Abstract
Poxviruses are a family of double stranded DNA viruses that include active human pathogens such as monkeypox, molluscum contagiousum, and Contagalo virus. The family also includes the smallpox virus, Variola. Due to the complexity of poxvirus replication, many questions still remain regarding their gene expression strategy. In this article we describe the conceptualization and usage of recombinant vaccinia viruses that enable real-time measurement of single and multiple stages of viral gene expression in a high-throughput format. This is enabled through the use of spectrally distinct fluorescent proteins as reporters for each of three stages of viral replication. These viruses provide a high signal-to-noise ratio while retaining stage specific expression patterns, enabling plate-based assays and microscopic observations of virus propagation and replication. These tools have uses for antiviral discovery, studies of the virus-host interaction, and evolutionary biology.
Keywords: Immunology, Issue 87, vaccinia; poxvirus; infection; virus-host interaction; screen; inhibitor; gene expression; cell biology; fluorescence; antiviral; reporter, mCherry, Venus, TagBFP
Introduction
Traditionally, virus expression is studied using molecular biology techniques (e.g. northern blotting, western blotting, microarray hybridization, etc.) 1. While these methods are capable of providing detailed information with respect to categorizing expression changes of individual mRNAs or proteins, they are typically not amenable to real-time and high-throughput processes. Alternative approaches using fluorescence-based reporters have been applied previously when working with poxviruses; however, their development and usage has been motivated by varied aims. Several such methods were designed for selection of recombinant viruses 2,3. These techniques express EGFP upon proper incorporation and expression of exogenous DNA from recombinant vaccinia clones. Similarly, several vaccinia strains stably expressing soluble EGFP or GFP-tagged proteins expressed under a native vaccinia promoter have been widely used. These are typically used to quantify virus entry and replication during antibody-based neutralization assays, chemical inhibition, or comparison of antiviral effectiveness 4–6. While these viruses have proven useful, they are limited in their ability to provide detailed information about the point of inhibition due to their usage of a single ambiguous early/late viral promoter. Previous methods have also made use of EGFP protein with suboptimal signal-to-noise characteristics.
Due to the complexity of poxvirus replication and the lack of existing tools available to assay real-time changes in viral gene expression, we have developed a suite of single and multi stage reporter viruses 7,8. As described in previous publications, these viruses express one, two, or three spectrally distinct fluorescent proteins from native vaccinia promoters during early, intermediate, or late stages in infection. These viruses can be used as indicators of virus replication progress using a fluorescence microscope and they are equally well suited for high-throughput plate-and-reader based assays. These viruses are easy to use in place of wild type virus, growing with similar kinetics to reach equivalent titers 7. During the planning and creation of these viruses, much care was taken in selecting fluorophores that have high signal-to-background characteristics with superior folding efficiency to facilitate reliable quantification with rapid feedback to changes in expression (Venus, mCherry and TagBFP). Additionally, combinations of viral promoters were chosen which produce high fidelity, unambiguous stage-specific expression, providing information about the full range of early (C11R promoter), intermediate (G8R promoter) and late (F17R promoter) gene expression, in contrast to ambiguous early/late promoters.
These viruses can be used as a tool for investigating the life cycle of the prototypical poxvirus, vaccinia virus. Much is still not known about the host-virus interaction despite its long history of study. Vaccinia is complex, producing over 200 unique proteins, many of which are immunomodulatory and host antagonistic. Upon infection, vaccinia virus immediately begins early mRNA transcription. This is facilitated by an RNA polymerase and transcription factors that were loaded on the viral genome during virion packaging and held in a paused state until subsequent infection. This early expression mainly produces proteins required for suppression of the host immune system (mRNA decapping, dsRNA sequestration, and decoy receptor proteins as well as inhibitors of apoptosis, stress response, and Toll, IL, and NF-κB signaling) and genome replication. Early expression also produces transcription factors necessary for intermediate expression. Intermediate expression includes the expression of late stage transcription factors. This expression cascade leads to production of structural and enzymatic virus proteins during late stages of infection, which are necessary for complete assembly of the mature vaccinia virion.
