Abstract
These studies were initiated to elucidate the mechanism of DNA nuclear transport in mammalian cells. Biotin- or gold-labeled plasmid and plasmid DNA expression vectors for Escherichia coli beta-galactosidase or firefly luciferase were microinjected into the cytoplasm of primary rat myotubes in culture. Plasmid DNA was expressed in up to 70% of the injected myotubes, which indicates that it entered intact, postmitotic nuclei. The nuclear transport of plasmid DNA occurred through the nuclear pore by a process common to other large karyophilic macromolecules. The majority of the injected plasmid DNA was sequestered by cytoplasmic elements. This understanding of plasmid DNA nuclear transport provides a basis for increasing the efficiency of gene transfer.
Full text
PDF

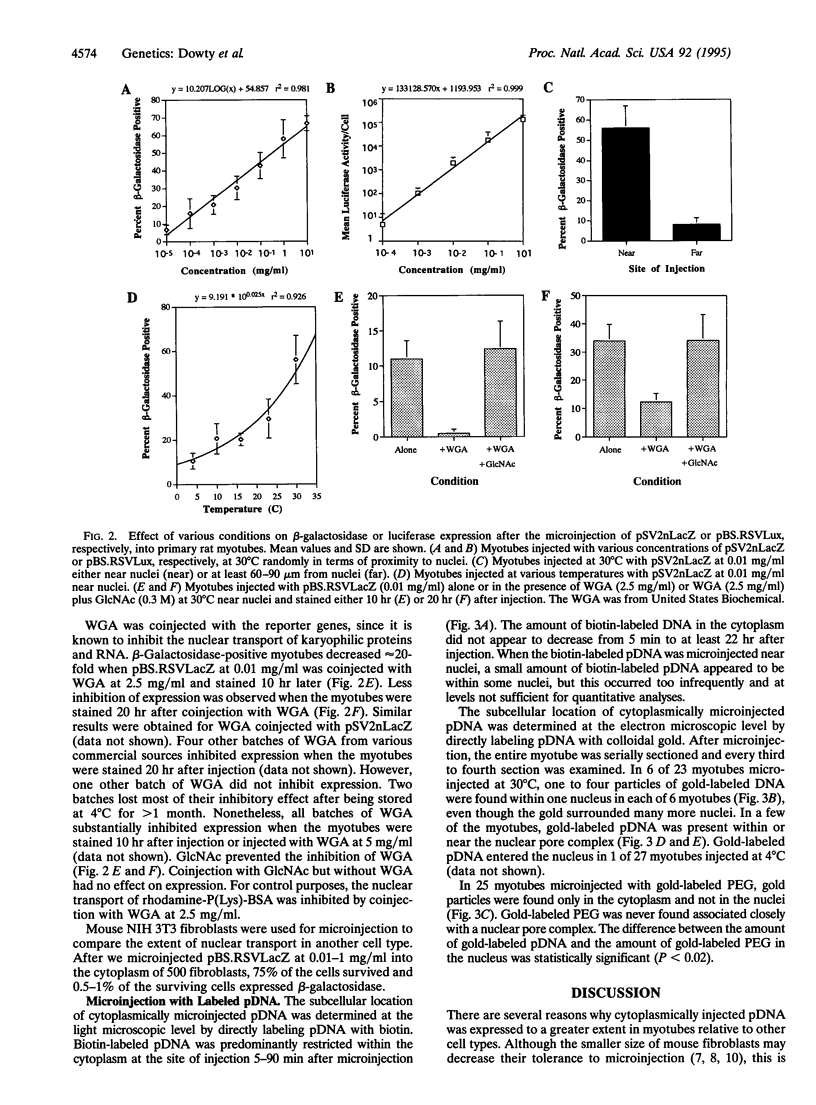


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Breeuwer M., Goldfarb D. S. Facilitated nuclear transport of histone H1 and other small nucleophilic proteins. Cell. 1990 Mar 23;60(6):999–1008. doi: 10.1016/0092-8674(90)90348-i. [DOI] [PubMed] [Google Scholar]
- Brinster R. L., Chen H. Y., Trumbauer M. E., Yagle M. K., Palmiter R. D. Factors affecting the efficiency of introducing foreign DNA into mice by microinjecting eggs. Proc Natl Acad Sci U S A. 1985 Jul;82(13):4438–4442. doi: 10.1073/pnas.82.13.4438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bukrinsky M. I., Haggerty S., Dempsey M. P., Sharova N., Adzhubel A., Spitz L., Lewis P., Goldfarb D., Emerman M., Stevenson M. A nuclear localization signal within HIV-1 matrix protein that governs infection of non-dividing cells. Nature. 1993 Oct 14;365(6447):666–669. doi: 10.1038/365666a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capecchi M. R. High efficiency transformation by direct microinjection of DNA into cultured mammalian cells. Cell. 1980 Nov;22(2 Pt 2):479–488. doi: 10.1016/0092-8674(80)90358-x. [DOI] [PubMed] [Google Scholar]
- Chin D. J., Green G. A., Zon G., Szoka F. C., Jr, Straubinger R. M. Rapid nuclear accumulation of injected oligodeoxyribonucleotides. New Biol. 1990 Dec;2(12):1091–1100. [PubMed] [Google Scholar]
- Clever J., Yamada M., Kasamatsu H. Import of simian virus 40 virions through nuclear pore complexes. Proc Natl Acad Sci U S A. 1991 Aug 15;88(16):7333–7337. doi: 10.1073/pnas.88.16.7333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cotten M., Wagner E., Zatloukal K., Phillips S., Curiel D. T., Birnstiel M. L. High-efficiency receptor-mediated delivery of small and large (48 kilobase gene constructs using the endosome-disruption activity of defective or chemically inactivated adenovirus particles. Proc Natl Acad Sci U S A. 1992 Jul 1;89(13):6094–6098. doi: 10.1073/pnas.89.13.6094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Danko I., Fritz J. D., Jiao S., Hogan K., Latendresse J. S., Wolff J. A. Pharmacological enhancement of in vivo foreign gene expression in muscle. Gene Ther. 1994 Mar;1(2):114–121. [PubMed] [Google Scholar]
