Abstract
α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors are considered to play a crucial role in synaptic plasticity in the developing visual cortex. In this study, we established a rat model of binocular form deprivation by suturing the rat binocular eyelids before eye-opening at postnatal day 14. During development, the decay time of excitatory postsynaptic currents mediated by α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors of normal rats became longer after eye-opening; however, the decay time did not change significantly in binocular form deprivation rats. The peak value in the normal group became gradually larger with age, but there was no significant change in the binocular form deprivation group. These findings indicate that binocular form deprivation influences the properties of excitatory postsynaptic currents mediated by α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors in the rat visual cortex around the end of the critical period, indicating that form stimulation is associated with the experience-dependent modification of neuronal synapses in the visual cortex.
Keywords: α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors, excitatory postsynaptic currents, whole-cell recording, visual cortex, binocular form deprivation, visual development, neural development, regeneration, neural regeneration
Research Highlights
-
(1)
Binocular form deprivation influenced the properties of excitatory postsynaptic currents mediated by α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors in rat visual cortex around the end of the critical period.
-
(2)
During development, the decay time of excitatory postsynaptic currents mediated by α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors became longer and the peak value became larger after eye-opening. This age-dependent change was inhibited by a lack of form stimulation.
Abbreviations
AMPA, α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid; EPSCs, excitatory postsynaptic currents; AMPA-EPSCs, excitatory postsynaptic currents mediated by the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor; PSCs, postsynaptic currents; PD, postnatal day
INTRODUCTION
The visual systems of humans and mammals develop from an immature to a mature state at an early postnatal stage[1], adapting to the external environment and internal gene modulation[2,3,4]. This sensitive stage is called the critical period of visual development[5]. During the critical period, most amblyopia resulting from an abnormal visual environment can be cured. However, visual function is not enhanced beyond the critical period because of changes in visualcortical plasticity[6].
α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors are considered to play a crucial role in synaptic plasticity in the developing visual cortex[7]. Excitatory postsynaptic currents (EPSCs) were rapidly depressed by a test pulse stimulation in an early postnatal stage via a Ca(2+)-permeable AMPA receptor-dependent mechanism[7,8]. Changes in sensory experience can regulate the synaptic levels of Ca(2+)-permeable AMPA receptors, using dark-reared animals[9]. Dark-rearing is a condition of complete deprivation of visual inputs while binocular suture is a condition of diffuse light stimulation without form vision[10,11]. However, binocular form deprivation rats cannot properly fixate at any time, and stimulation with diffuse light results in an abnormal binocular interaction in the visual cortex[10]. Binocular form deprivation can closely mimic the symptoms of amblyopia caused by ptosis and be applied to investigate the mechanisms underlying the onset of amblyopia[9,12]. Binocular form deprivation influences the developmental depressions of N-methyl-D-aspartate receptor subunit 2A in the rat visual cortex around the end of the critical period. Regarding abnormal visual experience, there is no report discussing the effects of binocular form deprivation on the EPSCs mediated by AMPA receptors (AMPA-EPSCs) in the rat visual cortex. In the visual cortex, the receptor component of postsynaptic currents (PSCs) consists of gamma-aminobutyric acid, AMPA and N-methyl-D-aspartate receptors[5]. AMPA-EPSCs can be isolated using bicuculline methiodide (a blocker of gamma-aminobutyric acid receptors) and D,L-2-amino-5-phosphonovalerate (a blocker of N-methyl-D-aspartate receptors), and assessed using 6-cyano-7-nitroquinoxaline-2,3-dione (a blocker of AMPA receptors). Therefore, we sutured rat binocular eyelids before eye-opening and reared them in the same conditions as normal rats. We recorded the PSCs of neurons located in layers II–IV of the visual cortex, in brain slices, using patch-clamp whole-cell recording, and then isolated AMPA-EPSCs pharmacologically to analyze the kinetic changes.
