Abstract
The γ-aminobutyric acid neurotransmitter in the spinal cord dorsal horn plays an important role in pain modulation through primary afferent-mediated presynaptic inhibition. The weakening of γ-aminobutyric acid-mediated presynaptic inhibition may be an important cause of neuropathic pain. γ-aminobutyric acid-mediated presynaptic inhibition is related to the current strength of γ-aminobutyric acid A receptor activation. In view of this, the whole-cell patch-clamp technique was used here to record the change in muscimol activated current of dorsal root ganglion neurons in a chronic constriction injury model. Results found that damage in rat dorsal root ganglion neurons following application of muscimol caused concentration-dependent activation of current, and compared with the sham group, its current strength and γ-aminobutyric acid A receptor protein expression decreased. Immunofluorescence revealed that γ-aminobutyric acid type A receptor α2 subunit protein expression decreased and was most obvious at 12 and 15 days after modeling. Our experimental findings confirmed that the γ-aminobutyric acid type A receptor α2 subunit in the chronic constriction injury model rat dorsal root ganglion was downregulated, which may be one of the reasons for the reduction of injury in dorsal root ganglion neurons following muscimol-activated currents.
Keywords: γ-aminobutyric acid, γ-aminobutyric acid type A receptor α2 subunit, neuropathic pain, dorsal root ganglion, whole-cell patch clamp, immunofluorescence, primary afferent depolarization, paw withdrawal latency, muscimol
Research Highlights
(1) Gamma-aminobutyric acid type A receptor α2 subunit participates in the pathogenesis of neurological pain.
(2) Gamma-aminobutyric acid type A receptor α2 subunit expression was downregulated in the dorsal root ganglion in rats with chronic constriction injury, which may be one of the reasons for the decreased current of dorsal root ganglion activation at the ipsilateral side after chronic constriction injury in the sciatic nerve.
(3) The activation current of the gamma-aminobutyric acid type A receptor α2 subunit was decreased on the ipsilateral side, but increased on the contralateral side. This change resulted from the compensation mechanism of nerves.
Abbreviations
GABA, γ-aminobutyric acid; GABAAR, γ-aminobutyric acid type A receptor; CCI, chronic constriction injury
INTRODUCTION
Neuropathic pain is defined as ‘pain initiated or caused by a primary lesion or dysfunction in the nervous system’. Neuropathic pain is often reported as having a lancinating or continuous burning character and is often associated with the appearance of abnormal sensory signs, such as spontaneous pain-related behavior, allodynia (pain as a result of a stimulus that does not normally provoke pain) or hyperalgesia (an increased response to a stimulus which is normally painful)[1,2,3,4]. In normal primary afferent neurons, it is rare for a firing threshold to be reached without the input of a stimulus. However, following nerve injury, it has been demonstrated that there is a large increase in the level of spontaneous firing in afferent neurons linked to the injury site. This has been termed ectopic discharge and has also been demonstrated in humans suffering from neuropathic pain.
γ-aminobutyric acid (GABA) is a classical inhibitory neurotransmitter. GABA receptors can be divided into GABAAR, GABABR and GABACR. GABAAR are ligand-gated ion channel receptors. Spinal dorsal horn GABAA receptors are found both postsynaptically on central neurons and presynaptically on axons and/or terminals of primary sensory neurons, where they mediate primary afferent depolarization and presynaptic inhibition. GABA can act at GABAAR localized on primary afferent neurons to inhibit presynaptic neurotransmitter release and produce analgesia via a process of primary afferent depolarization. GABA mediates fast synaptic inhibition by activating ionotropic GABAAR, which are assembled from a large family of constituent subunits[5,6]. GABAAR is made up from 19 known subunits (α1-6, β1-3, γ1-3, δ, ε, θ, π and ρ1-3)[7,8] with an integral channel that is permeable to Cl-ions. The majority of GABAAR consist of α2β3γ2[9] that can interact with a range of compounds via specific binding sites that include agonists (e.g. GABA and muscimol). After injury, GABAAR positive allosteric modulators inhibit robust analgesia in animals via restoration of post-synaptic GABAAR α2 and GABAAR α3 subunit function within the spinal cord[10]. Subunit α2 is expressed at pre- and post-synaptic elements within the spinal dorsal horn and is predominantly found within superficial layers that receive nociceptive input from primary afferents[11]. Recent molecular, genetic, and pharmacological data point to α1-containing GABAAR as the “sedative” and α2- and/or α3-containing receptors as the “anxiolytic” subtypes[12].
The contribution of primary afferent depolarization to the processing of nociceptive signals and to the antihyperalgesic effect of GABAAR modulators is unknown, mainly because of the lack of suitable tools for the specific targeting of presynaptic GABAAR. In this study, through the establishment of a chronic constriction injury CCI model of sciatic nerve, we monitored GABAAR α2 subunit function and expression changes in L4-6 dorsal root ganglion neurons using whole-cell patch-clamp and immunofluorescence methods to further understand the contribution of the GABAAR α2 subunit in pathological pain.
