Abstract
Aims
In the present study we evaluated the antiviral activity of subtilosin, a cyclical peptide isolated from Bacillus amyloliquefaciens, against herpes simplex virus type 2 (HSV-2) in cell cultures and we investigated subtilosin mode of action.
Methods and Results
We determined, using a virus yield inhibition assay, that non cytotoxic concentrations of subtilosin inhibit HSV-2 replication in Vero cell cultures. Subtilosin strongly inhibited extracellular and total virus production even when it was added at 8 h post-infection indicating that not only virus release but also viral particle formation is impeded by the antiviral peptide. Although viral glycoprotein gD level of expression is not affected by the bacteriocin, an altered pattern of gD intracellular localization was detected by immunofluorescence assay in subtilosin treated culture. On the other hand, at high concentrations subtilosin displays virucidal action.
Conclusions
Subtilosin displays antiviral and virucidal actions against HSV-2. The target of subtilosin inhibitory effect would be late stages of the viral replicative cycle such as viral glycoprotein intracellular transport.
Keywords: Herpes simplex virus, bacteriocin, antiviral activity, microbicide, subtilosin, antimicrobial peptide
Introduction
Subtilosin, a ribosomally-synthesized 3.4-kDa cyclical peptide (Marx et al., 2001) produced by both Bacillus subtilis (Babasaki et al. 1985) and Bacillus amyloliquefaciens (Sutyak et al. 2008a), is a bacteriocin that has the potential for applications in contraception and reproductive health due to its unique properties, such as natural origin, biodegradability, antimicrobial properties, lack of immunogenicity, overall safety, ease of production and formulation (Sutyak et al. 2008b; Noll et al. 2012; Turovskiy et al. 2012; van Kuijk et al. 2012). Previous studies have demonstrated that subtilosin reduces sperm motility in vitro in a dose dependent fashion (Sutyak et al. 2008b). This compound also displays antimicrobial activity against the foodborne pathogen Listeria monocytogenes Scott A (van Kuijk et al. 2012) and against bacterial vaginosis-associated pathogen Gardnerella vaginalis, including their antibiotic-resistant forms (Noll et al. 2012; Turovskiy et al. 2012).
Herpes simplex virus types 1 and 2 (HSV-1 and HSV-2) are human viral pathogens that cause serious clinical diseases comprising genital ulcerations, corneal blindness, cold sores and encephalitis. Patients with immune deficiencies have an increased risk of developing severe HSV infection and vertical transmission of HSV-1 or HSV-2 to the newborn can cause fatal neonatal encephalitis (Chentoufi and Benmohamed 2012). Over 530 million people worldwide are infected with HSV-2, which can cause recurrent and painful genital lesions, being recurrent genital herpes, the most prevalent sexually transmitted disease (STD) (Fife et al. 2008; Chentoufi and Benmohamed 2012). On the other hand, epidemiologic studies suggest there is synergy between HSV-2 and human immunodeficiency virus type 1 (HIV-1); HSV-2 reactivation increases HIV-1 concentrations in plasma and genital secretions whereas co-infection with HIV-1 would increase HSV-2 transmission risk (Freeman et al. 2006; Thurman and Doncel 2012; Des Jarlais et al. 2014).
The first antiviral chemotherapy for the management of herpetic infections was treatment with acyclovir (ACV), an analogue of the natural nucleoside guanosine. The triphosphate form of this analogue selectively inhibits the viral DNA polymerase activity thus inhibiting viral replication. Drug-resistant HSV isolates are frequently found in-vitro and in immunocompromised patients (Choong et al. 2010; van Velzen et al. 2012). Foscarnet (FOS), a pyrophosphate analogue, is often administered in ACV resistant HSV infections. Nevertheless, its use is reserved to patients where other drugs fail due to its high toxicity and low bioavailability (Chilukuri and Rosen 2003). Furthermore, it has been shown that antiviral treatment of herpetic infection fails to completely prevent HSV and HIV transmission (Johnston et al. 2012). Considering that even in treated patients the infection persists during the lifetime of the host (Efstathiou and Preston 2005), the lack of an effective vaccine (Belshe et al. 2012) and the reported emergence of HSV resistant mutants, there is a need to develop new antiherpetic compounds with different mechanisms of action.
Interestingly, in a previous study we reported that subtilosin affects HSV-1 infectivity by two mechanisms: the bacteriocin acts as a virucidal agent and as an inhibitor of HSV-1 multiplication in cell cultures (Torres et al. 2013). In addition to its antimicrobial properties, subtilosin does not harm vaginal tissues, and does not interfere with the healthy vaginal lactobacilli (Sutyak et al. 2008b; Noll et al. 2012; Torres et al. 2013). Therefore, subtilosin is a promising non-toxic spermicide and has concomitant antimicrobial properties that may promote healthy vaginal microbiota. In the present study we examined the susceptibility of HSV-2 to subtilosin treatment in cell cultures and explored subtilosin mode of action.
Materials and Methods
Compounds
Subtilosin was purified from cultures of Bacillus amyloliquefaciens KATMIRA1933, as previously described by Sutyak et al. (Sutyak et al. 2008a). Essentially, the overnight culture’s supernatant was filter-sterilized (0.45 μm filter, Millipore, Billerica, MA, USA), concentrated using 30% ammonium sulfate, re-suspended in water and fractionated using Sep-Pak® Light C18 cartridges (Waters, Milford, MA, USA). The sample was subjected to the step-gradient Tricin-SDS PAGE (Bio-Rad, Hercules, CA, USA) and silver-stain visualized as a single band. In the overlay test with the bacteriocin-sensitive reference strain of Micrococcus luteus this PAGE protein band produced a single zone of inhibition (data not shown). Subtilosin was diluted with maintenance medium (MM) consisting of minimum essential medium (MEM) (Gibco, Carlsbad, CA, USA) with 2% inactivated fetal bovine serum and 50µg/mL of gentamycin.
