ABSTRACT
Purpose
To determine normative reference ranges for higher-order wavefront error (HO-WFE), compare these values with those in common ocular pathologies, and evaluate treatments.
Methods
A review of 17 major studies on HO-WFE was made, involving data for a total of 31,605 subjects. The upper limit of the 95% confidence interval (CI) for HO-WFE was calculated from the most comprehensive of these studies using normal healthy patients aged 20 to 80 years. There were no studies identified using the natural pupil size for subjects, and for this reason, the HO-WFE was tabulated for pupil diameters of 3 to 7 mm. Effects of keratoconus, pterygium, cataract, and dry eye on HO-WFE were reviewed and treatment efficacy was considered.
Results
The calculated upper limit of the 95% CI for HO-WFE in a healthy normal 35-year-old patient with a mesopic pupil diameter of 6 mm would be 0.471 μm (471 nm) root-mean-square or less. Although the normal HO-WFE increases with age for a given pupil size, it is not yet completely clear how the concurrent influence of age-related pupillary miosis affects these findings. Abnormal ocular conditions such as keratoconus can induce a large HO-WFE, often in excess of 3.0 μm, particularly attributed to coma. For pterygium or cortical cataract, a combination of coma and trefoil was more commonly induced. Nuclear cataract can induce a negative spherical HO-WFE, usually in excess of 1.0 μm.
Conclusions
The upper limit of the 95% CI for HO-WFE root-mean-square is about 0.5 μm with normal physiological pupil sizes. With ocular pathologies, HO-WFE can be in excess of 1.0 μm, although many devices and therapeutic and surgical treatments are reported to be highly effective at minimizing HO-WFE. More accurate normative reference ranges for HO-WFE will require future studies using the subjects’ natural pupil size.
Key Words: wavefront refraction, cataract, keratoconus, higher-order aberrations of the eye, normal populations
Dr. Irvin Borish and his classic textbook Clinical Refraction helped teach many of us the art of refraction. He also taught us in his later years the use of new and evolving science and technologies to make clinical refraction more accurate and more efficient.1 The first significant implementation of wavefront science in vision care emerged in wavefront-guided corneal refractive laser surgery in the late 1990s.2 In the past 10 years, the increasing use of wavefront refraction has guided us toward a more accurate and efficient clinical refraction that Dr. Borish envisioned.
Wavefront refraction has been recognized as an optical science by Scheiner’s and Newton’s observations of aberrated light in the 17th and 18th centuries.3 The modern wavefront aberrometer applies a form of high-resolution autorefraction across the entire area of the patient’s pupil, giving the wavefront error (WFE) in terms of micrometers of deviation (root-mean-square [RMS]) from the ideal wavefront plane.4 The irregular portion of the WFE is termed higher-order wavefront error (HO-WFE), that is, not correctable with traditional sphero-cylindrical spectacles.
Visual acuity remains the most common method by which eye care professionals assess patients’ optical image quality. However, this technique does little to assess retinal image quality (blur, halos, poor edge discrimination, glare, and reduced contrast sensitivity) related to HO-WFE.5 Eye care professionals and researchers have described quality of vision issues in refraction for many years.6,7
In both the refraction and correction of lower-order WFE and HO-WFE, the pupil size is one of the most critical considerations.8,9 In wavefront refraction, the pupil size is an integral part of the specification of the WFE.10 Changes in the pupil size may cause subjective differences in the clinical refraction, that is, “day versus night” considerations. Mild pharmacological miosis may assist with subjective night vision symptoms like halos, glare, and reduced contrast sensitivity from aggregate spherical aberrations (SAs).10
New optical wavefront technologies have also been applied into visual correction. Such applications include, but are not limited to, optical devices such as “HO-WFE correcting” spectacles, contact lenses, intraocular lenses (IOLs), and diagnostic instruments and are applied in custom wavefront corneal laser surgery. These corrective applications have experienced mixed success owing to an array of dynamic pupillary, optical, physiological, immunological, and environmental variables and challenges, which are all difficult to control.11
This article reviews the major studies of HO-WFE in normal healthy patients and compiles normative data for a wide range of ages and pupil diameters. Also considered are possible future advances in the normal reference ranges for HO-WFE. The effects of common ocular pathologies on the HO-WFE are reviewed, and corrective procedures and devices are considered.
NORMAL HO-WFE
Many studies have been conducted to measure HO-WFE in normal healthy eyes; however, even in 2014, new studies continue to be published, in part because of the perception that there is a “paucity of data on the normal reference ranges.”12 Studies with different populations and methodologies are summarized in Table 1.4,13–28 The studies are tabulated by age group and ordered by pupil diameter, because these two factors account for most of the variance in HO-WFE in normal healthy eyes.14
TABLE 1.
Wavefront refraction: Larger population studies, ordered by pupil size

Studies with a 4- or 5-mm optical zone found the average HO-WFE to be less than 0.30 μm, often lower in the younger age groups (Table 1). There was more variability in the average HO-WFE for studies using a 6-mm optical zone, with the average HO-WFE being up to 0.518 μm. There were no studies that used the natural pupil size for subjects, not even the studies that concern age, where pupil size is known to be a variable.
It is difficult to define the normal HO-WFE in a population without studies that use the subjects’ natural pupil size. However, the normal HO-WFE can be estimated by using separate studies of the natural pupil size with age. Winn et al.29 reported that average pupil size decreases linearly as a function of age, for all luminance levels. On this basis, Applegate et al.14 estimated that the average HO-WFE at low-luminance levels would slightly reduce with age, whereas at high luminance levels, it may slightly increase with age.
