Abstract
The Nlrp3 inflammasome plays an important role in inflammation by controlling the maturation and secretion of the cytokines IL-1β and IL-18 in response to multiple stimuli including pore-forming toxins, particulate matter, and ATP. Although the pathways activated by the latter stimuli lead to a decrease inintracellular K+concentration,which is required for inflammasome activation,the mechanism by whichmicrobial RNA activates Nlrp3, remains poorly understood. Here we found that cytosolic poly(I:C), but not total RNA from healthy macrophages, macrophages undergoing pyroptosis, or mitochondrial RNA, induces caspase-1 activation and IL-1β release through the Nlrp3 inflammasome. Experiments with macrophages deficient in Tlr3, Myd88 orTrif, indicate that poly(I:C)induces Nlrp3 activation independently of TLR signaling. Further analyses revealed that the cytosolic sensorsRig-I andMda5act redundantly viathe common adaptor Mavsto induceNlrp3 activation in response topoly(I:C), but not ATP or Nigericin. Mechanistically, Mavstriggeredmembrane permeabilizationand K+ efflux independently of the inflammasome which were required for poly(I:C)-induced Nlrp3 activation. We conclude thatpoly (I:C)activates the inflammasome through an Mavs-dependent surveillance pathway that convergesinto a common K+ lowering step in the cytosol that is essential for the induction of Nlrp3 activation.
Keywords: Nlrp3; inflammasome; IL-1, dsRNA; Mavs; innate immunity
Introduction
A key step in the induction of inflammatory responses during infection is the activation of pattern-recognition receptors (PRRs) expressed on innate immune cells.PRRs sense conserved molecules produced by microbes and/or endogenous changes induced during cellular injury(1, 2). Upon activation, PRRs trigger multiple signaling pathways that promote pathogen clearance and tissue repair (3). A major signaling pathway induced in response to infectionis the inflammasome, a multi-protein complex that activates caspase-1 (4, 5). Once activated, caspase-1 proteolytically cleaves pro-interleukin-1β and pro-IL-18 into their biologically active forms (6). To date, four bonafide inflammasomes have been described of which three, the Nlrp1, Nlrp3 and Nlrc4 inflammasomes, are activated by members of the intracellular Nod-like receptor (NLR) family (5).The Nlrp3 inflammasome has received considerable attention due to its link to the pathogenesis of autoinflammatory syndromes and a variety of inflammatory diseases (7-9). Production of IL-1β through Nlrp3 is tightly regulated via a two-step process. The first step, referred as priming, involves the transcriptional induction of pro-IL-1β and Nlrp3by Toll-like receptor (TLR) agonists or certain cytokines such as TNF-α or IL-1β (10, 11). Both reactive oxygen species (ROS) and NF-κB activation have been shown to regulate the priming step (10-12). The second step involves the activation of Nlrp3 to assemble an active inflammasome complex that can process pro-IL-1β into mature IL-1β. Multiple stimuli including extracellular ATP, bacterial pore-forming toxins, particulate matter and microbial RNA have been reported to activate Nlrp3 independently of the priming step (5, 13, 14).
The mechanism that activates Nlrp3 has been the subject of intense investigation. Several events have been linked to the activation of Nlrp3including the production of ROS(15), mitochondrial damage(16), lysosomal damage(17), cell swelling (18)and cytosolic K+ efflux(19, 20). In addition, several molecules released upon mitochondrial and/or ROS-induced damage have been associated with Nlrp3 activation(21, 22). Understanding the steps required for Nlrp3 activation has been difficult because experiments often relied on chemical inhibitors or the use of activating stimuli such as ATP that trigger multiple cellular events some of which are induced in parallel or downstream of Nlrp3 activation (5, 13, 14, 20). Nonetheless, K+ efflux has been mechanistically linked to Nlrp3 activation induced by ATP, pore-forming toxin and particulate matter by multiple studies(23-25). Furthermore, recent studies showed that depletion of cytosolic K+ is sufficient to trigger the activation of Nlrp3(24). However, the mechanism by which microbial RNA triggers activation of Nlrp3 remains unclear. Microbial RNAs activate PRRs including Tlr3 and the RIG-like receptors (RLRs)Rig-I/Ddx58 and Mda5/Ifih1 (melanoma differentiation-associated gene 5). Rig-Iand Mda5 recognize double-stranded RNA (dsRNA) generated during viral replication and polyinosinic polycytidylic acid (poly I:C), a synthetic analog of dsRNA (26, 27) and induce type I IFNs and NF-κB via Mavs (mitochondrial antiviral signaling protein) (28).In addition Mavs and Trif/Ticamhave been linked to Nlrp3 activation in triggered by poly I:C(29), viruses (29-32) and bacteria(33), but it remains unclear whether Mavs and Trif act bypriming the inflammasome (10, 11),or byregulating Nlrp3 localization (34) or activation(32). Recent studies have linked microbial RNA to Nlrp3inflammasome activation in human macrophages through the binding of dsRNA to the DHX33 RNA helicase (35). Furthermore, stimulation of human macrophages with poly(I:C) induced the interaction of DHX33 with Nlrp3 and ASC, although it remains unclear whether such interactions are sufficient to activate the Nlrp3 inflammasome (35). Collectively, these studies suggestedthat dsRNA activates Nlrp3 by a mechanism that is distinct to that induced by ATP, pore-forming toxins and particulate matter. In the present studies, we have analyzed further the mechanism by which dsRNA activates the Nlrp3 inflammasome and provide evidence that cytosolic dsRNA inducesmembrane permeabilizationvia Mavsindependently of inflammasome activation which in turn leads to K+ efflux and Nlrp3 activation.
Material and Methods
Mice
Nlrp3−/−, Asc−/−, casp1/11−/− mice have been described (36, 37). P2rx7−/− mice wereobtained from G. Dubyak, Case Western Reserve University (Cleveland,OH). Tlr3−/−, Myd88/Ticam−/− and Mavs−/−/Ticam−/−, RigI/Ddx58−/−,Mda5/Ifih1−/−and RigI−/−Mda5−/− micehave been described (38-41). Pkr−/− mice from R. Kaufman, University of Michigan (Ann Arbor, MI). All mice were backcrossedonto the C57BL/6 background at least eight times. Wild-type (WT) C57BL/6 mice weremaintained in our animal facility. All animal studies were approvedby the University of Michigan Committee on Use and Care of Animals.
Reagents
ATP was from Sigma-Aldrich. Recombinant mouse TNF-α was obtained from Peprotech. Ultrapure LPS from Escherichia coli, Pam3CSK, flagellin, high and low molecular weight polyinosinic:polycytidylicacid (hereafter referred to as poly(I:C)), CpG, double-stranded B DNA poly(dA-dT)•poly(dT-dA) (poly(dA:dT)) and nigericin were from InvivoGen. Total Escherichia coli RNA and mouse heart total RNA were purchasedfrom Ambion. Lipofectamine 2000 was obtained from Invitrogen. The ROS inhibitor N-acetyl-L-cysteine (NAC) was purchased from Sigma.Z-VAD-FMK (carbobenzoxy-valyl-alanyl-aspartyl-[O-methyl]-fluoromethylketone) and Necrostatin were from Enzo Life Sciences.
RNA isolation
Liver mitochondria were purified as previously described (42). For isolation of total RNA from pyroptotic cells, BMDM were stimulated with Pam3CSK (10 μg/ml) for 1 h followed by incubation with ATP (5mM) for 30 min. RNA was isolated using a DNA/RNA isolation kit from Qiagen. To obtain RNA, total DNA/RNA was digested with RNase-Free DNase Set (Quiagen).
Cell culture and stimulation
BMDM were isolated as previouslydescribed (43). Cells were stimulated in IMDM supplemented with 10% FCS at a density of 1 × 106 per ml. Unless otherwise specified, BMDM were transfected with high molecular weight poly(I:C), bacterial RNA or mitochondrial RNA complexed with Lipofectamine 2000 or transfected with poly(dA:dT) complexed with Lyovec for 6-8 h at a concentration of 10 μg/ml. Where indicated, BMDM were prestimulated with LPS (100 ng/ml) for 1 h or with TNF (100 ng/ml)for 6 h and washed before restimulation with the indicated ligands. As a positive control for caspase-1 activation, WT macrophageswere incubated with 100 ng/ml LPS for 3 h and stimulatedfor 30 min either with 5 mM ATP or with nigericin (10 μg/ml). For inhibitor experiments, LPS-primed BMDM were preincubated with N-Acetyl-Cystein (NAC; 30 mM), pan-caspase inhibitor Z-VAD (40 μM) or necrostatin (20μM) for 1 h prior to stimulation with poly(I:C) or ATP.