Our set of fluorescent reporter viruses allows for rapid progress to be made in the understanding of poxvirus biology. One of the most common and time-consuming methods in the field of virology is the growth assay. This typically involves infecting cells, enacting a series of treatments, harvesting virus by lysing infected cells, and quantifying the resulting viral titer by plaque assay. Using the reporter viruses described here allows real-time measurement of virus growth that can be easily assayed and compared between numerous treatments performed in parallel. We foresee this set of reagents will be used in various protocols for identifying alterations in viral gene expression in response to drug treatment, RNAi knockdown, or host range restriction.
This method also enables high-content analysis on a larger scale than previously available, allowing for high-throughput anti-viral drug screens for specifically-defined target stages of interest. While numerous potential treatments to combat poxvirus infection have been identified, the only FDA approved therapies effective for treating poxvirus infection are the acyclic phosphonate nucleoside, Cidofovir, and treatment with vaccinia immune globulin 9,10. Despite the eradication of smallpox in 1977 11, poxviruses remain a significant threat to human health 12. Cessation of widespread vaccination against Variola virus has led to increased susceptibility to other poxviruses 13. For example, regions of Central Africa previously protected by vaccination are experiencing a surge in Monkeypox virus infections 14. Significant concern has also been raised regarding the latent susceptibility to intentional release of Variola virus. Due to the limited therapies currently available, there is an urgent need for development of novel treatments. These reporter viruses allow rapid and high-throughput small molecule inhibitor screening for inhibition of a specific stage of viral replication. Identification of inhibitors that target viral expression stages currently not the target of inhibition will facilitate development of combination therapies with increased potency.
Much can be gleaned about vaccinia cell biology by observing changes in each stage’s gene expression. Attenuation by chemical or genetic manipulation of an infected host cell is typically expressed in terms of reduced viral titers. However, by comparing changes in each stage of the viral expression cascade, one can obtain a more complete understanding of how virus fitness is impacted by a particular treatment. These data have been shown to correlate well with the traditional virus titer output, but provide more detailed mechanistic information as well as high throughput capabilities 7.
Protocol
1. Plate Cells
Dissociate HeLa cells from plate and dilute in growth media (DMEM, 2 mM L-glut, 10% FBS) to approximately 2.0 x 105 cells/ml. Dispense 100 μl/well in a black-walled, clear flat-bottom 96-well plate (20,000 cells/well).
Incubate cells for 24 hr until confluent in a 37 °C incubator + 5% CO2.
2. Infect Cells
- Dilute virus and infect cells.
- Thaw TrpV (Triple virus; Early Venus, Intermediate mCherry, Late TagBFP) and PLV (Promoter-less Venus) fluorescent virus and disaggregate using sonication for 5 minutes. Alternatively, crude virus stocks can be mixed 1:1 with 0.25 mg/ml trypsin in infection media and incubated at 37 °C for 30 min.
- Dilute crude virus stocks in 37 °C infection media (DMEM, 2 mM L-glut, 2% FBS). For high MOI infections (10 PFU/cell), dilute virus stock to 1.0 x 107 PFU/ml assuming 5 x 104 cells/well and an inoculum volume of 50 μl. Plan on infecting 3 replicate wells for each treatment with TrpV and another 3 replicate wells for the same treatment with PLV to account for background fluorescence specific to each treatment.
- Infect wells by adding 50 μl/well diluted virus in infection media. This is defined as time = 0 hr post infection (0 hpi).
- Dilute desired treatment compounds into infection media. For all treatments a single 2x master mix dilution should be made and applied to replicate wells (e.g. 350 μl total volume for six 96-wells with 50 μl each).
- Dilute experimental compounds into infection media.
- Dilute vehicle-control solvent (e.g. PBS, DMSO) into infection media. Note: Similar final concentrations of vehicle-control solvents should be used as applied for experimental compounds (e.g. if your IBT stock is made with DMSO and used at a final concentrations of 1 μl/ml, then use DMSO alone at 1 μl/ml as the vehicle control).
- Dilute control compounds into infection media. Recommended control compounds include: 3.6 μM (1 μg/ml) 1-β-D-Arabinofuranosylcytosine (AraC), 50 μM (11.7 μg/ml) isatin β -thiosemicarbazone (IBT), 60 μM (50 μg/ml) Rifampicin, or 5 μM (1.9 μg/ml) ST-246 8,15,16. See Representative Results for expected effects. Note: make this dilution at twice (2x) the desired final concentration since this is added at a 1:1 ratio to the volume already in the well.