RESULTS
Quantitative analysis of experimental animals
A total of 40 Long Evans rats were divided equally and randomly into normal and binocular form deprivation groups. At postnatal days (PD) 14, 21, 35 and 49, the numbers of rats in the normal group were 5, 5, 5 and 5, respectively. At PD 21, 35 and 49, the numbers of rats in the binocular form deprivation group were 6, 7 and 7, respectively. A total of 59 cells in layers II–IV of the rat visual cortex were recorded with the same stimulus of 0.5 mA and 100 μs duration, enabling isolation of AMPA-EPSCs by blocking the gamma-aminobutyric acid receptor-generated and N-methyl-D-aspartate-receptorgenerated current components (Figure 1). Among them, three cells were discarded because their input resistance and series resistance changed by more than 15%[9]; 56 cells met the criteria[9] (input resistance ≥ 200 MΩ, series resistance ≥ 25 MΩ) for analysis. At PD 14, 21, 35 and 49, the numbers of analyzed cells in the normal group were 8, 9, 7 and 7, respectively. At PD 21, 35 and 49, the numbers of cells in the binocular form deprivation group were 9, 8 and 8, respectively.
Figure 1.
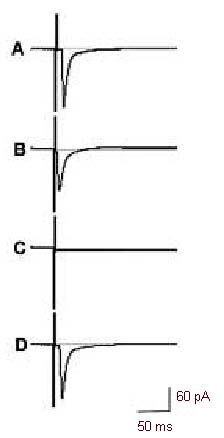
Isolation of AMPA-EPSCs in the rat visual cortex.
Postsynaptic currents (PSCs) were evoked with a stimulus of 0.5 mA and 100 μs duration, and the voltages were clamped at –30 mV. The vertical line of each trace represents the stimulus event.
In the same cell, trace A represents a PSC recorded without using any receptor blockers (AMPA or N-methyl-D-aspartate or gamma-aminobutyric acid receptor blocker bicuculline methiodide, or D,L-2-amino5-phosphonovalerate, or 6-cyano-7-nitroquinoxaline-2, 3-dione).
Trace B is an AMPA-EPSC recorded by adding bicuculline methiodide (20 μM) and D,L-2-amino-5phosphonovalerate (50 μM) to the artificial cerebrospinal fluid (at least 5 minutes).
Trace C indicates that all the components of the PSC were blocked by adding bicuculline methiodide (20 μM), 6-cyano-7-nitroquinoxaline-2,3-dione (20 μM) and D,L-2-amino-5-phosphonovalerate (50 μM) to the artificial cerebrospinal fluid.
Trace D represents a PSC recorded after washing out all the blockers showing almost complete recovery of the PSC and that the isolation of AMPA-EPSCs is stable.
AMPA-EPSCs: Excitatory postsynaptic currents (EPSCs) mediated by α-amino-3-hydroxy-5-methyl-4isoxazolepropionic acid (AMPA) receptors.
Electrophysiological properties of neuronal AMPA-EPSCs in the rat visual cortex
We analyzed the decay time (from the summit to the baseline), rise time (from the baseline to the summit) and peak value (amplitude) of neuronal AMPA-EPSCs to demonstrate their electrophysiological properties in rat visual cortex during development (Table 1). In the normal group, the decay time of AMPA-EPSCs in the visual cortex became longer after the rat eyes were opened and the rat matured (P < 0.05); however, the decay time did not change significantly in the binocular form deprivation group (P > 0.05). At PD 35 or PD 49, the decay time of AMPA-EPSCs in the binocular form deprivation group was lower than that in the normal group at the same PD (P < 0.05). The rise time of AMPA-EPSCs did not differ significantly between the normal and binocular form deprivation group at the same PD (P > 0.05). The peak value became larger with age in the normal group (P < 0.05), but it did not change significantly in the binocular form deprivation group (P > 0.05). At PD 35 or PD 49, the peak value of AMPA-EPSCs in the binocular form deprivation group was lower than that in the normal group at the same PD (P<0.05).
Table 1.