Cells and viruses
African green monkey kidney (Vero) cells were grown as monolayers in MEM supplemented with 5% inactivated fetal bovine serum and 50µg/mL of gentamycin. HSV-2 strain G was obtained from the American Type Culture Collection (Rockville, MD, USA). Virus stock was prepared in Vero cells.
Cell cytotoxicity assay
To assess the effect of subtilosin on cell viability, confluent monolayers of Vero cells grown in 96-well culture plates, were incubated with different concentrations of subtilosin for 48 h at 37°C. Then, cell viability was measured by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT, Sigma-Aldrich, St. Louis, MO) procedure (Denizot and Lang 1986). Cytotoxicity was expressed as the 50% cytotoxic concentration (CC50) which is the concentration of subtilosin that reduced cell viability by 50% with respect to the cellular control.
Virus yield inhibition assay
Antiviral activity was evaluated by a virus yield inhibition assay. To this end, Vero cells, grown in 24-well culture plates, were infected with HSV-2 at a multiplicity of infection (m.o.i.) of 1 PFU/cell. After 1 h of adsorption at 37°C, virus inoculum was discarded and cells were covered with MM (control) or MM containing serial dilutions of subtilosin. After 24 h of incubation at 37°C, the supernatants were harvested and extracellular virus yields were determined by a plaque formation assay. The antiviral activity was expressed as the 50% effective concentration (EC50), i.e. the compound concentration of subtilosin required to reduce virus yield by 50% compared to the untreated infected culture. The selectivity index (SI) was calculated as the ratio between CC50 and EC50 values.
Virucidal assay
To assay virucidal activity of the bacteriocin, HSV-2 was incubated with subtilosin at concentrations ranging from 25 to 200μg/mL or MM for 90 min at 37°C. After the incubation period, aliquots were conveniently diluted in MM and remaining infectivity was determined by plaque assay on Vero cells.
Time of addition experiment
Subtilosin (50μg/mL) was added to Vero cells, either during 6 h before the infection with HSV-2 (m.o.i.=1) or at 1, 3, 5 or 8 h post-infection (p.i.) Cultures were incubated up to 24 h p.i. and at that time supernatants were harvested to assess extracellular virus titer. Another set of identically infected-treated cultures were subjected to two freeze-thaw cycles, followed by low-speed centrifugation in order to quantify total (extracellular and intracellular) viral infectivity. Virus titers were determined by plaque formation assay.
Indirect immunofluorescence assay
Vero cells grown on glass coverslips were infected with HSV-2 at an m.o.i. of 1 PFU/cell. After 1 h adsorption at 37°C, cultures were incubated in MM containing or not subtilosin 25, 50 or 100μg/mL and incubated at 37°C for 24 h. After the removal of culture supernatants cells were washed with cold PBS, fixed with cold methanol (20 min at −20°C) and then incubated with a mouse monoclonal antibody reactive against gD viral glycoprotein (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) for 45 min at 37°C. The indirect staining was carried out by using goat anti-mouse immunoglobulins conjugated to FITC (Sigma Aldrich, St. Louis, MO, USA). Fluorescent cells were photographed with a Zeiss microscope with epifluorescence optics.
Western blot assay
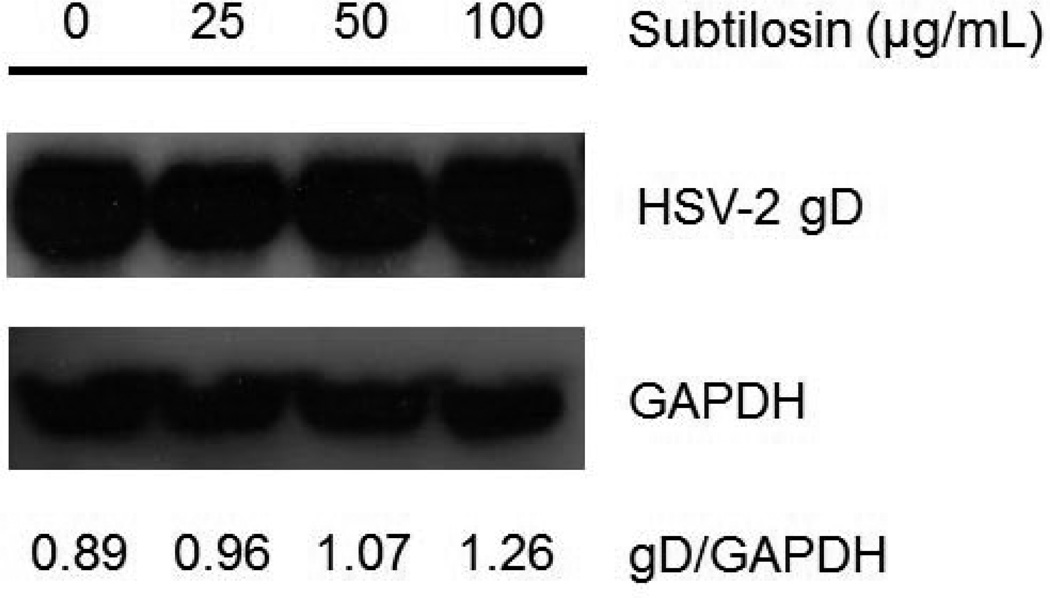
Vero cells were infected with HSV-2 (moi=1) and after virus adsorption cells were incubated in MM containing or not subtilosin 25, 50 or 100μg/mL and incubated at 37°C for 24 h. Then cells were lysed and samples were subjected to SDS-PAGE and transferred to PVDF membrane (Perkin Elmer Life Sciences, Inc., Waltham, MA, USA) in a dry system (LKB Multiphor II, Pharmacia, Sweden). Viral glycoprotein gD was revealed using mouse anti-gD (Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA) and GAPDH, used as loading control, was detected with mouse anti-GAPDH (Abcam, United Kingdom). Peroxidase-conjugated anti-mouse immunoglobulin G (Promega, Madison, WI, USA) was used as secondary antibody. The intensities of protein bands, visualized by chemiluminescence detection, were quantified by using Image J software and the relative intensity gD/GAPDH was calculated for each sample.
