Abstract
Background:
Junctional ectopic tachycardia occurs frequently after congenital cardiac surgery and can be a cause of increased morbidity and mortality. Dexmedetomidine (DEX) is an α2 adrenoreceptor agonist, has properties of controlling tachyarrhythmia by regulating the sympatho-adrenal system.
Objective:
To evaluate the efficacy of DEX for control of junctional ectopic tachycardia after repair of Tetralogy of Fallot (TOF).
Materials and Methods:
Two hundred and twenty pediatric cardiac patients with TOFs were enrolled in a prospective randomized control study. Patients underwent correction surgery. They were divided into two groups, i.e., Group 1 (DEX) and Group 2 (control). Heart rate, rhythm, mean arterial pressure (MAP) were recorded after the anesthetic induction (T1), after termination of bypass (T2), after 04 hours (T3), and 08 hours after transferring the patient to intensive care unit (ICU; T4).
Results:
Heart rate was comparable between two groups before starting the drug but statistically significant after bypass until 08 hours after transferring the patient to ICU. Junctional ectopic tachycardia occurred more in Group-2 (20%) as compared to Group-1 (9.09%; P = 0.022). Junctional ectopic tachycardia occurs early in Group-2 (0.14 ± 0.527 hours) as compared to Group 1 (0.31 ± 1.29 hours; P = 0.042). The duration of junctional ectopic tachycardia was more prolonged in Group-2 (1.63 ± 3.64 hours) as compared to Group-1 (0.382 ± 1.60 hours; P = 0.012). The time to withdraw from mechanical ventilation and ICU stay of Group 1 patient was less than of Group 2 patients (P = <0.001).
Conclusion:
DEX had a therapeutic role in the prevention of junctional ectopic tachycardia in patients undergoing repair for TOF.
Keywords: Congenital heart disease, dexmedetomidine, junctional ectopic tachycardia, tetralogy of Fallot, tachyarrhythmias
INTRODUCTION
Atrial and junctional tachyarrhythmias occur frequently after congenital cardiac surgery and can be a cause of increased morbidity and mortality. These rhythm disturbances often cause significant hemodynamic instability in patients with congenital heart defects and particularly after surgery of congenital heart disease. Dexmedetomidine (DEX) is an α2 adrenoreceptor agonist and has been successfully used in cardiac intensive care unit (CICU) for sedation and analgesia of infants and children after cardiac surgery.[1] It has a relatively high ratio of α2/α1 activity (1620:1 as compared to 220:1 for clonidine) and is therefore considered a full agonist of α2 receptor. This ratio ensures that its potent action is selective for the central nervous system, without unwanted cardiovascular effects from α1 receptor activation.
DEX has activity at the imidazoline receptors involved in central arterial blood pressure control.[2,3] Administration of a bolus drug result in a brief rise in the mean arterial blood pressure from α1 receptor binding, later during continuous infusion, it causes a dose-dependent decrease in mean arterial blood pressure (MAP) and heart rate (HR) and a reduction in sympathetic nervous system activity.[4] DEX also depresses both sinus and atrioventricular (AV) nodal function.[5] These properties of DEX may be effective in reducing the tachyarrhythmias after congenital heart surgery. Hence, we hypothesized that DEX will be effective in preventing junctional ectopic tachycardia (JET) after intracardiac repair of Tetralogy of Fallot (TOF).
Literature search revealed very few studies on the use of DEX in prevention of tachyarrhythmia such as JET in pediatric congenital cardiac surgery patients. Hence, we conducted the study to assess the efficacy of perioperative DEX therapy on prevention of JET after repair surgery of TOF.
MATERIALS AND METHODS
We conducted the prospective randomized double blind control study in 220 consecutive patients who underwent correction surgery for TOF after obtaining approval from the institute ethics committee and informed consent from the child's parents, between September 2012 and December 2013. The patients were allocated into two groups. The consistency in the surgical, anesthetic, and post-operative approach, during the study period was ensured the same team of cardiothoracic surgeons, cardiac anesthesiologists, and cardiac intensivist.
Inclusion criteria-Patients with TOF for intra-cardiac repair of age between 1-6 years were enrolled in the study. Exclusion criteria were patients undergoing re-operations, deep hypothermic circulatory arrest, renal dysfunction (Serum creatinine >2 mg/dl), pre-existing bleeding and coagulation abnormalities, patient with severe liver dysfunction, post-operative persistent heart block after weaning from cardiopulmonary bypass (CPB).
