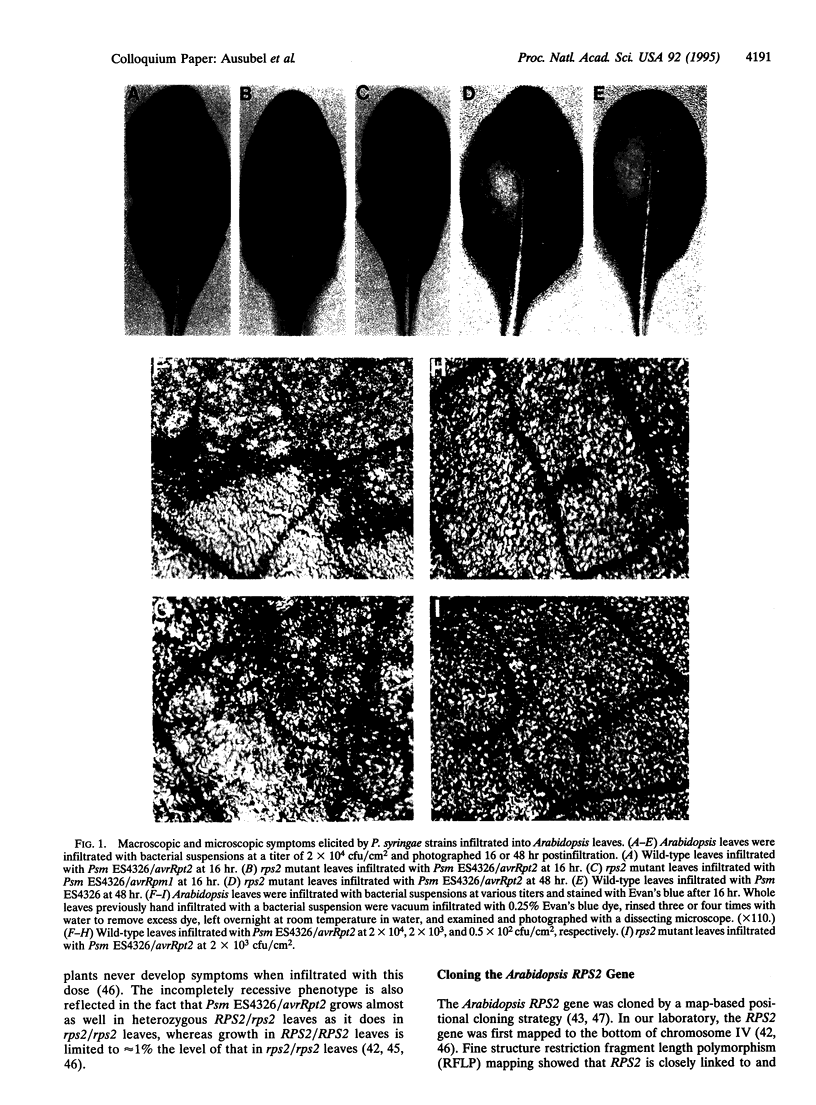
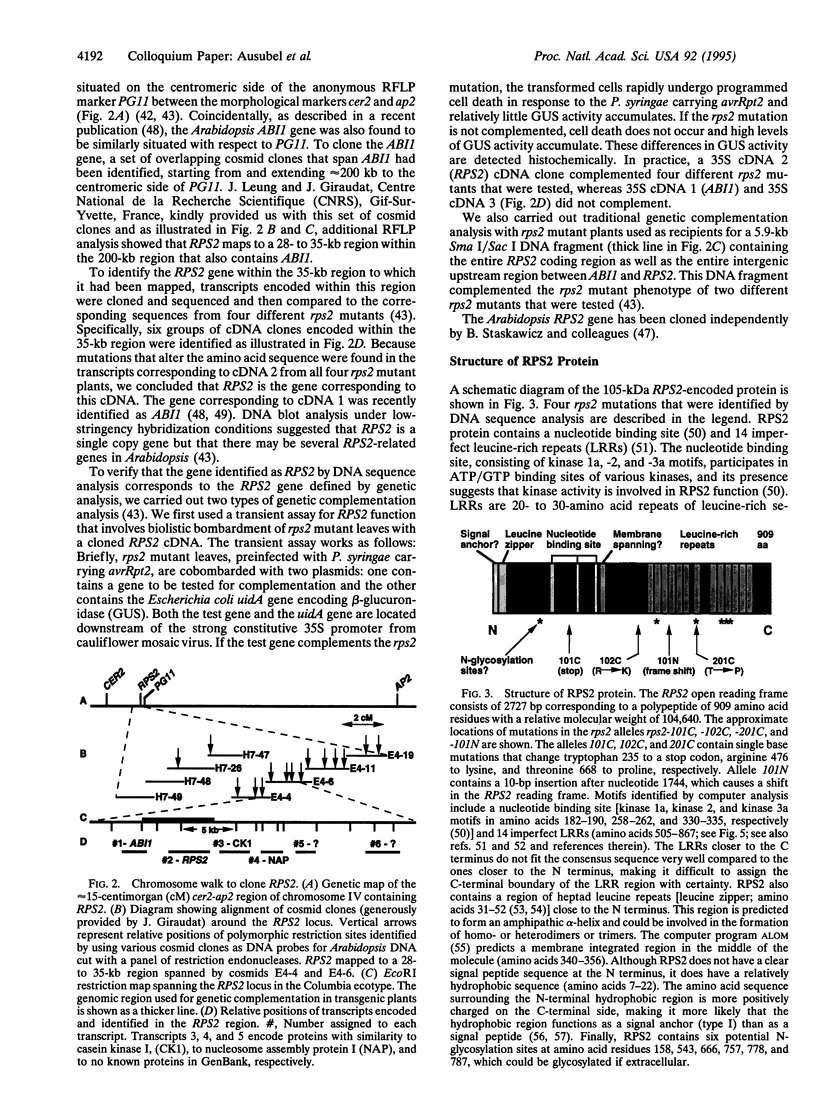
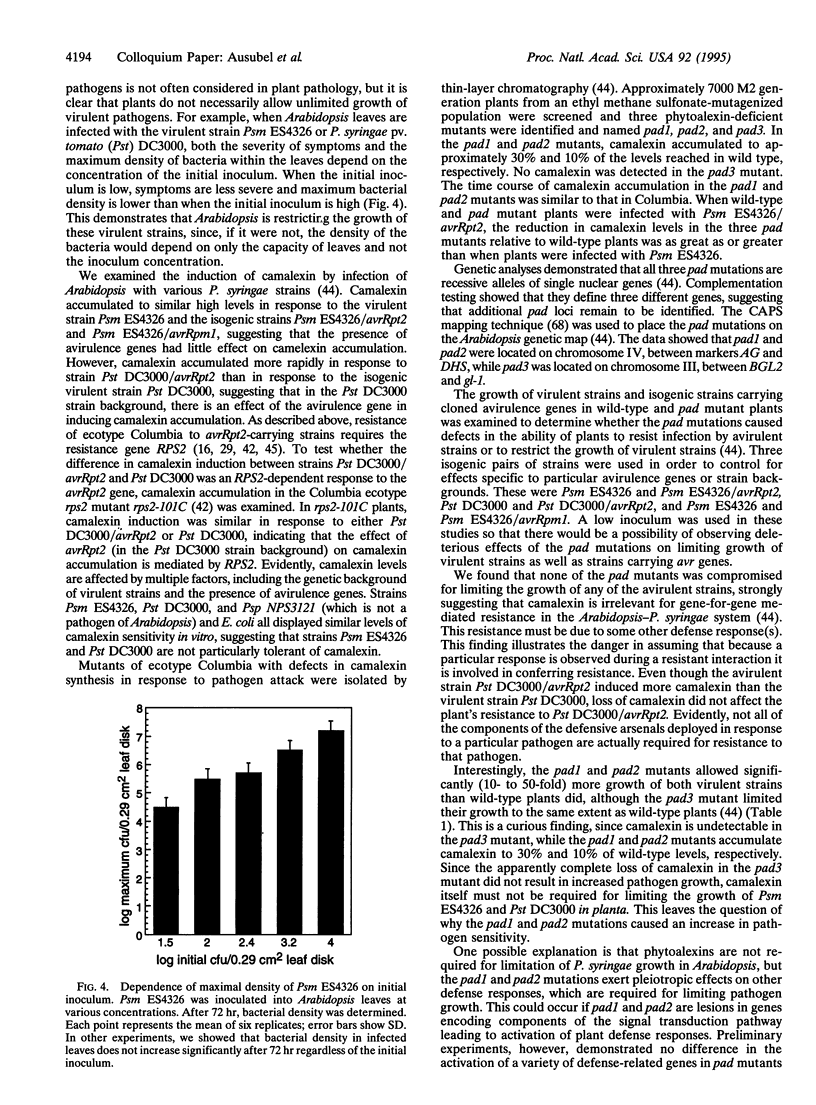
Abstract
The plant defense response to microbial pathogens had been studied primarily by using biochemical and physiological techniques. Recently, several laboratories have developed a variety of pathosystems utilizing Arabidopsis thaliana as a model host so that genetic analysis could also be used to study plant defense responses. Utilizing a pathosystem that involves the infection of Arabidopsis with pathogenic pseudomonads, we have cloned the Arabidopsis disease-resistance gene RPS2, which corresponds to the avirulence gene avrRpt2 in a gene-for-gene relationship. RPS2 encodes a 105-kDa protein containing a leucine zipper, a nucleotide binding site, and 14 imperfect leucine-rich repeats. The RPS2 protein is remarkably similar to the product of the tobacco N gene, which confers resistance to tobacco mosaic virus. We have also isolated a series of Arabidopsis mutants that synthesize decreased levels of an Arabidopsis phytoalexin called camalexin. Analysis of these mutants indicated that camalexin does not play a significant role in limiting growth of avirulent Pseudomonas syringae strains during the hypersensitive defense response but that it may play a role in limiting the growth of virulent strains. More generally, we have shown that we can utilize Arabidopsis to systematically dissect the defense response by isolation and characterization of appropriate defense-related mutants.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bent A. F., Kunkel B. N., Dahlbeck D., Brown K. L., Schmidt R., Giraudat J., Leung J., Staskawicz B. J. RPS2 of Arabidopsis thaliana: a leucine-rich repeat class of plant disease resistance genes. Science. 1994 Sep 23;265(5180):1856–1860. doi: 10.1126/science.8091210. [DOI] [PubMed] [Google Scholar]
- Braun T., Schofield P. R., Sprengel R. Amino-terminal leucine-rich repeats in gonadotropin receptors determine hormone selectivity. EMBO J. 1991 Jul;10(7):1885–1890. doi: 10.1002/j.1460-2075.1991.tb07714.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Damgaard O., Rasmussen O. Direct regeneration of transformed shoots in Brassica napus from hypocotyl infections with Agrobacterium rhizogenes. Plant Mol Biol. 1991 Jul;17(1):1–8. doi: 10.1007/BF00036800. [DOI] [PubMed] [Google Scholar]
