Abstract
Iodide uptake across the membranes of thyroid follicular cells and cancer cells occurs through an active transport process mediated by the sodium-iodide symporter (NIS). The rat and human NIS-coding genes were cloned and identified in 1996. Evaluation of NIS gene and protein expression is critical for the management of thyroid cancer, and several approaches to increase NIS levels have been tried. Identification of the NIS gene has provided a means of expanding its role in radionuclide therapy and molecular target-specific theragnosis (therapy and diagnosis using the same molecular target). In this article, we describe the relationship between NIS expression and the thyroid carcinoma treatment using I-131 and alternative therapeutic approaches.
Keywords: Differentiated thyroid cancer, Radioiodine therapy, Sodium-iodide symporter, Target-specific therapy, Theranosis
INTRODUCTION
The incidence of thyroid carcinoma is increasing in many countries, including Korea [1]. Differentiated thyroid carcinoma (DTC) accounts for most thyroid cancers and is characterized by slow growth and a good prognosis. The specific accumulation of iodide in the thyroid gland was first detected in 1915, and radioiodine was first applied in the diagnosis and treatment of thyroid cancer in 1942 [2]. Improvements in diagnosis and treatment using radioiodine have reduced DTC-associated mortality [3].
Iodide uptake across the membranes of thyroid follicular cells and cancer cells occurs through an active sodium-iodide symporter (NIS) [4]. The rat and human NIS-coding genes were cloned and identified in 1996 [5]. Low levels of NIS are present in thyroid carcinoma cells and are responsible for radioiodine uptake in metastatic lesions. However, one-third of all differentiated thyroid cancers and all anaplastic thyroid cancers do not concentrate radioiodine and, accordingly, have a poor prognosis. Thus, evaluation of NIS gene and protein expression is critical in the management of thyroid cancer [2].
RADIOIODINE THERAPY IN DTC
DTC is one of the most curable cancers, with a 10-year survival rate of 80% to 95% [4,6]. Surgical resection followed by radioiodine therapy is considered to be the ideal treatment for high-risk tumors. However, recurrence in the thyroid bed or cervical lymph nodes develops in 5% to 20% of patients with DTC, and some patients develop distant metastatic disease, which decreases the 10-year survival rate of patients by 50% [7,8]. Significant controversy persists regarding which patients will benefit from radioactive iodine treatment and the method of radioiodine application.
Postoperative remnant ablation
Radioiodine ablation of remnant thyroid following thyroidectomy is performed for several theoretical reasons. First, radioiodine is used to ablate any residual remnant thyroid tissues after surgical resection, which increases the sensitivity of subsequent tests based on serum thyroglobulin (Tg) measurement and radioiodine whole body scanning. Second, it also serves as an adjuvant treatment of potential residual tumors [4].
Over the past 20 years, 2,036 thyroid carcinoma patients have been treated at Seoul National University Hospital (SNUH) [4]: 1,873 with papillary thyroid carcinoma (92%) and 163 with follicular carcinoma (8%). Of these 2,036 patients, 468 (22.9%) had lymph node metastasis and/or distant metastasis according to I-131 posttherapy whole body scans. Specifically, 313 patients (15.4%) had regional lymph node metastasis, 83 (4.1%) mediastinal lymph node metastasis, 109 (5.3%) pulmonary metastasis, and 25 (1.2%) bone metastasis.
Radioiodine therapy for metastatic disease
The most common sites of distant metastases from DTC are the lungs and bone, followed by the brain, liver, kidneys, and muscle [8]. Older patients (>45 years old) with distant metastatic thyroid cancer are classified as having stage IVC disease according to the American Joint Committee on Cancer (AJCC) criteria (sixth edition). These patients have a 5-year survival rate of only ~30% to 40% [8,9,10]. Aggressive surgery, radioiodine therapy, and levothyroxine suppression therapy can improve overall survival and disease-specific survival in this subgroup of patients. The therapeutic options in patients with locally advanced or metastatic thyroid cancer with no response to radioiodine are limited. Unfortunately, many of these patients ultimately die from advanced disease. Other therapeutic approaches are needed.