Patients were premedicated with syrup promethazine 1 mg/kg. Sevoflurane in oxygen inhalation was used to place intravenous line and anesthesia was induced with ketamine 2 mg/kg, 1-2µg/kg fentanyl and 1 mg/kg rocuronium to facilitate tracheal intubation. Anesthesia was maintained using sevoflurane 1-2% in an oxygen-air mixture (1:1 ratio), fentanyl, midazolam, and vecuronium top-ups. Mechanical ventilation was provided by using a tidal volume of 10 mL/kg with the respiratory rate adjusted according to age, aiming to maintain PaCO2 between 30 and 35 mm Hg. Methyl prednisolone was administered in the dose of 30 mg/kg prior to skin incision and epsilon amino caproic acid (EACA) was administered in the dose of 100 mg/kg before, during and after CPB. Before initiation of CPB, heparin was administered into a central vein in the dosage of 400 IU/kg, to maintain the Kaolin-activated clotting time (ACT) above 480 seconds.
After insertion of arterial and central venous catheter, patients were allocated into either of two groups (n = 110). The patients were placed in either of two groups by drawing of a chit by operating room assistant, who was blinded to the study. The infusion was prepared by the same person. In Group 1, patient received an initial bolus of DEX (0.5 µg/Kg) over 10 minutes followed by a continuous infusion of 0.5 µg/Kg/hr. The infusion was continued throughout the operation upto weaning from ventilator in intensive care unit (ICU). In Group 2, a similar volume of normal saline was given in control group.
All patients were monitored using full disclosure telemetry (Philips Intellivue MP80) throughout the surgery until 08 hours after transferring the patient to ICU. Hemodynamic variables that included heart rate, rhythm, and MAP were measured at four time points, after anesthetic induction (T1), after termination of cardiopulmonary bypass (T2), 04 hours after transferring the patient to ICU (T3), and 08 hours after transferring the patient to ICU (T4). The development of JET episode was monitored by Holter and continuous electrocardiography (ECG) for 08 hours after weaning from CPB. JET was defined as narrow QRS complex tachycardia in the absence of surgically induced right bundle branch block, heart rate >170 beats per minute with baseline QRS complex, atrioventricular dissociation with a ventricular rate faster than the atrial rate. The diagnosis of JET was based on surface ECGs and atrial wire studies were not performed. The temperature of the child was monitored throughout the study period and any period of temperature >37°C (fever) was noted.
In all patients, a median sternotomy was performed. CPB was initiated after standard aorta-bicaval cannulation. Anesthetic and surgical management was standardized in both groups. The same surgical team performed all operations and they were blinded to the study protocol. A membrane oxygenator was used for all patients during the CPB. The CPB circuit was primed with lactated ringer solution 20 ml/kg, sodium bicarbonate7.5%, 1 ml/kg, mannitol 20%, 0.5 g/kg and 100 IU/kg of heparin. If required, packed red blood cell (PRBC) was added to pump volume during CPB and to maintain hematocrit (HCT) > 25%. All patients were cooled to 32°C while on CPB. Myocardial protection was achieved by using blood cardioplegia solution with a cardioplegia delivery system. Systemic pump flows was maintained between 2-2.4 liter/m2 body surface areas. After separation from CPB, protamine sulphate was administered in a dose of 1.3 mg per 100 IU of heparin to antagonize the effects of heparin.
Post CPB, appropriate inotrope and dilators were added to maintain patient's hemodynamic. A vasoactive inotrope score was calculated based on the type and dosage of inotrope utilized at the time of onset of arrhythmias using the following formula designed to account for relative potencies of various inotropes.[6]
[(dopamine+dobutamine)µg/Kg/min × 1] + (Milrinone µg/Kg/min × 20) + [(Epinephrine + Norepinephrine)µg/Kg/min × 100]2
Total amount of time patient spent on mechanical ventilation until successful extubation were taken as time of extubation. The criteria for tracheal extubation was conscious and responsive patient, adequate motor power, peripheral skin temperature > 33°C, stable hemodynamic parameters, no continuing bleeding, paO2 >60 mmhg, paCO2 <40 mmhg, and electrolytes within normal limits. The duration of ICU stay in hours after surgery was recorded in all patients. Electrolytes and blood gases were analyzed and normalized at 10 minutes after the administration of DEX, intraoperative period, after termination of CPB and after every 04 hours, till 08 hours after shifting the patient to ICU. Patients who developed JET and were hemodynamically unstable were managed with mild hypothermia, reduction in inotropes, magnesium, digoxin, and amiodarone.
