Introduction
Epidemiology
Bladder cancer is the most common malignancy of the urinary tract and the ninth most common malignancy in the world, with an estimated overall global 5-year prevalence of 1,110,265.1 In the United States, an estimated 74,690 new cases and 15,580 deaths from bladder cancer are expected in 2014.2 Urothelial (transitional cell) carcinoma is the predominant histologic type in the U.S, accounting for 90% of all bladder cancers.
Risk factors
Among a number of identified risk factors for urothelial cancers, environmental exposures account for most cases. Since the urothelial epithelium lining the mucosal surface of the entire urinary tract is exposed to carcinogens excreted in the urine, urothelial carcinoma is thought to arise from a field change (known as “field cancerization effect”) that affects the entire urothelium.
Cigarette smoking is the most prominent cause of bladder cancer, increasing risk an estimated 4-fold for both men and women.3 Exposure to occupational carcinogens is also strongly associated with bladder cancer occurrence. Potential carcinogenic compounds include benzene and polycyclic aromatic hydrocarbons.4,5 Other risk factors include pelvic radiation, cyclophosphamide and thiazolidimediones, chronic infection, and underlying genetic conditions such as Lynch syndrome.6
Clinical presentation
The most common presenting symptom is hematuria, which occurs in about 85% of patients. Hematuria is typically intermittent, gross, and painless. Bladder irritability, usually presenting as urinary frequency, urgency, and dysuria, occurs in about 20% of patients.6 In more advanced disease, patients can experience flank pain secondary to lymphadenopathy or ureter obstruction and various constitutional symptoms such as weight loss, fatigue, and anorexia.
Diagnosis, grading, and staging
Initial diagnostic workup usually involves cystoscopy and urine cytology. Cystoscopy is the gold standard for the initial diagnosis and staging of bladder cancer. If a bladder mass is detected, a transurethral resection of the bladder tumor (TURBT) is performed for full primary tumor staging. The resected bladder tumor specimen should include muscle to fully assess the depth of tumor invasion. If carcinoma in situ (CIS) is detected, multiple random biopsies, including several different areas of the bladder and the prostatic urethra, may be required to assess the extent of involvement.
After resection or biopsy of urothelial cancers by a urologist, a pathologist grades the tumor based on its microscopic appearance and assesses the depth of invasion. Tumors are classified as low-grade or high-grade. This system relies on the degree of atypia, hypertrophy of urothelial cell layers, and orthogonal orientation to stratify tumors into those at low or high risk of recurrence and/or progression. However, this stratification predominantly applies to non-muscle invasive tumors, as muscle invasive tumors by convention are automatically considered to be high grade. Cytology is commonly used as an adjunct to cystoscopy. Urine cytology has excellent specificity for bladder cancer (78.4%–99.4%) but lacks sensitivity (12.2%–79%). As a result, it is often helpful in the setting of high-grade disease, where it is usually positive, but of limited use in the setting of low-grade disease, where it is usually equivocal or negative.7 Its utility depends on the experience of the cytopathologist. Finding positive urine cytology in the absence of any identifiable lesions in the bladder on cystoscopy presents a difficult clinical dilemma. In this setting, further investigation is usually required in the form of upper urinary tract imaging such as CT urography, MR urography, or intravenous pyelography, and/or direct visualization by ureteroscopy, selective ureteral urine cytologies, and/or other urinary tumor markers.
Abdominal imaging with either CT or MRI is recommended in patients with a high-grade tumor or muscle invasive disease to assess for local lymph node involvement and locoregional extent of disease. These data can also be readily obtained from a CT or MRI urogram. Patients should also undergo a CT chest to assess for lung metastases prior to undergoing definitive treatment for muscle-invasive bladder cancer (MIBC), Depending on presenting symptoms, physical examination, and cystoscopic findings, further imaging such as radionuclide bone scans, and/or FDG PET/CT scan may be considered to evaluate for metastatic disease.
Non-muscle invasive bladder cancer
Bladder cancers are often classified as non-muscle invasive, muscle invasive, or metastatic. Non-muscle invasive bladder cancer (NMIBC) may involve the mucosa (Ta), submucosa or lamina propria (T1), or be characterized as flat CIS (Tis). Overall, NMIBC accounts for 70%–75% of all bladder cancers at initial presentation,8 about 60% of which are Ta, 30% are T1, and 10% are CIS. These are treated locally with TURBT, often in conjunction with intravesical chemotherapy or immunotherapy (Fig 1). Most NMIBCs recur within 6–12 months at the same stage, but 10%–15% of patients may develop invasive or metastatic disease, for which radical cystectomy and systemic chemotherapy are the standard treatments.
Fig. 1.
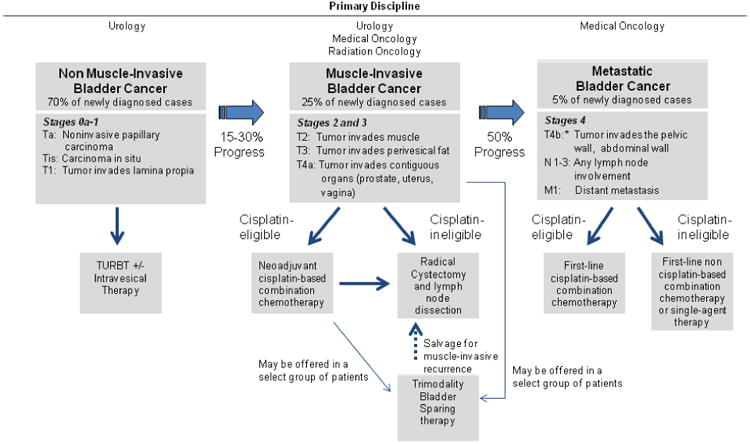
Bladder cancer management by stage.6
Muscle invasive bladder cancer
Approximately 25% of patients with bladder cancer present with a tumor invading the muscle layer of the bladder wall (T2–T4).9 MIBC is associated with a high rate of recurrence and poor overall prognosis despite aggressive local and systemic therapies. Even after radical cystectomy, the 5-year mortality rate of patients with MIBC remains about 50%–70%.10,11 Recently, recognition of the high rate of systemic failure for patients with MIBC has led to a new multidisciplinary management paradigm in which systemic therapy plays an increasingly important role. It is hoped that integrating surgery, medical oncology, and radiation oncology may enhance clinical outcomes for patients with MIBC.12 Simultaneously, bladder-preserving therapies have been introduced with promising outcomes in carefully selected patients with MIBC. This review will outline the multimodal management of MIBC.
Surgical Management
Radical cystectomy
For decades, radical cystectomy has been the mainstay of treatment for MIBC. Yet despite providing excellent local control, surgery alone does not result in optimal survival rates, especially for non-organ-confined (T3–T4) and lymph node-positive disease. An analysis of outcomes for patients who undergo radical cystectomy and pelvic lymph node dissection revealed that 5-year overall survival for patients with non-organ-confined, lymph node-negative disease was 49% and only 26% for patients with lymph node-positive disease, compared to 62% for patients with T2 and N0 disease.13
Pelvic lymph node dissection
Lymph node metastases are predictive of poor outcome; thus, pathologic assessment of resected lymph nodes is critical to staging and can only be performed intraoperatively.14 A series of studies evaluated whether the extent of pelvic lymph node dissection is an important prognostic factor.14-17 However, whether or not the extent of pelvic lymph node dissection has a significant impact on clinical outcome is still controversial.
Herr et al. analyzed correlations between surgical factors and clinical outcomes from the Southwest Oncology Group (SWOG) 8710 neoadjuvant chemotherapy trial.18 Multivariate analysis revealed that a greater number of lymph nodes removed (≥ 10 nodes vs. < 10 nodes) was prognostic for improved survival after cystectomy (HR 2.0; 95% CI: 1.4–2.8; P < 0.0001). Interestingly, when the extent of lymph node dissection was substituted in this model for number of lymph nodes (standard resection of distal common iliac, external iliac, obturator, and hypogastric nodes vs. limited resection of only nodes medial to the external iliac vein and obturator nodes), this difference was no longer prognostic, suggesting that extended pelvic lymph node dissection may be potentially therapeutic. However, in the SWOG 8710 study the extent of pelvic lymph node dissection did not correlate well with the cutoff of 10 lymph nodes removed. This is most likely related to the tremendous variability in pathologic assessment, the amount of lymph node tissue present in individual patients, and the extent and quality of lymph node dissection among surgeons despite defined limits of dissection.
Although there is no consensus regarding the optimal number of lymph nodes to dissect during radical cystectomy, an earlier analysis of lymph node metastases in patients who underwent extended lymphadenectomy (removal of all nodal regions below the inferior mesenteric artery) highlighted problems inherent in this procedure. Highly experienced surgeons from multiple centers were instructed to dissect nodes from 12 well-defined anatomical locations. Only 73% of surgeons successfully dissected all 12 regions. Although this study revealed that up to 25% of positive lymph nodes may lie outside the usual templates of node dissection (presacral and paracaval, interaortocaval, and para-aortic below the inferior mesenteric artery), the study provided no data that would have clarified whether or not removal of these nodes translated into a survival benefit.15
In contrast, a more recent study by Dhar et al. provides the most convincing evidence to date of the benefits of extended lymphadenectomy. The study compared outcomes of stage-matched patients from two different institutions using two different pelvic lymph node dissection templates. Patients at the Cleveland Clinic underwent limited pelvic lymph node dissection (proximal dissection to the bifurcation of the common iliac vessels), while patients at a hospital in Bern, Switzerland underwent a more extensive pelvic lymph node dissection (proximal dissection to crossing of ureters over the common iliac vessels). Patients with pT3 disease (extravesical disease into perivesical fat) who underwent the more extensive pelvic lymph node dissection showed a 30% improvement in 5-year recurrence-free survival, regardless of node status.16 Based on such findings, extended pelvic lymph node dissection, including the standard pelvic lymph node template and the presacral and common iliac lymph nodes, is becoming more widely accepted. The additional value of removing lymph nodes between the common iliac vessels and the inferior mesenteric artery along the aorta and vena cava is unclear. The effect of extended pelvic lymph node dissection on survival compared to standard or limited pelvic lymph node dissection is currently under investigation by SWOG in a randomized, prospective fashion.19
Improved surgical techniques
Radical cystectomy with pelvic lymph node dissection and urinary diversion is associated with considerable morbidity. The operation has the highest morbidity among all genitourinary surgeries, with complication rates of 30%–64%.20 In a study of a high-volume tertiary medical center that performed ∼750 radical cystectomies over 6 years, the 90-day readmission rate was ∼27% and the 90-day mortality rate was ∼7%.21 In males who undergo radical cystectomy, erectile dysfunction is a prevalent problem.22 Protocols for enhanced recovery after surgery (ERAS) may improve outcomes and result in less surgical morbidity for these patients.23 Improved surgical techniques such as nerve-sparing and prostate-sparing radical cystectomy have been successfully performed in selected patients, with excellent results in terms of potency.24,25 Additionally, minimally invasive robot-assisted laparoscopic radical cystectomy (RARC) is becoming more common worldwide and may result in shorter hospital stays and reduced blood loss for this typically morbid procedure. Results collected by the International Robotic Cystectomy Consortium demonstrate lymph node yield and oncologic safety on par with open surgery.26 Furthermore, a 48% 90-day complication rate was noted in this pooled analysis, with most complications being low-grade.27 A randomized trial comparing clinical outcomes with RARC and with open radical cystectomy is ongoing.28 Although radical cystectomy poses a sizeable risk of morbidity, it can result in cure for some patients. ERAS protocols, nerve-sparing surgical techniques, and RARC may make the risks of cystectomy more acceptable to both patients and surgeons.
Options for urinary diversion
Following cystectomy, a surgically constructed urinary diversion can establish reliable urinary drainage. These diversions can be incontinent or continent and are constructed from bowel segments from the stomach, small bowel, or colon. The choice of bowel segment depends on the presence of underlying renal insufficiency, as well as associated metabolic consequences and symptomatology associated with the various segments of bowel. For incontinent diversion, a segment of bowel, usually the ileum, is isolated from the remainder of the bowel on its own pedicled mesentery. The ureters are then surgically anastomosed to one end of the isolated segment of bowel. The other end is brought out through an opening in the abdominal wall, forming a stoma where an external appliance is attached to collect urine. This incontinent cutaneous urinary diversion, called an ileal conduit, is the most common type of urinary diversion performed, given the relative ease of the procedure and the shorter operative time compared to more complicated urinary reconstructions. In situations of previous pelvic irradiation, the transverse colon may be used as a conduit as well.
For continent urinary diversion, the ureters are anastomosed to a large segment of bowel that has been isolated to create a urinary reservoir. When a segment of large bowel and a portion of the terminal ileum are used, the resulting reservoir is called an Indiana pouch. The proximal portion of the terminal ileum is tapered and brought to the skin to serve as a cutaneous channel. At the distal portion of the terminal ileum, the one-way ileocecal valve then serves as the mechanism of continence for the reservoir. An Indiana pouch maintains continence and does not require an external appliance. It does, however, require intermittent catheterization for drainage and periodic irrigation to prevent accumulation of mucus, which can lead to stone formation over time. With the Indiana pouch, loss of the terminal ileum sometimes leads to malabsorption of lipids with consequent steatorrhea and lipid-soluble vitamin deficiency. A single-institution series of Indiana pouches had complications in up to 90% of patients, the most common being incontinence caused by problems with the valve.29 Continent cutaneous urinary diversion is also possible using different valve mechanisms and other portions of the bowel.
Continent urinary diversion can also be achieved with an orthotopic neobladder, another type of urinary reservoir created from a large segment of bowel, most commonly the ileum with the terminal ileum preserved. The new urinary reservoir is placed in the same location as the original bladder, then surgically anastomosed to the preserved urethral stump. Patients with an orthotopic neobladder can void spontaneously through the urethra by relaxing the external striated sphincter. There are many different variations of the orthotopic neobladder, including the Hautmann “W” neobladder, the Mansoura ileal neobladder, the “T” ileal neobladder, and the “Studer” ileal neobladder. Some series have reported rates of day- and night-time continence ranging from 70%–100%.30-34 Overall, the risk of significant complications over a lifetime is 18%–36% for patients with continent cutaneous reservoirs and 10%–18% for patients with orthotopic neobladders.35
A number of factors must be considered when choosing among types of urinary diversion. The surgeon must carefully review the patient's previous surgical history, prior pelvic radiation therapy, need for adjuvant therapy, risk of recurrence, pre-existing bowel conditions, and underlying medical conditions. The surgeon must also assess the patient's lifestyle and body image, support system, and willingness and ability to perform self-catheterization.36
Perioperative Therapies
Despite surgical improvements, the overall prognosis for MIBC treated with radical cystectomy alone remains suboptimal.37 The 5-year survival rate with radical cystectomy in MIBC is 50%–70%, depending on tumor stage, despite its high 90% 5-year local control rate. This suggests that the low cure rate is due in large part to micrometastasis at the time of surgery.38 A long-term follow-up study at the University of Southern California of > 1000 patients with MIBC who underwent radical cystectomy and pelvic lymph node dissection demonstrated a similar pattern: distant recurrence in 22% of patients and local recurrence in 7% of patients.11 Several neoadjuvant and adjuvant therapeutic approaches have been explored to optimize local and systemic control in patients with MIBC.
Neoadjuvant Therapies
Neoadjuvant radiation therapy
Several small studies have evaluated the role of neoadjuvant radiotherapy. Although neoadjuvant radiotherapy as a single agent is effective at pathologically downstaging disease at the time of resection,39 subsequent studies have reported disparate results. One large trial showed improved local control with this approach, but no obvious benefit in overall survival compared to radical cystectomy alone.40 A second randomized trial found no benefit for 20-Gy preoperative radiation.41 These trials all used doses of radiation lower than those considered effective today. Nevertheless, given the lack of firm evidence of therapeutic benefit with neoadjuvant radiotherapy alone, this treatment option is not recommended.
Neoadjuvant chemotherapy
In contrast, the role of neoadjuvant chemotherapy prior to radical cystectomy has been well established in the management of MIBC. Neoadjuvant chemotherapy has several potential advantages, including in vivo assessment of tumor response to a systemic regimen and downstaging of the primary tumor.
Cisplatin-based chemotherapy
Although bladder cancer is highly sensitive to cisplatin, a meta-analysis of four randomized trials (n = 479) failed to show a statistically significant survival benefit for cisplatin monotherapy prior to radical cystectomy.42 Several combination regimens containing cisplatin have been subsequently investigated.
The British Medical Research Council (MRC)/European Organization for Research and Treatment of Cancer (EORTC) reported a randomized phase III trial (BA06 30894) that evaluated a three-drug regimen of cisplatin, methotrexate and vinblastine (CMV) in 976 patients with MIBC.43 In this study, three cycles of CMV prior to cystectomy or curative full-dose radiation resulted in a complete pathologic response rate of 32.5% compared to 12.3% in the non-chemotherapy control arm. In a median follow-up of four years, neoadjuvant CMV failed to demonstrate significant survival benefit (HR 0.85; P = 0.075). However, in a recent update of the study after more than eight years of follow-up, the reduction in the risk of death has reached statistical significance (HR 0.84; P = 0.037), corresponding to an increase in 10-year survival from 30% to 36%.
