Abstract
Background
The effect of breast cancer subtype on margin status after lumpectomy remains unclear. This study aims to determine whether approximated breast cancer subtype is associated with positive margins after lumpectomy, which could be used to determine if there is an increased risk of developing local recurrence (LR) following breast-conserving surgery.
Methods
We studied 1,032 consecutive patients with invasive cancer who received lumpectomies and cavity margin (CM) assessments from January 2003 to November 2012. The following data were collected: patient age, cT stage, pT stage, grade, status of CM, lymph node status, menopausal status, ER, PR, HER-2, and Ki67, as well as the presence of extensive intraductal component (EIC) and lymphovascular invasion (LVI). A χ2 test was used to compare categorical baseline characteristics. Univariate and multivariate logistic regression analyses were performed to evaluate associations between pathologic features of CM status. Kaplan-Meier actuarial cumulative rates of LR (ipsilateral in-breast) were calculated.
Results
A total of 7,884 pieces of marginal tissue were collected from 1,032 patients, and 209 patients had positive CMs. Of the patients tested, 52.3% had luminal A subtype, 14.9% were luminal B, 12.8% were luminal-HER-2, 8.1% were HER-2 enriched, and 11.8% were triple negative. Univariate analysis showed that EIC (P <0.001), LVI (P = 0.026), pN stage (N1 vs. N0: P = 0.018; N3 vs. N0: P <0.001), and luminal B (P = 0.001) and HER-2 (P <0.001) subtypes were associated with positive CMs. Multivariable analysis indicated that only EIC (P <0.001), pN stage (P = 0.003), and HER-2 subtype (P <0.001) were significantly correlated with positive CMs. On multivariable analysis, HER-2 subtype was an independent prognostic factor in LR (P = 0.031).
Conclusions
The HER-2 subtype was the predictive factor most associated with positive CMs and an independent prognostic factor for LR. This result suggests that the increased risk of LR in HER-2 breast cancer is due to an increased microscopic invasive tumor burden, which is indicated by margin status after lumpectomy.
Electronic supplementary material
The online version of this article (doi:10.1186/1477-7819-12-289) contains supplementary material, which is available to authorized users.
Keywords: Breast cancer subtype, Breast-conserving surgery, Cavity margin, HER-2
Background
DNA microarray profiles have been used to classify breast tumors into distinct biologic subtypes [1, 2]. This testing may not often be feasible in a clinical setting, and these subtypes can be approximated by the expression of immunohistochemically-defined biological markers, such as the estrogen receptor (ER), the progesterone receptor (PR), and the human epidermal growth factor receptor 2 (HER-2), to classify tumors as luminal A (ER+ or PR+ and HER-2−), luminal B (ER+ or PR+ and HER-2+), HER-2+ (ER− and PR− and HER-2+), or triple-negative (TN) (ER− and PR− and HER-2−) subtypes [3]. Most reports show that the luminal A subtype is associated with the best prognosis, whereas significantly worse prognoses have been observed for the HER-2 and TN subgroups [4–6].
Randomized trials have validated breast-conserving surgery (BCS) as the standard treatment for early stage breast cancer (BC) [7, 8]. Minimizing local recurrence (LR) in the breast is very important in clinical settings because LR is associated with reduced survival and emotional distress [9]. The status of the surgical margin has been shown to be an independent predictor of LR [10, 11]. In a review including 34 related studies, LR was increased in cases involving a persistent, positive margin [12]. Negative margins should be achieved during BCS as recommended by the National Comprehensive Cancer Network guidelines.
Many studies have demonstrated that patients with HER-2 overexpression and TN BCs are at increased risk of developing LR following BCS [6, 13, 14]. Does the higher risk of LR in the two subtypes result from an increased microscopic invasive tumor burden that could be indicated by margin status after lumpectomy? We have no definite answer at present. Positive margins were reported to be significantly associated with large tumor size, young age, positive nodes, presence of lymphovascular invasion (LVI), and presence of an extensive intraductal component (EIC) [15–17]. In addition to the above analyzed clinical and pathologic variables, molecular phenotype may be a relevant factor of positive margins. The purpose of this study was to determine whether BC subtype approximation is associated with positive margins after initial lumpectomy and the extent of initial surgery that should be considered according to molecular subtypes.
Methods
Patient selection
We retrospectively reviewed the clinical and histopathologic data of 1,032 consecutive women ranging from 22 to 89 years (mean 48.2 years, median 47 years) at the time of diagnosis with clinical stage I or II invasive BC deemed suitable for BCS treated with lumpectomy and cavity margin (CM) excision between January 2003 and November 2012 in our center. The following data were included in the analysis: patient age, clinical T stage, pathological T stage, tumor grade, CM status, lymph nodes status, menopausal status, ER, PR, HER-2, and Ki67, as well as the presence of EIC and LVI (Table 1). Patients treated with prior neoadjuvant chemotherapy, endocrine therapy or radiation therapy to the breast or chest wall were excluded from the analysis, and patients with synchronous bilateral BC, more than one malignancy in one breast, or the presence of a non-palpable tumor were also excluded. We obtained approval for this study from the Institutional Review Board at Sun Yat-sen Memorial Hospital.
Table 1.
Patient baseline characteristics (n = 1,032)
| Characteristics | n | % |
|---|---|---|
| Age (y) | ||
| ≤35 | 110 | 10.7 |
| 36–50 | 527 | 51.1 |
| >50 | 395 | 38.3 |
| cT stage | ||
| T1 | 575 | 55.7 |
| T2 | 398 | 38.5 |
| Tx | 59 | 5.7 |
| pT stage | ||
| T1 | 658 | 63.8 |
| T2 | 271 | 26.2 |
| Tx | 103 | 10.0 |
| pN stage | ||
| N0 | 736 | 71.3 |
| N1 | 209 | 20.3 |
| N2 | 50 | 4.8 |
| N3 | 30 | 3.9 |
| Unknown | 7 | 0.7 |
| Grade | ||
| G1 | 83 | 8.1 |
| G2 | 590 | 57.4 |
| G3 | 354 | 34.5 |
| Menopausal status | ||
| Pre- | 652 | 63.2 |
| Post- | 379 | 36.8 |
| Margin status | ||
| Negative | 823 | 79.7 |
| Carcinoma in situ | 87 | 8.4 |
| Invasive carcinoma | 122 | 11.8 |
| Presence of LVI | 114 | 11.9 |
| Presence of EIC | 102 | 9.9 |
| ER or PR positive | 826 | 80.0 |
| HER-2 positive | 216 | 12.0 |
| Histological subtype | ||
| Invasive ductal carcinoma | 805 | 78.0 |
| Presence of DCIS component | 83 | 8.0 |
| Invasive lobular carcinoma | 29 | 2.8 |
| Other | 115 | 11.2 |
| Breast cancer subtype | ||
| Luminal A | 540 | 52.3 |
| Luminal B | 154 | 14.9 |
| Luminal-HER-2 | 132 | 12.8 |
| HER-2 positive | 84 | 8.1 |
| TN | 122 | 11.8 |
EIC, Extensive intraductal component; LVI, Lymphovascular invasion; ER, Estrogen receptor; PR, Progesterone receptor; HER-2, Human epidermal growth factor receptor 2; DCIS, Ductal carcinoma in situ; IDC, Invasive ductal carcinoma; TN, Triple-negative.
