Abstract
Flavonoids and other polyphenols are ubiquitous plant chemicals that fulfill a range of ecologic roles for their home plant, including protection from a range of biotic and abiotic stressors and a pivotal role in the management of pathogenic and symbiotic soil bacteria and fungi. They form a natural part of the human diet, and evidence suggests that their consumption is associated with the beneficial modulation of a number of health-related variables, including those related to cardiovascular and brain function. Over recent years, the consensus as to the mechanisms responsible for these effects in humans has shifted away from polyphenols having direct antioxidant effects and toward their modulation of cellular signal transduction pathways. To date, little consideration has been given to the question of why, rather than how, these plant-derived chemicals might exert these effects. Therefore, this review summarizes the evidence suggesting that polyphenols beneficially affect human brain function and describes the current mechanistic hypotheses explaining these effects. It then goes on to describe the ecologic roles and potential endogenous signaling functions that these ubiquitous phytochemicals play within their home plant and discusses whether these functions drive their beneficial effects in humans via a process of “cross-kingdom” signaling predicated on the many conserved similarities in plant, microbial, and human cellular signal transduction pathways.
Introduction
It is generally accepted that the consumption of dietary polyphenols derived from fruit, vegetables, and other plant-derived foods may confer a number of health benefits, including to cardiovascular and brain function. An extensive and expanding literature describes how, in mechanistic terms, polyphenols may exert these effects, with the predominant current theory being that they do so via interactions with mammalian cellular signal transduction pathways. However, the question of why these phytochemicals have these effects has been primarily overlooked. Therefore, the current review summarizes both the evidence that polyphenols do indeed affect human brain function (with reference to cardiovascular function when relevant) and their suggested modes of action. It then goes on to consider why these compounds exert these effects, specifically whether their modulation of diverse physiologic variables in mammals is in fact related to the ecologic “secondary metabolite” roles and the endogenous signaling roles that the polyphenols are trying to play for, and within, their own home plant.
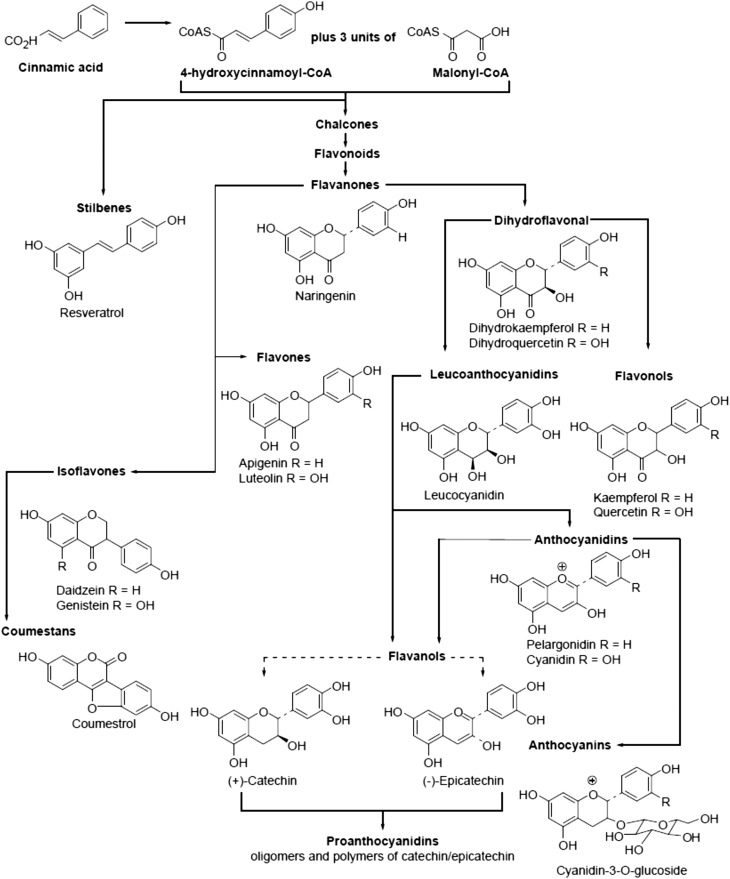
The “phenolics” represent a large group of ubiquitous phytochemicals that incorporate within their structure ≥1 phenyl aromatic hydrocarbon ring with ≥1 hydroxyl group attached. Within this broad chemical group, polyphenols represent a group of more complex phenolic structures that combine a shikimate pathway–derived cinnamic acid starter unit (cinnamoyl-CoA) with malonyl-CoA, which is derived via the acetate pathway. The majority of polyphenol groups incorporate 3 malonyl-CoA units and start with an identical polyketide before differentiating into stilbenes, which have a comparatively restricted distribution, and chalcones on the basis of the their first enzymatic step (stilbene synthase vs. chalcone synthase). The chalcones go on to act as the precursors for the entire flavonoid group of structures, which incorporates >8000 secondary metabolite compounds that are found ubiquitously across plants and plant tissues. The flavonoids share a common underlying structure of 2 six-carbon rings, with a three-carbon bridge, which usually forms a third ring (1), and the wider group can be further subdivided into the groups represented in Figure 1.
FIGURE 1.
Structure and synthetic pathways of the flavonoid and stilbene polyphenols, with selected example structures. Arrows indicate synthetic pathways. Reproduced from reference 139 with permission.
Polyphenols are an unavoidable component in the human diet. We obtain them from fruit, vegetables, cereals, seeds and beans, spices and herbs, oils, and all of the food products made from these basic components. Typically, the greatest quantities are consumed in the form of alcoholic and nonalcoholic beverages, such as wine and tea, fruit and fruit juices, and vegetables (2). Food diary studies suggest a wide variability in flavonoid consumption. For instance, the populations of the United States, Spain, and Australia were estimated to consume ∼190, 313, and 454 mg/d flavonoids, respectively, with the largest part being taken in the form of flavanols and their oligomers and polymers (2–4). Evidence also suggests that the consumption of dietary flavonoids is typically outweighed by simpler phenolics. For instance, a study conducted in Finland demonstrated a mean consumption of 222 mg/d flavonoids and 640 mg/d phenolic acids (mainly via chlorogenic acid from coffee) (5). Similarly, a French cohort consumed a mean of 1193 mg/d phenolics, of which phenolic acids contributed 639 mg (6).
Current Status of Knowledge
Polyphenols and human brain function
Epidemiologic evidence suggests that the consumption of polyphenols confer a wide range of health benefits. For example, the consumption of polyphenol-rich foods and drinks, such as chocolate (7) and tea (8), are inversely related to cardiovascular disease. Overall self-reported consumption of flavonoids has also been consistently shown to be associated with reduced mortality due to cardiovascular disease (9, 10), with the inverse relation between flavonoid consumption and hypertension/blood pressure confirmed by urinary analysis (11). Intervention trials have also confirmed these cardiovascular benefits, although much of this research has focused on flavanols, most often derived from cocoa (12). Taken as a whole, these trials suggest a consistent beneficial effect of cocoa flavanols on cardiovascular variables, including inflammatory biomarkers related to atherosclerosis, insulin resistance, lipid profiles, blood pressure, and vasodilation/endothelial function (9, 13, 14). Meta-analyses suggest that these effects are achievable with an optimal dose of 500 mg/d flavanols (14) and within 2 h of consumption (15).
Naturally, cardiovascular variables are inextricably linked to cerebral blood flow and metabolism in the brain, and therefore, they covary with the incidence of age-related cognitive decline, dementia, and mood disorders (16–20). In line with this, the consumption of tea, polyphenol-rich foods, fruit and vegetables, and total amounts of flavonoids have been shown to be associated with protection against, or slowed progression of, cerebrovascular diseases, such as strokes, and neurologic disorders, including dementia (21–28), and cognitive impairment/decline in elderly populations (22, 24, 29–31).
Intervention studies
Flavonoids.
A wealth of evidence in animals has shown that flavonoid-rich foods, extracts, and individual polyphenols can beneficially modulate cognitive function, typically in older and impaired rats that are suffering cognitive decrements as a consequence of age, brain insults, or induced pathologies. For instance, flavanol and anthocyanin–rich fruits and berries and single flavonoids consistently improve or prevent declines in all aspects of memory performance (32–35). These effects can also be accompanied by morphologic changes to structures relevant to memory, for example, by promoting or protecting neurogenesis and synaptic plasticity in the hippocampus (32, 33, 36, 37).
There is less evidence pertaining to humans in this regard, and flavanols have attracted the most attention. Two studies, 1 using transcranial Doppler (38) and the other using fMRI (39), demonstrated that both high (900 mg) and low (150 mg) doses of flavanols administered for a short period (2 wk and 5 d, respectively) can increase cerebral blood flow in healthy older and younger adults. In terms of direct modulation of cognitive function, 2 placebo-controlled, crossover design studies demonstrated benefits in the cognitive function of healthy adults as measured by a “cognitive demand battery” (40) and by spatial memory and choice reaction time tasks (41) after single doses of flavanol-enriched drinks and chocolate, respectively. In the case of the latter study (41), 2 visual tests also demonstrated that the high-flavanol treatment increased the speed of detecting the direction of motion of stimuli and led to an improvement in visual contrast sensitivity. The evidence with regards longer-term administration is less persuasive. Although in 1 study cocoa flavanols (990/520 mg/d) reduced insulin resistance, blood pressure, and lipid peroxidation and improved performance on 2 of 3 cognitive tasks (42), several studies failed to elicit any improvements in cognitive function. For instance, there were no cognitive improvements reported after administration of chocolate products containing 750 mg/d flavanols for 6 wk in participants ≥60 y (43) and no improvement on a working memory task after 30-d administration of 250 or 500 mg cocoa flavanols to middle-aged volunteers, although steady-state visually evoked potentials assessed by electroencephalography (EEG) were modulated in a manner interpreted by the authors as showing increased neural efficiency (44). Finally, a recently reported study also assessed the effects of both single doses and 30-d administration of 250/500 mg/d cocoa flavanols in healthy middle-aged participants and failed to demonstrate any substantial cognitive effects, although the subjective psychological state of the participants was improved in terms of “calmness” and “contentedness” (45).
For other sources of flavanols, 2 studies reported modulation of brain function by single doses of the tea catechin epigallocatechin gallate (EGCG) in young participants, as assessed by EEG (40) and near-infrared spectroscopy (46), and 2 studies reported modulation of activity after green tea extracts (containing flavanols and other components) as assessed by fMRI after a single dose (47) and EEG after 16-wk administration (48) in the absence of cognitive/mood effects.
Two placebo-controlled trials also demonstrated improved subjective climacteric symptoms in menopausal females after 3-mo (60 mg/d) (49) and 6-mo (200 mg/d) (50) administration of Pycnogenol, a proanthocyanidin-rich pine bark extract. Memory function and oxidative stress markers were also improved after 3-mo administration of 150 mg of Pycnogenol to elderly participants (51). However, 2 studies assessing the effects of high doses (∼1000 mg/d) of a similar extract, Enzogenol, for 5 or 6 wk did not show any improvements on a range of psychological or physiologic variables in healthy adults (52) and sufferers from mild traumatic brain injury (53).
Anthocyanins, the glycosides of anthocyanidins, are typically consumed by humans in brightly colored berries, grapes, fruits, and colored vegetables (54), and their consumption has been consistently shown to improve cognitive function in animal models of aging and neuropathology (55). However, human research has been methodologically inadequate, with the small sample sizes and control conditions in the 3 studies that involved the repeated administration of anthocyanin and phenolic acid–rich juice drinks rendering the modest findings essentially uninterpretable (56–58). Only 1 study assessed the effects of single doses with a comparatively healthy sample of 35 participants in a crossover design study, but this study found no substantial effects on a restricted range of cognitive, mood, and satiety measures (59).
Phytoestrogens.
A range of flavonoids and other polyphenols have estrogenic properties as a consequence of their direct binding affinity at mammalian nuclear estrogen receptors (ER) α and β. These polyphenols include the isoflavones genistein and daidzein and the structurally related coumestans, such as coumestrol, all of which are particularly richly expressed in the legume (Fabaceae) family. Other phytoestrogens include lignans, which are converted by intestinal bacteria to estrogenic metabolites, such as enterodiol, enterolactone, and secoisolariciresinol (60), as well as a number of ubiquitous flavones, including, in order of descending potency, kaempferol, quercetin, apigenin, and luteolin, several flavanones, including naringenin, and several stilbenes, including resveratrol (61).
The phytoestrogens typically exhibit weak ER binding, with a preference for ERβ, but exist at comparatively high concentrations (62). They can also act as agonists or antagonists, the latter via blockade of the activity of endogenous ligands, can bind to and inhibit the activity of androgen or estrogen metabolizing enzymes, such as aromatase (60), and can exert non-receptor–binding properties, potentially via modulation of signaling cascades related to estrogenic functioning (63). This multiplicity of effects means that the activity of the phytoestrogen can be highly variable and can depend on a number of factors, including tissue type, receptor subtype, dose, and the amount of the endogenous ligand present (60).
Most animal and human research in this area has tended to use the “soy” isoflavones coumestrol, genistein, and daidzein, found most abundantly in the soya bean plant (Glycine max), although these compounds are actually expressed in substantial amounts by a wide range of plants, including the majority of the legume (Fabaceae) family, and other food crops, such as the Caffea (coffee) genus.
Daidzein itself can be metabolized by intestinal bacteria into the more potent estrogenic compound equol, although only 30–50% of individuals are equipped with the requisite intestinal flora to accomplish this, with the percentage rising in cultures that eat more daidzein-containing plant products (64).
Soy isoflavones have been shown to exert a number of effects relevant to general health, including upregulation of endogenous antioxidant activity, and modulation of cardiovascular and immune function variables, and the mechanisms underlying carcinogenesis, including the inhibition of aberrant mitogenic activity related to ERα activity (65). These factors may underlie the epidemiologic observations of a potential relation between soy-isoflavone consumption and breast cancer (66–69), in particular those cases that feature the overexpression of ER and progesterone receptors within tumor cells (67). Soy-isoflavone supplementation has also been associated with decreased bodyweight and improved glucoregulation in postmenopausal non-Asian women (70), decreased menopausal symptoms (71), and improved vascular variables, such as endothelial function (72) and blood pressure (73).