Grossman et al. reported the results of the SWOG 8710 study that evaluated a four-drug combination of methotrexate, vinblastine, doxorubicin, and cisplatin (MVAC) in 307 patients with MIBC (T2N0M0 to T4aN0M0).10 The activity of MVAC in urothelial carcinoma was first reported in 1985 by Sternberg el al.44 and has been validated in subsequent randomized studies, showing superior activity to cisplatin alone or in combination with cyclophosphamide and doxorubicin (CISCA) in metastatic disease.45,46 The SWOG randomized phase III study showed that three cycles of MVAC chemotherapy can be given without adversely affecting a patient's chance of undergoing racial cystectomy, and achieved pathologic T0 rates of 38% compared to 9% in the surgery-alone group (P < 0.001). Neoadjuvant MVAC also showed superior survival, with a median survival of 77 months vs. 46 months in the cystectomy-alone arm (P = 0.05). A stratified proportional hazards model showed that patients in the cystectomy arm had a 33% greater risk of death than those in the combination-therapy arm (HR 1.33; 95% CI 1.0–1.76).
The survival benefit of platinum-based neoadjuvant chemotherapy has been further validated by two meta-analyses.47,48 The Advanced Bladder Cancer Meta-Analysis Collaboration analyzed 2688 patients from ten clinical trials and found that cisplatin-based combination neoadjuvant chemotherapy was associated with a significant overall survival benefit (HR 0.87; P = 0.016), although the HR for all trials, including those using single-agent cisplatin, was not significant (HR 0.91; P = 0.084).47 The meta-analysis by Winquist et al. evaluating 2605 patients from 11 randomized controlled trials also showed an overall survival benefit, with a pooled HR of 0.90 (P = 0.02) and HR of 0.87 (P = 0.006) for eight trials using cisplatin-based combination regimens.48
Despite its proven survival benefit, the MVAC combination regimen is associated with high toxicities and treatment-related mortality of 3%–4% in patients with advanced urothelial carcinoma.45 Thus, the alternative two-drug combination of gemcitabine and cisplatin (GC) has been widely accepted for neoadjuvant use. In a randomized study of > 400 patients with advanced or metastatic bladder cancer, the MVAC and GC regimens had comparable response rates (GC, 49%; MVAC, 46%); however, GC showed better safety and tolerability.49
No prospective study has directly compared GC to MVAC in the neoadjuvant setting. Several retrospective analyses have investigated the relative efficacy of GC compared to MVAC in patients with MIBC, with conflicting results.50-52 Fairey et al. (University of Southern California) evaluated 116 patients who received neoadjuvant GC (n = 58) or MVAC (n = 58) and found that complete response rates were higher for GC (27.3%) than for MVAC (17.1%), which was not statistically significant (P = 0.419).52 In contrast, a retrospective analysis by Yeshchina et al. (Comprehensive Cancer Center, Columbia University) evaluated 114 patients treated with either neoadjuvant GC (32%) or neoadjuvant MVAC (68%). Patients receiving MVAC showed higher pT0 rates than patients receiving GC (31% vs. 25%, respectively), which was not statistically significant (P = 0.645), and there was no difference in overall survival between the two regimens (HR 1.3; P = 0.33).50 However, Dash et al. (Memorial Sloan-Kettering Cancer Center) reported that 96 patients treated with GC (n = 42) or MVAC (n = 54) showed comparable complete response (pT0) rates of 26% for GC vs. 28% for MVAC.51
A phase II randomized, neoadjuvant chemotherapy trial by SWOG, scheduled to open in June 2014, will prospectively evaluate the power of an algorithm, “Co-eXpression ExtrapolatioN” or COXEN, based on the tumor's molecular profile, to predict tumor sensitivity to neoadjuvant dose-dense (dd) MVAC vs. GC in patients with MIBC.
Dose-dense regimens
Dose intensification has been evaluated for its ability to achieve higher response rates and reduce the duration of therapy. In the EORTC study by Sternberg et al., ddMVAC given every two weeks with growth-factor support resulted in a higher response rate with fewer dose delays and less toxicity compared to the standard schedule of MVAC given over four weeks in advanced metastatic bladder cancer.53 The feasibility and clinical efficacy of a dose-dense schedule in the neoadjuvant setting in MIBC has been evaluated in several phase II single-arm trials. Plimack et al. (Fox Chase Cancer Center) reported a single-arm phase II study of neoadjuvant ddMVAC.54 Treatment was generally well tolerated. Among 33 evaluable patients, 30 completed the full schedule of ddMVAC and 13 had a complete pathologic response (39.4%); three were downstaged to < T1. In a similar multicenter phase II study of ddMVAC in patients with MIBC, Qu et al. (Dana-Farber Cancer Institute) reported a pathologic and radiologic response rate of 49% and 69%, respectively.55 Both pathologic and radiographic responses were associated with significantly improved disease-free survival compared to nonresponders.
Siefker-Radtke et al. (M.D. Anderson) reported similar efficacy for the ddMVAC regimen.56 In a phase II study, ddMVAC in combination with bevacizumab resulted in pathologic downstaging to ≤ pT1 in 53% of patients and complete pathologic response in 38%, confirming the efficacy of ddMVAC. The role of additional bevacizumab is not clear; longer follow-up will be required to assess its effect on recurrence and survival. Meanwhile, a meta-analysis of 80 consecutive patients treated with ddMVAC as neoadjuvant therapy (Oxford Cancer Centre, U.K.) showed a complete pathologic response rate of 43% and a radiographic response rate of 83%.57
A dose-dense GC regimen has also been evaluated. A prospective, open label, phase III study (Hellenic Cooperative Oncology Group) comparing ddMVAC and dose-dense GC (ddGC) in patients with advanced urothelial cancer showed no difference in response rate (27% for ddMVAC vs. 32% for ddGC; P = 0.36) or overall survival (19 months for ddMVAC vs. 18 months for ddGC; P = 0.98).58 However, randomization was terminated early in this study due to low accrual. The feasibility and efficacy of neoadjuvant ddGC is currently being evaluated in several single-arm phase II studies.59
Neoajvuant chemotherapy for patients ineligible for cisplatin therapy
Despite high rates of tumor response and survival benefit with cisplatin-based combination chemotherapy, certain patients are not suitable candidates for this treatment. Contraindications to cisplatin include hearing loss/dysfunction, cardiac dysfunction, poor performance status, and renal insufficiency. Renal insufficiency is of particular concern since a large proportion of patients with urothelial cancer have impaired renal function due to multiple factors, including medical comorbidities, age-related decline in glomerular filtration rate, and ureteral obstruction.12 The combination of paclitaxel, carboplatin, and gemcitabine (PCaG) demonstrated an overall response rate of 69% and a complete response rate of 32% in a phase II trial in advanced urothelial cancers.60 However, when administered prior to cystectomy in patients with MIBC, PCaG resulted in seven deaths and removal of many patients from the study due to severe toxicities, raising concerns for the feasibility of this regimen.61 In addition, a phase II SWOG study (S0219) evaluating the clinical complete response (T0) rate after three cycles of PCaG followed by immediate cystectomy or cystoscopic surveillance resulted in an unacceptably high rate of understaging; 60% of patients who were presumed to have pT0 status had persistent cancer at cystectomy.62 A phase II study evaluating neoadjuvant gemcitabine, carboplatin, and panitumumab (GCaP) in patients with MIBC is underway,63 but so far there have been no large randomized studies of non-cisplatin-based chemotherapeutic regimens.
Adjuvant Therapies
For patients who undergo up-front radical cystectomy and are at high risk for recurrence, adjuvant chemotherapy is often the best course. The potential advantage of adjuvant chemotherapy is the possibility of complete pathologic staging, which can aid in the accurate selection of patients at risk for recurrence, given the high rate of clinical understaging.64 However, the delayed treatment of micrometastases is a hypothetical concern, and unlike with neoadjuvant chemotherapy, tumor response to a specific chemotherapy cannot be assessed. Furthermore, radical cystectomy has a high complication rate that prevents timely administration of adjuvant therapy. In a retrospective analysis of 1142 patients (Memorial Sloan-Kettering) who underwent up-front cystectomy, 64% experienced one or more complications within 90 days of radical cystectomy, rendering 30% unable to receive adjuvant chemotherapy.65
Prospective trials of cisplatin-based adjuvant chemotherapy
In a randomized trial, single-agent cisplatin as an adjuvant therapy failed to demonstrate a survival benefit in patients with MIBC.66 Patients received three cycles of cisplatin given on three consecutive days at monthly intervals after cystectomy (P = 0.65). The 5-year survival rate was 54% in the control group (n = 40) and 57% in the treatment group (n = 37).
Various adjuvant cisplatin-based combinations have been evaluated in randomized trials but have not met their survival endpoints, mainly due to methodological flaws or failure to achieve accrual goals. Most of these trials were small and thus severely underpowered. In a randomized study by Skinner et al., CISCA was evaluated in 99 patients with high-risk MIBC (pT3–4 or node-positive) treated with radical cystectomy and pelvic lymph node dissection, failed to show a significant survival benefit.67 Although the adjuvant chemotherapy arm showed a significant delay in time to progression compared to the observation arm (P = 0.001), the study was criticized for methodological flaws. Millikan et al. conducted a phase III study investigating perioperative vs. postoperative chemotherapy in patients with high-risk MIBC.68 The study showed no difference in outcome between the two arms: five cycles of adjuvant MVAC (n = 70) vs. two cycles of neoadjuvant and three cycles of adjuvant MVAC (n = 70). Overall survival was four years in both groups (P = 0.54). Notably, only 77% of patients randomized to the adjuvant arm actually received two or more cycles of chemotherapy, while 97% of patients randomized to the neoadjuvant/adjuvant arm had at least two cycles, suggesting that neoadjuvant chemotherapy is more feasible than adjuvant chemotherapy.
While a number of prospective randomized trials have failed to prove clinical benefit for various adjuvant chemotherapeutic regimens, some retrospective analyses of cisplatin-based adjuvant chemotherapy suggest a potential survival benefit. However, the cohort studies had inherent limitations.
An Advanced Bladder Cancer Meta-Analysis Collaboration report of 491 patients from six randomized controlled trials69 and data on 3947 patients from 11 centers70 showed a significant survival benefit for cisplatin-based adjuvant chemotherapy in high-risk patients. The meta-analysis data showed a 9% absolute survival improvement at three years, which translates to a 25% relative reduction in the risk of death with cisplatin-based adjuvant chemotherapy (HR 0.75; P = 0.019). The analysis was, however, limited by a small sample size with only 283 deaths.69 Data from a much larger patient cohort from multiple institutions in the off-protocol setting also showed that adjuvant chemotherapy was independently associated with improved survival (HR 0.83; P = 0.017).70 However, the effect of adjuvant chemotherapy was significantly modified by patients' risk features. Patients with the highest risk features, such as advanced pathologic staging (≥ T3) and node-positive disease, experienced the most significant survival benefit from adjuvant chemotherapy (HR 0.75; P = 0.002). Several large, more recent randomized trials were not included in the above meta-analysis. Unfortunately, these studies failed to meet their accrual goals and were prematurely terminated, leaving the benefit of adjuvant chemotherapy uncertain.
Paz-Ares et al. reported a trial (Spanish Oncology GU Group 99/01) evaluating adjuvant paclitaxel, gemcitabine, and cisplatin (PGC) that showed a progression-free survival benefit at five years compared to control (P < 0.0001) after four cycles of adjuvant PGC in patients with high-risk MIBC (pT3–T4 and/or pN+). Results also showed a prolonged five-year overall survival in the PCG arm (60%) compared to observation (31%) (P < 0.0009).71 However, this study was terminated early due to poor recruitment (140 enrolled out of an expected 340).
A randomized phase III trial reported by Cognetti et al. evaluating the benefit of adjuvant chemotherapy with GC also suffered from slow accrual.72 This study enrolled only 190 patients, significantly fewer than the planned 610 patients, and was thus underpowered to demonstrate survival benefit. Furthermore, only 62% of patients could complete chemotherapy as planned, and more than half of the patients required a dose reduction, further limiting the power of the study. The control and adjuvant chemotherapy arms showed similar five-year disease-free survival (42.3% and 37.2%, respectively; HR 1.08; P = 0.70) and overall survival (48.5%; HR 1.29; P = 0.24).
A phase III randomized study (EORTC 30994) was planned to evaluate adjuvant cisplatin-containing chemotherapy (MVAC, ddMVAC, or GC at the treating physician's discretion) compared to observation after radical cystectomy. This study was closed in 2008 due to poor accrual (298 patients enrolled over seven years out of a planned 664). The results of this study will be presented at the 2014 Annual Meeting of the American Society of Clinical Oncology. Similarly, a Cancer and Leukemia Group B trial (CALGB 90104) comparing sequential doxorubicin and gemcitabine followed by paclitaxel and cisplatin with GC alone in the adjuvant setting was closed due to slow accrual; only 42 patients out of a planned 800 were enrolled.
It has been suggested that p53 positivity is associated with a high recurrence rate in patients with organ-confined bladder cancer, especially in early-stage disease (T1/T2N0).73 p53 positivity has also been suggested as a predictive marker for cisplatin-based chemotherapy in bladder cancer.74 Stadler et al. conducted a study evaluating the role of molecularly targeted adjuvant MVAC in patients with early-stage disease.75 In this study, patients with pT1/T2N0M0 disease whose tumors were p53-positive, defined as ≥ 10% nuclear reactivity on immunohistochemistry (IHC), were randomized to either adjuvant MVAC (n = 58) or observation (n = 56), while p53-negative patients were observed. Only 67% of patients randomly assigned to MVAC received all three cycles, and 12 patients received no treatment. In the end, overall five-year probability of recurrence did not differ according to p53 status (P = 0.41), failing to validate the prognostic value of p53. Among p53-positive patients, adjuvant MVAC did not improve recurrence-free (P = 0.62) or overall survival (P = 0.89) compared to surgery alone, suggesting no value for adjuvant chemotherapy in patients with early-stage disease without high-risk features.75 However, the high rate of patients who refused randomization, the lower-than-expected event rate, and failures to receive assigned therapy severely compromised study power.
Generally, adjuvant chemotherapy is recommended for patients with high-risk features such as T3 or T4 disease and/or lymph node involvement, who have not been treated with neoadjuvant chemotherapy. Routine use of adjuvant chemotherapy in patients with MIBC without such high-risk features is controversial due to the lack of evidence of clear benefit in this group. There are limited data on the management of patients who are not candidates for cisplatin-based adjuvant chemotherapy. Non-cisplatin alternatives such as a carboplatin-based regimen or gemcitabine monotherapy lack data for standard use.76
Underuse of perioperative chemotherapy
As previously discussed, the SWOG 8710 trial that demonstrated a survival benefit with neoadjuvant chemotherapy was reported in 2003,10 followed by reports of two supporting meta-analyses in 2003 and 2004.47,48 Yet despite the well-established role of neoadjuvant chemotherapy in MIBC, up-front surgery is often the treatment of choice, due in part to the low rate of referral of patients from urologists to medical oncologists.
The National Cancer Data Base analyzed treatment patterns in 7161 patients diagnosed with stage III bladder cancer between 1997 and 2003. According to their report, which was published before SWOG 8710 was reported, only 10.4% of patients received adjuvant chemotherapy and a mere 1.2% were treated with neoadjuvant chemotherapy.77 Data from the Surveillance, Epidemiology, and End Results-Medicare database obtained between 1992 and 2002 showed that few patients with MIBC (1.2% with stage II and 7.3% with stage III) received chemotherapy prior to cystectomy.78 Fedeli et al. evaluated the trend of chemotherapy use in patients with MIBC between 2003 (after the publication of SWOG 8710) and 2007 using the National Cancer Data Base.79 This study showed that the percentage of patients with MIBC who received neoadjuvant chemotherapy increased each year after 2003 (6%) and doubled in 2007 (13%), although this number is still unacceptably low given the high-level evidence of survival benefit with neoadjuvant chemotherapy.
Various efforts have been made to increase the use of perioperative chemotherapy. A multi-institutional quality care initiative (QCI) has launched a two-phase effort. Phase 1 is the evaluation of baseline patterns of perioperative chemotherapy use in patients with MIBC between 2003 and 2008, and phase 2 is a prospective QCI. The quality indicators include (1) referral to a medical oncologist for consideration of multimodal therapy, (2) neoadjuvant (if recommended) cisplatin-based chemotherapy of at least three cycles, (3) adjuvant (if recommended) cisplatin-based chemotherapy of at least three cycles, and (4) all treatment to be completed within six months. A report by Bochner et al. showed that of 4344 patients in phase 1, only 34% received perioperative chemotherapy, 14% neoadjuvantly. Of those treated neoadjuvantly, 70% received cisplatin-based chemotherapy. Of the > 700 patients evaluated after initiation of the QCI (phase 2), 56% received perioperative cisplatin-based chemotherapy, 47% neoadjuvantly, which was a significant increase. There was a 64.7% increase in the use of any perioperative chemotherapy and a 3.4-fold increase in neoadjuvant chemotherapy.80
Apolo et al. evaluated the management patterns of medical oncologist in patients with MIBC. The results showed a reassuring increase in the adoption of recommendations that follow best evidence.81 A majority of referrals to medical oncologists were made by urologists (79%) for assessment for perioperative chemotherapies. Neoadjuvant and adjuvant chemotherapy were offered to patients with MIBC by 79% and 46% of respondents, respectively. The reasons given for not recommending neoadjuvant chemotherapy included poor performance status and renal dysfunction. The report suggested that a majority of medical oncologists offer perioperative chemotherapy, with a trend toward increased use of neoadjuvant chemotherapy over adjuvant chemotherapy when patients are referred by urologists and chemotherapy is considered appropriate.