The study by Applegate et al.14 gives the most comprehensive HO-WFE data set for subjects aged 20 to 80 years. Data sets such as this enable the upper limit of the 95% confidence interval (CI) for a normal population to be calculated.30–32 Table 2 shows the upper limit of the 95% CI for HO-WFE, as a function of age and pupil size, calculated from the study by Applegate et al.14 For example, an average 35-year-old patient would be expected to have a mesopic pupil size of about 6 mm33; the expected HO-WFE for such a patient would be 0.471 μm (471 nm) or less (Table 2). A measurement greater than this level (for this age and pupil diameter) would be suspicious for abnormality, at the 95% level.
TABLE 2.
What is normal? The upper limit of the 95% CI for WFE as a function of age and pupil size

Table 2 also presents the upper limit of the 95% CI of the Applegate et al.14 data, for several other HO-WFE individual terms—coma, spherical, and trefoil. These terms describe distinctive wavefront shapes. Although the terms are less widely used than, for example, astigmatism, the effect on vision for each may be no less profound.
Refracting clinicians are familiar with the clinical experience that some patients can detect blur before acuity is lost,34 for example, where the patient may say: “I can read it, but it’s not clear.” Patients will be able to identify their HO-WFE at an RMS level of 0.43 μm or greater,35 and experienced observers under laboratory conditions may detect HO-WFE blur at even lower levels.34 The linear equation, Me (defocus) = 4π √3 RMS error/area of pupil, may be used to convert RMS values to dioptric power.9 Therefore, an RMS value of greater than or equal to 0.43 μm is equivalent, for a 5-mm pupil aperture, to about greater than or equal to 0.50 diopters. A qualitative illustration of what the patient sees is the point spread function, which can reveal unique refractive information.9,36
The cortical interpretation of human vision means that WFE measurements require confirmation through the “gold standard” of subjective refraction.36,37 However, the RMS data provide insight into the classic refractive question: “Which is better, one or two, or no difference?,” or simply “Which is better?”3 The “just noticeable difference” (jnd) for experienced subjects may be as small as 0.04 μm of RMS aberration.38 In terms of visual acuity, 0.25 μm of RMS defocus error over a 6-mm pupil results in a loss of an average of eight letters (1.6 lines).39
In summary, normative data such as those presented in Table 2 may be applied to help in the diagnosis of abnormal conditions. The next advance in normal reference ranges for HO-WFE may use the subjects’ natural physiological pupil diameter, because the pupil diameter is an integral part of the WFE.
WAVEFRONT REFRACTION IN COMMON CLINICAL CONDITIONS
In clinical vision care, refraction is directly affected by numerous conditions of the eyelids, ocular surface, tear film, cornea, crystalline lens, and beyond. These effects on vision produce varying degrees of lower-order WFE and HO-WFE. Irrespective of their etiology, the wavefront refraction can provide an accurate means of measuring the effects of such conditions. Some of the more common diseases and their effects include, but are not limited to, the following examples.
Keratoconus
Keratoconus can have a devastating effect on vision. The impact is so large because keratoconus has a central effect on the corneal front surface, the most important refractive element of the eye. No condition probably has the potential to cause greater HO-WFE, that is, irregular astigmatism not correctable with spectacles. Indeed, severe keratoconus can cause extreme WFEs, beyond the range of measurement for standard instruments.40,41
Characteristically, keratoconus causes a significant coma WFE, owing to the cone being displaced from the center of the cornea (Fig. 1). Jafri et al.42 found for early keratoconus that the mean coma was 2.034 μm RMS (±1.532 SD), with a 6.5-mm pupil. Gobbe and Guillon43 assessed 45 patients with keratoconus, ranging from early keratoconus to very advanced cases, and measured total HO-WFE for a 6-mm pupil to be 3.744 μm (±3.086). Pantanelli et al.41 concluded that coma accounted for 62 ± 30% of the higher-order variance in their sample of 19 patients with keratoconus. A high correlation has been found between the abnormal corneal curvature and the total WFE and hence the HO-WFE can be useful in diagnosis.40,42
FIGURE 1.

Keratoconus in the right eye of a 20-year-old man. (A) Corneal topography map showing an apical power of 65 diopters. (B) HO-WFE map for a 6-mm pupil, with total higher order, 2.928 μm; total coma, 2.087 μm; total spherical, 0.870 μm; and total trefoil, 1.744 μm. The HO-WFE is significantly abnormal in relation to the keratoconus.
Wavefront Correction
Higher-order wavefront error in keratoconus is correctable, giving the patient good vision, as has been demonstrated using adaptive optics.44 Gas permeable corneal lenses and scleral contact lenses typically correct visual acuity in keratoconus more effectively than soft lenses or spectacles.31 However, even with gas permeable lenses, there may be a residual uncorrected HO-WFE, in part owing to irregularity in the posterior corneal surface.31,45
Wavefront-corrected contact lenses for keratoconus have been described, in custom hydrogel or rigid scleral lens designs. The visual acuity is typically improved by 1 to 2 lines of acuity over the standard contact lenses, reaching 20/20 to 20/30.30,46 Because static rotation or lateral displacements represent a significant limitation to the efficacy of a wavefront contact lens correction,47 it seems likely that further advances in contact lens correction for keratoconus will be made with the most stable fitting lenses in modern lens materials, which are scleral designs in either a rigid or a silicone-hydrogel material.
Corneal surgery for keratoconus, such as penetrating keratoplasty or intracorneal ring segments, usually improves the unaided visual acuity and reduces the regular refractive error (sphere and astigmatism), but the procedures often still result in a significant HO-WFE.41,48 Deep anterior lamellar keratoplasty is also associated with a significant postoperative HO-WFE,49 although it does have the advantage of improved endothelial integrity. Collagen cross-linking is a treatment for progression in keratoconus, which may also slightly reduce HO-WFE, although the visual acuity may not improve.50,51
Pterygium
A pterygium can cause an asymmetric distortion of the cornea, because the lesion typically extends radially from the corneal periphery (Fig. 2). The disruption to the corneal surface topography induces both coma and trefoil WFEs.52,53 There is commonly an increase in corneal and refractive astigmatism with pterygium, as well as an increase in HO-WFE, with an effect proportional to the extent of the lesion.54,55 Because the condition approaches lateral to the visual axis, a change in SA is not usually a feature of the condition.56 Higher-order wavefront error is associated with corresponding reductions in visual acuity and contrast sensitivity.52
FIGURE 2.
