Abstract
Background: High intake of cow-milk protein in formula-fed infants is associated with higher weight gain and increased adiposity, which have led to recommendations to limit protein intake in later infancy. The impact of protein from meats for breastfed infants during complementary feeding may be different.
Objective: We examined the effect of protein from meat as complementary foods on growth and metabolic profiles of breastfed infants.
Design: This was a secondary analysis from a trial in which exclusively breastfed infants (5–6 mo old from the Denver, CO, metro area) were randomly assigned to receive commercially available pureed meats (Meat group; n = 14) or infant cereal (Cereal group; n = 28) as their primary complementary feedings for ∼5 mo. Anthropometric measures and diet records were collected monthly from 5 to 9 mo of age; intakes from complementary feeding and breast milk were assessed at 9 mo of age.
Results: The Meat group had significantly higher protein intake, whereas energy, carbohydrate, and fat intakes from complementary feeding did not differ by group over time. At 9 mo of age, mean (±SEM) intakes of total (complementary feeding plus breast milk) protein were 2.9 ± 0.6 and 1.4 ± 0.4 g · kg−1 · d−1, ∼17% and ∼9% of daily energy intake, for Meat and Cereal groups, respectively (P < 0.001). From 5 to 9 mo of age, the weight-for-age z score (WAZ) and length-for-age z score (LAZ) increased in the Meat group (ΔWAZ: 0.24 ± 0.19; ΔLAZ: 0.14 ± 0.12) and decreased in the Cereal group (ΔWAZ: −0.07 ± 0.17; ΔLAZ: −0.27 ± 0.24) (P-group by time < 0.05). The change in weight-for-length z score did not differ between groups. Total protein intake at 9 mo of age and baseline WAZ were important predictors of changes in the WAZ (R2 = 0.23, P = 0.01).
Conclusion: In breastfed infants, higher protein intake from meats was associated with greater linear growth and weight gain but without excessive gain in adiposity, suggesting that potential risks of high protein intake may differ between breastfed and formula-fed infants and by the source of protein.
INTRODUCTION
The rate of weight gain in early infancy, specifically in the first year of life, has critical influences on the obesity trajectory in childhood that carries on to adulthood (1). Infant dietary intakes have a significant impact on infant growth and weight gain. Some observational studies (2–4) showed a greater weight gain in formula-fed than breastfed infants. In one study, breastfed and formula-fed infants were followed over the first year of life (5), and the average weight of breastfed infants was significantly lower than that of formula-fed infants after 6 mo of age. Because formula (∼1.5 g/100 mL) has a higher content of protein than breast milk (∼0.9 g/100 mL) (6), the difference in protein intakes has been considered a potential key contributor to the greater weight gain in formula-fed infants. A proposed potential mechanism includes the stimulation of insulin-like growth factor I (IGF-I)5 (7). The correlation between weight gain and protein intake has been reported in breastfed infants and a greater weight gain in formula fed compared with breast fed infants during the first year of life (8). Recently, Koletzko et al (9) conducted a large, randomized, controlled trial to examine the protein quantity on infant growth by using an isocaloric infant formula with different cow milk–based protein contents and reported a dose-dependent correlation between protein and weight gain. In a subsample from the study, the high-protein group also showed higher fat mass and possibly higher adiposity (10). The sum of these and other findings has led to recommendations to limit protein to <15% of energy during later infancy and the second year of life (11). Although dairy protein has been acknowledged to potentially pose more risk of excessive weight gain than meat protein is, recommendations have not distinguished between the protein source or between formula fed and breastfed infants.
Complementary foods refer to nutrient- and energy-containing solid or semisolid foods fed to infants in addition to human milk or formula (12) and are typically introduced between 4–6 mo of life. The complementary feeding period includes the largest proportion of the 1000-d critical window and may, thus, be a potentially critical period to influence obesity risks. The protein content of human milk gradually declines over the course of lactation in contrast to the higher and unchanging protein content of infant formula. For infants who are breastfed only (no formula), meats not only are a good source of high-quality protein but also provide highly bioavailable iron and zinc at a time when human milk alone no longer meets the needs of infants (13). Very limited evidence is available to evaluate the effect of protein quantity specifically from complementary foods on the growth of breastfed infants. Thus, the purpose of this report was to examine the effect of a lower- compared with higher-protein complementary feeding regimen, with animal flesh being the primary protein source, on infant growth and metabolic profile in older breastfed-only infants.
SUBJECTS AND METHODS
Study design
This study represents a secondary data analysis of a randomized control trial that was originally designed to compare the effect of fortified cereal–based compared with meat-based complementary foods on zinc homeostasis and iron status in breastfed-only infants. Primary outcomes of this study have been published elsewhere (13, 14). Exclusively breastfed infants who were 5 mo old were assigned to receive either commercially prepared pureed meats or fortified infant cereals. These foods served as the first complementary food and were continued with 1–2 servings/d throughout the remainder of the study. Monthly visits were conducted to assess dietary compliance, obtain anthropometric measurements, and record infant morbidity. In addition, between 9 and 10 mo of age, each infant had his or her blood drawn to assess biochemical markers during a metabolic testing period. Daily intakes of breast milk and complementary foods were also obtained during the metabolic period.
