Abstract
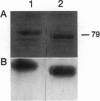
Resistance to bacterial speck in tomato is governed by a gene-for-gene interaction in which a single resistance locus (Pto) in the plant responds to the expression of a specific avirulence gene (avrPto) in the pathogen. Disease susceptibility results if either Pto or avrPto are lacking from the corresponding organisms. Leaves of tomato cultivars that contain the Pto locus also exhibit a hypersensitive-like response upon exposure to an organophosphorous insecticide, fenthion. Recently, the Pto gene was isolated by a map-based cloning approach and was shown to be a member of a clustered multigene family with similarity to various protein-serine/threonine kinases. Another member of this family, termed Fen, was found to confer sensitivity to fenthion. The Pto protein shares 80% identity (87% similarity) with Fen. Here, Pto and Fen are shown to be functional protein kinases that probably participate in the same signal transduction pathway.
Full text
PDF
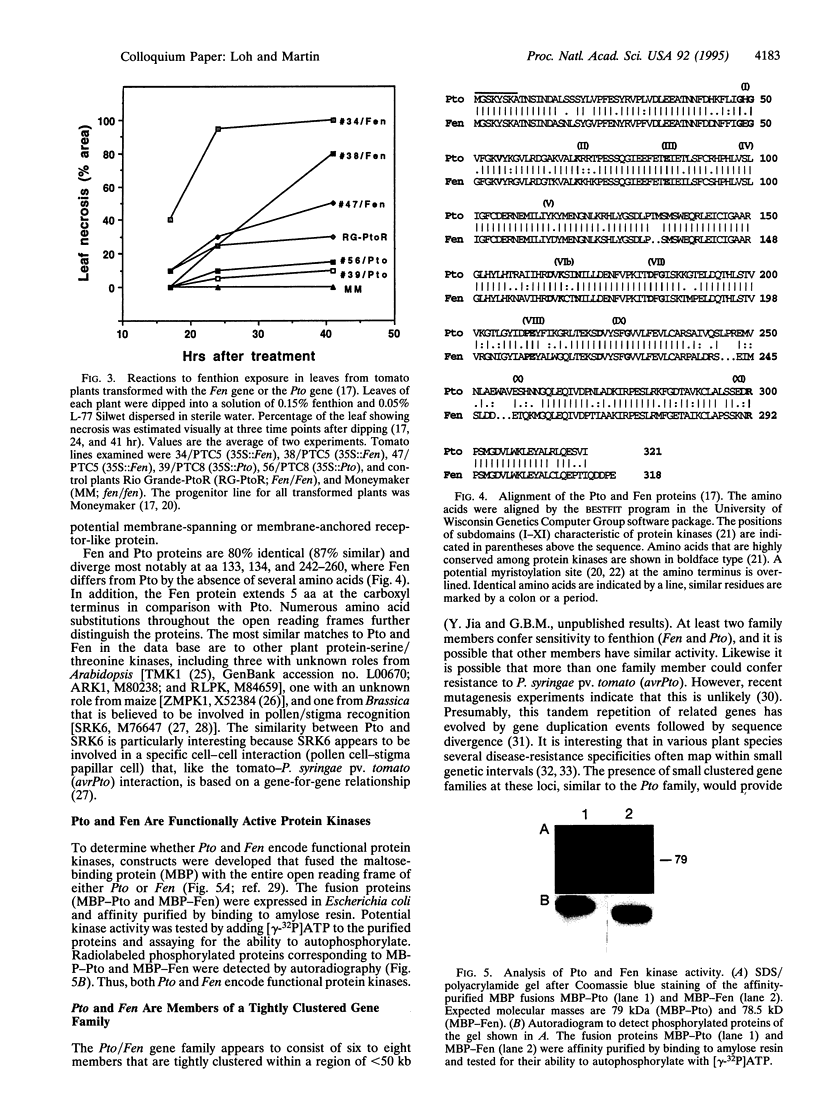

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Altschul S. F., Gish W., Miller W., Myers E. W., Lipman D. J. Basic local alignment search tool. J Mol Biol. 1990 Oct 5;215(3):403–410. doi: 10.1016/S0022-2836(05)80360-2. [DOI] [PubMed] [Google Scholar]
- Bennetzen J. L., Hulbert S. H. Organization, instability and evolution of plant disease resistance genes. Plant Mol Biol. 1992 Nov;20(4):575–578. [PubMed] [Google Scholar]
- Buss J. E., Kamps M. P., Gould K., Sefton B. M. The absence of myristic acid decreases membrane binding of p60src but does not affect tyrosine protein kinase activity. J Virol. 1986 May;58(2):468–474. doi: 10.1128/jvi.58.2.468-474.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carland F. M., Staskawicz B. J. Genetic characterization of the Pto locus of tomato: semi-dominance and cosegregation of resistance to Pseudomonas syringae pathovar tomato and sensitivity to the insecticide Fenthion. Mol Gen Genet. 1993 May;239(1-2):17–27. doi: 10.1007/BF00281596. [DOI] [PubMed] [Google Scholar]
- Chang C., Schaller G. E., Patterson S. E., Kwok S. F., Meyerowitz E. M., Bleecker A. B. The TMK1 gene from Arabidopsis codes for a protein with structural and biochemical characteristics of a receptor protein kinase. Plant Cell. 1992 Oct;4(10):1263–1271. doi: 10.1105/tpc.4.10.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickinson M. J., Jones D. A., Jones J. D. Close linkage between the Cf-2/Cf-5 and Mi resistance loci in tomato. Mol Plant Microbe Interact. 1993 May-Jun;6(3):341–347. doi: 10.1094/mpmi-6-341. [DOI] [PubMed] [Google Scholar]
- Grand R. J. Acylation of viral and eukaryotic proteins. Biochem J. 1989 Mar 15;258(3):625–638. doi: 10.1042/bj2580625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanks S. K., Quinn A. M., Hunter T. The protein kinase family: conserved features and deduced phylogeny of the catalytic domains. Science. 1988 Jul 1;241(4861):42–52. doi: 10.1126/science.3291115. [DOI] [PubMed] [Google Scholar]