With regards effects on brain function, soy isoflavones have been shown to improve memory performance in intact and ovariectomized rodents (74–77). In humans, they have been shown to substantially improve the physical, but not psychological, symptoms of premenstrual syndrome (78), improve ratings of quality-of-life in postmenopausal women (79), decrease both follicle-stimulating hormone and luteinizing hormone, and increase circulating concentrations of 17β-estradiol (80). In terms of neurocognitive function and mood, a review by Lamport et al. (81) identified 13 methodologically adequate studies that assessed the effects of isoflavone treatments on cognitive function. Seven of these studies demonstrated modest beneficial effects of supplementation. The authors interpret the findings as being inconclusive, although it is interesting to note that most of the studies involved postmenopausal females (81). In addition, Greendale et al. (82) found that isoflavone consumption could be related to either cognitive benefits or decrements, depending on the stage of menopause. Taken with results showing that several years after ovariectomy primates lose the ability to respond to either 17β-estradiol or equol in terms of hippocampal receptor binding and activity (83), this would seem to confirm a theorized “window of opportunity” for hormone replacement therapy and isoflavone supplementation. This factor may also contribute to the somewhat equivocal nature of the human intervention literature. Interestingly, in the review by Lamport et al. (81), 3 of the 4 studies that included males in their samples demonstrated cognitive benefits, including a demonstration of selective isoflavone-related benefits on a spatial working memory task on which females usually outperform males (84). Evidence actually suggests both cognitive benefits and deficits in male rats after phytoestrogen consumption (74), so taken together, it would be interesting to see more data from male humans and, indeed, younger premenopausal females.
Nonflavonoid polyphenols.
Consumption of the stilbene resveratrol (3,4′,5-trihydroxystilbene) is associated with numerous protective health benefits in animal models. These include increased longevity (85), anti-inflammatory (86) and antiviral (87) properties, and a protection against cancer and tumorogenesis (88), cardiovascular disease (89), and atherosclerosis (90). In terms of cognitive performance, a number of in vivo studies in rodents have demonstrated preserved behavior and cognitive performance in aged rats after laboratory-induced brain insults (91–94).
Despite resveratrol being deemed safe for human consumption, the huge body of literature surrounding this compound mainly deals with in vitro and in vivo/ex vivo animal research. Human intervention trials include 2 studies that demonstrated that both acute (30, 90, and 270 mg/d) (95) and chronic (75 mg/d) (96) administration of resveratrol improved peripheral vasodilation as measured by flow-mediated dilation in obese individuals. Similarly, 2 studies assessed the effects of resveratrol on cerebral blood flow and cognitive function. In the first, single doses of 250 and 500 mg of resveratrol increased cerebral blood flow in the frontal cortex in a dose-related manner during cognitive task performance, whereas both doses also increased deoxygenated hemoglobin concentrations, indicative of increased oxygen utilization, as measured by near-infrared spectroscopy (97). In the second study, 250 mg of resveratrol combined with 20 mg of the alkaloid piperine had exactly the same effect on cerebral blood flow variables (98). In neither study was cognitive function or mood modulated.
Interestingly, the less intensively researched stilbene pterostilbene, a dimethylether analogue of resveratrol, has greater oral bioavailability in plasma and brain tissue (99, 100), has more potent cancer cell inhibitory effects (101) and exerts greater cognitive/neuroprotective and cellular signal transduction effects in rodent models than resveratrol (100). To date, its efficacy has not been tested in humans.
The curcuminoid curcumin is formed from 1 cinnamic acid starter unit with 2 malonyl-CoA units (1). It is responsible for the bright yellow color of the Indian spice turmeric (Curcuma Longa) and has been used for centuries within the Ayurvedic system of medicine in the treatment for a host of ailments, including inflammation (102).
Despite a number of clinical trials that generated results suggesting potential utility in treating inflammation, cardiovascular disease, and diabetes (103), little research has focused on brain function. Curcumin is associated with an attenuation of cognitive deficits in rodent models of Alzheimer’s disease (104, 105). Epidemiologic data also suggest a relation between better cognitive performance and curry consumption in humans (106). However, to date, the results of only 2 small studies in humans, both assessing the effects of curcumin in Alzheimer’s sufferers, have been reported, and neither demonstrated symptomatic or biochemical efficacy (107, 108).
Although the evidence from intervention studies of direct modulation of human brain function by polyphenols, as reviewed above, is currently weak, it is notable that this may primarily reflect the limited research in this area and the methodologic limitations of the small number of studies. Certainly, the results of the many animal studies investigating brain function, the epidemiologic evidence, and the evidence of cardiovascular benefits after polyphenol interventions all point in the direction of a specific benefit to central nervous system function associated with polyphenol consumption. Naturally, this raises the question of how these benefits are achieved.
Polyphenols: mechanisms of action relevant to human brain function
The notion that polyphenols owe their beneficial effects on physiologic variables and disease states to direct antioxidant effects has been replaced by a growing consensus that their effects are more likely attributable to direct interactions with cellular signal transduction pathways (109). Comparatively, low bioavailability certainly precludes antioxidant properties as a key factor in their neuroprotective and neuroenhancing effects (110). Naturally, access to the brain is a necessary precondition for exerting direct effects on brain function, and it is notable that, after dietary supplementation, flavonoids and their metabolites were shown to be present in the brain at the low concentrations (10–300 nM) that would be sufficient for them to exert pharmacologic effects at receptors and within signaling pathways (33). This ability to cross the blood–brain barrier seems to be dictated by both the lipophilicity of the individual molecule and its interactions with transmembrane proteins, such as the multidrug resistance permeability glycoproteins, which transport molecules across cellular membranes and the blood–brain barrier (36, 111). Interestingly, several classes of flavonoids become more concentrated and are retained in neural tissue longer than they persevere in plasma (32). In addition to direct interactions with neurotransmitter receptors, polyphenols can also interact directly within diverse downstream neuronal and glial protein kinase and lipid kinase signaling cascades, such as the ubiquitous MAPK and phosphatidylinositide 3-kinase (PI3K)/protein kinase B (PKB) and target of rapamycin (TOR) signaling cascades (36, 112, 113). These ubiquitous signaling cascades transduce signals received either by receptors spanning the membrane of the cell or in its cytosol via a chain reaction in which a series of kinases activate each other in turn, by phosphorylation, ultimately leading to either the removal of proteins attached to transcription factors in the cytosol, allowing them to translocate to the nucleus, or direct interactions with transcription factors already in the nucleus. The summed and interacting activity in multiple signaling pathways dictates the response of the cell to environmental or stressor-related information, for instance, by modulating the activity of transcription factors, such as NF-κB or CREB (cAMP response element binding protein), in turn leading to a wide range of cellular responses, including cell proliferation, apopotosis, and the synthesis of growth factors, such as neurotrophins, and inflammatory molecules, such as induced NO synthase (iNOS), cytokines, and cyclooxegenase-2 (COX-2). Overactivity or dysregulation within these signaling pathways is implicated in the pathogenesis of cardiovascular and neurodegenerative diseases and cancers (36, 114). Recent research demonstrates that a range of flavonoids bind directly to individual protein kinases within these cascades, modulating their phosphorylation state and thereby modifying the activity and outcome of the signaling pathway (115). The effects of polyphenols within the central nervous system can then be primarily attributed to interactions with signal transduction pathways that both have a direct effect on cognitive function and an indirect effect via the attenuation of inflammatory processes and the enhancement of cerebrovascular function.
Cognitive function.
Flavonoids modulate cellular signaling pathways most directly by interacting with a range of receptors to which neurotransmitters and other signaling molecules bind. Dietary flavonoids have been shown to interact with estrogen (see above), GABAA (116), adenosine (117), opioid (118), nicotinic (119), and receptor tyrosine kinases. The latter include receptors such as tyrosine-related kinase B, which responds to key neurotrophins such as brain-derived neurotrophic factor (120). Evidence also suggests the existence of brain plasma membrane and nuclear binding sites that have yet to be fully characterized but for which flavonoids have a high affinity (121). In terms of downstream effects within signal transduction pathways, flavonoids may exert beneficial effects on cognitive function by activating components of the extracellular signal-regulated kinase (ERK) signaling cascade that leads to increased activity of transcription factors, such as cAMP response element binding protein, with a resultant increase in the expression of neurotrophins, such as brain-derived neurotrophic factor. This can ultimately lead to an increase in the synaptic plasticity and long-term potentiation that underlies long-term memory consolidation. Alternatively, upregulatory interactions within the PI3K/PKB pathways may lead to increased activity in the nutrient-sensing TOR signaling pathways or the increased expression of endothelial NO synthase (eNOS) and therefore NO synthesis (36, 112).
Cerebrovascular function.
The modulation of “good” NO synthase activity via eNOS leads to a brain-specific increase in local blood flow, angiogenesis, and neurogenesis, all of which may contribute to neuroprotection and neuronal repair in the face of aging and insults (36, 112). Modulation of eNOS synthesis also underpins the ability of flavonoids to improve peripheral vascular variables, such as endothelial function, blood pressure, and platelet aggregation, and in turn beneficially modulate gross cerebral blood flow and hemodynamic responses to neural activity. Notably, polyphenol-related increases in vasodilation, cerebral blood flow, and NO synthesis are also implicated in hippocampal angiogenesis and neurogenesis, processes that are implicated in learning, memory, and neuroprotection (36, 122).
Neuroinflammation.
Flavonoids can also selectively inhibit deleterious overactivity in signaling pathways. The beneficial effects of flavonoids on cardiovascular health and cancer has been attributed to an attenuation of the inflammatory cascades implicated in the pathogenesis of cardiovascular disease and tumorogenesis (123). As with other tissues, short-term inflammation of brain tissue can be a beneficial, natural, defensive reaction to injury, infection, stroke, and toxins. However, sustained neuroinflammation as a consequence of continued activation of microglia, the primary immune cells of the nervous system, and their subsequent sustained release of damaging proinflammatory mediators may contribute to the neuronal damage associated with neurodegenerative diseases, such as multiple sclerosis, Parkinson’s disease, and various dementias, as well as the deterioration in cognitive function seen with aging (124).
Evidence suggests that disparate flavonoids, including a number that act as integral parts of the human diet, such as flavanols and isoflavones and their metabolites, can suppress the neuroinflammatory activity of microglia by inhibiting each stage of the inflammatory signaling process in activated glia: inhibiting the activity of proinflammatory transcription factors, the release of cytokines, the generation of reactive oxygen species, and the synthesis or activity of iNOS, COX-2, and lypoxygenase and the resultant overproduction of NO, PGs, and leukotrienes. These effects in turn may be related to the ability of polyphenols to modulate the activity of multiple components of a range of neuronal and glial signaling pathways, including the individual components of MAPK and other kinase pathways involved in the inflammatory and apoptotic responses described above (114, 123). As an example, polyphenol-rich acai fruit pulp was shown to attenuate inflammatory responses in microglial cells and protect neurons from induced stress by inhibiting both MAPK and TOR pathways (125, 126).
Evidence does suggest that interactions with the components of these signaling pathways is dependent on the exact structure of the polyphenol molecule in question, for instance, in terms of the number and location of hydroxyl groups or the nature of specific bonds. This means that different polyphenols will exert markedly different cellular effects (36). However, a recent meta-analysis of 25 human intervention studies that included an assessment of the effects of a wide range of flavonoids on inflammatory biomarkers noted a substantial reduction in TNF-α associated with consuming flavonoid-rich foods or supplements and several flavonoid subgroups (127). Similarly, oral administration of 40 mg of resveratrol to humans was shown to lead to reductions in oxidative stress variables, the expression of proinflammatory kinases in mononuclear cells, the activity of the proinflammatory transcription factor NF-κB, and concentrations of related downstream proinflammatory cytokines (128), and increase nuclear factor-E2-related factor-2 activity with a consequent upregulation of endogenous antioxidant variables (129). The attenuation of neuroinflammation could certainly underpin the epidemiologic evidence of protected brain function in elderly humans.
Metabolites and the role of the gut microbiota.
Of course, there is a proviso to the above interpretation of the mechanisms of action of polyphenols. In reality, only a small percentage of polyphenols are absorbed intact in the upper gastrointestinal tract, with the vast majority hydrolyzed to aglycones and/or metabolized by gut microbiota before absorption and additional phase I/II metabolism (130, 131). Emerging evidence suggests that the gut microbiota makes a substantial contribution to the eventual palette of circulating, potentially bioactive, compounds, including the generation of entirely new compounds, such as a range of conjugated lactones and simple phenolic acids (131–133). To add more complications, the eventual palette of polyphenol derivatives will depend on the individual’s own gut microbiota profile, and the consumption of differing polyphenols will change the makeup of the microbiota itself (131, 134). Given that the composition of the gut microbiota plays multifarious roles in organism development, digestion, the bioavailability of bioactive nutritional components, immune function (130), and even brain development and behavior (135, 136), it has been suggested that many of the health benefits of polyphenols may be related to modulation of gut microbe populations (130).
The extensive metabolism of polyphenols does bring into question the relevance of much of the in vitro research that has informed the above. This research has often used naturally unattainable concentrations of polyphenols and parent compounds rather than the many metabolites that would be present in vivo (114). Similarly, it is not possible to attribute the many in vivo biologic effects, including those seen in human trials, directly to the consumed parent molecules rather than their metabolites. However, it is notable that the conjugated forms of polyphenols and the phenolic metabolic derivatives created by the gut microbiota typically have similar or greater bioactivity than the parent compounds, including within signaling pathways (133, 134, 137). It is also notable that the gut microbiota play an important evolved role in the tailoring of a wide range of exogenous dietary nutrients for increased bioactivity, including essential amino acids and several vitamins (138). Given that polyphenols formed part of the human diet throughout evolution, it would be surprising if the microbiota community did not fulfill similar roles for this group of compounds.
Why Do Polyphenols Affect Human Brain Function?
The ecologic roles of polyphenols and other phenolics
Although they are the dominant life form on earth, covering and harvesting the light from the vast majority of the terrestrial surface of the planet, plants have 1 major problem: they are autotrophs, rooted in place and feeding themselves by synthesizing the complex organic compounds that they require for life, from simple inorganic molecules that they find in their immediate vicinity. Their inability to move means that plants evolved the ability to synthesize a vast array of secondary metabolite chemicals (“secondary” because they do not take part in primary metabolism) that play ecologic roles, protecting the plant from physical challenges and allowing it to interact with its environment. Among the 3 major classes of secondary metabolite chemicals (phenolics, alkaloids, and terpenes), the phenolics play by far the widest range of ecologic roles, as befits their provenance as the most ancient of the 3 groups in evolutionary terms (139). From an external perspective, flavonoids concentrated in the epidermis of leaves or the cuticle of flowers and fruits provide a range of colors that attract pollinators and fruit-dispersing animals but deter herbivores. These range from the rich colors associated with anthocyanins and anthocyanidins, to the whites, yellows, and “ultraviolet” nectar guides provided by flavones and flavonols (140). All of these pigments also function as sunscreens and antioxidants, absorbing dangerous wavelengths of ultraviolet light and mopping up reactive oxygen species (141). Phenolics also comprise an important antimicrobial and antioxidant component of the sugar-rich nectar that manages the relation of the flower to symbiotic pollinators and “nectar robbers” (142–144). The more volatile simple phenolics also contribute generously to the “volatilome” of airborne chemicals that the plant releases to attract and deter pollinators and herbivores. This complex cocktail of chemicals includes a number of signaling molecules, such as methyl salicylate, the volatile ester of the hormone salicylic acid, and phenylpropenes, such as eugenol and estragole, all of which are released into the air around the plant as a rapid signaling system designed to deliver information about stressors and physical integrity rapidly to the distant parts of the plant, triggering a range of local responses in the home plant (145, 146). These chemicals can also orchestrate similar stress-related responses in the local community of plants (147).