Surgical and pathological considerations
There are two primary approaches that are currently used for surgical margin assessment after initial lumpectomy: lumpectomy margin [18, 19] (LM, excision of the specimen containing the tumor) and CM (breast tissue sampled from resection bed cavity) [20, 21]. CMs are increasingly used for a comprehensive assessment of margin status, avoiding an unnecessary second operation in nearly half of patients to achieve negative margins and with the belief that these represent true margins and supersede LMs [20–23]. In our center, CM excision was employed as a routine part of lumpectomies. Physical examination, ultrasound, mammogram, and occasionally magnetic resonance imaging were used for BC diagnosis. For the lumpectomy, 1 cm of macroscopically normal tissue was removed to ensure that the margins of the removed specimens would be tumor free. The superficial and deep margins of the excision extended up to the skin and to the pectoralis fascia. Following excision of the main tumor, seven to nine rectangular CMs [length × width × thickness: (5–10 mm) × (5–10 mm) × (5 mm)] within the perimeter of the lumpectomy resection cavity were excised in a clockwise direction at the time of the lumpectomy, as reported previously [23]. After the resection of each CM, a silk suture was stitched to mark its location within the lumpectomy cavity, and CMs were submitted separately for histopathologic analysis. CMs were defined as positive when tumor cells were observed, regardless of whether they were carcinoma in situ or microscopic invasive carcinoma and independent of their distance from the true margin. Patients were recommended for further surgery for re-excision or mastectomy when one or more positive CMs were found. This procedure was well described in our prior study [24].
Classification of subtypes
Breast cancers expressing high levels of Ki67 have been found to be associated with worse outcomes [25, 26]. In 2009, Cheang et al. [27] determined that the optimal cutoff point for the Ki67 labeling index was 13.25% for distinguishing luminal B from luminal A subtype. Based on this classification, the 12th St. Gallen International Breast Cancer Conference (2011) Expert Panel adopted a new immunohistochemical (IHC) classification of intrinsic subtypes, following application of the Ki67 labeling index using 14% as the cutoff value [28]. Therefore, biological cancer subtypes are approximated as follows: luminal A (ER+ or PR+ and HER-2−, Ki67 < 14%), luminal B (ER+ or PR+ and HER-2−, Ki67 ≥ 14%), luminal-HER-2 (ER+ or PR+ and HER-2+), HER-2 (ER− and PR− and HER-2+), and triple-negative (TN, ER− and PR− and HER-2−). This new classification was employed in our study. ER and PR statuses were determined using IHC staining. Positive ER or PR status was defined as ≥10% of tumor cell nuclei showing specific staining. An intensity of 0 to 1+ was considered HER-2 negative. Tumors were considered HER-2 positive if they were scored as 3+ by IHC or as 2+ by IHC and confirmed by fluorescence in situ hybridization amplification [29]. Grading of tumors was based on the modified Black’s nuclear grading system [30].
Statistical analysis
A χ2 test was used to compare baseline characteristics among categorical variables. Margin status were analyzed by using univariate and multivariate logistic regression models. Associations with LR (ipsilateral in-breast) after BCS were evaluated using univariate and multivariate Cox proportional hazards regression models and summarized with hazard ratios and 95% confidence intervals (CIs). Kaplan-Meier actuarial cumulative rates of LR were calculated. All statistical tests were two-sided and considered statistically significant at 0.05. We performed all data analysis using SPSS 19.0 for Windows.
Results
Baseline characteristics stratified according to breast cancer (BC) subtype
There were significant differences between the five BC subtypes in the distribution of age (P = 0.030), histological subtype (P <0.001), cT stage (P = 0.034), pT stage (P = 0.006), grade (P <0.001), and EIC (P <0.001; Additional file 1: Table S1). In the study, we found that compared to the other BC subtypes, the TN subtype was most commonly observed at an age of 36 to 50 years, and the HER-2 subtype was commonly observed at an age of >50 years and frequently exhibited EIC, larger tumor size, and positive margins.
Baseline characteristics stratified according to age quartile
There were significant differences among the three age quartiles in the distribution of BC subtype (P = 0.030), LVI (P = 0.006), and pT stage (P = 0.002; Additional file 2: Table S2). Compared to older patients, younger women more frequently had BC exhibiting LVI. To our surprise, in our study, older women more frequently had BC with larger tumors.
Rate of positive cavity margins (CMs) by age quartile and breast cancer (BC) subtype
Table 2 provides an analysis of positive CMs by age quartile and BC subtype. We did not see any differences in positive CMs by BC subtype in the age group ≤35 years (P = 0.204). In contrast, there were significant differences in positive CMs between the two older age quartiles, middle age (P <0.001) and >50 years (P = 0.001). In the middle age quartile, 40.6% and 42.9% of patients with luminal B and HER-2 subtypes, respectively, had positive CMs, which was higher than in older age quartile. In the >50 years age group, only patients with HER-2 subtypes had higher CM positivity.
Table 2.
Rate of positive CMs by age quartile and BC subtype
| Age (years) | Luminal A n = 540 (%) | Luminal B n = 154 (%) | Luminal-HER-2 n = 132 (%) | HER-2 n = 83 (%) | TN n = 122 (%) | Pvalue |
|---|---|---|---|---|---|---|
| ≤35 (n = 110) | 18.0 | 20.0 | 0.0 | 22.2 | 0.0 | 0.204 |
| 36–50 (n = 527) | 17.8 | 40.6 | 24.7 | 42.9 | 12.2 | <0.001 |
| >50 (n = 395) | 14.5 | 18.3 | 23.4 | 42.5 | 11.8 | 0.001 |
Positive CMs include ductal carcinoma in situ and invasive carcinoma.
