Abstract
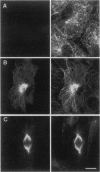
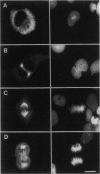
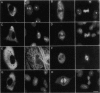
Eg5, a member of the bimC subfamily of kinesin-like microtubule motor proteins, localizes to spindle microtubules in mitosis but not to interphase microtubules. We investigated the molecular basis for spindle localization by transient transfection of Xenopus A6 cells with myc-tagged derivatives of Eg5. Expressed at constitutively high levels from a cytomegalovirus promoter, mycEg5 protein is cytoplasmic throughout interphase, begins to bind microtubules in early prophase, and remains localized to spindle and/or midbody microtubules through mitosis to the end of telophase. Both N- and C-terminal regions of Eg5 are required for this cell-cycle-regulated targeting. Eg5 also contains within its C-terminal domain a sequence conserved among bimC subfamily proteins that includes a potential p34cdc2 phosphorylation site. We show that mutation of a single threonine (T937) within this site to nonphosphorylatable alanine abolishes localization of the mutant protein to the spindle, whereas mutation of T937 to serine preserves spindle localization. We hypothesize that phosphorylation of Eg5 may regulate its localization to the spindle in the cell cycle.
Full text
PDF

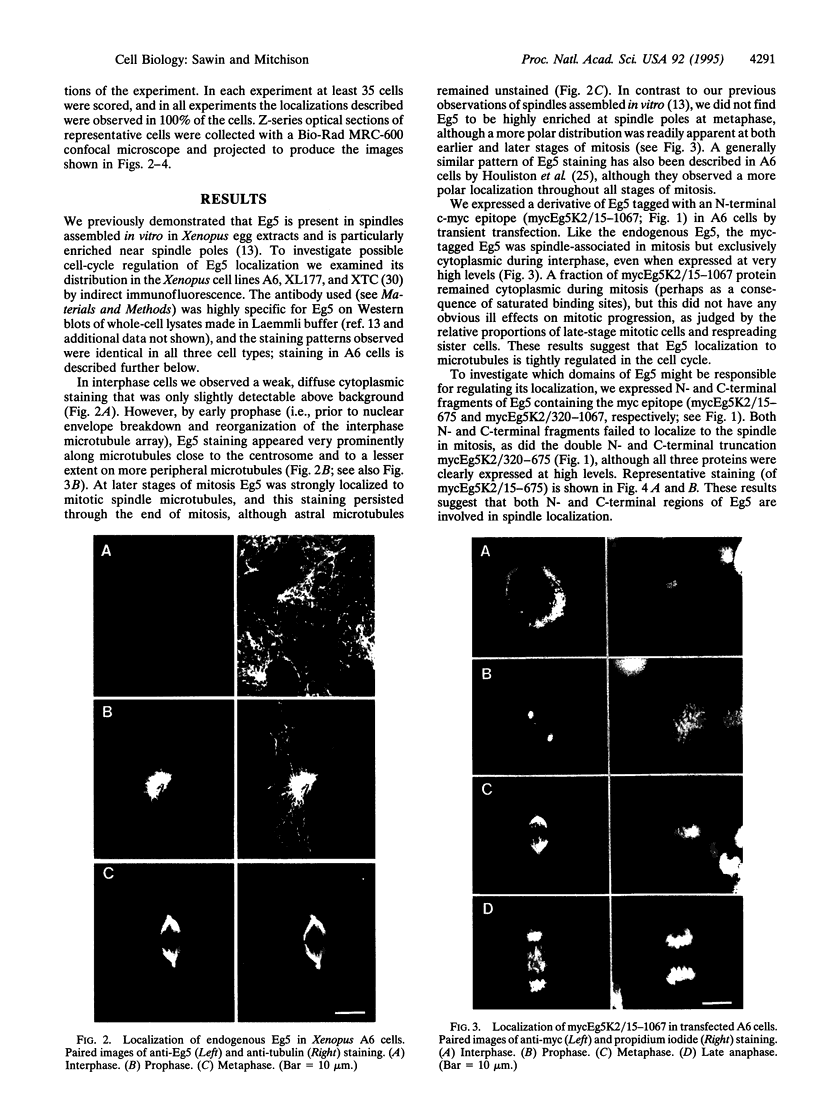
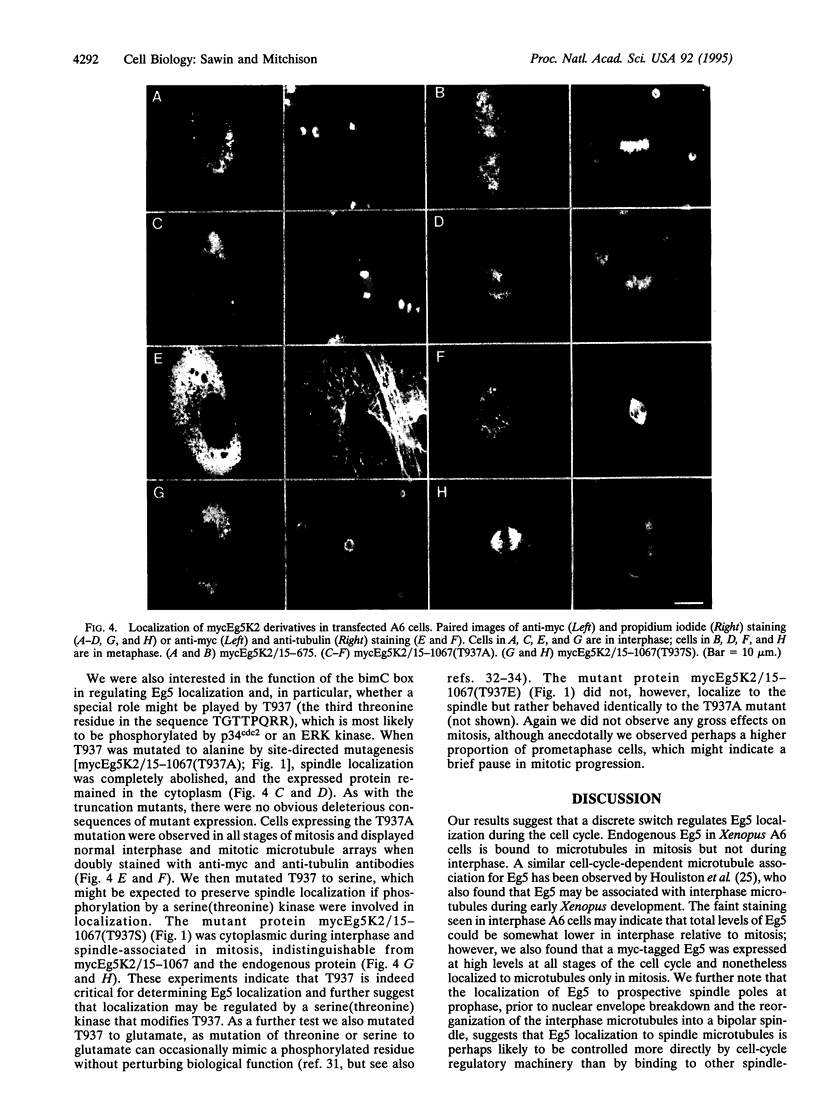

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Clark-Lewis I., Sanghera J. S., Pelech S. L. Definition of a consensus sequence for peptide substrate recognition by p44mpk, the meiosis-activated myelin basic protein kinase. J Biol Chem. 1991 Aug 15;266(23):15180–15184. [PubMed] [Google Scholar]
- Cole D. G., Saxton W. M., Sheehan K. B., Scholey J. M. A "slow" homotetrameric kinesin-related motor protein purified from Drosophila embryos. J Biol Chem. 1994 Sep 16;269(37):22913–22916. [PMC free article] [PubMed] [Google Scholar]
- Connell-Crowley L., Solomon M. J., Wei N., Harper J. W. Phosphorylation independent activation of human cyclin-dependent kinase 2 by cyclin A in vitro. Mol Biol Cell. 1993 Jan;4(1):79–92. doi: 10.1091/mbc.4.1.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crews C. M., Alessandrini A., Erikson R. L. Erks: their fifteen minutes has arrived. Cell Growth Differ. 1992 Feb;3(2):135–142. [PubMed] [Google Scholar]