Immediately after addition of virus, add 50 μl infection media containing desired treatments and controls as diluted in step 2.2. Note: To assay for inhibition before early stage expression, compound(s) can either be added directly to the diluted virus inoculum (step 2.1.2) or to host cells before infection (before step 2.1.3).
Incubate 6-24 hr in a 37 °C incubator + 5% CO2.
3. Fix Cells
Fix cells by adding 100 μl 8% paraformaldehyde to infection media already in each well. Incubate at room temperature for 15 min protected from light. Note: It is recommended to add 2x PFA (8%) directly to infection media to prevent aerosolization of infectious vaccinia which can occur if high-titer media is inverted directly into waste dish prior to fixation.
Remove fixative by inverting plate into the waste dish.
Add 100 μl of room temperature PBS.
Seal plate with optically clear adhesive film. Note: Plates can be stored at 4 °C if not read immediately. Be sure to return sealed plates to room temperature before reading to prevent condensation from distorting spectrophotometer measurements.
4. Quantify Virus Growth
Measure endpoint fluorescence at 515:530 for Venus, 587:610 for mCherry, and 415:457 for TagBFP (Excitation:Emission wavelength in nm). Four measurements should be averaged per well using optimized gain settings for each channel. Note: The appropriate emission wavelength may be different depending on the specific model and filter characteristics of your plate reader. This can be determined empirically by performing an emission wavelength scan to determine the point where there is the greatest difference between TrpV and PLV for each channel.
- Normalize the raw data to facilitate comparison between experiments.
- For each channel, determine the mean of replicate TrpV and PLV wells.
- Subtract the mean PLV-infected background measurements from the mean TrpV-infected experimental measurements for each treatment.
- Normalize the data by dividing each background subtracted value by the vehicle-only value well.
- Once an experiment has been repeated multiple times, a one-way analysis of variance (one-way ANOVA) test and multiple comparison post-test can be performed to determine if any of the treatments reflects a statistically significant difference from the vehicle-only treatment for each channel.
5. Alternative Protocol: Kinetic Plate Assays
Perform all steps through addition of treatments (step 2.3).
Seal plate with adhesive film and place in plate reader chamber equilibrated to 37 °C. Note: Special care must be taken to keep plates consistently at 37 °C to prevent undue cell stress and condensation. For kinetic assays, it is particularly important to use a buffered medium (sodium bicarbonate and HEPES) to maintain proper pH without addition of 5% CO2.
Set plate reader gain manually to prevent saturation of later time points. Note: Accurate gain optimization can be obtained by performing an endpoint assays under similar conditions.
Acquire hourly readings for 8-24 hpi and normalize using similar procedure as in endpoint assay (step 4.2).
Individual time points can be analyzed for statistical significance using similar methods as the previously described endpoint assay.
Representative Results
As an example of the typical usage of these viruses, the triple fluorescence-reporter virus was used to compare the point of inhibition of several well-defined poxvirus inhibitors.
HeLa cells were plated in tissue culture treated black-walled clear flat-bottom 96-well plates and incubated overnight. Confluent monolayers were infected at a multiplicity of infection (MOI) of 10 using either triple reporter virus (TrpV; Table 1) or promoter-less Venus (PLV) as detailed on the plate map (Figure 1). Infection media containing treatments was added to obtain a final concentration of 0.1% DMSO, 3.6 μM (1 μg/ml) AraC, 50 μM (11.7 μg/ml) IBT, 5 μM (1.9 μg/ml) ST-246 or 60 μM (50 μg/ml) Rifampicin in triplicate. 0.1% DMSO was used as the vehicle control since all inhibitor stocks were used at 1 μl/ml in DMSO. The plate was returned to a 37 °C incubator until 18 hpi. Cells were fixed for 15 minutes by addition of 100 μl 8% PFA to all wells and stored in PBS protected from light until read on a plate reader. Fluorescence readings were performed using a Tecan Infinite M1000 plate reader and i-control software (v1.5.14.0) by bottom-reading 4 points per well at excitation:emission wavelengths of 515:530, 587:610, 415:457nm for Venus, mCherry, and TagBFP fluorophores, respectively. Prior to final measurements, gain was optimized to allow maximal signal without saturation of sensors (gain was typically 235, 255, 235 for Green, Red and Blue measurements, respectively).
