Abstract
Background
Enteric neuromuscular disease is characteristic of several disease states, including Hirschsprung’s disease, esophageal achalasia, Chagas disease, and gastroparesis. Medical therapy for these conditions is limited, and surgical intervention may incur significant morbidity. Alternatively, transplantation of neural progenitor cells may regenerate enteric ganglia. Existing aganglionosis model systems are limited by swift animal demise or by spontaneous regeneration of native ganglia. We propose a novel protocol to induce permanent aganglionosis in a segment of rat jejunum, which may serve as an experimental transplantation target for cellular therapy.
Materials and Methods
This protocol was performed in 17 adult female Sprague-Dawley rats. A laparotomy was performed and a one-centimeter segment of jejunum was isolated from continuity. Among 14 rats, the isolated segments were treated with benzalkonium chloride (BAC) for 20 minutes to induce aganglionosis. Jejunal segment isolation was performed without BAC treatment in three rats. The animals were euthanized at post-treatment days 21-166. Muscle layer diameter was compared among normal, isolated, and BAC-treated isolated jejunal segments. The presence of jejunal ganglia was documented by immunohistochemical staining (IHC) for beta-III tubulin (TUJ1) and S100, markers of neuronal and glial cell lineages, respectively.
Results
Ganglia were identified by IHC in normal and isolated jejunal segments. Isolated segments had significantly hypertrophied smooth muscle layers compared to normal jejunum (diameter 343 ± 53 μm vs 211 ± 37 μm, P<0.0001). BAC-treated jejunal segments had no IHC evidence of ganglionic structures. Aganglionosis was persistent in all specimens up to 166 days post-treatment.
Conclusions
The exclusion of a jejunal segment from continuity and concurrent treatment with BAC results in an effective, reproducible, and permanent model of aganglionosis. Muscular hypertrophy and aganglionosis in the isolated jejunal segment make it an ideal recipient site for transplantation of neuroglial precursor cells.
Keywords: Hirschsprung’s disease, Aganglionosis, Regenerative medicine, Stem cell, Cell-based therapy, Aganglionosis
Introduction
A broad spectrum of disease processes results in enteric neuromuscular dysfunction. Esophageal achalasia, gastroparesis, Chagas disease, scleroderma, parkinsonism, and Hirschsprung’s disease are all characterized by neurogenic failure of peristaltic motility or secretory function. In each of these disease entities, spontaneous functional restoration is implausible; however, therapeutic options are often extremely limited. Medical therapeutic options for each of these conditions are limited, and their outcomes are variable.1-5 Surgical correction of achalasia and Hirschsprung’s disease, while potentially definitive, entails the risk of developing bleeding, infectious, or functional complications.1,5-6 Cell-based alternatives may be an effective treatment strategy for these conditions.
Recently, investigators have demonstrated that cells with neurogenic potential successfully engraft, survive, migrate, and differentiate into neurons and glia within the aganglionic intestine.7-10 Preliminary evidence suggests that transplanted cell-based therapies may regenerate enteric function.9,11 Nevertheless, while cell therapy is a promising avenue of scientific inquiry, existing experimental model systems are limited by a number of variables.
Current model systems of neurogenic enteric dysfunction include a variety of genetic manipulations. In a 2007 review, Heanue and colleagues summarized the extent of existing genetic knockout models of intestinal and colonic aganglionosis, citing among others mutations in Ret, Ednrb, Edn3, Gdnf, and Sox10 genes.12 These genetic models effectively mimic several phenotypes common to Hirschsprung’s disease. Animals with mutations sufficient to produce an aganglionic phenotype often succumb early in life.12-13 Consequently, studies of cell therapy are time-limited using these models.
Alternatively, trans-serosal chemical denervation using benzalkonium chloride (BAC) effectively renders the bowel aganglionic. This procedure was first described by Sato and colleagues in 1978 and corroborated the following year via histological analysis.14-15 The advantages of this procedure include the ability to control the extent of induced aganglionosis, resultant muscularis propria hypertrophy, and preservation of muscularis contractility.16-19 Because of the small bowel’s length and mobility within the abdomen, several investigators established an aganglionosis model by chemically denervating jejunum and ileum.10,16-22 While these model systems are relatively expedient to establish, they are comparable to few pathologic conditions. Other models of neurogenic dysfunction have been described throughout the alimentary tract. In 1989, Goto and Grosfeld showed that esophageal denervation using BAC mimicked achalasia.23 Several studies have also described BAC treatment of the colon and rectum to simulate Hirschsprung’s disease.7-9,11,14-15,24-27 The model systems in these studies have become standard for experiments of regenerative therapy; nevertheless, they are also limited.