Immunoblotting and cytokine measurements
For Western Blot analysis, cells were lysed by adding NP-40 to the media to a final concentration of 1 % and protease inhibitors. Cleared lysates were separated by 12% SDS-PAGE, transferred to PVDF membranes and probed with a rabbit anti-caspase-1 antibody generated in our laboratory. Concentrations of IL-1β(R&D Systems) and IL-18 (MBL International) were measuredby ELISA in cell-free culture supernatant.
Membrane permeabilization
Membrane permeabilizationwas assessed by measuring the release of cytosolic lactate dehydrogenase (LDH) into cell culture supernatants using the The CytoTox 96® Non-Radioactive Cytotoxicity Assay Kit from Promega according to the manufacturer’s instructions.
Determination of intracellular K+ content
Intracellular K+ measurements were performed by inductively-coupled plasma optical emission spectrometry (ICP-OES) with a Perkin-Elmer Optima 2000 DV spectrometer using yttrium as internal standard. The culture media were thoroughly aspirated and cells were extracted 30 min in 3% ultrapure HNO3. K+ determinations were done in 48-well plates. For accurate measurement of the intracellular K+, a control was performed in every experiment to determine the extracellular amount of K+ after aspiration and this value was subtracted from every measurement. In experiments using high and low extracellular K+, osmolarities were measured by freezing-point depression osmometry using a μOsmette (Precission systems). In experiments using high and low extracellular K+, media osmolarities were adjusted to 300 mOsm after adding the respective salts using a μOsmette freezing-point depression osmometer (Precission systems).
Statistical analysis
Statistical significance between groups was determined by two-tailedStudent’s t-test. Differences were considered significantwhen p< 0.05.
RESULTS
Microbial RNA, but not mammalian RNA or RNA species generated during pyroptosis, induces the activation of the Nlrp3 inflammasome
Consistent with previous studies(36, 44, 45), we found that transfection of bone marrow-derived macrophages (BMDM) with poly(I:C), a synthetic dsRNA analog mimicking viral RNA, but not that of total RNA prepared from healthy macrophagesinduced the release of IL-1β in WT, but not in Nlrp3-deficient, macrophages (Supplemental Fig. 1A). We next tested whether RNA from macrophages stimulated with ATP to induce Nlrp3 activation could elicit inflammasome activation. We found that transfection of total RNA from macrophages stimulated with ATP to undergo Nlrp3 activation, or mitochondrial RNA did not induce IL-1β release in LPS-primed macrophages (SupplementalFig. 1A). As a control, a preparation enriched in mitochondrial DNA, induced IL-1β release independently of NLRP3 (SupplementalFig. 1A). Consistently, caspase-1 activation induced by bacterial RNA and poly(I:C), but not pyroptotic RNA, was abolished in LPS-primed macrophages deficient in Nlrp3 or Asc(SupplementalFig. 1B). In contrast, stimulation with flagellin, an activator of the Nlrc4 inflammasome(46, 47), or poly(dA:dT), an activator of the Aim2 inflammasome(48), induced comparable caspase-1 activation in WT and Nlrp3-deficient macrophages (SupplementalFig. 1B). As expected, both activation of caspase-1 by cytosolic poly(dA:dT) and flagellin required the common adaptor Asc (SupplementalFig. 1B). These results indicate that microbial RNA, but not mammalian RNA or RNA species generated during pyroptosis, induces the activation of the Nlrp3 inflammasome.
Cytosolic poly(I:C) activates the Nlrp3 inflammasome independently of TLR signaling
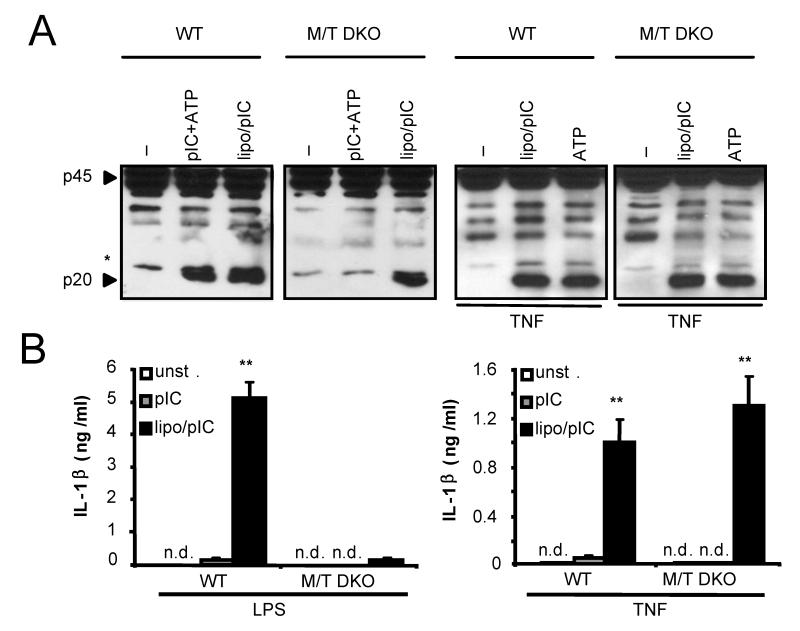
Poly(I:C) activates both Tlr3-Trif and cytosolic Rig-I/Mda5 signaling pathways. We first addressed whether TLRsare required for the activation of the Nlrp3 inflammasome induced by poly(I:C). To this end, we compared the activation of caspase-1 in WT BMDM or macrophagesdeficient in Myd88 and Trif, the two adaptors required for TLR signaling. Priming of macrophages with extracellular poly(I:C) promoted the activation of caspase-1 by ATP in WT, but not in macrophagesdeficient in Myd88/Trif (Fig.1A), which is consistent with the fact that extracellular poly (I:C) signals via Tlr3. In contrast, transfection of macrophageswith poly(I:C) conjugated with lipofectamine to mediate cytosolic stimulation induced comparable activation of caspase-1 in WT and Myd88/Trif-deficient cells(Fig.1A). Notably, cytosolic, but not extracellular, poly(I:C)induced the activation of caspase-1 even in in unprimed Myd88/Trif-deficient DCs further supporting the conclusion that cytosolic poly(I:C)-induced caspase-1 activation is independent of TLR signaling (Fig.1A). To test whether cytosolic poly(I:C) is sufficient to induce cytokine production, we assessed the production of IL-1β and IL-18 in unprimed BMDM, which do not express pro-IL1β but express low levels of pro-IL-18. In unprimed cells, cytosolic poly(I:C) induced the secretion of IL-18, but not of IL-1β (Supplemental Fig. 2). Consistent with these results, cytosolic poly(I:C) stimulation induced the upregulation of Nlrp3mRNA (Supplemental Fig. 2). These results indicate that cytosolic poly(I:C) is sufficient to induce Nlrp3 priming, activation of caspase-1 and IL-18 release, but not IL-1β production. In macrophagesprimed with LPS, transfection with poly(I:C) and lipofectamine, but not extracellular poly(I:C) alone, induced IL-1β release which required Myd88 consistent with the requirement of TLR signalling for LPS or extracellular poly(I:C) priming (Fig.1B). Likewise, both ATP or cytosolic poly(I:C) stimulation induced comparable caspase-1 activation and IL-1βrelease in WT and Myd88/Trif-deficient cells primed with TNF-α (Fig.1A,B). Consistent with the inability of extracellular poly(I:C) to induce caspase-1 activation, little or no secretion of IL-1β was detected when TNF-primed DCswere stimulated with poly(I:C)in the absence of lipofectamine (Fig.1B). These results indicate that cytosolic poly(I:C), but not extracellular poly(I:C), induces the activation of the Nlrp3 inflammasome independently of TLR signalling.
Figure 1.