Lungs
At SUNH, patients with pulmonary metastasis underwent several courses of I-131 therapy (range, 3.7 to 7.4 GBq) over 40 months (range, 6 to 171) and were treated on average 5.1 times (range, 1 to 14). During follow-up, pulmonary metastasis completely disappeared in 38 of the 109 patients (35%), and partial remission occurred in 44 (40%) (Table 1). Of the 109 patients, 45 (41%) had a diffuse pattern of I-131 lung uptake on whole body scans. Combined nodular and diffuse uptake was observed in 35 patients (32%), and nodular uptake without diffuse uptake in 29 (27%). The response to and benefit of I-131 treatment in pulmonary metastases is variable and at least partially dependent on the degree and pattern of radioiodine uptake in metastases as determined by radioiodine whole body scan (Fig. 1). Lesions with diffuse uptake were found to respond better than those with nodular uptake: of the 45 patients with diffuse lung uptake, 51% achieved complete remission [4].
Table 1.
Radioiodine Treatment Results for Lung Metastases in Patients with Differentiated Thyroid Cancer

Adapted from Chung et al. Nucl Med Mol Imaging 2010;44:4-14, with permission from Springer [4].
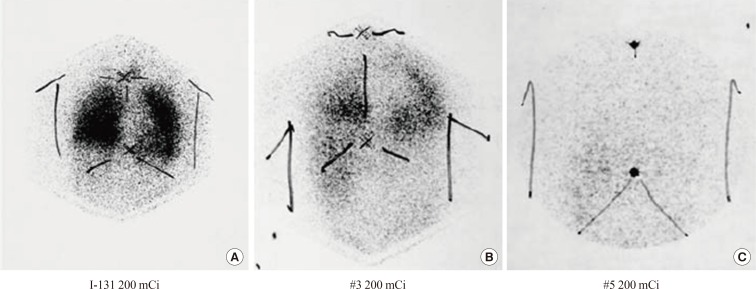
Fig. 1.
(A-C) A 62-year-old male postthyroidectomy papillary cancer patient with lymph node and lung metastases. After serial I-131 treatment (200 mCi), the metastatic lesions disappeared gradually, resulting in complete remission. Adapted from Chung et al. Nucl Med Mol Imaging 2010;44:4-14, with permission from Springer [4].
Age is an important prognostic factor, and pediatric patients with radioiodine-avid pulmonary metastases have an excellent prognosis, even without I-131 treatment [11].
Bone
I-131 therapy can benefit selected patients with bone metastases. The benefit from I-131 treatment appears to be greater in radioiodine positive, fewer in number, smaller in size, and negative on X-ray. For a patient with a single bone metastasis, other therapies such as surgical excision, external radiotherapy, radiofrequency ablation, cryotherapy, and/or arterial embolization should be considered combined with I-131.
Of the 25 patients with bone metastasis from DTC at SNUH [4], 16 (64%) had multiple bone metastases in vertebrae and pelvic bones. A total of 105 bone lesions were detected on posttherapy scans: 40 in vertebrae, 24 in pelvic bones, 12 in femurs, and 10 in the skull; others were detected in the sternum, ribs, or clavicles. Of these 105 bone lesions, 75 were treated with I-131 alone; 34 lesions (45%) improved, and 41 showed no response or were aggravated. Notably, nine of 25 bone lesions treated by surgical resection and I-131 therapy disappeared completely.
We found that I-131 therapy plays a key role in the management of metastatic DTC. Furthermore, our experience is that lung metastases are relatively well controlled by I-131 treatment, and that bone metastases require I-131 therapy plus surgical resection or external radiotherapy [12].