As with genetic knockout models, in situ chemical denervation models are limited by the animal’s reduced lifespan. When the gastrointestinal viscus is exposed to BAC, the animal subsequently experiences proximal visceral distention and significant weight loss, and most succumb within weeks.8,10,14-15 Furthermore, the denervation achieved by BAC exposure in situ may be transient. Investigations of the long-term effects of BAC on intestinal tissue revealed spontaneous regeneration of enteric ganglia among surviving animals.21,22,25 The origin of this so-called “neuroplasticity” effect is indistinct; however, it may be attributable to either proliferation of ganglion cells from adjacent bowel or from surviving precursors within the denervated segment. Notwithstanding the source, the potential for spontaneous gangliogenesis may interfere with investigations of exogenous cell-based therapies. Finally, studies of cell transplantation in denervated bowel explants eliminate physiologic processes that may exert effects on the delicate cellular milieu. In the present study, we describe a protocol of chemical denervation resulting in intestinal aganglionosis. Unlike previously described chemical denervation models, ours is the first to be performed in an isolated intestinal segment and to preserve the animal’s alimentary function. This model has the advantages of permanent denervation and preservation of the animal’s lifespan, thereby permitting long-term assessment of cell-based therapies.
Materials and Methods
Animals
The following was conducted with the approval of our Institutional Animal Care and Use Committee under protocol #2006-061. Adult female Sprague-Dawley (n=17) rats at 3 months of age were purchased from Charles River Laboratories (Wilmington, MA).
Operative Procedure
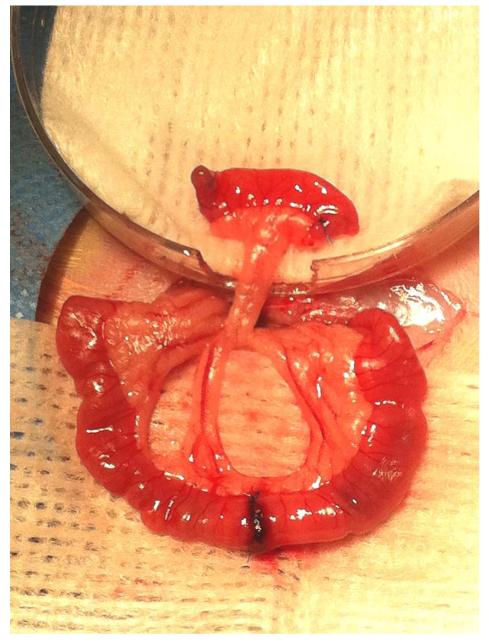
Each rat received a midline laparotomy under inhaled isoflurane anesthesia. The jejunum was transected twice to isolate a 1-cm discontinuous segment and its mesentery, approximately 10-15 cm caudal to the ligament of Treitz. In 14 rats, the isolated segment was wrapped with gauze soaked with benzalkonium chloride (BAC, 0.2% w/v, Sigma-Aldrich) solution for 20 minutes. In the remaining three rats, a 1-cm jejunal segment was isolated from continuity without BAC exposure. The ends of the isolated segment were closed and an end-to-end bypass jejunojejunostomy was performed with 6-0 polypropylene suture to restore intestinal continuity (Figure 1). The isolated segment was irrigated with 0.9% normal saline and wrapped with omentum. Upon replacement of the bowel into the peritoneal cavity, the abdominal fascia was closed with 3-0 polyglactin, and the skin closed with 3-0 nylon suture. A standard solid feeding regimen and trimethoprim sulfa (TMS, 1% v/v) were administered postoperatively for 14 days. Rat jejuna were procured 21-166 days after BAC treatment (Table 1). The early time point of 21 days was selected to allow for sufficient smooth muscle hypertrophy. Rats were sacrificed incrementally thereafter to the endpoint of 166 days. This final time point was selected arbitrarily to represent the extremely long-term condition. Retrieved jejunal segments were fixed in 10% formalin (Fisher Scientific) for 24 hours at 4°C.
Figure 1.
Jejunal segment isolation and bypass jejunojejunostomy. Gauze soaked in benzalkonium chloride is wrapped circumferentially around the isolated segment while the bypass anastomosis is created.
