Abstract
Our understanding of the molecular mechanisms underlying the pharmacological actions of estrogen receptor (ER) ligands has evolved considerably in recent years. Much of this knowledge has come from a detailed dissection of the mechanism(s) of action of the Selective Estrogen Receptor Modulators (SERMs) tamoxifen and raloxifene, drugs whose estrogen receptor (ER) agonist/antagonist properties are influenced by the cell context in which they operate. These studies have revealed that notwithstanding differences in drug pharmokinetics, the activity of an ER ligand is determined primarily by (a) the impact that a given ligand has on the receptor conformation and (b) the ability of structurally distinct ER-ligand complexes to interact with functionally distinct coregulators. Exploitation of the established relationships between ER structure and activity has led to the development of improved SERMs with more favorable therapeutic properties and of tissue-selective estrogen complexes, drugs in which a SERM and an ER agonist are combined to yield a blended activity that results in distinct clinical profiles. Remarkably, endogenous ligands that exhibit SERM activity have also been identified. One of these ligands, 27-hydroxycholesterol (27HC), has been shown to manifest ER-dependent pathological activities in the cardiovascular system, bone and mammary gland. Whereas the physiological activity of 27HC remains to be determined, its discovery highlights how cells have adopted mechanisms to allow the same receptor ligand complex to manifest different activities in different cells, and also how these processes can be exploited for new drug development.
Introduction
The estrogen receptor (ER) is a well-validated therapeutic target that has been exploited in the development of drugs that are currently used as (a) treatments for the climacteric symptoms associated with menopause, (b) oral contraceptives, (c) fertility agents and (d) breast cancer therapeutics. Until relatively recently it was considered that the pharmacology of ER ligands was relatively simple in that classical agonists (steroidal or non-steroidal) phenocopied the actions of the potent agonist 17β-estradiol, while antagonists exerted their activity primarily through competitively inhibiting the binding of estrogens to their cognate receptors. Not surprisingly, therefore, the pharmaceutical development of most of the ER modulators currently used in the clinic was driven by the simple premise that, when corrected for affinity, all agonists were qualitatively the same and likewise antagonists differed only in their affinity for the receptor. Thus it was long considered that, other than enhancements to delivery and formulation, only minor improvements could be made to the therapeutic activity of ER modulators. This became a particular issue for hormone therapy (HT) in postmenopausal women, where a significantly increased risk of endometrial cancer was observed in women taking unopposed estrogens, an activity that was a property of all estrogens. This liability led to the incorporation of progestins in HT regimens administered to women with an intact uterus in order to prevent estrogen-induced endometrial hypertrophy. Unexpectedly, the inclusion of progestins in these medicines was associated with a whole new series of clinical problems, the significance of which was highlighted by the results of the Women’s Health Initiative (WHI) in 2002 in which a slight, but significant increase in the risk of invasive breast cancer was observed in women taking conjugated estrogens (CE) together with medroxyprogesterone acetate (MPA) [1]. Whereas efforts to develop ER ligands that functioned in a tissue selective manner preceded the WHI, the results of this trial reinvigorated efforts to exploit the complexities of the ER signal transduction pathway as a means to develop safe and effective medicines for HT. From these efforts emerged the third generation Selective Estrogen Receptor Modulators (SERMs) and more recently the Tissue Selective Estrogen Complexes (TSECs), drugs whose actions on ER are manifest in a cell-selective manner and which do not require the inclusion of a progestin. A discussion of how the development of these new drugs was influenced by an increased understanding of the molecular pharmacology of ER is the subject of this perspective.
The discovery of first and second generation SERMs
The SERM concept emerged from a series of preclinical clinical studies which revealed that the “antiestrogen” tamoxifen actually exhibited substantial ER agonist activity in bone and in the uterus [2–6]. Thus, while able to oppose estrogen action in the mammary gland, tamoxifen exhibited agonist activity in other tissues. The potential therapeutic utility of this tissue selective action was first highlighted by clinical studies that reported a significant increase in bone mineral density (BMD) in the lumbar spine of tamoxifen-treated breast cancer patients when compared to controls [7, 8]. These were followed by a very informative placebo controlled trial in which the bone sparing activity of tamoxifen in breast cancer patients was confirmed [9]. Together the clinical and preclinical pharmacology of tamoxifen provided strong evidence that it was possible to develop molecules whose ER agonist activity was manifest in a cell-selective manner. Indeed, were it not for the fact that tamoxifen exhibited significant uterotrophic activity in rodents and in humans, it may have been developed to treat and prevent osteoporosis [3]. Interestingly, a second “antiestrogen” keoxifene was also shown to protect against ovariectomy-induced bone loss in rodents [2]. This drug had originally been developed as a treatment for patients with tamoxifen-resistant metastatic breast cancer. Although the initial clinical trials in breast cancer were inconclusive, this drug did distinguish itself from tamoxifen in that it did not exhibit uterotrophic activity [10, 11]. The bone protective, uterine sparing, activities of keoxifene were confirmed in the MORE trial, and keoxifene (renamed raloxifene) was subsequently approved for the prevention and treatment of osteoporosis [12]. Interestingly, both tamoxifen and raloxifene were also approved for use as breast cancer chemopreventatives in women at elevated risk for breast cancer [13]. It was also clear from these results that it was inappropriate to classify tamoxifen and raloxifene as “antagonists”, and thus a new class label “SERMs” was proposed to reflect their complex pharmacology [14–16]. Several other SERMs, notably idoxifene and droloxifene, were evaluated at around the same time for activity as bone sparing agents in postmenopausal women. However, whereas all of these first/second generation SERMs were found to exhibit similar activities in bone, raloxifene alone functioned as a pure antagonist in the uterus. Raloxifene remains the only SERM mono-therapy registered in the US for the treatment and prevention of osteoporosis.
Unlike classical estrogens, the SERMs were ineffective in the treatment of vasomotor instability and dyspareunia, two of the primary reasons women seek HT. For this reason, it was unlikely, even considering their favorable profiles in breast, bone and uterus, that these early SERMs would find significant use in the pharmacotherapy of the symptomatology associated with menopause. However, it was generally considered that, since the SERM actions of tamoxifen and raloxifene (as well as toremifene, idoxifene and droloxifene among others) were discovered in a serendipitous manner, and further that their activities were optimized for antagonist activity in the breast, with additional exploration drugs with improved action in other tissues could be developed.
Molecular mechanisms of SERM action
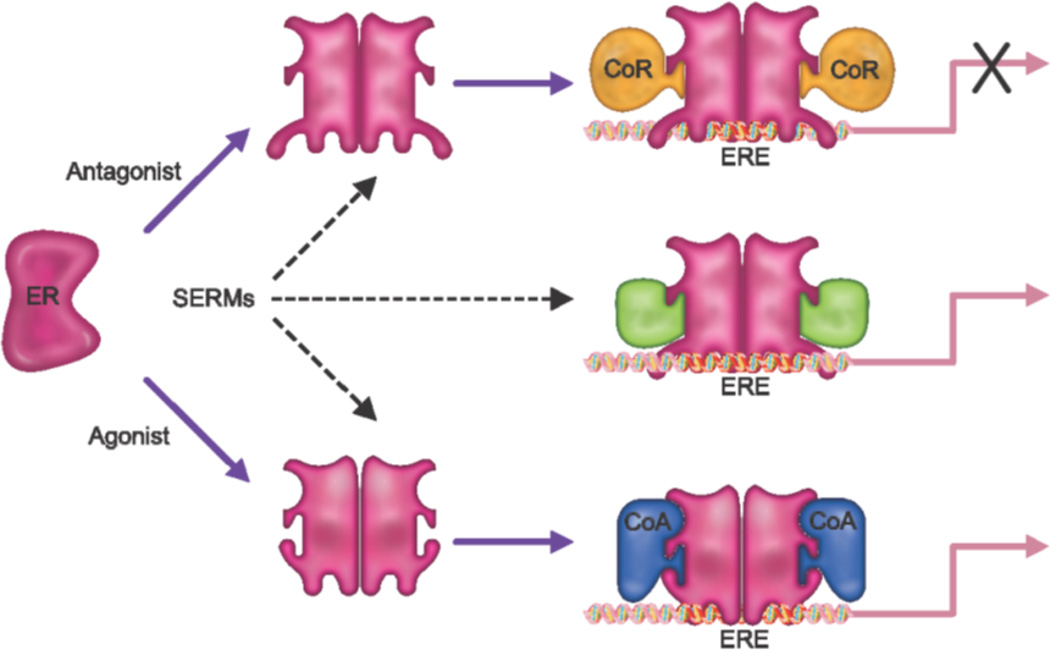
It was apparent even from the earliest studies that the pharmacology of SERMs was complex and that they were capable of exhibiting agonist, partial agonist or antagonist activities in different tissues [3, 17–19]. One of the key experiments that shed light on this complexity was performed by Gottardis and Jordan in the late 1980s in which they showed in xenograft models of breast cancer that over time, tamoxifen “switched” from an antagonist to an agonist [20]. The ability of serially-passaged tumors to recognize tamoxifen as an agonist indicated that resistance was a cell intrinsic process and suggested that dissection of the mechanisms underlying this activity would be informative with respect to ER pharmacology. The first evidence supporting a role for coregulators in NR pharmacology came from genetic studies in which disruption of a transcriptional corepressor switched tamoxifen from an antagonist to an agonist when assessed using a reconstituted ERα responsive transcription system in yeast [21]. Soon thereafter, Onate et al identified the first mammalian coregulator SRC-1, a protein which interacted directly with ERα (and other nuclear receptors) and increased its transcriptional activity [22]. It was subsequently shown that the relative agonist/antagonist activity of tamoxifen could be manipulated by increasing or decreasing the expression of SRC-1 within target cells [23]. This suggested that although tamoxifen induces a conformational change in ERα that dramatically reduces its ability to interact with coactivators, the impact of this disruptive conformational change can be overcome by increasing the cellular concentration of a specific coactivator. This, coupled with the fact that tamoxifen enables binding of the receptor to DNA and that it also increases ERα levels in cells, explains how this drug can induce significant activation of ER target genes [24]. It was further noted that elevated expression of SRC-1 and/or SRC-3 in breast tumors is associated with tamoxifen resistance and that the locus encoding SRC-3 is amplified in a large number of breast cancers [25–27]. However, even considering the role of coregulators, it remained unclear how the relative agonist/antagonist activities of different SERMs could be dramatically different within the same cell. The answer to this problem was revealed in studies which demonstrated that, contrary to the classical “binary on/off” models of ER action, the overall shape of the receptor was influenced by the nature of the ligand to which is was bound, and that this manifested in the differential presentation of protein-protein interaction surfaces on the receptor. Thus, as a consequence of their impact on ER structure, different ligands can facilitate the interaction of ER with different, functionally distinct, coregulators (Figure 1). To date over 300 coregulators have been identified although the functions of only a few have been explored in detail. Definition of the specific roles of individual coregulators in ER pharmacology will inform the development of screens for ligands that facilitate the interaction/disengagement of specific coregulators involved in processes of interest.