RESULTS
Quantitative analysis of experimental animals
Ninety experimental Sprague-Dawley rats were randomly divided into three groups: sham-surgery group (n = 30), CCI ipsilateral side model group (n = 30) and CCI contralateral side model group (n = 30). CCI models were established on the ipsilateral side and contralateral side of the latter groups, and the number of modeling success was both 26. The sham-surgery group was treated the same as that in the CCI modeling method, with the exception of mp ligation with chromium suture, and 26 rats were randomly selected from the sham group. A total of 78 rats in three groups were involved in the final experiment, 20 in each group for electrophysiology experiments and six in each group for immunofluorescence experiments.
Thermal hyperalgesia of the rat CCI model
We explored the behavioral performance of hyperalgesia after CCI. Thermal hyperalgesia was measured before operation (baseline) and on 1, 3, 7, 12 and 15 days after operation. Thermal hyperalgesia was assessed using the hot plate test. Heat-evoked thermal withdrawal latency of CCI rats began to significantly decrease from 1 to 15 days post-surgery. The sham-operated and CCI contralateral side showed no changes in baseline and after surgery (P > 0.05); the thermal withdrawal latency of the CCI ipsilateral side were significantly different before and after surgery (P < 0.01), and compared with sham-operated rats, thermal withdrawal latency of CCI rats significantly decreased (P < 0.05; Figure 1).
Figure 1.
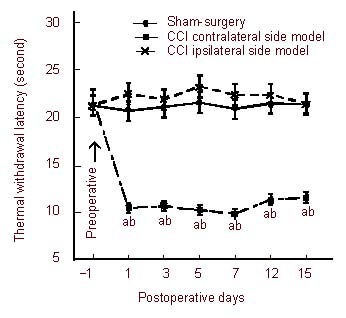
Paw withdrawal latency in rats following chronic constriction injury (CCI) in the sciatic nerve.
Withdrawal thresholds to thermal stimulation were measured by the hot plate test. Tests were carried out on day 1 before operation and on days 1, 3, 5, 7, 12 and 15 after operation. Thermal hyperalgesia is indicated by a significant reduction in the withdrawal latency.
aP < 0.01, vs. sham-surgery group; bP < 0.01, vs. preoperative CCI ipsilateral side. Data were expressed as mean ± SEM, n = 20. A homogeneity test for variance was performed followed by one-way analysis of variance, and a two-group comparison was conducted using the least significant difference t-test.
Changes in GABAAR electrophysiological features on dorsal root ganglion neurons in CCI rats
Freshly isolated neurons from the L4-6 dorsal root ganglion in the diameter range of 15–45 μm were used in the present study. In the majority of neurons examined (82.4%, 89/108), muscimol (10-8-10-3 M) induced a concentration-dependent inward current. We recorded three typical types of muscimol-activated currents: fast desensitization type (3/108), mixed (93/108) and slow desensitization type (12/108) (Figure 2A). This muscimol-activated current could be reversibly blocked by bicuculline (10-4 M), a selective antagonist of GABAAR, indicating this current was mediated by GABAAR (Figure 2B).
Figure 2.
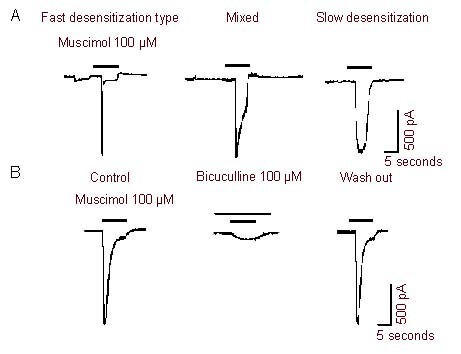
Muscimol-activated currents in rat dorsal root ganglion neurons.
(A) Three typical types of muscimol-activated currents: fast desensitization type (n = 3), mixed type (n = 93) and slow desensitization type (n = 12). n represents the number of positive cells of this type of current.
(B) The inward current evoked by muscimol (10-4 M) could be blocked by the γ-aminobutyric acid type A receptor antagonist bicuculline (10-4 M) (n = 6). Bars above each current indicated the drugs and duration of drug application.