I-131 therapy of patients with negative iodine scans
At present, there is no consensus regarding radioiodine therapy for Tg-positive and radioiodine-negative cancer [13,14]. The preference for empirical I-131 therapy in DTC patients with raised Tg levels and negative scan results is based on reducing the Tg levels and the increased likelihood of positive scan results after a high dose of radioiodine.
The role of fluorine-18 fluorodeoxyglucose positron emission tomography (F-18 FDG PET) in patients with DTC, high serum Tg levels, and negative radioiodine scans is still controversial. F-18 FDG uptake by DTC is associated with more aggressive biological behavior of the tumor and a worse prognosis. Moreover, high expression of the glucose transporter type 1 gene supports the use of PET with specific tracers in the clinical management of such cancers, and BRAF-V600E point mutations may lead to less differentiated phenotypes and lower expression of NIS, suggesting a worse prognosis [15]. However, discordant findings between PET and traditional nuclear medicine radioiodine imaging (the "flip/flop phenomenon": uptake of I-131 with no FDG uptake and vice versa) are observed frequently (Fig. 2) [16]. We suggest that the evaluation of NIS expression will guide I-131 therapy.
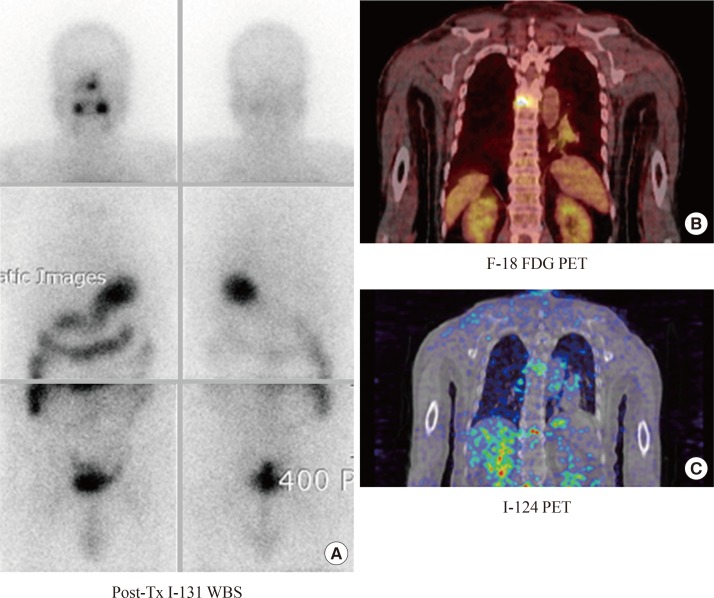
Fig. 2.
(A-C) Iodine scan-negative and fluorodeoxyglucose positron emission tomography (FDG PET)-positive metastasis. A patient with DTC had bone pain in the back after total thyroidectomy. An iodine scan was negative. By contrast, on an fluorine-18 FDG PET (F-18 FDG PET) scan, there was a focal hypermetabolic bone lesion, suggesting malignant tumors. I-124, a PET radiotracer iodine isotope, demonstrated faint iodine uptake on PET scans. This discrepancy between the I-131 scan and I-124 PET may have been caused by a difference in the resolution of the instruments. The patient underwent high-dose radioiodine treatment, and the bone pain improved. WBS, whole body scan. Adapted from Cheon, Korean J Endocr Surg 2011:11:139-45 [9].
NIS expression and its effectiveness for radioiodine therapy
NIS is an intrinsic membrane protein with 13 transmembrane domains and is composed of 643 amino acids. The recent cloning of the NIS gene enabled better understanding of the molecular mechanisms underlying iodide transport and has provided a means of expanding its role in the management of thyroid cancer [5].
The efficacy of I-131 is dependent in part on translocation of NIS protein to the cell membrane. In thyroid cancer, native NIS expression and radioiodide uptake are reduced. Stimulation of NIS expression by increasing serum levels of thyroid stimulating hormone (TSH) is required. Normal thyroid tissue incorporates the trapped iodide into Tg, a process referred to as organification, resulting in longer iodide retention. Iodide in most thyroid cancers is not incorporated efficiently into proteins and hence is more easily discharged from cancer tissues [17]. Greater NIS expression in thyroid cancer is associated with a greater uptake of radioiodide as well as a better prognosis.