Figure 1. Estrogen receptor (ER) pharmacology.
Until relatively recently it was considered that the role of an ER-agonist was that of a “switch” which upon binding converted the receptor into an active state. Antagonists, on the other hand were believed to function by competitively inhibiting agonist binding thus freezing the receptor in an inactive state. However, it is now known that the overall conformation of ER is determined by the ligand to which it is bound, which in turn impacts the ability of the receptor to engage functionally different coregulators. Pure agonists enable the interaction of the receptor with coactivators (CoA), while antagonists allow the receptor to interact with only corepressors (CoR). Selective estrogen receptor modulators (SERMs) permit the bound ERs to interact with different subsets of coactivators and corepressors, determined by the overall receptor conformation associated with a given SERM, thereby permitting these drugs to elicit different activities in different tissues. Thus, the cellular response to an ER-agonist complex will be primarily determined by the relative expression level and activity of functionally distinct coregulators in different target tissues.
The transition to mechanism-based discovery of clinically useful SERMs
The first clinical experience with raloxifene (keoxifene) was an unsuccessful attempt to identify agents that could be used to treat tamoxifen-resistant breast cancer [28]. The primary rationale at the time for this approach was that resistance to tamoxifen was thought to occur either as a consequence of ERα mutations that disrupted tamoxifen binding or was due to the production of an estrogenic metabolite of the drug within tumors. Initially, the failure of keoxifene as a breast cancer therapeutic was thought to reflect its unfavorable pharmaceutical properties. However, we now know from an abundance of structural studies that the overall conformation of the ERα-tamoxifen and ERα-raloxifene complexes are similar, allowing them to potentially interact with the same cofactors [29–31]. Therefore, the cross-resistance observed in the clinic was not surprising, and neither was their similar efficacy in breast cancer prevention as noted in the Study of Tamoxifen and Raloxifene (STAR) trial [13, 32, 33]. It was inferred, however, from the studies of tamoxifen/raloxifene pharmacology that compounds that enabled ERα to adopt a distinctly different conformation, and which disrupted the receptor cofactor interactions enabled by these SERMs, may have utility in the treatment of tamoxifen refractory tumors. To test this hypothesis, we developed a series of in vitro screens to identify compounds that enabled ER to adopt a conformational state distinct from those induced by tamoxifen, raloxifene or estradiol [16, 34, 35]. In this manner, GW5638/DPC974 was identified, a compound that was subsequently shown to interfere with ERα action by directly disrupting the folding of the critical helix 12 in the ligand binding domain of the receptor, ultimately inducing proteasomal degradation of the receptor, an activity that more accurately classifies this molecule as a SERM-like selective estrogen receptor degrader (SERD) [36–38]. While the first in class SERD fulvestrant has been approved for the treatment of advanced breast cancer, the therapeutic utility of this drug is limited by its poor pharmaceutical properties [39–41]. Importantly, GW5638/DPC974 inhibited the growth of both tamoxifen-sensitive and -resistant tumor xenografts [42, 43]. Most notably, in a small investigator-initiated clinical trial of the drug there was evidence of efficacy in patients with heavily treated metastatic disease. GW5638/DPC974 and fulvestrant differ in the mechanism by which they induce ER degradation and also in their pharmacological profile, in that GW5638/DPC974 exhibited SERM-like properties in bone [38, 44]. Unfortunately, development of GW5638/DPC974 was discontinued, a casualty of consolidation in the industry, with the ultimate owner of the drug (BMS) unconvinced of the utility of ER as a therapeutic target in advanced breast cancer. Regardless, the work with this drug firmly established the concept that it was possible to manipulate ERα structure and identify compounds that could be used in the pharmacotherapy of tamoxifen-resistant ER-positive tumors. Building on this work, and exploiting the relationship between ER structure and activity, resulted in the identification of ARN-810 (Seragon Pharmaceuticals), a drug that is currently being evaluated in phase I trials in advanced breast cancer patients who have progressed during treatment with tamoxifen or aromatase inhibitors [45]. Although not yet evaluated in breast cancer patients, pre-clinical data also suggest that more recently developed SERMs, such as bazedoxifene, that induce unique structural changes in ERα will also be effective in the treatment of tamoxifen refractory breast cancers [46, 47]. The latter drug has been approved for the prevention of osteoporosis in many countries outside of the US, and considering its favorable activity in preclinical models of tamoxifen resistant breast cancer, it could see near term “off-label” use as a breast cancer treatment.
Third generation SERMs
Definition of the molecular mechanisms underlying the pharmacological activities of the second generation SERMs revealed that the pharmacology of a given ER modulator is determined by (a) the effect of the bound ligand on ER structure and how this modulates the engagement of functionally distinct coregulators, (b) the impact of signaling pathways on the activity of different ER-coregulator complexes and, (c) the nucleotide sequence of the ERE within the target gene and the transcription factor landscape close to the DNA bound receptor. Additional complexity is introduced when one considers that there exist two genetically distinct estrogen receptors, ERα and ERβ, which are unlikely to interact with SERMs in the same manner [48, 49]. Both isoforms have distinct activities when acting as homodimers or can modulate each other’s activity through heterodimerization in cells where they are both expressed [50, 51]. The relevance of the interplay between ERα and ERβ noted in vitro is unclear and significant progress in resolving this issue has been impeded by the lack of consensus as to the target tissues/physiological processes in which ERβ is engaged. There are additional factors that influence ER pharmacology but those outlined have endured as the most important [52–56]. As these rules evolved, they were exploited in the development of mechanism-based screens for SERMs with unique activities, studies that were driven largely by efforts to develop compounds that exhibited the activity of tamoxifen in bone but which exhibited a more favorable profile in the uterus.
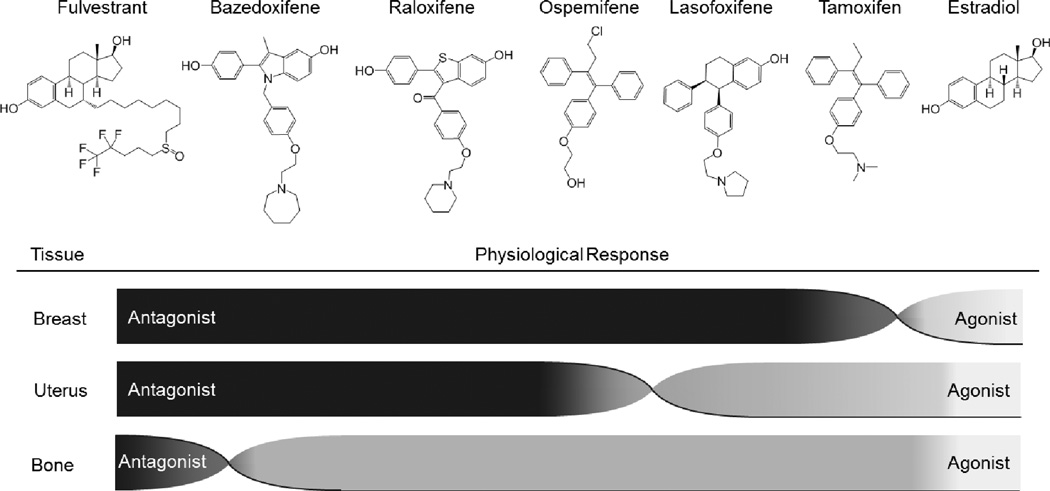
The first mechanism based screens for SERMs were established with the goal of identifying compounds whose pharmacological activities were similar to raloxifene but which had improved pharmaceutical properties. Arzoxifene, lasofoxifene and bazedoxifene, emerged from these studies with the latter two drugs being discovered utilizing a series of in vitro molecular assays similar to those described above [16]. In contrast, ospemifene was identified as a metabolite of the second generation SERM toremifene. These clinically relevant SERMs exhibit a spectrum of activities that have been exploited to address different clinical needs (Figure 2). Given their relatively recent appearance, a brief description of their attributes is warranted.