Muscimol induced a concentration-dependent (10-8–10-3 M) inward current on L4-6 dorsal root ganglion neurons from the sham-operated, CCI ipsilateral side model, and CCI contralateral side model. Figure 3A shows the sham-operated, CCI ipsilateral side, and CCI contralateral side curve traces for muscimol. The muscimol-activated current amplitude (Imuscimol) was significantly reduced in the CCI ipsilateral side model, while Imuscimol significantly increased on the CCI contralateral side. Imuscimol (10-4 M) in the sham-operated, CCI ipsilateral side model, and CCI contralateral side model rats was 1 487 ± 92.9 pA (n = 6), 541 ± 63.3 pA (n = 6) and 1 945 ± 28.4 pA (n = 6), respectively, among the three groups, which were significantly different (P < 0.01). From concentration-response curves of muscimol-activated neurons (Figure 3B), it can be seen that, (a) the dose-response curve for the CCI ipsilateral side is shifted downwards compared with sham-operated rats; the CCI contralateral side is shifted upwards; (b) the EC50 value in the sham-operated, CCI ipsilateral side model, and CCI contralateral side model groups are very close (14.7 ± 1.8 μM; 14.3 ± 0.9 μM; 14.4 ± 1.6 μM; n = 6), and are not significantly different between groups (P > 0.05).
Figure 3.
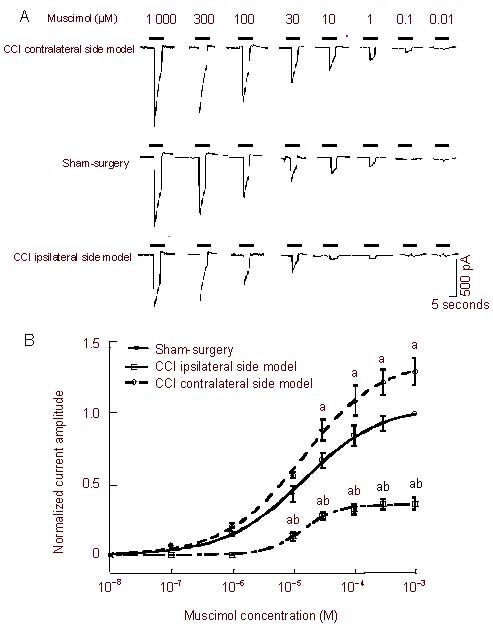
Comparison of muscimol-activated currents of dorsal root ganglion neurons.
(A) Sequential current-traces illustrating the concentration-dependent muscimol-activation current amplitude.
(B) The concentration-response curves for muscimol-activation. Data at each point are represented as mean ± SEM of 6–17 neurons. All muscimol-induced currents were normalized. aP < 0.05, vs. sham-surgery group; bP < 0.01, vs. chronic constriction injury (CCI) contralateral side model group.
The EC50 (half number of effective concentrations) value in the sham-surgery, CCI ipsilateral side model, and CCI contralateral side model groups are very close (14.7 ± 1.8 μM, 14.3 ± 0.9 μM, 14.4 ± 1.6 μM; n = 6), and no significant difference among the three groups was observed (P > 0.05). A homogeneity test for variance was performed followed by one-way analysis of variance, and two-group comparison was conducted using the least significant difference t-test.
Distribution of GABAAR α2 subunit expressing neurons in the dorsal root ganglion of CCI rats
Immunofluorescence staining displayed that GABAAR α2 subunit expression was widely distributed in dorsal root ganglion neurons of all three groups, and were mainly located in the cell membranes of various diameters at 12 days after modeling (Figure 4). Quantitative analysis demonstrated that GABAAR α2 subunit expression was decreased in the CCI ipsilateral side model group when compared with the sham-surgery group at 12 days after modeling (P < 0.01), and expression levels were highest in the CCI contralateral side model group.
Figure 4.
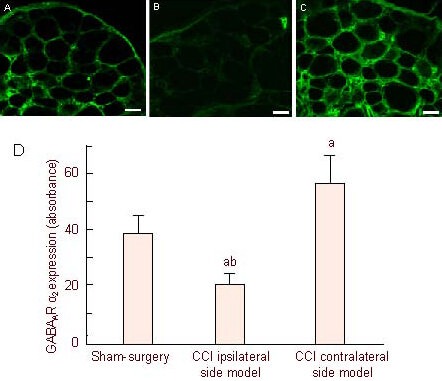
Confocal images of γ-aminobutyric acid type A receptor α2 (GABAAR α2) subunit expression in the chronic constriction injury (CCI)-induced L4-6 dorsal root ganglion (immunofluorescence staining, fluorescence microscope, × 20) at day 12 after injury.
GABAAR α2 subunit immunoreactivity was seen in dorsal root ganglion neuron cell membranes. Neurites of dorsal root ganglion neurons formed networks. Positive cells presented green fluorescent labeling: Mild fluorescence intensity in the sham group (A), weak fluorescence intensity in the CCI surgery group (B), high fluorescence intensity in the CCl contralateral group(C). Scale bars (A–C): 50 μM.
(D) The histogram shows the expression of GABAAR α2 in the dorsal root ganglion. aP < 0.01, vs. sham-surgery group; bP < 0.01, vs. CCl contralateral side model group. Data are expressed as mean ± SEM of six rats in each group.