- Endow S. A., Titus M. A. Genetic approaches to molecular motors. Annu Rev Cell Biol. 1992;8:29–66. doi: 10.1146/annurev.cb.08.110192.000333. [DOI] [PubMed] [Google Scholar]
- Enos A. P., Morris N. R. Mutation of a gene that encodes a kinesin-like protein blocks nuclear division in A. nidulans. Cell. 1990 Mar 23;60(6):1019–1027. doi: 10.1016/0092-8674(90)90350-n. [DOI] [PubMed] [Google Scholar]
- Evan G. I., Lewis G. K., Ramsay G., Bishop J. M. Isolation of monoclonal antibodies specific for human c-myc proto-oncogene product. Mol Cell Biol. 1985 Dec;5(12):3610–3616. doi: 10.1128/mcb.5.12.3610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodson H. V., Kang S. J., Endow S. A. Molecular phylogeny of the kinesin family of microtubule motor proteins. J Cell Sci. 1994 Jul;107(Pt 7):1875–1884. doi: 10.1242/jcs.107.7.1875. [DOI] [PubMed] [Google Scholar]
- Gould K. L., Moreno S., Owen D. J., Sazer S., Nurse P. Phosphorylation at Thr167 is required for Schizosaccharomyces pombe p34cdc2 function. EMBO J. 1991 Nov;10(11):3297–3309. doi: 10.1002/j.1460-2075.1991.tb04894.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hackney D. D., Levitt J. D., Suhan J. Kinesin undergoes a 9 S to 6 S conformational transition. J Biol Chem. 1992 Apr 25;267(12):8696–8701. [PubMed] [Google Scholar]
- Hagan I., Yanagida M. Kinesin-related cut7 protein associates with mitotic and meiotic spindles in fission yeast. Nature. 1992 Mar 5;356(6364):74–76. doi: 10.1038/356074a0. [DOI] [PubMed] [Google Scholar]
- Hagan I., Yanagida M. Novel potential mitotic motor protein encoded by the fission yeast cut7+ gene. Nature. 1990 Oct 11;347(6293):563–566. doi: 10.1038/347563a0. [DOI] [PubMed] [Google Scholar]
- Heck M. M., Pereira A., Pesavento P., Yannoni Y., Spradling A. C., Goldstein L. S. The kinesin-like protein KLP61F is essential for mitosis in Drosophila. J Cell Biol. 1993 Nov;123(3):665–679. doi: 10.1083/jcb.123.3.665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houliston E., Le Guellec R., Kress M., Philippe M., Le Guellec K. The kinesin-related protein Eg5 associates with both interphase and spindle microtubules during Xenopus early development. Dev Biol. 1994 Jul;164(1):147–159. doi: 10.1006/dbio.1994.1187. [DOI] [PubMed] [Google Scholar]
- Hoyt M. A. Cellular roles of kinesin and related proteins. Curr Opin Cell Biol. 1994 Feb;6(1):63–68. doi: 10.1016/0955-0674(94)90117-1. [DOI] [PubMed] [Google Scholar]