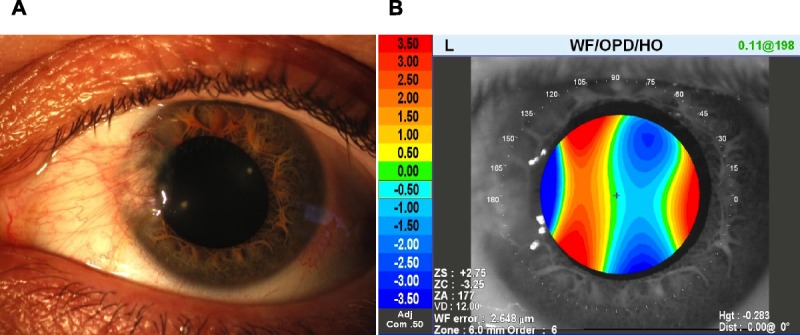
Pterygium in the left eye of a 42-year-old woman. (A) Anterior eye image. (B) HO-WFE map for a 6-mm pupil, with total higher order, 2.648 μm; total coma, 1.202 μm; total spherical, 0.223 μm; and total trefoil, 2.149 μm. The HO-WFE is significantly abnormal owing to the pterygium.
Zare et al.56 specified the pterygium in terms of the uninvolved optical zone of the cornea, measured from the slit lamp view, contending that this approach removed the influence of corneal size from the visual impact of the pterygia. Despite measuring only within a 5-mm-diameter optical zone, they found that the patient group with an uninvolved optical zone of 7 to 9 mm was associated with a total HO-WFE averaging 2.58 μm, and for patients grouped into the uninvolved optical zone of 5 to 7 mm, the total HO-WFE averaged 3.35 μm.
Wavefront Correction
Kwon et al.55 found a more effective reduction in WFE and astigmatism if there was surgical intervention in eyes with pterygia sized less than 3.0 mm. Although surgical excision of larger pterygia greater than 3 mm provided a significant benefit, reducing the total mean higher-order error with a 6.5-mm pupil from 3.73 μm preoperatively to 1.92 μm at 3 months postop did not result in the irregular WFE fully returning to the reference range. Surgical intervention in eyes with pterygia sized less than 3.0 mm reduced the total mean HO-WFE with a 6.5-mm pupil from 2.21 μm preoperatively to 1.18 μm at 3 months postop. Similarly, Gumus et al.57 showed that pterygium excision with conjunctival autograft plus corneal polishing significantly reduced most of the pterygium-induced HO-WFE, with improvements continuing into the 1-year postoperative period.
Cataract
Initial reports of the wavefront analysis of cataract were provided by Kuroda et al.58 in 2002. Nuclear sclerosis was associated with negative SA of −1.274 ± 0.733 μm for a 6-mm pupil in four patients.58 This finding was significant because the SA in normal healthy eyes is usually positive in sign.19,28 The negative SA appears because of the increased refractive index in the nuclear cataract, causing an increased refractive power in central lens compared with the periphery (Fig. 3).58 Other studies have since confirmed increased negative SA in nuclear cataract.5
FIGURE 3.
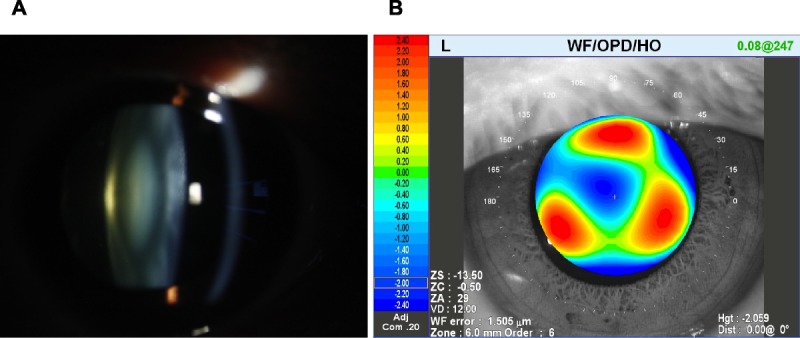
Moderate nuclear sclerosis cataract in the left eye of a 45-year-old woman. The patient had experienced a −8.00-diopter myopic shift in 2 years, with best-corrected acuity of 20/70 (pinhole 20/40). (A) Anterior eye image. (B) HO-WFE map for a 6-mm pupil, with total higher order, 1.505 μm; total coma, 0.521 μm; total spherical, −0.893 μm; and total trefoil, 1.059 μm. The WFE is significantly abnormal owing to the cataract.
Clinically, the visual effect of nuclear cataract depends on the pupil size. If the pupil is small, such as for patients older than 70 years, then a nuclear cataract may manifest as a myopic shift because of the increased central refractive power.59 However, visible crystalline lens changes may commence as early as the age of 40 owing to factors including high ambient temperature or medications.60,61 Nuclear cataract in middle-aged patients, or those with a larger pupil, may cause monocular triplopia, attributed to a combination of SA and trefoil.62
Cortical cataract tends to not affect SA, but to increase other WFEs, particularly coma (Fig. 4).63 Rocha et al.5 found increased coma and trefoil associated with cortical cataract, compared with nuclear cataract or control patients. This is consistent with the observation that cortical cataract tends to cause changes in astigmatism, rather than a myopic shift.59 Monocular diplopia can be associated with cortical cataract or the water cleft subtype of cataract, which may be attributed to the increased trefoil WFE.64,65
FIGURE 4.