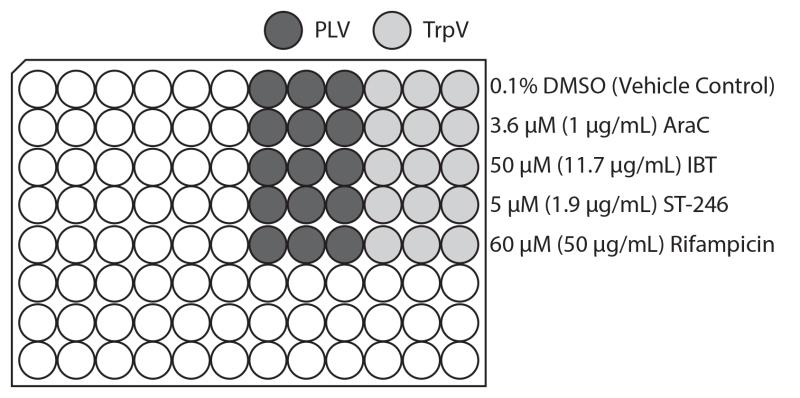 Figure 1. Representative layout of infections and drug treatments on 96-well plate. Diagram of the 96-well plate infection and small molecule addition scheme used for creating representative results. Triple fluorescent virus (TrpV; lighter gray) expressing early Venus, intermediate mCherry, and late TagBFP or promoter-less Venus (PLV; darker gray) are used in triplicate columns. 1-β-D-Arabinofuranosylcytosine (AraC), isatin β-thiosemicarbazone (IBT), Rifampicin, ST-246 and DMSO vehicle control with tested concentration are indicated at right. Click here to view larger image.
Figure 1. Representative layout of infections and drug treatments on 96-well plate. Diagram of the 96-well plate infection and small molecule addition scheme used for creating representative results. Triple fluorescent virus (TrpV; lighter gray) expressing early Venus, intermediate mCherry, and late TagBFP or promoter-less Venus (PLV; darker gray) are used in triplicate columns. 1-β-D-Arabinofuranosylcytosine (AraC), isatin β-thiosemicarbazone (IBT), Rifampicin, ST-246 and DMSO vehicle control with tested concentration are indicated at right. Click here to view larger image.
Relative fluorescence units (RFU) for each replicate were normalized for each channel. First, the mean fluorescence intensity for PLV wells was subtracted from the mean intensity of the TrpV wells for each treatment to account for background intensity contributed by each drug. Next, this value was divided by the background subtracted mean intensity of DMSO-treated TrpV wells (wild type growth). The results obtained after treatment with known poxvirus inhibitors was consistent with previously published findings and their understood mechanism of action. The cessation of early gene transcription and transition to intermediate gene expression has been shown to require DNA replication 17. Consistent with this, AraC, an inhibitor of DNA replication, showed a dramatic increase in early gene expression (210% of DMSO treated early expression; Figure 2) and a complete lack of intermediate and late expression (0.4% and 0.3% of DMSO treated intermediate and late expression, respectively; Figure 2). While the exact mechanism of action is not defined for IBT, it is thought to promote read-through transcription, resulting in increased amounts of dsRNA, activation of the RNase L pathway and apoptosis 18–20. In response to IBT treatment a progressively severe phenotype was observed in which intermediate and late expression were significantly less than DMSO alone (24.7% and 2.9% of DMSO treated for intermediate and late expression, respectively; Figure 2). A consistent, but not statistically significant decrease early fluorescence was also observed; however this it likely due to IBT-induced apoptosis at this later time point (74.0 % of DMSO at 18 hpi). In contrast to AraC and IBT, the poxvirus drugs ST-246 and Rifampicin both inhibit after late gene expression during virion assembly and maturation 15,16. No changes in gene expression were seen during all stages when cells were treated with either ST-246 (100.9%, 98.5%, and 94.5% of DMSO treated early, intermediate and late expression, respectively; Figure 2) or Rifampicin (107.6%, 104.2%, and 106.5% of DMSO treated early, intermediate and late expression, respectively).