Based on the above experiments, GABAAR α2 subunit levels in sensory neurons may underlie the development of pain. We examined alterations to GABAAR α2 subunits in the ipsilateral L4-6 dorsal root ganglion on days 3, 7, 12 and 15 after operation using immunofluorescence. GABAAR α2 subunit-positive signals in the membrane of dorsal root ganglion neurons decreased (Figure 5).
Figure 5.
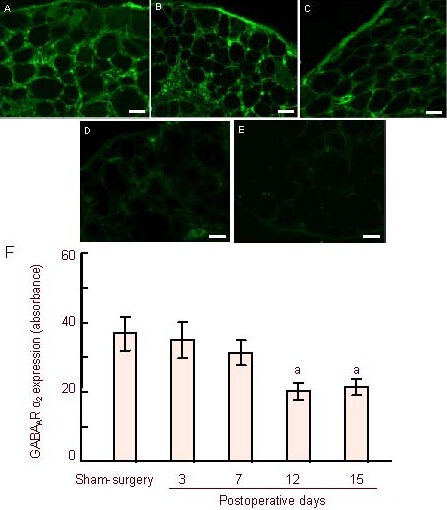
Changes in γ-aminobutyric acid type A receptor α2 (GABAAR α2) subunit-positive cells in the ipsilateral L4-6 dorsal root ganglion from day 3 before injury to day 15 after injury.
Positive cells labeled green (A): GABAAR α2 subunit expression in the sham-surgery group at 12 days. (B–E) GABAAR α2 subunit expression in the ipsilateral side from 3, 7, 12, and 15 days. GABAAR α2 immunoreactivity did not change significantly in the rat L4-6 dorsal root ganglion at 3 days, decreased at 7 days, but did not reach statistical significance after injury. A significant decrease was observed at day 12 and continued until day 15. Scale bars in A–E: 50 μm.
(F) Histogram shows the mean absorbance of GABAAR α2-positive neurons from the dorsal root ganglion. aP < 0.01, vs. sham-surgery group. Data are expressed as mean ± SEM of six rats in each group. A homogeneity test for variance was performed followed by one-way analysis of variance, and a two-group comparison was conducted using the least significant difference t-test.
Changes in absorbance values on the CCI contralateral side were not seen on day 3 (34.9 ± 5.21; n = 6) or day 7 (31.1 ± 3.50; n = 6) after surgery. However, the mean absorbance began to significantly attenuate by day 12 (20.2 ± 2.24; n = 6) after operation when compared with the sham-surgery group (37.2 ± 5.31; n = 6; P < 0.05). This significant decrease continued from 12 to 15 days (20.2±2.24; n = 6).
DISCUSSION
In the present study, we found that the CCI ipsilateral side muscimol-activated current amplitudes were significantly lower than that in the sham-surgery group, while the CCI contralateral side model group was greater. Although there was no nerve damage to the contralateral side, it still produced a series of changes in the electrophysiological properties of GABAAR and the expression of the GABAAR α2 subunit. Such compensatory changes may be due to injury information from the unilateral nerve into the bilateral spinal cord. Several possible explanations could account for this compensatory enhancement. Synaptic plasticity of neurons is believed to be fundamental to pathological pain and the importance of spinal sensitization is well recognized. This synaptic arrangement can be the substrate of local disinhibition, which may play a key role in sensory processing[13,14]. In the spinal cord, the GABAAR α2 subunit is densely expressed in the superficial layers of the dorsal horn, the main termination area of primary nociceptors[15].
At this site, the GABAAR α2 subunit is found not only postsynaptically on central neurons, where they cause classical hyperpolarization, but most likely presynaptically on the terminals of primary sensory neurons[15,16]. These terminals are depolarized by GABAA receptors[17], because their intracellular chloride concentration is kept above the electrochemical equilibrium by the chloride importer NKCC1. This primary afferent depolarization causes presynaptic inhibition, i.e., a reduction in synaptic glutamate release, possibly through inactivation of presynaptic sodium channels and/or through activation of a shunting conductance, both of which can result in inhibition of action potential propagation into presynaptic terminals[18]. Both processes will result in the reduction of nociceptive input to the spinal cord.