Effects of binocular form deprivation on the properties of AMPA-EPSCs in rat visual cortex

DISCUSSION
To investigate the AMPA-EPSCs around the end of the critical period, we performed experiments at PD 14–49 in rats, because rats had their peak plasticity at PD 14–21, with the critical period ending at PD 35[12]. From PD 14 to PD 49, we found that, following eye-opening, blockage of the gamma-aminobutyric acid and N-methyl-D-aspartate components resulted in longer decay times and larger peak AMPA-EPSC values, but the rise time and peak values of AMPA-EPSCs of visual cortical neurons did not change markedly with visual inputs in normal rats. During early postnatal life, there are many more silent synapses in the visual cortex compared with other developmental stages and proteins responsive to AMPA receptors are lacking[13,14]. The spine head size of dendritic spines is correlated with the synaptic AMPA receptor content[15]. With increasing visual experience, the synaptic transmission mediated by AMPA gradually increases[14]. After the second postnatal week, a significant increase in the relative contribution of the AMPA conductance[13], and AMPAR-mediated responses showed age-dependent changes in the rectification properties of their I–V relationships[16]. These changes in AMPA-EPSCs may play an important role in the experience-dependent modification of neuronal synapses in the visual cortex[17].
Binocular form deprivation inhibits normal developmental changes such that the decay time of AMPA-EPSCs became longer and the peak values became larger with age. Because the visual development of binocular form deprivation rats is driven mainly by light vision, with a complete lack of form vision, form stimulation may play an important role in the experience-dependent modification of neuronal synapses in the visual cortex. The critical period of visual development may be delayed by form deprivation, such as by dark-rearing[10].
MATERIALS AND METHODS
Design
This was a randomized, controlled, animal study.
Time and Setting
Experiments were performed in the Laboratory of Southwest Eye Hospital from August 2010 to May 2011.
Materials
A total of 40 healthy specific pathogen-free Long Evans rats were supplied by the Experimental Animal Center of Daping Hospital, Third Military Medical University of Chinese PLA (animal license No. SCXK 2007-0005) at postnatal 2–7 weeks, irrespective of sex. The rats were reared in a light-controlled room before the start of experiments, with a fixed lighting schedule (8:00 to 20:00). Light was generated by two fluorescent lamps with approximately 90 Ix of intensity at the animal level[10,18,19]. The room temperature was controlled at 22–25°C, and humidity at 50–60%[20,21]. All experimental protocols were conducted in accordance with the Guidance Suggestions for the Care and Use of Laboratory Animals, formulated by the Ministry of Science and Technology of China[22].
Methods
Establishment of binocular form deprivation rats
At PD 14, before eye-opening, binocular form deprivation Long Evans rats were established by suturing the binocular eyelids under sodium pentobarbital anesthesia[5,23]. The binocular form deprivation group was inspected twice a day for holes in the sutured eyelids. We immediately repaired the holes, if necessary. Closed binocular eyelids after suturing indicated successful binocular form deprivation model establishment[24].
Preparation of rat visual cortical slices
Visual cortical slices were prepared from normal rats at PD 14, 21 (7 days after eye-opening), 35 and 49, as described previously[10,25], and from binocular form deprivation rats at PD 21, 35 and 49. Animals were initially anesthetized with ether[26] and then decapitated. Brains were rapidly removed and placed into cold (4°C) carbogenated artificial cerebrospinal fluid, supplemented with 124 mM NaCI, 26 mM NaHCO3, 3 mM KCI, 1.25 mM NaH2PO4, 1.3 mM MgCI2, 2 mM CaCI2, 10 mM dextroglucose, pH 7.4[27,28]. Subsequently, visual cortical slices[29,30,31] (300 μm) were dissected using a vibroslicer (Ted Pella Instrument, CA, USA), and then incubated at room temperature for at least 1 hour in carbogenated artificial cerebrospinal fluid.