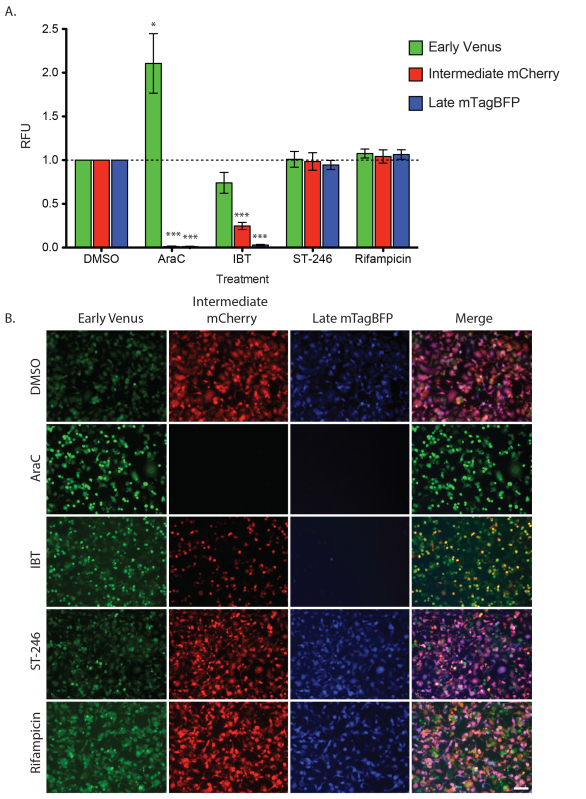 Figure 2. Stage of inhibition resulting from poxvirus antiviral drugs. HeLa cells were infected and treated as described in representative results section. A) Chart showing relative fluorescence units (RFU; background subtracted each channel based on PLV growth for each treatment and normalized to TrpV+DMSO) for early (green), intermediate (red), and late (blue) viral expression. Cells were grown in the presence of indicated compounds for 0-18 hpi. Mean with standard deviation is shown for four biological replicates each performed in triplicate. One-sample t-tests were performed for each treatment and DMSO channel pair and statistically significant differences are marked with asterisks (*p < 0.05, ***p < 0.0005). B) Images of each well treatment at 18 hpi. All images were captured with 10X objective on a Zeiss 200M epifluorescence microscope and exposures scaled similarly. Scalebar = 100 μm. Click here to view larger image.
Figure 2. Stage of inhibition resulting from poxvirus antiviral drugs. HeLa cells were infected and treated as described in representative results section. A) Chart showing relative fluorescence units (RFU; background subtracted each channel based on PLV growth for each treatment and normalized to TrpV+DMSO) for early (green), intermediate (red), and late (blue) viral expression. Cells were grown in the presence of indicated compounds for 0-18 hpi. Mean with standard deviation is shown for four biological replicates each performed in triplicate. One-sample t-tests were performed for each treatment and DMSO channel pair and statistically significant differences are marked with asterisks (*p < 0.05, ***p < 0.0005). B) Images of each well treatment at 18 hpi. All images were captured with 10X objective on a Zeiss 200M epifluorescence microscope and exposures scaled similarly. Scalebar = 100 μm. Click here to view larger image.
Historically, a combination of low MOI (0.01-0.1 PFU/cell) and high MOI (5-10 PFU/cell) growth assays are used when exploring the inhibitory effect of a new small molecule or virus mutant. At a low MOI, overall inhibitory effects are often exaggerated by even minor inhibition since only a subset of cells is initially infected. When trying to define the point during an infection cycle at which a virus is inhibited, a high MOI infection is likely more appropriate, as all the cells are infected by your primary inoculum. The experiment shown in Figure 3 was performed as in Figure 2 with the addition of a set of wells that were infected at a lower MOI = 1. As an inhibitor that functions post-transcriptionally, ST-246 should not affect any stage of gene expression; however, it is clear that when only a subset of cells is infected (Figure 3, MOI = 1) there is apparent inhibition of all stages of expression (38.4%, 48.9%, and 52.9% of DMSO compared to 100.9%, 98.5%, and 94.5% for MOI = 1 vs. MOI = 10 infection level of early, intermediate and late expression, respectively; Figure 3). Assuming 100% inhibition of mature virion formation by ST-246, this means 100% of the cells were infected by the MOI = 10 infections, and somewhere between 50 and 70% of the cells were productively infected during the MOI = 1 infection.