Statistical analysis
Power analysis was performed on JET because it was main outcome variable; incidence of JET is approximately 22% from the study of Khatami A et al.[7] , with a power of 80% and α error of 5%; and to detect 50% difference in JET in two groups, a sample size of 110 patients in each group should be adequate.
Statistical comparison between data was analyzed using the Chi-square test. Quantitative data like duration of JET, ICU stay, and inotropic score were analyzed by Student t-test. Data were expressed as mean ± SD. A value of P < 0.05 was considered statistically significant. One-way analysis of variance (ANOVA) was used for statistical analysis of patient characteristic HR, JET at different time interval. The Benferroni correction was used for post-hoc analysis.
Normal distributed outcome were analyzed using unpaired t-test or ANOVA. If normality was not achieved using a log transformation, we used a non-parametric Mann-Whitney test. Cumulative long-term survival was estimated according to Kaplan Meir method comparing difference between groups with log Rank test. All statistical comparisons were performed using the statistical analysis software statistical package for the social science (SPSS) version 17.0 (SPSS Inc., Chicago, IL).
RESULTS
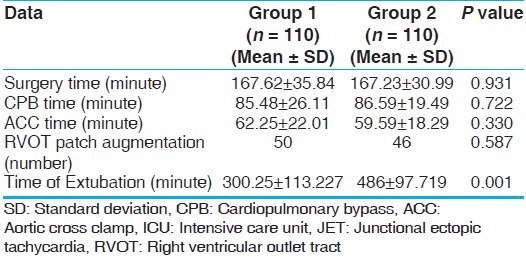
All 220 patients who were enrolled had completed the study. There was no surgical mortality in either group during the study period. Demographic data were comparable between the two groups [Table 1]. The surgical time, CPB time, aortic cross clamp (ACC) time, and number of patients undergoing right ventricular outlet tract (RVOT) resection with patch augmentation were comparable between two groups [Table 2].
Table 1.
Demographic data

Table 2.
Intra-operative and post-operative data
Heart rate between the two groups was statistically significant different at T2, T3, and T4 (P < 0.001) [Table 3]. A repeated measure ANOVA with Greenhouse-Geisser correction determined that HR differed statistically significant between time points (P < 0.005). Post-hoc test by Bonferroni correction revealed that DEX elicited a reduction in heart rate from T1 to subsequent time points (P < 0.001), but there was no significant difference between T2, T3, and T4 (P = 0.53, 0.718, 1.000), respectively when pair wise comparison was done.
Table 3.
Hemodynamic data
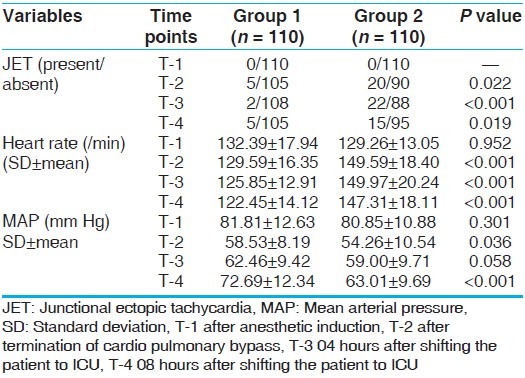
MAP was comparable during T1 between two groups. But MAP was statistically significant in T2 and T4 time interval (58.53 ± 8.19 vs. 54.26 ± 10.54, P < 0.036) and (72.69 ± 12.34 mm Hg vs. 63.01 ± 9.69 mm of Hg, P < 0.001) [Table 3]. The MAP was higher in Group 1 at T3 (62.46 ± 9.42 vs. 59.00 ± 9.71, P < 0.058) but this was not statistically significant.
JET was not detected in either group at T1 after induction of anesthesia. JET occurred significantly more in Group 2 (20%) as compared to Group 1 (9.1%; P = 0.022) and occurred earlier after coming off CPB in Group 2 (0.14 ± 0.527 hours) as compared to Group 1 (0.31 ± 1.29 hours; P = 0.042) [Table 4]. The duration of JET was also more prolonged in Group 2 (1.63 ± 3.64 hours) as compared to Group 1 (0.382 ± 1.60 hours; P = 0.012) [Table 4].
Table 4.