Electrophysiological recording
For electrophysiological recordings, slices were transferred to a submerged-type recording chamber and continually perfused with carbogenated artificial cerebrospinal fluid (1.5 mL/min) at 20–25°C[32]. Pipettes were pulled from borosilicate glass capillaries (World Precision Instruments, USA) with the use of a P-97 micropipette puller (Sutter Instrument, Novato, USA). The electrodes had tip impedances of 5–10 MΩ when filled with a solution containing 140 mM K+ gluconate, 2 mM MgCI2, 1.1 mM ethyleneglycol-bis(β-aminoethyl ether)-n, n’-tetraacetic acid, 0.1 mM CaCI2, 10 mM hydroxyethyl piperazine ethanesulfonic acid, 2 mM K2ATP, pH 7.4. The pipette was aimed at layers II–IV of the visual cortex and the stimulating electrode was placed in the white matter under the guidance of a stereomicroscope (Olympus Corporation, Tokyo, Japan).
Whole-cell recording was achieved using the patch-clamp technique[13,33,34] with an Axoclamp-200B amplifier (Axon Instrument, Foster City, CA, USA). Postsynaptic currents were evoked with a stimulus of 0.5 mA and 100 μs duration, and the voltages were clamped at-30 mV. Series resistance (10 to 20 megohms) was compensated and checked for constancy throughout the experiments.
Bicuculline methiodide (20 μM)(Sigma, St. Louis, MO, USA) and D,L-2-amino-5-phosphonovalerate (20 μM) (Sigma) were added to artificial cerebrospinal fluid for at least 5 minutes, sufficient to block gamma-aminobutyric acid receptors and N-methyl-D-aspartate receptors, and the AMPA-EPSCs were recorded. The AMPA-EPSCs were also assessed in the presence of bicuculline methiodide (20 μM), 6-cyano-7-nitroquinoxaline-2,3-dione (20 μM)(Sigma) and D,L-2-amino-5-phosphonovalerate (20 μM) to block gamma-aminobutyric acid, AMPA and N-methyl-D-aspartate receptors, respectively. Finally, all blockers were washed out to test the recovery of postsynaptic currents[28].
Statistical analysis
Data analysis was carried out using Clampfit8.1 (Axon Instrument) and SPSS 13.0 software (SPSS, Chicago, IL, USA). Data are expressed as mean ± SD using one-way analysis of variance with Dunnett's post-hoc test for peak value, rise time (10–90%), and decay time (10–90%) of AMPA-EPSCs.
Footnotes
Funding: This project was funded by the National Natural Science Foundation of China, No. 30772350.
Conflicts of interest: None declared.
Ethical approval: This study was approved by the Animal Ethics Committee of the Third Military Medical University of Chinese PLA.
(Edited by Lin JT, Li L/Qiu Y/Song LP)
REFERENCES
- [1].Hubel DH, Wiesel TN. The period of susceptibility to the physiological effects of unilateral eye closure in kittens. J Physiol. 1970;206(2):419–436. doi: 10.1113/jphysiol.1970.sp009022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Baroncelli L, Braschi C, Spolidoro M, et al. Nurturing brain plasticity: impact of environmental enrichment. Cell Death Differ. 2010;17(7):1092–1103. doi: 10.1038/cdd.2009.193. [DOI] [PubMed] [Google Scholar]
- [3].Di Garbo A, Mainardi M, Chillemi S, et al. Environmental enrichment modulates cortico-cortical interactions in the mouse. PLoS One. 2011;6(9):e25285. doi: 10.1371/journal.pone.0025285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Bélanger MC, Di Cristo G. Sensory experience differentially modulates the mRNA expression of the polysialyltransferases ST8Siall and ST8SialV in postnatal mouse visual cortex. PLoS One. 2011;6(9):e24874. doi: 10.1371/journal.pone.0024874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Hensch TK. Critical period regulation. Annu Rev Neurosci. 2004;27:549–579. doi: 10.1146/annurev.neuro.27.070203.144327. [DOI] [PubMed] [Google Scholar]
- [6].Kanonidou E. Amblyopia: a mini review of the literature. Int Ophthalmol. 2011;31(3):249–256. doi: 10.1007/s10792-011-9434-z. [DOI] [PubMed] [Google Scholar]