Statistical analysis
The 95 % confidence intervals (CI) of virus titers were calculated according to Poisson distribution.
Results
Antiviral activity of subtilosin against HSV-2
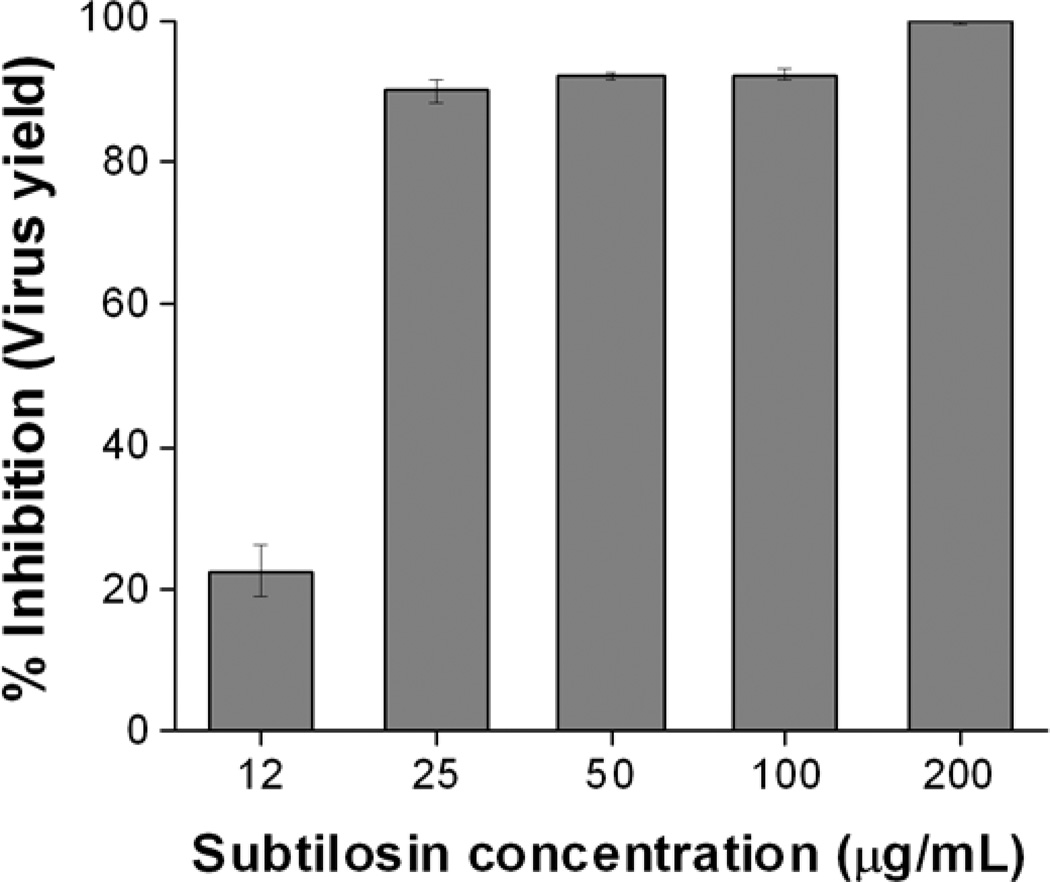
First, we evaluated the effect of treatment with different concentrations of subtilosin on Vero cell viability using the MTT method. The CC50 value of subtilosin in Vero cells was 316.8 µg/mL. Then we performed a virus yield inhibition assay using non-cytotoxic concentrations of subtilosin. A dose-dependent inhibition of virus yield was observed in subtilosin treated cultures (Figure 1). At 25μg/mL, subtilosin inhibited virus replication by almost 90%, whereas at 200μg/mL a reduction over 99.9% in virus titer was achieved (Figure 1). The EC50 and SI values of subtilosin against HSV-2 were 18.2μg/mL and 17.4, respectively.
Figure 1. Dose-dependent inhibition of HSV-2 multiplication by subtilosin.
Vero cells infected with HSV-2 (m.o.i. =1) were incubated with different concentrations of subtilosin. At 24 h p.i. virus yield was quantified by plaque assay. Data represent mean values from duplicate determinations ± standard deviation (SD).