Univariate and multivariate analysis: clinicopathological features associated with positive margins
By univariate analysis, age, cT stage, menopausal status, and tumor grade were not statistically significantly correlated with positive CMs. However, the presence of EIC (OR = 2.77, 95% CI: 1.80–4.27, P <0.001), LVI (OR = 1.64, 95% CI: 1.06–2.55, P = 0.026), pN stage (N1 vs. N0: OR = 1.56, 95% CI: 1.08–2.25, P = 0.018; N3 vs. N0: OR = 6.15, 95% CI: 2.92–12.98, P <0.001), histological subtype (presence of ductal carcinoma in situ (DCIS) component vs. IDC: OR = 2.42, 95% CI: 1.50–3.90, P <0.001) and BC subtype (luminal B vs. luminal A: OR = 2.03, 95% CI: 1.34–3.08, P = 0.001; HER-2 vs. luminal A: OR = 3.45, 95% CI: 2.11–5.63, P <0.001, Table 3) had a statistically significant correlation with positive CMs.
Table 3.
Univariate analysis: clinicopathological features correlated with positive cavity margins
| Variable | n | OR (95% CI) | Pvalue |
|---|---|---|---|
| BC subtype | |||
| Luminal B vs. luminal A | 154 | 2.03 (1.34–3.08) | 0.001 |
| Luminal-HER-2 vs. luminal A | 132 | 1.43 (0.89–2.28) | 0.139 |
| HER-2 vs. luminal A | 84 | 3.45 (2.11–5.63) | <0.001 |
| Triple-negative vs. luminal A | 122 | 0.60 (0.33–1.12) | 0.110 |
| Grade | |||
| G2 vs. G1 | 590 | 0.98 (0.56–1.73) | 0.947 |
| G3 vs. G1 | 354 | 0.99 (0.55–1.79) | 0.977 |
| Histological subtype | |||
| Presence of DCIS vs. IDC | 83 | 2.42 (1.50–3.90) | <0.001 |
| ILC vs. IDC | 29 | 1.83 (0.82–4.09) | 0.142 |
| Others vs. IDC | 115 | 0.39 (0.20–0.76) | 0.006 |
| Menopausal status | |||
| Post- vs. pre- | 379 | 0.98 (0.71–1.34) | 0.894 |
| cT stage | |||
| T2/3 vs. T1 | 398 | 0.97 (0.71–1.34) | 0.873 |
| EIC | 102 | 2.77 (1.80–4.27) | <0.001 |
| LVI | 114 | 1.64 (1.06–2.55) | 0.026 |
| Age | |||
| ~50 vs. ≤35 | 527 | 1.71 (0.97–3.02) | 0.063 |
| >50 vs. ≤35 | 395 | 1.35 (0.75–2.44) | 0.311 |
| pN stage | |||
| N1 vs. N0 | 209 | 1.56 (1.08–2.25) | 0.018 |
| N2 vs. N0 | 50 | 1.03 (0.49–2.18) | 0.932 |
| N3 vs. N0 | 30 | 6.15 (2.92–12.98) | <0.001 |
| pT stage | |||
| T2 vs. T1 | 271 | 1.17 (0.82–1.66) | 0.382 |
Positive CMs include DCIS and invasive carcinoma.
EIC, Extensive intraductal component; LVI, Lymphovascular invasion; ER, Estrogen receptor; PR, Progesterone receptor; HER-2, Human epidermal growth factor receptor 2; DCIS, Ductal carcinoma in situ; IDC, Invasive ductal carcinoma; ILC, Invasive lobular carcinoma; TN, Triple-negative.
Only significant variables in the univariate analysis were applied to the multivariate analysis with logistic regression model. By multivariate analysis, EIC (OR = 2.58, 95% CI: 1.54–4.32, P <0.001), pN stage (N3 vs. N0: OR = 3.92, 95% CI: 1.60–9.62, P = 0.003), and the HER-2 BC subtype (HER-2 vs. luminal A: OR = 2.60, 95% CI: 1.48–4.57, P <0.001; Table 4) were significantly correlated with positive CMs.
Table 4.
Multivariate analysis: clinicopathological features correlated with positive cavity margins
| Variable | OR | 95% CI | Pvalue |
|---|---|---|---|
| A. Positive CMs including DCIS and invasive carcinoma | |||
| EIC | 2.58 | 1.54–4.32 | <0.001 |
| Luminal B vs. luminal A | 1.22 | 0.73–2.04 | 0.456 |
| Luminal-HER-2 vs. luminal A | 1.08 | 0.63–1.86 | 0.774 |
| HER-2 vs. luminal A | 2.60 | 1.48–4.57 | <0.001 |
| Triple-negative vs. luminal A | 0.71 | 0.37–1.39 | 0.320 |
| N1 vs. N0 | 1.43 | 0.92–2.20 | 0.109 |
| N2 vs. N0 | 1.17 | 0.51–2.70 | 0.706 |
| N3 vs. N0 | 3.92 | 1.60–9.62 | 0.003 |
| B. Positive CM including DCIS only | |||
| EIC | 4.50 | 2.59–7.84 | <0.001 |
| Luminal B vs. luminal A | 2.42 | 1.26–4.67 | 0.008 |
| Luminal-HER-2 vs. luminal A | 2.06 | 1.07–3.99 | 0.032 |
| HER-2 vs. luminal A | 5.58 | 2.89–10.77 | <0.001 |
| Triple-negative vs. luminal A | 0.64 | 0.22–1.87 | 0.414 |
| C. Positive CM including invasive cancer only | |||
| Presence of DCIS component vs. IDC | 2.19 | 1.04–4.63 | 0.040 |
| ILC vs. IDC | 2.82 | 1.10–7.23 | 0.031 |
| Other vs. IDC | 0.75 | 0.33–1.70 | 0.488 |
| pN stage | |||
| N1 vs. N0 | 2.15 | 1.30–3.53 | 0.003 |
| N2 vs. N0 | 1.41 | 0.53–3.77 | 0.490 |
| N3 vs. N0 | 8.42 | 3.54–20.05 | <0.001 |
DCIS, Ductal carcinoma in situ; IDC, Invasive ductal carcinoma; EIC, Extensive intraductal component; ILC, Invasive lobular carcinoma.