Anterior cortical cataract in the left eye of a 72-year-old female patient, with best-corrected acuity of 20/70. (A) Anterior eye image. (B) HO-WFE map for a 6-mm pupil, with total higher order, 1.572 μm; total coma, 0.099 μm; total spherical, 0.121 μm; and total trefoil, 1.528 μm. The WFE is significantly abnormal, mainly attributed to trefoil.
Posterior subcapsular cataract is not well characterized in terms of the wavefront effects, because it can be quite dense, causing missing data points in the central area of the scan and a lack of reproducibility.5 It may be that the visual effects of posterior subcapsular cataract are not so much refractive, but more related to light absorption and scatter.
Wavefront Correction
Cataract surgery is a frequently performed procedure in many countries, and numerous studies evaluate the IOL implants and surgical techniques. An aspheric IOL may reduce postoperative SA and improve contrast sensitivity under mesopic and scotopic light conditions.66,67 The amount of negative SA in an aspheric IOL is reported as either zero (Akreos AO, Bausch & Lomb, Rochester, NY), −0.20 μm (SN60WF, Alcon Labs, Fort Worth, TX), or −0.27 μm (Tecnis ZA9003, Advanced Medical Optics, Santa Ana, CA) for a 6-mm optical zone.66–68 However, an aspheric IOL requires more accurate positioning in the eye, in terms of displacement or tilt, to minimize the HO-WFE.67,69
The corneal incision associated with cataract surgery can also impact the final HO-WFE, inducing spherical, trefoil, or tetrafoil HO-WFE for a 5.0- or 6.0-mm optical zone, although the effect may not be significant for a 3.5-mm pupil size.70 Posterior capsular opacification of the IOL can increase the HO-WFE, particularly trefoil, but this can be remediated with Nd:YAG laser posterior capsulotomy.71
Multifocal IOLs can provide targeted amounts of negative SA to increase a patient’s depth of focus and assist the patient’s near vision.72 Conversely, Santhiago et al.67 contend that the positive SA associated with spherical and a zero SA IOL may be helpful in terms of maintaining depth of focus for the patient, although they suggest that further studies are needed using the patients’ natural physiological pupil size.
Dry Eye
When the tear film breaks up in the normal healthy eye, there can be a huge dynamic effect on HO-WFE compared with the relatively small effect of the normal cornea and crystalline lens. Thibos et al.73,74 were among the first to demonstrate that wavefront refraction was influenced by tear film stability. They also documented microscopic change in the tear film, with distortion of the individual dots in the Shack-Hartmann instrument matrix array.
The effect of tear film breakup on HO-WFE appears to be related to local thinning of the tear film, as well as refractive effects at the edges of the areas of tear film breakup and changes in the tear meniscus.4,74 Tear film instability causes not only “macroscopic” WFEs but also microscopic changes to the WFE, which Himebaugh et al.75 termed very high order aberrations or microaberrations.
Koh et al.76 showed an increase in HO-WFE associated with tear film breakup. They compared higher-order errors immediately after blinking and 15 seconds later in 20 normal subjects. Regardless of the pupil size, all higher-order errors including spherical and coma were significantly increased. Higher-order wavefront error was increased 44% in the central 4-mm zone after tear film breakup and increased 23% in the central 6-mm zone.
More rapid changes in HO-WFE were demonstrated for dry eye patients by Montés-Micó et al.77 They measured soon after tear film breakup, 5 to 10 seconds after the blink. Higher-order wavefront error was increased 158% in the central 4-mm zone after tear film breakup and increased 129% in the central 6-mm zone. Again, the results were relatively independent of pupil size and both spherical and coma errors were affected. The authors used the term tear film irregularity to describe the effect of dry eye on higher-order aberrations. Similar results were demonstrated by Denoyer et al.78 who used serial wavefront measures in the first 10 seconds after the blink in mild-moderate dry eye patients. Higher-order wavefront error increased from 0.095 ± 0.015 μm immediately after blinking to 0.23 ± 0.06 μm within 10 seconds.
Wavefront Correction
Improvement in tear film regularity with lubricant treatment was demonstrated by Montés-Micó et al.79 They instilled 0.18% sodium hyaluronate into the eyes of 15 patients with mild-moderate dry eye. The initial total HO-WFE of 0.81 ± 0.1 μm was improved to 0.40 ± 0.12 μm at 10 minutes. More viscous eyedrops, such as 0.3% sodium hyaluronate or 0.5% carboxymethylcellulose sodium/0.9% glycerin may cause an initial brief increase in HO-WFE at instillation,80,81 although with prolonged use, this effect may diminish.
A further issue in dry eye is the effect of superficial punctate keratopathy, which exacerbates HO-WFE.82 Lekhanont et al.83 assessed the effect of 0.18% sodium hyaluronate in a group of 50 moderate to severe dry eye patients. There was a significant improvement in patient symptoms with the test drop for the 2-hour duration of the study, but the HO-WFE of 1.78 ± 0.54 μm at baseline was not changed by the treatment, an effect attributed in part to the presence of punctate epithelial keratopathy.