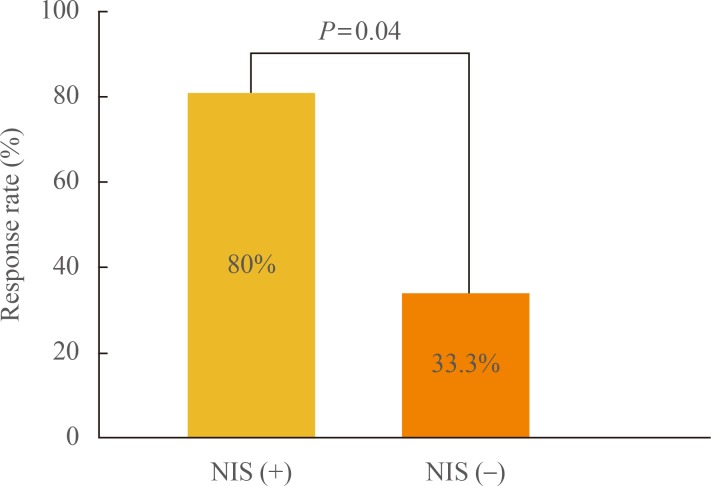
Positive immunostaining of human NIS (hNIS) in primary thyroid cancer could predict I-131 accumulation and the effectiveness of I-131 therapy in recurrent lesions (Fig. 3). At SNUH, while one-third of patients with metastasis but without NIS expression on tissue immunostaining responded to radioiodine therapy, 80% of patients with NIS expression responded to the therapy [18].
Fig. 3.
Comparison of I-131 treatment outcomes according to human sodium-iodide symporter (NIS) expression. Adapted from Min et al. Eur J Nucl Med 2001;28:639-45, with permission from Springer [18].
ALTERNATIVE THERAPEUTIC APPROACHES
NIS expression on thyroid cancer cell membranes depends on the differentiation status of the cells, and poorly differentiated thyroid cancer cells lose their ability to accumulate radioiodine. In such cases, other therapeutic options may be attempted, such as supportive chemotherapy using tyrosine kinase inhibitors [19] or preconditioning using retinoic acid (RA) for redifferentiation and restoration of NIS expression [20].
Lesion-specific therapy
Several studies have reported alterations in gene expression and phenotypic heterogeneity between metastatic and primary cancer sites [21]. The heterogeneity is primarily caused by differences in gene expression at different metastatic sites. The therapeutic effect of β-emitting radionuclides is correlated with tumor size [22].
In patients with multiple metastases, a heterogeneous response to I-131 therapy is not so uncommon, and other therapies should be considered for the lesions that do not respond to I-131 therapy. In such cases, lesion-specific evaluation is essential, and F-18 FDG PET can be a useful method to discriminate lesional differences in tumor characteristics.
Enhancement of NIS in thyroid cancer
One of the main methods for increasing NIS levels in tissues is to enhance NIS promoter activity such as redox factor-1 (Ref-1), early growth response 1 (Egr1), paired box-8 (Pax-8), NIS TSH-responsive factor-1 (NTF-1), or thyroid transcription factor-1 (TTF-1) [23]. However, these proteins are still not applicable clinically. The second method involves the use of exogenous agents to enhance the expression of NIS mRNA and protein.
Several medications have been investigated as potential redifferentiation agents [24]. The basic mechanisms involved are unclear but appear to be related to blocked access of tumor cells to free glutamine, isoprenylation of RAS family proteins, RA receptor activation, histone acetylation, peroxisome proliferator-activated receptor-γ activation, and demethylation. Clinical pilot studies have shown that the ability to accumulate iodide may be restored by RA treatment [25]. However, the effectiveness and legitimacy of using RA in clinical practice remain controversial.