Bladder-Preserving Strategies
Despite improvements in surgical techniques and supportive care, radical cystectomy continues to have high rates of morbidity and mortality.82,83 Further, urinary diversion may have its own complications and additional effects on quality of life. The desire to provide patients with a therapeutic option that preserves a functioning bladder has led to the development of several bladder-preserving strategies that, for a carefully selected subgroup of patients, provide an alternative to radical cystectomy.
Bladder-preserving strategies generally include limited surgical approaches such as TURBT or partial cystectomy, with or without radiation therapy and/or chemotherapy. The most effective bladder-preserving strategies often involve a trimodal or multimodal approach to maximize disease control. Although none of these approaches has been evaluated in comparison with radical cystectomy or with each other in a randomized fashion, the results of several prospective series suggest that appropriately selected patients with MIBC can achieve long-term disease control with an intact bladder. We review below (1) the various combinations of surgery, radiation, and chemotherapy that have been explored and (2) the current standard approach of trimodal therapy.
Bladder-preserving surgical approaches
Modified surgical approaches have been evaluated as alternatives to radical cystectomy. Conservative tumor resection involving either definitive TURBT or partial cystectomy has been employed in highly selected patients with solitary muscle invasive tumors without evidence of associated CIS.
A retrospective cohort study (Memorial Sloan-Kettering) of 151 patients with MIBC treated by definitive TURBT showed a 10-year survival probability that is comparable to that of radical cystectomy.84 The 10-year disease-specific survival was 76% for patients who underwent definitive TURBT, with a 57% rate of bladder preservation compared to 71% of patients who had immediate radical cystectomy. Among patients who had a complete pathologic response on restaging TURBT, 82% survived to 10 years. However, among patients with residual tumors, 69% had relapse, with a 10-year survival of only 57%. Solsona et al. performed a phase II nonrandomized study to determine the efficacy of cisplatin-based chemotherapy in patients with microscopic residual tumor after definitive TURBT.85 Patients with positive biopsies of the tumor bed after TURBT received either immediate radical cystectomy or three cycles of cisplatin-based chemotherapy in a nonrandomized fashion. The cancer-specific survival rate with bladder preservation was 64.5% and 59.8% at five and 10 years, respectively, with no significant difference compared to radical cystectomy (P = 0.544). The authors speculated that, based on findings in the radical cystectomy arm, 98% of patients in the chemotherapy arm had residual tumor, 89% of which was invasive. With cisplatin-based chemotherapy, 50.5% of patients achieved durable complete responses, suggesting effective control of microscopic residual invasive tumor in about 45% of patients.85
Partial cystectomy can be performed in highly selected cases where the bladder has been adequately sampled by random biopsies (including the prostatic urethra) and where there is no evidence of cancer other than the tumor of interest. This approach also allows complete pathologic staging with potential concurrent pelvic lymph node dissection while preserving urinary function. Partial cystectomy is contraindicated in the presence of any CIS. Finally, the lesion has to be removed with negative margins and the remaining bladder has to have adequate capacity to allow for normal voiding. With partial cystectomy, bilateral extended lymphadenectomy is recommended to adequately sample locoregional lymph nodes, based on several reports supporting such a strategy.
Several single-institution studies (Memorial Sloan-Kettering; M.D. Anderson) with a small number of patients have reported a five-year survival rate of ∼70%, with a bladder preservation rate of approximately 65% in highly selected patients with MIBC.86-89 A population-based matched analysis of 7243 patients with MIBC treated with partial cystectomy (n = 1573) or radical cystectomy (n = 5670) showed comparable overall survival and cause-specific survival for partial and radical cystectomy. The five-year estimated overall survival and cause-specific survival for partial cystectomy patients was 57.2% and 70.3%, respectively, and for radical cystectomy patients 54.6% and 69.2%, respectively (overall survival: HR 1.1; P = 0.3; cause-specific survival: HR 1.1; P = 0.5). Cohorts were matched for age, race, pT stage, pN stage, tumor grade, year of surgery, and number of lymph nodes removed.90 More recent data from Zhang et al. showed that partial cystectomy followed by adjuvant chemotherapy and radiation achieved comparable clinical outcomes.91 In this study, patients with pT3–4 tumors after partial cystectomy were treated with adjuvant MVAC and subsequent radiation of 45–50 Gy. The five-year bladder-preservation survival was 63%, with a 14% MIBC recurrence rate. It must be stressed that strict criteria are needed to select appropriate candidates for this nonstandard, bladder-preserving approach.
Sternberg et al. evaluated the feasibility of selecting patients for definitive TURBT or partial cystectomy on the basis of response to neoadjuvant chemotherapy.92 Patients with T2–T4, N0 MIBC were treated with neoadjuvant MVAC, followed by restaging with a repeat CT and TURBT. Based on response at restaging, patients underwent either a bladder-preservation protocol with TURBT (n = 52), or partial cystectomy (n = 13), or radical cystectomy (n = 39). About half of patients had T0 with MVAC chemotherapy. Among patients who had TURBT, 25% had superficial recurrences, 10% developed invasive disease, 23% developed metastases, and 6% developed both invasive and metastatic disease. Among patients who had partial cystectomy, 15% had superficial recurrences and 23% developed locally invasive disease. The five-year survival rate was 67% in the TURBT group and 69% in the partial cystectomy group. The authors concluded that bladder sparing in selected patients on the basis of response to neoadjuvant chemotherapy is feasible, although the study could not assess the benefit of neoadjuvant chemotherapy in this setting.
Radiation with or without chemotherapy
Radiation alone as a method of bladder preservation has been described. However, comparisons to radical cystectomy are problematic, as patients referred for radiotherapy alone as an alternative to radical cystectomy tend to be older and sicker than their counterparts who undergo radical cystectomy. The largest series reporting the outcomes of radiotherapy alone in patients with urothelial carcinoma of the bladder was from Scotland, where the institutional policy was primary radiotherapy as the sole treatment in patients with T1–T4 urothelial carcinoma of the bladder.93 This study reported on 961 patients, all treated in the era before CT scans. The presence of metastatic disease was assessed by skeletal radiography, chest x-ray, and bone scan as indicated. Following radiation, complete regression of the bladder tumor was observed in 45.9% of patients. The five-year survival rate for all patients, including those receiving palliative treatment only, was 30.3%. Survival was highly dependent on tumor stage, with five-year survival ranging from 30% for patients with T4 tumors with complete regression after radiation to 62.8% for patients with T1 tumors with complete regression. Several other series have shown similar rates of regression after primary radiotherapy.94,95
A major concern with radiation as a single modality for bladder preservation is the relatively high rate of local recurrence or persistence and the need for post-radiotherapy salvage cystectomy. Long-term follow-up of 159 patients with MIBC treated in a randomized trial with radical radiotherapy with or without neoadjuvant cisplatin showed that 24% of patients underwent salvage cystectomy, 20% in the radiotherapy-alone group and 28% in the combination-therapy group.95
Efforts to improve the efficacy of primary radiotherapy initially focused on the addition of sensitizing chemotherapy during radiotherapy. Results of multi-institutional retrospective trials employing this approach have suggested an improvement in local control, but variable results in terms of survival benefit compared to radiotherapy alone.96,97
Coppin et al. (National Cancer Institute of Canada) reported a prospective randomized trial that showed improved local control with concurrent addition of cisplatin to definitive radiotherapy in MIBC.98 The study randomized 99 patients to radiotherapy with or without cisplatin, followed by elective cystectomy or further radiotherapy at the investigator's and patient's discretion. Although concurrent chemoradiation significantly reduced the risk of pelvic failure compared to radiotherapy alone, there was no evidence of increased survival with the addition of concurrent chemotherapy.
Hussain et al. reported a phase I/II study of synchronous chemoradiation therapy with mitomycin C and 5-FU in 41 patients, 17 of whom had a glomerular filtration rate of < 50 mL/min, and 76% of whom had T3–T4 disease.99 This regimen achieved high rates of local control, with a local progression-free rate of 82% at one year and 79% at two years. The rate of organ preservation was also high: only five patients underwent salvage cystectomy. Overall one-, two-, and five-year survival rates were 68%, 49%, and 36%, respectively.
Similarly, the Bladder Cancer 2001 trial (BC2001) randomized 360 patients with MIBC to radiotherapy alone or concurrent chemoradiation with mitomycin C and 5-FU.100 In this trial, the addition of chemotherapy to radiotherapy resulted in improved local control, good long-term bladder function, and low rates of salvage cystectomy. Two-year recurrence-free survival was 67% with chemoradiation therapy vs. 54% with radiotherapy (HR 0.68; P = 0.03). Chemoradiation therapy was associated with a trend toward a reduction in cystectomy, with a two-year survival rate of 11.4% in the chemoradiation therapy group vs. 16.8% in the radiotherapy group, with no significant difference in overall survival. Based on data from these trials and other similar reports, primary radiotherapy alone for bladder preservation has largely been abandoned for patients who can tolerate multimodal therapy. The BC2001 trial confirmed significantly improved locoregional control of bladder cancer with 5-FU and mitomycin C compared to radiotherapy alone, with no significant increase in adverse events.100
Trimodal strategies
The suboptimal bladder preservation obtained with single modalities has led to an integrated approach involving surgery, radiation, and chemotherapy, with the goal of improving both local and distant disease control. Various strategies involving definitive non-cystectomy surgical resection in combination with chemotherapy and radiation for maximum local and systemic control in selected patients with MIBC have shown encouraging clinical outcomes and reasonable bladder-preservation rates. Multimodal bladder-preserving strategies developed in the 1980s by several groups, including Massachusetts General Hospital, the University of Paris, and the Radiation Therapy Oncology Group (RTOG), consisted of an induction phase of TURBT and cisplatin-based concurrent chemoradiation therapy, followed by a consolidation chemoradiation therapy based on tumor response at repeat cystoscopy.
In 1993, Tester et al. published the first report of a phase II study (RTOG 85-12) evaluating the tolerability and effectiveness of this strategy.101 Patients received 40 Gy of radiation with concomitant cisplatin after maximal TURBT. Complete responders received an additional 24-Gy boost with concurrent cisplatin, while nonresponders underwent salvage radical cystectomy. The complete response rate after induction therapy was 67% in evaluable patients. The treatment was well tolerated and resulted in a 64% three-year survival rate and a 42% five-year survival rate with an intact bladder. The RTOG has since evaluated multiple regimens based on radiosensitizing chemotherapy agents combined with radiotherapy. The RTOG has also tested different dosing of chemotherapy and radiotherapy, as well as novel combinations and additional adjuvant and neoadjuvant chemotherapies (Table 3).
Table 3.
Multimodal bladder-sparing trials.
| Study | Phase | Protocol | No of patients | CR (%) | Survival (%) | Author | Year | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| a Neoadjuvant chemotherapy | Concurrent chemotherapy | Radiation dose (Gy) | b Adjuvant chemotherapy | 5-year | 3-year | ||||||
| MGH189 | II | MCV × 2 | Cisplatin | 40.0 + 24.8 | 53 | 78 | 48 | Kaufman | 1993 | ||
| France103 | II | Cisplatin/5 FU | 24.0 + 20.0 | 54 | 74 | 59 | Housset | 1993 | |||
| RTOG 85-12101 | II | Cisplatin | 40.0 + 24.0 | 42 | 66 | 52 | Tester | 1993 | |||
| RTOG 88-02190 | II | MCV × 2 | Cisplatin | 39.6 + 25.2 | 91 | 75 | c62 | Tester | 1996 | ||
| MGH191 | N/A | MCV × 2 | Cisplatin | 39.6 + 25.2 | 106 | 66 | 52 | Kachnic | 1997 | ||
| Italy192 | II | MCV × 2 | Cisplatin | 40.0 + 24.0 | 56 | 50 | 55 | Fellin | 1997 | ||
| RTOG 89-03116 | III | MCV × 2 None | Cisplatin | 39.6 + 25.2 | 123 | 61 55 |
49 48 |
Shipley | 1998 | ||
| RTOG 95-06108 | 1/II | Cisplatin/5-FU | 24.0 + 40.0 | 34 | 67 | 83 | Kaufman | 2000 | |||
| German193 | N/A | d Various | e54 | 415 | 72 | 51 | Rodel | 2002 | |||
| RTOG 97-06194 | 1/II | Cisplatin | 40.8 + 24.0 | MCV × 3 | 46 | 74 | 61 | Hagan | 2003 | ||
| Italy96 | N/A | MCV × 2 | Cisplatin or carboplatin | f65 | 121 | 86 | 68 | Perdona | 2008 | ||
| RTOG99-06109 | I/II | Cisplatin/Pa clitaxel | 40.3 + 24.0 | GC × 4 | 80 | 81 | 56 | Kaufman | 2009 | ||
| Italy P1 P2195 |
II | MCV × 3 None | None Cisplatin |
60 40.8 + 24.0 |
41 33 |
66 76 |
75 65 |
Zapatero | 2010 | ||
| UK196 | II | Gemcitabine | 25.5 | 50 | 88 | 65 | Choudhury | 2011 | |||
| BC2001197 | III | 5-FU/mitomyc in None | 55 or64 | 360 | 48 35 |
James | 2012 | ||||
| RTOG02-33106 | II | Cisplatin/Pa clitaxel Cisplatin/5-FU | 40.3 + 24.0 | PCG × 4 | 93 | 72 62 |
71 75 |
Mitin | 2013 | ||
BPS = bladder preservation survival; MGH = Massachusetts General Hospital; MCV = methotrexate, cisplatin, and vinblastine; GC = gemcitabine and cisplatin; PCG = paclitaxel, cisplatin, and gemcitabine.
Chemotherapy given prior to initiation of radiation therapy
Chemotherapy given after to completion of radiation therapy
Four-year survival
Cisplatin or carboplatin or cisplatin/5-FU
Median (range, 45 to 69.4 Gy)
Median (range, 34 to 67 Gy)
Shipley et al. have reported and summarized a number of trimodal bladder-preservation trials at Massachusetts General Hospital,102 including a study of 190 patients treated with multimodal approaches between 1986 and 1997. All of the regimens used maximal TURBT followed by cisplatin-based concurrent chemoradiation therapy and reassessment. Consolidation with boost chemoradiation to a total dose of 64–65 Gy or salvage cystectomy was employed depending on tumor response to induction therapy (Fig 2). The primary differences among the various regimens related to the use of neoadjuvant or adjuvant chemoradiation therapy and the addition of 5-FU to cisplatin in concurrent chemoradiation therapy. In this series, a visibly complete TURBT was achieved in 57% of patients. Radical cystectomy was performed in 66 patients (35%) due to incomplete response to induction therapy (n = 41) and as salvage for recurrent invasive tumor (n = 25). The 5- and 10-year disease-specific survival rates for patients undergoing radical cystectomy were 48% and 41%, respectively. Among patients with complete remission after induction, 60% remained recurrence-free, 24% developed a superficial recurrence, and 16% developed an invasive tumor. The five-year overall survival rate was 54%, disease-specific survival rate was 63%, and five-year survival rate with a preserved native bladder was 45%.
Fig. 2.
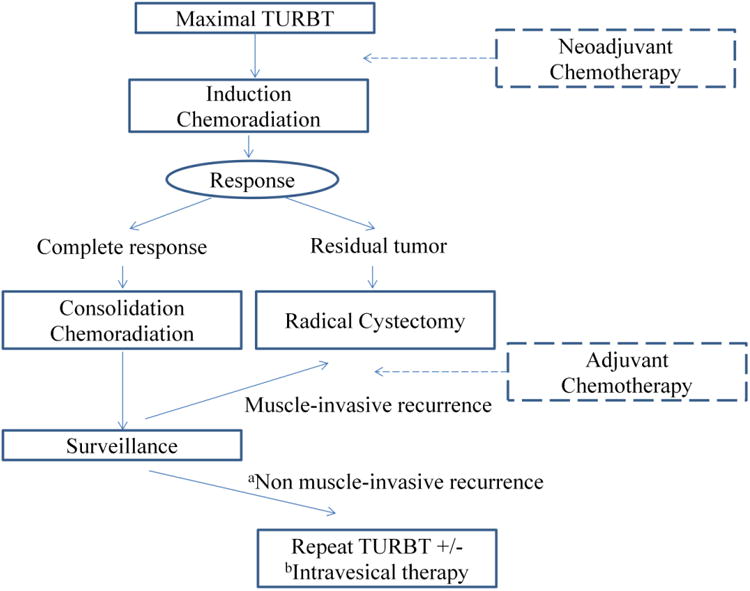
Algorithm for trimodality bladder-sparing therapy. TURBT = transurethral resection of the bladder tumor, aTa, Tis, T1, bIntravesical BCG or intravesical chemotherapy
Housset et al. (University of Paris) reported the results of a similar bladder-preserving trimodal strategy in 54 elderly patients with T2–T4 tumors treated with TURBT followed by chemoradiation of 24 Gy with concurrent cisplatin and 5-FU.103 Patients with persistent tumor on cystoscopic re-evaluation underwent radical cystectomy. Patients with a complete response (74%) on re-evaluation received either an additional 20 Gy (group A) or underwent radical cystectomy (group B) (Fig 2). After a median follow-up of 27 ± 12 months, three responding patients in group A and one in group B developed local recurrence. Metastatic disease occurred more frequently in patients who failed to achieve complete response (71%) than in responders (23%). The three-year overall survival rate was 59%; the disease-free survival rate was 62%. There was no difference in survival between groups A and B (81% and 77%, respectively).