Subjects
Forty-two infants (18 boys and 27 girls) between the ages of 5 and 6 mo from the Denver metropolitan area were enrolled and completed the study between January 2008 and September 2010. Infants were included in the study if the mother could show an intent or history of exclusive breastfeeding (no formula use) through 1 y and the infant was born at term (37–42 wk of gestation) with birth weight appropriate for gestational age. Infants were excluded if they had current or planned formula use, had low birth weight, used vitamin-mineral supplements (except vitamin D), or had significant congenital anomalies or known chronic conditions that would affect feeding, growth, or the developmental potential. The study was approved by the Colorado Multiple Institutional Review Board, and written and informed consent was obtained from mothers or fathers of infants. The sample-size calculation was based on zinc-absorption data in infants (13).
Dietary intervention
Breastfed infants were randomly assigned to receive 1 of 3 complementary dietary regimens previously described [1) meat, 2) iron- and zinc-fortified cereal, or 3) iron-only–fortified cereal]. For the purpose of this secondary analysis, the 2 cereal groups (2 and 3) were combined and considered the lower-protein complementary feeding group (the Cereal group), and the meat group (1) was considered the higher-protein complementary feeding group (the Meat group). Parents were asked to introduce complementary feeding at 6 mo of age with the assigned food. Main dietary components (cereal or meat purees) were provided at monthly visits, and mothers were given monthly written guidelines on appropriate amounts for the age of the infant. The Meat group was encouraged to aim for the consumption of one jar of pureed meat and gravy (71 g total, equivalent to 8 g protein) per day by 7 mo of age and 1–2 jars/d by 9 mo of age. The Cereal group was encouraged to offer 1 serving/d (15 g dry weight according to the manufacturer's label) by 7 mo of age and 2 servings/d by 9 mo of age. Infants in the Cereal group were instructed to avoid single-ingredient meats. For all groups, unfortified fruit, vegetables, yogurt, and cheese were allowed ad libitum as developmentally appropriate. Three-day diet records were completed by mothers before monthly visits (6, 7, 8, and 9 mo) and were reviewed with the research coordinator at each visit.
Between 9 and 10 mo of age, while infants underwent a metabolic study to measure zinc absorption, weighed duplicate diets (adjusted for plate waste and losses) were collected for 5 d. Breast-milk intake was measured by test weighing for 4 consecutive days (13). Energy and macronutrient intakes from complementary foods were calculated by a registered dietitian at the Clinical Translational Research CenterBio-nutrition Unit by using the Nutrient Data System for Research (University of Minnesota) dietary analyses program. The average energy density of human milk was estimated from the following equation:
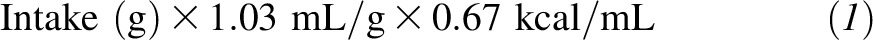 |
The average macronutrient composition of human milk was assumed to be as follows: 35 g fat/L, 68 g carbohydrate/L, and 9 g protein/L (15).
Anthropometric measurements
Length, weight, head circumference, and waist circumference were measured at 5 mo of age and at each subsequent monthly visit (6, 7, 8, and 9 mo). All measurements were performed in duplicate by trained research personnel. Length was measured in a recumbent position by using an infant stadiometer accurate to 0.1 cm (Holtain Ltc). An electronic digital balance (Sartorious Corp) was used to obtain naked infant weight. z scores were calculated on the basis of WHO/CDC breastfed-infant growth standards (16).
Sample collection and analyses
Morning blood samples collected were immediately centrifuged and stored at – 80°C until analyzed. The following markers were analyzed by the Colorado Clinical and Translational Science Institute's Core Laboratory: IGF-I (chemilluminescence; DiaSorin Liaison); IGF-I binding protein 3 (chemilluminescence; Siemen); TNF-α (ELISA; R&D Systems); IL-6 (ELISA; R&D Systems); leptin (radioimmunoassay; Millipore); glucose (hexokinase, ultraviolet; Beckman Coulter); insulin (radioimmunoassay; Millipore), blood urea nitrogen, HDL (enzymatic; Beckman Coulter), and triglyceride (enzymatic; Beckman Coulter). More information of sample processing has been published elsewhere (13).
Statistical analysis
A power analysis was based on the primary outcome of the original protocol to test total absorbed zinc per day between the 3 complementary feeding groups. A sample size of 15 per group yielded 82–93% power (α = 0.05; 2 tail) to detect a difference of 0.20 mg in total daily absorbed zinc (13).
Statistical analyses were performed with SAS software (version 9.3; SAS Institute Inc). Group data are presented as means (±SDs). Baseline variables were compared by using an independent Student's t test between Cereal and Meat groups, and variables that were different at baseline were included in additional analyses. Sex was tested as a categorical variable in the subsequent analysis, and results remained unaffected. Repeated-measures ANOVA (PROC GLM procedure; SAS Institute Inc) were used to evaluate the main effects of time, group, and their interactions on dependent variables. An independent Student's t test was used to compare values between groups as a post hoc analysis. Equal variance was checked by using Levene's test. The Wilcoxon-Mann-Whitney test was conducted when the sample was not normally distributed. A linear regression model was used between total protein intake at 9 mo of age and the change of the weight-for-age z score (WAZ) and length-for-age z score (LAZ) over time, with baseline WAZ and LAZ included, respectively. P < 0.05 was considered statistically significant.
