Abstract
Purpose
Phase-contrast optical coherence tomography (PC-OCT) provides volumetric imaging of the retinal vasculature without the need for intravenous injection of a fluorophore. Here, we compare images from PC-OCT and fluorescein angiography (FA) for normal individuals and patients with age-related macular degeneration and diabetic retinopathy.
Design
This is an evaluation of a diagnostic technology.
Participants
4 patients underwent comparative retinovascular imaging using FA and PC-OCT. Imaging was performed on 1 normal individual, 1 patient with dry age-related macular degeneration, 1 patient with exudative age-related macular degeneration and 1 patient with non-proliferative diabetic retinopathy.
Methods
FA imaging was performed using a Topcon (TRC-50IX) camera having resolution of 1280 (H) x 1024 (V) pixels. PC-OCT images were generated by software data processing of the entire cross-sectional image from consecutively acquired B-scans. Bulk axial motion was calculated and corrected for each transverse location, reducing the phase noise introduced from eye motion. Phase contrast was calculated through the variance of the motion-corrected phase changes acquired within multiple B-scans at the same position. Repeating these calculations over the entire volumetric scan produced a three-dimensional PC-OCT representation of the vasculature.
Main Outcome Measures
Feasibility of rendering retinal and choroidal microvasculature using PC-OCT was compared qualitatively to FA, the current gold standard for retinovascular imaging.
Results
PC-OCT rendered a two-dimensional depth color-coded vasculature map of the retinal and choroidal vasculature non-invasively. The choriocapillaris was imaged with better resolution of microvascular detail using PC-OCT. Areas of geographic atrophy and choroidal neovascularization imaged by FA were depicted by PC-OCT. Regions of capillary non-perfusion from diabetic retinopathy were shown by both imaging techniques; there was not complete correspondence between microaneurysms shown on FA and PC-OCT images.
Conclusion
PC-OCT yields high resolution imaging of the retinal and choroidal microvasculature that compares favorably to FA.
Introduction
Since the initial studies by Novotny and Alvis over 50 years ago, fluorescein angiography (FA) remains the gold standard for retinovascular imaging.1,2 An estimated 1 million FA studies are performed annually in the United States.3 While of obvious value in revealing fine details of the microvasculature, FA requires an intravenous injection, a skilled photographer, and is time consuming. Minor side effects such as nausea, vomiting, and multiple needle sticks in patients with challenging venous access are not uncommon.4 Because fluorescein leaks readily through the fenestrations of the choriocapillaris, it is not suitable for showing the anatomy of this important vascular layer that supplies the outer retina. Indocyanine green (ICG) angiography provides improved visualization of choroidal anatomy because this dye is more extensively protein bound than fluorescein and does not leak into the extravascular space as readily.5 Furthermore, it fluoresces at a longer wavelength than fluorescein and imaging can take place through pigment and thin layers of blood. Nevertheless, ICG imaging fails to depict the fine anatomic structure of the choriocapillaris.6,7
Following its introduction in 1991, optical coherence tomography (OCT) has become an indispensable clinical imaging tool.8,9 Use of spectral domain OCT (SD-OCT) enables high-resolution imaging of retinal morphology that is nearly comparable to histologic analysis. Despite the rapid evolution of OCT imaging, SD-OCT does not provide adequate visualization of retinal and choroidal microvasculature. Thus, as clinicians, we are often compelled to order both an OCT and FA in patients with common retinovascular diseases such as age-related macular degeneration, diabetic retinopathy, and retinovascular occlusions.
Recently, there has been increased interest in using data generated during SD-OCT imaging to generate angiographic images of the fundus. These angiograms are implemented non-invasively without injection of fluorescent dye. Fingler and co-workers have used phase-contrast optical coherence tomography (PC-OCT), also called phase- variance OCT, to image retinal microvasculature.10–14 This technique uses software processing of data normally acquired, but not utilized, during SD-OCT imaging. Using a different scanning protocol than found in commercial instruments, PC-OCT identifies regions of motion between consecutive B-scans that are contrasted with less mobile regions. In the retina and choroid the regions with motion correspond to the vasculature; these vessels are readily differentiated from other retinal tissues that are relatively static. An alternative method to acquire images of the retinal microvasculature is Doppler OCT, which measures the change in scatter position between successive depth scans and uses this information to calculate the flow component parallel to the imaging direction (called axial flow).15–17 Doppler OCT has been used to image large axial flow in the retina, but this technique without dedicated scanning protocols18 is limited in cases of slow flow or flow-oriented transverse to the imaging direction. Since this technique depends on measuring motion changes between successive depth scans, as imaging speed improvements continue for SDOCT systems, the scatterers have less time to move between measurements and the slowest motions become obscured by noise. This reduces the visualization capabilities of typical Doppler OCT techniques. In contrast, PC-OCT will be able to achieve the same time separations between phase measurements with increased SD-OCT imaging speeds, maintaining the demonstrated ability to visualize fast blood vessel as well as slow microvascular flow, independent of vessel orientation.