- Dong X., Mindrinos M., Davis K. R., Ausubel F. M. Induction of Arabidopsis defense genes by virulent and avirulent Pseudomonas syringae strains and by a cloned avirulence gene. Plant Cell. 1991 Jan;3(1):61–72. doi: 10.1105/tpc.3.1.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freialdenhoven A., Scherag B., Hollricher K., Collinge D. B., Thordal-Christensen H., Schulze-Lefert P. Nar-1 and Nar-2, Two Loci Required for Mla12-Specified Race-Specific Resistance to Powdery Mildew in Barley. Plant Cell. 1994 Jul;6(7):983–994. doi: 10.1105/tpc.6.7.983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glazebrook J., Ausubel F. M. Isolation of phytoalexin-deficient mutants of Arabidopsis thaliana and characterization of their interactions with bacterial pathogens. Proc Natl Acad Sci U S A. 1994 Sep 13;91(19):8955–8959. doi: 10.1073/pnas.91.19.8955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hain R., Reif H. J., Krause E., Langebartels R., Kindl H., Vornam B., Wiese W., Schmelzer E., Schreier P. H., Stöcker R. H. Disease resistance results from foreign phytoalexin expression in a novel plant. Nature. 1993 Jan 14;361(6408):153–156. doi: 10.1038/361153a0. [DOI] [PubMed] [Google Scholar]
- Hammond-Kosack K. E., Jones D. A., Jones JDG. Identification of Two Genes Required in Tomato for Full Cf-9-Dependent Resistance to Cladosporium fulvum. Plant Cell. 1994 Mar;6(3):361–374. doi: 10.1105/tpc.6.3.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartmann E., Rapoport T. A., Lodish H. F. Predicting the orientation of eukaryotic membrane-spanning proteins. Proc Natl Acad Sci U S A. 1989 Aug;86(15):5786–5790. doi: 10.1073/pnas.86.15.5786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joosten M. H., Cozijnsen T. J., De Wit P. J. Host resistance to a fungal tomato pathogen lost by a single base-pair change in an avirulence gene. Nature. 1994 Jan 27;367(6461):384–386. doi: 10.1038/367384a0. [DOI] [PubMed] [Google Scholar]
- Kawalleck P., Keller H., Hahlbrock K., Scheel D., Somssich I. E. A pathogen-responsive gene of parsley encodes tyrosine decarboxylase. J Biol Chem. 1993 Jan 25;268(3):2189–2194. [PubMed] [Google Scholar]
- Keen N. T. The molecular biology of disease resistance. Plant Mol Biol. 1992 May;19(1):109–122. doi: 10.1007/BF00015609. [DOI] [PubMed] [Google Scholar]