We combined RA therapy with radioiodine in 47 patients with radioiodine-refractory DTC. The overall response rate was 21.3% (n=10/47); one patient achieved a complete remission and nine patients showed a partial remission. Interestingly, age was the most important factor determining the response to RA/radioiodine combined therapy: patients less than 45 years of age were found to have more clinical benefit from RA application [26].
Some kinase inhibitors were reported very recently to have the potential to restore NIS expression in radioiodide-refractory thyroid cancer. Approximately 70% of papillary thyroid cancers have mutually exclusive gene mutations encoding the growth factor receptors RET and NTRK1, the three isoforms of RAS (N, H, and K), and BRAF [27]. Constitutive activation of these proteins stimulates mitogen-activated protein kinase (MAPK) signaling, which inhibits the expression of thyroid hormone biosynthesis genes, including NIS and thyroid peroxidase, which facilitate iodine uptake and organification, respectively [28]. Selumetinib, a selective MAPK pathway antagonist, was reported to cause clinically meaningful increases in iodine uptake and retention in a subgroup of patients with radioiodine-refractory thyroid cancer. These results suggest the potential of combined therapy with enhanced radioiodide uptake by small molecule-targeted drugs.
Radionuclide gene therapy using NIS
Cloning and characterization of the NIS gene have paved the way for the development of a novel radionuclide gene therapy, because the targeted expression of functional NIS in cancer cells enables these cells to concentrate iodide from the plasma, thus offering the possibility of radioiodine therapy [4]. This strategy could also be applied to non-thyroid cancers. The NIS gene can be transfected into a variety of cell types using nonviral or viral vectors, which can increase radioiodine uptake up to several hundred-fold [4]. Combination therapies based on targeting and expression of the NIS gene plus radioiodine treatment could also be used to treat nonthyroid malignant diseases, such as melanoma and cervical, breast, liver, and colon cancers.
CONCLUSIONS
Radioiodine remains a mainstay of therapy in patients with radioiodine-avid DTC because of the presence of the target molecule NIS. In fact, this is the first and still the most widely used targeted therapy in oncology. NIS expression is quite variable among DTC patients and even among different lesions in the same patient. As a result, there are controversies regarding I-131 therapy. Evaluation of hNIS expression or functional integrity in cancer tissues by immunohistochemical staining would enhance patient management and is recommended. Molecular imaging modalities including radioiodine scanning and F-18 FDG PET should be used for this purpose. Patients with progressive, radioiodine-resistant metastatic disease could be considered for clinical trials evaluating alternative approaches.
Footnotes
No potential conflict of interest relevant to this article was reported.
References
- 1.Ministry of Health and Welfare, National Cancer Center. Cancer facts & figures 2010. Seoul: National Cancer Center; 2013. [Google Scholar]
- 2.Chung JK. Sodium iodide symporter: its role in nuclear medicine. J Nucl Med. 2002;43:1188–1200. [PubMed] [Google Scholar]