LR based on breast cancer (BC) subtype
After a median follow-up of 63 months, a total of 831 patients who underwent successful conservative surgery with available follow-up records were reviewed for survival analysis. There were 32 LRs (ipsilateral in breast). The 5-year cumulative incidence of LR for all patients was 6.1% (95% CI: 3.3–8.9%). For patients in the luminal A subgroup, the 5-year cumulative incidence of LR was 3.9% (95% CI: 0.3–7.5%), compared with 4.4% (95% CI: 0–10.5%) for luminal B, 4.7% (95% CI: 0–10%) for luminal-HER-2, 13.4% (95% CI: 0–27.0%) for HER-2, and 8.8% (95% CI: 1.5–16.1%) for TN patients, respectively (Figure 1).
Figure 1.
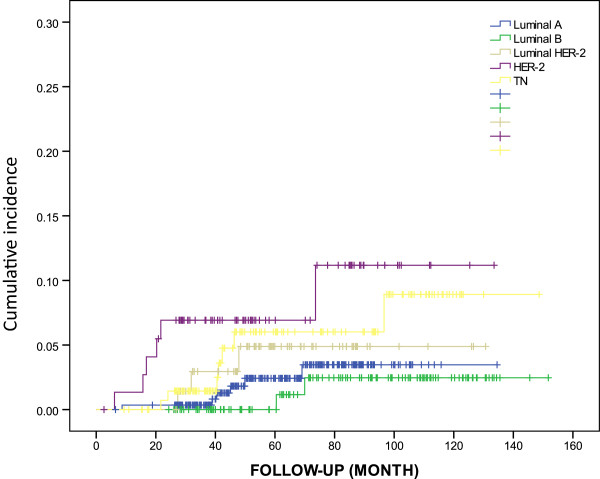
Cumulative incidence of local recurrence (ipsilateral in breast) by breast cancer subtype.
On univariable analysis, young age (36–50 vs. ≤35, P = 0.042), tumor size (T2 vs. T1, P = 0.014), positive nodes (LN+ vs. LN−, P = 0.032) and HER-2 subtype (HER-2 vs. luminal A, P = 0.016) were independently associated with increased risk of LR (Table 5). A multivariate Cox model revealed independent prognostic roles for tumor size (T2 vs. T1, P = 0.001), node status (LN+ vs. LN−, P = 0.044), and HER-2 subtype (HER-2 vs. luminal A, P = 0.031) in LR (Table 6).
Table 5.
Univariate survival analysis for LR (ipsilateral in breast)
| Variable | LR | |
|---|---|---|
| HR (95% CI) | P | |
| ~50 vs. ≤35 | 0.33 (0.12–0.96) | 0.042 |
| ≥50 vs. ≤35 | 0.78 (0.28–2.08) | 0.589 |
| T2 vs. T1 | 1.22 (1.04–1.44) | 0.014 |
| LN+ vs. LN− | 2.41 (1.08–5.38) | 0.032 |
| G3 vs. G1 | 1.90 (0.95–3.80) | 0.069 |
| Luminal B vs. luminal A | 0.59 (0.12–2.94) | 0.517 |
| Luminal-HER-2 vs. luminal A | 1.86 (0.46–7.43) | 0.381 |
| HER-2 vs. luminal A | 4.04 (1.30–12.54) | 0.016 |
| TN vs. luminal A | 2.30 (0.77–6.87) | 0.137 |
Table 6.
Multivariate survival analysis of LR (ipsilateral in breast)
| Variable | LR | |
|---|---|---|
| HR (95% CI) | P | |
| ~50 vs. ≤35 | 0.384 (0.13–1.15) | 0.086 |
| ≥50 vs. ≤35 | 1.031 (0.36–2.96) | 0.955 |
| T2 vs.T1 | 1.392 (1.15–1.69) | 0.001 |
| LN+ vs. LN− | 2.348 (1.02–5.39) | 0.044 |
| Luminal B vs. luminal A | 0.368 (0.07–1.99) | 0.245 |
| Luminal-HER-2 vs. luminal A | 1.662 (0.40–6.84) | 0.482 |
| HER-2 vs. luminal A | 3.650 (1.13–11.80) | 0.031 |
| TN vs. luminal A | 2.025 (0.64–6.41) | 0.230 |
Discussion
In this study, we determined whether BC subtype, as approximated by ER, PR, HER-2, and Ki67, was associated with positive CMs of 1,032 consecutive women who underwent lumpectomies for early stage invasive BC. Compared to all other subtypes, the HER-2 positive subtype was an independent predictor of positive CMs (OR = 2.60, P <0.001) and an independent prognostic factor for LR (P = 0.016).
Many reports have shown that patients with the HER-2 subtype have an increased risk for LR after BCS and radiotherapy (RT) [6, 14]. In our study we found that HER-2 positive patients had a significantly higher recurrence risk, which is consistent with above studies. Randomized trials have demonstrated that the addition of trastuzumab to chemotherapy decreases LR by approximately 50% compared to treatment with chemotherapy alone [29]. The mechanisms underlying the high rate of LR in patients with the HER-2 subtype have not been conclusively determined. In a study, HER-2 status was reported to be the only primary tumor characteristic that correlated with the presence of circulating tumor cells [31]. Some groups have found that circulating tumor cells in operable BC patients are associated with worse prognosis [32]. In another study, patients with the HER-2 subtype were found to be more likely to have multicentric disease [33]. It was also reported that patients with the HER-2 subtype may be relatively resistant to post-lumpectomy RT [34, 35]. The above studies may partly explain the high rate of LR. Our finding that the HER-2 subtype was associated with an increase in positive CMs may lead to interpreting HER-2 BC with multicentric disease, which would result in increased residual microscopic tumors and higher LR to some extent. The follow-up results in our study showed that the HER-2 positive cancer had the highest LR, and maybe it is a reasonable verification of the above theory.
In a previous study, luminal BCs were reported to have a better prognosis [4, 5]. Interestingly, increased LR with the luminal B subtype among young women after BCS has been reported [35, 36]. In the current study, 40.6% of patients between the ages of 36 to 50 years with the luminal B subtype had positive CMs, which was higher than in the >50 years subgroup. Using univariate analysis with the luminal A subtype as the baseline, the luminal B subtype was associated with an increased rate of positive CMs, with an odds ratio of 2.03 (95% CI: 1.34–3.08, P = 0.001). This finding may partially explain the increased LR with the luminal B subtype. The mechanisms are still not well understood and need further study.