Table 1.
Presence of Ganglia and Axons over Time
| Animal ID | Procedure | Days Post- procedure |
Ganglia | Axons |
|---|---|---|---|---|
| 01 | Isolation | 21 | Present | Present |
| 02 | Isolation | 28 | Present | Present |
| 03 | Isolation | 58 | Present | Present |
| 04 | Denervation | 21 | Absent | Present |
| 05 | Denervation | 28 | Absent | Present |
| 06 | Denervation | 39 | Absent | Absent |
| 07 | Denervation | 66 | Absent | Absent |
| 08 | Denervation | 73 | Absent | Absent |
| 09 | Denervation | 86 | Absent | Absent |
| 10 | Denervation | 87 | Absent | Absent |
| 11 | Denervation | 93 | Absent | Absent |
| 12 | Denervation | 94 | Absent | Absent |
| 13 | Denervation | 107 | Absent | Absent |
| 14 | Denervation | 107 | Absent | Absent |
| 15 | Denervation | 114 | Absent | Absent |
| 16 | Denervation | 114 | Absent | Absent |
| 17 | Denervation | 166 | Absent | Absent |
Isolation refers to jejunal isolation procedure in the absence of BAC
Denervation refers to a jejunal isolation procedure with administration of BAC
Immunohistochemistry
Fixed tissue intended for immunohistochemical staining was embedded in paraffin wax and cut into 5-μm sections. Paraffin was dissolved in xylenes for 10 minutes, followed by serial washes in 100%, 95%, 70% and 0% ethanol for two minutes each (Fisher Scientific). Slides were incubated for 20 minutes at 95°C in citrate buffer (Biogenex, San Ramon, CA) for antigen retrieval, then cooled in a water bath for 30 minutes. A hydrophobic PAP pen (Vector Laboratories, Burlingame, CA) barrier was placed on each section. Slides were incubated in a blocking solution of 5% normal goat serum (Vector Laboratories) and 2% bovine serum albumin in PBS with 0.05% Tween-20 (PBS/T) for one hour at room temperature. Primary antibodies were anti-S100 (1:200; Dako), anti-neuron specific β-III-tubulin (5 ug/ml; Abcam), and anti-glial fibrillary acid protein (1:400; Sigma-Aldrich). Slides with diluted antibody suspensions were incubated in a humidified slide chamber at 4°C overnight. Slides were washed three times for 30 minutes in PBS/T. Secondary antibodies were diluted in PBS/T and exposed to the tissue sections for 30 minutes at room temperature. Slides were washed three more times with PBS/T for 30 minutes. Secondary antibodies included goat anti-rabbit Alexa Fluor® 488, goat anti-mouse Alexa Fluor® 488, goat anti-rabbit Alexa Fluor® 594, and goat anti-mouse Alexa Fluor® 594 (all 1:200; Invitrogen). Each section was treated with Prolong Gold with DAPI (Invitrogen) and covered with a glass coverslip.
Microscopic Tissue Evaluation
Muscularis propria thickness was measured on cross-sectional slides stained with hematoxylin and eosin (H&E), and compared between normal, isolated, and isolated BAC-treated specimens using unpaired t-tests. Between 20 and 30 immunohistochemically stained tissue sections per animal were visualized under fluorescence microscopy (Leica Microsystems, Bannockburn, IL) to contrast the presence of ganglia among normal, isolated, and isolated BAC-treated jejunal specimens.