Indeed, GABAergic interneurons intercalated within the circuitry linking primary afferents and spinal projection neurons may play a dual role in controlling network excitability[13,19]. This study found that the GABAAR α2 subunit was involved in neuropathic pain. The GABAAR containing α2 subunit and α3 subunit are current targets in the battle to develop new pain medications, as they are expressed in the spinal cord where increasing inhibitory drive should result in analgesia[20]. We have found by immunofluorescence staining that the GABAAR α2 subunit is widely distributed in dorsal root ganglion neuron cell membranes of various diameters. Being consistent with electrophysiological experiments, at 12 days after CCI, the positive signal of the CCI ipsilateral side is reduced, however the contralateral side shows a compensatory increase. Fukuoka et al[21] examined the effects of unilateral L5 spinal nerve ligation using in situ hybridization, and found that GABAAR α2 subunit mRNA decreased in ipsilateral L5 dorsal root ganglion neurons but did not reach statistical significance. This result may explain why different damage models cause varying degrees of neuropathic pain. The CCI model is reported to be more sensitive to mechanical stimuli than the spinal nerve ligation model[22]. According to time-dependent changes of GABAAR α2 subunit expression, a significant decrease was observed at day 12 and continued until day 15. However, in the hot plate test, values for heat-evoked thermal withdrawal latency of CCI rats began to significantly decrease from 1 to 15 days post-surgery. This result may be due to the fact that there was no damage to dorsal root ganglion GABAAR involved in neuropathic pain occurrence and development of hyperalgesia, and that thermal hyperalgesia could not afford a leading role.
GABA, the most abundant and important inhibitive neurotransmitter in the central nervous system, plays a regulatory role in the regeneration of various nerve cells[23,24]. GABA receptor activation has been shown to lead to the depolarization of primary afferent terminals in the spinal dorsal horn, and significantly reduce the transmission of excitatory impulses in primary afferent fibers[25]. If GABA receptor-mediated presynaptic inhibition reduced, this may cause damage to transfer to the message center and the spinal cord, causing sensitization of neurons, leading to neuropathic pain. We have found a loss of presynaptic GABAAR-mediated inhibition in the dorsal root ganglion of CCI rats. This can be regarded as a key feature that contributes to the signs and symptoms of pain associated with neuropathic injury. After CCI, there is no specific loss to GABAergic neurons in the rat spinal dorsal horn after CCI in the sciatic nerve[26]. Other studies have shown this in the spared nerve injury model (e.g. partial sciatic nerve injury) suggesting that GABA synthesis is down-regulated[27]. However, in wild-type (α2fl/fl) mice and mice lacking α2-GABAAR, specifically in primary nociceptors (sns-α2-/-), GABAAR currents and dorsal root potentials are of normal amplitude in vitro, and mice show normal response thresholds to thermal and mechanical stimulation in vivo, and develop normal inflammatory and neuropathic pain sensitization. However, the effect of benzodiazepines such as diazepam and midazolam was significantly reduced[28].
Studies have demonstrated that the GABAAR α2 subunit participates in pain perception conduction in CCI rats. In patch clamp experiments, we found that there is a change in GABAAR function in CCI rats. At the same time, molecular biology studies confirmed the change in GABAAR α2 subunit expression. In addition, weakening of presynaptic inhibition of primary afferents can be due to the reduction in presynaptic GABAAR expression. On the other hand, weakening of presynaptic inhibition of primary afferents can also be interpreted as presynaptic GABAAR phosphorylation or dephosphorylation to reduce the activity of GABAAR[29]. In the progression of neuropathic pain, a large number of intracellular signaling pathways are involved in GABAAR subunit phosphorylation, such as cAMP, PKA, cGMP/PKC, NO, CaMKII and tyrosine kinase[30,31]. GABAAR phosphorylation or dephosphorylation will be our future research direction, and we hope to shed further light on neuropathic pain development.
MATERIALS AND METHODS
Design
A randomized, controlled, animal experiment.
Time and setting
Animal experiments were performed at the Department of Physiology, Shihezi University Medical College, China from August 2010 to November 2011.
Materials
Female, healthy, Sprague-Dawley rats, aged 8–10 weeks, weighing 250–280 g, were provided by the Experimental Animal Center of Xinjiang Medical University, China (license No. SCXK 2003-0001). Rats were housed in separate cages with a specific pathogen-free level barrier environment at 24 ± 3°C, relative humidity of 40–70%, 100–120 lx/12-hour light illumination, and allowed free access to food and water. The protocol was conducted in accordance with the Guidance Suggestions for the Care and Use of Laboratory Animals, formulated by the Ministry of Science and Technology of China[32].
Methods
Establishment of the rat CCI model
Surgical procedures were performed as previously described by Bennett et al[33]. Rats were anesthetized with an intraperitoneal injection of 1% (w/v) sodium pentobarbital, and the left sciatic nerve trunk was exposed under sterile conditions. Proximal to the sciatic trifurcation, approximately 7 mm of nerve was freed, and 4 tight ligatures of chrome catgut were placed around the sciatic nerve with about 1 mm spacing. The same surgical procedure was followed until the sciatic nerve was exposed, but no ligatures were applied. All surgical procedures were performed by the same person under aseptic conditions. There were significant differences between the CCI contralateral side model and sham-surgery groups, and paw withdrawal latency values in the CCI ipsilateral side model group were declined by at least 30%, which indicated modeling success. Six rats were randomly selected from each group.