In the studies described above, response to TURBT and chemoradiation therapy was evaluated after the induction phase, which allowed early identification of non-responding patients. The rationale for this approach was to allow for rapid salvage radical cystectomy and to reduce the total dose of radiation delivered prior to radical cystectomy to minimize surgical complications and improve the feasibility of urinary reconstruction.104
In contrast, a German group employed a bladder-preserving strategy of up-front complete chemoradiation therapy followed by response assessment, providing radical cystectomy only in cases of residual disease in order to maximize the chance of complete response and avoid radical cystectomy whenever possible.105 Rödel et al. (University of Erlangen) reported a study of > 400 patients with high-risk T1 or T2–T4 disease treated from 1982 to 2000 with radiation alone (n = 126) or concurrent chemoradiation therapy (n = 289) following TURBT. Chemotherapy was given during the first and fifth week of radiation and consisted of cisplatin (n = 145) or carboplatin in patients with renal insufficiency or heart failure (n = 95), or a combination of cisplatin and 5-FU (n = 49). The median dose of radiation was 54 Gy. Response was evaluated by restaging TURBT six weeks after completion of treatment. Salvage radical cystectomy was recommended for cases of persistent or recurrent invasive tumor (Fig 2). Complete resection of tumor was achieved in 28% with initial TURBT. After completion of treatment, 72% of patients achieved complete remission, 5% showed non-muscle invasive residual tumor, and 23% showed muscle invasive tumor. Concurrent chemoradiation therapy resulted in an increased rate of complete response and significantly improved overall survival compared to radiation alone. The complete response rate was 61% for patients treated with radiation alone, 66% after chemoradiation therapy with carboplatin, 82% with cisplatin, and 87% with 5-FU/cisplatin. Ten-year disease-specific survival was 42%, and > 80% of survivors had intact bladders.
Recent and ongoing combined modality trials have attempted to optimize chemotherapy regimens to minimize toxicity while improving rates of complete response and survival. Many patients with MIBC suffer from renal insufficiency or other morbidities for which cisplatin may be contraindicated. Based on these limitations, non-cisplatin regimens have been further explored and have been shown to be safe and effective in patients with renal insufficiency.
The safety and effectiveness of adding paclitaxel or 5-FU to cisplatin-based chemoradiation therapy was reported in a recent randomized phase II trial by Mitin et al. (RTOG 0233).106 This study consisted of an induction phase of TURBT and 40.3 Gy of chemoradiation therapy with either paclitaxel or 5-FU plus cisplatin. Tumor response was assessed after induction. Patients with a complete response (T0) or near-complete response (Tcis or Ta) received consolidation chemoradiation therapy to 64.3 Gy, with the same chemotherapy regimen as in the induction phase followed by adjuvant paclitaxel, cisplatin, and gemcitabine (PCG). In this study, the majority of patients in both the cisplatin/paclitaxel and cisplatin/5-FU groups have completed the induction and consolidation phases (85% and 83%, respectively). As expected, the complete response rate after induction therapy was high (72% with cisplatin/paclitaxel; 62% with cisplatin/5-FU), which is comparable to historic data from other cisplatin-based trimodal studies.107 However, only 58% of patients completed the treatment course and 19 patients did not complete adjuvant chemotherapy after chemoradiation therapy. In addition, a high rate of grade 3 and 4 toxicity was observed during adjuvant chemotherapy. The five-year overall survival rate in patients who completed all treatment courses was 81% compared with 49% in patients who completed chemoradiation therapy but not adjuvant chemotherapy.
The RTOG 0233 protocol was based on previous RTOG studies that tested the feasibility and efficacy of combining a second radiosensitizing agent to cisplatin. RTOG 9506 was a phase I/II trial of trimodal therapy that employed combined cisplatin and 5-FU as radiosensitizing agents.108 The regimen resulted in grade 3 or 4 hematologic toxicity in 21% of patients. The outcome was encouraging, with a 67% complete response rate to induction therapy and 66% three-year survival. RTOG 9906 was a phase I/II trial of cisplatin and paclitaxel delivered concurrently with radiation, with the addition of adjuvant gemcitabine and cisplatin.109 Seventy percent of patients completed the entire treatment course, including adjuvant chemotherapy. The post-induction complete response rate was 81% and the five-year survival rate was 56%. As in RTOG 9906, RTOG 0233 incorporated adjuvant chemotherapy after chemoradiation therapy or radical cystectomy, based on the observation that distant metastasis is the most common sign of treatment failure in MIBC. Mitin et al. recently presented a retrospective pooled analysis of RTOG 9906 and RTOG 0233, both of which allowed patients with near-complete response after the induction phase to proceed to consolidation.110 Of 119 eligible patients, 101 (85%) achieved complete response and 18 (15%) achieved near-complete response after induction chemoradiation and proceeded to consolidation. After a median follow-up of 5.9 years, 36% of patients with complete response and 28% of patients with near-complete response experienced tumor recurrence (P = 0.52). Among complete responders, 14 eventually required late salvage cystectomy for tumor recurrence vs. one patient among near-complete responders (P = 0.47). Disease-specific, bladder-intact, and overall survivals were not significantly different.
The three-drug combination PCG used as an adjuvant regimen in RTOG 0233 was initially evaluated by the Spanish Oncology GU Group in advanced bladder cancer patients in phase I/II studies and was shown to be feasible and highly active, with an overall response rate of 77.6%.111,112 In a subsequent EORTC phase III randomized study, PCG led to higher response rates than GC (55.5% vs. 43.6%, respectively) and a preferable survival trend in patients with advanced urothelial carcinoma of the bladder vs. the upper tract.113
Despite encouraging data from studies of bladder-preserving approaches, there are several concerns associated with bladder-preserving therapy. One is the potential for delay of definitive radical cystectomy in patients who will not achieve a complete response to induction treatment. The pelvic tissue changes caused by radiation not only make surgery technically challenging, but are also associated with higher postoperative complications compared to radical cystectomy without radiation.114 Rates of surgical complications after pelvic radiotherapy increase with dose, with higher rates of complications expected at the doses delivered in the setting of bladder preservation.115 Even after successful bladder-preserving therapy with a complete response, frequent, long-term surveillance is required for timely and effective management of invasive and non-invasive recurrence.
Patient selection for trimodal therapy
Not all patients with MIBC are appropriate candidates for bladder preservation. Considerations when determining eligibility for trimodal bladder-preserving strategies include factors that may predict for recurrence or for poor bladder function following treatment. Patient selection is key to ensuring that only those with a realistic chance of durable local control with an adequately functioning bladder are exposed to this aggressive therapy.
Tumor-associated hydronephrosis is a contraindication for trimodal approaches, as it is associated with a low rate of complete response and thus a low rate of bladder preservation.107,116 Although a visibly complete TURBT is a classic inclusion criterion for many trials, some protocols suggest that visible disease after TURBT is not an absolute contraindication117 as local control may be achieved, albeit at a lower rate than in patients with a visibly complete TURBT. Despite these conflicting data, most trials continue to require a visibly complete TURBT for enrollment. Patients with poor bladder capacity or poor urinary function are often suboptimal candidates as their urinary function may decline further. Extensive CIS or advanced local disease (T3b) is also considered a relative contraindication to bladder preservation.117 Importantly, a patient's ability to tolerate chemoradiotherapy must also be considered before embarking on this approach. Inability to tolerate chemotherapy or prior pelvic radiotherapy, as well as active collagen vascular disease and inflammatory bowel disease, are further contraindications to this approach.
Biomarker Studies in MIBC
Bladder cancer comprises a heterogeneous group of diseases beyond conventional histopathology. Advances in molecular technologies that allow more rapid and extensive analyses have identified various genetic and epigenetic changes and aberrant protein expressions associated with urothelial carcinoma. Deletion, mutation, or aberrant methylation of tumor suppressor genes such as TP53, RB1, CDKN2A, and PTEN and activation, mutation, or overexpression of oncogenes such as FGFR3, Her2, and CCND1 are commonly associated with MIBC. Some of these alterations seem to play a critical role in tumorigenesis, disease progression, and development of treatment resistance. A better understanding of these underlying molecular alterations may lead to the discovery of prognostic and predictive markers and the development of individualized targeted therapies.118
Gene expression signature
There is growing interest in using a gene expression signature to predict tumor characteristics and clinical outcomes in bladder cancer.119-121 Smith et al. have developed a 20-gene expression model to predict lymph node involvement in patients with clinically lymph node-negative (cN0) MIBC who are undergoing neoadjuvant chemotherapy.119 This model showed a predictive value for pathologic lymph node involvement (area under the curve [AUC]: 0.67). Riester et al. reported that novel cancer gene expression patterns could predict disease recurrence in high-risk bladder cancer patients after radical cystectomy.122 They identified gene expression patterns associated with clinical and prognostic variables in 93 patients with MIBC managed by radical cystectomy. The results were compared to previously published data sets, and a survival-associated gene expression subtype was identified using hierarchical clustering. Used together, survival-associated gene expression and the clinical nomogram validated by the International Bladder Cancer Nomogram Consortium123 make predictions of recurrence significantly more accurate.
The Cancer Genome Atlas
The Cancer Genome Atlas (TCGA) is a multi-institutional, comprehensive, coordinated effort to understand the molecular basis of cancer through various high-throughput genome analysis techniques. The project started in 2006 to identify genomic changes in more than 20 different types of human cancers, including bladder cancer. TCGA network has recently published a comprehensive, integrated study of 131 chemotherapy-naïve high-grade MIBCs (T2–T4a, Nx, Mx).124 Several genomic alterations with potential therapeutic implications have been identified, including the PI3K/AKT/mTOR, RTK/RAS pathways, ERBB2 (Her2), ERBB3, and FGFR3.
Mutations, copy number alterations, or RNA expression changes affecting the PI3K/AKT/mTOR pathway were observed in 42% of tumors, including activating point mutations in PIK3CA (17%), mutation or deletion of TSC1 or TSC2 (9%), and overexpression of AKT3 (10%). ERBB2 mutation or amplification was detected in 9% of tumors, which is approximately as frequent as in TCGA breast cancers, but with fewer amplifications and more mutations. Other genetic alterations with potential therapeutic implications include amplification of EGFR (9%) and FGFR3 mutation. FGFR3 mutation, commonly found in low-grade NMIBC, was found at a lower frequency in high-grade MIBC (17%) and is strongly associated with papillary morphology. In addition, multiple mutations in epigenetic pathways were commonly found. Of the tumors analyzed, 76% had an inactivating mutation in one or more chromatin-regulatory genes, which could potentially be targeted by newly developed agents that target chromatin modifications.
Vascular endothelial growth factor (VEGF)
VEGF binds with high affinity to one of three tyrosine kinase receptors (VEGFR-1, VEGFR-2, and VEGFR-3) and mediates normal and pathogenic angiogenesis in various cancers, mostly through interaction with VEGFR-2. VEGF is often overexpressed in bladder cancer. A high level of VEGF in both serum and urine is correlated with tumor progression, high recurrence rate, and poor survival.125-128 VEGFR-2 is expressed in 50% of urothelial carcinomas and expression is significantly higher in MIBC than in NMIBC.129,130
VEGF has been investigated as a diagnostic marker for bladder cancer. VEGF level is elevated in the urine of patients with bladder cancer and correlates with tumor recurrence rates.125 Several studies have compared the diagnostic potential of urinary VEGF vs. standard voided urine cytology, which has low sensitivity, particularly for low-grade and low-stage tumors.131 In a study analyzing VEGF level in urinary supernatants from 219 patients by enzyme-linked immunosorbent assay (ELISA) and Western blot, sensitivity and specificity were 76.7% and 61.5%, respectively, for VEGF compared to 70.8% and 90.8%, respectively, for voided urine cytology.132 In a case-control study with voided urine samples from 127 patients (63 control and 64 tumor-bearing), urinary concentration of VEGF as measured by ELISA showed accuracy of detection, with AUC of 0.886 and sensitivity and specificity of 83% and 87%, respectively.133
Inoue et al. evaluated the prognostic value of tumor VEGF expression in patients with MIBC treated with neoadjuvant MVAC chemotherapy and radical cystectomy.126 They examined VEGF expression before and after neoadjuvant chemotherapy, along with basic fibroblast growth factor and interleukin-8 expression and microvessel density by IHC in biopsy specimens from 55 patients with MIBC. VEGF expression and microvessel density in pretreated biopsy samples showed a correlation with disease recurrence (P = 0.032 and P = 0.015, respectively). Thus, VEGF appears to be a rational therapeutic target. Various VEGF-targeting agents are currently being evaluated in MIBC.
In a phase II study, bevacizumab, a monoclonal antibody targeting VEGF-A, was evaluated in combination with ddMVAC prior to radical cystectomy.56 Although this combination regimen demonstrated good tumor response with complete pathologic response in 38% of patients, the actual role of bevacizumab could not be assessed due to the study's singlearm design. Neoadjuvant bevacizumab was also tested in combination with a GC regimen.134 For 13 patients, 11 of whom underwent cystectomy, the bevacizumab-GC combination resulted in a mild antitumor effect, with pTis in three patients (27%) and no pathologic complete response. This combination was associated with a high rate (42%) of postoperative complications.
Sunitinib, an oral inhibitor of multiple tyrosine kinase receptors, including VEGFR, has shown synergistic antitumor effects with both cisplatin and gemcitabine in preclinical urothelial cancer models.135 Sunitinib monotherapy had mixed effects in patients with advanced urothelial cancers, either as a first-line therapy in patients who were not candidates for cisplatin-based chemotherapy or as second-line therapy after chemotherapy.136,137 A randomized phase II study evaluating the role of sunitinib as a maintenance therapy after chemotherapy in advanced urothelial carcinoma was closed prematurely due to slow accrual.138 The results of this study showed no benefit for sunitinib maintenance vs. observation.
Sunitinib in combination with chemotherapy is associated with high rates of toxicity. In a phase II trial, sunitinib in combination with GC as first-line therapy in metastatic disease and as neoadjuvant therapy in MIBC caused grade 3–4 hematologic toxicities in 70% of patients, causing the study to be discontinued.139 Another phase II trial of sunitinib in combination with GC as neoadjuvant therapy also closed prematurely due to withdrawal of financial support after accrual of only 18 patients. Despite the limited number of evaluable patients, sunitinib in combination with GC resulted in only one pT0 and was associated with significant hematologic toxicity.140
Her2
Several studies have reported Her2 overexpression rates of 5%–80% in bladder cancer, using various techniques and criteria.141-145 Lae et al. analyzed 1005 invasive bladder cancer tissue samples for overexpression of Her2 and Her2 gene amplification using standard IHC and fluorescence in situ hybridization (FISH), following current guidelines of the American Society of Clinical Oncology/College of American Pathologists.146 Her2 protein overexpression was found by IHC in 11.4% of tissue samples, and Her2 gene amplification was found by FISH in 5.1% of invasive bladder carcinomas. A study evaluating 80 cystectomy and lymph node dissection specimens found 28% (22/80) of cystectomy cases were Her2-positive by IHC and 53% (17/32) were positive in the lymph nodes. In this study, Her2 overexpression was not predictive of survival.142 In an ongoing phase II study (NCT01353222) using DN24-02, an autologous cellular immunotherapy targeting Her2 in the adjuvant setting after cystectomy, all patients had to have ≥1+ Her2 expression by IHC to be eligible. Of 61 patients screened, 49 (80%) had a Her2 expression score of ≥1+, 27 (44%) had a score of ≥2+ and 3 (5%) a 3+ score in the primary tumor. Of 23 patients who had Her2 expression levels evaluated in tumor from involved lymph nodes, 20 (87%) had a Her2 expression score of ≥1+, 12 (52%) had a score of ≥2+, and 3 (15%) had a 3+ score in the lymph nodes.145
Several agents that target Her2 have been evaluated in advanced bladder cancer. In a phase II trial, trastuzumab, a humanized monoclonal antibody targeting Her2, used in combination with paclitaxel, gemcitabine, and carboplatin resulted in a 70% response rate, including complete responses in patients with metastatic Her2-positive bladder cancer.147 Trastuzumab is being evaluated in combination with concurrent chemoradiation therapy with paclitaxel in patients with MIBC who have undergone TURBT and are not candidates for radical cystectomy.148 Lapatinib, an oral dual EGFR and Her2 tyrosine kinase inhibitor, is also being studied in combination with chemotherapy for metastatic urothelial carcinoma.149,150
Epidermal growth factor receptor (EGFR)
EGFR is a member of the ErbB family of tyrosine kinase receptors and is overexpressed in various epithelial tumors, including breast, lung, gastrointestinal, cervical, and bladder cancer.151,152 In one study, EGFR positivity was observed in 14 of 22 bladder tumors (74%).153 EGFR overexpression in urothelial carcinoma is associated with higher grade, higher rate of relapse, and poor overall survival.154-156 EGFR gene expression is closely associated with cisplatin resistance.157
Pruthie et al. conducted a phase II open-label trial of neoadjuvant erlotinib, an EGFR tyrosine kinase inhibitor, in patients with MIBC.158 A total of 20 patients received the full four-week neoadjuvant course of erlotinib prior to radical cystectomy. Complete pathologic response was reached in five patients (25%); two patients (10%) had pTis–T1. A similar trial at M.D. Anderson is assessing complete pathologic response rate after three to five weeks of erlotinib therapy in patients with MIBC.159
Cetuximab is a monoclonal antibody against EGFR. Although it has modest antitumor effects as a single agent, when combined with paclitaxel cetuximab showed moderate antitumor efficacy, with an overall response rate of 25% in previously treated urothelial cancers.160 A phase II trial (TUXEDO) of cetuximab in combination with concurrent chemoradiation therapy with either mitomycin C and 5-FU or cisplatin in MIBC is underway in the United Kingdom.