RESULTS
Dietary intakes
Forty-two subjects (17 boys; 25 girls) completed the study with 28 subjects in the Cereal group and 14 subjects in the Meat group. The detailed timing of recruitment was published elsewhere (13). No adverse effects were reported from any feeding regimens. Birth weight (the Meat group: 3.41 ± 0.32 kg; the Cereal group: 3.52 ± 0.29 kg) and maternal BMI (in kg/m2) (Meat: 23 ± 2; Cereal: 24 ± 3) were not different between groups. Dietary intakes from complementary foods that were based on monthly 3-d diet records collected monthly (6, 7, 8, and 9 mo of age) are presented in Table 1. Energy, carbohydrate, and fat intakes increased over time from 6 to 8 mo of age with no effect of group (P-effect of time < 0.05); intakes did not differ between 8 and 9 mo. There was a group-by-time interaction of protein intake (g/d or g · kg−1 · d−1) whereby protein intake increased over time from 6 to 8 mo with the Meat group consuming more protein at all intervention time points (Table 1; P-group by time < 0.01). In addition, fat intake (g/d) was positively correlated with protein intake (g/d; P < 0.0001). Carbohydrate and fat intakes as percentages of energy from complementary feeding were not affected by time or group, whereas the percentage of protein intake was consistently higher in the Meat group (Table 1). Complete dietary intake during the metabolic testing period, including breast-milk consumption, indicated virtually identical energy intakes for the 2 groups but with differences in macronutrient contributions to energy intake (Table 2). Protein intake was higher in the Meat group, and the percentage of energy from carbohydrate was higher in the Cereal group; fat intakes did not differ by group (Table 2).
TABLE 1.
Complementary food intakes from 3-d diet records1
| Age |
||||
| Variable and group | 6 mo | 7 mo | 8 mo | 9 mo |
| Energy (kcal/d)2 | ||||
| Meat | 168 ± 180a | 274 ± 238b | 431 ± 225c | 440 ± 217c |
| Cereal | 129 ± 115a | 189 ± 165b | 314 ± 215c | 331 ± 104c |
| Percentage of carbohydrate3 | ||||
| Meat | 38 ± 14 | 43 ± 12 | 48 ± 11 | 50 ± 12 |
| Cereal | 66 ± 14 | 71 ± 12 | 69 ± 9 | 69 ± 9 |
| Percentage of protein4 | ||||
| Meat | 24 ± 10 | 24 ± 11 | 24 ± 11 | 23 ± 5 |
| Cereal | 8 ± 1 | 8 ± 2 | 10 ± 2 | 11 ± 4 |
| Percentage of fat2 | ||||
| Meat | 38 ± 12a | 35 ± 10b | 30 ± 10b | 29 ± 9b |
| Cereal | 27 ± 16a | 22 ± 13b | 22 ± 10b | 22 ± 10b |
| Carbohydrate (g/d)2 | ||||
| Meat | 19 ± 24a | 30 ± 29b | 52 ± 32c | 56 ± 33c |
| Cereal | 19 ± 15a | 31 ± 23b | 31 ± 23c | 56 ± 18c |
| Protein (g/d)5 | ||||
| Meat | 7.8 ± 6.8a | 15.3 ± 13.0b | 24.3 ± 11.6c | 24.2 ± 10.4c |
| Cereal | 2.4 ± 2.0a | 3.7 ± 3.1b | 7.3 ± 4.6c | 8.8 ± 3.2c |
| Protein (g · kg−1 · d−1)5 | ||||
| Meat | 1.12 ± 1.10a | 2.00 ± 1.66b | 3.07 ± 1.61c | 2.96 ± 1.34c |
| Cereal | 0.32 ± 0.26a | 0.47 ± 0.37b | 0.92 ± 0.54c | 1.08 ± 0.42c |
| Fat (g/d)2 | ||||
| Meat | 7.2 ± 8.3a | 11.3 ± 10.5a | 14.7 ± 9.4b | 14.2 ± 7.8b |
| Cereal | 5.0 ± 6.0a | 6.0 ± 8.3a | 9.2 ± 11.3b | 8.4 ± 5.7b |
All values are means ± SDs. Meat group: n = 14; Cereal group: n = 28. A repeated-measures ANOVA was used to test the effect of group and time. Cereal group, lower-protein group; Meat group, higher-protein group.
Significant change over time (P < 0.05); values with different superscript letters were significantly different (P < 0.05).
No effect of time or group.
The Meat group was higher than the Cereal group at all time points (P < 0.0001); there was no change over time.
Significant group-by-time interaction (P < 0.01): the Meat group had higher protein intake than the Cereal group at all time points; protein intakes increased over time in both groups from 6 to 8 mo of age. Values with different superscript letters were significantly different.
TABLE 2.