In the case of direct herbivorous assault, flavonoids in plant tissue will act as first-line-of-defense general-purpose antifeedants, digestibility reducers, and toxins (148). The direct reaction of the plant to the herbivorous assault will be dictated by whether the assault involves chewing or sucking, with enzymes within the insect’s saliva, such as β-glucosidases or FA/amino-acid conjugates, potentially triggering a specific immune-like reaction driven by jasmonic acid or salicylic acid, respectively (see below) (149, 150). These hormonal signals might result in the synthesis and eventual polymerization of phenolic cinnamic acid alcohol monomers that will interlock to form woody lignin as a physical defense (151), and they will inevitably result in an increase in the synthesis of defensive secondary metabolites, including the many stress-induced phenolic compounds that directly protect plant tissue. The low-molecular-weight phenolics will also contribute to the volatile emissions that attract the predatory enemies of the herbivorous insect (152).
If the plant is damaged, microbial pathogens penetrating the wound will immediately encounter chemical defenses in the form, for instance, of the anthocyanidins that are distributed throughout plant tissue (148). Their molecular fingerprint will also be identified by transmembrane “pattern recognition receptors,” leading, over several days, to jasmonate hormone–driven synthesis of “phytoalexin” secondary metabolites that will accumulate around the site of infection (153). Phenolics comprise the phytoalexins most commonly used by plants, playing this role in all gymnosperms, and the majority of families of angiosperm plants (154).
Dropping below ground, the “rhizosphere,” the root system and surrounding soil, houses an extensive microbiota that bears striking functional similarities to the human gut microbiota, including a role in the metabolism and reabsorption of phenolics and the metabolic tailoring of key nutrients (138). This is a world that is particularly rich in phenolic secondary metabolites (155, 156), many of which are exuded into the soil in response to specific signals, for instance, from symbiotic and pathogenic microbes, and a range of abiotic stresses, such as nutrients and the mineral status of the soil, temperature, and water stress (157). Among the full range of phenolic structures in the rhizosphere, the flavonoids play specific benign roles in the management of microbial symbionts, attracting mutualist bacteria and triggering the germination of fungal spores. These microbes then colonize the roots, facilitating the uptake of nitrogen, water, nutrients, and minerals, in return for photosynthates, such as carbohydrate, from the plant (158, 159). These relations can be quite specific; in the legume (Fabaceae) family, the plant grows root nodules for the bacteria to inhabit, with individual flavonoids managing relations with individual species of Rhizobium bacteria (148, 157). Alternatively, they can be more general; for instance, ubiquitous flavonoids, such as quercetin and rutin, manage the relation of the plant with the arbuscular mycorrhizal fungi that contribute to plant phosphorus uptake by colonizing the surface of roots in the vast majority of plants (148, 159). Naturally, the protective reaction of the plant to infection by underground pathogenic fungi and bacteria is identical to that seen aboveground and often also involves the synthesis of phenolics.
At a greater distance from the plant, the soil still harbors a variety of phenolic secondary metabolites that were exuded by roots or leached from leaves and fallen plant tissue. For instance, flavanols and tannins protect against nematodes (148) and molluscs (160), whereas a number of phenolics in the soil inhibit the growth and germination of competitor plants. Many flavonoids exert these allelopathic properties by interfering with the signaling and transport of the hormone auxin in the roots of competitors or acting as prooxidants, among many other potential mechanisms of action.
In all of these many and varied interactions, individual phenolics are often multifunctional, multitalented compounds.
The plant signaling roles of polyphenols.
Although a huge research effort has been directed toward understanding the effects of polyphenols on mammalian cellular signaling, we know very little about the potential endogenous signaling roles that polyphenols play in their home plant. Nevertheless, a number of strands of evidence suggest that flavonoids may play multiple “primary” plant signaling roles. For example, it is notable that these chemicals are often synthesized locally by specialized cells or tissues at specific times dictated by developmental processes or stressors. They are often then actively transported long distances in the plant, becoming “integral components of the plant signaling machinery,” functioning both as signaling molecules in their own right and by interfering with the activity of other signaling molecules (161). As an example, flavonoids play recently established cellular signaling roles in pollen germination and dormancy, the sanctioning of the transmembrane movement of the hormone auxin, the nodulation process that allows the colonization of root systems by symbiotic bacteria, and the lignification process, and they partake in intraplant communications (161, 162). As an example of the latter, a recent study investigating gene expression showed that the allelopathic effects of juglone, a phenolic derivative that stunts the growth of plants in the vicinity of walnut trees by preventing root elongation, are related in major part to the ability of juglone to interfere with MAPK cellular signal transduction and gene expression within the jasmonic acid, abscisic acid, and gibberellic acid hormonal pathways of the encroaching recipient plant (163). A putative wide-ranging signaling role for flavonoids is also strongly supported by their detection, along with their synthetic enzymes, in the nucleus of plant cells, suggesting that they may function in endogenous gene transcription (164, 165).
Similarities in hormonal stress signaling between plants and animals.
Both plants and animals have well-developed autocrine, paracrine, and endocrine hormonal systems. In plants, the typical net effect of hormonal signaling will include modulation of the synthesis of numerous secondary metabolites, including flavonoids and other polyphenols. The key hormones upregulating defensive reactions are the oxylipin “jasmonates,” jasmonic acid, and its conjugated and hydroxylated derivatives, such as methyl-jasmonate and jasmonoyl-isoleucine. The jasmonates are formed from the eighteen-carbon PUFA α-linolenic acid when it is released from cellular glycerides by lipases in plant tissue as a consequence of biotic and abiotic stressors and subsequently metabolized via the octadecanoid pathway, which commences with oxidation by lipoxygenase enzymes (166, 167). The jasmonate derivatives then travel to other cells and degrade cellular proteins [jasmonate ZIM (zinc-finger expressed in inflorescence meristem)-domain proteins] that are suppressing the transcription factors that dictate the many physical and chemical plant responses to stressors, unleashing the appropriate defensive reactions (168, 169).
Therefore, although jasmonates are necessary for the induced synthesis of protective secondary metabolites, they represent only 1 strand of a complex interplay between hormones. These include salicylic acid, abscisic acid, auxin, and ethylene, as well as ubiquitous signaling molecules, such as NO. These chemicals are synthesized in varying quantities in response to differing stressors. For instance, jasmonic acid predominates in the response of a plant to insect herbivory and abiotic stressors, jasmonic acid and ethylene combined are synthesized in response to necrotrophic pathogen attack, and salicylic acid is induced after infestation with biotrophic pathogens (170). The synergistic and antagonistic crosstalk between these hormones and signaling molecules fine tunes the response of the plant to match the stressor (171), leading to complex stress-specific patterns of gene transcription (168, 170). As an example, whereas abscisic acid tends to work synergistically with the jasmonates (167), salicylic acid has an antagonistic relationship, in which 1 of its functions is to downregulate the jasmonate pathways, in effect switching the response of the plant from a short-term synthesis of phytochemicals suited to abiotic stressors and herbivory and redirecting it toward a longer-term “immune” response that will be more suited to resisting biotrophic and viral pathogens. Naturally, the converse is also true, with the jasmonates working to downregulate the salicylates (167, 172–174) and salicylic acid modulating the gene expression associated with other hormones, such as auxins (175).
These plant hormonal systems bear some striking similarities to animal systems (139). The most straightforward example is the role that the “plant” hormone abscisc acid plays in the mammalian nervous system. This genetically conserved signaling molecule is synthesized and operates via the same signaling pathways and conserved genes across plants and animals (176, 177). In both taxa, it guides the cellular reactions to abiotic stressors, such as heat and light. It also has wide-ranging functions within the mammalian immune system and modulates insulin release from the pancreas and the proliferation of stem cells. A number of these effects are related to its modulation, including upregulation, of the synthesis or function of PGs (176, 178).
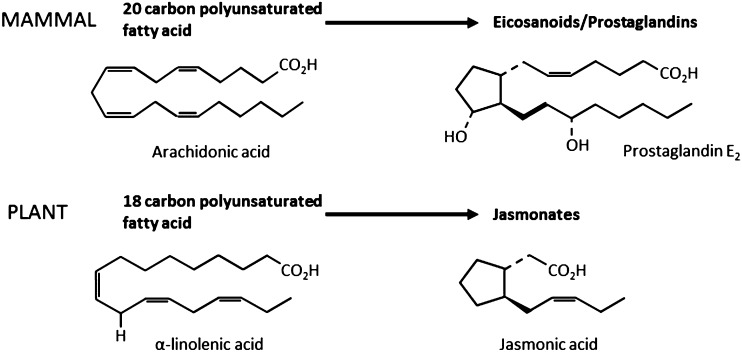
No less intriguingly, the oxylipin pathways that synthesize jasmonates in plants and the PGs in mammals are also genetically conserved orthologs (179). In mammals, this pathway leads to the synthesis of eicosanoids from the twenty-carbon PUFA arachadonic acid, which is released from cellular phospholipids by phospholipases and metabolized via pathways very similar to those seen in the formation of jasmonates in the plant, but in this case featuring both lipoxygenase and the COX-1 and COX-2 enzymes (180). In keeping with this shared heritage, the jasmonates in plants and the PGs in animals are closely related in structural (Fig. 2) and functional (179) terms. In animals, PGs and the COX enzymes contribute to the modulation of blood flow via the dilation or constriction of blood vessels and by determining the aggregation, or stickiness, of blood platelets, and they contract or relax bronchial and smooth muscle. Directly in keeping with the plant roles of jasmonates, they also govern a number of responses to stressors, including the regulation of inflammation and immune function, and the response to wounding (180, 181). Therefore, the most striking difference in the response of the plant jasmonate and animal PG systems to stressors is primarily seen in the end product, with the animal response most often associated with inflammation or immune system activation and the plant response, although including many analogous cellular responses, also being typified by the additional synthesis of secondary metabolite chemicals (182).
FIGURE 2.
Conserved signaling molecules: the plant jasmonate hormones (e.g., jasmonic acid) and the orthologous mammalian eicosanoids/PGs (e.g., PGE2) showing the analogous PUFA precursors. Reproduced from reference 139 with permission.
A wealth of emerging evidence suggests that each of the key hormonal mediators of secondary metabolite synthesis in plants can have reciprocal effects within mammalian tissue. Modulation of endogenous abscisic acid function in humans is being investigated from a therapeutic point of view with respect to a variety of diseases (176), and both the jasmonates and salicylates exert multifarious, potentially beneficial effects on mammalian cancerous cells in a manner that resembles their innate activity in plant cells (182). The activity of salicylic acid in terms of inducing tumor cell apoptosis, in particular, resembles the hypersensitive response, or programmed cell death, that it coordinates in response to pathogens in plants (182). Salicylic acid itself, particularly in the structurally close form of acetylsalicylic acid, or aspirin, has also attracted a huge amount of research over many decades. Its activity in mammals systems can be seen as a direct extension of its role within plants (183), with many of its actions involving interactions with mammalian cellular processes that are directly conserved or very similar to those seen in plants. The most readily appreciable example is the inhibition of COX activity by acetylsalicylic acid, which leads to reduced PG synthesis, and the celebrated anti-inflammatory properties of aspirin (182). This can be seen as a direct reflection of the analogous antagonistic effect of salicylic acid within the jasmonate pathways that induce secondary metabolite synthesis in plants, in much the same way that the upregulation of PG function by abscisic acid in the animal mirrors its synergistic relation with the jasmonates in the plant (180).
Is the modulation of brain function by polyphenols a consequence of unintended “cross-kingdom” signaling between plants and humans?
Interference by the phenolic plant hormone salicylic acid in the synthesis of PGs is clearly an example of unintended “cross-kingdom” signaling by the phytochemical. The concordance in hormonal signaling between plants and humans is not surprising given the similarities in cellular function exhibited by the 2 taxa. Both plants and humans inherited in excess of ∼3000 genes from their last common ancestor (184, 185). This shared genetic inheritance comprises a large core of common “housekeeping” genes that are essential for survival and that dictate a raft of processes, including central metabolism, genome replication and expression, and the many commonalities that we see in the molecular and physiologic properties of plant and animal cells (186). This does raise the question as to the extent to which this common genetic inheritance may underlie the mammalian physiologic effects of polyphenols.
The notion that some form of cross-kingdom signaling underpins the effects of polyphenols has been advanced previously (161). For instance, Howitz and Sinclair (187) proposed the concept of “xenohormesis” whereby animals and fungi read the chemical evidence of environmental stress from the plants on which they feed in the form of stress-induced secondary metabolites, such as polyphenols, which then trigger a hormetic response in the consuming organism (hormesis being the process by which a small amount of a potential stressor, such as a toxin, physical exercise, or starvation, enhances functioning by triggering an adaptive stress response). A favored example here is the ability of several polyphenols to interact in vitro with the mammalian sirtuin genes [silent information regulator two protein (SIRT or SRT)] that regulate a number of transcription factors (e.g., p53, NF-κB, PPARγ, and PPARγ coactivator 1-α) that play key roles in stress responses, cellular differentiation, and metabolism (188). Sirtuins ultimately modulate a range of critical metabolic and physiologic processes, including cellular metabolism, survival and aging, stress resistance, inflammation, immune function, and endothelial function. It is notable that much of the huge interest in the polyphenol resveratrol was sparked by the observation that it extends the lifespan of yeast and animals, such as the fruit fly (Drosophila melanogaster) and roundworm (Caenorhabditis elegans), in a manner similar to caloric restriction, apparently via a hormetic response mediated via the activation of sirtuin genes (189, 190).
The notion of xenohormesis as an adaptive mechanism is intriguing, but as an explanatory tool of human/phytochemical interactions, it falls foul of the law of parsimony. It seems more likely that any beneficial effects are simply predicated on the similarities between signaling pathways in the taxa, with polyphenol/signaling interactions driven by the function the phytochemicals are trying to fulfill for their home plant, including their endogenous roles as signaling molecules within the plant itself. As described above, a wealth of evidence suggests that flavonoids owe their bioactivity in mammals to interactions with the transduction and signaling pathways that mediate cellular responses to stressors, such as the ubiquitous protein and lipid kinase signaling cascades (e.g., MAPK, PI3K/PKB, and TOR) (36, 112, 191). Many of the key components of cellular signaling are included in the common ancestral endowment of cellular housekeeping functions, and the same signaling cascades often play identical roles in mediating the cellular responses of plants and animals to stressors (192, 193). Plants have a particularly rich complement of >1000 kinase genes (194), and a striking 2.5% of the genome of the model plant Arabidopsis thaliana is given over solely to the ≥600 genes that express the kinase subfamily of receptor-like kinases. These receptors bridge the cellular membrane and transduce extracellular signals into cellular secondary messenger MAPK signaling cascades. Phylogenetic evidence shows that the entire kinase superfamily and the specific receptor-like kinases evolved in a unicellular ancestor of both plants and animals. Beyond this point, the majority of plant receptor-like kinases remained specific to serine/threonine, whereas the majority of animal receptors evolved specificity to tyrosine (i.e., receptor tyrosine kinases) (195). Similarly, animals and plants also share the MAPK enzyme signaling pathways. Indeed, these are highly conserved across all eukaryotes (192, 193), with plant MAPKs being most closely homologous to the ERK MAPK subfamily in mammals (196). In addition, plants also possess conserved PI3Ks and their interacting PKBs (197). The Arabidopsis genome also encodes 39 distinct, conserved “animal” AGC kinases (named after the member kinases: cAMP-dependent protein kinase A, cGMP-dependent protein kinase G, and phospholipid-dependent protein kinase C). These kinases modulate the activity of other intracellular second messengers, including cAMP, cGMP, and phospholipids, and make their own independent (to MAPK) contribution to stress signaling and engage in crosstalk interactions with the MAPK signaling pathways (194, 198).