Virucidal action of subtilosin
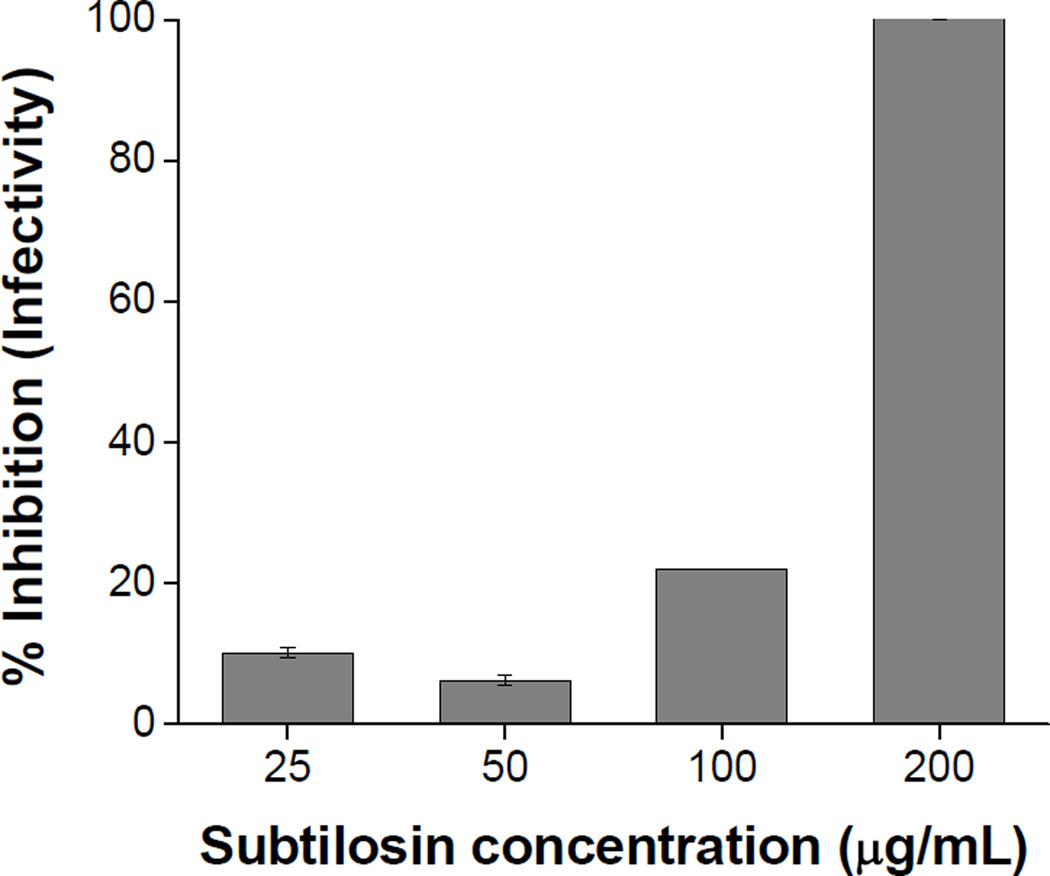
Taking into account previous studies that showed that at high concentrations subtilosin display virucidal activity against HSV-1 (Torres et al. 2013), we evaluated the effect of subtilosin on HSV-2 particles by incubating aliquots of viral stock with different concentrations of subtilosin during 90 min at 37°C. As can be seen in Fig. 2, at the highest concentration tested (200μg/mL), subtilosin exhibited a direct inactivating effect on viral particles since after 90 min of treatment a 99,99% reduction of HSV-2 infectivity was observed. However, at 100, 50 or 25μg/mL, concentrations that displayed antiviral activity (Figure 1), subtilosin showed no relevant virucidal effect (Figure 2).
Figure 2. Virucidal activity of subtilosin.
HSV-2 suspensions were incubated with different concentrations of subtilosin during 90 min at 37°C. Remaining infectivity was determined by plaque formation assay. Data represent mean values from duplicate determinations ± SD.