Several groups in recent years have developed OCT imaging methods to push beyond conventional Doppler OCT imaging limitations. Some approaches involve increasing the flow contrast though hardware modifications of SD-OCT machines such as in two beam scanning19,20, or producing a heterodyne frequency for extracting flow components 21. Others have utilized non-conventional scanning patterns22 or repeated B-scan acquisitions23,24 such as used in PC-OCT to increase the time separation between phase measurements and enhance Doppler flow contrast of microvascular flow. In addition to phase-based contrast techniques to visualize vasculature, intensity-based visualization of microvasculature has been developed for OCT using segmentation25, speckle-based temporal changes26,27, decorrelation-based techniques28, and contrast based on both phase and intensity changes29. Each of these methods have varying capabilities in regards to microvascular visualization, noise levels and artifacts while imaging retinal tissues undergoing typical motion during acquisition. Some of the noise and artifact limitations can be overcome with selective segmentation of the volumetric data or increased statistics through longer imaging times, but further analysis is required to be able to compare all of the visualization capabilities from all these different systems.
Herein, we wish to show the preliminary capabilities of PC-OCT to image retinal and choroidal microvasculature in normals and patients with age-related macular degeneration (AMD) and diabetic retinopathy.
Methods
The tenets of the Declaration of Helsinki were observed, and written informed consent approval by the institutional review board (IRB) was obtained. For FA imaging, each subject’s pupils were dilated with a combination of 1% tropicamide and 2.5% phenylephrine. Sodium fluorescein 10% in water (500 mg/5 mL) was injected intravenously, followed by a flush of normal saline. Fundus FA images were acquired with a Topcon (TRC-50IX) camera having resolution of 1280 (H) x 1024 (V) pixels.
Two different OCT instruments were used in the acquisition of PC-OCT data presented here. The primary system was a high-speed (125-kHz A-scan rate) SD-OCT system constructed at the University of California, Davis (UCD) that acquired in vivo human retinal images with scanning areas of 1.5 x 1.5 mm2 or 3 x 3 mm2.13 Figures 1, 2, 3, 5, and 6 of this manuscript were acquired with this 125kHz OCT system. Another system operating at 25kHz at the University of California, Davis was used to acquire retinal scanning areas of 1x1 mm2.12 We used this system to acquire Figure 4. A bite-bar and a forehead rest were used to stabilize head position for both systems. The image acquisition time of each PC-OCT volumetric scan was less than 5 seconds for each system.
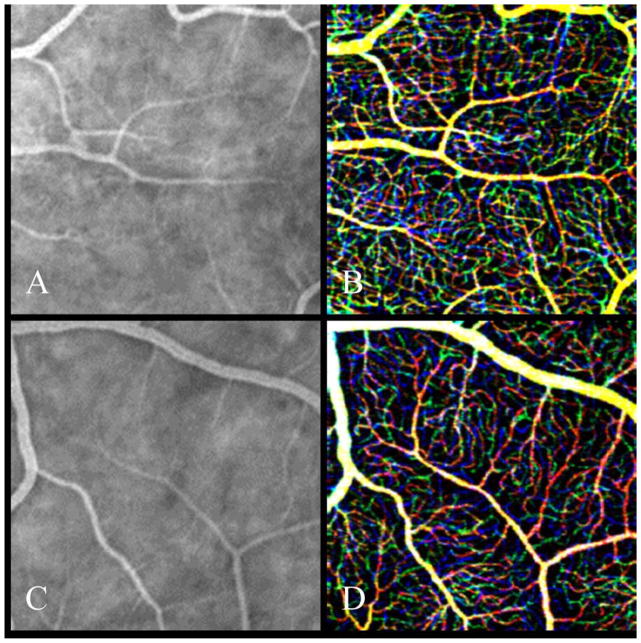
Figure 1.