Complete dietary intakes between 9 and 10 mo of age on the basis of 5-d weighed dietary intakes including breast milk1
| Variable | Meat | Cereal |
| Energy from CF (kcal/d) | 316 ± 97 | 277 ± 113 |
| Energy from BM (kcal/d) | 240 ± 86 | 293 ± 125 |
| Energy from CF + BM (kcal/d) | 556 ± 127 | 570 ± 151 |
| Energy from CF + BM (kcal · kg−1 · d−1) | 66 ± 17 | 68 ± 15 |
| Carbohydrate from CF + BM (g/d) | 65 ± 24 | 78 ± 16 |
| Protein from CF + BM (g/d)2 | 23 ± 5 | 12 ± 4 |
| Protein from CF + BM (g · kg−1 · d−1)2 | 2.7 ± 0.6 | 1.4 ± 0.4 |
| Fat from CF + BM (g/d) | 22 ± 4 | 22 ± 6 |
| Carbohydrate from CF + BM (%)2 | 46 ± 8 | 55 ± 9 |
| Protein from CF + BM (%)2 | 17 ± 4 | 8 ± 2 |
| Fat from CF + BM (%) | 37 ± 5 | 35 ± 6 |
All values are means ± SDs. Meat group: n = 14; Cereal group: n = 28. An independent Student's t test was used to compare groups. BM, breast milk; Cereal group, lower-protein group; CF, complementary feeding; Meat group, higher-protein group.
Significant difference between the Meat and Cereal groups (P < 0.001).
Infant growth
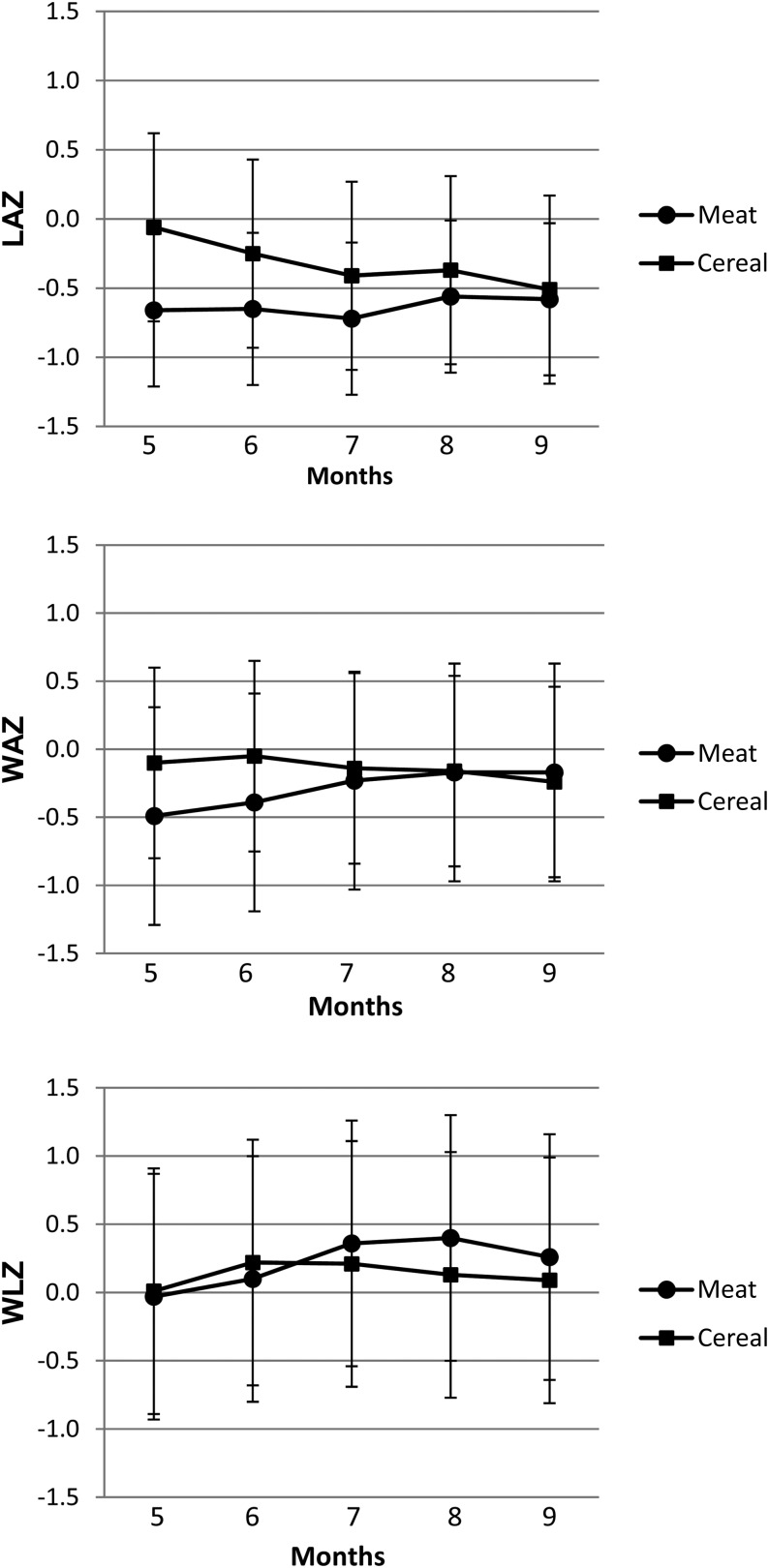
Infant growth longitudinal z scores are presented in Figure 1. The mean LAZ in the Cereal group was higher at baseline (5 mo of age; P = 0.03). This difference disappeared after 6 mo of age. A group-by-time interaction was observed for WAZs (P < 0.05) whereby WAZs increased in the Meat group and decreased in the Cereal group. There was no significant effect of time or group on weight-for-length z scores (WLZs) or BMI z scores, which indicated proportional gain in weight relative to length (see Supplemental Table 1 under “Supplemental data” in the online issue). Waist circumference increased from baseline (41 ± 2 cm) to postintervention (43 ± 3 cm) for all participants (P < 0.05).