Cytosolic poly(I:C) induces Nlrp3 activation independent of TLR signaling. (A) BMDC derived from WT or Myd88−/−/Ticam−/− DKO mice were primed with poly(I:C) (10μg/ml) or TNF (100ng/ml) for 6 hours and then stimulated with ATP (5 mM) for 30 minutes or stimulated directly with poly(I:C) conjugated with lipofectamine for 6 hours. Extracts were prepared from cells plus culture supernatants and immunoblotted with an antibody detecting active caspase-1 (p20). (B) WT or Myd88−/−/Ticam−/− DKO BMDC were primed with LPS or TNF and stimulated with poly(I:C) conjugated, or not, with lipofectamine for 6 hours. IL-1β was measured in cell-free supernatants by ELISA. Values represent the mean of triplicate wells ± SD. (**) p<0.01. Results are representative of three independent experiments.
Microbial RNA induces Nlrp3 activation via Mavs
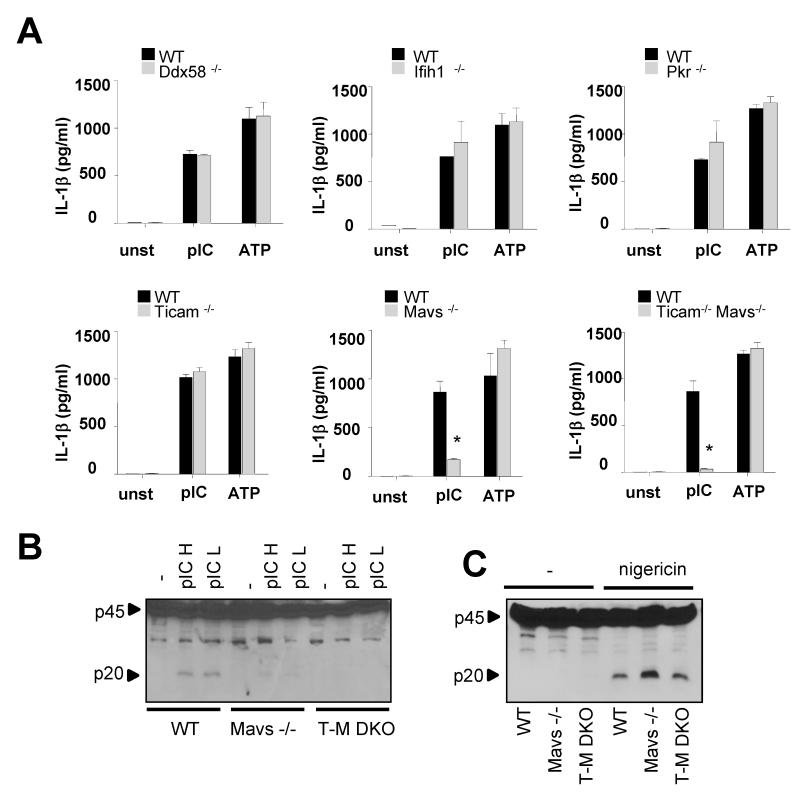
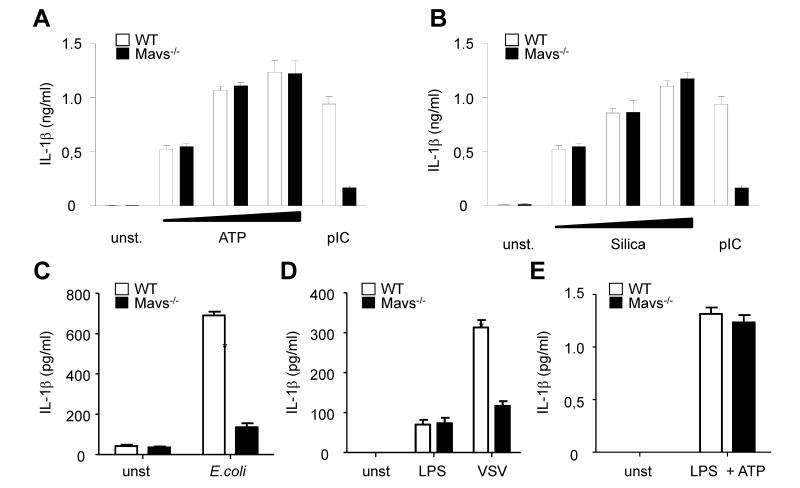
We next assessed the contribution of the cytosolic surveillance pathways in the activation of the Nlrp3 inflammasome induced by poly(I:C). To this end, LPS-primed WT macrophages or LPS-primed macrophages deficient in the cytosolic RNA sensors Rig-I, Mda5, or Pkr were stimulated with cytosolic poly(I:C) and the secretion of IL-1β was measured. We found that macrophagesdeficient in Rig-I, Mda5, or Pkr were not impaired in the release of IL-1β compared to WT cells, suggesting that the recognition of dsRNA by these receptors is dispensable for the activation of the Nlrp3inflammasome (Fig.2A). We next assessed the role of Mavs and Trif in inflammasome activation, as these two adaptors are used respectively by Rig-I/Mda5and Tlr3 to initiate signal transduction pathways(49). The secretion of IL-1β induced by cytosolic poly(I:C) proceeded unabated in Trif-deficient macrophages (Fig.2A). In contrast, the secretion of IL-1β induced by cytosolic poly(I:C) was greatly reduced in Mavs deficient macrophages and abrogated in Mavs/Trif-deficient macrophages (Fig.2A). Consistent with the IL-1β results, the activation of caspase-1 induced by cytosolic poly(I:C) was greatly reduced in Mavs deficient and abolished in macrophages double deficient in Mavs and Trif (Fig.2B). In contrast, the activation of the Nlrp3 inflammasome induced by Nigericin was comparable in WT and macrophages deficient in Mavs or Mavs/Trif (Fig.2C). Similarly, dose response experiments showed that Mavs was dispensable for the activation of the Nlrp3inflammasome induced by ATP or silica (Fig.3A,B). These results indicate that Mavs plays an importantrole in the activation of the Nlrp3inflammasome induced by cytosolic poly(I:C), but is dispensable for caspase-1 activation triggered by other Nlrp3 activators. To further confirm the role of Mavs in the activation of the Nlrp3 inflammasome induced by microbial RNA, we infected macrophages with Escherichia coli or DCs with Vesicular Stomatitis Virus (VSV), in which microbial RNA has been previously shown to induce Nlrp3 activation (29, 33, 36, 44, 50). We found that IL-1β secretion in macrophages infected with E. coli or DCs infected with VSV was severely impaired in Mavs-deficient cells compared to WT cells (Fig.3C,D). As observed in BMDM, in BMDC the activation of caspase-1 induced by cytosolic poly (I:C) was Nlrp3 dependent (Supplemental Fig. 3). Consistent with previous results in macrophages, the secretion of IL-1β induced by LPS+ATP was comparable in WT and Mavs deficient DCs (Fig.3E). Altogether, these results indicate thatMavs is important for Nlrp3 inflammasome activation induced by microbial RNA but dispensable for the activation of Nlrp3 induced by ATP, particulate matter and bacterial pore-forming toxins.
Figure 2.
Poly(I:C) induces inflammasome activation via Mavs. (A) LPS-primed BMDM derived from WT, Ddx58−/−, Ifih1−/−, Pkr−/−, Ticam−/−, Ips1−/− or Ticam−/−/Ips1−/− DKO mice were stimulated with high (pIC H) or low (pIC L) molecular weight poly(I:C) conjugated with lipofectamine for 6 hours or ATP or nigericin for 30 min. (A) IL-1β secretion was assessed by ELISA in cell free supernatant. Values represent the mean of triplicate wells ± SD. (*) p<0.01. (B-C), extracts were prepared from cells plus culture supernatants and immunoblotted with an antibody detecting active caspase-1 (p20). All results are representative of three independent experiments.
Figure 3.
VSV and E. coli, but not silica or ATP, induce inflammasome activation via Mavs. (A C) LPS-primed BMDM or (D-E) DC derived from WT or Mavs−/− mice were stimulated with ATP (A) (1,25mM; 2,5mM; 5mM) for 30 minutes, or Silica (B) (100μg/ml; 200μg/ml, 400 μg/ml) for 6 hours, or poly(I:C) conjugated with lipofectamine for 6 hours or infected with E. coli or VSV for 24 hours (C-D). IL-1β secretion was assessed by ELISA in cell free supernatant. Values represent the mean of triplicate wells ± SD. (*) p<0.01. (A-D) Results are representative of three independent experiments.