This extended protein kinase family plays analogous cellular transduction and signaling roles in plants and animals. One particularly pertinent example is the TOR kinase signaling pathway, a crucial growth-regulating cellular system that simultaneously collects information on stressors, nutrients, and internal energy states from multiple extracellular and intracellular inputs, including from the PI3K/PKB pathways. In benign conditions of nutrient and energy availability, TOR upregulates a vast range of energy-consuming activities, such as metabolism, cell proliferation, and the translation of mRNA, ultimately dictating a wealth of developmental and metabolic processes by regulating other key kinases (199, 200). The TOR kinase pathways are, again, highly conserved, having originated before the last common eukaryotic ancestor, and both their function and core molecular components, including the upstream PI3K/PKB pathway, are shared across the cells of nearly all eukaryotic clades (201). For instance, in plants, TOR signaling pathways play a large part in regulating growth, with the TOR gene expressed most abundantly in rapidly growing and dividing tissues. As with mammalian cells, TOR also mediates plant cellular responses to stressors and directs autophagy, possibly via interactions with plant hormones, such as abscisic acid (202). This pathway comes sharply into focus for humans when we consider that, in mammals, TOR signaling is aberrantly upregulated in cancer (203) and dysregulated in obesity and diabetes, contributing to insulin resistance and cardiovascular disease. It also plays a key role in the process of cellular ageing (200). Conversely, TOR inhibitors may provide novel treatments for these same diseases, and it has previously been observed that the life-extending properties of caloric restriction are mediated by reduced TOR signaling (200).
In mammals, the functioning of the TOR signaling pathway is also modulated by a wide range of flavonoids and other polyphenols. For instance, ubiquitous catechins, such as EGCG, and flavonols, such as quercetin, attenuate cell proliferation and tumorogenesis by inhibiting both PI3K/PKB and TOR signaling in mammalian cells, potentially by binding to the ATP binding sites of the kinase proteins (204, 205). Similarly, quercetin, curcumin, and resveratrol were all shown to promote protective autophagy–mediated cell death in mammalian cancer cells via TOR inhibition (206). Resveratrol may also have its beneficial effects on cellular senescence, cell growth, glucose homeostasis, and cardiovascular function through its inhibition of TOR signaling via both sirtuin-dependent and -independent mechanisms. In the latter case, established mechanisms include modulation by resveratrol of PI3K/PKB signaling upstream of TOR and direct interactions with the TOR kinase itself (207, 208). Similarly, anthocyanin-rich berries protect the hippocampus in irradiation models of aging by inhibiting TOR and attenuating inflammation, oxidative stress, and a loss of protective autophagy (209, 210). Conversely, anthocyanin and flavanol supplementation in rodents activates TOR, promoting “growth” in terms of synaptic plasticity in cognition-relevant brain regions (112). This last finding suggests that flavonoids can exert biphasic, homeostatic effects within this single system.
Although the TOR and MAPK pathways and the other protein kinase pathways are heavily involved in directing gene transcription across taxa in response to a wide variety of stressors, the ultimate downstream products of this signaling effort differs between animals and plants. In animals, 1 consequence of activation will be the synthesis of COX and the PGs, whereas in the plant, the same signaling pathways will lead to the expression of lipoxygenase and the synthesis of jasmonates. In the case of plants, this, in turn, will ultimately lead to the synthesis of a raft of protective secondary metabolite chemicals (211, 212), with crosstalk with other hormones shaping the specific response via their own interactions with the plant kinome (196, 213, 214).
In general, across taxa, stress/immune signaling and gene transcription is a complex affair that is typified by an abundance of feedback and feedforward loops that closely regulate the expression of signaling molecules and transcription factors. As an example, in plants, the expression of salicylic acid genes is essential for many stress responses, but an overaccumulation of salicylic acid is toxic to the cell, so feedback and feedforward loops between the components of pathways responsible for synthesizing salicylates, other hormones, and cellular factors carefully regulate its synthesis (175). Polyphenols and other phenolics represent the ultimate downstream products of a multitude of plant hormonal defense communications. They also travel within the plant, penetrate cells and cell nuclei (101), and may exert a raft of independent signaling properties. Therefore, they represent ideal candidates to both feedback within their own cellular synthetic pathways and interact with the cellular signaling pathways in neighboring and distant cells.
An interesting analogy can be drawn with the “vitamins”, which are typically plant-derived primary metabolite chemicals that play broadly analogous essential cellular roles when consumed by humans. The synthesis of several of these compounds cannot only be induced by a variety of stressors in the plant, but they can also self-regulate their own synthesis, preventing wasteful or damaging overexpression via direct feedback interactions within their own biosynthetic pathways (215). The possibility that polyphenols, and other secondary metabolites, interact with their own synthetic pathways in this manner has simply not been investigated in plants to date. However, it is clearly a possibility that the modulation of animal stress signaling pathways by polyphenols may simply echo the role of the phytochemicals within the genetically conserved or analogous signaling pathways within their own plant. In this context, it would make absolute sense for the polyphenol products of stress signaling to provide 1 of the many endogenous signals that contribute in the plant to the activity of the global stress, nutrition, and energy-sensing TOR kinase pathways. This activity may then be transferred directly after consumption into modulation of the conserved PI3K/PKB and TOR pathways in animals. Similarly, viewed in the context of the conserved nature of the jasmonate and PG synthetic pathways, the inhibition of the enzyme COX2 in animal tissue by a wide variety of flavonoids (114) can also be interpreted as an attempt by the flavonoids to inhibit lipoxygenase enzymes within the orthologous plant jasmonate pathways. This interaction within the plant may be part of the complex crosstalk between the antagonistic jasmonate and salicylate hormone systems (as suggested with regards salicylic acid itself) (180) or it may simply be feedback or feedforward within and across the jasmonate system.
It is also notable that the net effect of flavonoids within several animal signaling pathways is modulation of the synthesis of NO (114). This ubiquitous signaling molecule, which represents a key downstream product of signaling cascades in both taxa, plays diverse modulatory roles in both the synthesis and function of jasmonates, salicylates, and ethylene (216, 217). Therefore, it is intrinsically tied to secondary metabolite synthesis pathways in plants. Any effects on NO synthesis in animal tissue may well also represent an attempt by the phytochemicals to interact with the various pathways dictating NO synthesis in their home plant.
Returning briefly to the yeast/animal sirtuin genes that fostered so much interest in the polyphenol resveratrol, it is also the case that, whereas mammals express 7 distinct, structurally similar sirtuin genes (SIRT1 to SIRT7), of which SIRT1 is the best characterized, plants also possess 2 conserved sirtuin genes that are homologs of the human equivalents (188). These are expressed in the reproductive tissue (SRT1) and throughout plant tissue (SRT2), respectively (218). The specific roles of these sirtuins in plants are poorly understood, but evidence does suggest that they play roles in plant development via hormonal signaling and in the hypersensitive immune response (218). Intriguingly, the SRT2 plant homolog was shown to suppress salicylic acid–dependent defense against pathogens, suggesting that it may play a role in the mutually inhibitory crosstalk between salicylic acid and the jasmonates (219). Once again, this would seem to suggest that any effects of polyphenols on sirtuin-mediated stress responses in animals may simply reflect an echo of polyphenol/sirtuin interactions in the plant.
Finally, just to confirm that polyphenols do exert direct cross-kingdom signaling effects as a consequence of the role they are trying to play in their home plant, it is interesting to note that 1 of the few plant signaling roles of flavonoids that has attracted any research attention is their modulation of transmembrane auxin movement. Flavonoids accomplish this by binding to multidrug resistance permeability glycoproteins in cell membranes, with the consequence that they inhibit auxin efflux from the cell. The same transporters are genetically conserved in plants and animals, and they form the established target underlying the potential medicinal utility of flavonoids in terms of reducing drug efflux in mammalian multidrug resistant cells (162, 220).
Is the modulation of brain function by polyphenols a consequence of intentional cross-kingdom signaling?
Although the examples immediately above suppose that any effects of polyphenols in mammals are simply a consequence of the close similarities in plant and animal signal transduction pathways, a second class of interactions would seem to be predicated on intentional cross-kingdom signaling by plants, albeit, in this case, that the consuming animals are not the intended target.
The phytoestrogens.
The most obvious example here is the benign part that flavonoids play in the evolutionarily ancient symbiotic relations of the plant with both the bacteria and fungi that colonize the rhizosphere, facilitating the uptake of nitrogen, water, nutrients, and minerals. The most ancient of these relations, that between plants and arbuscular mycorrhizal fungi, is ubiquitous across all plant lineages, whereas the more specialized relations between nitrogen fixing rhizobial bacteria and plants arose at a later date. Both relations work via the same mechanisms. Flavonoids released into the rhizosphere are detected by microbial receptors, such as the bacterial nodulation D protein. The flavonoids either attract bacteria by chemotaxis or stimulate the germination of fungal spores in the soil. The plant then perceives the presence of the symbiotic fungus or bacteria through the chemical emission by the microbe of lipochitooligosaccharide nod or myc (standing for nodulation and mycorrhizal, respectively) factors that bind to and activate lysine motif receptor kinases in the plant (221). Several iterations of this autoregulatory feedback ultimately govern the population level of microbes and the amounts of hyphal branching within the root system. In the legume (Fabaceae) family, this process also results in the creation of specialized root nodules designed specifically to accommodate the bacterial colony (148, 157). In a similar manner, a number of flavonoids are synthesized in response to the bacterial “quorum-sensing” signaling molecules that direct the population size and activity of bacteria in the rhizosphere. These flavonoids bind to bacterial receptors in a positive feedback mechanism that directs both bacterial behavior and flavonoid synthesis (159).
The most salient point here is that both the bacterial and fungal receptors involved in these processes are estrogen-like receptors that bear a striking similarity to mammalian ERα and ERβ in terms of the palette of chemicals and ligands that they recognize and to which they respond. They also exhibit the same ligand concentration-dependent activity, they co-occur with chaperone proteins in their unactivated state, and the nature of their gene transcription effects are similar (222, 223). These microbial receptors also bind mammalian estrogens, such as 17β-estradiol (223). Indeed, it has been suggested that many of the proteins within the respective plant and microbe signaling pathways are orthologous (224) and that the bacterial estrogen-like receptor, the nodulation D protein, is a partial ortholog of the mammalian ER (225) (although this is disputed in reference 226). Certainly, the key structural elements that dictate the binding of phytoestrogens to mammalian ERs, for instance, the aromatic ring with a hydroxyl group corresponding to the C3 position in the 17β-estradiol ring system (61, 223), also dictate the binding of the same compounds to microbial estrogen-like receptors (223). It is also notable that all of the key flavonoids that exert estrogenic effects in humans also take part in the rhizosphere plant/microbe estrogen-like receptor interactions described above (222, 223, 227). Therefore, the estrogenic effects of these phytoestrogens are predicated on an unintended transfer of the intended cross-kingdom plant/microbe signaling, predicated on the similarity in the cellular signal transduction equipment possessed by microbes and mammals.
Defense against microbial pathogens.
Of course, the ability to disrupt quorum-sensing and estrogen-like receptor signaling in pathogenic microbes may be a useful defense mechanism (227), and it is notable that several antimicrobial phytoalexins with more restricted distributions, such as kievitone, phaseollin, and resveratrol, also have estrogenic properties (228). However, the TOR pathways may provide a better example of intentional cross-kingdom signaling in the defensive disruption of microbial signaling. The TOR kinase signaling pathway itself was originally identified in the 1990s when researchers were trying to discover the mechanism of action of a potent antifungal chemical produced by bacteria found 20 years previously in a soil sample from Easter Island (also known as Rapa Nui). They named the antifungal macrolide molecule rapamycin, and after many years of research, identified the targets of rapamycin as novel protein kinases that they named “target of rapamycin”, as well as a specific binding protein (FK binding protein 12) within the TOR pathway (229). To the fungus, the inhibition by rapamycin of the TOR pathway signals a lack of environmental nutrients and prevents protein synthesis and cellular proliferation, thereby inhibiting growth and handing an advantage to the rapamycin-synthesizing bacteria.
It also transpired that all of the components of the TOR pathway targeted by rapamycin are conserved in fungi, animals, and plants (201, 230). If we accept that the bacteria evolved the synthesis of rapamycin as an antifungal strategy, then any effects of rapamycin in mammals would represent a cross-kingdom transfer of the intended cross-kingdom inhibition of TOR signaling in fungi. Rapamycin certainly has numerous useful medicinal applications for humans, all of which are predicated on TOR pathway inhibition. It started life in the late 1990s as an antifungal treatment, but it soon became apparent that its major use was as an immune suppressant and antiproliferative that could be used to prevent organ rejection after transplant surgery.
Recent research has also demonstrated that rapamycin has a number of properties in common with TOR-inhibiting polyphenols, such as EGCG, quercetin, and resveratrol, including the ability to increase longevity and protect against cancer in mammals (231). Conversely, the synthesis of the same polyphenols is upregulated by biotic stressors, including fungi, and they all exhibit antifungal properties (232, 233). Unfortunately, as yet, we do not know whether these antifungal properties are predicated on interference with TOR signaling. However, this does raise the possibility that the cross-kingdom kinase signaling roles of many polyphenols may simply reflect an unintended transfer of their intended role as antimicrobials, in the same manner as rapamycin, due to the conserved nature of the TOR pathways.
Conclusions
The foregoing presents a prima facie case that the modulatory effects of polyphenols within human cellular signal transduction pathways, and therefore their beneficial effects on cardiovascular and brain function, are predicated on a cross-kingdom transfer of the signaling role that these phytochemicals play, either endogenously within their own plant or exogenously as they perform ecologic roles related to the management by the plant of its symbiotic and pathogenic microbial neighbors.