Mammalian target of rapamycin (mTOR)
mTOR is a serine/threonine protein kinase and part of the PI3K/Akt/mTOR pathway, which plays a critical role in cell growth, angiogenesis, protein synthesis, and cell survival.161 Mutations, copy number alterations, or RNA expression changes affecting the PI3K/Akt/mTOR pathway are commonly found in various malignancies, including bladder cancer.124,162 Data from Memorial Sloan-Kettering showed mutations or copy number gains/losses of genes in the PI3K/Akt/mTOR pathway, including PIK3CA, PIK3R1, TSC1, PTEN, and the AKT3 isoforms in 26 of 95 patients (27%) with high-grade MIBC. These genetic alterations were associated with a trend toward longer time to recurrence (HR 0.53; P = 0.08).163 In a single-arm, non-randomized phase II study at the same institution, everolimus, an oral mTOR inhibitor, exhibited only mild antitumor activity in metastatic urothelial cancer patients refractory to cytotoxic chemotherapy.164 In another single-institution phase II study, everolimus showed antitumor activity in only a small number of patients with advanced bladder cancer.165 However, some genetic alterations, such as TSC1 mutation166 or mTOR activation mutations (E2419K and E2014K),167 have been identified by whole-genome sequencing in a subset of patients. These alterations are associated with exceptional responses to everolimus, which validates the clinical value of next-generation molecular techniques in achieving more precise and personalized therapy.
Several trials of mTOR inhibitors, including sirolimus, temsirolimus, and everolimus, are underway in advanced bladder cancer, either in combination with a standard chemotherapy regimen168,169 or as monotherapy.170,171 A study of neoadjuvant sirolimus in MIBC is ongoing.172
Imaging
Advanced imaging techniques are improving pretreatment staging of MIBC. PET/CT, which is not recommended for routine staging, has proven prognostic value in MIBC. In small studies, positive PET/CT prior to planned cystectomy in patients with no evidence of metastatic disease by conventional staging methods has been associated with poor survival.173,174 Mertens et al. investigated the ability of 18F-fluorodeoxyglucose (FDG)–PET/CT to detect extravesicular lesions and their association with overall survival.175 Of 211 patients with MIBC, 98 (46.4%) had one or more extravesicular lesions on PET/CT, and 113 (53.5%) had a negative PET/CT. Patients with a positive PET/CT had significantly shorter overall and disease-specific survival (median overall survival: 14 vs. 50 months, P = 0.001; disease-specific survival: 16 vs. 50 months, P < 0.001). In a multivariate analysis, the presence of extravesicular lesions on PET/CT was confirmed as an independent prognostic indicator of mortality (HR 3.0). In another study by Apolo et al., FDG PET/CT lesion-based analysis showed an overall sensitivity and specificity of 87% (95% CI: 76% to 94%) and 88% (95% CI: 78% to 95%), respectively176. In this study, 10 patients with organ-confined disease diagnosed on CT were found to have more extensive disease on FDG-PET/CT. As a result of the findings on FDG-PET/CT, patients scheduled for curative treatment with radical cystectomy had their surgery cancelled and instead were treated with systemic chemotherapy for more advanced disease.
Conclusion
MIBC is an aggressive tumor with a high rate of recurrence and poor overall prognosis. Radical cystectomy is the standard treatment for MIBC. Even with substantial improvements in surgical techniques, mortality in MIBC remains high; radical cystectomy alone has an especially high rate of systemic failure. Despite a clearly demonstrated survival benefit, cisplatin-based neoadjuvant chemotherapy has been underutilized. However, numerous institutions are increasingly adopting perioperative chemotherapy regimens. In carefully selected patients with MIBC, various bladder-preserving strategies can be considered. Trimodal approaches including definitive non-cystectomy surgical resection in combination with chemotherapy and radiation have shown encouraging clinical outcomes. Various prognostic and predictive markers in MIBC have been discovered as our understanding of molecular biology improves. Furthermore, multiple molecular therapeutic targets have been identified and clinical trials are ongoing to assess their safety and efficacy in MIBC patients. Lastly, advances in imaging techniques, such as FDG PET/CT, used in addition to standard CT may not only improve detection of more advanced disease, but may also have prognostic value in MIBC.
Table 1.
Selected neoadjuvant chemotherapy studies.
| Study Design | Regimen | No of Patients | pCR (%) | Median OS (months) | Survival Benefit | Author | Year |
|---|---|---|---|---|---|---|---|
| Phase III RCT177 | Cisplatin | 62 | 19.6 | 34.2 in Cisplatin arm 23.1 in Control arm |
No | Martinez- Pireiro | 1995 |
| Phase III RCT10 | MVAC | 153 | 38 | 77 in MVAC arm 46 in Control arm |
Yes | Grossman | 2003 |
| Phase III RCT178 | CMV | 491 | 32.5 | 44 in CMV arm 37 in Control arm |
Yes | MRC/EORTC | 2011 |
| Phase II61 | PCaG Arm 1(T2-3N0) Arm 2 (T4 or N+) |
22 29 |
32 21 |
18.8 28.5 |
Smith | 2008 | |
| Phase II179 | GC | 15 | 26.7 | 36 | Herchenhorn | 2007 | |
| Phase II180 | ddDVAC + bevacizumab | 44 | 39 | Not yet reached | Siefker-Radtke | 2011 | |
| Phase II181 | ddDVAC | 44 | 36.1 | Plimack | 2012 | ||
| Retrospective182 | GC | 25 | 20 | Scosyrev | 2012 | ||
| Retrospective51 | GC MVAC |
42 54 |
26.2 28 |
Dash | 2008 | ||
| Retrospective183 | GC MVAC |
22 9 |
50 22.2 |
Kaneko | 2011 | ||
| Retrospective50 | GC MVAC |
37 77 |
25 31 |
Yeshchina | 2012 | ||
| Retrospective184 | GC MVAC |
24 22 |
25 22.5 |
35.3 | Pal | 2012 |
RCT = randomized controlled trial; GC = gemcitabine and cisplatin; MVAC = methotrexate, vinblastine, doxorubicin, and cisplatin; PCaG = paclitaxel, carboplatin, and gemcitabine; CMV = cisplatin, methotrexate, and vinblastine; ddDVAC = dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin.
Table 2.
Selected adjuvant chemotherapy studies.
| Study design | Regimen | No of patients | Survival | Survival Benefit | Author | Year | |
|---|---|---|---|---|---|---|---|
| Median OS | 5-year OS | ||||||
| Phase II RCT67 | CAP | 91 | 4.3years in CAP arm 2.4 years in Control arm | Yes | Skinner | 1991 | |
| Phase II RCT66 | Cisplatin | 77 | 57% in Cisplain arm 54% in Control arm | No | Studer | 1994 | |
| Phase II RCT185 | M-VAC/M- VEC | 49 | No | Stockle | 1995 | ||
| Phase II RCT186 | CMV | 55 | 63months in CMV arm 36 months in Control arm | No | Freiha | 1996 | |
| Phase II76 | Arm A; AG→PC (nRF) Arm B; AG→PCa (iRF) |
20 30 |
4.6 years in AG→PC arm 2.5 years in Control arm 3.4 years in AG→PCa arm 2 years in Control arm |
b Yes b Yes |
Gallagher | 2009 | |
| Phase III RCT187 | PGC | 142 | Not yet reached in PGC arm 26 months in Control arm | Yes | Paz-Ares | 2010 | |
| Phase III RCT75 | MVAC | a114 | P=0.41 p53 + vs. – P=0.89 MVAC vs. Control | No | Stadler | 2011 | |
| Phase III RCT72 | GC | 194 | 53.7% in GC Arm 43.4% in Control arm | No | Cognetti | 2012 | |
| Phase III RCT188 | Gemcitabine | 115 | 32 months in G-adj arm 31 months in G-prog arm | No | Lehmann | 2013 | |
RCT = randomized controlled trial; CAP = cyclophosphamide, doxorubicin (Adriamycin), and cisplatin; M-VAC = methotrexate, vinblastine, doxorubicin (Adriamycin), and cisplatin; M-VEC = methotrexate, vinblastine, epirubicin, and cisplatin; CMV = cisplatin, methotrexate, and vinblastine; PGC = paclitaxel, gemcitabine, and cisplatin; GC = gemcitabine and cisplatin; AG = doxorubicin and gemcitabine; PC = paclitaxel and cisplatin; PCa = paclitaxel and carboplatin; OS = overall survival; mOS = median overall survival; G-adj = adjuvant gemcitabine; G-prog = gemcitabine in progress; nRF = normal renal function; iRF = impaired renal function
p53 IHC positive, pT1/T2N0M0 patients
compared to matched non-adjuvant chemotherapy control
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Ploeg M, Aben KK, Kiemeney LA. The present and future burden of urinary bladder cancer in the world. World J Urol. 2009;27:289–93. doi: 10.1007/s00345-009-0383-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. doi: 10.3322/caac.21166. [DOI] [PubMed] [Google Scholar]
- 3.Freedman ND, Silverman DT, Hollenbeck AR, et al. Association between smoking and risk of bladder cancer among men and women. JAMA. 2011;306:737–45. doi: 10.1001/jama.2011.1142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zeegers MP, Swaen GM, Kant I, et al. Occupational risk factors for male bladder cancer: results from a population based case cohort study in the Netherlands. Occup Environ Med. 2001;58:590–6. doi: 10.1136/oem.58.9.590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gaertner RR, Trpeski L, Johnson KC. A case-control study of occupational risk factors for bladder cancer in Canada. Cancer Causes Control. 2004;15:1007–19. doi: 10.1007/s10552-004-1448-7. [DOI] [PubMed] [Google Scholar]
- 6.Apolo AB, Dahut W. Bladder cancer. In: Allegra C, editor. The Bethesda Handbook of Clinical Oncology. Philadelphia: Lippincott Williams & Wilkins; 2014. pp. 208–18. [Google Scholar]
- 7.Agarwal PK, Black PC, Kamat AM. Considerations on the use of diagnostic markers in management of patients with bladder cancer. World J Urol. 2008;26:39–44. doi: 10.1007/s00345-007-0232-1. [DOI] [PubMed] [Google Scholar]
- 8.Kirkali Z, Chan T, Manoharan M, et al. Bladder cancer: epidemiology, staging and grading, and diagnosis. Urology. 2005;66:4–34. doi: 10.1016/j.urology.2005.07.062. [DOI] [PubMed] [Google Scholar]
- 9.Raghavan D. Chemotherapy and cystectomy for invasive transitional cell carcinoma of bladder. Urol Oncol. 2003;21:468–74. doi: 10.1016/s1078-1439(03)00145-5. [DOI] [PubMed] [Google Scholar]
- 10.Grossman HB, Natale RB, Tangen CM, et al. Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N Engl J Med. 2003;349:859–66. doi: 10.1056/NEJMoa022148. [DOI] [PubMed] [Google Scholar]
- 11.Stein JP, Lieskovsky G, Cote R, et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. J Clin Oncol. 2001;19:666–75. doi: 10.1200/JCO.2001.19.3.666. [DOI] [PubMed] [Google Scholar]
- 12.Apolo AB, Grossman HB, Bajorin D, et al. Practical use of perioperative chemotherapy for muscle-invasive bladder cancer: summary of session at the Society of Urologic Oncology annual meeting. Urol Oncol. 2012;30:772–80. doi: 10.1016/j.urolonc.2012.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Madersbacher S, Hochreiter W, Burkhard F, et al. Radical cystectomy for bladder cancer today--a homogeneous series without neoadjuvant therapy. J Clin Oncol. 2003;21:690–6. doi: 10.1200/JCO.2003.05.101. [DOI] [PubMed] [Google Scholar]
- 14.Karl A, Carroll PR, Gschwend JE, et al. The impact of lymphadenectomy and lymph node metastasis on the outcomes of radical cystectomy for bladder cancer. Eur Urol. 2009;55:826–35. doi: 10.1016/j.eururo.2009.01.004. [DOI] [PubMed] [Google Scholar]
- 15.Leissner J, Ghoneim MA, Abol-Enein H, et al. Extended radical lymphadenectomy in patients with urothelial bladder cancer: results of a prospective multicenter study. J Urol. 2004;171:139–44. doi: 10.1097/01.ju.0000102302.26806.fb. [DOI] [PubMed] [Google Scholar]
- 16.Dhar NB, Klein EA, Reuther AM, et al. Outcome after radical cystectomy with limited or extended pelvic lymph node dissection. J Urol. 2008;179:873–8. doi: 10.1016/j.juro.2007.10.076. discussion 8. [DOI] [PubMed] [Google Scholar]
- 17.Canter D, Guzzo TJ, Resnick MJ, et al. A thorough pelvic lymph node dissection in presence of positive margins associated with better clinical outcomes in radical cystectomy patients. Urology. 2009;74:161–5. doi: 10.1016/j.urology.2009.01.024. [DOI] [PubMed] [Google Scholar]
- 18.Herr HW, Faulkner JR, Grossman HB, et al. Surgical factors influence bladder cancer outcomes: a cooperative group report. J Clin Oncol. 2004;22:2781–9. doi: 10.1200/JCO.2004.11.024. [DOI] [PubMed] [Google Scholar]
- 19.Horsted F, West J, Grainge MJ. Risk of venous thromboembolism in patients with cancer:a systematic review and meta-analysis. PLoS Med. 2012;9:e1001275. doi: 10.1371/journal.pmed.1001275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shabsigh A, Korets R, Vora KC, et al. Defining early morbidity of radical cystectomy for patients with bladder cancer using a standardized reporting methodology. Eur Urol. 2009;55:164–74. doi: 10.1016/j.eururo.2008.07.031. [DOI] [PubMed] [Google Scholar]
- 21.Stimson CJ, Chang SS, Barocas DA, et al. Early and late perioperative outcomes following radical cystectomy: 90-day readmissions, morbidity and mortality in a contemporary series. J Urol. 2010;184:1296–300. doi: 10.1016/j.juro.2010.06.007. [DOI] [PubMed] [Google Scholar]
- 22.Zippe CD, Raina R, Massanyi EZ, et al. Sexual function after male radical cystectomy in a sexually active population. Urology. 2004;64:682–5. doi: 10.1016/j.urology.2004.05.056. discussion 5-6. [DOI] [PubMed] [Google Scholar]
- 23.Pruthi RS, Nielsen M, Smith A, et al. Fast track program in patients undergoing radical cystectomy: results in 362 consecutive patients. J Am Coll Surg. 2010;210:93–9. doi: 10.1016/j.jamcollsurg.2009.09.026. [DOI] [PubMed] [Google Scholar]
- 24.Rozet F, Lesur G, Cathelineau X, et al. Oncological evaluation of prostate sparing cystectomy: the Montsouris long-term results. J Urol. 2008;179:2170–4. doi: 10.1016/j.juro.2008.01.112. discussion 4-5. [DOI] [PubMed] [Google Scholar]
- 25.Schoenberg MP, Walsh PC, Breazeale DR, et al. Local recurrence and survival following nerve sparing radical cystoprostatectomy for bladder cancer: 10-year followup. J Urol. 1996;155:490–4. [PubMed] [Google Scholar]
- 26.Johar RS, Hayn MH, Stegemann AP, et al. Complications after robot-assisted radical cystectomy: results from the International Robotic Cystectomy Consortium. Eur Urol. 2013;64:52–7. doi: 10.1016/j.eururo.2013.01.010. [DOI] [PubMed] [Google Scholar]
- 27.Marshall SJ, Hayn MH, Stegemann AP, et al. Impact of surgeon and volume on extended lymphadenectomy at the time of robot-assisted radical cystectomy: results from the International Robotic Cystectomy Consortium (IRCC) BJU Int. 2013;111:1075–80. doi: 10.1111/j.1464-410X.2012.11583.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Soler S, Delgado C, Ballaz A, et al. Unsuspected pulmonary embolism in patients with cancer. Thromb Res. 2012;129(Suppl 1):S16–9. doi: 10.1016/S0049-3848(12)70010-5. [DOI] [PubMed] [Google Scholar]
- 29.Holmes DG, Thrasher JB, Park GY, et al. Long-term complications related to the modified Indiana pouch. Urology. 2002;60:603–6. doi: 10.1016/s0090-4295(02)01945-3. [DOI] [PubMed] [Google Scholar]
- 30.Cancrini A, De Carli P, Pompeo V, et al. Lower urinary tract reconstruction following cystectomy: experience and results in 96 patients using the orthotopic ileal bladder substitution of Studer et al. Eur Urol. 1996;29:204–9. [PubMed] [Google Scholar]