FIGURE 1.
Mean (±SD) longitudinal z scores of anthropometric measures by group. Meat group: n = 14; Cereal group: n = 28. A repeated-measures ANOVA used group (Meat group compared with Cereal group) and time (pre compared with post) as the 2 variables and showed a group-by-time interaction for both the LAZ and WAZ (P < 0.05) as follows: the LAZ decreased in the Cereal group, and the WAZ increased in the Meat group and decreased in the Cereal group. Cereal group, lower-protein group; LAZ, length-for-age z score; Meat group, higher-protein group; WAZ, weight-for-age z score; WLZ, weight-for-length z score.
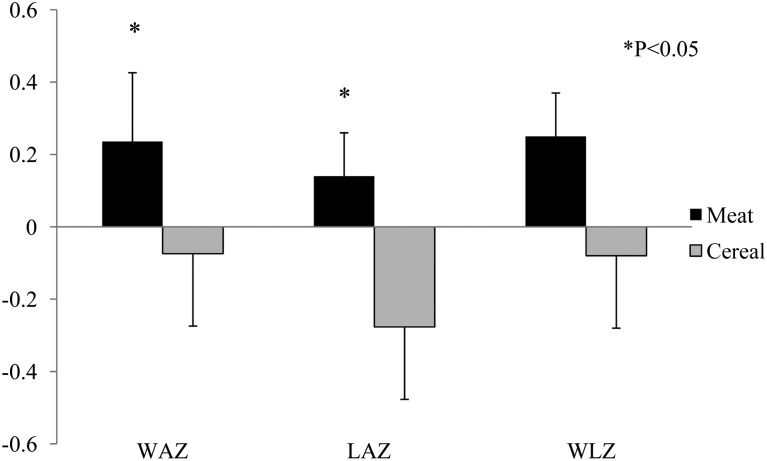
Changes of growth z scores (LAZ, WAZ, and WLZ) from baseline (5 mo of age) to the end of the study (9 mo of age) between Meat and Cereal groups are shown in Figure 2. Significance between groups was observed for changes of the WAZ and LAZ. A linear regression also showed that total protein intake (g · kg−1 · d−1) at 9 mo of age (complementary feeding plus breast milk) was a significant predictor of WAZ (P = 0.02) and LAZ (P = 0.09) changes over time. There was a significant linear relation (P = 0.01, R2 = 0.23) between total protein intake at 9 mo of age (P = 0.02) and changes of the WAZ over time with the baseline WAZ included in the model (P = 0.05):
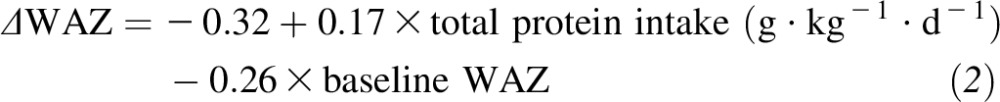 |
However, the relation between total protein intake at 9 mo of age and the ΔLAZ was only borderline significant (P = 0.06), and the baseline LAZ was not a significant variable for this model (P = 0.36).
FIGURE 2.
Mean (±SD) changes in WAZ, LAZ, and WLZ over time (5–9 mo of age) between groups. Meat group: n = 14; Cereal group: n = 28. A 1-factor ANOVA of group and baseline (5 mo of age) LAZs or WAZs as covariates showed that changes in the WAZ and LAZ were both different between groups (P < 0.05). Mean values presented for WAZs and LAZs were adjusted means. Cereal group, lower-protein group. LAZ, length-for-age z score; Meat group, higher-protein group; WAZ, weight-for-age z score; WLZ, weight-for-length z score.
Blood biomarkers
Results of blood biomarkers at 9 mo of age are presented in Table 3. There was no difference between groups; a trend of a higher leptin concentration in the Meat group was observed (P = 0.08).
TABLE 3.
Blood biomarkers at 9 mo1
| Variable | Meat | Cereal |
| IGF-I (ng/mL) | 30 ± 12 (11) | 29 ± 17 (17) |
| IGF-I BP3 | 1.70 ± 0.34 (8) | 1.73 ± 0.55 (20) |
| TNF-α (pg/mL) | 17 ± 5 (11) | 16 ± 5 (17) |
| IL-6 (pg/mL) | 4.1 ± 2.4 (6) | 3.9 ± 3.0 (12) |
| Leptin2 (ng/mL) | 2.90 ± 0.88 (7) | 2.24 ± 0.83 (20) |
| Glucose (mg/dL) | 80 ± 6 (7) | 83 ± 7 (19) |
| Insulin (μU/mL) | 11 ± 9 (8) | 11 ± 5 (18) |
| HDL (mg/dL) | 46 ± 13 (13) | 41 ± 6 (25) |
| Triglyceride (mg/dL) | 124 ± 59 (13) | 170 ± 132 (25) |
All values are means ± SDs; numbers of samples available are in parentheses. An independent Student's t test was used to compare groups. Cereal, lower-protein group; IGF-I, insulin-like growth factor I; IGF-I BP3, insulin-like growth factor I binding protein 3; Meat group, higher-protein group.
P-trend of difference between groups = 0.08.