Poly(I:C) induces membrane permeabilization independent of the inflammasome
In addition to inducing the secretion of IL-1β and IL-18, the activation of the inflammasome triggers a form of cell death called pyroptosis, which is characterized by the caspase-1 dependent disruption of cell membrane integrity and the release of intracellular contents into the extracellular milieu(51). We next investigated whether poly(I:C) induces membrane permeabilization by measuring the release of LDH which in healthy cells is found in the cytosol. We found that cytosolic poly(I:C) and Nigericin,which was used as a positive control, compromisedthe cell membrane integrity as assessed by the release of LDH (Supplemental Fig. 4). As expected, membrane permeabilization induced by Nigericin was blocked in Nlrp3- or caspase-1-deficient macrophages (Supplemental Fig. 4A,B). Surprisingly, loss of cell membrane integrity induced by cytosolic poly(I:C) was unimpaired in Nlrp3 or caspase-1 deficient macrophages (Supplemental Fig. 4A,B), indicating that cytosolic poly(I:C) induced membrane permeabilization is independent of pyroptosis. To further explore the mechanism by which cytosolic poly(I:C) induces membrane permeabilization, we tested whether the release of LDH was caused by necroptosis, a form of cell death in which disruption of cell membrane integrity is dependent on RIP1 and RIP3 (52). As expected with previous studies (53), necrostatin, an inhibitor of RIP1, blocked necroptosis and membrane permeabilization induced by co-stimulation of L929 cells with TNF-α and ZVAD which was used as a positive control (Supplemental Fig. 4C). Necrostatin, however, had no effect on membrane damage induced by cytosolic poly(I:C) in macrophages (Supplemental Fig. 4D). Furthermore, ZVAD, a pan-caspase inhibitor that efficiently prevents apoptosis, was not effective in blocking cytosolic poly(I:C)-induced membrane permeabilization, excluding the possibility that membrane damage was caused by secondary necrosis of apoptotic cells. These results suggest that cytosolic poly(I:C)-induced membrane permeabilization is independent of caspase-1, necroptosis or apoptosis.
Poly(I:C) induces membrane permeabilization primarily via Mavs
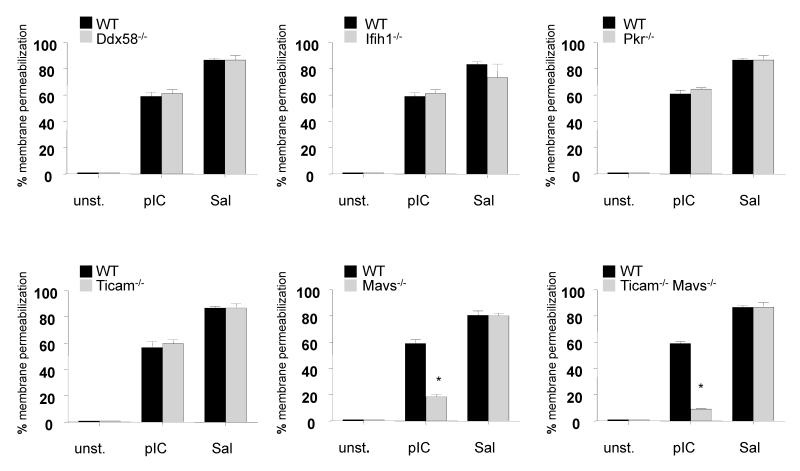
We next investigated whether membrane permeabilization was caused by the detection of poly(I:C) via sensors of dsRNA. To this end, we stimulated WT and macrophages deficient in the cytosolic RNA sensorsRig-I, Mda5,Pkr or the adaptor protein Trif and Mavs with cytosolic poly(I:C) or Salmonella as a control and measured membrane permeabilization by measuring the release of LDH. Cytosolic poly(I:C)-induced LDH release was reduced in macrophage deficient in Mavsand Mavs/Trif, but not in Rig-I, Mda5, Pkr or Trif (Fig.4). The role of Mavs was specific in that LDH release induced by Salmonella that triggers inflammasome activation via Nlrc4 did not require Mavs (Fig.4). Collectively these results indicate thatMavs is important for both poly(I:C)-induced membrane permeabilization and the activation of the Nlrp3inflammasome.
Figure 4.
dsRNA induces membrane permeabilization via Mavs. LPS-primed BMDM derived from WT, Ddx58−/−, Ifih1−/−, Pkr−/−, Ticam−/−, Mavs−/− or Ticam−/−/Mavs−/− DKO (T/M DKO) mice were stimulated with poly(I:C) (10 μg/ml) conjugated with lipofectamine or infected with Salmonella. Membrane permeabilization was evaluated after 6 hours by analyzing the release of cytosolic LDH in the supernatant. Values represent the mean of triplicate wells ± SD. (*) p<0.01 Results are representative of three independent experiments.
Poly(I:C)-induced membrane permeabilization causes potassium efflux which is required for inflammasome activation
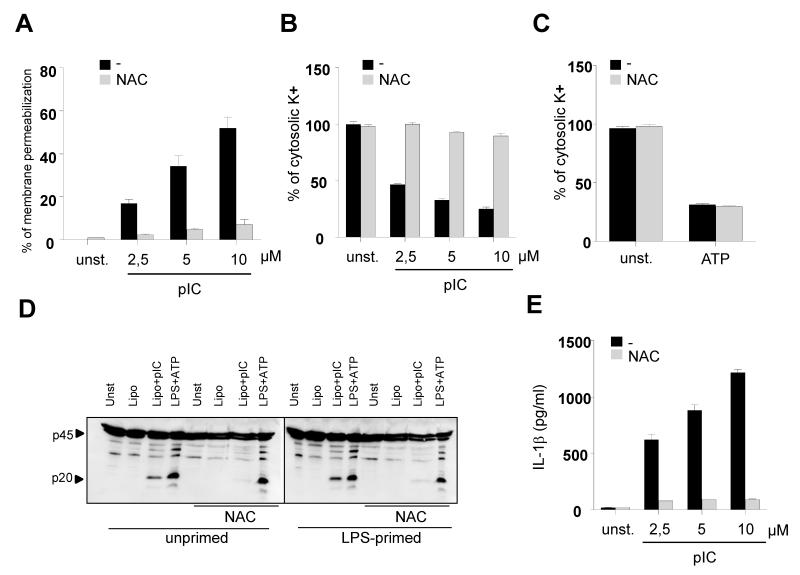
The K+ concentration inside cells is actively controlled by the Na+/K+-ATPase, a membrane transport protein that pumps Na+ outside and K+ inside the cell to achieve relatively high concentrations of K+ and low concentrations of Na+ in the cytosol. However, a compromise in cell membrane integrity can cause the efflux of K+. Because a decrease in the intracellular concentration ofK+is important for Nlrp3 activation induced by ATP, pore-forming toxins and particulate matter(24), we investigated whether cytosolic Mavs-dependent membrane permeabilization induced by poly(I:C) could cause a net decrease of intracellular K+. Cytosolic stimulation of macrophages with poly(I:C) induced membrane permeabilization and a decrease in the intracellular content of K+in a dose-dependent manner (Fig.5A, B). As ROS have been previously shown to cause membrane damage(54, 55), we then tested whether poly(I:C)-induced membrane damage was prevented by inhibiting ROS with the free radical scavenger N-acetylcysteine (NAC). NAC blocked poly(I:C)-induced membrane permeabilization (Fig.5A) and prevented K+ efflux (Fig.5B). Consistent with a specific effect of NAC in blocking poly(I:C)-induced K+ efflux, the lowering of intracellular K+ induced by ATP, which occurs in response to P2RX7 receptor stimulation, was not affected by NAC (Fig.5C). To further confirm the role of ROS in Mavs-dependent activation of Nlrp3, we stimulated macrophages with cytosolic poly(I:C) or ATP in the presence of NAC. Consistent with a previous publication (12, 24), NAC did not inhibit caspase-1 activation induced by ATP in primed cells (Fig.5D), but blocked effectively cytosolic poly(I:C)-induced caspase-1 activation (Fig.5D) and IL-1β secretion (Fig.5E). These results confirm that in primed cells ROS are not necessary for Nlrp3 activation and suggests that in response to poly IC Mavs-dependent ROS production triggers K+ efflux by causing cell membrane damage.