- 31.Hautmann RE, de Petriconi R, Gottfried HW, et al. The ileal neobladder: complications and functional results in 363 patients after 11 years of followup. J Urol. 1999;161:422–7. doi: 10.1016/s0022-5347(01)61909-8. discussion 7-8. [DOI] [PubMed] [Google Scholar]
- 32.Stein JP, Dunn MD, Quek ML, et al. The orthotopic T pouch ileal neobladder: experience with 209 patients. J Urol. 2004;172:584–7. doi: 10.1097/01.ju.0000131651.77048.73. [DOI] [PubMed] [Google Scholar]
- 33.Carrion R, Arap S, Corcione G, et al. A multi-institutional study of orthotopic neobladders: functional results in men and women. BJU Int. 2004;93:803–6. doi: 10.1111/j.1464-410X.2003.04743.x. [DOI] [PubMed] [Google Scholar]
- 34.Madersbacher S, Mohrle K, Burkhard F, et al. Long-term voiding pattern of patients with ileal orthotopic bladder substitutes. J Urol. 2002;167:2052–7. [PubMed] [Google Scholar]
- 35.Lee RK, Abol-Enein H, Artibani W, et al. Urinary diversion after radical cystectomy for bladder cancer: options, patient selection, and outcomes. BJU Int. 2014;113:11–23. doi: 10.1111/bju.12121. [DOI] [PubMed] [Google Scholar]
- 36.Parekh DJ, Donat SM. Urinary diversion: options, patient selection, and outcomes. Semin Oncol. 2007;34:98–109. doi: 10.1053/j.seminoncol.2006.12.010. [DOI] [PubMed] [Google Scholar]
- 37.Raghavan D, Shipley WU, Garnick MB, et al. Biology and management of bladder cancer. N Engl J Med. 1990;322:1129–38. doi: 10.1056/NEJM199004193221607. [DOI] [PubMed] [Google Scholar]
- 38.Kaufman DS. Challenges in the treatment of bladder cancer. Ann Oncol. 2006;17(Suppl 5):v106–12. doi: 10.1093/annonc/mdj963. [DOI] [PubMed] [Google Scholar]
- 39.Shipley WU, Cummings KB, Coombs LJ, et al. 4,000 RAD preoperative irradiation followed by prompt radical cystectomy for invasive bladder carcinoma: a prospective study of patient tolerance and pathologic downstaging. J Urol. 1982;127:48–51. doi: 10.1016/s0022-5347(17)53598-3. [DOI] [PubMed] [Google Scholar]
- 40.Cole CJ, Pollack A, Zagars GK, et al. Local control of muscle-invasive bladder cancer: preoperative radiotherapy and cystectomy versus cystectomy alone. Int J Radiat Oncol Biol Phys. 1995;32:331–40. doi: 10.1016/0360-3016(95)00086-E. [DOI] [PubMed] [Google Scholar]
- 41.Crawford ED, Das S, Smith JA., Jr Preoperative radiation therapy in the treatment of bladder cancer. Urol Clin North Am. 1987;14:781–7. [PubMed] [Google Scholar]
- 42.Neoadjuvant cisplatin for advanced bladder cancer. Cochrane Database Syst Rev. 2000:CD001426. doi: 10.1002/14651858.CD001426. [DOI] [PubMed] [Google Scholar]
- 43.Neoadjuvant cisplatin, methotrexate, and vinblastine chemotherapy for muscle-invasive bladder cancer: a randomised controlled trial. International collaboration of trialists. Lancet. 1999;354:533–40. [PubMed] [Google Scholar]
- 44.Sternberg CN, Yagoda A, Scher HI, et al. Preliminary results of M-VAC (methotrexate, vinblastine, doxorubicin and cisplatin) for transitional cell carcinoma of the urothelium. J Urol. 1985;133:403–7. doi: 10.1016/s0022-5347(17)48996-8. [DOI] [PubMed] [Google Scholar]
- 45.Loehrer PJ, Sr, Einhorn LH, Elson PJ, et al. A randomized comparison of cisplatin alone or in combination with methotrexate, vinblastine, and doxorubicin in patients with metastatic urothelial carcinoma: a cooperative group study. J Clin Oncol. 1992;10:1066–73. doi: 10.1200/JCO.1992.10.7.1066. [DOI] [PubMed] [Google Scholar]
- 46.Logothetis CJ, Dexeus FH, Finn L, et al. A prospective randomized trial comparing MVAC and CISCA chemotherapy for patients with metastatic urothelial tumors. J Clin Oncol. 1990;8:1050–5. doi: 10.1200/JCO.1990.8.6.1050. [DOI] [PubMed] [Google Scholar]
- 47.Neoadjuvant chemotherapy in invasive bladder cancer: a systematic review and meta-analysis. Lancet. 2003;361:1927–34. doi: 10.1016/s0140-6736(03)13580-5. [DOI] [PubMed] [Google Scholar]
- 48.Winquist E, Kirchner TS, Segal R, et al. Neoadjuvant chemotherapy for transitional cell carcinoma of the bladder: a systematic review and meta-analysis. J Urol. 2004;171:561–9. doi: 10.1097/01.ju.0000090967.08622.33. [DOI] [PubMed] [Google Scholar]
- 49.von der Maase H, Hansen SW, Roberts JT, et al. Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol. 2000;18:3068–77. doi: 10.1200/JCO.2000.18.17.3068. [DOI] [PubMed] [Google Scholar]
- 50.Yeshchina O, Badalato GM, Wosnitzer MS, et al. Relative efficacy of perioperative gemcitabine and cisplatin versus methotrexate, vinblastine, adriamycin, and cisplatin in the management of locally advanced urothelial carcinoma of the bladder. Urology. 2012;79:384–90. doi: 10.1016/j.urology.2011.10.050. [DOI] [PubMed] [Google Scholar]
- 51.Dash A, Pettus JAt, Herr HW, et al. A role for neoadjuvant gemcitabine plus cisplatin in muscle-invasive urothelial carcinoma of the bladder: a retrospective experience. Cancer. 2008;113:2471–7. doi: 10.1002/cncr.23848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Fairey AS, Daneshmand S, Quinn D, et al. Neoadjuvant chemotherapy with gemcitabine/cisplatin vs. methotrexate/vinblastine/doxorubicin/cisplatin for muscle-invasive urothelial carcinoma of the bladder: a retrospective analysis from the University of Southern California. Urol Oncol. 2013;31:1737–43. doi: 10.1016/j.urolonc.2012.07.005. [DOI] [PubMed] [Google Scholar]
- 53.Sternberg CN, de Mulder PH, Schornagel JH, et al. Randomized phase III trial of high-dose-intensity methotrexate, vinblastine, doxorubicin, and cisplatin (MVAC) chemotherapy and recombinant human granulocyte colony-stimulating factor versus classic MVAC in advanced urothelial tract tumors: European Organization for Research and Treatment of Cancer Protocol no. 30924. J Clin Oncol. 2001;19:2638–46. doi: 10.1200/JCO.2001.19.10.2638. [DOI] [PubMed] [Google Scholar]
- 54.Plimack ER, Hoffman-Censits J, Viterbo R, et al. Neoadjuvant accelerated MVAC (AMVAC) in patients with muscle invasive bladder cancer: Results of a multicenter phase II study. J Clin Oncol. 2012;30 doi: 10.1200/JCO.2013.53.2465. abstr 4526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Qu A, Jacobus S, Signoretti S, et al. Phase II study of neoadjuvant dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin (ddMVAC) chemotherapy in patients with muscle-invasive urothelial cancer (MI-UC): Pathologic and radiologic response, serum tumor markers, and DNA excision repair pathway biomarkers in relation to disease-free survival (DFS) J Clin Oncol. 2013;31 abstr 4530. [Google Scholar]
- 56.Siefker-Radtke A, Kamat AM, Corn P, et al. Neoadjuvant chemotherapy with DDMVAC and bevacizumab in high-risk urothelial cancer: Results from a phase II trial at the M. D. Anderson Cancer Center. J Clin Oncol. 2012;30(suppl 5) abstr 261. [Google Scholar]
- 57.Blick C, Hall P, Pwint T, et al. Accelerated methotrexate, vinblastine, doxorubicin, and cisplatin (AMVAC) as neoadjuvant chemotherapy for patients with muscle-invasive transitional cell carcinoma of the bladder. Cancer. 2012;118:3920–7. doi: 10.1002/cncr.26675. [DOI] [PubMed] [Google Scholar]
- 58.Bamias A, Dafni U, Karadimou A, et al. Prospective, open-label, randomized, phase III study of two dose-dense regimens MVAC versus gemcitabine/cisplatin in patients with inoperable, metastatic or relapsed urothelial cancer: a Hellenic Cooperative Oncology Group study (HE 16/03) Ann Oncol. 2013;24:1011–7. doi: 10.1093/annonc/mds583. [DOI] [PubMed] [Google Scholar]
- 59.Sahut D'Izarn M, Caumont Prim A, Planquette B, et al. Risk factors and clinical outcome of unsuspected pulmonary embolism in cancer patients: a case-control study. J Thromb Haemost. 2012;10:2032–8. doi: 10.1111/j.1538-7836.2012.04868.x. [DOI] [PubMed] [Google Scholar]
- 60.Hussain M, Vaishampayan U, Du W, et al. Combination paclitaxel, carboplatin, and gemcitabine is an active treatment for advanced urothelial cancer. J Clin Oncol. 2001;19:2527–33. doi: 10.1200/JCO.2001.19.9.2527. [DOI] [PubMed] [Google Scholar]
- 61.Smith DC, Mackler NJ, Dunn RL, et al. Phase II trial of paclitaxel, carboplatin and gemcitabine in patients with locally advanced carcinoma of the bladder. J Urol. 2008;180:2384–8. doi: 10.1016/j.juro.2008.08.075. discussion 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.deVere White RW, Lara PN, Jr, Goldman B, et al. A sequential treatment approach to myoinvasive urothelial cancer: a phase II Southwest Oncology Group trial (S0219) J Urol. 2009;181:2476–80. doi: 10.1016/j.juro.2009.01.115. discussion 80-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Nagane M, Nishikawa R, Narita Y, et al. Phase II study of single-agent bevacizumab in Japanese patients with recurrent malignant glioma. Jpn J Clin Oncol. 2012;42:887–95. doi: 10.1093/jjco/hys121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wood DP. Discrepancy between clinical and pathological stage: external validation of the impact on prognosis in an international radical cystectomy cohort. J Urol. 2011;186:2211–2. doi: 10.1016/j.juro.2011.10.012. [DOI] [PubMed] [Google Scholar]
- 65.Donat SM, Shabsigh A, Savage C, et al. Potential impact of postoperative early complications on the timing of adjuvant chemotherapy in patients undergoing radical cystectomy: a high-volume tertiary cancer center experience. Eur Urol. 2009;55:177–85. doi: 10.1016/j.eururo.2008.07.018. [DOI] [PubMed] [Google Scholar]
- 66.Studer UE, Bacchi M, Biedermann C, et al. Adjuvant cisplatin chemotherapy following cystectomy for bladder cancer: results of a prospective randomized trial. J Urol. 1994;152:81–4. doi: 10.1016/s0022-5347(17)32822-7. [DOI] [PubMed] [Google Scholar]
- 67.Skinner DG, Daniels JR, Russell CA, et al. The role of adjuvant chemotherapy following cystectomy for invasive bladder cancer: a prospective comparative trial. J Urol. 1991;145:459–64. doi: 10.1016/s0022-5347(17)38368-4. discussion 64-7. [DOI] [PubMed] [Google Scholar]
- 68.Millikan R, Dinney C, Swanson D, et al. Integrated therapy for locally advanced bladder cancer: final report of a randomized trial of cystectomy plus adjuvant M-VAC versus cystectomy with both preoperative and postoperative M-VAC. J Clin Oncol. 2001;19:4005–13. doi: 10.1200/JCO.2001.19.20.4005. [DOI] [PubMed] [Google Scholar]
- 69.Adjuvant chemotherapy in invasive bladder cancer: a systematic review and meta-analysis of individual patient data Advanced Bladder Cancer (ABC) Meta-analysis Collaboration. Eur Urol. 2005;48:189–99. doi: 10.1016/j.eururo.2005.04.005. discussion 99-201. [DOI] [PubMed] [Google Scholar]
- 70.Svatek RS, Shariat SF, Lasky RE, et al. The effectiveness of off-protocol adjuvant chemotherapy for patients with urothelial carcinoma of the urinary bladder. Clin Cancer Res. 2010;16:4461–7. doi: 10.1158/1078-0432.CCR-10-0457. [DOI] [PubMed] [Google Scholar]
- 71.Paz-Ares L, Solsona E, Esteban E, et al. Randomized phase III trial comparing adjuvant paclitaxel/gemcitabine/cisplatin (PGC) to observation in patients with resected invasive bladder cancer: Results of the Spanish Oncology Genitourinary Group (SOGUG) 99/01 study. J Clin Oncol. 2010;28(18S) abstr LBA4518. [Google Scholar]
- 72.Cognetti F, Ruggeri EM, Felici A, et al. Adjuvant chemotherapy with cisplatin and gemcitabine versus chemotherapy at relapse in patients with muscle-invasive bladder cancer submitted to radical cystectomy: an Italian, multicenter, randomized phase III trial. Ann Oncol. 2012;23:695–700. doi: 10.1093/annonc/mdr354. [DOI] [PubMed] [Google Scholar]
- 73.Esrig D, Elmajian D, Groshen S, et al. Accumulation of nuclear p53 and tumor progression in bladder cancer. N Engl J Med. 1994;331:1259–64. doi: 10.1056/NEJM199411103311903. [DOI] [PubMed] [Google Scholar]
- 74.Cote RJ, Esrig D, Groshen S, et al. p53 and treatment of bladder cancer. Nature. 1997;385:123–5. doi: 10.1038/385123b0. [DOI] [PubMed] [Google Scholar]
- 75.Stadler WM, Lerner SP, Groshen S, et al. Phase III study of molecularly targeted adjuvant therapy in locally advanced urothelial cancer of the bladder based on p53 status. J Clin Oncol. 2011;29:3443–9. doi: 10.1200/JCO.2010.34.4028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Gallagher DJ, Milowsky MI, Iasonos A, et al. Sequential adjuvant chemotherapy after surgical resection of high-risk urothelial carcinoma. Cancer. 2009;115:5193–201. doi: 10.1002/cncr.24570. [DOI] [PubMed] [Google Scholar]
- 77.David KA, Milowsky MI, Ritchey J, et al. Low incidence of perioperative chemotherapy for stage III bladder cancer 1998 to 2003: a report from the National Cancer Data Base. J Urol. 2007;178:451–4. doi: 10.1016/j.juro.2007.03.101. [DOI] [PubMed] [Google Scholar]
- 78.Porter MP, Kerrigan MC, Donato BM, et al. Patterns of use of systemic chemotherapy for Medicare beneficiaries with urothelial bladder cancer. Urol Oncol. 2011;29:252–8. doi: 10.1016/j.urolonc.2009.03.021. [DOI] [PubMed] [Google Scholar]
- 79.Fedeli U, Fedewa SA, Ward EM. Treatment of muscle invasive bladder cancer: evidence from the National Cancer Database, 2003 to 2007. J Urol. 2011;185:72–8. doi: 10.1016/j.juro.2010.09.015. [DOI] [PubMed] [Google Scholar]
- 80.Bochner B, Feifer A, Sperling D, et al. Multi-institutional quality care initiative (QCI) to improve the care of patients with invasive bladder cancer (BlCa) J Clin Oncol. 2014;32(suppl 4) abstr 298. [Google Scholar]
- 81.Apolo AB, Bochner B, Steinberg S, et al. Examining the management of muscle-invasive bladder cancer (MIBC) by medical oncologists (MO) J Clin Oncol. 2012;30(suppl 5) doi: 10.1016/j.urolonc.2013.12.012. abstr 298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Konety BR, Allareddy V, Herr H. Complications after radical cystectomy: analysis of population-based data. Urology. 2006;68:58–64. doi: 10.1016/j.urology.2006.01.051. [DOI] [PubMed] [Google Scholar]
- 83.Manoharan M, Ayyathurai R, Soloway MS. Radical cystectomy for urothelial carcinoma of the bladder: an analysis of perioperative and survival outcome. BJU Int. 2009;104:1227–32. doi: 10.1111/j.1464-410X.2009.08625.x. [DOI] [PubMed] [Google Scholar]
- 84.Herr HW. Transurethral resection of muscle-invasive bladder cancer: 10-year outcome. J Clin Oncol. 2001;19:89–93. doi: 10.1200/JCO.2001.19.1.89. [DOI] [PubMed] [Google Scholar]
- 85.Solsona E, Climent MA, Iborra I, et al. Bladder preservation in selected patients with muscle-invasive bladder cancer by complete transurethral resection of the bladder plus systemic chemotherapy: long-term follow-up of a phase 2 nonrandomized comparative trial with radical cystectomy. Eur Urol. 2009;55:911–9. doi: 10.1016/j.eururo.2008.08.027. [DOI] [PubMed] [Google Scholar]
- 86.Knoedler JJ, Boorjian SA, Kim SP, et al. Does partial cystectomy compromise oncologic outcomes for patients with bladder cancer compared to radical cystectomy? A matched case-control analysis. J Urol. 2012;188:1115–9. doi: 10.1016/j.juro.2012.06.029. [DOI] [PubMed] [Google Scholar]