- Hoyt M. A., He L., Loo K. K., Saunders W. S. Two Saccharomyces cerevisiae kinesin-related gene products required for mitotic spindle assembly. J Cell Biol. 1992 Jul;118(1):109–120. doi: 10.1083/jcb.118.1.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- L'Allemain G., Her J. H., Wu J., Sturgill T. W., Weber M. J. Growth factor-induced activation of a kinase activity which causes regulatory phosphorylation of p42/microtubule-associated protein kinase. Mol Cell Biol. 1992 May;12(5):2222–2229. doi: 10.1128/mcb.12.5.2222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Guellec R., Paris J., Couturier A., Roghi C., Philippe M. Cloning by differential screening of a Xenopus cDNA that encodes a kinesin-related protein. Mol Cell Biol. 1991 Jun;11(6):3395–3398. doi: 10.1128/mcb.11.6.3395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levin L. R., Zoller M. J. Association of catalytic and regulatory subunits of cyclic AMP-dependent protein kinase requires a negatively charged side group at a conserved threonine. Mol Cell Biol. 1990 Mar;10(3):1066–1075. doi: 10.1128/mcb.10.3.1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao H., Li G., Yen T. J. Mitotic regulation of microtubule cross-linking activity of CENP-E kinetochore protein. Science. 1994 Jul 15;265(5170):394–398. doi: 10.1126/science.8023161. [DOI] [PubMed] [Google Scholar]
- Moreno S., Nurse P. Substrates for p34cdc2: in vivo veritas? Cell. 1990 May 18;61(4):549–551. doi: 10.1016/0092-8674(90)90463-o. [DOI] [PubMed] [Google Scholar]
- Navone F., Niclas J., Hom-Booher N., Sparks L., Bernstein H. D., McCaffrey G., Vale R. D. Cloning and expression of a human kinesin heavy chain gene: interaction of the COOH-terminal domain with cytoplasmic microtubules in transfected CV-1 cells. J Cell Biol. 1992 Jun;117(6):1263–1275. doi: 10.1083/jcb.117.6.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Connell M. J., Meluh P. B., Rose M. D., Morris N. R. Suppression of the bimC4 mitotic spindle defect by deletion of klpA, a gene encoding a KAR3-related kinesin-like protein in Aspergillus nidulans. J Cell Biol. 1993 Jan;120(1):153–162. doi: 10.1083/jcb.120.1.