In our study, the TN subtype had a low rate of positive CMs, and the result did not seem to coincide with the higher LR of the TN subtype reported by most papers [4, 37, 38]. We observed the clinicopathologic features of the TN subgroup in the present study and found that this low rate of positive CMs may be related to the fact that most TN patients had T1 stage tumors (60.7%), less presence of LVI (TN vs. HER-2: 10.7% vs. 14.3%), and EIC (TN vs. HER-2: 4.1% vs. 22.6%). TN patients with large tumors may have received immediate mastectomies or neoadjuvant chemotherapy and would have thus been excluded from our study. This finding may reflect selection bias, but we have performed multivariate analyses to adjust for the confounding factors.
Our univariate analysis showed that the BC subtype, the presence of EIC or LVI, histopathology subtype, and pN stage were significantly associated with positive CMs. This result was not completely consistent with a previous study [20]. Cao et al. [20] reported that younger patient age, higher number of positive LMs, higher tumor grade, and the presence of EIC were predictive of residual carcinoma in CM specimens. In our study, age and high tumor grade were not predictive factors of positive CMs. Several previous studies have also demonstrated that the presence of EIC [17, 20] and larger tumor size [22, 39] were predictive factors for positive CMs. So far, we have only found one paper, reported by Sioshansi et al. [39], that was specifically looking for associations of different BC subtypes with the risk of residual tumors. Sioshansi et al. [39] showed that age (P = 0.003), tumor size (P <0.001), LVI (P = 0.007), nodal status (P <0.001), and TN subtype (P = 0.006) were associated with an elevated risk of residual invasive cancer by univariate analysis [39]. In our univariate analysis, EIC (P <0.001) was also an important predictive component of positive CMs, and this was not shown in the previous study. Using multivariable analysis, only nodal status (OR = 3.06, 95% CI: 1.77–5.30, P <0.001), TN status (TN vs. non-TN, OR = 3.28, 95% CI: 1.56–6.89, P = 0.02), and tumor size (tumor size >2.0 cm vs. <1.0 cm, OR = 3.49, 95% CI: 1.65–7.38, P = 0.001) maintained statistical significance on multivariate analysis [39]. However, tumor size was not a significant predictive factor associated with positive CMs in our multivariate analysis. EIC (OR = 2.58, 95% CI: 1.53–4.32, P <0.001), pN stage (N3 vs. N0: OR = 3.92, 95% CI: 1.60–9.62, P = 0.003), and HER-2 subtype (HER-2 vs. luminal A: OR = 2.60, 95% CI: 1.48–4.57, P <0.001) were significantly correlated with positive CMs. The difference between associated BC subtypes may be due to the following: i) Classification by different immunohistochemical markers. In previous studies, approximated molecular phenotypes were defined by ER, PR, and HER-2, which was different from our new classification. On the basis of recent data suggested by the 12th St. Gallen International Breast Cancer Conference (2011) Expert Panel, the Ki67 index was used in our study, which additionally discriminated partial luminal B patients from luminal A patients. The use of the Ki67 index is unique to this study. ii) Distribution of BC subtypes. Sioshansi et al. [39] reported that 73.5% of patients in their study were luminal A, 9.5% were luminal B, 4.5% were HER-2 enriched, and 12.5% were TN. In our study, 52.3% were luminal A, 14.9% were luminal B, 12.8% were luminal-HER-2, 8.0% were HER-2, and 11.8% were TN.
Among different age groups, positive rate of CMs in different molecular subtypes is not clear yet. We analyzed positive rates of CMs (including invasive cancer and carcinoma in situ) by age quartile and BC subtype in the current study. Women aged ≤35 years with BC are reported to have a poor prognosis and for most women, and menopause happens around age 50. According to this, we divided patients into three groups. In the youngest age quartile (≤35 years), the positive CM rate demonstrated no significant difference (P = 0.204). In contrast, the quartile containing ages 36 to 50 years had positive CM rates of 40.6% and 40.9% in luminal B and HER-2 subtypes, respectively (P <0.001), and the quartile with patients older than 50 years had a positive CM rate of 42.5% with the HER-2 subtype, which reached statistical significance (P = 0.001). Thus, younger age (≤35 years) was not a risk factor for positive CMs in our study.
The risk of residual disease, including carcinoma in situ and invasive cancer (residual disease, including carcinoma in situ alone, was excluded from one study [39]), after lumpectomy has been examined in many studies [24, 40]. In recent decades, positive re-excision rates from 17% to 39% have been reported [20, 41–43]. In our current series, 20.3% (209/1,032) of patients had positive CMs, including carcinoma in situ and invasive cancer. This result was similar to those of previously published literature. For a comprehensive assessment, we also evaluated the positive CM rate with carcinoma in situ or invasive cancer alone using multivariate analysis. The positive rates were 9.6% (87/910, CMs with carcinoma in situ alone) and 12.9% (122/945, CMs with invasive cancer alone). For patients with positive CMs, including carcinoma in situ, EIC (P <0.001) and BC subtypes (HER-2 vs. luminal A: P <0.001; luminal B vs. luminal A: P = 0.008; luminal-HER-2 vs. luminal A: P = 0.032, Table 4) showed a significant association with positive CMs. For patients with positive CMs, including invasive cancer alone, histological subtype (presence of DCIS component vs. IDC, P = 0.040; invasive lobular carcinoma vs. IDC, P = 0.031) and pN stage (N1 vs. N0: P = 0.003; N3 vs. N0: P <0.001, Table 4) showed statistical correlation with positive CMs. BC subtype was no longer a relevant factor, which was not consistent with Sioshansi et al. [39].
There are several inherent limitations to this study. i) Although many surgeons increasingly prefer to use CMs for margin assessment, without information from long-term clinical follow-up, it is not clear whether CMs or LMs are superior. We used only the CM method without corresponding LM section analysis. ii) This is a single-center study, and the population was not representative of Chinese or Asian demographics. iii) BC subtypes approximated according to ER, PR, HER-2, and Ki67 are only a substitute for genotype-based molecular BC subtypes. Further studies will be needed to confirm the findings based on these new definitions.