Figure 5.
dsRNA-induced membrane permeabilization is required for K+ ellux and inflammasome activation. Unprimed and LPS-primed BMDM were pretreated with NAC (30 mM) and stimulated with poly(I:C) (10 μg/ml) conjugated with lipofectamine for 6 hours (A-E) or with ATP (C-D) (5 mM) for 30 minutes. (A and E) Membrane permeabilization was evaluated by analyzing the release of cytosolic LDH in the supernatant. (B-C) intracellular K+ content was evaluated by ICP/MS. (D) Caspase-1 activation was evaluated by immunblotting with an antibody detecting active caspase-1 (p20). (E) IL-1β was evaluated by ELISA in cell free supernatant. (A-C and E) Values represent the mean of triplicate wells ± SD. (*) p<0.01 (A-E) Results are representative of three independent experiments.
Activation of the Nlrp3-inflammasome by poly(I:C) requires K+ efflux
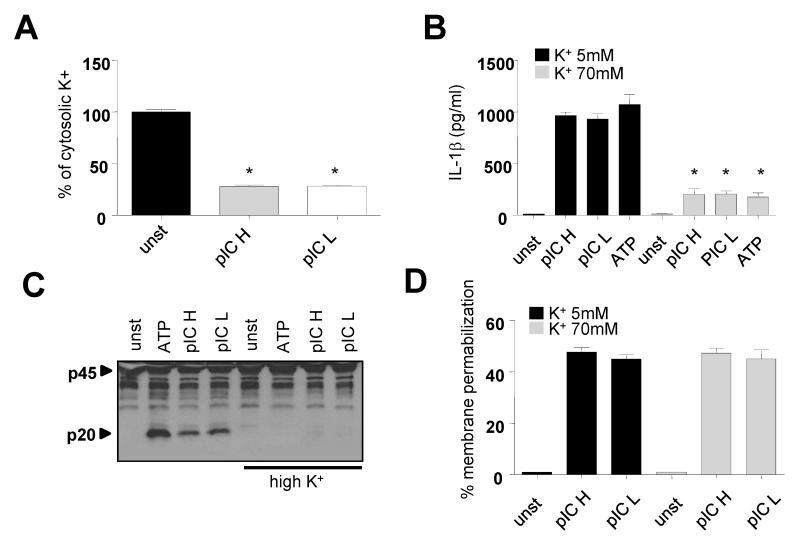
We have shown that Mavs-induced membrane permeabilization triggers K+ efflux. We next investigated whether Mavs induced K+ efflux was required for Nlrp3 activation. To prevent K+ efflux caused by cytosolic poly(I:C),we stimulated macrophages inisotonic medium containing 70 mM K+ and assessed the activation of the Nlrp3 inflammasome and LDH release. As expected, transfection of high and low molecular weight poly(I:C) with lipofectamine induced K+ efflux, caspase-1 activation and IL-1β secretion (Fig. 6A-C). In contrast, caspase-1 and IL-1β secretion induced by cytosolic poly(I:C) were prevented by incubation of macrophages in high (70 mM), but not low (5 mM) extracellular K+ (Fig.6B,C). Notably, media containing high (70 mM) K+ concentration did not prevent poly(I:C)-induced LDH release (Fig.6D), further confirming that LDH release occurs upstream of K+ efflux. Collectively, these results indicate that Mavs-induced membrane permeabilization causes K+ efflux which in turn induces Nlrp3 activation.
Figure 6.
Activation of the Nlrp3-inflammasome by poly(I:C) requires K+ efflux. A, LPS- primed BMDM were stimulated with high (pIC H) or low (pIC L) molecular weight poly(I:C) conjugated with lipofectamine for 6 hours (A-D) or ATP for 30 min (B-C), in buffer containing low (5mM) or high (70mM) K+ concentration (B-D). (A) Intracellular K+ was evaluated by ICP/MS, (B) IL-1β secretion was evaluated by ELISA in cell free supernatant. (C), caspase-1 activation was evaluated by immunblotting with an antibody detecting active caspase-1 (p20) and (D) membrane permeabiliziation was evaluated by analyzing the release of cytosolic LDH in the supernatant. (A, B and D). Values represent the mean of triplicate wells ± SD. (*) p<0.01. (A-D) Results are representative of three independent experiments.
Rig-I and Mda5 act redundantly via the common adaptor Mavs to induce membrane permeabilization, K+ efflux and Nlrp33 activation
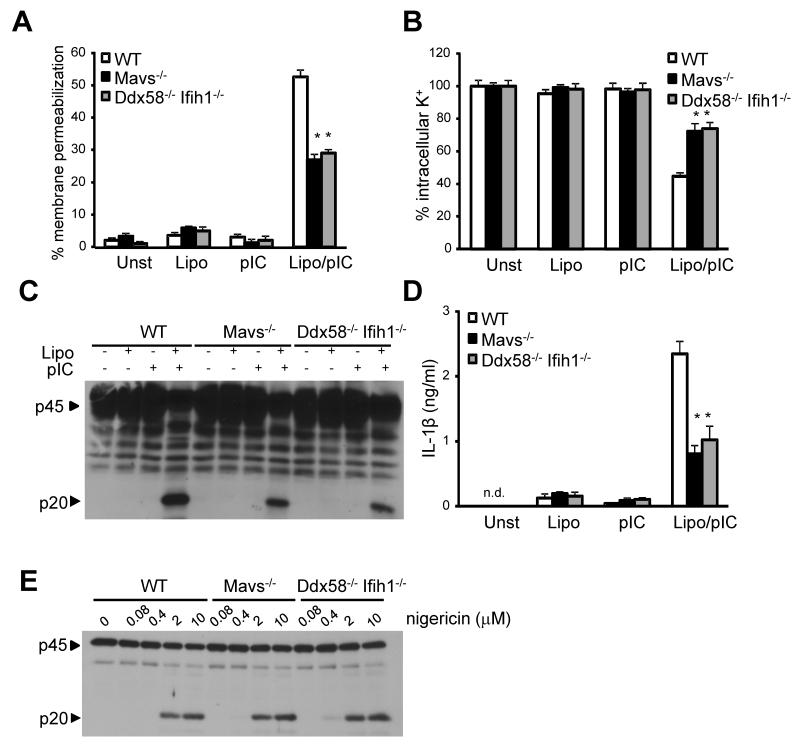
We have shown Nlrp3 activation induced by cytosolic poly(I:C) proceeds unabated in Rig-I or Mda5 deficient cells, but is greatly impaired in the Mavs deficient cells, a common adaptor in both Rig-I and Mda5 signaling pathway. Next we explore the possibility that Rig-I and Mda5 act redundantly to induce Nlrp3 activation in response ot cytosolic poly(I:C). To this end, we stimulated BMDM deficient in Mavsor deficient in both Rig-I and Mda5and assessed membrane permeabilization, K+ efflux, caspase-1 activation and IL-1β secretion after cytosolic poly(I:C) stimulation. We found that membrane permeabilization (Fig. 7A), K+ efflux (Fig. 7B), caspase-1 activation (Fig. 7C) and IL-1β secretion (Fig. 7D) was impaired in BMDM deficient in both Rig-I and Mda5. Furthermore, we observed a comparable reduction of membrane permeabilization (Fig. 7A), K+ efflux (Fig. 7B), caspase-1 activation (Fig. 7C) and IL-1β secretion (Fig. 7D) in BMDM doubly deficient in Rig-I and Mda5 and deficient in Mavs. The partial reduction in caspase-1 activation and IL-1β secretion in BMDM doubly deficient in Rig-I and Mda5 and deficient in Mavs is consistent with results shown in Fig. 2 that revealed the involvement of a Trif-dependent pathway in poly(I:C)-induced caspase-1 activation. In contrast, Nlrp3 activation induced by Nigericin proceeded unabated in BMDM deficient in both Rig-I and Mda5 or BMDM deficient in Mavs (Fig. 7E).Collectively, these results indicate that Rig-I and Mda5 act redundantly via the common adaptor Mavs to induce membrane permeabilization and K+ efflux triggering Nlrp3 activation.
Figure 7.
Rig-I and Mda5 act redundantly via the common adaptor Mavs to induce NLRP3 activation. (A-D) LPS-primed BMDM derived from WT, Ddx58−/−Ifih1−/− DKO or Mavs−/− mice were stimulated poly(I:C) conjugated, or not, with lipofectamine for 6 hours (A-D) (A) membrane permeabilization was evaluated by analyzing the release of cytosolic LDH in the supernatant (B) Intracellular K+ was evaluated by ICP/MS. (C) Caspase-1 activation was evaluated by immunoblotting with an antibody detecting active caspase-1 (p20). (D) IL-1β secretion was evaluated by ELISA in cell free supernatant. (A, B and D). Values represent the mean of triplicate wells ± SD. (*) p<0.01. (A-D) Results are representative of three independent experiments.