- Featherstone C., Darby M. K., Gerace L. A monoclonal antibody against the nuclear pore complex inhibits nucleocytoplasmic transport of protein and RNA in vivo. J Cell Biol. 1988 Oct;107(4):1289–1297. doi: 10.1083/jcb.107.4.1289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldherr C. M., Akin D. The permeability of the nuclear envelope in dividing and nondividing cell cultures. J Cell Biol. 1990 Jul;111(1):1–8. doi: 10.1083/jcb.111.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldherr C. M., Kallenbach E., Schultz N. Movement of a karyophilic protein through the nuclear pores of oocytes. J Cell Biol. 1984 Dec;99(6):2216–2222. doi: 10.1083/jcb.99.6.2216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forbes D. J. Structure and function of the nuclear pore complex. Annu Rev Cell Biol. 1992;8:495–527. doi: 10.1146/annurev.cb.08.110192.002431. [DOI] [PubMed] [Google Scholar]
- Garcia-Bustos J., Heitman J., Hall M. N. Nuclear protein localization. Biochim Biophys Acta. 1991 Mar 7;1071(1):83–101. doi: 10.1016/0304-4157(91)90013-m. [DOI] [PubMed] [Google Scholar]
- Goldfarb D. S. Karyophilic peptides: applications to the study of nuclear transport. Cell Biol Int Rep. 1988 Sep;12(9):809–832. doi: 10.1016/0309-1651(88)90090-2. [DOI] [PubMed] [Google Scholar]
- Jiao S., Schultz E., Wolff J. A. Intracerebral transplants of primary muscle cells: a potential 'platform' for transgene expression in the brain. Brain Res. 1992 Mar 13;575(1):143–147. doi: 10.1016/0006-8993(92)90434-b. [DOI] [PubMed] [Google Scholar]
- Jiao S., Williams P., Safda N., Schultz E., Wolff J. A. Co-transplantation of plasmid-transfected myoblasts and myotubes into rat brains enables high levels of gene expression long-term. Cell Transplant. 1993;2(3):185–192. doi: 10.1177/096368979300200302. [DOI] [PubMed] [Google Scholar]
- Kam Z., Borochov N., Eisenberg H. Dependence of laser light scattering of DNA on NaCl concentration. Biopolymers. 1981 Dec;20(12):2671–2690. doi: 10.1002/bip.1981.360201213. [DOI] [PubMed] [Google Scholar]
- Kaneda Y., Iwai K., Uchida T. Increased expression of DNA cointroduced with nuclear protein in adult rat liver. Science. 1989 Jan 20;243(4889):375–378. doi: 10.1126/science.2911748. [DOI] [PubMed] [Google Scholar]
- Lang I., Scholz M., Peters R. Molecular mobility and nucleocytoplasmic flux in hepatoma cells. J Cell Biol. 1986 Apr;102(4):1183–1190. doi: 10.1083/jcb.102.4.1183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leonetti J. P., Mechti N., Degols G., Gagnor C., Lebleu B. Intracellular distribution of microinjected antisense oligonucleotides. Proc Natl Acad Sci U S A. 1991 Apr 1;88(7):2702–2706. doi: 10.1073/pnas.88.7.2702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luby-Phelps K., Castle P. E., Taylor D. L., Lanni F. Hindered diffusion of inert tracer particles in the cytoplasm of mouse 3T3 cells. Proc Natl Acad Sci U S A. 1987 Jul;84(14):4910–4913. doi: 10.1073/pnas.84.14.4910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maul G. G. The nuclear and the cytoplasmic pore complex: structure, dynamics, distribution, and evolution. Int Rev Cytol Suppl. 1977;(6):75–186. [PubMed] [Google Scholar]