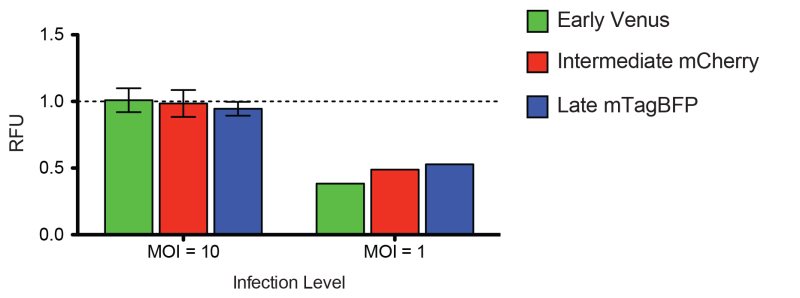 Figure 3. Effect of infection level on apparent ST-246 inhibition. HeLa cells infected as in Figure 2 with both high (MOI=10) and low (MOI=1) TrpV treated with ST-246. Relative fluorescence units (RFU; background subtracted each channel based on PLV growth for each treatment and normalized to TrpV+DMSO) for early (green), intermediate (red), and late (blue) viral expression in cells infected at either MOI=1 or MOI=10 and treated with 20 μM ST-246. Click here to view larger image.
Figure 3. Effect of infection level on apparent ST-246 inhibition. HeLa cells infected as in Figure 2 with both high (MOI=10) and low (MOI=1) TrpV treated with ST-246. Relative fluorescence units (RFU; background subtracted each channel based on PLV growth for each treatment and normalized to TrpV+DMSO) for early (green), intermediate (red), and late (blue) viral expression in cells infected at either MOI=1 or MOI=10 and treated with 20 μM ST-246. Click here to view larger image.
| Name | Description | Ref. |
| TrpV | Triple virus; C11R (Early) promoted Venus, G8R (Intermediate) promoted mCherry, F17R (Late) promoted mTagBFP | 7 |
| PLV | Promoter-less Venus | |
| EV | C11R (Early) promoted Venus | |
| IV | G8R (Intermediate) promoted Venus | |
| LV | F17R (Late) promoted Venus | |
| IREV | G8R (Intermediate) promoted mCherry and C11R (Early) promoted Venus | |
| LREV | F17R (Late) promoted mCherry and C11R (Early) promoted Venus | |
| LR | F17R (Late) promoted mCherry | |
| mCherry-A4L | N-terminal fusion of the VENUS fluorophore to the late-expressed viral core protein A4L |
Table 1. List of vaccinia virus reporter strains. Table describing fluorescence reporter viruses.
Discussion
Here, we have described the practical usage of a multi-stage vaccinia reporter virus (TrpV), which provides reproducible, real-time feedback measures of virus replication. By using well-defined poxvirus inhibitors, we were able to show that TrpV responds in a manner consistent with the understood mechanism of action for each inhibitor. While the TrpV virus provides the most comprehensive information about virus stage progression, two-stage (IREV, LREV) and single-stage (EV, IV, LV) viruses can also be used similarly, taking advantage of the optimal protein folding, stability, and superior signal-to-noise ratio of the Venus fluorophore.
The proof-of-concept assay in this protocol indicates the feasibility of identifying the stage of viral gene expression effected by known poxvirus inhibitors in a simple, high throughput plate assay. This assay can be extended to complete quick, comprehensive screening of unknown chemical libraries as well. The viruses can be used to infect small-molecule treated cells at low MOI, allowing for the specific identification of compounds that block any stage of the viral lifecycle. The inhibitors can then be screened using TrpV at several MOIs to determine the stage of the virus lifecycle inhibited, from entry (no gene expression), to early, intermediate, or late gene expression or assembly (full gene expression but no virus spread). We have used this technique to successfully identify a new anti-poxviral inhibitor with activity against several poxviruses, including Monkeypox 8.
While we have provided details for using these viruses in a plate-reader format, it is equally well suited for microscopic examination. Infection and observation via fluorescence microscope (see Figure 2B) allows for a cell-by-cell examination of viral stage progression, which could be useful in a mixed-cell culture setting, such as infection of a tissue, or in viral or bacterial co-infection conditions. Additionally, while these viruses were created for the purpose of antiviral screening 8, we expect them to be of equal usefulness in basic research investigating the life cycle of vaccinia virus. For example, these viruses can easily be modified using traditional techniques to either lack or overexpress viral proteins; the kinetics of virus gene expression and spread can then be monitored in real time for all stages of viral transcription.