Figure 2. SERMs exert a spectrum of activities in estrogen target tissues.
Like estradiol, all selective estrogen receptor modulators (SERMs) described to date increase and/or maintain bone mineral density (BMD), although SERMs do not elicit as robust of response as do full agonists. Fulvestrant is associated with decreased BMD. Similarly, the antagonist activities of different SERMs in the breast are not equivalent. Stimulation of uterine hypertrophy is the primary response that distinguishes SERMs, a liability of tamoxifen that has been reduced in second and third generation SERMs. It is the uterine response that most clearly determines the safety profile of a given SERM.
Lasofoxifene
Lasofoxifene (Pfizer, Inc.) was initially identified as a SERM able to elicit an ER conformation similar to that induced by raloxifene. Clinical trials have demonstrated that lasofoxifene significantly increased BMD and decreased levels of bone turnover markers, as compared to placebo, while also being associated with a favorable lipid profile [57]. The Postmenopausal Evaluation and Risk-reduction with Lasofoxifene (PEARL) study revealed that not only did lasofoxifene reduce vertebral fracture risk, but that both doses of the drug evaluated were also associated with decreased risk of ER-positive breast cancer as compared to placebo [58, 59]. Counterposing the favorable effects of lasofoxifene on bone quality, lipid profile and breast cancer risk was the observation of clinically important increases in endometrial thickness and a higher incidence of endometrial polyps, uterine leiomyoma, and vaginal bleeding [60]. Thus, this SERM lacks the favorable uterine profile observed for raloxifene. Lasofoxifene received approval from the European Medical Association in 2009 for the treatment of postmenopausal women at increased risk of fracture; however, the drug was not launched and its marketing authorization has since lapsed. Although lasofoxifene was also shown to decrease vaginal atrophy, it did not receive approval for this indication.
Bazedoxifene
Bazedoxifene (Pfizer, Inc.) has been evaluated in several clinical trials for the prevention and treatment of postmenopausal osteoporosis. In all of these studies, bazedoxifene treatment was associated with improved BMD, reduced levels of bone turnover markers, and favorable effects on lipid profiles. Similar increases in BMD and reduction of vertebral fractures were observed with bazedoxifene or raloxifene treatment of postmenopausal women at risk for or diagnosed with osteoporosis [61, 62]. Furthermore, bazedoxifene was not associated with stimulation of endometrial or breast tissues [63–65]. In 2009, bazedoxifene was approved in Europe and Japan for the treatment of postmenopausal women at risk for, or presenting with, osteoporosis. This drug, although receiving an approvable letter from the FDA, has not been registered as a monotherapy for osteoporosis in the United States.
Arzoxifene
Similar to bazedoxifene and lasofoxifene, arzoxifene (Eli Lilly & Co.) was identified as a SERM lacking ER agonist activity in breast and endometrial tissues while exhibiting agonist activity in bone and preserving a positive lipid profile. Phase 2 and 3 studies of arzoxifene demonstrated reduced bone turnover and increased lumbar spine BMD as compared to placebo [66, 67]. Similar to bazedoxifene, arzoxifene was not associated with increased endometrial thickness, but neither was it associated with a difference in the incidence of non-vertebral fractures as compared to placebo, and in fact a higher incidence of adverse events was observed for arzoxifene (as compared to placebo) [66, 67]. Although the manufacturer has elected to discontinue development of arzoxifene, a 56% decrease in the risk of invasive breast cancer as compared to placebo, as well as demonstrated efficacy in the treatment of recurring or advanced endometrial cancer, illustrates the utility of developing additional SERMs for use as cancer therapeutics [68, 69].
Ospemifene
Ospemifene (Shionogi & Co.) was identified as a weakly estrogenic metabolite of toremifene. Although toremifene displays the same pharmacodynamic liabilities (endometrial stimulation) as tamoxifen, clinical trials of ospemifene have reported only a weak uterine response resulting in a detectible increase in endometrial lining without hypertrophy in some patients, comparable to raloxifene [70, 71]. Ospemifene was approved in the United States in 2013 for the treatment of dyspareunia associated with vulvar and vaginal atrophy in postmenopausal women [72]. Preclinical data and phase 2 trials would suggest that, as with a majority of SERMs to date, ospemifene is bone protective [73–75]. As with raloxifene, however, an increased incidence of vasomotor symptoms was observed for ospemifene [71].
Tissue Selective Estrogen Complexes (TSECs)
The early termination of the WHI study due to an increased incidence of invasive breast cancer in the CE/MPA arm led to a dramatic reduction (90% between 2002–2007) in the use of estrogen containing medicines for HT [76]. Interestingly, a significant reduction in breast cancer incidence was noted in the CE only arm of the WHI, and recent follow-up studies have indicated that this protective activity has endured [77]. Regardless, HT use by postmenopausal women remains low despite its clear overall benefit beyond its ability to treat vasomotor symptoms [78, 79]. In the last decade, multiple studies have compared medicines containing different estrogens and progestins with respect to safety and efficacy. One general conclusion from these studies is that it appears to be the progestin component that underlies the increased breast cancer risk associated with HT [80, 81].
The primary indication prompting HT use is vasomotor symptoms associated with the cessation of ovarian estrogen production. Less obvious, but clinically more important, is the decrease in BMD and alterations in lipid profile that occur with estrogen deprivation, which respectively underlie the increased risk of osteoporosis and fracture and of cardiovascular events in postmenopausal women. SERMs have nearly universally been found to increase BMD and generate a favorable lipid profile (Figure 2). However, while certain SERMs such as ospemifene may alleviate some menopausal symptoms, efforts to identify a uterine sparing SERM that is also able to reduce vasomotor symptoms have been unsuccessful. Thus, there remains an unmet medical need for pharmaceutical approaches to mitigate the impact of estrogen deprivation in women with an intact uterus following the loss of ovarian function. An interesting new approach that has emerged recently is the Tissue Selective Estrogen Complexes (TSECs), medicines that combine an estrogen and SERM to elicit a blend of ER agonist activities. To date, two different TSEC combinations have been described (17β-estradiol/raloxifene and CE/bazedoxifene). Increased endometrial thickness and endometrial hyperplasia was observed in patients treated with a TSEC comprised of raloxifene and estradiol, apparently limiting its clinical utility [82, 83]. In contrast, the CE/bazedoxifene TSEC has been extensively evaluated in the clinic, was found to have an overall favorable safety profile, and was recently approved in the US for the treatment of menopausal symptoms. In a trial of over 3000 patients, it was observed that CE/bazedoxifene exhibited activities in the endometrium that were similar to placebo while vasomotor symptoms were reduced by as much as 86% [84]. This is an exceptionally important finding as it makes obsolete the need for a progestin in HT administered to postmenopausal women without hysterectomy. While a direct comparison of the two TSEC formulations has not been performed, it is clear that a) TSECs are likely to exhibit different clinical activities, and b) the overall activity of the combination is greatly dependent on the ratio and activities of the constituent components. The apparent clinical success of CE/bazedoxifene, as compared to raloxifene/estradiol, may relate to differences in:
antagonist/agonist profile of the constituent SERM– Co-administration of bazedoxifene with raloxifene in ovariectomized rats demonstrated that bazedoxifene reversed the small uterine stimulation observed for raloxifene [85].
mechanism of SERM action – Bazedoxifene treatment results in proteasomal degradation of ERα in breast cancer cells whereas raloxifene has little effect [46, 47].
estrogenic constituents – Whereas other estrogen formulations have been associated with increased breast cancer risk [80, 81], a 10-year follow up analysis of the CE only arm of the WHI revealed a significant reduction in breast cancer risk [77].