CONCLUSIONS
The wavefront refraction combines the critical refractive metrics of lower-order refraction, HO-WFE, pupillometry, point spread function, jnd, and beyond. The technique is designed to improve the accuracy and efficiency of refraction in clinical practice. Population studies of wavefront refraction for normal healthy eyes have shown that the upper limit of the 95% CI for HO-WFE RMS is about 0.5 μm with normal physiological pupil sizes. Other clinical studies have established that the threshold of awareness for a HO-WFE error is also about 0.5 μm, providing some concordance and giving an important baseline with which to evaluate clinical disorders. More accurate normal reference ranges for HO-WFE may require studies using the subjects’ natural physiological pupil sizes across the age groups. Wavefront refractive studies have shown a HO-WFE in excess of 1.0 μm for a range of abnormal and irregular conditions, including keratoconus, pterygium, cataract, and dry eye. Wavefront corrections are increasingly being used with contact lenses, therapeutic and surgical treatments, retinal imaging, and other methodologies. Dr. Borish would be pleased with the advances in refraction. Today’s eye care practitioners should be as well.
Adrian S. Bruce
Australian College of Optometry
Corner of Keppel and Cardigan Sts
Carlton, Victoria 3053
Australia
e-mail: abruce@aco.org.au
ACKNOWLEDGMENTS
This review of wavefront refraction is dedicated to the memory of Dr. Irvin Borish who taught so many the art of clinical refraction. Dr. Bruce thanks his colleagues, mentors, and students at the Australian College of Optometry and the Department of Optometry and Vision Sciences, University of Melbourne, for their support, teaching, and enthusiasm. Dr. Catania thanks Larry Thibos and Ray Applegate for the inspiration they provided him over the years in his attempt to bring wavefront science and their excellent research to clinical eye care practitioners. Dr. Catania consults for Marco Ophthalmic and Johnson and Johnson Vision Care. The authors have no financial or proprietary interest in this work.
REFERENCES
- 1. Thibos LN, Himebaugh NL, Coe CD. Wavefront refraction. In: Benjamin WJ, Borish IM, eds. Borish’s Clinical Refraction, 2nd ed. Oxford, UK: Butterworth-Heinemann; 2006: 765– 89 [Google Scholar]
- 2. Krueger RR, Rabinowitz YS, Binder PS. The 25th anniversary of excimer lasers in refractive surgery: historical review. J Refract Surg 2010; 26: 749– 60 [DOI] [PubMed] [Google Scholar]
- 3. Thibos LN. The 2012 Charles Prentice medal lecture: wavefront measurement of refractive state. Optom Vis Sci 2013; 90: 911– 23 [DOI] [PubMed] [Google Scholar]
- 4. Thibos LN, Hong X, Bradley A, Cheng X. Statistical variation of aberration structure and image quality in a normal population of healthy eyes. J Opt Soc Am (A) 2002; 19: 2329– 48 [DOI] [PubMed] [Google Scholar]
- 5. Rocha KM, Nosé W, Bottós K, Bottós J, Morimoto L, Soriano E. Higher-order aberrations of age-related cataract. J Cataract Refract Surg 2007; 33: 1442– 6 [DOI] [PubMed] [Google Scholar]
- 6. Applegate RA, Marsack JD, Ramos R, Sarver EJ. Interaction between aberrations to improve or reduce visual performance. J Cataract Refract Surg 2003; 29: 1487– 95 [DOI] [PubMed] [Google Scholar]
- 7. Applegate RA, Ballentine C, Gross H, Sarver EJ, Sarver CA. Visual acuity as a function of Zernike mode and level of root mean square error. Optom Vis Sci 2003; 80: 97– 105 [DOI] [PubMed] [Google Scholar]
- 8. Charman N, Jennings JAM, Whitefoot H. The refraction of the eye in relation to spherical aberration and pupil size. Brit J Physiol Optics 1978; 32: 78– 93 [PubMed] [Google Scholar]
- 9. Cheng X, Bradley A, Ravikumar S, Thibos LN. Visual impact of Zernike and Seidel forms of monochromatic aberrations. Optom Vis Sci 2010; 87: 300– 12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Randazzo A, Nizzola F, Rossetti L, Orzalesi N, Vinciguerra P. Pharmacological management of night vision disturbances after refractive surgery. Results of a randomized clinical trial. J Cataract Refract Surg 2005; 31: 1764– 72 [DOI] [PubMed] [Google Scholar]
- 11. O’Brart DP. Excimer laser surface ablation: a review of recent literature. Clin Exp Optom 2014; 97: 12– 7 [DOI] [PubMed] [Google Scholar]
- 12. Yazar S, Hewitt AW, Forward H, McKnight CM, Tan A, Mountain JA, Mackey DA. Comparison of monochromatic aberrations in young adults with different visual acuity and refractive errors. J Cataract Refract Surg 2014; 40: 441– 9 [DOI] [PubMed] [Google Scholar]
- 13. Hartwig A, Atchison DA. Analysis of higher-order aberrations in a large clinical population. Invest Ophthalmol Vis Sci 2012; 53: 7862– 70 [DOI] [PubMed] [Google Scholar]