Conclusions
In summary, although there are potential limitations to this study, the findings showed that that the poor prognosis of the HER-2 subtype is due to increased residual microscopic tumor burden after lumpectomy. More clinical trials will be required to confirm our conclusion. This information may help surgeons to choose the most appropriate surgical treatment for each patient. Further study and follow-up data are required to confirm the findings from our study. Oncoplastic breast surgery and an increased “boost” in radiotherapy may be good choices for patients with the HER-2 subtype to reduce the microscopic tumor burden and to improve prognosis and cosmetic results.
Electronic supplementary material
Additional file 1: Table S1.: Patient baseline characteristics stratified by subtype. (DOCX 29 KB)
Additional file 2: Table S2.: Patient baseline characteristics stratified by age quartile. (DOCX 43 KB)
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grants 81172524/H1622, 81172537/H1622, 81272900/H1622, and 81201758/H1610).
Abbreviations
- BC
Breast cancer
- BCS
Breast-conserving surgery
- CI
Confidence intervals
- CM
Cavity margin
- DCIS
Ductal carcinoma in situ
- EIC
Extensive intraductal component
- ER
Estrogen receptor
- HER-2
Human epidermal growth factor receptor 2
- IDC
Invasive ductal carcinoma
- IHC
Immunohistochemistry
- LM
Lumpectomy margin
- LR
Local recurrence
- LVI
Lymphovascular invasion
- PR
Progesterone receptor
- TN
Triple-negative.
Footnotes
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
J-HX, J-WJ, and S-FX designed the research and drafted the manuscript. J-HX, J-WJ, Y-YP, L-SR, and F-HY collected the clinical materials and follow-up. L-JQ, R-NY, J-L, W-JN, G-R, Z-LL, C-K, D-HR, and Z-YJ attended immunohistochemistry in this study. L-Q and S-EW modified the manuscript. All authors read and approved the final manuscript.
Contributor Information
Haixia Jia, Email: xiaohaijia@126.com.
Weijuan Jia, Email: jiaweijuan@gmail.com.
Yaping Yang, Email: yypapple1225@163.com.
Shunrong Li, Email: lisoon2000@163.com.
Huiyi Feng, Email: fjeff@163.com.
Jieqiong Liu, Email: liujieqiong01@gmail.com.
Nanyan Rao, Email: raonany@126.com.
Liang Jin, Email: jinl2001@qq.com.
Jiannan Wu, Email: king8702@163.com.
Ru Gu, Email: ccaibai@qq.com.
Liling Zhu, Email: vivovivo@163.com.
Kai Chen, Email: ckaichen@126.com.
Heran Deng, Email: dengheran0930@sina.com.
Yunjie Zeng, Email: zenyunjie@126.com.
Qiang Liu, Email: victorlq@hotmail.com.
Erwei Song, Email: songerwei02@yahoo.com.cn.
Fengxi Su, Email: fengxisu@vip.163.com.
References
- 1.Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, Zhu SX, Lønning PE, Børresen-Dale AL, Brown PO, Botstein D. Molecular portraits of human breast tumours. Nature. 2000;406:747–752. doi: 10.1038/35021093. [DOI] [PubMed] [Google Scholar]
- 2.Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey SS, Thorsen T, Quist H, Matese JC, Brown PO, Botstein D, Eystein Lønning P, Børresen-Dale AL. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci U S A. 2001;98:10869–10874. doi: 10.1073/pnas.191367098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brenton JD, Carey LA, Ahmed AA, Caldas C. Molecular classification and molecular forecasting of breast cancer: ready for clinical application? J Clin Oncol. 2005;23:7350–7360. doi: 10.1200/JCO.2005.03.3845. [DOI] [PubMed] [Google Scholar]
- 4.Sorlie T, Tibshirani R, Parker J, Hastie T, Marron JS, Nobel A, Deng S, Johnsen H, Pesich R, Geisler S, Demeter J, Perou CM, Lønning PE, Brown PO, Børresen-Dale AL, Botstein D. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci U S A. 2003;100:8418–8423. doi: 10.1073/pnas.0932692100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Carey LA, Perou CM, Livasy CA, Dressler LG, Cowan D, Conway K, Karaca G, Troester MA, Tse CK, Edmiston S, Deming SL, Geradts J, Cheang MC, Nielsen TO, Moorman PG, Earp HS, Millikan RC. Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA. 2006;295:2492–2502. doi: 10.1001/jama.295.21.2492. [DOI] [PubMed] [Google Scholar]
- 6.Nguyen PL, Taghian AG, Katz MS, Niemierko A, Abi Raad RF, Boon WL, Bellon JR, Wong JS, Smith BL, Harris JR. Breast cancer subtype approximated by estrogen receptor, progesterone receptor, and HER-2 is associated with local and distant recurrence after breast-conserving therapy. J Clin Oncol. 2008;26:2373–2378. doi: 10.1200/JCO.2007.14.4287. [DOI] [PubMed] [Google Scholar]
- 7.Fisher B, Anderson S, Bryant J, Margolese RG, Deutsch M, Fisher ER, Jeong JH, Wolmark N. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation for the treatment of invasive breast cancer. N Engl J Med. 2002;347:1233–1241. doi: 10.1056/NEJMoa022152. [DOI] [PubMed] [Google Scholar]
- 8.Veronesi U, Cascinelli N, Mariani L, Greco M, Saccozzi R, Luini A, Aguilar M, Marubini E. Twenty-year follow-up of a randomized study comparing breast-conserving surgery with radical mastectomy for early breast cancer. N Engl J Med. 2002;347:1227–1232. doi: 10.1056/NEJMoa020989. [DOI] [PubMed] [Google Scholar]
- 9.Darby S, McGale P, Correa C, Taylor C, Arriagada R, Clarke M, Cutter D, Davies C, Ewertz M, Godwin J, Gray R, Pierce L, Whelan T, Wang Y, Peto R. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet. 2010;378:1707–1716. doi: 10.1016/S0140-6736(11)61629-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mechera R, Viehl CT, Oertli D. Factors predicting in-breast tumor recurrence after breast-conserving surgery. Breast Cancer Res Treat. 2009;116:171–177. doi: 10.1007/s10549-008-0187-y. [DOI] [PubMed] [Google Scholar]