Discussion
In this study we have identified a signaling pathway that mediates the sensing of dsRNA in the cytosol and activates the Nlrp3 inflammasome. Experiments with Tlr3 and Myd88/Trif deficient mice showed that cytosolic poly(I:C) induced Nlrp3 activation proceedsindependently of TLR signalling. Furtheremore, experiments using LPS-primed cells, in which pro-IL-1β and Nlrp3 are upregulated independently of Mavs, indicate that cytosolic poly(I:C) induces Nlrp3 activation. An important component of the dsRNA-induced Nlrp3 pathway is the adaptor Mavs which was critical for the induction of membrane permeabilization, K+ efflux and caspase-1 activation. In our studies, we found that Mavs was critical for Nlrp3 activation, but RigI or Mda5 were dispensable for the induction of membrane permeabilization, caspase-1 activation and IL-1β release. Rig-I and Mda5 recognize dsRNA and upon activation they initiate signaling by recruiting Mavs(26). Experiment using BMDM deficient in both Rig-I and MDA5stimulated with cytosolic poly (I:C) showed an inhibition of membrane permeabilization, K+ efflux, caspase-1 activation and IL-1β secretionthat were comparable to that observed in Mavs deficient cells, indicating that Rig-I and MDA5 act redundantly via the common adaptor Mavs to induce Nlrp3 activation. Recently, Subramanian et al. showed that Nigericin and ATP-induced Nlrp3 activation wereMavs dependent(34). In contrast, our results and recent studies (32, 56), found that Mavs was not required for Nlp3 activation induced by Nigericin or ATP. In addition to the major Rig-I/Mda-5/Mavs pathway, we found a role for a Trif-dependent pathway in Nlrp3 activation induced by poly(I:C). Further experiments are needed to determine if the Trif pathway involves TLR3 stimulation or a different Trif-dependent pathway activated by poly(I:C). A recent model suggested that Nlrp3 is activated via a direct interaction of Nlrp3 with the DHX33 helicase induced upon recognition of dsRNA by DHX33 in human cells (35). Thus, it is possible that both Mavs-dependent K+ efflux and direct interaction between DHX33 with Nlrp3 are important in triggering the activation of Nlrp3 in response to cytosolic dsRNA. Collectively, these results indicate that recognition of dsRNA by cytosolic RLRs induces via Mavs the activation of the Nlrp3 inflammasome as well as signaling pathways leading to type I IFNs and NF-κB activation.
Previous studies showed that K+ efflux is critical for the activation of the Nlrp3 inflammasome induced by ATP, pore-forming toxin and particulate matter (24). However, the mechanism by which dsRNA triggers activation of Nlrp3remains unclear. We showed in the current work that cytosolic dsRNA triggers K+ efflux, which was required for Nlrp3 activation. Notably, K+ efflux induced by dsRNA required Mavs-mediated membrane permeabilization which waseffectively blocked by NAC, but was independent of caspase-1. Intriguingly, cytosolic stimulation of DCs with poly(I:C) can induce an Mavs-dependent cell death pathway that enhances the production of type I IFNs, IL-6 and NF-κB activation (57). However, it is unclear whether the cytosolic pathway described by Zou et al. induces caspase-1 activation and IL-1β release. Unlike the latter Mavs-mediated pathway, the cytosolic poly(I:C)-induced pathway leading to Nlrp3 activation was unaffected by treatment with inhibitors of necroptosis(57). These results suggest that the Mavs-dependent cell death pathways enhancing type I IFNs/NF-κB activation and Nlrp3 activation are distinct. An interesting observation is that polyIC and bacterial dsRNA, but not RNA from pyroptotic macrophages or mitochondria, trigger Nlrp3 activation. These results suggest that RNA structuressuch as the 5′ triphosphate group and blunt end of short dsRNAs or long-duplex RNAs that are important for Rig-I and Mda-5 recognition are critical for Nlrp3 activation (58, 59). The observation that NAC inhibits K+ efflux suggests that Mavs activation induced by dsRNA mediates membrane damage through ROS which leads to K+efflux to induce Nlrp3 activation. Consistent with recent studies (12, 24), Nlrp3 activation induced by ATP was unaffected by NAC. Furthermore, extracellular media containing 70 mM K+ prevented caspase-1 activation, but not dsRNA-induced membrane permeabilization, indicating that K+ efflux is induced downstream of membrane damage.Collectively, these results indicate thatcytosolic dsRNA sensing triggersmembrane permeabilization via Mavs and ROS which in turn leads to K+ efflux and Nlrp3 activation. Consistent with this model, treatment of macrophages with certain chemical agents that induce necrosis which is associated with plasma membrane damage, or high dose of lipofectamine (data not shown),can elicit caspase-1 activation and IL-1β release via the Nlrp3inflammasome(60). Further studies will be necessary to better understand the mechanism by which ROS are generated in response to cytosolic dsRNA and how ROS induce membrane permeabilization.
In summary, our studies provide evidence that sensing of dsRNA in the cytosol induces a signaling pathway via the sensors RigI and Mda5 and the common adaptor Mavs that activates the Nlrp3 inflammasome by inducting membrane permeabilization and K+ efflux. Thus, the cytosolic dsRNA pathway triggered by dsRNA converges into a common K+ lowering step that is essential for the induction of Nlrp3 activation by a large array of stimuli including ATP, pore-forming toxins, and particulate matter.
Supplementary Material
Acknowledgements
We thank the members of the Núñez laboratory for advice and G. Dubyak and R. Kaufman for generous gift of mutant mice
Abbreviations used in this paper
- BMDMs
bone marrow derived macrophages
- LDH
lactate dehydrogenase
- dsRNA
double-stranded RNA
- Mavs
mitochondrial antiviral signaling protein
- Mda5
melanoma differentiation-associated gene 5
- NLRs
nucleotide-binding oligomerization domain-like receptors
- Nlrp3
NLR pyrin domain-containing 3
- poly (I:C)
polyinosinic:polycytidylicacid
- RLRs
RIG-like receptors
- ROS
reactive oxygen species
- TLRs
Toll-like receptors
- WT
wild-type
Footnotes
This work was supported by grants R01AI063331 and R01DK091191 from the National Institutes of Health. L. F. was supported by a Research Career Development Award from the Crohn’s and Colitis Foundation of America.