- 14. Applegate RA, Donnelly WJ, 3rd, Marsack JD, Koenig DE, Pesudovs K. Three-dimensional relationship between high-order root-mean-square wavefront error, pupil diameter, and aging. J Opt Soc Am (A) 2007; 24: 578– 87 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Atchison DA, Markwell EL. Aberrations of emmetropic subjects at different ages. Vision Res 2008; 48: 2224– 31 [DOI] [PubMed] [Google Scholar]
- 16. Brunette I, Bueno JM, Parent M, Hamam H, Simonet P. Monochromatic aberrations as a function of age, from childhood to advanced age. Invest Ophthalmol Vis Sci 2003; 44: 5438– 46 [DOI] [PubMed] [Google Scholar]
- 17. Salmon TO, van de Pol C. Normal-eye Zernike coefficients and root-mean-square wavefront errors. J Cataract Refract Surg 2006; 32: 2064– 74 [DOI] [PubMed] [Google Scholar]
- 18. Wan XH, Li SM, Xiong Y, Liang YB, Li J, Wang FH, Li J, Jhanji V, Wang NL. Ocular monochromatic aberrations in a rural Chinese adult population. Optom Vis Sci 2014; 91: 68– 75 [DOI] [PubMed] [Google Scholar]
- 19. Porter J, Guirao A, Cox IG, Williams DR. Monochromatic aberrations of the human eye in a large population. J Opt Soc Am (A) 2001; 18: 1793– 803 [DOI] [PubMed] [Google Scholar]
- 20. Lyall DAM, Srinivasan S, Gray LS. Changes in ocular monochromatic higher-order aberrations in the aging eye. Optom Vis Sci 2013; 90: 996– 1003 [DOI] [PubMed] [Google Scholar]
- 21. Athaide HV, Campos M, Costa C. Study of ocular aberrations with age. Arq Bras Oftalmol 2009; 72: 617– 21 [DOI] [PubMed] [Google Scholar]
- 22. Yamane N, Miyata K, Samejima T, Hiraoka T, Kiuchi T, Okamoto F, Hirohara Y, Mihashi T, Oshika T. Ocular higher-order aberrations and contrast sensitivity after conventional laser in situ keratomileusis. Invest Ophthalmol Vis Sci 2004; 45: 3986– 90 [DOI] [PubMed] [Google Scholar]
- 23. Philip K, Martinez A, Ho A, Conrad F, Ale J, Mitchell P, Sankaridurg P. Total ocular, anterior corneal and lenticular higher order aberrations in hyperopic, myopic and emmetropic eyes. Vision Res 2012; 52: 31– 7 [DOI] [PubMed] [Google Scholar]
- 24. Hashemian SJ, Soleimani M, Foroutan A, Joshaghani M, Ghaempanah MJ, Jafari ME, Yaseri M. Ocular higher-order aberrations and mesopic pupil size in individuals screened for refractive surgery. Int J Ophthalmol 2012; 5: 222– 5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kingston AC, Cox IG. Population spherical aberration: associations with ametropia, age, corneal curvature, and image quality. Clin Ophthalmol 2013; 7: 933– 8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Nakano EM, Bains H, Nakano K, Nakano C, Portellinha W, Oliveira M, Alvarenga L. Wavefront analysis in Asian-Brazilians. J Refract Surg 2006; 22: S1024– 6 [DOI] [PubMed] [Google Scholar]
- 27. Plainis S, Pallikaris IG. Ocular monochromatic aberration statistics in a large emmetropic population. J Mod Opt 2008; 55: 759– 72 [Google Scholar]
- 28. Wang L, Koch DD. Ocular higher-order aberrations in individuals screened for refractive surgery. J Cataract Refract Surg 2003; 29: 1896– 903 [DOI] [PubMed] [Google Scholar]
- 29. Winn B, Whitaker D, Elliott DB, Phillips NJ. Factors affecting light-adapted pupil size in normal human subjects. Invest Ophthalmol Vis Sci 1994; 35: 1132– 7 [PubMed] [Google Scholar]
- 30. Marsack JD, Parker KE, Niu Y, Pesudovs K, Applegate RA. On-eye performance of custom wavefront-guided soft contact lenses in a habitual soft lens-wearing keratoconic patient. J Refract Surg 2007; 23: 960– 4 [DOI] [PubMed] [Google Scholar]
- 31. Marsack JD, Parker KE, Pesudovs K, Donnelly WJ, 3rd, Applegate RA. Uncorrected wavefront error and visual performance during RGP wear in keratoconus. Optom Vis Sci 2007; 84: 463– 70 [DOI] [PubMed] [Google Scholar]
- 32. Smolek MK. Method for expressing clinical and statistical significance of ocular and corneal wave front error aberrations. Cornea 2012; 31: 212– 21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Linke SJ, Baviera J, Munzer G, Fricke OH, Richard G, Katz T. Mesopic pupil size in a refractive surgery population (13,959 eyes). Optom Vis Sci 2012; 89: 1156– 64 [DOI] [PubMed] [Google Scholar]
- 34. Ravikumar A, Applegate RA, Shi Y. Six just-noticeable differences in retinal image quality in 1 line of visual acuity: toward quantification of happy versus unhappy patients with 20/20 acuity. J Cataract Refract Surg 2011; 37: 1523– 9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Bühren J, Pesudovs K, Martin T, Strenger A, Yoon G, Kohnen T. Comparison of optical quality metrics to predict subjective quality of vision after laser in situ keratomileusis. J Cataract Refract Surg 2009; 35: 846– 55 [DOI] [PubMed] [Google Scholar]
- 36. Artal P, Chen L, Fernández EJ, Singer B, Manzanera S, Williams DR. Adaptive optics for vision: the eye’s adaptation to point spread function. J Refract Surg 2003; 19: S585– 7 [DOI] [PubMed] [Google Scholar]
- 37. Chen L, Singer B, Guirao A, Porter J, Williams DR. Image metrics for predicting subjective image quality. Optom Vis Sci 2005; 82: 358– 69 [DOI] [PubMed] [Google Scholar]