- 11.Komoike Y, Akiyama F, Iino Y, Ikeda T, Akashi-Tanaka S, Ohsumi S, Kusama M, Sano M, Shin E, Suemasu K, Sonoo H, Taguchi T, Nishi T, Nishimura R, Haga S, Mise K, Kinoshita T, Murakami S, Yoshimoto M, Tsukuma H, Inaji H. Ipsilateral breast tumor recurrence (IBTR) after breast-conserving treatment for early breast cancer: risk factors and impact on distant metastases. Cancer. 2006;106:35–41. doi: 10.1002/cncr.21551. [DOI] [PubMed] [Google Scholar]
- 12.Singletary SE. Surgical margins in patients with early-stage breast cancer treated with breast conservation therapy. Am J Surg. 2002;184:383–393. doi: 10.1016/S0002-9610(02)01012-7. [DOI] [PubMed] [Google Scholar]
- 13.Freedman GM, Anderson PR, Li T, Nicolaou N. Locoregional recurrence of triple-negative breast cancer after breast-conserving surgery and radiation. Cancer. 2009;115:946–951. doi: 10.1002/cncr.24094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim HJ, Han W, Yi OV, Shin HC, Ahn SK, Koh BS, Moon HG, You JH, Son BH, Ahn SH, Noh DY. Young age is associated with ipsilateral breast tumor recurrence after breast conserving surgery and radiation therapy in patients with HER2-positive/ER-negative subtype. Breast Cancer Res Treat. 2011;130:499–505. doi: 10.1007/s10549-011-1736-3. [DOI] [PubMed] [Google Scholar]
- 15.Park CC, Mitsumori M, Nixon A, Recht A, Connolly J, Gelman R, Silver B, Hetelekidis S, Abner A, Harris JR, Schnitt SJ. Outcome at 8 years after breast-conserving surgery and radiation therapy for invasive breast cancer: influence of margin status and systemic therapy on local recurrence. J Clin Oncol. 2000;18:1668–1675. doi: 10.1200/JCO.2000.18.8.1668. [DOI] [PubMed] [Google Scholar]
- 16.Peterson ME, Schultz DJ, Reynolds C, Solin LJ. Outcomes in breast cancer patients relative to margin status after treatment with breast-conserving surgery and radiation therapy: the University of Pennsylvania experience. Int J Radiat Oncol Biol Phys. 1999;43:1029–1035. doi: 10.1016/S0360-3016(98)00519-7. [DOI] [PubMed] [Google Scholar]
- 17.Smitt MC, Nowels K, Carlson RW, Jeffrey SS. Predictors of reexcision findings and recurrence after breast conservation. Int J Radiat Oncol Biol Phys. 2003;57:979–985. doi: 10.1016/S0360-3016(03)00740-5. [DOI] [PubMed] [Google Scholar]
- 18.Fisher ER, Sass R, Fisher B, Gregorio R, Brown R, Wickerham L. Pathologic findings from the National Surgical Adjuvant Breast Project (protocol 6). II. Relation of local breast recurrence to multicentricity. Cancer. 1986;57:1717–1724. doi: 10.1002/1097-0142(19860501)57:9<1717::AID-CNCR2820570902>3.0.CO;2-H. [DOI] [PubMed] [Google Scholar]
- 19.Wright MJ, Park J, Fey JV, Park A, O’Neill A, Tan LK, Borgen PI, Cody HS, 3rd, Van Zee KJ, King TA. Perpendicular inked versus tangential shaved margins in breast-conserving surgery: does the method matter? J Am Coll Surg. 2007;204:541–549. doi: 10.1016/j.jamcollsurg.2007.01.031. [DOI] [PubMed] [Google Scholar]
- 20.Cao D, Lin C, Woo SH, Vang R, Tsangaris TN, Argani P. Separate cavity margin sampling at the time of initial breast lumpectomy significantly reduces the need for reexcisions. Am J Surg Pathol. 2005;29:1625–1632. doi: 10.1097/01.pas.0000180448.08203.70. [DOI] [PubMed] [Google Scholar]
- 21.Huston TL, Pigalarga R, Osborne MP, Tousimis E. The influence of additional surgical margins on the total specimen volume excised and the reoperative rate after breast-conserving surgery. Am J Surg. 2006;192:509–512. doi: 10.1016/j.amjsurg.2006.06.021. [DOI] [PubMed] [Google Scholar]
- 22.Tengher-Barna I, Hequet D, Reboul-Marty J, Frassati-Biaggi A, Seince N, Rodrigues-Faure A, Uzan M, Ziol M. Prevalence and predictive factors for the detection of carcinoma in cavity margin performed at the time of breast lumpectomy. Mod Pathol. 2009;22:299–305. doi: 10.1038/modpathol.2008.186. [DOI] [PubMed] [Google Scholar]
- 23.Povoski SP, Jimenez RE, Wang WP, Xu RX. Standardized and reproducible methodology for the comprehensive and systematic assessment of surgical resection margins during breast-conserving surgery for invasive breast cancer. BMC Cancer. 2009;9:254. doi: 10.1186/1471-2407-9-254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chen K, Zeng Y, Jia H, Jia W, Yang H, Rao N, Song E, Cox CE, Su F. Clinical outcomes of breast-conserving surgery in patients using a modified method for cavity margin assessment. Ann Surg Oncol. 2012;19:3386–3394. doi: 10.1245/s10434-012-2331-5. [DOI] [PubMed] [Google Scholar]
- 25.Trihia H, Murray S, Price K, Gelber RD, Golouh R, Goldhirsch A, Coates AS, Collins J, Castiglione-Gertsch M, Gusterson BA. Ki-67 expression in breast carcinoma: its association with grading systems, clinical parameters, and other prognostic factors–a surrogate marker? Cancer. 2003;97:1321–1331. doi: 10.1002/cncr.11188. [DOI] [PubMed] [Google Scholar]