Results
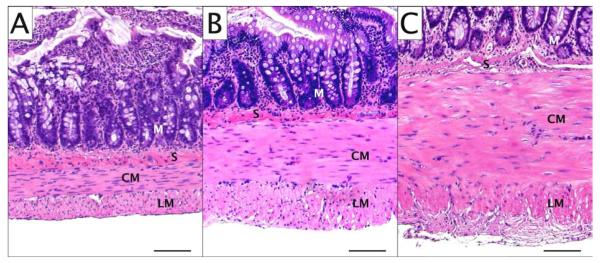
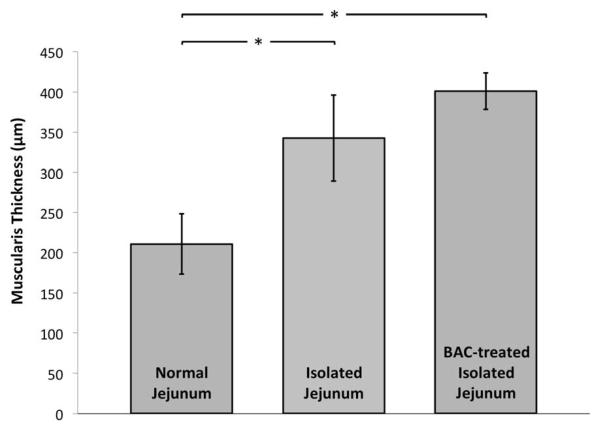
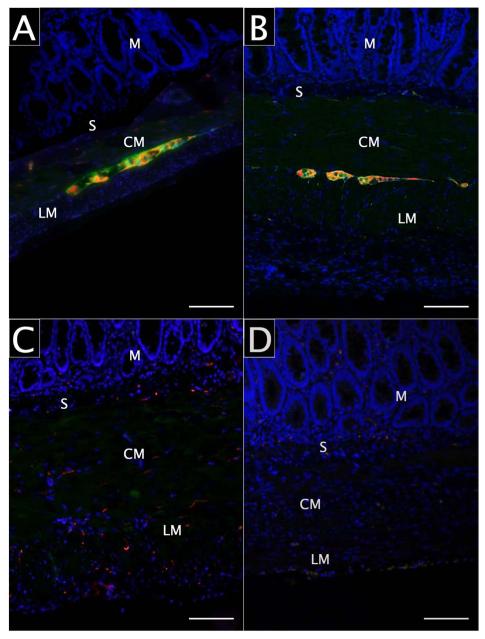
All rats underwent successful laparotomy, jejunal segment isolation, and BAC treatment as intended. No rat succumbed prior to planned sacrifice and tissue retrieval. Histologic sections of normal, isolated, and BAC-treated isolated jejunal segments are shown in Figure 2. Segmental isolation alone (without BAC treatment) resulted in increased muscularis propria thickness (343 vs 211μm; P<0.001) with discernable myenteric and submucosal ganglia (Figure 3). Isolated segments treated with BAC also demonstrated muscularis thickening (Figure 3) compared to normal jejunum (401 vs 211μm; P<0.001). Neuronal and glial cell bodies were immunohistochemically undetectable in any layer of isolated BAC-treated jejunal segments at 28 days, while axonal elements persisted in the smooth muscle layers (Figure 4.C). At 166 days, neither intramuscular axons nor ganglia were identifiable in any layer of the jejunal wall (Figure 4.D). Table 1 demonstrates the presence of ganglion cells and intramuscular axons in experimental tissue segments over time.
Figure 2.
Jejunal muscularis propria hypertrophy, hematoxylin and eosin (H&E) staining. A) Normal jejunum; B) Isolated jejunum; C) Isolated BAC-treated jejunum. Scale bars represent 100 μm. M: mucosa, S: submucosa, CM: circular smooth muscle, LM: longitudinal smooth muscle.
Figure 3.
Muscularis propria thickness in normal, isolated, and isolated BAC-treated jejunum.
Figure 4.
Jejunal aganglionosis. A) Normal jejunum; B) Isolated jejunum, 28 days; C) Isolated BAC-treated jejunum, 28 days; D) Isolated BAC-treated jejunum, 166 days. S100 appears green, β-III-tubulin (TUJ1) appears red, DAPI appears blue, and scale bars represent 100 μm. M: mucosa, S: submucosa, CM: circular smooth muscle, LM: longitudinal smooth muscle.
At all time points, when externally compressed by grasping forceps, the isolated segments undergo isolated tonic contraction at the site of stimulus that neither propagates nor remits. In the postoperative period, the animals lost an average of 5% body weight in the first two days after laparotomy, and then achieved their preoperative weights within 12 days. All animals continued to gain weight at a 2-4% weekly rate thereafter, and none succumbed prior to planned euthanasia.
Discussion
We found that neuronal and glial cell bodies are effectively obliterated, while intramuscular axonal elements are identifiable 28 days after segmental bowel isolation and BAC exposure. Several months later, the muscularis propria and myenteric layers remain denervated, and no axons are perceptible. We conclude that jejunal isolation and exposure to BAC results in irreversible aganglionosis. The denervation induced by BAC entails an immunologic process that takes place after an initial chemical insult to the neuronal cell body.16-17,20 While lymphocyteand macrophage-mediated cell body degeneration has been reported to take place within 1-2 weeks, our observations suggest that subsequent axonal degeneration occurs between 4-6 weeks after BAC exposure.