- [7].Meng K, Li YH, Zhang L, et al. Ca2+-permeable AMPA receptors mediate induction of test pulse depression of naive synapses in rat visual cortical slices at early postnatal stage. Neuroscience. 2010;165(3):684–691. doi: 10.1016/j.neuroscience.2009.11.030. [DOI] [PubMed] [Google Scholar]
- [8].Asrar S, Zhou Z, Ren W, et al. Ca(2+) permeable AMPA receptor induced long-term potentiation requires PI3/MAP kinases but not Ca/CaM-dependent kinase II. PLoS One. 2009;4(2):e4339. doi: 10.1371/journal.pone.0004339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Goel A, Xu LW, Snyder KP, et al. Phosphorylation of AMPA receptors is required for sensory deprivationinduced homeostatic synaptic plasticity. PLoS One. 2011;6(3):e18264. doi: 10.1371/journal.pone.0018264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Sato MT, Tokunaga A, Kawai Y, et al. The effects of binocular suture and dark rearing on the induction of c-fos protein in the rat visual cortex during and after the critical period. Neurosci Res. 2000;36(3):227–233. doi: 10.1016/s0168-0102(99)00126-1. [DOI] [PubMed] [Google Scholar]
- [11].Giannakopoulos M, Kouvelas ED, Mitsacos A. Experience-dependent regulation of NMDA receptor subunit composition and phosphorylation in the retina and visual cortex. Invest Ophthalmol Vis Sci. 2010;51(4):1817–1822. doi: 10.1167/iovs.09-4438. [DOI] [PubMed] [Google Scholar]
- [12].Hensch TK. Critical period plasticity in local cortical circuits. Nat Rev Neurosci. 2005;6(11):877–888. doi: 10.1038/nrn1787. [DOI] [PubMed] [Google Scholar]
- [13].Golshani P, Warren RA, Jones EG. Progression of change in NMDA, non-NMDA, and metabotropic glutamate receptor function at the developing corticothalamic synapse. J Neurophysiol. 1998;80(1):143–154. doi: 10.1152/jn.1998.80.1.143. [DOI] [PubMed] [Google Scholar]
- [14].Kielland A, Heggelund P. AMPA receptor properties at the synapse between retinal afferents and thalamocortical cells in the dorsal lateral geniculate nucleus of the rat. Neurosci Lett. 2001;316(2):59–62. doi: 10.1016/s0304-3940(01)02337-0. [DOI] [PubMed] [Google Scholar]
- [15].He K, Lee A, Song L, et al. AMPA receptor subunit GluR1 (GluA1) serine-845 site is involved in synaptic depression but not in spine shrinkage associated with chemical long-term depression. J Neurophysiol. 2011;105(4):1897–1907. doi: 10.1152/jn.00913.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Kumar SS, Huguenard JR. Properties of excitatory synaptic connections mediated by the corpus callosum in the developing rat neocortex. J Neurophysiol. 2001;86(6):2973–2985. doi: 10.1152/jn.2001.86.6.2973. [DOI] [PubMed] [Google Scholar]
- [17].Liu X, Chen C. Different roles for AMPA and NMDA receptors in transmission at the immature retinogeniculate synapse. J Neurophysiol. 2008;99(2):629–643. doi: 10.1152/jn.01171.2007. [DOI] [PubMed] [Google Scholar]
- [18].Bengoetxea H, Argandoña EG, Lafuente JV. Effects of visual experience on vascular endothelial growth factor expression during the postnatal development of the rat visual cortex. Cereb Cortex. 2008;18(7):1630–1639. doi: 10.1093/cercor/bhm190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Argandoña EG, Bengoetxea H, Lafuente JV. Physical exercise is required for environmental enrichment to offset the quantitative effects of dark-rearing on the S-100beta astrocytic density in the rat visual cortex. J Anat. 2009;215(2):132–140. doi: 10.1111/j.1469-7580.2009.01103.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Gelfo F, De Bartolo P, Giovine A, et al. Layer and regional effects of environmental enrichment on the pyramidal neuron morphology of the rat. Neurobiol Learn Mem. 2009 May;91(4):353–65. doi: 10.1016/j.nlm.2009.01.010. [DOI] [PubMed] [Google Scholar]