Post-operative outcome of patients
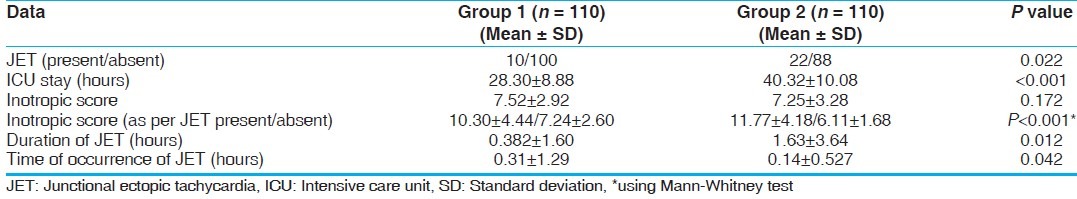
The inotropic score between two groups was not significant (P = 0.172) [Table 4]. But when inotropic score were compared between the patients who developed JET and who did not developed JET using Mann-Whitney test there was statistically significant difference (P < 0.001) [Table 4]. Kaplan-Meir survival analysis for time of JET showed that Group 1 developed JET much later after the surgery (approximately 4 hours post-operative in majority of patients). Group 2 patients experienced JET within 01 hours after weaning from CPB [Figure 1].
Figure 1.
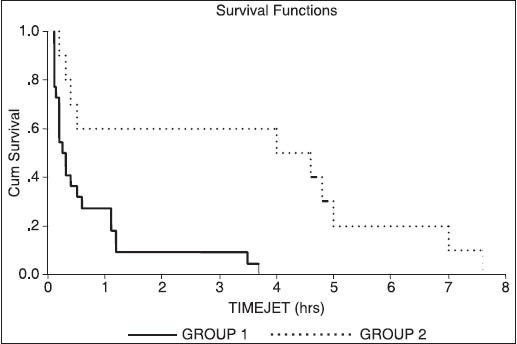
Survival analysis for time of JET. The horizontal axis shows time of occurring JET in hours and vertical axis shows probability of occurring JET. Group 1 experienced JET much later after the surgery (4 hours postop), this group does not show JET between 1-03 hours after coming out of CPB, as compared to patients who did not received Dexmed (Group 2) JET occurred within 01 hours after coming out of cardiopulmonary bypass. (Group 1-Dexmed group, Group 2-Normal saline group)
The patient in Group 1 stayed less time on mechanical ventilation and extubated earlier as compared to Group 2 (P = 0.001). This early extubation leads to early discharge from ICU. Patients in Group 1 stayed for lesser time in ICU as compared to Group 2 (P = 0.001) [Table 4].
DISCUSSION
JET is a tachyarrhythmia related with surgery for congenital heart disease. JET causes serious hemodynamic deterioration in the immediate post-operative period.[8,9] JET is a narrow QRS complex tachycardia with atrioventricular dissociation and slower atrial rate as compared to ventricular rate. Atrioventricular asynchrony and consequent loss of the contribution of atrial contraction to cardiac output can lead to hemodynamic instability.[10,11] The precise mechanism of JET is not known, but it probably results from surgical trauma to the proximal conduction tissue related to suture placement or indirect stretch injury.[12] The incidence of JET has been reported to vary from 8% to 24% depending on patient selection and surgical procedure.[13,14,15] Several risk factors have been associated with JET, both surgical and non-surgical. The surgical risk factors include resection of muscle bundles, correction of RVOT, and closure of ventricular septal defect.[12] The non-surgical risk factors are younger patient, longer CPB time and ACC time, hypomagnesemia, use of catecholamines and hyperthermia.[11,14,15]
DEX is a α2 adrenoreceptor agonist with primarily sedative, analgesic, and anxiolytics properties. The hemodynamic effect of DEX, i.e., hypotension and bradycardia are predictable from the pharmacological of α2 adrenorecptor agonist.[16] However, Chyrosostomou C et al. demonstrated that DEX can be used efficiently and without any significant change in hemodynamic parameters in pediatric cardiac surgery, as demonstrated in our study.[17] The present study demonstrated that use of DEX in pediatric cardiac surgery result in reduction in HR (from tachycardia to normal heart rate) with a concomitant reduction in occurrence of JET.