The extent of muscularis hypertrophy we observed after jejunal treatment with BAC further substantiates our colleagues’ prior observations.28-30 In combination with segmental isolation, we observed BAC denervation produced a 90% increase in muscularis thickness. Our observations of jejunal isolation and denervation suggest substantial hypertrophy develops to counteract the radial force generated by accumulating mucus within the isolated segment; however, the hypertrophy we observed over time had no discernable predilection for either the circular or longitudinal smooth muscle layer. Previous studies of BAC denervation in situ reported that smooth muscle hypertrophy predominates in the longitudinal smooth muscle layer, and the overall degree of hypertrophy is considerably less than what we have observed.18-19 We must conclude that the differential development of hypertrophy in denervated smooth muscle layers is unpredictable when the intestinal segment is isolated from continuity.
Jejunal muscularis hypertrophy is a favorable characteristic for the purposes of cell delivery, as broad recipient tissue thickness permits controlled intramuscular injection.10 Furthermore, smooth muscle function is preserved, as the muscular tissue remains contractile after BAC treatment, while the absence of peristalsis owes to its denervation.16 To our knowledge, the ideal recipient muscularis thickness profile for transplanted cell engraftment has not been established. Nevertheless, we have demonstrated the utility of this model system as a recipient for transplanted skin-derived precursor cells (SKPs) to regenerate enteric ganglia.31 It remains unclear whether muscularis hypertrophy will influence our ability to detect motility changes induced by cellular regenerative therapies. We anticipate the behaviors of a variety of cellular therapeutic agents within this recipient model will require significant scientific scrutiny.
An additional advantage of this experimental model is that the animal remains free of the deleterious effect of aganglionosis in continuity. When a segment of intestine is rendered aperistaltic, the proximal bowel dilates as intraluminal secretions accumulate. Severe distention of the intestine results in ineffective muscle contraction and the development of persistent ileus. The animal becomes incapable of deriving nutrition from digestion of food, loses weight, and ultimately perishes. In our model, isolation of the experimental jejunal segment and intestinal bypass preserve the animal’s alimentary function, resulting in normal weight gain. Accordingly, in vivo experimentation may continue for several months after segmental denervation.
In studies of the long-term effects of in situ chemical denervation, spontaneous gangliogenesis was observed as early as 45-60 days after BAC treatment.21-22,25 Adjacent jejunum represents the only potential source of spontaneous gangliogenesis that would be inhibited by our isolation procedure. The absence of spontaneous gangliogenesis at all time points in our study suggests that this phenomenon indeed derives from adjacent ganglionic bowel. This mechanism of neuroplastic regeneration remains to be characterized further. Nevertheless, our model prohibits the spontaneous migration of adjacent ganglionic precursors, resulting in ostensibly permanent aganglionosis.
Our model of intestinal aganglionosis is particularly pertinent for trials of regenerative treatment. Using this model, we are now capable of evaluating the long-term potential of cell-based therapy to thrive and to restore intestinal function in vivo. We infer that stable aganglionosis is attainable throughout the intraabdominal gastrointestinal tract using a similar method of isolation from continuity, chemical denervation, and bypass anastomosis.
Acknowledgements
We thank the California Institute of Regenerative Medicine and the Eli and Edythe Broad Stem Cell Research Center at UCLA for support. Additional support was provided by NIH R01 DK083319 and the Sun West Company.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Contributions include conception and experimental design, data collection, data interpretation and analysis, manuscript writing, and obtaining funding.
Contributions include conception and experimental design, data interpretation and analysis, and critical revision of the manuscript.
Contributions include conception and experimental design, data collection, data interpretation and analysis, critical revision of the manuscript, and obtaining funding.
Contributor Information
Justin P. Wagner, Department of Surgery, University of California, Los Angeles.
Veronica F. Sullins, Department of Surgery, University of California, Los Angeles.
James C. Y. Dunn, Department of Surgery, Division of Pediatric Surgery, and Department of Bioengineering, University of California, Los Angeles.