- Miller A. D. Human gene therapy comes of age. Nature. 1992 Jun 11;357(6378):455–460. doi: 10.1038/357455a0. [DOI] [PubMed] [Google Scholar]
- Minagawa K., Matsuzawa Y., Yoshikawa K., Masubuchi Y., Matsumoto M., Doi M., Nishimura C., Maeda M. Change of the higher order structure of DNA induced by the complexation with intercalating synthetic polymer, as is visualized by fluorescence microscopy. Nucleic Acids Res. 1993 Jan 11;21(1):37–40. doi: 10.1093/nar/21.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirzayans R., Aubin R. A., Paterson M. C. Differential expression and stability of foreign genes introduced into human fibroblasts by nuclear versus cytoplasmic microinjection. Mutat Res. 1992 Feb;281(2):115–122. doi: 10.1016/0165-7992(92)90045-j. [DOI] [PubMed] [Google Scholar]
- Mulligan R. C. The basic science of gene therapy. Science. 1993 May 14;260(5110):926–932. doi: 10.1126/science.8493530. [DOI] [PubMed] [Google Scholar]
- Paine P. L., Moore L. C., Horowitz S. B. Nuclear envelope permeability. Nature. 1975 Mar 13;254(5496):109–114. doi: 10.1038/254109a0. [DOI] [PubMed] [Google Scholar]
- Paine P. L. Nuclear protein accumulation by facilitated transport and intranuclear binding. Trends Cell Biol. 1993 Oct;3(10):325–329. doi: 10.1016/0962-8924(93)90096-j. [DOI] [PubMed] [Google Scholar]
- Picard D., Yamamoto K. R. Two signals mediate hormone-dependent nuclear localization of the glucocorticoid receptor. EMBO J. 1987 Nov;6(11):3333–3340. doi: 10.1002/j.1460-2075.1987.tb02654.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt-Zachmann M. S., Dargemont C., Kühn L. C., Nigg E. A. Nuclear export of proteins: the role of nuclear retention. Cell. 1993 Aug 13;74(3):493–504. doi: 10.1016/0092-8674(93)80051-f. [DOI] [PubMed] [Google Scholar]
- Thorburn A. M., Alberts A. S. Efficient expression of miniprep plasmid DNA after needle micro-injection into somatic cells. Biotechniques. 1993 Mar;14(3):356–358. [PubMed] [Google Scholar]
- Wolff J. A., Malone R. W., Williams P., Chong W., Acsadi G., Jani A., Felgner P. L. Direct gene transfer into mouse muscle in vivo. Science. 1990 Mar 23;247(4949 Pt 1):1465–1468. doi: 10.1126/science.1690918. [DOI] [PubMed] [Google Scholar]
- Yoneda Y., Imamoto-Sonobe N., Yamaizumi M., Uchida T. Reversible inhibition of protein import into the nucleus by wheat germ agglutinin injected into cultured cells. Exp Cell Res. 1987 Dec;173(2):586–595. doi: 10.1016/0014-4827(87)90297-7. [DOI] [PubMed] [Google Scholar]
- de Wet J. R., Wood K. V., DeLuca M., Helinski D. R., Subramani S. Firefly luciferase gene: structure and expression in mammalian cells. Mol Cell Biol. 1987 Feb;7(2):725–737. doi: 10.1128/mcb.7.2.725. [DOI] [PMC free article] [PubMed] [Google Scholar]