Comparison between fluorescein angiography (FA) (A,C) and phase-contrast optical coherence tomography (PC-OCT) (B,D) over 1.5x1.5 mm2 areas in parafovea (top: 8° nasal eccentricity, bottom: 8° temporal and superior eccentricity) of a 60-year-old normal subject. PC-OCT is red-green-blue color-coded to represent the depth imaging, which is not captured in FA data sets. Red is the most superficial capillary bed, green is the intermediate capillary plexus, and blue is the deeper capillary network. Larger diameter vessels are yellowish due to positioning in both superficial and intermediate layers.
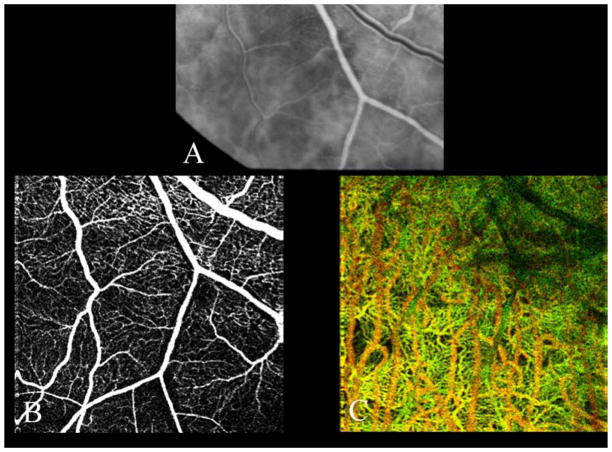
Figure 2.
Comparison of fluorescein angiography (FA) and phase-contrast optical coherence tomography (PC-OCT) imaging of retinal and choroidal vasculature just outside the inferotemporal arcade. (A) FA in laminar phase. En face projection of PC-OCT (B) shows retinal vasculature of the same region. The color-coded image (C) shows choroidal vasculature, superficial choroidal vessels (choriocapillaris, Sattler’s layer) in green and larger choroidal vessels (Haller’s layer) in black. Notice that shadows of retinal vasculature generate artifacts in black in the upper right area in (C).
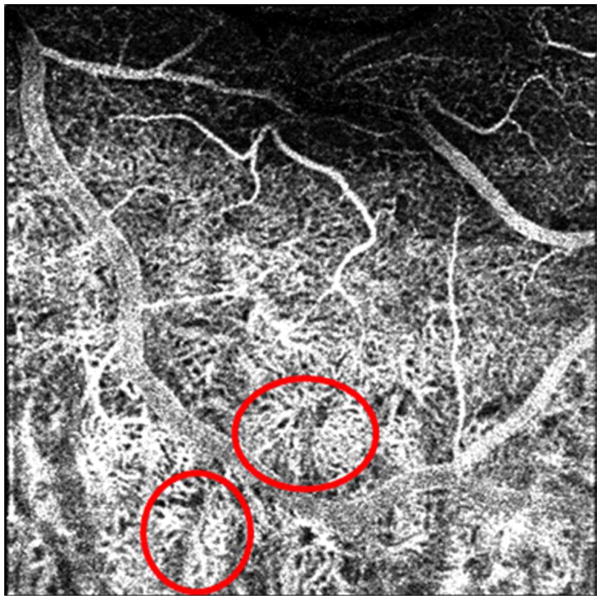
Figure 3.
Phase-contrast optical coherence tomography showing depth imaging of retinal and choroidal vasculature. Putative choriocapillaris lobules are circled in red.
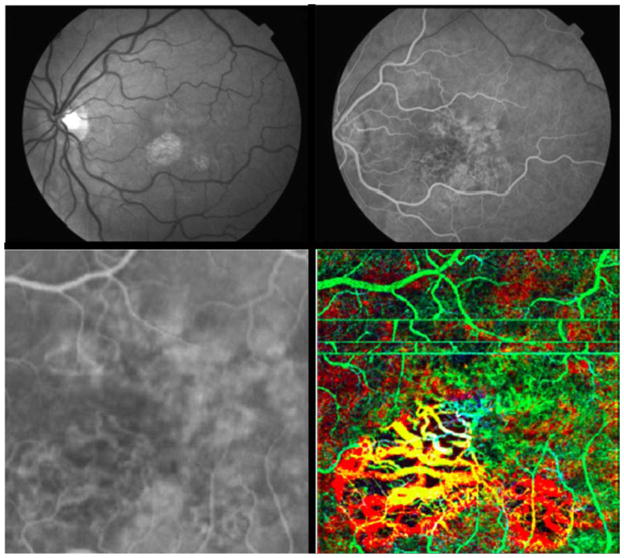
Figure 5.