Because of a vast research effort over the past decades, we now know a great deal about the interactions of polyphenols within mammalian signal transduction pathways. In contrast we know very little about their interactions within the orthologous plant (and to a lesser extent microbe) signaling pathways. But why is this important? Elucidating the ecologic and endogenous signaling roles of phytochemicals is obviously of interest from a plant science perspective, but beyond that a fuller understanding of these endogenous signaling roles must also inform the analogous mammalian research. Refocusing just a small fraction of the huge research effort directed at polyphenol/mammal interactions toward the analogous systems in plants may well offer an exponential increase in our understanding of the potential of these phytochemicals regarding human health. Added to this, the plant research would be substantially cheaper and, in some cases, less ethically contentious.
It is also notable that the physiologic effects and mechanisms of action of the many metabolites created by the gut microbiota and phase I/II metabolism of polyphenols have been largely overlooked in favor of concentrating on the parent compounds. Given that many of these metabolites, including a number of ubiquitous simple phenolic acids, are found naturally in plants, it seems reasonable to suggest that plant models might make a reasonable starting point for rectifying the imbalance in this literature.
Acknowledgments
Figures 1 and 2 and the broad arguments presented herein were originally published in the book Plants and the Human Brain by Dr. Kennedy and have been reproduced with permission from Oxford University Press. The sole author had responsibility for all parts of the manuscript.
References
- 1.Dewick PM. Medicinal natural products: a biosynthetic approach. Chichester, UK: John Wiley and Sons; 2009 [Google Scholar]
- 2.Chun OK, Chung SJ, Song WO. Estimated dietary flavonoid intake and major food sources of us adults. J Nutr 2007;137:1244–52 [DOI] [PubMed] [Google Scholar]
- 3.Zamora-Ros R, Andres-Lacueva C, Lamuela-Raventós RM, Berenguer T, Jakszyn P, Barricarte A, Ardanaz E, Amiano P, Dorronsoro M, Larrañga N, Martínez C, Sánchez MJ, Navarro C, Chirlaque MD, Tormo MJ, Quirós JR, González CA. Estimation of dietary sources and flavonoid intake in a spanish adult population (EPIC-spain). J Am Diet Assoc 2010;110:390–8 [DOI] [PubMed] [Google Scholar]
- 4.Johannot L, Somerset SM. Age-related variations in flavonoid intake and sources in the australian population. Public Health Nutr 2006;9:1045–54 [DOI] [PubMed] [Google Scholar]
- 5.Ovaskainen M-L, Torronen R, Koponen JM, Sinkko H, Hellstrom J, Reinivuo H, Mattila P. Dietary intake and major food sources of polyphenols in Finnish adults. J Nutr 2008;138:562–6 [DOI] [PubMed] [Google Scholar]
- 6.Pérez-Jiménez J, Fezeu L, Touvier M, Arnault N, Manach C, Hercberg S, Galan P, Scalbert A. Dietary intake of 337 polyphenols in French adults. Am J Clin Nutr 2011;93:1220–8 [DOI] [PubMed] [Google Scholar]
- 7.Djoussé L, Hopkins PN, North KE, Pankow JS, Arnett DK, Ellison RC. Chocolate consumption is inversely associated with prevalent coronary heart disease: The National Heart, Lung, and Blood Institute Family Heart Study. Clin Nutr 2011;30:182–7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Deka A, Vita JA. Tea and cardiovascular disease. Pharmacol Res 2011;64:136–45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bauer SR, Ding EL, Smit LA. Cocoa consumption, cocoa flavonoids, and effects on cardiovascular risk factors: an evidence-based review. Curr Cardiovasc Risk Rep 2011;5:120–7 [Google Scholar]
- 10.McCullough ML, Peterson JJ, Patel R, Jacques PF, Shah R, Dwyer JT. Flavonoid intake and cardiovascular disease mortality in a prospective cohort of us adults. Am J Clin Nutr 2012;95:454–64 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Medina-Remón A, Zamora-Ros R, Rotches-Ribalta M, Andres-Lacueva C, Martinez-Gonzalez MA, Covas MI, Corella D, Salas-Salvado J, Gomez-Gracia E, Ruiz-Gutierrez V, et al. Total polyphenol excretion and blood pressure in subjects at high cardiovascular risk. Nutr Metab Cardiovasc Dis 2011;21:323–31 [DOI] [PubMed] [Google Scholar]
- 12.Kay CD, Hooper L, Kroon PA, Rimm EB, Cassidy A. Relative impact of flavonoid composition, dose and structure on vascular function: a systematic review of randomised controlled trials of flavonoid-rich food products. Mol Nutr Food Res 2012;56:1605–16. [DOI] [PubMed] [Google Scholar]
- 13.Monagas M, Khan N, Andres-Lacueva C, Casas R, Urpi-Sarda M, Llorach R, Lamuela-Raventos RM, Estruch R. Effect of cocoa powder on the modulation of inflammatory biomarkers in patients at high risk of cardiovascular disease. Am J Clin Nutr 2009;90:1144–50 [DOI] [PubMed] [Google Scholar]
- 14.Shrime MG, Bauer SR, McDonald AC, Chowdhury NH, Coltart CEM, Ding EL. Flavonoid-rich cocoa consumption affects multiple cardiovascular risk factors in a meta-analysis of short-term studies. J Nutr 2011;141:1982–8 [DOI] [PubMed] [Google Scholar]
- 15.Hooper L, Kay C, Abdelhamid A, Kroon PA, Cohn JS, Rimm EB, Cassidy A. Effects of chocolate, cocoa, and flavan-3-ols on cardiovascular health: a systematic review and meta-analysis of randomized trials. Am J Clin Nutr 2012;95:740–51 [DOI] [PubMed] [Google Scholar]
- 16.Knopman D, Boland L, Mosley T, Howard G, Liao D, Szklo M, McGovern P, Folsom A. Cardiovascular risk factors and cognitive decline in middle-aged adults. Neurology 2001;56:42–8 [DOI] [PubMed] [Google Scholar]
- 17.Barnes DE, Alexopoulos GS, Lopez OL, Williamson JD, Yaffe K. Depressive symptoms, vascular disease, and mild cognitive impairment: findings from the Cardiovascular Health Study. Arch Gen Psychiatry 2006;63:273–9. [DOI] [PubMed] [Google Scholar]
- 18.Cukierman-Yaffe T, Gerstein HC, Williamson JD, Lazar RM, Lovato L, Miller ME, Coker LH, Murray A, Sullivan MD, Marcovina SM. Relationship between baseline glycemic control and cognitive function in individuals with type 2 diabetes and other cardiovascular risk factors the action to control cardiovascular risk in diabetes-memory in diabetes (ACCORD-MIND) trial. Diabetes Care 2009;32:221–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bruehl H, Sweat V, Hassenstab J, Polyakov V, Convit A. Cognitive impairment in nondiabetic middle-aged and older adults is associated with insulin resistance. J Clin Exp Neuropsychol 2010;32:487–93 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pearson S, Schmidt M, Patton G, Dwyer T, Blizzard L, Otahal P, Venn A. Depression and insulin resistance cross-sectional associations in young adults. Diabetes Care 2010;33:1128–33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hollman PCH, Geelen A, Kromhout D. Dietary flavonol intake may lower stroke risk in men and women. J Nutr 2010;140:600–4 [DOI] [PubMed] [Google Scholar]
- 22.Kuriyama S, Hozawa A, Ohmori K, Shimazu T, Matsui T, Ebihara S, Awata S, Nagatomi R, Arai H, Tsuji I. Green tea consumption and cognitive function: a cross-sectional study from the tsurugaya project. Am J Clin Nutr 2006;83:355–61 [DOI] [PubMed] [Google Scholar]
- 23.Commenges D, Scotet V, Renaud S, Jacqmin-Gadda H, Barberger-Gateau P, Dartigues JF. Intake of flavonoids and risk of dementia. Eur J Epidemiol 2000;16:357–63 [DOI] [PubMed] [Google Scholar]
- 24.Ng TP, Feng L, Niti M, Kua EH, Yap KB. Tea consumption and cognitive impairment and decline in older chinese adults. Am J Clin Nutr 2008;88:224–31 [DOI] [PubMed] [Google Scholar]
- 25.Barberger-Gateau P, Raffaitin C, Letenneur L, Berr C, Tzourio C, Dartigues JF, Alperovitch A. Dietary patterns and risk of dementia: the Three-City cohort study. Neurology 2007;69:1921–30 [DOI] [PubMed] [Google Scholar]
- 26.Dai Q, Borenstein AR, Wu YG, Jackson JC, Larson EB. Fruit and vegetable juices and Alzheimer’s disease: the Kame Project. Am J Med 2006;119:751–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Letenneur L, Proust-Lima C, Le Gouge A, Dartigues JF, Barberger-Gateau P. Flavonoid intake and cognitive decline over a 10-year period. Am J Epidemiol 2007;165:1364–71 [DOI] [PubMed] [Google Scholar]
- 28.Arab L, Liu W, Elashoff D. Green and black tea consumption and risk of stroke a meta-analysis. Stroke 2009;40:1786–92 [DOI] [PubMed] [Google Scholar]
- 29.Devore EE, Kang JH, Breteler M, Grodstein F. Dietary intakes of berries and flavonoids in relation to cognitive decline. Ann Neurol 2012;72:135–43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kesse-Guyot E, Fezeu L, Andreeva VA, Touvier M, Scalbert A, Hercberg S, Galan P. Total and specific polyphenol intakes in midlife are associated with cognitive function measured 13 years later. J Nutr 2012;142:76–83 [DOI] [PubMed] [Google Scholar]
- 31.Nurk E, Refsum H, Drevon CA, Tell GS, Nygaard HA, Engedal K, Smith AD. Intake of flavonoid-rich wine, tea, and chocolate by elderly men and women is associated with better cognitive test performance. J Nutr 2009;139:120–7 [DOI] [PubMed] [Google Scholar]
- 32.Miller MG, Shukitt-Hale B. Berry fruit enhances beneficial signaling in the brain. J Agric Food Chem 2012;60:5709–15 [DOI] [PubMed] [Google Scholar]
- 33.Spencer JP. The impact of fruit flavonoids on memory and cognition. Br J Nutr 2010;104:S40–7 [DOI] [PubMed] [Google Scholar]
- 34.Smoliga JM, Baur JA, Hausenblas HA. Resveratrol and health – a comprehensive review of human clinical trials. Mol Nutr Food Res 2011;55:1129–41 [DOI] [PubMed] [Google Scholar]
- 35.Smith JM, Stouffer EM. Concord grape juice reverses the age-related impairment in latent learning in rats. Nutr Neurosci 2014;17(2):81–87 [DOI] [PubMed] [Google Scholar]
- 36.Williams RJ, Spencer JPE. Flavonoids, cognition, and dementia: actions, mechanisms, and potential therapeutic utility for alzheimer disease. Free Radic Biol Med 2012;52:35–45 [DOI] [PubMed] [Google Scholar]
- 37.Stangl D, Thuret S. doi: 10.1007/s12263-009-0134-5. Impact of diet on adult hippocampal neurogenesis. Genes Nutr 2009;4:271–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sorond FA, Lipsitz LA, Hollenberg NK, Fisher ND. Cerebral blood flow response to flavanol-rich cocoa in healthy elderly humans. Neuropsychiatr Dis Treat 2008;4:433. [PMC free article] [PubMed] [Google Scholar]
- 39.Francis ST, Head K, Morris P, Macdonald I. The effect of flavanol-rich cocoa on the fmri response to a cognitive task in healthy young people. J Cardiovasc Pharmacol 2006;47:S215–20 [DOI] [PubMed] [Google Scholar]
- 40.Scholey A, Downey LA, Ciorciari J, Pipingas A, Nolidin K, Finn M, Wines M, Catchlove S, Terrens A, Barlow E, et al. Acute neurocognitive effects of epigallocatechin gallate (egcg). Appetite 2012;58:767–70 [DOI] [PubMed] [Google Scholar]
- 41.Field DT, Williams CM, Butler LT. Consumption of cocoa flavanols results in an acute improvement in visual and cognitive functions. Physiol Behav 2011;103:255–60 [DOI] [PubMed] [Google Scholar]
- 42.Desideri G, Kwik-Uribe C, Grassi D, Necozione S, Ghiadoni L, Mastroiacovo D, Raffaele A, Ferri L, Bocale R, Lechiara MC. Benefits in cognitive function, blood pressure, and insulin resistance through cocoa flavanol consumption in elderly subjects with mild cognitive impairmentnovelty and significance the cocoa, cognition, and aging (cocoa) study. Hypertension 2012;60:794–801 [DOI] [PubMed] [Google Scholar]
- 43.Crews WD, Harrison DW, Wright JW. A double-blind, placebo-controlled, randomized trial of the effects of dark chocolate and cocoa on variables associated with neuropsychological functioning and cardiovascular health: clinical findings from a sample of healthy, cognitively intact older adults. Am J Clin Nutr 2008;87:872–80 [DOI] [PubMed] [Google Scholar]
- 44.Camfield DA, Scholey A, Pipingas A, Silberstein R, Kras M, Nolidin K, Wesnes K, Pase M, Stough C. Steady state visually evoked potential (ssvep) topography changes associated with cocoa flavanol consumption. Physiol Behav 2012;105:948–57 [DOI] [PubMed] [Google Scholar]
- 45.Pase MP, Scholey AB, Pipingas A, Kras M, Nolidin K, Gibbs A, Wesnes K, Stough C. Cocoa polyphenols enhance positive mood states but not cognitive performance: a randomized, placebo-controlled trial. J Psychopharmacol 2013;27:451–8 [DOI] [PubMed] [Google Scholar]
- 46.Wightman EL, Haskell CF, Forster JS, Veasey RC, Kennedy DO. Epigallocatechin gallate, cerebral blood flow parameters, cognitive performance and mood in healthy humans: a double-blind, placebo-controlled, crossover investigation. Hum Psychopharmacol 2012;27:177–86 [DOI] [PubMed] [Google Scholar]
- 47.Borgwardt S, Hammann F, Scheffler K, Kreuter M, Drewe J, Beglinger C. Neural effects of green tea extract on dorsolateral prefrontal cortex. Eur J Clin Nutr 2012;66:1187–92 [DOI] [PubMed] [Google Scholar]
- 48.Park SK, Jung IC, Lee WK, Lee YS, Park HK, Go HJ, Kim K, Lim NK, Hong JT, Ly SY, et al. A combination of green tea extract and l-theanine improves memory and attention in subjects with mild cognitive impairment: a double-blind placebo-controlled study. J Med Food 2011;14:334–43 [DOI] [PubMed] [Google Scholar]