Characterization of subtilosin antiviral activity
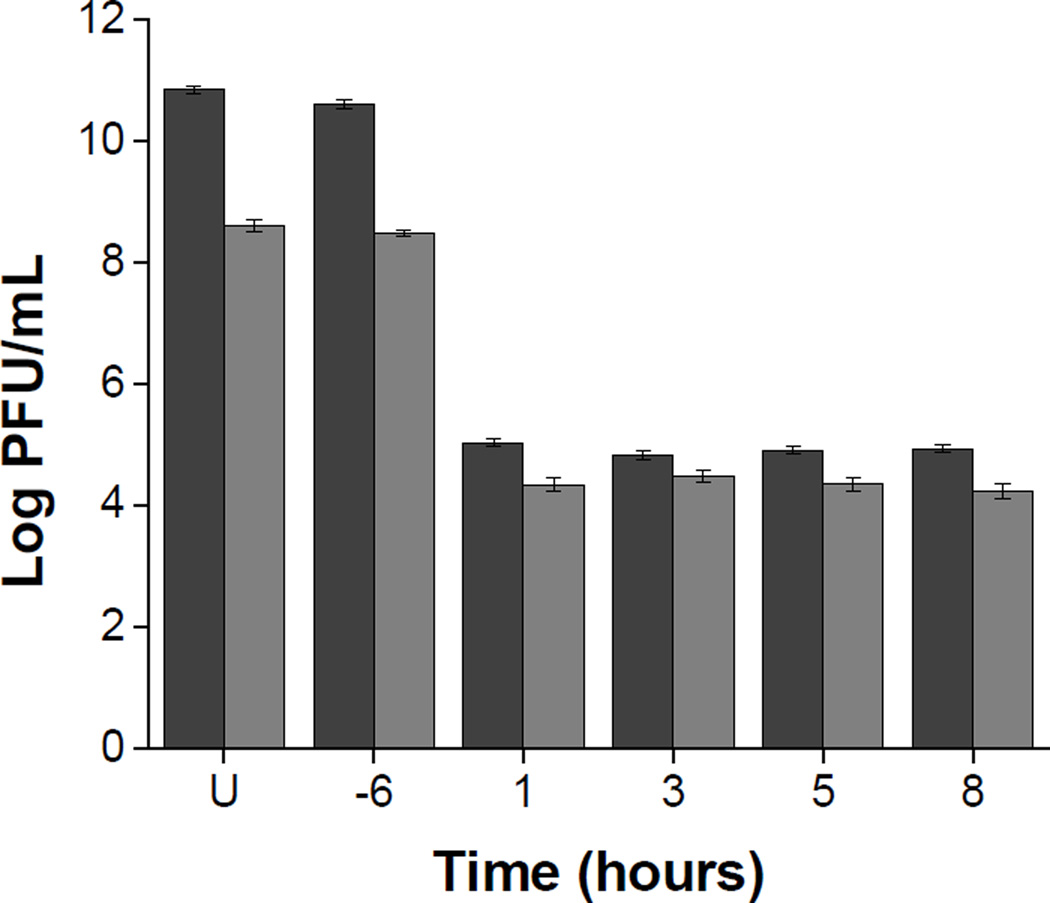
To characterize subtilosin inhibitory action against HSV-2, a time of addition experiment was performed. Vero cells were treated with subtilosin (50μg/mL) for 6 h prior to infection or at different times after infection. In all cases, extracellular and total virus production was determined at 24 h p.i. Subtilosin treatment of cells before infection did not affect virus production indicating that the peptide does not induce an antiviral state on the cell culture. On the contrary, the presence of subtilosin from 1, 3, 5 or 8 h p.i reduced total virus titer by approximately 6 log units and the amount of infectious virus released to the extracellular medium was also strongly inhibited (Figure 3). The same level of reduction in virus production was observed when subtilosin was added at 1 or 8 h p.i., thus a possible explanation for these results is that a late stage on the viral replicative cycle would be blocked by subtilosin.
Figure 3. Effect of time of addition of subtilosin on HSV-2 production.
Vero cells were treated with subtilosin during 6 h previous to infection (−6) or were incubated with subtilosin from different times after infection (1, 3, 5 or 8 h p.i.). At 24 h p.i. extracellular (grey bars) and total virus (black bars) production were determined. U = untreated infected culture. Data are mean values of virus titers from duplicate determinations ± 95% CI.
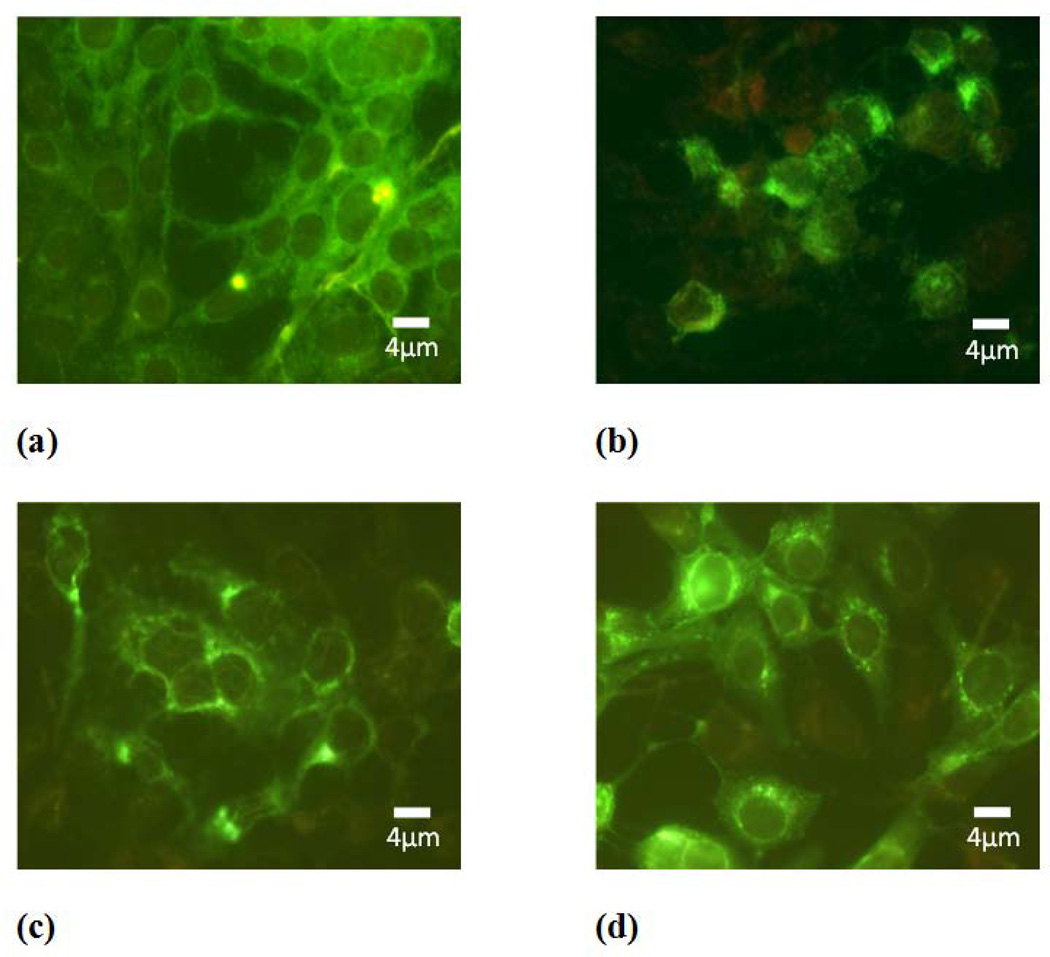
In order to further investigate which is the step of viral replication affected by subtilosin, we performed an IF assay to analyze the effect of the bacteriocin on viral protein expression employing a monoclonal antibody that recognizes gD glycoprotein. We observed that gD was localized predominately in the perinuclear region of cells treated with subtilosin, whereas in untreated cells a more diffuse and homogenous pattern of cytoplasmic fluorescence was detected. Changes in gD intracellular distribution were more evident in cultures treated with the highest concentration of subtilosin assayed (Figure 4 b). To determine whether treatment with subtilosin also affects gD production, we performed a comparative analysis of gD total level of expression between cultures treated with different concentrations of subtilosin and untreated infected cells. The analysis by western blot shown in figure 5 revealed similar gD levels in treated and untreated cells indicating that the antimicrobial peptide does not impair viral protein synthesis. Altogether these results suggest that subtilosin would not affect gD production but interferes with gD intracellular transport therefore affecting formation of mature viral particles.
Figure 4. Intracellular localization of gD glycoprotein in subtilosin treated cells.
Cytoplasmic immunofluorescence staining of HSV-2 infected Vero cells in the absence (a) or in the presence of 100μg/mL (b), 50μg/mL (c) or 25μg/mL (d) subtilosin. Magnification 400×.
Figure 5. gD production in subtilosin treated cells.
HSV-2 infected Vero cells were treated with different concentrations of subtilosin and at 24 h p.i. the expression of viral protein gD and cellular protein GAPDH, used as loading control, was assessed by Western blot. The relative intensity of gD band with respect to GAPDH band for each sample of one representative experiment is shown.