The incidence of post-operative JET after repair of TOF is relatively high (22%) in our study, it is similar to study by Dodge-Khatani et al. who reported an incidence of 21.9% in patients undergoing TOFs repair.[12,18] Resection of RVOT obstruction was not an independent risk factor for JET, in the present study (P = 0.587) which was demonstrated by Dodge-Khatami et al.[12]
Hammer et al. found that DEX depressed both sinus and atrioventricular (AV) nodal function.[5] Atrioventricular nodal depression seemed to be in agreement with our study. The decrease in heart rate that we witnessed in patients with DEX group was likely secondary to suppression of the AV node, neither our study nor the study by Hammer et al. documented any episode of complete heart block.[5]
Dodge-Khatini A et al.[7] in their study noted that cardiac ICU stay increased from 4.5 days to 8.8 days when compared those without and with post-operative JET, respectively as did mechanical ventilation time (3.5-7.8 days). The present study showed ICU stay and time of extubation was significantly reduced in patients received DEX compare to control group (P < 0.001) due to better hemodynamic without JET.
Chryostosmou et al.[19] retrospectively reviewed their experience with post-operative DEX infusion in pediatric patients undergoing cardiac surgery. DEX was administered in the post-operative period at a dose of 0.1-0.5 µg/Kg/hr for 3-26 hours and they reported successful post-operative sedation in 93% of the patients with absent or minimal pain scores. They also reported that 87% of the patients on DEX infusion were easily weaned and extubated.[19] This finding of DEX for better sedation and analgesia in ICU might have helped the DEX group patients for shorter mechanical ventilation time and ICU stay in our study.
In our study, patient who developed JET have high inotropic score as compared to patient who did not developed JET this is in accordance with study by Hoffman et al. who examined that perioperative risk factor associated with JET were younger age and increase use of dopamine as a risk factors for development of JET.[13]
Limitations
The current study included only TOF patients. Our observation period was only for 08 hours and thus we may have missed JET that occurred afterwards. The diagnosis of JET was based on surface ECG and atrial wire studies were unavailable. This could influence the accuracy of the diagnosis of JET.
In conclusion, the present study suggests that DEX has a therapeutic role in the prevention of JET in patients undergoing intracardiac repair for TOF. DEX therapy results in better control of hemodynamic parameters resulting in lower inotropic score; shorter mechanical ventilation time; and ICU stay.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared
REFERENCES
- 1.Savola JM, Vertanen R. Central alpha-2 adrenoreceptors are highly stereoselective for dexmedetomidine, the dextro enantiomer of medetomidine. Eur J Pharmacol. 1991;195:193–9. doi: 10.1016/0014-2999(91)90535-x. [DOI] [PubMed] [Google Scholar]
- 2.Venn RM, Bradshow CJ, Spencer R. Preliminary UK experience of dexmedetomidine, a novel agent for postoperative sedation in the intensive care unit. Anaesthesia. 1999;54:1136–42. doi: 10.1046/j.1365-2044.1999.01114.x. [DOI] [PubMed] [Google Scholar]
- 3.Khan ZP, Ferguson CN, Jones RM. Alpha-2 and imidazoline receptor agonists: Their pharmacology and therapeutic role. Anaesthesia. 1999;54:146–65. doi: 10.1046/j.1365-2044.1999.00659.x. [DOI] [PubMed] [Google Scholar]
- 4.Kallio A, Scheinin M, Koulu M, Ponkilainen R, Ruskoaho H, Viinamäki O, et al. Effects of dexmedetomidine, a selective alpha-2 adrenoreceptor agonist, on hemodynamic control mechanisms. Clin Pharmacol Ther. 1989;46:33–42. doi: 10.1038/clpt.1989.103. [DOI] [PubMed] [Google Scholar]