DISCUSSION
This secondary analysis of a randomized trial of breastfed infants in a Westernized setting resulted in 2 major findings. First, compared with a lower-protein, cereal-based diet, high protein intake provided primarily from meats as complementary foods from 6 to 9 mo of age was associated with greater linear growth and proportional weight gain (Figure 2). Second, despite significantly different distributions of macronutrient intakes at 9 mo of age, the combined intake of breast milk and complementary foods resulted in caloric intakes that were virtually indistinguishable between the 2 groups, which reinforced the notion of the ability of breastfed infants to effectively regulate energy intake. Furthermore, these data suggest that a higher intake of protein from meats can be safely recommended for breastfed infants for whom rich sources of bioavailable zinc and iron are particularly important during the complementary feeding period (13, 14, 17).
Results of studies that compared the effects of animal protein source on children's linear growth and development have been mixed with milk protein being cited as having more of an effect on linear growth and meat associated with greater lean body mass (18). Randomized controlled trials to test the effect of different complementary feeding regimens in breastfed infants in high-resource settings have been especially limited. An earlier short-term randomized trial in breastfed-only infants showed good acceptance of meat as an early complementary food, but compared with a traditional iron-fortified-cereal–based regimen, no impact on linear or ponderal growth at 12 mo of age was detected. However, the interpretation was limited by the very short duration of the intervention of ∼4–6 wk (19). Controlled trials of meats as complementary foods in low-resource settings, including those with high stunting rates, have reported modest or no effects on linear growth (20–22). These result may reflect the limits of a food-based approach to affect growth in older infants with early and severe stunting and a greater likelihood of nonnutritional factors affecting postnatal growth. Data from the current study suggest that dietary quality, including the amount of protein intake, may positively affect linear growth in older breastfed-only infants in the United States. In contrast, for those infants who consumed the conventional, cereal-based diet, the LAZ steadily decreased over the intervention period (Figure 1), which suggested that the cereal-based diet may not have optimally supported linear growth in these breastfed infants.
Protein has been considered to be a risk factor for excessive weight gain in infancy, which may be associated with increased obesity risk later in life (23). The current analysis showed a significant increase in the WAZ in the Meat group, but changes in the WAZ and LAZ were highly correlated and should not be viewed separately. The LAZ increase in the Meat group was likely to have been the driver for the significantly greater increase in WAZ. No difference was observed between groups for adiposity as represented by weight-for-length (WLZ), BMI, or waist circumference. In addition, infants from both Meat and Cereal groups had mean WAZs and LAZs <0 throughout the intervention (Figure 1); the mean WLZ was modestly >0 and did not differ between groups. Together, the data do not suggest that these infants had gained excessive weight or adiposity. However, body-composition measurements were not obtained and would have been necessary to directly examine adiposity differences. Emerging evidence suggests that the protein source is an important factor in infant growth regulation, possibly mediated through the IGF-I axis (24). Günther et al (25) followed 203 participants from 6 mo to 7 y with diet information at multiple time points and showed that early dairy protein intake, not meat intake, at 12 mo of age was positively correlated with fatness at 7 y. In the current study, IGF-I and IGF-I binding protein 3 were not different between groups, but this nonsignificance could have been attributable to a lack of power. Although the current study was not designed to compare protein sources (ie, by controlling the quantity to compare the source), it agrees with previous findings that meat is not associated with IGF-I stimulation in infants.
The optimal protein intake for infants has been under debate for a long time, partially because of difficulties in conducting protein-requirement research in this particular population (26). The 2007 WHO/FAO recommendation of protein for 6-mo-old infants is 7.8% of energy for boys and 7.6% of energy for girls (11). The Feeding Infants and Toddler Study reported, for US infants aged 6–11 mo, mean protein intake ∼10% total energy (∼22 g/d) similar to intake of the Meat group at 24 g/d (27). However, because of lower energy intake of infants in the current study, protein intake constituted a higher percentage of energy at 17%. Nonetheless, infants from both Meat and Cereal groups consumed higher amounts of protein than the Adequate Intake (11g/d) and WHO recommendations (28).
Participating infants were allowed to consume ad libitum amounts of complementary foods and breast milk. Although the Meat group consumed a significantly greater amount of protein, total energy intake (kcal · kg−1 · d−1) was essentially the same between groups (Table 2). Notably, the average total caloric intake for both groups at 9 mo of age was between 85% and 90% of the estimated energy requirement for 7–12-mo-olds (28). Significantly higher protein intake from complementary foods in the Meat group was associated with a tendency for higher fat and caloric intakes, although neither intake was significantly different between groups. Dietary data at 9–10 mo of age suggested that this intake was compensated by lower breast-milk and carbohydrate consumption in the Meat group, which resulted in comparable total energy intakes between groups. The estimated total energy intake (66–68 kcal · kg−1 · d−1) was relatively lower than the WHO energy requirement for 9-mo-old breastfed infants (77 kcal · kg−1 · d−1) (29). This finding is consistent with previous reports that breastfed infants are more likely to self-regulate energy intake with a reduction in breast-milk intake as complementary foods are introduced. An equivalent downregulation of formula intake is less predictable and presents higher risk of excessive caloric intake (8). Consistent with reduced self-regulation is the reported estimated energy intake of predominantly formula-fed 6–11-mo-old infants in the Feeding Infants and Toddler Study, which was >20% above the estimated energy requirement for age (27).