- 49.Kohama T, Negami M. Effect of low-dose french maritime pine bark extract on climacteric syndrome in 170 perimenopausal women a randomized, double-blind, placebo-controlled trial. J Reprod Med 2013;58:39–46 [PubMed] [Google Scholar]
- 50.Yang HM, Liao MF, Zhu SY, Liao MN, Rohdewald P. A randomised, double-blind, placebo-controlled trial on the effect of pycnogenol® on the climacteric syndrome in peri-menopausal women. Acta Obstet Gynecol Scand 2007;86:978–85 [DOI] [PubMed] [Google Scholar]
- 51.Ryan J, Croft K, Mori T, Wesnes K, Spong J, Downey L, Kure C, Lloyd J, Stough C. An examination of the effects of the antioxidant pycnogenol® on cognitive performance, serum lipid profile, endocrinological and oxidative stress biomarkers in an elderly population. J Psychopharmacol 2008;22:553–62 [DOI] [PubMed] [Google Scholar]
- 52.Pipingas A, Silberstein RB, Vitetta L, Rooy CV, Harris EV, Young JM, Frampton CM, Sali A, Nastasi J. Improved cognitive performance after dietary supplementation with a pinus radiata bark extract formulation. Phytother Res 2008;22:1168–74 [DOI] [PubMed] [Google Scholar]
- 53.Theadom A, Mahon S, Barker-Collo S, McPherson K, Rush E, Vandal AC, Feigin VL. doi: 10.1111/ene.12099. Enzogenol for cognitive functioning in traumatic brain injury: a pilot placebo-controlled rct. Eur J Neurol 2013:20:1135–44. [DOI] [PubMed] [Google Scholar]
- 54.Manach C, Williamson G, Morand C, Scalbert A, Rémésy C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am J Clin Nutr 2005;81:230S–42S [DOI] [PubMed] [Google Scholar]
- 55.Shukitt-Hale B, Lau FC, Joseph JA. Berry fruit supplementation and the aging brain. J Agric Food Chem 2008;56:636–41 [DOI] [PubMed] [Google Scholar]
- 56.Krikorian R, Nash TA, Shidler MD, Shukitt-Hale B, Joseph JA. Concord grape juice supplementation improves memory function in older adults with mild cognitive impairment. Br J Nutr 2010;103:730. [DOI] [PubMed] [Google Scholar]
- 57.Krikorian R, Shidler MD, Nash TA, Kalt W, Vinqvist-Tymchuk MR, Shukitt-Hale B, Joseph JA. Blueberry supplementation improves memory in older adults. J Agric Food Chem 2010;58:3996–4000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Krikorian R, Boespflug EL, Fleck DE, Stein AL, Wightman JD, Shidler MD, Sadat-Hossieny S. Concord grape juice supplementation and neurocognitive function in human aging. J Agric Food Chem 2012;60:5736–42 [DOI] [PubMed] [Google Scholar]
- 59. Hendrickson SJ, Mattes RD. No acute effects of grape juice on appetite, implicit memory and mood. Food Nutr Res 2008;52. [DOI] [PMC free article] [PubMed]
- 60.Rochester JR, Millam JR. Phytoestrogens and avian reproduction: exploring the evolution and function of phytoestrogens and possible role of plant compounds in the breeding ecology of wild birds. Comp Biochem Physiol A Mol Integr Physiol 2009;154:279–88 [DOI] [PubMed] [Google Scholar]
- 61.Fang H, Tong W, Shi LM, Blair R, Perkins R, Branham W, Hass BS, Xie Q, Stacy L, Moland CL. Structure-activity relationships for a large diverse set of natural, synthetic, and environmental estrogens. Chem Res Toxicol 2001;14:280–94 [DOI] [PubMed] [Google Scholar]
- 62.Collins-Burow BM, Burow ME, Duong BN, McLachlan JA. Estrogenic and antiestrogenic activities of flavonoidphytochemicals through estrogen receptor binding-dependent and-independent mechanisms. Nutr Cancer 2000;38:229–44 [DOI] [PubMed] [Google Scholar]
- 63.Collins-Burow BM, Antoon JW, Frigo DE, Elliott S, Weldon CB, Boue SM, Beckman BS, Curiel TJ, Alam J, McLachlan JA, et al. Antiestrogenic activity of flavonoid phytochemicals mediated via the c-jun n-terminal protein kinase pathway. Cell-type specific regulation of estrogen receptor alpha. J Steroid Biochem Mol Biol 2012;132:186–93 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Shor D, Sathyapalan T, Atkin SL, Thatcher NJ. Does equol production determine soy endocrine effects? Eur J Nutr 2012;51:389–98 [DOI] [PubMed] [Google Scholar]
- 65.Barnes S. The biochemistry, chemistry and physiology of the isoflavones in soybeans and their food products. Lymphat Res Biol 2010;8:89–98 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Guha N, Kwan ML, Quesenberry CP, Jr, Weltzien EK, Castillo AL, Caan BJ. Soy isoflavones and risk of cancer recurrence in a cohort of breast cancer survivors: the life after cancer epidemiology study. Breast Cancer Res Treat 2009;118:395–405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Anderson LN, Cotterchio M, Boucher BA, Kreiger N. Phytoestrogen intake from foods, during adolescence and adulthood, and risk of breast cancer by estrogen and progesterone receptor tumor subgroup among ontario women. Int J Cancer 2013;132:1683–92 [DOI] [PubMed] [Google Scholar]
- 68.Boucher BA, Cotterchio M, Anderson LN, Kreiger N, Kirsh VA, Thompson LU. Use of isoflavone supplements is associated with reduced postmenopausal breast cancer risk. Int J Cancer 2013;132:1439–50 [DOI] [PubMed] [Google Scholar]
- 69.Dong JY, Qin LQ. Soy isoflavones consumption and risk of breast cancer incidence or recurrence: a meta-analysis of prospective studies. Breast Cancer Res Treat 2011;125:315–23 [DOI] [PubMed] [Google Scholar]
- 70.Zhang YB, Chen WH, Guo JJ, Fu ZH, Yi C, Zhang M, Na XL. Soy isoflavone supplementation could reduce body weight and improve glucose metabolism in non-asian postmenopausal women-a meta-analysis. Nutrition 2013;29:8–14 [DOI] [PubMed] [Google Scholar]
- 71.Taku K, Melby MK, Kronenberg F, Kurzer MS, Messina M. Extracted or synthesized soybean isoflavones reduce menopausal hot flash frequency and severity: systematic review and meta-analysis of randomized controlled trials. Menopause 2012;19:776–90 [DOI] [PubMed] [Google Scholar]
- 72.Beavers DP, Beavers KM, Miller M, Stamey J, Messina MJ. Exposure to isoflavone-containing soy products and endothelial function: a bayesian meta-analysis of randomized controlled trials. Nutr Metab Cardiovasc Dis 2012;22:182–91 [DOI] [PubMed] [Google Scholar]
- 73.Taku K, Lin N, Cai DL, Hu JW, Zhao XH, Zhang YM, Wang PY, Melby MK, Hooper L, Kurzer MS, et al. Effects of soy isoflavone extract supplements on blood pressure in adult humans: systematic review and meta-analysis of randomized placebo-controlled trials. J Hypertens 2010;28:1971–82 [DOI] [PubMed] [Google Scholar]
- 74.Sumien N, Chaudhari K, Sidhu A, Forster MJ. Does phytoestrogen supplementation affect cognition differentially in males and females? Brain Res 2013;1514:123–7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sarkaki A, Badavi M, Aligholi H, Moghaddam AZ. Preventive effects of soy meal (+/− isoflavone) on spatial cognitive deficiency and body weight in an ovariectomized animal model of Parkinson’s disease. Pak J Biol Sci 2009;12:1338–45 [DOI] [PubMed] [Google Scholar]
- 76.Neese SL, Wang V, Doerge D, Woodling K, Andrade J, Helferich W, Korol D. Impact of dietary genistein and aging on executive function in rats. Neurotoxicol Teratol 2010;32:200–11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Lee YB, Lee HJ, Won MH, Hwang IK, Kang TC, Lee JY, Nam SY, Kim KS, Kim E, Cheon SH, et al. Soy isoflavones improve spatial delayed matching-to-place performance and reduce cholinergic neuron loss in elderly male rats. J Nutr 2004;134:1827–31 [DOI] [PubMed] [Google Scholar]
- 78.Bryant M, Cassidy A, Hill C, Powell J, Talbot D, Dye L. Effect of consumption of soy isoflavones on behavioural, somatic and affective symptoms in women with premenstrual syndrome. Br J Nutr 2005;93:731–9 [DOI] [PubMed] [Google Scholar]
- 79.Basaria S, Wisniewski A, Dupree K, Bruno T, Song MY, Yao F, Ojumu A, John M, Dobs AS. Effect of high-dose isoflavones on cognition, quality of life, androgens, and lipoprotein in post-menopausal women. J Endocrinol Invest 2009;32:150–5 [DOI] [PubMed] [Google Scholar]
- 80.Hooper L, Ryder JJ, Kurzer MS, Lampe JW, Messina MJ, Phipps WR, Cassidy A. Effects of soy protein and isoflavones on circulating hormone concentrations in pre- and post-menopausal women: a systematic review and meta-analysis. Hum Reprod Update 2009;15:423–40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lamport DJ, Dye L, Wightman JD, Lawton CL. The effects of flavonoid and other polyphenol consumption on cognitive performance: a systematic research review of human experimental and epidemiological studies. Nutr Aging 2012;1:5–25 [Google Scholar]
- 82.Greendale GA, Huang MH, Leung K, Crawford SL, Gold EB, Wight R, Waetjen E, Karlamangla AS. Dietary phytoestrogen intakes and cognitive function during the menopausal transition: Results from the study of women’s health across the nation phytoestrogen study. Menopause 2012;19:894–903 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Hamilton RT, Rettberg JR, Mao Z, To J, Zhao L, Appt SE, Register TC, Kaplan JR, Brinton RD. Hippocampal responsiveness to 17β-estradiol and equol after long-term ovariectomy: implication for a therapeutic window of opportunity. Brain Res 2011;1379:11–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Thorp AA, Sinn N, Buckley J, Coates A, Howe P. Soya isoflavone supplementation enhances spatial working memory in men. Br J Nutr 2009;102:1348–54 [DOI] [PubMed] [Google Scholar]
- 85.Baur JA, Pearson KJ, Price NL, Jamieson HA, Lerin C, Kalra A, Prabhu VV, Allard JS, Lopez-Lluch G, Lewis K, et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature 2006;444:337–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Udenigwe CC, Ramprasath VR, Aluko RE, Jones PJH. Potential of resveratrol in anticancer and anti-inflammatory therapy. Nutr Rev 2008;66:445–54 [DOI] [PubMed] [Google Scholar]
- 87.Nijveldt RJ, van Nood E, van Hoorn DEC, Boelens PG, van Norren K, van Leeuwen PAM. Flavonoids: a review of probable mechanisms of action and potential applications. Am J Clin Nutr 2001;74:418–25 [DOI] [PubMed] [Google Scholar]
- 88.Soleas GJ, Diamandis EP, Goldberg DM. Resveratrol: a molecule whose time has come? And gone? Clin Biochem 1997;30:91–113 [DOI] [PubMed] [Google Scholar]
- 89.Saiko P, Szakmary A, Jaeger W, Szekeres T. Resveratrol and its analogs: defense against cancer, coronary disease and neurodegenerative maladies or just a fad? Mutat Res 2008;658:68–94 [DOI] [PubMed] [Google Scholar]
- 90.Fan E, Zhang LJ, Jiang S, Bai YH. Beneficial effects of resveratrol on atherosclerosis. J Med Food 2008;11:610–4 [DOI] [PubMed] [Google Scholar]
- 91.Ates O, Cayli S, Altinoz E, Gurses I, Yucel N, Sener M, Kocak A, Yologlu S. Neuroprotection by resveratrol against traumatic brain injury in rats. Mol Cell Biochem 2007;294:137–44 [DOI] [PubMed] [Google Scholar]
- 92.Jin F, Wu Q, Lu YF, Gong QH, Shi JS. Neuroprotective effect of resveratrol on 6-ohda-induced Parkinson’s disease in rats. Eur J Pharmacol 2008;600:78–82 [DOI] [PubMed] [Google Scholar]
- 93.Sharma M, Gupta YK. Chronic treatment with trans rosveratrol prevents intracerebroventricular streptozotocin induced cognitive impairment and oxidative stress in rats. Life Sci 2002;71:2489–98 [DOI] [PubMed] [Google Scholar]
- 94.Sönmez U, Sonmez A, Erbil G, Tekmen I, Baykara B. Neuroprotective effects of resveratrol against traumatic brain injury in immature rats. Neurosci Lett 2007;420:133–7 [DOI] [PubMed] [Google Scholar]
- 95.Wong RHX, Howe PRC, Buckley JD, Coates AM, Kunz I, Berry NM. Acute resveratrol supplementation improves flow-mediated dilatation in overweight/obese individuals with mildly elevated blood pressure. Nutr Metab Cardiovasc Dis 2011;21:851–6 [DOI] [PubMed] [Google Scholar]
- 96.Wong RH, Berry NM, Coates AM, Buckley JD, Bryan J, Kunz I, Howe PR. Chronic resveratrol consumption improves brachial flow-mediated dilatation in healthy obese adults. J Hypertens 2013;31:1819–27 [DOI] [PubMed] [Google Scholar]
- 97.Kennedy DO, Wightman EL, Reay JL, Lietz G, Okello EJ, Wilde A, Haskell CF. Effects of resveratrol on cerebral blood flow variables and cognitive performance in humans: a double-blind, placebo-controlled, crossover investigation. Am J Clin Nutr 2010;91:1590–7 [DOI] [PubMed] [Google Scholar]
- 98.Wightman EL, Reay J, Haskell CF, Williamson G, Dew TP, Kennedy DO. Effects of resveratrol alone or in combination with piperine on cerebral blood flow parameters and cognitive performance in humans: a randomised, double blind, placebo controlled cross-over investigation. Br J Nutr 2014;112:203–13 [DOI] [PubMed] [Google Scholar]
- 99.Kapetanovic IM, Muzzio M, Huang Z, Thompson TN, McCormick DL. Pharmacokinetics, oral bioavailability, and metabolic profile of resveratrol and its dimethylether analog, pterostilbene, in rats. Cancer Chemother Pharmacol 2011;68:593–601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Chang J, Rimando A, Pallas M, Camins A, Porquet D, Reeves J, Shukitt-Hale B, Smith MA, Joseph JA, Casadesus G. Low-dose pterostilbene, but not resveratrol, is a potent neuromodulator in aging and Alzheimer’s disease. Neurobiol Aging 2012;33:2062–71 [DOI] [PubMed] [Google Scholar]