61-year-old woman with dry age-related macular degeneration (AMD). Top left shows red free photo; top right is laminar flow phase of fluorescein angiography (FA). Lower left is high magnification FA image of foveal region; lower right shows a 3 x 3 mm phase-contrast optical coherence tomography (PC-OCT) depth image of same region, color coded. (Green is superficial/anterior choroidal region, which includes horizontal motion artifacts. Yellow is deeper into choroid, and red is the deepest imaging plane.) All slices are fixed distances relative to the anterior surface of retina.
Figure 6.
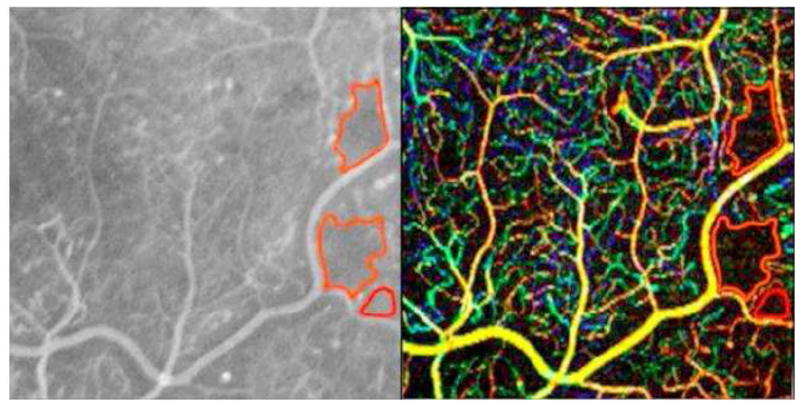
View of temporal retina in a patient with proliferative diabetic retinopathy. Fluorescein angiography (FA) on left shows multiple microaneurysms; regions of capillary non-perfusion outlined in red. 3 x 3 mm phase-contrast optical coherence tomography (PC-OCT) on right shows similarly shaped regions of capillary non-perfusion outlined in red. While some microaneurysms are shown by FA and PC-OCT (white circles), others are only shown in the FA image (blue circles) or PC-OCT (red circles).
Figure 4.
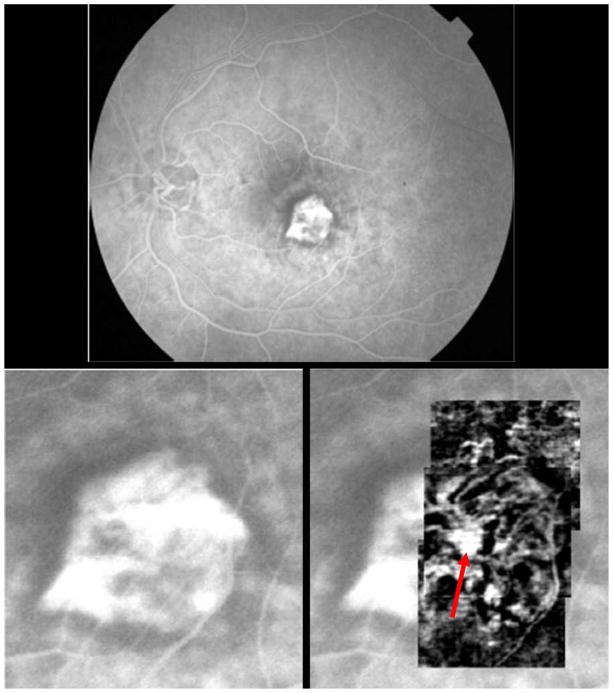
Wet age-related macular degeneration with subfoveal choroidal neovascularization (CNV). Top shows early laminar flow transit fluorescein angiography (FA) of subfoveal classic CNV. Lower left shows magnification of FA, zoomed into region of CNV. Lower right is overlay of phase-contrast optical coherence tomography (PC-OCT) montage of the subfoveal CNV. Note cartwheel shape of multiple vascular spokes emanating from probable central feeder vessel (red arrow). (PC-OCT data are based upon consecutive B-scans, each acquired with 25kHz University of California, Davis system over 1x1 mm2).