Whereas TSECs manifest a very useful clinical profile, their mechanism of action and the functional differences between different SERM/estrogen combinations remain to be determined. In the case of CE/bazedoxifene, evidence would suggest that, whereas the CNS is clearly responding to the estrogen component as evidenced by decreased vasomotor symptoms, the activities of the SERM appear to predominate in the periphery. Further, while CE treatment alone was associated with increased breast density in the WHI, neither bazedoxifene nor CE/bazedoxifene administration resulted in significant changes in breast density [65, 86, 87]. Similarly, the BMD improvement observed for CE/bazedoxifene resembles that associated with bazedoxifene therapy alone and is less robust than that observed in patients treated with CE/MPA [88]. Thus, it appears that the activities of the SERM predominate in the periphery, while the CNS receives sufficient estrogen exposure to alleviate symptoms of estrogen deprivation. The lack of breast stimulation and uterine quiescence that is observed for CE/bazedoxifene has led to the suggestion that this formulation may present a viable HT option for breast cancer survivors and also a potentially useful treatment for other estrogenopathies. Notably, in preclinical models of endometriosis CE/bazedoxifene treatment leads to regression of lesions, suggesting that CE/bazedoxifene may have utility for the treatment of endometriosis in patients [89].
One of the major impediments to understanding the existing TSECs is information regarding the occupancy of the ERs in various tissues. Further complicating this picture is the fact that in some cells both ERα and ERβ are expressed. Clearly, in conditions where drug is not saturating it is reasonable to believe that mixed heterodimers of the same receptor, one monomer occupied by an estrogen and the other by a SERM, could exist and that their biological output would be different than homodimers of a receptor occupied by the same ligand. Whereas evidence in support of this latter hypothesis has been established in vitro, it is unlikely, especially for CE/bazedoxifene, that this mechanism will be as important as it would be expected; given the affinity of bazedoxifene (and the drug formulation), it will likely fully occupy the receptor. This likely explains why the profile of the CE/bazedoxifene TSEC is very similar to bazedoxifene alone in most tissues. There are data that SERMs vary in their ability to penetrate the blood brain barrier, and thus it might be expected that the ability of the TSECs to treat hot flashes may be the result of favorable pharmacokinetics, where the estrogen has more efficient entry and/or privileged access to the brain. Regardless, the TSECs represent the first innovation in the pharmacotherapy of the climacteric in nearly 50 years and they are likely to have a significant impact on the treatment of the symptomatology of the climacteric.
Oxysterols as Endogenous SERMs
The complex pharmacology of synthetic SERMs suggested the possibility that endogenous molecules may exist that possess SERM-like activities. The first evidence for such an activity came from studies by Umetani et al. aimed at understanding why estrogens were cardioprotective in experimental models but were not associated with benefit in the WHI [90]. They noted that 27-hydroxycholesterol (27HC), the most abundant oxysterol, was able to inhibit ERα- and ERβ-dependent transcriptional activity at concentrations that could reasonably be expected to exist in humans [91, 92]. These data, and the observation that 27HC induces a distinct conformational change in ER, led to the hypothesis that this oxysterol may have unique, ER-dependent biological activity [93]. Indeed, using a series of cell-based assays several investigators have determined that, like other SERMs, 27HC can function as either an agonist or antagonist in a manner that is influenced by cell context. These data confirmed that 27HC was a bona fide SERM, a finding that justifies studies directed at defining potential ER-dependent (patho)physiological actions of 27HC in estrogen responsive target organs.
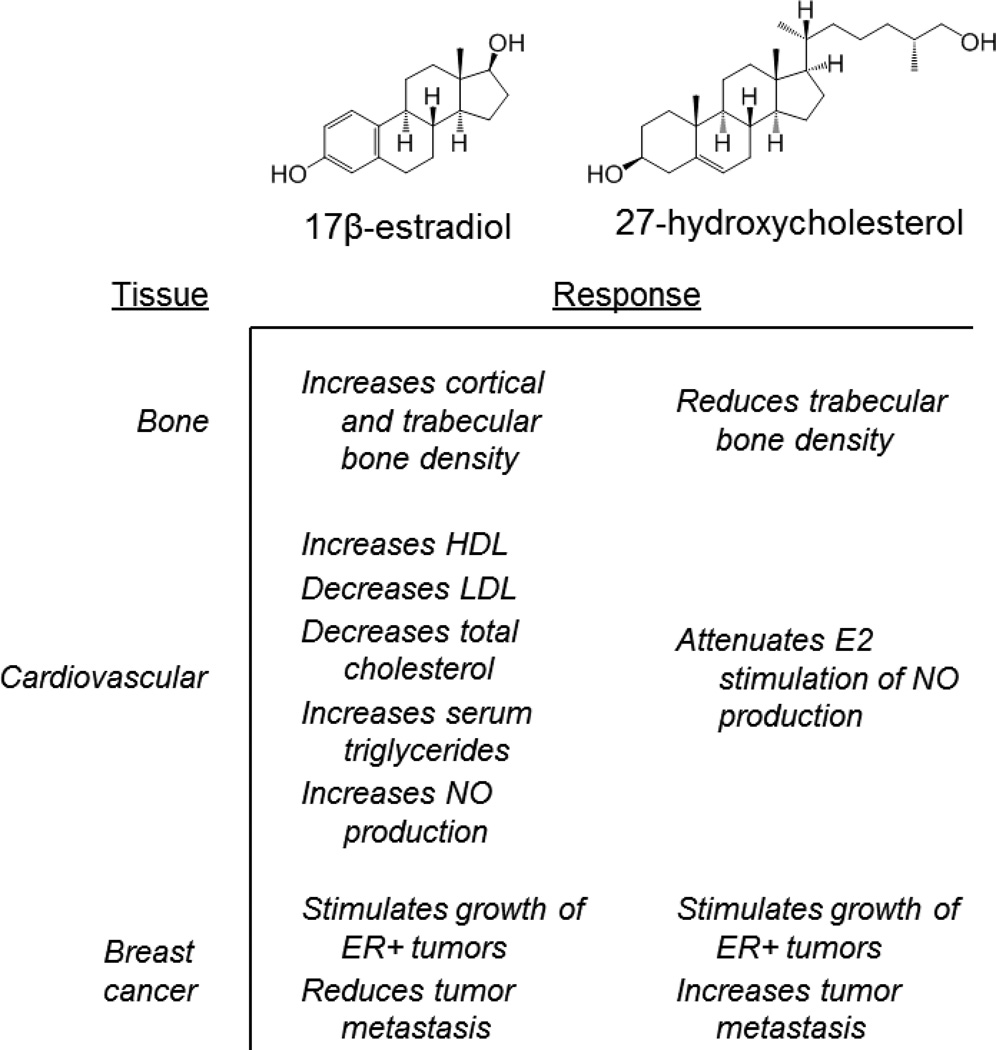
In mouse models of the cardiovascular system, 27HC was shown to attenuate the protective effects of 17β-estradiol on vascular repair (re-endothelialization) by inhibiting ER-dependent production of nitric oxide [90, 94]. Based on these data it was proposed that when 27HC levels are increased, such as in patients with elevated cholesterol, the cardio-protective effects of estradiol are likely to be diminished by direct competitive antagonism of estradiol action on ER by 27HC. Additional studies demonstrated that 27HC attenuated estrogen action in bone. Elevation of 27HC, either pharmacologically (by injection), or by genetically disrupting the CYP7B1 locus (the enzyme responsible for the catabolism of 27HC), results in significantly decreased trabecular and cortical bone [95]. Importantly, in the CYP7B1−/− mouse model the effects of 27HC on bone were partially reversed by administering pharmacological doses of 17β-estradiol, a result that implicates ER as one target of the pathological actions of 27HC. In these 28-day studies it was also observed that while ovariectomy alone had minimal effects on cortical bone in wild type mice, a dramatic loss of bone was observed in ovariectomized mice in which 27HC levels were elevated. By extrapolation, this latter finding suggests that post-menopausal women with elevated cholesterol, and by inference 27HC, may be at increased risk for cortical fractures. Hypercholesterolemia is in fact an established risk factor for osteoporosis in post-menopausal women [96–98]. As a primary cholesterol metabolite, systemic 27HC levels mirror those of circulating cholesterol and are increased in obese individuals [99]. Not surprisingly therefore, the use of 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors (statins), is associated with decreased fracture risk and increased BMD [100, 101].
As opposed to its ER-antagonist action in the cardiovascular system and bone, it has been shown in breast cancer cells that 27HC functions as a partial agonist, inducing the expression of ER target genes and stimulating cell proliferation [93]. It has also been shown in relevant animal models that 27HC increases the growth and metastasis of ER-positive breast cancer tumors [102, 103]. CYP27A1, the enzyme responsible for the conversion of cholesterol to 27HC is highly expressed in macrophages. Thus, given the relationship between increased macrophage number and negative outcome in breast cancer it was of interest that the levels of 27HC found in breast tumors were significantly higher than in normal breast tissue or uninvolved tissue adjacent to tumors [103]. Further, expression of CYP27A1 in breast cancer tissue was associated with higher grade tumors, while high expression of the enzyme responsible for 27HC catabolism, CYP7B1, was associated with a better prognosis [102, 103]. Collectively, these findings have led to the conclusion that 27HC, is an endogenous SERM that functions as a biochemical link between obesity and breast cancer (Figure 3).
Figure 3.