- Maina C. V., Riggs P. D., Grandea A. G., 3rd, Slatko B. E., Moran L. S., Tagliamonte J. A., McReynolds L. A., Guan C. D. An Escherichia coli vector to express and purify foreign proteins by fusion to and separation from maltose-binding protein. Gene. 1988 Dec 30;74(2):365–373. doi: 10.1016/0378-1119(88)90170-9. [DOI] [PubMed] [Google Scholar]
- Martin G. B., Brommonschenkel S. H., Chunwongse J., Frary A., Ganal M. W., Spivey R., Wu T., Earle E. D., Tanksley S. D. Map-based cloning of a protein kinase gene conferring disease resistance in tomato. Science. 1993 Nov 26;262(5138):1432–1436. doi: 10.1126/science.7902614. [DOI] [PubMed] [Google Scholar]
- Martin G. B., Frary A., Wu T., Brommonschenkel S., Chunwongse J., Earle E. D., Tanksley S. D. A member of the tomato Pto gene family confers sensitivity to fenthion resulting in rapid cell death. Plant Cell. 1994 Nov;6(11):1543–1552. doi: 10.1105/tpc.6.11.1543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin G. B., Ganal M. W., Tanksley S. D. Construction of a yeast artificial chromosome library of tomato and identification of cloned segments linked to two disease resistance loci. Mol Gen Genet. 1992 May;233(1-2):25–32. doi: 10.1007/BF00587557. [DOI] [PubMed] [Google Scholar]
- Martin G. B., Williams J. G., Tanksley S. D. Rapid identification of markers linked to a Pseudomonas resistance gene in tomato by using random primers and near-isogenic lines. Proc Natl Acad Sci U S A. 1991 Mar 15;88(6):2336–2340. doi: 10.1073/pnas.88.6.2336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ronald P. C., Salmeron J. M., Carland F. M., Staskawicz B. J. The cloned avirulence gene avrPto induces disease resistance in tomato cultivars containing the Pto resistance gene. J Bacteriol. 1992 Mar;174(5):1604–1611. doi: 10.1128/jb.174.5.1604-1611.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salmeron J. M., Barker S. J., Carland F. M., Mehta A. Y., Staskawicz B. J. Tomato mutants altered in bacterial disease resistance provide evidence for a new locus controlling pathogen recognition. Plant Cell. 1994 Apr;6(4):511–520. doi: 10.1105/tpc.6.4.511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salmeron J. M., Staskawicz B. J. Molecular characterization and hrp dependence of the avirulence gene avrPto from Pseudomonas syringae pv. tomato [corrected]. Mol Gen Genet. 1993 May;239(1-2):6–16. doi: 10.1007/BF00281595. [DOI] [PubMed] [Google Scholar]
- Stein J. C., Howlett B., Boyes D. C., Nasrallah M. E., Nasrallah J. B. Molecular cloning of a putative receptor protein kinase gene encoded at the self-incompatibility locus of Brassica oleracea. Proc Natl Acad Sci U S A. 1991 Oct 1;88(19):8816–8820. doi: 10.1073/pnas.88.19.8816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stein J. C., Nasrallah J. B. A plant receptor-like gene, the S-locus receptor kinase of Brassica oleracea L., encodes a functional serine/threonine kinase. Plant Physiol. 1993 Mar;101(3):1103–1106. doi: 10.1104/pp.101.3.1103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker J. C., Zhang R. Relationship of a putative receptor protein kinase from maize to the S-locus glycoproteins of Brassica. Nature. 1990 Jun 21;345(6277):743–746. doi: 10.1038/345743a0. [DOI] [PubMed] [Google Scholar]