References
- 1.Villenueve PJ, Sundaresan RS. Dysphagia. In: Ashley SW, editor. ACS Surgery: Principles & practice [Online] Decker; Hamilton, ON, Canada: Sect. 2010. pp. 1–12. Ch 1. [Google Scholar]
- 2.Tan SA, Sarosi GA. Gastrointestinal motility disorders. In: Mulholland MW, Lillemoe KD, Doherty GM, et al., editors. Greenfield’s surgery: Scientific principles & practice. 5th ed Lippincott Williams & Wilkins; Philadelphia: 2010. pp. 1026–37. [Google Scholar]
- 3.Chokhavatia S, Anuras S. Neuromuscular disease of the gastrointestinal tract. Am J Med Sci. 1991;301:201–14. doi: 10.1097/00000441-199103000-00010. [DOI] [PubMed] [Google Scholar]
- 4.Camilleri M. Diagnosis and treatment of enteric neuromuscular diseases. Clin Auton Res. 2003;13:10–5. doi: 10.1007/s10286-003-0073-y. [DOI] [PubMed] [Google Scholar]
- 5.Sato TT, Oldham KT. Pediatric abdomen. In: Mulholland MW, Lillemoe KD, Doherty GM, et al., editors. Greenfield’s surgery: Scientific principles & practice. 5th ed Lippincott Williams & Wilkins; Philadelphia: 2010. pp. 1884–945. [Google Scholar]
- 6.Georgeson KE. Hirschsprung’s disease. In: Holcomb GW, Murphy JP, Ostlie DJ, editors. Ashcraft’s Pediatric Surgery. Elsevier Health Sciences; Philadelphia: 2010. pp. 456–67. [Google Scholar]
- 7.Liu W, Wu RD, Dong YL, et al. Neuroepithelial stem cells differentiate into neuronal phenotypes and improve intestinal motility recovery after transplantation in the aganglionic colon of the rat. Neurogastroenterol Motil. 2007;19:1001–9. doi: 10.1111/j.1365-2982.2007.00981.x. [DOI] [PubMed] [Google Scholar]
- 8.Shu X, Meng Q, Jin H, et al. Treatment of aganglionic megacolon in mice via neural stem cell transplantation. Mol Neurobiol. 2013;48:429–37. doi: 10.1007/s12035-013-8430-x. [DOI] [PubMed] [Google Scholar]
- 9.Pan WK, Zheng BJ, Gao Y, et al. Transplantation of neonatal gut neural crest progenitors reconstructs ganglionic function in benzalkonium chloride-treated homogenic rat colon. J Surg Res. 2011;167:e221–30. doi: 10.1016/j.jss.2011.01.016. [DOI] [PubMed] [Google Scholar]
- 10.Geisbauer C, Wu BM, Dunn JCY. Transplantation of enteric cells into the aganglionic rodent small intestines. J Surg Res. 2012;176:20–8. doi: 10.1016/j.jss.2011.05.014. [DOI] [PubMed] [Google Scholar]
- 11.Dong YL, Liu W, Gao YM, et al. Neural stem cell transplantation rescues rectum function in the aganglionic rat. Transpl Proc. 2008;40:3646–52. doi: 10.1016/j.transproceed.2008.06.107. [DOI] [PubMed] [Google Scholar]
- 12.Heanue TA, Pachnis V. Enteric nervous system development and Hirschsprung’s disease: Advances in genetic and stem cell studies. Nat Rev Neurosci. 2007;8:466–79. doi: 10.1038/nrn2137. [DOI] [PubMed] [Google Scholar]
- 13.Won KJ, Torihashi S, Mitsui-Saito M, et al. Increased smooth muscle contractility of intestine in the genetic null of the endothelin ETB receptor: A rat model for long segment Hirschsprung’s disease. Gut. 2002;50:355–60. doi: 10.1136/gut.50.3.355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sato A, Yamamoto M, Imamura K, et al. Pathophysiology of aganglionic colon and anorectum: An experimental study on aganglionosis produced by a new method in the rat. J Pediatr Surg. 1978;13:399–405. doi: 10.1016/s0022-3468(78)80464-3. [DOI] [PubMed] [Google Scholar]
- 15.Sakata K, Kunieda T, Furuta T, et al. Selective destruction of intestinal nervous elements by local application of benzalkonium solution in the rat. Experientia. 1979;35:1611–3. doi: 10.1007/BF01953222. [DOI] [PubMed] [Google Scholar]