Discussion
The lifelong HSV-2 infection is the principal cause of genital ulcer disease and it is also a contributing factor in the increase of acquisition and transmissibility of HIV infection (Gottlieb et al. 2014; Des Jarlais et al. 2014). In the last years, great efforts have been made to develop new antiviral molecules effective against HSV including those viral strains that are resistant to nucleoside analogues (Wald et al. 2014). In a previous work we have described the ability of subtilosin to inhibit the multiplication of ACV-susceptible and ACV-resistant strains of HSV-1 (Torres et al. 2013). Here we demonstrate that subtilosin is also an effective inhibitor of HSV-2 multiplication and we further characterize the antiviral mode of action of this bacterial peptide.
Although antiviral activity of several antimicrobial peptides isolated from different bacteria genera has been previously reported (Wachsman et al. 1999, 2003; Serkedjieva et al. 2000; Todorov et al. 2005, 2010; Saeed et al. 2007; Férir et al. 2013) no virucidal effect was described for these bacteriocins and up to now much remains unknown about their inhibitory mechanism of action. CRL5, peptide isolated from Enterococcus faecium, exhibits an inhibitory effect on HSV-1 late viral protein synthesis (Wachsman et al. 2003) whereas the lantibiotic peptide Labyrinthopeptin A1 produced by Actinomadura namibiensis blocks an early after-adsorption step of HIV multiplication (Férir et al. 2013).
Our results show that pre-treatment with subtilosin did not protect cells from infection. This indicates that subtilosin should be present during viral multiplication to exert its inhibitory action and also suggests that the bacteriocin does not cause an irreversible effect on host cell functions. However, the addition of subtilosin after HSV-2 infection strongly inhibited both extracellular and total virus production, indicating that not only virus release but also viral particle formation is blocked by bacteriocin treatment. The same level of inhibition was achieved when subtilosin was added between 1 and 8 h p.i. indicating that early events of HSV-2 multiplication cycle are not affected by the bacteriocin.
Furthermore, the analysis of the expression of viral glycoprotein gD showed that although gD production is not affected by the bacteriocin an altered pattern of gD intracellular localization in subtilosin treated cultures was observed suggesting that subtilosin would affect gD intracellular transport. Future studies will be necessary to establish the precise cellular compartment in which gD accumulates after subtilosin treatment. Taking together these results indicate that the target of subtilosin antiviral action are late stages of virus replication, thus hindering viral particle formation.
Besides its inhibitory effect and in coincidence with the results obtained with HSV-1 (Torres et al. 2013), at high concentrations, subtilosin also exhibits virucidal action against HSV-2. Altogether our results show that subtilosin mode of antiviral action seems to be clearly different to those previously described for other bacteriocins (Serkedjieva et al. 2000; Wachsman et al. 2003; Férir et al. 2013).