Optical Coherence Tomography Instrumentation
A schematic of the SD-OCT instruments used in this study has been reported previously.12,13 The OCT systems operated at a center wavelength of 855 nm and with a full-width-half-maximum of 75 nm, resulting in an axial resolution of approximately 4.5 μm in tissue and focused in the retina with an approximate lateral resolution of 15 μm. Scanning protocols used to acquire the PC-OCT data were volumetric scans comprised of BM-scans (B-M-mode scans), which are a series of sequential B-scans acquired over the same spatial region in the retina. The phase differences between sequential B-scans within each BM-scan were extracted for the phase-variance contrast calculation. The spacing between consecutive A-scans and BM-scans for the scan patterns presented here ranged from as small as 3 μm for the densely sampled scans to as large as 15 μm for scans with the lowest sampling density. Graphical programming–based software (LabVIEW; National Instruments, Austin, TX) was used to acquire and process PC-OCT data sets.
Phase-Contrast Method
Phase changes were calculated for the entire cross-sectional image from consecutively acquired B-scans. Bulk axial motion was calculated and corrected for each transverse location, reducing the phase noise introduced from eye motion. Phase contrast was calculated through the variance of the motion-corrected phase changes acquired within a BM-scan. Intensity thresholding of the average OCT intensity image was used to create a mask for the PC-OCT image that removes contributions of phase noise caused by low signal-to-noise regions. Repeating these calculations over the entire volumetric scan produces a three-dimensional PC-OCT representation of the vasculature. Further details regarding BM-scans and the phase variance contrast processing have been reported previously. 10,12
Image Processing
The volumetric data set processed by the phase-variance contrast method was segmented manually from the nerve fiber layer to the outer plexiform layer for characterization of the retinal circulation. The choroidal vasculature was imaged by segmenting the data deep to the retinal pigment epithelium (RPE) layer. Pseudo-color coding of depth position showed distinct locations of vascular networks. Its en face projection view produced a two-dimensional depth color-coded vasculature map.
Results
Normals: Comparison of retinal and choroidal vasculature imaged by FA and PC-OCT
Figure 1 shows a comparison between FA and PC-OCT imaging of the retinal vasculature along the inferotemporal arcade from a 60-year-old healthy male. Color coding of the PC-OCT images delineates capillary architecture of different retinal depths. This level of resolution is not depicted using conventional FA where microvascular resolution is partially “washed out” by background choroidal fluorescence.
Figure 2 demonstrates the capability of PC-OCT to image the choroid. FA (Fig. 2A) is shown of the same region for comparison with projection (Fig. 2B) of retinal vessels from PC-OCT data. As shown in Figure 1, PC-OCT imaging of the retinal vasculature compares favorably to resolution by FA of the same region. The depth-coded vasculature (Fig. 2C) of the choroid is shown by PC-OCT, where overlying retinal vessels are shown as shadows. Superficial vessels of the choriocapillaris and Sattler’s layer are shown, as are deeper, larger choroidal vessels of Haller’s layer.
Figure 3 shows another region of choroidal imaging from the same individual. Note the fine structure of apparent choroidal lobules (circled) with feeding arterioles at its center and radiating capillaries extending toward draining venules.
Dry and Wet Age-Related Macular Degeneration
Figure 4 shows an 85-year-old male with subfoveal choroidal neovascularization (CNV); vision is 20/100. Comparative imaging with FA and PC-OCT is shown. On the right, the CNV imaged with PC-OCT is overlaid over the high magnification FA. The ability of PC-OCT to capture the fine vascular detail of this classic CNV is evident. The enhanced clarity of microvascular detail revealed by PC-OCT compared to FA is in part because PC-OCT does not detect a breakdown of the blood retinal barrier. Fluorescein at this stage of the angiogram leaks from CNV and extravascular dye obscures fine microvasculature.
Figure 5 shows a case of AMD with geographic atrophy (GA) in a 61-year-old woman with vision of 20/60 OS. A red-free photograph shows two regions of well delineated GA, one centered just inferonasal to fixation and the other smaller area, just inferotemporal to the fovea. An early phase FA displays the regions of GA as well as adjacent areas of patchy atrophy. The two lower frames compare high magnification views by FA and PC-OCT. The PC-OCT image is color coded for depth; the areas of geographic atrophy show large choroidal vessels and loss of the overlying superficial choriocapillaris.
Diabetic Retinopathy
Figure 6 shows a case of non-proliferative diabetic retinopathy with regions of capillary non-perfusion in a 29-year-old woman with type 1 diabetes for 17 years; vision is 20/25 OS.
While regions of capillary non-perfusion, delineated with red contours in the right side of Figure 6, are comparably imaged using PC-OCT, microaneurysms are generally more apparent in the FA. Some microaneurysms seen on FA are not imaged by PC-OCT, and conversely others are visible by PC-OCT, but less so by FA.