- 16.Fox DA, Bass P. Pharmacological characterization of rat jejunal contractility after chronic ablation of the myenteric plexus. J Pharmacol Exp Ther. 1986;238:372–7. [PubMed] [Google Scholar]
- 17.Fox DA, Epstein ML, Bass P. Surfactants selectively ablate enteric neurons of the rat jejunum. J Pharmacol Exp Ther. 1983;227:538–44. [PubMed] [Google Scholar]
- 18.Holle GE. Changes in muscularis externa of rat small intestine after myenteric ablation with benzalkonium chloride: Electron microscopic and morphometric study. Dig Dis Sci. 1998;43:2666–75. doi: 10.1023/a:1026603327807. [DOI] [PubMed] [Google Scholar]
- 19.Michopoulou A, Triggas B, Hra E, et al. Early intestinal morphological changes following benzalkonium chloride treatment in a rat model of short bowel syndrome. Ann Ital Chir. 2009;80:135–9. [PubMed] [Google Scholar]
- 20.Parr EJ, Sharkey KA. Multiple mechanisms contribute to myenteric plexus ablation induced by benzalkonium chloride in the guinea-pig ileum. Cell Tissue Res. 1997;289:253–64. doi: 10.1007/s004410050872. [DOI] [PubMed] [Google Scholar]
- 21.Ramalho FS, Santos GC, Ramalho LNZ, et al. Myenteric neuron number after acute and chronic denervation of the proximal jejunum induced by benzalkonium chloride. Neurosci Lett. 1993;163:74–6. doi: 10.1016/0304-3940(93)90232-a. [DOI] [PubMed] [Google Scholar]
- 22.Luck MS, Dahl JL, Boyeson MG, et al. Neuroplasticity in the smooth muscle of the myenterically and extrinsically denervated rat jejunum. Cell Tissue Res. 1993;271:363–74. doi: 10.1007/BF00318623. [DOI] [PubMed] [Google Scholar]
- 23.Goto S, Grosfeld JL. The effect of a neurotoxin (benzalkonium chloride) on the lower esophagus. J Surg Res. 1989;47:117–9. doi: 10.1016/0022-4804(89)90073-5. [DOI] [PubMed] [Google Scholar]
- 24.Yoneda A, Shima H, Nemeth L, et al. Selective chemical ablation of the enteric plexus in mice. Pediatr Surg Int. 2002;18:234–7. doi: 10.1007/s003830100681. [DOI] [PubMed] [Google Scholar]
- 25.Garcia SB, Demarzo MMP, Vinhadeli WS, et al. No reduction with ageing of the number of myenteric neurons in benzalkonium chloride treated rats. Neurosci Lett. 2002;331:66–8. doi: 10.1016/s0304-3940(02)00832-7. [DOI] [PubMed] [Google Scholar]
- 26.Qin HH, Lei N, Mendoza J, et al. Benzalkonium chloride-treated anorectums mimicked endothelin-3-deficient aganglionic anorectums on manometry. J Pediatr Surg. 2010;45:2408–11. doi: 10.1016/j.jpedsurg.2010.08.045. [DOI] [PubMed] [Google Scholar]
- 27.Xiong HL, Guo ZY, Li SX, et al. In vivo detection of Hirschsprung’s disease by optical coherence tomography in rats. Phys Med Biol. 2013;58:1549–61. doi: 10.1088/0031-9155/58/5/1549. [DOI] [PubMed] [Google Scholar]
- 28.Jabaji Z, Stark R, Dunn JCY. Regeneration of enteric ganglia in mechanically lengthened jejunum after restoration into intestinal continuity. J Pediatr Surg. 2013;48:118–23. doi: 10.1016/j.jpedsurg.2012.10.027. [DOI] [PubMed] [Google Scholar]
- 29.Shekherdimian S, Scott A, Chan A, et al. Intestinal lengthening in rats after massive small intestinal resection. Surgery. 2009;146:291–5. doi: 10.1016/j.surg.2009.03.034. [DOI] [PubMed] [Google Scholar]
- 30.Shekherdimian S, Panduranga MK, Carman GP, et al. The feasibility of using an endoluminal device for intestinal lengthening. J Pediatr Surg. 2010;45:1575–80. doi: 10.1016/j.jpedsurg.2010.03.015. [DOI] [PubMed] [Google Scholar]
- 31.Wagner JP, Sullins VF, Dunn JCY. Transplanted skin-derived precursor stem cells generate enteric ganglion-like structures in vivo. J Pediatr Surg. 2014 doi: 10.1016/j.jpedsurg.2014.01.061. [In Press] [DOI] [PMC free article] [PubMed] [Google Scholar]