Behavioral testing of thermal sensitivity of the rat CCI model
Thermal hyperalgesia was measured using a 58°C hot plate[34,35], and expressed as paw withdrawal latency of the left hind paw. The constant temperature water bath apparatus was produced in Jintan, China (type SHZ-82). Three measurements of thermal nociceptive threshold were taken for each rat, at 4-minute intervals, and the mean of three measurements was regarded as the paw withdrawal latency. Maximum latency was defined as 30 seconds, after which time the animals were removed from the hot plate to prevent tissue damage. Behavioral testing at the following different time points was performed before operation on 1, 3, 5, 7, 12 and 15 days after operation. After behavioral testing at each time point, all six rats from each group were killed, and L4-6 dorsal root ganglions were dissected out for experimentation.
Immunofluorescence staining for GABAAR α2 expression in the dorsal root ganglion
Rats were anaesthetized with 1% (w/v) sodium pentobarbital, and then perfused through the aorta with 0.9% (w/v) normal saline, followed by fresh 4% (w/v) paraformaldehyde in PBS for 10 minutes for tissue fixation. The lumbar dorsal root ganglion at the level of L4-6 to nerve injury was removed rapidly and placed in 4% (w/v) paraformaldehyde in PBS for 24 hours. The L4-6 dorsal root ganglia were cut into 5 μm slices with a freezing microtome. The sections were treated with 0.3% (v/v) H2O2 for 10 minutes and blocked with 3% (w/v) bovine serum albumin in 0.3% (v/v) H2O2. Immunofluorescence staining was performed with goat anti-GABAAR α2 polyclonal antibody (1:200; Santa Cruz Biotechnology, CA, USA), overnight at 4°C. The sections were incubated for 1 hour in solution containing FITC-labeled rabbit anti-goat secondary antibody (1:100; Zhongshan Golden Bridge, Beijing, China) at room temperature. Slides were then examined by confocal microscopy. Quantitative analysis of GABAAR α2 expression in the dorsal root ganglion was performed by measuring the mean absorbance following laser confocal microscopy (LSM510; Carl Zeiss, Jena, Germany) and using analysis software (hp9001; Carl Zeiss, Jena, Germany).
Electrophysiological recordings of dorsal root ganglion neurons
Whole-cell patch clamp recordings were carried out at room temperature (22–24°C) using a whole-cell patch clamp amplifier[36]. Currents were recorded from single dorsal root ganglion neurons in vitro using an Axon 700B amplifier (Axon, San Jose, USA) and the pCLAMP 10.2 hardware and software (Axon, Silicon Valley, USA). The micropipettes were filled with internal solution containing (mM): KCI 140, MgCI2 2.5, HEPES 10, EGTA 11 and ATP 5. The pH was adjusted to 7.2 with KOH, and osmolarity was adjusted to 310 mOsm/L with glucose. Cells were bathed in an external solution containing (mM): NaCI 150, KCI 5, CaCI2 2.5, MgCI2 2, HEPES 10, D-glucose 10. Osmolarity was adjusted to 340 mOsm/L with glucose, and pH was adjusted to 7.4 with NaOH. The resistance of the recording pipette was in the range of 2-5 MΩ. A small patch of membrane underneath the tip of the pipette was aspirated to form a gigaseal, and then a negative pressure was applied to rupture it, thus establishing a whole-cell configuration. The adjustment of capacitance compensation and series resistance compensation was carried out before the membrane currents were recorded. The holding potential was set at –60 mV, except when indicated otherwise. Membrane currents were filtered at 10 kHz.
Statistical analysis
Data were analyzed with SPSS 13.0 software (SPSS, Chicago, IL, USA) and presented as mean ± SEM. A homogeneity test for variance was performed followed by one-way analysis of variance, and two-group comparison was conducted using the least significant difference t-test. A P < 0.05 was considered statistically significant.
Acknowledgments
We wish to thank Dr. Hongzhen Hu from the Department of Integrative Biology and Pharmacology, University of Texas Health Science Center for reading the manuscript and for valuable suggestions.
Footnotes
Funding: This study was supported by the Youth Science and Technology Innovation Special Foundation of Xinjiang Production and Construction Corps, China, No. 2010JC33.
Conflicts of interest: None declared.
Ethical approval: The experiment is approved by the Policy for Rodent Survival Surgery provided by the Animal Care Committee of Shihezi University School of Medicine, China.