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Page B. D., Satterwhite L. L., Rose M. D., Snyder M. Localization of the Kar3 kinesin heavy chain-related protein requires the Cik1 interacting protein. J Cell Biol. 1994 Feb;124(4):507–519. doi: 10.1083/jcb.124.4.507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roof D. M., Meluh P. B., Rose M. D. Kinesin-related proteins required for assembly of the mitotic spindle. J Cell Biol. 1992 Jul;118(1):95–108. doi: 10.1083/jcb.118.1.95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawin K. E., Endow S. A. Meiosis, mitosis and microtubule motors. Bioessays. 1993 Jun;15(6):399–407. doi: 10.1002/bies.950150606. [DOI] [PubMed] [Google Scholar]
- Sawin K. E., LeGuellec K., Philippe M., Mitchison T. J. Mitotic spindle organization by a plus-end-directed microtubule motor. Nature. 1992 Oct 8;359(6395):540–543. doi: 10.1038/359540a0. [DOI] [PubMed] [Google Scholar]
- Sawin K. E., Mitchison T. J. Microtubule flux in mitosis is independent of chromosomes, centrosomes, and antiparallel microtubules. Mol Biol Cell. 1994 Feb;5(2):217–226. doi: 10.1091/mbc.5.2.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawin K. E., Mitchison T. J. Mitotic spindle assembly by two different pathways in vitro. J Cell Biol. 1991 Mar;112(5):925–940. doi: 10.1083/jcb.112.5.925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawin K. E., Scholey J. M. Motor proteins in cell division. Trends Cell Biol. 1991 Nov;1(5):122–129. doi: 10.1016/0962-8924(91)90117-r. [DOI] [PubMed] [Google Scholar]
- Schroer T. A. Structure, function and regulation of cytoplasmic dynein. Curr Opin Cell Biol. 1994 Feb;6(1):69–73. doi: 10.1016/0955-0674(94)90118-x. [DOI] [PubMed] [Google Scholar]
- Smith J. C., Tata J. R. Xenopus cell lines. Methods Cell Biol. 1991;36:635–654. [PubMed] [Google Scholar]
- Toyoshima I., Yu H., Steuer E. R., Sheetz M. P. Kinectin, a major kinesin-binding protein on ER. J Cell Biol. 1992 Sep;118(5):1121–1131. doi: 10.1083/jcb.118.5.1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trybus K. M., Lowey S. Conformational states of smooth muscle myosin. Effects of light chain phosphorylation and ionic strength. J Biol Chem. 1984 Jul 10;259(13):8564–8571. [PubMed] [Google Scholar]
- Vulliet P. R., Hall F. L., Mitchell J. P., Hardie D. G. Identification of a novel proline-directed serine/threonine protein kinase in rat pheochromocytoma. J Biol Chem. 1989 Sep 25;264(27):16292–16298. [PubMed] [Google Scholar]