- Keith B., Dong X. N., Ausubel F. M., Fink G. R. Differential induction of 3-deoxy-D-arabino-heptulosonate 7-phosphate synthase genes in Arabidopsis thaliana by wounding and pathogenic attack. Proc Natl Acad Sci U S A. 1991 Oct 1;88(19):8821–8825. doi: 10.1073/pnas.88.19.8821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiedrowski S., Kawalleck P., Hahlbrock K., Somssich I. E., Dangl J. L. Rapid activation of a novel plant defense gene is strictly dependent on the Arabidopsis RPM1 disease resistance locus. EMBO J. 1992 Dec;11(13):4677–4684. doi: 10.1002/j.1460-2075.1992.tb05572.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein P., Kanehisa M., DeLisi C. The detection and classification of membrane-spanning proteins. Biochim Biophys Acta. 1985 May 28;815(3):468–476. doi: 10.1016/0005-2736(85)90375-x. [DOI] [PubMed] [Google Scholar]
- Knorr D. A., Dawson W. O. A point mutation in the tobacco mosaic virus capsid protein gene induces hypersensitivity in Nicotiana sylvestris. Proc Natl Acad Sci U S A. 1988 Jan;85(1):170–174. doi: 10.1073/pnas.85.1.170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobe B., Deisenhofer J. Crystal structure of porcine ribonuclease inhibitor, a protein with leucine-rich repeats. Nature. 1993 Dec 23;366(6457):751–756. doi: 10.1038/366751a0. [DOI] [PubMed] [Google Scholar]
- Koch E., Slusarenko A. Arabidopsis is susceptible to infection by a downy mildew fungus. Plant Cell. 1990 May;2(5):437–445. doi: 10.1105/tpc.2.5.437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konieczny A., Ausubel F. M. A procedure for mapping Arabidopsis mutations using co-dominant ecotype-specific PCR-based markers. Plant J. 1993 Aug;4(2):403–410. doi: 10.1046/j.1365-313x.1993.04020403.x. [DOI] [PubMed] [Google Scholar]
- Kunkel B. N., Bent A. F., Dahlbeck D., Innes R. W., Staskawicz B. J. RPS2, an Arabidopsis disease resistance locus specifying recognition of Pseudomonas syringae strains expressing the avirulence gene avrRpt2. Plant Cell. 1993 Aug;5(8):865–875. doi: 10.1105/tpc.5.8.865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamb C. J., Lawton M. A., Dron M., Dixon R. A. Signals and transduction mechanisms for activation of plant defenses against microbial attack. Cell. 1989 Jan 27;56(2):215–224. doi: 10.1016/0092-8674(89)90894-5. [DOI] [PubMed] [Google Scholar]