- 101.Nutakul W, Sobers HS, Qiu P, Dong P, Decker EA, McClements DJ, Xiao H. Inhibitory effects of resveratrol and pterostilbene on human colon cancer cells: a side-by-side comparison. J Agric Food Chem 2011;59:10964–70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Ammon HP, Wahl M. Pharmacology of curcuma longa. Planta Med 1991;57:1–7 [DOI] [PubMed] [Google Scholar]
- 103.Gupta SC, Patchva S, Aggarwal BB. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J 2013;15:195–218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Ishrat T, Hoda MN, Khan MB, Yousuf S, Ahmad M, Khan MM, Ahmad A, Islam F. Amelioration of cognitive deficits and neurodegeneration by curcumin in rat model of sporadic dementia of Alzheimer’s type (sdat). Eur Neuropsychopharmacol 2009;19:636–47 [DOI] [PubMed] [Google Scholar]
- 105.Ahmed T, Gilani AH. Inhibitory effect of curcuminoids on acetylcholinesterase activity and attenuation of scopolamine-induced amnesia may explain medicinal use of turmeric in Alzheimer’s disease. Pharmacol Biochem Behav 2009;91:554–9 [DOI] [PubMed] [Google Scholar]
- 106.Ng TP, Chiam PC, Lee T, Chua HC, Lim L, Kua EH. Curry consumption and cognitive function in the elderly. Am J Epidemiol 2006;164:898–906 [DOI] [PubMed] [Google Scholar]
- 107.Ringman JM, Frautschy SA, Teng E, Begum AN, Bardens J, Beigi M, Gylys KH, Badmaev V, Heath DD, Apostolova LG. Oral curcumin for Alzheimer’s disease: tolerability and efficacy in a 24-week randomized, double blind, placebo-controlled study. Alzheimers Res Ther 2012;4:43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Baum L, Lam CWK, Cheung SKK, Kwok T, Lui V, Tsoh J, Lam L, Leung V, Hui E, Ng C. Six-month randomized, placebo-controlled, double-blind, pilot clinical trial of curcumin in patients with alzheimer disease. J Clin Psychopharmacol 2008;28:110–3 [DOI] [PubMed] [Google Scholar]
- 109. Maraldi T, Vauzour D, Angeloni C. Dietary polyphenols and their effects on cell biochemistry and pathophysiology 2013. Oxid Med Cell Longev. 2014;2014:Article ID 576363, 3 pages. [DOI] [PMC free article] [PubMed]
- 110.Spencer JPE. Flavonoids and brain health: multiple effects underpinned by common mechanisms. Genes Nutr. 2009;4:243–50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Youdim KA, Qaiser MZ, Begley DJ, Rice-Evans CA, Abbott NJ. Flavonoid permeability across an in situ model of the blood–brain barrier. Free Radic Biol Med 2004;36:592–604 [DOI] [PubMed] [Google Scholar]
- 112.Khurana S, Venkataraman K, Hollingsworth A, Piche M, Tai TC. (2013). Polyphenols: benefits to the cardiovascular system in health and in aging. Nutrients 2013;5(10):3779–827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Baptista FI, Henriques AG, Silva AM, Wiltfang J, da Cruz e, Silva OA. Flavonoids as therapeutic compounds targeting key proteins involved in Alzheimer’s disease. ACS Chem Neurosci 2014;5:83–92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Spencer JPE, Vafeiadou K, Williams RJ, Vauzour D. Neuroinflammation: modulation by flavonoids and mechanisms of action. Mol Aspects Med 2012;33:83–97 [DOI] [PubMed] [Google Scholar]
- 115.Hou DX, Kumamoto T. Flavonoids as protein kinase inhibitors for cancer chemoprevention: direct binding and molecular modeling. Antioxid Redox Signal 2010;13:691–719 [DOI] [PubMed] [Google Scholar]
- 116.Wasowski C, Marder M. Flavonoids as GABA A receptor ligands: the whole story? J Exp Pharmacol 2012;4:9–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Karton Y, Jiang JL, Ji XD, Melman N, Olah ME, Stiles GL, Jacobson KA. Synthesis and biological activities of flavonoid derivatives as a3 adenosine receptor antagonists. J Med Chem 1996;39:2293–301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Katavic PL, Lamb K, Navarro H, Prisinzano TE. Flavonoids as opioid receptor ligands: identification and preliminary structure-activity relationships. J Nat Prod 2007;70:1278–82 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Lee BH, Choi SH, Shin TJ, Pyo MK, Hwang SH, Lee SM, Paik HD, Kim HC, Nah SY. Effects of quercetin on α9α10 nicotinic acetylcholine receptor-mediated ion currents. Eur J Pharmacol 2011;650:79–85 [DOI] [PubMed] [Google Scholar]
- 120.Andero R, Daviu N, Escorihuela RM, Nadal R, Armario A. 7,8-dihydroxyflavone, a trkb receptor agonist, blocks long-term spatial memory impairment caused by immobilization stress in rats. Hippocampus 2012;22:399–408 [DOI] [PubMed] [Google Scholar]
- 121.Han YS, Bastianetto S, Dumont Y, Quirion R. Specific plasma membrane binding sites for polyphenols, including resveratrol, in the rat brain. J Pharmacol Exp Ther 2006;318:238–45 [DOI] [PubMed] [Google Scholar]
- 122.Vauzour D. Effect of flavonoids on learning, memory and neurocognitive performance: relevance and potential implications for Alzheimer’s disease pathophysiology. J Sci Food Agric 2014;94:1042–56 [DOI] [PubMed] [Google Scholar]
- 123.García-Lafuente A, Guillamón E, Villares A, Rostagno MA, Martínez JA. Flavonoids as anti-inflammatory agents: implications in cancer and cardiovascular disease. Inflamm Res 2009;58:537–52 [DOI] [PubMed] [Google Scholar]
- 124.Frank-Cannon TC, Alto LT, McAlpine FE, Tansey MG. Does neuroinflammation fan the flame in neurodegenerative diseases. Mol Neurodegener 2009;4:47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Poulose SM, Fisher DR, Larson J, Bielinski DF, Rimando AM, Carey AN, Schauss AG, Shukitt-Hale B. Anthocyanin-rich açai (euterpe oleracea mart.) fruit pulp fractions attenuate inflammatory stress signaling in mouse brain bv-2 microglial cells. J Agric Food Chem 2012;60:1084–93 [DOI] [PubMed] [Google Scholar]
- 126.Poulose SM, Fisher DR, Bielinski DF, Gomes SM, Rimando AM, Schauss AG, Shukitt-Hale B. Restoration of stressor-induced calcium dysregulation and autophagy inhibition by polyphenol-rich açaí (euterpe spp.) fruit pulp extracts in rodent brain cells in vitro. Nutrition 2014;30:853–62 [DOI] [PubMed] [Google Scholar]
- 127. Peluso I, Raguzzini A, Serafini M. Effect of flavonoids on circulating levels of tnf-#x03B1 and il-6 in humans: a systematic review and meta-analysis. Mol Nutr Food Res 2013;57:784#x2013801. [DOI] [PubMed]
- 128.Ghanim H, Sia CL, Abuaysheh S, Korzeniewski K, Patnaik P, Marumganti A, Chaudhuri A, Dandona P. An antiinflammatory and reactive oxygen species suppressive effects of an extract of polygonum cuspidatum containing resveratrol. J Clin Endocrinol Metab 2010;95:E1–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Ghanim H, Sia CL, Korzeniewski K, Lohano T, Abuaysheh S, Marumganti A, Chaudhuri A, Dandona P. A resveratrol and polyphenol preparation suppresses oxidative and inflammatory stress response to a high-fat, high-carbohydrate meal. J Clin Endocrinol Metab 2011;96:1409–14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Bolca S, Van de Wiele T, Possemiers S. Gut metabotypes govern health effects of dietary polyphenols. Curr Opin Biotechnol 2013;24:220–5 [DOI] [PubMed] [Google Scholar]
- 131.Faria A, Fernandes I, Norberto S, Mateus N, Calhau C. Interplay between anthocyanins and gut microbiota. J Agric Food Chem 2014;62:6898–902 [DOI] [PubMed] [Google Scholar]
- 132.van Duynhoven J, Vaughan EE, van Dorsten F, Gomez-Roldan V, de Vos R, Vervoort J, van der Hooft JJ, Roger L, Draijer R, Jacobs DM. Interactions of black tea polyphenols with human gut microbiota: implications for gut and cardiovascular health. Am J Clin Nutr 2013;98:1631S–41S [DOI] [PubMed] [Google Scholar]
- 133.Monagas M, Urpi-Sarda M, Sánchez-Patán F, Llorach R, Garrido I, Gómez-Cordovés C, Andres-Lacueva C, Bartolomé B. Insights into the metabolism and microbial biotransformation of dietary flavan-3-ols and the bioactivity of their metabolites. Food Funct 2010;1:233–53 [DOI] [PubMed] [Google Scholar]
- 134.Chiou YS, Wu JC, Huang Q, Shahidi F, Wang YJ, Ho CT, Pan MH. Metabolic and colonic microbiota transformation may enhance the bioactivities of dietary polyphenols. J Funct Foods 2014;7:3–25 [Google Scholar]
- 135. Bercik P, Denou E, Collins J, Jackson W, Lu J, Jury J, Deng Y, Blennerhassett P, Macri J, McCoy KD. The intestinal microbiota affect central levels of brain-derived neurotropic factor and behavior in mice. Gastroenterology 2011;141:599#x2013609. [DOI] [PubMed]
- 136.Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci 2012;13:701–12 [DOI] [PubMed] [Google Scholar]
- 137.Delmas D, Aires V, Limagne E, Dutartre P, Mazué F, Ghiringhelli F, Latruffe N. Transport, stability, and biological activity of resveratrol. Ann N Y Acad Sci 2011;1215:48–59 [DOI] [PubMed] [Google Scholar]
- 138.Ramírez-Puebla ST, Servín-Garcidueñas LE, Jiménez-Marín B, Bolaños LM, Rosenblueth M, Martínez J, Rogel MA, Ormeño-Orrillo E, Martínez-Romero E. Gut and root microbiota commonalities. Appl Environ Microbiol 2013;79:2–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Kennedy DO. Plants and the human brain. New York: Oxford University Press; 2014.
- 140.Harborne JR. Introduction to ecological biochemistry. London: Elsevier; 1993.
- 141.Agati G, Tattini M. Multiple functional roles of flavonoids in photoprotection. New Phytol 2010;186:786–93 [DOI] [PubMed] [Google Scholar]
- 142.Liberato MC, de Morais SM, Siqueira SMC, de Menezes J, Ramos DN, Machado LKA, Magalhaes IL. Phenolic content and antioxidant and antiacetylcholinesterase properties of honeys from different floral origins. J Med Food 2011;14:658–63 [DOI] [PubMed] [Google Scholar]
- 143.Adler LS. The ecological significance of toxic nectar. Oikos 2000;91:409–20 [Google Scholar]
- 144.Heil M. Nectar: generation, regulation, and ecological functions. Trends Plant Sci 2011;16:191–200 [DOI] [PubMed] [Google Scholar]
- 145.Heil M, Silva Bueno JC. Within-plant signaling by volatiles leads to induction and priming of an indirect plant defense in nature. Proc Natl Acad Sci USA 2007;104:5467–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Maffei ME. Sites of synthesis, biochemistry and functional role of plant volatiles. S Afr J Bot 2010;76:612–31 [Google Scholar]
- 147.Glinwood R, Ninkovic V, Pettersson J. Chemical interaction between undamaged plants–effects on herbivores and natural enemies. Phytochemistry 2011;72:1683–9 [DOI] [PubMed] [Google Scholar]
- 148.Treutter D. Significance of flavonoids in plant resistance: a review. Environ Chem Lett 2006;4:147–57 [Google Scholar]
- 149.Musser RO, Cipollini DF, Hum-Musser SM, Williams SA, Brown JK, Felton GW. Evidence that the caterpillar salivary enzyme glucose oxidase provides herbivore offense in solanaceous plants. Arch Insect Biochem Physiol 2005;58:128–37 [DOI] [PubMed] [Google Scholar]
- 150.Smith CM, Boyko EV. The molecular bases of plant resistance and defense responses to aphid feeding: current status. Entomol Exp Appl 2007;122:1–16 [Google Scholar]
- 151.Walters DR. Plant defence: warding off attack by pathogens. Herbivores, and parasitic plants. Chichester, UK: John Wiley and Sons; 2011.
- 152.Turlings TCJ, Tumlinson JH, Lewis WJ. Exploitation of herbivore-induced plant odors by host-seeking parasitic wasps. Science 1990;250:1251–3 [DOI] [PubMed] [Google Scholar]
- 153.Iriti M, Faoro F. Review of innate and specific immunity in plants and animals. Mycopathologia 2007;164:57–64 [DOI] [PubMed] [Google Scholar]
- 154.Harborne JB. The comparative biochemistry of phytoalexin induction in plants. Biochem Syst Ecol 1999;27:335–67 [Google Scholar]
- 155.Cesco S, Neumann G, Tomasi N, Pinton R, Weisskopf L. Release of plant-borne flavonoids into the rhizosphere and their role in plant nutrition. Plant Soil 2010;329:1–25 [Google Scholar]
- 156.Li ZH, Wang QA, Ruan XA, Pan CD, Jiang DA. Phenolics and plant allelopathy. Molecules 2010;15:8933–52 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Weston LA, Mathesius U. Flavonoids: their structure, biosynthesis and role in the rhizosphere, including allelopathy. J Chem Ecol 2013;39:283–97 [DOI] [PubMed] [Google Scholar]
- 158.Mandal SM, Chakraborty D, Dey S. Phenolic acids act as signaling molecules in plant-microbe symbioses. Plant Signal Behav 2010;5:359–68 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Hassan S, Mathesius U. The role of flavonoids in root-rhizosphere signalling: Opportunities and challenges for improving plant-microbe interactions. J Exp Bot 2012;63:3429–44 [DOI] [PubMed] [Google Scholar]
- 160.Martens S, Mithofer A. Flavones and flavone synthases. Phytochemistry 2005;66:2399–407 [DOI] [PubMed] [Google Scholar]
- 161.Pourcel L, Grotewold E. Participation of phytochemicals in plant development and growth. New York: Springer; 2009.
- 162.Taylor LP, Grotewold E. Flavonoids as developmental regulators. Curr Opin Plant Biol 2005;8:317–23 [DOI] [PubMed] [Google Scholar]
- 163.Chi WC, Fu SF, Huang TL, Chen YA, Chen CC, Huang HJ. Identification of transcriptome profiles and signaling pathways for the allelochemical juglone in rice roots. Plant Mol Biol 2011;77:591–607 [DOI] [PubMed] [Google Scholar]
- 164.Peer WA, Murphy AS. Flavonoids as signal molecules. New York: Springer; 2008.