This study was not without limitations. First, this was a secondary analysis of a clinical trial designed to measure zinc homeostasis in breastfed infants, and the power calculation was based on the primary outcome (zinc absorption). Thus, the sample size was relatively small and may have been underpowered to detect potential differences between groups. For example, we only had 50% and 60% of power to test the potential difference between changes of the LAZ and WAZ, respectively, between groups. Thus, any firm conclusions need to be made with caution, and larger prospective trials are needed to confirm these findings. A second limitation of the study was that body composition, specifically fat mass, was not measured in participants. Body-composition measurements provide an important additional insight compared with that for growth data because fat mass and fat-free mass play completely different roles in health and metabolic regulation such as insulin resistance. Future studies investigating the impact of complementary feeding choices on growth and obesity risk should include reliable methods of body-composition measurement (30). In addition, because only breastfed infants were studied, it was not clear whether formula-fed infants would have followed a different growth pattern when exposed to the same complementary feeding regimens. The strengths of this study were as follows: 1) the study was a randomized controlled trial in contrast to most research available for the complementary feeding period; 2) breast milk intake was objectively measured and counted as part of total energy and protein intake; 3) blood biomarkers that could contribute to the potential differential growth and metabolic responses were measured; and 4) this study was conducted in a Westernized population.
In conclusion, our findings suggest that a high-protein complementary diet, with meat as the primary source of protein, increases the linear growth in breastfed-only infants without showing excessive weight gain or adiposity. This growth is in comparison with growth of breastfed-only infants consuming a more traditional feeding pattern with fortified infant cereal as the main complementary food. The 2 complementary feeding regimens differed substantially in macronutrient distributions, but differences in energy intakes from complementary foods appeared to be balanced by altered intakes of breast milk. This adjustment resulted in very similar total energy intakes, which averaged modestly less than the estimated energy requirement (28). In contrast to dairy products, the value of meats to address the challenge of meeting iron and zinc needs for older breastfed infants is well recognized. Findings from this study require additional confirmation, but they reinforce the potential value of introducing flesh foods early and differences in nutritional needs and risks between formula-fed and breastfed infants. Accordingly, recommendations for complementary feeding should recognize these distinctions.
Acknowledgments
We thank the following co-investigators who contributed to the conduct of the primary trial: Diana Culberston, Jamie Westcott, and K Michael Hambidge. Likewise, we gratefully acknowledge the dedication and cooperation of participating infants and their parents.
The authors’ responsibilities were as follows—MT and NFK: analyzed data and wrote the manuscript. Neither author declared a conflict of interest.
Footnotes
Abbreviations used: CF, complementary feeding; IGF-I, insulin-like growth factor I; LAZ, length-for-age z score; WAZ, weight-for-age z score; WLZ, weight-for-length z score.
REFERENCES
- 1.Ong KK, Loos RJ. Rapid infancy weight gain and subsequent obesity: systematic reviews and hopeful suggestions. Acta Paediatr 2006;95:904–8. [DOI] [PubMed] [Google Scholar]
- 2.Kramer MS, Guo T, Platt RW, Vanilovich I, Sevkovskaya Z, Dzikovich I, Michaelsen KF, Dewey K. Feeding effects on growth during infancy. J Pediatr 2004;145:600–5. [DOI] [PubMed] [Google Scholar]
- 3.Victora CG, Morris SS, Barros FC, Horta BL, Weiderpass E, Tomasi E. Breast-feeding and growth in Brazilian infants. Am J Clin Nutr 1998;67:452–8. [DOI] [PubMed] [Google Scholar]
- 4.Dewey KG, Heinig MJ, Nommsen LA, Peerson JM, Lonnerdal B. Growth of breast-fed and formula-fed infants from 0 to 18 months: the DARLING Study. Pediatrics 1992;89:1035–41. [PubMed] [Google Scholar]
- 5.Dewey KG. Growth characteristics of breast-fed compared to formula-fed infants. Biol Neonate 1998;74:94–105. [DOI] [PubMed] [Google Scholar]
- 6.American Academy of Pediatrics, Committee on Nutrition, Barness LA. Pediatric nutrition handbook. 6th ed. Elk Grove Village, IL: American Academy of Pediatrics, 2009. [Google Scholar]
- 7.Socha P, Grote V, Gruszfeld D, Janas R, Demmelmair H, Closa-Monasterolo R, Subias JE, Scaglioni S, Verduci E, Dain E, et al. European Childhood Obesity Trial Study G: milk protein intake, the metabolic-endocrine response, and growth in infancy: data from a randomized clinical trial. Am J Clin Nutr 2011;94:1776S–84S. [DOI] [PubMed] [Google Scholar]
- 8.Heinig MJ, Nommsen LA, Peerson JM, Lonnerdal B, Dewey KG. Intake and growth of breast-fed and formula-fed infants in relation to the timing of introduction of complementary foods: the DARLING study. Davis Area Research on Lactation, Infant Nutrition and Growth. Acta Paediatr 1993;82:999–1006. [DOI] [PubMed] [Google Scholar]