Discussion
We have shown the capability of PC-OCT to provide imaging comparable to FA in normals, and patients with AMD and diabetic retinopathy. Obtaining PC-OCT angiography does not require hardware alterations in current SD-OCT instruments; instead, software analysis of data acquired during routine SD-OCT imaging is further processed to yield angiographic images. Indeed, we have recently begun using a commercial SD-OCT platform to generate PC-OCT images. In contrast to FA, PC-OCT angiographic imaging is accomplished non-invasively. Because it is technically easier to perform OCT imaging than FA, which requires a skilled photographer, PC-OCT is obtained with minimal training.
PC-OCT angiographic imaging is based upon the contrast of moving versus more static tissues. Therefore, it is particularly well suited to display the fine structure of the capillary networks. 3D imaging with PC-OCT generates high resolution depth imaging of retinal and choroidal vasculature using SD-OCT. While angiographic imaging with PC-OCT seems to compare favorably to conventional FA, processing methods currently used in PC-OCT do not reveal vascular leakage. Therefore, FA would be preferable for imaging cases of central serous retinopathy where detection of a leakage site is used to guide laser therapy.
Imaging of microaneurysms in diabetic retinopathy does not show exact correspondence between FA and PC-OCT. While some microaneurysms were shown by both imaging techniques, others were shown by only FA or only by PC-OCT. The reason for this is probably related to the plane of imaging by PC-OCT. While FA images all retinal and choroidal vasculature simultaneously, PC-OCT imaging is depth-based (volumetric). Thus microvascular alterations seen on FA could be potentially detected by PC-OCT if other retinal planes were imaged. We are exploring this possibility currently as more patients are imaged. Accurate depiction of microaneurysms by PC-OCT is important if it were to be used as an alternative to FA for guiding laser therapy.
How would PC-OCT be used in clinical practice? Because images are obtained with standard SD-OCT imaging, PC-OCT imaging of diabetic patients might allow improved characterization of disease severity. Specifically, the extent of retinal capillary non-perfusion could be characterized better than by ophthalmoscopy alone. While this could also be achieved using FA, this is not commonly done because of cost and complexity of this test. Furthermore, while not demonstrated herein, PC-OCT could be used to follow patients with high-risk drusen as a means to detect early CNV, perhaps before it becomes symptomatic and causes visual loss. Were this the case, then anti-vascular endothelial growth factor (VEGF) therapy might be initiated earlier in the course of wet AMD with possible preservation of better vision. The ability of PC-OCT to characterize choroidal microvasculature is also intriguing. Imaging could potentially improve our understanding of geographic atrophy and how alterations in the choriocapillaris might also contribute to development of wet AMD.30
Because of its simplicity, non-invasive character, and apparent improved ability to render microvascular detail, PC-OCT may be used increasingly as an alternative to FA.
Acknowledgments
Financial Support: Supported by the National Eye Institute (EY 014743), Research to Prevent Blindness (RPB), Beckman Institute, That Man May See Foundation, Howard Hughes Medical Institute (HHMI) Med-into-Grad Initiative (HHMI-MIG 56006769). The sponsors and funding agencies had no role in the design or conduct of this research.
Footnotes
Conflicts of Interest: Drs. Schwartz, Fraser and Fingler hold an issued patent on the PC-OCT technology. They also hold founders shares in a company with an interest in the technology. Drs. Kim, Zawadski, Morse, Park and Werner have no conflict of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Alvis D. Happy 50th birthday [letter] Ophthalmology. 2009;116:2259. doi: 10.1016/j.ophtha.2009.09.038. [DOI] [PubMed] [Google Scholar]
- 2.Marmor MF, Ravin JG. Fluorescein angiography: insight and serendipity a half century ago. Arch Ophthalmol. 2011;129:943–8. doi: 10.1001/archophthalmol.2011.160. [DOI] [PubMed] [Google Scholar]