Comparison of the pharmacological activities of 17β-estradiol and the endogenous selective estrogen receptor modulator 27-hydroxycholesterol (27HC) in estrogen target tissues
Final comments
Despite the fact that the first SERMs (then considered non-steroidal antiestrogens) were identified over 60 years ago and have been in the clinic for nearly 35 years, it is only relatively recently that the molecular basis for their tissue selective actions has become apparent. In the past 25 years, our understanding of the pharmacological profile of these molecules has evolved from the initial recognition of their tissue selective actions, and these studies have informed contemporary mechanism-based approaches to identify SERMs that exhibit specific profiles [3, 14, 15, 104, 105]. Of particular importance in this regard is the observation that the overall shape of ER is influenced by a) the nature of the ligand with which it interacts and b) the impact which this has on the differential presentation of protein-protein interaction surfaces on the receptor. This has provided information as to the mechanism(s) by which information, contained within chemically distinct ligands, is interpreted by the receptor. The further recognition that differently conformed ER-ligand complexes exhibit unique coregulator interaction preferences describes how cells/target genes ultimately discriminate between different ER ligands. Understanding the specific roles of different coregulators in physiological and pathological processes of interest and definition of the processes that converge upon and regulate the activity of different ER-coregulator complexes is a key next step in the development of SERMs/ER ligands with improved functional selectivity.
Acknowledgments
Some of the research discussed in this paper was supported by an NIH grant R37 DK048807.
SEW and DPM have served as ad-hoc consultants for Pfizer, Inc.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Rossouw JE, Anderson GL, Prentice R, LaCroix AZ, Kooperberg C, Stefanick ML, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women's Health Initiative randomized controlled trial. JAMA. 2002;288:321–333. doi: 10.1001/jama.288.3.321. [DOI] [PubMed] [Google Scholar]
- 2.Jordan VC, Phelps E, Lindgren JU. Effects of anti-estrogens in castrated and intact female rats. Breast Cancer Res Treat. 1987;10:31–35. doi: 10.1007/BF01806132. [DOI] [PubMed] [Google Scholar]
- 3.Gottardis MM, Robinson SP, Satyaswaroop PG, Jordan VC. Contrasting actions of tamoxifen on endometrial and breast tumor growth in the athymic mouse. Cancer Res. 1988;48:812–815. [PubMed] [Google Scholar]
- 4.Ferrazzi E, Cartei G, Mattarazzo R, Fiorentino M. Oestrogen-like effect of tamoxifen on vaginal epithelium. Br Med J. 1977;1:1351–1352. doi: 10.1136/bmj.1.6072.1351-e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Black LJ, Goode RL. Uterine bioassay of tamoxifen, trioxifene and a new estrogen antagonist (LY117018) in rats and mice. Life Sci. 1980;26:1453–1458. doi: 10.1016/0024-3205(80)90049-1. [DOI] [PubMed] [Google Scholar]
- 6.Kurl RN, Borthwick NM. Clomiphene and tamoxifen action in the rat uterus. J Endocr. 1980;85:519–524. doi: 10.1677/joe.0.0850519. [DOI] [PubMed] [Google Scholar]
- 7.Gotfredsen A, Christiansen C, Palshof T. The effect of tamoxifen on bone mineral content in premenopausal women with breast cancer. Cancer. 1984;53:853–857. doi: 10.1002/1097-0142(19840215)53:4<853::aid-cncr2820530406>3.0.co;2-m. [DOI] [PubMed] [Google Scholar]
- 8.Turken S, Siris E, Seldin D, Flaster E, Hyman G, Lindsay R. Effects of tamoxifen on spinal bone density in women with breast cancer. J Natl Cancer Inst. 1989;81:1086–1088. doi: 10.1093/jnci/81.14.1086. [DOI] [PubMed] [Google Scholar]
- 9.Love RR, Mazess RB, Barden HS, Epstein S, Newcomb PA, Jordan VC, et al. Effects of tamoxifen on bone mineral density in postmenopausal women with breast cancer. New Engl J Med. 1992;326:852–856. doi: 10.1056/NEJM199203263261302. [DOI] [PubMed] [Google Scholar]
- 10.Black LJ, Sato M, Rowley ER, Magee DE, Bekele A, Williams DC, et al. Raloxifene (LY139481 HCI) prevents bone loss and reduces serum cholesterol without causing uterine hypertrophy in ovariectomized rats. J Clin Invest. 1994;93:63–69. doi: 10.1172/JCI116985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Buzdar AU, Marcus C, Holmes F, Hug V, Hortobagyi G. Phase II evaluation of Ly156758 in metastatic breast cancer. Oncology. 1988;45:344–345. doi: 10.1159/000226637. [DOI] [PubMed] [Google Scholar]
- 12.Ettinger B, Black DM, Mitlak BH, Knickerbocker RK, Nickelsen T, Genant HK, et al. Reduction of vertebral risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. Multiple Outcomes of Raloxifene Evaluation (MORE) Investigators. JAMA. 1999;282:637–645. doi: 10.1001/jama.282.7.637. [DOI] [PubMed] [Google Scholar]
- 13.Vogel VG, Costantino JP, Wickerham DL, Cronin WM, Cecchini RS, Atkins JN, et al. Update of the National Surgical Adjuvant Breast and Bowel Project Study of Tamoxifen and Raloxifene (STAR) P-2 trial: Preventing breast cancer. Cancer Prev Res (Phila Pa) 2010;3:696–706. doi: 10.1158/1940-6207.CAPR-10-0076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Miller CP. SERMs: evolutionary chemistry, revolutionary biology. Curr Pharm Des. 2002;8:2089–2111. doi: 10.2174/1381612023393404. [DOI] [PubMed] [Google Scholar]
- 15.Miller CP, Collini MD, Tran BD, Harris HA, Kharode YP, Marzolf JT, et al. Design, synthesis, and preclinical characterization of novel, highly selective indole estrogens. J Med Chem. 2001;44:1654–1657. doi: 10.1021/jm010086m. [DOI] [PubMed] [Google Scholar]
- 16.McDonnell DP, Clemm DL, Hermann T, Goldman ME, Pike JW. Analysis of estrogen receptor function in vitro reveals three distinct classes of antiestrogens. Mol Endocrinol. 1995;9:659–668. doi: 10.1210/mend.9.6.8592512. [DOI] [PubMed] [Google Scholar]
- 17.Harper MJK, Walpole AL. Contrasting endocrine activities of cis and trans isomers in a series of substituted triphenylethylenes. Nature. 1966;212:87–89. doi: 10.1038/212087a0. [DOI] [PubMed] [Google Scholar]
- 18.Turner CH, Sato M, Bryant HU. Raloxifene preserves bone strength and bone mass in ovariectomized rats. Endocrinology. 1994;135:2001–2005. doi: 10.1210/endo.135.5.7956922. [DOI] [PubMed] [Google Scholar]
- 19.Turner RT, Wakley GK, Hannon KS, Bell NH. Tamoxifen prevents the skeletal effects of ovarian hormone deficiency in rats. J Bone Miner Res. 1987;2:449–456. doi: 10.1002/jbmr.5650020513. [DOI] [PubMed] [Google Scholar]
- 20.Gottardis MM, Jordan VC. Development of tamoxifen-stimulated growth of MCF-7 tumors in athymic mice after long-term antiestrogen administration. Cancer Res. 1988;48:5183–5187. [PubMed] [Google Scholar]
- 21.McDonnell DP, Vegeto E, O'Malley BW. Identification of a negative regulatory function for steroid receptors. Proc Natl Acad Sci USA. 1992;89:10563–10567. doi: 10.1073/pnas.89.22.10563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Onate SA, Tsai S, Tsai M-J, O'Malley BW. Sequence and characterization of a coactivator for the steroid hormone receptor superfamily. Science. 1995;270:1354–1357. doi: 10.1126/science.270.5240.1354. [DOI] [PubMed] [Google Scholar]
- 23.Smith CL, Nawaz Z, O'Malley BW. Coactivator and corepressor regulation of the agonist/antagonist activity of the mixed antiestrogen, 4-hydroxytamoxifen. Mol Endocrinol. 1997;11:657–666. doi: 10.1210/mend.11.6.0009. [DOI] [PubMed] [Google Scholar]
- 24.Wijayaratne AL, Nagel SC, Paige LA, Christensen DJ, Norris JD, Fowlkes DM, et al. Comparative analyses of the mechanistic differences among antiestrogens. Endocrinology. 1999;140:5828–5840. doi: 10.1210/endo.140.12.7164. [DOI] [PubMed] [Google Scholar]