(Edited by Zou WY, Jia DL/Yang Y/Song LP)
REFERENCES
- [1].Bridges D, Thompson SW, Rice AS. Mechanisms of neurpathic pain. Br J Anaesth. 2001;87(1):12–26. doi: 10.1093/bja/87.1.12. [DOI] [PubMed] [Google Scholar]
- [2].Wilson M. Overcoming the challenges of neuropathic pain. Nurs Stand. 2002;16(33):47–53. doi: 10.7748/ns2002.05.16.33.47.c3190. [DOI] [PubMed] [Google Scholar]
- [3].Randic M, Hecimovic H, Ryu PD. Substance P modulates glutamate-induced currents in acutely isolated rat spinal dorsal horn neurons. Neurosci Lett. 1990;117(1-2):74–80. doi: 10.1016/0304-3940(90)90122-p. [DOI] [PubMed] [Google Scholar]
- [4].Yang HW, Hu XD, Zhang HM, et al. Roles of CaMKII, PKA, and PKC in the induction and maintenance of LTP of C-fiber-evoked field potentials in rat spinal dorsal horn. J Neurophysiol. 2004;91(3):1122–1133. doi: 10.1152/jn.00735.2003. [DOI] [PubMed] [Google Scholar]
- [5].Barnard EA, Skolnick P, Olsen RW, et al. International Union of Pharmacology. XV. Subtypes of γ-aminobutyric acid A receptors: classification on the basis of subunit structure and function. Pharmacol Rev. 1998;50(2):291–313. [PubMed] [Google Scholar]
- [6].Duveau V, Laustela S, Barth L, et al. Spatiotemporal specificity of GABAA receptor-mediated regulation of adult hippocampal neurogenesis. Eur J Neurosci. 2011;34(3):362–373. doi: 10.1111/j.1460-9568.2011.07782.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Olsen RW, Sieghart W. GABAA receptors: subtypes provide diversity of function and pharmacology. Neuropharmacology. 2009;56(1):141–148. doi: 10.1016/j.neuropharm.2008.07.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Olsen RW, Sieghart W. International Union of Pharmacology. LXX. Subtypes of γ-aminobutyric acid A receptors: classification on the basis of subunit composition, pharmacology, and function. Update. Pharmacol Rev. 2008;60(3):243–260. doi: 10.1124/pr.108.00505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Marutha Ravindran CR, Ticku MK. Tyrosine kinase phosphorylation of GABAA receptor subunits following chronic ethanol exposure of cultured cortical neurons of mice. Brain Res. 2006;1086(1):35–41. doi: 10.1016/j.brainres.2006.02.106. [DOI] [PubMed] [Google Scholar]
- [10].Munro G, Erichsen HK, Rae MG, et al. A question of balance-positive versus negative allosteric modulation of GABA(A) receptor subtypes as a driver of analgesic efficacy in rat models of inflammatory and neuropathic pain. Neuropharmacology. 2011;61(1-2):121–132. doi: 10.1016/j.neuropharm.2011.03.017. [DOI] [PubMed] [Google Scholar]
- [11].Knabl J, Witschi R, Hösl K, et al. Reversal of pathological pain through specific spinal GABAA receptor subtypes. Nature. 2008;451(17):330–334. doi: 10.1038/nature06493. [DOI] [PubMed] [Google Scholar]
- [12].Atack JR. GABAA receptor subtype-selective modulators. I. α2/α3-selective agonists as non-sedatinganxiolytics. Curr Top Med Chem. 2011;11(9):1176–1202. doi: 10.2174/156802611795371350. [DOI] [PubMed] [Google Scholar]
- [13].Labrakakis C, Lorenzo LE, Bories C, et al. Inhibitory coupling between inhibitory interneurons in the spinal cord dorsal horn. Mol Pain. 2009;12(5):24–31. doi: 10.1186/1744-8069-5-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Zeilhofer HU, Wildner H, Yévenes GE. Fast synaptic inhibition in spinal sensory processing and pain control. Physiol Rev. 2012;92(1):193–235. doi: 10.1152/physrev.00043.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Bohlhalter S, Weinmann O, Mohler H, et al. Laminar compartmentalization of GABAA-receptor subtypes in the spinal cord: an immunohistochemical study. J Neurosci. 1996;16(1):283–297. doi: 10.1523/JNEUROSCI.16-01-00283.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Persohn E, Malherbe P, Richards JG. In situ hybridization histochemistry reveals a diversity of GABAA receptor subunit mRNAs in neurons of the rat spinal cord and dorsal root ganglia. Neuroscience. 1991;42(2):497–507. doi: 10.1016/0306-4522(91)90392-2. [DOI] [PubMed] [Google Scholar]