Control of transmission of STDs includes the use of topically applied microbicides that inactivate the relevant pathogens, thus virucidal agents provides a good source of developing new microbicidal formulations. Bacteriocins are attractive natural alternatives to classic antibiotics and virucidal agents, which could be used for the treatment of bacterial and viral infections. The ability of subtilosin to inactivate both HSV-1 and HSV-2 particles, together with its spermicidal activity and safety of subtilosin based nanofibers for use on human skin tissues (Sutyak et al. 2008b; Torres et al. 2013) makes this peptide a promissory candidate to be included in new studies concerning the design of nanofiber systems containing a combination of antimicrobial and anti-inflammatory drugs to be proved in animal models.
Significance and Impact of Study.
Given its antimicrobial activity and its safety for human tissues, subtilosin could represent a valuable alternative to be considered in the development of new microbicide formulations.
Acknowledgements
This work was funded by grant from Universidad de Buenos Aires (20020110100076); P.J.S. and M.L.C. were supported by the Bill and Melinda Gates Foundation Grand Challenges Exploration (Round 5, Phase I Grant OPP1025200) and by the National Institutes of Health/National Institute of Allergy and Infectious Diseases (1R01AI084137).
Footnotes
Conflict of Interest
We have no conflicts of interest to disclose.
References
- Babasaki K, Takao T, Shimonishi Y, Kurahashi K. Subtilosin A, a new antibiotic peptide produced by Bacillus subtilis: isolation, structural analysis, and biogenesis. J Biochem. 1985;98:585–603. doi: 10.1093/oxfordjournals.jbchem.a135315. [DOI] [PubMed] [Google Scholar]
- Belshe RB, Leone PA, Bernstein DI, Wald A, Levin MJ, Stapleton JT, Gorfinkel I, Morrow RL, Ewell MG, Stokes-Riner A, Dubin G, Heineman TC, Schulte JM, Deal CD Herpevac Trial for Women. Efficacy results of a trial of a herpes simplex vaccine. N Engl J Med. 2012;366:34–43. doi: 10.1056/NEJMoa1103151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chentoufi AA, Benmohamed L. Mucosal herpes immunity and immunopathology to ocular and genital herpes simplex virus infections. Clin Dev Immunol. 2012;2012:149135. doi: 10.1155/2012/149135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chilukuri S, Rosen T. Management of acyclovir-resistant herpes simplex virus. Dermatol Clin. 2003;21:311–320. doi: 10.1016/s0733-8635(02)00093-1. [DOI] [PubMed] [Google Scholar]
- Choong K, Walker N, Apel A, Whitby M. Aciclovir-resistant herpes keratitis. Clin Experiment Ophtalmol. 2010;38:309–313. doi: 10.1111/j.1442-9071.2010.02209.x. [DOI] [PubMed] [Google Scholar]
- Denizot F, Lang R. Rapid colorimetric assay for cell growth and survival. J Inmunol Methods. 1986;89:271–277. doi: 10.1016/0022-1759(86)90368-6. [DOI] [PubMed] [Google Scholar]
- Des Jarlais DC, Arasteh K, McKnight C, Perlman DC, Feelemyer J, Hagan H, Cooper HL. HSV-2 Co-Infection as a Driver of HIV Transmission among Heterosexual Non-Injecting Drug Users in New York City. PLoS One. 2014;9:e87993. doi: 10.1371/journal.pone.0087993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Efstathiou S, Preston CM. Towards an understanding of the molecular basis of herpes simplex virus latency. Virus Res. 2005;111:108–119. doi: 10.1016/j.virusres.2005.04.017. [DOI] [PubMed] [Google Scholar]
- Férir G, Petrova MI, Andrei G, Huskens D, Hoorelbeke B, Snoeck R, Vanderleyden J, Balzarini J, Bartoschek S, Brönstrup M, Süssmuth RD, Schols D. The lantibiotic peptide labyrinthopeptin A1 demonstrates broad anti-HIV and anti-HSV activity with potential for microbicidal applications. PLoS One. 2013;8:e64010. doi: 10.1371/journal.pone.0064010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fife KH, Warren TJ, Justus SE, Heitman CK. An international, randomized, double-blind, placebo controlled, study of valacyclovir for the suppression of herpes simplex virus type 2 genital herpes in newly diagnosed patients. Sex Transm Dis. 2008;35:666–673. doi: 10.1097/OLQ.0b013e31816d1f42. [DOI] [PubMed] [Google Scholar]
- Freeman EE, Weiss HA, Glynn JR, Cross PL, Whitworth JA, Hayes RJ. Herpes simplex virus 2 infection increases HIV acquisition in men and women: systematic review and meta-analysis of longitudinal studies. AIDS. 2006;20:73–83. doi: 10.1097/01.aids.0000198081.09337.a7. [DOI] [PubMed] [Google Scholar]