- 38. Legras R, Chateau N, Charman WN. Assessment of just-noticeable differences for refractive errors and spherical aberration using visual simulation. Optom Vis Sci 2004; 81: 718– 28 [DOI] [PubMed] [Google Scholar]
- 39. Applegate RA, Sarver EJ, Khemsara V. Are all aberrations equal? J Refract Surg 2002; 18: S556– 62 [DOI] [PubMed] [Google Scholar]
- 40. Maeda N, Fujikado T, Kuroda T, Mihashi T, Hirohara Y, Nishida K, Watanabe H, Tano Y. Wavefront aberrations measured with Hartmann-Shack sensor in patients with keratoconus. Ophthalmology 2002; 109: 1996– 2003 [DOI] [PubMed] [Google Scholar]
- 41. Pantanelli S, MacRae S, Jeong TM, Yoon G. Characterizing the wave aberration in eyes with keratoconus or penetrating keratoplasty using a high-dynamic range wavefront sensor. Ophthalmology 2007; 114: 2013– 21 [DOI] [PubMed] [Google Scholar]
- 42. Jafri B, Li X, Yang H, Rabinowitz YS. Higher order wavefront aberrations and topography in early and suspected keratoconus. J Refract Surg 2007; 23: 774– 81 [DOI] [PubMed] [Google Scholar]
- 43. Gobbe M, Guillon M. Corneal wavefront aberration measurements to detect keratoconus patients. Cont Lens Anterior Eye 2005; 28: 57– 66 [DOI] [PubMed] [Google Scholar]
- 44. Roorda A. Adaptive optics for studying visual function: a comprehensive review. J Vis 2011; 11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Nakagawa T, Maeda N, Kosaki R, Hori Y, Inoue T, Saika M, Mihashi T, Fujikado T, Tano Y. Higher-order aberrations due to the posterior corneal surface in patients with keratoconus. Invest Ophthalmol Vis Sci 2009; 50: 2660– 5 [DOI] [PubMed] [Google Scholar]
- 46. Sabesan R, Johns L, Tomashevskaya O, Jacobs DS, Rosenthal P, Yoon G. Wavefront-guided scleral lens prosthetic device for keratoconus. Optom Vis Sci 2013; 90: 314– 23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. López-Gil N, Castejón-Mochón JF, Fernández-Sánchez V. Limitations of the ocular wavefront correction with contact lenses. Vision Res 2009; 49: 1729– 37 [DOI] [PubMed] [Google Scholar]
- 48. Pérez-Merino P, Ortiz S, Alejandre N, de Castro A, Jiménez-Alfaro I, Marcos S. Ocular and optical coherence tomography-based corneal aberrometry in keratoconic eyes treated by intracorneal ring segments. Am J Ophthalmol 2014; 157: 116– 27 [DOI] [PubMed] [Google Scholar]
- 49. Bahar I, Kaiserman I, Srinivasan S, Ya-Ping J, Slomovic AR, Rootman DS. Comparison of three different techniques of corneal transplantation for keratoconus. Am J Ophthalmol 2008; 146: 905– 12 [DOI] [PubMed] [Google Scholar]
- 50. Vinciguerra P, Albè E, Trazza S, Rosetta P, Vinciguerra R, Seiler T, Epstein D. Refractive, topographic, tomographic, and aberrometric analysis of keratoconic eyes undergoing corneal cross-linking. Ophthalmology 2009; 116: 369– 78 [DOI] [PubMed] [Google Scholar]
- 51. Ghanem RC, Santhiago MR, Berti T, Netto MV, Ghanem VC. Topographic, corneal wavefront, and refractive outcomes 2 years after collagen crosslinking for progressive keratoconus. Cornea 2014; 33: 43– 8 [DOI] [PubMed] [Google Scholar]
- 52. Applegate RA, Hilmantel G, Howland HC, Tu EY, Starck T, Zayac EJ. Corneal first surface optical aberrations and visual performance. J Refract Surg 2000; 16: 507– 14 [DOI] [PubMed] [Google Scholar]
- 53. Pesudovs K, Figueiredo FC. Corneal first surface wavefront aberrations before and after pterygium surgery. J Refract Surg 2006; 22: 921– 5 [DOI] [PubMed] [Google Scholar]
- 54. Gumus K, Erkilic K, Topaktas D, Colin J. Effect of pterygia on refractive indices, corneal topography, and ocular aberrations. Cornea 2011; 30: 24– 9 [DOI] [PubMed] [Google Scholar]
- 55. Kwon SM, Lee DJ, Jeung WJ, Park WC. Power vector and aberrations using corneal topographer and wavefront aberrometer before and after pterygium surgery. J Korean Ophthalmol Soc 2008; 49: 1737– 45 [Google Scholar]
- 56. Zare M, Zarei-Ghanavati S, Ansari-Astaneh MR, Baradaran-Rafiee A, Einolahi B. Effects of pterygium on ocular aberrations. Cornea 2010; 29: 1232– 5 [DOI] [PubMed] [Google Scholar]
- 57. Gumus K, Topaktas D, Göktaş A, Karakucuk S, Oner A, Mirza GE. The change in ocular higher-order aberrations after pterygium excision with conjunctival autograft: a 1-year prospective clinical trial. Cornea 2012; 31: 1428– 31 [DOI] [PubMed] [Google Scholar]
- 58. Kuroda T, Fujikado T, Maeda N, Oshika T, Hirohara Y, Mihashi T. Wavefront analysis in eyes with nuclear or cortical cataract. Am J Ophthalmol 2002; 134: 1– 9 [DOI] [PubMed] [Google Scholar]
- 59. Pesudovs K, Elliott DB. Refractive error changes in cortical, nuclear, and posterior subcapsular cataracts. Br J Ophthalmol 2003; 87: 964– 7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Miranda MN. The geographic factor in the onset of presbyopia. Trans Am Ophthalmol Soc 1979; 77: 603– 21 [PMC free article] [PubMed] [Google Scholar]