- 26.De Azambuja E, Cardoso F, De Castro G, Colozza M, Jr, Mano MS, Durbecq V, Sotiriou C, Larsimont D, Piccart-Gebhart MJ, Paesmans M. Ki-67 as prognostic marker in early breast cancer: a meta-analysis of published studies involving 12,155 patients. Br J Cancer. 2007;96:1504–1513. doi: 10.1038/sj.bjc.6603756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cheang MC, Chia SK, Voduc D, Gao D, Leung S, Snider J, Watson M, Davies S, Bernard PS, Parker JS, Perou CM, Ellis MJ, Nielsen TO. Ki67 index, HER2 status, and prognosis of patients with luminal B breast cancer. J Natl Cancer Inst. 2009;101:736–750. doi: 10.1093/jnci/djp082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Goldhirsch A, Wood WC, Coates AS, Gelber RD, Thurlimann B, Senn HJ. Strategies for subtypes–dealing with the diversity of breast cancer: highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann Oncol. 2011;22:1736–1747. doi: 10.1093/annonc/mdr304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE, Jr, Davidson NE, Tan-Chiu E, Martino S, Paik S, Kaufman PA, Swain SM, Pisansky TM, Fehrenbacher L, Kutteh LA, Vogel VG, Visscher DW, Yothers G, Jenkins RB, Brown AM, Dakhil SR, Mamounas EP, Lingle WL, Klein PM, Ingle JN, Wolmark N. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005;353:1673–1684. doi: 10.1056/NEJMoa052122. [DOI] [PubMed] [Google Scholar]
- 30.Black MM, Speer FD. Nuclear structure in cancer tissues. Surg Gynecol Obstet. 1957;105:97–102. [PubMed] [Google Scholar]
- 31.Lang JE, Mosalpuria K, Cristofanilli M, Krishnamurthy S, Reuben J, Singh B, Bedrosian I, Meric-Bernstam F, Lucci A. HER2 status predicts the presence of circulating tumor cells in patients with operable breast cancer. Breast Cancer Res Treat. 2009;113:501–507. doi: 10.1007/s10549-008-9951-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Xenidis N, Perraki M, Kafousi M, Apostolaki S, Bolonaki I, Stathopoulou A, Kalbakis K, Androulakis N, Kouroussis C, Pallis T, Christophylakis C, Argyraki K, Lianidou ES, Stathopoulos S, Georgoulias V, Mavroudis D. Predictive and prognostic value of peripheral blood cytokeratin-19 mRNA-positive cells detected by real-time polymerase chain reaction in node-negative breast cancer patients. J Clin Oncol. 2006;24:3756–3762. doi: 10.1200/JCO.2005.04.5948. [DOI] [PubMed] [Google Scholar]
- 33.Wiechmann L, Sampson M, Stempel M, Jacks LM, Patil SM, King T, Morrow M. Presenting features of breast cancer differ by molecular subtype. Ann Surg Oncol. 2009;16:2705–2710. doi: 10.1245/s10434-009-0606-2. [DOI] [PubMed] [Google Scholar]
- 34.Albert JM, Gonzalez-Angulo AM, Guray M, Sahin A, Strom EA, Tereffe W, Woodward WA, Tucker SL, Hunt KK, Hortobagyi GN, Buchholz TA. Estrogen/progesterone receptor negativity and HER2 positivity predict locoregional recurrence in patients with T1a, bN0 breast cancer. Int J Radiat Oncol Biol Phys. 2010;77:1296–1302. doi: 10.1016/j.ijrobp.2009.12.011. [DOI] [PubMed] [Google Scholar]
- 35.Cancello G, Maisonneuve P, Rotmensz N, Viale G, Mastropasqua MG, Pruneri G, Veronesi P, Torrisi R, Montagna E, Luini A, Intra M, Gentilini O, Ghisini R, Goldhirsch A, Colleoni M. Prognosis and adjuvant treatment effects in selected breast cancer subtypes of very young women (<35 years) with operable breast cancer. Ann Oncol. 2010;21:1974–1981. doi: 10.1093/annonc/mdq072. [DOI] [PubMed] [Google Scholar]
- 36.Arvold ND, Taghian AG, Niemierko A, Abi Raad RF, Sreedhara M, Nguyen PL, Bellon JR, Wong JS, Smith BL, Harris JR. Age, breast cancer subtype approximation, and local recurrence after breast-conserving therapy. J Clin Oncol. 2011;29:3885–3891. doi: 10.1200/JCO.2011.36.1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hattangadi-Gluth JA, Wo JY, Nguyen PL, Abi Raad RF, Sreedhara M, Niemierko A, Freer PE, Georgian-Smith D, Bellon JR, Wong JS, Smith BL, Harris JR, Taghian AG. Basal subtype of invasive breast cancer is associated with a higher risk of true recurrence after conventional breast-conserving therapy. Int J Radiat Oncol Biol Phys. 2012;82:1185–1191. doi: 10.1016/j.ijrobp.2011.02.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lin NU, Vanderplas A, Hughes ME, Theriault RL, Edge SB, Wong YN, Blayney DW, Niland JC, Winer EP, Weeks JC. Clinicopathologic features, patterns of recurrence, and survival among women with triple-negative breast cancer in the National Comprehensive Cancer Network. Cancer. 2012;118:5463–5472. doi: 10.1002/cncr.27581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sioshansi S, Ehdaivand S, Cramer C, Lomme MM, Price LL, Wazer DE. Triple negative breast cancer is associated with an increased risk of residual invasive carcinoma after lumpectomy. Cancer. 2012;118:3893–3898. doi: 10.1002/cncr.27376. [DOI] [PubMed] [Google Scholar]
- 40.Wazer DE, Schmidt-Ullrich RK, Schmid CH, Ruthazer R, Kramer B, Safaii H, Graham R. The value of breast lumpectomy margin assessment as a predictor of residual tumor burden. Int J Radiat Oncol Biol Phys. 1997;38:291–299. doi: 10.1016/S0360-3016(97)82498-4. [DOI] [PubMed] [Google Scholar]
- 41.Beck NE, Bradburn MJ, Vincenti AC, Rainsbury RM. Detection of residual disease following breast-conserving surgery. Br J Surg. 1998;85:1273–1276. doi: 10.1046/j.1365-2168.1998.00876.x. [DOI] [PubMed] [Google Scholar]
- 42.Barthelmes L, Al Awa A, Crawford DJ. Effect of cavity margin shavings to ensure completeness of excision on local recurrence rates following breast conserving surgery. Eur J Surg Oncol. 2003;29:644–648. doi: 10.1016/S0748-7983(03)00122-7. [DOI] [PubMed] [Google Scholar]
- 43.Hewes JC, Imkampe A, Haji A, Bates T. Importance of routine cavity sampling in breast conservation surgery. Br J Surg. 2009;96:47–53. doi: 10.1002/bjs.6435. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1.: Patient baseline characteristics stratified by subtype. (DOCX 29 KB)
Additional file 2: Table S2.: Patient baseline characteristics stratified by age quartile. (DOCX 43 KB)