- 87.Holzbeierlein JM, Lopez-Corona E, Bochner BH, et al. Partial cystectomy: a contemporary review of the Memorial Sloan-Kettering Cancer Center experience and recommendations for patient selection. J Urol. 2004;172:878–81. doi: 10.1097/01.ju.0000135530.59860.7d. [DOI] [PubMed] [Google Scholar]
- 88.Smaldone MC, Jacobs BL, Smaldone AM, et al. Long-term results of selective partial cystectomy for invasive urothelial bladder carcinoma. Urology. 2008;72:613–6. doi: 10.1016/j.urology.2008.04.052. [DOI] [PubMed] [Google Scholar]
- 89.Kassouf W, Swanson D, Kamat AM, et al. Partial cystectomy for muscle invasive urothelial carcinoma of the bladder: a contemporary review of the M. D. Anderson Cancer Center experience. J Urol. 2006;175:2058–62. doi: 10.1016/S0022-5347(06)00322-3. [DOI] [PubMed] [Google Scholar]
- 90.Capitanio U, Isbarn H, Shariat SF, et al. Partial cystectomy does not undermine cancer control in appropriately selected patients with urothelial carcinoma of the bladder: a population-based matched analysist. Urology. 2009;74:858–64. doi: 10.1016/j.urology.2009.03.052. [DOI] [PubMed] [Google Scholar]
- 91.Zhang M, Tao R, Zhang C, et al. Lymphovascular invasion and the presence of more than three tumors are associated with poor outcomes of muscle-invasive bladder cancer after bladder-conserving therapies. Urology. 2010;76:902–7. doi: 10.1016/j.urology.2010.05.007. [DOI] [PubMed] [Google Scholar]
- 92.Sternberg CN, Pansadoro V, Calabro F, et al. Can patient selection for bladder preservation be based on response to chemotherapy? Cancer. 2003;97:1644–52. doi: 10.1002/cncr.11232. [DOI] [PubMed] [Google Scholar]
- 93.Duncan W, Quilty PM. The results of a series of 963 patients with transitional cell carcinoma of the urinary bladder primarily treated by radical megavoltage X-ray therapy. Radiother Oncol. 1986;7:299–310. doi: 10.1016/s0167-8140(86)80059-7. [DOI] [PubMed] [Google Scholar]
- 94.van der Steen-Banasik E, Ploeg M, Witjes JA, et al. Brachytherapy versus cystectomy in solitary bladder cancer: a case control, multicentre, East-Netherlands study. Radiother Oncol. 2009;93:352–7. doi: 10.1016/j.radonc.2009.04.020. [DOI] [PubMed] [Google Scholar]
- 95.Cooke PW, Dunn JA, Latief T, et al. Long-term risk of salvage cystectomy after radiotherapy for muscle-invasive bladder cancer. Eur Urol. 2000;38:279–86. doi: 10.1159/000020294. [DOI] [PubMed] [Google Scholar]
- 96.Perdona S, Autorino R, Damiano R, et al. Bladder-sparing, combined-modality approach for muscle-invasive bladder cancer: a multi-institutional, long-term experience. Cancer. 2008;112:75–83. doi: 10.1002/cncr.23137. [DOI] [PubMed] [Google Scholar]
- 97.Gamal El-Deen H, Elshazly HF, Abo Zeina EA. Clinical experience with radiotherapy alone and radiochemotherapy with platin based regimens in organ-sparing treatment of invasive bladder cancer. J Egypt Natl Canc Inst. 2009;21:59–70. [PubMed] [Google Scholar]
- 98.Coppin CM, Gospodarowicz MK, James K, et al. Improved local control of invasive bladder cancer by concurrent cisplatin and preoperative or definitive radiation. The National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol. 1996;14:2901–7. doi: 10.1200/JCO.1996.14.11.2901. [DOI] [PubMed] [Google Scholar]
- 99.Hussain SA, Stocken DD, Peake DR, et al. Long-term results of a phase II study of synchronous chemoradiotherapy in advanced muscle invasive bladder cancer. Br J Cancer. 2004;90:2106–11. doi: 10.1038/sj.bjc.6601852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.James ND, Hussain SA, Hall E, et al. Radiotherapy with or without chemotherapy in muscle-invasive bladder cancer. N Engl J Med. 2012;366:1477–88. doi: 10.1056/NEJMoa1106106. [DOI] [PubMed] [Google Scholar]
- 101.Tester W, Porter A, Asbell S, et al. Combined modality program with possible organ preservation for invasive bladder carcinoma: results of RTOG protocol 85-12. Int J Radiat Oncol Biol Phys. 1993;25:783–90. doi: 10.1016/0360-3016(93)90306-g. [DOI] [PubMed] [Google Scholar]
- 102.Shipley WU, Kaufman DS, Zehr E, et al. Selective bladder preservation by combined modality protocol treatment: long-term outcomes of 190 patients with invasive bladder cancer. Urology. 2002;60:62–7. doi: 10.1016/s0090-4295(02)01650-3. discussion 7-8. [DOI] [PubMed] [Google Scholar]
- 103.Housset M, Maulard C, Chretien Y, et al. Combined radiation and chemotherapy for invasive transitional-cell carcinoma of the bladder: a prospective study. J Clin Oncol. 1993;11:2150–7. doi: 10.1200/JCO.1993.11.11.2150. [DOI] [PubMed] [Google Scholar]
- 104.Koga F, Kihara K. Selective bladder preservation with curative intent for muscle-invasive bladder cancer: a contemporary review. Int J Urol. 2012;19:388–401. doi: 10.1111/j.1442-2042.2012.02974.x. [DOI] [PubMed] [Google Scholar]
- 105.Rodel C, Grabenbauer GG, Kuhn R, et al. Combined-modality treatment and selective organ preservation in invasive bladder cancer: long-term results. J Clin Oncol. 2002;20:3061–71. doi: 10.1200/JCO.2002.11.027. [DOI] [PubMed] [Google Scholar]
- 106.Mitin T, Hunt D, Shipley WU, et al. Transurethral surgery and twice-daily radiation plus paclitaxel-cisplatin or fluorouracil-cisplatin with selective bladder preservation and adjuvant chemotherapy for patients with muscle invasive bladder cancer (RTOG 0233): a randomised multicentre phase 2 trial. Lancet Oncol. 2013;14:863–72. doi: 10.1016/S1470-2045(13)70255-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Efstathiou JA, Spiegel DY, Shipley WU, et al. Long-term outcomes of selective bladder preservation by combined-modality therapy for invasive bladder cancer: the MGH experience. Eur Urol. 2012;61:705–11. doi: 10.1016/j.eururo.2011.11.010. [DOI] [PubMed] [Google Scholar]
- 108.Kaufman DS, Winter KA, Shipley WU, et al. The initial results in muscle-invading bladder cancer of RTOG 95-06: phase I/II trial of transurethral surgery plus radiation therapy with concurrent cisplatin and 5-fluorouracil followed by selective bladder preservation or cystectomy depending on the initial response. Oncologist. 2000;5:471–6. doi: 10.1634/theoncologist.5-6-471. [DOI] [PubMed] [Google Scholar]
- 109.Kaufman DS, Winter KA, Shipley WU, et al. Phase I-II RTOG study (99-06) of patients with muscle-invasive bladder cancer undergoing transurethral surgery, paclitaxel, cisplatin, and twice-daily radiotherapy followed by selective bladder preservation or radical cystectomy and adjuvant chemotherapy. Urology. 2009;73:833–7. doi: 10.1016/j.urology.2008.09.036. [DOI] [PubMed] [Google Scholar]
- 110.Mitin T, George A, Zietman AL, et al. Long-term outcomes among patients who achieve complete or near-complete responses after the induction phase of bladder-preserving combined modality therapy for muscle-invasive bladder cancer: A pooled analysis of RTOG 9906 and 0233. J Clin Oncol. 2014;32(suppl 4) doi: 10.1016/j.ijrobp.2015.09.030. abstr 284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Bellmunt J, Albanell J, Paz-Ares L, et al. Pretreatment prognostic factors for survival in patients with advanced urothelial tumors treated in a phase I/II trial with paclitaxel, cisplatin, and gemcitabine. Cancer. 2002;95:751–7. doi: 10.1002/cncr.10762. [DOI] [PubMed] [Google Scholar]
- 112.Bellmunt J, Guillem V, Paz-Ares L, et al. Phase I-II study of paclitaxel, cisplatin, and gemcitabine in advanced transitional-cell carcinoma of the urothelium. Spanish Oncology Genitourinary Group. J Clin Oncol. 2000;18:3247–55. doi: 10.1200/JCO.2000.18.18.3247. [DOI] [PubMed] [Google Scholar]
- 113.Bellmunt J, von der Maase H, Mead GM, et al. Randomized phase III study comparing paclitaxel/cisplatin/gemcitabine and gemcitabine/cisplatin in patients with locally advanced or metastatic urothelial cancer without prior systemic therapy: EORTC Intergroup Study 30987. J Clin Oncol. 2012;30:1107–13. doi: 10.1200/JCO.2011.38.6979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Kim HL, Steinberg GD. Complications of cystectomy in patients with a history of pelvic radiation. Urology. 2001;58:557–60. doi: 10.1016/s0090-4295(01)01269-9. [DOI] [PubMed] [Google Scholar]
- 115.Eisenberg MS, Dorin RP, Bartsch G, et al. Early complications of cystectomy after high dose pelvic radiation. J Urol. 2010;184:2264–9. doi: 10.1016/j.juro.2010.08.007. [DOI] [PubMed] [Google Scholar]
- 116.Shipley WU, Winter KA, Kaufman DS, et al. Phase III trial of neoadjuvant chemotherapy in patients with invasive bladder cancer treated with selective bladder preservation by combined radiation therapy and chemotherapy: initial results of Radiation Therapy Oncology Group 89-03. J Clin Oncol. 1998;16:3576–83. doi: 10.1200/JCO.1998.16.11.3576. [DOI] [PubMed] [Google Scholar]
- 117.Chen RC, Shipley WU, Efstathiou JA, et al. Trimodality bladder preservation therapy for muscle-invasive bladder cancer. J Natl Compr Canc Netw. 2013;11:952–60. doi: 10.6004/jnccn.2013.0116. [DOI] [PubMed] [Google Scholar]
- 118.Apolo AB, Milowsky M, Bajorin DF. Clinical states model for biomarkers in bladder cancer. Future Oncol. 2009;5:977–92. doi: 10.2217/fon.09.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Smith SC, Baras AS, Dancik G, et al. A 20-gene model for molecular nodal staging of bladder cancer: development and prospective assessment. Lancet Oncol. 2011;12:137–43. doi: 10.1016/S1470-2045(10)70296-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Kim WJ, Kim EJ, Kim SK, et al. Predictive value of progression-related gene classifier in primary non-muscle invasive bladder cancer. Mol Cancer. 2010;9:3. doi: 10.1186/1476-4598-9-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Blaveri E, Simko JP, Korkola JE, et al. Bladder cancer outcome and subtype classification by gene expression. Clin Cancer Res. 2005;11:4044–55. doi: 10.1158/1078-0432.CCR-04-2409. [DOI] [PubMed] [Google Scholar]
- 122.Riester M, Taylor JM, Feifer A, et al. Combination of a novel gene expression signature with a clinical nomogram improves the prediction of survival in high-risk bladder cancer. Clin Cancer Res. 2012;18:1323–33. doi: 10.1158/1078-0432.CCR-11-2271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.International Bladder Cancer Nomogram C. Bochner BH, Kattan MW, et al. Postoperative nomogram predicting risk of recurrence after radical cystectomy for bladder cancer. J Clin Oncol. 2006;24:3967–72. doi: 10.1200/JCO.2005.05.3884. [DOI] [PubMed] [Google Scholar]
- 124.Comprehensive molecular characterization of urothelial bladder carcinoma. Nature. 2014;507:315–22. doi: 10.1038/nature12965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Crew JP, O'Brien T, Bicknell R, et al. Urinary vascular endothelial growth factor and its correlation with bladder cancer recurrence rates. J Urol. 1999;161:799–804. [PubMed] [Google Scholar]
- 126.Inoue K, Slaton JW, Karashima T, et al. The prognostic value of angiogenesis factor expression for predicting recurrence and metastasis of bladder cancer after neoadjuvant chemotherapy and radical cystectomy. Clin Cancer Res. 2000;6:4866–73. [PubMed] [Google Scholar]
- 127.Jeon SH, Lee SJ, Chang SG. Clinical significance of urinary vascular endothelial growth factor in patients with superficial bladder tumors. Oncol Rep. 2001;8:1265–7. doi: 10.3892/or.8.6.1265. [DOI] [PubMed] [Google Scholar]
- 128.Bernardini S, Fauconnet S, Chabannes E, et al. Serum levels of vascular endothelial growth factor as a prognostic factor in bladder cancer. J Urol. 2001;166:1275–9. [PubMed] [Google Scholar]
- 129.Kopparapu PK, Boorjian SA, Robinson BD, et al. Expression of VEGF and its receptors VEGFR1/VEGFR2 is associated with invasiveness of bladder cancer. Anticancer Res. 2013;33:2381–90. [PubMed] [Google Scholar]
- 130.Xia G, Kumar SR, Hawes D, et al. Expression and significance of vascular endothelial growth factor receptor 2 in bladder cancer. J Urol. 2006;175:1245–52. doi: 10.1016/S0022-5347(05)00736-6. [DOI] [PubMed] [Google Scholar]
- 131.Rife CC, Farrow GM, Utz DC. Urine cytology of transitional cell neoplasms. Urol Clin North Am. 1979;6:599–612. [PubMed] [Google Scholar]
- 132.Eissa S, Salem AM, Zohny SF, et al. The diagnostic efficacy of urinary TGF-beta1 and VEGF in bladder cancer: comparison with voided urine cytology. Cancer Biomark. 2007;3:275–85. doi: 10.3233/cbm-2007-3601. [DOI] [PubMed] [Google Scholar]
- 133.Urquidi V, Goodison S, Kim J, et al. Vascular endothelial growth factor, carbonic anhydrase 9, and angiogenin as urinary biomarkers for bladder cancer detection. Urology. 2012;79:1185e1–6. doi: 10.1016/j.urology.2012.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Chaudhary U, Golshayan A, Brisendine A, et al. Phase II trial of neoadjuvant cisplatin, gemcitabine, and bevacizumab followed by radical cystectomy (RC) in patients with muscle-invasive transitional cell carcinoma (TCC) of the bladder. J Clin Oncol. 2011;29(suppl 7) abstr 276. [Google Scholar]
- 135.Sonpavde G, Jian W, Liu H, et al. Sunitinib malate is active against human urothelial carcinoma and enhances the activity of cisplatin in a preclinical model. Urol Oncol. 2009;27:391–9. doi: 10.1016/j.urolonc.2008.03.017. [DOI] [PubMed] [Google Scholar]
- 136.Bellmunt J, Gonzalez-Larriba JL, Prior C, et al. Phase II study of sunitinib as first-line treatment of urothelial cancer patients ineligible to receive cisplatin-based chemotherapy: baseline interleukin-8 and tumor contrast enhancement as potential predictive factors of activity. Ann Oncol. 2011;22:2646–53. doi: 10.1093/annonc/mdr023. [DOI] [PubMed] [Google Scholar]
- 137.Gallagher DJ, Milowsky MI, Gerst SR, et al. Phase II study of sunitinib in patients with metastatic urothelial cancer. J Clin Oncol. 2010;28:1373–9. doi: 10.1200/JCO.2009.25.3922. [DOI] [PubMed] [Google Scholar]
- 138.Grivas PD, Daignault S, Tagawa ST, et al. Double-blind, randomized, phase 2 trial of maintenance sunitinib versus placebo after response to chemotherapy in patients with advanced urothelial carcinoma. Cancer. 2014;120:692–701. doi: 10.1002/cncr.28477. [DOI] [PubMed] [Google Scholar]
- 139.Galsky MD, Hahn NM, Powles T, et al. Gemcitabine, cisplatin, and sunitinib for metastatic urothelial carcinoma and as preoperative therapy for muscle-invasive bladder cancer. Clin Genitourin Cancer. 2013;11:175–81. doi: 10.1016/j.clgc.2012.10.001. [DOI] [PubMed] [Google Scholar]
- 140.Balar AV, Iyer G, Apolo AB, et al. Phase II trial of neoadjuvant gemcitabine (G) and cisplatin (C) with sunitinib in patients (pts) with muscle-invasive bladder cancer (MIBC) J Clin Oncol. 2012;30 abstr 45817̂. [Google Scholar]
- 141.Underwood M, Bartlett J, Reeves J, et al. C-erbB-2 gene amplification: a molecular marker in recurrent bladder tumors? Cancer Res. 1995;55:2422–30. [PubMed] [Google Scholar]
- 142.Jimenez RE, Hussain M, Bianco FJ, Jr, et al. Her-2/neu overexpression in muscle-invasive urothelial carcinoma of the bladder: prognostic significance and comparative analysis in primary and metastatic tumors. Clin Cancer Res. 2001;7:2440–7. [PubMed] [Google Scholar]