References
- 1.Chen GY, Nunez G. Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol. 2010;10:826–837. doi: 10.1038/nri2873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nature immunology. 2010;11:373–384. doi: 10.1038/ni.1863. [DOI] [PubMed] [Google Scholar]
- 3.Inohara N, Nunez G. NODs: intracellular proteins involved in inflammation and apoptosis. Nat Rev Immunol. 2003;3:371–382. doi: 10.1038/nri1086. [DOI] [PubMed] [Google Scholar]
- 4.Martinon F, Mayor A, Tschopp J. The inflammasomes: guardians of the body. Annual review of immunology. 2009;27:229–265. doi: 10.1146/annurev.immunol.021908.132715. [DOI] [PubMed] [Google Scholar]
- 5.Franchi L, Munoz-Planillo R, Nunez G. Sensing and reacting to microbes through the inflammasomes. Nature immunology. 2012;13:325–332. doi: 10.1038/ni.2231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Franchi L, Eigenbrod T, Munoz-Planillo R, Nunez G. The inflammasome: a caspase-1-activation platform that regulates immune responses and disease pathogenesis. Nature immunology. 2009;10:241–247. doi: 10.1038/ni.1703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Masters SL, Simon A, Aksentijevich I, Kastner DL. Horror autoinflammaticus: the molecular pathophysiology of autoinflammatory disease (*) Annual review of immunology. 2009;27:621–668. doi: 10.1146/annurev.immunol.25.022106.141627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ozkurede VU, Franchi L. Immunology in clinic review series; focus on autoinflammatory diseases: role of inflammasomes in autoinflammatory syndromes. Clinical and experimental immunology. 2012;167:382–390. doi: 10.1111/j.1365-2249.2011.04535.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hao LY, Liu X, Franchi L. Inflammasomes in inflammatory bowel disease pathogenesis. Curr Opin Gastroenterol. 2013;29:363–369. doi: 10.1097/MOG.0b013e32836157a4. [DOI] [PubMed] [Google Scholar]
- 10.Bauernfeind FG, Horvath G, Stutz A, Alnemri ES, MacDonald K, Speert D, Fernandes-Alnemri T, Wu J, Monks BG, Fitzgerald KA, Hornung V, Latz E. Cutting edge: NF-kappaB activating pattern recognition and cytokine receptors license NLRP3 inflammasome activation by regulating NLRP3 expression. Journal of immunology. 2009;183:787–791. doi: 10.4049/jimmunol.0901363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Franchi L, Eigenbrod T, Nunez G. Cutting edge: TNF-{alpha} Mediates Sensitization to ATP and Silica via the NLRP3 Inflammasome in the Absence of Microbial Stimulation. Journal of immunology. 2009 doi: 10.4049/jimmunol.0900173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bauernfeind F, Bartok E, Rieger A, Franchi L, Nunez G, Hornung V. Cutting Edge: Reactive Oxygen Species Inhibitors Block Priming, but Not Activation, of the NLRP3 Inflammasome. Journal of immunology. 2011;187:613–617. doi: 10.4049/jimmunol.1100613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wen H, Miao EA, Ting JP. Mechanisms of NOD-like receptor-associated inflammasome activation. Immunity. 2013;39:432–441. doi: 10.1016/j.immuni.2013.08.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Latz E, Xiao TS, Stutz A. Activation and regulation of the inflammasomes. Nat Rev Immunol. 2013;13:397–411. doi: 10.1038/nri3452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dostert C, Petrilli V, Van Bruggen R, Steele C, Mossman BT, Tschopp J. Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science. 2008;320:674–677. doi: 10.1126/science.1156995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhou R, Yazdi AS, Menu P, Tschopp J. A role for mitochondria in NLRP3 inflammasome activation. Nature. 2011;469:221–225. doi: 10.1038/nature09663. [DOI] [PubMed] [Google Scholar]
- 17.Hornung V, Bauernfeind F, Halle A, Samstad EO, Kono H, Rock KL, Fitzgerald KA, Latz E. Silica crystals and aluminum salts activate the NALP3 inflammasome through phagosomal destabilization. Nature immunology. 2008;9:847–856. doi: 10.1038/ni.1631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Compan V, Baroja-Mazo A, Lopez-Castejon G, Gomez AI, Martinez CM, Angosto D, Montero MT, Herranz AS, Bazan E, Reimers D, Mulero V, Pelegrin P. Cell volume regulation modulates NLRP3 inflammasome activation. Immunity. 2012;37:487–500. doi: 10.1016/j.immuni.2012.06.013. [DOI] [PubMed] [Google Scholar]
- 19.Kahlenberg JM, Lundberg KC, Kertesy SB, Qu Y, Dubyak GR. Potentiation of caspase-1 activation by the P2X7 receptor is dependent on TLR signals and requires NF-kappaB-driven protein synthesis. Journal of immunology. 2005;175:7611–7622. doi: 10.4049/jimmunol.175.11.7611. [DOI] [PubMed] [Google Scholar]
- 20.Perregaux D, Gabel CA. Interleukin-1 beta maturation and release in response to ATP and nigericin. Evidence that potassium depletion mediated by these agents is a necessary and common feature of their activity. The Journal of biological chemistry. 1994;269:15195–15203. [PubMed] [Google Scholar]
- 21.Shimada K, Crother TR, Karlin J, Dagvadorj J, Chiba N, Chen S, Ramanujan VK, Wolf AJ, Vergnes L, Ojcius DM, Rentsendorj A, Vargas M, Guerrero C, Wang Y, Fitzgerald KA, Underhill DM, Town T, Arditi M. Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity. 2012;36:401–414. doi: 10.1016/j.immuni.2012.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Iyer SS, He Q, Janczy JR, Elliott EI, Zhong Z, Olivier AK, Sadler JJ, Knepper-Adrian V, Han R, Qiao L, Eisenbarth SC, Nauseef WM, Cassel SL, Sutterwala FS. Mitochondrial cardiolipin is required for Nlrp3 inflammasome activation. Immunity. 2013;39:311–323. doi: 10.1016/j.immuni.2013.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Franchi L, Kanneganti TD, Dubyak GR, Nunez G. Differential requirement of P2X7 receptor and intracellular K+ for caspase-1 activation induced by intracellular and extracellular bacteria. The Journal of biological chemistry. 2007;282:18810–18818. doi: 10.1074/jbc.M610762200. [DOI] [PubMed] [Google Scholar]
- 24.Munoz-Planillo R, Kuffa P, Martinez-Colon G, Smith BL, Rajendiran TM, Nunez G. K(+) efflux is the common trigger of NLRP3 inflammasome activation by bacterial toxins and particulate matter. Immunity. 2013;38:1142–1153. doi: 10.1016/j.immuni.2013.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Petrilli V, Papin S, Dostert C, Mayor A, Martinon F, Tschopp J. Activation of the NALP3 inflammasome is triggered by low intracellular potassium concentration. Cell death and differentiation. 2007 doi: 10.1038/sj.cdd.4402195. [DOI] [PubMed] [Google Scholar]
- 26.Loo YM, Gale M., Jr. Immune signaling by RIG-I-like receptors. Immunity. 2011;34:680–692. doi: 10.1016/j.immuni.2011.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yoneyama M, Fujita T. RNA recognition and signal transduction by RIG-I-like receptors. Immunological reviews. 2009;227:54–65. doi: 10.1111/j.1600-065X.2008.00727.x. [DOI] [PubMed] [Google Scholar]
- 28.Kawai T, Takahashi K, Sato S, Coban C, Kumar H, Kato H, Ishii KJ, Takeuchi O, Akira S. IPS-1, an adaptor triggering RIG-I- and Mda5-mediated type I interferon induction. Nature immunology. 2005;6:981–988. doi: 10.1038/ni1243. [DOI] [PubMed] [Google Scholar]