- 3.Swanson EA, Huang D. Ophthalmic OCT reaches $1 billion per year. Retin Physician. 2011;8(4):45, 58–59, 62. [Google Scholar]
- 4.Lipson BK, Yannuzzi LA. Complications of intravenous fluorescein injections. Int Ophthalmol Clin. 1989;29(3):200–5. doi: 10.1097/00004397-198902930-00011. [DOI] [PubMed] [Google Scholar]
- 5.Owens SL. Indocyanine green angiography. Br J Ophthalmol. 1996;80:263–6. doi: 10.1136/bjo.80.3.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pauleikhoff D, Spital G, Radermacher M, et al. A fluorescein and indocyanine green angiographic study of choriocapillaris in age-related macular disease. Arch Ophthalmol. 1999;117:1353–8. doi: 10.1001/archopht.117.10.1353. [DOI] [PubMed] [Google Scholar]
- 7.Flower RW, Fryczkowski AW, McLeod DS. Variability in choriocapillaris blood flow distribution. Invest Ophthalmol Vis Sci. 1995;36:1247–58. [PubMed] [Google Scholar]
- 8.Huang D, Swanson EA, Lin CP, et al. Optical coherence tomography. Science. 1991;254:1178–81. doi: 10.1126/science.1957169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.van Velthoven ME, Faber DJ, Verbraak FD, et al. Recent developments in optical coherence tomography for imaging the retina. Prog Retin Eye Res. 2007;26:57–77. doi: 10.1016/j.preteyeres.2006.10.002. [DOI] [PubMed] [Google Scholar]
- 10.Fingler J, Schwartz D, Yang C, Fraser SE. Mobility and transverse flow visualization using phase variance contrast with spectral domain optical coherence tomography. [Accessed August 24, 2013];Opt Express [serial online] 2007 15:12636–53. doi: 10.1364/oe.15.012636. Available at: http://www.opticsinfobase.org/oe/abstract.cfm?uri=oe-15-20-12636. [DOI] [PubMed] [Google Scholar]
- 11.Fingler J, Readhead C, Schwartz DM, Fraser SE. Phase-contrast OCT imaging of transverse flows in the mouse retina and choroid. Invest Ophthalmol Vis Sci. 2008;49:5055–9. doi: 10.1167/iovs.07-1627. [DOI] [PubMed] [Google Scholar]
- 12.Fingler J, Zawadzki RJ, Werner JS, et al. Volumetric microvascular imaging of human retina using optical coherence tomography with a novel motion contrast technique. [Accessed August 24, 2013];Opt Express [serial online] 2009 17:22190–200. doi: 10.1364/OE.17.022190. Available at: http://www.opticsinfobase.org/oe/abstract.cfm?uri=oe-17-24-22190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kim DY, Fingler J, Werner JS, et al. In vivo volumetric imaging of human retinal circulation with phase-variance optical coherence tomography. [Accessed August 24, 2013];Biomed Opt Express [serial online] 2011 2:1504–13. doi: 10.1364/BOE.2.001504. Available at: http://www.opticsinfobase.org/boe/fulltext.cfm?uri=boe-2-6-1504&id=213832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim DY, Fingler J, Zawadzki RJ, et al. Noninvasive imaging of the foveal avascular zone with high-speed, phase-variance optical coherence tomography. Invest Ophthalmol Vis Sci. 2012;53:85–92. doi: 10.1167/iovs.11-8249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.White B, Pierce M, Nassif N, et al. In vivo dynamic human retinal blood flow imaging using ultra-high-speed spectral domain optical coherence tomography. [Accessed August 24, 2013];Opt Express [serial online] 2003 11:3490–7. doi: 10.1364/oe.11.003490. Available at: http://www.opticsinfobase.org/oe/abstract.cfm?uri=oe-11-25-3490. [DOI] [PubMed] [Google Scholar]
- 16.Leitgeb RA, Schmetterer L, Hitzenberger CK, et al. Real-time measurement of in vitro flow by Fourier-domain color Doppler optical coherence tomography. Opt Lett. 2004;29:171–3. doi: 10.1364/ol.29.000171. [DOI] [PubMed] [Google Scholar]
- 17.Tao YK, Kennedy KM, Izatt JA. Velocity-resolved 3D retinal microvessel imaging using single-flow imaging spectral domain optical coherence tomography. [Accessed August 24, 2013];Opt Express [serial online] 2009 17:4177–88. doi: 10.1364/oe.17.004177. Available at: http://www.opticsinfobase.org/oe/abstract.cfm?uri=oe-17-5-4177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Grulkowski I, Gorczynska I, Szkulmowski M, et al. Scanning protocols dedicated to smart velocity ranging in spectral OCT. [Accessed August 24, 2013];Opt Express [serial online] 2009 17:23736–54. doi: 10.1364/OE.17.023736. Available at: http://www.opticsinfobase.org/oe/abstract.cfm?uri=oe-17-26-23736. [DOI] [PubMed] [Google Scholar]