- 165.Saslowsky DE, Warek U, Winkel BS. Nuclear localization of flavonoid enzymes in arabidopsis. J Biol Chem 2005;280:23735–40 [DOI] [PubMed] [Google Scholar]
- 166.Avanci NC, Luche DD, Goldman GH, Goldman MHS. Jasmonates are phytohormones with multiple functions, including plant defense and reproduction. Genet Mol Res 2010;9:484–505 [DOI] [PubMed] [Google Scholar]
- 167.Zhao J, Davis LC, Verpoorte R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol Adv 2005;23:283–333 [DOI] [PubMed] [Google Scholar]
- 168.Pauwels L, Goossens A. The jaz proteins: a crucial interface in the jasmonate signaling cascade. Plant Cell 2011;23:3089–100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Katsir L, Chung HS, Koo AJK, Howe GA. Jasmonate signaling: a conserved mechanism of hormone sensing. Curr Opin Plant Biol 2008;11:428–35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.De Vos M, Van Oosten VR, Van Poecke RMP, Van Pelt JA, Pozo MJ, Mueller MJ, Buchala AJ, Metraux JP, Van Loon LC, Dicke M, et al. Signal signature and transcriptome changes of arabidopsis during pathogen and insect attack. Mol Plant Microbe Interact 2005;18:923–37 [DOI] [PubMed] [Google Scholar]
- 171.An C, Mou Z. Salicylic acid and its function in plant immunity. J Integr Plant Biol 2011;53:412–28 [DOI] [PubMed] [Google Scholar]
- 172.Vlot AC, Dempsey DA, Klessig DF. Salicylic acid, a multifaceted hormone to combat disease. Annu Rev Phytopathol 2009;47:177–206 [DOI] [PubMed] [Google Scholar]
- 173.El Oirdi M, Abd El Rahman T, Rigano L, El Hadrami A, Rodriguez MC, Daayf F, Vojnov A, Bouarab K. Botrytis cinerea manipulates the antagonistic effects between immune pathways to promote disease development in tomato. Plant Cell 2011;23:2405–21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Traw MB, Bergelson J. Interactive effects of jasmonic acid, salicylic acid, and gibberellin on induction of trichomes in arabidopsis. Plant Physiol 2003;133:1367–75 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Moore JW, Loake GJ, Spoel SH. Transcription dynamics in plant immunity. Plant Cell 2011;23:2809–20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Li HH, Hao RL, Wu SS, Guo PC, Chen CJ, Pan LP, Ni H. Occurrence, function and potential medicinal applications of the phytohormone abscisic acid in animals and humans. Biochem Pharmacol 2011;82:701–12 [DOI] [PubMed] [Google Scholar]
- 177.Bassaganya-Riera J, Skoneczka J, Kingston DGJ, Krishnan A, Misyak SA, Guri AJ, Pereira A, Carter AB, Minorsky P, Tumarkin R, et al. Mechanisms of action and medicinal applications of abscisic acid. Curr Med Chem 2010;17:467–78 [DOI] [PubMed] [Google Scholar]
- 178.Scarfi S, Fresia C, Ferraris C, Bruzzone S, Fruscione F, Usai C, Benvenuto F, Magnone M, Podesta M, Sturla L, et al. The plant hormone abscisic acid stimulates the proliferation of human hemopoietic progenitors through the second messenger cyclic adp-ribose. Stem Cells 2009;27:2469–77 [DOI] [PubMed] [Google Scholar]
- 179.Lee D-S, Nioche P, Hamberg M, Raman CS. Structural insights into the evolutionary paths of oxylipin biosynthetic enzymes. Nature 2008;455:363–8 [DOI] [PubMed] [Google Scholar]
- 180.Schultz JC. Shared signals and the potential for phylogenetic espionage between plants and animals. Integr Comp Biol 2002;42:454–62 [DOI] [PubMed] [Google Scholar]
- 181.Lee JY, Hwang DH. Dietary fatty acids and eicosanoids. In: Chow CK, ed. Fatty acids in foods and their health implications. Boca Raton (FL): CRC Press; 2008.
- 182.Lin L, Tan RX. Cross-kingdom actions of phytohormones: a functional scaffold exploration. Chem Rev 2011;111:2734–60 [DOI] [PubMed] [Google Scholar]
- 183.Pierpoint WS. The natural history of salicylic acid—plant product and mammalian medicine. Interdiscip Sci Rev 1997;22:45–52 [Google Scholar]
- 184.Fritz-Laylin LK, Prochnik SE, Ginger ML, Dacks JB, Carpenter ML, Field MC, Kuo A, Paredez A, Chapman J, Pham J, et al. The genome of naegleria gruberi illuminates early eukaryotic versatility. Cell 2010;140:631–42 [DOI] [PubMed] [Google Scholar]
- 185.Koonin EV. The incredible expanding ancestor of eukaryotes. Cell 2010;140:606–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Koonin EV, Fedorova ND, Jackson JD, Jacobs AR, Krylov DM, Makarova KS, Mazumder R, Mekhedov SL, Nikolskaya AN, Rao BS, et al. A comprehensive evolutionary classification of proteins encoded in complete eukaryotic genomes. Genome Biol 2004;5:R7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Howitz KT, Sinclair DA. Xenohormesis: sensing the chemical cues of other species. Cell 2008;133:387–91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Greiss S, Gartner A. Sirtuin/sir2 phylogeny, evolutionary considerations and structural conservation. Mol Cells 2009;28:407–15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Howitz KT, Bitterman KJ, Cohen HY, Lamming DW, Lavu S, Wood JG, Zipkin RE, Chung P, Kisielewski A, Zhang LL, et al. Small molecule activators of sirtuins extend saccharomyces cerevisiae lifespan. Nature 2003;425:191–6 [DOI] [PubMed] [Google Scholar]
- 190.Wood JG, Rogina B, Lavu S, Howitz K, Helfand SL, Tatar M, Sinclair D. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature 2004;430:686–9 [DOI] [PubMed] [Google Scholar]
- 191.Rahman I, Biswas SK, Kirkham PA. Regulation of inflammation and redox signaling by dietary polyphenols. Biochem Pharmacol 2006;72:1439–52 [DOI] [PubMed] [Google Scholar]
- 192.Nakagami H, Pitzschke A, Hirt H. Emerging map kinase pathways in plant stress signalling. Trends Plant Sci 2005;10:339–46 [DOI] [PubMed] [Google Scholar]
- 193.Ausubel FM. Are innate immune signaling pathways in plants and animals conserved? Nat Immunol 2005;6:973–9 [DOI] [PubMed] [Google Scholar]
- 194.Bögre L, Okresz L, Henriques R, Anthony RG. Growth signalling pathways in arabidopsis and the agc protein kinases. Trends Plant Sci 2003;8:424–31 [DOI] [PubMed] [Google Scholar]
- 195.Shiu SH, Bleecker AB. Receptor-like kinases from arabidopsis form a monophyletic gene family related to animal receptor kinases. Proc Natl Acad Sci USA 2001;98:10763–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Zhang T, Liu Y, Yang T, Zhang L, Xu S, Xue L, An L. Diverse signals converge at mapk cascades in plant. Plant Physiol Biochem 2006;44:274–83 [DOI] [PubMed] [Google Scholar]
- 197.Brown JR, Auger K. Phylogenomics of phosphoinositide lipid kinases: perspectives on the evolution of second messenger signaling and drug discovery. BMC Evol Biol 2011;11:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Forzani C, Carreri A, van Bentem SD, Lecourieux D, Lecourieux F, Hirt H. The arabidopsis protein kinase pto-interacting 1–4 is a common target of the oxidative signal-inducible 1 and mitogen-activated protein kinases. FEBS J 2011;278:1126–36 [DOI] [PubMed] [Google Scholar]
- 199.Robaglia C, Thomas M, Meyer C. Sensing nutrient and energy status by snrk1 and tor kinases. Curr Opin Plant Biol 2012;15:301–7 [DOI] [PubMed] [Google Scholar]
- 200.Zoncu R, Efeyan A, Sabatini DM. Mtor: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol 2011;12:21–35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.van Dam TJ, Zwartkruis FJ, Bos JL, Snel B. Evolution of the tor pathway. J Mol Evol 2011;73:209–20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Bassham DC. Function and regulation of macroautophagy in plants. Biochim Biophys Acta 2009;1793:1397–403 [DOI] [PubMed] [Google Scholar]
- 203.Ballou LM, Lin RZ. Rapamycin and mtor kinase inhibitors. J Chem Biol 2008;1:27–36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Van Aller GS, Carson JD, Tang W, Peng H, Zhao L, Copeland RA, Tummino PJ, Luo L. Epigallocatechin gallate (egcg), a major component of green tea, is a dual phosphoinositide-3-kinase/mtor inhibitor. Biochem Biophys Res Commun 2011;406:194–9 [DOI] [PubMed] [Google Scholar]
- 205.Brüning A. Inhibition of mtor signaling by quercetin in cancer treatment and prevention. Anticancer Agents Med Chem 2013;13(7):1025–31 [DOI] [PubMed] [Google Scholar]
- 206.Zhang X, Chen LX, Ouyang L, Cheng Y, Liu B. Plant natural compounds: Targeting pathways of autophagy as anti-cancer therapeutic agents. Cell Prolif 2012;45:466–76 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Ghosh HS, McBurney M, Robbins PD. Sirt1 negatively regulates the mammalian target of rapamycin. PLoS One 2010;5:e9199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Liu M, Wilk SA, Wang A, Zhou L, Wang R-H, Ogawa W, Deng C, Dong LQ, Liu F. Resveratrol inhibits mtor signaling by promoting the interaction between mtor and deptor. J Biol Chem 2010;285:36387–94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Poulose SM, Bielinski DF, Gomes SM, Carrihill-Knoll K, Rabin BM, Shukitt-Hale B. Protective effects of berries and walnuts against the accelerated aging and age-associated stress caused by irradiation in critical regions of rat brain [abstract]. FASEB J 2010;26:255.6 [Google Scholar]
- 210.Poulose SM, Bielinski D, Carrihill-Knoll KL, Rabin BM, Shukitt-Hale B. Neuronal stress following exposure to 56fe particles and the effects of antioxidant-rich diets. J Radiat Res 2014;55(Suppl 1):73–4 [Google Scholar]
- 211.Ren D, Liu Y, Yang K-Y, Han L, Mao G, Glazebrook J, Zhang S. A fungal-responsive mapk cascade regulates phytoalexin biosynthesis in arabidopsis. Proc Natl Acad Sci USA 2008;105:5638–43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Ahuja I, Kissen R, Bones AM. Phytoalexins in defense against pathogens. Trends Plant Sci 2012;17:73–90 [DOI] [PubMed] [Google Scholar]
- 213.Nadarajah K, Sidek HM. The green MAPKs. Asian J Plant Sci 2010;9:1–10 [Google Scholar]
- 214.Ritsema T, van Zanten M, Leon-Reyes A, Voesenek L, Millenaar FF, Pieterse CMJ, Peeters AJM. Kinome profiling reveals an interaction between jasmonate, salicylate and light control of hyponastic petiole growth in arabidopsis thaliana. PLoS One 2010;5:314255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Smith AG, Croft MT, Moulin M, Webb ME. Plants need their vitamins too. Curr Opin Plant Biol 2007;10:266–75 [DOI] [PubMed] [Google Scholar]
- 216.Delledonne M. No news is good news for plants. Curr Opin Plant Biol 2005;8:390–6 [DOI] [PubMed] [Google Scholar]
- 217.Huang X, Stettmaier K, Michel C, Hutzler P, Mueller MJ, Durner J. Nitric oxide is induced by wounding and influences jasmonic acid signaling in arabidopsis thaliana. Planta 2004;218:938–46 [DOI] [PubMed] [Google Scholar]
- 218.Hollender C, Liu ZC. Histone deacetylase genes in arabidopsis development. J Integr Plant Biol 2008;50:875–85 [DOI] [PubMed] [Google Scholar]
- 219.Wang C, Gao F, Wu J, Dai J, Wei C, Li Y. Arabidopsis putative deacetylase atsrt2 regulates basal defense by suppressing PAD4, EDS5 and SID2 expression. Plant Cell Physiol 2010;51:1291–9 Erratum in: Plant Cell Physiol 2010;51:1820 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Conseil G, Baubichon-Cortay H, Dayan G, Jault JM, Barron D, Di Pietro A. Flavonoids: a class of modulators with bifunctional interactions at vicinal atp- and steroid-binding sites on mouse p-glycoprotein. Proc Natl Acad Sci USA 1998;95:9831–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Venkateshwaran M, Volkening JD, Sussman MR, Ané JM. Symbiosis and the social network of higher plants. Curr Opin Plant Biol 2013;16:118–27 [DOI] [PubMed] [Google Scholar]
- 222.Fox JE. Chemical communication threatened by endocrine-disrupting chemicals. Environ Health Perspect 2004;112:648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Poulin MJ, Simard J, Catford JG, Librie F, Piché Y. Response of symbiotic endomycorrhizal fungi to estrogens and antiestrogens. Mol Plant Microbe Interact 1997;10:481–7 [Google Scholar]
- 224.Baker ME. Endocrine activity of plant-derived compounds: an evolutionary perspective. Proc Soc Exp Biol Med 1995;208:131–38 [DOI] [PubMed] [Google Scholar]
- 225.Györgypal Z, Kondorosi A. Homology of the ligand-binding regions of rhizobium symbiotic regulatory protein nodd and vertebrate nuclear receptors. Mol Gen Genet 1991;226:337–40 [DOI] [PubMed] [Google Scholar]
- 226.Fox JE, Starcevic M, Jones PE, Burow ME, McLachlan JA. Phytoestrogen signaling and symbiotic gene activation are disrupted by endocrine-disrupting chemicals. Environ Health Perspect 2004;112:672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Makoi JH, Ndakidemi PA. Biological, ecological and agronomic significance of plant phenolic compounds in rhizosphere of the symbiotic legumes. Afr J Biotechnol 2010;6:1358–68. [Google Scholar]
- 228.Boué SM, Burow ME, Wiese TE, Shih BY, Elliott S, Carter-Wientjes CH, McLachlan JA, Bhatnagar D. Estrogenic and antiestrogenic activities of phytoalexins from red kidney bean (phaseolus vulgaris l.). J Agric Food Chem 2011;59:112–20 [DOI] [PubMed] [Google Scholar]
- 229.Heitman J, Movva N, Hall MN. Targets for cell cycle arrest by the immunosuppressant rapamycin in yeast. Science 1991;253:905. [DOI] [PubMed] [Google Scholar]
- 230.Cruz MC, Cavallo LM, Görlach JM, Cox G, Perfect JR, Cardenas ME, Heitman J. Rapamycin antifungal action is mediated via conserved complexes with fkbp12 and tor kinase homologs incryptococcus neoformans. Mol Cell Biol 1999;19:4101–12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Harrison DE, Strong R, Sharp ZD, Nelson JF, Astle CM, Flurkey K, Nadon NL, Wilkinson JE, Frenkel K, Carter CS. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 2009;460:392–5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Jung HJ, Hwang I, Sung W, Kang H, Kang B, Seu Y, Lee D. Fungicidal effect of resveratrol on human infectious fungi. Arch Pharm Res 2005;28:557–60 [DOI] [PubMed] [Google Scholar]
- 233.Araújo C, Leon L. Biological activities of curcuma longa l. Mem Inst Oswaldo Cruz 2001;96:723–8 [DOI] [PubMed] [Google Scholar]