- 143.Latif Z, Watters AD, Dunn I, et al. HER2/neu overexpression in the development of muscle-invasive transitional cell carcinoma of the bladder. Br J Cancer. 2003;89:1305–9. doi: 10.1038/sj.bjc.6601245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Fleischmann A, Rotzer D, Seiler R, et al. Her2 amplification is significantly more frequent in lymph node metastases from urothelial bladder cancer than in the primary tumours. Eur Urol. 2011;60:350–7. doi: 10.1016/j.eururo.2011.05.035. [DOI] [PubMed] [Google Scholar]
- 145.Press MF, ODP, Plimack ER, Gomella LG, Quinn DI, Sharma P, DeVries T, Sims RB, Chen M, Bajorin DF. HER2 expression in patients (pts) with surgically resected urothelial cancer at high risk of recurrence screened for the phase II randomized, open-label trial of DN24-02, an autologous cellular immunotherapy targeting HER2. J Clin Oncol. 2013;31(suppl 6) abstr 292. [Google Scholar]
- 146.Lae M, Couturier J, Oudard S, et al. Assessing HER2 gene amplification as a potential target for therapy in invasive urothelial bladder cancer with a standardized methodology: results in 1005 patients. Ann Oncol. 2010;21:815–9. doi: 10.1093/annonc/mdp488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Hussain MH, Mac Vicar GR, Petrylak DP, et al. Trastuzumab, paclitaxel, carboplatin, and gemcitabine in advanced human epidermal growth factor receptor-2/neu-positive urothelial carcinoma: results of a multicenter phase II National Cancer Institute trial. J Clin Oncol. 2007;25:2218–24. doi: 10.1200/JCO.2006.08.0994. [DOI] [PubMed] [Google Scholar]
- 148.Sawant SP, Banumathy S, Daddi A, et al. Pulmonary embolism in cancer patients. Indian J Cancer. 2012;49:119–24. doi: 10.4103/0019-509X.98936. [DOI] [PubMed] [Google Scholar]
- 149.Thaler J, Ay C, Pabinger I. Venous thromboembolism in cancer patients - risk scores and recent randomised controlled trials. Thromb Haemost. 2012;108:1042–8. doi: 10.1160/TH12-04-0241. [DOI] [PubMed] [Google Scholar]
- 150.Halton J, Nagel K, Brandao LR, et al. Do children with central venous line (CVL) dysfunction have increased risk of symptomatic thromboembolism compared to those without CVL-dysfunction, while on cancer therapy? BMC Cancer. 2012;12:314. doi: 10.1186/1471-2407-12-314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Neal DE, Marsh C, Bennett MK, et al. Epidermal-growth-factor receptors in human bladder cancer: comparison of invasive and superficial tumours. Lancet. 1985;1:366–8. doi: 10.1016/s0140-6736(85)91386-8. [DOI] [PubMed] [Google Scholar]
- 152.Colquhoun AJ, Mellon JK. Epidermal growth factor receptor and bladder cancer. Postgrad Med J. 2002;78:584–9. doi: 10.1136/pmj.78.924.584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Chaux A, Cohen JS, Schultz L, et al. High epidermal growth factor receptor immunohistochemical expression in urothelial carcinoma of the bladder is not associated with EGFR mutations in exons 19 and 21: a study using formalin-fixed, paraffin-embedded archival tissues. Hum Pathol. 2012;43:1590–5. doi: 10.1016/j.humpath.2011.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Mellon K, Wright C, Kelly P, et al. Long-term outcome related to epidermal growth factor receptor status in bladder cancer. J Urol. 1995;153:919–25. [PubMed] [Google Scholar]
- 155.Kassouf W, Black PC, Tuziak T, et al. Distinctive expression pattern of ErbB family receptors signifies an aggressive variant of bladder cancer. J Urol. 2008;179:353–8. doi: 10.1016/j.juro.2007.08.087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Kramer C, Klasmeyer K, Bojar H, et al. Heparin-binding epidermal growth factor-like growth factor isoforms and epidermal growth factor receptor/ErbB 1 expression in bladder cancer and their relation to clinical outcome. Cancer. 2007;109:2016–24. doi: 10.1002/cncr.22627. [DOI] [PubMed] [Google Scholar]
- 157.Kim WT, Kim J, Yan C, et al. S100A9 and EGFR gene signatures predict disease progression in muscle invasive bladder cancer patients after chemotherapy. Ann Oncol. 2014 doi: 10.1093/annonc/mdu037. [DOI] [PubMed] [Google Scholar]
- 158.Pruthi RS, Nielsen M, Heathcote S, et al. A phase II trial of neoadjuvant erlotinib in patients with muscle-invasive bladder cancer undergoing radical cystectomy: clinical and pathological results. BJU Int. 2010;106:349–54. doi: 10.1111/j.1464-410X.2009.09101.x. [DOI] [PubMed] [Google Scholar]
- 159.Strowd RE, 3rd, Knovich MA, Lesser GJ. The therapeutic management of bleeding and thrombotic disorders complicating CNS malignancies. Curr Treat Options Oncol. 2012;13:451–64. doi: 10.1007/s11864-012-0207-3. [DOI] [PubMed] [Google Scholar]
- 160.Wong YN, Litwin S, Vaughn D, et al. Phase II trial of cetuximab with or without paclitaxel in patients with advanced urothelial tract carcinoma. J Clin Oncol. 2012;30:3545–51. doi: 10.1200/JCO.2012.41.9572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Gingras AC, Raught B, Sonenberg N. Regulation of translation initiation by FRAP/mTOR. Genes Dev. 2001;15:807–26. doi: 10.1101/gad.887201. [DOI] [PubMed] [Google Scholar]
- 162.Garcia JA, Danielpour D. Mammalian target of rapamycin inhibition as a therapeutic strategy in the management of urologic malignancies. Mol Cancer Ther. 2008;7:1347–54. doi: 10.1158/1535-7163.MCT-07-2408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Balar AV, Iyer G, Al-Ahmadie H, et al. Alterations in the PI3K/Akt signaling pathway and association with outcome in invasive high-grade urothelial cancer (UC) J Clin Oncol. 2012;30(suppl 5) abstr 277. [Google Scholar]
- 164.Milowsky MI, Iyer G, Regazzi AM, et al. Phase II study of everolimus in metastatic urothelial cancer. BJU Int. 2013;112:462–70. doi: 10.1111/j.1464-410X.2012.11720.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Seront E, Rottey S, Sautois B, et al. Phase II study of everolimus in patients with locally advanced or metastatic transitional cell carcinoma of the urothelial tract: clinical activity, molecular response, and biomarkers. Ann Oncol. 2012;23:2663–70. doi: 10.1093/annonc/mds057. [DOI] [PubMed] [Google Scholar]
- 166.Iyer G, Hanrahan AJ, Milowsky MI, et al. Genome sequencing identifies a basis for everolimus sensitivity. Science. 2012;338:221. doi: 10.1126/science.1226344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Wagle N, Grabiner BC, Van Allen EM, et al. Activating mTOR Mutations in a Patient with an Extraordinary Response on a Phase I Trial of Everolimus and Pazopanib. Cancer Discov. 2014 doi: 10.1158/2159-8290.CD-13-0353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Mandala M, Grosso F, Vitalini C, et al. Venous thromboembolism is a relevant and underestimated adverse event in cancer patients treated in phase I studies. Br J Cancer. 2012;107:612–6. doi: 10.1038/bjc.2012.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Reinke CE, Karakousis GC, Hadler RA, et al. Incidence of venous thromboembolism in patients undergoing surgical treatment for malignancy by type of neoplasm: An analysis of ACS-NSQIP data from 2005 to 2010. Surgery. 2012;152:186–92. doi: 10.1016/j.surg.2012.05.003. [DOI] [PubMed] [Google Scholar]
- 170.Drosik A, Trzepiora B, Kozielski J. Thromboembolic venous disease as a first sign of neoplastic disease--a case report. Wiad Lek. 2012;65:15–8. [PubMed] [Google Scholar]
- 171.Lovely JK, Nehring SA, Boughey JC, et al. Balancing venous thromboembolism and hematoma after breast surgery. Ann Surg Oncol. 2012;19:3230–5. doi: 10.1245/s10434-012-2524-y. [DOI] [PubMed] [Google Scholar]
- 172.Kyriazi V. Breast cancer as an acquired thrombophilic state. J Breast Cancer. 2012;15:148–56. doi: 10.4048/jbc.2012.15.2.148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Drieskens O, Oyen R, Van Poppel H, et al. FDG-PET for preoperative staging of bladder cancer. Eur J Nucl Med Mol Imaging. 2005;32:1412–7. doi: 10.1007/s00259-005-1886-9. [DOI] [PubMed] [Google Scholar]
- 174.Kibel AS, Dehdashti F, Katz MD, et al. Prospective study of [18F]fluorodeoxyglucose positron emission tomography/computed tomography for staging of muscle-invasive bladder carcinoma. J Clin Oncol. 2009;27:4314–20. doi: 10.1200/JCO.2008.20.6722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Mertens LS, Mir MC, Scott AM, et al. 18F-fluorodeoxyglucose--positron emission tomography/computed tomography aids staging and predicts mortality in patients with muscle-invasive bladder cancer. Urology. 2014;83:393–8. doi: 10.1016/j.urology.2013.10.032. [DOI] [PubMed] [Google Scholar]
- 176.Apolo AB, Riches J, Schoder H, et al. Clinical value of fluorine-18 2-fluoro-2-deoxy-D-glucose positron emission tomography/computed tomography in bladder cancer. J Clin Oncol. 2010;28:3973–8. doi: 10.1200/JCO.2010.28.7052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Martinez-Pineiro JA, Gonzalez Martin M, Arocena F, et al. Neoadjuvant cisplatin chemotherapy before radical cystectomy in invasive transitional cell carcinoma of the bladder: a prospective randomized phase III study. J Urol. 1995;153:964–73. [PubMed] [Google Scholar]
- 178.International Collaboration of T, Medical Research Council Advanced Bladder Cancer Working P, European Organisation for R et al. International phase III trial assessing neoadjuvant cisplatin, methotrexate, and vinblastine chemotherapy for muscle-invasive bladder cancer: long-term results of the BA06 30894 trial. J Clin Oncol. 2011;29:2171–7. doi: 10.1200/JCO.2010.32.3139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Herchenhorn D, Dienstmann R, Peixoto FA, et al. Phase II trial of neoadjuvant gemcitabine and cisplatin in patients with resectable bladder carcinoma. Int Braz J Urol. 2007;33:630–8. doi: 10.1590/s1677-55382007000500004. discussion 8. [DOI] [PubMed] [Google Scholar]
- 180.Rajput MZ, Kamat AM, Clavell-Hernandez J, et al. Perioperative outcomes of laparoscopic radical nephroureterectomy and regional lymphadenectomy in patients with upper urinary tract urothelial carcinoma after neoadjuvant chemotherapy. Urology. 2011;78:61–7. doi: 10.1016/j.urology.2011.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Plimack ER, Hoffman-Censits JH, Viterbo R, et al. Neoadjuvant accelerated MVAC (AMVAC) in patients with muscle invasive bladder cancer: Results of a multicenter phase II study. ASCO Meeting Abstracts. 2012;30:4526. [Google Scholar]
- 182.Scosyrev E, Messing EM, van Wijngaarden E, et al. Neoadjuvant gemcitabine and cisplatin chemotherapy for locally advanced urothelial cancer of the bladder. Cancer. 2012;118:72–81. doi: 10.1002/cncr.26238. [DOI] [PubMed] [Google Scholar]
- 183.Kaneko G, Kikuchi E, Matsumoto K, et al. Neoadjuvant gemcitabine plus cisplatin for muscle-invasive bladder cancer. Jpn J Clin Oncol. 2011;41:908–14. doi: 10.1093/jjco/hyr068. [DOI] [PubMed] [Google Scholar]
- 184.Pal SK, Ruel NH, Wilson TG, et al. Retrospective analysis of clinical outcomes with neoadjuvant cisplatin-based regimens for muscle-invasive bladder cancer. Clin Genitourin Cancer. 2012;10:246–50. doi: 10.1016/j.clgc.2012.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Stockle M, Meyenburg W, Wellek S, et al. Adjuvant polychemotherapy of nonorgan-confined bladder cancer after radical cystectomy revisited: long-term results of a controlled prospective study and further clinical experience. J Urol. 1995;153:47–52. doi: 10.1097/00005392-199501000-00019. [DOI] [PubMed] [Google Scholar]
- 186.Freiha F, Reese J, Torti FM. A randomized trial of radical cystectomy versus radical cystectomy plus cisplatin, vinblastine and methotrexate chemotherapy for muscle invasive bladder cancer. J Urol. 1996;155:495–9. discussion 9-500. [PubMed] [Google Scholar]
- 187.Paz-Ares LG, Solsona E, Esteban E, et al. Randomized phase III trial comparing adjuvant paclitaxel/gemcitabine/cisplatin (PGC) to observation in patients with resected invasive bladder cancer: Results of the Spanish Oncology Genitourinary Group (SOGUG) 99/01 study. ASCO Meeting Abstracts. 2010;28:LBA4518. [Google Scholar]
- 188.Lehmann J, Kuehn M, Fischer C, et al. Randomized phase III study of adjuvant versus progression-triggered treatment with gemcitabine (G) after radical cystectomy (RC) for locally advanced bladder cancer (LABC) in patients not suitable for cisplatin-based chemotherapy (CBC) (AUO-trial AB22/00) ASCO Meeting Abstracts. 2013;31:250. [Google Scholar]
- 189.Kaufman DS, Shipley WU, Griffin PP, et al. Selective bladder preservation by combination treatment of invasive bladder cancer. N Engl J Med. 1993;329:1377–82. doi: 10.1056/NEJM199311043291903. [DOI] [PubMed] [Google Scholar]
- 190.Tester W, Caplan R, Heaney J, et al. Neoadjuvant combined modality program with selective organ preservation for invasive bladder cancer: results of Radiation Therapy Oncology Group phase II trial 8802. J Clin Oncol. 1996;14:119–26. doi: 10.1200/JCO.1996.14.1.119. [DOI] [PubMed] [Google Scholar]
- 191.Kachnic LA, Kaufman DS, Heney NM, et al. Bladder preservation by combined modality therapy for invasive bladder cancer. J Clin Oncol. 1997;15:1022–9. doi: 10.1200/JCO.1997.15.3.1022. [DOI] [PubMed] [Google Scholar]
- 192.Fellin G, Graffer U, Bolner A, et al. Combined chemotherapy and radiation with selective organ preservation for muscle-invasive bladder carcinoma. A single-institution phase II study. Br J Urol. 1997;80:44–9. doi: 10.1046/j.1464-410x.1997.00221.x. [DOI] [PubMed] [Google Scholar]
- 193.Rodel C, Grabenbauer GG, Kuhn R, et al. Organ preservation in patients with invasive bladder cancer: initial results of an intensified protocol of transurethral surgery and radiation therapy plus concurrent cisplatin and 5-fluorouracil. Int J Radiat Oncol Biol Phys. 2002;52:1303–9. doi: 10.1016/s0360-3016(01)02771-7. [DOI] [PubMed] [Google Scholar]
- 194.Hagan MP, Winter KA, Kaufman DS, et al. RTOG 97-06: initial report of a phase I-II trial of selective bladder conservation using TURBT, twice-daily accelerated irradiation sensitized with cisplatin, and adjuvant MCV combination chemotherapy. Int J Radiat Oncol Biol Phys. 2003;57:665–72. doi: 10.1016/s0360-3016(03)00718-1. [DOI] [PubMed] [Google Scholar]
- 195.Zapatero A, Martin de Vidales C, Arellano R, et al. Updated results of bladder-sparing trimodality approach for invasive bladder cancer. Urol Oncol. 2010;28:368–74. doi: 10.1016/j.urolonc.2009.01.031. [DOI] [PubMed] [Google Scholar]
- 196.Choudhury A, Swindell R, Logue JP, et al. Phase II study of conformal hypofractionated radiotherapy with concurrent gemcitabine in muscle-invasive bladder cancer. J Clin Oncol. 2011;29:733–8. doi: 10.1200/JCO.2010.31.5721. [DOI] [PubMed] [Google Scholar]
- 197.James ND, Hussain SA, Hall E, et al. Results of a phase III randomized trial of synchronous chemoradiotherapy (CRT) compared to radiotherapy (RT) alone in muscle-invasive bladder cancer (MIBC) (BC2001 CRUK/01/004) ASCO Meeting Abstracts. 2010;28:4517. [Google Scholar]


