, Dawson NA, et al. Prognostic model for predicting survival in men with hormone-refractory metastatic prostate cancer. J Clin Oncol. 2003;21:1232–7. doi: 10.1200/JCO.2003.06.100. [DOI] [PubMed] [Google Scholar]
- 24.Schellhammer PF, Chodak G, Whitmore JB, Sims R, Frohlich MW, Kantoff PW. Lower baseline prostate-specific antigen is associated with a greater overall survival benefit from sipuleucel-T in the immunotherapy for prostate adenocarcinoma treatment (IMPACT) trial. Urology. 2013;81:1297–302. doi: 10.1016/j.urology.2013.01.061. [DOI] [PubMed] [Google Scholar]
- 25.Harrop R, Treasure P, de Belin J, Kelleher M, Bolton G, Naylor S, et al. Analysis of pre-treatment markers predictive of treatment benefit for the therapeutic cancer vaccine MVA-5T4 (TroVax) Cancer Immunol Immunother. 2012;61:2283–94. doi: 10.1007/s00262-012-1302-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Harrop R, Chu F, Gabrail N, Srinivas S, Blount D, Ferrari A. Vaccination of castration-resistant prostate cancer patients with TroVax (MVA-5T4) in combination with docetaxel: a randomized phase II trial. Cancer Immunol Immunother. 2013;62:1511–20. doi: 10.1007/s00262-013-1457-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Koh YW, Kang HJ, Park C, Yoon DH, Kim S, Suh C, et al. The ratio of the absolute lymphocyte count to the absolute monocyte count is associated with prognosis in Hodgkin’s lymphoma: correlation with tumor-associated macrophages. The Oncologist. 2012;17:871–80. doi: 10.1634/theoncologist.2012-0034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Johnson ME, Zhu F, Li T, Wu H, Galloway TJ, Farma JM, et al. Absolute lymphocyte count: A potential prognostic factor for Merkel cell carcinoma. J Am Acad Dermatol. 2014;70:1028–35. doi: 10.1016/j.jaad.2014.01.890. [DOI] [PubMed] [Google Scholar]
- 29.Watanabe R, Tomita N, Itabashi M, Ishibashi D, Yamamoto E, Koyama S, et al. Peripheral blood absolute lymphocyte/monocyte ratio as a useful prognostic factor in diffuse large B-cell lymphoma in the rituximab era. Eur J Haematol. 2014;92:204–10. doi: 10.1111/ejh.12221. [DOI] [PubMed] [Google Scholar]
- 30.Jacobsen EA, Helmers RA, Lee JJ, Lee NA. The expanding role(s) of eosinophils in health and disease. Blood. 2012;120:3882–90. doi: 10.1182/blood-2012-06-330845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Simson L, Ellyard JI, Dent LA, Matthaei KI, Rothenberg ME, Foster PS, et al. Regulation of carcinogenesis by IL-5 and CCL11: a potential role for eosinophils in tumor immune surveillance. J Immunol. 2007;178:4222–9. doi: 10.4049/jimmunol.178.7.4222. [DOI] [PubMed] [Google Scholar]
- 32.McNeel D, Rubio MT, Damaj G, Emile JF, Belanger C, Varet B, et al. Hypereosinophilia as a presenting sign of acute graft-versus-host disease after allogeneic bone marrow transplantation. Transplantation. 2002;74:1797–800. doi: 10.1097/00007890-200212270-00028. [DOI] [PubMed] [Google Scholar]
- 33.Dubucquoi S, Desreumaux P, Janin A, Klein O, Goldman M, Tavernier J, et al. Interleukin 5 synthesis by eosinophils: association with granules and immunoglobulin-dependent secretion. J Exp Med. 1994;179:703–8. doi: 10.1084/jem.179.2.703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Thielen C, Radermacher V, Trimeche M, Roufosse F, Goldman M, Boniver J, et al. TARC and IL-5 expression correlates with tissue eosinophilia in peripheral T-cell lymphomas. Leuk Res. 2008;32:1431–8. doi: 10.1016/j.leukres.2008.02.016. [DOI] [PubMed] [Google Scholar]
- 35.McNeel DG, Dunphy EJ, Davies JG, Frye TP, Johnson LE, Staab MJ, et al. Safety and immunological efficacy of a DNA vaccine encoding prostatic acid phosphatase in patients with stage D0 prostate cancer. J Clin Oncol. 2009;27:4047–54. doi: 10.1200/JCO.2008.19.9968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fong L, Brockstedt D, Benike C, Breen JK, Strang G, Ruegg CL, et al. Dendritic cell-based xenoantigen vaccination for prostate cancer immunotherapy. J Immunol. 2001;167:7150–6. doi: 10.4049/jimmunol.167.12.7150. [DOI] [PubMed] [Google Scholar]
- 37.Vanderlugt CL, Miller SD. Epitope spreading in immune-mediated diseases: implications for immunotherapy. Nat Rev Immunol. 2002;2:85–95. doi: 10.1038/nri724. [DOI] [PubMed] [Google Scholar]
- 38.Smith HA, Maricque BB, Eberhardt J, Petersen B, Gulley JL, Schlom J, et al. IgG responses to tissue-associated antigens as biomarkers of immunological treatment efficacy. J Biomed Biotech. 2011:454861. doi: 10.1155/2011/454861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.GuhaThakurta D, Fan L-Q, Vu T, Sheikh NA, Trager JB. MC13-0071 Immune response against non-targeted tumor antigens after treatment with sipuleucel-T and its association with improved clinical outcome. Eur J Cancer. 2013;49(S4):S12. doi: 10.1158/1078-0432.CCR-14-2334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Strasser EF, Zimmermann R, Weisbach V, Ringwald J, Zingsem J, Eckstein R. Mononuclear cell variability and recruitment in non-cytokine-stimulated donors after serial 10-liter leukapheresis procedures. Transfusion. 2005;45:445–52. doi: 10.1111/j.1537-2995.2005.04202.x. [DOI] [PubMed] [Google Scholar]
- 41.Strauss RG. Effects on donors of repeated leukocyte losses during plateletpheresis. J Clin Apher. 1994;9:130–4. doi: 10.1002/jca.2920090208. [DOI] [PubMed] [Google Scholar]
- 42.Flanigan RC, Polcari AJ, Shore ND, Price TH, Sims RB, Maher JC, et al. An analysis of leukapheresis and central venous catheter use in the randomized, placebo controlled, phase 3 IMPACT trial of Sipuleucel-T for metastatic castrate resistant prostate cancer. J Urol. 2013;189:521–6. doi: 10.1016/j.juro.2012.09.029. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.