- 5.Hammer GB, Drover DR, Cao H, Jackson E, Williams GD, Ramamoorthy C, et al. The effects of dexmedetomidine on cardiac electrophysiology in children. Anesth Analg. 2008;106:79–83. doi: 10.1213/01.ane.0000297421.92857.4e. [DOI] [PubMed] [Google Scholar]
- 6.Gaies MG. Vasoactive-inotropic score as a predictor of morbidity and mortality in infants after cardiopulmonary bypass. Pediatr Crit Care Med. 2010;11:234–8. doi: 10.1097/PCC.0b013e3181b806fc. [DOI] [PubMed] [Google Scholar]
- 7.Dodge-Khatami A, Miller OI, Anderson RH, Gil-Jaurena JM, Goldman AP, de Leval MR. Impact of junctional ectopic tachycardia on postoperative morbidity following repair of congenital heart defects. Eur J Cardiothorac Surg. 2002;21:255–9. doi: 10.1016/s1010-7940(01)01089-2. [DOI] [PubMed] [Google Scholar]
- 8.Till JA, Ho SY, Rowland E. Histopathological findings in three children with his bundle tachycardia occurring subsequent to cardiac surgery. Eur Heart J. 1992;13:709–12. doi: 10.1093/oxfordjournals.eurheartj.a060239. [DOI] [PubMed] [Google Scholar]
- 9.Dorman BH, Sade RM, Burnette JS, Wiles HB, Pinosky ML, Reeves ST, et al. Magnesium supplementation in the prevention of arrhythmias in pediatric patients undergoing surgery for congenital heart defects. Am Heart J. 2000;139:522–8. doi: 10.1016/s0002-8703(00)90097-8. [DOI] [PubMed] [Google Scholar]
- 10.Haas NA, Plumpton K, Justo R, Jalali H, Pohlner P. Postoperative junctional ectopic tachycardia. Z Kardiol. 2004;93:371–80. doi: 10.1007/s00392-004-0067-3. [DOI] [PubMed] [Google Scholar]
- 11.Garson A, Gillette PC. Junctional ectopic tachycardia in children: Electrocardiography, electrophysiology and pharmacologic response. Am J Cardiol. 1979;44:298–302. doi: 10.1016/0002-9149(79)90320-5. [DOI] [PubMed] [Google Scholar]
- 12.Dodge-Khatami A, Miller OI, Anderson RH, Goldman AP, Gil-Jaurena JM, Elliott MJ, et al. Surgical substrates of postoperative junctional ectopic tachycardia in congenital heart defects. J Thorac Cardiovasc Surg. 2002;123:624–30. doi: 10.1067/mtc.2002.121046. [DOI] [PubMed] [Google Scholar]
- 13.Hoffman TM, Bush DM, Wernowsky G, Cohen MI, Wieand TS, Gaynor JW, et al. Postoperative junctional ectopic tachycardia in children: Incidence, risk factors and treatment. Ann Thorac Surg. 2002;74:1607–11. doi: 10.1016/s0003-4975(02)04014-6. [DOI] [PubMed] [Google Scholar]
- 14.Rekawek J, Kansy A, Miszczak-Knecht M, Manowska M, Bieganowska K, Brzezinska-Paszke M, et al. Risk factors for cardiac arrhythmias in children with congenital heart disease after surgical intervention in the early postoperative period. J Thorac Cardiovasc Surg. 2007;133:900–4. doi: 10.1016/j.jtcvs.2006.12.011. [DOI] [PubMed] [Google Scholar]
- 15.Andreasen JB, Johnsen SP, Ravn HB. Junctional ectopic tachycardia after surgery for congenital heart disease in children. Int Care Med. 2008;34:895–902. doi: 10.1007/s00134-007-0987-2. [DOI] [PubMed] [Google Scholar]
- 16.Bloor BC, Ward DS, Belleville JP, Maze M. Effects of intravenous dexmedetomidine in humans: Hemodynamic changes. Anesthesiology. 1992;77:1134–42. doi: 10.1097/00000542-199212000-00014. [DOI] [PubMed] [Google Scholar]
- 17.Chrysostomou C, Sanchez De Toledo J, Avolio T, Motoa MV, Berry D, Morell VO, et al. Dexmedetomidine use in a pediatric cardiac intensive care unit: Can we use it in infants after cardiac surgery? Pediatr Crit Care Med. 2009;10:654–60. doi: 10.1097/PCC.0b013e3181a00b7a. [DOI] [PubMed] [Google Scholar]
- 18.Pfammater JP, Wagner B, Berdat P, Bachmann DC, Pavlovic M, Pfenninger J, et al. Procedural factors associated with early postoperative arrhythmias after repair of congenital heart defects. J Cardiovasc Surg. 2002;123:258–62. doi: 10.1067/mtc.2002.119701. [DOI] [PubMed] [Google Scholar]
- 19.Chryostosmou C, Di Filippo S, Manrique AM, Schmitt CG, Orr RA, Casta A, et al. Use of dexmedetomidine in children after cardiac and thoracic surgery. Pediatr Crit Care Med. 2006;7:126–31. doi: 10.1097/01.PCC.0000200967.76996.07. [DOI] [PubMed] [Google Scholar]