- 61. Weale RA. Human ocular aging and ambient temperature. Br J Ophthalmol 1981; 65: 869– 70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Kim A, Bessho K, Okawa Y, Maeda N, Tano Y, Hirohara Y, Mihashi T, Fujikado T. Wavefront analysis of eyes with cataracts in patients with monocular triplopia. Ophthalmic Physiol Opt 2006; 26: 65– 70 [DOI] [PubMed] [Google Scholar]
- 63. Ninomiya S, Maeda N, Kuroda T, Saito T, Fujikado T, Tano Y, Suita J, Hirohara Y, Mihashi T. Evaluation of lenticular irregular astigmatism using wavefront analysis in patients with lenticonus. Arch Ophthalmol 2002; 120: 1388– 93 [PubMed] [Google Scholar]
- 64. Fujikado T, Shimojyo H, Hosohata J, Hirohara Y, Mihashi T, Maeda N, Tano Y. Wavefront analysis of eye with monocular diplopia and cortical cataract. Am J Ophthalmol 2006; 141: 1138– 40 [DOI] [PubMed] [Google Scholar]
- 65. Qu J, Sasaki H, Sakamoto Y, Kawakami Y, Sasaki K, Jonasson F. Higher-order ocular aberrations caused by crystalline lens waterclefts. J Cataract Refract Surg 2010; 36: 799– 805 [DOI] [PubMed] [Google Scholar]
- 66. Ohtani S, Gekka S, Honbou M, Kataoka Y, Minami K, Miyata K, Oshika T. One-year prospective intrapatient comparison of aspherical and spherical intraocular lenses in patients with bilateral cataract. Am J Ophthalmol 2009; 147: 984– 9 [DOI] [PubMed] [Google Scholar]
- 67. Santhiago MR, Netto MV, Barreto J, Jr, Gomes BAF, Mukai A, Guermandi APC, Kara-Junior N. Wavefront analysis, contrast sensitivity, and depth of focus after cataract surgery with aspherical intraocular lens implantation. Am J Ophthalmol 2010; 149: 383– 9 [DOI] [PubMed] [Google Scholar]
- 68. Kohnen T, Klaproth OK, Bühren J. Effect of intraocular lens asphericity on quality of vision after cataract removal: an intraindividual comparison. Ophthalmology 2009; 116: 1697– 706 [DOI] [PubMed] [Google Scholar]
- 69. McKelvie J, McArdle B, McGhee C. The influence of tilt, decentration, and pupil size on the higher-order aberration profile of aspheric intraocular lenses. Ophthalmology 2011; 118: 1724– 31 [DOI] [PubMed] [Google Scholar]
- 70. Pesudovs K, Dietze H, Stewart OG, Noble BA, Cox MJ. Effect of cataract surgery incision location and intraocular lens type on ocular aberrations. J Cataract Refract Surg 2005; 31: 725– 34 [DOI] [PubMed] [Google Scholar]
- 71. Levy J, Lifshitz T, Klemperer I, Knyazer B, Ashkenazy Z, Kratz A, Belfair N. The effect of Nd:YAG laser posterior capsulotomy on ocular wave front aberrations. Can J Ophthalmol 2009; 44: 529– 33 [DOI] [PubMed] [Google Scholar]
- 72. Villegas EA, Alcón E, Mirabet S, Yago I, Marín JM, Artal P. Extended depth of focus with induced spherical aberration in light-adjustable intraocular lenses. Am J Ophthalmol 2014; 157: 142– 9 [DOI] [PubMed] [Google Scholar]
- 73. Thibos LN, Hong X. Clinical applications of the Shack-Hartmann aberrometer. Optom Vis Sci 1999; 76: 817– 25 [DOI] [PubMed] [Google Scholar]
- 74. Tutt R, Bradley A, Begley C, Thibos LN. Optical and visual impact of tear break-up in human eyes. Invest Ophthalmol Vis Sci 2000; 41: 4117– 23 [PubMed] [Google Scholar]
- 75. Himebaugh NL, Nam J, Bradley A, Liu H, Thibos LN, Begley CG. Scale and spatial distribution of aberrations associated with tear breakup. Optom Vis Sci 2012; 89: 1590– 600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Koh S, Maeda N, Kuroda T, Hori Y, Watanabe H, Fujikado T, Tano Y, Hirohara Y, Mihashi T. Effect of tear film break-up on higher-order aberrations measured with wavefront sensor. Am J Ophthalmol 2002; 134: 115– 7 [DOI] [PubMed] [Google Scholar]
- 77. Montés-Micó R, Caliz A, Alio JL. Wavefront analysis of higher order aberrations in dry eye patients. J Refract Surg 2004; 20: 243– 7 [DOI] [PubMed] [Google Scholar]
- 78. Denoyer A, Rabut G, Baudouin C. Tear film aberration dynamics and vision-related quality of life in patients with dry eye disease. Ophthalmology 2012; 119: 1811– 8 [DOI] [PubMed] [Google Scholar]
- 79. Montés-Micó R, Caliz A, Alio JL. Changes in ocular aberrations after instillation of artificial tears in dry-eye patients. J Cataract Refract Surg 2004; 30: 1649– 52 [DOI] [PubMed] [Google Scholar]
- 80. Koh S, Maeda N, Ikeda C, Takai Y, Fujimoto H, Oie Y, Nishida K. Effect of instillation of eyedrops for dry eye on optical quality. Invest Ophthalmol Vis Sci 2013; 54: 4927– 33 [DOI] [PubMed] [Google Scholar]
- 81. Ridder WH, 3rd, LaMotte J, Hall JQ, Jr, Sinn R, Nguyen AL, Abufarie L. Contrast sensitivity and tear layer aberrometry in dry eye patients. Optom Vis Sci 2009; 86: 1059– 68 [DOI] [PubMed] [Google Scholar]
- 82. Kaido M, Matsumoto Y, Shigeno Y, Ishida R, Dogru M, Tsubota K. Corneal fluorescein staining correlates with visual function in dry eye patients. Invest Ophthalmol Vis Sci 2011; 52: 9516– 22 [DOI] [PubMed] [Google Scholar]
- 83. Lekhanont K, Chuckpaiwong V, Vongthongsri A, Sangiampornpanit T. Effects of sodium hyaluronate on wavefront aberrations in dry eye patients. Optom Vis Sci 2014; 91: 39– 46 [DOI] [PubMed] [Google Scholar]