- Landschulz W. H., Johnson P. F., McKnight S. L. The leucine zipper: a hypothetical structure common to a new class of DNA binding proteins. Science. 1988 Jun 24;240(4860):1759–1764. doi: 10.1126/science.3289117. [DOI] [PubMed] [Google Scholar]
- Leung J., Bouvier-Durand M., Morris P. C., Guerrier D., Chefdor F., Giraudat J. Arabidopsis ABA response gene ABI1: features of a calcium-modulated protein phosphatase. Science. 1994 Jun 3;264(5164):1448–1452. doi: 10.1126/science.7910981. [DOI] [PubMed] [Google Scholar]
- Martin G. B., Brommonschenkel S. H., Chunwongse J., Frary A., Ganal M. W., Spivey R., Wu T., Earle E. D., Tanksley S. D. Map-based cloning of a protein kinase gene conferring disease resistance in tomato. Science. 1993 Nov 26;262(5138):1432–1436. doi: 10.1126/science.7902614. [DOI] [PubMed] [Google Scholar]
- Melan M. A., Dong X., Endara M. E., Davis K. R., Ausubel F. M., Peterman T. K. An Arabidopsis thaliana lipoxygenase gene can be induced by pathogens, abscisic acid, and methyl jasmonate. Plant Physiol. 1993 Feb;101(2):441–450. doi: 10.1104/pp.101.2.441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metzler M. C., Cutt J. R., Klessig D. F. Isolation and Characterization of a Gene Encoding a PR-1-Like Protein from Arabidopsis thaliana. Plant Physiol. 1991 May;96(1):346–348. doi: 10.1104/pp.96.1.346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer K., Leube M. P., Grill E. A protein phosphatase 2C involved in ABA signal transduction in Arabidopsis thaliana. Science. 1994 Jun 3;264(5164):1452–1455. doi: 10.1126/science.8197457. [DOI] [PubMed] [Google Scholar]
- Meyerowitz E. M. Arabidopsis, a useful weed. Cell. 1989 Jan 27;56(2):263–269. doi: 10.1016/0092-8674(89)90900-8. [DOI] [PubMed] [Google Scholar]
- Mindrinos M., Katagiri F., Yu G. L., Ausubel F. M. The A. thaliana disease resistance gene RPS2 encodes a protein containing a nucleotide-binding site and leucine-rich repeats. Cell. 1994 Sep 23;78(6):1089–1099. doi: 10.1016/0092-8674(94)90282-8. [DOI] [PubMed] [Google Scholar]