Investigations of virus-host interaction can benefit from the use of these reporter viruses as well. These reporters allow rapid identification of the stage of viral replication completed during infection of non-permissive cell types, such as certain primary cell lines, peripheral blood leukocytes, or divergent species 21,22. Information gathered from using this tool would further our knowledge of poxvirus host range restriction factors and the function of viral immuno-modulatory proteins. The viruses will also be useful to identify the stage of virus replication inhibited by knockdown of host factors required for infection, in either a whole-genome scale screen or in a directed small library screen.
Several steps in this protocol should be considered critical when beginning to use these viruses in a new system. Differences in plate format, growth media, and cell type may require alteration of MOI, inoculation time, or incubation duration. It becomes especially important to optimize endpoint incubation time before scaling up to high-throughput format. To determine the ideal incubation time, a pilot kinetic plate assay should be performed (see step 5). During a kinetic assay, fluorescence measurements are made every hour for 12-24 hours and can be used to pinpoint the time at which the greatest difference is observed between control and treatment virus expression. The importance of this step can be seen in treatment with IBT, where inhibition due to increased rates of apoptosis lead to an apparent, although not statistically significant decrease in early expression. If earlier time points (4-8 hpi) were observed there would likely be no observed difference. Additionally, testing a novel inhibitory compound should include multiple compound concentrations. While these fluorescence reporter viruses are able to detect minimal changes in gene expression, the additional information obtained by multiple concentrations can lead to a better understanding of their overall effect.
During the planning and creation of these viruses, much care was taken in selecting fluorophores that have high signal-to-background characteristics 7. The Venus protein was identified as significantly better than any other tested fluorophore, and therefore was used throughout the development of triple, double, and single fluorescent reporter viruses. One limitation to using this fluorophore is that it cannot be used to infect any cell line already containing a green fluorescent reporter of fusion protein. To address analysis under these circumstances, several alternative viruses were created using red fluorescent proteins (Table 1). By expressing either soluble mCherry from the F17R late promoter or a mCherry-A4L fusion protein from the natural A4L late promoter we are able to accomplish similar measurements without Venus. Although the usage of mCherry does not have the added benefit provided by the Venus protein we are now able to accommodate measurement in green-expressing cell lines.
In summary, we have developed a set of reporter vaccinia viruses with various applications, including high-throughput plate reader or high content imaging assays. These viruses will make examination of the viral lifecycle much faster than traditional methods, and can be used for basic science as well as applied antiviral research.
Disclosures
The authors have nothing to disclose and no patents are pending for viruses or screening methods.
Acknowledgments
We thank SIGA Technologies (Corvallis, OR) for providing ST-246. D.K.R. was supported by an NIH training grant in immunology to Boston University (5T32AI 7309). This work was supported in part by P41 086180, NIH RO1AI1096159-01, and RO3 (to J.H.C.).
References
- Yen J, Golan R, Rubins K. Vaccinia virus infection & temporal analysis of virus gene expression: part 1. J Vis Exp. 2009;26(26) doi: 10.3791/1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hansen SG, Cope TA, Hruby DE. BiZyme: a novel fusion protein-mediating selection of vaccinia virus recombinants by fluorescence and antibiotic resistance. BioTechniques. 2002;32(5):1182–1187. doi: 10.2144/02325dd08. [DOI] [PubMed] [Google Scholar]