- 25.Anzick SL, Kononen J, Walker RL, Azorsa DO, Tanner MM, Guan X-Y, et al. AIB1, a steroid receptor coactivator amplified in breast and ovarian cancer. Science. 1997;277:965–968. doi: 10.1126/science.277.5328.965. [DOI] [PubMed] [Google Scholar]
- 26.Osborne CK, Bardou V, Hopp TA, Chamness GC, Hilsenbeck SG, Fuqua SAW, et al. Role of the estrogen receptor coactivator AIB1 (SRC-3) and HER-2/neu in tamoxifen resistance in breast cancer. J Natl Cancer Inst. 2003;95:353–361. doi: 10.1093/jnci/95.5.353. [DOI] [PubMed] [Google Scholar]
- 27.Redmond AM, Bane FT, Stafford AT, McIlroy M, Dillon MF, Crotty TB, et al. Coassociation of estrogen receptor and p160 proteins predicts resistance to endocrine treatment: SRC-1 is an independent predictor of breast cancer recurrence. Clin Cancer Res. 2009;15:2098–2106. doi: 10.1158/1078-0432.CCR-08-1649. [DOI] [PubMed] [Google Scholar]
- 28.Buzdar AU, Marcus C, Holmes F, Hug V, Hortobachi G. Phase II evaluation of LY156758 in metastatic breast cancer. Oncology. 1988;45:144–145. doi: 10.1159/000226637. [DOI] [PubMed] [Google Scholar]
- 29.Norris JD, Paige LA, Christensen DJ, Chang C-Y, Huacani MR, Fan D, et al. Peptide antagonists of the human estrogen receptor. Science. 1999;285:744–746. doi: 10.1126/science.285.5428.744. [DOI] [PubMed] [Google Scholar]
- 30.Brzozowski AM, Pike AC, Dauter Z, Hubbard RE, Bonn T, Engstrom O, et al. Molecular basis of agonism and antagonism in the oestrogen receptor. Nature. 1997;389:753–758. doi: 10.1038/39645. [DOI] [PubMed] [Google Scholar]
- 31.Shiau AK, Barstad D, Loria PM, Cheng L, Kushner PJ, Agard DA, et al. The structural basis of estrogen receptor/coactivator recognition and the antagonism of this interaction by tamoxifen. Cell. 1998;95:927–937. doi: 10.1016/s0092-8674(00)81717-1. [DOI] [PubMed] [Google Scholar]
- 32.Vogel VG. The NSABP Study of Tamoxifen and Raloxifene (STAR) trial. Expert Rev Anticancer Ther. 2009;9:51–60. doi: 10.1586/14737140.9.1.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Vogel VG, Costantino JP, Wickerham DL, Cronin WM, Cecchini RS, Atkins JN, et al. Effects of tamoxifen vs raloxifene on the risk of developing invasive breast cancer and other disease outcomes. JAMA. 2007;295:2727–2741. doi: 10.1001/jama.295.23.joc60074. [DOI] [PubMed] [Google Scholar]
- 34.Chang C-Y, Norris JD, Gron H, Paige LA, Hamilton PT, Kenan DJ, et al. Dissection of the LXXLL nuclear receptor-coactivator interaction motif using combinatorial peptide libraries: Discovery of peptide antagonists of estrogen receptors αand β. Mol Cell Biol. 1999;19:8226–8239. doi: 10.1128/mcb.19.12.8226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Norris JD, Paige LA, Christensen DJ, Chang CY, Huacani MR, Fan D, et al. Peptide antagonists of the human estrogen receptor. Science. 1999;285:744–746. doi: 10.1126/science.285.5428.744. [DOI] [PubMed] [Google Scholar]
- 36.Wu Y-L, Yang X, Ren Z, McDonnell DP, Norris JD, Willson TM, et al. Structural basis for an unexpected mode of SERM-mediated ER antagonism. Mol Cell. 2005;18:413–424. doi: 10.1016/j.molcel.2005.04.014. [DOI] [PubMed] [Google Scholar]
- 37.Connor CE, Norris JD, Broadwater G, Willson TM, Gottardis MM, Dewhirst MW, et al. Circumventing tamoxifen resistance in breast cancers using antiestrogens that induce unique conformational changes in the estrogen receptor. Cancer Res. 2001;61:2917–2922. [PubMed] [Google Scholar]
- 38.Wittmann B, Sherk A, McDonnell D. Definition of functionally important mechanistic differences among selective estrogen receptor down-regulators. Cancer Res. 2007;67:9549–9560. doi: 10.1158/0008-5472.CAN-07-1590. [DOI] [PubMed] [Google Scholar]
- 39.Chia S, Gradishar W, Mauriac L, Bines J, Amant F, Federico M, et al. Double-blind, randomized placebo controlled trial of Fulvestrant compared with exemestane after prior nonsteroidal aromatase inhibitor therapy in postmenopausal women with hormone receptorpositive, advanced breast cancer: results from EFECT. J Clin Oncol. 2008;26:1664–1670. doi: 10.1200/JCO.2007.13.5822. [DOI] [PubMed] [Google Scholar]
- 40.Di Leo A, Jerusalem G, Petruzelka L, Torres R, Bondarenko I, Khasanov R, et al. Results of the CONFIRM Phase III trial comparing fulvestrant 250 mg with fulvestrant 500 mg in postmenopausal women with estrogen receptor positive advanced breast cancer. J Clin Oncol. 2010;28 doi: 10.1200/JCO.2010.28.8415. [DOI] [PubMed] [Google Scholar]
- 41.Leo AD, Jerusalem G, Petruzelka L, Torres R, Bondarenko I, Khasanov R, et al. CONFIRM: A Phase III, randomized, parallel-group trial comparing fulvestrant 250 mg vs fulvestrant 500 mg in postmenopausal women with estrogen receptor-positive advanced breast cancer. Cancer Res. 2009;69 doi: 10.1200/JCO.2010.28.8415. Abstract nr 25. [DOI] [PubMed] [Google Scholar]
- 42.Connor CE, Norris JD, Broadwater G, Willson TM, Gottardis MM, Dewhirst MW, et al. Circumventing tamoxifen resistance in breast cancers using antiestrogens that induce unique conformational changes in the estrogen receptor. Cancer Res. 2001;61:2917–2922. [PubMed] [Google Scholar]
- 43.Dardes RC, O'Regan RM, Gajdos C, Robinson SP, Bentrem D, Reyes Adl, et al. Effects of a new clinically relevant antiestrogen (GW5638) related to tamoxifen on breast and endometrial cancer growth in vivo. Clin Cancer Res. 2002;8:1995–2001. [PubMed] [Google Scholar]
- 44.Willson TM, Norris JD, Wagner BL, Asplin I, Baer P, Brown HR, et al. Dissection of the molecular mechanism of action of GW5638, a novel estrogen receptor ligand, provides insights into the role of ER in bone. Endocrinology. 1997;138:3901–3911. doi: 10.1210/endo.138.9.5358. [DOI] [PubMed] [Google Scholar]
- 45.Mayer I, Bardia A, Dickler M, Manning H, Mahmood U, Ulaner G, et al. Abstract OT3-2-07: Phase I study of ARN-810, a novel selective estrogen receptor degrader, in post-menopausal women with locally advanced or metastatic estrogen receptor positive breast cancer. Cancer Res. 2013;73 Abstract nr OT3-2-07. [Google Scholar]
- 46.Wardell SE, Nelson ER, Chao CA, McDonnell DP. Bazedoxifene exhibits antiestrogenic activity in animal models of tamoxifen-resistant breast cancer: implications for treatment of advanced disease. Clin Cancer Res. 2013;19:2420–2431. doi: 10.1158/1078-0432.CCR-12-3771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lewis-Wambi J, Kim H, Curpan R, Grigg R, Sarker M, Jordan V. The Selective Estrogen Receptor Modulator Bazedoxifene Inhibits Hormone-Independent Breast Cancer Cell Growth and Downregulates Estrogen Receptor {alpha} and Cyclin D1. Mol Pharmacol. 2011 doi: 10.1124/mol.111.072249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ramsey TL, Risinger KE, Jernigan SC, Mattingly KA, Klinge CM. Estrogen receptor beta isoforms exhibit differences in ligand-activated transcriptional activity in an estrogen response element sequence-dependent manner. Endocrinology. 2004;145:149–160. doi: 10.1210/en.2003-1043. [DOI] [PubMed] [Google Scholar]
- 49.Barkhem T, Carlsson B, Nilsson Y, Enmark E, Gustafsson J, Nilsson S. Differential response of estrogen receptor alpha and estrogen receptor beta to partial estrogen agonists/antagonists. Mol Pharmacol. 1998;54:105–112. doi: 10.1124/mol.54.1.105. [DOI] [PubMed] [Google Scholar]
- 50.Kuiper GGJM, Carlsson B, Grandien K, Enmark E, Häggblad J, Nilsson S, et al. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors αand β. Endocrinology. 1997;138:863–870. doi: 10.1210/endo.138.3.4979. [DOI] [PubMed] [Google Scholar]
- 51.Hall JM, McDonnell DP. The estrogen receptor β-isoform (ERβ) of the human estrogen receptor modulates ERα transcriptional activity and is a key regulator of the cellular response to estrogens and antiestrogens. Endocrinology. 1999;140:5566–5578. doi: 10.1210/endo.140.12.7179. [DOI] [PubMed] [Google Scholar]