- Niyogi K. K., Fink G. R. Two anthranilate synthase genes in Arabidopsis: defense-related regulation of the tryptophan pathway. Plant Cell. 1992 Jun;4(6):721–733. doi: 10.1105/tpc.4.6.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Shea E. K., Klemm J. D., Kim P. S., Alber T. X-ray structure of the GCN4 leucine zipper, a two-stranded, parallel coiled coil. Science. 1991 Oct 25;254(5031):539–544. doi: 10.1126/science.1948029. [DOI] [PubMed] [Google Scholar]
- Ohl S., Hedrick S. A., Chory J., Lamb C. J. Functional properties of a phenylalanine ammonia-lyase promoter from Arabidopsis. Plant Cell. 1990 Sep;2(9):837–848. doi: 10.1105/tpc.2.9.837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parker J. E., Barber C. E., Fan M. J., Daniels M. J. Interaction of Xanthomonas campestris with Arabidopsis thaliana: characterization of a gene from X. c. pv. raphani that confers avirulence to most A. thaliana accessions. Mol Plant Microbe Interact. 1993 Mar-Apr;6(2):216–224. doi: 10.1094/mpmi-6-216. [DOI] [PubMed] [Google Scholar]
- Rothberg J. M., Jacobs J. R., Goodman C. S., Artavanis-Tsakonas S. slit: an extracellular protein necessary for development of midline glia and commissural axon pathways contains both EGF and LRR domains. Genes Dev. 1990 Dec;4(12A):2169–2187. doi: 10.1101/gad.4.12a.2169. [DOI] [PubMed] [Google Scholar]
- Salmeron J. M., Barker S. J., Carland F. M., Mehta A. Y., Staskawicz B. J. Tomato mutants altered in bacterial disease resistance provide evidence for a new locus controlling pathogen recognition. Plant Cell. 1994 Apr;6(4):511–520. doi: 10.1105/tpc.6.4.511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samac D. A., Hironaka C. M., Yallaly P. E., Shah D. M. Isolation and Characterization of the Genes Encoding Basic and Acidic Chitinase in Arabidopsis thaliana. Plant Physiol. 1990 Jul;93(3):907–914. doi: 10.1104/pp.93.3.907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samac D. A., Shah D. M. Developmental and Pathogen-Induced Activation of the Arabidopsis Acidic Chitinase Promoter. Plant Cell. 1991 Oct;3(10):1063–1072. doi: 10.1105/tpc.3.10.1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki N., Choe H. R., Nishida Y., Yamawaki-Kataoka Y., Ohnishi S., Tamaoki T., Kataoka T. Leucine-rich repeats and carboxyl terminus are required for interaction of yeast adenylate cyclase with RAS proteins. Proc Natl Acad Sci U S A. 1990 Nov;87(22):8711–8715. doi: 10.1073/pnas.87.22.8711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traut T. W. The functions and consensus motifs of nine types of peptide segments that form different types of nucleotide-binding sites. Eur J Biochem. 1994 May 15;222(1):9–19. doi: 10.1111/j.1432-1033.1994.tb18835.x. [DOI] [PubMed] [Google Scholar]