- 9.Koletzko B, von Kries R, Closa R, Escribano J, Scaglioni S, Giovannini M, Beyer J, Demmelmair H, Gruszfeld D, Dobrzanska A, et al. Lower protein in infant formula is associated with lower weight up to age 2 y: a randomized clinical trial. Am J Clin Nutr 2009;89:1836–45. [DOI] [PubMed] [Google Scholar]
- 10.Escribano J, Luque V, Ferre N, Mendez-Riera G, Koletzko B, Grote V, Demmelmair H, Bluck L, Wright A, Closa-Monasterolo R. Effect of protein intake and weight gain velocity on body fat mass at 6 months of age: the EU Childhood Obesity Programme. Int J Obes (Lond) 2012;36:548–53. [DOI] [PubMed] [Google Scholar]
- 11.Michaelsen KF, Larnkjaer A, Molgaard C. Amount and quality of dietary proteins during the first two years of life in relation to NCD risk in adulthood. Nutrition, metabolism, and cardiovascular diseases. Nutr Metab Cardiovasc Dis 2012;22:781–6. [DOI] [PubMed] [Google Scholar]
- 12.Young BE, Johnson SL, Krebs NF. Biological determinants linking infant weight gain and child obesity: current knowledge and future directions. Adv Nutr 2012;3:675–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Krebs NF, Westcott JE, Culbertson DL, Sian L, Miller LV, Hambidge KM. Comparison of complementary feeding strategies to meet zinc requirements of older breastfed infants. Am J Clin Nutr 2012;96:30–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Krebs NF, Sherlock LG, Westcott J, Culbertson D, Hambidge KM, Feazel LM, Robertson CE, Frank DN. Effects of different complementary feeding regimens on iron status and enteric microbiota in breastfed infants. J Pediatr 2013;163:416–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Picciano MF. Nutrient composition of human milk. Pediatr Clin North Am 2001;48:53–67. [DOI] [PubMed] [Google Scholar]
- 16. Grummer-Strawn LM, Reinold C, Krebs NF: Use of World Health Organization and CDC growth charts for children aged 0-59 months in the United States. MMWR Recomm Rep 2010;59:1ndash15. [PubMed]
- 17.Dewey KG. The challenge of meeting nutrient needs of infants and young children during the period of complementary feeding: an evolutionary perspective. J Nutr 2013;143:2050–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Allen L. Comparing the value of protein sources for maternal and child nutrition. Food Nutr Bull 2013;34:263–6. [DOI] [PubMed] [Google Scholar]
- 19.Krebs NF, Westcott JE, Butler N, Robinson C, Bell M, Hambidge KM. Meat as a first complementary food for breastfed infants: feasibility and impact on zinc intake and status. J Pediatr Gastroenterol Nutr 2006;42:207–14. [DOI] [PubMed] [Google Scholar]
- 20.Penny ME, Creed-Kanashiro HM, Robert RC, Narro MR, Caulfield LE, Black RE. Effectiveness of an educational intervention delivered through the health services to improve nutrition in young children: a cluster-randomised controlled trial. Lancet 2005;365:1863–72. [DOI] [PubMed] [Google Scholar]
- 21.Dror DK, Allen LH. The importance of milk and other animal-source foods for children in low-income countries. Food Nutr Bull 2011;32:227–43. [DOI] [PubMed] [Google Scholar]
- 22.Krebs NF, Mazariegos M, Chomba E, Sami N, Pasha O, Tshefu A, Carlo WA, Goldenberg RL, Bose CL, Wright LL, et al. Randomized controlled trial of meat compared with multimicronutrient-fortified cereal in infants and toddlers with high stunting rates in diverse settings. Am J Clin Nutr 2012;96:840–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brisbois TD, Farmer AP, McCargar LJ. Early markers of adult obesity: a review. Obesity reviews: an official journal of the International Association for the Study of Obesity 2012;13:347–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rolland-Cachera MF, Deheeger M, Akrout M, Bellisle F. Influence of macronutrients on adiposity development: a follow up study of nutrition and growth from 10 months to 8 years of age. Obes Rev 1995;19:573–8. [PubMed] [Google Scholar]
- 25.Günther AL, Buyken AE, Kroke A. Protein intake during the period of complementary feeding and early childhood and the association with body mass index and percentage body fat at 7 y of age. Am J Clin Nutr 2007;85:1626–33. [DOI] [PubMed] [Google Scholar]
- 26.Kalhan SC. Optimal protein intake in healthy infants. Am J Clin Nutr 2009;89:1719–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Butte NF, Fox MK, Briefel RR, Siega-Riz AM, Dwyer JT, Deming DM, Reidy KC. Nutrient intakes of US infants, toddlers, and preschoolers meet or exceed dietary reference intakes. J Am Diet Assoc 2010;110:S27–37. [DOI] [PubMed] [Google Scholar]
- 28.Institute of Medicine (U.S.), Panel on Macronutrients, Institute of Medicine (U.S.). Standing Committee on the Scientific Evaluation of Dietary Reference Intakes: Dietary Reference Intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein, and amino acids. Washington, D.C.: National Academies Press, 2005. [Google Scholar]
- 29.Food and Agriculture Organization of the United Nations. United Nations University, World Health Organization. Human energy requirements: report of a Joint FAO/WHO/UNU Expert Consultation: Rome, 17-24 October 2001. Rome, Italy: Food and Agricultural Organization of the United Nations, 2004. [Google Scholar]
- 30.Butte NF, Hopkinson JM, Wong WW, Smith EO, Ellis KJ. Body composition during the first 2 years of life: an updated reference. Pediatr Res 2000;47:578–85. [DOI] [PubMed] [Google Scholar]