- Gottlieb SL, Low N, Newman LM, Bolan G, Kamb M, Broutet N. Toward global prevention of sexually transmitted infections (STIs): The need for STI vaccines. Vaccine. 2014;32:1527–1535. doi: 10.1016/j.vaccine.2013.07.087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston C, Saracino M, Kuntz S, Magaret A, Selke S, Huang ML, Schiffer JT, Koelle DM, Corey L, Wald A. Standard-dose and high-dose daily antiviral therapy for short episodes of genital HSV-2 reactivation: three randomised, open-label, cross-over trials. Lancet. 2012;379:641–647. doi: 10.1016/S0140-6736(11)61750-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marx R, Stein T, Entian KD, Glaser SJ. Structure of the Bacillus subtillis peptide antibiotic subtilosin A determined by 1H NMR and matrix assisted laser desorption/ionization time-of-flight mass spectrometry. J Protein Chem. 2001;20:501–506. doi: 10.1023/a:1012562631268. [DOI] [PubMed] [Google Scholar]
- Noll KS, Prichard MN, Khaykin A, Sinko PJ, Chikindas ML. The natural antimicrobial peptide subtilosin acts synergistically with glycerol monolaurate, lauric arginate, and ε-poly-L-lysine against bacterial vaginosis-associated pathogens but not human lactobacilli. Antimicrob Agents Chemother. 2012;56:1756–1761. doi: 10.1128/AAC.05861-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saeed S, Rasool SA, Ahmad S, Zaidi SZ, Rehmani S. Antiviral activity of staphylococcin 188: A purified bacteriocin like inhibitory substance isolated from Staphylococcus aureus AB188. Res J Microbiol. 2007;2:796–806. [Google Scholar]
- Serkedjieva J, Danova S, Ivanova I. Antiinfluenza virus activity of a bacteriocin produced by Lactobacillus delbrueckii. Appl Biochem Biotechnol. 2000;88:285–298. [Google Scholar]
- Sutyak KE, Wirawan RE, Aroutcheva AA, Chikindas ML. Isolation of the Bacillus subtilis antimicrobial peptide subtilosin from the dairy product-derived Bacillus amyloliquefaciens. J Appl Microbiol. 2008a;104:1067–1074. doi: 10.1111/j.1365-2672.2007.03626.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sutyak KE, Anderson RA, Dover SE, Feathergill KA, Aroutcheva AA, Faro S, Chikindas ML. Spermicidal activity of the safe natural antimicrobial peptide subtilosin. Infect Dis Obstet Gynecol. 2008b;2008:540758. doi: 10.1155/2008/540758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thurman AR, Doncel GF. Herpes simplex virus and HIV: genital infection synergy and novel approaches to dual prevention. Int J STD AIDS. 2012;23:613–619. doi: 10.1258/ijsa.2012.011356. [DOI] [PubMed] [Google Scholar]
- Todorov SD, Wachsman M, Tomé E, Dousset X, Destro MT, Dicks LM, Franco BD, Vaz-Velho M, Drider D. Characterisation of an antiviral pediocin-like bacteriocin produced by Enterococcus faecium. Food Microbiology. 2010;27:869–879. doi: 10.1016/j.fm.2010.05.001. [DOI] [PubMed] [Google Scholar]
- Todorov SD, Wachsman MB, Knoetze H, Meincken M, Dicks LM. An antibacterial and antiviral peptide produced by Enterococcus mundtii ST4V isolated from soya beans. Int J Antimicrob Agents. 2005;25:508–513. doi: 10.1016/j.ijantimicag.2005.02.005. [DOI] [PubMed] [Google Scholar]
- Torres NI, Noll KS, Xu S, Li J, Huang Q, Sinko PJ, Wachsman MB, Chikindas ML. Safety, formulation, and in vitro antiviral activity of the antimicrobial peptide subtilosin against herpes simplex virus type 1. Probiotics Antimicrob Proteins. 2013;5:26–35. doi: 10.1007/s12602-012-9123-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turovskiy Y, Cheryian T, Algburi A, Wirawan RE, Takhistov P, Sinko PJ, Chikindas ML. Susceptibility of Gardnerella vaginalis biofilms to natural antimicrobials subtilosin, ε-poly-L-lysine, and lauramide arginine ethyl ester. Infect Dis Obstet Gynecol. 2012;2012:284762. doi: 10.1155/2012/284762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Kuijk S, Noll KS, Chikindas ML. The species-specific mode of action of the antimicrobial peptide subtilosin against Listeria monocytogenes Scott A. Lett Appl Microbiol. 2012;54:52–58. doi: 10.1111/j.1472-765X.2011.03170.x. [DOI] [PubMed] [Google Scholar]
- van Velzen M, van Loenen FB, Meesters RJ, de Graaf M, Remeijer L, Luider TM, Osterhaus AD, Verjans GM. Latent acyclovir-resistant herpes simplex virus type 1 in trigeminal ganglia of immunocompetent individuals. J Infect Dis. 2012;205:1539–1543. doi: 10.1093/infdis/jis237. [DOI] [PubMed] [Google Scholar]
- Wachsman MB, Farías ME, Takeda E, Sesma F, de Ruiz Holgado AP, de Torres RA, Coto CE. Antiviral activity of enterocin CRL35 against herpesviruses. Int J Antimicrob Agents. 12:293–299. doi: 10.1016/s0924-8579(99)00078-3. [DOI] [PubMed] [Google Scholar]
- Wachsman MB, Castilla V, de Ruiz Holgado AP, de Torres RA, Sesma F, Coto CE. Enterocin CRL35 inhibits late stages of HSV-1 and HSV-2 replication in vitro. Antiviral Res. 2003;58:17–24. doi: 10.1016/s0166-3542(02)00099-2. [DOI] [PubMed] [Google Scholar]
- Wald A, Corey L, Timmler B, Magaret A, Warren T, Tyring S, Johnston C, Kriesel J, Fife K, Galitz L, Stoelben S, Huang ML, Selke S, Stobernack HP, Ruebsamen-Schaeff H, Birkmann A. Helicase-primase inhibitor pritelivir for HSV-2 infection. N Engl J Med. 2014;370:201–210. doi: 10.1056/NEJMoa1301150. [DOI] [PubMed] [Google Scholar]