- 3.Fernandes JK, Day TA, Richardson MS, Sharma AK. Overview of the management of differentiated thyroid cancer. Curr Treat Options Oncol. 2005;6:47–57. doi: 10.1007/s11864-005-0012-3. [DOI] [PubMed] [Google Scholar]
- 4.Chung JK, Youn HW, Kang JH, Lee HY, Kang KW. Sodium iodide symporter and the radioiodine treatment of thyroid carcinoma. Nucl Med Mol Imaging. 2010;44:4–14. doi: 10.1007/s13139-009-0016-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dai G, Levy O, Carrasco N. Cloning and characterization of the thyroid iodide transporter. Nature. 1996;379:458–460. doi: 10.1038/379458a0. [DOI] [PubMed] [Google Scholar]
- 6.Sampson E, Brierley JD, Le LW, Rotstein L, Tsang RW. Clinical management and outcome of papillary and follicular (differentiated) thyroid cancer presenting with distant metastasis at diagnosis. Cancer. 2007;110:1451–1456. doi: 10.1002/cncr.22956. [DOI] [PubMed] [Google Scholar]
- 7.Van Nostrand D. The benefits and risks of I-131 therapy in patients with well-differentiated thyroid cancer. Thyroid. 2009;19:1381–1391. doi: 10.1089/thy.2009.1611. [DOI] [PubMed] [Google Scholar]
- 8.Haugen BR, Kane MA. Approach to the thyroid cancer patient with extracervical metastases. J Clin Endocrinol Metab. 2010;95:987–993. doi: 10.1210/jc.2009-2305. [DOI] [PubMed] [Google Scholar]
- 9.Cheon GJ. New trends in radioiodine treatment for the advanced differentiated thyroid cancer. Korean J Endocr Surg. 2011;11:139–145. [Google Scholar]
- 10.Massin JP, Savoie JC, Garnier H, Guiraudon G, Leger FA, Bacourt F. Pulmonary metastases in differentiated thyroid carcinoma. Study of 58 cases with implications for the primary tumor treatment. Cancer. 1984;53:982–992. doi: 10.1002/1097-0142(19840215)53:4<982::aid-cncr2820530427>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- 11.Massin JP, Savoie JC, Garnier H, Guidraudon G, Leger FA, Bacourt F. Pulmonary metastases in differentiated thyroid carcinoma: study of 58 cases with implications for the primary tumor treatment. Cancer. 1984;53:982–992. doi: 10.1002/1097-0142(19840215)53:4<982::aid-cncr2820530427>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- 12.Bernier MO, Leenhardt L, Hoang C, Aurengo A, Mary JY, Menegaux F, Enkaoua E, Turpin G, Chiras J, Saillant G, Hejblum G. Survival and therapeutic modalities in patients with bone metastases of differentiated thyroid carcinomas. J Clin Endocrinol Metab. 2001;86:1568–1573. doi: 10.1210/jcem.86.4.7390. [DOI] [PubMed] [Google Scholar]
- 13.Chao M. Management of differentiated thyroid cancer with rising thyroglobulin and negative diagnostic radioiodine whole body scan. Clin Oncol (R Coll Radiol) 2010;22:438–447. doi: 10.1016/j.clon.2010.05.005. [DOI] [PubMed] [Google Scholar]
- 14.Schlumberger MJ. Papillary and follicular thyroid carcinoma. N Engl J Med. 1998;338:297–306. doi: 10.1056/NEJM199801293380506. [DOI] [PubMed] [Google Scholar]
- 15.Schonberger J, Ruschoff J, Grimm D, Marienhagen J, Rümmele P, Meyringer R, Kossmehl P, Hofstaedter F, Eilles C. Glucose transporter 1 gene expression is related to thyroid neoplasms with an unfavorable prognosis: an immunohistochemical study. Thyroid. 2002;12:747–754. doi: 10.1089/105072502760339307. [DOI] [PubMed] [Google Scholar]
- 16.Feine U, Lietzenmayer R, Hanke JP, Held J, Wohrle H, Müller-Schauenburg W. Fluorine-18-FDG and iodine-131-iodide uptake in thyroid cancer. J Nucl Med. 1996;37:1468–1472. [PubMed] [Google Scholar]