- 19.Zotter S, Pircher M, Torzicky T, et al. Visualization of microvasculature by dual-beam phase-resolved Doppler optical coherence tomography. [Accessed August 24, 2013];Opt Express [serial online] 2011 19:1217–27. doi: 10.1364/OE.19.001217. Available at: http://www.opticsinfobase.org/oe/fulltext.cfm?uri=oe-19-2-1217&id=209402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Makita S, Jaillon F, Yamanari M, et al. Comprehensive in vivo micro-vascular imaging of the human eye by dual-beam-scan Doppler optical coherence angiography. [Accessed August 24, 2013];Opt Express [serial online] 2011 19:1271–83. doi: 10.1364/OE.19.001271. Available at: http://www.opticsinfobase.org/oe/fulltext.cfm?uri=oe-19-2-1271&id=209407. [DOI] [PubMed] [Google Scholar]
- 21.Wang RK, Jacques SL, Ma Z, et al. Three dimensional optical angiography. [Accessed August 24, 2013];Opt Express [serial online] 2007 15:4083–97. doi: 10.1364/oe.15.004083. Available at: http://www.opticsinfobase.org/oe/abstract.cfm?uri=oe-15-7-4083. [DOI] [PubMed] [Google Scholar]
- 22.Braaf B, Vermeer KA, Vienola KV, de Boer JF. Angiography of the retina and the choroid with phase-resolved OCT using interval-optimized backstitched B-scans. [Accessed August 24, 2013];Opt Express [serial online] 2012 20:20516–34. doi: 10.1364/OE.20.020516. Available at: http://www.opticsinfobase.org/oe/fulltext.cfm?uri=oe-20-18-20516&id=240889. [DOI] [PubMed] [Google Scholar]
- 23.Kurokawa K, Sasaki K, Makita S, et al. Three-dimensional retinal and choroidal capillary imaging by power Doppler optical coherence angiography with adaptive optics. [Accessed August 24, 2013];Opt Express [serial online] 2012 20:22796–812. doi: 10.1364/OE.20.022796. Available at: http://www.opticsinfobase.org/oe/fulltext.cfm?uri=oe-20-20-22796&id=242390. [DOI] [PubMed] [Google Scholar]
- 24.Braaf B, Vienola KV, Christy K, et al. Real-time eye motion correction in phase-resolved OCT angiography with tracking SLO. [Accessed August 24, 2013];Biomed Opt Express [serial online] 2013 4:51–65. doi: 10.1364/BOE.4.000051. Available at: http://www.opticsinfobase.org/boe/fulltext.cfm?uri=boe-4-1-51&id=246986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schmoll T, Singh AS, Blatter C, et al. Imaging of the parafoveal capillary network and its integrity analysis using fractal dimension. [Accessed August 24, 2013];Biomed Opt Express [serial online] 2011 2:1159–68. doi: 10.1364/BOE2.001159. Available at: http://www.opticsinfobase.org/boe/fulltext.cfm?uri=boe-2-5-1159&id=211741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mariampillai A, Leung MK, Jarvi BA, et al. Optimized speckle variance OCT imaging of microvasculature. Opt Lett. 2010;35:1257–9. doi: 10.1364/OL.35.001257. [DOI] [PubMed] [Google Scholar]
- 27.Motaghiannezam R, Fraser S. Logarithmic intensity and speckle-based motion contrast methods for human retinal vasculature visualization using swept source optical coherence tomography. [Accessed August 24, 2013];Biomed Opt Express [serial online] 2012 3:503–21. doi: 10.1364/BOE.3.000503. Available at: http://www.opticsinfobase.org/boe/fulltext.cfm?uri=boe-3-3-503&id=227627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jia Y, Tan O, Tokayer J, et al. Split-spectrum amplitude-decorrelation angiography with optical coherence tomography. [Accessed August 24, 2013];Opt Express [serial online] 2012 20:4710–25. doi: 10.1364/OE.20.004710. Available at: http://www.opticsinfobase.org/oe/fulltext.cfm?uri=oe-20-4-4710&id=227624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liu G, Chou L, Jia W, et al. Intensity-based modified Doppler variance algorithm: application to phase instable and phase stable optical coherence tomography systems. [Accessed August 24, 2013];Opt Express [serial online] 2011 19:11429–40. doi: 10.1364/OE.19.011429. Available at: http://www.opticsinfobase.org/oe/fulltext.cfm?uri=oe-19-12-11429&id=214327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bhutto I, Lutty G. Understanding age-related macular degeneration (AMD): Relationships between the photoreceptor/retinal pigment epithelium/Bruch’s membrane/choroiocapillaris complex. Mol Aspects Med. 2012;33:295–317. doi: 10.1016/j.mam.2012.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]