- 52.Bruning JB, Parent AA, Gil G, Zhao M, Nowak J, Pace MC, et al. Coupling of receptor conformation and ligand orientation determine graded activity. Nat Chem Biol. 2010;6:837–843. doi: 10.1038/nchembio.451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Gee AC, Carlson KE, Martini PGV, Katzenellenbogen BS, Katzenellenbogen JA. Coactivator peptides have a differential stabilizing effect on the binding of estrogens and antiestrogens with the estrogen receptor. Mol Endocrinol. 1999;13:1912–1923. doi: 10.1210/mend.13.11.0373. [DOI] [PubMed] [Google Scholar]
- 54.Likhite VS, Stossi F, Kim K, Katzenellenbogen BS, Katzenellenbogen JA. Kinase-specific phosporylation of the estrogen receptor changes receptor interactions with ligand, deoxyribonucleic acid, and coregulators associated with alterations in estrogen and tamoxifen activity. Mol Endocrinol. 2006;20:3120–3132. doi: 10.1210/me.2006-0068. [DOI] [PubMed] [Google Scholar]
- 55.Reese JC, Katzenellenbogen BS. Differential DNA-binding abilities of estrogen receptor occupied with two classes of antiestrogens: Studies using human estrogen receptor overexpressed in mammalian cells. Nucleic Acids Res. 1991;19:6595–6602. doi: 10.1093/nar/19.23.6595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hall JM, McDonnell DP, Korach KS. Allosteric regulation of estrogen receptor structure, function, and coactivator recruitment by different estrogen response elements. Mol Endocrinol. 2002;16:469–486. doi: 10.1210/mend.16.3.0814. [DOI] [PubMed] [Google Scholar]
- 57.McClung MR, Siris E, Cummings S, Bolognese M, Ettinger M, Moffett A, et al. Prevention of bone loss in postmenopausal women treated with lasofoxifene compared with raloxifene. Menopause. 2006;13:377–386. doi: 10.1097/01.gme.0000188736.69617.4f. [DOI] [PubMed] [Google Scholar]
- 58.LaCroix AZ, Powles T, Osborne CK, Wolter K, Thompson JR, Thompson DD, et al. Breast cancer incidence in the randomized PEARL Trial of lasofoxifene in postmenopausal osteoporotic women. J Natl Cancer Inst. 2010 doi: 10.1093/jnci/djq415. In Press. [DOI] [PubMed] [Google Scholar]
- 59.Cummings SR, Ensrud K, Delmas PD, LaCroix AZ, Vukicevic S, Reid DM, et al. Lasofoxifene in postmenopausal women with osteoporosis. N Engl J Med. 2010;362:686–696. doi: 10.1056/NEJMoa0808692. [DOI] [PubMed] [Google Scholar]
- 60.Goldstein SR, Neven P, Cummings S, Colgan T, Runowicz CD, Krpan D, et al. Postmenopausal Evaluation and Risk Reduction With Lasofoxifene (PEARL) trial: 5-year gynecological outcomes. Menopause. 2011;18:17–22. doi: 10.1097/gme.0b013e3181e84bb4. [DOI] [PubMed] [Google Scholar]
- 61.Miller PD, Chines AA, Christiansen C, Hoeck HC, Kendler DL, Lewiecki EM, et al. Effects of bazedoxifene on BMD and bone turnover in postmenopausal women: 2-yr results of a randomized, double-blind, placebo-, and active-controlled study. J Bone Miner Res. 2008;23:525–535. doi: 10.1359/jbmr.071206. [DOI] [PubMed] [Google Scholar]
- 62.Silverman SL, Christiansen C, Genant HK, Vukicevic S, Zanchetta JR, de Villiers TJ, et al. Efficacy of bazedoxifene in reducing new vertebral fracture risk in postmenopausal women with osteoporosis: results from a 3-year, randomized, placebo-, and active-controlled clinical trial. J Bone Miner Res. 2008;23:1923–1934. doi: 10.1359/jbmr.080710. [DOI] [PubMed] [Google Scholar]
- 63.Archer DF, Lewis V, Carr BR, Olivier S, Pickar JH. Bazedoxifene/conjugated estrogens (BZA/CE): incidence of uterine bleeding in postmenopausal women. Fertil Steril. 2009;92:1039–1044. doi: 10.1016/j.fertnstert.2009.05.093. [DOI] [PubMed] [Google Scholar]
- 64.Christiansen C, Chesnut CH, 3rd, Adachi JD, Brown JP, Fernandes CE, Kung AW, et al. Safety of bazedoxifene in a randomized, double-blind, placebo- and active-controlled Phase 3 study of postmenopausal women with osteoporosis. BMC Musculoskelet Disord. 2010;11:130. doi: 10.1186/1471-2474-11-130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Harvey JA, Holm MK, Ranganath R, Guse PA, Trott EA, Helzner E. The effects of bazedoxifene on mammographic breast density in postmenopausal women with osteoporosis. Menopause. 2009;16:1193–1196. doi: 10.1097/gme.0b013e3181a7fb1e. [DOI] [PubMed] [Google Scholar]
- 66.Bolognese M, Krege JH, Utian WH, Feldman R, Broy S, Meats DL, et al. Effects of arzoxifene on bone mineral density and endometrium in postmenopausal women with normal or low bone mass. J Clin Endocrinol Metab. 2009;94:2284–2289. doi: 10.1210/jc.2008-2143. [DOI] [PubMed] [Google Scholar]
- 67.Kendler DL, Palacios S, Cox DA, Stock J, Alam J, Dowsett SA, et al. Arzoxifene versus raloxifene: effect on bone and safety parameters in postmenopausal women with osteoporosis. Osteoporos Int. 2012;23:1091–1101. doi: 10.1007/s00198-011-1587-0. [DOI] [PubMed] [Google Scholar]
- 68.Powles TJ, Diem SJ, Fabian CJ, Neven P, Wickerham DL, Cox DA, et al. Breast cancer incidence in postmenopausal women with osteoporosis or low bone mass using arzoxifene. Breast Cancer Res Treat. 2012;134:299–306. doi: 10.1007/s10549-012-2041-5. [DOI] [PubMed] [Google Scholar]
- 69.McMeekin DS, Gordon A, Fowler J, Melemed A, Buller R, Burke T, et al. A phase II trial of arzoxifene, a selective estrogen response modulator, in patients with recurrent or advanced endometrial cancer. Gynecol Oncol. 2003;90:64–69. doi: 10.1016/s0090-8258(03)00203-8. [DOI] [PubMed] [Google Scholar]
- 70.Goldstein SR, Archer DF, Simon JA, Constantine G. Endometrial safety of ospemifene and the ability of transvaginal ultrasonography to detect small changes in endometrial thickness. Obstet Gynecol. 2014;123(Suppl 1):96S–97S. [Google Scholar]
- 71.Voipio SK, Komi J, Kangas L, Halonen K, DeGregorio MW, Erkkola RU. Effects of ospemifene (FC-1271a) on uterine endometrium, vaginal maturation index, and hormonal status in healthy postmenopausal women. Maturitas. 2002;43:207–214. doi: 10.1016/s0378-5122(02)00206-2. [DOI] [PubMed] [Google Scholar]
- 72.Portman D, Palacios S, Nappi RE, Mueck AO. Ospemifene, a non-oestrogen selective oestrogen receptor modulator for the treatment of vaginal dryness associated with postmenopausal vulvar and vaginal atrophy: A randomised, placebo-controlled, phase III trial. Maturitas. 2014 doi: 10.1016/j.maturitas.2014.02.015. [DOI] [PubMed] [Google Scholar]
- 73.Qu Q, Zheng H, Dahllund J, Laine A, Cockcroft N, Peng Z, et al. Selective estrogenic effects of a novel triphenylethylene compound, FC1271a, on bone, cholesterol level, and reproductive tissues in intact and ovariectomized rats. Endocrinology. 2000;141:809–820. doi: 10.1210/endo.141.2.7342. [DOI] [PubMed] [Google Scholar]
- 74.Komi J, Lankinen KS, DeGregorio M, Heikkinen J, Saarikoski S, Tuppurainen M, et al. Effects of ospemifene and raloxifene on biochemical markers of bone turnover in postmenopausal women. J Bone Miner Metab. 2006;24:314–318. doi: 10.1007/s00774-006-0689-9. [DOI] [PubMed] [Google Scholar]
- 75.Komi J, Heikkinen J, Rutanen EM, Halonen K, Lammintausta R, Ylikorkala O. Effects of ospemifene, a novel SERM, on biochemical markers of bone turnover in healthy postmenopausal women. Gynecol Endocrinol. 2004;18:152–158. doi: 10.1080/09513590410001672197. [DOI] [PubMed] [Google Scholar]
- 76.Sarrel PM, Njike VY, Vinante V, Katz DL. The mortality toll of estrogen avoidance: an analysis of excess deaths among hysterectomized women aged 50 to 59 years. Am J Public Health. 2013;103:1583–1588. doi: 10.2105/AJPH.2013.301295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Anderson GL, Chlebowski RT, Aragaki AK, Kuller LH, Manson JE, Gass M, et al. Conjugated equine oestrogen and breast cancer incidence and mortality in postmenopausal women with hysterectomy: extended follow-up of the Women's Health Initiative randomised placebo-controlled trial. The lancet oncology. 2012;13:476–486. doi: 10.1016/S1470-2045(12)70075-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Barbaglia G, Macia F, Comas M, Sala M, del Mar Vernet M, Casamitjana M, et al. Trends in hormone therapy use before and after publication of the Women's Health Initiative trial: 10 years of follow-up. Menopause. 2009;16:1061–1064. doi: 10.1097/gme.0b013e3181a02b44. [DOI] [PubMed] [Google Scholar]