- Popov S, Mirshahidi S, Essono S, Song R, Wang X, Ruprecht RM. Generation of recombinant vaccinia viruses via green fluorescent protein selection. DNA and Cell Biology. 2009;28(3):103–108. doi: 10.1089/dna.2008.0792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartee E, Mohamed MR, Lopez MC, Baker HV, McFadden G. The addition of tumor necrosis factor plus beta interferon induces a novel synergistic antiviral state against poxviruses in primary human fibroblasts. Journal of Virology. 2009;83(2):498–511. doi: 10.1128/JVI.01376-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward BM, Moss B. Visualization of intracellular movement of vaccinia virus virions containing a green fluorescent protein-B5R membrane protein chimera. Journal of Virology. 2001;75(10):4802–4813. doi: 10.1128/JVI.75.10.4802-4813.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson MC, Damon IK, Karem KL. A rapid, high-throughput vaccinia virus neutralization assay for testing smallpox vaccine efficacy based on detection of green fluorescent protein. Journal of Virological Methods. 2008;150(1-2):14–20. doi: 10.1016/j.jviromet.2008.02.009. [DOI] [PubMed] [Google Scholar]
- Dower K, Rubins KH, Hensley LE, Connor JH. Development of Vaccinia reporter viruses for rapid, high content analysis of viral function at all stages of gene expression. Antiviral Research. 2011;91(1):72–80. doi: 10.1016/j.antiviral.2011.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dower K, et al. Identification of a pyridopyrimidinone inhibitor of orthopoxviruses from a diversity-oriented synthesis library. Journal of Virology. 2012;86(5):2632–2640. doi: 10.1128/JVI.05416-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Clercq E. Clinical Potential of the Acyclic Nucleoside Phosphonates Cidofovir, Adefovir, and Tenofovir in Treatment of DNA Virus and Retrovirus Infections. Clinical Microbiology Reviews. 2003;16(4):569–596. doi: 10.1128/CMR.16.4.569-596.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prichard MN, Kern ER. Orthopoxvirus Targets for the Development of New Antiviral Agents. Antiviral Research. 2012;10 doi: 10.1016/j.antiviral.2012.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fenner F. A successful eradication campaign. Global eradication of smallpox. Reviews of Infectious Diseases. 1982;4(5):916–930. doi: 10.1093/clinids/4.5.916. [DOI] [PubMed] [Google Scholar]
- Breman JG, Henderson DA. Poxvirus dilemmas--monkeypox, smallpox, and biologic terrorism. The New England. Journal of Medicine. 1998;339(8):556–559. doi: 10.1056/NEJM199808203390811. [DOI] [PubMed] [Google Scholar]
- Baker RO, Bray M, Huggins JW. Potential antiviral therapeutics for smallpox, monkeypox and other orthopoxvirus infections. Antiviral Research. 2003;57:1–2. doi: 10.1016/S0166-3542(02)00196-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rimoin AW, et al. Major increase in human monkeypox incidence 30 years after smallpox vaccination campaigns cease in the Democratic Republic of Congo. Proceedings of the National Academy of Sciences of the U S A. 2010;107(37):16262–167. doi: 10.1073/pnas.1005769107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sodeik B, Griffiths G, Ericsson M, Moss B, Doms RW. Assembly of vaccinia virus: effects of rifampin on the intracellular distribution of viral protein p65. Journal of Virology. 1994;68(2):1103–1114. doi: 10.1128/jvi.68.2.1103-1114.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grosenbach DW, Jordan R, Hruby DE. Development of the small-molecule antiviral ST-246 as a smallpox therapeutic. Future Virology. 2011;6(5):653–671. doi: 10.2217/fvl.11.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broyles SS. Vaccinia virus transcription. Journal of General Virology. 2003;84(9):2293–2303. doi: 10.1099/vir.0.18942-0. [DOI] [PubMed] [Google Scholar]
- Condit RC, Niles EG. Regulation of viral transcription elongation and termination during vaccinia virus infection. Biochimica et Biophysica Acta (BBA) - Gene Structure and Expression. 2002;1577(2):325–336. doi: 10.1016/s0167-4781(02)00461-x. [DOI] [PubMed] [Google Scholar]
- Díaz-Guerra M, Rivas C, Esteban M. Inducible expression of the 2-5A synthetase/RNase L system results in inhibition of vaccinia virus replication. Virology. 1997;227(1):220–228. doi: 10.1006/viro.1996.8294. [DOI] [PubMed] [Google Scholar]
- Cohrs RJ, Condit RC, Pacha RF, Thompson CL, Sharma OK. Modulation of ppp(A2’p)nA-dependent RNase by a temperature-sensitive mutant of vaccinia virus. Journal of Virology. 1989;63(2):948–951. doi: 10.1128/jvi.63.2.948-951.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sánchez-Puig JM, Sánchez L, Roy G, Blasco R. Susceptibility of different leukocyte cell types to Vaccinia virus infection. Virology Journal. 2004;1(1) doi: 10.1186/1743-422X-1-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bengali Z, Satheshkumar PS, Yang Z, Weisberg AS, Paran N, Moss B. Drosophila S2 cells are non-permissive for vaccinia virus DNA replication following entry via low pH-dependent endocytosis and early transcription. PLoS One. 2011;6(2) doi: 10.1371/journal.pone.0017248. [DOI] [PMC free article] [PubMed] [Google Scholar]