- 17.Schlumberger M, Lacroix L, Russo D, Filetti S, Bidart JM. Defects in iodide metabolism in thyroid cancer and implications for the follow-up and treatment of patients. Nat Clin Pract Endocrinol Metab. 2007;3:260–269. doi: 10.1038/ncpendmet0449. [DOI] [PubMed] [Google Scholar]
- 18.Min JJ, Chung JK, Lee Y, Jeong J, Lee D, Jang J, Lee M, Cho B. Relationship between expression of the sodium/iodide symporter and (131)I uptake in recurrent lesions of differentiated thyroid carcinoma. Eur J Nucl Med. 2001;28:639–645. doi: 10.1007/s002590100509. [DOI] [PubMed] [Google Scholar]
- 19.Paeng JC, Kang KW, Park do J, Oh SW, Chung JK. Alternative medical treatment for radioiodine-refractory thyroid cancers. Nucl Med Mol Imaging. 2011;45:241–247. doi: 10.1007/s13139-011-0107-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fernandez CA, Puig-Domingo M, Lomena F, Estorch M, Camacho Marti V, Bittini AL, Marazuela M, Santamaria J, Castro J, Martinez de Icaya P, Moraga I, Martin T, Megia A, Porta M, Mauricio D, Halperin I. Effectiveness of retinoic acid treatment for redifferentiation of thyroid cancer in relation to recovery of radioiodine uptake. J Endocrinol Invest. 2009;32:228–233. doi: 10.1007/BF03346457. [DOI] [PubMed] [Google Scholar]
- 21.Swoboda A, Rasin-Streden D, Schanab O, Okamoto I, Pehamberger H, Petzelbauer P, Mikula M. Identification of genetic disparity between primary and metastatic melanoma in human patients. Genes Chromosomes Cancer. 2011;50:680–688. doi: 10.1002/gcc.20890. [DOI] [PubMed] [Google Scholar]
- 22.O'Donoghue JA, Bardies M, Wheldon TE. Relationships between tumor size and curability for uniformly targeted therapy with beta-emitting radionuclides. J Nucl Med. 1995;36:1902–1909. [PubMed] [Google Scholar]
- 23.Puppin C, Arturi F, Ferretti E, Russo D, Sacco R, Tell G, Damante G, Filetti S. Transcriptional regulation of human sodium/iodide symporter gene: a role for redox factor-1. Endocrinology. 2004;145:1290–1293. doi: 10.1210/en.2003-1250. [DOI] [PubMed] [Google Scholar]
- 24.Haugen BR. Redifferentiation therapy in advanced thyroid cancer. Curr Drug Targets Immune Endocr Metabol Disord. 2004;4:175–180. doi: 10.2174/1568008043339811. [DOI] [PubMed] [Google Scholar]
- 25.Simon D, Koehrle J, Reiners C, Boerner AR, Schmutzler C, Mainz K, Goretzki PE, Roeher HD. Redifferentiation therapy with retinoids: therapeutic option for advanced follicular and papillary thyroid carcinoma. World J Surg. 1998;22:569–574. doi: 10.1007/s002689900436. [DOI] [PubMed] [Google Scholar]
- 26.Oh SW, Moon SH, Park do J, Cho BY, Jung KC, Lee DS, Chung JK. Combined therapy with 131I and retinoic acid in Korean patients with radioiodine-refractory papillary thyroid cancer. Eur J Nucl Med Mol Imaging. 2011;38:1798–1805. doi: 10.1007/s00259-011-1849-2. [DOI] [PubMed] [Google Scholar]
- 27.Kimura ET, Nikiforova MN, Zhu Z, Knauf JA, Nikiforov YE, Fagin JA. High prevalence of BRAF mutations in thyroid cancer: genetic evidence for constitutive activation of the RET/PTC-RAS-BRAF signaling pathway in papillary thyroid carcinoma. Cancer Res. 2003;63:1454–1457. [PubMed] [Google Scholar]
- 28.Durante C, Puxeddu E, Ferretti E, Morisi R, Moretti S, Bruno R, Barbi F, Avenia N, Scipioni A, Verrienti A, Tosi E, Cavaliere A, Gulino A, Filetti S, Russo D. BRAF mutations in papillary thyroid carcinomas inhibit genes involved in iodine metabolism. J Clin Endocrinol Metab. 2007;92:2840–2843. doi: 10.1210/jc.2006-2707. [DOI] [PubMed] [Google Scholar]