- 29.Rajan JV, Rodriguez D, Miao EA, Aderem A. The NLRP3 inflammasome detects encephalomyocarditis virus and vesicular stomatitis virus infection. J Virol. 2011;85:4167–4172. doi: 10.1128/JVI.01687-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Delaloye J, Roger T, Steiner-Tardivel QG, Le Roy D, Knaup Reymond M, Akira S, Petrilli V, Gomez CE, Perdiguero B, Tschopp J, Pantaleo G, Esteban M, Calandra T. Innate immune sensing of modified vaccinia virus Ankara (MVA) is mediated by TLR2-TLR6, MDA-5 and the NALP3 inflammasome. PLoS Pathog. 2009;5:e1000480. doi: 10.1371/journal.ppat.1000480. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 31.Pothlichet J, Meunier I, Davis BK, Ting JP, Skamene E, von Messling V, Vidal SM. Type I IFN triggers RIG-I/TLR3/NLRP3-dependent inflammasome activation in influenza A virus infected cells. PLoS Pathog. 2013;9:e1003256. doi: 10.1371/journal.ppat.1003256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Park S, Juliana C, Hong S, Datta P, Hwang I, Fernandes-Alnemri T, Yu JW, Alnemri ES. The mitochondrial antiviral protein MAVS associates with NLRP3 and regulates its inflammasome activity. Journal of immunology. 2013;191:4358–4366. doi: 10.4049/jimmunol.1301170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rathinam VA, Vanaja SK, Waggoner L, Sokolovska A, Becker C, Stuart LM, Leong JM, Fitzgerald KA. TRIF licenses caspase-11-dependent NLRP3 inflammasome activation by gram-negative bacteria. Cell. 2012;150:606–619. doi: 10.1016/j.cell.2012.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Subramanian N, Natarajan K, Clatworthy MR, Wang Z, Germain RN. The adaptor MAVS promotes NLRP3 mitochondrial localization and inflammasome activation. Cell. 2013;153:348–361. doi: 10.1016/j.cell.2013.02.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mitoma H, Hanabuchi S, Kim T, Bao M, Zhang Z, Sugimoto N, Liu YJ. The DHX33 RNA helicase senses cytosolic RNA and activates the NLRP3 inflammasome. Immunity. 2013;39:123–135. doi: 10.1016/j.immuni.2013.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kanneganti TD, Ozoren N, Body-Malapel M, Amer A, Park JH, Franchi L, Whitfield J, Barchet W, Colonna M, Vandenabeele P, Bertin J, Coyle A, Grant EP, Akira S, Nunez G. Bacterial RNA and small antiviral compounds activate caspase-1 through cryopyrin/Nalp3. Nature. 2006;440:233–236. doi: 10.1038/nature04517. [DOI] [PubMed] [Google Scholar]
- 37.Ozoren N, Masumoto J, Franchi L, Kanneganti TD, Body-Malapel M, Erturk I, Jagirdar R, Zhu L, Inohara N, Bertin J, Coyle A, Grant EP, Nunez G. Distinct roles of TLR2 and the adaptor ASC in IL-1beta/IL-18 secretion in response to Listeria monocytogenes. Journal of immunology. 2006;176:4337–4342. doi: 10.4049/jimmunol.176.7.4337. [DOI] [PubMed] [Google Scholar]
- 38.Kumar H, Koyama S, Ishii KJ, Kawai T, Akira S. Cutting edge: cooperation of IPS-1- and TRIF-dependent pathways in poly IC-enhanced antibody production and cytotoxic T cell responses. Journal of immunology. 2008;180:683–687. doi: 10.4049/jimmunol.180.2.683. [DOI] [PubMed] [Google Scholar]
- 39.Kato H, Sato S, Yoneyama M, Yamamoto M, Uematsu S, Matsui K, Tsujimura T, Takeda K, Fujita T, Takeuchi O, Akira S. Cell type-specific involvement of RIG-I in antiviral response. Immunity. 2005;23:19–28. doi: 10.1016/j.immuni.2005.04.010. [DOI] [PubMed] [Google Scholar]
- 40.Gitlin L, Barchet W, Gilfillan S, Cella M, Beutler B, Flavell RA, Diamond MS, Colonna M. Essential role of mda-5 in type I IFN responses to polyriboinosinic:polyribocytidylic acid and encephalomyocarditis picornavirus. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:8459–8464. doi: 10.1073/pnas.0603082103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Errett JS, Suthar MS, McMillan A, Diamond MS, Gale M., Jr. The essential, nonredundant roles of RIG-I and MDA5 in detecting and controlling West Nile virus infection. J Virol. 2013;87:11416–11425. doi: 10.1128/JVI.01488-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Frezza C, Cipolat S, Scorrano L. Organelle isolation: functional mitochondria from mouse liver, muscle and cultured fibroblasts. Nature protocols. 2007;2:287–295. doi: 10.1038/nprot.2006.478. [DOI] [PubMed] [Google Scholar]
- 43.Franchi L, Kamada N, Nakamura Y, Burberry A, Kuffa P, Suzuki S, Shaw MH, Kim YG, Nunez G. NLRC4-driven production of IL-1beta discriminates between pathogenic and commensal bacteria and promotes host intestinal defense. Nature immunology. 2012;13:449–456. doi: 10.1038/ni.2263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kanneganti TD, Body-Malapel M, Amer A, Park JH, Whitfield J, Franchi L, Taraporewala ZF, Miller D, Patton JT, Inohara N, Nunez G. Critical role for Cryopyrin/Nalp3 in activation of caspase-1 in response to viral infection and double-stranded RNA. The Journal of biological chemistry. 2006;281:36560–36568. doi: 10.1074/jbc.M607594200. [DOI] [PubMed] [Google Scholar]
- 45.Allen IC, Scull MA, Moore CB, Holl EK, McElvania-TeKippe E, Taxman DJ, Guthrie EH, Pickles RJ, Ting JP. The NLRP3 inflammasome mediates in vivo innate immunity to influenza A virus through recognition of viral RNA. Immunity. 2009;30:556–565. doi: 10.1016/j.immuni.2009.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Franchi L, Amer A, Body-Malapel M, Kanneganti TD, Ozoren N, Jagirdar R, Inohara N, Vandenabeele P, Bertin J, Coyle A, Grant EP, Nunez G. Cytosolic flagellin requires Ipaf for activation of caspase-1 and interleukin 1beta in salmonella-infected macrophages. Nature immunology. 2006;7:576–582. doi: 10.1038/ni1346. [DOI] [PubMed] [Google Scholar]
- 47.Miao EA, Alpuche-Aranda CM, Dors M, Clark AE, Bader MW, Miller SI, Aderem A. Cytoplasmic flagellin activates caspase-1 and secretion of interleukin 1beta via Ipaf. Nature immunology. 2006;7:569–575. doi: 10.1038/ni1344. [DOI] [PubMed] [Google Scholar]
- 48.Schattgen SA, Fitzgerald KA. The PYHIN protein family as mediators of host defenses. Immunological reviews. 2011;243:109–118. doi: 10.1111/j.1600-065X.2011.01053.x. [DOI] [PubMed] [Google Scholar]
- 49.Kawai T, Akira S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity. 2011;34:637–650. doi: 10.1016/j.immuni.2011.05.006. [DOI] [PubMed] [Google Scholar]
- 50.Sander LE, Davis MJ, Boekschoten MV, Amsen D, Dascher CC, Ryffel B, Swanson JA, Muller M, Blander JM. Detection of prokaryotic mRNA signifies microbial viability and promotes immunity. Nature. 2011;474:385–389. doi: 10.1038/nature10072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fink SL, Cookson BT. Apoptosis, pyroptosis, and necrosis: mechanistic description of dead and dying eukaryotic cells. Infect Immun. 2005;73:1907–1916. doi: 10.1128/IAI.73.4.1907-1916.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kaczmarek A, Vandenabeele P, Krysko DV. Necroptosis: the release of damage-associated molecular patterns and its physiological relevance. Immunity. 2013;38:209–223. doi: 10.1016/j.immuni.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 53.Vandenabeele P, Declercq W, Vanden Berghe T. Necrotic cell death and ‘necrostatins’: now we can control cellular explosion. Trends Biochem Sci. 2008;33:352–355. doi: 10.1016/j.tibs.2008.05.007. [DOI] [PubMed] [Google Scholar]
- 54.Demidchik V, Shabala SN, Coutts KB, Tester MA, Davies JM. Free oxygen radicals regulate plasma membrane Ca2+- and K+-permeable channels in plant root cells. J Cell Sci. 2003;116:81–88. doi: 10.1242/jcs.00201. [DOI] [PubMed] [Google Scholar]
- 55.van der Zee J, Dubbelman TM, van Steveninck J. Peroxide-induced membrane damage in human erythrocytes. Biochim Biophys Acta. 1985;818:38–44. doi: 10.1016/0005-2736(85)90135-x. [DOI] [PubMed] [Google Scholar]
- 56.Allam R, Lawlor KE, Yu EC, Mildenhall AL, Moujalled DM, Lewis RS, Ke F, Mason KD, White MJ, Stacey KJ, Strasser A, O’Reilly LA, Alexander W, Kile BT, Vaux DL, Vince JE. Mitochondrial apoptosis is dispensable for NLRP3 inflammasome activation but non-apoptotic caspase-8 is required for inflammasome priming. EMBO Rep. 2014 doi: 10.15252/embr.201438463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zou J, Kawai T, Tsuchida T, Kozaki T, Tanaka H, Shin KS, Kumar H, Akira S. Poly IC triggers a cathepsin D- and IPS-1-dependent pathway to enhance cytokine production and mediate dendritic cell necroptosis. Immunity. 2013;38:717–728. doi: 10.1016/j.immuni.2012.12.007. [DOI] [PubMed] [Google Scholar]
- 58.Schlee M, Roth A, Hornung V, Hagmann CA, Wimmenauer V, Barchet W, Coch C, Janke M, Mihailovic A, Wardle G, Juranek S, Kato H, Kawai T, Poeck H, Fitzgerald KA, Takeuchi O, Akira S, Tuschl T, Latz E, Ludwig J, Hartmann G. Recognition of 5′ triphosphate by RIGI helicase requires short blunt double-stranded RNA as contained in panhandle of negative-strand virus. Immunity. 2009;31:25–34. doi: 10.1016/j.immuni.2009.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kato H, Takahasi K, Fujita T. RIG-I-like receptors: cytoplasmic sensors for non-self RNA. Immunological reviews. 2011;243:91–98. doi: 10.1111/j.1600-065X.2011.01052.x. [DOI] [PubMed] [Google Scholar]
- 60.Li H, Ambade A, Re F. Cutting edge: Necrosis activates the NLRP3 inflammasome. Journal of immunology. 2009;183:1528–1532. doi: 10.4049/jimmunol.0901080. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.