- 79.Whiteley J, Wagner JS, Bushmakin A, Kopenhafer L, Dibonaventura M, Racketa J. Impact of the severity of vasomotor symptoms on health status, resource use, and productivity. Menopause. 2013;20:518–524. doi: 10.1097/GME.0b013e31827d38a5. [DOI] [PubMed] [Google Scholar]
- 80.Bakken K, Fournier A, Lund E, Waaseth M, Dumeaux V, Clavel-Chapelon F, et al. Menopausal hormone therapy and breast cancer risk: impact of different treatments. The European Prospective Investigation into Cancer and Nutrition. Int J Cancer. 2011;128:144–156. doi: 10.1002/ijc.25314. [DOI] [PubMed] [Google Scholar]
- 81.Beral V Million Women Study C. Breast cancer and hormone-replacement therapy in the Million Women Study. Lancet. 2003;362:419–427. doi: 10.1016/s0140-6736(03)14065-2. [DOI] [PubMed] [Google Scholar]
- 82.Davis S, O'Neill S, Eden J, Baber R, Ekangaki A, Stocks J, et al. Transition from estrogen therapy to raloxifene in postmenopausal women: effects on treatment satisfaction and the endometrium-a pilot study. Menopause. 2004;11:167–175. doi: 10.1097/01.gme.0000087981.28957.cf. [DOI] [PubMed] [Google Scholar]
- 83.Stovall DW, Utian WH, Gass ML, Qu Y, Muram D, Wong M, et al. The effects of combined raloxifene and oral estrogen on vasomotor symptoms and endometrial safety. Menopause. 2007;14:510–517. doi: 10.1097/GME.0b013e318031a83d. [DOI] [PubMed] [Google Scholar]
- 84.Levine J. Treating menopausal symptoms with a tissue-selective estrogen complex. Gender medicine. 2011;8:57–68. doi: 10.1016/j.genm.2011.03.008. [DOI] [PubMed] [Google Scholar]
- 85.Komm BS, Kharode YP, Bodine PV, Harris HA, Miller CP, Lyttle CR. Bazedoxifene acetate: a selective estrogen receptor modulator with improved selectivity. Endocrinology. 2005;146:3999–4008. doi: 10.1210/en.2005-0030. [DOI] [PubMed] [Google Scholar]
- 86.McTiernan A, Chlebowski RT, Martin C, Peck JD, Aragaki A, Pisano ED, et al. Conjugated equine estrogen influence on mammographic density in postmenopausal women in a substudy of the women's health initiative randomized trial. J Clin Oncol. 2009;27:6135–6143. doi: 10.1200/JCO.2008.21.7166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Harvey JA, Pinkerton JV, Baracat EC, Shi H, Chines AA, Mirkin S. Breast density changes in a randomized controlled trial evaluating bazedoxifene/conjugated estrogens. Menopause. 2013;20:138–145. doi: 10.1097/gme.0b013e318271f5e7. [DOI] [PubMed] [Google Scholar]
- 88.Pinkerton JV, Harvey JA, Lindsay R, Pan K, Chines AA, Mirkin S, et al. Effects of bazedoxifene/conjugated estrogens on the endometrium and bone: a randomized trial. J Clin Endocrinol Metab. 2014;99:E189–E198. doi: 10.1210/jc.2013-1707. [DOI] [PubMed] [Google Scholar]
- 89.Naqvi H, Sakr S, Presti T, Krikun G, Komm B, Taylor HS. Treatment with Bazedoxifene and Conjugated Estrogens Results in Regression of Endometriosis in a Murine Model. Biol Reprod. 2014 doi: 10.1095/biolreprod.113.114165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Umetani M, Domoto H, Gormley AK, Yuhanna IS, Cummins CL, Javitt NB, et al. 27-Hydroxycholesterol is an endogenous SERM that inhibits the cardiovascular effects of estrogen. Nat Med. 2007;13:1185–1192. doi: 10.1038/nm1641. [DOI] [PubMed] [Google Scholar]
- 91.Dzeletovic S, Breuer O, Lund E, Diczfalusy U. Determination of cholesterol oxidation products in human plasma by isotope dilution-mass spectrometry. Analytical biochemistry. 1995;225:73–80. doi: 10.1006/abio.1995.1110. [DOI] [PubMed] [Google Scholar]
- 92.Karuna R, Holleboom AG, Motazacker MM, Kuivenhoven JA, Frikke-Schmidt R, Tybjaerg-Hansen A, et al. Plasma levels of 27-hydroxycholesterol in humans and mice with monogenic disturbances of high density lipoprotein metabolism. Atherosclerosis. 2011;214:448–455. doi: 10.1016/j.atherosclerosis.2010.10.042. [DOI] [PubMed] [Google Scholar]
- 93.DuSell CD, Umetani M, Shaul PW, Mangelsdorf DJ, McDonnell DP. 27-hydroxycholesterol is an endogenous selective estrogen receptor modulator. Mol Endocrinol. 2008;22:65–77. doi: 10.1210/me.2007-0383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Brouchet L, Krust A, Dupont S, Chambon P, Bayard F, Arnal JF. Estradiol accelerates reendothelialization in mouse carotid artery through estrogen receptor alpha but not estrogen receptor beta. Circulation. 2001;103:423–428. doi: 10.1161/01.cir.103.3.423. [DOI] [PubMed] [Google Scholar]
- 95.DuSell CD, Nelson ER, Wang X, Abdo J, Modder UI, Umetani M, et al. The endogenous selective estrogen receptor modulator 27-hydroxycholesterol is a negative regulator of bone homeostasis. Endocrinology. 2010;151:3675–3685. doi: 10.1210/en.2010-0080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Tarakida A, Iino K, Abe K, Taniguchi R, Higuchi T, Mizunuma H, et al. Hypercholesterolemia accelerates bone loss in postmenopausal women. Climacteric. 2011;14:105–111. doi: 10.3109/13697137.2010.507888. [DOI] [PubMed] [Google Scholar]
- 97.Yamaguchi T, Sugimoto T, Yano S, Yamauchi M, Sowa H, Chen Q, et al. Plasma lipids and osteoporosis in postmenopausal women. Endocr J. 2002;49:211–217. doi: 10.1507/endocrj.49.211. [DOI] [PubMed] [Google Scholar]
- 98.Orozco P. Atherogenic lipid profile and elevated lipoprotein (a) are associated with lower bone mineral density in early postmenopausal overweight women. Eur J Epidemiol. 2004;19:1105–1112. doi: 10.1007/s10654-004-1706-8. [DOI] [PubMed] [Google Scholar]
- 99.Karuna R, Eckardstein Av, Rentsch KM. Dpant assisted-atmospheric pressure photoionization (DA-APPI) liquid chromatography-mass spectrometry for the quantification of 27-hydroxycholesterol in plasma. J Chromatography B. 2009;877:261–268. doi: 10.1016/j.jchromb.2008.12.033. [DOI] [PubMed] [Google Scholar]
- 100.Bauer DC, Mundy GR, Jamal SA, Black DM, Cauley JA, Ensrud KE, et al. Use of statins and fracture: results of 4 prospective studies and cumulative meta-analysis of observational studies and controlled trials. Arch Intern Med. 2004;164:146–152. doi: 10.1001/archinte.164.2.146. [DOI] [PubMed] [Google Scholar]
- 101.Lupattelli G, Scarponi AM, Vaudo G, Siepi D, Roscini AR, Gemelli F, et al. Simvastatin increases bone mineral density in hypercholesterolemic postmenopausal women. Metabolism. 2004;53:744–748. doi: 10.1016/j.metabol.2004.01.010. [DOI] [PubMed] [Google Scholar]
- 102.Nelson ER, Wardell SE, Jasper JS, Park S, Suchindran S, Howe MK, et al. 27-Hydroxycholesterol links hypercholesterolemia and breast cancer pathophysiology. Science. 2013;342:1094–1098. doi: 10.1126/science.1241908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wu Q, Ishikawa T, Sirianni R, Tang H, McDonald JG, Yuhanna IS, et al. 27-Hydroxycholesterol promotes cell-autonomous, ER-positive breast cancer growth. Cell reports. 2013;5:637–645. doi: 10.1016/j.celrep.2013.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Smith CL, O'Malley BW. Coregulator function: a key to understanding tissue specificity of selective receptor modulators. Endocr Rev. 2004;25:45–71. doi: 10.1210/er.2003-0023. [DOI] [PubMed] [Google Scholar]
- 105.Jordan VC, Fritz NF, Tormey DC. Endocrine effects of adjuvant chemotherapy and longterm tamoxifen administration on node-positive patients with breast cancer. Cancer Res. 1987;47:624–630. [PubMed] [Google Scholar]