- [17].Labrakakis C, Tong CK, Weissman T, et al. Localization and function of ATP and GABAA receptors expressed by nociceptors and other postnatal sensory neurons in rat. J Physiol. 2003;549(Pt 1):131–142. doi: 10.1113/jphysiol.2002.031963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Kullmann DM, Ruiz A, Rusakov DM, et al. Presynaptic, extrasynaptic and axonal GABAA receptors in the CNS: where and why? Prog Biophys Mol Biol. 2005;87(1):33–46. doi: 10.1016/j.pbiomolbio.2004.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Personius KE, Chang Q, Mentis GZ. Reduced gap junctional coupling leads to uncorrelated motor neuron firing and precocious neuromuscular synapse elimination. Proc Natl Acad Sci U S A. 2007;104(28):11808–11813. doi: 10.1073/pnas.0703357104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Nickolls S, Mace H, Fish R, et al. A comparison of the α2/3/5 selective positive allosteric modulators L-838, 417 and TPA023 in preclinical models of inflammatory and neuropathic pain. Adv Pharmacol Sci. 2011;2011:608912. doi: 10.1155/2011/608912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Fukuoka T, Tokunaga A, Kondo E, et al. Change in mRNAs or neuropeptides and the GABA(A) receptor in dorsal root ganglion neurons in a rat experimental neuropathic pain model. Pain. 1998;78(1):13–26. doi: 10.1016/S0304-3959(98)00111-0. [DOI] [PubMed] [Google Scholar]
- [22].Kim KJ, Yoon YW, Chung JM. Comparison of three rodent neuropathic pain models. Exp Brain Res. 1997;113(2):200–206. doi: 10.1007/BF02450318. [DOI] [PubMed] [Google Scholar]
- [23].Luyt K, Slade TP, Dorward JJ, et al. Developing oligodendrocytes express functional GABA(B) receptors that stimulate cell proliferation and migration. J Neurochem. 2007;100(3):822–840. doi: 10.1111/j.1471-4159.2006.04255.x. [DOI] [PubMed] [Google Scholar]
- [24].Chiba C, Matsushima O, Muneoka Y, et al. Time course of appearance of GABA and GABA receptors during retinal regeneration in the adult newt. Brain Res Dev Brain Res. 1997;98(2):204–210. doi: 10.1016/s0165-3806(96)00195-2. [DOI] [PubMed] [Google Scholar]
- [25].Willis WD. John Eccles’ studies of spinal cord presynaptic inhibition. Prog Neurobiol. 2006;78(3-5):189–214. doi: 10.1016/j.pneurobio.2006.02.007. [DOI] [PubMed] [Google Scholar]
- [26].Schoffnegger D, Heinke B, Sommer C, et al. Physiological properties of spinal lamina II GABAergic neurons in mice following peripheral nerve injury. J Physiol. 2006;577(3):869–878. doi: 10.1113/jphysiol.2006.118034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Polgár E, Hughes DI, Arham AZ, et al. Loss of neurons from laminas I-III of the spinal dorsal horn is not required for development of tactile allodynia in the spared nerve injury model of neuropathic pain. J Neurosci. 2005;25(28):6658–6666. doi: 10.1523/JNEUROSCI.1490-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Witschi R, Punnakkal P, Paul J, et al. Presynaptic α2-GABAA receptors in primary afferent depolarization and spinal pain control. J Neurosci. 2011;31(22):8134–8142. doi: 10.1523/JNEUROSCI.6328-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Brandon NJ, Delmas P, Kittler JT, et al. GABAA receptor phosphorylation and functional modulation in cortical neurons by a protein kinase C-dependent pathway. J Biol Chem. 2000;275(49):38856–38862. doi: 10.1074/jbc.M004910200. [DOI] [PubMed] [Google Scholar]
- [30].Marutha Ravindran CR, Ticku MK. CaMKII phosphorylation of the GABA(A) receptor: receptor subtype-and synapse-specific modulation. J Physiol. 2009;587(10):2115–2125. doi: 10.1113/jphysiol.2009.171603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Marutha Ravindran CR, Mehta AK, Ticku MK. Effect of chronic administration of ethanol on the regulation of tyrosine kinase phosphorylation of the GABAA receptor subunits in the rat brain. Neurochem Res. 2007;32(7):1179–1187. doi: 10.1007/s11064-007-9288-y. [DOI] [PubMed] [Google Scholar]
- [32].The Ministry of Science and TechnoIogy of the People's Republic of China. Guidance Suggestions for the Care and Use of Laboratory Animals. 2006 Sep 30; [Google Scholar]
- [33].Bennett GJ, Xie YK. A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain. 1988;33(1):87–107. doi: 10.1016/0304-3959(88)90209-6. [DOI] [PubMed] [Google Scholar]
- [34].Bianchi M, Sacerdote P, Ricciardi-Castagnoli P, et al. Central effects of tumor necrosis factor alpha and interleukin-1 alpha on nociceptive thresholds and spontaneous locomotor activity. Neurosci Lett. 1992;148(1-2):76–80. doi: 10.1016/0304-3940(92)90808-k. [DOI] [PubMed] [Google Scholar]
- [35].Su L, Wang C, Yu YH, et al. Role of TRPM8 in dorsal root ganglion in nerve injury-induced chronic pain. BMC Neurosci. 2011;12(20):1186–1471. doi: 10.1186/1471-2202-12-120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Si JQ, Zhang ZQ, Li CX, et al. Modulatory effect of substance P on GABA-activated currents from rat dorsal root ganglion. Acta Pharmacol Sin. 2004;25(5):623–629. [PubMed] [Google Scholar]