- Tsuji J., Jackson E. P., Gage D. A., Hammerschmidt R., Somerville S. C. Phytoalexin Accumulation in Arabidopsis thaliana during the Hypersensitive Reaction to Pseudomonas syringae pv syringae. Plant Physiol. 1992 Apr;98(4):1304–1309. doi: 10.1104/pp.98.4.1304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uknes S., Mauch-Mani B., Moyer M., Potter S., Williams S., Dincher S., Chandler D., Slusarenko A., Ward E., Ryals J. Acquired resistance in Arabidopsis. Plant Cell. 1992 Jun;4(6):645–656. doi: 10.1105/tpc.4.6.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van den Ackerveken G. F., Van Kan J. A., De Wit P. J. Molecular analysis of the avirulence gene avr9 of the fungal tomato pathogen Cladosporium fulvum fully supports the gene-for-gene hypothesis. Plant J. 1992 May;2(3):359–366. doi: 10.1111/j.1365-313x.1992.00359.x. [DOI] [PubMed] [Google Scholar]
- Verburg J. G., Huynh Q. K. Purification and Characterization of an Antifungal Chitinase from Arabidopsis thaliana. Plant Physiol. 1991 Feb;95(2):450–455. doi: 10.1104/pp.95.2.450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whalen M. C., Innes R. W., Bent A. F., Staskawicz B. J. Identification of Pseudomonas syringae pathogens of Arabidopsis and a bacterial locus determining avirulence on both Arabidopsis and soybean. Plant Cell. 1991 Jan;3(1):49–59. doi: 10.1105/tpc.3.1.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitham S., Dinesh-Kumar S. P., Choi D., Hehl R., Corr C., Baker B. The product of the tobacco mosaic virus resistance gene N: similarity to toll and the interleukin-1 receptor. Cell. 1994 Sep 23;78(6):1101–1115. doi: 10.1016/0092-8674(94)90283-6. [DOI] [PubMed] [Google Scholar]
- Yu G. L., Katagiri F., Ausubel F. M. Arabidopsis mutations at the RPS2 locus result in loss of resistance to Pseudomonas syringae strains expressing the avirulence gene avrRpt2. Mol Plant Microbe Interact. 1993 Jul-Aug;6(4):434–443. doi: 10.1094/mpmi-6-434. [DOI] [PubMed] [Google Scholar]
- von Heijne G. The signal peptide. J Membr Biol. 1990 May;115(3):195–201. doi: 10.1007/BF01868635. [DOI] [PubMed] [Google Scholar]