- [21].Koehnle TJ, Rinaman L. Early experience alters limbic forebrain Fos responses to a stressful interoceptive stimulus in young adult rats. Physiol Behav. 2010;100(2):105–115. doi: 10.1016/j.physbeh.2010.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].The Ministry of Science and Technology of the People's Republic of China. Guidance Suggestions for the Care and Use of Laboratory Animals. 2006 Sep 30; [Google Scholar]
- [23].Krahe TE, Medina AE. Activation of NMDA receptors is necessary for the recovery of cortical binocularity. J Neurophysiol. 2010;103(5):2700–2706. doi: 10.1152/jn.00442.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Wang WF, Kiyosawa M, Ishiwata K, et al. Glucose metabolism in the visual structures of rat monocularly deprived by eyelid suture after postnatal eye opening. Jpn J Ophthalmol. 2005;49(1):6–11. doi: 10.1007/s10384-004-0146-z. [DOI] [PubMed] [Google Scholar]
- [25].Fathollahi Y, Salami M. The role of N-methyl-D-aspartate receptors in synaptic plasticity of rat visual cortex in vitro: effect of sensory experience. Neurosci Lett. 2001;306(3):149–152. doi: 10.1016/s0304-3940(01)01894-8. [DOI] [PubMed] [Google Scholar]
- [26].Tsurugizawa T, Uematsu A, Uneyama H, et al. Effects of isoflurane and alpha-chloralose anesthesia on BOLD fMRI responses to ingested L-glutamate in rats. Neuroscience. 2010;165(1):244–251. doi: 10.1016/j.neuroscience.2009.10.006. [DOI] [PubMed] [Google Scholar]
- [27].Wang Y, Neubauer FB, Lüscher HR, et al. GABAB receptor-dependent modulation of network activity in the rat prefrontal cortex in vitro. Eur J Neurosci. 2010;31(9):1582–1594. doi: 10.1111/j.1460-9568.2010.07191.x. [DOI] [PubMed] [Google Scholar]
- [28].Qin W, Yin ZQ, Wang S, et al. Effects of binocular form deprivation on the excitatory post-synaptic currents mediated by N-methyl-D-aspartate receptors in rat visual cortex. Clin Experiment Ophthalmol. 2004;32(3):289–293. doi: 10.1111/j.1442-9071.2004.00819.x. [DOI] [PubMed] [Google Scholar]
- [29].Cheetham CE, Fox K. Presynaptic development at L4 to 12/3 excitatory synapses follows different time courses in visual and somatosensory cortex. J Neurosci. 2010;30(38):12566–12571. doi: 10.1523/JNEUROSCI.2544-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].McCoy PA, McMahon LL. Sympathetic sprouting in visual cortex stimulated by cholinergic denervation rescues expression of two forms of long-term depression at layer 2/3 synapses. Neuroscience. 2010;168(3):591–604. doi: 10.1016/j.neuroscience.2010.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Osanai M, Tanaka S, Takeno Y, et al. Spatiotemporal properties of the action potential propagation in the mouse visual cortical slice analyzed by calcium imaging. PLoS One. 2010;5(10):e13738. doi: 10.1371/journal.pone.0013738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Ali AB, Bannister AP, Thomson AM. Robust correlations between action potential duration and the properties of synaptic connections in layer 4 interneurones in neocortical slices from juvenile rats and adult rat and cat. J Physiol. 2007;580(Pt 1):149–169. doi: 10.1113/jphysiol.2006.124214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Jiang B, Huang S, de Pasquale R, et al. The maturation of GABAergic transmission in visual cortex requires endocannabinoid-mediated LTD of inhibitory inputs during a critical period. Neuron. 2010;66(2):248–259. doi: 10.1016/j.neuron.2010.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Runyan CA, Schummers J, Van Wart A, et al. Response features of parvalbumin-expressing interneurons suggest precise roles for subtypes of inhibition in visual cortex. Neuron. 2010;67(5):847–857. doi: 10.1016/j.neuron.2010.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]


